1
Introduction
It is widely understood that stem cell treatments have the potential to revolutionize medicine. Doctors and medical researchers think, for example, that it could be possible to develop stem cell–based treatments for such diseases as chronic heart disease, Type I diabetes, and Parkinson’s disease. Stem cells could also prove valuable in repairing various injuries, such as spinal cord damage, the brain damage caused by a stroke, and the damage to heart muscles caused by a heart attack. And cell lines created from stem cells could be used in the testing of drugs and in various types of biomedical research.
Because of this potential, in 2004 California voters approved Proposition 71 to set up a 10-year, $3 billion program to fund research on stem cells. Under the direction of the California Institute for Regenerative Medicine, this program will pay to build facilities for stem cell research and will fund doctors and scientists to carry out research with the ultimate goal of helping to develop therapies based on stem cells.
For this research to move forward, however, will require a steady supply of stem cells, particularly human embryonic stem cells. Those stem cells are collected from developing human embryos created from eggs—or oocytes—harvested from the ovaries of female donors. Thus much of the promise of stem cells depends on women choosing to donate oocytes to the research effort.
The oocyte donation process is not without risk, however. Donors are given doses of hormones to trigger the production of more eggs than would normally be produced, and this hormone treatment can have various side effects. Once the eggs have matured in the ovary, they must be
retrieved via a surgical procedure that is typically performed under anesthesia, and both the surgery and the anesthesia carry their own risks. Furthermore, given the very personal nature of egg donation, the experience may carry psychological risks for some women as well.
With this in mind, in 2006 the California Institute for Regenerative Medicine contracted with the National Academies to organize a workshop that would bring together experts from various areas to speak about the potential risks of oocyte donation and to summarize what is known and what needs to be known about this topic. The Committee on Assessing the Medical Risks of Human Oocyte Donation for Stem Cell Research was formed to plan the workshop, which was held in San Francisco on September 28, 2006. This report is a summary and synthesis of that workshop.
SOURCES OF STEM CELLS
Stem cells are the body’s resource for all other types of cells. That is, stem cells are unspecialized cells that can self-replicate and give rise to specialized types of cells, from neurons to white blood cells. Stem cells come in several varieties, including embryonic, fetal, and adult stem cells, but most of the interest in possible medical applications has focused on: embryonic stem cells and adult stem cells. Embryonic stem cells can give rise to any type of cell in the body, whereas adult stem cells are generally more limited, giving rise to only certain types of cells, depending on where in the body they are located. Although adult stem cells may have many important therapeutic uses, embryonic stem cells are generally considered to have more potential at this time, in large part because it is relatively easier to grow large numbers of embryonic stem cells in a cell culture. And, in particular, Proposition 71 gives priority to human embryonic stem cell research.
As Linda Giudice, the committee chair, explained in her introductory remarks at the workshop, human embryonic stem cells are generally collected from the inner cell mass of the blastocyst. A blastocyst is a spherical preimplantation embryo containing 200 to 250 cells. It consists of an outer layer of cells, the trophectoderm, and an inner fluid-filled cavity (blastocoel) containing an interior cluster of cells called the inner cell mass. It is the inner cell mass from which embryonic stem cells are derived.
The embryos used as a source of embryonic stem cells can be created in two ways (see Figure 1-1). The most common way—and, indeed, the only proven way with human embryos at this point—is by in vitro fertilization (IVF), in which an egg is fertilized with sperm cells in a culture dish. A second technique, called somatic cell nuclear transfer, works by replacing the nucleus of the egg with the nucleus of a somatic cell (i.e., a cell that is neither an egg nor a sperm) from the same or another person. Since the nucleus of a cell contains its nuclear DNA, an egg used for somatic cell nuclear transfer has all of its DNA (except for that associated with another cell structure called mitochondria) from the person donating the somatic cell and none from the egg donor. This technique may one day make it possible to grow tissues that are genetically nearly identical to a donor—allowing doctors, for example, to create replacement organs that would not be rejected by a patient’s body—but at this point no one has succeeded in making somatic cell nuclear transfer work with human oocytes.
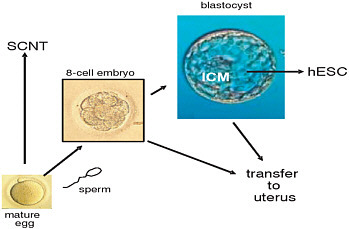
FIGURE 1-1 The procurement process and options for oocytes and embryos for research.
Note: Retrieved oocytes can be inseminated with sperm and subsequent embryos can be transferred to a women’s uterus at the 8 cell stage or at the blastocyst stage. Alternatively, oocytes can be used for research and undergo somatic cell nuclear transfer (SCNT). Also, the inner cell mass (ICM) of the blastocyst can be used to derive human embryonic stem cell (hESC) lines.
The major source of human embryonic stem cells to date has been excess IVF embryos that are donated by couples who have completed their treatment for infertility. In cases in which female patients cannot produce their own eggs, these embryos are made using donated eggs from other women. If stem cells are to be made by IVF purely for research, however, and not as a part of infertility treatment, this would necessarily require the donation of eggs. To make stem cells by nuclear transfer would also require the donation of eggs. So research on human embryonic stem cells may eventually demand a supply of eggs that are donated by women for research purposes.
EGG DONATION
Over the past two decades, millions of women have had oocytes collected for the purpose of assisted reproduction. Most of those women were IVF patients whose eggs were viable but who were unable to achieve a pregnancy for some other reason, such as blocked fallopian tubes or a partner with a low sperm count. But a significant minority of the women having their eggs harvested were not themselves trying to get pregnant but rather were donating their eggs to help another woman get pregnant.
In 2003, the latest year for which statistics are available, the Society for Assisted Reproductive Technology reported that there were 115,392 assisted reproduction cycles, or attempts, at 428 clinics around the United States. Of those, nearly 12 percent—or about 13,000 assisted reproduction cycles—involved oocytes provided by egg donors.
Whether a woman’s eggs are to be harvested for her own infertility treatment or for donation to another woman—or for research—the donation process is the same. The woman self-injects hormones (gonadotropins) to stimulate the growth of ovarian follicles, plus a gonadotropin-releasing hormone (GnRH) agonist to block the normal surge of luteinizing hormone (LH), which could cause the woman to ovulate before the physician retrieves the eggs. In many instances, GnRH agonists are administered a week before stimulation to control the stimulation cycle and avoid a spontaneous LH surge. A woman subsequently self-injects the hormone human chorionic gonadotropin (hCG, similar to LH) to effect egg maturation. When the eggs are ready, the woman is brought into surgery, where she receives intravenous sedation, after which a transvaginal probe is placed in her vagina. A hollow needle emerges from the probe,
travels through the back of the vagina and into the ovary, where, under the guidance of ultrasound technology, the eggs are aspirated. Typically, a woman who has undergone the usual hormone treatment will have a dozen or so eggs that can be collected.
Once the oocytes have been retrieved, they are prepared for fertilization. Each egg is placed in a culture medium along with prepared sperm cells and incubated for about 18 hours. At the end of this time, the eggs have been fertilized, and they are put into a growth medium for another 1-2 days, until they have reached the four- to eight-cell stage. At this point, they can be transferred into the woman’s uterus, although a number of assisted reproduction facilities wait another two days until the fertilized embryo has reached the blastocyst stage, with approximately 100 cells. At this stage, the embryo can be used for the collection of embryonic stem cells from the blastocyst’s inner cell mass.
POTENTIAL RISKS
Years of experience with assisted reproduction have identified a number of potential risks associated with egg donation (see Box 1-1), which fall into three main categories. The first category of potential risks arises from the hormone regimen that women are given to stimulate egg production. The risks include ovarian hyperstimulation syndrome; breast, ovarian, and endometrial cancers; and perhaps problems with long-term fertility. The second category is associated with the surgical procedure, including the anesthesia, and involves many of the same issues that anyone having surgery faces. The third set of potential risks is psychological in nature and includes anxiety, mood swings, and post-donation adjustment.
|
BOX 1-1 Potential Risks of Oocyte Donation Acute Risks Ovarian hyperstimulation syndrome Surgical Anesthetic Psychological Long-Term Risks Breast, ovarian, and endometrial cancers Future fertility |
The workshop panelists were asked to discuss these potential risks, evaluating the seriousness of each and discussing the uncertainties involved in each. Dr. Giudice summarized the issues before the committee as “what is known, what needs to be known, and what can be done to reduce the potential risks over time.” The panelists’ answers to those questions are described in the pages that follow.






