6
Global Systems for the Purchase and Delivery of TB Drugs
Paul Zintl of Partners In Health reported that in fall 2007, largely as a result of problems with GLC-approved projects around the world, the Working Group on Multidrug-Resistant Tuberculosis formed a Drug Management Subcommittee to help identify and quantify shortages of second-line drugs for such projects. The working group has worked with UNITAID, the Global Drug Facility (GDF) hosted through WHO, to resolve delivery and logistical issues. While the situation has improved, there is still a significant shortage of quality-assured drugs for GLC-approved projects. With the growth of diagnostic capacity and the increase in both projects and numbers of patients now being approved by the GLC, this shortage could be a real bottleneck.
The working group identified a combination of delivery and logistical problems. The GDF and the GLC were very understaffed to handle these problems, and the problems persist. But the real issue is the lack of consistent demand for quality-assured drugs. Suppliers are producing drugs on order because the demand is sporadic and unpredictable. In addition, once a project has been approved, actual enrollment often lags behind projected enrollment, and payments are slow.
As seen in Table 6-1, there has been substantial growth in the number of GLC-approved projects and the numbers of patients treated. In 2006, when WHO changed its global TB policy to recommend treatment of MDR TB patients under proper programmatic conditions, there were just over 5,500 patients enrolled in 32 approved projects. By 2007, there were 104 projects with 30,000 patients enrolled, including a rapid ramp-up in the
TABLE 6-1 Green Light Committee Projects and Patients, 2006–2009
African region—from 0 to 15 projects. The large number of projects and patients in Eastern Europe, which includes Russia and the countries of the former Soviet Union, is also notable.
Despite the growth in GLC projects, they represent only a tiny fraction of the more than 400,000 MDR TB cases estimated to occur each year, as shown in Figure 3-1 in Chapter 3. The vast majority of patients are being treated through non-GLC-approved projects under programmatic conditions that may not be ideal for treatment of MDR TB and with drugs that are not quality assured. As shown in Figure 3-1, in 2008 there were an estimated 500,000 MDR TB cases worldwide. Of these, only 12,000 were expected to receive GLC-approved treatment; 34,000 were expected to be treated with a non-GLC regimen; and the remainder represent new, untreated cases.
The non-GLC market for second-line drugs is substantial. IMS Health has given WHO and the Drug Management Subcommittee data that show quite robust sales of all second-line TB drugs. There are, for example, a large number of manufacturers of second-line drugs in Russia, both domestic and foreign. In the Pathway to Patients report, the TB Alliance also confirms a very large and growing market for second-line drugs (TB Alliance and the Global Alliance for TB Drug Development, 2007).
PROCUREMENT PROBLEMS
Zintl noted that the increased incidence of MDR TB has already led to a significant growth in the world supply of second-line TB drugs. Unfortunately, however, most of this increased production and consumption is of second-line drugs that are not quality assured.
As countries confront their epidemics of MDR TB, there will be a significant expansion of public-sector treatment programs, as well as growth in the government- and donor-sponsored purchase and procurement of second-line TB drugs. This expansion could help boost the desperately short supply of quality-assured drugs if efforts to stimulate demand for such drugs in MDR TB priority countries are successful. This is likely to happen only if the GLC/GDF approval and procurement processes are overhauled. The existing GLC approval and GDF/International Dispensary Association (IDA) procurement processes were, appropriately, designed for the pilot project era when DOTS-Plus1 was being evaluated, before WHO had revised its TB control policy calling for treatment of patients with MDR TB in the context of a strong DOTS TB control program. But the pilot project approval and procurement model is no longer appropriate, practical, or effective. The approach is too fragmented and has failed to achieve necessary scale. There is only one quality-assured supplier for each of the second-line drugs for GLC projects (except for one drug that has two quality-assured suppliers). All GLC-approved projects go through a single procurement agent, IDA, and there is only one source of supply. Such limited supply depth carries risks, as evidenced by a recent manufacturing bottleneck. Manufacturers of two key drugs shut down their plants—one because of a batch problem, the other because of factory expansion. As a result, deliveries of those drugs were put on hold for a considerable period of time, causing serious supply shortages and stock-outs in treatment projects.
THE DRUG QUALITY ISSUE
It is important that MDR TB treatment projects follow WHO treatment guidelines and that they use drugs that are quality assured. Poor treatment or treatment with poor-quality drugs will not cure patients and will ultimately generate XDR TB. For some countries and projects, the existing GLC/GDF approval and procurement mechanisms will still be adequate, but this will not be the case for other countries—notably those with the highest burdens of MDR TB. To address this shortcoming, countries could be enlisted to participate in a revamped system that would monitor their compliance with WHO treatment guidelines and the extent of their use of (or failure to use) quality-assured drugs. Not all quality-assured second-line TB drugs can be channeled through the GLC, nor do they need to be.
At the same time, calling for the existing GLC/GDF approval and procurement system to be revamped is not to minimize the seriousness of the challenge posed by nonstandard treatment protocols and poor-quality
drugs. Treatment and drug quality vary tremendously across programs and countries. The markets for second-line drugs in priority countries are large and growing rapidly; they are fragmented, and regulation and monitoring of these markets is nonexistent or inconsistent at best. The problem stems from largely insufficient insistence by countries that their MDR TB patients be treated with second-line drugs that are quality assured. In some cases, there are significant disagreements among regulators in different countries about what constitutes quality assurance.
A workshop participant from Management Sciences for Health asked whether suppliers face any risk associated with not participating in the prequalification process. Zintl replied that it should be made clear that the market is moving toward becoming totally quality assured through requests for national commitments from all high-burden countries. The expanding epidemic and increasing diagnostic capacity are creating a very attractive market for manufacturers, and there will be an incentive for both domestic markets and export production. If demand forecasting is reliable, companies such as Lupin Limited and Cipla will be eager to expand and to seek prequalification. But unless manufacturers see a growing market for quality-assured drugs—and, consequently, a declining market for drugs that are not quality assured—they will have no incentive to become prequalified. Thus if India, China, Russia, and the other priority countries continue to be willing to buy and treat patients with drugs of uncertain quality, it is the market for drugs of uncertain quality that will continue to grow, and manufacturers will see little return on investments in ensuring quality. Zintl added that in the short term, it may be necessary to sacrifice pricing objectives to achieve quality objectives, particularly in those high-burden countries where companies need a financial incentive to raise their quality standards.
Nacy asked whether there is a way to reward companies for bearing the expense of quality assurance. Zintl replied that there certainly is, and that such rewards are important interim steps. It is necessary to work with funders such as UNITAID to develop incentives. One such proposal is to provide, for example, a 25 percent premium over 2 years or to offer a minimum-purchase contract. The Center for Global Development, the Clinton Foundation, and others are already exploring such ideas.
Iain Richardson of Eli Lilly and Company noted the wide discrepancy between prices in local private markets, where many manufacturers are competing, and the prices of drugs provided through the GDF. To ensure that these companies produce drugs of high quality, it is necessary both to assure them of a good return on their investments in quality and to let them know that unless drugs are quality assured, they will not be able to be sold.
Need for Better Data on Drug Quality
Data on the quality of second-line drugs that have not gone through GLC prequalification are limited. Keshavjee noted that Management Sciences for Health studied this issue in Brazil—a market that is reasonably well regulated—and found that there have been problems with drugs. Nunn added that unpublished data from more than 10 years ago on first-line drug fixed-dose combinations show that many products, particularly from the Indian market, were substandard. Looking at the high rates of MDR TB in the former Soviet Union, one of the major hypotheses is drug quality. Cassell added that anecdotal evidence from Kazakhstan, for example, makes clear that substandard products are contributing to resistance and poor patient outcomes.
Sloutsky discussed what is really meant by “quality” and “assured.” The ultimate issue regarding the quality of an antibiotic drug is its potency. In package inserts, drug manufacturers do not provide data on exact potency but list the potency within a range of concentrations. This range can be rather wide, with the minimum and maximum values being significantly different. For example, a drug may be listed as active in the range of 0.5–20.0 micrograms per milliliter. When one is dealing with second-line drugs and working close to critical concentrations, each drug dilution is meaningful, and such a wide range does not help in assessing the real potency. Sloutsky suggested that, if the drug manufacturer’s package insert cannot be relied upon as a measure of quality assurance, this assessment must be conducted by an independent laboratory, and the test must be performed anonymously to ensure the integrity of the process. This testing can be requested by the GLC, the GDF, or other governing bodies, which can make it mandatory for drug manufacturers.
Nunn described current efforts to collect data on drug quality by looking randomly at TB drugs from various sites in different countries and measuring their active ingredients—similar to what was done with AIDS and malaria. But results from those studies are months away. Nunn added that much more data is needed, not only on first-line but also on second-line drugs, for which there are reasons to suppose the quality may be worse. Castro emphasized the need for a strong data collection effort going forward. He noted that intuitively, the contribution of poor-quality drugs to drug resistance appears clear, but data are needed to make an airtight case for the link—both baseline data and ongoing monitoring as progress is achieved. Cassell added that the private sector is making a strong commitment of effort and dollars to address the issue of counterfeit and substandard drugs as a result of contamination of heparin and other incidents. She suggested that this would be a fruitful area for collaboration between the private sector and the global health community.
Quality Enforcement
Observing that TB epidemics are spreading across borders, Jim Yong Kim of the Harvard School of Public Health and Harvard Medical School inquired about the possibility of the International Health Regulations (IHR) being invoked to address the cross-border spread of MDR and XDR TB. Nunn noted that the possibility of XDR TB being brought under the IHR had been discussed by the first of the task forces on XDR TB only a couple of years ago. He said, “The feeling then was that [XDR TB] doesn’t easily fall under the IRH because it is not a question of a disease going from A to B. The likelihood is that the disease is already [at] B.” The issue arose again in 2007 when a U.S. civilian who was thought to have XDR TB traveled internationally. Nunn added that to make a stronger case for invoking the IHR, it is first necessary to prove that drug quality is poor. This represents both a challenge and a strategic opening, as the results of quality assessments are beginning to emerge.
Quality Strategies
Zintl suggested that, to address the challenges raised above, countries must first commit to the purchase of quality-assured drugs. A proposal has been made to the GLC and the GDF to allow some large countries to purchase directly from manufacturers as long as they can demonstrate that the drugs are quality assured. This proposal raises a major economic issue, as indicated by the high prices of these drugs in Russia. Economic interests can be expected to cause countries to fight this proposal—particularly countries with very high burdens that will want to buy drugs from domestic manufacturers. It is essential, Zintl said, first to obtain evidence that products from manufacturers are already or on their way to being quality assured and then, over time, begin the difficult work toward further regulatory harmonization. Large, high-priority countries are not likely to commit immediately to procuring drugs only from an approved GDF list. But once they become willing to commit to quality-assured drugs, they may be cautiously willing to shift more and more of their second-line TB drug procurement to products that meet stringent regulatory standards.
In addition to quality assurance of drugs, it is important to obtain commitments to appropriate treatment of MDR TB. Such a commitment will include proper programmatic treatment and the use of drugs that are quality assured, and it will require engaging large, priority countries in pressing their pharmaceutical companies to become prequalified or, at a minimum, to be approved by stringent national regulatory authorities. A ministerial meeting is being held in Beijing in April 2009 to urge countries to make these commitments.
Countries are rapidly scaling up MDR TB treatment, but they are doing so mainly with drugs of unknown quality and without proper programmatic management. According to Zintl, it will be difficult to change this situation without a significant financial commitment and a willingness to alter the dynamics of the robust markets that are selling drugs of unknown quality. It is critical to engage countries in this process and then to track their progress over time. This effort should begin even before the Beijing meeting in 2009.
NEED FOR ACCURATE DEMAND FORECASTING
Levine discussed the critical role of accurate demand forecasting, drawing on lessons learned from dealing with malaria. In a recent report, a CGD global health working group examines issues related to demand forecasting, analyzing the underlying incentives and risks borne by the various parties involved with providing or using information for demand forecasting (CGD, 2007).
The past 5–7 years have seen major changes not only in the absolute volume of resources, but also in their use. While donor funding for the development and purchase of vaccines, diagnostics, and drugs traditionally represented a relatively small portion of donor contributions, it now accounts for approximately 60–65 percent of the total. While greater access to modern medicines benefits millions of people around the world, there is a discontinuity between demand and supply as manufacturers attempt to understand the current demand for their products and how to respond. Complicating matters is the entry of a range of new suppliers into the market, including both innovative multinational and emerging firms. With respect to the pipeline for malaria drugs, the good news is that, after a long period with limited new prospects, a large number of new products are now poised to come on line. While public–private partnerships have been working hard to develop new molecules into viable products, however, they have paid little attention to whether there will be a sustainable market for all of those products in the future and what the capacity requirements will be.
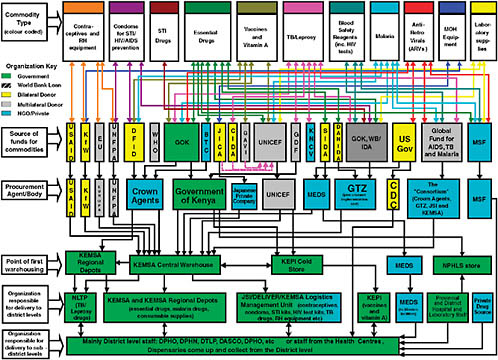
Companies face growing but fragmented demand, a dynamic supplier landscape, and an accelerating pipeline. As a result, they are finding it difficult, according to Levine, to estimate demand with sufficient clarity to make a strong business case for investment within the firm or to know how to engage with the global health players, whether in a commercial or a corporate social responsibility capacity. To illustrate one aspect of the complexity these companies face, Figure 6-1 depicts the procurement process in Kenya. It details the fragmentation of funders, the diversity of service providers, and the range of commodities that are being purchased in varying amounts at varying times, mainly from international sources.
Each of these funders has its own budget cycle, and unpredictable increases or decreases in the willingness or ability to procure products have obvious implications for reliable supply.
Levine highlighted disconnects between the information suppliers receive about demand and the amounts for which there is money to purchase needed products, as well as the supply actually offered by the firms. In the case of malaria, WHO’s demand forecasts have been off by orders of magnitude. For example, the original demand for Coartem was estimated to be 55 million doses; the actual orders turned out to total 14 million doses. The following year, WHO estimated that 100 million doses would be demanded and purchased; the actual number turned out to be 55million. Sanofi had to discard 10 million tablets of artesunate because of overforecasts. Likewise, Uganda ended up with $1–2 million worth of expired AIDS drugs and other donor-purchased items because of overforecasts. Similar stories exist for other products. One possible exception is many vaccine products, for which demand is somewhat easier to project and UNICEF has a longer track record.
Many of those within industry with a strong commitment to participating in the global health effort identify weak demand forecasting as one of
FIGURE 6-1 Commodity logistics system in Kenya (as of April 2004).
NOTE: BTC = Belgian Technical Cooperation, CDC = Centers for Disease Control and Prevention, CIDA = Canadian International Development Agency, DANIDA = Danish International Development Agency, DASCO = District AIDS/STD Coordinator, DFID = UK Department for International Development, DLTP = District Leprosy and TB Program, DPHN = district public health nurse, DPHO = district public health officer, EU = European Union, GAVI = Global Alliance for Vaccines and Immunization, GDF = Global Drug Facility, GOK = Government of Kenya, GTZ = Deutsche Gesellschaft für Technische Zusammenarbeit, IDA = International Dispensary Association, JICA = Japan International Cooperation Agency, JSI = John Snow Inc., KEMSA = Kenya Medical Supplies Agency, KEPI = Kenya Expanded Program on Immunization, KFW = Kreditanstalt für Wiederaufbau, KNCV = Koninklijke Nederlandse Centrale Vereniging tot bestrijding der Tuberculose (Dutch Tuberculosis Foundation), MEDS = Mission for Essential Drugs and Supplies, MOH = Ministry of Health, MSF = Médecins sans Frontières (Doctors Without Borders), NGO = nongovernmental organization, NLTP = National Leprosy and TB Program, NPHLS = National Public Health Laboratories Services (Kenya), RH= reproductive health, SIDA = Swedish International Development Cooperation Agency, STI = sexually transmitted infection, UNFPA = United Nations Population Fund, UNICEF = United Nations Children’s Fund, USAID = U.S. Agency for International Development, WB = World Bank.
SOURCE: Global Health Forecasting Working Group, 2007. Constructed and produced by Steve Kinzett, JSI/Kenya. Copyright 2007 CGD, reprinted with permission.
the leading, if not the greatest, challenges in providing global health products. It is difficult for a company to make the case for investing in either research and development or manufacturing capacity because of the poor track record of demand forecasting.
According to Levine, one of the central problems is that industry, public health specialists, and others in the global health community define needs differently. Governments, donors, funders, and procurement intermediaries in global health often think in terms of estimated need—how much one would ideally like to provide—as opposed to genuine demand, or how much one realistically expects will be purchased. Translating need to actual demand is complex, and involves applying multiple layers of information about both the available financing and the capacity of the health system to provide drugs and scale up when new products are being introduced or new programs are being initiated.
Demand forecasts, then, are an essential link in the supply chain and play five critical roles:
-
Facilitate the matching of supply with demand and eliminate lag time that often occurs.
-
Lead to new products because manufacturers have a realistic picture of future market potential.
-
Enable developing-country health systems to expand to meet changing supply and demand requirements.
-
Permit funders to plan purchases and allocate resources more efficiently.
-
Make bottlenecks more transparent so they can be addressed through advocacy and policy.
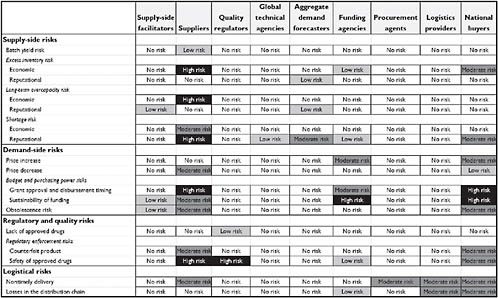
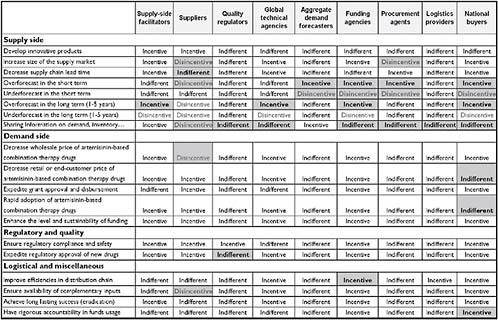
If all stakeholders would benefit from better demand forecasting, one might ask why it does not improve. To answer this question, one must examine incentives that drive behavior. Figure 6-2 shows the different stakeholders and the different kinds of risks they face, such as batch failure and leakage of funds. The one stakeholder that consistently faces the most risk is the manufacturer. Given this risk environment, Figure 6-3 shows the various incentives the different players have. Many of the stakeholders along the supply chain have an incentive to overestimate potential demand. In contrast, suppliers—who face the greatest risk—have an incentive to underestimate demand and reduce their exposure. According to Levine, while this example focuses on the artemisinin combination therapy (ACT) supply chain for malaria, similar incentives apply to the supply chain for TB drugs.
Levine offered three recommendations. One was to take forecasting seriously as critical to having a functioning global health system and to
apply some good-practice principles that have been used in other sectors. The second was to create a global health infomediary—basically a clearinghouse for key demand information that would be completely separated from any advocacy functions and would be viewed as credible by industry. The third was to ensure that those who are providing the money and the information about demand have a clear stake in accurate forecasting. It is essential to redistribute risk to those who are paying for products so they share some of the risks associated with incorrect forecasts; currently, these risks are borne exclusively by suppliers.
Reflecting on Eli Lilly’s experience with its MDR TB initiative over the past 5 years, Richardson suggested that greater transparency in the supply chain is needed. Second-line drug manufacturers are unsure of how to get drugs approved, who is going to pay for them, and what the demand is. It is important to ensure that this system is simple and that companies understand how to get their drugs procured. Standards to which suppliers will be held must be clear and unequivocal. And there must be some mechanism for dealing efficiently with changes once a manufacturer has been prequalified. Prequalification testing is only a partial solution to ensuring continued supply—there must be ongoing monitoring of both manufacturers and drugs. Ongoing monitoring of Good Manufacturing Practices at facilities is just as critical, or even more critical, than prequalification in ensuring a sustainable drug supply. Companies must learn how to navigate approval and procurement both at a local level and through an entity such as the GDF. It is important to engage people who are capable of steering a path through both sets of regulations. It is also important that the amount of uncertainty in the forecast and demand management aspects of procurement be reduced. If companies understand the path forward and know who will be paying, they will be better able to manufacture and supply the drugs that are needed.