3
The Global Application of Knowledge, Tools, and Technology: Opportunities and Obstacles
Changes in travel and trade and the disruption of economic and cultural norms have accelerated and made it much more difficult to control the emergence and spread of infectious diseases, as described in Chapters 1 and 2 of this report. Even as progress is made, the public health community will likely encounter further setbacks, such as growing antimicrobial resistance. Yet there is a positive side to these developments as well. While globalization intensifies the threat of infectious disease, it also results in stronger tools for addressing that threat. From technological advances in information dissemination (e.g., the Internet) to the growing number of bidirectional infectious disease training programs that are bringing clinicians, scientists, and students from both sides of the equator together, the opportunities made available by globalization appear as endless as the challenges are daunting.
At the same time, the opportunities afforded by globalization do not necessarily come easily. Workshop participants identified obstacles that, if not addressed, may prevent or retard the ability to take full advantage of some of these new global tools. Global surveillance capabilities made possible by advances in information and communications technologies, for example, are still fraught with numerous challenges. This chapter summarizes the workshop presentations and discussions pertaining to some of these opportunities and obstacles.
One of the most enthusiastically discussed opportunities made available by our increasingly interconnected world is the type of transnational public health research, training, and education program exemplified by the Peru-based Gorgas Course in Clinical Tropical Medicine. This program not
only benefits its northern participants, but also helps build a sustainable public health capacity in the developing world. Historically, the goal of many tropical disease training programs was to strengthen the northern country’s capacity for tropical disease diagnosis and treatment. The trend toward a bidirectional, more egalitarian approach that benefits the developing-country partner as much as its northern collaborator reflects a growing awareness that a sustainable global public health capacity can be achieved only with the full and equal participation of the developing world. Thus, not only are the Gorgas Course and other, similar programs becoming more popular, both politically and among students, but their nature is also changing in significant and telling ways. The shifting focus of many of the international training programs of the Fogarty International Center (FIC) within the National Institutes of Health (NIH) further reflects the increased awareness, funding, and efforts needed to strengthen bidirectional international training in epidemiology, public health, and tropical medicine in particular.
Are there enough of these programs to go around, however? In addition, despite the clear and growing need and interest, are U.S. medical and veterinary students receiving enough training in public health, let alone in tropical infectious diseases? The Gorgas Course, FIC programs, and other, similar initiatives are summarized here. The chapter also addresses the need to better incorporate public health training into U.S. medical and veterinary school curricula and ways in which Russian scientists could contribute to a transnational public health education program.
Workshop participants identified other opportunities for progress as well. These include worldwide access to antiretroviral agents and vaccines; an increased capacity for global and regional surveillance; and technological advances in information and communications technology, namely, the Internet.
Rather than focusing on specific opportunities, several of the presentations and discussions revolved around the various ways in which certain organizations and regions are capitalizing on such opportunities. The Pan American Health Organization (PAHO), for example, is relying on the development of regional political networks to aid in the construction of regional surveillance networks and to facilitate the sharing of diagnostic and treatment techniques across borders. Despite these promising developments, many obstacles impeding regional efforts to strengthen infectious disease control capacity remain.
Another regional example is Russia, which, despite its current general state of public health as described in the previous chapter, has experienced some recent achievements and taken advantage of opportunities for the development of international collaborations in infectious disease control. The State Research Center of Virology and Biotechnology (VECTOR) plays a leading role in these efforts, which are summarized here.
A final example is the relationship between the Massachusetts state public health laboratory system and public health clinics in Peru, whereby sputum specimens and isolates from Peru are sent to Massachusetts on a daily basis for drug resistance testing. This arrangement could serve as a model for a much larger, market-based approach to sharing limited public health resources.
TRANSNATIONAL PUBLIC HEALTH TRAINING PARTNERSHIPS1
As noted above, the trend toward bidirectionality in transnational education and training, whereby southern partners have as much to gain as their northern collaborators, reflects a growing awareness that a sustainable global capacity to respond to infectious disease threats requires the full and equal participation of developing countries where infectious diseases are endemic. It is vitally important that the intellectual, technological, and health care workforce capacities of the developing world be strengthened, both for the sake of improving the health of local populations and because so many of the world’s infectious diseases arise in tropical countries and spread via nonindigenous travelers. One-third of all new infectious diseases identified over the past 25 years were discovered in Latin America. The Gorgas Course in Clinical Tropical Medicine and other international training programs offered by the Instituto de Medicina Tropical “Alexander von Humboldt” (IMT) in Lima, Peru, and several of the overseas training programs sponsored by FIC exemplify this trend toward bidirectionality, as described in this section.
Nevertheless, in addition to improving the capacity of the developing world, one of the primary goals of these programs remains the education and training of northern students, researchers, and practitioners. Despite the progress made over the past decade in providing increasing numbers of opportunities for U.S. students and health care practitioners to gain experience overseas, including experience with the treatment and control of tropical infectious diseases, much work remains to be done. Efforts of the American Society of Tropical Medicine and Hygiene (ASTMH) and others to improve the training of U.S. students in tropical infectious disease medicine are also summarized here.
Workshop participants expressed serious concern that too few medical and veterinary school students receive adequate public health training in general, let alone training in tropical infectious diseases. It is vital for front-
line practitioners to receive such training. As one participant observed, it was an astute physician who identified inhalational anthrax in Florida in the fall of 2001. This section includes a summary of the discussion pertaining to the need for public health training and the challenges, mainly monetary, that make meeting this need difficult.
Although most of the discussion of transnational training partnerships focused on collaborations between the developing and developed worlds, particularly between the United States and Latin America, participants also discussed how a global approach to public health education and training would benefit Russia, especially in light of rapidly developing antiglobalist and xenophobic sentiment in that country. Some examples of ways in which Russia could, in turn, contribute to a mutual exchange of information and skills were also outlined.
The Gorgas Course and Other Recent Bidirectional Training Initiatives: A Peruvian Perspective
Although U.S. and Latin American scientists have been collaborating for more than two decades, the collaborations have tended to involve only certain U.S. universities interacting with certain Latin American countries. This same colonial attitude has been true of European–African collaborations as well. The situation is changing, however. Developed-world partners are adopting new approaches in their interactions with the developing world and realizing that respecting the decisions made by their developing-country partners is critical to the long-term sustainability of these partnerships.
For example, IMT, on the campus of the Universidad Peruana Cayetano Heredia in Lima, where the Gorgas Course in Clinical Tropical Medicine is held, maintains a strong collaboration with the Belgian-run Institute of Tropical Medicine in Antwerp. An important component of this four-year collaboration is the way in which the Belgian partners have respected IMT’s decisions. When the collaboration was initiated, the Belgians asked, “What is your priority? How would you like your Institute to develop?” A workshop ensued, IMT devised a plan, and the Belgians agreed with and supported the plan.
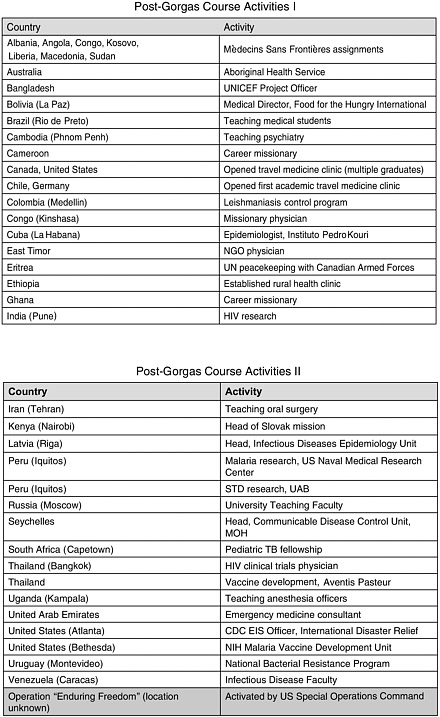
Another, more popular example is Peru’s interaction with the University of Alabama (UAB) in support of the Gorgas Course in Clinical Tropical Medicine. This annual nine-week diploma course provides 380 contact hours (in English) and daily bedside teaching. Over the last six years, the Gorgas Course has trained 185 medical doctors from 44 countries. About 50 percent of the participants are from the United States, 17 percent from Canada, 12 percent from Latin America, 10 percent from Europe, six percent from Asia, five percent from Australia and New Zealand, and two percent from Africa. Most of the trainees are midcareer professionals with
about 10 to 15 years of experience. Participants range from United Nations Children’s Fund (UNICEF) project officers to missionary physicians, U.N. peacekeeping forces, and vaccine developers (see Figure 3-1). The age range of the participants is from under 30 to over 60.
The Gorgas Course does more than train. In two months alone there were some 10,000 visitors to cases published on its website. And since the anthrax attacks in the United States, there have been 7,000 U.S. inquiries regarding the anthrax photos provided by the Gorgas Course case collection. IMT also sponsors several other training programs and courses for professionals and students. For example, small grants are available for U.S. students to go to Peru for three or four months at a time and conduct research; two U.S. students participate each year and together have coauthored six published papers to date.
The bidirectional nature of IMT’s relationship with UAB is illustrated by the approximately 20 Peruvian students who have trained at UAB over the last three years. In 2002 there were eight Peruvian residents in the UAB Department of Medicine, and the chief resident was from Peru. IMT also offers its own master’s degree in infectious disease control, although this program is not yet as well supported as others. In recognition of the fact that IMT provides sustainable, credible training and is expected to continue to grow, UAB is undertaking several other new initiatives. In 2003, for example, IMT initiated a separate program for trainees from other Latin American countries, including Bolivia, Paraguay, Ecuador, and Colombia. UAB is also developing a plan to provide clinical training in HIV/AIDS and tuberculosis (TB).
Other Peruvian collaborations with the United States include a ten-year partnership with the University of Washington School of Public Health and Community Medicine; some 40 published papers have resulted from this collaboration. Several personnel from the University of Washington have also trained in Peru. Other examples include collaborations with the University of Maryland (cholera and typhoid fever), Johns Hopkins University (cysticercosis), and Harvard-affiliated Partners in Health (TB and multidrug-resistant TB).
In addition to respecting the decisions made by participants from the developing world, the adequate transfer of economic support is vital to the continued success of these types of programs. The failure to transfer sufficient funds to developing countries is one of the key issues regarding international partnerships. This is especially true of partnerships with the United States, as about 90 percent of U.S. funds designated for training programs in Latin American developing countries are actually spent in U.S. institutions. This is the case even when the award recipients are Latin Americans. In reality, the amount of funds transferred is very small, as the award money still belongs to U.S. institutions and private companies. The situa-
tion is analogous to American travelers who visit Peru but fly American Airlines, stay at the Sheraton or Marriott, and travel with a U.S. tour group. Although the trip may cost the tourist $3,000, only about $20 stays in Peru; the rest goes back to the United States (Gotuzzo, 2002).
The transfer of technology, skills, and knowledge gained from research endeavors in the developing world is another key issue. With regard to the transfer of technology, for example, the BACTEC system for drug susceptibility testing of Mycobacterium tuberculosis is available but cannot be used in Peru. With regard to the transfer of knowledge, it is critical that research conducted in developing countries have a significant, positive local impact. Even though European and U.S. countries have spent $1 billion on malaria research in just one African country over the past 50 years, the rates of mortality and morbidity from the disease have not changed, and the quality of life has not improved. This situation must be changed.
The Changing Nature of Fogarty International Center’s Transnational Training Partnerships
The 35-year-old FIC has a specific mandate to promote and support international scientific research and training in the global health sciences and reduce global health disparities. In its early years, the agency focused largely on the exchange of scientists among developed countries. Over the last 15 years, however, it has increasingly emphasized scientific capacity building and training in developing countries by awarding traditional training grants, mainly to universities within the United States. This shifting programmatic emphasis reflects the changing nature of training partnerships between developed and developing countries. In addition, FIC maintains several mathematical modeling research programs addressing a wide range of issues, from disease prevalence to rates of global mortality from influenza and the biomedical modeling of bioterrorism.
FIC’s traditional training grants for capacity building and training have been highly successful. Foreign trainees come to the United States, where they receive clinical, laboratory, and research training in public health and then return to their countries, where they use their new skills in leadership roles. As a measure of FIC’s success, one-quarter of the scientific presentations at the World AIDS Congress in Durbin, South Africa, in 2000 had one or more authors who had been trained with FIC support. In fact, these capacity-building programs have been so successful that they have reached the point at which the foreign sites are now able to assume much more responsibility and autonomy.
Because of this success, FIC, in collaboration with its 10 NIH partners and the Centers for Disease Control and Prevention (CDC), has established
a new, next-generation grants program known as the International Clinical, Operational, and Health Services Research Training Award for AIDS and Tuberculosis (ICOHRTA-AIDS/TB). There are several important differences between this new approach to training individuals from overseas and the earlier, more traditional grants:
-
Training site—Most significant is that, although the grants still fund collaborations, or partnerships, between sites in the United States and abroad, they are increasingly centered in the low-resource country. In the past, foreign trainees were brought to the United States, where they enrolled in courses and, in some cases, degree programs. Now, the training sites are increasingly located in the developing countries, and the developing-country participants set the agenda, decide whom to work with, and assume much more ownership of their efforts than in the past. This shift in emphasis empowers resource-limited countries to sustain their own health care initiatives without continued reliance on CDC and other outside agencies.
-
Research agenda—In the past, research agendas were set mainly by grant recipients in the United States. The agendas of ICOHRTA-AIDS/TB will increasingly be set by individuals in the developing countries.
-
Faculty mentoring—Faculty mentoring was previously performed mainly by faculty in the developed country. It is now being done increasingly by faculty in the developing country.
-
NIH funding—In the past, NIH funding went to the site in the developed country. NIH funding now goes to sites in both the developing and developed countries; paired grants will include direct funding, including overhead costs, to both countries.
-
Types of awards—The long-term sustainability of the grants program at a foreign site requires a long-term commitment from the United States. Traditional training grants are typically for five years, but these new-generation grants will be 10-year (or longer) cooperative agreement programs.
-
Research emphasis—The earlier grants program focused mainly on epidemiology and prevention, important work without which these new-generation grants would not be possible. One of the primary goals of the new program, however, is to expand this focus and integrate clinical, operational, and health services research in an effort to apply results obtained at the benchtop to the bedside. The emphasis is on the integration of therapy and care with prevention efforts.
In addition to FIC’s training grants programs, NIH oversees several other, similar international efforts, including the Prevention Trials Network, the Vaccine Trials Network, the Comprehensive International Program of Research on AIDS, Popular Opinion Leaders, International Cen-
ters for Excellence in Research, and Partnerships for HIV/AIDS Research in Africa. FIC also helps foster many major U.S. government and other international AIDS research efforts, including those of CDC’s Global AIDS Program; the U.S Agency for International Development’s Rapid Response Initiative; the Bill and Melinda Gates Foundation; the American Foundation for AIDS Research; the Elizabeth Glaser Pediatric AIDS Foundation; the Academic Alliance for AIDS Care and Prevention in Africa; and the Global AIDS, Tuberculosis, and Malaria Fund.
One of the most recently established FIC programs is the Global Health Research Initiative Program for New Foreign Investigators, a reentry grant program with the aim of reversing the “brain drain” from foreign countries that occurs when scientists come to the United States for graduate school and NIH training and do not return home. The program provides five years of support at $50,000 a year to scientists who reenter their home country after receiving training in the United States.
Also relevant is the International Training and Research Program in Emerging Infectious Diseases, a traditional program carried out at university sites conducting research overseas. In addition, FIC has partnered with the National Science Foundation and created the Ecology of Infectious Disease program. The purpose of this program is to support efforts toward understanding the ecological and biological mechanisms that govern the relationships between human-induced environmental changes and the emergence and transmission of infectious disease. Finally, FIC funds the International Studies on Health and Economic Development program, which supports projects examining the effects of health on microeconomic agents (individuals, households, and enterprises) and aggregate growth, as well as the effects of health finance and delivery systems on health outcomes.
Russia’s Potential Contributions to Transnational Training
Although most of the discussion of international bidirectional training programs during the workshop focused on north–south collaborations, and U.S.–Latin American partnerships in particular, some participants commented on the need for a transnational public health research and education agenda in Russia, especially with regard to infectious disease control in migrant populations. Many Russian experts are capable of participating in such international collaborative programs, and Russia could contribute to this type of program in many ways:
-
Russia’s unique experience with protecting its population from extremely dangerous infections as part of the former Soviet Union’s so-called “counterplot service” could be a very useful source of information for other countries.
-
Russia could play a leading role in prevention and control programs for HIV/AIDS and other infectious diseases throughout Eastern Europe.
-
Russia’s stored smallpox specimens—one of the world’s two supplies of wild virus—could be used for international collaborative research.
-
Russian experts who are developing a register of sites in Russia where anthrax (Bacillus anthracis) spores are stored could contribute to an international effort to register and monitor anthrax sites around the world. Such a registry could be used in bioterrorism prevention and control, for example, to differentiate between bioterrorist and natural outbreaks.
-
Russian experts could also aid in the development of a register of infectious disease carriers for use in preventing the dissemination of infectious diseases beyond national borders as a result of migration.
Recent Overseas Training Initiatives: A U.S. Perspective
After the Institute of Medicine (IOM) announced in a 1987 report, U.S. Capacity to Address Tropical Infectious Disease Problems, that only 300 people in the United States had the capability to diagnose and treat tropical diseases, ASTMH formed a committee to formulate recommendations for remedying the situation (IOM, 1987).2 It was clear that the United States had no truly excellent program offering the kind of diploma training course, including laboratory and overseas experiences, called for by the IOM report. The ASTMH committee recommended that an examination in clinical tropic medicine be administered and that a diploma in tropical medicine and hygiene be offered. ASTMH distributed a request for proposal to 370 U.S. and Canadian medical schools; 22 schools responded, and the proposals from seven U.S. and five overseas medical schools were accepted. Today, there are strict requirements for a diploma course and a separate two-month overseas course. Since 1995, 619 individuals have taken the examination, 412 have passed, and 387 have had the overseas experience.
The Gorgas Course, which prepares students for the ASTMH examination, is an excellent example of the type of training program in tropical medicine and hygiene envisioned by ASTMH. In addition to the Gorgas Course, other ASTMH-accredited diploma courses are offered at the following universities: Bernard Nocht Institute (Germany), Case Western Re-
serve University, Humboldt University (Germany), Johns Hopkins University, Liverpool School of Tropical Medicine, London School of Hygiene and Tropical Medicine, Mahidol University (Thailand), Tulane University, University of Virginia, West Virginia University, and Uniformed Services University of the Health Sciences.
A number of other ASTMH training initiatives were inspired by the 1987 IOM report. These include the Ben H. Kean Traveling Fellowships in Tropical Medicine (for clinical or research experiences for medical students and residents); the Centennial Fellowship (for senior undergraduate and graduate students to work on immunomolecular parasitology with colleagues in the developing world); the Burroughs-Wellcome Fund Fellowship (for overseas projects at collaborating field sites); the Gorgas Memorial Research Award (for Latin American and Caribbean scientists to work with their North American colleagues on collaborative projects); and travel awards for overseas colleagues to present their research at ASTMH annual meetings.
Many non-ASTMH educational initiatives have also emerged recently worldwide. They include the Yale World Fellows Program and the Yale/ Johnson & Johnson Physician Scholars Program. The former attempts to bring together midcareer professional leaders from academia, government, nongovernmental organizations, business, and media from throughout the world in an effort to create an international dialogue and network among fellows and broaden the general understanding of globalization. Participants receive a generous stipend for themselves and their families so they can attend Yale for a semester or a full year. Fellows participate in seminars and independent studies during an initial visit; they revisit the campus after having worked back at their home sites for two years.
The Yale/Johnson & Johnson Physician Scholars program, which started in 1981, sends physicians-in-training in a variety of subspecialties to 16 sites around the world for overseas rotations. In 2002, when the program was broadened to include a national competition, it funded 65 scholars, including senior midcareer physicians. The goal is to place physicians in underserved areas where they can learn to recognize tropical diseases and develop long-standing bilateral relationships between their home school or hospital and the host area.
Not only there is a need for more training in tropical infectious diseases among health care providers in the United States and other countries in the developed world, but there is also a strong student demand for such training. By going abroad, students learn about and encounter diseases and conditions, such as measles, that do not occur regularly or at all in the United States. In 1984, just four percent of graduating medical students participated in overseas training. This percentage has risen dramatically. According to an unpublished 2002 study of nearly 400 internal medicine
residency programs, 45 percent of those programs offered overseas training electives; another 45 percent expressed interest in developing such electives.
Students who have spent time abroad usually return to their home institution full of excitement about international and other career possibilities that they might otherwise not have considered. According to one study, physicians who participate in the Yale School of Medicine’s International Health Program are much more likely, as they pursue their careers, to care for patients who require public assistance, immigrants, HIV-positive patients, and substance abusers (Gupta et al., 1999). The results of that study, however, are obviously affected by a selection bias, as not all physicians-intraining choose to undertake such overseas electives.
Despite the successes of the various overseas training programs described above (e.g., the Gorgas Course and FIC’s overseas programs) and the need and student demand for more of these opportunities, many ongoing programs have several problems that need to be addressed:
-
Participants often do not receive a sufficient preparatory curricular and cultural introduction to their programs and host sites.
-
Trainees often lack the ability to speak the language of the host site.
-
The programs lack a standardized curriculum across schools.
-
Training places a potential burden on host sites. As the demand for overseas training and the number of students involved increase, this burden will become greater.
-
The demand for overseas training opportunities is very high, but the supply of good mentorships is very low.
-
There is a lack of bidirectional support—for example, funding to bring overseas faculty back to the United States for minisabbaticals.
Content of Medical and Veterinary School Public Health Curricula
The concern that most medical and veterinary school students are not receiving enough general public health education, let alone training in tropical infectious diseases, was discussed above. Most medical school curricula still offer only an hour or less of instruction in antibiotic resistance and the mechanisms of action of antibiotics. As noted earlier, some physicians practicing today have never seen a case of measles, and some veterinarians practicing today have never encountered foot-and-mouth disease.
Although training in tropical infectious diseases should constitute a significant component of the public health education of medical and veterinary school students, it is important to keep in mind that not all infectious disease problems emerge in the developing world. The United States is experiencing many problems within its own borders that need to be
addressed. For example, the Alabama state health commissioner indicated at a recent public health forum on emerging infections that not enough diphtheria-pertussis-tetanus vaccine boosters are available to be administered to the next generation of Alabamians, and several audience members at that forum said they had taken their children to receive tetanus shots only to be told that there was no tetanus vaccine available.
A workshop participant noted that as a result of the 2001 anthrax attacks and the increased threat of bioterrorism, both real and perceived, instructors are generally spending more time on the pathogenesis and treatment of infectious diseases, particularly those that would result from the release of potential bioterrorist agents, and that the American Medical Association has been working on additions to the curriculum. However, the general perception is that the curriculum is already full, and in fact, the Liaison Committee on Medical Education has indicated that students today have too much contact time and need fewer lectures and more hours of small-group learning sessions.
Another participant suggested that perhaps medical and veterinary school curricula could be modified to incorporate public health training programs—both general programs and those focused on tropical infectious diseases—without necessarily increasing contact time. However, the cost of doing so could be prohibitive. U.S. academic centers are currently under great financial stress because of the obligations placed on them as a result of requirements promulgated at both the state and national levels. Most of this financial stress is a consequence of state balanced budget amendments and continuing reductions in levels of reimbursement by federal payers, despite inflationary pressures. This situation is occurring in the face of growing numbers of uninsured and indigent patients, who are disproportionately and largely cared for by academic health centers. Moreover, fewer physician-scientists in the United States are applying for NIH grants and conducting research than in the past, and the number of M.D.’s applying for grants is declining in proportion to the number of applying Ph.D.’s. The recently increased NIH budget and the new influx of funding for biodefense (see the discussions of funding in Chapters 2 and 4) will certainly help alleviate this situation to some extent, but they are not solutions. It is worrisome that medical research institutions may be too preoccupied with other matters to take full advantage of the unprecedented opportunity offered by the new biodefense funding.
Thus, although the participants generally agreed that medical schools have an obligation to teach physicians about global public health and to equip them with cultural competence within the realm of health care, they also acknowledged the severe financial pressures that currently plague academic health centers. These financial pressures, which are expected to worsen in the future, combined with an already full curriculum, make curriculum modification a difficult challenge.
Finally, a participant suggested that training and research consortia and collaborations among U.S. academic institutions be established to make better use of limited funding and resources. This has already been accomplished to a limited extent but could be done much more effectively. The key may lie with NIH, CDC, or other funding programs with the power to promote these kinds of collaborations. One goal of such collaborative programs would be to take advantage of the expertise that exists in different places so as to avoid duplicated or fragmented efforts. The focus would be on one or two collaborative sites and on doing what needs to be done to make those sites truly excel, rather than on dividing the funding among multiple sites.
REGIONAL EFFORTS TO TAKE ADVANTAGE OF GLOBAL OPPORTUNITIES3
Three regional or organizational efforts have been undertaken to take advantage of some of the newly available global tools and opportunities: (1) PAHO’s efforts to prevent the spread of infectious diseases in the Americas; (2) Russia’s effort to build new international bridges in the wake of post-Soviet reform; and (3) a partnership between the Massachusetts state public health laboratories and Peruvian health clinics that serves as a model for a novel, market-based approach to sharing limited public health resources. This section summarizes these efforts.
Regional Efforts in the Americas: Global Opportunities and Obstacles
The original mandate of the 100-year-old PAHO was to help countries work together to prevent the spread of communicable diseases. While having broadened over time, the original mandate remains fundamental. Communicable diseases are still a major public health problem throughout the Americas, as recent outbreaks of emerging and reemerging infectious diseases in the region attest. PAHO’s 1995 Regional Plan to Control Emerging and Reemerging Diseases identified four major steps necessary to address this continuing threat. Other organizations, such as the World Health Organization (WHO) and CDC, have identified the same steps as priority needs worldwide:
-
Improve surveillance.
-
Strengthen the public health infrastructure through laboratory capacity building and training.
-
Improve the means used to translate the findings from applied research into simple diagnosis and treatment techniques, including methods that can be used in the field.
-
Develop a capacity for outbreak detection, primarily by formulating prevention and control guidelines or manuals and conducting training programs.
PAHO’s efforts to take these steps illustrate how some of the new opportunities made available by globalization are being or can be used on a regional scale. For example, improved information and communications technologies allow faster cross-border communication. PAHO is currently assisting Central American countries with the development of INFOCAM, an electronic system that will allow routine sharing of surveillance information. Also being used are e-mail, electronic chat rooms, and the Program for Monitoring Infectious Diseases (ProMED)—an Internet-based reporting system dedicated to rapid and global dissemination of information on outbreaks of infectious diseases that affect human health. Despite these many opportunities and early success stories, however, much remains to be done.
Opportunities
Several political networks have developed throughout the region, and these, in turn, have encouraged more cross-border cooperative efforts to prevent and control the emergence and spread of infectious diseases. Examples include the following:
-
Caribbean Community (CARICOM), RESSCAD (meeting of the health sector of Central America), Mercosur in the southern cone of South America, and Hipolito Unanue in the Andes comprise countries that have joined together not only to improve their economic situations, but also to enhance infectious disease surveillance efforts and develop regional health plans.
-
Caribbean countries have developed a subregional plan for HIV/ AIDS, which has been signed by all their health ministers. Such a collaborative effort is especially critical in this region, where migration among countries is an important factor in the transmission of HIV.
-
With CDC support, countries have banded together and agreed on the priority syndromes they will strive to address. For example, the countries in the southern cone of South America have assigned priority to efforts against hemolytic-uremic syndrome and influenza, and the countries of the Amazon basin to efforts against sudden adult death, hemorrhagic fevers,
-
and malaria. The countries run certain laboratory tests to detect the agents responsible for these syndromes and then report their results. Extensive in-country training occurs to strengthen laboratory capacity and the capabilities of laboratory clinicians.
-
Nineteen countries are participating in two networks that evaluate the antimicrobial resistance of several organisms.
-
Nine countries in the Amazon basin have held regional meetings to address malaria-related issues.
-
Three subregional laboratory surveillance networks have been established in the Americas: one in the Amazon basin, one in the southern cone, and one in Central America.
-
A regional laboratory network for food analysis has been set up in the Americas, and all countries in the region are now using the Hazard Analysis Critical Control Point Inspection model for the inspection of food-processing establishments. Several regional meetings have been held to determine how the risk of bovine spongiform encephalopathy can be assessed and what steps must be taken to investigate any reports or suspicions.
-
Southern cone countries have agreed to work together and meet annually to discuss efforts to control Chagas’ disease. Their collective efforts have already reduced the spread of the disease vector (through the spraying of thatched houses with insecticide), and the seroprevalence of the disease among schoolchildren has decreased by 90 percent.
Regional political developments have made it easier to share various diagnostic and treatment techniques. This, coupled with increased, faster travel, has also made it easier for people from different countries to come together for training; for example, training in detecting and responding to outbreaks has occurred in all South American and several Central American countries. The sending of specimens to regional reference laboratories for analysis has been facilitated as well. Along with CDC in Atlanta, Georgia, several regional laboratories are now acting as reference laboratories as well as training centers.
Finally, because of globalization, free trade, and the critical role of participation in the world market in a country’s economic development, countries throughout the Americas have generally become more motivated to improve their public health. For example, as was witnessed in the United Kingdom, the economic repercussions of foot-and-mouth disease can be devastating. The rapid control of the 2000–2001 South American outbreak of foot-and-mouth disease demonstrated not only the importance of communicating and working together, but also the economic benefits of doing so. The certification that Uruguay and Argentina were free of disease, for example, increased their exports to the United States by billions of dollars.
Obstacles
Despite the many successful regional efforts to strengthen the capacity to prevent and control the emergence and spread of infectious diseases in the Americas, numerous obstacles remain. These obstacles are not necessarily unique to PAHO or the Americas. In a sense, although globalizing forces have in many ways made countries working together to control the spread of local outbreaks more feasible, in other ways they have made it more difficult:
-
As important as immediate intercountry communication is to the rapid detection and control of local outbreaks, many countries hesitate to report such outbreaks because they fear the potential economic repercussions. If a country were to announce that it was experiencing a dengue, cholera, or foot-and-mouth disease outbreak, for example, not only would tourism plummet, but so, too, would the country’s exports. Most countries would prefer to try to control an outbreak without broadcasting its occurrence.
-
It is important to note that the purpose of rapid intercountry communication is not just to prevent the spread of local outbreaks, but also to aid in the detection of local outbreaks that may already have spread. A generation ago, the epidemiology of food-poisoning outbreaks might have been limited to church picnics, where everybody got sick in the same place. Now, with so much international travel and the global transport of food, infectious disease cases arising from one source may appear in other locations very far away. Identifying cases of disease in disparate geographic locations that have arisen from the same source poses a great challenge.
-
As the world becomes more international, there is a growing need to comply with international standards for the interregional and transcontinental movement of goods and products, such as standards imposed by the World Trade Organization (WTO). Many countries are being forced to improve previously ignored conditions in order to export goods. They are also being forced to develop credibility. When a country says it is free of a certain disease, has tested a certain product, or has carried out a certain inspection, people must be able to believe it.
-
Sometimes, when a food product does not meet export standards, it is consumed or sold locally. Another major challenge has therefore become ensuring that food consumed or sold locally is of standard export quality, and that improvements countries are making in their production and food-processing practices are countrywide and not confined to their export industries or factories.
-
Finally, to improve public health, it has become necessary to work outside regular health channels and target everyone at the community level, from vendors to street children and favelas.
After the workshop presentation on PAHO’s regional efforts, a participant asked about PAHO’s role in Cuba, a country that has the lowest rate of HIV infection in the Americas and that has done a reasonably good job of controlling dengue, and yet either is not recognized for its public health efforts or is being punished in other ways. In response, it was pointed out that PAHO is simply a forum for the exchange of ideas among countries and that U.S. policy toward Cuba is not a PAHO issue. Cuba does in fact take part in many of the same PAHO training sessions and joint meetings in which the United States participates.
Russia: Opportunities for International Partnerships in a Borderless World
A critical but little appreciated factor in the achievement of smallpox eradication was a remarkable collaboration between WHO, the USA, and the USSR beginning in 1966 and extending through the certification of eradication in 1980. Although many years during this period were especially tense ones in East-West relationships, professionals from two countries worked together … to bring the smallpox eradication campaign to a successful conclusion.
—Henderson, 1998, p. 113
Over the past decade, Russia has been witnessing the emergence and reemergence of multiple infectious diseases, from hepatitis B to HIV/AIDS. These crises, coupled with the fact that the percentage of research funding allocated to the life sciences is far less than that in most other developed countries, has put the country in a precarious public health situation. Yet as much as globalization and the breakdown of cold war borders and social structures may have contributed to this situation, they have also provided opportunities for the development of international collaboration in emerging infectious disease research and prevention.
VECTOR plays a leading role in Russia’s participation in various international cooperative efforts to prevent and control the emergence and reemergence of infectious diseases, and serves as an excellent example of the type of international collaboration made possible in a borderless world. VECTOR is located in Koltsovo, in the Novosibirsk region, the geographic center of the Russian Federation. Novosibirsk is the third-largest city in the Russian Federation and is located about 1,000 kilometers from the border with Mongolia and China and about 400 kilometers from the border with Kazakhstan.
VECTOR is organized into three main divisions: Research, Production, and Support. The Research Division includes six research institutes (Research Institute of Molecular Biology; Research Institute of Aerobiology;
Research Institute of Bioengineering; Research, Design, and Technology Institute of Biologically Active Substances; Research Institute of Cell Cultures; and Research Institute: Collection of Cultures of Microorganisms), as well as the WHO Collaborating Center for Orthopoxvirus Diagnosis and Repository for Variola Virus Strains and DNA. The Production Division produces pharmaceuticals and provides energy, water, and heat. The Support Division includes an animal breeding and holding facility and service and transport departments.
The center is involved in multiple public health research and disease prevention endeavors encompassing a wide range of infectious agents, including a collection of about 120 smallpox virus strains that SCR VB Vector scientists are studying in close cooperation with the United States and WHO. Moreover, the center is the only civilian institution in Russia where researchers work with Ebola and Marburg viruses. Additional agents with which VECTOR works include other human and animal pathogenic orthopoxviruses, tickborne encephalomyelitis virus, Japanese encephalomyelitis virus, Crimean-Congo hemorrhagic fever virus, hemorrhagic fever virus with renal syndrome, hepatitis viruses (A, B, and C), cytomegalovirus, herpesviruses, measles and mumps viruses, Venezuelan equine encephalomyelitis virus, rubella virus, HIV types 1 and 2, influenza viruses, some endemic animal and fish viruses, and multidrug-resistant TB.
The center is producing two immunobiological products for public health. The first is an innovative hepatitis A vaccine (Hep A-in-Vac), a cultured, inactivated, injectable vaccine for use against viral hepatitis A in adults and children. Hep A-in-Vac is unique to Russia, where VECTOR has been producing it since 1997. Second is an injectable vaccine against measles. VECTOR is the second-largest producer of a vaccine against measles in Russia, after the Moscow Enterprise of Viral Preparations.
VECTOR has also developed a range of diagnostic test kits that are currently being produced in the Production Division, and in some cases by shareholder companies. Infections or agents for which kits have been developed include: HIV, hepatitis (A, B, C, and D), tickborne encephalitis, measles, cytomegalovirus, toxoplasmosis, lues (syphilis), chlamydiosis, herpes, rubella, toxocariasis, opisthorchiasis, trichinosis, and helminthiasis.
Funding for Life Sciences in Russia and the United States
Funding for life science research in the United States has increased almost constantly over the last four decades in terms of both the absolute amount and the relative percentage of all (except defense-related) science research funding. By 2000, funding for life science research had reached almost 50 percent of all nondefense science research funding. The situation
in Russia is very different. According to the Russian Foundation of Basic Research (RFFI), the main source of competitive research funding in Russia, life science funding in 2001 represented less than 22percent of all science research funding. Because RFFI does not fund defense science, these figures indicate that the relative percentage of life science funding in Russia is more than two times lower than that in the Untied States.
Unfortunately, the same is true of foreign and international funds awarded to Russian scientists. For example, in 2000 only 25 percent of all Cooperative Grants Program awards to Russia were in the biological sciences. In 1999, only 18.6 percent of all funding from the International Science and Technology Center (ISTC), the main funding source for science research in Russia, was for biotechnology and life sciences. The latter figure has risen during the past couple of years to about 24 percent, but it is still well below the 50 percent level in the United States. Over the past several years, the number of ISTC-funded VECTOR projects has increased from less than five in 1995 to 25 or 26 in 2002. In addition to ISTC, other international programs from which VECTOR receives funding include the Civilian Research and Development Foundation in the United States; the International Association (INTAS) of the European Community; the North Atlantic Treaty Organization science program; the Initiatives for Proliferation Prevention (IPP) Program of the U.S. Department of Energy; FIC; and the Volkswagen Foundation in Germany.
International Collaboration
Several recent achievements in public health in Russia can be attributed to the increasing collaboration between VECTOR and the various international funding agencies and programs cited above:
-
One of the first ISTC grants helped VECTOR complete preclinical trials and organize clinical trials for its hepatitis A vaccine, which has been in production since 1998. Although VECTOR started developing the vaccine in the mid-1980s, a shortage of funds in the mid-1990s delayed its dissemination.
-
VECTOR has been collaborating closely with several international and foreign agencies, including WHO, CDC, and the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID), on smallpox virus research. One of the first grants was from WHO in the early 1990s, with which VECTOR started sequencing smallpox virus genomes in cooperation with some Moscow-based institutions. The current collaboration (with CDC and USAMRIID) involves daily e-mail exchanges and sometimes direct television connections.
-
International collaborations have allowed the rapid development
-
of new and improved Enzyme-Linked Immunosorbent Assay (ELISA) diagnostic test kits for the detection of disease markers.
-
International collaborations have led to the sequencing of the genomes of many viruses endemic in Russia, including orthopoxviruses and current isolates of hantaviruses, viral hepatitis, and Congo-Crimean hemorrhagic fever virus. This sequencing information will be helpful in the development of new diagnostic kits and initiation of the development of recombinant vaccines.
-
VECTOR has participated in international efforts to develop methods for the rapid detection of multidrug-resistant TB. The Novosibirsk region has one of the largest prevalences of TB in the Russian Federation and a high percentage of multidrug-resistant TB.
-
VECTOR has participated in about 30 training courses on good manufacturing practices (GMP) and good labor practices (GLP) to improve the quality of production of pharmaceuticals, diagnostic kits, and vaccines.
-
Finally, VECTOR was the second Russian institution to register its ethical committee with NIH. This move has helped the center take an active role in introducing ethical principles and share its knowledge with medical clinics and other institutions in the region.
VECTOR has many current and proposed goals and activities, including:
-
Study of the prevalence and the properties of pathogens that represent either a natural or a potential bioterrorist threat to public health and animal husbandry in the Asian part of Russia and neighboring countries.
-
Continued laboratory and diagnostic support for the identification and localization of infectious disease outbreaks.
-
Continued development of a new generation of techniques for the diagnosis, prevention, and treatment of infectious diseases in both humans and animals, which could also be used in the event of a bioterrorist attack.
-
Continued participation in improving both national and international biosafety regulations and developing local epidemiological response plans.
-
Continued development of information technology–assisted means of predicting infectious disease outbreaks and identifying emerging human and animal pathogens.
-
Creation and operation of a research and educational complex for the improvement of professionally skilled experts in the fields of biosafety, epidemiology, GMP, GLP, and good control practices.
-
In the event that the proposed International Center in VECTOR is created, a long-term strategic collaboration that is far less subject to the political and economic fluctuations of member states.
The Need for a New Public Health Paradigm in Russia4
Despite the achievements of VECTOR and its participation in various international collaborative efforts, an enormous amount of work remains to be done in Russia with regard to strengthening public health capacity. Of the four sectors that will play key roles in shaping the next stage of post-Soviet public health reform—private enterprise, civil society, the state, and international players—the state must play the central role in breaking the vicious cycle of poverty, inequality, disease, premature death, and depopulation that currently plagues Russia. The capacity of private enterprise remains compromised in Russia, and civil society is still underdeveloped. International partnerships, such as those sustained by VECTOR, will be vital to the state’s success.
To capitalize on the international partnership opportunities made available by globalization, however, it will be crucial to overcome the gap between the reality and the image of post-Soviet changes in Russia within both the Russian political and international arenas. Within the former arena, the Russian dialogue on globalization has been inadequate and has been centered on accession to WTO and globalization’s impact on specific economic interests. The effect of globalization on health is not mentioned in many official documents, including those of the Russian Ministry of Health, or in a number of other important, recently released government documents. In the international arena, public health and globalization issues should be considered for inclusion in the activities of the Russian–U.S. intergovernmental commission on health, and major international organizations and foreign governments implementing programs in Russia must direct more attention toward the social and public health aspects of their proposed changes. Programs based on irrelevant standards and developed for other types of societies at different stages and levels of development are unsustainable in Russia and collapse when external support and funding are exhausted.
A participant noted that the Cooperative Threat Reduction program, the scientific cooperation between former Soviet scientists and their western counterparts that was established for the purpose of fighting infectious disease and sharing knowledge, is in considerable jeopardy at the moment. The Bush Administration can no longer certify that Russia is committed to honoring and abiding by the terms of the Biological Weapons Convention and, as a result, will no longer request money for new projects or additional spending on ongoing cooperation projects on which approximately $350
|
4 |
See also Appendix D. |
million has already been spent. This situation will continue until either the U.S. Congress provides the administration with a waiver of the certification requirement or Russia becomes more forthcoming about its previous activities during the Soviet era and permits greater transparency into its ongoing biological research programs. This is a very worrisome development.
A Public Health “Bank”
Because limited global public health resources must be used in a cost-effective and conservative manner, the only way the public health sector will be able to respond effectively to the increasing globalization of infectious disease is by pooling the resources of all participants. If this cannot be accomplished, individual countries, especially those in the developing world, will never be able to afford everything they need to manage increasingly globalized infectious diseases, nor will public health as an institution be able to protect its credibility. A workshop participant introduced as a possible solution establishing a public health “bank” to pool resources that could be shared across borders.
Although the majority of public health tasks are accomplished by the private sector and the amount of work done in the public sector is small by comparison, the public sector has a unique credibility. Public health is called upon when problems cannot be readily solved or when unexpected outbreaks overwhelm either short- or long-term capacities. Its credibility has been earned because the public health system has maintained timely, effective responses and good disease prevention policies and practices.
To sustain this credibility, however, public health needs a critical mass of resources, including sufficient technology, staff, and capital, to continue to provide effective services and products. Technology must keep pace with the rapid changes that continually occur, staff are needed to maintain expertise over time, and capital is required to sustain the services and products provided through economic ups and downs. It is virtually impossible to maintain this critical mass of resources on an individual statewide or even countrywide basis. Collaborative efforts may be the best if not the only solution.
In this light, a participant suggested that public health adopt a market-like architecture that would be operable on a global level and would allow the movement of resources across political barriers. The establishment of some type of public health “currency” would facilitate the sharing of resources and provide a conduit through which the developed and developing worlds could exchange experiences and information. When public health professionals learn how to manage problems in the developed world, where they have enormous resources at their disposal and are exposed to relatively few infectious diseases, it is difficult for them to attain the proficiency
necessary to deal adequately with problems in the developing world, which has few resources and an enormous infectious disease burden.
Moreover, a market-like architecture would make use of the extensive capabilities and expertise of state public health laboratories, which today often are not being fully utilized. Members would contribute their assets to the bank and make them accessible on demand; a global health broker, such as WHO or some newly created organization, would manage the enterprise. A daily, ongoing, collaborative commitment would be necessary. For example, even though case rates of TB are minuscule in the United States, TB testing and intensive high-technology diagnostics are routine nationwide. This represents an enormous U.S. public health resource waiting to be used in a more meaningful way.
The relationship between the Massachusetts state public health laboratory and clinics throughout Peru can serve as a model for this market-like architecture. As noted earlier, the Massachusetts state public health laboratory receives on a daily basis sputum specimens and isolates that have been collected from Peruvian clinics for the detection of Mycobacterium tuberculosis, which causes TB. After the isolates in the samples have been tested for drug resistance, the results are reported back to practitioners in Peru, again on a daily basis. By using specimens from Peru that are much more difficult to assess than those to which they would otherwise be exposed, the Massachusetts laboratory workers are benefiting from a specific 4-week training program. Thus, the capacity of the Massachusetts laboratory is being developed while it is simultaneously providing real-time testing for drug-resistant TB in Peruvian patients. That the bank is mutually beneficial to all partners is an important selling point to political leaders (Timperi, 2002).
Other participants expressed interest in this notion of a global public health resource bank, even though it is unclear how the bank would operate. One participant suggested that a bank of this sort might be a useful construct for establishing bidirectional training programs in infectious disease control.
DRUG AND VACCINE ACCESS AND DELIVERY IN THE DEVELOPING WORLD5
Vaccines and antiretroviral agents have enormous proven public health benefits and could potentially save millions of lives in the developing world. Yet while globalization should be making it more feasible to bridge the gap in their access and delivery between the developed and
developing worlds—for example, by increasing technology transfer—many obstacles remain.
Barriers to Implementing Vaccination Programs in the Developing World
Vaccine programs are one of the most successful tools for addressing public health concerns in an equitable manner. The history of global vaccination initiatives—particularly the Children’s Vaccine Initiative (CVI) and the Global Alliance for Vaccines and Immunizations (GAVI)—suggests potential areas of improvement for the global public health system.
Before 1974 and the eradication of smallpox, routine vaccination for childhood diseases was implemented in less than 5 percent of the global birth cohort and in very few developing countries. An expanded immunization program, which initially included six antigens—those for diphtheria, pertussis, and tetanus; oral poliovirus; Mycobacterium bovis BCG (bacillus Calmette-Guérin), and measles—and subsequently yellow fever was launched in the mid-1970s.
A decade later, in 1985, the IOM report Vaccine Supply and Innovation (IOM, 1985) led to the rapid expansion of NIH research and the development of new vaccines by the U.S. military. In 1990, UNICEF introduced universal childhood immunization, which achieved about 80 percent coverage throughout the world. During the same year, those efforts led to the World Summit of Children and the launch of CVI, whose charge was to accelerate the development and deployment of vaccines globally.
The global history of vaccine usage over the past two decades, however, reveals a growing gap between developing and developed countries in the numbers of vaccines that have been introduced successfully (see Figure 3-2). Although countries have been encouraged during this time to recognize the value of immunization for infectious disease control, most countries have faced operational and financial constraints.
GAVI was formed in 2000 in response to this situation; its financial arm, the Global Vaccine Fund, was initially capitalized by the Bill and Melinda Gates Foundation with more than $750 million. This vaccine purchase fund was designed to serve the 78 poorest countries, that is, countries with a gross national product (GNP) under $1,000. Other barriers to implementing vaccines on a global basis besides per-dosage cost were not taken into account.
To help identify such barriers, a study on routine hepatitis B vaccination worldwide was conducted in the late 1990s. It was found that, despite a recommendation by WHO that all countries introduce universal vaccination against hepatitis B, in 1998 only about 75 of 179 countries or territories with populations of more than 150,000 were actually administering the vaccine. This is a vaccine that costs 50 cents per dose and prevents a
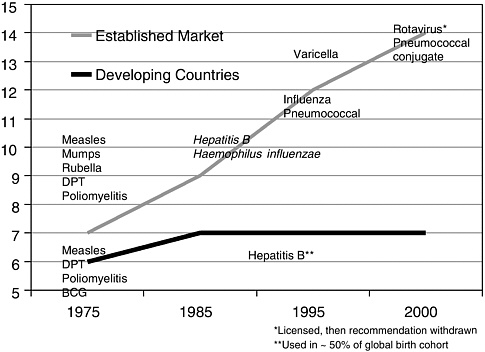
FIGURE 3-2 Number of vaccines used routinely in developing and developed countries.
SOURCE: Miller (2002).
bloodborne, sexually transmitted disease that leaves about 20 percent of those infected carriers for life. Yet it was not being used (Miller, 2002).
The study revealed that the two most important predictors of whether a country would adopt the hepatitis B vaccine were its coverage rates for other vaccines and the cost of the vaccine relative to per capita Gross Domestic Product (GDP). Also, countries that spent more money on treatment costs were five times as likely to adopt the vaccine, and those with a greater disease burden, as measured in years of life lost, were more likely to adopt than those with a lesser burden. When these factors were incorporated into a logistic regression model that was then applied to other vaccines, such as that for Haemophilus influenzae type B, the model achieved 87 percent accuracy in predicting which countries would be most likely to adopt new vaccines and in what order. It is interesting to note that these countries were not always those GAVI had targeted; that is, they did not necessarily have a per capita GNP below $1,000. Rather, with few exceptions, they were the countries with the worst infrastructures.
Since its inception, GAVI has recognized that infrastructure is an im-
portant, albeit complex, issue that must be addressed. How the needed infrastructure should be built, however, and what role GAVI can play must be determined. Numerous challenges exist, including the following:
-
Translation or adaptation of new public health interventions—The implementation of any type of health intervention in a new setting must take local conditions into account.
-
Technology transfer—Building infrastructure requires understanding the technical limitations of the setting and the need to use locally appropriate technologies. Recently, for example, large-scale campaigns have been carried out in response to outbreaks of meningitis that have ravaged the sub-Saharan African region; many of these campaigns have relied on 50-dose vials of vaccine with five syringes. One can imagine the number of iatrogenic transmissions of bloodborne pathogens that result from such a program.
-
Political commitment—Building local infrastructure requires political commitment and a certain level of civil society and governance to oversee the effort. Often, even though United Nations (UN) countries’ organizations and ministries of health are involved, there is no involvement at the national level. It may be necessary to approach the problem at a much lower level of government or society.
-
Human resources—The ongoing lack of human resources and appreciation for local health interventions is another key constraint. To attain GAVI’s goal of vaccination coverage rates in excess of 80 or 90 percent, there must be personnel on the ground. Although an epidemiologist can be parachuted in to meet critical, short-term needs, such an approach does not address long-term infrastructural needs and the sustainability of personnel at the local level.
-
Economic barriers—The creation of the Global Fund to Fight AIDS, Tuberculosis, and Malaria (Global Fund) and the contributions of new global public health partners will help alleviate economic barriers to building infrastructure.
-
Oversight—Some type of evaluation process that is not self-serving will be needed to achieve long-term accountability.
-
Commitments of private suppliers—Vaccination programs rely on public–private partnerships and commitments from private suppliers, which in turn expect credible demands. As a result of the sudden ramp-up of GAVI, critical vaccines are in short supply. Without credible demands that are backed financially, serious problems will be encountered. For example, even though whole-cell pertussis vaccines are still being promoted in much of the developing world, soon no company may be manufacturing them.
-
Absorptive capacity—In the rush to achieve desirable short-term outcomes, local programs tend to be scaled up through increased humani-
-
tarian assistance. The sudden introduction of billions of dollars into these systems, however, raises the question of how rapidly this money can really be spent wisely. Although these efforts may be well intentioned, they often exceed the absorptive capacity at the local level and lead to what is termed “programmatic iatrogenesis,” in which the cure is worse than the problem (Miller, 2002). For example, although it was a noble goal, the introduction of a malaria eradication program promoted both vector and parasite resistance (when, for example, local farmers started using DDT for purposes other than malaria control). As another example, a lack of long-term planning, combined with a lack of local recognition of unsafe drug injection practices and the need for appropriate technology, has raised many issues regarding schistosomiasis control in Egypt. Finally, expanded vaccination efforts, including eradication programs, raise questions about whether vaccination should cease following eradication. Economically, it makes sense to push for eradication efforts. Once an agent has been eradicated, poor countries have no incentive to continue vaccinating. If the agent comes back in one or two generations, however, hundreds of millions of people around the world will be susceptible to the disease.
Several steps must be taken to address the above challenges:
-
Governance—Many countries do not have a functional ministry of health or planning that can adequately oversee health programs. Therefore, other levels of government or even local academic institutions or nongovernmental organizations will need to take on this role.
-
Appropriate policy for local needs—It is critical that policy be directed toward local needs. To this end, FIC intiated the Disease Control Priorities Project (DCPP) in response to a 1993 World Bank Development Report. Currently DCPP’s partners include the World Bank, WHO, FIC, and the National Library of Medicine (NLM); they examine disease control priorities in terms of both intervention needs and science and technology capabilities.
-
Economic barriers—Long-term planning and a long-term financial commitment based on science, not politics, are crucial. The new influx of global funds directed toward international public health is a step in the right direction (see Chapter 4). However, there is some question as to how long the Gates Foundation, for example, and other large foundations are going to remain committed to GAVI and other programs.
-
Human resources—FIC recently undertook a study to demonstrate to WHO and its Commission on Macroeconomics and Health that human resource capacity is as much a global good as is product development. Indeed, product development in the absence of personnel at the local level who can use the product is only half the job. FIC has several programs in
-
place to address this issue. Most recently, for example, to reverse the brain drain, the Global Health Research Initiative Program for New Foreign Investigators has been providing grants for foreign nationals trained in the United States to return to their home countries (see the discussion earlier in this chapter). Ultimately, one of the greatest hopes for the future lies in training foreign nationals who then assume prominent positions within the governments of their own countries.
-
Appropriate technologies—The implementation of successful vaccine programs requires not only improvements to appropriate technologies, but also assurance that adequate funding for these technologies and a commitment to their proper use are directed specifically at resource-poor populations. Public–private partnerships are important to this end, but unless additional support and incentives are provided specifically for communities with few resources, this will be a difficult barrier to overcome. For example, although safer equipment for vaccine delivery may not be a priority in the United States, where safe injection practices are already in place, it may be critical for resource-poor populations.
-
Understanding of local incentives—Infectious diseases and infectious disease control can be understood only within the larger social science or anthropological context (see Chapter 4 for additional discussion of the social context of epidemiology). This context includes local incentives to implement vaccination programs.
Access to and Delivery of Antiretroviral Agents in the Developing World
Antiretroviral therapy is currently the only tool available for mitigating the economic and social destabilization due to HIV/AIDS in sub-Saharan Africa, a situation that may spread to India and other parts of the world. This is the case despite the tremendous resources that have been expended on development of an HIV vaccine. Despite its promise, however, the introduction of antiretroviral therapy into resource-limited settings poses many challenges. Of primary concern is the emergence of drug-resistant virus (IOM, 2005).
It is expected that over the course of the next 10 years, 30–50 million Africans will die from HIV/AIDS. Although poor health and death do not necessarily lead to political instability, they do maintain the economics of poverty. The continuously decreasing life expectancy in the region will progressively affect both the industrial workforce and the family unit, leading to the destabilization of society. As many who have worked in sub-Saharan Africa have witnessed, orphans are already raising orphans, and fathers have lost the ability to train their children in many agricultural skills. In the absence of intervention, this destabilization will worsen and, as noted, may spread to other regions.
From a foreign policy perspective on global health, antiretroviral therapy has several goals:
-
To reduce the impact of the HIV/AIDS pandemic on declining life expectancies in the countries affected.
-
To reverse the tragic trend of the increasing number of orphans whose parents have died from HIV-related causes.
-
To reverse the trend of the loss of key members of the workforce, including local industrial workers and teachers.
-
To minimize the impact of the pandemic on local governments and global economics.
-
To avert the development of economic and political instability in sub-Saharan Africa and the spread of this instability to other regions of the world.
Lessons to Be Learned from Antiretroviral Use in the United States
The introduction of antiretroviral therapy in the United States has had a major, positive impact on mortality (death from advanced HIV infection) and morbidity (conditions secondary to HIV infection), but it has also raised many important issues. First, disparities in access to health care have made it more difficult to implement antiretroviral therapy effectively in the African American community, where a significant number of individuals are experiencing treatment failure as a result of drug resistance. Second, increased morbidity and mortality have occurred secondary to drug-induced toxicities. Third, the rate of transmission of drug-resistant virus has increased. Finally, there have been significant shifts in consensus recommendations on how antiretroviral therapy should be used. To further complicate matters, there are currently 18 antiretroviral drugs available, but they are not interchangeable; each has its own profile that makes it more or less suitable for use in resource-limited settings.
Both in the United States and in Europe, antiretroviral therapy has been associated with a high rate of treatment failure in the first year (e.g., 63 percent of the patients in a Baltimore clinic, 53 percent of the patients in a Cleveland clinic, 50 percent of the patients in San Francisco General Hospital, and 40 percent of the patients in an Amsterdam clinic). A key factor contributing to such high failure rates in the first year of therapy is impatience for progress and the premature introduction of new treatments into the HIV-infected population. Before antiretroviral agents were introduced into the United States in 1985, there existed a lack of significant public health debate regarding consensus recommendations for use of the drugs, a poor understanding of the goals of treatment for the prevention of HIV replication in vivo, and a lack of debate about the premature use of anti-
retroviral therapy and its effect on the future ability to control the epidemic. The wide-scale application of consensus treatment guidelines occurred before the effectiveness of several drug therapies—zidovudine [AZT] monotherapy, sequential monotherapy, and combination therapy with two nucleoside analogues and one protease inhibitor—had been validated by clinical data. For these therapies, particularly those involving protease inhibitors, the pharmacokinetics were pushed beyond their capacities. Furthermore, the lack of approval or preapproval of drugs as a result of the findings of drug–drug interaction studies resulted in significant alterations in pharmacokinetic interactions. This, in turn, led to the use of subtherapeutic doses and the development of drug resistance.
The requirement for strict adherence to the treatment regimen for these drugs has also contributed to viral resistance, an especially important point to keep in mind when the drugs are introduced into resource-limited settings. The drugs should be used in the context of directly observed therapy and in the presence of treatment helpers, mother’s helpers, or treatment support systems. According to one study, even 95 percent adherence (e.g., one or fewer missed pills per week in a 21-pill-a-week regimen) can lead to a 20 percent failure rate (Paterson et al., 1999), while 90 percent adherence (missing two or fewer pills per week) can lead to a 50 percent failure rate.
Because of drug resistance, antiretroviral treatments have since their inception evolved from the initial AZT monotherapy to combination therapy to salvage therapy. Between 30 and 60 percent of all patients receiving treatment in the United States are infected with a drug-resistant HIV strain; approximately 10–15 percent of patients are infected with strains with cross-resistance to at least three classes of drugs and are on deep salvage therapy because their resistance to virtually all antiretroviral drugs leaves them no other treatment options; and about 14 percent of new infections are caused by drug-resistant virus. The negative repercussions of the premature introduction of antiretrovirals must be kept in mind when these drugs are made available in resource-limited areas.
In addition to drug resistance, other constraints on the use of combination antiretroviral therapy in resource-limited areas of the world include toxicity and side effects, drug–drug interactions, issues of adherence to and the complexities of the regimens, a lack of long-term efficacy in real-life settings, a loss of HIV-specific immunity, and cost. Thus there are real questions about the feasibility of antiretroviral therapy for most people in the world in need of treatment.
The Need for an African Solution
There is growing awareness, both within and outside of Africa, of the need for an African solution for HIV/AIDS. A number of African leaders
are interested in the potential of antiretroviral therapy, largely because they are concerned about the survival of their democracies. The Group of Eight (G8) countries are also becoming more aware of the problem; the Global Fund, for example, was developed specifically to provide assistance for the treatment of AIDS, along with TB and malaria.
In fact, now that HIV/AIDS has become a national security and foreign policy issue for the developed world and the resources for intervention are no longer limited to traditional international health programs, this is an opportune time for the introduction of antiretroviral agents into Africa. Moreover, antiretroviral therapy has become more feasible from a clinical standpoint. Three or four very potent reverse transcriptase regimens with low pill burdens are available, as are some excellent drugs with good potencies that can be administered once a day, some with high mutation thresholds. Cost reduction efforts by the pharmaceutical industry and potential contributions from the Global Fund also enhance the feasibility of introducing the widespread use of antiretroviral therapy into Africa.
Although the development of an African solution involving the use of antiretroviral agents must proceed aggressively, the effort must also be undertaken with great caution. The current American approach of sequential combination chemotherapy and sequential drug failure is obviously not a durable formula for slowing or reversing the economic and social destabilization caused by HIV/AIDS in Africa. If viral resistance were to emerge in sub-Saharan Africa, no amount of economic or political will be able to control the epidemic. Thus the most critical aspect of an African antiretroviral solution is to take the necessary steps to prevent the emergence of drug-resistant virus. These steps include the following:
-
A sustainable strategy for antiretroviral treatment in resource-limited settings needs to be developed, implemented, and evaluated; this strategy should be aggressive but prudent and should be different from the U.S. approach of sequential combination antiretroviral therapy. For example, one approach would be to evaluate the feasibility of alternative strategies for reducing mother-to-child transmission among pregnant and nursing women, and then expand feasible strategies to the greater adult population.
-
There is an urgent need for debate on timing antiretroviral therapy so as to avoid the premature introduction of suboptimal therapy and the resultant emergence of drug-resistant virus. The best available knowledge needs to be applied to identify the optimum regimens for wide-scale use. A number of African countries have proudly provided zidovudine-lamivudine (Combivir) to their patient populations; however, this practice should be curtailed until a more effective strategy can be developed. Nigeria has decided to use stavudine, lamivudine, and nevirapine, but again, this may
-
not be an optimal decision based on the drugs’ mutation thresholds and current understanding of how the drugs work. At the very least, the effectiveness of the drugs should be demonstrated prior to their wide-scale use by a population. Only drugs that have excellent potency profiles, high mutation thresholds, and limited toxicity profiles and to which there is limited cross-class viral resistance should be used. Use of the currently available protease inhibitors is not feasible in resource-limited areas.
-
In light of the above issues, the evaluation of alternative treatment strategies in resource-limited settings ought to be a scientific priority.
-
Directly Observed Treatment-Short Course (DOTS) is perhaps the most critical element of any antiretroviral treatment strategy, at least initially. The mutation thresholds for individuals with viral loads greater than 50 copies of RNA per milliliter of blood plasma are much higher than those for individuals with viral loads below 50 copies per milliliter, and pilot studies have shown that DOTS can achieve 100 percent suppression rates. DOTS enhances adherence; prevents drug leakage from public pharmacies into the informal sector, which will likely be a significant problem in resource-limited areas; prevents the use of counterfeit drugs; maximizes the ability to recognize early drug toxicities; decreases the need to monitor patients for their viral loads and viral resistance, given that the 100 percent suppression capacity of the regimens has been validated; and provides a support system for individuals while teaching them the skills required to maintain successful therapy.
-
Product profiles and selection criteria need to be developed for resource-limited areas. For example, one of the most important criteria as regards preventing the development of resistance is for the drugs to have high mutation thresholds.
-
Attainment of generic status for selected products should be accelerated, if necessary.
-
The development of new treatments designed for use in resource-limited settings needs to be prioritized.
-
The primary care infrastructure in Africa needs to be strengthened.
-
African health professions education and the local health professions workforce need to be improved.
-
The political will to do what is necessary over the long run, including making the priority decisions required to prevent economic and political instability, needs to be developed.
-
The operational capacity of the Global Fund needs to be enhanced.
Whatever action is taken in sub-Saharan Africa will have historic consequences; indeed, it will shape the world of the twenty-first century. If the approach used in sub-Saharan Africa is successful, it could be extended to other regions, with broad economic and political consequences around the
world. If it is unsuccessful, the AIDS epidemic will continue to widen the health and economic gaps between the developed and developing worlds and exacerbate the growing political instability in Africa and beyond.
Antiretroviral Therapy as a Global Public Good
Some of the issues addressed during the discussion of antiretroviral therapy in Africa raised questions about drug development and drug delivery in general. Given the complexities and implications of the economics of the pharmaceutical and vaccine industries, it was suggested that an in-depth discussion of this important topic be reserved for a separate workshop or other venue involving economists, among others. Nonetheless, some participants made a few comments on the subject, which are summarized below.
The terrible inequities and disparities that characterize the HIV/AIDS crisis in Africa have led many people to argue that antiretroviral therapy should be considered a global public good, and that as such, the drugs should be provided free of charge to all who need them. There is concern, however, that identifying antiretroviral agents as a global public good would create a situation whereby pharmaceutical companies would begin undergoing the same adverse selection phenomenon currently being experienced by health insurance plans in the United States.6 Public health interventions that are cost-effective for the public are not a source of profit for manufacturers. As people turned to companies that provided free antiretroviral products, the companies might consider those products a wasted investment and decide to abandon their production. At the workshop, this situation was compared to the pressure that has been exerted on vaccine companies to provide vaccines for only a few cents per dose. In the 1980s, there were about 20 vaccine manufacturers; today there are only four major manufacturers, and only two of these are in the United States. Neglecting to
consider the market conditions for vaccine products may drive even these few remaining vaccine manufacturers out of business.
The same situation applies to companies that sell products for both the prevention and treatment of the same disease. If it is more cost-effective, from a public health standpoint, to take preventive measures for a given disease, these companies will make more money by developing and producing preventive products and will have less incentive to develop and produce the corresponding treatment products.
Finally, it was noted that when these issues are discussed, people often start thinking immediately about the need for new drugs. However, the 23 percent rate of implementation of DOTS noted several times during the workshop illustrates that, at least in the case of multidrug-resistant TB, the main problem is not the production of new drugs but the implementation of the therapy. Even if the pharmaceutical industry produces a new anti-TB drug every year, multidrug-resistant TB is going to continue to emerge worldwide unless the implementation of DOTS is improved. As anti-HIV therapies are introduced into countries, it must be kept in mind that for many reasons, there may be fewer innovative efforts to develop new therapies in the future. The same is true of antibiotics. Thus it is crucial that the first steps taken be the right ones.
OPPORTUNITIES FOR AND OBSTACLES TO GLOBAL SURVEILLANCE7
Effective in-country surveillance systems are one of the many reasons smallpox was and remains eradicated.8 As noted throughout this report, however, emerging infectious diseases are no longer geographically contained; therefore, effective in-country surveillance systems alone cannot prevent and control their global spread. A global surveillance system is necessary to better protect against infectious diseases through a collaborative response.
An effective global surveillance and response system has four key components: accurate surveillance, appropriate information dissemination, a
reasonable international response, and adequate enforcement by multilateral agencies. Ideally, clinical identification of an infectious disease in the field would be followed by diagnostic laboratory support, and finally diagnostic confirmation by a regional, national, or international laboratory, such as WHO, which would also establish an international strategy appropriate for the situation. The international response would involve WHO or another agency that would disseminate the information and coordinate the support. Nations would then institute their own control procedures on the basis of their national policies, international regulations, WTO policies, and, if necessary, Codex Alimentarius Commission food standards. Eventually, the outbreak would be controlled with minimal international spread and disruption of trade.
However, there are several impediments to the establishment of such a global surveillance and response system. Perhaps most important, regional and local health officials sometimes face strong political pressure to suppress outbreak reports so as to protect the country from the potentially damaging economic and political costs that often ensue. These costs stem in part from the lack of dissemination of information or the dissemination of inappropriate information by health officials and the media, as well as the limited diagnostic, treatment, and prevention capacities available in developing countries. The media are often prone to sensationalism when reporting on outbreaks. Other nations may issue travel advisories or impose trade sanctions, even when such measures are unnecessary, in an effort to protect their populations, public confidence, political interests, and industries. Indeed, the measures taken all too often are far more stringent than is necessary or appropriate.
The 1994 outbreak of plague in India serves as a disturbing example of what can happen when a country reports an outbreak. The outbreak, which caused 56–60 deaths, occurred in the town of Surat, which at the time was experiencing economic and environmental conditions among the worst in the country. India’s diagnostic capacity was not very good at the time, and there was some question as to whether the outbreak actually involved plague. On the basis of its assessment that the outbreak was plague, however, India responded in accordance with WHO health regulations and ensured the adequate monitoring of both people and goods. Nonetheless, when the public was informed of the outbreak, many people fled from Surat, including physicians, who were among the first to leave (after treating suspected cases and instituting control measures such as the distribution of antibiotics). Although WHO recommended that there be no restrictions on trade and travel, trade restrictions were imposed by Qatar, the United Arab Emirates, Bangladesh, Oman, Italy, Sweden, and others. Moreover, Italy, France, Germany, Canada, the United Kingdom, the United States, and other countries restricted travel to India, and more than 2.2 million
tourist trips to the country were canceled. Overall, the reported outbreak cost India an estimated $2.3 billion in lost trade and travel; the country experienced a record trade deficit in 1994.
Why was the response to India’s reporting so extreme, given that its actions were appropriate according to the recommendations of WHO, and especially given that it was unclear whether the outbreak was in fact plague? Ironically, a small, confirmed outbreak of plague occurred in the western United States at the same time.
The cholera epidemic in Peru in 1991 serves as another example of the negative repercussions of outbreak reporting. The epidemic began on the Peruvian coast and then traveled south through the Amazon basin into Brazil (even though the reported mortality rate was less than one percent, which may have been due to underreporting). Peruvians made the diagnoses, informed the public, treated suspected cases, and instituted control measures. Signs indicating that people should treat their food and water to prevent cholera were posted throughout Peru and the rest of Latin America. Despite these measures, several of Peru’s neighbors, including Bolivia, Ecuador, Chile, and Argentina, imposed trade restrictions (although the epidemic spread to Ecuador and Chile anyway). The European Union also imposed trade restrictions. Peru lost more than $700 million in trade, and Chile lost $330 million. Travel from North America and the European Union was restricted, and half of all tourists scheduled to travel to the region canceled their plans. These travel restrictions were instituted even though both CDC and WHO stated that “on no account should travel be restricted because of cholera.” The United States implemented testing procedures and placed restrictions on imported food, despite the claim of the UN Food and Agriculture Organization that “there are no documented outbreaks of cholera resulting from commercially imported food. Epidemiological data suggest that the risk from contaminated imported food is negligible.” The trade restrictions, lost tourism, and increased inspection that resulted from the reported cholera outbreak cost Peru more than $1.5 billion.
Other examples of the economic costs of outbreak reporting include the shift in coffee prices and subsequent trade-related economic losses that occurred as a result of the 1998 cholera outbreak in Ethiopia. Similarly, the 1998 Nipah virus outbreak in Malaysia, which led to trade and travel restrictions, as well as the loss of the local swine production–related infrastructure, cost the country hundreds of millions of dollars. The current situation with regard to tourism in the Caribbean is another good example of the tension between reporting an outbreak and withholding information to avert economic loss. Because tourism is such a significant industry in the Caribbean—more than 50 million people visit the region each year, many of whom arrive on massive cruise ships—the economic impact of outbreak
reporting would likely be quite significant. For this and other reasons, much remains to be done to develop an effective regional surveillance system in the Caribbean.
Anecdotal evidence suggests that many countries suppress the reporting of information on outbreaks. Pakistan, for example, has experienced many cholera outbreaks over the past 35 years, none of which have been reported. HIV/AIDS is commonly underreported or not reported at all in many countries, while known plague outbreaks in Africa and elsewhere have gone unreported as well. This is not surprising, as there is no reason to expect that a farmer raising livestock in Argentina, for example, would let it be known that his cattle are dying from what appears to be bovine spongiform encephalopathy.
To encourage the rapid and accurate reporting of outbreaks and to strengthen global surveillance and response capacities, three main challenges must be met:
-
The economic and political costs of outbreak reporting must be minimized:
-
Governments and trade associations must be better educated.
-
WTO and WHO need to be proactive in preventing inappropriate economic responses.
-
Economic aid should be granted to countries that are treated unfairly.
-
Mechanisms for the rapid redress of inappropriate responses need to be developed. Decision makers and policy makers need to be aware that if such a situation is not resolved quickly, individuals may lose their livelihoods through the loss of their perishable products.
-
-
The dissemination of inaccurate information must be remedied:
-
Transmitting accurate information early in an outbreak is critical to a prompt and appropriate response. Press releases and other reporting information (e.g., from WHO, CDC, national health organizations, and nongovernmental organizations) need to be reliable and credible.
-
The media (television, print, and radio, as well as the Internet and the World Wide Web) need to be better educated with regard to their role in outbreak reporting, and it must be determined who is responsible (e.g., the government or schools of public health) for educating those who are doing the reporting. Reporters need to be educated in the principles of surveillance, the true threat of outbreaks, the importance of transmitting accurate information, and the impact of stigmatization and sensationalism. The last point is especially important: too often the victim is blamed, and too often the victim is a poor person in a poor country.
-
-
The limited assistance available for diagnostic, treatment, and prevention capabilities must be rectified. The ability to diagnose, treat, and prevent outbreaks locally must be strengthened by improving the training of local field staff; strengthening national and local laboratory capacities; supporting research on inexpensive, easy-to-use detection methods and equipment; and increasing the transfer of existing biotechnology and genomics. For example, the Sustainable Sciences Institute in San Francisco, California, teaches investigators throughout Latin American about simple, inexpensive polymerase chain reaction techniques. Such programs need to be expanded and further supported, and the transfer of biotechnology and genomics needs to increase. Countries must be empowered to make diagnoses themselves rather than depending on CDC and others. Such dependency is not sustainable and does not give countries the capacity to deal with future outbreaks.
In addition to meeting the above challenges, workshop participants suggested a few other steps that could be taken to improve global surveillance. For example, clinical microbiology laboratories, which lie at the foundation of the U.S. laboratory response network and are the recipients of some of the recent biodefense funding, could be considered potential sources of surveillance data. Thus even if the tens of thousands of tests conducted in these laboratories each day failed to yield data that could be used for research purposes, they could be used to save lives and treat patients. Quality assurance tests could be used to monitor the data as necessary. In fact, global surveillance efforts would benefit from tighter links to all components of the clinical arena, including health care providers and physicians.
Because of the increasing opportunities for animal–human disease transmission resulting from globalization, a participant suggested that programs be established to encourage and facilitate the exchange of information between the animal and human disease surveillance communities. This information exchange would be of great benefit for disease detection, at least in the United States. Foot-and-mouth disease, for example, could be introduced into the United States with frightening ease, especially if it were done intentionally. Rapid detection, containment, and eradication would require shared intelligence between the animal and human disease surveillance communities.
Obstacles to Regional Surveillance: The Caribbean Region as a Case Study
Development of the PAHO-administered Caribbean Surveillance System (CARISURV), based at the Caribbean Epidemiology Center in Port of Spain, Trinidad and Tobago, represents an attempt to regionalize infectious
disease surveillance in the Caribbean. However, the system faces multiple challenges, both financial and organizational. For example, very little money is being spent on dengue surveillance, even though recent economic analyses suggest that the economic impact of dengue in the Caribbean exceeds that of the common cold. Perhaps one of the greatest challenges is that the Caribbean, like many developing regions, is a large area with diverse characteristics. It comprises about 35 countries whose people speak six different languages and have allegiances to different previous colonial powers.
Moreover, although most nations in the region are characterized as middle-income developing countries where public health and other problems are generally not as acute as in poorer countries, there are exceptions. Haiti’s annual per capita GDP is about $700, the lowest in the hemisphere; other nations, such as Honduras, are also quite poor. Even among the so-called middle-income Caribbean countries, most governments lack sufficient resources to implement the public health measures that are considered their primary responsibility and for which they generally do not receive assistance from the private sector.
The region has very limited human resources devoted to the reporting of infectious diseases, and the statistics produced are notoriously unreliable. In most countries in the region, only a single person is responsible for reporting all infectious diseases, including HIV/AIDS. Laboratory capacity and technology support are also underdeveloped. This situation poses a great challenge for the region’s foreign collaborators, including the United States. The capacity to conduct U.S. Food and Drug Administration– certified clinical trials for HIV vaccines in places such as Jamaica and Trinidad is limited by the fact that very few laboratories meet the criteria of the certifying agencies in the United States.
Strengthening of the regional surveillance capacity will require, among other things, the establishment of long-lasting training programs of the type described earlier in this chapter. Because there is a very limited number of trained personnel in the Caribbean—a situation typical of most countries in the developing world—training is extremely important for strengthening not just surveillance capacity, but also public health capacity in general.
Surveillance Success Stories
Although most observers would acknowledge the considerable challenges to meeting the need for an effective global surveillance system, there are some success stories that serve as good models for what can be achieved. The successes of polio and influenza surveillance efforts, for example, are attributed primarily to clear-cut goals and rewards (eradication, improved health for all). When attempts are made to enforce surveillance in places where there
is no clear benefit—for example, where reporting creates a significant economic backlash—the challenges can be more difficult to overcome.
It was also pointed out that although the 1994 plague outbreak in India had a devastating economic impact, illustrating the negative global reaction that can result from sensationalized media reporting, few people are aware that India experienced another, more recent plague outbreak that was handled quietly, professionally, and appropriately. There was very little backlash in the form of international sanctions and the like. The pathogen was recognized, isolated, and confirmed within Indian laboratories using modern, state-of-the art resources; there was no need for international assistance. This success was due largely to extended collaborations with CDC and others aimed at improving India’s surveillance and response capabilities.
INFORMATION AND COMMUNICATIONS TECHNOLOGY9
Workshop participants emphasized the potential of the Internet and the World Wide Web to change the way the work of public health is conducted, enabling enormous strides in the ability to understand, track, and fight infectious diseases. ProMED, e-mail, electronic chat rooms, the Global Outbreak Alert and Response Network (GOARN), and the Global Atlas of Infectious Diseases are just a few of the ways the Internet is being exploited for infectious disease control on a global scale. ProMED, for example, is a global public health information network that scans English, French, and Spanish media worldwide for reports of infectious disease outbreaks. Although not all ProMED reports are accurate, they allow investigations to begin more rapidly. Furthermore, ProMED reports are more public than country reports, and in many countries the press has a better surveillance capacity than the government. Reports can be obtained either by accessing the ProMED website or by subscribing to receive regular e-mails. (GOARN and the Global Atlas of Infectious Diseases are discussed below.)
Because the Internet and other communications technologies can be used to raise general awareness of the fundamental problems of global health, a participant suggested that they could also be used as leverage to ensure that those who can alleviate problems remain accountable—for example, governments that fail to provide resources for the Global Fund. Thus far, the groups that have used the Internet most effectively for such leverage have been targeting pharmaceutical companies, not governments.
Finally, although not discussed in detail during the workshop, telemedi-
cine was mentioned as a new, potentially very powerful tool. Telemedicine is the use of telecommunications technologies to deliver medical information and services when the provider and patient are separated by distance. It was suggested, for example, that recent advances in telemedicine may provide innovative solutions for improved screening of migrants. (See Chapter 2 for a discussion of migrant health.)
The Global Outbreak Alert and Response Network
In 2000, because the new International Health Regulations (IHRs) were not expected to be finalized and approved for another couple of years, WHO developed GOARN as the framework for some of the organizational activities necessary to be able to respond to urgent public health problems on a global level. GOARN is a technical partnership of 110 institutions and networks that work together to mobilize and pool resources for outbreak alerts and responses. Its purpose is to contain outbreaks by rapidly identifying, verifying, and communicating threats; to deliver appropriate technical assistance to affected areas; and to contribute to long-term outbreak preparedness.
GOARN is coordinated by a WHO operational team in Geneva, Switzerland, which works closely with WHO’s six regional offices, 141 country offices, and liaison offices. The network’s outbreak event management system has four main components: intelligence, verification, response, and follow-up. Information is gathered from various sources, both formal (e.g., WHO laboratory networks and regional and subregional networks) and informal (e.g., nongovernmental organizations and the media). The information is then verified by WHO offices and the countries involved, after which a risk assessment is conducted and the appropriate response determined. The appropriate response could include an investigation, preventive measures, case management, and/or the provision of information to the public.
Much (39 percent) of the information gathered by the network is transmitted to WHO from the Global Public Health Intelligence Network in cooperation with Health Canada. This real-time early-warning system scans more than a million Internet websites daily for news and other reports of infectious disease events. Other sources of intelligence include WHO (33 percent); ProMED (six percent); and various other sources, such as laboratories and nongovernmental organizations (22 percent).
To avoid the spread of rumors, information is not made available to the public until after it has been verified. Verification is a systematic process by which WHO confirms the occurrence of an outbreak, its etiology, and the need for assistance in an affected state(s). The network monitors only events of potential international public concern. The criteria used to determine
whether an event is of international public concern include an unknown etiology, unexpectedly high morbidity or mortality rates, the need for assistance, the potential effect on international travel and trade, the current status of the outbreak relative to the IHRs, and any suspicion that the event was caused by an intentionally released biological agent.
Between March 2001 and March 2002, GOARN verified 195 events, most of which occurred in Africa (47 percent); some occurred in Asia and a very few in the Americas. The geographic distribution of the events reflects in part the quality of the public health infrastructure in these regions and the capacity to identify the events and respond appropriately at the national level.
The response component of GOARN involves providing rapid, appropriate, and effective assistance to the affected state(s); ensuring a level of response geared to the needs of the affected state(s); meeting daily to verify new intelligence and coordinate any responses; providing a field presence and coordination when needed; and maintaining systematic information management. From 1998 to 2001 there were more than 20 WHO-facilitated epidemic response missions, for which WHO was only one of several response partners. CDC, for example, was involved in more than half of these missions. All responses are tracked in a computerized outbreak event management system. WHO is currently developing a secure website for the network at which network members will be able to find timely information about ongoing responses.
Global Atlas of Infectious Diseases
The Global Atlas of Infectious Diseases is intended to be a web-based information and interactive mapping system that will support the global surveillance of infectious diseases. It will be used to provide access to infectious disease data and related information; standardized analyses of diseases, including maps, charts, and tables; a standardized dissemination mechanism; and an interactive data entry system for all partners.
At the website, one can find information about a disease in any particular area of the world or even a certain region within a larger geographic area. For HIV/AIDS, for example, one could select the African region of WHO and then find places where sentinel surveillance for HIV/AIDS was conducted for pregnant women in 1998. One could then select a particular country and locate the different sentinel sites within that country, or find a report identifying the sites by name. One could also develop charts that would enable comparisons across sites or over time, as well as link to other sources of information, such as epidemiology fact sheets for particular diseases.
REFERENCES
Barry M. 2002 (April 16). Considering the Resources and Capacity for the Response. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Cash R. 2002 (April 16). Impediments to Global Surveillance and Open Reporting of Infectious Diseases. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Cleghorn F. 2002 (April 16). Considering the Resources and Capacity for the Response. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Corber S. 2002 (April 16). A Response to Shifting Threats. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Demin A. 2002 (April 16). Social Aspects of Public Health Challenges in a Period of Globalization: The Case of Russia. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Gardner P. 2002 (April 17). New Directions in Capacity Building. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Gotuzzo E. 2002 (April 17). The Global Application of Tools, Technology, and Knowledge to Counter the Consequences of Infectious Diseases: A Discussion of Priorities and Options. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Gupta AR, Wells CK, Horwitz RI, Bia FJ, Barry M. 1999. The International Health Program: The fifteen-year experience with Yale University’s Internal Medicine Residency Program. Am J Trop Med Hyg 61(6):1019–1023.
Henderson DA. 1998. Smallpox eradication—A cold war story. In: World Health Forum. Vol. 19. Baltimore, MD: Johns Hopkins School of Hygiene and Public Health. P. 113.
IOM (Institute of Medicine). 1985. Vaccine Supply and Innovation. Health Promotion and Disease Prevention. Washington, DC: National Academy Press.
IOM. 1987. The U.S. Capacity to Address Tropical Infectious Disease Problems. Report by a steering committee of the Board on Science and Technology for International Development. Washington, DC: National Academy Press.
IOM. 2005. Scaling Up Treatment for the Global AIDS Pandemic. Report by the Board on Global Health. Washington, DC: The National Academies Press.
Klaucke D. 2002. Globalization and Health: A Framework for Analysis and Action. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
LeDuc J. 2002 (April 17). The Global Application of Tools, Technology, and Knowledge to Counter the Consequences of Infectious Diseases: A Discussion of Priorities and Options. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Miller M. 2002 (April 17). Considerations for Shaping the Agenda. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Netesov S. 2002 (April 17). The Current Situation and Perspectives of International Collaboration in the Field of Biomedical Sciences: The Example of the State Research Center of Virology and Biotechnology, VECTOR. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Paterson D, Swindels S, Mohr J. 1999 (Unpublished Data). How Much Adherence is Enough? A Prospective Study of Adherence to Protease Inhibitor Therapy Using MEMSCaps. Poster presented at 6th Conference on Retroviruses and Opportunistic Infections. Chicago, IL, Abstract 92.
Redfield R. 2002 (April 16). Considerations for Drug Access and Delivery in the Developing World. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.
Timperi R. 2002 (April 17). Considerations for Shaping the Agenda. Presentation at the Institute of Medicine Workshop on the Impact of Globalization on Infectious Disease Emergence and Control: Exploring the Consequences and Opportunities, Washington, D.C. Institute of Medicine Forum on Emerging Infections.