3
Methylmercury Contamination of Aquatic Ecosystems: A Widespread Problem with Many Challenges for the Chemical Sciences
David P. Krabbenhoft
U.S. Geological Survey, Water Resources Division
INTRODUCTION
Mercury, as an issue, had a rebirth in the late 1980s, especially in Wisconsin’s most precious tourist area, the northern Wisconsin Lake District. More than 50 percent of the fish in the district during that time were contaminated with levels of mercury exceeding those provided in advisories limiting the consumption of fish for certain people. With such a pristine area being contaminated, people became very concerned and wanted some explanation.
In this paper, the development and current understanding of this problem are explored. Two examples are given of current projects that are at the crossroads of politics and policy; both are directly trying to answer the question of just how responsible current emissions are in driving this problem versus historic contamination that now blankets the world’s sediments and soils from a legacy of mercury releases.
Mercury Contamination
It is difficult to not currently be aware of this issue. It is on the front page of about every newspaper and magazine, and it is there for a very good reason. Methylmercury formed by bacterial action on ionic mercury (Hg(II)) is far and away the leading environmental contaminant for consumption advisories of fish in the United States, as well as globally. This is because mercury emissions are ubiquitous, and almost no other contaminant bioaccumulates to the extent of methylmercury. Typical bioconcentration factors are a million- to a hundred million-fold more than water concentrations, giving levels of toxicological concern even in the most remote sections of the planet. This makes the issue highly visible today, along with discussion of reducing mercury emissions from coal-burning facilities and compliance with the Clean Air Act and its proposed amendments with the Clear Skies Initiative.
There are direct health effects on humans and fish-eating wildlife from methylmercury contamination of fish. Last year it was found that mercury is, in fact, a major endocrine disrupter and that mercury, specifically methylmercury, is a significant inhibitor of selenium uptake by humans, wildlife, or any invertebrate system. Selenium deficiency can thus come from mercury exposure. In addition, concern has increased about sociocultural damage to people in fish-eating communities whose culture would be affected if they had to switch their food base.
Global Loading Sources
The impacts on the globe from mercury releases before and after industrialization are shown in Figure 3.1. There is three- to ten-fold more mercury now compared to pre-industrialization.
It is not clear how loadings from mining, atmospheric, and other sources compare to each other. Most of the effort for geochemists now lies in trying to understand what controls the availability of various mercury sources once they are released to the environment. It is known that mercury sources cannot be traced back to mercury releases atom for atom. However, it is not known why. That is an area in which the chemical sciences can play a very big role in helping to understand what happens to mercury, especially how it is made biologically available.
Where does mercury come from globally? About a third of the mercury currently released to the world’s atmosphere comes directly from human activities. Another third comes from the oceans through photoreduction at the surface and volatilization because mercury is incredibly photosensitive. Speciation changes take place by the hour whether in a lake, a stream, or an ocean and are difficult to study because they depend on time of day and light intensity. For a geochemist this is a big challenge. The third source comes from soil emissions, again, mostly through photoreduction. The ma-
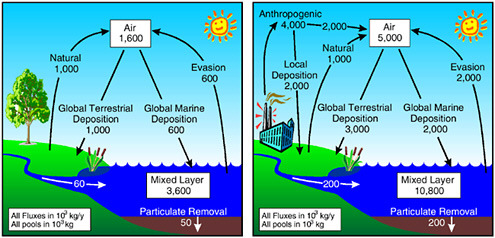
FIGURE 3.1 Pre-industrial (left) and current (right) global mercury cycle. SOURCE: Adapted from Mason, R. P.; W. F. Fitzgerald; F. M. M. Morel. 1994. The biogeochemical cycling of elemental mercury: Anthropogenic influences, Geochimica et Cosmochimica Acta, 58(15): 3191-3198.
jority, then, probably two-thirds of the emissions are actually reemissions of anthropogenic sources of mercury in the oceans and soils of the world. Current efforts are focused on determining how quickly these emissions can be reduced. To do this a better quantitative context is needed for establishing how quickly the world’s ecosystems will respond to any proposed mercury emissions reduction strategies.
A number of developing countries, particularly China, are the biggest mercury emitters worldwide, primarily because of their use of energy generated from coal combustion. The use of coal is by far the most significant source of mercury emitted to the atmosphere and for the last decade has contributed to the emission and deposition of an immense amount of mercury into the ecosystems of China. When the third largest reservoir in the world, Three Gorges Dam, is filled, the mercury now present in those areas that are going to be flooded will be methylated at a high rate since the highest-efficiency route to methylate mercury is to flood an area. Many of the people who live on the Yangtze River are fishermen, and they will undoubtedly receive more methylmercury as a result of that dam construction, coupled with the large-scale use of coal in the last decade. Such a result has already been demonstrated in Canada where there were five-to twenty-fold increases in mercury concentrations in fish after the creation of a reservoir.
Modeling
More information is available for the United States than most other countries because a great deal of effort is put into understanding mercury emissions and deposition patterns. Data from the Toxic Release Inventory coupled with an atmospheric mercury model can be used to predict mercury deposition patterns around the United States. It turns out that mercury deposition is as much a function of rainfall patterns in the United States as it is of the actual sources of emissions.
Rainfall amount alone can completely account for the differences observed because the atmosphere is presently never depleted of mercury from rainfall. The amount that can be stripped out from a rainfall event is an infinite pool. There is heterogeneity to the earth’s skies, particularly in industrialized areas such as the United States, but the stripping mechanism that brings mercury to the earth, most importantly, is rainfall.
The model suggests that mercury deposition rates differ by up to three orders of magnitude across the United States. It might be on the order of a factor of 10 or so, but the greatest deposition occurs in the eastern United States, where there are more mercury emissions due not only to more incineration and more coal combustion, but also to more rainfall.
Mercury Cycling
It is now known that mercury coming out of stacks is speciated primarily in three different forms: elemental (zerovalent Hg) as a vapor, particulate (organic Hg(II) compounds), and ionic (inorganic Hg(II) compounds, primarily mercuric chloride). Elemental mercury travels around the globe, while particulate mercury is not transported very far and ionic mercury is transported intermediate distances. The
ionic form of mercury probably has a significant impact on ecosystems since it is a very soluble species and quickly becomes part of a methylation cycle.
Mercury would not be such a concern if it were not methylated. All of the mercury in consumable fish tissues and just about all vertebrate systems is methylmercury. This is the only form of mercury that bioaccumulates in humans and wildlife, the only form of mercury that crosses the blood-brain barrier where it causes most of its problems, and the only form that accumulates in human neurological tissues. Not only does methylmercury bioaccumulate more once it is in the body, but it can also access areas that elemental mercury cannot. About half of the mercury in humans probably comes from dental fillings, but that inorganic mercury does not get to the more sensitive parts of the body.
It is very important to understand the connection between where methylmercury is produced, where it is absorbed, and its behavior in the body (Figure 3.2). The path from release to the formation of ionic mercury involves photochemical processes, deposition, and then formation of methymercury at the sediment-water interface in most aquatic ecosystems. The primary methylating agents in most of these aquatic ecosystems are sulfate-reducing bacteria, which are ubiquitous in the top centimeter where most sediments transition from aerobic to anaerobic. Sulfate diffuses down from the water just above this area, and if mercury is present, the sulfur will be in the form of Hg(II) compounds such as HgS(HS)−,
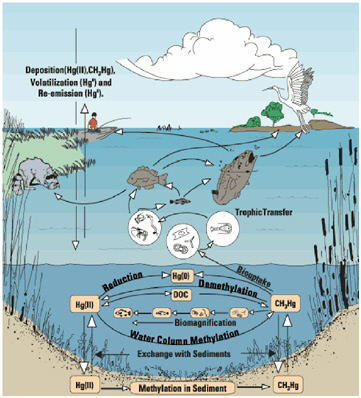
FIGURE 3.2 Aquatic cycling of mercury.
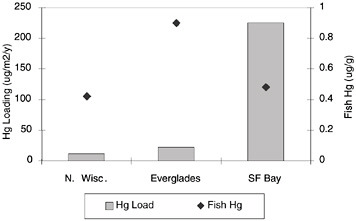
FIGURE 3.3 Relating sources and loading to bioaccumulation—bioavailability is the key.
Hg(HS)2, and Hg(Sn)HS− that are then converted to methylmercury.1
Mercury Loading and Ecological Restoration
Until 15 years ago, mercury was not considered as much of a problem as it is today. About 27 million pounds of mercury were largely unaccounted for in the Sierra Nevadas from the 1849 Gold Rush in California. That mercury is still making its way out of the Sierra Nevadas through the central valley drainage system and into San Francisco Bay where it is now a very large issue.
Hundreds of tons of mercury currently enter the California bay delta system. At the same time, the bay is slated for a substantial effort of ecological restoration, and the number-one activity for spending money in that ecological restoration is the construction of wetlands. It is now known that the best way to make methylmercury in the environment is to put mercury into wetlands. When this was realized a few years ago, in the San Francisco area, it created a major problem for local, state, and federal agencies.
However, this is not just a case of mercury loading in the water or on land. Figure 3.3 shows estimates of the atmospheric loading rates. The value for Wisconsin is only milligrams per square meter per year anywhere in the state. The riverine load from the Sierras draining the historical contaminated areas of the Gold Rush, by comparison, dwarfs that in Wisconsin.
However, the average mercury concentration in a predator fish or sport fish represented by diamonds in Figure 3.3 is as high for the bay delta of California as it is for northern
Wisconsin. More effective means than controlling mercury load are thus needed to address mercury toxicity in ecosystems.
EXAMPLES
Ideally, available research findings could prescribe a “safe” level of mercury exposure to ecosystems that would result in an acceptable amount of mercury in fish. However, it is presently not known if “new,” recently deposited mercury is what drives mercury levels in fish or if vast pools of “old” mercury currently residing in soils and sediments from decades to centuries of deposition are the driving factor and could sustain the problem for many decades to come. To fill this information gap, researchers are currently conducting two different mercury loading studies in the United States (the Aquatic Cycling of Mercury in the Everglades [ACME] project) and Canada (the Mercury Experiment to Assess Atmospheric Loading in Canada and the United States [METAALICUS] Project), using stable isotopes of mercury that effectively distinguish between new and old mercury.2
Aquatic Cycling of Mercury in the Everglades (ACME) Project
The ACME project began in 1995 and has changed tremendously over the last eight years. This is a completely infield experimental approach to understand mercury cycling processes and is ready to interface directly with the $8 billion Everglades restoration program. This is the largest restoration program ever attempted in the United States, and although it is not intended to make the mercury conditions in the Everglades worse, it may.
The large series of in-field experiments mimic what may happen as a result of restoring this ecosystem. This is being done to see what impact the current approach will have. When a wetland is drained and its soil is oxidized, the wetland cannot be recreated from that soil. Now about 50 percent remains of what was once the Florida Everglades, often described as a “river of grass.”
Agricultural Impacts
There are many complicating factors in the attempt to restore this large wetland area, and many come from the Everglades agricultural area. This is a very important vegetable and sugarcane growing area for the United States and is the source of many vegetables in winter.
One of the detrimental actions in the Everglades agricultural area occurs because these former Everglades soils lack free nutrients. To grow crops, farmers must make phosphate available. Because alkaline soils bind all the nutrients, farmers add elemental sulfur by the ton to adjust the soil pH and free the nutrients. Sulfur is converted to sulfate (sulfuric acid) by bacterial action. This feeds the sulfate-reducing bacteria that make methylmercury. As it turns out, it is evident that sulfate additions in this agricultural area have more to do with the methylmercury problem in the Everglades than mercury falling from the sky. Efforts are now being made to work out what the primary controlling set of processes is and the external factors that led to this large problem of mercury contamination in the Everglades.
South Florida is an area with ecosystem-wide postings for mercury, unique to fish consumption advisories for mercury in the United States. Only in South Florida does it say that no one should eat fish. Everywhere else, advisories state that one can eat one fish per month if you are not pregnant or of childbearing age. The eating and catching of fish are curtailed as revenue of hundreds of millions of dollars is being lost because tourists no longer come to fish.
Drainage canals in the agricultural area keep fields from becoming flooded, but they also convey the sulfate put on fields to the Everglades. The result is a 100-mile-long sulfate gradient that runs from the agricultural area just south of Lake Okeechobee all the way down to Everglades National Park. Right now the only way for the national park to be affected by agricultural activities is by short-circuiting the canals, draining nutrients straight down into the park. In fact, this is the first step that was undertaken in the Everglades restoration, opening these floodgates to get water down there as quickly as possible.
Hydrologic Restoration Versus Water Quality Restoration: Sulfates
It has been difficult to express discontent with the approach being used in the hydrologic restoration of the Everglades, which ignores the quality of that water. It is understandable that Everglades National Park people want water as soon as possible; their ecosystem is greatly diminished by the lack of water. Water and appropriate breeding habitat for those fish-eating wading birds that historically were found in great numbers in South Florida are needed for the success of the ecosystem.
However, if water is shunted down there too fast, sulfur is going to be there for a very long time. It will continue to cycle and may eventually get buried, but it is going to have a very long history and cause continuing problems.
Population Impacts
An additional point is that in South Florida presently, there are plans for a very large scale aquifer storage and recovery system to support what is anticipated to be a dou-
|
2 |
For additional details on the ACME project, see http://sofia.usgs.gov/people/krabbenhoft.html, and for the METAALICUS project, see http://www.biology.ualberta.ca/old_site/metaalicus//metaalicus.htm. |
bling of the population over the next 25 years. To meet and integrate with sustainable water development needs, the waters that are being sent into the aquifer will be recovered later, not only for use as drinking water, but also to feed the ecosystem when it needs its water laden with sulfate. Efforts are being made to see just how great an impact this will have in the near future.
Flooding
Flooding previously exposed lands can have a huge impact. Flooding also occurs naturally in the Everglades. Every year, every other year, every third year, depending on whether it is a wet or a dry cycle, the Everglades naturally go through this dry-down and rewetting, moving the whole ecosystem into much, much higher levels of methylmercury, much higher than in environments that remain wet.
To most people in South Florida restoring the Everglades is equivalent to restoring the natural hydrology of the system. Water quality in the Everglades is synonymous with phosphorus abatement, but phosphate has no impact on methylmercury formation.
There are many other chemical concerns in the Everglades that have not been addressed because phosphorus gets all the attention. In South Florida, the hydrologic restoration will call for specifically more wetting and drying, higher high waters, and longer periods of dry-out and oxidation of the soils. As the soils become wet again, it is necessary to know what mercury is the source of the newly formed methylmercury.
Is it mercury already in the soil, derelict contamination, or mercury in rainfall that rewet the system? If it is mercury in rainfall that rewet the system, it is still possible to improve the situation. If it is mercury that is already in the soil, it becomes a much more difficult problem.
By the use of the stable isotopes in experiments in meterwide tanks (mesocosms), it is possible to distinguish between mercury that has been added and mercury that is already in the ecosystem. It was found that the older mercury is less available to Gambusia, the little mosquito fish ubiquitous across the Everglades and a very good biomarker across the system (Figure 3.4). What was observed however is that over this range of loading in the Everglades, there was a very linear and positive relationship between the amount of mercury added and the amount of mercury in the fish. This is mercury added, not remobilized mercury from sediments.
It was very important to establish that, in fact, in contaminated ecosystems there is still a positive response between the amount of mercury that is added and the amount of mercury found in fish.
Mercury Aging
Studies were also carried out to understand the effects of the age of mercury sources on bioavailability. A comparison
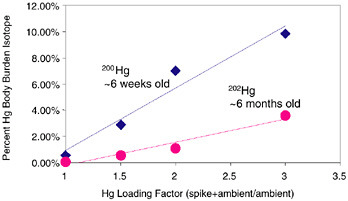
FIGURE 3.4 Bioaccumulation of new versus old mercury by Gambusia.
was made between the percentage of two sources of mercury in fish, one six weeks old (200Hg) and the other six months old (202Hg). The same amount of each mercury isotope was added to the same mesocosm. It was found that six-week-old mercury was three times more prevalent in fish than mercury that was four and a half months older. This is the so-called mercury aging effect. It has been found in the last few years that ecosystems do not cycle old mercury and new mercury equivalently.
These experiments have been repeated with the same results. In these very same mesocosms—now two years old—fish were gathered again and it is no longer possible to find either one of these isotopes that are still present in sediment. They are seen in sediment, but not in fish.
Mercury accumulation in fish is being driven by new mercury coming in. Now a third isotope can be added and it ends up in fish at the same amount added, but the mercury that gets into sediments is stabilized, probably by reduced sulfur groups in the soils. It does not cycle anywhere near as actively as the new mercury.
Then a series of mixed experiments was conducted and it was found, for example, that adding dissolved organic carbon (DOC) with mercury greatly increased the amount of mercury that became methylated and bioaccumulated in fish.
Low amounts of added sulfate resulted in considerable methylmercury formation. By adding more, sometimes there would be more, but if there is a high amount of sulfate in the mesocosms, methylation is shut down because the bioavailability of the mercury decreases. What does this mean for the Everglades? A series of hypotheses are being tested presently with a model to see whether any of this can be predicted.
It is now believed that if sulfate loads are reduced coming into the ecosystem, the peak of methylmercury formation will be moved further north, but there will still be a zone in which sulfur levels are so high that there will be inhibition. There will also be an optimal zone for methylmercury for-
mation, but the overall size of the peak will be less. Under a restorative flow condition with no attention to water quality, sulfate transport into the system will increase; instead of having just a peak of methylmercury formation in one area, there will be an entire southern Everglades system with high methylmercury levels. This is undesirable.
METAALICUS Project—Ecosystem Level Response to Changes in Mercury Loading
One of the criticisms of the work conducted in the Florida Everglades is that the experiments are run inside meter-widediameter cylinders giving “mesocosm effects,” with results that are not representative of the surrounding environment. An approach to deal with this criticism is to dose an entire watershed in northwestern Ontario with mercury, and not only do that, but dose a whole watershed with mercury isotopes to answer two questions: (1) What is the true ecosystem-scale response to a change in mercury load, looking at the same time at new versus old mercury? (2) Which is more important at the watershed scale?
The project is known as METAALICUS. The list of participants is very long. It is a large project, and the mercury isotopes alone cost about a million dollars a year, but this is the only way to really answer these questions. The major questions to be answered deal with the ecosystem-level response to a change in mercury loading. Next, it has been stated over and over again that a hundred years of derelict contamination exists in soils and sediments across the globe. Will things improve if mercury emissions are reduced? Third, can the vast sedimentary pools of mercury sustain the problem for very long periods of time? Last, do new and old mercury behave similarly in the environment, and if not, why?
Mercury-202 is being added to the lake; mercury-199 to the wetlands, which are on the western edge of the lake; and mercury-200 to the entire forested catchment. The mercury is added to the wetland and the catchment by a very brave aviator who flies right above the treetops in driving rainstorms (Figure 3.5).
This is being done at a rate of about 20 µg/m2 per year; similar to what is seen in the eastern United States. An area that presently gets about 5 µg of mercury is being increased to about 20 µg. It should be noted that the amount of mercury being added to the ecosystem is so small that the best mass spectrometers would not detect it. The added mercury is thus not an environmental concern.
Mercury Aging
It was found that mercury-202 added directly in the lake is methylated at a five to seven times higher rate than native mercury, providing further evidence in a completely different ecosystem (not confined by a cylinder or anything else) to show that new mercury is responsible for the formation of methylmercury. It is not old mercury that has facilitated the problem. This is not to say that old mercury has nothing to do with it, but atom for atom, the old mercury is not nearly as involved.
There continues to be a significant amount of ambient mercury moving off the uplands into the lake, at about 1,500 mg per year. However, almost none of the spike put down it has moved—only about 1 percent. Mercury, when applied to the surface of a green plot, whether it is grassland, wetland, or just a place with plants, tends not to move until it penetrates deeper into the soil, into the humus zone where the great mobilizer of mercury in the environment, dissolved organic carbon, is generated.
Deep Temperate Lake versus South Florida Wetlands
In this project a cohort of yellow perch was followed that were born in the spring of 2001 with no mercury. At the end of their first growing season, the vast majority of the mercury that they had accumulated was ambient mercury not the spike. Even though a four- to five-year pulse was added to the lake, it contributed very little of the mercury in the fish.
Now about a third of the bioaccumulated methylmercury in the second year is coming from the spike. This differs from the situation in the Everglades where both very rapid responses to additions and very quick dissipations of the effect of that new mercury are seen. Here, a deep temperate lake and arboreal ecosystem seem to have longer time lags, which can probably be expected in lake settings across the northern United States and southern Canada.
Next Steps
This project, now in its third year, is going to be a five- to ten-year effort. Thus far, it has been a mercury loading study. The debate in Washington right now centers on the effect of a reduction. The group of scientists that planned this study was thinking about that seven years ago when it was decided that a mercury reduction study cannot be done until a loaded system is available. So the idea all along with this project was to load up an ecosystem in order to be able to watch the response when reduction occurs. This was the last year of additions; next year, the reduction phase and probably the more interesting part of this study starts.
At each site, scientists have observed the same basic processes operating on the added mercury, but at different rates due to the very different environmental conditions—the warm, hot wetlands of the Everglades and the cold, boreal lake in Canada. At both study sites, however, the new mercury gets converted into methylmercury (the most toxic and bioaccumulated form of mercury) more efficiently than the old (Figure 3.5).
FIGURE 3.5 Movement of mercury isotopes.
DISCUSSION
Incident at Lake Apopka
Tom Dillion, of Science Applications International Corporation, noted that in late 1998 at Lake Apopka in Florida, some mud farms were flooded resulting in the worst bird kill ever. He questioned whether the incident was a result of the mercury phenomenon.
Dr. Krabbenhoft responded that the decline in wading birds got people’s attention, and mercury was assumed to be the culprit. Mercury loading studies have been performed on adult birds. It was determined that adult birds have a unique way of dealing with methylmercury; they shunt all their methylmercury straight to their feathers and can withstand almost anything. However, eggs put all the methylmercury into body growth and internal organ development, resulting in a substantial decline in the survival rate of eggs upon exposure to methylmercury. He does not think the Lake Apopka response was methylmercury, although many may disagree.
Policy Implications
Debbie Elcock, of Argonne National Laboratory, requested some conclusions about the policy implications.
Dr. Krabbenhoft replied that there are three ways to deal with the mercury problem: fish consumption advisories, emissions reductions, and landscape management. Unfortunately, fish consumption advisories do not help the wildlife. Emissions reductions will decrease the new mercury available for methylation in the environment; however, old mercury still exists there. For instance, it is necessary to consider whether hydrologic restoration in South Florida needs to move forward more quickly than the water quality restoration. Florida’s Governor Bush signed an amendment to the Everglades Forever Act delaying achieving the water quality criteria until 2016 instead of meeting those criteria in two more years. From Dr. Krabbenhoft’s vantage point delaying achieving those water quality criteria but continuing with the hydrologic restoration could potentially be a disaster. It might be better if they did not proceed with hydrologic restoration at all until they are comfortable moving ahead with water quality restoration at the same time.
Furthermore, mercury is a concern of the current administration and many federal agencies. The proposed Clear Skies Act and the proposed Clean Air Act have specific emission reduction levels in them, and Dr. Krabbenhoft is not aware of any studies that pinpoint the appropriate level of reduction. He and his colleagues are very curious about where these reduction levels came from.
The United Nations has an environmental program that is also addressing the mercury issue. Two or three meetings have been held in the last year at which mercury was one of the topics. The United States and other U.N. nations are entertaining thoughts of global mercury reductions as a way of addressing the problem.
Alan Hecht, of the White House Council on Environmental Quality (now at the Environmental Protection Agency), commented that engineers are already starting to release water in the marshlands in Iraq to try to restore them. Releasing the water without a look at the chemistry may lead to greater problems down the road. The Everglades is an area that has to be drawn into the policy arena more quickly because actions are being taken that may have unintended consequences.
Dr. Krabbenhoft replied that Iraq has experienced that many times with mercury. About 40 or 50 years ago, the United States sent seeds to Iraq to help them grow wheat. The Iraqis thought the seed would also be a good source of wheat for flour. Unfortunately, labels on the seeds were only printed in English, so the Iraqis could not read that the seed was sterilized with phenylmercuric acetate. Use of the seed for flour resulted in the deaths of thousands of people. He gave this example to illustrate that good intentions sometimes lead to misfortune because of lack of follow-through.
Reactionary Responses
Dennis Hjeresen, of the Green Chemistry Institute, thought it seemed that attention to various chemical contaminants in the environment goes to the “current poster child.” In the past, dichlorodiphenyltrichloroethane (DDT) and trichloroethylene (TCE) were focused on, while currently mercury, perchlorates, and nitrates get the attention. However, it does not seem that, within the scientific community, there is a very good way of anticipating, setting priorities, or setting up an infrastructure to study the broader issues. It would appear that many of the biological and ecosystem issues are going to be the same across contami-
nants, but the money seems to flow only when there is a crisis.
Dr. Krabbenhoft’s big breakthrough as a researcher came when the Florida panther died of mercosis in the early 1990s. He said the response occurred because there was a crisis: something as precious and as artful as a Florida panther died of a specific contaminant. He might not have been able to study the problem if a Florida panther had not died.
However, he felt that things may be changing. Some colleagues are now doing research on emerging contaminants, hormones, and pharmaceuticals. In this way, they are working in more of an anticipatory rather than a reactionary framework. However, it is highly unlikely that a researcher will receive funding unless the research can meet a need. Generally, it is more common to react than to prevent.








