3
Lessons Learned
With the many policy, research, and funding initiatives now supporting and recommending antiretroviral therapy (ART) scale-up for resource-constrained settings, it will be critical to understand the experience gained by both the developed and developing worlds in treating people with HIV/AIDS. One lesson learned is that poor early treatment decisions can limit a patient’s future therapeutic options and irrevocably alter the course of the disease and treatment response. One fear associated with use of ART in all settings is treatment failure as a result of the emergence of resistant virus. If widespread drug resistance were to emerge, the long-term public health impact of and financial capacity for HIV/AIDS treatment could be threatened. Preventing treatment failure—whether because of resistance or other factors—will be paramount to ensuring the long-term durability and sustainability of efforts to combat this pandemic. Thus it is essential that the many lessons learned about the causes and implications of treatment success, treatment failure, and drug resistance in the developed and developing worlds guide ART scale-up programs. While the successes achieved in more resource-constrained settings may have occurred on a small scale, they, too, can provide invaluable lessons for the scale-up of treatment to the millions in need now and in the future.
This chapter first describes the problem of drug resistance and then examines its implications for ART scale-up. The final section reviews lessons learned from early scale-up efforts around the world.
THE PROBLEM OF RESISTANCE
Highly Active Antiretroviral Therapy
During the initial years of treating the HIV/AIDS epidemic in the United States and other parts of the developed world where treatment was available, ART consisted of the use of one or two drugs, known as mono or dual therapy. By the mid-1990s, as additional drugs were developed for HIV, it became clear that other therapy regimens might be better. Highly active antiretroviral therapy (HAART), a regimen based on using at least three ARVs from a minimum of two classes in combination, was developed in the mid- to late 1990s following the introduction of additional classes of ARVs and the recognition that using more drugs in combination is more effective in restoring immune function, decreasing HIV burden, and preventing the emergence of drug resistance. The three main classes are nucleoside reverse transcriptase inhibitors (NRTIs), nonnucleoside reverse transcriptase inhibitors (NNRTIs), and protease inhibitors (PIs). (A drug from a fourth, newer class was approved in the United States in 2003 but is not yet widely used clinically and is not discussed further in this report.) HAART is now the standard treatment recommended in the United States. Appendix B provides a more thorough discussion of HAART.
Overview of Resistance
Drug resistance can render treatment ineffective for individual patients and has implications for the treatment of whole populations should they receive a high burden of transmitted drug-resistant virus. Resistance to HIV drugs arises in the presence of incomplete suppression of viral replication in the face of the selective pressure of drug treatment. The resistance-conferring mutations accumulate during rapid, error-prone HIV replication. The longer viral replication continues in the presence of inadequate ART regimens (i.e., failure of drugs to completely suppress viral replication), the more resistance will emerge. Even with low levels of virus present (i.e., between 50 and 500 copies/ml), drug resistance can develop (Fischl, 1999; Hirsch et al., 1998, 2000, 2003; Richman et al., 2004). The emergence of drug-resistant virus is one factor that can contribute to the failure of ART (Deeks, 2003). The fact that drug-resistant HIV-1 viral variants can still replicate well contributes to the rapid evolution of HIV in the face of potent drug pressure.
Although most resistance mutations confer a functional virus, the mutations can produce a virus with less replicative capacity than its wild-type relative. In theory, new mutations that emerge over time may often but not completely compensate for this defect. The lower fitness (or infectivity) of
drug-resistant variants, as well as the lower viral load in partially treated patients (compared with untreated patients), may contribute to a lower rate of transmission of drug-resistant virus (Leigh Brown et al., 2003). Nevertheless, public health efforts should be focused on trying to minimize the rate of acquiring resistance (i.e., by treating drug-sensitive cases appropriately), rather than on trying to prevent the transmission of drug-resistant strains.
Thus resistance to HIV drugs can be acquired, and HIV-resistant viruses can be transmitted. And most HIV-infected individuals with drug-resistant virus are initially infected with a drug-sensitive virus that acquires resistance during ART (Deeks, 2003).
The prevalence of drug resistance among HIV strains in the United States may be as high as 50 percent, based on the results of a large cohort study of HIV-infected adults receiving HAART (Richman et al., 2004). Drug resistance has been found for each class of drugs available for ART, and resistance may be present for more than one drug or class—a phenomenon known as multidrug resistance (Richman et al., 2004).
Acquired resistance is dependent upon several factors, including patient adherence to therapy, the mutation rate of the virus, and the efficacy of the ART regimen (Blower et al., 2003a; Deeks, 2003; Leigh Brown et al., 2003; Hirsch et al., 1998, 2000, 2003) (see Box 3-1). Adherence rates of less than 90 percent and less than 80 percent have been shown to result in drug resistance and virological failure (Hirsch et al., 1998, 2000, 2003; Paterson et al., 2000; Sethi et al., 2003). As discussed above, ineffective regimens that do not completely suppress viral replication will lead to resistant virus, and the use of sequential mono or dual therapy is no longer recommended because of its now-recognized association with the development of a high rate of drug resistance (Deeks, 2003; Hirsch et al., 1998, 2000, 2003; Leigh Brown et al., 2003).
Transmitted resistance occurs when a person is infected with a virus that has previously acquired resistance. Cases of drug-resistant virus in the United States were first documented in the mid-1990s, and several recent reports have suggested that the percentage of new infections demonstrating some form of drug resistance has risen significantly since then (Grant et al., 2002; Little et al., 2002; Simon et al., 2002). If transmitted drug-resistant virus is not faced with the continuous selection pressure of ART, it can revert back to the drug-sensitive, wild type of virus (Hirsch et al., 2003). Hence, the drug-resistant virus may not be measured at a particular point in time. However, the first documented case of a resistant virus that was stably transmitted from one person to a another, who then passed it on to a third person, was reported in 2003 (Taylor et al., 2003). See Appendix B for a more-detailed discussion of resistance.
|
BOX 3-1 Logistics
Medication factors
Patient factors
Virus factors
Provider factors
SOURCE: Deeks, 2003. |
Resistance to Antiretroviral Drugs and Their Classes
As noted above, drug resistance has been found for all classes of drugs available for treatment of HIV/AIDS (Clavel and Hance, 2004). The NRTI drug lamivudine (3TC) and drugs within the NNRTI class are the preferential components of regimens for resource-constrained settings. The ease with which resistance can emerge to 3TC and NNRTIs when regimens containing these drugs are not used effectively underscores the critical need to focus on adherence with these programs. NNRTI-based regimens are the most frequently used first-line regimens in North America and Europe;
however, PI-based regimens are necessary for patients with either acquired or transmitted NNRTI-resistant HIV infection.
The World Health Organization (WHO) recommends that, despite their cost, PI-based regimens be considered first-line regimens in populations where the prevalence of NNRTI resistance is greater than 5 to 10 percent (Hirsch et al., 2003). In the developed world, a drug resistance test would be used to select a regimen (Hirsch et al., 2003). Other reasons for considering a PI-based regimen as a first-line regimen include the presence of viral types with known insensitivity to NNRTIs (e.g., HIV-2 or HIV-1 group O) and situations in which the patient cannot tolerate NNRTIs. Although nevirapine, for example, is a potent NNRTI with demonstrated clinical efficacy, it is also associated with rash and hepatotoxicity, both of which can be severe and life-threatening (Ena et al., 2003; Law et al., 2003; Martin-Carbonero et al., 2003). Even mild rashes can cause poor adherence and treatment failure.
Given concerns about resistance, particularly with the WHO-recommended NNRTI-based regimens, high adherence rates will be absolutely critical to the success of ART scale up. There is no known reason not to expect that, if patients adhere to currently available triple-combination drug regimens, success rates could approach 80 to 90 percent (in terms of complete viral suppression), thus minimizing the rates of emergence of drug resistance over time. Yet while studies have shown that good adherence is quite feasible in resource-constrained settings under certain circumstances (e.g., in clinical settings where the studies are conducted) (Laniece et al., 2003; Orrell et al., 2003), the replicability of these results in other circumstances has been questioned.
Questions have been raised regarding the durability of NNRTI-versus PI-based regimens for ART scale-up in resource-constrained settings and the extent to which the WHO-recommended first-line NNRTI-based regimens appropriately address concerns about resistance (Redfield, 2004). How should concerns about resistance and the need for excellent adherence be balanced with the reality that the NNRTI-based regimens are otherwise the best choice for use in resource-constrained settings (i.e., with respect to toxicity, effectiveness, side effects, pill burden, and affordability) (Barreiro et al., 2002)? Such regimens have similarly been selected as the preferred first-line regimens in developed country settings.
Nevirapine Resistance: Implications for Mother, Child, and the Pandemic
While single-dose nevirapine may be a cost-effective and simple regimen for preventing HIV transmission from mother to child in resourceconstrained settings, concerns regarding nevirapine resistance have arisen
and may limit the drug’s future utility for this purpose (Jackson et al., 2000). In a recent study from Thailand, women with a history of limited nevirapine exposure for a single pregnancy had reduced rates of successful treatment responses subsequently with a treatment regimen containing nevirapine (Jourdain et al., 2004).
In a prior study conducted in Uganda, although 19 percent of the women and 46 percent of the infants who had received single-dose nevirapine had detectable nevirapine resistance at 6 weeks postpartum, resistance had faded from detection in all women and infants by 12 to 18 months (Eshleman et al., 2001). In this case, it is possible that drug resistance was not detected when the mother discontinued the nevirapine prophylaxis because drug-sensitive wild-type virus had reemerged in the viral population. Rather than having been eliminated, the nevirapine-resistant virus may simply have been “archived,” with the potential to reappear as a therapeutic challenge in the future when infected mothers and children were placed on therapy (Ekouvi et al., 2004).
Work in Cote d’Ivoire by Chair and colleagues revealed that 21 of 74 women had a detectable nevirapine resistance mutation at 4 weeks postpartum, as did 6 (23 percent) of the offspring evaluated. As reported, follow-up plasma and DNA-(PBMC) samples collected at 3 months (1 child) and until 12 months (1 child) revealed the persistence of NVP-resistance mutations (Ekouevi et al., 2004). Other studies have also documented high rates of nevirapine-resistance mutations (Gordon et al., 2004; Loubser et al., 2004).
Another factor to consider is the consequence of the emergence of nevirapine-resistant virus among different HIV subtypes, since the pattern of nevirapine-resistance mutations varies among subtypes. One recent study showed that women with subtype D had a higher rate of nevirapine resistance than women with subtype A (Eshleman et al., 2004a,b). Interestingly, nevirapine-resistance mutations differ between babies and their mothers, suggesting that resistance is not transmitted. Rather, babies have active, ongoing viral replication at the time of intra- and/or postpartum exposure to nevirapine.
Clearly, concern is rapidly growing that the use of ARVs that readily select for resistance and have long half-lives in settings where mother-to-child transmission is common and the support of potent combinations is lacking may compromise future treatment of mothers and children. The search for less problematic alternatives to prevent mother-to-child transmission should thus be a top priority. More needs to be known about the prevalence of nevirapine and 3TC resistance at the initiation of HAART in therapy-naïve but prophylaxis-experienced women and perhaps in their partners as well.
Modeling Resistance
Mathematical models have been used to describe and predict the HIV/ AIDS pandemic. Of particular interest for scale-up of ART in resourceconstrained settings is the implication of ARV usage rates for HIV incidence and the emergence of resistance. The prevalence of resistant HIV and the level of transmission of drug-resistant strains are a direct function of the number of persons treated and the amount of therapy provided (Blower and Farmer, 2003; Blower et al., 2000, 2001, 2003a,b; Velasco-Hernandez et al., 2002). Both high usage rates (as modeled in the United States) and low usage rates (as modeled in the developing world) have demonstrated this relationship. Since the ARV usage rate in Africa will be low (less than 10 percent of infected persons for at least the first several years), the level of transmitted resistance will be low.
It is true that modeling studies have shown that—as demonstrated in a U.S. population of gay men—high rates of usage can significantly reduce the total number of new HIV infections, but will result in increases in the prevalence of drug resistance (Blower and Farmer, 2003; Blower et al., 2000, 2003b). However, the low usage rates expected in Africa under even the best of circumstances suggest that in this setting, therapy will not have much impact on the total number of new HIV infections, and that for this reason, other interventions that support prevention must continue to receive a strong emphasis. According to these same modeling studies, most drug resistance is acquired, not due to transmission of resistant strains (Blower et al., 2003a). Finally, based on modeling studies, it is expected that the level of drug-resistant strains of HIV will first rise and then level off and stabilize. This phenomenon is expected because of the competitive dynamics between drug-resistant and drug-sensitive strains of HIV.
One modeling study (Blower et al., 2003a) considered the potential impact of the rate of ARV use on (1) the future prevalence of drug-resistant HIV, (2) the future transmission rate of drug-resistant strains of HIV, and (3) the cumulative number of HIV infections prevented over time by widespread use of ART. If 10 percent of the infected population is treated, the prevalence of ARV resistance can be as low as 4 percent after 10 years. If the proportion of individuals treated increases to 50 percent, the prevalence of ARV resistance can be as high as 53 percent after 10 years. The prevalence of drug resistance rises as risky behaviors increase, as the relative fitness (or “infectivity”) of ARV-resistant strains increases, and as the effectiveness of the ARV regimen in suppressing viral load decreases. In this same study, assuming that 10–50 percent of the infected population receives ART (again, a level of coverage not realistic anytime soon) and that the rate of emergence of acquired resistance varies from 20 to 70 percent, the models indicate the likelihood of very little transmitted resistance even
after 10 years of ARV usage (median of 5.9 percent of new cases expected to be drug-resistant). However, extremely high levels of transmission (greater than 40 percent) could occur in the following scenarios: increased ARV usage, greater transmissibility (or fitness) of the virus (resistant variants generally tend to be less transmissible than their wild-type relatives), increased risky behaviors, higher levels of acquired resistance, and increased use of ineffective treatment regimens (i.e., those that do not adequately suppress viral replication). The predicted relatively small impact of anticipated levels of ART on HIV incidence at the population level is illustrated in Figure 3-1.
Even under the best possible conditions, WHO’s 3-by-5 campaign aims to treat no more than 50 percent of the world’s HIV-infected population currently in need of treatment and a much smaller fraction (less than 10 percent) of all those HIV-infected. Thus it is unlikely that the growth of the HIV epidemic in sub-Saharan African will be curtailed to a major degree by the use of ART (Blower and Farmer, 2003; Blower et al., 2003a, 2004).
Based on the experiences and studies cited above, it would appear that appropriate interventions to minimize resistance include ensuring that patients with failed first-line regimens are treated with effective second-line
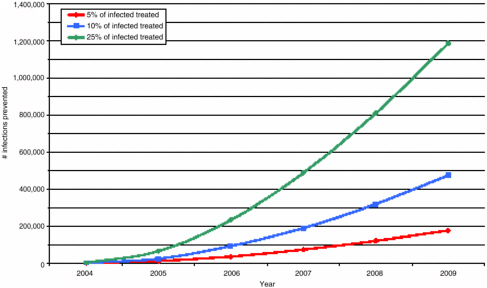
FIGURE 3-1 Potential for prevention of HIV infections by percent of HIV-infected persons under ART.
SOURCE: Sally Blower, David Geffen School of Medicine at UCLA.
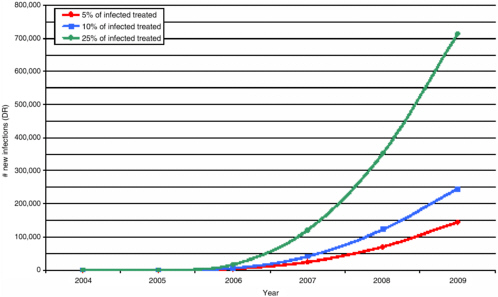
FIGURE 3-2 Drug-resistant new infections over time as a function of the percent of infected persons treated.
SOURCE: Sally Blower, David Geffen School of Medicine at UCLA.
regimens, that increases in risky behavior are prevented, and that high levels of adherence to effective regimens are maintained. Finally, because modeling studies demonstrate increased emergence of HIV resistance only with implausibly high levels of use of ART, widespread emergence of resistance may not occur during ART scale-up, when treatment rates will be relatively low (see Figure 3-2).
IMPLICATIONS OF RESISTANCE FOR SCALE-UP
While HAART has improved the lives of thousands with HIV/AIDS, HAART regimens are complex and as such have the potential for failure. As emphasized in this chapter, one reason for failure that is of particular concern as planning occurs for wide-scale introduction of ART into resource-constrained settings is the emergence of drug resistance.
The emergence of drug resistance is a concern mainly for the individual patient being treated, but also has implications for the durability of standard treatment regimens for the population as a whole. Second-line drug regimens needed once resistance to first-line regimens has developed may be unavailable or unaffordable in resource-limited settings. Moreover, resistance detection using laboratory tests will be of highly limited availability in resource-constrained settings.
While aggressive and innovative price-reduction mechanisms have lowered the cost of many ARVs, the drugs needed for a second-line ART regimen should initial treatment fail are particularly expensive. According to a November 2003 Médecins sans Frontières report, costs for second-line regimens average US$1,203 in South Africa, $1,875 in Malawi, $1,594 in Kenya, and $4,763 in Cameroon, versus $400–1,000, $288, $1,594, and $292, respectively, for first-line drugs (Médecins sans Frontières, 2003b). Not only are these second-line regimens more expensive, but their increased complexity with regard to dosing, scheduling, and monitoring requirements makes them a difficult choice for resource-constrained settings. As suggested above, moreover, resistance may go undetected in resource-constrained settings, where resistance testing may be unavailable because of costs and technical and infrastructure needs. Thus while the many lessons learned in the developed world about the causes and implications of drug resistance should guide the use of ART in resource-limited settings, the resource-intensive, laboratory-focused monitoring for drug resistance that is typical in wealthy countries may not, at this point in time, be as appropriate or necessary for more resource-constrained settings.
Although the emergence of some degree of drug resistance is inevitable, as is common with most drug-treated pathogens, widespread resistance should not be a necessary consequence of a broad treatment program. As noted earlier, despite years of (now-recognized) inadequate mono and dual ART in the United States and Europe, no cases of drug-resistant HIV transmission were recognized until the mid-1990s (Little, 2000). Thus even if ART scale-up in resource-constrained settings is conducted poorly, widespread population- or epidemic-level resistance will not likely emerge with HAART for several years. In part this is because only a small fraction of the 40 million people worldwide infected with HIV will actually be exposed to ART in the next several years; thus, HIV transmission will be largely from persons with susceptible virus. Although there are very limited data on the prevalence of resistant viral strains, even in the United States, one recent South African study showed that among 37 drug-näive HIV-positive women screened in an antenatal clinic, none had any of 23 mutations known to be associated with ARV resistance, suggesting that even after 3 years of ART, resistance is still low to undetectable at the level of untreated patients (Pillay et al., 2002).
At the same time, the possibility that higher levels of resistance and increases in transmitted resistance could eventually emerge, although not viewed as a major issue for the near term, raises the question of when general clinical screening for drug-resistant strains should be implemented. The question also arises of what should be done now, short of laboratory-intensive resistance screening, to predict and prepare (in terms of surveillance) for the emergence of a higher level of resistance should it occur.
Certain measures can be taken now to monitor conditions that foster emergence without implementing extensive, formal, laboratory-intensive resistance surveillance systems. WHO’s Global HIV Resistance Surveillance Network (HIV Resnet), a group of global experts convened to identify and implement resistance-evaluation methods, has developed a non–laboratory-based early warning system. This system is based on the use of “indicators,” most of which consist of various data being collected for other purposes that can provide indirect but helpful indications of situations in which resistance is likely to emerge. For example, given that the use of substandard treatment regimens (e.g., monotherapy) has been associated with resistance, data on the use of such regimens, such as prescription records from pharmacies, could be used to alert program managers of conditions that foster resistance. The relative yield of different laboratory and nonlaboratory markers for identifying an emerging problem with drug resistance should be considered an important topic for operations research.
Meanwhile, few studies have measured population-level resistance at a large scale in areas where ART has already been available for many years, even in the United States. Most studies involve insufficient sample sizes to allow accurate estimation of the prevalence of resistant virus within the larger HIV-infected population. Moreover, the proportions of risk groups in most study samples do not represent the actual proportions in the HIV population at large. For example, among the widely cited population-level studies of transmitted resistance, the overwhelming majority of subjects within the study samples are white men who have sex with men (Boden et al., 1999; Grant et al., 2002; Little et al., 2002; Simon et al., 2002). Yet male-to-male sexual contact now accounts for only about 40 percent of new HIV diagnoses in the United States, while fewer than 30 percent of newly diagnosed individuals in the United States are white (CDC, 2004). Large, representative cohort studies are needed to derive more accurate estimates of the prevalence of HIV drug resistance and achieve a better understanding of population factors that impact such prevalence.
Recommendation 3-1. For individual patients about to embark on therapy, general clinical screening for resistance to antiretroviral drugs is not recommended at this time for two reasons: because the prevalence of resistance in HIV-infected individuals not previously exposed to antiretroviral therapy is expected to be undetectable or low, and because the proportion of total persons with HIV who are receiving therapy in a given country will also be relatively small in the short term. Coordinated, systematic testing for resistance to antiretroviral drugs should, however, be conducted among a subset of patients failing treatment. These latter results will be critical in evaluating ART
programs and in determining whether and when routine population-based resistance testing might eventually prove effective. Sentinel surveillance of treatment-naïve HIV-positive persons may also be indicated in the future.
LEARNING BY DOING: SCALE-UP AROUND THE WORLD
This section reviews lessons learned from ART scale-up in five countries: Brazil, Haiti, South Africa, Nigeria, and Uganda.
Brazil
Brazil, with a per capita income of about US$3,000, is much less resource-constrained than most of sub-Saharan Africa, the Caribbean, and other countries being targeted for current ART scale-up programs, and its national HIV prevalence rate is much lower. Nontheless, the success of that country’s ART program—considered the most advanced such national program in the developing world—serves as an important example of how sound policy can profoundly improve treatment and prevention efforts, save lives and money, and halt the expansion of an epidemic.
In 1996, the Brazilian public health system began widespread and free provision of ARVs for all citizens. Late in 1996, the Brazilian president signed a law establishing this free distribution. By 2001, public and private laboratories were manufacturing 63 percent (43 percent of expenditures) of the ARVs available to Brazilians (Galvão, 2002). Seven of the 13 ARVs being used to treat Brazilians are domestically produced; others have been procured by the government on the international market through aggressive negotiation of prices. As one example, the threat to break patent law for efavirenz resulted in Merck, the drug’s producer, reducing its price by 60 percent (Galvão, 2002). Overall, Brazil saw the median cost of ART drop 67 percent from 1997 to 2002 (Schechter, 2004). In part because of Brazil’s leadership in addressing patent law, in 2001 the UN Human Rights Commission announced that access to medical drugs during pandemics is a human right.
The ARVs in Brazil are distributed by 486 centers located in public health hospitals or health centers. A structural component integral to the distribution effort is the Unified Health System, established in 1988. This system provides free comprehensive care to the entire population of Brazil.
An estimated 35,900 Brazilians with HIV/AIDS received medication in 1997, 1 year after program initiation. In 2003, with 280,000 cases of AIDS and 600,000 cases of HIV infection, Brazil provided 128,000 people with medication (Lima, 2004). (It is predicted that 148,500 persons with HIV/
AIDS will receive therapy in 2004.) In addition, because of free access to effective therapy, more Brazilians have been tested for HIV, and epidemiologic surveillance of the disease has improved. As an example, in 1996 there was a 40 percent increase in the number of AIDS cases reported. From 2000 to 2002, epidemiologic surveillance revealed a decline in HIV incidence from 17,504 to 7,361 cases (Boletin Epidemiologico, 2004).
There are many reasons for Brazil’s success in addressing its HIV/AIDS epidemic. Community advocacy and the government’s leadership are particularly noteworthy (Levi and Vitória, 2002). Especially active in the process have been religious organizations, sexual minorities, and organizations representing persons living with HIV/AIDS. The Brazilian government acknowledged the problem, and citizens participated in committees designing the country’s response. Following passage of the law guaranteeing free, universal ART in 1996, an independent advisory committee established national treatment guidelines and criteria, and the treatment program was integrated into the existing health care infrastructure.
Early on in program implementation, prevention and treatment initiatives were integrated. In the 1990s, it was predicted that there would be over 1 million cases of HIV/AIDS in Brazil by 2000. The actual number of cases in 2000 was only about half that figure, largely as a result of wide-scale prevention efforts (Schechter, 2004). One successful prevention initiative was a significant increase in the use of condoms, as demonstrated by minimal use in the early 1980s and a rate of use at first intercourse of over 70 percent by the late 1990s (Schechter, 2004).
A unique feature of the Brazilian program that has contributed to its success is the use of information technology for program management (Lima, 2004). Two computerized systems were developed to assist in monitoring patient care (see the discussion in Chapter 5).
Government spending for ART and HIV/AIDS care in Brazil increased from US$34 million in 1996 to $232 million in 2001. Because of a substan-
|
The Brazilian Experience: Summary of Lessons Learned
|
tial reduction in opportunistic infections and hospital admissions, this increased spending has been cost-effective. The Brazilian Ministry of Health estimates cost savings of over US$1 billion (Galvão, 2002).
Haiti
Haiti is one of the world’s poorest countries, with an annual per capita income of around US$440 (World Bank, 2004). The estimated prevalence of HIV among adults in Haiti is 6 percent (Fitzgerald, 2004; WHO, 2003a). About 60 percent of HIV-infected people live in Port-au-Prince and surrounding urban areas, while the other 40 percent live in the rural country-side.
Pre-HAART cohort studies in Haiti showed that the estimated survival time from HIV infection to death was 7½ years for adults and less than 1 year for newborns—both of these rates being dramatically accelerated compared with people living with HIV/AIDS in the pre-HAART era in the United States and Europe (Fitzgerald, 2004). Despite these alarming statistics, however, the HIV epidemic in Haiti has not doubled in size, as was predicted in the mid-1990s. Indeed, seroprevalence data indicate that the epidemic is contracting rather than expanding (Fitzgerald, 2004; UNAIDS, 2002).
Two noteworthy initiatives have been established in Haiti to help address the epidemic. In urban Haiti, the Haitian Study Group on Kaposi’s Sarcoma and Opportunistic Infections, commonly known as GHESKIO, was established in 1982 with the support of the Cornell University Medical College (GHESKIO, 2004). In rural Haiti, the HIV Equity Initiative was established in 1998 with assistance from the Boston-based Partners in Health, which is affiliated with Harvard Medical School (WHO, 2003a).
Urban Haiti: GHESKIO
The GHESKIO Center operates in a central slum district in Port-au-Prince. Patients visit the clinic each month. Some patients are counseled more aggressively and are visited at home. Patients are followed by a physician every 3 months and by a nurse every month. Therapy is monitored by clinical signs and symptoms, as well as routine CD4 counts at baseline and every 6 months. Adherence is ensured through a team approach that includes pharmacists, community health workers, and a growing number of patients who are employed as social workers and peer educators (Fitzgerald, 2004). The GHESKIO program provides comprehensive care. One component of the program is an evaluation of patients’ nutritional needs and the provision of nutritional support when necessary. Although physicians take a very active role during the first 3 months of ART, the frequency of contact
is reduced as treatment progresses. The ownership in this program with respect to care belongs to nurses, social workers, pharmacists, and community health workers.
As of January 2004, 972 patients were on ART under the program (Fitzgerald, 2004). The first-line regimen comprises zidovudine, lamivudine, and efavirenz. Pregnant women are given nevirapine instead of efavirenz. Fully 75 percent of all patients had clinical AIDS at the start of therapy, and the median CD4 count was 114. Approximately 10 percent of patients have required a regimen change because of drug toxicity (anemia and rash are the most common complications). During a 1-year follow-up examination, mortality was found to be 6 percent; many of these deaths were among patients with advanced disease at the time therapy was initiated (Fitzgerald, 2004).
Rural Haiti: The HIV Equity Initiative
Partners in Health’s HIV Equity Initiative is frequently cited as a remarkable success story in the introduction and implementation of ART in a resource-constrained setting (WHO, 2003a; Farmer et al., 2001a,b). Its mission was to provide integrated preventive and comprehensive treatment services to those living with HIV/AIDS, using the already successful directly observed therapy (DOT) model for tuberculosis. The aim was to deliver HAART to those people in the direst need of therapy—the sickest and closest to death from AIDS. The clinical facility in Cange, Clinique Bon Sauveur, was set up in a province with no electricity and limited road access, and began to offer HAART in 1999. Within the first 2 years of the program’s establishment, the availability of HAART contributed to a more than 300 percent increase in use of HIV counseling and testing services (Farmer et al., 2001b; Walton et al., 2004).
The HIV Equity Initiative in Haiti harnessed the capacity of human infrastructure and trained a cadre of community health workers in the administration and follow-up of ART. These workers, called accompagnateurs, provided HAART and served as an essential link between those with HIV/AIDS in the community and the resources of the Clinique Bon Sauveur, which is more than 5 hours away by foot for some of those served. Building on the success of the directly observed therapy short course (DOTS) for tuberculosis, HAART was provided by directly observed interventions.
Prior to 2002, initiation of therapy was based on clinical status. Patients were selected for DOT-HAART if they met the following criteria (WHO, 2003a):
-
Absence of active tuberculosis
-
Recurrent opportunistic infections that were difficult to manage with antibacterial or antifungal agents
-
Chronic enteropathy with wasting
-
Otherwise unexplained significant weight loss
-
Severe nervous system complications attributable to HIV disease
-
Severe leucopenia, anemia, or thrombocytopenia
Laboratory determination of HIV serology, hematocrit, and white blood cell count was prerequisite to beginning HAART. Of note, a CD4 determination was not necessary. If symptoms of gastrointestinal side effects, anemia, or thrombocytopenia developed, a liver function test or complete blood count was ordered as appropriate. To monitor response to therapy, again a CD4 determination was not the tool. Rather, the patient’s weight, a studied predictor of survival and disease progression, was used.
Patients enrolled in the HIV Equity Initiative received a three-drug regimen consisting of stavudine, lamivudine, and nevirapine.1 Approximately 10 percent of patients initiated on this regimen switched regimens because of toxicity (Fitzgerald, 2004). In their three-drug regimens, pregnant women received zidovudine instead of stavudine, and patients with tuberculosis received efavirenz instead of nevirapine.
While two physicians assessed persons with HIV/AIDS, it was the accompagnateurs who provided the care. For those on HAART, an accompagnateur would visit daily and observe the patients taking their medications. In some cases, a second daily visit would occur to observe subsequent taking of medication. While visiting patients, the accompagnateurs would provide educational and emotional support. They also were trained in recognizing complications of the disease and its therapies; as complications arose, patients would be referred back to the Clinique Bon Sauveur for a more skilled evaluation. Routinely, however, patients treated through the HIV Equity Initiative were followed up in clinic on a monthly basis. In addition to daily visits by accompagnateurs, efforts to improve adherence to HAART included assessments of the patients’ living quarters, social support system, and financial system. Monthly meetings involving people living with HIV/AIDS also were held to facilitate information exchange and provide support.
The program has been lauded for reducing AIDS-associated stigma and for achieving success in reducing mortality from HIV/AIDS in a rural area without electricity and paved roads at low cost. All services provided
through the HIV Equity Initiative (e.g., laboratory monitoring, home visits, medical consultations) were provided free of charge.
The Partners in Health program currently provides ART to about 700 patients living with HIV/AIDS. The majority of these patients now have undetectable viral loads (86 percent of a small subset of 65 patients whose viral loads were measured with at least 3 months of follow-up) and are able to live normal, active lives. One-year mortality is about 5 percent, with almost all of this mortality associated with very advanced disease (i.e., severe wasting and low CD4 counts) at the initiation of ART (Fitzgerald, 2004).
With the international and multilateral financial support that has been provided and is forthcoming, the Partners in Health program hopes to provide comprehensive care for HIV and tuberculosis on a much larger scale in Haiti. Several strategies are being employed to make the scale-up feasible. ART as a fraction of program costs has decreased from 80 percent to below 15 percent because of three endeavors: purchase of drugs from WHO-prequalified manufacturers, concessional pricing agreements with pharmaceutical companies, and collaboration with a wholesale distributor of essential medicines based in the Netherlands (WHO, 2003a). Also, in anticipation of further scale-up of treatment, Partners in Health has established a logistical monitoring system that includes web-based medical records, an application allowing for off-line data entry, and a system to monitor drug inventory.
Summary
GHESKIO and the HIV Equity Initiative provide ART to over 1,600 people living in resource-constrained settings and serve as helpful examples for ART scale-up. As the result of two national conferences attended by representatives from the Haitian Ministry of Health, U.S. and European
|
The Haitian Experience: Summary of Lessons Learned
|
universities, and people living with HIV/AIDS, Haitian national guidelines based on the WHO-recommended regimens have been established. Appropriate modifications for strengthening adherence have been made in accordance with lessons learned in both Port-au-Prince and rural Haiti.
South Africa
Khayelitsha, South Africa, is a resource-poor, periurban township just outside of Cape Town. It has a population of approximately 500,000, 50,000 of whom are estimated to be HIV-infected (WHO, 2003b). The majority of people in the township live in informal housing such as shacks built from corrugated iron, and many live without electricity and running water. With the help of Médecins sans Frontières (see Box 3-2), HIV/AIDS
|
BOX 3-2 Médecins sans Frontières (MSF) is an international, independent medical humanitarian organization founded in 1971. With the help of 2,500 international volunteers per year and 15,000 national staff, it currently has 400 relief projects ongoing in 80 countries. Because of its efforts worldwide, MSF received the Nobel Peace Prize in 1999. One activity in which MSF is engaged is the provision of ART and HIV/AIDS care to countries in need. All of MSF’s ART programs, the first of which was initiated in 2001, are pilot projects designed to demonstrate the efficacy and feasibility of ART in resource-poor settings. Currently there are 42 ART programs in 19 countries throughout Africa, Asia, Latin America, and the former Soviet Union serving some 11,000 people on ART. Key components of MSF-sponsored programs for HIV/AIDS are as follows:
SOURCE: Cohen, 2004. |
clinics were set up within community health centers in Khayelitsha in 2000, and in 2001, ART began to be provided at these clinics to patients in advanced stages of the disease. The clinics provide comprehensive care for patients with HIV/AIDS and were initially staffed in each case by one physician, one nurse, and one lay counselor. Additional staff members have since been added, and the clinics are now more heavily nurse based. They currently serve more than 1,800 patients per month among three clinics (WHO, 2003b).
Eligibility for ART is based on both clinical and social indicators; ability to adhere to therapy is also a factor. To be clinically eligible, patients must have CD4 cell counts of less than 200/mm3 and Stage III or IV disease based on WHO criteria. (See Appendix C for the WHO guidelines.) A clinic staff member visits each patient’s home to assess the social support network. Potential ability to adhere to therapy is based on an assessment of adherence to cotrimoxazole prophylaxis and tuberculosis treatment, as well as regular attendance at HIV clinics in the period leading up to the beginning of treatment (WHO, 2003b; Médecins sans Frontières, 2003a). The first-line treatment regimen is stavudine, lamivudine, and either efavirenz or nevirapine. In the case of the latter two drugs, the decision on which to use takes into account whether a patient is also being treated for tuberculosis, has indications of abnormal liver function, or is pregnant. Patients receive clinical assessments either weekly or biweekly for the first 2 months of treatment, after which they are evaluated on a monthly basis.
By the end of 2003, 776 individuals had been started on ART. A full analysis of the 287 treatment-naïve adult patients who initiated therapy during the first year and a half of the program has been performed (Médecins sans Frontières, 2003a). Among these 287, HIV RNA levels were reduced to undetectable in 89.2 percent of patients at 6 months and remained undetectable in 84.2 percent of patients at 12 months, 75.0 percent at 18 months, and 69.7 percent at 24 months (Coetzee et al., 2004). Patients showed a median weight gain of 5.0 kg and 9.0 kg at 6 and 12 months, respectively, as well as a median gain in CD4 cell count of 288 cells/mm3 after 24 months. The survival rate at 24 months was 86.3 percent for all patients and 91.4 percent for patients who began treatment with a CD4 count greater than 50 cells/mm3 (Coetzee et al., 2004). Nearly three-fourths of patient deaths occurred in the first 3 months after treatment initiation.
The Khayelitsha program has also witnessed encouraging rates of both patient retention and adherence to therapy. Of the patient cohort just described, only one individual was entirely lost to follow-up by July 2003, while a dozen more either moved to a different province or stopped therapy for other reasons. Separately, a prospective study of self-reported adherence to ART regimens found that 87.5 percent of patients reported “high” levels
of adherence (having taken at least 95 percent of their drug doses in a 4-day recall period) at 1 month, 89.0 percent at 3 months, and 87.7 percent at 12 months (Médecins sans Frontières, 2003a). To achieve these levels of adherence, Médecins sans Frontières has promoted a dual strategy of simple, standardized pill regimens and a robust patient education program combining several elements (Médecins sans Frontières, 2003a; WHO, 2003b):
-
Individual support—Patients are required to identify another individual close to them (such as a friend or other household member) who will act as a treatment assistant, accompanying them during the enrollment process and learning basic ART principles. Lay counselors are also provided to assist with individual adherence plans.
-
Peer support—Clinics host regular meetings for individuals on ART, where participants can discuss adherence barriers, adverse events, and community and psychosocial issues, such as disclosing their condition to others.
-
Material support—Individuals receiving ART are provided with educational materials and supplies, including pillboxes, schedules, drug-related information charts, and educational materials such as pamphlets and newsletters on the risks and benefits of ART.
The success of the program in Khayelitsha has been the result of a number of efforts. Médecins sans Frontières played a pivotal role in guiding the program and procuring affordable ARVs, and the Treatment Action Campaign, a grassroots advocacy group, was instrumental in mobilizing the community to be aware of HIV as a political issue and pressuring the government to address it. The latter group has also established education programs to combat the stigma and discrimination associated with the disease and to promote its prevention and treatment.
Nigeria
As of 2001, there were an estimated 3.5 million HIV-infected Nigerians, including an estimated 1.2 million with symptomatic AIDS. In 2002, the government of Nigeria initiated a nationwide ART program with the goal of treating 10,000 adults and 5,000 children with HIV (Idoko, 2004). Because there had been no pilot initiatives on which to base programmatic policies and guidelines, the program was based largely on the WHO recommendations. Decisions on exceptions to those recommendations were made by a committee. Treatment eligibility, for example, was based on a CD4 count below 350 cells/mm3, given that many patients with CD4 counts above 200 still suffer significant immunological damage. The priority is to treat symptomatic patients.
The first-line regimen is a WHO-recommended combination of generic forms of lamivudine, stavudine, and nevirapine. Thus far, this regimen has proven potent, while its pill burden has proven acceptable. Within 12 months following the initiation of therapy, patients were gaining weight; CD4 cell counts were increasing; and, based on viral load testing at one center, about 80 percent of patients had fully suppressed viral loads (Idoko, 2004). Major adverse events that occurred with this treatment regimen included hepatotoxicity (reported as most likely from nevirapine) and a case of severe Stevens-Johnson syndrome. Other concerns with the use of this treatment regimen in Nigeria include the following:
-
The risk of resistance to nevirapine, which is also used for prevention of mother-to-child transmission
-
Tuberculosis coinfection (about 50 percent) and drug–drug interactions with the first-line regimen—particularly nevirapine, which can cause hepatotoxicity
-
Hepatitis B coinfection
-
Hepatitis C coinfection
-
Price differentials between public and private purchasers
Despite these concerns, scale-up of ART has proceeded. The greatest challenges faced by Nigeria have been the establishment of a regular, continuous drug supply and the delayed procurement of second-line drugs due to delays in receipt of donor monies.
Uganda
Over the past decade and a half, Uganda has witnessed remarkable declines in HIV prevalence among its population. Surveillance data from antenatal clinics in many cases show prevalence rates as high as 20 to 30 percent in 1991 declining to 10 percent or lower just 10 years later (Uganda AIDS Commission, 2003). Despite declining rates of new infections, however, the country continues to face serious challenges, including a total number of HIV-infected individuals estimated at 600,000, many of whom are progressing to AIDS (WHO, 2003c). In an effort to improve access to ART in resource-constrained settings, UNAIDS launched a pilot project—the HIV Drug Access Initiative—to introduce structured therapy programs in four countries: Uganda, Cote d’Ivoire, Chile, and Vietnam (Katzenstein et al., 2003).
When the Uganda initiative began in June 1998, five health care facilities were accredited to provide ART, all of them located in or around Kampala. As the program has expanded in recent years, more facilities have
been accredited so that by 2003, a total of 23 facilities, including six regional hospitals, were providing ART (WHO, 2003c).
In the program’s initial stages, only four NRTIs and one PI were available, but by February 2000, the number of available treatments had grown to six NRTIs, four PIs, and three NNRTIs (Weidle et al., 2002). These drugs were initially employed in one of two alternative approaches. The first targeted complete suppression of viral replication and used three-drug combination therapy regimens, including two NRTIs and either a PI or an NNRTI. The second approach, which is no longer recommended, targeted partial viral suppression and focused on the use of two NRTIs, with the option of adding hydroxyurea. Given that many people could not afford the triple-drug combination, the partially suppressive therapy was felt to be justified in Uganda at the program’s outset and was recommended on the grounds that partial viral suppression was better than none (WHO, 2003c). As a result of significant decreases in the cost of ART, however, more individuals have been treated with triple-drug regimens since 2000.
There are two primary suppliers of ARVs in Uganda: the Joint Clinical Research Centre and Medical Access Uganda Limited. The latter organization was established under the UNAIDS Drug Access Initiative as a not-for-profit company that would procure drugs at reduced cost from participating pharmaceutical companies and then sell these drugs to treatment centers accredited by Uganda’s National Advisory Board (WHO, 2003c). Four multinational pharmaceutical groups have contributed to the company’s operating costs: Glaxo-Wellcome, Bristol Myers Squibb, Roche Products Ltd., and Merck Sharpe and Dohme. In this manner, Medical Access Limited has become Uganda’s principal supplier of ARVs from the pharmaceutical industry. By comparison, the Joint Clinical Research Centre is now the country’s principal supplier of generic ARVs. It was first established through collaborative efforts between Uganda’s Ministry of Health, Makerere University, and the Ministry of Defence and began importing generic ARVs in 2000 (WHO, 2003c).
In 1997, prior to the beginning of the Drug Access Initiative, 450 people were enrolled in ART programs in Uganda. With the initiative’s assistance, this figure rose to 900 by 2000, when the UNAIDS pilot project came to an end. Early clinical assessments of patients treated through the Uganda initiative have quantified the project’s efficacy and illustrated the extent to which well-organized drug procurement and distribution programs can have an impact even in such resource-constrained settings (Weidle et al., 2002, 2003; Katzenstein et al., 2003). After the project’s end, further developments, including substantial reductions in drug prices and governmental efforts to train more staff and accredit additional clinical centers, helped raise the number of treated individuals to 3,000 by the end of 2001
(WHO, 2003c). This rapid escalation has continued, with an estimated 17,000 Ugandans receiving ART by the end of 2003 (Kamya, 2003).
|
The U.S. Experience: Summary of Lessons Learned
|
REFERENCES
Barreiro P, Garcia-Benayas T, Soriano V, Gallant J. 2002. Simplification of antiretroviral treatment: How to sustain success, reduce toxicity and ensure adherence avoiding PI use. AIDS Review 4:233–241.
Blower SM, Farmer P. 2003. Predicting the public health impact of antiretrovirals: Prevention in developing countries. AIDS Science 3(11).
Blower SM, Gershengorn HB, Grant RM. 2000. A tale of two futures: HIV and antiretroviral therapy in San Francisco. Science 287:650–654.
Blower SM, Aschenbach AN, Gershengorn HB, Kahn JO. 2001. Predicting the unpredictable: Transmission of drug-resistant HIV. Nature Medicine 7:1016–1020.
Blower SM, Ma L, Farmer P, Koenig S. 2003a. Predicting the impact of antiretrovirals in resource poor settings: Preventing HIV infections whilst controlling drug resistance. Current Drug Targets-Infectious Disorders 3:345–353.
Blower SM, Schwartz EJ, Mills J. 2003b. Forecasting the future of HIV epidemics: The impact of antiretroviral therapies and imperfect vaccines. AIDS Review 5:113–125.
Blower SM, Bodine E, Kahn J, McFarland W. 2004. The impact of the antiretroviral rollout on drug resistant HIV in Africa: Insights from empirical data and theoretical models. AIDS (in press).
Boden D, Hurley A, Zhang L, Cao Y, Guo Y, Jones E, Tsay J, Ip J, Farthing C, Limoli K, Parkin N, Markowitz, M. 1999. HIV-1 drug resistance in newly infected individuals. Journal of the American Medical Association 282(12):1135–1141.
Boletin Epidemiologico. 2004. AIDS. [Online]. Available: http://www.aids.gov.br/ [accessed August 24, 2004].
CDC (Centers for Disease Control and Prevention). 2004. HIV-AIDS Surveillance: General Epidemiology. [Online]. Available: http://www.cdc.gov/hiv/graphics/surveill.htm [accessed August 2, 2004].
Clavel F, Hance A. 2004. HIV drug resistance. New England Journal of Medicine 350(10): 1023–1035.
Coetzee D, Hildebrand K, Boulle A, Maartens G, Louis F, Labatala V, Reuter H, Ntwana N, Goemaere E. 2004. Outcomes after two years of providing antiretroviral treatment in Khayelitsha, South Africa. AIDS 18(6):887–895.
Cohen R. 2004. Positive Replication: The MSF Experience Providing HAART in Developing Countries. Paper presented at the Institute of Medicine Workshop on Antiretroviral Use in Resource Constrained Settings, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Deeks S. 2003. Treatment of antiretroviral-drug-resistant HIV-1 infection. Lancet 362:2002–2011.
Ekouevi DK, Peyatvin G, Rouet F, Bequet L, Montcho C, Viho I, Fassinou P, Leroy V, Dabis F, Rouzioux C, Ditrame Plus Tsudy Grou. 2004. Persistence of nevirapine-resistant virus and pharmacokinetic analysis in women who received intrapertum NVP associated to a short course of zidovudine (ZDV) to prevent HIV-1 transmission: The Ditrame Plus ANRS 1201/02 Study, Abidjan, Cote d’Ivoire. Abstract 160. Antiviral Therapy 9:S176.
Ena J, Amador C, Benito C, Fenoll V, Pasquau F. 2003. Risk and determinants of developing severe liver toxicity during therapy with nevirapine-and efavirenz-containing regimens in HIV-infected patients. International Journal of STD & AIDS 14(11):776–781.
Eshleman SH, Mracha M, Guay LA, Deseyve M, Cunningham S, Mirochnick M, Musoke P, Fleming T, Glenn Fowler M, Mofenson LM, Mmiro F, Jackson JB. 2001. Selection and fading of resistance mutations in women and infants receiving nevirapine to prevent HIV-1 vertical transmission (HIVNET 012). AIDS 15:1951–1957.
Eshleman SH, Wang J, Guay LA, Cunningham SP, Mwatha A, Brown ER, Musoke P, Mmiro F, Jackson JB. 2004a. Distinct patterns of selection and fading of K103N and Y181C are seen in women with subtype A vs D HIV-1 after single dose nevirapine: HOVNET 012. Abstract 50. Antiviral Therapy 9:S59.
Eshleman SH, Guay LA, Mwatha A, Brown ER, Cunningham SP, Musoke P, Mmiro F, Jackson JB. 2004b. Characterization of nevirapine resistance mutations in women with subtype A vs. D HIV-1 6–8 weeks after single-dose nevirapine. HIV Journal of Acquired Immune Deficiency Syndromes 35(2):126–130.
Farmer P, Léandre F, Mukherjee J, Claude M, Nevil P, Smith-Fawzi M, Koenig S, Castro A, Becerra M, Sachs J, Attaran A, Kim JY. 2001a. Community-based approaches to HIV treatment in resource-poor settings. Lancet 358:404–409.
Farmer P, Léandre F, Mukherjee J, Gupta R, Tarter L, Kim JY. 2001b. Community-based treatment of advanced HIV disease: Introducing DOT-HAART (directly observed therapy with highly active antiretroviral therapy). Bulletin of the World Health Organization 79(12):1145–1151.
Fischl MA. 1999. Antiretroviral therapy in 1999 for antiretroviral-naïve individuals with HIV infection. AIDS 13(Supplement 1):s49–s59.
Fitzgerald D. 2004 (January 27). Lessons Learned from the Use of ARVs in Very LowResource Settings: The Haiti Experience. Paper presented at the Institute of Medicine Workshop on Antiretroviral Scale-up in Resource Constrained Settings, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Galvão J. 2002. Access to antiretroviral drugs in Brazil. Lancet 360(9348):1862–1865.
GHESKIO (Haitian Study Group on Kaposi’s Sarcoma and Opportunistic Infections). 2004. Homepage. [Online]. Available: http://www.haitimedical.com/gheskio/ [accessed July 1, 2004].
Gordon M, Graham N, Bland R, Rollins N, De Oliveira T, Monosi B, Van Laethem K, Vandamme A, Cassol S. 2004. Surveillance of resistance in KZN South Africa, including mother-infant pairs 6 weeks after single-dose NVP. Abstract 71. Antiviral Therapy 9:S80.
Grant RM, Hecht FM, Warmerdram M, Liu L, Liegler T, Petropoulos CJ, Hellmann NS, Chesney M, Busch MP, Kahn JO. 2002. Time trends in primary HIV-1 drug resistance among recently infected persons . Journal of the American Medical Association 288(2): 181–188.
Hirsch MS, Conway B, D’Aquila RT, Johnson VA, Brun-Vezinet F, Clotet B, Demeter LM, Hammer SM, Jacobsen DM, Kuritzkes DR, Loveday C, Mellors JW, Vella S, Richman DD. 1998. Antiretroviral drug resistance testing in adults with HIV infection. Journal of the American Medical Association 279:1984–1991.
Hirsch MS, Brun-Vezinet F, D’Aquila RT, Hammer SM, Johnson VA, Kuritzkes DR, Loveday C, Mellors JW, Clotet B, Conway B, Demeter LM, Vella S, Jacobsen DM, Richman DD. 2000. Antiretroviral drug resistance testing in adult HIV-1 infection. Journal of the American Medical Association 283:2417–2426.
Hirsch MS, Brun-Vezinet F, Clotet B, Conway B, Kuritzkes DR, D’Aquila RT, Demeter LM, Hammer SM, Johnson VA, Loveday C, Mellors JW, Jacobsen DM, Richman DD. 2003. Antiretroviral drug resistance testing in adults infected with human immunodeficiency virus type 1:2003 recommendations of an International AIDS Society-USA panel. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America 37:113–128.
Idoko J. 2004. The 2003 WHO Guidelines for ARV Use: Perspectives from a User. Paper presented at the Institute of Medicine Workshop on Antiretroviral Scale-up in Resource Constrained Settings, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Jackson JB, Becker-Pergola G, Guay LA, Musoke P, Mracna M, Fowler MG, Mofenson LM, Mirochnick M, Mmiro F, Eshleman SH. 2000. Identification of the K103N mutation in Ugandan women receiving nevirapine to prevent HIV-1 vertical transmission. AIDS 14:FT111–FT115.
Jourdain G, Ngo-Giang-Huong N, Tungyai P, Kummee A, Bowonwatanwong C, Kantipong P, Lechanachai P, Hammer S, Lallemant M; Perinatal HIV Prevention Trial Group. 2004 (February 8–11). Exposure to Intrapartum Single-Dose Nevirapine and Subsequent Maternal 6-Month Response to NNRTI-Based Regimens. Paper presented at the 11th Conference on Retroviruses and Opportunistic Infections, San Francisco, CA.
Kamya M. 2003. The National Strategic Framework for HIV/AIDS Activities in Uganda: 2000/1-2005/6. Mid-Term Review Report—Theme 2: Care and Treatment Technical Working Group. [Online]. Available: http://www.aidsuganda.org/pdf/Annex_2_TWG_2_Report.pdf [accessed August 6, 2004].
Katzenstein D, Laga M, Moatti JP. 2003. The evaluation of the HIV/AIDS drug access initiatives in Côte d’Ivoire, Senegal and Uganda: How access to antiretroviral treatment can become feasible in Africa. AIDS 17(Supplement 3):S1–S4.
Laniece I, Ciss M, Desclaux A, Diop K, Mbodj F, Ndiaye B, Sylla O, Delaporte G, Ndoye I. 2003. Adherence to HAART and its principal determinants in a cohort of Senegalese adults. AIDS 17:S103-S108.
Law W, Dore G, Duncombe C, Mahanontharit A, Boyd M, Ruxrungtham K, Lange J, Phanuphak P, Cooper D. 2003. Risk of severe hepatotoxicity associated with antiretroviral therapy in the HIVNAT cohort, Thailand, 1996–2001. AIDS 17(15):2191–2199.
Leigh Brown AJ, Frost SDW, Mathews WC, Dawson K, Hellmann NS, Daar ES, Richman DD , Little SJ. 2003. Transmission fitness of drug-resistant human immunodeficiency virus and the prevalence of resistance in the antiretroviral-treated population. Journal of Infectious Diseases 187:683–686.
Levi G, Vitória M. 2002. Fighting against AIDS: The Brazilian experience. AIDS 16:2373–2383.
Lima R. 2004. Information Management and Technology Considerations for ARV Programs in Resource-Constrained Settings. Paper presented at the Institute of Medicine Workshop on Antiretroviral Scale-up in Resource Constrained Settings, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Little SJ. 2000. Transmission and prevalence of HIV resistance among treatment-naïve subjects. Antiviral Therapy 5(1)33–40.
Little SJ, Holte S, Routy JP, Daar ES, Markowitz M, Collier AC, Koup RA, Mellors JW, Connick E, Conway B, Kilby M, Wang L, Whitcomb JM, Hellmann NS, Richman DD. 2002. Antiretroviral-drug resistance among patients recently infected with HIV. New England Journal of Medicine 347:385–394.
Loubser S, Sherman G, Chezzi C, Jones S, Cohen S, Puren S, Morris L. 2004. Characterization of nevirapine resistance mutations using RT-PCR and DNA sequencing methods in a mother-infant cohort following single dose nevirapine. Abstract 131. Antiviral Therapy 9:S145.
Martin-Carbonero L, Nunez M, Gonzalez-Lahoz J, Soriano V. 2003. Incidence of liver injury after beginning antiretroviral therapy with efavirenz or nevirapine. HIV Clinical Trials 4(2):115–120.
Médecins sans Frontières. 2003a. Providing HIV Services Including Antiretroviral Therapy at Primary Health Care Clinics in Resource-Poor Settings: The Experience from Khayelitsha. [Online]. Available: http://www.msf.org/source/countries/africa/southafrica/2004/1000/khayelitsha1000.pdf [accessed August 5, 2004].
Médecins sans Frontières. 2003b. Surmounting Challenges: Procurement of Antiretroviral Medicines in Low-and Middle-Income Countries. [Online]. Available: http://www.accessmed-msf.org/documents/procurementreport.pdf [accessed July 1, 2004].
Orrell C, Bangsberg DR, Badri M, Wood R. 2003. Adherence is not a barrier to successful antiretroviral therapy in South Africa AIDS 17(9):1369–1375.
Paterson DL, Swindells S, Mohr J, Brester M, Vergis EN, Squier C, Wagener MM, Singh N. 2000. Adherence to protease inhibitor therapy and outcomes in patients with HIV infection. Annals of Internal Medicine 136(3):253.
Pillay C, Bredell H, McIntyre J, Gray G, Morris L. 2002. HIV-1 subtype C reverse transcriptase sequences from drug-naive pregnant women in South Africa. AIDS Research and Human Retroviruses 18(8):605–610.
Redfield R. 2004 (January 27). Durability of ARV Therapy: U.S. Experience and its Implications for Resource-Constrained Settings. Paper presented at the Institute of Medicine Workshop on Antiretroviral Scale-up in Resource Constrained Settings, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Richman D, Morton S, Wrin T, Hellmann N, Berry S, Shapiro M, Bozzette S. 2004. The prevalence of antiretroviral drug resistance in the United States. AIDS 18:1–7.
Schechter M. 2004 (January 27). Lessons Learned from the Scale-up of Antiretroviral Treatment in Brazil. Paper presented at the Institute of Medicine Workshop on Antiretroviral Scale-up in Resource Constrained Settings, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Sethi A, Celentano D, Gange S, Moore R, Gallant J. 2003. Association between adherence to antiretroviral therapy and human immunodeficiency virus drug resistance. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America 37:1112–1118.
Simon V, Vanderhoeven J, Hurley A, Ramratnam B, Louie M, Dawson K, Parkin N, Boden D, Markowitz M. 2002. Evolving patterns of HIV-1 resistance to antiretroviral agents in newly infected individuals. AIDS 16:1511–1519.
Taylor S, Cane P, Hue S, Xu L, Wrin T, Lie Y, Hellmann N, Petropoulos C, Workman J, Ratcliffe D, Choudhury B, Pillay D. 2003. Identification of a transmission chain of HIV type 1 containing drug resistance-associated mutations. AIDS Research and Human Retroviruses 19:353–361.
Uganda AIDS Commission. 2003. Follow-up to the Declaration of Commitment on HIV/ AIDS (UNGASS): Uganda Country Report, January–December 2002. [Online]. Available: http://www.unaids.org/html/pub/topics/ungass2003/sub-saharan-africa/uganda_ungassreport_2003_en_pdf.pdf [accessed August 6, 2004].
UNAIDS (Joint United Nations Programme on HIV/AIDS). 2002. HIV/AIDS and Sexually Transmitted Infections: Haiti. 2002 Update. [Online]. Available: http://www.unaids.org/ [accessed August 4, 2004].
Velasco-Hernandez JX, Gershengorn HB, Blower SM. 2002. Could widespread use of combination antiretroviral therapy eradicate HIV epidemics? Lancet Infectious Diseases 2:487–493.
Walton D, Farmer P, Lambert W, Léandre F, Koenig S, Mukherjee J. 2004. Integrated HIV prevention and care strengthens primary health care: Lessons from rural Haiti. The Journal of Public Health Policy 25(2):137–158.
Weidle PJ, Malamba S, Mwebaze R, Sozi C, Rukundo G, Downing R, Hanson D, Ochola D, Mugyenyi P, Mermin J, Samb B, Lackritz E. 2002. Assessment of a pilot antiretroviral drug therapy programme in Uganda: Patients’ response, survival, and drug resistance. Lancet 360(9326):34–40.
Weidle P, Downing R, Sozi C, Mwebaze R, Rukundo G, Malamba S, Despess R, Hertogs K, Larder B, Ochola D, Mermin J, Samb B, Lackritz E. 2003. Development of phenotypic and genotypic resistance to antiretroviral therapy in the UNAIDS HIV Drug Access Initiative—Uganda. AIDS 17(Supplement 3):S39–S48.
WHO (World Health Organization). 2003a. Access to Antiretroviral Treatment and Care: The Experience of the HIV Equity Initiative, Cange, Haiti. [Online]. Available: http://www.who.int/hiv/pub/prev_care/en/Haiti_E.pdf [accessed July 1, 2004].
WHO. 2003b. Antiretroviral Therapy in Primary Health Care: Experience of the Khayelitsha Programme in South Africa. [Online]. Available: http://www.who.int/hiv/pub/prev_care/en/South_Africa_E.pdf [accessed July 1, 2004].
WHO. 2003c. Scaling up Antiretroviral Therapy: Experience in Uganda. [Online]. Available: http://www.who.int/hiv/pub/prev_care/en/Uganda_E.pdf [accessed July 1, 2004].
World Bank. 2004. World Development Report 2004: Making Services Work for Poor People. Washington, DC: World Bank.



























