Monitoring and Assessing Particulate Matter
JUDITH C. CHOW and JOHN G. WATSON
Desert Research Institute
University of Nevada
Suspended particulate matter (PM) is a pollutant that adversely affects health, regional haze, global climate, ecosystems, and property. Mass measurements of PM were first made in the late nineteenth century by drawing ambient air through filter paper that was weighed before and after sampling. Measurements of total suspended particulates (TSP, particles with an aerodynamic diameter of less than 40 µm) for radioactivity analyses were first done in the United States during the 1950s to evaluate the effects of above-ground nuclear weapons tests. The technology was soon adopted for measuring mass concentrations in major urban areas. The availability of these data were the basis for designating TSP as the first indicator of PM for the 1970 U.S. national ambient air quality standard (NAAQS).
Different particle size fractions, such as PM10 and PM2.5 (particles with aerodynamic diameters of less than 10 µm and 2.5 µm, respectively) have different properties with respect to penetration into the human respiratory system, effects on visibility, and deposition on surfaces. The composition of PM, mostly carbon, sulfate, nitrate, ammonium, soil-related elements with associated oxides, industrial metals, and liquid water, also influences PM effects (Chow, 1995; Watson et al., 1995).
PM is both emitted directly and formed from gases, such as sulfur dioxide (SO2), oxides of nitrogen (NOx), ammonia (NH3), and volatile organic compounds (VOCs), that are emitted directly into the atmosphere. PM that is emitted directly is called primary aerosol; PM formed from gases is called secondary aerosol. Anthropogenic sources of PM and its precursor gases include industrial emissions, transportation, cooking, heating, the burning of vegetation, and dust from disturbed land. Natural sources include windblown dust from undisturbed
deserts, natural vegetation, wildfires, sea salt, and volcanoes. Human activities and industrial development influence both anthropogenic and natural emissions.
Secondary nitrate is related to ozone that derives from NOx and VOC precursor gases. A large quantity of precursor gases and primary particles comes from combustion sources that also emit carbon dioxide, a greenhouse gas. The problem of high PM concentrations must be addressed in the context of other air pollution problems.
Measuring PM concentrations, identifying their sources, and evaluating the effects of emission-reduction measures are difficult under any circumstances, but they present major challenges in China, where many small industries are not equipped with pollution-control devices and may not be documented in an emissions inventory. In addition, many urban areas use an unknown combination of domestic coal (honeycomb), bottled gas, and natural gas combustion for cooking and heating. Agricultural burning and refuse incineration are also common, even in areas where they have been outlawed.
Environmental planners and policy makers in China must first define the issues related to PM and precursor emissions. They must then adopt valid technical methods to make cost-effective decisions about where to locate roads and facilities, how to implement regulations and pollution-control measures, and where and when to monitor pollution levels to determine hazards, quantify source contributions, and track improvements.
This paper summarizes several approaches to common problems and provides references to more detailed information. Several key topics are addressed as they relate to rapid industrial development in China:
-
approaches to designing a PM monitoring network
-
zones of influence of different source emissions and zones of representation for ambient monitors
-
methods of obtaining and validating PM measurements
-
methods of quantifying organic and elemental carbon
DESIGNING A MONITORING NETWORK
The U.S. Environmental Protection Agency provides guidance on locating stations to monitor compliance with NAAQS (Koch and Rector, 1987; Watson et al., 1998). These guidance documents define the concepts and terms of network design and describe methods of defining planning areas and evaluating and selecting monitoring sites for an ambient air-quality network. In addition, the documents suggest how existing resources can be used for network design and provide examples of practical applications of various methodologies.
The first step in designing a network is to define monitoring objectives. Objectives may include: determining compliance with standards; forecasting pollution episodes; and quantifying adverse health effects, source attribution, visibility intensity and causes, materials soiling, and climate change. The next
step is site selection. Sites can be selected to represent source influence, community exposure, boundary/transport, and background conditions. Next, the monitoring strategy must be specified. There are three levels of complexity for monitoring networks (EPA, 2002):
-
Level III. A network of portable, inexpensive filters for continuous sampling at a large number of locations requires only a small investment in site infrastructure and maintenance. The hardware used for indoor and exposure studies can be used for these sites. The trade-off for greater spatial coverage is reduced accuracy and precision. Temporary, dense networks of this type surrounding Level I and Level II sites could establish zones of representation for the permanent monitors.
-
Level II. The network would have fixed sites with proven technology, similar to compliance sites, but with locations and observables that would serve multiple purposes. Sites would have real-time access for forecasting and providing pollution alerts. Resources no longer needed for compliance at urban sites could be used to establish background, boundary, and transport sites. Discontinued Level II compliance sites could be replaced with Level III monitors to address community concerns and issues of environmental justice.
-
Level I. The fixed sites in this network would have both proven and novel technologies, similar to U.S. supersites (EPA, 1998). A few of these sites would be located in contrasting environments—with different sources, meteorological conditions, and PM composition—to test new technologies, study atmospheric processes, and support health studies. These sites would have instrumentation similar to that of Level III and Level II sites to determine comparability, as well as detailed particle size ranges, PM chemistry, and precursor gases. They would provide an infrastructure for testing and evaluating new measurement concepts and developing procedures to implement them at Level II and III sites.
ZONES OF INFLUENCE OF SOURCE EMISSIONS AND ZONES OF REPRESENTATION FOR AMBIENT MONITORS
Multiple proximate and distant sources contribute to PM concentrations at any given location. Each monitoring site has a specific zone of representation, depending on the relative amounts contributed by emission sources of different spatial scales. In the United States, these scales are defined in regulation 40 C.F.R., Part 58. Sites outside of urban areas and away from sources provide background monitoring at regional scales (100 km to 1,000 km); sites next to roadways or downwind of factories monitor PM at middle scales (100 m to 500 m) superimposed on contributions from neighborhood (500 m to 4 km), urban (4 km to 100 km), and regional scales.
To estimate the spatial and temporal distribution of PM10 concentrations,
especially near fugitive dust sources, Chow et al. (1999) set up a closely spaced monitoring network with 29 Level III neighborhood-scale satellite sites surrounding two Level II urban sites with a study domain of 12 km × 13 km. The resultant data showed that the zone of influence around an individual emitter (e.g., a construction site) was less than 1 km. Measurements taken in residential and commercial areas showed that PM10 concentrations were similar in neighborhoods.
Another method of determining zones of representation is to examine samples from high time-resolution (e.g., 1 to 5 minute average) measurements. These are available using inexpensive nephelometers, which measure particle light scattering, and aethalometers, which measure particle light absorption. Watson and Chow (2002a) showed how data from neighborhood-scale and urban-scale sites can be compared by subtracting the short-duration spikes that represent middle-scale sources. High time-resolution measurements can also be used to create pollution roses, which can be used to determine the direction from which most of the material originates.
Figure 1 shows how light-scattering measurements taken at a Level I
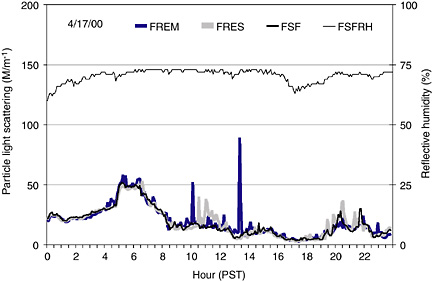
FIGURE 1 Light-scattering measurement taken at the Fresno, California, supersite (FSF), at a nearby residential site (FRES, 0.5 km E of FSF), and at a roadside site (FREM, 1 km WSW of FSF). The relative humidity at FSF (FSFRH) is kept at less than 74 percent by heating the sample inlet when ambient relative humidity exceeds 74 percent. This removes liquid water that would interfere with the relationship between PM2.5 and light scattering.
monitoring site (FSF, Fresno, California, supersite) represents the same PM concentrations as were measured in the same neighborhood at a Level III site near a highway (FREM, ~1 km WSW of site FSF) and at a Level III urban residential site (FRES, ~0.5 km east of FSF) (Watson et al., 2000). Neighborhood measurements at FSF show elevated concentrations in the early morning (~0600 hours) and late evening (~2000 hours). However, Figure 1 also shows multiple high-concentration spikes (mostly from the roadside site and typically during periods of heavy traffic) that were not observed at the Fresno supersite (FSF).
OBTAINING AND VALIDATING MEASUREMENTS
Several reviews identify the challenges for PM sampling and analysis (e.g., Chow, 1995; Chow et al., 2002a; EPA, 1994; Watson and Chow 2001; Wilson et al., 2002). The 24-hour, integrated, filter-based technology is gradually evolving toward in-situ, continuous, hourly measurements. In the interim, precision, equivalence, comparability, and predictability must be established among different measurement principles and methods. Short-duration (3 to 6 hours), rather than 24-hour, integrated measurements, are often used to monitor diurnal variations of PM concentrations and composition. Flow rates for short-duration samples must be increased to ensure adequate loadings for chemical speciation.
No single filter medium is appropriate for all chemical analyses; chemical characterization often requires sampling of multiple substrates (Chow, 1995; Fehsenfeld et al., 2003). Selection of the appropriate sampling substrates depends on particulate sampling efficiency, mechanical stability, chemical stability, temperature stability, and cost/availability. Because Teflon-membrane filters are inert to the absorption of gases, do not absorb water, and have low blank weights and low blank levels, they have been commonly used for mass measurement by gravimetry and elemental measurement by x-ray fluorescence (XRF), photo-induced x-ray emission (PIXE), and instrumental neutron-activation analysis (INAA) (Watson et al., 1998). Prewashed and prebaked quartz-fiber filters, which have low blank levels for ions and carbon and do not absorb water, are used for anion and cation analysis by ion chromatography (IC) (Chow and Watson, 1998) and carbon analysis (Chow et al., 1993, 2001). Several sampling substrates are subject to positive and negative interferences, such as artifact formation, loss of volatile species, filter fragility, particle loss in transport, and water absorption when the relative humidity is high.
Chow et al. (2002b) showed that 20 to 30 percent of sampled ammonium nitrate may evaporate if left in the field for 24 hours; 40 to 50 percent is volatilized if the sample is left in the field for 72 hours. Volatilized nitrate is not significant (within ± 5 percent) with diurnal monitoring in the early morning (0000 to 0600 hours) and late evening (1800 to 2400 hours). However, PM2.5 volatilized nitrate may be significant (~20 to 30 percent during the morning hours and up to ~ 40 percent during the afternoon hours when temperatures exceed
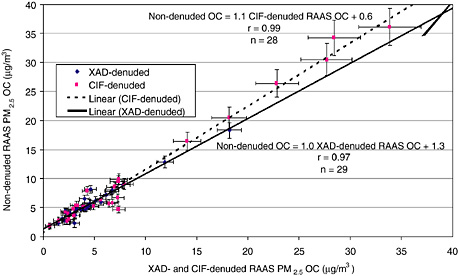
FIGURE 2 Measurements of denuded and nondenuded organic carbon (OC) are similar when field blanks are subtracted (CY2000). Denuders consisting of XAD resin or charcoal-impregnated cellulose-fiber filter (CIF) remove organic vapors from the incoming airstream.
20°C). Because volatilized ammonium nitrate is not measured as part of the gravimetric mass, PM2.5 mass concentrations may be underestimated.
Denuders and backup filters are often used to evaluate and quantify positive and negative artifacts. Denuders remove gases that might absorb onto a filter; backup filters capture gases that have evaporated from sampled particles. Positive and negative artifacts may partially cancel each other out in measurements of organic carbon. At the FSF site, for example, Watson and Chow (2002b) found that organic carbon samples were similar with and without preceding organic-gas denuders when a field blank was subtracted (Figure 2). Denuded and nondenuded samples of organic carbon were highly correlated (0.97 < r < 0.99) with a slope close to unity (within ± 10 percent). Detailed measurements of organic compounds coupled with accurate thermodynamic models that explain gas particle-phase relationships are necessary for measuring different aerosol compositions in various environments.
QUANTIFYING ORGANIC AND ELEMENTAL CARBON
Carbon, a large component of PM (30 to 40 percent), is often divided into several fractions, most commonly organic carbon and elemental carbon. Elemental carbon, the black, light-absorbing fraction, is measured by many
different methods that do not always have the same results (Chow et al., 1993, 2001; Currie at al., 2002; Fung et al., 2002; Schmid et al., 2001; Sharma et al., 2002; Watson and Chow, 2002b). Some of the more detailed carbon fractions have been found to represent different source contributions (Chow et al., 2003, 2004a; Chow and Watson, 2002; Watson et al., 1994).
Fung et al. (2002) compared the interagency monitoring of protected visual environments (IMPROVE) thermal optical reflectance (TOR) method (Chow et al., 1993) with the thermal manganese dioxide oxidation (TMO) method (Fung, 1990) for simulated black carbon and ambient samples from Hong Kong, Korea, and the United States. The results for elemental carbon compared well with the simulated sample, but the TMO measurement was higher than the IMPROVE TOR measurement for the samples in urban, vehicle-dominated Hong Kong and lower than the IMPROVE TOR measurement in the remote background Korean and U.S. samples. This comparison showed that different carbon analysis methods can vary markedly as a function of the composition and concentration of the sample.
Chow et al. (2004b) showed that elemental carbon differs within a temperature protocol for reflectance (TOR) and transmittance (TOT) pyrolysis corrections, as shown in Figure 3. The Environmental Protection Agency (EPA) uses two different protocols for thermal/carbon analysis: (1) the IMPROVE network protocol and (2) the speciation trends network (STN) protocol. Both protocols sample air through quartz-fiber filters. Punches from these filters are then submitted for analysis (Chow et al., 1993; 2001). The two methods differ in terms of combustion temperatures, analysis durations, and optical monitoring. As shown in Figure 3, the underestimation of elemental carbon is ~30 percent with the IMPROVE monitoring protocol; this increases to ~70 percent with the STN protocol. When the cross-sections of the filter punches were examined: (1) charring was found throughout the filter, either from adsorbed organic vapors during sampling or adsorbed vaporized particles during analysis or liquid organic particles; (2) elemental carbon near the filter surface evolves earlier than some of the charred organic carbon embedded in the filters during thermal analysis; and (3) charred organic carbon attenuates red laser light more efficiently than elemental carbon (Chow et al., 2004b).
Many operating parameters can individually or collectively cause variations of up to 30 percent in test results. These include differences in combustion atmospheres, temperature ramping rates, and temperature plateaus. Unfortunately, no systematic research is being done to resolve these discrepancies. Such research should include: (1) a review and evaluation of the published literature, much of which is not in the air-quality journals; (2) documentation of the method used, especially for thermal evolution carbon comparisons with different combustion temperatures, residence times, ramp rates, and optical-pyrolysis corrections; (3) establishment of standards for different sources of black carbon (e.g., diesel fuel, wood burning, tar, gasoline engines) on different filter media (e.g., Teflon
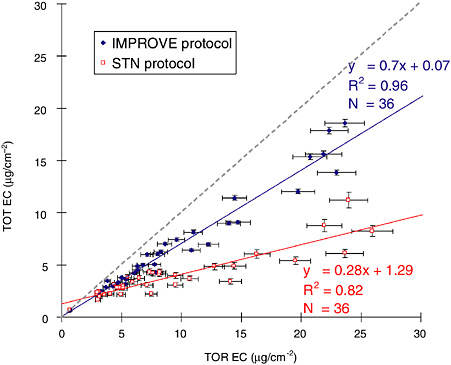
FIGURE 3 Comparison of transmission (TOT) and reflectance (TOR) corrected measurements of elemental carbon (EC) in samples from Fresno, California, using the IMPROVE and STN protocols. (The dashed line indicates the 1:1 correspondence.) Source: Chow et al., 2004b.
membranes, quartz fibers) at different deposit levels (light, medium, and dark appearances) for measurements of absolute absorption with a photoacoustic monitor (Moosmüller et al., 1998); (4) optical modeling of changes in absorption properties of particles on and within a filter compared to particles suspended in air (e.g., Chen et al., 2004); (5) experiments to examine the distribution of particles and pyrolysis artifact in a filter; (6) an evaluation of the effects of nonabsorbing particles, transmittance wavelengths, initial darkness, carbonate deposits, and oxygen-supplying minerals; and (7) optimizing fractions to identify source contributions.
RELEVANCE TO CHINA
China is a large, diverse country with many local, urban, regional, and international pollution problems for which there are no simple solutions. In this paper we have presented some possible starting points for PM monitoring and assessments.
Because of China’s rapid urbanization, pollution control will require collaboration among academic, regulatory, and commercial entities in various cities and provinces, and across international borders.
An integrated assessment, similar to the North American Research Strategy for Tropospheric Ozone (NARSTO) public/private partnership (Fehsenfeld et al., 2003), would be a good starting point for determining what is known and unknown about PM in China and what steps should be taken to minimize its adverse effects on health, visibility, and the environment.
REFERENCES
Chen, L.-W.A., J.C. Chow, W.P. Arnott, H. Moosmüller, and J.G. Watson. 2004. Modeling reflectance and transmittance of quartz-fiber filter samples containing elemental carbon particles: implications for thermal/optical analysis. Journal of Aerosol Science. In press.
Chow, J.C. 1995. Measurement methods to determine compliance with ambient air quality standards for suspended particles. Journal of the Air and Waste Management Association 45(5): 320–382.
Chow, J.C., and J.G. Watson. 1998. Ion Chromatography in Elemental Analysis of Airborne Particles. Pp. 97–137 in Elemental Analysis of Airborne Particles, Vol. 1, edited by S. Landsberger and M. Creatchman. Amsterdam: Gordon and Breach Science.
Chow, J.C., and J.C. Watson. 2002. Review of PM2.5 and PM10 apportionment for fossil fuel combustion and other sources by the chemical mass balance receptor model. Energy and Fuels 16(2): 222–260. Available online at: http://pubs3.acs.org/acs/journals/doilookup?in_doi=10.1021/ef0101715.
Chow, J.C., J.G. Watson, L.C. Pritchett, W.R. Pierson, C.A. Frazier, and R.G. Purcell. 1993. The DRI thermal/optical reflectance carbon analysis system: description, evaluation and applications in U.S. air quality studies. Atmospheric Environment 27A(8): 1185–1201.
Chow, J.C., J.G. Watson, M.C. Green, D.H. Lowenthal, D.W. DuBois, S.D. Kohl, R.T. Egami, J.A. Gillies, C.F. Rogers, C.A. Frazier, and W. Cates. 1999. Middle- and neighborhood-scale variations of PM10 source contributions in Las Vegas, Nevada. Journal of the Air and Waste Management Association 49(6): 641–654.
Chow, J.C., J.G. Watson, D. Crow, D.H. Lowenthal, and T. Merrifield, T. 2001. Comparison of IMPROVE and NIOSH carbon measurements. Aerosol Science and Technology 34(1): 23–34.
Chow, J.C., J.P. Engelbrecht, J.G. Watson, W.E. Wilson, N.H. Frank, and T. Zhu. 2002a. Designing monitoring networks to represent outdoor human exposure. Chemosphere 49(9): 961–978.
Chow, J.C., J.G. Watson, S.A. Edgerton, and E. Vega. 2002b. Chemical composition of PM2.5 and PM10 in Mexico City during winter 1997. Science and the Total Environment 287(3): 177–201.
Chow, J.C., J.G. Watson, L.L. Ashbaugh, and K.L. Magliano. 2003. Similarities and differences in PM10 chemical source profiles for geological dust from the San Joaquin Valley, California. Atmosphere and Environment 37(9-10): 1317–1340.
Chow, J.C., J.G. Watson, H.D. Kuhns, V. Etyemezian, D.H. Lowenthal, D.J. Crow, S.D. Kohl, J.P. Engelbrecht, and M.C. Green. 2004a. Source profiles for industrial, mobile, and area sources in the Big Bend Regional Aerosol Visibility and Observational Study. Chemosphere 54: 185–208.
Chow, J.C., J.G. Watson, L.-W.A. Chen, W.P. Arnott, H. Moosmüller, and K. Fung. 2004b. Equivalence of elemental carbon by thermal/optical reflectance and transmittance with different temperature protocols. Environmental Science and Technology. Accepted.
Currie, L.A., B.A. Benner, Jr., H. Cachier, R. Cary, J.C. Chow, E.R.M. Druffel, T.I. Eglinton, O. Gustafsson, P.C Hartmann, J.I. Hedges, J.D. Kessler, T.W. Kirchstetter, D.B. Klinedinst, G.A. Klouda, J.V. Marolf, C.A. Masiello, T. Novakov, A. Pearson, K.M. Prentice, H. Puxbaum, J.G. Quinn, C.M. Reddy, H. Schmid, J.F. Slater, J.G. Watson, and S.A. Wise. 2002. A critical evaluation of interlaboratory data on total, elemental, and isotopic carbon in the carbonaceous particle reference material, NIST SRM 1649a. Journal of Research of the National Institute of Standards and Technology 107(3): 279–298.
EPA (Environmental Protection Agency). 1994. Guidelines for PM10 Sampling and Analysis Applicable to Receptor Modeling, Final Report. EPA-452/R-94-009. Research Triangle Park, N.C.: Environmental Protection Agency, Office of Air Quality Planning and Standards.
EPA. 1998. Draft Supersites Conceptual Plan. Research Triangle Park, N.C.: Environmental Protection Agency, Office of Air Quality Planning and Standards.
EPA. 2002. National Air Quality Monitoring Strategy. Available online at: http://www.epa.gov/ttn/amtic/stratdoc.html.
Fehsenfeld, F.C., D.R. Hastie, P.A. Solomon, and J.C. Chow. 2003. Gas and Particle Measurments. Pp. 5-1–5-37 in Particulate Matter Science for Policy Makers: A NARSTO Assessment, Part 2. Pasco, Wash.: NARSTO. Also available online at: http://www.cgenv.com/Narsto.
Fung, K.K. 1990. Particulate carbon speciation by MnO2 oxidation. Aerosol Science and Technology 12(1): 122–127.
Fung, K.K., J.C. Chow, and J.G. Watson. 2002. Evaluation of OC/EC speciation by thermal manganese dioxide oxidation and the IMPROVE method. Journal of the Air and Waste Management Association 52(11): 1333–1341.
Koch, R.C., and H.E. Rector. 1987. Optimum network design and site exposure criteria for particulate matter. EPA 450/4-87-009. Research Triangle Park, N.C.: Environmental Protection Agency, Office of Air Quality Planning and Standards.
Moosmüller, H., W.P. Arnott, C.F. Rogers, J.C. Chow, C.A. Frazier, L.E. Sherman, and D.L. Dietrich. 1998. Photoacoustic and filter measurements related to aerosol light absorption during the Northern Front Range Air Quality Study (Colorado 1996/1997). Journal of Geophysical Research 103(D21): 28149–28157.
Schmid, H.P., L. Laskus, H.J. Abraham, U. Baltensperger, V.M.H. Lavanchy, M. Bizjak, P. Burba, H. Cachier, D.J. Crow, J.C. Chow, T. Gnauk, A. Even, H.M. ten Brink, K.P. Giesen, R. Hitzenberger, C. Hueglin, W. Maenhaut, C.A. Pio, and J. Puttock. 2001. Results of the “Carbon Conference” international aerosol carbon round robin test: Stage 1. Atmospheric Environment 35(12): 2111–2121.
Sharma, S., J.R. Brook, H. Cachier, J.C. Chow, A. Gaudenzi, and G. Lu. 2002. Light absorption and thermal measurements of black carbon in different regions of Canada. Journal of Geophysical Research 109(D24): AAC 11-1–AAC 11-11.
Watson, J.G., and J.C. Chow. 2001. Ambient Air Sampling. Pp. 821–844 in Aerosol Measurement: Principles, Techniques, and Applications, 2nd ed, edited by P. Baron and K. Willeke. New York: John Wiley & Sons.
Watson, J.G., and J.C. Chow. 2002a. A wintertime PM2.5 episode at the Fresno, CA supersite. Atmospheric Environment 36(3): 465–475.
Watson, J.G., and J.C. Chow. 2002b. Comparison and evaluation of in situ and filter carbon measurements at the Fresno supersite. Journal of Geophysical Research 107(D21): ICC 3-1–ICC 3-15.
Watson, J.G., J.C. Chow, D.H. Lowenthal, L.C. Pritchett, C.A. Frazier, G.R. Neuroth, and R. Robbins. 1994. Differences in the carbon composition of source profiles for diesel- and gasoline-powered vehicles. Atmosphere and Environment 28(15): 2493–2505.
Watson, J.G., G.D. Thurston, N.H. Frank, J.P. Lodge, R.W. Wiener, F.F. McElroy, M.T. Kleinman, P.K. Mueller, and J.C. Chow. 1995. Measurement methods to determine compliance with ambient air quality standards for suspended particles: 1995 critical review discussion. Journal of the Air and Waste Management Association 45(9): 666–684.
Watson, J.G., J.C. Chow, H. Moosmuller, M.C. Green, N.H. Frank, and M.L. Pitchford. 1998. Guidance for using continuous monitors in PM2.5 monitoring networks. EPA-454/R-98-012. Research Triangle Park, N.C.: Environmental Protection Agency, Office of Air Quality Planning and Standards.
Watson, J.G., J. C. Chow, and C.A. Frazier. 1999. X-ray Fluorescence Analysis of Ambient Air Samples. Pp. 67–96 in Elemental Analysis of Airborne Particles, Vol. 1, edited by S. Landsberger and M. Creatchman. Amsterdam: Gordon and Breach Science.
Watson, J.G.,s J.C. Chow, J.L. Bowen, D.H. Lowenthal, S.V. Hering, P. Ouchida, and W. Oslund. 2000. Air quality measurements from the Fresno supersite. Journal of the Air and Waste Management Association 50(8): 1321–1334.
Wilson, W.E., J.C. Chow, C.S. Claiborn, W. Fusheng, J.P. Engelbrecht, and J.G. Watson. 2002. Monitoring of particulate matter outdoors. Chemosphere 49(9): 1009–1043.














