3
Policy Precedents in Decontamination
The subject matter of this report, determining when a facility is safe for use, is not a new policy-making dilemma. The prospect of remediating a biologically contaminated public facility, however, requires that policy makers consider both the biological nature of the hazard and the public nature of the building. The public nature of the building adds a social dimension to policy and decision making because the public’s perception of the event and its aftermath must be taken into account. Another dimension is national security. The range of policy precedents in decontamination reviewed here provides lessons relevant to each dimension:
-
Routine microbial decontamination of water and food supplies
-
Biosafety practices in laboratories and environmental infection control in hospitals
-
Decontamination procedures and safety programs for biological weapons research and testing facilities
-
Cleanup of hazardous-waste sites
-
Remediation of radioactive waste within the nuclear weapons complex
Although not related to biological hazards, the remediation of waste sites under the Superfund program and of radioactive waste within the nuclear weapons complex are instructive in the special considerations that arise in the context of affected individuals who face involuntary exposures to unfamiliar hazards. The legacy of secrecy within the nuclear weapons complex (Fehner and Gosling, 1996; O’Leary, 1997)—as it relates to public confidence in government declara-
tions regarding safety—is instructive in the need for transparency in public health matters that arise against a national security backdrop.
MICROBIAL DECONTAMINATION IN FOOD AND WATER SUPPLIES
Foodborne and waterborne infections are commonplace examples of the need to reduce pathogen occurrence in environments that have a direct effect on human health. Microbial risk assessment, addressed in greater detail in Chapter 4, is a useful tool for setting standards in food and water safety, making it appropriate for use in the current application.
The first direct use of microbial-risk-based criteria for regulating exposure to pathogens in the United States was in the U.S. Environmental Protection Agency ( EPA) water quality criteria for recreational waters (EPA, 1986a). That document listed water quality limits that were set so that a risk (from recreational contact such as swimming) of 0.008 illnesses/bather-day (for fresh water) to 0.019 illnesses/bather-day (for marine water) per exposure resulted; experts deemed that an acceptable risk to the public (EPA, 1986a).
Shortly thereafter, the Surface Water Treatment Rule mandated that all water purveyors that used surface water introduce treatment sufficient to provide adequate control of pathogens. In the rule’s development, treatment was to achieve a residual risk of infection of less than 1/10,000 per year based on microbial risk assessment, even if there was a high concentration of pathogens in raw water (Macler and Regli, 1993).
In food safety, canning, the first large-scale method of industrial food preservation, relies on heat to minimize risk from pathogens and to reduce spoilage. A criterion that is sufficient to provide 12 logs of inactivation (that is, only one out of 1 × 1012 spores would remain active) of Clostridium botulinum spore—referred to as a “botulinum cook”—is commonly applied even though the criterion does not necessarily result in a sterile product (Farkas, 1997). More recent concerns on food safety—particularly for less thoroughly processed foods—have led to increasing attention to providing a scientific basis for food safety criteria.
A committee of the Institute of Medicine (IOM, 2003) has examined food safety. Among its conclusions are two that are germane to the current report:
There is a need to define “acceptable levels” of hazard reduction at critical points linked to public health objectives. The Food Safety Objective concept can help establish this link and define these levels, and it can also provide a theoretical framework to relate performance standards to public health objectives.
Quantitative microbial risk assessment offers the scientific tools to define the most effective solutions for lowering consumer exposure to foodborne microbial hazards.
A food safety objective is a quality measure to control risk at a particular surveillance point in the food-processing chain. Specifically, it is “the maximum frequency and/or concentration of a (microbiological) hazard in a food at the time of consumption that provides the appropriate level of protection” (International Commission on Microbiological Specifications for Foods, 2002). The objective can be derived from a dose–response relationship to the pathogen in question (Havelaar et al., 2004). However, a risk management decision on acceptable risk must still be made.
BIOSAFETY IN MICROBIOLOGICAL AND BIOMEDICAL LABORATORIES
Humans and microorganisms cohabit the world, and their interactions lead to disease occasionally. Society takes steps to reduce the chances of disease among individuals and population (for example, by sterilizing food and water supplies to kill harmful microorganisms). Some people, however, do have extensive intentional contact with infectious agents during work or study in microbiology and biomedical laboratories. Biological safety policies and procedures have evolved to ensure that laboratory researchers and technicians are protected against infectious disease and prevented from unintentionally releasing pathogens into the environment or the community. Two facts are relevant. First, safety is a top priority for people who routinely face the possibility of exposure to pathogens. Second, laboratory decontamination protocols are essential parts of a comprehensive biological safety program.
Safety procedures used in performing necessary tasks, along with laboratories’ physical containment features, provide the framework in which people can work in biologically contaminated environments. The development and implementation of laboratory safety guidelines form the foundation for biological safety in any laboratory that uses microbiological organisms that can cause infectious diseases (DHHS, 1999). Basic elements of biosafety include the following:
-
Staff have an in-depth understanding of the microorganisms with which they work.
-
Staff are well versed in safety and procedures, including the correct use of personal protective equipment.
-
Continuing education is provided in new technologies, including new equipment or procedures that promote laboratory safety.
-
Architecture and equipment designs are chosen for the physical containment of pathogens.
-
Procedures are established for the decontamination of work surfaces.
Microbiological and biomedical laboratory operations demonstrate that under carefully controlled situations, humans can come into extensive contact with
pathogens without harm. Protective features, however, are eminent in a laboratory’s physical structure, equipment, personal protective gear, personnel training, and accepted professional practice. There also are prescribed procedures in the event of a spill or other accident. One cannot generalize from this special environment to public buildings whose occupants would not necessarily have extensive knowledge of microbiology or biosafety procedures, and who have not consented as such, to exposure to dangerous microorganisms.
ENVIRONMENTAL INFECTION CONTROL IN HEALTH CARE FACILITIES
Hospitals and other health care facilities implement administrative and engineering procedures to eliminate or control infectious agents that cause disease (Sehulster and Chinn, 2003). Health care settings are characterized both by a higher likelihood of the introduction or presence of infectious agents and by their institutional duty to protect patients, particularly those whose immune systems are compromised and vulnerable to infection. The health of workers and patients can be at stake whether the threat is an environmental pathogen such as Legionella spp. or an airborne pathogen like Mycobacterium tuberculosis. Health care facilities have developed infection control and epidemiological functions to protect a broad range of building occupants—from health care professionals, to nonmedically trained workers, to patients and visitors.
Most relevant to this report is the fact that health care facilities, to a greater or lesser degree, retain a cadre of professionals, many of whom work on site, who are trained specifically to address issues of microbial contamination and infectious disease. Those facilities often employ infection control practitioners, infectious-disease experts, epidemiologists, employee health and safety personnel, and facility engineers. There are also third-party oversight mechanisms, such as the Joint Commission on Accreditation of Healthcare Organizations, that evaluate the infection control performance of health care facilities. In contrast, public buildings, such as airports, are not routinely staffed by people who would be familiar with infectious agents and disease transmission. Nor are the owners or operators of public facilities necessarily familiar with microbiological, epidemiological, or larger public health principles.
DECONTAMINATION OF U.S. ARMY BIOLOGICAL WARFARE LABORATORIES
The U.S. Army Biological Laboratories at Fort Detrick, Maryland, were in operation from March 1943 until July 1972. Their mission was to conduct offensive and defensive research with highly pathogenic agents and/or their toxins, with the ultimate goal of protecting the United States. At the start of the program, safety procedures, vaccines, antibiotics, medical treatment regimens, and con-
tainment facilities were limited, and there were many unknown operational elements and unrecognized risks to military and civilian employees. Because of the high-risk nature of the research and the sizeable population facing possible exposure (1500-1700 personnel), a comprehensive safety program was put in place and continuously improved. That program—which included decontamination policies and protocols—was successful in terms of technical rigor, health and safety outcomes, and employee confidence, according to records available to this committee.
Several factors contributed to the effectiveness and credibility of the program, beginning with the fundamental priority given to health and safety: Any research activity that would compromise employee safety, cause damage to facilities, allow release of agents into the environment, or permit cross-contamination among research materials or laboratory animals could not be initiated—any of those incidents halted the research. Employee safety was not compromised for program expedience or financial savings.
A dedicated, well-educated, large scientific safety staff (up to 30 people) was appointed at the start of the program. The staff included well-trained laboratory technicians and Ph.D. scientists. A physician served as the safety director. Safety responsibilities enveloped examination of every task and the conduct of research studies to evaluate hazards associated with laboratory operations, production, equipment, and facilities design. Procedures and decisions were transparent to those who might have been affected. Also, safety staff members were available to all employees, outside the chains-of-command structure, to address concerns. That open-door policy provided a forum in which to evaluate employee concerns, identify deficiencies regardless of magnitude, answer inquiries, render assistance, and provide daily safety awareness.
Senior management instituted the operating principle that no punitive reprisal, punishment, or fault finding was to occur in an aftermath of an accident, error in judgments, or equipment or facility damage, thus allowing the laboratories to learn from every experience to prevent reoccurrence. The policy was approved by the Military and Civil Service Commission as an exception for Fort Detrick, and it mandated reporting of incidents to the safety staff for evaluation.
A biological safety research program assessed all operational aspects, equipment, and facilities development, and it provided investigative mechanisms for each laboratory or production procedure. The program eventually evolved into the scientific discipline now known as biological safety or biosafety. It identified procedures to ensure safety in every component of work with pathogenic agents, including tasks that involved toxins, genetic manipulation, and production of agents and vaccines. An extensive laboratory safety training program was maintained for the lifetime of the program.
Before agent research could be initiated, a screening evaluation was conducted. The evaluation involved library research to glean knowledge and assess risk; identify a disinfectant of choice if possible; assess biological decay param-
eters, sterilization conditions, heat stability, vaccine availability, antibiotic sensitivity, or resistance; and delineate a medical treatment regimen. The evaluation usually was performed by a principal investigator, and often it involved members of the safety staff before an agent could be used or placed in the program.
Apart from administrative and engineering controls to protect against exposure, from inception, the Biological Warfare Laboratories established a comprehensive medical surveillance and treatment program that encompassed prophylactic vaccination; complete medical surveillance for any illness, either suspect or frank; and complete treatment for known or suspected illness. Before any employee could seek medical assistance from a private physician, he or she had to obtain clearance from post physicians. Employees had amounted to free medical care because of the responsibility of the facility physician to rule out all possibility of laboratory-acquired illness. The facility maintained a full medical staff, an outpatient clinic, and a complete isolation-quarantine hospital.
The Fort Detrick case is instructive on several matters. The health and safety of occupants were core objectives in that environment, where weaponized pathogens were produced and studied. A comprehensive set of safeguards was in place both to prevent exposure in the first place and to monitor for any untoward effects should exposure occur by accident. Medical care for occupationally acquired infection was a given. Employees trusted the decisions and interventions of a dedicated and trained safety staff, against the backdrop of a larger policy that no research activity should proceed that might endanger staff.
There are limits to generalizing from this case to the matter at hand. Those facilities were used exclusively for biodefense purposes by a typically healthy, robust, vaccinated population and employees were under the control of a central authority, the U.S. Army. One implication of that important fact is that it is probable that there was more cultural homogeneity at Fort Detrick than would be the case in a major American airport. People at Fort Detrick generally would be expected to share assumptions about what counted as relevant expertise and who had the ultimate authority to make decisions regarding acceptable risk. In the case of an American airport, there would be more diversity among people involved, thus there would be more points of view and more potential for social conflict.
DEVELOPMENT OF SUPERFUND AND REMEDIATION PLANS
The policy question “How clean is clean enough?” arose early in the development of the Superfund program. The Comprehensive Environmental Response, Compensation and Liability Act (CERCLA, 43 USC 103) was enacted in 1980 in response to growing public and government concern over several highly visible and dangerous contaminated sites, most notably in the community of Love Canal, New York. CERCLA provided a mechanism for site identification and, ultimately, financing for response and remediation through mechanisms for assignment of liability and the establishment of trust funds. At some Superfund sites, it
was necessary to decontaminate the interior of buildings. Those cases are directly analogous to the problem addressed in this report, and the remedy chosen is (at least in part) decontamination of the building and interior components to permit at least partial reoccupancy. One example was the cleanup of the Grand Street Mercury site in Hoboken, New Jersey (NJ0001327733). In that case, mercury contamination of a residential structure was caused by that building’s prior use as a factory for manufacture of mercury vapor lamps. Cleanup criteria (including allowable concentrations of mercury in ambient air) were set for the remediation so that rehabitation could be permitted. CERCLA also provided guidance on the extent to which remediation was to be conducted. A study by the General Accounting Office (GAO) noted, however, that the original National Contingency Plan consistently left cleanup targets vague (GAO, 1985). The plan required only that the selected action “be cost-effective and mitigate and minimize damage to provide adequate protection of public health and welfare and the environment.”
Great heterogeneity thus emerged among the design goals of the initial remediation projects, fueling local and national controversy over the quality of cleanup operations. How could the public and other interested parties be sure that remediation was effective when there was no consensus about the endpoint? GAO identified four principal options for resolving the controversy (GAO, 1985):
-
Restore sites to their “original” condition by completely removing all contaminants.
-
Set uniform national standards on a contaminant-by-contaminant basis.
-
Require application of “best available technology” to the cleanup.
-
Deal with immediate and significant problems, but defer further action until more detailed criteria were developed.
The 1986 reauthorization (Superfund Amendments and Reauthorization Act [SARA]) was designed, in part, to help address the gap in safety and performance standards (EPA, 1986b). The key points of SARA (EPA, 1986b) that are relevant to this report are as follows:
-
SARA gives weight to the use of permanent remedies and innovative treatment technologies.
-
It requires Superfund actions to account for other state and federal standards and regulations in setting cleanup goals.
-
It increases state and local involvement in investigation and site selection.
The passage of SARA caused EPA to review other environmental programs and develop cleanup guidelines concordant with goals for public health established in other programs (Applicable or Relevant and Appropriate Requirements), to weigh the qualitative attributes of the potential remedies in the decision-making process (permanence and innovation), and to involve stakeholders in
|
BOX 3-1 “Risk assessment and site cleanup will usually have to proceed on the basis of very limited knowledge for determining the precise level of cleanup necessary. There is simply not enough technical and health-related information available to know precisely what the level of cleanup at any specific site should ultimately be. The selection of appropriate cleanup technologies and the ultimate evaluation of cleanup performance remain somewhat of an art rather than a science. Restoring sites to pristine or background levels or requiring the use of best available technology is probably not practical or economical based on a rational cost, benefit analysis.” “An ideal remedial cleanup should provide complete and total protection of human health and the environment from the remediated site contamination. However, complete protection is neither technically feasible nor affordable. There will always be some level of risk remaining at a remediated site.” (Wentz, 1989). |
decision making. However, even after SARA, the dominance of qualitative and subjective aspects of decision making remained (Box 3-1).
The practice of applying technically based standards within the selection of remedies, for the most part, remains unchanged since SARA, and EPA does not generally clean up to below natural background concentrations. However, where the anthropogenic background concentrations exceed acceptable risk-based concentrations—and where EPA has determined that a response action is appropriate—the agency’s goal is to develop a comprehensive response to area wide contamination (EPA, 1997). There has been increasing emphasis in recent years on stakeholder involvement in determining the fate of Superfund sites. Inclusion of interested and affected parties is meant to address two aspects of hazardous-waste-site management. The first acknowledges that cleanup is, ultimately, a values-driven endeavor. EPA has recognized and the National Research Council’s recommendations have affirmed (NRC, 1983, 1996) that when public policy incorporates public health and environmental aims, decision making necessarily involves factors beyond the technical aspects of risk assessment, that is, a technically validated definition of potential danger (Figure 3-1).
Many discussions on the possibility of restoring a given site to its “original” condition by complete removal of all contaminants have included assessments of technical feasibility and cost effectiveness. There is not necessarily agreement, from a public policy standpoint, about what constitutes acceptable cleanup. In-
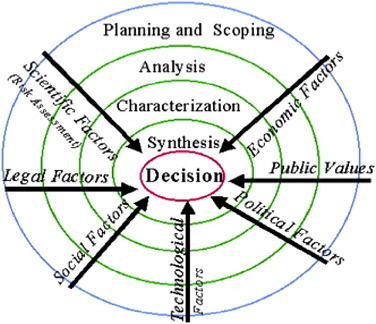
FIGURE 3-1 EPA risk management decision framework. Source: EPA, 2000.
volving the full range of interested and affected parties is seen as vital to reaching consensus. A second dimension for inclusive decision making is the realization that the people and groups that face the most immediate or direct health consequences from exposure to a hazardous waste site have the highest stakes in the cleanup of a Superfund site. Their confidence in whether or not remediation decisions adequately protect their health could not be ensured unless they participated in deliberations about what constituted reasonable action by responsible parties in the absence of perfect solutions (NRC, 1996).
REMEDIATION EXPERIENCES IN THE U.S. NUCLEAR WEAPONS COMPLEX
The U.S. Department of Energy (DOE) manages more than 100 sites contaminated with radiological and chemical by-products that have been amassed during a half-century of nuclear weapons production (Burger et al., 2003). The largest of those sites (such as Oak Ridge, Tennessee; Savannah River, South Carolina; Hanford, Washington) have hundreds of individual waste sites within their boundaries that require remediation. Cleanup challenges facing DOE include the sheer magnitude of remediation projects—DOE cleanup constitutes 20% of the world’s environmental remediation market. But there also are the compounded hazards and technological difficulties posed by long-lived radioac-
tive contaminants that are mixed with toxic chemicals and, most important for this study, the lack of public and regulator trust in DOE and its site operators.
From its inception, the management of the nuclear weapons complex was characterized by secrecy and self-regulation (Fehner and Gosling, 1996). The wartime Manhattan Engineer District, under the U.S. Army Corps of Engineers, carried out projects without any appraisal by local populations or public regulators. Reorganized after the World War II into the civilian-controlled Atomic Energy Commission (AEC), the weapons complex continued to be exempt from external licensing and regulation. AEC—a predecessor to DOE—held the authority to establish its own standards, oversee contractor health and safety, and manage the distribution of nuclear materials. The commission’s work was cast in terms of national security, and AEC managers and operators saw the manufacture and development of nuclear materials as their primary mission; waste and environmental concerns were of less immediate consequence or importance.
DOE’s claim to self-regulation was challenged in the 1980s after public disclosures about toxic releases, in reaction to the work of environmental activist groups, and as a result of landmark legal decisions that opened the agency up to EPA and state regulation. The question of who exactly should perform risk assessments for hazardous-waste sites within the weapons complex generated significant controversy (Henry et al., 1997; NRC, 1994). The public generally has been unwilling to accept DOE’s official assurances about the health effects of exposure to nuclear-related activities because of a core conflict of interest for the agency and its predecessors. The government is responsible for the production and testing of nuclear weapons, and it has been the nearly exclusive source of funding for U.S. radiation research in general and for radioactive fallout monitoring, dose reconstructions, and epidemiological investigations, in particular (Hoffman et al., 2002).
Revelations about deliberate deception about environmental and worker exposures also greatly undermined public confidence in DOE pronouncements about health matters (Ledwidge et al., 2004; Thomas, 2001). Scientists who failed to acknowledge publicly the limits of their knowledge regarding radiation health effects also engendered mistrust. Public skepticism about DOE as a source of information on radiation exposure and health risks has, in some cases, necessitated independent reevaluations of cleanup standards (Till and Meyer, 2001). Requests for reevaluations, although reasonable, incur additional costs as do the resulting delays in cleanup operations.
In reviewing the major lessons from the U.S. experience on long-term management of areas contaminated with radioactive materials, one top DOE official characterized the “absolute key to success” as “the establishment of open, honest and inclusive communications and decision-making” with affected populations (Jones, 2004). The corollaries to this are the need to create opportunities to reach mutual agreement on expectations for cleanup endpoints and measures of success before taking action, and to provide the necessary financial and technical support
so that the involved parties are confident that technical assessments are uncompromised; that is, that the assessments are based on science and free from conflicts of interests (Burger, 2002; Jones, 2004). Stakeholder involvement provides valuable returns in the form of local knowledge that can lead to better assessments of risk, greater public confidence in the science-based tools that support decision making, and the cultivation of participants who can champion the result within the larger affected community (Till and Meyer, 2001).
Review of policy precedents for a range of decontamination experiences—radiological, chemical, and biological—presents guiding principles that are applicable to the remediation of biologically contaminated public facilities. It is important that parties whose personal health and property are affected have adequate representation in decision making about decontamination and reopening of a facility. Restoration decisions that solicit neither consent nor input from affected parties are likely to be questioned or dismissed altogether. The history of radioactive-waste remediation within the nuclear weapons complex underscores the importance of open and inclusive decision making: Government secrecy, evoked in the name of national security, greatly diminished public confidence in official declarations about environmental health and safety. Finally, biological safety policies and practices to protect people in hospitals and laboratories—places that present a greater chance of exposure to pathogens—incorporate decontamination and sterilization as part of comprehensive safety systems that include medical surveillance to identify inadvertent exposures and resulting illnesses. Postevent medical monitoring is a wise practice in the context of a biologically contaminated public building.
FINDINGS AND RECOMMENDATIONS
Finding 3-1
Determining acceptable risk is a complex issue: Willingness to accept risk varies from person to person, from situation to situation, and from culture to culture. Managing risk also is complex: Different people have different ideas about how much responsibility the government or the owners and operators of public facilities and lands have to limit public exposure to risk. Those issues have been considered in many situations, and many policy-making lessons can be learned from events involving Superfund and the U.S. Department of Energy.
Recommendation 3-1
In contemplating how to respond to potential biological attacks, authorities should base their plans on lessons from the experiences of others who have dealt with decontamination issues in the broadest sense; they should not consider their charge a completely novel task. Decision making about a facility contaminated as the result of a biological attack should be mindful of the critical policy dimen-
sions of the biological quality of the hazard, the public nature of the building, the public’s perception of an attack, and the event’s national security implications.
Finding 3-2
If safety-related standards and protocols are devised and implemented behind closed doors, without the consent or input of affected and interested parties, those standards are likely to be questioned or rejected outright. Lack of transparency for policy decisions that directly affect public health—even in the context of a proclaimed national security interest—can severely erode public confidence. The establishment of a formal planning procedure that involves relevant stakeholders before an event should expedite the response and confer legitimacy for decisions made during and after decontamination.
Recommendation 3-2
Representatives of affected parties should be involved in risk management decision making, and they should participate in the technical discussions needed to make decisions. Engaging the people whose well-being is most at stake helps ensure their greater confidence in the outcome of risk-based decisions. Those who provide the technical information should be independent experts who are free of conflicts of interest, so that they can give the highest priority to protecting public health. Stakeholder involvement in risk assessment and management provides valuable returns: local knowledge that can contribute to a more robust definition of the danger, greater public confidence in scientific tools that support public policy, and more widespread acceptance of the legitimacy of the results.
Finding 3-3
People and microorganisms cohabit the world; their interactions sometimes result in human disease. Nonetheless, in settings where people risk exposure to pathogens (laboratories, hospitals), biological safety policies can protect against human disease. Decontamination is not a standalone activity, but part of a larger set of controls over dangerous microorganisms and their potential health effects. The domestic institution that routinely dealt with weaponized pathogens—the U.S. Army Biological Warfare Laboratories—developed a comprehensive set of biological safety programs to control those pathogens. Protective measures ranged from preemptive vaccination to medical monitoring and treatment for inadvertent exposures.
Recommendation 3-3
Integrated protection for human health is the most prudent policy in the context of a facility contaminated as the result of a biological attack. After a facility has been decontaminated, some type of medical monitoring is critical to ensure confidence that a facility is safe, and the purpose and outcome of medical monitoring
should be made transparent to affected parties. In the event of any incident in the future, a centralized and sustained effort should be organized to track the health of those exposed, or potentially exposed, to pathogens.
REFERENCES
Burger, J. 2002. Restoration, stewardship, environmental health, and policy: understanding stakeholders’ perceptions. Environmental Management 30(5): 631-640.
Burger, J., T.M. Leschene, M. Greenberg, J.R. Karr, M. Gochfeld, and C.W. Powers. 2003. Shifting priorities at the Department of Energy’s bomb factories: protecting human and ecological health. Environmental Management 31(2): 157-167.
DHHS (U.S. Department of Health and Human Services). 1999. Biosafety in Microbiological and Biomedical Laboratories. Washington, DC: U.S. Government Printing Office.
EPA (U.S. Environmental Protection Agency). 1986a. Ambient Water Quality Criteria for Bacteria. Washington, DC: Office of Water. EPA440/5-84-002.
EPA. 1986b. Superfund Amendments and Reauthorization Act. [Online]. Available at: http://www.epa.gov/superfund/action/law/sara.htm.
EPA. 1997. Rules of Thumb for Superfund Remedy Selection. Washington, DC: Office of Solid Waste and Emergency Response. EPA 540-R-97-013.
EPA. 2000. Risk Characterization Handbook. Washington, DC: Science Policy Council.
Farkas, J. 1997. Physical Methods of Food Preservation. Pp. 497-519 in Food Microbiology: Fundamentals and Frontiers, M.P. Doyle, L.R. Beuchat, and T.J. Montville, eds. Washington, D.C.: ASM Press.
Fehner, T.R., and F.G. Gosling. 1996. Coming in from the cold: regulating U.S. Department of Energy nuclear facilities, 1942-96. Environmental History 1(2): 5-33.
GAO (U.S. Government Accounting Office). 1985. Cleaning Up Hazardous Wastes: An Overview of Superfund Reauthorization Issues. Washington, DC: U.S. General Accounting Office.
Havelaar, A.H., M.J. Nauta, and J.T. Jansen. 2004. Fine-tuning food safety objectives and risk assessment. International Journal of Food Microbiology 93: 11-29.
Henry, C.J., M.S. Moses, and R. Henderson. 1997. The need for policy and risk analysis - the Department of Energy experience. Toxicologic Pathology 25(1): 27-31.
Hoffman, F., A. Owen, I. Apostoaei, and B.A. Thomas. 2002. A perspective on public concerns about exposure to fallout from the production and testing of nuclear weapons. Health Physics 82(5): 736-748.
International Commission on Microbiological Specifications for Foods. 2002. Microorganisms in Foods 7: Microbiological Testing in Food Safety Management. New York: Kluwer Academic/Plenum Publishers.
IOM (Institute of Medicine). 2003. Scientific Criteria to Ensure Safe Food. Washington, DC: The National Academies Press.
Jones, C.R. 2004. A United States perspective on long-term management of areas contaminated with radioactive materials. Radiation Protection Dosimetry 109(1-2): 75-77.
Ledwidge, L., L.R. Moore, and L. Crawford. 2004. Stakeholder perspectives on radiation protection. Health Physics 87(3): 293-299.
Macler, B.A., and S. Regli. 1993. Use of microbial risk assessment in setting United States drinking water standards. International Journal of Food Microbiology 18(4): 245-256.
NRC (National Research Council). 1983. Risk Assessment in the Federal Government: Managing the Process. Washington, DC: National Academy Press.
NRC. 1994. Building Consensus Through Risk Assessment and Management of the Department of Energy’s Environmental Remediation Program. Washington, DC: National Academy Press.
NRC. 1996. Understanding Risk: Informing Decisions in a Democratic Society. Washington, DC: National Academy Press.
O’Leary, H.R. 1997. DOE nurtures openness to mend past miscues. Forum for Applied Research and Public Policy 12(3): 103-108.
Sehulster, L., and R.Y.W. Chinn. 2003. Guidelines for environmental infection control—health care facilities. Morbidity and Mortality Weekly Report 52(RR10): 1-42.
Thomas, J.P. 2001. Rebuilding trust in established institutions. Health Physics 80(4): 379-383.
Till, J.E., and K.R. Meyer. 2001. Public involvement in science and decision making. Health Physics 80(4): 370-378.
Wentz, C.A. 1989. Hazardous Waste Management. New York: McGraw-Hill.














