3
Nanotechnology: Expanding Scientific Understanding
Today, science is at the optimal time to begin to study the impact of nanomaterials on human health, said Vicki Colvin of Rice University. Science is looking at the birth of a new industry, and beginning to address risk in a way that has not been done with any other developing technologies before (i.e. well before large-scale amounts of these materials are introduced into the environment or onto consumers). This provides public health with a unique opportunity to shape a new, emerging area with research on environmental health issues that we might encounter, while avoiding the problems that have plagued chemistry in the past.
Kenneth Olden of the National Institute of Environmental Health Sciences echoed this, and noted that very few studies have been done thus far on nanoparticles and their effect on human health and the environment. Science needs to start investing in research of these issues. Toxicity and bioavailability of engineered nanomaterials is largely unknown. He noted that it will be a daunting, costly, and time-consuming challenge to assess every single nanomaterial that is made individually, which means there will be a need to set priorities. NIH is trying to develop some effective, high output, more informative, and less costly systems. Two of its primary objectives are to maintain a comprehensive database and develop quantitative ways of measuring nanomaterials. Efforts such as this will help science develop standard materials for assessing environmental health impacts.
Unlike incidental particles, very little is known about engineered nanoparticles and how they interact with cells or human organisms. There are only about 30 papers written on the environmental and health impacts of these particles; however, there is a wealth of knowledge on incidental nanoparticles and how these particles interact with biological organisms, noted Colvin. Questions remain whether the engineered nanoparticles will act as a bulk solid or a molecular system (see Toxicology below).
SIZE ISN’T EVERYTHING
In most cases, nanoscale systems will alter in physical size upon interaction with an aqueous system. For example, it is very common for many nanostructures to adopt a different chemical form simply through relatively minor interactions; consequently, size is not a constant factor in biological interactions, noted Colvin. Furthermore, the surface area can make up a sizeable fraction of these materials, and they can be derived to make many different biomedical systems. By changing surface coatings the nanomaterial toxicity can almost be completely altered. For example, changing the surface features of the materials can change a hydro-phobic particle into a hydrophilic one. Hypothetically, surface coats could, for instance, make it possible to eat nanoscale mercury if it has the right surface coating, while it may be dangerous to eat nanoscale table salt if the surface coating was not correct. For this reason, the scientists’ typical view of toxicology, which is driven by the composition of an inorganic particle, may have to be modified for nanoscale materials, because the surface is going to affect different dimensions of environmental and health effects, according to Colvin.
INTERACTIONS WITH BIOLOGICAL SYSTEMS
Chemists and engineers interested in creating biocompatible nanostructures need to understand their interactions with biological systems. Colvin suggested that the challenge that nanomaterials pose to environmental health is that they are not one material. It is difficult to generalize about them because, similar to polymers, they represent a very broad class of systems. Many engineered nanomaterials have precisely controlled internal structures, which are structures of perfect solids. Over a third of the atoms in a nanoparticle are at the surface, and these are extremely reactive systems, which in some cases can generate oxygen radicals (see oxidative stress later in the chapter); however, nanoparticles can be tied up very tightly in covalent bonds and wrapped with a polymer. Because of the size of nanostructures, it is possible to manipulate the surface interface to allow for interactions with biological systems. Colvin noted that with the correct coating particles below 50 nm can translocate into cells relatively easily and are able to interact with channels, enzymes, and other cellular proteins. Those particles above 100 nm, based primarily on size of the particles, have more difficulty. Through the interactions with cellular machinery, there is potential for medical uses, such as drug delivery and cellular imaging.
ENVIRONMENTAL INFLUENCE OF TOXICOLOGY OF NANOMATERIALS
For applications such as drug delivery devices and therapeutics, there are well-established testing regimes utilizing whole animal and in vitro testing. How-
ever, for some of the environmental applications, scientists do not have those testing procedures in place, noted Eva Oberdörster, Southern Methodist University.
While coating or covalently modifying the outer surfaces of nanomaterials eliminates the toxicity of most particles, questions remain about whether under environmental conditions—as opposed to laboratory conditions, the nanomaterials will still be benign. In a recent study, it was demonstrated that if surface-modified C60 materials were irradiated to UVA (i.e., ultraviolet radiation of 320–400 nm in wavelength) for 11 min or UVB (i.e. ultraviolet radiation of 290–320 nm) for 22 min, cytotoxicity returns (Rancan et al., 2002). Additional research by Drefus et al. (2004) suggests that air exposure and nanoparticle dose are also important for cytotoxic effects. When cadmium selenide (CdSe) quantum dots in a liver culture model are exposed to air or ultraviolet light, hepatocyte viability decreases as assessed by mitochondrial activity of QD-treated cultures (Figure 3-1). In this study, exposure to ultraviolet light for only 4 hours significantly decreased hepatocyte viability even at very low doses of nanomaterial. So, while these nanomaterials may be safe under laboratory conditions, a more reliable or maybe environmentally relevant endpoint is to weather these compounds under environmental conditions, concluded Oberdörster.
While these nanomaterials may be safe under laboratory conditions, a more reliable or maybe environmentally relevant endpoint is to weather these compounds under environmental conditions.
—Eva Oberdörster
ROUTES OF ADMINISTRATION AND POTENTIAL HEALTH EFFECTS
Fullerenes and other nanomaterials can accumulate in the body, depending on the dosing route. For oral administration, 98 percent of fullerenes are eliminated within 48 hours via feces and urine (Yamago et al., 1995). The 2 percent that is not excreted is found throughout the rest of the body, noted Oberdörster. Intravenous dosing is rapidly transported to the liver (73–92 percent), the spleen (up to 2 percent), lung (up to 5 percent), kidney (up to 3 percent), heart (approximately 1 percent), and the brain (approximately 0.84 percent) within 3 hours. After 1 week, 90 percent of intravenously administered fullerenes are still in the body, noted Oberdörster.
Inhalation of nanoparticles may also be problematic because the particles are often small enough that alveolar macrophages cannot detect or scavenge the particles for elimination. By evading alveolar macrophages, nanoparticles can enter the lymphatic and circulatory systems to be distributed throughout the body within 24 hours, noted Oberdörster. This has been studied with a number of
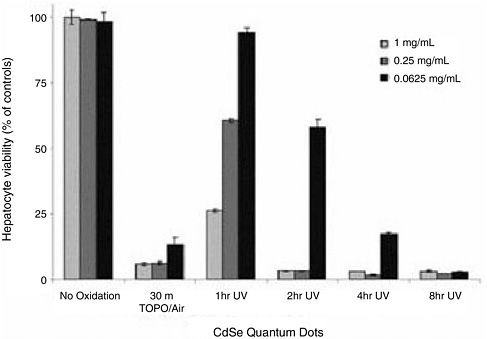
FIGURE 3-1 Toxicity of cadium selenide (CdSe) quantum dots in liver culture model is dependent on processing conditions and nanoparticle dose. Hepatocyte viability as assessed by mitochondrial activity of QD-treated cultures relative to untreated controls. Thirty minutes of exposure to air while TOPO-capped renders QDs highly toxic at all concentrations tested. Ultraviolet light exposure also induces toxicity that increases with exposure time and is QD-concentration dependent. Biochemical assays of viability were confirmed via phase contrast microscopy where control hepatocyte cultures exhibited distinct intercellular boundaries, well-defined nuclei, and polygonal morphology. SOURCE: Drefus et al., 2004. Reprinted with permission from Nano Letters 4:11–18. ©2004 American Chemistry Council.
particles, including titanium dioxide. It should be noted that not all nanomaterials evade phagocytosis, and in studies using nano- vs. bulk TiO2, ~20% of nano-sized TiO2 can be recovered by repeated lung lavages to obtain alveolar macrophages, as compared to ~80% of bulk-sized TiO2. Depending on the quantity of inhaled materials, it is also possible to overload macrophages. Inhaled particles can also be transported to the brain via the olfactory or trigeminal nerves. This process was first noted in studies from the 1940s, and the current hypothesis is that these nano-sized particles move in a similar mechanism as viruses. This is one mechanism to bypass the blood–brain barrier, Oberdörster suggested, and could be exploited for drug delivery.
OXIDATIVE STRESS
Nanoparticles, including C60, metal Qdots, and TiO2 can be redox active, which may lead to DNA cleavage, oxidative stress, and/or an inflammatory response. For example, C60 fullerenes, if exposed to light, can either make singlet oxygen or be electron donors to make super oxide radicals. The potential dilemma is that not only does the immune system use super oxide radicals to kill foreign toxicants; the super oxide radicals can cause hydroxyl radicals, which can lead to DNA cleavage. The good news is that the body has some ability to prevent the undesired DNA cleavage through super oxide dismutase, part of the antioxidant defense system, noted Oberdörster.
Oxidative stress is potentially a larger issue. A number of ambient or laboratory-produced ultra-fine particles cause oxidative stress, a mechanism that leads to cell damage or cell death. The severity of damage is dependent on the chemistry, as some particles result in more oxidative stress than others. In a recent study in her laboratory, Oberdörster looked at oxidative damage, brain translocation, and gene expression changes in large mouth bass exposed to fullerene C60.
Glutathione (GSH), a molecule involved with antioxidant activity, can be used as a marker of redox activity. The gill region, where fullerenes would probably be present, showed decreased levels of GSH, which was unchanged in the liver and brain. Protein oxidation, another marker of oxidative stress, was unchanged in the liver, brain, and gills. What was interesting, noted Oberdörster, was the increase in lipid peroxidation in the brain, an increase not seen in the gills or the liver. She hypothesized that lower lipid peroxidation in the gills and liver of fullerenes-exposed animals suggests that some tissues are more capable of responding to and repairing fullerene-induced cellular damage.
Complementing this data, she found that in the liver the expression of some inflammation-related genes was modulated. Genes associated with hormone regulation, immune cell response, and clotting and anti-clotting pathways were found to alter their expression after exposure to fullerenes. Expression of repair enzymes was increased, suggesting that the fish were starting to repair damage incurred by the response to fullerene molecules. As this was a preliminary study, the fish were euthanized for experimental study before further response to the fullerene molecules could be studied.
Genes associated with hormone regulation, immune cell response, and clotting and anti-clotting pathways were found to alter their expression after exposure to fullerenes.
—Eva Oberdörster
NANOTECHNOLOGY, INHALATION, AND CYTOTOXICITY
Particle size determines whether particles are respirable or inhalable. This difference can be critical, as inhaled particles can be trapped and cleared, whereas respirable particles are often more likely to be retained. In rats, particle sizes less than 3 µm are considered to be respirable. For humans less than 5–10 µm is respirable, while 10–50 µm is inhalable, noted David Warheit of the DuPont Company. These particles tend to be deposited at the junctions of the terminal bronchioles and alveolar ducts in rats and the level of respiratory bronchioles in humans. The alveolar regions of the lung, which are primarily used for exchange of oxygen and carbon dioxide comprise 95 percent of the lung surface area and are the pulmonary compartment where diseases such as asbestosis and silicosis are manifested.
Particle size and aggregation is an important consideration for deposition in the alveolar region. Particles such as fine-sized iron (i.e., 1 µm) are primarily deposited as discrete particles though they have some limited potential to aggregate. Conversely, although there is a potential for some types of particles to disaggregate, ultra-fine or nanoparticles generally tend to deposit as aggregates due to high Van Der Waals forces, rather than discrete particles. This means that if an inhaled particle with a diameter of 50–100 nm forms an aggregate of 5–10 particle types, in terms of deposition it may have the properties of a 200–500 nm particle, suggested Warheit.
Whether particles aggregate and then disaggregate once they reach the lung fluids and the process for generation of nanoparticles will become increasingly important.
—David Warheit
In addition to concern about deposition, particle clearance from the lungs may also be important for assessing nanoparticles. Normal clearance in the lung is performed by phagocytosis of particles by alveolar macrophages. These cells exit the lungs via the mucociliary escalator to be coughed up or swallowed. This process is effective for the fine-sized (100 nm to 3 µm), low toxicity particles if the clearance system is not overloaded; however, it is unknown if this happens with ultra-fine particles or nanoparticles.
Some current hypotheses suggest that nanoparticles are more toxic (inflammatory, tumorigenic) than fine-sized particles of identical composition, noted Warheit. This concept is based on a systematic evaluation of only three particle types: titanium dioxide, carbon black, and diesel particles. Thus, he noted that the current hypotheses are based on a paucity of data.
Determining toxicity can be complicated because not all nanoparticles are more toxic than fine-sized particles of the same chemical composition. The surface coatings of particles, exposure to UV radiation, and dispersion properties
can change the behavior of the particles. Whether particles aggregate and then disaggregate once they reach the lung fluids as well as the process for generation of nanoparticles—for example, gas phase versus liquid phase synthesis—will become increasingly important. He suggested that developing a working hypothesis for determination of particle toxicity will depend on the capacity of the particles to cause cell and lung injury, promote inflammation, inhibit macrophage function, and persist in the lung. Finally, Warheit observed that species’ differences complicate research because some species, such as rats are particularly sensitive to particles, sometimes making it difficult to extrapolate the results to humans.
Toxicity of Carbon Nanotubes
In a recent study, Warheit investigated the toxicity of intratracheally instilled carbon nanotubes, which are approximately 1 nm by 1–5 µm as a singular particle. However, due to strong electrostatic potential, they rarely exist as individual discrete particles and agglomerate into nanoropes.
Following instillation of the carbon nanotubes into the lung, the tissue was analyzed by looking at cell proliferation, histopathology, lung weights, etc. at 24 hours, 1 week, 1 month, and 3 months post instillation. Through this paradigm, the researchers would be able to determine the initial, transient reaction, but also ask whether the toxicity was sustained or progressive.
Fifteen percent of the animals died within the first 12 hours due to high agglomeration from electrostatic attraction, which essentially coated the airways of these animals. This was not because of the toxicity of the material, but rather because the material coated their airways. Thus, these animals died from suffocation because of the unique properties of carbon nanotubes, said Warheit.
The animals that survived the first 24 hours post instillation survived through the 3 months. Exposure to carbon nanotubes produced only a transient inflammatory response at 24 hours, but this was acute, with no inflammatory effects seen at 3 months. Interestingly, Warheit reported that they did see multifocal granulomatous lesions in the lung tissue studies. These lesions are not commonly seen in dust-exposed rats. In the center of the lesions were the agglomerated carbon nanoropes walled off by foamy multi-nucleated macrophages cells. The distributions of the lesions were not consistent, nor were they dose dependent or progressive from 1 to 3 months.
Warheit put these results into the context of possible exposure in the workplace. Since carbon nanotubes are used in the electronics field for diode, transistors, cellular-phone signal amplifier, and ion storage for batteries, his DuPont colleagues, as well as NIOSH
Particles need to be thought of as having inherent toxicity, and being carriers for organic molecules and metals.
—John Froines
researchers, performed exposure assessments in the workplace. The results suggested that the dust was less than 53 µg/m3, which was extremely low. Most of the nanotubes were aggregated into nanoropes, which may not be respirable. Warheit concluded by stating that scientists cannot assume that all nanomaterials are the same as their bulk counterparts, which suggests that materials will need to be tested on a case-by-case basis, a process that may be infeasible because of resource constraint. He suggested that priorities for studying particles based on surface coating, surface charge, and particle aggregation will need to be made.
WHAT CAN WE LEARN FROM DIESEL PARTICLES?
John Froines of UCLA raised the question, does the research that scientists are doing on airborne particulate matter related health effects have relevance to understanding potential issues with respect to nanotechnology? He suggested that there are areas where there are significant differences, but there are also places with commonalities. For example, most of the ultra-fine particles from diesel emissions are in the 10 nm range, classifying them as nanoparticles. He suggested that a number of biochemical processes may be similar between the air pollution particles and the engineered particles.
Redox Cycling
Nanoparticles from diesel exhaust may contain pro-oxidative chemicals. Quinones, one such chemical, can undergo redox cycling to semiquinones, subsequently generating reactive oxygen species (ROS) such as superoxide radical anions. This cycling can lead to a build-up of ROS that result in oxidative stress. Oxidative stress then produces pro-inflammatory effects, such as allergic airway disease, adjuvant effects in asthma, and propagation of cardiovascular disease.
Airborne particulate matter has a coarse, a fine, and an ultra-fine or nanoparticle mode. These particle modes differ in the carbon, organic carbon, metals, inorganics and polycyclic aromatic hydrocarbon (PAH) content. Ultra-fine particles have a greater organic carbon and PAH content then the coarse or fine particles. More interestingly, noted Froines, is that the redox activity is greater in the ultra-fine particles, which leads to an increase in glutathione depletion and mitocondrial damage in cells exposed to these particles.
The redox activity in nanoparticles was confirmed in a recent study by Li et al. (2003). Redox activity was examined in the coarse, fine, and ultra-fine particles at 50 or 150 meters from a freeway. At both distances, the redox activity per microgram was greatest in the nanoparticle region. At a distance of fifty miles from downtown Los Angeles, Froines collected particles with the same size distribution, and once again, the ultra-fine particles had the greatest redox activity.
Persistent Redox Activity
Diesel particles need to be thought of as having inherent toxicity, because of their being carriers for organic molecules and metals, noted Froines.
Organic extraction of the particulate matter only removes 10–30 percent of the redox activity from the particles. The remaining redox activity cannot be extracted from the particles, noted Froines. Mucociliary fluid is even less effective in extracting capability. Thus, the active chemical species remain on the particles themselves, which means that the particles retain their toxicity.
Particles need to be thought of as having inherent toxicity, because of their being carriers for organic molecules and metals.
—John Froines
Health Endpoints
There have been a number of health endpoints associated with ultra-fine particles. Animals exposed to ultra-fine particles from freeways show enhanced allergic airway responses and CNS inflammation. In human clinical studies with particles from freeways, particles have also been associated with a statistically significant decrease in heart rate variability and other cardiovascular parameters in normal subjects. Further, exposure to mobile sources has been associated with increased risk to preterm birth and low birth weight. Froines concluded by suggesting that as nanotechnology continues to evolve there is accumulating evidence in the area of air pollution from ultra-fine particle work that there are health endpoints that need further research.
NANOTECHNOLOGY AND STRATEGIES TO ENSURE OCCUPATIONAL HEALTH
The people currently most affected by nanomaterials, nanoparticles, and their potential impact on the environment and health are the groups that generate and handle the materials, that is the people in the workforce and the laboratories, noted Andrew Maynard, National Institute for Occupational Safety and Health (NIOSH). NIOSH has established a very active research agenda to reduce the potential health impact in these groups. Yet as they begin to look at the research agenda, he noted that there are a number of pressing issues.
Since the 1950s, industrial hygienists have mostly concentrated on inhalation exposure related to mass of the material, because this has been the most useful information for relating exposure to health effects. The use of mass as a paradigm marginalizes nanoparticles and nanostructured materials, because even if there are many of these particles there is very little mass associated with them. A 1 nanometer particle has only 1 billionth of the mass of a 1 micron-sized
Under the mass paradigm nanometersized particles are not looked upon as dangerous; however, we cannot ignore the chemistry of nanoscale materials that have other unique and unusual structures and properties which may cause health problems.
–Andrew Maynard
particle. Therefore, under the mass paradigm nanometer-sized particles are not looked upon as dangerous, noted Maynard. However, scientists cannot ignore the chemistry of nanoscale materials that have other unique and unusual structures and properties, which may cause health problems. As noted above, most nanomaterials are insoluble and the surfaces of these materials are very different from the core properties. Nanomaterials have very specific heterogeneous structures, and they are exclusively engineered to specification.
Mass, particle size, and surface structure are all important properties for determining where these particles deposit in the respiratory system upon inhalation and how the body will react, as discussed previously. This is where occupational health faces its first challenge. Health scientists need to understand how these materials may impact health in the workplace or the laboratory, and they may need to undergo a philosophical change in the way these materials are viewed, said Maynard.
NIOSH is concerned with identifying the risks from nanoparticle exposure and considering how to control the risks, whether through reducing risk or reducing the impact. However, to be in a position to assess risk, additional information is needed regarding toxicity of the materials including: how they interact biologically in the body and what the health effects are resulting from toxicity, environmental exposure, and potential exposure routes (i.e., whether the material is inhaled, ingested or absorbed cutaneously), noted Maynard.
He noted that the toxicity of nanomaterials needs to be understood in a framework of the materials’ characterization. If scientists do not understand the materials from a physical and chemical perspective, they cannot interpret exposure or toxicity measurements. Scientists already know how some of the small structure materials behave, but they have a long way to go before understanding the risks posed by these materials, said Maynard.
There are a number of steps that need to be taken in order to understand the occupational health impacts of the nanomaterials, according to Maynard. These include the need to:
-
Understand the problem from different perspectives (e.g., industry, public, workers, and regulatory).
-
Perform gap analyses to determine a course of action.
-
Create a strategic plan for achieving short-, medium-, and long-term goals.
In order to achieve these goals, there is a need to coordinate activities between stakeholders (i.e., the people who are directly impacted by the processes), the materials, and the service providers (i.e., the people who have the knowledge that allows us to take action to impact health effects at the workplace). He concluded by suggesting that nanotechnology is a multidisciplinary area that covers a large range of disciplines and that a multidisciplinary approach to the problem is necessary in order to be effective.
Unless we have a multidisciplinary approach to the problem we are not going to make any significant impact.
—Andrew Maynard
AN OPEN PROCESS
There is a need for increased levels of cooperation between the industries involved, public interest groups, and government parties to find solutions that are economically viable, but still protect the environment and health, asserted John Balbus, Environmental Defense. This is not a small goal, because it is important that nanotechnology development is done right the first time. Modern history has produced a number of technological advances that held such great promise for revolutionizing society; that they were accompanied by safety short-cuts. Not all technological advances proved to be health concerns, but DDT, polychlorinated biphenyls, tetra-ethyl lead, and chlorofluorocarbons are just a few examples where wide-scale release had a number of unintended side-effects.
Potential Problems
These past problems have implications for nanotechnology. The federal government, through various agencies in the National Nanotechnology Initiative, has increased nanotechnology research funding in the last 5 years from $200 million to $850 million. During the same time period, the investment in environmental health and safety implications research funding had only a slight increase from 0.2 percent to less than 1 percent of total investment in nanotechnology. Balbus noted that the public expects the federal government to anticipate and to be proactive about the risks of any future commercial products and he questioned whether the funding levels were adequate for environment and health research.
Balbus noted there are a number of reasons why the public and public interests groups have reason for concern about nanotechnology. Currently, there is a lack of knowledge about these products that will need to be addressed before scientists can adequately address environmental health concerns. Nanomaterials are:
-
Deliberately manufactured so that they have novel physical-chemical properties, and many times unpredicted physical-chemical properties.
-
Heterogeneous, and this is not only in terms of the different kinds of nanomaterials that are deliberately engineered, but it is very likely that different nanomaterials will be heterogeneous in terms of fate, transport, and degradation. Should these materials be distributed in the environment, they will be in different stages of transformation.
-
Poorly soluble in water, at least for the carbon-based nanomaterials. These nanoparticles have to have surface coatings or surface modifications in order to make them biocompatible.
These properties have a number of implications for environmental health. Nanomaterials are the size of viruses, and as such are able to be transported through the body along nerves and through the lymphatic system, providing an opportunity for these materials to cross the blood–brain barrier, which may result in a number of unintended consequences. Poor water solubility may lead to similar problems—seen with other poorly water soluble chemicals such as PCBs—with increased potential for bioaccumulation through repeated cycle absorption, evaporation, and transpiration. These are only two examples, but Balbus questioned whether science is in a position to answer to these questions, since there is a paucity of data presently. The current observations are not “doom and gloom,” but they haven’t been reassuring from a health and safety standpoint, he concluded.
The current observations are not “doom and gloom,” but they haven’t been reassuring from a health and safety standpoint.
—John Balbus
Regulatory Framework
The regulatory framework for bringing these products to market also needs to be discussed. The statute that covers new chemicals, other than pesticides, cosmetics, and pharmaceuticals, including nanotechnology, is the Toxic Substances Control Act (TSCA), but Balbus questioned whether TSCA could meet the challenges for nanotechnology. For example, pre-manufacture notices require the company to provide only information on the process, likely uses, and physical-chemical properties of the substance, but not to generate new data on the toxicity of the substance. Furthermore, he noted that there is no requirement for post-marketing surveillance, which means that once the compounds are in commerce, there is no requirement to assess whether they are causing any kind of damage. The EPA does have the capacity to generate a test rule, and insist on
toxicity data, but the burden for findings is high for the agency and these provisions only very rarely have been invoked.
Balbus noted that one model that has been put forward is the European Commission’s decision algorithm for bringing nanomaterials to market. This decision algorithm considers solubility, ability to be transported within the body, and probability of ecotoxicity. The products are assigned low, intermediate, and high priority (see Figure 3-2), and level of priority is then used to drive the research agenda. It remains to be seen if the actual implementation and enforcement of the algorithm will be reached, concluded Balbus.
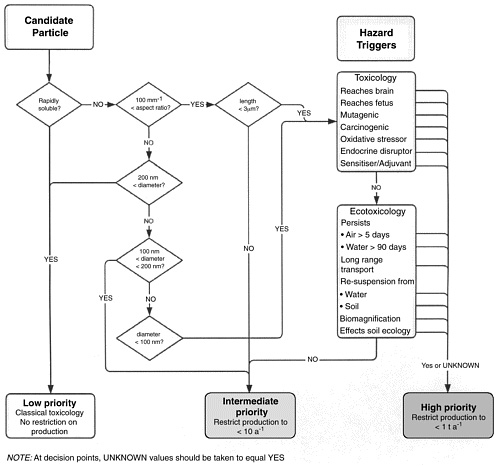
FIGURE 3-2 Flow chart utilized by the European Commission on Community Health and Consumer Protection to assess solubility, ability to be transported within the body, ecotoxic probability, and other hazard probability and priorities and is assigned low, intermediate, and high priority. SOURCE: European Commission, Community Health and Consumer Protection, 2004. Reprinted with permission.
Moving Forward
Balbus noted that it is not a question of whether we develop nanotechnology, or we move forward. Rather, it is a question of how does the science move forward in a way that best protects the public, and gets the health and safety right the first time. While assuring health and safety, scientists need to be reminded that lack of toxicity evidence is not the same as lack of toxicity. We also need to recognize that industry moves faster than government. Scientists know that it is going to take several years to get the nomenclature in place. This will complicate the application of TSCA1 for these compounds. The government will need to develop strategies that harness some of the nimbleness and speed of industry.
One way to accomplish this is to create a road map that draws from the strengths of government, academia, and non-governmental organizations. There are at least four steps to accomplish this goal, noted Balbus. First, science needs to define the categories based on potential for environmental health effects. Second, there is a need to develop a testing strategy to answer the basic questions. A component of this may require developing new techniques or methods. Third, industry and government need to commit to pre-manufacture testing that examines ecological and human health. There should be some consensus about what testing needs to be done and when. Fourth, industry should consider voluntary use restrictions in the absence of data, especially for those uses that are likely to be highly dispersive.
Risk Communication for Emerging Technologies
During the course of the day’s discussion, roundtable members, speakers and participants centered their discussions on public trust and risk communication. Continuing on the examples discussed by John Balbus early in his presentation, participants also raised the examples of the times when nuclear energy was introduced in the United States and there was a lot of positive publicity about the peaceful atom which later on produced a large backlash when the public realized that there were also hazards in the new technology. Another example was when biotechnology was advertised as being absolutely safe and, again, later the public realized about the negative sides of biotechnology. Nanotechnology also holds promises but on the other hand there is a lot of uncertainty from the scientific and technology development communities about the downsides of the new technology being communicated to the public. Vicki Colvin said that the scientific
community maintains communication with the public about both positive and negative sides of the new technology but she argued that it is better to articulate the positive benefits of nanotechnology. However, she agreed that in order to build public trust, the scientific community needs to assess and determine the risks of nanotechnology as well.
Finally, trust takes more than just good risk communication skills. The best way to get the public to perceive things correctly is by right action, and by a clear demonstration that everything in the power of government and industry is being done to insure protection of consumers and the environment, concluded Balbus.















