3
Morning Breakout Sessions with Invited Speakers
IMPLEMENTING THE CANCER SURVIVORSHIP CARE PLAN AND COORDINATING CARE
Moderator: Sheldon Greenfield, University of California, Irvine
Dr. Greenfield: We’re going to have each of the speakers, Deborah Schrag, Peter Bach, Phyllis Torda, and Doug Ulman, talk for about five minutes. We will leave a little time for clarification or one or two questions after each speaker, and then hopefully we will be able to have some more long-term discussion following the presentations.
Deborah Schrag, Memorial Sloan-Kettering Cancer Center
Thank you for the opportunity to present to you this morning. I would like to discuss an initiative spearheaded by Patricia Ganz and Ellen Stovall, called the Treatment Plan/Summary. It is directly related to the transition to survivorship treatment summary. Although I am a medical oncologist at Memorial Sloan-Kettering Cancer Center, in a sense, I am here representing ASCO and the NCCS on this initiative. Many of you in the room have participated in this initiative, and I welcome you to chime in. The goals of the Treatment Plan/Summary are to improve patient-physician communication; to improve coordination of care across disparate healthcare settings; to streamline the burden of documentation; and to foster standards for documentation in our increasingly digital age; and ultimately, they are to facilitate our ability to evaluate outcomes.
So, what is the proposal? Essentially, the goal is that oncologists should prepare a written treatment plan at the initiation of a course of cancer therapy. Typically, that includes chemotherapy, but it might include radiation, alternative treatment; other modalities are possible. Furthermore, oncologists should prepare a treatment summary at the end of a treatment course. The transition to survivorship care plans, which you heard so much about from Dr. Ganz and others this morning, we conceive of as a more detailed and extensive type of this particular summary. To get there, oncology professionals need to achieve consensus about the key elements for a treatment plan and summary. Professionals also need to specify standard formats that can accommodate free text, synoptic as well as fully digitized versions, that are made available to a variety of practice settings and types. And that these should adopt open source models.
What would be the key elements of a Treatment Plan/Summary? Well, the idea is not to produce 15-page informed consent documents. The goal is for something relatively succinct. Think about operative notes, pathology reports, radiation oncology summaries, which are absolutely standard. Think about the people in Hurricane Katrina, who all of a sudden had to change their sites of care quickly. The idea is not full disclosure of everything, but to provide a succinct summary. We have vetted this around quite widely, trying to achieve consensus about what those elements ought to be, and the ones that appear again and again are shown in Box 3-1.
So, what is the development strategy? How are we going forward with this? Well, I would say that having spoken about this in a number of settings, I think it is fair to say that we have achieved consensus among oncology practitioners, and I include physicians and nurses, that this is a good idea. And that if we could accomplish this and streamline the burden of documentation, it would be a good thing both for us as providers, and for our patients, and for our ability to communicate with our colleagues in other medical disciplines, dentists, teachers, all kinds of people, who may want these documents and need them.
What we are grappling with are the specifics in what those ought to look like. We are in the process of trying to develop some standard formats and templates, and are incorporating feedback we have received as part of this process. Then there will be some pilot testing, which we hope to conduct in a variety of settings, specifically through some of the NCCN institutions, and through QOPI, which is ASCO’s Quality Oncology Practice Initiative. It is a group of private practitioners who are very motivated to participate in quality improvement initiatives. And then ultimately, when we think about dissemination and implementation of this, we have to get involved with software developers and, healthcare payers to, as Dr. Ganz said earlier, make sure this activity is appropriately reimbursed. And, of course, advocacy organizations have spearheaded this effort from the very
|
BOX 3-1 Plan
Summary
|
beginning. This is something that Ellen has been talking about for years and years. So, let me stop there. Hopefully, I have time to take questions. I invite those of you who have participated in this initiative to chime in.
Dr. Greenfield: Is there a question or two?
Dr. James Talcott, Massachusetts General: When you are describing the development of critical content and achieving consensus, you have a list of people involved in the process. And it seems to be people who are trying to provide input. I wonder if it would be helpful to think a little bit about the audience for what we are trying to deliver, rather than people who have information to add to it. We talk an awful lot about patient-centered care,
and obviously the purpose of these things is to serve the interests of the patients. But we are trying to communicate to other oncologists, primary care doctors, and so on. I just wondered if you had some thoughts about the process of identifying the content from those perspectives?
Dr. Schrag: I would say there has been some very spirited discussion in various venues about the extent to which these documents should be focused on and designed specifically for patients. And as you know from informed consent documents, the standards often are that things need to be written at an eighth-grade or a tenth-grade reading level. In our discussions so far, this is not the approach we have taken. These documents are really aimed at and are to be written at the level of the primary care physician. So, they should not have a lot of “onco-speak” and jargon that would only be understood by sophisticated oncologists, with all kinds of abbreviations. They should be understood and intelligible to your typical primary care practitioner. In addition, many dentists, before doing a root canal or tooth extraction would want to know about anticoagulant therapy (e.g., Innohep [tinzaparin], coumadin), the cancer history, recent chemotherapy, and the patient’s white count. These providers also would need to understand the treatment plan/summary and have a contact to call if they saw that there was something over their head.
Patients, in looking at the treatment plan/summary ought to recognize the list of drugs that they received, for example, cytoxan and tamoxifen and bleomycin. But the goal was not to pitch it so that patients would necessarily understand every single word. I think having such a document helps fulfill the goal of fostering patient-physician communication. Some patients will look at such a document, and really not want to get involved with all the nitty-gritty issues and the details. We all have patients like that. And others will really push us and ask, “What is that drug? Why is it given this often? What are the side effects?,” and it may prompt some conversation. So, the short answer to your question is, at this point the Treatment Plan/Summary is not pitched at the patient level. It is pitched at the primary care physician level. The goals of this effort are to enhance coordination, both doctor-to-doctor and patient-to-physician. A lot of research describes just how problematic coordination is, and I think we can make patients a lot happier if we improve the doctor-to-doctor coordination.
Ms. Stovall: I think that the age of paternalism in medicine may have passed, hopefully, and with it, using such confusing terms as “intent to treat.” There has also been resistance to discussing palliative care. We have hopefully moved beyond that. And it will take institutions and leaders, people like Deborah and many of our cancer centers to lead the way, and hopefully those in oncology practice in the community will adopt and adapt
that attitude as well. We have to let patients know about their treatment and what we are really trying to do for our patients. The goals of treatment need to be communicated, and I think we have to be more honest in using these tools to communicate these goals.
Dr. Johnson: I am a surgical oncologist from St. Louis University. Have the legal ramifications of this effort been considered by any of the development entities? If you give a patient a statement of what should be done, and you omit something that might happen, are you legally liable? If you do not suggest that patients have their gated cardiac scan twice a year, and they only get it once a year, and they have a sudden death, would you be legally liable? Have these issues been discussed?
Dr. Schrag: In a preliminary way some of the legal advisors to ASCO and NCCS have taken a look at this. The legal specific disclaimer language has not been specified to the final comma, but essentially, there is a way to avoid liability issues by saying this is a summary and that it does not describe every single thing. Such a summary already exists in radiation oncology, so there is a way to provide clinicians some legal protections. This is an obstacle that will have to be encountered, but I do not think we need to get bogged down in those sort of legal details. We want to do something that we think makes sense for our patients.
We think it makes sense in terms of how I, as a medical oncologist, communicate with you as a surgical oncologist, and with my colleague Dr. Ayanian as a primary care physician. We think that it is just the right thing to do, and we think it ultimately will streamline office practice and office culture. When people call up for notes and records, we all have people who work in our practices who are very busy photocopying, and trying to figure out what to photocopy. And when it comes to your practice as a surgeon, they know what to go for. They go for the operation note, and they go for the pathology report. It is great, and it is easy. When I call your office, I get what I need in three seconds because you are a surgeon. However, when you call my office, you are likely to obtain a large stack of documents that you will have to sift through to find the four to five essential documents that you really need. And what you need may not necessarily be the first new patient note, because at that point the diagnosis and plan are not all formulated. Alternatively, you might receive a flow sheet, but that might not provide any detail regarding the reasons for underlying treatment recommendations. So, you have to spend a lot of time fishing and sifting. I presume you have had that experience. We do not want to get bogged down too much in the legal details before we have achieved consensus on what the content of this document should actually look like, but I agree that we will need to sort legal issues out.
Dr. Greenfield: I think we are going to have to move on, but I just want to say that in the diabetes field, we have encountered this problem, and we have a partial solution. I do not have time to talk about it now. We can talk about it later in the discussion. But we have gone part of the way toward addressing the problem of multiple guidelines, legal threats, and so forth. Let’s go on to Peter Bach’s presentation.
Peter Bach, Center for Medicare & Medicaid Services
Thank you. I am the senior advisor on cancer policy to the administrator at the Centers for Medicare & Medicaid Services. I want to thank you for having me here, and I am excited about reading today’s USA Today and seeing banner-like announcements of some major progress and innovation in the cancer survivor arena (Szabo, 2005). I would like to talk about our recently announced demonstration project and how it is directly relevant to some of the ideas emanating from this group. We announced Wednesday, November 2, that in 2006 hematologists and oncologists in office-based practices are going to have another opportunity like they did in 2005, to submit additional information about patients and their care to CMS using our billing system (CMS, 2005). And they will receive an additional payment for doing so.
The information we are gathering focuses on three interrelated areas. And I think this directly bears on the issue of monitoring and management of survivors and patients in an expectant mode, as well as other periods of disease surveillance. We are asking the oncologists in the context of evaluation and management visits, the visits between doctors and patients, to tell us first, the primary purpose of the visit. Dr. Schrag offhandedly mentioned a couple of things that might go on in a practice like a new diagnosic visit, or a surveillance visit, or a visit focused on counseling about potential complications. We are asking doctors to report to us, in the context of particular visits, the primary focus of that visit, with an understanding that all visits are complex and touch on many issues. We want to better understand the spectrum of cancer care, and also appropriately credit doctors for the range of what they do. We are linking the response to that question to another question we are asking them to answer, and again, answer through our billing system, which is whether or not what they are doing follows well-established and accepted practice guidelines that are evidence-based. So, when they are focusing on therapy, are they following practice guidelines, like those from NCCN or ASCO? When they are in a surveillance mode, are they following practice guidelines?
As we elaborate tighter and tighter, more evidence-based, more knowledge-based, and more clinically relevant practice guidelines, we can continue to link what doctors are doing, what they say they are doing, to
practice guidelines that are directly relevant to what they are doing; something we could do in a survivorship mode, something we could do when patients are being actively managed.
We are asking that doctors report a third piece of information, which was on Dr. Schrag’s slide as well. We are asking them to clarify the patient’s disease status. Currently, what we have in our process of billing, and we do not even have it on every patient on every visit, is the ICD-9 code, which tells us the primary cancer diagnosis in most cases. There are subtypes of ICD-9 codes that give us some discrimination in some cancers. But we do not really know within the context of a visit between a doctor and a patient, and a treatment plan, whether or not doctors are following guidelines, or what the extent of a patient’s disease is. For example, patients could be newly diagnosed, they could have a recurrence, they could have new metastatic disease, or they could be in a period where they have no evidence of disease. We now do not have any way of determining disease status through our claims system. Without this information on disease status, we can not understand what is actually going on when the doctors are saying, well, this is the focus of the visit, and these are the guidelines I am following.
We are trying to move, through using our billing system, and using a demonstration project like this, to get a better understanding of what is happening in doctors’ offices, and what is happening with the care of patients across the spectrum of illness. And in a sense, we are taking a horizontal approach as was described in Dr. Schrag’s treatment summary proposal idea, because embedded in this demonstration is the entire treatment history and treatment course for a patient. That is a quick summary of the demonstration. It pays $23 per event reported, somewhat lower that the 2005 demonstration, which paid $130 per event, but was linked to chemotherapy. Those of you who are in practice know that there are many more evaluation management events with patients, or there certainly should be, than there are chemotherapy events. So, the math works out slightly differently. That is a quick summary of the demonstration. I am happy to take questions.
Dr. Ganz: Peter, if we wanted to develop a mechanism for having a visit that would focus on the survivorship care plan visit, which would be linked obviously to an end-of-treatment summary of some sort, would there be some potential for a demonstration that would create that visit, or could be coded as a specialized visit in that way? Could you see something like that possibly happening in the future?
Dr. Bach: Maybe. This is an issue that we have pondered. We are wrestling with these issues. We compare the practices of radiation oncology to medical oncology providers, and ask if there should be a dedicated visit for treatment planning in medical oncology, because there is an analogous one
in radiation oncology. But that, of course, is not the full story of how we compensate the two professional groups in providing different kinds of services. We have also considered special visit categories. There are a handful of them in Medicare, for example, we have a “Welcome to Medicare” visit where you get to go to the doctor even if there is nothing wrong with you, which we do not really have in other places. My personal view is that the mechanism for getting these sorts of visits in the system is to embed them in the current evaluation management strategy, but also within something like this year’s demonstration project, which we hope will be something that continues over time, and we can continue to have a new focus of the visit added, linked to a relevant guideline. This is again where I see we could go. We have a suite of five possible foci for visits in the 2006 demonstration. I could easily see either going to another one, or embedding this in a current category, and linking to a set of relevant guidelines. It is probably easier than creating a separate code. It is challenging to create separate codes for different specialties. I am a pulmonologist, not an oncologist and we do things that may justify separate codes as well. So, this approach is challenging. I would rather redefine the spectrum of what it is that oncologists and primary care providers do.
Joan McClure, National Comprehensive Cancer Network (NCCN): I have been looking at your document and have some questions about how one would code partially concordant care. For instance, the NCCN guidelines, in the work-up phase, frequently call for a wide variety of tests. Sometimes a physician will choose to do many, but not all of the tests. And it was just something that kind of jumped out at me, and I wondered if you thought about it?
Dr. Bach: We have, and by December 1, we will have specific—I do not want to use the word “rules”—but algorithms that oncologists will be able to use to compare what they are doing to the guidelines, and to determine whether or not they are following the guidelines. Obviously, the NCCN and ASCO guidelines are quite elaborate, and are multidimensional. It would not be reasonable to either apply a standard that says that unless you do every single thing listed in the next 30 pages, you are not following the guidelines. Not only is that not reasonable, we will not learn anything of value. So, we are actually going through the guidelines and identifying those areas where we say this constitutes concordant care, and this does not, for the purpose of learning what we want to learn from this demonstration project.
Dr. Greenfield: As Phyllis comes up, let me ask you, Peter, on the third topic in terms of extent, stage, and so forth, are you going to provide specifications for the patient classification?
Dr. Bach: Of course. Those are coming. We did not publish those with the fact sheet, in part, because it was 16 pages long, and they are disease-specific categories that map appropriately to the guidelines, and have been defined by the medical oncology specialty community. But they will be far too broad to finally discriminate between patients the way we do as clinicians, but sufficient to at least stratify. The average cancer has four categories.
Phyllis Torda, National Committee for Quality Assurance (NCQA)
Good afternoon. I am Vice President for Product Development at NCQA, which means that I have responsibility for taking what we know about how to measure quality, and wrapping it into programs on an ongoing basis to improve quality of care, or in Patti’s words this morning, routinizing quality care and making it part of ongoing practice. That is really what we are all about. I have been at NCQA doing this for 10 years. Prior to that, I worked for two different consumer advocacy organizations, and I am also a relatively recent cancer survivor myself. In putting together my remarks this morning, I tried to meld my personal thoughts as a cancer survivor, with what I have learned professionally about how to measure quality. And on a personal basis, and thinking about the cancer survivorship plan, I want one. Why didn’t I get one? Why can’t I have one tomorrow? And I consider myself a very informed consumer. Professionally, I know what the barriers are, so in my remarks I am going to try to summarize a little bit about what we know about how to put one foot in front of the other, and make progress in this area.
We would all like to think about the systematic practice as described in Figure 3-1 that very much builds on the chronic care model that Patti talked about earlier. The systematic inputs of information that the physician has include such things as medical evidence, patient data, customized reminders, and self-management resources. These inputs facilitate an ongoing partnership for health with the patient. Systematic outputs of this model include care management and quality improvement. That is the vision.
Findings from a 2003 survey of U.S. physicians conducted by the Commonwealth Fund summarized here show how short of that vision we are today (Audet et al., 2005):
-
85% of physicians can’t generate registry lists by test results or current medications
-
33% of physicians repeat tests because results are unavailable
-
15% of physicians observed abnormal test results not followed up
-
Only 18% of physicians have data on patients’ outcomes
-
Only 13% of physicians can generate their own performance measures
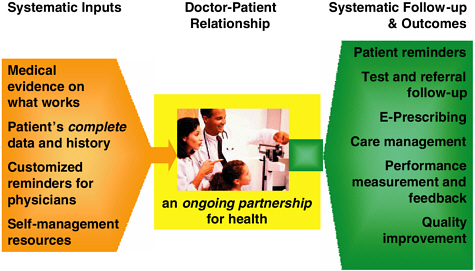
FIGURE 3-1 The systemic practice.
What do we know? We know that measurement leads to improvement. Figure 3-2 shows data from a program that the NCQA operates along with the American Diabetes Association in which physicians apply for what we call recognition in diabetes. On each of the six measures, cohorts of physician applicants have improved their performance over the three years of
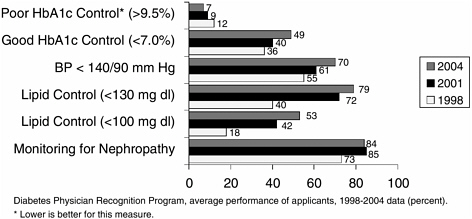
FIGURE 3-2 Measurement leads to improvement: Physicians achieving “Diabetes Physician Recognition” show substantial improvement in key clinical measures.
FIGURE 3-3 Health plans and employers use recognition information.
measurement, 1998, 2001, and 2004. So, we know that measurement leads to improvement.
We also know that if we had programs that define excellence in care, that we can get payers—payers being health plans, self-insured employers, potentially Medicare—to use the information. Figure 3-3 shows the various ways that we have been focusing on working with payers to get them to feature the outputs or the lists of recognized physicians that we have developed in our programs. And those ways range from on the left, just featuring the information in provider directories, to helping the practices collect the data, to paying monetary rewards, to using the information to construct products which could be narrow networks or differential co-pays. So, we can get people to use the information in ways that matter.
I tried to think about the vehicles that we could use to begin to implement the cancer survivorship care plan. The NCQA has a program known as Physician Practice Connections. It was created for a group of self-insured employers known as Bridges to Excellence. The program is about systematic practice and about how practices use information in a systematic way to provide care. So, just beginning with that program, and looking over the requirements, I tried to think about where the cancer survivorship plan fit in? There is a requirement in our newest version, which we are just finalizing, that requires practices to have a procedure in place, a process in place for identifying their high-risk patients (Figure 3-4).

FIGURE 3-4 Physician practice connections.
So, in terms of where the survivorship care plan would come in, I think it would have to start at identifying high-risk patients. If identified as high-risk, physicians caring for cancer survivors in primary care practices would have to think about what needs to be done to care for these high-risk patients in an ongoing way. So, that is one vehicle.
Another vehicle that Deborah Schrag mentioned earlier is the ASCO QOPI program, which evaluates medical oncology practices according to measures in the categories of care shown in Box 3-2. I agree with Deborah that the QOPI program could be expanded to include requirements relating to practices taking an active role in developing the survivorship care plan. This would begin to get at routinizing it, as Patti mentioned earlier.
In thinking about implementation issues, we have discussed a program for medical oncologists whereby they document their care and recommendations. We need to discuss further the role of the primary care physician. If the patient comes in to see his or her primary care physician, and the patient has been identified as a high-risk patient, does the physician say, “Let me see your survivorship care plan?” Does the primary care physician then have any responsibility for reminding the patient of their need for a follow-up referral visit, of tracking those, of doing anything in that regard? Does the primary care physician have a responsibility to think about what self-management resources they can provide to a patient based on their survivorship care plan?
|
BOX 3-2
|
Likewise, what is the specialist’s responsibility on an ongoing basis? They develop the plan. If you miss a visit, does the specialist have a responsibility to contact the patient, and say, “You missed a visit” at least once? What does it mean beyond preparing the initial plan?
And then of course, what is the survivor’s responsibility? I think to have effective implementation, all of these roles need to be thought through and delineated, and delineated as a partnership where each party understands their own contribution.
And with that I’ll stop and take any immediate questions, or let us move on.
Dr. Jacobs: I am the Director of the Cancer Survivorship Program at the University of Pennsylvania Abramson Cancer Center which has been in existence for about five years. I think this is an incredibly good plan. We do notes, and actually a care plan of sorts that we do send the providers after we see patients. So, we do something very similar to what you are proposing here. And we send it to providers, and we give it to patients if they want one. Many patients say they do not want one. I think there are huge obstacles to the plan. That is not to say that it should not happen, because I think it should. But I am wondering what you have thought in terms of obstacles posed by enormous differences in the way in which people document. Some providers do not document at all, or they barely document just in long-hand, what they need to for insurance purposes, but they do not necessarily dictate notes. I am talking about what happens at our institution. Maybe your experiences are different. And just among 10 people who are caring for the same population of patients, you could have such broad differences.
In addition to that, communicating with say primary care providers, we feel very strongly that many of our patients or most of our patients should be following up with their primary care provider after we have seen them, and we have evaluated them, and provided this care plan. But then we get from patients that they do not want to do that. They want to come back to the oncologist. They do not feel comfortable going elsewhere for care. And then we have the oncologists who do not want to let go of their patients; who want their patients to come back to them. So, it is an enormous problem that we have been encountering for the last few years, and I think that we do need to educate. My point here is to emphasize the need to educate providers, patients, the subspecialists, as well as the primary care providers. I do not really know how that can be done.
Dr. Greenfield: Let’s have Phyllis respond to that, and maybe if we have time for questions and answers, other people can respond to that as well.
Dr. Torda: I am sure everybody in this room has thought about the obstacles as much as I have. I think what I am suggesting is beginning to make roles very concrete, to embody them in standardized forms, and to have some measurement programs that incorporate carrying out those responsibilities, and measuring against them, and making those results public in some way or another as a way to move forward. There are lots of barriers.
Dr. Greenfield: Okay, let’s go on to Doug Ulman.
Doug Ulman, Lance Armstrong Foundation
Thank you very much. I am the Director of Survivorship at the Lance Armstrong Foundation, and a three-time cancer survivor. It is a privilege to be here, and I want to thank the IOM and ASCO for having me, and for the wonderful report. We are obviously very committed to all of the recommendations, and know that it is imperative that this be an ongoing dialogue that is actually fueled and turned into action.
I want to talk about two issues in terms of implementing the care plan recommendation. I think it is a very exciting recommendation, but also a very complicated one. So, the two things that I want to talk about briefly are empowerment and infrastructure. I think the survivors, and we have talked a little bit about it, and we have heard a little bit just recently about this, but survivors need to be empowered. So, the notion that we can create a care plan and just hand it to individuals and expect that it will alleviate or reduce anxiety, or provide a higher quality of care I think is a little bit unrealistic. And so, I think we need to develop very complex educational
programs that are implemented with survivors and their families in the long-term, so that it is not just a hand off of a document or an electronic record, but yet an ongoing dialogue. And we also need to evaluate when it is appropriate to revisit that plan. Is it a year later? Is it 5 years later? Is it 10 years later?
The care obviously needs to be coordinated, and we have heard a lot about that. But I would advocate strongly that the survivor does need to understand what they have been through, and understand that care plan. And I think Linda’s point is interesting that some survivors choose not to receive that. There may be any number of reasons for that, but I think if a survivor or their family understood that there was a companion education piece or program that would help them work through and educate themselves, maybe they would be more likely to want that information, and feel like they were empowered with that information.
Just briefly, I want to give you a case study. A 19-year-old diagnosed while in college, 400 miles from home, treated at three different institutions by six different providers, returns to college and is diagnosed with two additional primary cancers. He graduates, relocates three times in the next three years, and travels more than 4,000 miles to three cities. He is now 28, sees an oncologist in two different cities in addition to a primary care physician for follow-up. That is my story in a nutshell. And I have a cardboard box filled with records and x-rays and scans and documents. On a personal level, having a survivorship care plan would be incredibly exciting for me and reduce a lot of time and energy in terms of showing up at doctors’ offices an hour early to complete information.
In terms of infrastructure, we need the guidelines and coordinated program development (to include academic centers, community practices, and primary care physicians). I think the piece about electronic systems is more interesting and exciting now. There is the Passport for Care Program on the pediatric side, which is putting the infrastructure in place to track and update guidelines automatically, so that survivors can have that electronically. I think as was mentioned earlier, the example of Hurricane Katrina, we saw that hospitals and individuals lost everything. So, the paper route, long-term, is probably not the most sustainable.
But I think that we have to empower the survivors, and then build the necessary infrastructure in order to not only implement the care plan, but also to provide quality survivorship care. So, I will stop there and take any questions.
Dr. Greenfield: Actually, as Doug sits down, let me comment before Julia poses her question. Sherry Kaplan and I have given patients guidelines in the form of algorithms, patient level, not doctor level, much oversimplified. And we found the same thing that Doug said. Many of them, maybe even
most of them, never used them. So, we are trying to create solutions. We think we found several around that. But I would also make one other comment. I am very pleased we had this discussion, because what looks like a rather simple notion, turns out to be very complex, and therefore, a lot of research and demonstrations, and if you will, a lot of good thought, have to go into bringing this about.
Dr. Rowland: I want to thank the panel for all their thoughtful commentary here. But I also want to make an impassioned plea. This is a wonderful moment. It comes back to Anna’s call for empirical studies. All of us sitting here—educated consumers—are saying we would like these reports. One example of an interactive information system for survivors of childhood cancer, the Passport for Care Program, a wonderful web-based engine that we all drool over when we see it, has already been built. But we do not know if people will use it. You can build it, but will they come to it? I do not want us to be trapped into building something that people do not want. There are important empirical questions to ask: Who does or may not want the report? What would be meaningful to them? What information would they like and when in the course of their care?
I am reminded of some of my work with Patti Ganz. We tried to educate women about what to expect as they were finishing their cancer treatment, because survivors told us, “Gee, I wish I had had that knowledge when I finished treatment.” Yet when we did approach them as they were finishing treatment, they said, “I am not ready to hear this information.” So the questions we need to ask are: When in the course of care is it best to deliver this information? How should it be delivered? How often might you have to repeat it? These are necessary first order research questions, and I would love to see us build delivery models that would allow us to answer them.
Dr. Greenfield: Do any of you want to respond to that?
Dr. Bach: I agree, but I think there is an analogous research question that you would want to address, which is what is going on in the physician community, and the culture of medical caring that creates impediments for this kind of communication? And it is certainly true that patients at different points in their discourse are ready for different kinds of information. But it is also the case that doctors behave differently during different periods of the disease course that may make patients more afraid, or choose to be less empowered than they could be were the doctor to handle the entire spectrum of their care differently. So, it is challenging for us at CMS who worry about these sorts of issues, to have people respond and say, “Well, if you just paid for this other thing, then we would do it too,” when I think the real question is “Is this
something that doctors should intrinsically be focused on doing? And shouldn’t that be a part of the culture of the entire community?”
Susan Leigh: I am going to wear my nursing hat. When patients often come in for their follow-up visit, and they see their physician, they are expecting their physician to look for disease. They are expecting them to look for what is wrong with them. And it is a really scary time for lots of people to come in, and so we lose a lot of people. We lose people to follow-up, because they do not want somebody to find something that is wrong. When they come into some nursing clinics, the nurses then say, “Well, we will do a review of the systems here. But let’s see how we can help you be well.” And we then offer different ways to modify diet, or modify exercise. And if you take the time to help people learn how to be well, and I think that is maybe what Doug was alluding to with some of the issues he was dealing with. So, I think that the way we approach the survivor for that visit is incredibly important. And I think we need to have nurses at the table along with our physician colleagues, because we are working together as a team, and very often the nurse has the time and maybe a different kind of expertise to add to the team.
Dr. Greenfield: A very good point, and I think the role of nursing actually in this report, and in more general terms, has not been stated strongly enough.
Dr. Anna Meadows: When I was the director of the Office of Cancer Survivorship, who and when an individual was considered a survivor was up for argument. Was it now, the minute of diagnosis, the point of ending therapy, or maybe some arbitrary period after? And the point that was just made is important, because until about three, four, or five years after the diagnosis, no oncologist is going to give up their patient. They are going to continue to follow them. Pediatric oncologists definitely will. Medical oncologists also will. So, the time of ending therapy or some short time after that may not be the most appropriate time. They are not ready.
We are actually doing a study in pediatrics at the Children’s Hospital now where we are talking to parents—now, admittedly this is parents of children—on their off-therapy visit and about the long-term clinical plan. And we are giving some parents just paper information, and we are having a conference with another group of parents, a randomized trial to see which of these works better, and whether this facilitates their follow-up.
Admittedly, we will not be able to answer that question for a long time, because we do not refer those patients to our follow-up clinic until they are two years off therapy at least, and five years from diagnosis. So, they do not see the survivorship program until then. But we want to see whether this
promotes their coming to the survivorship program and getting all that information. So, that is a small research study, but as always, children will lead the way.
Kym Martin: I am a new member of the National Coalition for Cancer Survivorship. I am a pediatric childhood cancer survivor, a 22-year Hodgkin’s survivor. To Julia’s point about really taking a look at what the patients/survivors would like in their treatment plan, and when they would like it, yesterday I had a visit with a new oncologist here in the area. I am new to Maryland and like Doug, 22 years of records have followed me around various parts of the country. And to sit down and talk about where I have been, and where I need to go, it was difficult for me to actually go back and review my records again before I had to meet with him. And I am an advocate. I am all for empowering survivors, and I am all for having the information.
I think one of the things that we should also be focusing on is bringing the family of survivors into the fold, because if patients are not ready, at least a family member can act as an adjunct for their care, and kind of keep them on track. So, from my personal experience even though you may think you are ready, you may not be as a survivor. So, let’s bring the other survivors that we recognize as family members, and get them on board and find out from their perspective too, how they would like to help the patients if they are not able.
Dr. Greenfield: Thank you for that. I want to ask Phyllis to respond to this, and I will lead into her response. In other diseases there have been ways found to deal with the timing problem, basically which is to say you have a whole year, or some period to do this. In other words, you do not have to do it on the first visit. That is what Linda was talking about, or maybe the second. You can make up a specification, or something like that, that says within a year you will have at least had to offer a survivorship care plan. That would be a quality measure, something like this. Do you have any more comments about that?
Phyllis Torda: Well, certainly there is an art to measurement, and Rodger talked about it a little bit earlier this morning too. Conceptually you need to get agreement on what you want to do, and where the disagreements are. And then you can usually figure out how to design a measure that will be better than nothing, and will be good for a start.
I would just like to comment briefly on the need for research. It is very important to not just assess how people feel in a static way, but to also look at “what ifs.” So, can you set expectations that after a course of active treatment, there will be an appointment with a medical oncologist? And the medical oncologist will go over what the care plan is for the future, so you
are not just again, looking at things in a static mode, but how can you help people to move along.
Kevin Stein, American Cancer Society: I would like to thank you for some excellent talks. I want to build on something mentioned earlier and that is that I feel that this treatment plan should be more multidisciplinary, because just doctors talking to doctors is not going to include the full spectrum of issues that survivors have. In fact, many issues are psychosocial in nature. And you are going to make referrals to pain management specialists, to psychologists, to psychiatrists, and to social workers. And a lot of those individuals are going to be involved in these patients’ care, especially as they move away from the acute phase where the physical symptoms are important, and the psychological symptoms of cancer-specific distress, things of that nature, become more important. So, we need to include these people in this treatment plan, and make it more comprehensive. And that includes not just the other treatment providers, but also building on what Kym Martin said about the family, and taking into consideration the individual differences, the cultural, the ethnic background of these individuals, and how that impacts on how they are going to engage in the follow-up care.
Dr. Greenfield: Thank you for the comment. Any responses to that? Are there other questions?
Diana Jeffery, National Cancer Institute, Office of Cancer Survivorship: As I am listening, I am thinking of lots of research questions. But the number one question I have is, if this was instituted, whether in demonstration projects or a national kind of effort, would there be a difference at the end of the day or the end of the year, or at the end of 5 years or 10 years? And what would that difference be? And what would those indicators be? What facets of quality of life would be considered success? And who would pick those indicators? I am hoping it is not the provider. I am hoping it is the survivor.
Dr. Greenfield: But even that is a researchable question. For example, you might hypothesize that treatment of certain late side effects could lead to a reduction of symptoms and improvement in function. There are multiple endpoints and I think the point you are making is we have to decide what those endpoints are.
Dr. Schrag: I think there are lots of ways to operationalize that question. One obvious thing is to make sure that cancer survivors go on and get the care that they need. That is, that they are appropriately assessed and receive screening mammograms and cholesterol checks. And if they are Hodgkin’s
survivors that there is special attention paid to breast cancer. So, I think we already have a set of quality metrics. And Rodger Winn and many other people in this room are working hard on figuring out what those quality metrics should be. But I think the other thing is that increasingly the science of measurement is really a more complicated and sophisticated construct than patient satisfaction. But I point to things like the Picker Institute measures. John Ayanian and others of you in this room have used them very successfully. These are based on patient surveys. They are more often focused on inpatient care. The American public still trust their doctors, they like their doctors, they think their doctors are doing the right things. Where we fall down, more doctors than nurses, but nurses too, is on communication and coordination. We have got countless surveys and countless disciplines that show that. I think we need to do before and after assessments, and use some of those metrics to see whether this stuff makes a difference.
And what I would say to the NCI folks is “You put out the RFAs (requests for applications), and let those of us who are out there respond to them.” And we will try, and be sure to help us, because I would say, having tried to do this research, and Mary McCabe who is in the audience can probably speak to it better than I, when I think about doing these kinds of research projects, a lot of it is pilot work.
Let’s try this and see whether it makes a difference. And we want to be nimble about it. And we have to decide whether we are going to do this as operations research, sort of quick and dirty. Let’s try it, measure the impact, regroup, revise, and do it again. Or, consider whether we are going to do it in the context of Institutional Review Board (IRB)-approved research. And IRB-approved research, whether you are at a big place or a small place, it just takes six months to get it through an IRB. By the time you have written it, and approved it, and vetted it, and then the patient has to sign a very scary looking form. And that is a tremendous obstacle to us being nimble. So, what I would say to our NCI colleagues is “Help us be nimble.” We would love to do this research. Help us intervene with IRBs to explain that maybe this does not really need six months of an IRB protocol, that this area may fall under the area of operations research.
Dr. Greenfield: Let me respond to what Deborah said. In many places, what is called quality-of-care research is either exempt or it is expedited. So, we and others have built a lot of pilot work on the backs of that. But your plea has got to be heeded.
Dr. Antonio Wolff: We clinical trialists have implemented, and I think it is working very well, the concept of a central IRB. We are talking about low-risk research. We are not talking about giving poisons to people. This model may be applicable in the survivorship context. The other issue is that
resources are needed in this area. I was wondering about the beautiful video that was produced to accompany the IOM report (IOM, 2006a). It was very moving for me to watch, and I am very curious to hear what is going to happen with that, how widespread is it going to be disseminated?
We are all preaching to the converted here. And when we have congressional representatives and others saying we cannot shake up the status quo, those are the people that need to be talked to so that the NCI and others will have the money they need for research.
Dr. Greenfield: Let’s have Ellen respond to that with the final word.
Ms. Stovall: Well, the NCCS has prevailed upon the Institute of Medicine to give us enough copies of both the DVD and the report so that it will be distributed to our friends on Capitol Hill.
Dr. Greenfield: And thank you very much, and we thank the panelists for their comments.
BUILDING BRIDGES BETWEEN ONCOLOGY AND PRIMARY CARE PROVIDERS
Moderator: Steven Woolf, Virginia Commonwealth University
The topic that we are going to focus on now is this notion of building bridges between primary care and survivorship care. Our first speaker is Kevin Oeffinger. Kevin is a family physician and medical director, as of July, for the Living Beyond Cancer Program at the Memorial Sloan-Kettering Cancer Center.
Kevin Oeffinger, Memorial Sloan-Kettering Cancer Center
Thank you so much, Steve. Let me begin by providing a rationale for why bridging oncologists and primary care physicians is important. First, we will talk about the study by Craig Earle and associates where they looked at 15,000 five-year survivors of colorectal cancer (Earle and Neville, 2004). They looked at the type of health care that survivors had received and the type of healthcare provider delivering that care. When you look at the acute care, preventive care, or overall care, the survivors that had a primary care physician and an oncologist were most likely to have recommended care (Figure 3-5).
This finding of better care associated with both specialty and primary care is not limited to cancer. In a study of over 35,000 patients who had had a myocardial infarction, when investigators followed up and looked at the two-year mortality rate, patients that were followed by both a generalist
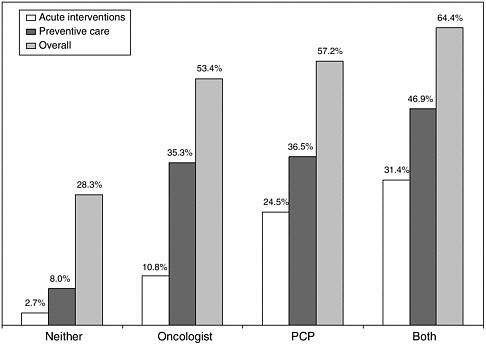
FIGURE 3-5 Percent of patients receiving recommended care by type of provider. PCP=primary care physicians.
SOURCE: Earle and Neville, 2004.
and a cardiologist within three months after hospital discharge had a lower mortality rate (Figure 3-6).
There are a number of other studies that have shown that patients have improved outcomes when they are followed by both a specialist and primary care physician for a chronic disease. What I would like to emphasize though, is that most of the studies have focused on either the simple presence or absence of a healthcare provider, and have not systematically looked at whether there was shared care.
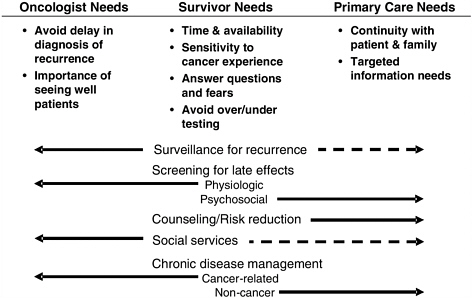
I have illustrated in Figure 3-7 the distinct needs of oncologists, survivors, and primary care providers. We must acknowledge that survivors need physicians with time to devote to them, some knowledge of the dynamics of the cancer experience, and someone to answer questions and address fears. Are my symptoms the results of my cancer experience? Is this shortness of breath something that is a sequela of my cancer therapy? In other words, how to evaluate that patient with a perspective of what their cancer therapy was.
From the oncologist’s point of view, if there is a recurrence, there will not be a delay in diagnosis given their expertise in surveillance. And importantly, seeing survivors in an oncology practice also provides a psychological balance in a setting where death is a frequent occurrence.
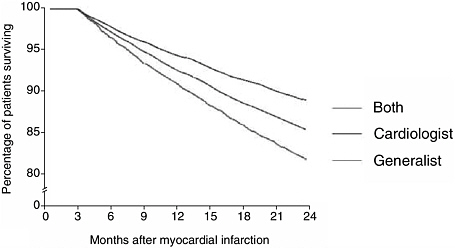
FIGURE 3-6 Patient surviving after myocardial infarction by type of provider.
SOURCE: Ayanian et al., 2002.
Primary care physicians have a need for continuity with their patients, not only after cancer diagnosis, but through cancer therapy, and then on to survivorship. We need targeted information to understand how cancer chemotherapy or radiation affects the survivor’s long-term health.
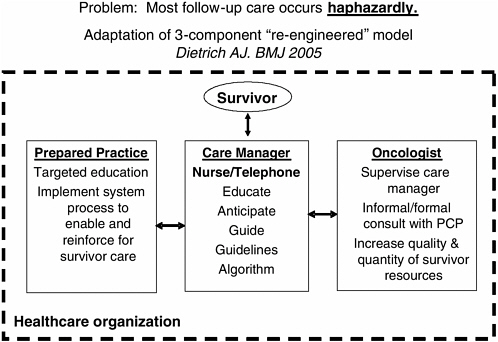
So, if we look at the different ways that oncologists or primary care physicians might fill this role, we recognize that both are important and needed in this model. The problem that was highlighted earlier is that most care in the United States is provided in a haphazard fashion. So, what I am showing in Figure 3-8 is an adaptation of a three-component “reengineered” model proposed by Dietrich that has been shown to be effective in reducing poor outcomes with patients with chronic disease.
In this model, a care manager, usually a nurse, educates, anticipates, and guides the cancer survivors by telephone using guidelines and algorithms. The oncologist provides supervision for the care manager, consults with the primary care physician, and takes the lead to increase the quality and the quantity of survivorship resources. In some primary care practices, education is provided not only for clinicians, but also for the office staff in order to implement processes that not only enable, but reinforce the quality of survivor care.
The electronic health record has a key role to play in promoting survivorship care. The technology is currently available and the cancer care team can create an electronic health record and then populate the record with key elements of the cancer survivorship plan outlined earlier. Guidance for the primary care physician regarding possible late effects, recommendations
for screening, and a problem and medication list can become part of a shared record. Through the use of simple computer software you can translate this into lay terms. And survivors, through a HIPAA compliant web-based interface, can access this information, deposit new information if they are monitoring symptoms or signs, and communicate in an asynchronous fashion with the cancer team. Very importantly, there is a physician portal where a primary care physician can access this information, contribute outcome data, including information on any meeting with the cancer team and the survivor. This system unifies the survivor, cancer team, and primary care provider and provides an opportunity for communication. It enhances outcomes data, and it empowers the survivor.
I would like to address research as it provides an important bridge between the work of oncology and primary care. One of the success stories of the National Cancer Institute in the last 20 years has been the development of the Community Clinical Oncology Program (CCOP), which encourages cancer centers to engage in community-based cancer research, in the areas of both cancer care and cancer control. There are currently about 4,000 physicians, primarily community oncologists, participating in 61 CCOPs.
Practice-based research networks (PBRNs) are a mechanism to develop research opportunities in primary care, family medicine, internal medicine, and primary care pediatrics. Within the Federation of PBRNs there are now 65 networks, including over 6,500 physicians providing care for over 16 million patients in the United States and Canada. The PBRNs represent an opportunity to launch new research to test models of care and to conduct generalizable survivorship research. Importantly, they enhance our work by fostering a working together towards a common goal of maximizing the health of our cancer survivors.
Dr. Woolf: Thank you, Kevin. That is a helpful perspective from your experience as a family physician in a cancer center. Now, we are going to hear from Pat Legant, who is a medical oncologist in community practice in Salt Lake City, Utah.
Pat Legant, Community-Based Medical Oncologist
I have practiced medical oncology in the community for over 20 years, and I am pleased to report that I follow a great many cancer survivors. In this way, I think that my experience parallels that of most oncology specialists, in that most adult cancer survivorship care in this country is supervised by an oncology specialist who works in cooperation with a primary care provider, in some version of the model of shared care that is described in the report from the Institute of Medicine.
This model has much to recommend it, and in fact there is some evidence that patients prefer it. There are a number of advantages of the shared care model. In particular, the physician supervising the cancer care has specific expertise and experience in oncology. The primary care doctor and the oncology specialist see the patient with a different set of eyes. We need both perspectives.
The chief disadvantage of the shared care model, as cited in the IOM report, is a dwindling supply of oncology specialists in the face of an increasing population of cancer survivors. However, one can make the same comment about primary care providers in that they will be faced with the non-cancer related care of us baby boomers.
The IOM report maintains that cancer care in this country is poorly coordinated and often fragmented. This may be less a failure of the shared care model than of the way it functions. There are certain requirements for effective shared care. First, to work optimally, the model requires close cooperation among the oncology specialists, the primary care provider, and the patient. It assumes a culture of team work, in which the oncologist serves as the primary provider for the cancer-related care, and the primary physician supervises the non-cancer-related care.
Second, there must be clear communication between the oncology specialist and the primary care provider, whether by letters and faxes as now, or electronic medical records in the future. This communication must include individualized content, providing the survivor’s disease status, suggested tests for follow-up, and also management guidance for this patient specifically as opposed to cookbook recommendations. So, for example, if I have a patient on tamoxifen, I have to know who is doing the pelvic examination, because somebody needs to do it every year. Communication must also specifically address the recipient to guarantee that responsibility is passed securely. Communication must be multidirectional. Right now, most of the communication goes from the oncology specialist to the primary care provider, but it should be back and forth among all the specialists. So, for example, if my patient gets congestive heart failure, I need to be sure that it is not a late side effect of treatment or relapsed cancer, or I may simply need to assure the patient that the heart failure has nothing to do with the past cancer.
Third, the system should allow for flexibility of relative involvement of the oncology specialist and the primary provider. The baton of responsibility should pass back and forth over time, rather than just once, to reflect the variability and the unpredictability of malignancy. Involvement should also depend on the course of the cancer, the course of the co-morbidities, the risk of relapse, and the patient’s geographic and social circumstances. If I have a patient who lives in Wyoming, I will communicate extensively with the primary provider in the winter in hopes of sparing the patient a three-
hour drive to my office in the snow. There should be no artificial barriers to this kind of give and take, for example, things like insurance limits on how many times a patient can see the oncology specialist per year, or higher co-pays to see the oncologist specialist, who after all is acting as the primary provider for the cancer-related care.
Fourth, the patient should be explicitly recognized as part of the communication team. This then mandates patient education on how and what to report. If there is a new lump or back pain persists after a couple of weeks, I want the patient to call me, as well as the primary provider. The proposed treatment care summary is an excellent first step, but this is an ongoing process over a very long time.
Finally, communication and education are critical elements of optimal cancer care. Some of this can be streamlined, with checklists and electronic medical records and the like, but much of it is not delegable, and it is time intensive. Insurers must offer adequate reimbursement, or else these activities cannot happen.
The shared care model has served us well for many years, and will continue to be the predominant model in the foreseeable future. We owe it to our patients to identify the critical elements of the model and to make the model better. Improving communication within the oncology care team is one way to do this.
Dr. Woolf: Thank you, Pat. Our next speaker, Ann Partridge, is going to give us a perspective about additional special issues in thinking about this linkage. Ann is a medical oncologist at the Dana Farber Cancer Institute. She is also instructor of medicine at Harvard Medical School. Her clinical research has focused on psychosocial, adherence, and communication issues in oncology, and breast cancer in particular.
Ann Partridge, Dana-Farber Cancer Institute
Thank you. It is a true pleasure to be here today. I want to focus on two particular issues. You have all addressed today how survivorship, up until the last several years, has been something that has been somewhat of an afterthought for the vast majority, aside from the people in this room. The two issues that I want to address are viewed even more so as afterthoughts, but nevertheless very, very important. The first one, and I think we could all agree as we watched that video this morning, relates to the generally tremendous psychosocial impact of cancer on an individual. This is something that we pay attention to on some level, because sometimes you can not help but pay attention to it, because someone is crying, or because they are not coming in for treatments, because they can not deal with it. But short of that, especially during or after treatment, it is something that gets
pushed aside so you get someone through their treatment. Or people themselves push it aside so they can buckle down and get through their active treatment.
A couple of the survivors on the video talked about it today, especially early in that transition time, that is when a lot of people really reach a crisis point in terms of how they are coping with their cancer diagnosis and moving forward. What I would like to introduce today is that we not push the psychosocial issues off to the side as we agree about this care program and care plan. That we face the fact that the anxieties about the future and the psychosocial distress that comes with a cancer diagnosis is not something that goes away, and actually can become bigger when one gets through the active treatment, because they are no longer focusing on the acute side effects and toxicities and will survive. Now, they are focusing on the “what does it mean to me” questions. So, not only do we need to help our patients through a rich communication with primary care physicians and oncologists and other care providers, help them to know who to call when they have an ache or a pain, we also need to help them know where they can seek additional psychosocial support as they grapple with their diagnosis and moving forward in life.
The second point I would like to make is even further out on the trajectory. And it is the long-term follow-up of patients. And in particular, the issue of communication with patients either following a trial in a research study where they received standard care, when ultimately research tells us that those patients are at greater risk for some new disease. The perfect example is Hodgkin’s survivors, young women who received mantle radiation for Hodgkin’s disease in their teens, who are at dramatically higher risk of breast cancer 15 years down the line. In the future, after we learn more things like this, and follow our survivors better for their long-term outcomes, how are we going to communicate those risks with those survivors?
Another critical piece as we do the “hand off” or the shared care transitions is how we will make sure that primary care physicians and whoever is following these patients 15 years down the line have access to relevant cancer-related information? There are ways to build these communications in either through web sites or staying in contact in some way with an oncologist in the long-term. As you heard Dr. Ganz say today, she still sees many of her patients yearly. But I’ll bet that there are many survivors that are 15 years out from their cancer that she does not see any more. They probably go back to their primary care physicians.
Finally, we need to figure out a way that the long-term information about potential side effects from treatments survivors may have received 15 years ago is conveyed. And we can not expect that it all will make the New England Journal of Medicine, USA Today, or other outlet such that they will hear about it in the lay press. I think we have to find ways through
this burgeoning world of communication to help facilitate the transfer of that information as well. Thank you.
Dr. Woolf: Thank you, Ann. Our last speaker is Regina Benjamin, who is a family physician in Bayou La Batre, Alabama, just outside Mobile. It is a treat to have all of our speakers here, but I do not know if you realize how special it is to have Regina here. She will comment on that. Regina is going to comment about survivorship from her perspective as a practicing primary care physician.
Regina Benjamin, Bayou La Batre Rural Health Clinic
I am a family physician, and when I finished residency, I opened my practice in a little town, Bayou La Batre, Alabama. It is the furthermost village on the Gulf Coast of Alabama. They filmed the movie, “Forrest Gump,” there, if some of you saw it. It is a pretty place, but it is a poor place. I found a community of “working poor,” that is, people who are too poor to afford medical care, but too rich to qualify for Medicaid.
Much of what I was going to say to you has been said earlier in our sessions, and by the other panelists. I will just tell you which of the recommendations that I think are most important for me, and why I think they are important. The first one is number one, to raise the awareness of the needs of the cancer survivor. We have 10 million cancer survivors out there. I did not even know we had that many, and many people did not know that. And just the fact that we have that many gives us an opportunity to start to address the issues and needs there. Many of my patients will come in with an acute problem for example sinusitis or a cold, and they do not tell me they have had cancer. I only find out when I do an in-depth history and physical. Patients themselves do not think it matters as far as their acute health problem is concerned. So, raising awareness of patients will really help. They are not hiding it. It is just not a top priority.
It is the same with physicians. There is a patient I saw last week who I have been seeing for several years. I know she has had breast cancer. But until I went to do her annual pap smear and her breast exam did it occur to me, oh yes, this person has had breast cancer, and I need to start to follow-up on some things. And that is why recommendation number two is really important, that a survivorship care plan is needed. If I could put the plan on the front of the chart it would remind me of the things that I need to be looking for, the things I need to be doing, what the patient has experienced, and just continuously have a reminder. We do that for cholesterol. We do it for pap smears. We do it with mammograms. It would be a good document for us to have. I think that this recommendation for a survivorship care plan, in particular, is the most important.
Primary care providers often lose our patients to follow-up. When a patient of mine has an elevated PSA I send him to a urologist, and he is diagnosed with prostate cancer. It can be a year or two before I even get him back out of the oncology system, unless his family calls me up and says, “Do you know what those doctors want to do? Do you think they should do it or not?” Otherwise, I may lose them for a couple of years until they have completed their cancer treatment and care. The cancer survivorship plan would really open the communication between all providers, because the oncologist did not get the patient from me, they got the patient from the urologist to whom I had sent him. So, the oncologist may not even know I am in the loop. So, I think that the care plan would really help pull it together, so that when I do finally get this patient back in a year or so, I will have something to go by.
The clinical guidelines and assessment tools that are called for as part of recommendation number three, can help primary care providers manage the late effects. These could really help those of us in the boondocks, if you will, to basically know what we need to do. I am not an oncologist. I do not plan to be an oncologist, but I want to know what to look for, and I want to give my patient the best care I can. I want someone to give me some guidelines so I know what to look for, and I know when to send them back. And if my patient starts feeling fatigue or has other symptoms, I need to know what to look for, what tests I need to be doing, what laboratory tests I should conduct, and generally, how to follow up. I think survivorship guidelines would really help us as primary care physicians very much.
And the last thing that is important to me, because it is important to my entire medical career, has been access to adequate and affordable health insurance. The patients that I see work, but the majority of them do not have insurance. And when you get a diagnosis of cancer, you can often not get insurance. And if patients are uninsured, we have to find ways to advocate for them to make sure that they can always purchase insurance if it is available, and make sure that it is affordable.
Hurricane Katrina hit my town and brought us a 25 foot surge of salt water, and of the 2,300 people in the town, 2,000 have no homes. Many of my patients are cancer survivors, and they did not have insurance in the beginning. Now, unemployment has doubled, making health coverage even harder to get. And as we have discussed, stress from such difficult circumstances can manifest as physical symptoms. So, we have to be sure to identify anything that comes up, particularly with cancer.
Lastly, I would just like to say that many of us know cancer survivors who eventually lost that battle. I just want to remind us that as we talk about these issues, that we remember those folks that have contributed so much for us to get here today, and always keep them in our thoughts. They
had struggles and they survived for a while, oftentimes a long time, and then they lost that battle.
Dr. Woolf: I want to thank the speakers for achieving our goals and staying on schedule. We have a good block of time for discussion.
Dr. Susan Weiner, Children’s Cause for Cancer Advocacy: I would like to address some questions about this notion of a shared care model to the speakers. There seem to be two versions of it. In one version, there is a nurse intermediary, and in another version there is not such a nurse. I typically do not speak in public about my personal experience. I was the parent of a child with cancer, who was diagnosed as an infant, and then died when he was nearly 14. I was his case manager, of course. And one of the things about not having an intermediary in coordinating specialty care and primary care is that the burden of decision-making is on the family as to who to call and what to do. And from a parent’s perspective, I can tell you that is one of the most stressful things one can endure. But one also hears from adults, as well as parents, that one does not want to “bother” the physician. So, this is a situation in which there is no intermediary, where there is no nurse. When there is an intermediary, the risks of course are that that person may serve as an inappropriate gatekeeper. There have to be personal dynamics that work. There has to be the bond of trust. So, that is my first set of concerns about the shared care model, and things that I think have to be worked out.
The second set of challenges has to do with the notion of the culture of team work. One of the things that I have experienced, and that others have experienced as well, is that there is frequent and intense competition between caring physicians, especially if it is a rare cancer. And in some instances, if it is between institutions, it can be like the army and the navy. It is extremely difficult to manage, because one wants to have not a competitive view, but an insightful view that is empirically based or based in practice. It is tremendously difficult to sort that out from the patient’s perspective.
And then the final point about the culture of collaboration relates to patient loyalty. Will I be unfaithful? Will he be hurt if I went to see X in order to ask a question about my condition? This is a very difficult problem to face, since one does not want to bias the care that one receives for a loved one. So, I think that in looking at the shared care model, as it becomes more articulated, I am not sure that those things need to be alternatively evaluated, but they sure need to be looked at a priori.
Dr. Woolf: Those are good points. Panelists, do you want to address them, or do you have some additional comments?
Dr. Partridge: I just wanted to address the first point and the last point related to the need for an intermediary as a part of the shared care model. Based on my experience, one of the most important things, as we think about caring for the adult survivorship population, is that the volume is tremendous. And in terms of allocation of resources, there are going to have to be intermediaries to some degree to try and care for the volume and to give people the appropriate attention they need to deal with all the survivorship issues for various cancers. What is probably the best way to fold that into the long-term care of survivors is, if one has the opportunity, to build it in from the beginning. And if you work with a physician or a nurse-practitioner or a physician’s assistant or a nurse, to have the patients know that person from the beginning. Such teamwork helps with time and resource issues, and also to facilitate better care. This early introduction makes it less of an acute transition to another provider. This model, at least in our institution (Dana Farber), seems to work in terms of how we follow our patients long-term, and does not make them feel like they are seeing someone who is unfamiliar or an inappropriate gatekeeper, because they already know this gatekeeper and they are part of the team.
Dr. Woolf: Kevin, do you want to make a comment?
Dr. Oeffinger: I will make a couple of comments. Nurses are very important, as are many other providers. Survivorship care, more than probably most other areas of medicine, involves a team approach. And whether that is a nurse within the cancer team, or whether it is a nurse in the prepared primary care practice that works as an intermediary, it is going to vary from practice to practice. The other thing that I want to highlight is we used the term “shared care,” and I am going to be the devil’s advocate, and say we do not know what shared care is. We have not tested shared care. What we describe in most of the studies is “both” care. And it is truly haphazard, it is not systematic, and it is not planned. It usually is “I talk with you, you talk with me,” but we do not pay attention to those that get lost to follow-up. The biggest group that is lost to follow-up from every study, my study included, is that large group of patients that do not go to the oncologist, that do not go to the primary care physician, and nobody knows it.
Dr. Benjamin: There is also a large group out there called “caretakers.” The Rosalynn Carter Foundation focuses on rural caretakers, but there is an international organization as well. We need to make sure we involve family caretakers in any model that we intend to put together, because oftentimes they know more about how to get around the system than we do, and we can learn a lot from them. Oftentimes we do not think of these systems
issues as scientists, as physicians, or as nurses. We need to encourage utilizing the caretaker in our models as well.
Dr. Woolf: If I may use my liberty to stand outside my role as moderator and also respond. As a primary care physician, we often have this habit of looking across diseases, because we take care of multiple conditions. Some of the concerns you raised when I listened to you are specific to cancer survivorship. Many others I see encountered by other patients and families and caregivers who are dealing with chronic, ongoing, recurring, and potentially life-threatening diseases that are other than cancer. So, it might be congestive heart failure, diabetes, or Alzheimer’s. The primary care physicians and specialists work together in managing those conditions all the time. And this is the theme that I am going to come to in my talk later.
Many of the challenges that you have raised, the need for a case manager to deal with navigating the system, deciding who to go to, and whose feelings are going to be hurt, and so on, are encountered by these families as well. My point is to say that the solution to the general problems that have been raised is a system solution. That it is not necessarily a cancer care solution, but a healthcare solution.
Dr. Susan Weiner:: I agree with you entirely. I think though that there is one difference. And one encounters this difference in the pediatric community between the care of kids with special healthcare needs, and the care of kids with cancer. And that is the societal perception that cancer is like an infection. It is an acute disease. It is treated, and then it is done. And I think if anything, a big impact this report can have is to change that perception.
Dr. Woolf: Yes, that is what we intended to be the most important message of the report.
Bob Weiss, National Lymphedema Network: There is something that I do not hear here in terms of shared care that I think is very important from my own experience. And I am talking now as a patient advocate, my wife being the patient. I heard communication between the various people, but I do not hear knowledge. And in our experience—my wife is a 13-year breast cancer survivor, and she has had lymphedema, from the very beginning—the medical people that we deal with have virtually no knowledge of first, lymphedema, its pathologies, its physiology, and second, its modern treatment. And the reason for this, in my opinion, is the fact that in Europe there is in medical schools, a department of lymphology. Every medical student that goes through school in Europe is exposed to the lymphatic system, its
pathologies, and its treatment. In Europe there is also a method of treatment that was developed over the last 50 years that has been found to be very effective. The insurance companies cover the specialized treatment. In the United States it is viewed as alternative medicine, and we don’t believe in alternative medicine. That is a quote from a newly graduated MD that treated my wife for a respiratory health problem. And when I told her what the treatment of lymphedema was, she said, “Oh, we don’t believe in that. We doctors do not believe in that.” Now, there is an information gap here. My wife’s oncologist said, “The further away you get from chemotherapy, the fewer the symptoms will be.” She completely neglected the long-term effects of the radiation my wife had, which are far worse than any of the chemotherapy she had. My wife’s treatment was involved, she had 21 affected nodes and had chemotherapy, a mastectomy, hormonal treatment, radiation, she had the works. And thank God, she is still alive and still kicking. And she very often reminds me that all of the effects of chemotherapy and the radiation and everything pales in terms of her quality of life in comparison to the daily care that she has to take for lymphedema.
Dr. Woolf: Let’s get a reaction to the panel on the issue of gaps in knowledge, and how the partnership between the primary care physician and the oncologist can help deal with those gaps in understanding.
Dr. Benjamin: There is a case study in the book. I do not know if it is your wife, but it sure sounds almost just like it. We had a similar case study with lymphedema, arm swelling, and nobody diagnoses it for a while. All of a sudden it is lymphedema several years later after her breast cancer treatment. The gaps are there, and that is what we are trying to highlight, trying to bring to the attention of the scientific community, to each other that we do need to close those gaps, because they are there. And patients are the ones who will tell us.
Bob Weiss: Yes, but if you do not have the knowledge, very often the doctor, the physician, says, “Oh, your arm or your leg is swollen? Please take this diuretic I will prescribe for you. Raise your limb when it starts swelling, and go home, goodbye.” And it is stage two lymphedema. It is not going to do any good. Furthermore, the diuretic is contraindicated and makes the lymphedema worse. So, there is misinformation also given by the physicians in the case of treatment of this.
Dr. Benjamin: Recommendation number three in the IOM report is give us some clinical guidelines, some scientific, evidence-based clinical guidelines, so that we can close some of these gaps.
Dr. Partridge: There do need to be efforts based, in part, on the experiences of the Institute of Medicine and ASCO and this report, to improve the knowledge base of general oncologists and primary care physicians providing long-term survivorship care. We need to, for example, increase CME credits and access to this information. In our state, you have to have all your CME credits handed in when you want to get relicensed and some of those credits have to include risk management. At some point, some of the CME credits may have to include long-term issues for cancer, or long-term issues for something else. I hate to have to have things mandated. You can not force people to learn.
Dr. Woolf: This also circles back to Kevin’s point about the electronic health record. There will be a point in time where integrated information systems will provide prompts to the clinician about lymphedema for example, and the ability to quickly pull out current recommendations and evidence-based guidelines on how to manage that.
Dr. Diller, Dana-Farber: In response to your point about lymphedema, this may seem tangential, but recently there was an article published by Lois Travis on the risk of breast cancer in survivors of Hodgkin’s disease. And I am a pediatric oncologist with an interest in care of survivors. I got an e-mail from a primary care provider who had read the article, or read the press release, or read some lay version of it, and wrote me and said, “Should I be getting mammograms in patients who had been treated a number of years ago that I now follow in primary care?” So, in some ways, the system does work. We do research. We publish. The publication gets read, or it gets disseminated, and then that gets back to the primary care provider taking care of the patient, who then starts to think, “I have a patient like that. I may think about taking care of them a way I haven’t considered before.” So, the processes that Dr. Meadows described this morning of research driving the process in a lot of ways, the process I described is one of the most traditional ones in academic medicine, but one that sometimes ends up working.
Dr. Grunfeld: I’d like to suggest an approach to the concept of shared care, which is somewhat different. I would like to suggest that along with shared care, we focus on appropriate care, which brings in the expertise that is needed at the time that it is needed. If we look at the epidemiology chapter in this book, what really comes to my mind, and really was the impetus for the research I have done for years on this topic, is that for the major prevalent cancers, breast, colon, and prostate cancer, the majority of survivors are over 65. The majority of people have one if not multiple co-morbidities. Regina brought up the point, which we have certainly experi-
enced in Canada, which is that the diagnosis of cancer often creates a separation between patients and their primary care physician, so that the cancer treatment, diagnosis, and follow-up care become the focus of attention. If we really look at what the patient’s needs are, ultimately after the acute treatment phase, for those people who do not go on to develop recurrence, it is management of multiple co-morbidities. So, I would suggest our idea of the shared care should encompass the concept of obtaining the relevant expertise at the time that they are needed. During the diagnostic and treatment phase, absolutely, secondary/tertiary level expertise are required. But I would challenge the concept of Patti Ganz seeing patients 10, 15 years after their diagnosis, and say, “What is the real benefit for the patient? And what are the opportunity costs?” Because if Patti is seeing patients 10 or 15 years after diagnosis, what about the patients who she won’t be able to see. She will make a huge quality of care and symptom management difference if she sees them during the palliative phase.
Dr. Woolf: Did everybody in the back hear the question? Let me do my best to restate it. Over the course of cancer survivorship health needs are changing. There are co-morbidities. And at any given point in time, it might be the case that the patient’s co-morbid conditions other than cancer may be the dominant health need. And the expertise that might be required at that time might be different than say during an acute recurrence of the cancer at some point. So, the model of shared care needs to be in flux, depending on what expertise is necessary to deal with the care needs at the time. Is that fair?
Dr. Grunfeld: I think that is fair. I would just make an ancillary point to that. There were two figures that Kevin showed from administrative data on what preventive care cancer survivors get (Figures 3-5 and 3-6). This research illustrated how when cancer survivors see the family doctor, they receive recommended general preventive care, and when they see the oncologist, they receive the recommended cancer specific preventive care. I would say that both are important for the patient. And a facilitation process of guidelines should include patient responsibility. It can help achieve continuity.
Dr. Woolf: Does anyone on the panel want to respond?
Dr. Legant: In my presentation, I talked about the flexibility of involvement and was trying to get at exactly this point. During the acute treatment phase of care, to a certain degree when a medical oncologist is involved, he or she will actually take over the co-morbidity management, which is largely in the field of internal medicine. We may not do quite as good a job as the
internist, but at least we know to call somebody when we get in over our heads. But down the road a piece, you need even more give and take. And long-term, I think there are still reasons to follow people once a year. It is not very often that they will touch base with their oncologist. I hope when people do studies of survivorship care, they will look not only at whether screening tests were getting done, but also whether late relapses are picked up quickly. We also need to consider reassurance of the patient, because many patients just like that sort of connection still with their old oncologist. Which is very important just to reassure them that when their best friend down the block gets a brain mass from a breast cancer 20 years ago, that it is not going to happen to them given their circumstances.
Dr. Benjamin: I agree with your points, particularly, your point that it is not an either/or situation. It is both. And it is this team concept that we oftentimes find in medicine. If someone has a heart attack, I send them to a cardiologist. But they come back. And if a year later they start to have some chest pain and symptoms, I send them back to the cardiologist. The same thing should occur with cancer. When you need them, you need to be able to refer them back, and it should not be either/or, it should be both.
Dr. Woolf: In the interest of time, because we have to break for lunch, let me just make John’s the last question. I am sure the panel will be available afterwards.
Dr. John Ayanian: My question is in follow-up to Regina’s point. And that, as we talk about these models of shared care, I think it is going to be very important to bring the primary care physician organizations to the table in partnership with ASCO. It will not be enough for ASCO to educate its members, the American College of Physicians, the American Academy of Famliy Physicians, and the Society of General Internal Medicine. Essentially the AMA (American Medical Association) is an overarching organization that brings physicians from different disciplines and fields together. I think there is a good model for this in terms of the way the American Heart Association, the American College of Cardiology, and the American College of Physicians have worked together on evidence-based guidelines for patients with heart disease. They together bring both the primary care and specialty care perspective to the table to weigh the evidence as a group to get to what the guidelines should be, and the models of care. So, my question would be what can we do, what will it take to bring the leading organizations of primary care physicians into this process? I think many primary care physicians are just grappling with the needs of cancer survivors in the ways as best they can as individuals. But we haven’t really made this
an area of focus within the leading physician organizations. What could we do to do that to form a partnership with ASCO and other oncology groups?
Dr. Benjamin: In the AMA and the Academy of Family Physicians we could take this report and present a resolution to the house of delegates in both organizations saying exactly what you just said. And then it becomes policy in those organizations. And the leadership then would be forced to work with the leadership of ASCO. So, that is the simplest mechanism to do it. And the AMA meeting is actually going on right now. The next one would not be until June, but it certainly could be done.
Dr. Woolf: I think the release of this report is a leverage point. And I think we can galvanize momentum to take that forward at an organization level. So, I think moving quickly on this might be a good idea. Please join me in thanking our speakers.