3
Research Opportunities
Six major research initiatives for the environmental sciences have been identified in recent NRC reports (NRC, 2001c, 2004), the first four of which cannot be met without using the geologic record of ecological dynamics:
-
Biological diversity
-
Biogeochemistry
-
Ecological impacts of climate variability
-
Habitat alteration
-
Invasive species
-
Infectious diseases
The current state of the biosphere is a consequence of both present day processes and prior conditions. A full understanding of current patterns and processes, therefore, requires geohistorical analysis to supplement knowledge based on shorter-term observations, experiments, and modeling. The geological record provides the rich source of information that is essential for developing an understanding of the origin and controls of biological diversity, the controls and dynamics of biogeochemical cycling, the ecological impacts of climate change and variability, and the extent and consequences of habitat alteration. It also has the potential to provide important insights into the dynamics of invasive species and the environmental context of infectious diseases.
There are three general areas of environmental science research in which addressing the historical antecedents of present day patterns and processes will be especially important. Each takes advantage of the rich
and detailed geological record of how past ecosystems responded to environmental change at a variety of timescales. Together they provide the basic knowledge needed to predict and manage the response of the biosphere to likely change in the future.
1. The Geologic Record as an Ecological Laboratory. The geological record comprises an immense array of natural laboratories for studying how ecological systems operate under diverse conditions and at broad timescales. Using these laboratories to answer fundamental questions about biological diversity and biogeochemical processes is both possible and urgently needed. Ecological and evolutionary processes at timescales beyond direct human observation have influenced biodiversity and biogeochemistry at all scales (see examples described below). Currently, most ecological theory is based on short-term observations and mechanics, which are then extrapolated to longer timescales. Ecological studies using geohistorical records are needed to characterize ecological processes that occur over longer timescales; identify patterns and mechanisms that are masked by the short timespans of direct observation; and recognize those aspects of modern ecological systems that are contingent on past events. The geologic record also contains a series of “alternative worlds” suitable for testing the universality of ecological theory.
2. Ecological Responses to Past Climate Change. The geologic record contains information on how ecological systems—from individual species to biomes—have responded to a wide array of climate changes in the past. Just as paleoclimatological studies have revealed sensitivities and vulnerabilities in the global climate system that could not have been identified from analysis of modern systems alone (NRC, 2002a), so too are paleoecological studies revealing ecological responses to past climate changes that could not have been predicted solely from modern ecological investigations and theory (see examples described below). Studies that link paleobiological and paleoclimatic records are urgently needed to assess the ecological consequences of ongoing and future climate changes. The past two centuries have experienced only a fraction of the potential variability within the global climate system. Therefore, direct observations of biotic responses to climate variability and change provide only a limited view of the full range of possible changes and responses. Parallel to the call for “extending the record of [climate] observations” using the geological record (NRC, 2001c), a concerted effort is needed to use geohistorical records to gather critical information on how ecosystems will respond to future change. Specific time intervals can also serve as model systems for understanding effects of climate changes of particular magnitude, rate, extent, and duration.
3. Ecological Legacies of Societal Activities. The geologic record contains a rich store of information on ecological changes occurring during periods of past societal activity. The relative timing and nature of these correlations provide abundant material for evaluating direct and indirect causation—such records are important for gauging the extent to which our modern world has already been altered by human activities (see examples described below). They are also needed to predict effects of future societal modification of habitats and biological systems themselves. Determining whether particular ecological phenomena are induced by ongoing societal activity, comprise legacies of past human activities, or would have occurred in the absence of societal activity is a vital first step toward appropriate management. Determining how societal activities have shaped modern ecological systems at local, regional, and global scales is essential for understanding the world we have inherited, for assessing ecological theory developed within altered ecosystems, and for predicting how ecosystems will change in the face of ongoing and future societal activities. It is also necessary for determining baselines of natural ecological variability against which human activities and management decisions can be evaluated.
THE GEOLOGIC RECORD AS AN ECOLOGICAL LABORATORY
Ecological studies have tended to focus on patterns and processes that are observable on the timescales of direct human experience—weeks to decades, and occasionally centuries. Although ecological succession, one of the core concepts of ecology, is concerned with changes occurring over timescales of decades to millennia (Cowles, 1899, 1901; Clements, 1916; Glenn-Lewin et al., 1992), successional studies have only rarely taken advantage of geohistorical records of actual change at individual sites. Instead, ecologists have relied on space-for-time substitution (Pickett, 1989) or direct observations of successional change at shorter timescales. Chronosequence studies have pitfalls, however (S.T. Jackson et al., 1988; Davis, 1989; Fastie, 1996), and the oldest “permanent plots” where long-term changes can be monitored systematically date only to the mid-19th century (Pickrell, 2001). Most of the remaining conceptual and empirical core of ecology, whether at the organismal, population, community, ecosystem, or global level, tends to be focused on the “here and now,” with only nominal acknowledgment of longer-term patterns and dynamics. Thus, we are building our understanding of ecology on a very small sample, comprising an ultra-thin and perhaps unrepresentative slice of the history of the biosphere.
Ecologists now recognize that many important ecological processes operate at timescales far beyond human life spans and that in many cases
we cannot simply scale up in a linear fashion from studies spanning a few years to phenomena spanning thousands to millions of years (see Box 3.1). Many recent trends in ecology are concerned with the search for general rules that govern ecological systems, for example, in the areas of community assembly (Weiher and Keddy, 1999), biodiversity dynamics (Rosenzweig, 1995; Hubbell, 2001), macroecology (Brown, 1995; Maurer, 1999), scaling (Brown and West, 2000; Enquist et al., 2003; Smith et al., 2004), biodiversity/productivity relationships (Loreau et al., 2001; Tilman et al., 2001), global biogeochemical cycles (Schlesinger, 1997), trophic interactions (Williams and Martinez, 2000; Brose et al., 2004), and ecological stoichiometry (Sterner and Elser, 2002). Rigorous and definitive testing of general theory of this kind in ecology can be extraordinarily difficult but the past, as represented in the fossil record, provides a potentially powerful means for assessing and refining such ecological hypotheses. So far, the considerable potential of the geologic record has not been fully exploited by ecologists. Furthermore, although ecological rules identified from modern observation and theory are generally treated as universal, they may be contingent on the modern environment and biota. They may have relict, non-equilibrium features from both natural events and human activities of the past. Subjecting these hypothesized rules to rigorous tests involving both “natural,” pre-human conditions as well as various “alternative worlds” of the recent and deep past will determine the extent to which ecological laws are analogous to physical laws, or whether they evolve as the biosphere evolves. Observations in deep time can help determine whether deep structure and principles exist in ecological systems.
Opportunities for Scientific Advance
Major opportunities exist for increased interactions between ecology and the geosciences, particularly paleoecology and paleobiology. Future research efforts should be particularly focused on studies and syntheses that have the capacity for:
-
testing fundamental ecological theory and principles at timescales greater than the past two centuries;
-
identifying important ecological patterns and processes that emerge only at timescales beyond those of direct human observation; and
-
determining whether the basic laws and principles identified in ecology today are universal and thus applicable throughout geologic time, or are contingent on the modern biosphere, and thus evolve through time as environments and biota change. If the latter, we need to know whether such evolution proceeds in a predictable and systematic fashion, and which major ecological concepts are universal and which are subject to change.
|
BOX 3.1 Ecological processes occur over a vast array of temporal scales, comprising at least 16 orders of magnitude. For example, a phosphate ion released into solution in the photic zone of the ocean may be absorbed by phytoplankton in less than a second (10−8 year), while an organically bound phosphate incorporated into ocean sediments may be locked up for 108 years or more and then released only after subduction, metamorphism, uplift, and ultimate release by weathering (Ruttenberg, 2004). The mean generation time of a single-celled prokaryote may be less than a day, while that of many tree species is more than a century. With a constant environment, interspecific competition may take days, years, or centuries to reach equilibrium, respectively, in communities of microbes, intertidal mollusks, or forest trees. Ecologists are increasingly recognizing that ecological systems are influenced by both “fast” and “slow” processes, and that many ecological patterns are dictated by interactions between processes operating at very different rates (Carpenter and Turner, 2001). Developing theoretical, observational, and experimental frameworks for understanding these interactions is a major challenge for ecology and related sciences. In practice, ecologists have dealt with this problem by focusing on particular timescales (usually short), treating slow processes as constants (parameters), and calculating equilibrium values for the faster variables (Carpenter and Turner, 2001). However, this approach has limitations. Because of disturbances, climate variability, and other factors, ecosystems are in a state of continual flux. Although ecologists have addressed this problem by incorporating random fluctuations or disturbances into their models, such variations may not be simple random variation about a constant mean. For example, climate variability is non-stationary at timescales from interannual to multimillennial and beyond. Disturbances (e.g., fire, windstorms, hurricanes, flood and wave events) are often tied to climate, and consequently disturbance frequencies and amplitudes cannot necessarily be modeled as random variables, even at routine ecological timescales of 101-103 years. Some events may be so infrequent (e.g., impacts, volcanic eruptions, methane releases from the seafloor) or slow-acting (e.g., sea-level change) that observation and experimentation are essentially out of the question. Many ecological processes, particularly biogeochemical, biogeographic, and evolutionary processes, occur over even longer timescales, ranging from 103-108 years (i.e., at geologic timescales). Such slow processes can be accelerated, decelerated, reversed, shunted to alternative pathways, or (in the case of phyletic evolution) terminated, as a consequence of climatic, tectonic, macroevolutionary, and other events. The controls on slow processes and the long-term effects of rare events can be understood only by using the geologic record. |
These issues cut across a wide range of timescales, ranging from recent millennia to the entire record of life on Earth. They are vitally important to appropriate application of ecological knowledge and theory to conservation biology, biosphere maintenance, and global change biology. As we enter a period of potentially rapid environmental change, with extensive biological invasions and impoverishment of biodiversity, we need to know which ecological rules will apply and which are likely to be altered.
A critical goal will be to improve dialogue between paleobiologists and ecologists, particularly in the realm of theory and concepts. But this must be accompanied by integrated studies aimed at addressing the central questions of ecology across a range of timescales. For example, use of the geologic record is vital to addressing national ecological research priorities in the areas of biodiversity and biogeochemical cycles. It can also provide important perspectives on infectious diseases and invasive species. One major goal is to integrate paleobiological studies with ecological experimentation and modeling.
Both communities will benefit from increased interactions. A danger in the traditional ecological focus on the “here and now” of experiments on existing systems and environments is that the role of historical contingency and long-term processes can be overlooked. On the other hand, paleoecologists may discount the implications of short-term ecological experiments simply because short-term events are often difficult to resolve in the geologic record. Greater collaboration and coordination between these disciplines can help determine where observable short-term processes can scale up to explain long-term patterns, suggest experimental tests of paleoecologically generated hypotheses, and identify the ecological consequences of past environmental events.
Biodiversity
While understanding patterns of extant biological diversity is one of the central themes of ecology, the factors and processes that govern it are still inadequately understood. Consequences of biodiversity loss are not fully known but may be substantial, ranging from elimination of potentially useful species to loss of ecosystem services and even collapse of ecosystem function. The geologic record provides information on speciation and extinction rates as well as biogeographic changes that regulate biodiversity. Resolving one of the Grand Challenges in Environmental Sciences (NRC, 2001c; p. 26)—“Produce a quantitative, process-based theory of biological diversity at the largest possible variety of spatial and temporal scales”—requires use of the geologic record.
The Evolutionary Dynamics of Biodiversity. Biodiversity is shaped by biogeographic and evolutionary dynamics that occur over thousands to millions of years. Although the formation, invasion, migration, substitution, and even extinction of species can potentially be inferred from phylogenetic structure, the fossil record is a direct and rich source of data needed to discover the biological principles underlying the origin, maintenance, collapse, recovery, and extinction of species. Paleobiological time series leading to the present day, together with time series embedded entirely in the geologic record provide opportunities to identify and test fundamental ecological principles underlying biodiversity (see Box 3.2). Isolated but well-situated snapshots and brief time exposures in the geologic record provide additional opportunities.
Virtually all the key variables in conventional “macroecological” analysis of species assemblages in the present day—namely, species composition, richness, relative abundance, geographic range, body size and many life history factors, growth or metabolic rates, environmental tolerance, trophic or functional group, speciation and extinction rate—are measurable in the fossil record (see Chapter 2). Moreover, some kinds of information can only be determined using paleobiological data. For example, although net diversification rates can be inferred from species richness in clades and communities without a long historic record, the more useful raw speciation and extinction rates can only be determined from the fossil record. Only data on raw rates can reveal whether the striking variation in richness observed among ecological communities reflects differences in the production of species, in rates of species loss, or some combination of the two. These are qualitatively different dynamics, and call for completely different management strategies. These issues can be addressed by studies in deep time (e.g., Valentine, 1990; Sepkoski, 1998; Jablonski and Roy, 2003) as well as in the late Quaternary (e.g., Colinvaux, 1997; Hooghiemstra, 1997; Willis and Niklas, 2004).
Ecological theory and experimentation has led to the recognition that certain species are “community keystones,” whose extinction might have profound and long-lasting effects (Paine, 1966; Chase and Leibold, 2003). However, other studies—including a number of paleoecological studies—suggest that communities can retain fundamental structure and function despite continual turnover in species composition driven by routine geographic range shifts and extinctions (Valentine and Jablonski, 1993; Holland and Patzkowsky, 2004; S.T. Jackson and Overpeck, 2000; Webb et al., 2004). This suggests that there can be considerable interchangeability among species within ecological and functional categories. Are these apparently conflicting perspectives an artifact of scale? Paleoecological studies can take advantage of natural experiments in which community
|
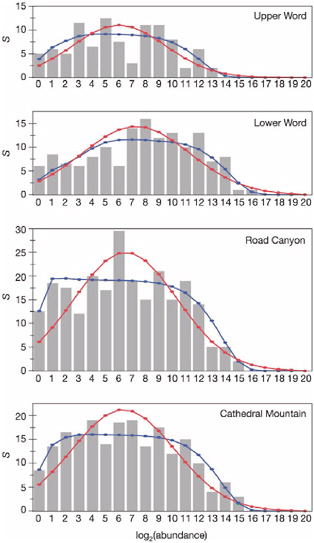
BOX 3.2 Paleobiological data can be used to provide tests of fundamental ecological theory. Permian rocks in west Texas, deposited 275-265 Ma ago, contain well-preserved and well-studied assemblages of brachiopods. More than 850,000 specimens are now housed at the National Museum of Natural History in Washington, D.C. Olszewski and Erwin (2004) tallied these specimens in order to determine the species abundance distributions and how these distributions changed through the 10-million year interval. Maximum likelihood fits to the species abundance curves (shown below) were closer to the zero-sum multinomial distribution predicted by Hubbell’s (2001) neutral model of ecological communities than to the classic log-normal distribution. The shapes of the distributions of the lower two intervals differ significantly from those of the upper two intervals, and these differences are attributed to restrictions in population size, decreased isolation, and decreased chances of immigration resulting from lower sea levels in the upper two intervals. Accordingly, the geologic record of Permian fossils in this area preserves the results of a natural experiment on how environmental change affects ecological community structure over long time periods. |
composition has changed with range shift or extirpation to assess this question. Similarly, little is known about the timescales and patterns of survival in remnant populations and the consequences for community structure. Is richness conserved? Do new species substitute for lost ones? Are functional groups thinned proportionally? Understanding these dynamics in terms of general principles, and at the timescales at which
species migrations and community turnover actually occur, will contribute significantly to our understanding of individual species behavior and the net outcomes for biodiversity and community structure in habitat fragments—an understanding that has obvious practical implications for management of natural systems in the face of human activities and impacts.
One reason why migration has emerged as such an important species response to environmental change is that species often exhibit evolutionary stasis, and are insufficiently malleable in an evolutionary sense to adapt to radically altered conditions. The fossil record clearly indicates pervasive evolutionary stasis in diverse groups in the face of substantial environmental change (Huntley et al., 1989; Coope 1995; J.B.C. Jackson and Cheetham, 1999; J.B.C. Jackson and Johnson, 2000). However, evolutionary responses have been documented in several cases (e.g., Smith et al., 1995; Rousseau, 1997; Benton and Pearson, 2001). What permits or drives some species to depart from stasis and thus adjust to changing physical and biological environments? Do the evolutionary responses observed represent expansion into new niche space or redistribution of existing variation (Huntley, 1999; S.T. Jackson and Overpeck, 2000)? These questions are particularly pressing in the tropics, where many species are linked in close ecological partnerships. What intrinsic biological or environmental factors promote the coevolution of “mutualists”1 or of producers and consumers? Paleoecological records suggest that coevolutionary adjustments may have been very rapid, given the rapid pace of Quaternary climatic change and community response documented in the tropics, but it is also possible that the coevolutionary associations observed today comprise a limited subset—those where the partners happened to migrate together over repeated glacial/interglacial cycles. These alternative explanations suggest radically different potentials for the regeneration capacity of coevolutionary partnerships that have been disrupted. This can only be tested—using the fossil record—by determining the antiquity of such partnerships. Although molecular data can provide information on the age of taxa, their mutual readjustments can only be documented using morphological and paleobiogeographic data from geohistorical records (e.g., Labandeira, 2002).
Rules of Extinction. When the rate or magnitude of the environmental change exceeds the ability of a species to adapt or migrate, the result is local, regional, or global extinction. Because global extinction is irreversible and because even local extinction can remove key genetic resources and severely perturb communities, one fundamental aim of conservation is to minimize extinction or at least to minimize its effects. Consequently, a high priority for managing the present day biota is a set
of predictive rules for species extinction and survival—both the overall rates (what proportion lost?) as well as patterns of selectivity (which species or kinds of species, which habitats, which precursor states?). In a given situation, these rules probably vary according to the magnitude and kind of disturbance, the intrinsic characteristics of the species involved (e.g., reproductive rates, geographic range sizes, niche parameters), or the original richness, spatial extent, and productivity of the community (i.e., the ecological context). The fossil record affords an opportunity to test these factors across a range of extinction intensities and drivers. Some commonalities are beginning to emerge among taxa and across time intervals (e.g., the effect of geographic range, body size, relative abundance; see Jablonski, 1995; McKinney, 1997; Purvis et al., 2000; Manne and Pimm, 2001; Harcourt et al., 2002), but more comparative work is needed to expand both taxonomic and ecological coverage, and to test whether survivorship patterns change qualitatively with disturbance intensity or type. This is an area where modeling efforts and simulations, through collaborations between paleontologists and ecologists could be particularly productive. Such models could act as a spur and guide for paleontological field work to iteratively refine the models. Determining the characteristics of demonstrably resilient systems or groups, particularly in relation to biotic or environmental crises, will be critical for effective management of diversity as a whole. Resilient groups may not have characteristics considered desirable from a human perspective.
Community Structure: Unity, Anarchy, or Both? Geologic records of different ecosystems and time periods often yield contrasting views of community unity and integrity. Paleobiological records indicate that terrestrial and temperate marine communities did not migrate as cohesive, integrated units in response to Quaternary environmental changes. Instead, species shifted their geographic ranges individualistically, producing species associations that do not occur today (Box 3.3). This fluid pattern of community assembly and disassembly was unexpected by ecologists, and speaks to fundamental questions of the inertia of community structure, the strength and particularity of biotic interactions among species, and the likely consequences of species extinctions and invasions for community resilience. Some systems, however, such as tropical coral reefs, show greater stability of community composition during the Quaternary (J.B.C. Jackson, 1995; Pandolfi, 1999; Pandolfi and Jackson, 2001), and many pre-Quaternary paleobiological studies suggest long intervals of community stability (Brett et al., 1996; Ivany, 1996; Schopf and Ivany, 1998). Assessment of the origin of this variation—whether it stems from the nature of the physical environment (S.T. Jackson, 2000; S.T. Jackson and Overpeck, 2000), differential resilience of communities to
|
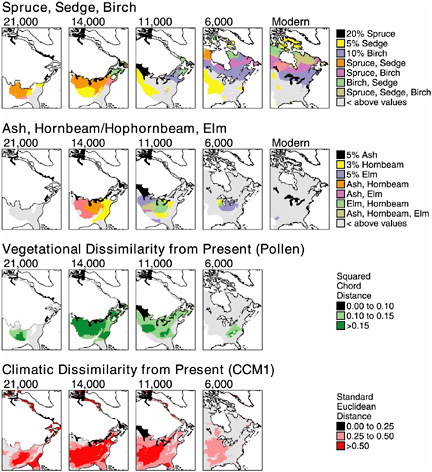
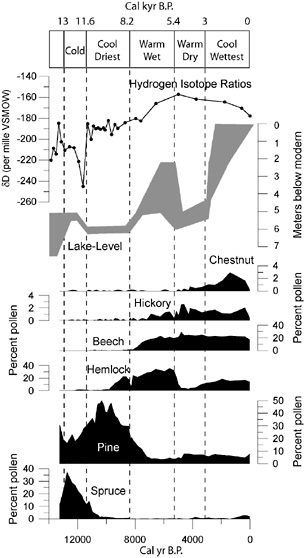
BOX 3.3 One of the most important insights to emerge from the late Quaternary fossil record is the existence in the recent past of biotic assemblages that have no modern counterparts. For example, sediments of Cheek Bend Cave in Tennessee that date to 17,500-17,000 years ago contain bones of yellow-cheeked voles, a tundra/boreal-forest rodent of northwest Canada and central Alaska, and plains pocket gophers, a grassland rodent of the central United States. Today, populations of these species do not occur within 1,000 km of each other. High-precision dating of the bones confirms that these species lived near the cave at the same time during the last glacial/interglacial transition (Stafford et al., 1999). Vertebrate faunal records from Cheek Bend Cave and numerous other sites contain diverse examples of communities with no modern analogs, and similarly singular assemblages have been documented for terrestrial insects (Coope, 1994) and marine mollusks (Roy et al., 1995). Similar results come from studies of past terrestrial vegetation; pollen and plant macrofossil assemblages with no modern analogs are well documented in a variety of continental settings (S.T. Jackson and Williams, 2004). For example, vegetation of the upper Midwest was occupied during the last deglaciation by forests dominated by boreal conifers (spruce) and cool-temperate hardwoods (elm, oak, ash, hornbeam) (Williams et al., 2001). Such forests do not occur anywhere in North America today. A critical question for both ecology and conservation biology is how these singular communities arise. Their existence in the past indicates that animal and plant species have responded individualistically to environmental changes of the late Quaternary, and that communities may be ephemeral at timescales beyond a few thousand years. Did they develop because species range adjustments through dispersal and colonization could not keep pace with the rate of environmental change? Or did they arise as a result of unique combinations of environmental variables? Paleoecologists have pursued these questions vigorously during the past two decades. A recent study by Williams et al. (2001) comparing fossil pollen data with paleoclimate simulations has supported the latter hypothesis (see figure below). Regions and time periods characterized by pollen assemblages lacking modern analogs closely match those with simulated climate regimes lacking modern analogs. Simulated climates of the late glacial period (18,000-10,000 years ago) were drier and had greater temperature seasonality than modern climates of the region. From this perspective, the modern suite of species associations is not fixed, and novel associations may be expected to arise if future climatic |
|
conditions move outside the modern range. Recent climatic trends and climate simulations all suggest that 21st century climates may have smaller diurnal and annual seasonal ranges, and the synoptic pattern of precipitation intensity and variability is likely to change (Easterling et al., 1997; IPCC, 2001; S.T. Jackson and Williams, 2004). An important question is whether we have sufficient ecological knowledge to predict which communities will be most sensitive to these changes, and which sorts of new communities will arise to replace them. Paleoecological studies can play two roles here. First, examination of the history of particular communities can indicate which communities have been most sensitive to climate changes of the recent past. Second, the no-analog communities of the past can serve as a laboratory for testing models relating modern biogeographic distributions and community composition to climate. Numerous opportunities exist for collaboration among ecologists, paleoecologists, paleoclimatologists, and modelers to determine whether past community composition can be predicted accurately from modern ecological observations and paleoclimate information. Such collaborations will help assess whether we have adequate knowledge to predict biotic responses to ongoing and future climate change. Transient effects are likely to dominate in the near term, and another pressing question is how rapidly communities will respond to climate change in the coming decades. Again, the past may serve as a guide. Analyses of high-resolution lacustrine records suggest that plant communities responded to past abrupt climate changes with lag times less than a century (Birks and Ammann, 2000; Tinner and Lotter, 2001; Williams et al., 2002) and that these responses were spatially complex (Shuman et al., 2002a). Response times of a few decades are short from a geological perspective, but highly significant on human and ecological timescales. Based on these findings, we can expect disruption of existing communities and emergence of new combinations of species associations in the near future in response to climate change, although other anthropogenic effects (land use, invasive species) will also contribute. More broadly, these studies underscore the value of Quaternary research for studying linkages between biological and physical systems at timescales intermediate between the human life span and the deep-time perspectives afforded by longer-term geohistorical records (S.T. Jackson, 2000). |
physical perturbation (J.B.C. Jackson, 1992), the taxonomic level of the investigation (species vs. genus vs. family vs. functional group), or alternations between stable and unstable environmental regimes in geologic time (Brett et al., 1996)—has important implications for ecological and evolutionary theory as well as for conservation strategies.
Recovery after Biotic Disturbance. Disturbance occurs on a variety of temporal and spatial scales. Many such disturbances are not amenable to contemporary analysis but require paleobiological data. An important unresolved issue is whether biotic response to perturbation scales from ecological to evolutionary events. Do the processes involved in response to ecological disturbances also apply to response to mass extinctions, or is there a threshold beyond which the processes differ? The variety and intensity of disturbances that species and communities can tolerate or withstand is best evaluated through the paleobiological record of past disturbances. This allows definition of the limits to disturbances beyond which the system may collapse. Repeated disturbances might weaken communities by, for example, reducing the number of connections within food webs, or may harden communities through progressive subtraction of more narrowly adapted species. Evidence for such behavior and its net consequences for community diversity and structure, and predictions for present day communities, can be acquired only from historical analysis.
In the historical analysis of recoveries, it is clear that some species that appear to be adapted for particular disturbance regimes may in reality simply have fortuitous exaptations (or pre-adaptations) that happen to be useful. For example, long-leaf pines in southeastern forests occur in a fire regime that paleobiological data suggest has only existed for the last 6-8 Ka, and to which they could not have evolved. Fire adaptations may have arisen in a very different environment. Similarly, high carbon-use efficiency in conifers—an adaptation to cold environments—may also confer advantages in the low-CO2 environments that arose during Quaternary glaciations (S.T. Jackson et al., 2000). The preferential survival of dino-flagellates relative to other phytoplankton during the end-Cretaceous extinction events may be attributable to their characteristic resting cysts, which appear to have evolved to permit dormancy under much less extreme stresses (Kitchell et al., 1986). These examples underscore that only paleobiological analysis can reveal the behavior of species over the timescales—and through the repeated natural experiments—that permit assessment of the relative importance of the different environmental and biological factors affecting biotic recovery.
Biogeochemistry
Biogeochemical cycles of carbon, oxygen, nitrogen, phosphorus, sulfur, and other elements play critical roles in the earth system, ranging from effects on local productivity to global biosphere functioning. Understanding these cycles and how they are influenced by heterogeneity in space and variability in time is a critical goal for ecology. It is also vitally important as human activities disrupt these cycles, altering their rates, pathways, and chemical transformations. Many of the processes involved in these cycles occur at timescales of hundreds to millions of years, and so geological perspectives are necessary. Two of the four “important areas of [biogeochemical] research” identified in the NRC Grand Challenges in Environmental Sciences report—“(1) Improve the quantification of sources and sinks of the nutrient elements, and gain a better understanding of the biological, chemical, and physical factors regulating transformations of nutrient reservoirs, and (2) Improve understanding of the interactions among the various biogeochemical cycles” (NRC, 2001c; p.19)—will require integration of information from the geologic record to facilitate improved understanding. Biogeochemical cycles are global in nature, but the underlying processes, particularly biological ones, are inherently local. Geohistorical records can address fundamental biogeochemical questions at spatial scales ranging from local to global, and temporal scales ranging from the recent past to the entirety of geobiological history.
Biogeochemical Fluxes and Controls: Retrospective Experiments and Chronosequences. Ecologists are concerned with understanding the processes governing biogeochemical cycles, and with assessing the sensitivity of biogeochemical reservoirs and fluxes to a wide array of perturbations—from local disturbances to global climate change. Their efforts are hampered by the wide range of temporal scales at which relevant processes occur and the limited array and duration of observational and experimental studies available. Geohistorical records can be exploited to solve some of these problems; in particular, they can be used to design retrospective experiments and for chronosequence analysis.
Retrospective experiments take advantage of natural or human-caused situations in which treatment effects and controls can be assessed using geohistorical records. Sedimentary basins—lakes, estuaries, marine basins, peatlands—provide a record of a variety of biogeochemical processes and fluxes in the form of fossil organisms, biomarkers, and sediment geochemistry. They simultaneously record information about adjacent terrestrial ecosystems (e.g., vegetation, soils) and climate. Quasi-experimental pairs or arrays of basins can be used to examine treatment versus control effects of perturbations (e.g., see Box 3.4), assess sensitivity of different systems to
perturbations, and identify dominant controls by examining differential responses of sites in contrasting environments (e.g., Whitehead et al., 1989; Ford, 1990; Ewing, 2002; Leavitt et al., 2003). This approach can even be used to provide estimates for variables not measured in long-term monitoring programs (e.g., Leavitt et al., 1989). The retrospective-experimental approach greatly extends our ability to understand the controls on biogeochemical processes.
The chronosequence approach—in which variation in space is substituted for change in time—is one of the oldest and most powerful tools employed by ecologists for studying temporal change. Historically, its primary applications have involved geological or disturbance-based contexts (respectively referring to the time since geologic origination of the land surfaces, and time since the last severe disturbance). The wide range in ages of the volcanic islands of the Hawaiian archipelago, for instance, have been used to identify rates of nutrient leaching, sources and rates of nutrient supply, and the climatic controls on these processes (Chadwick et al., 1999; Hotchkiss et al., 2000). Information from paleoecological studies comprises a vastly underutilized context for chronosequence studies of biogeochemical flux. Such studies provide site-specific information on the timing of immigration of dominant species, conversion of one vegetation type to another (e.g., steppe to woodland, tundra to forest), and invasion of new plant functional types (e.g., conifers into hardwood forests) (e.g., Björkman and Bradshaw, 1996; Peñalba and Payette, 1997; Davis et al., 1998; Lyford et al., 2003; Betancourt, 2004). Ecologists can utilize this information to devise sampling arrays to assess the effects of a range of factors (e.g., species composition, plant functional types, and vegetation physiognomy) on soil chemistry, watershed hydrology, decomposition rates, disturbance regimes, and other ecosystem properties important to biogeochemical cycling. Abundant opportunities exist for collaboration between ecologists, paleoecologists, and geologists to identify appropriate chronosequences at a broad range of scales.
The Evolution of Global Biogeochemical Cycles. The fundamental understanding of the dynamics and controls of biogeochemical cycles must rest on a foundation composed of the entire history of the biosphere (Schlesinger, 2004). Biogeochemical cycles have evolved together with the biota of the planet, and the evolution of these cycles has involved reciprocal effects of biogeochemical and environmental changes on the biota. Furthermore, because of the diversity of rates and response times among the various components of the earth system, these effects have played out over a vast array of timescales. Determining how these cycles have changed—and why—constitutes a major scientific challenge that requires
|
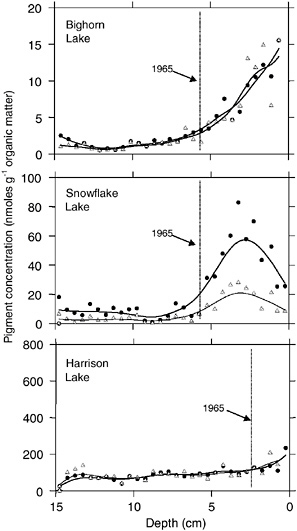
BOX 3.4 Fish introductions and invasions are widespread in North America, and they will continue as management agencies continue to stock lakes and streams, and as fish invade new waters as a result of human activities or by natural means (Rahel, 2000). Fish introductions can alter food-web structure and extirpate native amphibians, zooplankton, and benthic macroinvertebrates. A recent collaborative study involving surveys, experiments, modeling, and paleolimnology has revealed that trout introductions to oligotrophic lakes (i.e., clear lakes with low primary productivity as a result of nutrient limitations) can have important biogeochemical effects, ultimately leading to increased primary productivity in planktonic algae (Schindler et al., 2001). Trout feed on amphibians and benthic macroinvertebrates along the shallow margins of lakes, and they excrete phosphorus-rich wastes into the water column throughout the lake. In doing so, the fish transfer phosphorus from terrestrial ecosystems and the benthos—which would normally be recycled within the benthos or trapped in sediments—to the pelagic system. Experimental and observational studies in lakes indicate that this transfer can lead to a significant increase in phosphorus availability to phytoplankton (Schindler et al., 2001). A retrospective experiment was performed, using algal biomarkers in sediments to test whether fish stocking in the 1960s influenced primary productivity. Paleolimnological studies of three lakes, each with a different fish-stocking history, show clear responses consistent with the nutrient-transfer hypothesis: the unstocked lake (Harrison Lake; see figure below) showed no change, whereas a lake with successful stocking (Bighorn Lake) showed a persistent increase in algal productivity. Fish were stocked in a third lake (Snowflake Lake), but disappeared within two decades. The return of algal productivity to pre-stocking levels indicated that lakes can recover when fish populations decline or are removed. Schindler et al. (2001) concluded that management and restoration of historically fishless lakes should begin with a moratorium on additional stocking. Trout were introduced for the first time to Bighorn Lake in 1965, and brook trout populations have been self-sustaining in the lake ever since. Trout were also introduced repeatedly to Snowflake Lake in the early to mid-1960s. However, these populations were not self-sustaining and were extinct by the early 1980s. Harrison Lake has never been stocked, but it sustains a native population of bull trout. The unstocked lake—Harrison Lake—shows little change in algal productivity during recent decades. Bighorn Lake, with fish populations since 1965, shows a steady increase in productivity following the original stocking. Snowflake Lake shows an |
the marshaling of resources and fostering of collaborations among the earth and life science communities.
Great progress in understanding the biogeochemical evolution of the biosphere has been made in the past two decades. Carbon cycling has been a particular focus of attention (e.g., see Box 3.5), both because biogeochemical cycles of other elements are tied to it, and because of the need to understand controls of global carbon fluxes and reservoirs in the context of the anthropogenic CO2 increase (Berner and Konthava, 2001; Sundquist and Visser, 2004). However, many critical questions remain, and the biogeochemical history of other elements is more poorly known. Is it possible to predict past dynamics of oxygen, phosphorus, iron, and nitrogen cycles based on knowledge of past carbon cycling? Are interactions among these cycles contingent on particular configurations of climate, sea level, and continental position? Are couplings among cycles contingent on the existence (or absence) of particular types of organisms or metabolic pathways? Furthermore, development of improved paleobiogeochemical proxies is needed to extend our understanding in deep time.
Scientific advance in addressing these and other questions will proceed most rapidly and effectively using a “critical intervals” approach, in which resources are focused on particular periods of time that are likely to be particularly revealing (Stanley, 1997). These can include periods for which diverse, detailed information is readily available, periods with important system transitions or excursions, periods with unique configurations of the earth system, and periods for which processes can be linked with understanding and modeling of modern systems or biota. The latter can range from anaerobic, prokaryotic systems of the Proterozoic, to the modern biota of the late Quaternary. For the Phanerozoic, periods in which correlations of events or time series between marine and terrestrial domains should be emphasized. A hierarchical, “zoom-lens” approach can be taken in many cases. For example, the Eocene-Oligocene interval might be studied at a coarse scale, with intensified efforts focused on specific events such as the early Eocene thermal maximum and the rapid cooling event at the Eocene-Oligocene boundary (Zachos et al., 2001).
These efforts will require collaborations spanning much of the earth and environmental sciences, and integrating data collection, database development, and modeling. Some initial efforts at such collaborations have been made for some geologic intervals, notably the Proterozoic, Cenozoic, and Quaternary (e.g., Anbar and Knoll, 2002; Bigelow et al., 2003; Kaplan et al., 2003; Knoll, 2003; Rothman et al., 2003; Wing et al. 2003).
Dynamics of Biotic Invasions
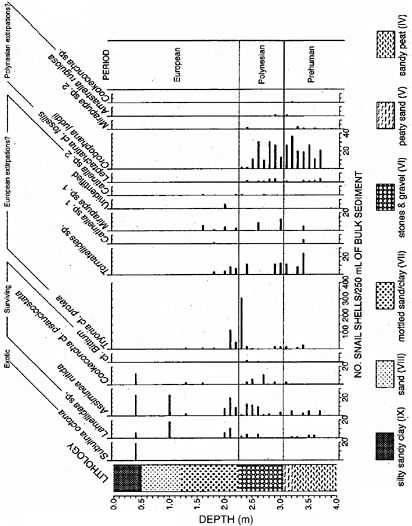
Species introductions, both deliberate and accidental, are having dramatic ecological effects across the globe (Mooney and Hobbs, 2000; Mack et al., 2003; NRC, 2004). The long-term ecological and evolutionary consequences of these exchanges are as yet poorly understood. Geohistorical records of past invasions induced by climate change, dispersal, and continental drift provide a series of natural experiments that can help assess future community and ecosystem consequences of biotic invasions. For example, the past biotic interchanges from North Pacific to North Atlantic, between North America and South America, and between eastern Asia and western North America, recognized on the basis of phylogenetic and biogeographic analyses of pre- and post-interchange biotas, are rich in insights into the asymmetry seen in most biotic interchanges, the differential invasive properties of species, phylads,2 and functional groups, and the contrasting regional biotic histories of donor and recipient areas (Vermeij, 1991; Jablonski and Sepkoski, 1996). Paleobiological records of species expansions into new territory following climatic changes of the late Quaternary provide lessons relevant to modern invasions, including the relative rapidity and underlying mechanisms (Davis, 1981a; Webb, 1988; Clark et al., 1998; Lyford et al., 2003). Paleobiological approaches can also be used to investigate species introductions that occurred before adequate historical documentation (Parkes et al., 1992; S.T. Jackson, 1997; Egan and Howell, 2001).
Infectious Diseases
There is increasing awareness of the risks of outbreaks of infectious diseases in humans and other species that are important for human use or conservation (NRC, 2001c). Although epidemiological studies will likely be central to this topic, the role of environmental change and human activities in triggering disease outbreaks is being increasingly appreciated. Our ability to identify past disease outbreaks and their consequences using the geologic record is certainly limited. Nevertheless, we should be alert to the opportunities that may exist for useful investigations on this topic. For example, sedimentary records of dinoflagellate tests or biomarkers in selected areas may help determine whether recent outbreaks of Pfiesteria red tides—harmful algal blooms—are unprecedented and presumably related to human activities, or have occurred in the past with
|
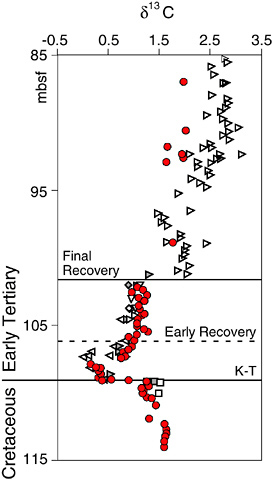
BOX 3.5 The ecological effects of the Cretaceous-Tertiary extinction in the oceans persisted for millions of years. D’Hondt et al. (1998) used carbon stable isotopes in the calcium carbonate hard parts of planktonic and benthic foraminifera to track the recovery of oceanic productivity and the flux of carbon to the deep sea. Although the productivity of surface waters was restored quickly, full recovery to pre-extinction levels of organic carbon flux to the deep sea was delayed by at least three million years. The figure above shows a sharp decrease in δ13C at the Cretaceous-Tertiary (K/T) boundary, suggesting that surface productivity was greatly reduced as a consequence of the events and extinctions occurring at the end of the Cretaceous. In the “Strangelove Ocean” (Hsü and McKenzie, 1985) that existed after the K/T event, the normal sinking of organic matter to the deep sea ceased and the difference in the δ13C composition of shallow planktic foraminifera and deep sea benthic foraminifera hard parts disappeared. D’Hondt et al. (1998) suggest that despite the return of surface productivity to pre-extinction levels within a few thousands of years (shown by the “Early Recovery” zone in the figure), final recovery to pre-extinction δ13C differences was delayed by for almost 3 million years by reduced fluxes of organic carbon. They argue that the delayed recovery was either a consequence of the small size of the surviving phytoplankton or the small size of phytoplankton grazers. The organic matter of very small phytoplankton is more likely to be recycled within the surface waters than exported to deeper water, and larger grazers are needed to aggregate biomass (as carcasses or fecal material) for rapid delivery to the deep sea. The “Final Recovery” of δ13C may reflect the evolution of larger species of phytoplankton and their grazers in the post-K/T ocean, and the consequent recovery of ecological mechanisms for the delivery of organic matter to the deep sea. The end-Cretaceous extinctions affected not only the composition of the earth’s biotas but also its ecological dynamics and carbon cycling—the ecological legacy of this mass extinction lingered in the oceans for millions of years. |
similar extent and magnitude. Similarly, molecular or serological analysis of debris from fossil rodent middens and archeological sites may provide clues on the antiquity, extent, and evolution of hantavirus. Prehistoric outbreaks of forest pathogens in North America (Davis, 1981b; Allison et al., 1986; Fuller, 1998) and Europe (Peglar, 1993) are well documented. Application of molecular techniques to the sedimentary record may help resolve the causal agents, and high-resolution paleoclimatological studies may help identify whether climate variations interacted with pathogens or vectors (e.g., Speer et al., 2001; Acuna-Soto et al., 2002). Late Quaternary megafaunal extinctions have been attributed to microbial pathogens spread by migrating humans (MacPhee and Marx, 1997), and attempts are being made to test this hypothesis using molecular analysis of fossil material (Greenwood et al., 2001).
ECOLOGICAL RESPONSES TO PAST CLIMATE CHANGE
Climate has an overarching effect on Earth’s biota—biodiversity and biogeochemistry are strongly influenced by temperature and precipitation at local to global scales. Geographic distributions and population sizes of organisms are constrained to varying degrees by climate. The tempo and pattern of biological invasions and disease outbreaks are frequently determined by climate, and effects of human land use and habitat alteration are contingent on climate. Atmospheric accretion of greenhouse gases, together with extensive land-cover changes by human activities, are raising concerns about human-induced climate change (IPCC, 2000, 2001; Pielke et al., 2002; Marland et al., 2003). However, the paleoclimate record indicates that, even in the absence of human influences on climate, climate change and variability are natural components of Earth’s system. Historical data have been crucial to disentangling potential anthropogenic and non-anthropogenic impacts.
Management of modern ecosystems (e.g., for maintenance of biodiversity, sequestration of carbon, utilization of timber, forage, fisheries, and other resources) in the face of ongoing and future climate change must be informed by knowledge of how such systems have responded to climatic variations in the past. Understanding how ecological systems have responded to past climatic and other environmental changes is necessary because the geologic record of ecological dynamics includes a vastly greater range of climatic conditions than are present today, or that are recorded in the instrumental and archival records of the past two centuries. Indeed, paleoclimatological studies of the past two decades have revealed properties of Earth’s climate system, including variability, sensitivity, control, feedbacks, and susceptibility to abrupt change, that
would have otherwise gone unnoticed (NRC, 2001c, 2002a). We would be utterly ignorant of the rates of abrupt climate change in the absence of paleoclimate data (NRC, 2002a).
The rapid, ongoing development of paleoclimatic and other paleoenvironmental records, at local to global scales, and from the Proterozoic to the late Holocene, provides important opportunities for linking records of biotic changes to the existing paleoclimatic and paleobiological datasets and records. Such opportunities exist throughout the entire geologic column. Most depositional settings where high-quality, well-dated paleoclimatic records can be extracted—whether these are high-resolution time series or more scattered samples across time—can also yield paleobiological records. Recent improvements in dating and chronostratigraphy have opened additional opportunities for correlating these records across different localities, regions, continents, and oceans, allowing climatic, environmental, and biotic dynamics to be studied together within a spatial framework. Finally, the recent and continuing development of a multitude of paleoclimate proxies (Parrish, 1998; Bradley, 1999) is driving an important transition in paleoclimatology and paleoecology. Paleoclimate inference is no longer heavily dependent on biological proxies, but can now draw on a greater variety of both biological and non-biological data. Thus, paleontological data on past distribution and abundance of organisms can now be used as response variables in studies in which climate change has been independently inferred (see Box 3.6).
Opportunities for Scientific Advance
Particular emphasis should be given to research efforts that focus on particularly well-documented and revealing time intervals in Earth history, and to work that contributes to our ability to forecast ecological responses to ongoing and future climate change. Integrated studies of paleoclimate, paleoenvironment, and paleobiology are not practical for every interval of Earth history. With limited resources and in light of variation in the quality of the geologic record at different time periods, efforts should be targeted at selected intervals in Earth history that:
-
provide high temporal resolution and easy correlation over broad areas and among terrestrial and marine records;
-
have existing or potential high-quality paleoclimate records, preferably involving multiple proxies and multiple inferred variables (e.g., temperature and precipitation; warm-season and cool-season temperatures, means and extremes);
|
BOX 3.6 Efforts to use geohistorical records to understand biotic responses to environmental change have often been hampered because the same data—pollen percentages, occurrences of plant or animal fossils—were used to infer both paleoenvironments and composition of past biotic communities. The proliferation of multiple paleoenvironmental proxies in the past two decades has now made it possible to link biotic responses to past environmental changes with greater confidence and precision. For example, pollen percentages from lake and peatland sediments—long a dominant source of paleoclimatic inferences for continental regions—are now being complemented by a variety of paleoclimatological and paleohydrological proxies, including stable isotopes, geochemical, mineralogical, and molecular markers, and microfossils of aquatic organisms (e.g., diatoms, chironomids, testate amoebae, chrysophytes). In the figure below, postglacial pollen percentages of several important forest trees are plotted for a pond in the Berkshires of western Massachusetts, together with independently derived estimates of paleoclimate from lake levels (paleohydrology) and hydrogen isotopes (paleotemperature) from a lake 200 km to the east in southeastern Massachusetts. Major vegetation transitions in the Berkshires coincide with climatic transitions recorded in southeastern Massachusetts, demonstrating the climatic control of vegetational composition and spatial coherence of climatic change at millennial scales (Shuman et al., 2004). |
-
contain existing or potential paleobiological records of adequate quality;
-
have potential for linking or nesting ecosystem dynamics across a range of timescales; and
-
provide opportunities for testing theory and integrating knowledge from ecology, evolutionary biology, and biogeochemistry.
These time intervals should collectively encompass a range of past climatic variations, both quantitative (magnitude and rate of change) and qualitative (nature of change; earth-system boundary conditions). The ideal suite of time intervals and studies can be envisioned as a set of natural experiments that together reveal the dynamics of biotic response in a wide range of habitats as driven by gradual, abrupt, and cyclical climatic changes at timescales ranging from years to millions of years.
The late Quaternary is particularly well suited for studies of biotic response to climate change because of high temporal precision, ease of precise correlation among sites, and opportunities to link past dynamics with modern ecosystems. It also provides the clearest links to ecological forecasting and risk assessment in the face of current global change. However, earlier intervals in the geologic record also provide important opportunities for studying biotic responses to climatic changes of different magnitude, quality, and duration. The pre-Quaternary record is a rich source of information on how biological diversity and biogeochemical cycling responded to changes in climate at different scales (e.g., transitions from greenhouse to icehouse worlds) and rates. Furthermore, because biodiversity and biogeochemistry both interact with climate change (NRC, 2001c), they must be studied in an integrated fashion.
Integrating Ecology and Climate Change
Ecologists have long recognized the influence of climate on global biodiversity, primary productivity, species distributions, and biogeochemical cycles. Although other ecological phenomena, including population size, population dynamics, community structure, and evolutionary adaptations have been traditionally ascribed to biotic interactions (competition, predation, mutualism), there is increasing recognition that climate plays an important, often dominating role (Post and Stenseth, 1999; Post et al., 1999; Ackerly, 2003; Hengeveld, 1990; Stenseth et al., 2002; Chavez et al., 2003). Because climate varies significantly at ecological and evolutionary timescales, and because climate exerts direct and indirect influences on ecological systems at all levels of organization (organismal to global), a clear need exists for studies targeting climatic influences
across a wide array of ecological systems, timescales, time periods, and spatial scales.
Biotic systems also affect climate through their effects on parts of the climate system such as carbon cycling, albedo, and the production of naturally occurring greenhouse gases. Geohistorical records provide abundant opportunities for learning about how the physical environment, particularly climate, constrains or determines features of the biosphere at scales ranging from genomic to global biogeochemistry and about how biotic systems affect climate. Following are select examples of how geohistorical approaches are contributing to fundamental understanding of ecological systems in the context of climate change and variability.
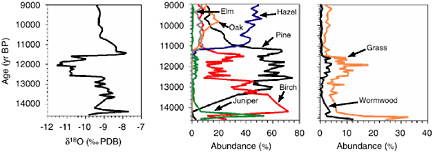
Climate Change and Community Reorganization. Studies of climate change during the last deglaciation reveal that millennial-scale climatic change leads to recombinations of climate variables at individual sites (e.g., different combinations of seasonal temperatures, or temperature and precipitation), and qualitative alteration of geographic and elevational gradients in climate (e.g., changes in the slope of the relationship between seasonal temperatures) (Kutzbach and Webb, 1993; Kutzbach et al., 1998). These patterns of change have profound ecological implications (Webb, 1987; S.T. Jackson and Overpeck, 2000; Webb et al., 2004). Paleoecologists had long recognized late Quaternary biotic assemblages that lacked modern counterparts (see Box 3.3). The existence of past climates lacking modern analogs provided a powerful potential explanation, which was corroborated by the demonstration that peculiar communities were concentrated in regions and time periods where prevailing climate was unlike any existing today (Williams et al., 2001; S.T. Jackson and Williams, 2004). It is now becoming increasingly evident that many—perhaps most—communities undergo extensive reorganizations in response to climatic changes at millennial timescales and beyond. The full ecological and evolutionary implications of this phenomenon have yet to be explored.
Community-level response to climate change represents a potentially fruitful area for integrated studies of climate change and paleontology both in the Quaternary and earlier in Earth’s history. Some communities may be less likely than others to undergo disassembly in response to environmental change and whether such differential responses show systematic patterns as a function of latitude, diversity, food-web structure, environmental plasticity, or other properties deserves further study. For communities that do undergo disassembly, we need to explore the underlying patterns and processes. For example, does disassembly consist of gradual turnover as local populations of some species are extirpated and other species colonize? Or do communities persist until some threshold is
reached, undergoing collapse and rapid replacement? What roles do disturbances and climate extremes play in these responses? Studies focused on these questions are needed to assess the role of non-linearities and threshold effects, and determine the roles of climate extremes, disturbance, and ecological properties (population size and connectedness, generation time, dispersal capacity) in governing community responses to climate change. They will also help assess community-level consequences of disassembly. Are there predictable consequences for species richness, representation of functional groups, or food-web structure? Understanding these dynamics will enrich our fundamental understanding of ecology as well as determine risks and consequences associated with future climate change.
Climate Change and Ecosystem Function. Ecosystem function depends on the prevailing climate regime (including variability) and on structure and composition of biotic communities, particularly vegetation. A central goal of global change biology is to develop capacity to predict how climate change will affect—both directly and indirectly—such ecosystem properties and services as primary productivity, carbon storage, biogeochemical cycles, disturbance regimes, atmospheric feedbacks, and soil stability. Climate exerts direct control on many of these properties for terrestrial systems, and influences physical properties of vegetation and composition of the biota, which influence other ecosystem functions. Aquatic ecosystems are also directly influenced by climate (temperature, hydrology) and by properties (productivity, disturbance, biogeochemical flux) of terrestrial ecosystems in catchment basins.
Changes in ecosystem function resulting from climate change and/or vegetational change are well documented from paleoecological records in both terrestrial and aquatic ecosystems. Climate changes of the recent past have often had dramatic ecosystem consequences, some far-reaching in nature. Fire regimes have undergone shifts in ecosystems across North America during the past 12,000 years (Clark et al., 1996, 2001b; Carcaillet et al., 2001; Veblen et al., 2003), with consequent changes in watershed hydrology and geomorphology (Meyer et al., 1995; Meyer and Pierce, 2003). Vegetation shifts involving major changes in important plant traits (Lavorel and Garnier, 2002; Chapin, 2003) have led to changes in landscape stability and biogeochemical flux. For example, early Holocene soil stabilization and humification associated with climate-driven replacement of tundra woodland by coniferous forests led within a few centuries to widespread ecosystem acidification (Whitehead et al., 1989; Ford, 1990), providing a baseline against which to evaluate industrial acidification (from “acid rain”) of the 20th century. More subtle changes in biogeochemical flux are associated with Holocene shifts in forest dominants
(Whitehead et al., 1989; Willis et al., 1997; Ewing, 2002; Ewing and Nater, 2002). Finally, climate-driven vegetational shifts have synergistic effects with the climate system, owing to vegetation/atmosphere feedbacks (Kabat et al., 2004). For example, such feedbacks were responsible for deep penetration of early Holocene monsoons into the African continent (Kutzbach et al., 2001).
Many of these ecosystem-level responses took several centuries to play out, owing to the slow rates of many biogeochemical and other processes and system-response lags. Such slow changes are difficult to fully understand in the absence of geohistorical records, and changes of the past century or two are confounded by direct human impacts (e.g., land clearance, damming/diversions, pollutants). Opportunities abound for further exploitation of the fossil record to understand climatic influences on ecosystem function, particularly with the rapid recent development of proxies for paleoclimatic inference, tools for paleoecological assessment, and ability to obtain precise dating of sediments and events. Furthermore, ongoing activity in studying and modeling ecosystem function in relation to climate change provides a rich source of theory and hypotheses for testing using the fossil record. The extent to which ecosystem function is directly controlled by climate versus indirectly influenced via vegetation dominants or individual species is still being debated. Geohistorical records provide opportunities for teasing apart the respective roles of climate and biota in controlling ecosystem properties. A variety of natural experiments exist in the past that can be studied to address these questions and test models of future climate-change impacts on ecosystems.
Evolutionary Effects of Climate Change—Integrating Paleobiology and Genetics. Biotic responses to climatic changes of the Quaternary have involved large- and small-scale shifts in geographic distributions, expansions and reductions in population sizes, and fragmentation and coalescence of populations. These responses have long been recognized as having significant evolutionary and genetic consequences, and Quaternary events have traditionally been viewed as key to explaining speciation events and patterns of genetic diversity within species. The past decade’s advances in molecular and mathematical tools for genetic analysis (Avise, 2000; Hewitt, 1999, 2000), together with the increasingly detailed records of biogeographical and ecological changes of the late Quaternary, are providing opportunities for synthesis of evolution, ecology, and climate change (Petit et al., 2002a,b). We are now in position to apply paleobiological and molecular-genetic approaches in an integrated fashion to track population-level dynamics in response to climatic change, and to assess the roles of isolation, bottle-necking, dispersal, migration, and other events in shaping the genetic structure and ecology of existing populations (Hewitt, 2000;
Jacobs et al., 2004). These efforts can shed light on how climate variation and change have influenced the evolutionary dynamics of populations and species, and provide a basis for forecasting genetic and conservation implications of future change.
Studies of DNA from fossil material, although apparently limited to a handful of paleontological settings in the late Quaternary, can reveal spatiotemporal patterns of genetic and evolutionary responses to environmental change (Poinar et al., 1998, 2001, 2003; Kuch et al., 2002; Hofreiter et al., 2003; Willerslev et al., 2003; Hadly et al., 2004). Application of these kinds of studies to sensitive species during past periods of abrupt climate change can determine the role of environmental extremes in shaping evolution and adaptation (Ackerly et al., 2000; Gutschick and BassiriRad, 2003). Similar efforts aimed at periods of non-analog climates and communities can be used to test whether novel environmental changes lead to predictable adaptive responses (Ackerly, 2003).
The fossil record of the pre-Quaternary provides important opportunities for learning about the impact of climate change on evolution and ecology (e.g., see Box 3.7). The modern biotas have evolutionary origins in the pre-Quaternary, with most intergeneric divergences and many species divergences dating to the Tertiary (Avise, 2000). Understanding how these divergences and associated adaptations arose in relation to pre-Quaternary climate changes is necessary to understand the ecological and genetic properties of extant species. Furthermore, the pre-Quaternary record contains a vast number of natural experiments in which the effects of climate changes varying widely in rate, magnitude, duration, and global context can be explored. These experiments can be utilized to answer fundamental questions about ecological and evolutionary patterns and theory (e.g., McKinney and Drake, 1998; Erwin and Wing, 2000; Gastaldo and DiMichele, 2000; J.B.C. Jackson and Johnson, 2000, 2001; Wilf et al., 2001).
Forecasting Biotic Responses to Future Climate Change
Climate change represents one of the great environmental challenges faced by human society, and successful planning and decision-making depends on our ability to anticipate future changes and their consequences (NRC, 2001c). A science of ecological forecasting is developing rapidly, focusing on predicting effects of climate, human activities, invasive species, and other factors on ecosystems, ecosystem services, and biological resources (Clark et al., 2001a). To date, with a few notable exceptions (e.g., Swetnam et al., 1999), ecological forecasting has made little explicit use of historical perspectives.
|
As the mean annual temperature of the site increased from an estimated 14.4 ± 2.5°C in the Paleocene to 21.3 ± 2.2°C in the Eocene, both the diversity of insect damage and the frequency of herbivory increased. The geologic record of global warming during the Paleocene-Eocene provides a laboratory in which to examine the response of plant-insect interactions to climate change. |
Paleoclimatological data and perspectives are playing an increasingly important role in climate forecasting and risk assessment, particularly in context of the ongoing severe drought in the western United States. Climate scientists have recognized that the instrumental record of the past century provides a miniscule sample of the range of natural climate variability and is too short to evaluate dynamics at decadal timescales and beyond. Furthermore, climate variations of the 20th century have been clouded by human activity, including trace gas emissions (e.g., carbon dioxide, methane), aerosols, and land-use/land-cover change, so it is difficult to differentiate non-anthropogenic influences from human ones. Thus, paleoclimate studies, ranging from records spanning the past few centuries to records covering the last few glacial/interglacial cycles, are central to climate forecasting. In fact, current concerns in climate policy over abrupt changes driven by non-linearities in the climate system (NRC, 2002a) have arisen entirely from paleoclimate records which indicate that such changes and responses are intrinsic features of the earth system.
The rich geologic record of biotic responses to past climate changes is an essential component of ecological forecasting. In its absence, we are left with a depauperate view of the full range of ecological consequences of climate change, and are hampered in separating direct effects of climate from other factors (e.g., human activities, direct response to trace gas concentrations). We discuss three areas of particularly critical need for historical and geohistorical perspectives: (1) natural resource management, (2) abrupt climate change, and (3) conservation of biodiversity. Paleobiological studies are already making substantial contributions in a fourth key area, the parameterization and testing of models of regional and global climate change (Kohfeld and Harrison, 2000; Kutzbach et al., 2001; Kaplan et al., 2003).
Climate Variability and Management of Natural Resources
Just as climate scientists have come to recognize that the instrumental records spanning the past century or two fails to include the full range of climate variability relevant to planning and risk assessment, applied ecologists and resource managers are realizing that 20th-century monitoring provides an inadequate baseline for assessing variability in population size, disturbance frequency, and productivity in ecosystems. Furthermore, human activities such as timber harvesting, fire suppression, and livestock grazing have altered natural variation and distorted baselines. Resource managers are turning to the concept of “range of natural variability” or “historic range of variability,” as a baseline for management (Swanson et al., 1993; Kaufmann et al., 1994; Landres et al., 1999). These applications often focus on the mid-19th century, or the decades immediately preceding extensive Euro-American settlement, as the natural baseline for management targets. However, most of the properties of interest to managers (e.g., population size, disturbance frequency, regeneration rate, discharge) are tied to climate, and climate variability of the 19th century is as unrepresentative of the preceding several centuries as it is of the 20th century (Bradley and Jones, 1992; Cook et al., 1999). Furthermore, some ecological phenomena vary at lower frequencies that require records spanning multiple centuries to detect. Resource management will benefit from records spanning several centuries or more to identify the controls on ecosystem properties and to assess the full range of inherent variability.
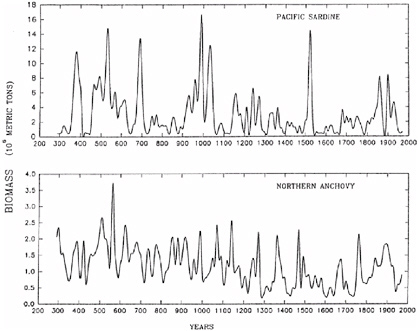
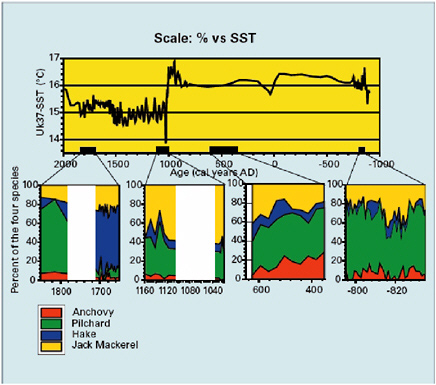
Perspectives spanning hundreds to thousands of years are being applied in fisheries management and vegetation management in a few areas. Fire scar, woodrat midden, sedimentary charcoal, and paleoclimate studies are influencing management of forests and woodlands in parts of western North America (Millar and Woolfenden, 1999; Swetnam et al., 1999). Reconstructions of marine fish populations (e.g., sardines, anchovies) from fish scales accumulated in sediments are being used in tandem with paleoclimatological, paleoceanographic, and modern ecological studies to develop predictive frameworks for fisheries management (Baumgartner et al., 1992; Schwartzlose et al., 1999; see Box 3.8). These syntheses and applications are important because they allow identification of the relative impacts of climate variability and human impacts (e.g., harvesting, fire suppression) on natural resources, and provide a great deal of predictive power by linking resource properties (population size, productivity, fire risk) to climate variability at interannual, decadal, and multidecadal timescales. Diverse ecological phenomena, ranging from population size to vertebrate sex ratios to food web structure are being linked to interannual climate variation (El Niño-Southern Oscillation [ENSO], North Atlantic Oscillation [NAO], Arctic Oscillation [AO]) (Lima
|
BOX 3.8 Many marine fisheries have undergone catastrophic declines in the past four decades. These events are often attributed to overfishing or poor management practices. The 1972-1973 collapse of the Peruvian anchoveta fishery is one of the most celebrated cases. This event, however, coincided with an unusually strong El Niño event, and the plummet of the anchoveta populations is now regarded as resulting from a combination of fishing practices and the interruption of nutrient upwelling during the El Niño event. Populations of fish and other marine organisms change in size and location in response to climate variations; for example, marine fish fluctuations of the past century have been linked to ENSO (El Niño-Southern Oscillation) variation as well as lower-frequency variations in the Pacific Basin (e.g., Pacific Decadal Oscillation [PDO]) (Mantua et al., 1997; Chavez et al., 2003; Lehodey et al., 2003). Pelagic fishes—such as sardines and anchovies—slough off scales as they grow, and these scales are preserved in sediments of deep, anoxic basins. Fish scales from sediment cores of these basins provide high-resolution (often annual) records of changing biomass, species composition, and age structure of pelagic fish communities, spanning hundreds to thousands of years. These geohistorical records are providing information on the natural variability and climatic controls of marine fisheries. In the Santa Barbara Basin off the California coast, sardine and anchovy production has fluctuated tenfold during the past 1,700 years, with rapid increases and equally rapid declines (see figure below). Spectral analysis of these records reveal high-frequency variation (period of 50-60 years), possibly linked to PDO variation, and lower-frequency trends, which are correlated with the Medieval Warm Period (10th to 14th centuries) and the Little Ice Age (15th to 19th centuries) (Baumgartner et al., 2004). |
et al., 2001; Post and Stenseth, 1999; Post et al., 1999; Grant et al., 2000). These interannual variations are superimposed upon lower-frequency variations, and their regional expressions are often contingent on the lower-frequency dynamics (e.g., Dettinger et al., 2001; Enfield et al., 2001). Thus, scaling up of ecological responses in a forecasting framework may not be straightforward, and will require longer-term data and perspectives. Forecasting must be supported wherever possible by longer-term historical studies. For many important ecological phenomena, it may be impossible to obtain appropriate paleontological records, so integration of ecological modeling with paleoclimate records will be necessary. But such applications will need to be validated, and opportunities for directly linking geohistorical studies to ecological and paleoclimatological studies should be exploited when they arise.
Ecological Consequences of Abrupt Climate Change
Earth’s recent climatic history has been punctuated by abrupt, high-magnitude changes, ranging from temperature shifts of up to 16°C and twofold changes in precipitation spanning decades during the last deglaciation to widespread multidecadal droughts during the past 2,000 years (NRC, 2002a). Documentation of these abrupt changes demonstrates that Earth’s climate system is strongly influenced by nonlinear effects, and that it is capable of rapid shifts that are likely to have vast societal, economic, and ecological consequences (NRC, 2002a; Alley et al., 2003). Abrupt climate change has become a major research focus in the context of global change, and paleoclimate studies continue to play a major role in understanding mechanisms of abrupt change as well as assessing risks.
Ecological consequences of abrupt climate changes have received comparatively little attention, even though such changes were first identified from paleontological records (Younger Dryas interval) and tree ring studies (late Holocene droughts). Yet the evidence available indicates that ecological impacts were large and widespread. The Younger Dryas interval, an abrupt cooling concentrated in the North Atlantic region 12,800-11,600 BP, resulted in vegetation changes over much of the globe (Peteet, 1995), many of them occurring within a century of the climate change (Birks and Ammann, 2000; Shuman et al., 2002a,b; Williams et al., 2002). The Younger Dryas represents a model system for exploring the biotic implications of large-magnitude, rapid change (see Box 3.9). Did widespread mortality of tree and shrub populations lead to subsequent wildfires? Did vegetational transitions involve rapid migration (e.g., Webb et al., 2004), or alternatively opportunistic expansion of local populations capable of tolerating the new conditions? Did local and regional population extirpations
increase risk of global extinction for species? How did populations and species survive abrupt changes of the past? These questions bear directly on the likely consequences of abrupt changes in the future (NRC, 2002a).
Abrupt changes in the Holocene are also suitable model systems for studying how climatic changes of smaller magnitude and shorter dura-
tion influenced ecosystems. A transient climate anomaly associated with collapse of the Laurentide ice sheet 8200 BP had effects in the North Atlantic region and possibly beyond (Tinner and Lotter, 2001; Shuman et al., 2002a). Other events are documented from geohistorical records in the mid- to late Holocene at regional to global scales (van Geel et al., 1996, 2000; NRC, 2002a; Booth et al., 2004). Severe droughts and other abrupt events of the past 1,000-2,000 years can be documented from both sedimentary records and tree ring analyses (Woodhouse and Overpeck, 1998). These events provide unique opportunities for linking demographic studies of extant populations (Swetnam and Betancourt, 1998; Allen and Breshears, 1998; Swetnam et al., 1999) with high-resolution paleoclimate records and paleobiological records from sediments. Such integrated studies will be valuable both in forecasting ecological changes associated with future abrupt climate change and in examining the mechanisms underlying responses recorded in sedimentary records predating the past 1,000-2,000 years.
Maintaining Biodiversity in a Changing World
Conserving biological diversity and maintaining critical ecosystem services in a time of global climate change, possibly including abrupt changes and climate “surprises” (Overpeck and Webb, 2000; NRC, 2002a), is an immense challenge for scientists, managers, and policy makers. Climate-driven disruption of community structure as incumbent species populations decline and new species immigrate may compromise ecosystem services, and put many species at risk of extinction. These problems are compounded by human activities and introduced species. Habitat fragmentation may inhibit dispersal of native species to suitable habitats, and climate change may interact with wildfire, exotic invasions, and pathogen or disease outbreaks in creating stress on species, populations, and communities.
The species living on Earth today have obviously been subjected to the effects of climate change repeatedly in the late Quaternary, and past climate changes have been accompanied by disturbances, invasions, and other stresses. However, we are almost completely ignorant of what may have been lost in terms of biodiversity—both species diversity and genetic diversity—during these past episodes, and hence are severely handicapped in assessing risks and vulnerabilities for ecological impacts of ongoing and future change. The fossil record has much to contribute to evaluating the potential losses in genetic diversity, species diversity, community stability, and ecosystem services owing to climate change, as well as (conversely) understand the potential role of climate change in generating genetic diversity and ecological novelty.
A wide variety of questions of both fundamental scientific interest and practical value can be addressed using the fossil record. Have species extinctions or genetic bottlenecking been concentrated during past periods of rapid or abrupt climate change? What are the relative capacities for adaptation to environmental changes versus extinction or migration (S.T. Jackson and Overpeck, 2000; Davis and Shaw, 2001; Ackerly 2003)? Does capacity for adaptive response vary predictably among species (Hadly et al., 2004)? Does rapid migration inevitably lead to attenuation of genetic diversity (Cwynar and McDonald, 1987)? Have severe pathogen outbreaks in the past (e.g., Davis, 1981b; Peglar, 1993) been triggered by climate anomalies? Are species-based, community-based, or habitat-based strategies for biodiversity conservation most likely to be effective during a period of climate change (e.g., Hunter et al., 1988)? We know that many species that are widespread and abundant today have been rare and restricted in the past (e.g., Pinus ponderosa var. ponderosa), while species that are rare and restricted today have been widespread and abundant in the recent past (e.g., Pinus remota) (Betancourt et al., 1990; Lanner and Van Devender, 1998). We also know that abundant and widespread species can undergo complete extinction within a few thousand years (S.T. Jackson and Weng, 1999). What are the extinction risks in the face of environmental change?
An important lesson from paleoecological studies of the late Quaternary is that most terrestrial communities and ecosystems have developed within the past 800 to 8,000 years, and that most species have attained their modern distributions within that same period. Thus, the geographic ranges of species, the composition of ecological communities, and the properties of ecosystems (including diversity, productivity, and disturbance regimes) are all contingent on particular climatic regimes that have prevailed for a few thousand years at most. Understanding the causal relations between ecological properties and climate regimes, and delineating the range of climatic conditions that existing systems are capable of tolerating, is critical to ecological forecasting in the context of global change. However, modern spatial patterns of species and communities, and modern climatic gradients provide an insufficient basis for inferring the climatic limits of existing systems.
Macroclimatic regimes as recent as 9000-6000 BP have no modern counterparts (see Box 3.3), and the climate changes—even those of the past few thousand years—that led to development of modern ecosystems are poorly documented. At least some modern communities developed rapidly, suggesting either non-linear relationships between climate change and community composition or a major role of abrupt or rapid climate change. In most cases we do not know how much, nor what kind of, climatic change might lead to collapse of natural populations, commu-
nities, or ecosystems in the future. Because climatic change within the next century may yield climate regimes as different as those of today from those of the last deglaciation 14,000-11,000 years ago (e.g., see figure 10 in S.T. Jackson and Williams, 2004), we cannot yet assess the resilience of biotic systems in the face of future change.
Historical and geohistorical studies are essential to address these questions. Integrated studies involving retrospective approaches (e.g., paleoecology, paleoclimatology, demographic reconstructions of population history) in tandem with ecological studies (long-term monitoring; experimentation in field, greenhouse, and laboratory; modeling) would accelerate progress in both ecology and paleoecology, and provide a firm foundation for ecological forecasting related to climate change.
ECOLOGICAL LEGACIES OF SOCIETAL ACTIVITIES
During the past few centuries, and possibly for the past several millennia, the biosphere has been experiencing a situation unprecedented in its history: the dominant ecological influence of a single eukaryotic species at a global scale. Human societies have undoubtedly affected the environment and biota at local scales since the emergence of Homo sapiens some 100,000 years ago, and other hominids had ecological effects as well. However, the spread of modern humans across the globe, the increase in their populations with the development of agriculture, and the proliferation of new technologies and life styles, have led to expansion of human ecological influences from highly localized to regional and, ultimately, global in scale.
The development of ecology and other environmental sciences during the past two centuries has led to recognition of these impacts, which has led in turn to a concern with identifying ecological baselines—systems, past or present, that show no discernible human influence—with which to compare modern systems for impact assessment. Restoration ecology, conservation biology, and ecosystem management all rely on these baselines, which often include estimates of past natural variability. However, geohistorical records are providing abundant evidence of widespread human impacts on populations, communities, and ecosystems, even in places long thought to be relatively unaffected by human activities (Douglas et al., 2004; Willis et al., 2004) (see Box 3.10). Recent evidence suggests that agricultural societies had global effects on biogeochemistry at least 5,000 years ago (Ruddiman and Thomson, 2001; Ruddiman, 2003).
Although a strong case has been made for the role of humans in the extinction of large mammals in North America starting about 13,000 years ago (Martin, 1967; Alroy, 2001b), evidence for a human role in such ancient extinctions remains mixed (e.g., Guthrie, 2003, 2004) and a scientific con-
|
BOX 3.10 Even some remote lakes of the high Arctic have been altered by human activity long before the advent of industrial society. Douglas et al. (2004) examined the sedimentary and fossil record of a high Arctic lake adjacent to an AD ~1200-1600 Thule Inuit whaling settlement on Somerset Island, Canada. The lake differs from most other isolated Arctic lakes in having unusually high concentrations of total phosphorus, organic carbon, and calcium. The site is scattered with whale bones, including bones used in the construction of dwellings, 10 bowhead whale crania that may have been used in ceremonies, and scattered bones, baleen, and skin of other marine mammals. A detailed chronology of the lake sediments was provided by 210Pb dating, and archaeological materials were dated with AMS radiocarbon dating. Strongly positive δ15N values in lake sediments indicate the influence of decaying marine organic matter on the lake while the relative abundance of several diagnostic species of diatoms document the response of the lake’s flora to the human-caused influx of nutrients from decaying whale carcasses. An increase in δ15N values in the lake sediments follows the initial Thule Inuit settlement in AD ~1200, and the increase in abundance of an epiphytic diatom marks the development of moss vegetation in the lake. Clearly, decaying whale carcasses provided the unusually high nutrients that characterize this lake. Despite the site’s abandonment in AD ~1600, the decay of carcasses and continuing decay of bones has sustained the lake’s high levels of phosphorous and high δ15N values for the past 400 years. Human impact on this high Arctic lake system long preceded the influence of Europeans and lingered long after the abandonment of the settlement. |
sensus has proven elusive (Grayson et al., 2001, Cardillo and Lister, 2002; Barnosky et al., 2004; see Figure 3.1). Regardless of the role of humans in Pleistocene extinctions, there is a growing realization that we have inherited a world that has already been profoundly altered by human activities.
Organized, systematic monitoring and documentation of ecological systems date only to the 19th century, although fragmentary information can be gleaned from documentary sources and museum collections dating back a few centuries. Even for the 20th century, important gaps remain in our observational network for ecology. Failure to recognize the extent to
FIGURE 3.1 Summary of the numbers of megafaunal genera (genera consisting of species with body sizes >44 kg) that became extinct on each continent, the strength of the extinction chronology, and a comparison of the timing of extinction with the timing of human arrival and late Pleistocene climatic change. SOURCE: Barnosky et al. (2004); used with permission.
which ecological systems have been altered by human activities can lead to confusion and error in ecological theory and application. The pioneers of ecology often overlooked the fact that systems they studied were far from pristine, leading them to misattribute patterns arising from human disturbance to processes such as succession (e.g., S.T. Jackson et al., 1988). We are not immune from this problem today, given the widespread impacts of humans throughout the globe.
Populations, communities, ecosystems, and the biosphere all have “memory;” they carry vestiges of past events and trends, owing to delayed response to forcings or perturbations, persistence of physical entities, and the contingent nature of many ecological phenomena on previous states or events. This “ecological memory” is encapsulated in the concept of ecological legacies, defined as current properties of an ecological system that can be explained only by events or conditions that are not present in the system today (see Box 3.11). Thus, many features of modern systems, from the absence of large size classes in coastal fish populations to the high fire risk in many western forests to the relatively high pre-industrial methane concentrations in the earth’s atmosphere, may all be regarded as legacies of past societal activities (respectively, fishing, fire prevention, and rice cultivation) (J.B.C. Jackson et al., 2001, Swetnam et al., 1999; Ruddiman and Thomson, 2001).
We can neither understand nor manage the ecological systems of our planet adequately without determining which features are ascribable to natural processes and patterns and which are ecological legacies of societal origin. The geologic record can and must play a critical role in assessing such legacies. In nearly all cases, geohistorical records provide the only source of information on ecological history that extends to pre-human (i.e., true baseline) conditions.
Opportunities for Scientific Advance
Geohistorical records can contribute to understanding of human impacts and legacies in innumerable ways, from individual lakes, wet-lands, and forest stands to regional, continental, and global scales. Particular demands will arise at the local level as resource managers and restoration ecologists come to appreciate the need for historical information and context (Egan and Howell, 2001). Greatly improved understanding and application can come from studies that integrate ecological, paleoecological, and archaeological approaches to:
-
discriminate effects of human activities from those of climate variability;
-
assess the magnitude and extent of impacts on specific ecological systems under different societies, cultures, and technologies;
-
determine how local effects of human land use and habitat alteration scale up to aggregated effects across heterogeneous regions, including entire catchment basins;
-
exploit opportunities to use past or ongoing human activities (e.g., top predator removal, landscape alteration, nutrient enrichment) to evaluate fundamental ecological theory;
|
BOX 3.11 The legacy concept in ecology originated from the observation that ecosystem recovery following large natural disturbances (e.g., the 1980 Mt. St. Helens eruption and the 1988 Yellowstone fires) was influenced by organisms that survived the disturbance as well as physical and biotic structures (windthrow mounds, dead trees, soil carbon, coarse woody debris) that persisted from the previous community (Franklin and Halpern, 1989; Foster et al., 1998). The concept has since been expanded to incorporate any property of an ecosystem that is attributable to a past event (e.g., a wildfire) or system state (e.g., a previous community at the site). For example, age structure of a forest or woodland stand may be linked to a disturbance event centuries ago (Swetnam and Betancourt, 1998; Frelich, 2002), soil structure of a forest stand may be attributable to occupancy of the site by prairie vegetation several thousand years ago, and nitrogen reservoirs in Antarctic lakes may be attributable to high lake levels of the Pleistocene (Moorhead et al., 1999). Human activities, including management practices, also impose legacies across a wide range of spatial scales (Wallin et al., 1994; Swetnam et al., 1999; Foster et al., 2003). Ecological systems carry these historical signatures for a variety of reasons. First, ecological processes occur across a wide range of timescales, so one component of a system may change rapidly in response to disturbance or environmental change while another may change very slowly. For instance, a crown fire kills most trees and burns most fine fuels in a forest stand within minutes, while coarse woody debris and soil carbon can persist for decades after the fire (Foster et al., 1998). Plant species composition can change in response to climate change within decades, while soil carbon reservoirs may require centuries or millennia to respond. Second, ecological systems can leave a strong imprint on the environment that may persist long after the originating system has disappeared or been replaced by another. For example, vegetation influences the physical and chemical structure of soils, and many species, ranging from prokaryotes to trees and mammals, have bioengineering properties, creating landforms that can persist for decades to millennia (Jones et al., 1994; Crooks, 2002). Third, many properties of biological systems are contingent on history. The genetic structure of populations contain overprints of population history (Hewitt, 2000; Petit et al., 2003), age structure of populations is often governed by previous mortality, recruitment, and/or dispersal events (Swetnam and Betancourt, 1998), and soil nutrient reservoirs are influenced by the climatic and vegetation history of the site. Human land use imposes legacies in soil and vegetation that can persist for hundreds to thousands of years (Motzkin et al., 1996; Dupouey et al., 2002; Foster and Aber, 2004). The concept of ecological legacies provides a vital link between ecology and the historical sciences. |
-
identify the differential sensitivity of ecosystems to persistent legacies of human activities; and
-
address human legacies that affect species, habitats, and ecosystems of particular management concern and societal value.
The kinds of systems and timescales at which geohistorical records can be applied to these problems are legion. Humans affect ecosystems in a variety of ways, ranging from inadvertent introduction of diseases and invasive organisms to direct utilization of native species to appropriation of entire habitats for conversion to agricultural or urban landscapes. Hunter/fisher/gatherer societies have different impacts from pastoral/agricultural societies, which differ again from industrial/urban ones. There is considerable diversity among cultural groups within each of these broad categories. And impacts of any given culture should be contingent on the local ecosystems and the prevailing environment, including climate and climate variability. All ecological studies and management efforts should be preceded or accompanied by serious investigation of the extent to which system properties represent legacies of past human activities (Hamburg and Sanford, 1986). Well-focused, integrative studies of the kind described above will put us in position to use the past to manage for the future.
Natural Variability and Shifting Baselines
Environments and biotas vary naturally around some ever-shifting mean value. The magnitude of this variation depends on characteristics of the environment as well as the frequency, duration, and temporal resolution of observations. Documenting this natural variability is important because the demonstration of human impact requires that we separate the “signal” of human action from the “noise” of non-anthropogenic variation.
Assessing the natural range of variability requires geohistorical perspectives because observational environmental and ecological records do not nearly encompass the full range of natural variability over the past few centuries, and there is abundant evidence for human impact on the environment throughout the entire observational record. Similar problems plague all ecological investigations because no living systems can be considered pristine. Completely “natural” systems (in the sense that they are unaffected by the consequences of human activity) are only available in the geologic record. Fortunately, as described earlier, the quality of environmental and biotic information in the record often allows the reconstruction of environmental variation with seasonal and annual resolution.
A common problem in measuring human impact is what fisheries biologist Daniel Pauly (1995) called the “Shifting Baseline Syndrome.”
Scientists and the general public alike tend to define a “natural” or “pristine” baseline as the way things were either when they were first observed, or from descriptions of the very recent past. Human impacts are then measured from this baseline of personal, short-term experience, despite the likelihood of prior human-caused change. For example, in fisheries, each new generation of fishers tends to resist limits to their catch because they consider the present day catch as the norm, rather than the enormous catches of the previous generations. The now-classic example concerns codfish from the Gulf of Maine—for the 5,000 years leading up to the 20th century, the average size of fish caught and eaten was about 1 m, whereas the average size steadily decreased in the 20th century (J.B.C. Jackson et al., 2001; see Figure 3.2). Ignorance of this change allowed continued fishing when the average size had already dropped by two thirds in the 1970s and 1980s, leading to the subsequent total collapse of the fishery.
We can still unambiguously define an environment and its associated biota as pristine based on the way things were at that place—plus or minus the natural range of variation—immediately before the first arrival of modern Homo sapiens. This admittedly discounts the environmental effects
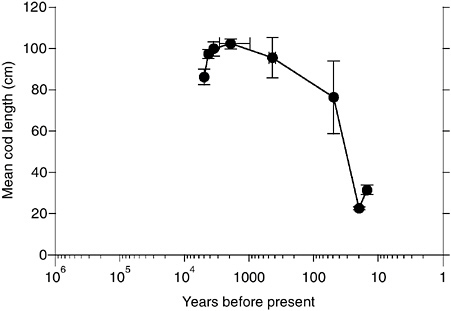
FIGURE 3.2 Time series of mean body length of Atlantic cod from kelp forests in the coastal Gulf of Maine. The earlier five data points are derived from archaeological records, whereas the last three points are from fisheries data. SOURCE: J.B.C. Jackson et al. (2001); used with permission.
of pre-modern humans in Africa before 150 Ka, but for our purposes here we can probably dismiss these effects as relatively minor. All other definitions of pristine depend on more arbitrary assessments of a lack of “significant” human impact, despite the presence of human populations, and are subject to the problem of shifting baselines noted above.
The value of recognizing the pristine condition of an environment and biota is that it provides a basic frame of reference to assess all subsequent human and natural environmental change. Most interestingly, such true baselines allow us to ask the question: “What was the world like just before the arrival of modern humans?” Of course, the first arrival of people around the world was not synchronous, and required almost the entirety of human existence from first origins in Africa approximately 150-100 Ka. Very approximately, modern humans arrived in Europe and Asia between about 100-50 Ka, in Melanesia and Australia about 60-40 Ka, in the Americas about 20-12 Ka, in Polynesia 2 Ka, and less than 1,000 years ago in New Zealand and Madagascar. This delayed arrival of people in progressively remote locations from Africa provides a powerful tool to distinguish between changes in climate and human impacts as causes for biotic change.
Assessing Human Impact
A reliable frame of reference needs to be established for evaluating human impacts over many timescales throughout the Holocene. Human land use and habitat alteration have left signatures on the landscape and in geohistorical records dating in many cases to the first human colonizations. Sedimentary charcoal indicates increased incidence and extent of wildfires upon colonization of Australia by aboriginal hunter-gatherers ~40,000 years ago. Hunter-gatherer societies throughout the world have used fire as a tool to manipulate habitat, with effects on vegetation composition and structure detectable in geohistorical records at local to regional scales (e.g., McGlone, 1989; Burney, 1999). Agricultural societies have had even greater impacts, converting natural vegetation to agricultural lands. Such conversions in the prehistoric record have ranged from transient and local (e.g., Iversen, 1973; McAndrews, 1988; McAndrews and Boyko-Diakonow, 1989) to persistent and regional (Berglund, 1992; Binford et al., 1987; Brenner et al., 2002). Societies with large populations and advanced technologies, both agricultural and industrial, have tended to have even greater and more persistent effects.
Because aquatic systems accumulate sediments and are influenced by land use in surrounding catchment basins, they are particularly effective recorders of human impacts. Lakes accumulate sediments continuously and thus are especially effective recorders of human activities in the sur-
rounding watershed (e.g., Smol, 2002; Alin et al., 1999, 2002; Cohen, 2003) as well as distant regions. For example, paleontological and geochemical studies of lake sediments in the Adirondacks and other regions provided critical confirmation that regional ecosystem acidification was a result of mineral acids from distant industrial sources (Charles et al., 1990, 1994; Battarbee and Charles, 1994). Signatures of human activities also show up in fluvial, wetland, and estuarine sediments, even though deposition may be irregular or discontinuous. Studies of estuarine sediments have revealed the extent to which coastal ecosystems have been altered by human agricultural, industrial, and fisheries activities (J.B.C. Jackson et al., 2001; Curtin et al., 2001; Cooper and Brush, 1991; Brush, 1997). Sediments of lakes and peat bogs have been used to document onset of metal mining and smelting (e.g., Shotyk et al., 1998; Martinez-Cortizas et al., 1999), providing biogeochemical baselines.
Human impacts on Holocene environments and biotas are generally much more closely related to cultural, technological, and economic attributes of societies than to the simple presence of humans (see Box 3.12). This is often most clearly evident from comparisons of human activities and their consequences during the rapid economic transitions associated with colonial occupation (e.g., the Americas before and after European conquest and colonization, Australia before and after British colonization). Other critical transitions include the advent of intensive agriculture, industrialization and resource exploitation based on fossil fuels, and transitions from local to regional to global markets.
Documentation of human-caused changes in the distribution, abundance, and size of species and the distribution and composition of communities offers the potential for reconstructing the structure and function of natural communities. This is fundamental for understanding the adaptations and limits of species that now inhabit strikingly different habitats than those lived in previously. Some possibly general patterns of the consequences of human activities have been identified, but require considerable further examination in a wide variety of settings:
-
the loss or ecological extinction of megafauna;
-
shortening of food webs;
-
distortion of biogeochemical cycles;
-
shifts from heterogeneous to homogeneous biogeographic distributions due to invasive species;
-
general loss of biodiversity; and
-
replacement of eukaryotes in the oceans by bacteria and archaea.
As pointed out by Margalef (1968), all these processes may act to reverse ecological succession while at the same time increasing produc-
|
BOX 3.12 Islands of the central tropical Pacific—Polynesia—have been invaded by two successive waves of human culture. The prehistoric Polynesian (or Lapita) culture swept westward across the region beginning about 4,000 years ago, reaching the most remote islands (Hawaii, Easter Island, New Zealand) in the past 1,000-2,000 years. The Polynesians cultivated imported crops, tended domesticated livestock (pigs, chickens), and foraged for wild plant and animal foods. They also imported dogs and rats. The late 18th and 19th centuries saw secondary colonization and conquest of the Pacific islands by European and Euro-American cultures. On the larger islands (e.g., Hawaiian islands, New Zealand), these latter cultures implemented extensive and intensive grazing (cattle, sheep) and cultivation (sugar cane) in the 19th century, as well as imported a wide variety of non-native plants and animal species. Paleontological studies provide rich records of the impacts of these activities on native fauna and flora. Easter Island saw particularly devastating impacts of Polynesians, with deforestation leading to extinction of all the native trees (Dransfield et al., 1984; Flenley et al., 1991). Native bird communities throughout Polynesia underwent rapid species extinctions as the Polynesians arrived; as many as 2,000 species of birds, comprising some 20 percent of global avian diversity, may have disappeared during the Polynesian invasions (Steadman, 1995). A recent integrated paleontological excavation on the island of Kauai (Hawaii) illustrates the severity of the losses associated with Polynesian activities, as well as the impacts of the second, European, colonization wave (Burney et al., 2001). Native species of crabs, marine mollusks, land snails, and birds that survived the Polynesian invasion were exterminated during the 19th century (see figures below). Pollen and macrobotanical data indicate that plant species that are now rare and restricted to remote montane sites were widespread in the coastal lowlands before human colonization. Few of the native vertebrate and invertebrate animals recorded in the pre-Polynesian sediments remain today. As Burney et al. (2001) observed, the paleontological record at their site “documents the purloined riches of a truly lost world.” |
tivity. Although this increases food supplies on land in the case of agriculture, the process greatly decreases food supplies in the ocean because microbes and jellyfish come to dominate the resulting communities. Simple food-web models can be employed to estimate the standing crops of species at different trophic levels using historical data for estimated abundances of apex predators that have greatly declined before ecological observations began (Christensen and Pauly, 1993). Historical data describing the composition and structure of communities offer powerful but under utilized tools for estimating the ecosystem consequences of human impacts.
Historical Ecology and Restoration Ecology
Truly pristine pre-human conditions may not provide realistic targets for resource management or ecosystem restoration. Such pristine conditions have not occurred more recently than centuries to thousands of years ago, depending on the ecosystem and its location, and the world is already in a different climate regime. Nevertheless, they are an essential frame of reference for evaluating the natural geographic ranges and habitat distributions of species, their characteristic size frequency distributions, and a host of other parameters that are the essentials of basic ecological research. Furthermore, such fundamental baselines provide goals for ecological restoration, to the extent that prevailing climatic conditions allow. And by allowing us to differentiate natural phenomena from legacies of human activities, and to determine the specific human activities to which particular legacies are attributable, they provide a sounder basis for determining realistic and appropriate management and restoration targets. Knowing whether a particular vegetation pattern or disturbance regime is attributable to natural conditions, pre-European Native American land use, or Euro-American impacts, for example, is an essential first step in management decisions. Decisions ultimately rest on a number of social, scientific, economic, and political considerations, but historical knowledge can provide more informed judgments. As an example, pinyon pines (Pinus edulis) do not grow at Chaco Canyon National Monument today. Yet paleontological studies indicate that they occurred there naturally from ~6,000 years ago until the populations were exterminated by Anasazi fuel harvesting 1,000-800 years ago (Betancourt and van Devender, 1981). This information complicates, but also enriches, decisions.
One of the greatest values of historical data describing ecological conditions before substantial human impacts is the formulation of goals for ecological restoration. This is particularly important in the case of habitats and ecosystems that were not described or monitored before human disturbance. Although much restoration can be guided by conditions in
nearby, relatively undisturbed settings, there is an increasing appreciation for the value of historical data from both documentary evidence and natural biotic and environmental archives (i.e., the proxy data described in Chapter 2) (e.g., Brenner et al., 1993; Brush, 1997). Indeed, for long-lived species such as trees, the long temporal framework provided by tree ring analysis is essential for understanding such important phenomena as fire frequency and migration rates (Swetnam et al., 1999). This historical approach to restoration has now matured to the point where techniques for reconstructing reference ecosystems and their historic range of variation are well established for a variety of terrestrial and coastal ecosystems (see Egan and Howell, 2001).
The application of historical ecology to issues of ecosystem restoration and management has enormous potential for both basic and applied research. In basic research, the integration and cross-calibration of observational, documentary, and proxy ecological data provides both a near-continuous and long-term ecological record as well as the opportunity to develop confidence limits for proxy indicators farther back in the geologic record. In applied research, the approaches of historical ecology demand close collaboration among biologists, geologists, and archaeologists engaged in ecosystem management and restoration—practical issues that are an increasingly important focus of the U.S. Geological Survey.