7
Training the Learning Health Professional
OVERVIEW
In a system that increasingly learns from data collected at the point of care and applies the lessons for patient care improvement, healthcare professionals will continue to be the linchpin of the front lines, assessing the needs, directing the approaches, ensuring the integrity of the tracking and the quality of the outcomes, and leading innovation. However, what these practitioners will need to know and how they learn will dramatically change. Orienting practice around a continually evolving evidence base requires new ways of thinking about how we can create and sustain a healthcare workforce that recognizes the role of evidence in decision making and is attuned to lifelong learning. Presentations throughout the workshop revealed concern from many different sectors on how to best fill the pipeline with students dedicated to building and applying an expanded evidence base for health care and establish a culture that encourages collaboration across the spectrum of care delivery and health professions. This session explored three of the many conduits for educating health professionals: existing and emerging decision support, formal educational requirements, and continuing education programs. In addressing this broad topic, speakers raised a wide range of issues and emphasized the pressing need for culture change throughout the healthcare system.
In the first paper, William Stead discusses the challenges that confront the health professional in acquiring the knowledge relevant to providing individualized care to patients. With the expansion of genomics and proteomics, he predicts a one- to two-order-of-magnitude change in the
amount of information needed for clinical decisions—far exceeding the capacity of the human mind. The electronic health record (EHR) will help health professionals accommodate this overload and deliver evidence-based, individualized care. In such a system, EHRs would change the practice ecosystem by making learning continuous, with clinical practice augmented by “just-in-time” access to information and curricula. In addition, when paired with clinical informatics tools, EHRs can support a number of learning strategies ranging from identification of variation in care, to hypothesis generation, to phenotype-genotype hypothesis testing. Achieving this potential will require a completely new approach, entailing discontinuous changes in how we define the roles of health professionals and how we learn.
Mary Mundinger of the Columbia School of Nursing then outlines some of the challenges in health professions education with regard to orienting training around an evolving evidence base. Grounding the teaching approach in evidence and adopting translational research as a guiding principle can lead to a continuous cycle in which students and faculty engage in research, implementation, dissemination, and inquiry. Yet most institutions find considerable variance in the level of integration of evidence into education across health professions. While intellectual engagement with evidence-based practice is evident in faculty publications, it often has not yet found its way substantially into the curriculum as a framework for training. In part, educational efforts always lag behind academic practice, but changes in the culture of medical practice are needed to ensure that educating health professionals about the benefits and methods of bringing evidence to their daily practice actually produces a skill that is recognized and utilized in the healthcare setting.
In the chapter’s final paper, Mark Williams discusses the shortfalls of current continuing medical education (CME) efforts and urges a shift to a knowledge translation approach that is integrated with practice and occurs on a daily basis. He notes that change is needed not only in the content and approach of CME but also in the culture of medical practice, advocating for a shift toward collaborative teamwork and increased cross-departmental collaboration, coupled with incentives for change and the provision of tools to facilitate such change.
CLINICIANS AND THE ELECTRONIC HEALTH RECORD AS A LEARNING TOOL
William W. Stead, M.D.
Vanderbilt University
The electronic health record is one key to a shift to systems approaches to evidence-based care that is nonetheless individualized. EHRs enable
change in the practice ecosystem (Stead 2007). They change roles and responsibilities, what the clinician needs to know, and how the clinician learns, while providing a new source of information. These changes are discontinuous, not an incremental improvement in what we do today. Hence, conversations about using the EHR are not generally focused on issues of providing evidence to, or generating evidence from, our current practice processes. Yet let us consider nine ways the EHR and clinical informatics tools could potentially be used to generate and apply evidence: using billing data to identify variability in practice; EHR data to direct care; EHR data to relate outcomes back to practice; EHR data to monitor open-loop processes; decision support systems for alerts and reminders within clinical workflow; decision support systems for patient-specific alerts to change in practice; decision support systems for links to evidence within clinical workflow; de-identified EHR data to detect unexpected events; and de-identified EHR and Biobank data for phenotype-genotype hypothesis generation. Illustrative examples will be drawn from how Vanderbilt uses informatics, coupled with electronic health records, to support learning in clinical workflow and population management.
Call for a Discontinuous Change in How Clinicians Learn
Figure 7-1 depicts the current medical decision-making model. The clinician is an integrator, aggregating information from the patients and their records with biomedical knowledge, recognizing patterns, making decisions, and trying to translate those decisions into action. Cognitive research has shown that the human mind can handle about seven facts at a time in a decision-making process (Miller 1956; Cowan 2000). We are bumping up against that limit today. This cognitive overload is one of the
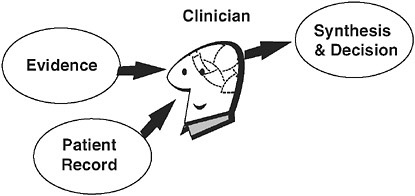
FIGURE 7-1 The decision model of the 1900s.
reasons we see the overuse, underuse, and misuse in health care that the Institute of Medicine (IOM) has highlighted in the Quality Chasm Series. This overload will get worse by one or two orders of magnitude as biomedical research turns functional genomics and proteomics into clinically useful information. We need a new decision-making model to deliver reproducible quality in the face of increasingly rich information sources.
Figure 7-2 depicts a possible alternative that emerged during Vanderbilt University Medical Center’s strategic planning process (Vanderbilt Medical Center September 2005) and the visioning phase of the National Library of Medicine’s long range planning process (NLM Board of Regents, National Institutes of Health 2006). Basically we envision a personal health knowledge base. This new resource would be much more than the personal health records emerging today. It would be a pre-computed intelligent integration of the individual’s health information, together with the subset of biomedical evidence relevant to that individual, presented in a way that lets the clinician and the patient—with very different learning levels and learning styles—make the right decisions. Such a model changes what the clinician needs to know to perform. For example, factual recall becomes less important and coaching skills become more important.
We also envision a change in how we learn. In 1973, I was taught in medical school a defined body of biomedical information, “just in case” I needed it. I was also trained as a scientist so that I could discover and learn through reading and practice. I was tested on the body of knowledge and credentialed as “knowing enough” through my degree, my license, and my boards in internal medicine and nephrology. If you think about it, you will realize that we can not achieve acceptable healthcare quality with that ap-
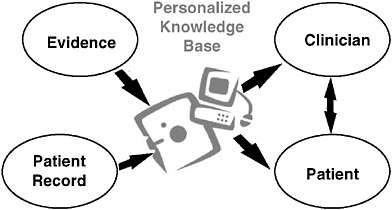
FIGURE 7-2 A possible model for the 2000s.
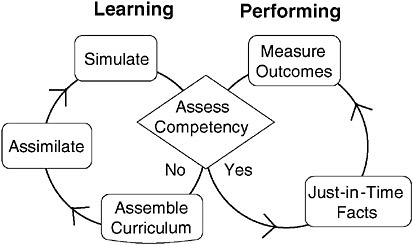
FIGURE 7-3 Continuous learning during performance.
proach to learning. It is not just that we have more biomedical information than anyone can learn or keep up with, even with ever-increasing specialization and a fifth year of medical school. The bigger problem is the variability in practice that comes from letting individuals learn, one by one, from their practice. Process reproducibility is a key to ensuring quality.
The clinician of tomorrow should be trained as a pilot to “fly” a system of care. In such a system, learning would be continuous and augmented with “just-in-time” access to information in clinical workflow. Credentials would be competency-based, reflecting current information about the individual’s learning record and outcomes. Figure 7-3 depicts how a systems approach to learning might work. At its center is assessment. The system would decide if the clinician knows what she needs to know to do what she is going to do next. If the answer is yes, based on her learning and outcome records, she proceeds to perform the clinical task (right-hand circle). Her knowledge of how to use facts is assisted by computer recall of details, such as specific drug-drug interactions. Electronic records track the patient’s progress and provide feedback regarding the effectiveness of the “system” and the clinician. After each cycle of clinical performance, her competency is reassessed. She flips into the learn cycle (left-hand circle) whenever additional knowledge or improvement is needed. The learn cycle begins by assembling a targeted curriculum using tools such as the Vanderbilt University School of Medicine’s KnowledgeMap (Denny et al. 2003). Next she reads and assimilates the information. Finally, she uses simulation to test understanding and technical skill. The simulator pushes her past her
limit of competency, ensuring individual understanding of that boundary. The simulator takes her back to the assessment point. If knowledge and skill are adequate, she flips back over into the performance mode. If not, she repeats the learn cycle.
Electronic Health Records as Learning Tools
Box 7-1 presents a framework of nine ways the EHR and clinical informatics tools can be used to support aspects of learning. It is a “splitter’s” view to point out the many data sources available and the “niche” of various tools. Certain learning strategies can be supported by billing data, others take full EHR, others take decision support systems, others take deidentified EHR data or a combination of de-identified health record and a bio bank.
The easiest step is to use billing data to identify variability in practice. You can use the data for a practice group to help members identify where they have variability. At Vanderbilt, we started with a top-to-bottom sort of range in resource utilization by Diagnosis-Related Group (DRG) or procedure code. Since this measures intragroup variability, it avoids the comparable population challenge of external benchmarks. We then got the group around the table, helped members look at areas where they had high variability, and asked what they wanted to do most of the time. We reflected these decisions in collaborative care pathways. In many cases, we took out
|
BOX 7-1 A Splitters View of EHR Data and Tools to Support Learning Billing Data
Electronic Health Records
Decision Support Systems
Decision Support Systems
|
as much as 40 percent of the work, while decreasing morbidity and mortality (Koch, Seckin, and Smith 1995). So utilizing just billing data—which almost everyone has today —we have seen significant improvement.
Full EHRs are more complicated and more powerful. For the purposes of this discussion, uses of EHR data can be separated from uses of the clinical workflow software that is commonly bundled in the purchase of an electronic medical record system. EHRs provide ready access to data about the patient, freeing up the clinician’s mind during direct care to focus on synthesis and pattern recognition. In addition, they provide a hypothesis generation resource as a “free” by-product of care. For example, the Duke Cardiovascular Databank (Rosati et al. 1975), begun in the mid-1970s, was probably the first large-scale case of using the computer as a time-lapse camera to tie clinical outcome back to the practice that produced it. Baseline data were captured for each patient admitted with cardiovascular disease, the results of their studies and procedures were entered, and a research team added outcome data with long-term follow-up. The Databank led to early ambulation post-myocardial infarction. Before the Databank, patients were put on prolonged bed rest. A query to the Databank showed a patient would not have complications if they did not occur in the first few hours. This “dry lab” hypothesis was then tested through a targeted controlled trial (McNeer et al. 1978). It is unlikely this finding would have been reached so quickly with conventional trails alone. Finally, data in the EHR can provide the feedback to trigger an external monitor of an open-loop process. Open-loop processes operate without internal feedback for real-time model adaptation. If an open-loop process makes frequent status reports, an external monitor can intervene if the status moves out of an acceptable range. Consider the requirement to administer antibiotics in a fixed time to patients presenting with pneumonia. One approach would be to force every clinician to use a programmed work process for every patient. This branching logic approach works at a microsystem level where a decision situation needs to be handled the same way over and over. By contrast, patients, who may ultimately turn out to have pneumonia, present in many ways and to all parts of the health system—a macrosystem problem. A better approach combines the open-loop process of the human clinician with a real-time event monitor. The clinician would be free to obtain data and recognize patterns. The monitor would check all records to see if recently arriving patients have been ruled in or out of the pneumonia population. If this event has not occurred within a specified period, the monitor could intervene to notify the clinician to make a decision whether or not the probability of pneumonia justified proceeding with administration of an initial dose of antibiotic.
Decision support systems, when informed by data from the EHR, permit alerts and reminders in clinical workflow as a helpful check on memory.
For example, the clinician is alerted to an allergy to a drug being ordered or reminded to order levels of aminoglycosides. Patient-specific alerts to information about a recommended change in practice can take the next step by supporting learning at a “teachable moment.” For example, the pharmacy and therapeutics committee at Vanderbilt recommends use of cephepime instead of ceftazidime for antipseudomonal treatment. When a clinician begins to order ceftazidime, a web page comes up with the recommendation, radio buttons pre-computed as to how to apply the recommendations to the particular patient, and links to the evidence leading to the recommendation. This approach closes the gap between new information, learning, and translation into practice. The direct link to the evidence provides the hook for processes to keep the recommendation up-to-date as information changes.
As we look to the future, we expect to aggregate de-identified extracts of EHR data on large populations and to use pattern discovery algorithms to detect unexpected events, an approach the National Library of Medicine is funding a dataset to test (Miller 2006). Derivatives of the electronic patient chart are being constructed—for example, converting all dates to an offset to a random start time. This approach maintains the temporal relationships within the record while removing reference points that might result in re-identification. Such abstracts might be aggregated on a large scale to detect more quickly problems such as the complications of Vioxx. It might then be possible to decrease pre-market testing by assuring robust post-market surveillance with systematic translation of problem detection into practice. As a next step, Vanderbilt is going live with a project to bank de-identified DNA samples of all patients who do not opt out. A one-way hash will link these samples to the synthetic derivative of the electronic chart, permitting phenotype-genotype hypothesis testing.
Challenges to Achieving the Potential
The potential use of EHRs as a tool for learning has been clear for decades. I include a number of older citations to underscore the maturity of many of these ideas. Until recently, the underlying information technology did not scale up to handle the amount or complexity of biomedical information. Such technical limits are behind us. Google shows the ease with which information can be aggregated from across the globe and made accessible. A number of challenging problems remain to work through. Examples include how to authenticate a patient to his or her record without requiring yet another identifier; how to support both confidentiality and access; and how to achieve interoperability for core items such as allergies while enabling access to data that are too highly dimensional to be regularized;
and so forth. However, these informatics challenges are not the major rate-limiting steps at this juncture.
Our capacity to envision a new way of working and to manage the transition is the rate-limiting step. The changes to systems approaches to care and learning are discontinuous. By discontinuous, I mean that we cannot achieve the ultimate potential by fixing aspects of our current health non-system. The goal is a completely new approach. People’s roles and responsibilities, the process, and the technology all need to change. These changes must be coordinated and take place in steps that can be accommodated within the current non-system while leading to another system. At the end of the journey we will see quite different professions, credentialing, decision-making strategies, et cetera.
The Institute of Medicine has taken a leadership role in highlighting the quality problems inherent in today’s health non-system and in calling for systems approaches to care. To date its reports recommend how to cross the chasm through a set of targeted fixes such as adoption of information technology or addition of certain competencies to the health science curriculum. It is not likely that we can achieve the discontinuous change in how we provide care without equally discontinuous changes in our definitions of professional roles and how we learn. We need to develop pictures of alternative visions of various combinations of roles, processes, and infrastructure that scale up to translate our scientific breakthroughs into the quality we want, at a price we can afford. Next, we need an actionable road map that shows how we can implement key aspects of these visions in the context of current reality.
EMBEDDING AN EVIDENCE PERSPECTIVE IN HEALTH PROFESSIONS EDUCATION
Mary Mundinger, Dr.P.H., R.N.
Columbia University School of Nursing
With rapid advances in medical knowledge, teaching health professionals to evaluate and use evidence in clinical decision making becomes one of the most crucial aspects of future efficacy and patient safety. This paper discusses Columbia University School of Nursing’s approach to teaching about evidence at the baccalaureate, masters, and clinical doctoral levels.
Fifty years ago in 1956, Sydney Burwell, dean of Harvard Medical School, said to his students, “Half of what you are taught as medical students will in ten years have been shown to be wrong. And the trouble is none of your teachers knows which half” (Sackett et al. 2001). What was once generally considered conventional care is no longer acceptable; many think of it as undisturbed ignorance. In 1985, Skolnick, in Medicine and
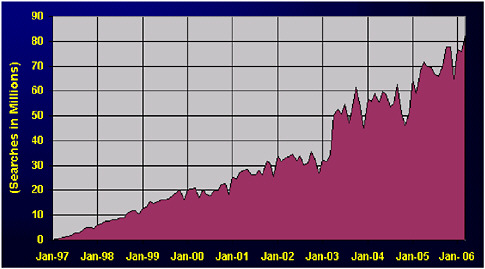
FIGURE 7-4 Medline searches.
SOURCE: NLM: www.nlm.nih.gov/bsd/medline_growth.html.
Law noted, “Failure to search the appropriate scientific literature is an obvious breach of the broader duty to perform at the level of knowledge and practice in a … clinical specialty” (Skolnick 1985). Searching the literature to guide practice is clearly a necessary first step, but only when sophisticated search and analysis skills are utilized. Evidence to direct practice is being sought more consistently as the most recent National Library of Medicine (2006a) graph on 10 years of database searches illustrates (Figure 7-4). But stronger educational efforts related to evidence assessment are needed to equip healthcare professionals with the tools and skills to continually bring the best evidence to bear on practice.
In thinking about teaching students to become evidence-based clinicians, there are three main questions. First is: Why teach it? This question has been quite cogently analyzed in this workshop; essentially, it is the best way to provide the best outcomes for our patients. The previous chapter addresses how it helps patients participate in treatment decisions and adopt best self-care practices. This is particularly important as we move beyond the era in which we expected passive patient compliance with physician-prescribed regimens. In addition, evidence-based practice is the way to achieve highest value with available resources. There isn’t enough money to deliver the kind of care we are delivering now and still achieve the best possible outcomes. To improve the value of care we need to rely more on evidence.
The second question is: What do we do? It is not enough to give one
course and expect the concept of evidence-based practice to take hold. At Columbia University School of Nursing, we found that we needed to examine the context and the core of how we engage in practice to get evidence-based practice fully integrated in our curricula. Our approach was to adopt a new guiding principle: translational research would inform everything we did. By this we do not mean the linear bench-to-bedside approach. Our school, in addition to having a program for college graduates to enter nursing through a second degree program, also has 12 master’s programs in a number of clinical specialties and a doctoral research program, and we were considering instituting the first clinical doctoral program in the country. The doctor of nursing practice degree uses a teaching approach entirely grounded in evidence. Translational research is essentially a process that moves from conventional data collection, analysis, conclusions, and recommendations to actively incorporating the new evidence into practice (the hardest part), then to institutionalizing the change into policy (regulatory, reimbursement, liability), and then to ensuring that the new ways of doing things are taught to students. New research questions emanating from the new “state of the art” practice close the circle.
The beauty of this continuous circular process is that it does not stop after research changes practice. In our school, both research and clinical faculty actively engage in this process. Research doctoral students select studies emanating from faculty practice themes, and clinical doctoral students engage in the process of generating data for faculty research studies. Some examples follow. Dr. Suzanne Bakken’s research on developing informatics-based approaches to patient care that enable data collection, secondary use of datasets, and building of evidence across evidence-based practice studies also includes teaching data collection to B.S. students using personal digital assistants. Doctoral research studies based on questions arising from faculty clinical practices include such topics as adult liver donation; testing acupressure for relief of AIDS-related nausea and vomiting; development of a fall and injury risk assessment instrument; diabetes treatment in the Hispanic population; and breast cancer screening practices. Changes in clinical practice guidelines resulting from faculty research include the cardiac effects of Parkinson’s drugs and depression screening for children. Policy change as a result of clinical research and faculty practice includes a new hospital emergency room (ER) discharge policy for pregnant women. The interaction and active progression of translational research is a vibrant core of faculty scholarship.
Students are taught to understand different levels of evidence and to distinguish among good and bad studies and good studies with flawed conclusions or wrong recommendations. If practitioners cannot make these distinctions, the literature as an instrument of putting best evidence into practice will be insufficient. Although understanding the five levels of evi-
dence is the first step in becoming an evidence-based clinician, students are also taught to focus on the critical relationship between design methods and conclusions and recommendations and to understand how the design of a trial might affect the ultimate strength of its recommendations. Simply having the gold standard of a randomized control trial is simply not enough. Strategies to analyzing and implementing evidence vary. Education of doctoral research students concentrates on analyzing evidence; training of clinical doctoral students focuses on implementing evidence.
The third and final question is: How will we get where we want to go? Clearly a main enabler is the electronic health record, which will be fundamental for data mining, evidence application, and evidence generation. Although the Institute of Medicine and some of the most sophisticated clinicians in the country have long advocated EHRs, we are still far from achieving broad utilization. We must help students become sophisticated readers of the literature and help them find systematic ways to phrase questions and collect data. We need to ensure that our advanced research courses teach the data-mining techniques that practitioners and the country will need to move forward.
Columbia’s experience—as an academic health center that includes medical, public health, nursing, and dentistry—is illustrative. Collectively we have about 2,500 students, and the schools are highly integrated. More than half the public health faculty are physicians; many faculty members in each school have dual appointments; in nursing, 40 clinical faculty have joint appointments in medical school departments. Yet the curricula of these schools are quite disparate. We may be a collegial, academic campus, but we could not be more different in how we teach evidence-based practice. In medicine, evidence-based practice is incorporated into the curricula of three intensive courses that span one month in the fourth year of training. In this one month, students are introduced to the clinician scientist role, the practicing physician role, and biomedical informatics. Nationwide, physicians are clearly involved in evidence-based practice publications and research, but medical school curricula do not reflect this. In dentistry, evidence-based practice is introduced in the first semester, first year with courses in “Scientific Inquiry and Decision Making in Dentistry” and “Informatics, Epidemiology, Ethics, and General Dental Practice.” Again while this school is cognizant of bringing discussion of evidence into its curricula, there isn’t a strong evidence-based component that is identifiable. In public health, elements of evidence-based medicine are integrated into courses on “Informatics” and “Health Information Technology.” Both the public health and the medical schools have National Institutes of Health (NIH) grants on evidence-based practice and clinical decision making. Columbia also has the best bioinformatics department in a medical school. The intellectual property for evidence-based practice certainly is present and is seen
in faculty research publications, but it has not yet found its way into the curricula as a very specific way to train students. Education lags somewhat behind academic practice, and this may be true nationally as well.
In nursing, we have three separate curricula to train students for our different-level nursing programs. To educate college graduates to become nurses and earn a second B.S. degree we require a research utilization course. Master’s degree students take a course titled “Assessing Clinical Evidence” (the ACE course). For our bachelor’s nursing program we help nurses understand established evidence-based care, understand the science and how care protocols evolve, and how to know when to deviate from protocols because of patient responses. In the master’s advanced practice nursing program, we are helping our students to learn, distinguish, and apply levels of evidence to literature analysis.
In the doctoral nursing program, students learn how to use evidence, how to assess literature using informatics, and how to collect data and generate evidence. They gain an understanding of research design and methods in relation to study outcomes and conclusions, the systematic use of data to adopt and generate evidence, and interdisciplinary and cross-site collaboration. In our research doctoral program we offer several biostatistics and research design and methods courses; our clinical doctoral students take some of these same courses and also “Synthesizing, Translating, and Integrating Clinical Evidence.” The research doctoral graduates, training to be principal investigators, will ultimately be designing and conducting trials; the clinical doctoral graduates will need to know when and how to use evidence.
One particularly important point is that, although Columbia provides this kind of training, often it is not utilized because of restrictions on the nurse’s role in providing patient care. If we allowed nurses the opportunity to employ evidence-based practice methods as part of their work, we might not have a nursing shortage. Nurses are trained to look at evidence, think critically and intelligently, and make decisions based on their knowledge, but they are not being allowed to do this in their jobs.
The three-tiered curricula progression (B.S.-M.S.-doctorate) at Columbia University Nursing School prepares graduates to distinguish and use evidence to guide practice. Applying the evidence, however, is very much limited by scope of practice. Hospital nurses, for example, are expected to abide by hospital policy, established nursing protocols, and physician orders. Professional responsibility requires the nurse to report any patient responses that could contraindicate following current protocols or orders. However actively initiating or changing regimens is not expected of nurses, and often not tolerated, even if the scientific evidence validates those actions. This kind of practice is deeply unsatifisfying to knowledgeable professionals. Shift work with its shared accountability, but without any authority
for nurses, and a hierarchy that negates the nurse’s decision making are all factors contributing to a broken system. In the 1970s, models with nursing accountability, M.D. partnerships, and flexibility in devising care regimens were developed. Hospitals found them too complex in the context of industrial organization, and physicians found giving up their total and easy determination of ordering care irksome. So the models disappeared (Mundinger 1973).
Nurses prepared as nurse practitioners with a master’s degree in a specialty (adult primary care, for example) have more authority and can utilize their decision-making ability in a more independent way. This level of practice is again limited by regulation—this time at the state level rather than institutional level. Each state determines the level of independence (full, shared with a physician, or supervised by a physician) and prescriptive authority (full, only certain categories of drugs, or cosigned by a physician). Many qualified nurse practitioners shun opportunities in states where practice is limited.
Doctoral-level clinicians are those educated for more sophisticated practice and decision making, and regulatory bodies (hospitals for admitting privileges and states for prescribing and Medicaid reimbursement) have not yet made distinctions between them and M.S.-prepared practitioners. Regulation almost always lags behind practice. The Balanced Budget Act of 1997 was an exception in authorizing master’s-prepared nurse practitioners for direct Medicare reimbursement in any site (United States Congress 1997), even though nurse practitioners are not educated for care of hospitalized patients. The doctor of nursing practice degree catches up with this regulation, educating nurses to care for patients across sites and over time—a true professional model of responsibility that, if the principles of educational philosophy are implemented, will be fully dedicated to advancing the application of evidence.
Several changes must occur in order to fully utilize the professional nursing workforce and ensure application of best evidence. Hospitals must adopt models of care that recognize nursing decision making. This is true for bedside nurses and for doctoral-level clinicians who admit and order care for their patients. Hospital medical boards must also change their bylaws and provide accessible consultations, as well as privileges for doctoral-level clinicians. Second, state regulations must be changed to standardize and authorize nurse decision making in outpatient settings, including increasing prescriptive authority, decreasing M.D. oversight, and nonrestrictive reimbursement from Medicaid. Third, private insurers and Medicare must recognize doctoral clinicians and grant them parity with physicians. Nurses have consistently demonstrated thoughtful responsibility for the care decisions they make, and a number of studies attest to this quality of care and outcomes (Mundinger et al. 2000). Nurses will be attracted
to positions where their competency is operationalized, and the nursing “shortage” would disappear.
The changes enumerated above all relate to increasing the focus by nurses on generation and application of evidence. Especially important to optimizing evidence-based practice is to ensure that nurses can also develop evidence. Given the time, hospital nurses are particularly attuned to nuanced changes in a patient’s condition and see themes of patient response to a variety of interventions. With a way to chart patient responses and to indicate their nursing-specific observations and treatment, they could be the frontline voice in guiding scientific evaluation of new or emerging evidence. The EHR can be developed to capture these data, but its utility depends on accurate input—a function that is often the responsibility of nurses.
Teaching baccalaureate nursing students how evidence is generated will spark their interest as graduates to play an important role. Master’s-prepared nurse practitioners generate evidence through the more complete and comprehensive data they collect on their own primary care patients. Knowing how to collect data and publish their practice perspectives will add to the science of given clinical conditions or context of care. Doctoral clinicians will be the major source of identifying emerging patterns or new insights of care outcomes. Researchers will use these carefully collected data to carry out analytical outcomes leading to new evidence-based practice guidelines.
KNOWLEDGE TRANSLATION: REDEFINING CONTINUING EDUCATION AROUND EVOLVING EVIDENCE
Mark V. Williams, M.D.
Emory University School of Medicine
It’s an incredibly simple idea and one that is blindingly obvious to most lay people. … Assess the existing evidence and concentrate on the reliable stuff.
Iain Chalmers, 1996
Dramatic increases in the generation of new medical knowledge practically overwhelm practicing clinicians; in fact, entire books have been published on how best to manage all the “evidence” available (Rennie and Guyatt 2001; Strauss et al. 2005). Each year, thousands of clinical trials are added to the already voluminous research literature in hundreds of journals. For example, to keep up with germane developments in the field of internal medicine, physicians now face the prospect of learning from thousands of relevant articles being published each month. Not surprisingly, numb
resignation may supplant the desired eager and interactive approach to lifelong learning. Recognizing that learning all the evidence is impossible, some experts recommend that healthcare providers develop information management skills while allowing others to help identify, review, and summarize salient and valid clinical information (Slawson and Shaughnessy 2005). Even with successful transfer of this responsibility for knowledge organization, clinicians must still know how to access and deliver recommended advances in clinical care and systems of delivery. Unfortunately, physicians are doing a mediocre job of delivering recommended care to patients, with one well-designed study showing success about half the time (McGlynn et al. 2003); national reports confirm this though there are signs of improvement (AHRQ 2003).
To foster the dissemination of innovation and application of new evidence, clinicians must undertake efforts to ensure that clinical practice reflects the best current evidence. While the best evidence should always inform medical decisions and health choices, simply providing more bedside evidence may only worsen the informational plight of busy clinicians. Continuing education after initial training plays an essential role in allowing them to apply such new evidence effectively to patient care. The standard approach to postgraduate physician education, traveling to a continuing medical education course and listening to presentations in a classroom setting, is endorsed by medical societies, supported by pharmaceutical companies, and required by many state licensing boards. Yet, previous systematic reviews have documented for decades that standard CME is ineffective at changing physician behavior and translating proven interventions into practice (Haynes et al. 1984; Davis et al. 1995; Davis et al. 1999). While typical didactic sessions unsuccessfully influence practice, interactive workshops (e.g., role playing, case discussion, practicing skills) do seem to generate moderate changes in performance and offer hope (O’Brien et al. 2006). A variant of CME, continuous professional development, has been advocated by the American Board of Internal Medicine and attempts to incorporate adult learning principles and reflection (Baron 2005), but its economic value is yet to be determined (Brown et al. 2002). Additionally, it employs self-directed learning as a principal method, but physicians have limited ability to self-assess their own competency (Davis et al. 2006). All of these approaches to continuing education tend to focus on the physician to the exclusion of other members of the healthcare team.
Demands from patients, insurers, and regulatory agencies such as the Joint Commission on Accreditation of Healthcare Organizations (JCAHO) and the Centers for Medicare and Medicaid Services (CMS), combined with increased emphasis on pay for performance to improve overall quality of care, mandate changes in methods for CME (Rowe 2006). Popular magazines such as Reader’s Digest suggest that hospitalization is equivalent
to gambling with your life and portray patients as feeling suspended above a chasm between known medical evidence and the care actually provided (IOM 2001). Healthcare professionals do not have it any better; as a cover of Time magazine proclaimed in 2006, doctors also fear for the safety of their family members and themselves when they enter a hospital. The overarching message is to stay out of the hospitals if you value your life. Despite the lack of certainty regarding whether or not pay for performance will actually improve care (Petersen et al. 2006), healthcare insurers will increasingly use it to control costs, attempting to align payment incentives to promote better-quality care by rewarding providers who perform well (IOM 2007). For hospitals and healthcare systems to succeed in this new environment, involvement of the entire healthcare team will be necessary to utilize innovative approaches and take advantage of evolving evidence, including both scientific advances and learning how to deliver existing effective therapies more consistently. Of note, governmental healthcare leaders support the reinvention of CME and linking it to care delivery (Clancy 2004).
Knowledge translation may represent an approach that combines the right tools with involvement of the entire healthcare team to yield truly effective CME (Davis et al. 2003). This approach moves CME to where we deliver care, it targets all participants (patients, nurses, pharmacists, and doctors), and the content is based around initiatives to improve health care. Such a model marks an important shift toward translating evidence into practice and crossing the current perceived quality chasm, while also promoting an interdisciplinary approach. The theoretical underpinnings to this approach maintain not only that we make physicians and other healthcare providers aware of the evidence, but also that it is important that adoption occurs, and adherence is encouraged through thoughtful incentives as well as reminders to accomplish these goals. A review of the various components of knowledge translation with comparison to standard CME elucidates the advantages.
First, this approach moves CME out of the classroom into the actual setting of care delivery. By focusing on changing participants’ behavior at the site of care (e.g., ordering and giving influenza vaccinations to eligible patients, measuring and treating pain post-operatively) and providing tools or toolkits to facilitate such changes, best evidence can be delivered where it is needed. The Society of Hospital Medicine, the medical society for hospitalists, now promotes the use of such tools on its web site (http://www.hospitalmedicine.org) to enhance patient safety and the quality of inpatient care, and examples of their use are being published (McKean et al. 2006). All that is lacking is the link to CME credits.
Knowledge translation also has a different target than the standard CME focus on physicians. This model can involve the entire healthcare
team and group learning (Davis et al. 2003). It may even include patients in addition to nurses, pharmacists, and other healthcare providers. By centering the content on evidence that improves patient health care, instead of purely clinical or pathophysiologic material (e.g., the need and indications for administering an angiotensin-converting enzyme [ACE] inhibitor to a patient with heart failure vs. the mechanism by which the medications act on the renin-angiotensin pathway), there is a more direct impact on outcomes. Finally, knowledge translation pursues an interdisciplinary approach with inclusion of all relevant staff in the care process, instead of the teacher determining the goals of instruction, and employs a model that guides implementation and evaluation of interventions. This broadens expertise to include the fields of informatics, organizational learning, social marketing, and quality improvement (Davis et al. 2003). Although mainly still in the conceptual phase, the continuing education model of knowledge translation should be advanced by recent changes introduced by the Accreditation Council for Graduate Medical Education (ACGME) (Leach and Philibert 2006).
With the formation of a Committee on Innovation in the Learning Environment, the ACGME seeks to achieve implementation of the expectation that physicians be taught six general competencies: patient care, medical knowledge, practice-based learning and improvement, interpersonal communication skills, professionalism, and systems-based practice (Batalden et al. 2002). This forces a shift from residents’ learning predominantly through clinical experiences to also include an emphasis on mastering and leading systems that deliver safe care (Leach and Philibert 2006). Fortunately, multiple quality improvement strategies, including some with robust research supporting their efficacy (e.g., audit and feedback), are available to be utilized in this effort (Stein 2006).
There are some anticipated challenges with the knowledge translation approach. We will need monitoring systems to support and document provider participation in these activities and evidence on specific mechanisms that promote learning and change. Yet, similar to healthcare delivery, the future of CME needs to function through collaborative teamwork in which we pull physicians out of their autonomous role and into collaboration with nurses, pharmacists, occupational and physical therapists, and dieticians. Achieving team-based, patient-centered, evidence-based care as the objective might occur at the local level and even nationally. The Institute for Healthcare Improvement attempted this with its 100,000 Lives Campaign and the newer 5 Million Lives Campaign (http://www.ihi.org/IHI/Programs/Campaign/). Collaborative projects by large organizations such as the Hospital Corporation of America (HCA; a large corporation with almost 200 hospitals) also have the potential to impact large numbers of patients profoundly. For example, HCA is partnering with expertise from
an academic medical center (Vanderbilt) on a project funded by the Agency for Healthcare Research and Quality (AHRQ) to improve care and reduce errors in intensive care units (ICUs) across many of their hospitals. Yet, neither of these efforts includes official CME credits in its implementation. Forging this link might dramatically augment their success because physicians would have the added push from their state medical boards and accreditation agencies to become involved.
As mentioned earlier, impending pay for performance (P4P) initiatives should also facilitate adoption of the knowledge translation approach, even outside academic medical centers (Rowe 2006). The old model of healthcare reimbursement follows the simple principle that essentially the more you do, the more you make. A new model of pay for performance could help us move beyond this, and in an important sign, CMS is considering lowering payments for cases with medical errors and tying the National Quality Forum’s “never events” to lower reimbursement. Tying knowledge translation to mandated CME seeking to accomplish this goal may powerfully drive healthcare systems both to apply evidence and to generate it through healthcare delivery. Instead of physicians traveling to a distant location (typically a vacation resort) for CME, healthcare delivery can become a learning experience as they experience practice-based learning with their colleagues. This will require consistent integration with care delivery occurring everyday. If data can be collected simultaneously with this implementation, we can also learn and have practice-based evidence to guide future practice improvements (Horn 2006). Not only would healthcare professionals be learning, but the entire system would also learn how to optimize care delivery.
REFERENCES
AHRQ (Agency for Healthcare Research and Quality). National Healthcare Quality Report. 2003. Available from http://www.ahrq.gov/qual/nhqr05/nhqr05.htm. (accessed December 19, 2006).
Baron, R. 2005. Personal metrics for practice: how’m I doing? New England Journal of Medicine 353:1992-1993.
Batalden, P, D Leach, S Swing, H Dreyfus, and S Dreyfus. 2002. General competencies a accreditation in graduate medical education. Health Affairs (Milwood) 21:103-111.
Brown, C, C Belfield, and S Field. 2002. Cost effectiveness of continuing professional development in health care: a critical review of the evidence. British Medical Journal 324:652-655.
Clancy, C. 2004. Commentary: Reinventing continuing medical education. British Medical Journal 4:181.
Cowan, N. 2000. The magical number 4 in short-term memory: a reconsideration of mental storage capacity. Behavioral and Brain Sciences 94:87-185.
Davis, D, M Thomson, A Oxman, and R Haynes. 1995. Changing physician performance: a systematic review of the effect of continuing medical education strategies. Journal of the American Medical Association 274:700-705.
Davis, D, M O’Brien, N Freemantle, F Wolf, P Mazmanian, and A Taylor-Vaisey. 1999. Impact of formal continuing medical education: do conferences, workshops, rounds, and other traditional continuing education activities change physician behavior or health care outcomes? Journal of the American Medical Association 282(9):867-874.
Davis, D, M Evans, A Jadad, L Perrier, D Rath, D Ryan, G Sibbald, S Strauss, S Rappolt, M Wowk, and M Zwarenstein. 2003. The case for knowledge translation: shortening the journey from evidence to effect. British Medical Journal 327:33-35.
Davis, D, P Mazmanian, M Fordis, R Van Harrison, K Thorpe, and L Perrier. 2006. Accuracy of physician self-assessment compared with observed measures of competence: a systematic review. Journal of the American Medical Association 296:1094-1102.
Denny, J, J Smithers, R Miller, and A Spickard 3rd. 2003. “Understanding” medical school curriculum content using KnowledgeMap. Journal of the American Medical Informatics Association 10(4):351-362.
Haynes, R, D Davis, A McKibbon, and P Tugwell. 1984. A critical appraisal of the efficacy of continuing medical education. Journal of the American Medical Association 251:61-64.
Horn, S. 2006. Performance measures and clinical outcomes. Journal of the American Medical Association 296:2731-2732.
IOM (Institute of Medicine). 2001. Crossing the Quality Chasm. A New Health System for the 21st Century. Washington, DC: National Academy Press.
———. 2007. Rewarding Provider Performance: Aligning Incentives in Medicare, Pathways to Quality Health Care. Washington, DC: The National Academies Press.
Koch, M, B Seckin, and J Smith Jr. 1995. Impact of a collaborative care approach to radical cystectomy and urinary reconstruction. Journal of Urology 154(3):996-1001.
Leach, D, and I Philibert. 2006. High-quality learning for high-quality health care: Getting it right. Journal of the American Medical Association 296:1132-1134.
McGlynn, E, S Asch, J Adams, J Keesey, J Hicks, A DeCristofaro, and E Kerr. 2003. The quality of health care delivered to adults in the United States. New England Journal of Medicine 348:2635-2645.
McKean, S, J Stein, G Maynard, T Budnitz, A Amin, S Johnson, and L Wellikson. 2006. Curriculum development: the venous thromboembolism quality improvement resource room. Journal of Hospital Medicine 1:124-132.
McNeer, J, G Wagner, P Ginsburg, A Wallace, C McCants, M Conley, and R Rosati. 1978. Hospital discharge one week after acute myocardial infarction. New England Journal of Medicine 298(5):229-232.
Miller, G. 1956. The magical number seven plus or minus two: some limits on our capacity for processing information. Psychological Review 63(2):81-97.
Miller, R. 2006. TIME (Tools for Inpatient Monitoring using Evidence) for Safe & Appropri ate Testing. NLM Grant 5R01LM007995-03.
Mundinger, M. 1973. Primary nurse role evolution. Nursing Outlook 21(10):642-645.
Mundinger, M, R Kane, E Lenz, A Totten, W Tsai, P Cleary, W Friedewald, A Siu, and M Shelanski. 2000. Primary care outcomes in patients treated by nurse practitioners or physicians: a randomized trial. Journal of the American Medical Association 283(1):59-68.
NLM Board of Regents, National Institutes of Health. 2006. Charting a Course for the 21st Century: NLM’s Long Range Plan 2006-2016. Available from http://www.nlm.nih.gov/pubs/plan/lrpdocs.html. (accessed April 4, 2007).
O’Brien, M, N Freemantle, A Oxman, F Wolf, D Davis, and J Herrin. 2006. Continuing education meetings and workshops: effects on professional practice and health care outcomes. Cochrane Database of Systematic Reviews 4.
Petersen, L, L Woodard, T Urech, C Daw, and S Sookanan. 2006. Does pay-for-performance improve the quality of healthy care? Annals of Internal Medicine 145:265-272.
Rennie, D, and G Guyatt. 2001. Users’ Guides: Manual for Evidence-Based Clinical Practice. Chicago, IL: American Medical Association.
Rosati, R, J McNeer, C Starmer, B Mittler, J Morris Jr., and A Wallace. 1975. A new information system for medical practice. Archives of Internal Medicine 135(8):1017-1024.
Rowe, J. 2006. Pay-for-performance and accountability: related themes in improving health care. Annals of Internal Medicine 145:695-699.
Sackett, D, W Richardson, W Rosenberg, and R Haynes. 2001. Evidence-Based Medicine: How to Practice and Teach EBM. New York: Churchill Livingstone.
Skolnick, M. 1985. Expanding physician duties and patients rights in wrongful life: Harbeson v. Parke-Davis, Inc. Medicine and Law 4(3):283-298.
Slawson, D, and A Shaughnessy. 2005. Teaching evidence-based medicine: should we be teaching information management instead? Academic Medicine 80:685-689.
Society of Hospital Medicine. Ongoing. Quality Improvement Tools. Available from http://www.hospitalmedicine.org/Content/NavigationMenu/HQPS/QualityImprovementTools/Quality_Improvement_.htm. (accessed December 18, 2006).
Stead, W. 2007. Rethinking electronic health records to better achieve quality and safety goals. Annual Review of Medicine 58(14):1-14.
Stein, J. 2006. The language of quality improvement: therapy classes. Journal of Hospital Medicine 1:327-330.
Strauss, S, W Richardson, P Glasziou, and R Haynes. 2005. Evidence Based Medicine. London, UK: Churchill Livingstone.
United States Congress. 1997. Balanced Budget Act of 1997. Washington, DC: Government Printing Office.
Vanderbilt Medical Center. 2005 (September). Strategic Plan for VUMC Informatics & Roadmap to 2010 September 2005. Available from http://www.mc.vanderbilt.edu/infocntr/IC_Strategic_Plan_05.pdf. (accessed April 4, 2007).






















