2
Earth Processes and Human Physiology
Except for radiant energy from the sun, the resources necessary for sustaining life are derived chiefly from the near-surface portions of the land, sea, and air. Intensive utilization of earth materials has enhanced the quality of human life, especially in the developed nations. However, natural background properties and earth processes such as volcanic eruptions, as well as human activities involving the extraction, refining, and manufacturing of mineral commodities, have led to unwanted side effects such as environmental degradation and health hazards. Among the latter are airborne dusts and gases, chemical pollutants in agricultural, industrial, and residential waters, and toxic chemical species in foodstuffs and manufactured products. Of course, at appropriate levels of ingestion and assimilation, most earth materials are necessary for life, but underdoses and overdoses have adverse effects on human health and aging.
Although the environmental concentration of a substance is important and relatively easy to measure, its specific chemical form (a function of the biogeochemical environment, complex species interactions, Eh, and pH) determines the substance’s reactivity and therefore its bioaccessibility. In the case of earth materials, specific mineralogical characteristics (e.g., mineralogy, grain size) must also be considered together with these chemical factors when assessing bioaccessibility. Thus, a number of analytical measurements are required to accurately assess the bioavailability of a naturally occurring chemical and mineralogical species. For most, an optimal dose range enhances health, whereas too little (deficiency) or too much (toxicity) have adverse impacts. Because the bioavailabilities of a
spectrum of earth materials present in the environment constitute critical variables that influence human health—particularly where regional and local “hotspots” of earth material deficiency or toxicity occur—the bioavailabilities of earth materials must be quantified by collaborative, integrated geological and biomedical research. To understand the physiological responses of the human body to the ingestion and assimilation of earth materials, this chapter begins by briefly describing the dynamic geological processes responsible for the areal disposition of earth materials in the near-surface environment, with particular attention to soil characteristics. This is followed by a brief description of those aspects of human physiology that are—through their responses to bioaccessible nutrients and hazardous materials—directly responsive to the biogeochemical environment.
EARTH PROCESSES
The near-surface portions of the planet and their complex couplings with—and feedbacks from—the atmosphere, hydrosphere, and biosphere make up the interactive earth systems so crucial for life. In turn, these dynamic systems are a reflection of the origin and geological evolution of the earth in the context of solar system formation. The following brief review of earth’s deep-seated and surficial processes provides the physical context for the public health component of human interactions with the earth.
Planetary Architecture and Crustal Dynamics
The solid earth consists of a series of nested shells. The outermost thin skin, or crust, overlies a magnesium silicate-rich mantle, the largest mass of the planet. Beneath the mantle is the earth’s iron-nickel core. The terrestrial surface is unique among the planets of our solar system, possessing an atmosphere, global oceans, and both continents and ocean basins. Incoming sunlight powers oceanic-atmospheric circulation. Solar energy absorbance and transfer mechanisms are responsible for the terrestrial climate and its variations, as well as for cyclonic storms and coastal flooding. In the solid portions of the planet, the escape of buried heat through mantle flow has produced the earth’s crust, as well as energy and mineral deposits and all terrestrial substances necessary for life in the biosphere.
Although imperceptible to humans without geophysical monitoring, continuous differential vertical and horizontal motions characterize the earth’s crust. This remarkable mobility explains the growth and persistence of long-lived, high-standing continents and the relative youth of low-lying ocean basins, although the former are being planed down by
erosion and the latter are being filled through sedimentary deposition. The earth’s surface is continuously being reworked, and a dynamic equilibrium has been established between competing agents of crustal erosion and deposition (external processes) versus crustal construction (an internal process). Crustal deformation, a consequence of mantle dynamics, is the ultimate cause of many geological hazards, including earthquakes and tsunamis, volcanic eruptions, and landslides. In addition to the direct fatalities and injuries, natural catastrophes result in the displacement of surviving populations into unhealthy environments where communicable diseases can—and often do—spread widely.
Plate Tectonics—Origins of Continental and Ocean Crust
Scientists have studied the on-land geology of the earth for more than two centuries, and much is known concerning the diverse origins of the continental crust, its structure, and constituent rocks and minerals (see Earth Materials below). Within the past 35 years, marine research has elucidated the bathymetry, structure, and physicochemical nature of the oceanic crust, and as a result we have a considerably improved appreciation of the manner in which various parts of the earth have evolved with time. A startling product of this work was the realization that, beneath the relatively stiff outer rind of the planet (the lithosphere), portions of the more ductile mantle (the asthenosphere) are slowly flowing. Both continental and oceanic crusts form only the uppermost, near-surface layers of great lithospheric plates; differential motions of these plates—plate tectonics— are coupled to the circulation of the underlying asthenosphere on which they rest. The eastern and western hemispheric continents are presently drifting apart across the Atlantic Ocean and have been doing so for more than 120 million to 190 million years. Locally, continental fragments came together in the past and others are presently colliding, especially around the Pacific Rim.
Mid-ocean ridges represent the near-surface expression of hot, slowly ascending mantle currents with velocities on the order of a few centimeters per year. Whether this upwelling is due to part of a convection cell that returns asthenosphere to shallower levels after it has been dragged to depths by a lithospheric plate sinking elsewhere, or is a consequence of deeply buried thermal anomalies that heat and buoy up the asthenosphere, is not known, but both processes probably occur to varying degrees. Approaching the seafloor, the rising mantle undergoes decompression and partial melting to generate basaltic liquid. The magma within the upwelling asthenosphere is less dense and thus even more buoyant. It rises toward the interface with seawater and solidifies to form the oceanic crust, capping the stiffer, less buoyant mantle. The mid-oceanic ridges—
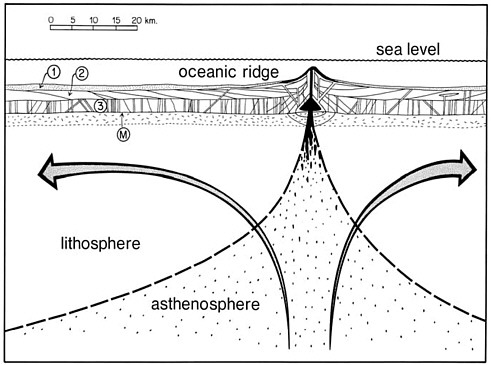
FIGURE 2.1 Schematic cross-section of a mid-oceanic ridge spreading center (a divergent plate boundary). Curvilinear mantle flow lines (arrows) show the circulation paths followed by rising asthenosphere and its cooling and conversion to lithosphere. Basaltic magma is shown as black coalescing blobs. Layers 1, 2, and 3 are deep-sea sediments, basaltic lava flows, and intrusive equivalents, respectively. M marks the Mohorovicic Discontinuity (the crust-mantle boundary).
SOURCE: Ernst (1990).
divergent plate boundaries—are spreading centers where the cooling lithospheric plates that overlie the ductily flowing mantle currents are transported at right angles away from the ridge (see Figure 2.1).
As it moves away from the ridge axis, the cooling oceanic lithosphere gradually thickens at the expense of the upper part of the asthenosphere. Heat is continuously lost, so the lithosphere-asthenosphere boundary (solid, rigid mantle above; incipiently molten, ductile mantle below), which is very close to the sea bottom beneath the oceanic ridge, descends to greater water depths away from the spreading center because its overall density increases. Unlike light continental lithosphere floating on a denser mantle, the oceanic lithosphere has a slightly greater density than the asthenosphere below, and so the oceanic plate will sink back into the deep mantle where geometrically possible.
An oceanic plate moves away from the ridge axis until it reaches a convergent plate boundary. Here, one slab must return to the mantle to conserve volume—the process of subduction (see Figure 2.2). A bathymetric low, or trench, marks the region where bending of the down-going oceanic slab is greatest. It is difficult for continental crust-capped lithosphere to sink because it is less dense than the mantle below; however, due to the descent of oceanic lithosphere, the dragging of a segment of continental crust into and down the inclined subduction zone occasionally takes place.
Production of new oceanic crust along submarine ridge systems results from this plastic flow of the deep earth, as does addition to—and deformation of—the continental crust in the vicinity of seismically and volcanically active continental margins and island arcs. Oceanic ridges are sited over upwelling mantle columns, whereas along subduction zones, lithospheric plates are descending beneath active continental mar-
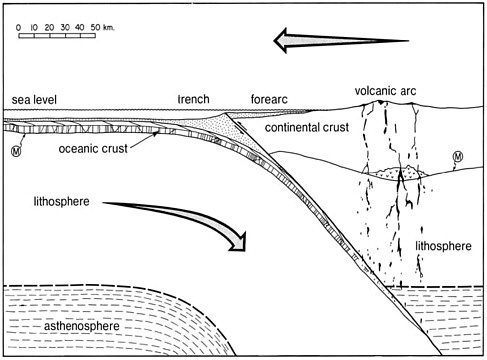
FIGURE 2.2 Schematic cross-section of a subduction zone, involving an oceanic trench and island arc-continental margin (a convergent plate boundary). Curvilinear mantle flow (arrow) shows the paths followed by cooling, descending lithosphere. Island arc magma is shown as black coalescing blobs. M is the Mohorovicic Discontinuity (the crust-mantle boundary).
SOURCE: Ernst (1990).
gins and island arcs. In contrast to submarine ridges, however, continents are also typified by mountain belts that display evidence of great crustal shortening and thickening (e.g., the Appalachians, Himalayas, and Alps). These compressional mountains contain great tracts of preexisting layered rocks, now contorted into fault-bounded blocks of folded rock. Such collisional mountain belts mark the sites of present or ancient plate boundaries.
Geological Catastrophes— Earthquakes, Volcanic Eruptions, and Landslides
Earthquakes are concentrated along plate boundaries, and earthquake locations in the oceans outline the edges of the rather young, approximately 50 km thick, homogeneous plates. In contrast, continental lithosphere may be as much as 200 to 300 km thick and is on average much older—up to 3.9 billion years old. As a consequence, these continent-capped plates consist of a diversity of rock types with variable strengths, being transected by numerous discontinuities and zones of weakness, which reflect a tortured history of repeated rifting, crustal amalgamation, and mountain building.
Where oceanic lithosphere descends beneath continents and island arcs along subduction zones, seismicity is situated at progressively greater depths farther inland as the stable lithospheric plate progressively overrides the sinking plate (see Figure 2.2). For this reason, active continental margins exhibit a broad zone of earthquakes. Seismicity around the Circumpacific is intense because oceanic lithosphere sinks beneath the continental edges of the Americas and Australasia. Large earthquakes— such as the Sumatran earthquake of December 26, 2004—are episodic, with intensities roughly proportional to the shaking time and the length of the crustal segment that ruptured in the specific seismic event.
Volcanism is a consequence of partial melting of the down-going lithospheric plate at depths approaching or exceeding 100 km. Such magmas rise buoyantly into the earth’s crust in island arcs and continental margins where they form volcanic chains and subjacent batholiths. This is also the region where plate convergence and contraction builds structural mountain belts, resulting in crustal thickening, rugged topography, and high erosion rates—such belts are characterized by landslides, mudflows, and other mass movements.
Earth Materials
The earth’s crust constitutes far less than 1 percent of the entire planetary mass but represents the nurturing substrate for virtually all life on
land and much of the life in the oceans. The biosphere predominantly occupies the near-surface skin of the solid crust—the upper, illuminated portions of the oceans and the lowermost zones of the atmosphere. To investigate and quantify the human health and longevity effects due to the presence and bioassimilation of earth materials, we need to understand the nature of the constituents that make up the earth’s crust—minerals and rocks.
A mineral is a naturally occurring, inorganically produced solid that possesses a characteristic chemistry or limited range of compositions, and a periodic, three-dimensional atomic order or polymerization (i.e., crystal structure). The diagnostic physical properties of a mineral, such as hardness, fracture, color, density, index of refraction, solubility, and melting temperature, are unique and specific consequences of a mineral’s chemical constitution, bonding, and crystal structure. Important minerals that make up the near-surface crust, and their chemical formulas, include quartz, SiO2; alkali feldspar, (K, Na)AlSi3O8; plagioclase feldspar, (Na, Ca)Al1-2Si3-2O8; olivine, (Mg, Fe)2SiO4; garnet, (Mg, Fe)3Al2Si3O8; pyroxene, (Mg, Fe)SiO3; amphibole, Ca2(Mg, Fe)5Si8O22(OH)2; muscovite, KAl2Si3AlO10(OH)2; biotite, K(Mg, Fe)3Si3AlO10(OH)2; talc, Mg3Si4O10(OH)2; serpentine, Mg6Si4O5(OH)8; kaolin, Al4Si4O10(OH)8; calcite, CaCO3; pyrite, FeS2; and hematite, Fe2O3. Some of these minerals are produced deep within the earth, some form as weathering products at the earth’s surface, and some are formed by biological processes (e.g., limestones containing the fossilized remains of marine organisms, formed by biomineralizing processes that are analogous to the human processes that form bones and teeth).
A mineraloid is a naturally occurring solid or liquid that lacks a rigorous, periodic atomic structure. The chemical compositions and physical properties of mineraloids range widely. Such substances are more weakly bonded than compositionally similar minerals; most behave like viscous fluids. Volcanic glass, amber, coal, and petroleum are examples of mineraloids.
A rock is a naturally occurring, cohesive, multigranular aggregate of one or more minerals and/or mineraloids, making up an important mapable part of the crust at some appropriate scale. The mineralogical and bulk compositions of a rock are a function of its origin. Geologists recognize three main rock-forming processes; hence, there are three principal classes of rocks:
-
Igneous—A molten, or largely molten solution (i.e., magma) that solidifies deep within the crust to form an intrusive rock, or is transported to the surface prior to completely solidifying to form an extrusive rock. Intrusive rocks cool slowly at depth to form relatively coarse-grained bod-
-
ies such as granite, granodiorite, and gabbro. Extrusive rocks are rapidly quenched, producing glassy or fine-grained volcanic ash and lava flows, such as rhyolite, rhyodacite, and basalt. Granites-rhyolites consist mainly of quartz and alkali feldspar, whereas gabbros-basalts contain olivine, pyroxene, and Ca-rich plagioclase. Rocks with mineralogical and bulk chemical bulk compositions intermediate between these end members are common.
-
Sedimentary—Such deposits form by the mechanical settling of particulate matter or precipitation of a solute from a fluid, typically water. Progressively finer grained clast sizes make up conglomerates, sandstones, siltstones, and mudstones (i.e., shales); the coarser sedimentary clasts are rich in quartz and feldspars, whereas the finer grained mudstones are dominated by clay minerals. Many chemical precipitates, but not all, are biologically generated; the major chemical sedimentary rocks are limestones (calcite) and chert (quartz).
-
Metamorphic—This group of rocks has been transformed at depth in the crust by deformation and/or physicochemical conditions that were distinctly different from those attending the formation of the preexisting igneous and sedimentary (or metamorphic) rock types. Greenstone, serpentinite, marble, gneiss, and slate are familiar examples of recrystallized (metamorphosed) basalt, mantle lithosphere, limestone, granite, and shale, respectively.
Surface interactions of minerals, mineraloids, and rocks with agents of the biosphere, atmosphere, and/or hydrosphere result in alteration of chemically reactive earth materials to produce a thin veneer of clay-rich soil. The process is termed weathering and results in removal and transportation of earth materials as soluble species in aqueous solution and as insoluble particles entrained in moving fluids (wind, water, and ice). The residue left behind over time builds up a soil profile. It is such weathering products that in many cases provide the ready supply of both nutrients and toxic chemical species that influence the existence of life in general and human health in particular. Geological mapping and remote sensing techniques provide the enhanced spatial understanding of the areal disposition and concentration of surficial earth materials that are an essential component of epidemiological investigations of environmentally related diseases and human senescence.
Soil and the Vadose Zone
The human environment is heavily dependent on the continuum between soil, water, and air that is located at the earth’s surface. Ultimately this continuum—and the interactions between the physical, chemical, and
biological properties of each component—moderates many of our activities. The geological zone between the land surface and subsurface groundwater—the vadose zone—consists of unsaturated organic and earth materials. A subset of this vadose zone is the near-surface soil environment, which is in direct contact with both surface water and the atmosphere.
Soil directly and indirectly influences our quality of life—it is taken for granted by most people but is essential for our daily existence. It is responsible for plant growth and for the cycling of nutrients through microbial transformations, and has a major effect on the oxygen/carbon dioxide balance of the atmosphere. Because of our reliance on soil, any disturbance of soil or the vadose zone, or modification of natural soil-forming processes, has the potential for adverse public health effects. Soil also plays a critical public health role in regard to pollutants that have been disposed of at the earth’s surface, as they can promote or restrict transport to groundwater, the atmosphere, or food crops.
Soil is a complex mixture of weathered rock particles, organic residues, air, water, and billions of living organisms that are the end product of the interaction of the parent rock material with climate, living organisms, topography, and time—the five soil-forming factors. The soil layer can be as thin as a few inches or may be hundreds of feet thick. Because soils are derived from unique sources of parent material under specific environmental conditions, no two soils are exactly alike—there are thousands of different kinds of soils within the United States.
Soils can be acidic (pH <5.5), neutral (pH of 6–8), or alkaline (pH >8.5). Soil pH affects the solubility of chemicals in soils by influencing the degree of ionization of compounds and their subsequent overall charge. The extent of ionization is a function of the pH of the environment and the dissociation constant (pK) of the compound. Consequently, soil pH can be critical for affecting the transport of potential pollutants through the soil and vadose zone and can also affect the transport of viruses with different overall charge. In high rainfall areas, the combination of acidic components and residues of organic matter, together with the leaching action of percolating water, leads to acidic soils. Conversely, soils in arid areas are more likely to be alkaline because of reduced leaching, lower organic contents, and the evaporative accumulation of salts.
Soil normally consists of about 95–99% inorganic and 1–5% organic matter. The inorganic material is composed of three particle size types— sand (0.05–2 mm), silt (0.002–0.05 mm), and clay (<0.002 mm; i.e., <2 microns)—that result from the weathering characteristics of the parent rock. In some geological terrains (e.g., some igneous and glacial areas), soils also contain larger (>2 mm) gravel- and cobble-sized particles inherited from the parent rock type. The percentage of sand, silt, and clay in a particular soil determines its texture (see Figure 2.3), which affects many of
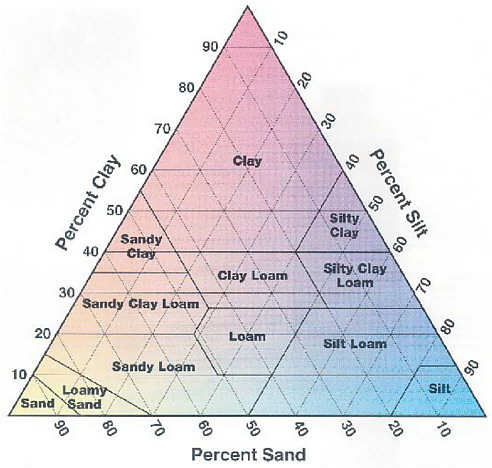
FIGURE 2.3 Soil textural triangle showing the relationship between soil texture categories and particle size. These textural classes characterize soil with respect to many of their physical properties.
SOURCE: Pepper et al. (2006).
the physical and chemical properties of the soil. Of the three primary particle types, clay is by far the dominant component for determining a soil’s properties because of the greater number of clay particles per unit weight. The increased surface area of soils with higher clay concentrations leads to increased chemical reactivity of the soil. In addition, clay particles are the primary soil particles that have an associated electric charge. This is the basis for a soils cation-exchange capacity (CEC), which is normally a negative charge that occurs because of isomorphic substitution or ionization of hydroxyl groups at the edge of the clay lattice.
Differences in the partitioning of elements among the different par-
TABLE 2.1 Size Fractionation of Soil Constituents
ticle size classes is an important component of understanding potential health effects from soils. Elemental variations result both from the mineralogical and geochemical characteristics inherited from parent rock materials and, particularly for the clay fraction, from macro and trace elements introduced by contamination. The distribution of inorganic and organic constituents among the different soil particle classes is summarized in Table 2.1.
The three types of primary particles do not normally remain as individual entities. Rather, they aggregate to form secondary structures, which occur because microbial gums, polysaccharides, and other microbial metabolites bind the primary particles together. In addition, particles can be held together physically by fungal hyphae and plant roots. These secondary aggregates, which are known as “peds,” can be of different sizes and
shapes, and give the soil its structure. Pore space within the aggregate structure (intraggregate pore space) and between the aggregates (interaggregate pore space) is crucial to the overall soil architecture. Pore space also regulates water movement and retention as well as air diffusion and microsite redox potentials.
Organic Matter in Soils
Organic compounds are incorporated into soil at the surface via plant residues such as leaves or grassy material. These organic residues are degraded by soil microorganisms, which use the organic compounds as food or microbial substrate. The main plant constituents—cellulose, hemicellulose, lignin, protein and nucleic acids, and soluble substances such as sugars—vary in their degree of complexity and ease of breakdown by microbes. In general, soluble constituents are easily metabolized and break down rapidly, whereas lignin, for example, is very resistant to microbial decomposition. The net result of microbial decomposition is the release of nutrients for microbial or plant metabolism, as well as the particle breakdown of complex plant residues.
The nutrient release that occurs as plant residues degrade has several effects on soil. The enhanced microbial activity causes an increase in soil structure, which affects most of the physical properties of soil, such as aeration and infiltration. The stable humic substances contain many constituents that contribute to the pH-dependent CEC of the soil. In addition, many of the humic and nonhumic substances can complex or chelate heavy metals and sorb organic contaminants. This retention affects their availability to plants and soil microbes as well as their potential for transport into the subsurface. Overall, the physical constituents control many of the chemical reactions that occur within soil, with fine particles (~2µm)—inorganic granulometric and fine clays as well as organic matter that results from microbial decomposition of plant residues—being particularly important.
Conversely, it is the soil microflora that control biochemical transformations in soil. Interestingly, the organic and biological constituents of soil mimic the mineral constituents with respect to size (Table 2.1). In order of increasing size, the major soil biota consists of viruses, bacteria, fungi, algae, and protozoa. As size decreases, the number of organisms increases to staggeringly large numbers—a gram of soil literally contains billions of organisms. The physical heterogeneity of soil results in microenvironments that allow diverse microbial communities to coexist in close proximity. Overall, the variable terminal electron requirements of aerobic and anaerobic microbes coupled to variable nutritional require-
ments (autotrophy and heterotrophy) result in extraordinary soil biological diversity. The soil microflora are responsible for many of the biochemical processes essential for human life, including plant growth, products for human health, and groundwater protection (Stirzaker et al., 1996; Bejat et al., 2000; Strobel and Daisy, 2003). Conversely, soils also contain human pathogens and are a source of bacterial antibiotic resistance.
Gases and Liquids in Soils
Because soil and the atmosphere are in direct contact, most of the gases found in the atmosphere are also found in the air phase within the soil (the soil atmosphere)—oxygen, carbon dioxide, nitrogen, and other volatile compounds such as hydrogen sulfide or ethylene. The concentrations of oxygen and carbon dioxide in the soil atmosphere are normally different from those in the atmosphere, reflecting oxygen use by aerobic soil organisms and subsequent release of carbon dioxide. In addition, gaseous concentrations in soil are altered by diffusion of oxygen into soil and carbon dioxide from soil. Because microbial degradation of many organic compounds in soil is carried out by aerobic organisms, the presence of oxygen in soil is necessary for such decomposition. Oxygen occurs either dissolved in the soil aqueous solution or in the soil atmosphere.
The total amount of pore space depends on soil texture and structure. Soils high in clays have greater total pore space but smaller pore sizes. In contrast, sandy soils have larger pore sizes, allowing more rapid water and air movement. Aerobic soil microbes require both water and oxygen, which are both found within the pore space, and therefore soil moisture content controls the amount of available oxygen in a soil. In soils saturated with water, all pores are full of water and the oxygen content is very low. In dry soils, all pores are essentially full of air, so the soil moisture content is very low. In soil with moderate moisture content, both air (oxygen) and moisture are readily available to soil microbes. In such situations, soil respiration via aerobic microbial metabolism is normally at a maximum.
Because they are unsaturated, vadose zones generally are aerobic. However, due to the heterogeneous nature of the subsurface, anaerobic zones can occur in clay lenses and so both aerobic and anaerobic microbial processes may occur in close proximity. At contaminated sites, volatile organic compounds can be present in the gaseous phase of the vadose zone. For example, chlorinated solvents, which are ubiquitous organic contaminants, are volatile and are typically found in the vadose-zone gaseous phase below hazardous waste sites.
Soil as a Genetic Resource
Soil is the home for billions of microorganisms with unimaginable diversity. It has been estimated that 4,000 completely different genomes of standard bacteria (Torsvik et al., 1990) and at least 1 million different species of fungi (Gunatilaka, 2006) are present in soil. Of these organisms, less than 1% of bacterial species and 5% of fungal species have been identified (Young, 1997). Despite this limitation, soil has proven to be a treasure chest of natural products critical to maintaining human health and welfare. Of the soil microbes, actinomycetes and fungi have proved to be particularly rich sources of metabolites with novel biological activities. The fungal antibiotic penicillin and the actinomycete antibiotic streptomycin were the first antibiotics to be discovered from soil. More recently, interest has centered on rhizosphere bacteria and endophytic microbes as a new source of natural products including antibiotics.
Biogeochemical Cycling
Over the past century, industrialization has accelerated and the environmental consequences are ubiquitous. Although some believe that environmental changes on our planet are caused entirely by anthropogenic excesses, the earth’s environment has in fact undergone profound changes—caused by processes such as those briefly described above— throughout its history. The superposition of earth materials—the stratigraphy of sediments and rocks—provides a record of past physical and biological processes. These in turn define the directions and magnitudes of the physical and biological changes that provide the context for modern conditions. This section notes the diversity of biological systems, emphasizing the metabolic processes of microorganisms and their roles in consuming earth materials to produce metabolites and influence the environment.
Diverse and abundant microorganisms, such as bacteria, viruses, fungi, algae, protozoa, and other groups of single-celled organisms, affect nearly all aspects of the environment from the atmosphere to deep within sediments beneath the ocean floor. To many people, microorganisms are viewed as harbingers of disease. But food technologists know that the life processes of bacteria and yeast are essential for the production of many dairy products, baked goods, and alcoholic beverages. Soil scientists study the complex symbioses involving microbes and plants. Few of us realize the importance of microorganisms in the formation of many natural resources, including petroleum, carbonate and silica minerals, and metallic ore deposits. They play crucial roles in the global cycling of carbon, sulfur, and nitrogen and in maintaining the composition and the dynamic
equilibrium among the chemical species present in the oceans, soils and rocks, and the atmosphere.
Six elements are of crucial importance for all life on earth—carbon (C), hydrogen (H2), oxygen (O2), nitrogen (N2), sulfur (S), and phosphorus (P). These elements, collectively referred to as CHONSP, are essential components of the building blocks of all cells from unicellular bacteria to multicellular mammals. In different combinations and ratios, they are present in carbohydrates, lipids, proteins, and nucleic acids. They combine to form skeletal materials such as lignin and cellulose in plants, chitin in insects and crustaceans, and keratin in mammals and—with the calcium cation (Ca)—form apatite in bones and calcite in invertebrates. A wide spectrum of metabolic systems has evolved to efficiently recycle these elements. Accordingly, the products of one set of biochemical processes are used as reactants for another set, thus ensuring that the elements are not irreversibly bound in a form that is unavailable to living matter.
Biogeochemical cycling is the recycling of elements by organisms in the context of geological processes. Various microorganisms participate in every chemical transformation of CHONSP. These tiny, incredibly abundant and diverse organisms are the workhorses of biogeochemistry. They degrade previously synthesized organic material or form new organic substances by fixing carbon dioxide, both photosynthetically and in the absence of light. In carrying out their wide range of metabolic functions, they consume and then release each element, thus returning it to the biosphere, the hydrosphere, the atmosphere, and/or the solid earth. An instructive example is represented by the development of soil—the substrate on which the terrestrial food chain is based.
HUMAN PHYSIOLOGICAL PROCESSES
All of us share a bipedal upright structure and a body with distinctive anatomical parts that have discrete morphologies and functions. Acting in concert, these components maintain a state of balance (homeostasis) within the organism. The loss of body structural integrity and function can occur when homeostasis is perturbed by internal factors or by physiological response to hazardous materials in the environment. Most earth materials—solids, liquids, or gases—are essential for the body or are benign. A few can become harmful, especially if in elevated amounts, where they impinge or enter the body and disturb the normal functions of the organs. There are three usual routes of exposure to earth materials—respiration (through the nose or mouth and into the breathing apparatus), ingestion (through the mouth into the digestive system), and dermal (through the skin).
The circulatory system that transports blood to all organs and tissues is the pathway for disseminating the nutrients—and any hazardous substances—throughout the body. Most hazardous substances that enter the body are either excreted, or protective mechanisms present in our bodies that refresh the cell populations in tissues will act over time to diminish the hazard. Unfortunately, some normal protective processes may exacerbate the action of the hazard, aiding, for example, local accumulation of the hazardous species, or provide unwanted long-term consequences such as occurrences of scarring from deposition of particulate matter in the lung. Although the level and duration of exposure (see Box 2.1) that can lead to disease are unique to an individual, there are some hazards—such as asbestos and arsenic—that are known public health issues at minimal exposure levels.
Human anatomy is the starting point within which complicated tissue, cellular, and molecular-level reactions take place that define specific diseases. Details of these specific reactions and diseases are supplied by specialists (e.g., Warwick and Williams, 1973; Goldman and Ausiello, 2004; Cohen, 2005) and are beyond the scope of this report. However, partial or total inhibition of one of these body systems would likely have dire results that we can all appreciate. A simplified overview of the physiology of three body systems and their normal functions—the respiratory, digestive, and integumentary (skin) systems—provides the human context for this report.
Inhalation Pathways
The human respiratory system consists of organs and tissues in the upper trunk that permit us to breathe ambient air, extract oxygen, and respire carbon dioxide back into the atmosphere. Under both voluntary and involuntary nervous system and muscular control, the continual motions of inspiration and exhalation of air provide the oxygen essential for metabolic activity throughout the body. The passageways from the nose or mouth through to the lungs, with their specialized air sacs or alveoli, are illustrated in Figure 2.4. These tiny balloonlike structures are surrounded by arterioles and venules where the essential gas exchange— into and from the blood—actually takes place. Hazardous materials in ambient air may access the inner portions of the body through the respiratory system sampled with each breath.
The respiratory system has a series of built-in defenses, natural mechanisms, and strategies that are very effective for minimizing inadvertent transport of a wide range of potential hazards, whether they are solids, liquids, or gases. The first line of defense is the nose, where hairs, and/or a sneeze, will expel unwanted materials. Ciliate cells that popu-
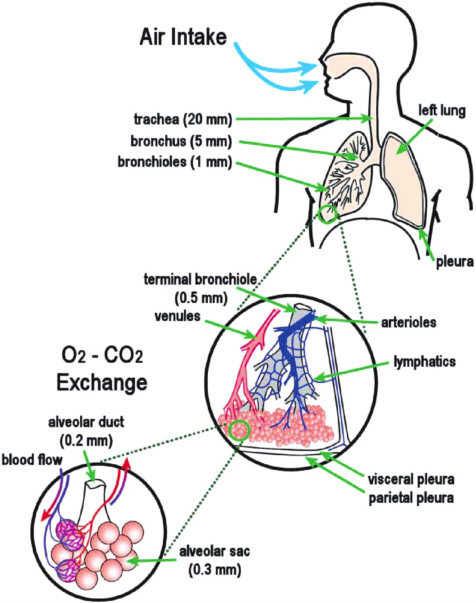
FIGURE 2.4 Schematic diagram of the human respiratory system, showing the gross anatomy of the lung, the covering membranes (pleura), airways, and air sacs (alveoli). The average diameter of portions of the air flow system are indicated—trachea, 20 mm; bronchus, 8 mm; terminal and respiratory bronchioles, 0.5 mm; alveolar duct, 0.2 mm; and alveolar sacs, 0.3 mm.
SOURCE: Modified after Skinner et al. (1988).
|
BOX 2.1 Exposure Assessment in Epidemiological Studies Epidemiological studies of environmental exposure—studies of adverse health outcomes in human populations—are designed to quantify the risk of disease at particular levels of exposure to the environmental threat. The success of such studies to detect an effect, or conversely to demonstrate that health consequences of specific exposures are likely to be minimal, depends on both the accuracy of the health outcome diagnosis and the accuracy of estimates of exposure to the environmental insult. Depending on the health outcome under study, there can be a wide range of diagnostic accuracy. However, in countries with advanced systems of medical care, diagnostic accuracy is typically quite high for health effects such as cancer, major birth defects, myocardial infarction (heart attack), and many neurological diseases, and the inaccuracies that do occur typically do not limit the ability to accurately quantify the effects of environmental exposures. Far more important in influencing the success of such studies is inaccuracy in exposure assessment, usually referred to as “misclassification of exposure.” That is, persons with high exposures can erroneously be classified as having had low or no exposure and persons with low exposure as having had high exposure. In addition, many studies use indicators of exposure rather than actual measurements of exposure, and such indicators can be less precise. Even in instances of equivalent exposure, differences in individual physiological characteristics (e.g., respiration rate, the capacity to metabolize specific xenobiotic compounds) can cause differences in effective dose that can produce varying health outcomes. How does such misclassification affect the findings of an epidemiological study? The basic measure of effect used by most epidemiological studies is the relative risk—an estimate of the ratio of disease (or death) rate among exposed persons relative to the unexposed rate. For example, the relative risk for lung cancer among pack-a-day lifetime smokers is about 15, meaning that smokers have about 15 times the rate of lung cancer as people who have never smoked. There is a gradient of risk in most real-world situations, with the relative risk increasing with the level of exposure. When exposure misclassification occurs in epidemiological studies without regard to whether subjects are diseased or not diseased, the result |
|
is usually to lower the estimates of the effect. Given a “true” relative risk, it is possible to calculate what a researcher would actually observe in an epidemiological study, given various levels of exposure misclassification. In a simplified example, where subjects are either “exposed” or “unexposed” (i.e., no gradient of exposure), and the correlation coefficient is 0.60 between the “true” and estimated exposures, a relative risk of 2.0 would appear to have a relative risk of 1.6 (Vineis, 2004). In some situations, such a difference could determine whether or not an exposure is identified as a significant risk factor. Thus, misclassification of exposure in epidemiological studies can have a major detrimental effect, and it is highly worthwhile to place major resources into the exposure assessment efforts when investigating environmental or occupational exposures. Earth scientists frequently play a crucial role in providing measures or estimates of exposure in epidemiological studies of environmental contaminants. A good example is the collaboration between the National Cancer Institute (NCI) and the U.S. Geological Survey (USGS) in an epidemiological study of bladder cancer in northern New England, which also includes collaborators from Dartmouth Medical School and the state health departments of Vermont, New Hampshire, and Maine. Since 1950, bladder cancer mortality in New England for both males and females has been above the U.S. average. Because some areas of New England have modestly elevated levels of arsenic in groundwater, arsenic in drinking water has been suggested as a reason for these excess bladder cancer rates. The identification of arsenic in drinking water as a cause of bladder cancer in parts of Taiwan, Chile, and Argentina, where it occurs (or has occurred) at considerably higher levels, has been proposed as supporting evidence for this suggestion. The NCI recently completed fieldwork in New England for a population-based case control study, over a three-year period, which involved about 1,200 newly diagnosed cases of bladder cancer and 1,200 disease-free persons. A water sample was collected at the current homes of patients and control subjects, and information about past residences and their water sources, together with much additional information, was obtained in personal home interviews. The USGS located past wells used by the subjects and, wherever possible, collected water samples. USGS scientists also developed geologically based statistical models to estimate arsenic levels in well water where sample collection was not feasible (Ayotte et al., 2006). |
late the surface of the upper bronchi constantly beat upward, also acting to remove substances that may not have been propelled out by a sneeze. They are often swallowed or expectorated with continually locally supplied mucus. If a particle is small enough to bypass these expulsion efforts, it may become lodged in the tissues along the passageways or in the lung tissue, where special cells such as macrophages may engulf the intruding particle and transport it through the lymph system to the lymph nodes. Another protection option occurs when offending materials become isolated by being engulfed in fibrous collagenous protein (collagen) produced by cells known as fibroblasts. This mechanism is part of the normal repair strategies employed by biological systems subjected to trauma (Skinner et al., 1988).
The structures of the respiratory system from the macro level (i.e., the muscles and the connective tissues forming the pleura that enclose and define the lung cavity), to the specialized cells for transport of gases and those that act in natural defense of the system, have a multitude of normal biological reactions. Some may become pathological and disadvantageous, decreasing the availability of oxygen. Lung disease is a product of the sensitivities and lung physiology of an individual and the duration and exposure level to a hazardous substance.
There are a host of potentially harmful substances whose presence can produce breathlessness, hacking cough, or difficulties in breathing. Respiratory malfunctions are usually diagnosed through physical examination, including auscultation (listening with a stethoscope), radiological examination (X-ray imaging) of the chest, and patient exposure histories. Clinical evaluation may be able to identify likely potential offenders through the specificity of organ responses, which may in turn be confirmed by histological analysis. With repeated exposures, especially at high doses over a long time, the incorporation of foreign materials usually causes cells in the respiratory system to die or to mutate, and diseases such as chronic obstructive pulmonary disease (COPD) or cancer may result. Smoking, of course, is the most egregious example of deliberate inhalation of hazardous material.
Inhalation of gaseous, liquid, or particulate matter can alter and perhaps reduce pulmonary function, especially if this foreign material causes irritation in the airways because normal clearance mechanisms are compromised (e.g., by smoking). Protection from irritation of the delicate tissues is part of the normal breathing apparatus—cilia in the nose and upper respiratory tract, coughing and expectoration, cellular (e.g., macrophage) activities all aid in the expulsion or isolation of potentially damaging foreign material. If these multiple preventive mechanisms prove unable to expel or mitigate the adverse action of the foreign materials on the respiratory system, chronic coughs, bronchitis, or asthma may result
from such irritations and/or continued exposures (Skinner et al., 1988; Koenig, 1999). Particle size determines whether particles are respirable (<10µm) or inhalable1 (10–100µm). Respirable particles tend to be deposited at the junctions of the respiratory bronchioles in humans (see Figure 2.4). The airways and the alveolar regions, where the air/carbon dioxide exchange takes place, are affected in diseases such as asbestosis and silicosis. Studies have estimated that with each 10 µg m−3 increase in PM10 above a base level of 20 mg m−3, daily respiratory mortality increases by 3.4%, cardiac mortality increases 1.4%, hospitalizations increase 0.8%, and school absenteeism increases 4.1% (Vedel, 1995).2 Particulate matter can also exacerbate the production of photochemical smog, the end product of the interaction of smoke, volatile hydrocarbons, and fog, under the influence of ultraviolet light.
Ingestion Pathways
The essential activities of the gastrointestinal system are absorption of nutrients and elimination of waste. Whenever something is ingested, at least a portion is usually digested and absorbed and is distributed to all parts of the body by the circulation system, either in its original form or as modified by chemical transformation in the liver or elsewhere.
Mastication of solid food by the teeth reduces the size so that it can be swallowed and enter the gastrointestinal tract. Saliva and other gland secretions that empty into the mouth moisten the chewed food, providing enzymes like amylase that dissolve starches and add to the disintegration. Complete solubilization of all the large molecules in foodstuffs leading to release of soluble constituents (e.g., small ions, or molecules) requires biochemical activity that commences in the saliva and continues into the small intestine. Specialized gland cells that line the inside of the tract secrete chemicals that break down the solids, while others are involved in absorption and transmission of the “chemicals,” some of which are nutrients, and make them available to the circulatory system. The mobilization of ingested materials, and the extraction and adsorption of nutrients, is the focus of the digestive tract. The tract is effectively a flexible tube that ranges in length up to about 9 m from the mouth to the anus in an adult.
The organs in the digestive tract are supported by fibrous connective tissue, and in some parts of the system there are double layers that also
carry blood vessels, lymph vessels, and nerves. Each of these organs performs specialized activities with specialized cells and each produces proteins and enzymes that have minor and trace elements as cofactors. Ingestion of hazardous materials or elements in food or water may induce local aberrations in the normal operations of each of these essential organs. High concentrations of arsenic (see Chapters 4 and 5), for example, must pass through the gastrointestinal system and can influence local biochemical reactions.
Clinically, examination of blood and urine is traditionally used to assay the amount of a potential hazard that has been ingested. However, despite the fact that fluids are consistently exchanged throughout body compartments, if there is a potential for accumulation intracellularly or within certain tissues, then these trace levels are not easily assessed. The fate of absorbed nutrients or hazardous materials may be modified in the body by the liver and dumped into the intestine for elimination in the feces. Alternatively they may be filtered and recycled by the kidney or actively secreted in the urine. Some unwanted chemicals are known to be stored in fats (e.g., DDT) or other cells in relatively small amounts.
Skin Absorption Pathways
The skin is the largest organ of the human body. More properly known as the integumentary system, the skin is composed of thin layers of tissue that cover the entire body and in some instances presents early clues to an individual’s health status. The outermost area of skin—the epidermis—contains keratinocytes3 that produce stratified squamous epithelium layers and a few other types of cells. The base of the epidermis is interdigitated with the underlying dermis, mostly connective tissue of mesodermal origin. The dermis supplies some nourishment to the lowest layers of the epidermis and is a framework of connective tissue that contains blood vessels, nerve endings, glands, and the roots of hair follicles. The skin has variable thickness—it may reach 1.5 mm at the soles of the feet and palms of the hands, whereas in other areas (such as around the eyes) the skin is a thin (< 0.1 mm) delicate covering that is relatively elastic.
Although one function of the skin is to assist in the formation of vitamin D, the major contribution of skin to human health is to protect the body from invasions of pathogens, prevent dehydration, and keep body temperature constant. The optimal performance of this organ requires continual regeneration to maintain complete body coverage to minimize
adventitious fluid and water loss or infection. Dermal toxicity results from local tissue responses through direct contact of a substance with skin or, alternatively, may represent a manifestation of systemic toxicity following ingestion or inhalation. Allergic contact dermatitis induced by nickel is an example of a local tissue response (Centeno, 2000).
GEOAVAILABILITY, BIOAVAILABILITY, AND BIOACCUMULATION
Many chemical elements occur in living tissues in such small concentrations that they are referred to as trace elements. Some trace elements are essential for human life because of their role as catalysts in cellular functions involving metabolic or biochemical processes. At present, less than one-third of the 90 naturally occurring elements obtained from the air, water, and food are known to be essential to life. The mineral elements currently considered essential for human health and metabolism include the major ions/anions sodium (Na+), calcium (Ca2+), chlorine (Cl−), magnesium (Mg2+), potassium (K+), silicon (Si4+), sulfate (SO4−), and nitrate (NO3−); trace elements such as phosphorus (P), iodine (I), and fluorine (F); and metals/metalloids such as iron (Fe), zinc (Zn), copper (Cu), manganese (Mn), vanadium (V), selenium (Se), cobalt (Co), nickel (Ni), chromium (Cr), tin (Sn), and molybdenum (Mo) (Moynahan, 1979). Some of these species occur predominantly in silicate minerals (e.g., Mn, boron [B]), some in silicates and sulfides (e.g., Zn, Se) or as trace element impurities in phyllosilicate minerals, and some predominantly as sulfides (e.g., Cu, Mo); others (e.g., Fe) are ubiquitous (Combs, 2005). The bioavailability and bioassimilation of these essential mineral elements are dependent upon each of their unique physiochemical properties.
Various definitions have been used to describe trace element concentrations within earth science materials and humans (see Box 2.2). Interestingly, both earth scientists and public health professionals have distinguished total elemental concentrations from bioavailable concentrations. In essence, bioavailable concentration in soils refers to the concentration of an element in solution that can be taken up by plants or microorganisms. Within the human body, the term “bioavailable” refers to the amount of a particular element that can be absorbed by the body and influence human health and welfare. For earth science materials and the human body, bioavailable concentrations are always less than total elemental concentrations.
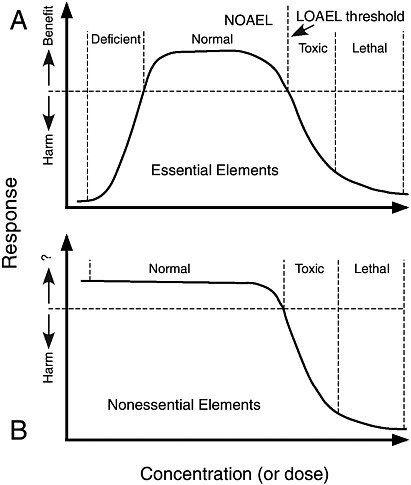
Many trace elements are metabolic requirements for humans, so too small an amount of these elements will result in deficiency (see Figure 2.5A). Although the biological response is optimal at higher concentra-
|
BOX 2.2 Bioavailability Definitions Earth Science Definitions Bioavailability—that fraction of an element or compound in solution that can be taken up by plants or soil micro-organisms. Biomagnification—the bioaccumulation of a substance up the food chain by transfer of residues of the substance in smaller organisms that are food for larger organisms in the chain. Geoavailability—that portion of an element or compound’s total content in an earth material that can be liberated to the surficial or near-surface environment (or biosphere) through mechanical, chemical, or biological processes. Health Science Definitions Bioaccessible fraction—that fraction of a metal or a metal compound that is soluble in various body fluids (gastrointestinal, respiratory, perspiration, etc.). Solubility is dependent on individual physiology. Bioavailable fraction—that fraction of a metal or metal compound that is absorbed by the body and transported within the body to a site of toxicological action. Absorption and transport are both dependent on individual physiology. |
tions, at still higher concentrations the element may become toxic. Toxicologists express the level at which these phenomena are observed in several different ways. The first is the No Observable Adverse Effect Level (NOAEL) or No Observable Adverse Effect Concentration (NOAEC). At higher concentrations, the biological response is expressed as the Lowest Observable Adverse Effect Level (LOAEL) or Lowest Observable Adverse Effect Concentration (LOAEC). The dose-response curve lacks a deficiency zone for nonessential elements (see Figure 2.5B).
Element toxicity depends on the bioavailability of the element, its distribution in the body, the physical and chemical form of the element, and its storage and excretion parameters. In recent years, considerable interest has been focused on assessing the human health risk posed by metals, metalloids, and trace elements in the environment. It has long been recognized that large areas of the globe contain human populations characterized by having trace element excess, deficiency, or chronic poisoning (e.g., Selinus et al., 2005).