AAALAC International Perspective
John G. Miller
The Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International is a not-for-profit corporation established in 1965 as the American Association for Accreditation of Laboratory Animal Care. From its inception, AAALAC has had sound science as a principal focus. A 1964 report of AAALAC’s predecessor, the Animal Care Panel, included the following statements: “As part of the scientific community, the Animal Care Panel has been working to define the conditions of animal care which promote sound and proper animal experimentation. … The Animal Care Panel cannot and will not proceed with this program [accreditation] without the consent and support of the scientific community.” AAALAC’s current mission statement continues this emphasis on science, stating that the organization’s purpose is to “enhance the quality of research, teaching and testing by promoting humane, responsible animal care and use.”
It is most appropriate that ILAR host this meeting, because the animal care and use standard most widely known in the global laboratory animal science community is ILAR’s Guide for the Care and Use of Laboratory Animals (Guide). AAALAC is proud to have developed the first edition of the Guide, under a 1962 contract from the National Institutes of Health (NIH). Serving as the principal standard used by both AAALAC and the US Public Health Service (PHS) in evaluating animal care and use, each of the seven editions of the Guide has been developed by scientists, with its guidance based on “published data, scientific principles, expert opinion
and experience with methods and practices that have proved to be consistent with high-quality, humane animal care and use.” This hierarchy of scientific support for its recommendations, from peer-reviewed data to experiential evidence, has been a hallmark of the Guide and has undoubtedly contributed to its widespread acceptance by the laboratory animal and more general scientific communities. Its utility as an international standard is demonstrated by the fact that the English version has been translated into nine additional languages.
As noted above, the Guide is the principal standard used by both AAALAC and the PHS, with both applying its provisions to all vertebrate animals. When one considers the number of animals being used at academic and other institutions that receive support from the NIH and other PHS agencies, and the fact that all major US pharmaceutical companies and commercial suppliers of animals are accredited by AAALAC, it is reasonable to estimate that 90% or more of the research animals in the United States are cared for and used in programs that apply the standards of the Guide. This percentage is likely higher for laboratory mice and rats, and refutes the claims of those who state that because the US Department of Agriculture’s (USDA’s) Animal and Plant Health Inspection Service does not “regulate” mice and rats, these species are not “protected” in the United States.
For nearly the first 15+ years of its existence, AAALAC accredited animal care and use programs only in the United States. Its first accredited program outside the United States was just across the border in Canada, but in 1986 Europe was included with the addition of a program in the United Kingdom. In 1996, the AAALAC Board of Trustees approved a new Strategic Plan that called for significantly increased emphasis on accreditation of programs internationally. Following the directions of that plan has led to remarkable success, with accredited programs currently in 18 countries on five continents. A new AAALAC service, begun in 1997 and called the Program Status Evaluation, has taken AAALAC representatives to additional countries, where institutions are striving to understand the accreditation process and bring their programs up to the AAALAC Standard. Through this international growth, AAALAC has had the opportunity to observe and evaluate programs from the Netherlands to the Philippines, and from Indiana to India. This experience has placed all of us involved with AAALAC, including the Executive Office staff, Council on Accreditation, and ad hoc consultants, in the position of de facto harmonization of different animal care and use standards into the referenced AAALAC Standard above.
AAALAC employs a variety of existing standards and related guidance in its assessment of animal care and use programs. As already mentioned, the Guide is our principal standard, and we apply its provisions
and principles worldwide. However, the Guide is intentionally written in general terms to allow flexibility in its application. Taken together with the Guide’s emphasis on performance as a measure of successful application, situations naturally occur in which professional judgments regarding appropriate implementation may differ. To assist AAALAC evaluators in these situations and to provide guidance to prospective and current accredited units, we have developed a list of publications and other documents we term our “Reference Resources.” The full list is available at http://www.aaalac.org/resources.htm, and includes references from Europe and Canada, in addition to US resources. They provide more specificity than the Guide in a wide variety of areas, and in many cases provide examples of appropriate outcomes that are useful when applying the Guide’s performance standards. The Reference Resources provide guidance in areas such as euthanasia methods (Report of the AVMA Panel on Euthanasia; Euthanasia of Experimental Animals (EC DGXI)); training (FELASA recommendations on the education and training of persons working with laboratory animals: Categories A and C); humane endpoints (Guidance document on the recognition, assessment, and use of clinical signs as humane endpoints for experimental animals used in safety evaluation (OECD)); and many more.
The Guide and AAALAC’s Reference Resources share a very important common characteristic—both are science based. The process for adding references to our list requires that the Council on Accreditation vote approval before such addition. The key factor in the Council’s consideration of a prospective reference is scientific documentation of its validity and value to an animal care and use program. This factor has led on occasion to the Council’s disapproval of proposed resources in which, although originally science based, the supporting data are outdated. Similarly, existing references that have become outdated or have been super-ceded by newer science-based publications are removed and/or replaced.
Evaluating an entire animal care and use program requires more than the application of the provisions and principles of the Guide and Reference Resources. A review of the process by which AAALAC assesses and accredits programs in the United States and internationally also helps demonstrate the mechanisms by which the wide variety of local standards, guidance, and policies are harmonized through the accreditation process to result in a common AAALAC Standard.
In the United States and internationally, the legal and regulatory requirements applicable to the unit being evaluated constitute the baseline for accreditation. No program can receive AAALAC accreditation if it is in violation of the law. Thus, in the United States, all provisions of the Animal Welfare Act Regulations must be met for species covered by the USDA, and for units receiving PHS support, all elements of their Assur-
ance of Compliance with the NIH Office of Laboratory Animal Welfare must also be met. Program elements are then evaluated based on the provisions of the Guide; and when necessary and appropriate, specific Reference Resources are used to evaluate performance outcomes in areas in which the Guide is nonspecific or institutionally approved deviations from its recommendations have been employed. Critically important is that all principles of the Guide must be met. Finally, the expert professional judgment of the AAALAC Council on Accreditation is applied through a peer-review process, and a final accreditation status is granted.
Internationally the process is practically identical. Again, no program can become AAALAC accredited if it is in violation of local legal and regulatory requirements. Use of individuals as ad hoc consultants who are familiar with these local requirements facilitates uniform and appropriate interpretation and application to the unit. Once these local baseline requirements are shown to have been met, the Guide becomes the next standard to be applied. It is important to note that when local requirements are more stringent than Guide recommendations, the former must be met to achieve accreditation. In some instances, the Guide includes provisions not addressed in national or supranational animal welfare legislation or regulations, for example, in the area of occupational health and safety. In such cases, two options are available. (1) Other local requirements may exist outside the animal welfare area, as is the case with occupational health and safety requirements in the European Union (Council Directive on the Introduction of Measures to Encourage Improvement in the Safety and Health of Workers at Work (Directive 89/391/EEC). (2) In the absence of alternative local standards, the Guide standards are used as the basis for evaluating program elements in these areas. As AAALAC has grown internationally, we have conducted assessments in countries without national regulatory standards or other requirements for animal care and use. In these instances—just as in the United States—the Guide and Reference Resources serve as the basis for our evaluation. Finally, the application of expert professional judgment through the peer review process by the Council on Accreditation determines a program’s final accreditation status. The key to maintaining consistency and uniformity of the AAALAC Standard across diverse international settings and standards is that all principles of the Guide must be met.
Notwithstanding the broad array of standards and guidance available in the area of animal care and use, circumstances occasionally arise for which there is no applicable published standard. In addition, professional judgments may differ regarding the acceptability of practices or procedures not specifically addressed in existing standards. In these circumstances, AAALAC again looks to science for solutions. In fact, the process used by AAALAC follows that used by ILAR in developing the
Guide, that is, we look for published data in the area in question. When no relevant reports are located, scientific principles and expert opinion form the basis for resolution, with the final decision often informed by Council members’ experience with proven methods or practices. An example of this process involves the use of alcohol as a disinfectant. The Guide states that “alcohol is neither a sterilant nor a high-level disinfectant,” yet it is used extensively for these purposes in rodent survival surgery. To answer questions about the suitability of such use, the Council formed a subcommittee to research and address this issue. Based on the information in six refereed scientific journal articles, two additional references, and the Manual of Clinical Microbiology, the Council determined that alcohol was acceptable as a skin disinfectant, but under certain circumstances may not be adequate to sterilize or disinfect surgical instruments. These determinations were published in the AAALAC newsletter, Connection, and became part of the AAALAC Standard.
Thus, the AAALAC Standard is not a static document. In fact, it is not based on a single document at all, but rather a compilation of many existing standards, guidelines, and policies that encompass all aspects of an animal care and use program. The majority of these are science based, a fact that not only gives credence to those, like the AAALAC Council on Accreditation, who interpret and apply them in an accreditation program, but also leads to the greater likelihood of acceptance and implementation by the scientists subject to their provisions. The AAALAC Standard is, therefore, an evolutionary product that is developing as internationally recognized standards are interpreted through the collective professional judgment of animal care and use experts and applied through an in-depth, multilayered, scientific peer-review process.
Before providing a list of areas in which I believe more science would be useful, I will comment on the modification of existing standards. It is my strong belief that when existing long-standing guidelines or requirements appear to be meeting the welfare needs of animals, any significant changes should meet three requirements: (1) the change must be of clear benefit to the animals; (2) it should not interfere unnecessarily with the research; and (3) it should be science based.
Finally, the following list comprises areas that I believe could benefit from additional scientific study and data. I provide them only as topics for consideration, with the hope that this workshop will serve as a venue for discussion.
-
Enclosure dimensions;
-
Wire-bottom cages;
-
Environmental enrichment;
-
Decapitation/cervical dislocation;
The Council of Europe: What Is It?
Wim deLeeuw
Shortly after the end of World War II, several movements and activities were born that were dedicated to European unification. As an overall result, the Council of Europe was founded as an international political institution in 1949. It is designed only with international cooperation in mind. The general aims of the Council of Europe are to:
-
Protect human rights, democracy, and the rule of law in all member states;
-
Promote awareness and encourage Europe’s cultural identity and diversity;
-
Seek solutions to (social) problems facing European society;
-
Consolidate democratic stability in Europe;
-
Promote social cohesion and social rights; and
-
Promote and develop a European cultural identity with emphasis on education.
The actual areas of concern are human rights, health, education, culture, youth, sport, the environment, local democracy, heritage, legal cooperation, bioethics, animal welfare, and regional planning. Today, the Council of Europe has 45 member states, including about 800 million people.
The Council of Europe must be distinguished from the European Union, which was set up in 1957 as the European Economic Community. First, it is not a supranational institution like the European Community. It
does not have legislative power. Its member states are cooperating on a voluntary basis. The Council of Europe cannot impose any rule on its member states. Second, unlike the European Union, the Council of Europe is not an economic organization.
The geographical confines of the Council of Europe are larger than the membership of the EU. The EU has 15 member states: Austria, Belgium, Denmark, Finland, France, Germany, Greece, Ireland, Italy, Luxembourg, the Netherlands, Portugal, Spain, Sweden, and the United Kingdom. All of these countries are also member states of the Council of Europe. Next to these member states, however, the Council of Europe also includes 30 other European countries: Albania, Andorra, Armenia, Azerbaijan, Bosnia and Herzegovina, Bulgaria, Croatia, Cyprus, the Czech Republic, Estonia, Georgia, Hungary, Iceland, Latvia, Liechtenstein, Lithuania, Malta, Moldova, Norway, Poland, Romania, the Russian Federation, San Marino, Serbia and Montenegro, Slovenia, Slovakia, Switzerland, the former Yugoslav Republic of Macedonia, Turkey, and Ukraine.
THE COUNCIL OF EUROPE: HOW DOES IT WORK?
The headquarters of the Council of Europe, Le Palais de l’Europe, is situated in Strasbourg, France. The Committee of Ministers is the decision-making body of the Council of Europe. It is composed of the Ministers of Foreign Affairs of the member states. This body officially adopts Conventions, Resolutions, Agreements, and Recommendations. The Committee of Ministers also ensures that the conventions and agreements are implemented. In addition, there are two other institutions: (1) The Parliamentary Assembly is the organization’s deliberative body, the members of which are appointed by national parliaments. (2) The Congress of Local and Regional Authorities of Europe is a consultative body that represents local and regional authorities. Governments, national parliaments, and local and regional authorities are thus represented separately at the Council of Europe level.
The main tools of the Council of Europe to achieve its objectives are the following legal instruments:
-
Recommendations—often referred to as “soft law.” There is no legal obligation to follow or implement these recommendations; and
-
Conventions or treaties concluded between states. The member states are not legally obliged to sign a Convention, although they may be expected to do so since under the Council of Europe’s Statute, they have undertaken to “collaborate sincerely and effectively in the realization of the aim of the Council.” Nonetheless, there are different ways a member can deal with a Convention. It may choose to ignore the Convention as
-
being not relevant or not applicable to the national situation. By taking that position, a member is not obliged to comply with its provisions. A member can sign the Convention, thus recognizing the value and existence of the Convention. After having signed a Convention, a member is still not obliged to comply with the provisions of the Convention. However, once a state has signed and ratified (i.e., its Parliament has approved the instrument) and the Convention has become effective, the state will be morally and legally bound under international law to implement the Convention. Thus, the state has become a Party to that Convention and must ensure that the provisions will be respected on its territory. Most Council of Europe Conventions are not directly applicable within a member state; they are not “self-executing.” The most common way for a state to implement them is to enact appropriate national legislation or to adapt its existing domestic law to make it correspond to the rules in the Convention.
In contrast to the European Union, practically spoken, there is little legal enforcement of Conventions, which probably leads to variability in compliance. Some Conventions are also open for adoption by nonmember states. The Conventions and recommendations are drafted by governmental experts responsible to the Committee of Ministers, thereby providing for the interaction of political interests with technical considerations. They only have a legal status after they are adopted by the Committee of Ministers.
More than 350 nongovernmental organizations (NGOs) are granted a consultative status. Within the context of the Council of Europe, there are several consultation arrangements, which enable NGOs to participate in intergovernmental activities and encourage dialogue. These NGOs are a vital link to the public at large and to specific parts of society.
THE COUNCIL OF EUROPE AND ANIMAL WELFARE
The work of the Council of Europe on animal protection was started in the 1960s. Since then, the following five Conventions on the protection of animals have been drawn up:
-
On animals during international transport (ETS 65, 1968), which establishes general conditions for the international transport of animals;
-
On animals kept for farming purposes (ETS 87, 1976), which is a framework convention. More detailed recommendations on species are given in separate guidelines;
-
On animals for slaughter (ETS 102, 1979);
-
On vertebrate animals used for experimental and other scientific purposes (ETS 123, 1986); and
-
On pet animals (ETS 125, 1987).
All of these Conventions are based on the principle that “for his own well-being, man may, and sometimes must, make use of animals, but that he has a moral obligation to ensure, within reasonable limits, that the animal’s health and welfare [are] in each case not unnecessarily put at risk.”
These Conventions were the first international legal instruments to establish ethical principles for the use and handling of animals. They are the result of very lengthy research, discussions, and negotiations, undertaken by governmental experts, delegates from animal welfare organizations, scientific researchers, and representatives of professional associations directly concerned. They are therefore the results of compromises. The political and technical value of the legal instruments working method adopted in the framework of these activities is based on the close collaboration between representatives of all the governmental and nongovernmental organizations that are involved. They have been used as a basis for, and continue to influence, all of the national relevant legislation in Europe.
THE COUNCIL OF EUROPE AND THE PROTECTION OF LABORATORY ANIMALS
As early as 1971, the parliamentary assembly recognized that to protect animals against abusive and unnecessary experimentations certain norms should be established at an international level, to enable states to regulate such experiments in an harmonious way in their domestic law. A first draft of the Convention was elaborated by the Ad Hoc Committee of Experts on the Protection of Animals, the CAHPA. After lengthy discussions, the Convention was finally adopted in May 1985. The Convention is accompanied by an explanatory report, and attached to it are technical appendices. Appendix A presents guidelines for the accommodation and care of animals. Existing German and US guidelines were used as a basis. Unlike the provisions of the Convention itself, the guidelines in Appendix A are not mandatory; they are recommendations. These guidelines are based on knowledge of that time and good practice. Appendix A explains and supplements the principles on accommodation and care as adopted in article 5 of the Convention. Appendix B contains tables for the presentation of the statistical data on the use of animals for experimental and other scientific purposes. The object of the Appendix is thus to help authorities, institutions, and individuals in their pursuit of the aims of the Council of Europe in this matter.
The European Convention for the protection of vertebrate animals used for experimental and other scientific purposes (1986, ETS 123) includes provisions concerning the scope, care, and accommodation of the animals, conduct of experiments, humane killing, authorization procedures, acquisition of animals, control of breeding or supplying and user
establishments, education and training, and statistical information. It is clearly visible from several provisions that the 3Rs of Russell and Burch are used as a basis for the Convention.
Currently, 15 countries have signed and ratified ETS 123 and thus are Parties to the Convention: Belgium, Cyprus, Czech Republic, Denmark, Finland, France, Germany, Greece, the Netherlands, Norway, Spain, Sweden, Switzerland, the United Kingdom, and the European Community. The Convention is signed by Bulgaria, Ireland, Portugal, Slovenia, and Turkey. The Convention provides for Multilateral Consultations of the Parties at least every 5 years, to examine the application of the Convention and the advisability of revising it or extending any of its provisions according to changes of circumstances and new scientific evidence. The Multilateral Consultations are prepared by a Working Party. For their work, the Parties have invited other member states of the Council of Europe and nonmember states and cooperate very closely with nongovernmental organizations that represent the fields concerned. In the preparatory meetings for the 4th Multilateral Consultation, the following observers participated:
Canadian Council on Animal Care (CCAC)
European Biomedical Research Association (EBRA)
European Federation of Animal Technologists (EFAT)
European Federation for Primatology (EFP)
European Federation of Pharmaceutical Industries and Associations (EFPIA)
European Science Foundation (ESF)
Federation of European Laboratory Animal Breeders Associations (FELABA)
Federation of European Laboratory Animal Science Associations (FELASA)
Federation of Veterinarians of Europe (FVE)
International Council for Laboratory Animal Science (ICLAS)
Institute for Laboratory Animal Research (ILAR)
International Society for Applied Ethology (ISAE)
World Society for the Protection of Animals (WSPA)
Eurogroup for Animal Welfare (Eurogroup)
Member States: Austria, Croatia, Hungary
Nonmember State: United States of America
The participation of representatives of observer states and nongovernmental organizations is of great value. It implies a very broad exchange of information at technical as well as legal and political levels. Therefore, their involvement in this work has to be associated with the
success of the Multilateral Consultations to ensure a common and satisfactory level of protection for animals used for scientific purposes, thus enabling the Council of Europe to maintain its position of initiator in Europe for the protection of these animals.
Until now, three Multilateral Consultations have been held. At the 1st Multilateral Consultation held in 1992, the Parties adopted a resolution in which the scope of the Convention was made more precise in respect for genetically modified animals, and certain tables for statistical data were remodeled.
At the 2nd Multilateral Consultation that was held in 1993, a resolution on education and training of persons working with laboratory animals was adopted. This resolution contained guidelines for the education and training of persons taking care of animals (Cat. A), persons carrying out procedures (Cat. B), and persons responsible for directing or designing procedures and animal science specialists (Cat. D). The guidelines included in the resolution were mainly based on a report that had been issued by FELASA.
At the 3rd Multilateral Consultation that was held in 1997, a resolution on the acquisition and transport of animals was adopted. This resolution contained guidelines that complemented the guidelines on this topic included in Appendix A.
Concerning the care and accommodation of animals, the Parties recognized that Appendix A had proven to be of great value and was widely used as a reference. At the same time, however, it was realized that the Appendix had been drafted more than 10 years ago. The Parties agreed that new scientific evidence and new experience since then made it necessary to revise the Appendix and to define the areas where further research is needed. They therefore agreed that this revision of Appendix A should be on the agenda of the 4th Multilateral Consultation. Pending this revision, a resolution was drafted presenting guidelines for the improvement of the accommodation and care of laboratory animals, which would complement the guidelines in Appendix A. The guidelines in the resolution were mainly based on the conclusions and recommendations of the International Expert Workshop on laboratory animal welfare that was held in 1993 in Berlin. It was concluded that the most important areas appeared to be the enrichment of the environment of the individual species according to their needs for the following:
-
Social interaction. Group or pair housing was considered to be preferable to individual housing for all gregarious species, as long as the groups are stable and harmonious;
-
Activity-related use. Cages should be structured to enable an activity-related use of the space available; and
-
Appropriate stimuli and materials. It was recognized that guidelines could never replace close and regular observations of the animals involved to ensure that the enrichment initiatives do not have adverse effects for groups or individuals.
Taking into account the evolution of scientific knowledge and changing circumstances, the Parties realized that the technical Appendices might need to be adapted more frequently than its main provisions. However, because these Appendices are an integral part of the Convention, such adaptations could result in complicated amendment procedures. Therefore, a Protocol of Amendment (ETS 170) providing for a simplified procedure for the amendment of the technical Appendices to the Convention was drafted and opened for signature in June 1998. Thus, the Parties are able to amend the technical Appendices, without formal adoption by the Committee of Ministers.
The finalized documents must be formally adopted at the 4th Multilateral Consultation. Thereafter they will be submitted to the Committee of Ministers. The text of the Convention and the related documents, such as resolutions adopted by the Committee of Ministers, as well as the draft proposals for the revision of the Appendix on which the discussion is finalized and the finalized background documents, are available on the website of the Council of Europe (www.coe.int/animalwelfare/) .
The work that has been done at the Council of Europe in the area of laboratory animal welfare was based on a very fruitful cooperation between member states and observers of various organizations. To be more effective, it will be very important that the cooperation between the European Union and the Council of Europe is intensified and that cooperation with other international umbrella organizations is developed further.
ICLAS and the International Community
Gilles Demers
HISTORY
Through an initiative of the United Nations Educational, Scientific, and Cultural Organizations (UNESCO), the Council for International Organizations of Medical Sciences (CIOMS), and the International Union of Biological Sciences (IUBS), the International Committee on Laboratory Animals (ICLA) was conceived in 1956 as a nongovernmental organization to promote high standards of laboratory animal quality, care, and health. Its activities have included collaboration with the World Health Organization since 1961. In 1979, ICLA was renamed the International Council for Laboratory Animal Science (ICLAS), because much new knowledge in biology and medicine requires planned experiments with organisms or their parts.
ICLAS is an international nongovernmental and nonprofit scientific organization. ICLAS exists to promote high standards of animal care and use in education, research, testing, and diagnosis, to promote good science and foster humane practices in scientific research. The ICLAS Mission and Aims are compatible with the highest possible standards of animal research internationally.
MISSION AND AIMS
ICLAS advances human and animal health by promoting the ethical care and use of animals in research worldwide. The aims of ICLAS are
-
To promote and coordinate the development of laboratory animal science throughout the world and as a matter of priority in developing countries;
-
To promote international collaboration in laboratory animal science;
-
To promote quality definition and monitoring of laboratory animals;
-
To collect and disseminate information on laboratory animal science;
-
To promote worldwide harmonization in the care and use of laboratory animals;
-
To promote the humane use of animals in research through recognition of ethical principles and scientific responsibilities; and
-
To promote the 3Rs tenets of Russell and Burch.
MEMBERSHIP
ICLAS is composed of four (4) categories of members: National members (30); Scientific/Union members (37); Associate members (34); and Honorary members (9). National members represent national perspectives. Scientific/Union members represent national or regional laboratory animal science and other scientific associations. Associate members represent commercial and academic organizations that support the aims of ICLAS.
List of ICLAS Members
National members: Argentina, Austria, Belgium, Canada, China, Costa Rica, Cuba, Cyprus, Denmark, Finland, Germany, Hong Kong, Hungary, India, Iran, Ireland, Israel, Italy, Japan, Netherlands, Norway, Poland, South Africa, Spain, Sweden, Thailand, the United Kingdom, the United States, Mexico, and Tunisia.
Scientific/Union members: AALAS/USA, ANZLAS/Australia-New Zealand, AGS/USA, CALAS-ACSAL/Canada, CALAS/China, GVSOLAS/Germany, JALAS/Japan, KALAS/Korea, LASA/U.K., NVP/ Netherlands, Scand-LAS/Sweden, SEEA/Spain, AFSTAL/France, CSLAS/ Taiwan, SECAL/Spain, FinLAS/Finland, BCLAS/Belgium, Balt-LAS/ Latvia, ACCMAL/Central America, AAALAC, ACLAM/USA, CSLAS/ Croatia, KRIBB/Korea, JSP/Japan, SGV/Switzerland, AACyTAL/Argentina, AMCAL/Mexico, COBEA/Brazil, BALAS/Bangladesh, TALAS/ Thailand, International Union of Biological Sciences (IUBS)/France,
International Union of Immunological Societies (IUIS)/Netherlands, International Union of Nutritional Sciences (IUNS)/Netherlands, International Union of Pharmacology (IUPHAR) Germany, International Union of Physiological Sciences (IUPS)/USA, and International Union of Toxicology (IUTOX)/Switzerland.
NEW ICLAS GOVERNING BOARD (2003-2007)
President: Gilles Demers (Canada)
Vice President: Norikazu Tamaoki (Japan)
Secretary-General: Patri Vergara (SECAL, Spain)
Treasurer: Cecilia Carbone (Argentina)
National Members
Gemma Perretta (Italy)
Czeslaw Radzikowski (Poland)
Norikazu Tamaoki (Japan)
Guy De Vroey (Belgium)
Scientific/Union Members
Denna Benn (CALAS, Canada)
Melvin Dennis (AALAS, USA)
Guy Dubreuil (AFSTAL, France)
J.R. Haywood (IUPHAR, USA)
Rafael Hernandez (ACCMAL, Mexico)
Toshio Itoh (JALAS, Japan)
STRATEGIC PLAN
The ICLAS Governing Board has developed a Strategic Plan to guide the organization through the next several years. The Strategic Plan includes the mission statement of ICLAS: “The International Council for Laboratory Animal Science advances human and animal health by promoting the ethical care and use of animals in research worldwide.” ICLAS strives to act as a worldwide resource for laboratory animal science knowledge; to be the acknowledged advocate for the advancement of laboratory animal science in developing countries and regions; and to serve as a premier source of laboratory animal science guidelines and standards, and as a general laboratory animal welfare information center.
ICLAS PROGRAMS
Meetings
International meeting. An international scientific meeting is held in association with the general assembly every 4 years. It is organized by a National or Scientific member and is often held in association with regional/ local organizations.
Regional meetings. Other regional scientific meetings and courses are organized by laboratory animal science organizations in the various regions of the world under the auspices of six ICLAS Regional Committees for the following regions: Europe, Asia, Africa (French and English regions), Oceania, and the Americas. This process has allowed ICLAS to focus on each region and to assure diffusion of scientific knowledge within all regions of the world. ICLAS provides funding and guidance for courses and meetings in these regions.
Communications
ICLAS FYI Bulletin. The ICLAS FYI Bulletin is an electronic instrument that provides worldwide distribution of timely information that may be of interest to ICLAS constituents and that may be passed on to their constituents. An average of five bulletins are sent each month, and these bulletins have led to interaction among laboratory animal scientists around the world. This international network is the most extensive in laboratory animal science in the world.
ICLAS Website: www.iclas.org. The ICLAS web page has been developed to provide various items of information on ICLAS programs and ICLAS activities. This information is important to existing and potential constituents.
ICLAS INITIATIVES
ICLAS-CCAC International Symposium on Regulatory Testing and Animal Welfare (Québec, Canada, June 2001)
This ICLAS initiative was a great success, and included 160 participants from 22 countries. The proceedings of this meeting were published in ILAR Journal in the fall of 2002, and included the following conclusions:
-
A definite link between good animal welfare and quality science;
-
Reduction of pain and distress is a higher priority than the reduction of numbers of animals;
-
Guidance documents on humane endpoints: CCAC (1998) and OECD (2000) guidelines recognized as effective refinement tools to minimize pain and distress;
-
Existing validated earlier endpoints should be used by all sectors;
-
Guidelines developed by the OECD and ICH to promote more humane methodologies for the testing of chemicals are reducing animal use by eliminating redundant testing;
-
Data sharing and training programs should be put in place quickly to assist regulators, toxicologists, and others to be comfortable with the new tests;
-
Animal care practices that improve animal welfare without jeopardizing the scientific design must be implemented.
ILAR International Workshop on Development of Science-based Guidelines for Laboratory Animal Care
ICLAS is a cosponsor of this workshop convened to discuss the available knowledge that can affect current and pending guidelines for laboratory animal care, identify gaps in that knowledge to encourage future research endeavors, and discuss the scientific evidence that can be used to assess the benefits and costs of various regulatory approaches affecting facilities, research, and animal welfare.
Meeting for Harmonization of Guidelines (FELASA 2004, France)
The harmonization of existing guidelines for the use of animals in research, teaching, and testing is an emerging issue in the context of the globalization of research around the world. ICLAS, as an international umbrella organization, could act as a facilitator in this area. Accordingly, ICLAS will be inviting one or two representatives of the principal organizations in the world that produce or use guidelines for the use of animals in research, to a 1-day meeting (June 13-14, 2004) held in conjunction with FELASA 2004, in Nantes, France. Representatives from ILAR, FELASA, CCAC, Council of Europe, OECD, ICH, AAALAC, and others will be invited. This will be an opportunity to open the dialogue on harmonization of some existing guidelines and to learn whether there are possibilities to reach a consensus on the recognition of these guidelines at an international level. According to the commitment of the participants, this initiative could be repeated on a regular basis.
CONCLUSION
In the context of the ILAR International Workshop on Development of Science-based Guidelines for Laboratory Animal Care, ICLAS can play an important role because of
-
Its role as an international umbrella organization:
-
ICLAS membership includes countries from every region of the world; and
-
The impact of ICLAS programs to ensure diffusion of good science and good animal welfare practices is felt worldwide, through ICLAS Meetings, Regional Programs, the Communication Program, and other ICLAS Initiatives.
-
-
The ICLAS Policy regarding harmonization versus standardization.
ICLAS supports the harmonization of animal care and use policies, guidelines, and other forms of regulation on a worldwide basis. However, because ICLAS is in constant liaison with countries and regions having different cultures, traditions, religions, legislations, regulations, and laws, ICLAS considers that each country should be able to maintain an animal welfare oversight system that reflects those elements and that suits the country’s own particular characteristics. The rigidity related to standardization for all does not fit with respect to the characteristics of individual countries.
Role of the National Institutes of Health Office of Laboratory Animal Welfare and the Public Health Service Policy on Humane Care and Use of Laboratory Animals
Nelson L. Garnett
The laws, regulations, and policies in the United States have three main sources of which two are government and one is a private voluntary accreditation body: (1) the US Department of Agriculture (USDA), (2) the Department of Health and Human Services, and (3) the American Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC). In this presentation, I will focus mostly on the Public Health Service (PHS) contribution, although there are many similarities among the three sources.
I am responsible for a portion of the US system. To reiterate, we start with a law, the Public Health Service Act. It is the same law that authorized the establishment of the National Institutes of Health (NIH) many years ago. The PHS Act was amended in 1985 to include the PHS Policy on Humane Care and Use of Laboratory Animals, implemented by my office, the Office of Laboratory Animal Welfare (OLAW). It is interesting to note that the PHS Policy preceded the legislation that authorized it, so it was ready to be implemented almost immediately after passage. We believe that Congress was very familiar with the contents of the PHS Policy and provided a strong endorsement by authorizing it virtually unchanged.
The latest version of the PHS Policy was reprinted in 2002 with minor changes to reflect updated references, addresses, and a name change for our office. Otherwise, it is unchanged from the 1986 version. I will pro-
vide the websites where you can download this and many other relevant documents in the discussion. The PHS Policy spells out the requirements for animal care that must be followed by all institutions wishing to be eligible to receive funding from any of the PHS agencies. The PHS Policy also requires that institutions receiving PHS support design their programs to conform with the Guide for the Care and Use of Laboratory Animals (the Guide).
You have already heard much about the Guide as it relates to AAALAC, but it has always been linked to compliance with the PHS Policy as well. One more example of the harmonization of the regulations and policies is the fact that almost all of the institutional animal care and use committee (IACUC) procedures in the USDA regulations were drawn from the PHS Policy. Many of the existing USDA cage size requirements were taken verbatim from the earlier version of the Guide.
OLAW is part of the NIH, the primary government funding source for biomedical research. My immediate boss is responsible for the arm of NIH that funds billions of dollars worth of research at universities and other institutions in the United States and abroad. About half of that research includes some animal component. OLAW is composed of three major components: Assurances, Compliance, and Education. The total staff has recently grown to 10, including seven professionals and three support staff. We are responsible for monitoring approximately 1000 institutions that receive PHS support.
The applicability of the PHS Policy includes all animal-related activities conducted or supported by the PHS. The main PHS agencies involved in funding animal research are the NIH, the Centers for Disease Control and Prevention (CDC), and the Food and Drug Administration (FDA). This coverage is very broad and includes intramural and extramural research, grants and contracts, subcontracts, training grants, cooperative agreements, domestic and foreign activities, and even some collaborations and purchase orders. It applies to all live vertebrate animals supported by the PHS, without exception. In addition to the PHS agencies, many others such as the National Science Foundation, Howard Hughes Medical Institute, and American Heart Association have adopted the PHS Policy for their own funding programs. These endorsements have greatly expanded the influence of the policy beyond its original intent.
All PHS-supported activities must be conducted at an assured institution and must be reviewed and approved by an IACUC. PHS awarding units are responsible for ensuring that these requirements have been met before considering proposals for funding. Each grant applicant must address “Five Points” within the body of the application before the scientific peer review. Applications without this information are considered “in-
complete.” These points are number and space rationale descriptions of use procedures to minimize pain methods of euthanasia.
The US Government Principles (at the back of both the PHS Policy and the Guide) provide the foundation for all federal regulations regarding animals, and the PHS Policy was expressly written to implement these principles. I should also mention that these principles are virtually identical to the International Guiding Principles, which are applicable worldwide. It is also important to note that these principles apply not only to biomedical research, but also to testing as well as training (teaching).
Briefly, OLAW is responsible for the implementation and interpretation of the PHS Policy, the negotiation and approval of Assurances, the evaluation of noncompliance, and a nationwide education program. The current PHS Policy was already in existence and ready to be put into place almost immediately after the legislation that passed in 1985 had mandated it. It was patterned after the widely accepted human subjects protections that were implemented out of the same office until several years ago. The key elements of the PHS Policy include the philosophy of enforced self-regulation, an Assurance mechanism with oversight by a local institutional committee, and appropriate reporting and documentation. The policy applies to all PHS-supported activities and covers all vertebrate animals.
The PHS concept of enforced self-regulation includes a reliance on performance standards wherever possible, and recognizes the need for flexibility and professional judgment. To be effective, it must be self-monitoring, self-correcting, and self-reporting.
WHAT IS AN ANIMAL WELFARE ASSURANCE?
The Assurance is a written document that provides the basis for a trust relationship between the institution and the government. It describes your unique program of animal care and use and, once approved, becomes a criterion for future evaluation. An approved Assurance is required for eligibility to receive PHS support. The Assurance describes, in some detail, the program of animal care and use and must address all of the following elements of that program: applicability, lines of authority and responsibility, the IACUC, procedures to implement PHS Policy, veterinary care, occupational health, personnel qualifications, facilities, and species. The Assurance must be signed by the Institutional Official, someone in the institution who is authorized to make commitments on behalf of the institution and to ensure that the conditions of the PHS Policy are met.
Many of you are familiar with the role of the IACUC in protocol review and facility inspections. What is not always understood is that the IACUC is usually advisory to, or acting on behalf of, the Institutional
Official in carrying out its duties. Two other responsibilities that do not always receive the attention they deserve are the review of programs and the investigation of animal welfare concerns.
Protocol review procedures include the following:
-
Review of all animal-related activities;
-
Provision for designated reviewer(s) to conduct a review (after all members have had an opportunity to call for a full review);
-
Ability to consult experts if needed;
-
Requirement to review all significant changes before their initiation; and
-
Monitoring ongoing activities (perhaps one of the most frequently overlooked responsibilities).
The IACUCs are expected to take the following considerations:
-
Animal procedures involving analgesia and anesthesia, euthanasia;
-
Environmental conditions;
-
Veterinary medical care;
-
Personnel qualifications.
-
Specific USDA requirements to verify that (1) proposals avoid unnecessary duplication, and (2) the Principal Investigator has considered alternatives to painful procedures.
-
Review of certain broad general elements of “the science” (e.g., relevance to human/animal health, advancement of knowledge, good of society, species and numbers, and consideration of nonanimal methods). IACUCs are not expected to conduct scientific peer review, but instead to consider certain basic ethical issues as inseparable from the science.
Every 6 months, the IACUC is required to conduct animal facility and program evaluations. Reports of these evaluations are submitted to the Institutional Official and describe the institution’s adherence to, or departures from, the Guide. Reasonable and specific plans and schedules are then developed for correcting those deficiencies.
Annual reporting is considered vital to OLAW’s ability to provide oversight. These reports are to include the dates of semiannual evaluations, significant program or facility changes, change in accreditation status, and changes in IACUC membership; and they must allow for minority views to be expressed. Prompt self-reporting of problems is an essential part of our trust relationship. The majority of compliance issues with which we deal are brought to our attention by the institutions themselves. We view the reporting of problems, along with the corrective actions taken, to be a positive sign that the system is working as expected.
Finally, I would like to add a few words about the most important ingredient in our animal welfare oversight system—education. Education is the preventive medicine in our business. OLAW cosponsors an entire series of animal welfare educational activities throughout the year, with active participation from all of the other players, including the regulated community. The wealth of information is beyond the scope of this presentation, but I invite you to visit this website (http://grants.nih.gov/grants/olaw/olaw.htm) and explore the many resources and links that it has to offer.
Regulatory Authority of the US Department of Agriculture Animal and Plant Health Inspection Service
Chester A. Gipson
Congress passes, and the President signs, all legislation authorizing activities of the Animal and Plant Health Inspection Service (APHIS) of the US Department of Agriculture (USDA). The laws authorize or direct the Secretary of Agriculture to take certain actions, which may include issuing regulations. The Secretary delegates authority to the Under Secretary of Marketing and Regulatory Programs (MRP), who then delegates authority to the Administrator of APHIS. The Administrator of APHIS delegates authority to the APHIS Associate Administrator and Deputy Administrators/Directors. The Animal Care (AC) program of APHIS receives its regulatory authority from the Animal Welfare Act (7 U.S.C. 2131-2159) and the Horse Protection Act (15 U.S.C.1821-1831).
ADMINISTRATIVE PROCEDURE ACT
The Administrative Procedure Act (APA) contains the basic requirements for federal rulemaking. For most rulemaking, the APA requires the following:
-
Publication in the Federal Register of a proposed rule, including either the terms or substance of the proposed rule;
-
Opportunity for public participation in rulemaking through submission of written comments on the proposed rule;
-
Publication in the Federal Register of a final rule, with an explana-
-
tion of any changes that the agency has made and a response to the public comments; and
-
An effective date for the final rule that is at least 30 days after publication in the Federal Register, unless the rule relieves restrictions, grants an exemption, or there is other good cause for making an exception. This kind of rulemaking is called “informal” or “notice and comment” rulemaking.
Publication of a rule in the Federal Register has certain legal effects. The rule provides official notice of the existence and content of a document. Publication indicates that the document was issued properly. Finally, publishing the rule provides evidence that it is judicially noticed by a court of law. Regulations that are not published in the Federal Register in accordance with the Administrative Procedure Act may not be upheld in a court of law. Therefore, any rules that an agency wishes to enforce should be published in the Federal Register.
OTHER ACTS AND EXECUTIVE ORDERS
Although the APA sets forth the basic requirements for federal rulemaking, other acts and executive orders also apply and include the following: (1) Executive Order 12866, which provides for review of federal rules by the Office of Management and Budget (OMB) (part of the Office of the President) and which requires the preparation of cost-benefit analyses for some rules; and (2) the Regulatory Flexibility Act, which requires analyses by agencies of the potential economic effects of their rules on small entities (small businesses, nonprofits, and small governmental jurisdictions).
TYPES OF RULES
Among the several types of rules are a proposed rule, a final rule, an interim rule, an advance notice of proposed rulemaking, and a direct final rule. Each type is described briefly below.
Proposed Rule
Most rulemaking in APHIS begins with a proposed rule. This document must contain a preamble that includes the following, at a minimum: an explanation of the proposed rule; an analysis of the anticipated economic effects of the proposed rule; a description of any information collection requirements; an invitation to the public to submit comments by a
specified date (usually 60 days after publication); and the proposed rule itself, as it would appear in the Code of Federal Regulations.
Final Rule
Most rulemaking in APHIS concludes with a final rule. This document must contain a preamble that includes the following, at a minimum: a response to the issues raised by the public comments; an analysis of the anticipated economic effects of the final rule; a statement concerning any information collection requirements contained in the rule; and an effective date for the rule. The effective date must be at least 30 days after publication, unless the final rule relieves restrictions or there is other good cause for making the rule effective sooner. The final rule document must also contain the rule text that will appear in the Code of Federal Regulations.
Interim Rule
An interim rule may be issued instead of a proposed rule when there is good cause for making a rule effective before the public has an opportunity to comment upon it. For example, APHIS may need to put immediate restrictions in place after an outbreak of an animal disease to prevent the spread of that disease. An interim rule may be followed by a final rule, which could contain changes to the interim rule based on public comments. If the final rule does not make any changes to the interim rule, APHIS calls the final rule an affirmation of the interim rule. An interim rule contains a preamble that must include the following, at a minimum: an explanation of the rule; an effective date (usually upon publication); a description of any information collection requirements and the emergency approval number from the OMB necessary for implementing them; and an invitation to the public to submit comments by a specified date.
Advance Notice of Proposed Rule
Yet another type of rulemaking document is the advance notice of proposed rulemaking. This type of document may be used when the agency seeks to obtain preliminary information before issuing a proposed rule, or even making a decision about whether to issue a proposed rule. This document contains a description of the rulemaking being considered; an invitation to the public to submit comments by a specified date; and specific questions or issues that APHIS believes the public should address.
Direct Final Rule
The direct final rule is a type of rule that provides a shortcut for noncontroversial rules that are unlikely to generate even one negative comment. The direct final rule must include the following, at a minimum: an explanation of the rule; an analysis of anticipated economic effects of the rule; a deadline for submitting comments; a tentative effective date; and the rule itself, as it would appear in the Code of Federal Regulations. If no adverse comments are received by the close of the comment period, the direct final rule becomes effective on the date specified. If any adverse comments are received by the close of the comment period, the direct final rule must be withdrawn. If APHIS chooses to proceed with rulemaking, APHIS must issue a proposed rule. To ensure that the public receives notice of whether a direct final rule will become effective as indicated, APHIS publishes a brief notice after the comment period closes, either affirming the effective date or, if APHIS receives adverse comments, withdrawing the direct final rule.
RULEMAKING PROCESS IN APHIS
The following steps occur within APHIS before any rule is issued:
-
A need is identified.
-
A regulatory work plan is prepared, cleared within the Department of Agriculture, and designated as “significant” or “not significant” by OMB.
-
A writer on the regulatory staff of APHIS receives the assignment and works with a technical expert from the relevant APHIS program area and others to develop the rule.
-
The proposed rule is drafted and cleared within APHIS.
-
The Office of General Counsel (OGC) of USDA reviews and clears the rule for legal sufficiency, and policy officials within the department also review and clear the rule. At a minimum, the policy officials include the Deputy Administrator for Animal Care or other program involved, the Administrator of APHIS, and the Under Secretary for Marketing and Regulatory Programs. If the proposed rule has been designated “significant” by OMB, it is also reviewed by other policy officials in the Department, including the Chief Economist of USDA, the Chief Information Officer, and the Secretary.
-
The proposed rule is then also reviewed by OMB.
-
After all clearances have been obtained, the proposed rule is signed by the Administrator or the Under Secretary and published in the Federal Register.
After the rule is published in the Federal Register, comments arrive. The comments undergo evaluation and, if changes are necessary, revisions will occur. A worksheet describing the number and nature of public comments received and the agency’s planned response to them is submitted through the OMB for a designation (which may or may not be the same as the designation for the proposed rule). The final rule is drafted and cleared within APHIS. It is then reviewed and cleared by OGC, policy officials within USDA, and, if “significant,” by OMB. The final rule is then signed and published in the Federal Register with a specified effective date.
TIME FRAMES
The time required for a given rulemaking varies, depending on the complexity of rule, the number and nature of comments received, the priority assigned by the agency (APHIS has an average of 150-200 actions in progress at any given time), and the designation assigned by OMB. Rules designated “significant” take longer than rules designated “not significant,” at least partly because the clearance process within the department involves more policy officials and because OMB also reviews the document. OMB normally has 90 days to complete its review. Regulations designated “significant” may take several years to complete, from initiation of the regulatory workplan to publication of a final rule.
SUMMARY
For most requirements that the agency imposes on the public, the APA requires APHIS to conduct rulemaking. Although APA contains the basic requirements for rulemaking, including publication in the Federal Register, other laws and executive orders also apply to rulemaking. Among the various types of rulemaking documents, the most typical is a proposed rule followed by a final rule. Regulations are reviewed within APHIS, by the Office of General Counsel (USDA) and other policy officials, and, if designated “significant,” by the OMB. Rules designated “significant” may take several years to complete.
ADDITIONAL INFORMATION
For more information about USDA-APHIS rulemaking, visit the APHIS website at www.aphis.usda.gov/index.html, or contact the Regulatory Analysis and Development Staff of APHIS at USDA-APHIS-RAD, 4700 River Road, Unit 118, Riverdale, MD 20737–1238, Phone: (301) 734-8682.
A Review and Comparison of Processes to Change Regulatory Guidelines: A European Perspective
Jonathan Richmond
The regulation of the use of animals for experimental and other scientific purposes and the determination of minimum required standards of animal care and accommodation across Europe are issues and processes noted more for their complexity and opaqueness than uniformity and transparency. Although each European member state devises and implements its own national legislation and standards of care and accommodation, these domestic provisions are informed, and in some cases determined, by supranational agreements and legislation at the level of the Council of Europe and the European Community.
The Council of Europe, which currently has 45 member states, was established in 1949 to protect human rights, to encourage both diversity and a common European identity, to seek problems to societal problems, and to consolidate democratic stability. Its focus is more societal and cultural than economical and political. A key Council instrument is the Convention—an agreement to impose common standards and practices that are binding only on the member states that choose to sign and ratify. Not only is ratification voluntary, but there are no legal penalties for not ratifying, or ratifying but not complying.
In contrast, the European Union (EU) is focused on economic and political union. Through directives and other statutory instruments, member countries are obliged to adopt common policies and approaches toward these ends. There are currently 15 member states, with a number of Eastern European “candidate countries” joining in the spring of 2004.
Although directives mandate WHAT has to be achieved, they allow each member state discretion as to HOW it is achieved. The European Commission monitors transposition and implementation, and noncompliance is dealt with through legal proceedings in the European courts. Matters are further complicated by the fact that the EU is itself a member of the Council of Europe and it seeks to represent EU countries within the Council of Europe on matters within the legal competency of the Union.
It is timely to reflect on how these processes work in practice, because current work on changing the Council of Europe Convention and the European Community Directive provides useful insights into how changes are made to the content of these European instruments.
COUNCIL OF EUROPE CONVENTION ETS 123 (1986)
Council of Europe Convention ETS 123, which dates from 1986, makes provision for the protection of animals produced and used for experimental and other scientific purposes, including both fundamental and applied research. Most members of the European Union, and the Commission itself, have signed and ratified this Convention. Appendix A of the Convention sets minimum provisions for the housing and care of animals. Strictly speaking, those who have ratified the Convention are required only to “take note” of these provisions.
The Council has been working for several years now on revising Appendix A. Initial hopes in some quarters that the revision would produce evidence-based optimum standards of animal care and accommodation quickly proved unrealistic. Even for the common laboratory species, there is a dearth of evidence-based material on laboratory animal care and accommodation (and the Council has neither the time nor the resources to generate evidence), there are numerous opinions about contemporary best practice, but there is no means of demonstrating optimum provision.
In an attempt to resolve this problem, the Council of Europe established a series of technical expert working groups that were supported by a Secretariat, a steering group including representatives of the parties to the Convention, and various observers, including the Institute for Laboratory Animal Research, the US Department of Agriculture, and the Canadian Council on Animal Care, and a separate drafting group. The working groups were charged with the task of producing both general and animal-specific guidance on care and accommodation. The expert groups are broadly based and consist of experts largely drawn from nongovernmental organizations but excluding the national competent authorities.
The technical work is nearing completion. Both general guidance and species-specific provisions are being drafted (backed up by separate detailed appendices that set out the information that was considered when
the revised specifications were determined). Once the technical content has been finalized, a multilateral meeting of the parties to the Convention will be convened to adopt or reject the proposals.
Two key considerations are evident in the outputs to date: (1) The quality and complexity of the space provided is as important as the area or volume of space; and (2) pair and group housing of social species will be considered to be the norm. Most importantly, to encourage and facilitate diversity and innovation, “performance-based” standards are being sought whenever practical. The Council of Europe is placing drafts of “completed” documents on its website. This process has been expensive, lengthy, and complex—and it is not finished yet.
Although the status of the relevant Appendix to the Convention is likely to be as before (i.e., something the Parties should take note of), the fact that the European Community is now a Party to the Convention is of significance. Specifically, the Community will be obliged to ensure that similar standards are incorporated into the relevant European Directive, and member states may then be obliged to conform.
EUROPEAN UNION DIRECTIVE 86/609/EEC (1986)
The statutory instrument EU Directive 86/609/EEC makes provision for the harmonization of laws for the protection of animals produced and used for experimental and other scientific purposes. In scope, it covers safety testing; work aimed at preventing, detecting, and controlling disease; the assessment, detection, and modification of physiological functions; and protection of humans and the natural environment. However, unlike the Convention, it probably does not cover fundamental research (including drug discovery), forensic enquiries, or education and training.
The Directive includes an “informative Annex” on standards of care and accommodation that the member states shall “pay regard to.” Furthermore “animal welfare” is not a Commission competence strictly speaking, and the Directive permits member states to “adopt stricter measures.” Work on reviewing and revising the Directive started recently. The early background work is being undertaken by four Technical Expert Working Groups, which are again weighted in favor of nongovernmental organizations rather than national competent authorities. Even at this stage, it is clear that the 3Rs of replacement, reduction, and refinement; a system of harm/benefit assessment; and a requirement for ethical review processes will underpin a revised Directive.
Another EU Directive that has an impact on animal use is the establishment of criteria to which products (e.g., chemicals, pharmaceuticals, and medical devices) must conform before they can be marketed or used within member states. Ensuring that these criteria make best provision for
a free market, consumer choice and safety, and animal welfare is a real challenge. Furthermore, the need for the EU to ensure that proposed test methods that replace, reduce, or refine animal use are scientifically valid before changing current test requirements is also a challenge. The role of the European Centre for the Validation of Alternative Methods and the Interagency Coordinating Committee on the Validation of Alternative Methods will also be considered.
HARMONIZATION AND DIVERSITY
Within Europe, there might be one Directive and one Convention to protect animals used for scientific purposes; but even within the EU, there are nevertheless 15 ways of doing it. Within the EU, the various national systems give a first impression of diversity, rather than harmony. Nevertheless, although the details vary, they have many elements in common; and much of the meeting presentation will compare and contrast key elements of several national systems. Their commonalities include regulation impacts on the place, the program of work, the personnel involved, and the training requirements for key staff. Authorization may be invested centrally, regionally, or even locally; but oversight by inspection is a common provision. Ethical review processes have become the norm, with different functions being discharged at local, regional, and national levels.
Hopefully, and mindful of the reasons for there being an EU, the revision of the Directive will focus on what needs to be regulated, rather than on what can be regulated; and it will produce a proportionate system not overly endowed with bureaucracy. Although all member states wel-come the flexibility of approach inherent in Directives, all are agreed that the outputs must be harmonized. Yet we live in interesting times, and it will be interesting to see what priority of the political agenda these issues are assigned when a newly elected European Parliament convenes next year. This Parliament will be composed of representatives of current member states and candidate countries.
Japanese Regulations on Animal Experiments: Current Status and Perspectives
Naoko Kagiyama and Tatsuji Nomura
In terms of ethics in animal experimentation, advanced countries have adopted the 3R principles of humane experimental technique first espoused by Russell and Burch in 1959, namely, replacement, reduction, and refinement. Twenty-six years later, in 1985, the 3Rs were translated into 11 basic principles by the Council of International Organizations for Medical Sciences (CIOMS). These items have become international principles that govern animal experimentation.
INTERNATIONAL COMPARISON OF REGULATIONS ON ANIMAL EXPERIMENTATION
Europe
The year 1986 was important in terms of the following: (1) the Council of Europe concluded the convention for the protection of vertebrate animals used for experimental and other scientific purposes; (2) the European Union formulated directives on the approximation of laws, regulations, and administrative provisions to rectify disparities in welfare policies among member states; and (3) in the United Kingdom, the Cruelty to Animals Act of 1876 was amended and its title changed to the Animals (Scientific Procedures) Act (Figure 1, left). The regulatory agency in the United Kingdom, the Home Office, issues three licenses—for the project, the personnel, and the premises where the animal experiment is to be
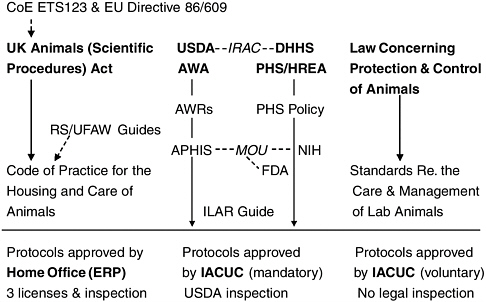
FIGURE 1 Comparison between regulations on animal experiments in the United Kingdom, the United States, and Japan. USDA, US Department of Agriculture; IRAC, Interagency Research Animal Committee; DHHS, Department of Health and Human Services; AWA, Animal Welfare Act; PHS, Public Health Service; HREA, Health Research Extension Act; AWR, Animal Welfare Regulations; UFAW, Universities Federation for Animal Welfare; APHIS, Animal and Plant Health Inspection Service; MOU, Memorandum of Understanding; NIH, National Institutes of Health; FDA, Food and Drug Administration; ILAR, Institute for Laboratory Animal Research; IACUC, institutional animal care and use committee.
conducted. All three licenses must be obtained before starting the animal experiment. Thus, the UK regulatory authority directly controls animal experimentation.
United States
In the United States, the Animal Welfare Act was enacted in 1966. The amendment in 1985 required research facilities to appoint an institutional animal care and use committee (IACUC) as well as to ensure the qualifications of personnel involved in animal experiments (Figure 1, center). Animal experiments can be performed based on a review and approval of the IACUC and the final approval of the institutional official. The regulatory agency, the US Department of Agriculture (USDA), conducts unannounced inspections of facilities every year.
Japan
In Japan, animal experimentation is also regulated by laws. The types of regulations in Japan (Figure 1, right) resemble the US system, which holds each institution responsible for self-regulation. However, the designation of an equivalent of the IACUC, registration, and legal inspections of laboratory animal facilities are not stipulated in the law.
LEGAL SYSTEMS IN JAPAN
The detailed legal system in Japan is described in Figure 2 from an historical viewpoint. The Law Concerning the Protection and Control of Animals enacted in 1973 was amended in 1999 and given the new title of the Law for the Humane Treatment and Management of Animals. This law protects all species of animals from cruelty (Investigative Committee 2001).
The law emphasizes respect for life, companionship with animals, and well-being of animals. It specifies the responsibility of the owner of the animal, and calls for the alleviation of pain and distress as well as the humane death of animals used for scientific purposes. Based on the law, the Standards Relating to the Care and Management of Experimental

FIGURE 2 Regulations related to animal experiments in Japan.
Animals were specified in 1980 (Figure 2, left). The Standards cover the care and management of laboratory animals, but not the use of animals (Investigative Committee 1980). The same year that the Standards were announced, the Science Council of Japan advised the Prime Minister to prepare administrative guidance for the use of animals for scientific purposes (Science Council of Japan 1981).
In 1987, in accordance with the advice of the Science Council of Japan, the Ministry of Education notified universities and other institutions to establish voluntary guidelines on animal experimentation (Ministry of Education 1987) (Figure 2, right). As a result of the notification, universities and even private research institutes formulated their own principles, guidelines, manuals, and other materials in accordance with the laws and with administrative guidance. Similarly, the scientific associations concerned have compiled guidelines for individual research fields to encourage members to balance science and animal welfare (JALAS 1987). Thus, animal experiments in Japan are regulated by a combination of legal and scientific developments.
CURRENT STATUS
Current regulations regarding animal experimentation in Japan are summarized in Table 1. The three categories include the following: laws consisting of the law, the standards, and the guide; administrative guidance issued by the Head of Science and International Affairs Bureau through their bulletin, which includes the notification; and voluntary guidelines formulated by individual scientific associations. Laboratory animal scientists in Japan have observed all three categories of regulations equally without the force of law but with a moral sense. The law, the standards, and the guide in the first category consist of 31, 10, and 4
TABLE 1 List of Regulations Regarding Animal Experiments in Japan
|
1. |
LAWS Law for the Humane Treatment and Management of Animals (Ministry of the Environment) —Standards Relating to the Care and Management, etc. of Experimental Animals —Guide for the Disposal of Animals |
|
2. |
ADMINISTRATIVE GUIDANCE Notification Concerning Animal Experimentation Conducted by Universities, etc. (Ministry of Education) |
|
3. |
VOLUNTARY GUIDELINES More than 12 guidelines on animal experiments formulated by individual scientific associations |
TABLE 2 Characteristics of Regulations on Animal Experiments in Japan
|
• |
Laws specify the responsibility of the owner of the animal |
|
• |
Laws call for 3Rs emphasizing the alleviation of pain and distress as well as humane death of animals used for scientific purposes |
|
• |
Administrative guidance encourages the ethical use of animals |
|
• |
Self-regulation system similar to the US and Canada |
|
• |
Recommendation for designating IACUC |
|
• |
Exemption from legal registration/inspection |
articles, respectively. Each has an associated explanatory handbook with 302, 115, and 68 pages of texts, respectively. The handbook for the Standards was edited by seven laboratory animal scientists, one medical doctor, and one representative of the Japan Animal Welfare Society to reflect the opinions of animal advocates.
In Table 2, the characteristics of Japanese regulations on animal experiments are listed. Our ethical principles, known as the 3Rs, are the same as in Western countries. The law emphasizes refinement that focuses on the alleviation of pain and distress as well as the humane death of animals.
The law adopts the self-regulation system for animal experimentation, and the notification only recommends designation of an IACUC (Ministry of Education 1987). Nevertheless, according to survey results, almost all medical schools and pharmaceutical companies as well as about one-third of breeders have established IACUCs even though the law does not mandate it. Laboratory animal and livestock facilities are exempted from registration and legal inspection. Instead, the Japanese Association for Laboratory Animal Science voluntarily conducts surveys every 3 years on the number of animals used for scientific purposes (CLACU 2003). It is therefore evident that laboratory animal scientists in Japan recognize the importance of replacement of live animals with insentient materials and reduction in the number of animals involved.
Although our regulations may appear somewhat lenient and ambiguous from the Western viewpoint (Nomura 1995) (a feeling that has sometimes annoyed Japanese scientists when collaborating with Western colleagues), the authors believe that certain religious implications may underlie animal experimentation ethics and the structure of regulations in individual countries, as described below.
RELIGIOUS IMPLICATIONS
A Thai venerable who graduated from medical school made a presentation about the philosophy of Karma in Buddhism at the 2003 annual
meeting of the American Association of Laboratory Animal Sciences. Researchers in Buddhist countries regard animals as existing on the same level as humans and generally treat animals based on the philosophy of Karma. Buddha rewards or punishes people based on their deeds (Karma). The logic of reincarnation is called samsara, the endless round of rebirth and redeath based on the impersonal judge of the Natural World, on which the destiny of humans depends.
Researchers in historically Christian countries, by comparison, appear to handle laboratory animals from the standpoint of the Lord of Creation. Researchers in Western countries give the impression of conducting animal experiments based on phylogenic domination of the animal. Therefore, they often cite the expression “humane care and responsible use.”
PERSPECTIVE OF ANIMAL EXPERIMENTS IN JAPAN
In Japan, we seek to combine Buddhist and Christian assumptions and to reach a point where humans should take responsibility for laboratory animals so that they can accomplish good Karma while using the animals for scientific purposes. If animals are suffering from infectious diseases, for example, they will not be able to provide reliable experimental data as their good Karma. Thus, microbiological control of the animal environment should be the responsibility as well as good Karma for people engaged in animal experimentation. Investigators in Japan may achieve the 3Rs principle in practice without any strict regulations because of the fear of samsara. However, the need to collaborate with Western colleagues requires compromise with the Western system. For this reason, we have been discussing appropriate strategies.
There is current disagreement over whether we should aim toward more stringent regulations, similar to European countries, or continue the current self-regulation system as in the United States and Canada. To ensure a convincing self-regulation system, we will need to clarify the responsibility of persons who engage in animal experiments, define the role of the IACUC, and implement animal welfare practices compatible with scientific needs, as shown in Figure 3. As mentioned above, none of these elements are strictly stipulated by the law but are regulated by administrative guidance and voluntary guidelines to encourage flexible animal research. It is now time for us to consider a certain mechanism to defend animal research as well as a validation system for self-regulation to reach a social consensus on the necessity of animal experimentation.

FIGURE 3 Reinforcement of the self-regulation system within animal research facilities in Japan.
REFERENCES
CLACU [Committee for Laboratory Animal Care and Use, Japanese Association for Laboratory Animal Science]. 2003. The number of live animals used in experiments in 2001 [text in Japanese, summary in English]. Exp Anim 52:143-158.
Investigative Committee [Investigative Committee on the Law for the Humane Treatment and Management of Animals]. 2001. Explanatory Handbook on the Law [text in Japanese]. Tokyo: Seirin Shoin.
Investigative Committee [Investigative Committee on the Standards Relating to the Care and Management of Experimental Animals]. 1980. Explanatory Handbook on the Standards [in Japanese]. Tokyo: Gyousei.
JALAS [Japanese Association for Laboratory Animal Science]. 1987. Guidelines on Animal Experimentation [in Japanese]. Exp Anim 36:285-288.
Ministry of Education [Ministry of Education, Science and Culture, Science and International Affairs Bureau]. 1987. Notification on Animal Experimentation in Universities [in Japanese].
Nomura, T. 1995. Laboratory animal care policies and regulations in Japan. ILAR J 37:60-61.
Science Council of Japan. 1981. Recommendation for the establishment of animal experiment guideline [in Japanese]. Exp Anim 30:173-178.
Process for Change—Development and Implementation of Standards for Animal Care and Use in Canada
Clément Gauthier
The Canadian Council on Animal Care (CCAC) is the national organization that has set and overseen the implementation of standards for the care and use of animals in science since 1968. It is a peer review organization involving close to 2000 scientists, veterinarians, animal care technicians, students, community representatives, and representatives of the animal welfare movement through its programs of assessment, guidelines development, education, and training. The CCAC pioneered the institutional animal care committee (ACC) as the local keystone of its decentralized ethical review and oversight system. For a detailed description, consult the following documents of the CCAC in the order indicated: The Assessment Program of the CCAC, 2000; Terms of Reference for Animal Care Committees, 2000; CCAC Guidelines on Animal Use Protocol Review, 1997; and Categories of Invasiveness in Animal Experiments, 1991. All of these documents are available on the CCAC web site [http://www.ccac.ca]. ACCs are now part of most legislated and voluntary frameworks for the regulation/oversight of animals used for scientific purposes worldwide.
Although compliance with CCAC standards is voluntary for government and industry, it is mandated through federal spending power for academic institutions. Over the past decade, provincial and federal governments have increasingly recognized the CCAC as a quasi-regulatory system. CCAC standards are now referenced in the regulations to provincial laws relating to the use of animals for scientific purposes (CCAC
2003; Russell and Burch 1959) and in the policies of relevant federal departments and agencies.
The underlying ethical basis of all CCAC guidelines and policies requires adherence to the 3Rs (reduction, replacement, and refinement) of Russell and Burch (1959). Maximizing animal well-being and minimizing pain and distress are the ethical drivers for the development of refinement measures. Housing conditions and environmental controls must be refined with the primary objective of meeting the social and behavioral needs of animals and of maximizing animal well-being. To minimize pain and distress, investigators frequently adopt practices believed to improve animal welfare based on anecdotal evidence. Although there is no commonly understood definition for the term “best practice,” per se, there is agreement on its content criteria and consensus on the fact that it must be evaluated by peers. The peer-based approach underlying the CCAC guidelines development process and the use of evidence-based learning loops in the evolution of best practices to implement these guidelines are two essential pillars of international harmonization of standards for the care and use of animals in science.
Compliance with CCAC standards is ensured at the local level through ACC peer-based review of protocols and mandatory site visits of facilities. National quality assurance is provided through CCAC’s external, peer-based assessment visits, which include the assessment of the functioning of the ACC. At the June 2001 first International Symposium on Regulatory Testing and Animal Welfare organized by the CCAC in collaboration with the International Council for Laboratory Animal Sciences, the Breakout Group on Best Practices for Animal Care Committees and Animal Use Oversight concluded that future progress requires encouraging diversity of frameworks as a source of continuous improvement, and the networking of ACCs to identify, encourage, and share best practices (ILAR 2002).
The institutional ACC plays a central role as the third pillar of international harmonization, for the following reasons:
-
It is representative of the scientific culture and moral values of home countries;
-
It facilitates communications and empowers informed decision making at the local level;
-
It is already integrated as an accountable keystone of most national oversight and regulatory systems worldwide; and
-
It provides each country with enhanced ability to influence international harmonization of best practices for animal care and use in science.
REFERENCES
CCAC [Canadian Council on Animal Care]. 2003. Legislation in Canada related to experimental animals. In: Experimental Animal User Training Core Topics Modules, Module 1: Guidelines, Legislation and Regulations. Electronic document (http://www.ccac.ca/english/educat/Module01E/module01-11.html).
ILAR [Institute for Laboratory Animal Research]. 2002. Proceedings of the ICLAS/CCAC International Symposium held in Québec City, Canada, June 21-23, 2001. ILAR J 43:S1-S136. Available at http://dels.nas.edu/ilar/jour_online.
Russell, W.M.S., and Burch, R. 1992 [1959]. Principles of Humane Experimental Technique. England: Universities Federation for Animal Welfare, Potters Bar, Herts, UK.
Building Credible Science from Quality, Animal-based Information
Paul Gilman
The research and development arm of the US Environmental Protection Agency (EPA) employs 1,950 employees at 13 laboratories and research facilities across the nation. Its $700 million budget for fiscal year 2003 included a $100 million extramural research grant program. The focus of those people, facilities, and programs is the generation of credible, relevant, and timely research results—and technical support—that inform EPA’s policies, decision making, and promulgation of regulations. Setting those regulations, making those policies, and accomplishing those decisions with sound science requires the Office of Research and Development (ORD) to fulfill the following responsibilities:
-
Production of relevant, high-quality, cutting-edge research results in human health, ecology, pollution control, and prevention investigations and in economics and decision sciences;
-
Characterization of scientific findings properly; and
-
Use of appropriate tools and approaches for the performance of science in the decision process.
High-priority areas for ORD include research in human health, particulate matter, drinking water, clean water, global change, endocrine disruptors, ecological risk, pollution prevention, and homeland security. Each of these important areas of investigation may include the use of animal models.
In its role as a leader in the environmental and human health protection research communities, ORD upholds three fundamental tenets in the use of animals in its research:
-
Animals represent a limited resource and are precious research tools;
-
Quality and validity of data collected from animals must be assured; and
-
Resultant research must have the full confidence of and acceptance by the scientific, regulated, and decision-making communities, and the public.
Understanding the specific needs for and value of animal use is balanced with associated responsibilities in conducting such research. ORD adheres to
-
Stringent review to assure responsible use;
-
Annual review and accreditation by the Association for Assessment and Accreditation of Laboratory Animal Care;
-
Close scrutiny of animal protocols; and
-
Continuous attention to the refinement of methodologies that reduce the number of or eliminate the use of animals.
















































