5
Framework for Event Management
The committee was directed to provide decision makers with practical advice about managing the final stages of decontamination and reoccupation of a facility after an act of bioterrorism. The case studies, field experience, and research outlined in Chapters 1-4 provide a basis for explaining how such an event might unfold and how various organizations and agencies have addressed real crises in public and private buildings. The succeeding chapters are meant to increase basic understanding about the specifics of buildings, the properties of biological agents that might contaminate buildings, and strategy for preparing for and managing events. The discussions concern risk assessment and risk management. Risk assessment involves identification and scaling of an event in terms of the biological agents involved and the effects on building systems and potentially exposed populations. Risk management broadly includes preparation, coordination, decontamination, clearance, communication, and medical monitoring.
This chapter introduces risk assessment and outlines the factors it should consider. Chapters 6-10 present some technical components of risk assessment and explain how characterization of the biological hazard, air movement in the building, dose–response modeling, sampling and identification of biological hazard, and decontamination methods used all contribute information to the assessment.
Chapters 5-10 lead to and support the discussions presented in Chapter 11, which offers decision makers guidance on response to and recovery from an act of bioterrorism. The committee recognizes there could be unforeseen circumstances or that some people might fill unfamiliar roles in an emergency. Laying out technical protocols will facilitate response in case of an attack. Developing
and practicing emergency plans and communication can lead to increased vigilance, security, and confidence among building occupants and can minimize anxieties during an event. We note that it is more likely that a building will have a flood, chemical spill, fire, or be subject to malodorous conditions, “sick building” problems, or disease clusters than that an attack with biological weapons will occur. The practical guidance offered here will have benefit if it is followed and applied to other untoward events that might occur.
Often, public health decisions (that result in regulatory or other actions) with respect to environmental exposures are made without complete knowledge. Some important factors often are not clear; others could be highly variable. A risk analysis methodology has been applied to environmental decision making in the United States over the past 20 years and by that approach, the estimated decreases in damage attributable to increasing regulation or control of exposure are balanced against the quantifiable, nonquantifiable, and noneconomic costs of an action to identify the most desirable decision. Although the metrics used to balance benefits and costs differ with the context, the overall process of assessing risks works the same way for many situations. The committee believes that the risk analysis framework is appropriate in the context of the question “How clean is safe?” for decontamination subsequent to the release of a biological agent.
A risk assessment that involves a microbiological pathogen—also known as quantitative microbial risk assessment (QMRA)—can follow the framework developed for assessing the risk attributable to exposure to harmful chemical agents as outlined by the National Research Council (NRC, 1983), and it broadly includes the following steps:
-
Hazard assessment involves identifying pathogens, determining how exposure occurred, and assessing the potential outcomes of infection (course of disease). The vectors and vehicles for secondary transmission also are identified for transmissible agents.
-
Exposure assessment evaluates the number of people who have ingested, inhaled, or otherwise been in contact with particular amounts (doses) of the infectious agents, and with what frequency.
-
Dose–response analysis examines the relationship between the dose of biological agent to an individual person and the probability of that person’s becoming ill. For populations, dose-response analysis characterizes the relationship between the dose of the agent in a given environment and the number of people who will become ill as a result of exposure to that agent in that place. Given a particular scenario for the distribution of doses and the associated uncertainties, the dose–response relationship provides an estimate of the expected number of adverse outcomes (disease cases) and their distribution and uncertainty.
-
Risk characterization is a “synthesis and summary of information about a hazard that addresses the needs and interests of decision makers and of interested
-
and affected parties “(NRC, 1996). Risk characterization is a prelude to decision making that involves communicating information about the hazard to decision makers and interested and affected parties so that they have a comprehensive understanding of the risks, variables, and uncertainties. Those parties would become informed participants and share their perspectives and concerns in the risk characterization process.
-
Risk management involves actions that should be taken to reduce the risks attributable to exposure to the biological hazard.
Using the terminology generally employed in the United States, risk assessment consists of the first four steps, and risk analysis encompasses risk assessment and risk management. Risk analysis includes policy and nonquantitative considerations, as described in other chapters of this report. Typically, a risk assessment is repeated several times for different management scenarios to determine the amount of remediation required to achieve a given reduction in risk. Risk assessment can be repeated for various degrees of remediation.
QMRA has been validated with several microorganisms:
-
Dose–response information for ingestion of cysts of Giardia lamblia correlated with illness rates associated with waterborne contact (Rose et al., 1991)
-
The attack rate during the massive waterborne outbreak of cryptosporidiosis in Milwaukee in 1993 matched projections from human dose–response information obtained from controlled human trials (Haas and Rose, 1994)
-
An animal dose-response relationship for Escherichia coli O157:H7 was consistent with the attack rate noted in a recreational-waterborne outbreak (Haas et al., 2000)
QMRA has been used to formulate guidelines or standards in several contexts. A risk assessment approach was used in the development of the surface water treatment rule, which requires utilities that process surface water to demonstrate specific treatment results for reduction of Giardia and viruses before the water is distributed (Macler and Regli, 1993). Proposed rationales for treatment of wastewater for direct or indirect reuse have applied QMRA methodologies (Tanaka et al., 1998). After a series of international consultations, the World Health Organization proposed a unified framework for the control of infectious disease transmitted in water (potable, recreational, and agricultural) that has microbial risk assessment as an underlying paradigm (Bartram et al., 2001). The selection of respirators as personal protective devices against airborne infection has been analyzed with microbial risk assessment (Nicas and Hubbard, 2002). That was derived after validation with reference to airborne transmission of Mycobacterium tuberculosis (Nicas, 1996).
Alternative protocols also have been presented—for example, the schematic
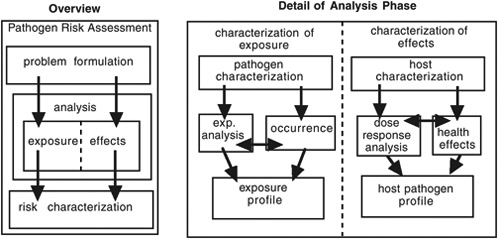
FIGURE 5-1 Schematic diagram of ILSI microbial risk analysis protocol. SOURCE: Adapted from ILSI, 2000.
protocol developed by the International Life Sciences Institute (ILSI) and shown in Figure 5-1 (ILSI, 2000). That protocol was based on the National Research Council framework (NRC, 1983) described above. It emphasizes the relationships between the technical and policy-making components of the risk assessment process, particularly at the problem formulation stage. It is designed to be useful for quantitative and qualitative risk assessments. As delineated by ILSI (2000), the process focuses on microorganisms in water, but much of it is relevant to the task of assessing harms caused by biological agents that could be released into public buildings or facilities. The first step is problem formulation, “a systematic planning step that identifies the goals, breadth, and focus of the risk assessment, [and] the regulatory and policy context of the assessment” (ILSI 2000). Figure 5-1 illustrates the second, or analysis, phase of ILSI’s approach. It shows how a decision-making body would need to consider both pathogen and host characteristics and the surrounding environment to properly assess exposure and effects.
To understand the flow chart, one must evaluate the elements within each box. Several elements could be relevant to the risk analysis for a contaminated public transportation facility. They would be used to create an exposure profile:
-
Pathogen characterization describes the virulence and pathogenicity of the microorganism, the diseases it cause, its survival and multiplication, its resistance to control or treatment, its host specificity, its infection mechanisms, its potential for secondary spread, and its taxonomy or strain variation.
-
Pathogen occurrence identifies concentration, spatial distribution (including clumping, aggregation, particles, and clustering), nonhuman reservoirs, survival, persistence, indicators, and surrogates for indirect evaluation.
-
Exposure analysis characterizes routes of exposure; the size and demographics of the exposed population; the spatial and temporal nature of exposure and whether single or multiple; the behavior of the exposed population; and treatment, processing, and recontamination.
The far-right-hand section of Figure 5-1 shows characterization of effects, which uses information from host characterization, dose–response analysis, and health effects to create a host–pathogen profile. ILSI’s protocol contains several elements that are relevant to risk analysis for a contaminated public transportation facility.
-
Host characterization describes the host population by age, immune status, concurrent illness, genetic background, pregnancy, nutritional status, demographics, and social and behavioral traits.
-
Health effects are identified, including the duration and severity of illness, infectivity, morbidity, mortality, sequelae of illness, extent of amount of secondary transmission, and quality of life.
-
Dose–response analysis is a statistical model that analyzes or quantifies dose–response relationships; human and animal dose-response data; use of outbreak or intervention data; route of exposure; source and preparation of material; organism type or strain, including virulence factors or other measures of pathogenicity; and characteristics of the exposed population.
The ILSI approach then pulls all of the relevant information together into a risk characterization that is subjected to two steps: risk estimation and risk description. That segment of the process would include characterization of uncertainty, variability, and confidence the decision-making group has in the estimates used above; sensitivity analysis to identify and evaluate the most important variables and information needs; and a decision analysis that evaluates alternative risk management strategies.
FINDINGS AND RECOMMENDATIONS
Finding 5-1
The QMRA process, developed over the past 20 years, has been used to inform decision making about events involving microbial hazards that affect food safety, drinking-water quality, and the use of isolation rooms in hospitals.
Recommendation 5-1
A risk assessment approach should be adopted as one component of decision making for determining the adequacy of decontamination efforts after a release or suspected release of a biological contaminant.
Finding 5-2
Thorough risk analysis requires critical information about each variable. This information is weak for certain variables when one considers agents that might be used in a biological attack.
Recommendation 5-2
More dose–response and sampling source data are needed to inform a practical, as opposed to a theoretical, risk analysis for any given biological attack.
REFERENCES
Bartram, J., L. Fewtrell, and T.-A. Stenstrom. 2001. Harmonised assessment of risk and risk management for water-related infectious disease: an overview. Pp. 1-16 in Water Quality. Guidelines, Standards and Health: Assessment of Risk and Risk Management for Water-Related Infectious Disease. L. Fewtrell and J. Bartram, eds. London: World Health Organization and IWA Publishing.
Haas, C.N., and J.B. Rose. 1994. Reconciliation of Microbial Risk Models and Outbreak Epidemiology: The Case of the Milwaukee Outbreak. Annual Conference—American Water Works Association, New York.
Haas, C.N., A. Thayyar-Madabusi, J.B. Rose, and C.P. Gerba. 2000. Development of a dose-response relationship for Escherichia coli O157:H7. International Journal of Food Microbiology 56(2-3): 153-159.
ILSI (International Life Sciences Institute). 2000. Revised Framework for Microbial Risk Assessment: An ILSI Risk Science Institute Workshop Report. Washington, DC: International Life Sciences Institute.
Macler, B.A., and S. Regli. 1993. Use of microbial risk assessment in setting United States drinking water standards. International Journal of Food Microbiology 18(4): 245-256.
Nicas, M. 1996. An analytical framework for relating dose, risk and incidence: an application to occupational tuberculosis infection. Risk Analysis 16(4): 527-538.
Nicas, M., and A. Hubbard. 2002. A risk analysis for airborne pathogens with low infectious doses: application to respirator selection against Coccidioides immitis spores. Risk Analysis 22(6): 1153-1163.
NRC (National Research Council). 1983. Risk Assessment in the Federal Government: Managing the Process. Washington, DC: National Academy Press.
NRC. 1996. Understanding Risk: Informing Decisions in a Democratic Society. Washington, DC: National Academy Press.
Rose, J.B., C.N. Haas, and S. Regli. 1991. Risk assessment and the control of waterborne giardiasis. American Journal of Public Health 81: 709-713.
Tanaka, H., T. Asano, E.D. Schroeder, and G. Tchobanoglous. 1998. Estimating the safety of wastewater reclamation and reuse using enteric virus monitoring data. Water Environment Research 70(1): 39-51.






