5
Consequences of Gestational Weight Gain for the Mother
Women whose weight gain during pregnancy is outside the recommended ranges may experience various adverse maternal outcomes, which may include increased risk for pregnancy-associated hypertension, gestational diabetes (GDM), complications during labor and delivery, and postpartum weight retention and subsequent maternal obesity as well as an increased risk for unsuccessful breastfeeding. As noted in Chapter 1 and discussed in detail in Chapter 2, there is an increased prevalence in the United States of women who are overweight or obese entering pregnancy, also putting them at greater risk for several of these same adverse pregnancy outcomes. Additionally, more women are becoming pregnant at an older age and are thus entering pregnancy with chronic conditions, such as type 2 diabetes that could contribute to increased morbidity during both the prenatal and postpartum periods.
The Agency for Healthcare Research and Quality (AHRQ) commissioned a comprehensive, systematic evidence-based review of the literature on outcomes related to absolute weight gain as well as gestational weight gain (GWG) within or outside the guidelines set in the Institute of Medicine (IOM) report Nutrition During Pregnancy. This review included evidence on the consequences of GWG for both the mother and infant (Viswananthan et al., 2008). The committee used this review as a foundation for discussion of the state of the science for GWG and maternal outcomes in this chapter as well as for infant outcomes in Chapter 6.
This chapter provides reviews of the state of the science before the IOM (1990) report and summaries of findings from the Viswanathan et al.
(2008) AHRQ evidence-based review on outcomes of gestational weight gain that are related to the mother during pregnancy, at delivery, and postpartum periods. Studies were rated “good,” “fair,” or “poor” based on a scoring algorithm developed by the AHRQ study reviewers using previously published guidelines (Downs and Black, 1998; Deeks et al., 2003). The methodological approach and system of rating articles used in the AHRQ review is provided in Appendix E. Discussions in this chapter also include articles published since release of the AHRQ report in which associations between GWG and maternal outcomes were examined (see Appendix F for summary data tables).
CONCEPTUAL FRAMEWORK: CONSEQUENCES OF GESTATIONAL WEIGHT GAIN FOR THE MOTHER
The committee’s conceptual framework (see Chapter 1) illustrates a model for maternal and child outcomes consequent to GWG outside the ranges recommended by the IOM (1990) report (Figure 5-1). There are numerous potential causal factors, including environmental factors, that can influence the determinants of GWG and its consequences and others that may affect those consequences by other routes. These consequences,
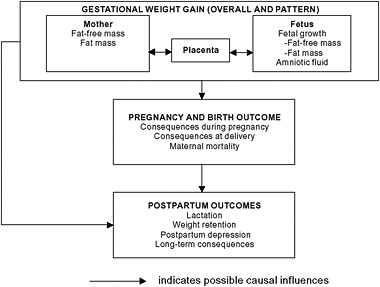
FIGURE 5-1 Schematic summary of maternal consequences associated with gestational weight gain.
i.e., adverse health outcomes to the mother, can arise in the prenatal and/or postpartum periods. Among the well-studied prenatal maternal outcomes that result from excessive GWG are pregnancy-associated hypertension (including preeclampsia and eclampsia) and risk of complications in labor and delivery. In the postpartum period, weight retention can lead to higher weight status in subsequent pregnancies as well as weight retention and other long-term maternal health consequences such as increased risk for type 2 diabetes and cardiovascular disease. Unfortunately the literature in this area does not allow inference of causality because it is based solely on observational studies.
The committee’s conceptual framework (see Chapter 1) illustrates a model for maternal and child outcomes consequent to GWG outside the ranges recommended by the IOM (1990) report (Figure 5-1). There are numerous potential causal factors, including environmental factors that can influence the determinants of GWG and its consequences, and others that may affect those consequences by other routes. These consequences, i.e., adverse health outcomes to the mother, can arise in the prenatal and/or postpartum periods. Among the most-studied prenatal maternal outcomes resulting from excessive GWG are pregnancy-associated hypertension (including preeclampsia and eclampsia) and the risk of complications in labor and delivery. In the postpartum period, potential consequences include weight retention and lactation performance. Weight retention can lead to higher weight status in subsequent pregnancies predisposing the woman to more adverse reproductive outcomes (creating a cycle of risk) and other long-term maternal health consequences such as increased risk for type 2 diabetes, cancer, cardiovascular disease, and mental health issues. Therefore these outcomes are also included in the model. Unfortunately the literature in this area does not allow inference of causality since it is based solely on observational studies, thus we can not say that gestational weight gain causes these outcomes but rather that it is associated with them. The committee has made inferences using the best data available in consideration of plausible biologic mechanisms, confounding and other aspects of the individual study methodology, and the patterns of results.
CONSEQUENCES DURING PREGNANCY
The committee’s evaluation of the evidence on associations between GWG and consequences for the mother during pregnancy showed that evidence for an association between GWG and pregnancy complications such as GDM and gestational hypertensive disorders is inconclusive because of inconsistent results and methodological flaws. The outcome of mental health during pregnancy is understudied.
Gestational Diabetes and Impaired Glucose Tolerance
Although pregnancy is frequently accompanied by a pronounced physiological decrease in peripheral insulin sensitivity (reviewed in Chapter 3), the combination of decreased peripheral insulin sensitivity and beta-cell dysfunction can lead to the development of abnormal glucose tolerance during pregnancy, or GDM. It is well established that women who are obese when they enter pregnancy tend to develop a more pronounced insulin resistance and are at greater risk for GDM than are non-obese women (Dahlgren, 2006; Chu et al., 2007). The incidence of GDM has increased dramatically in recent decades (see Chapter 2). From 1989 to 2004 there was a relative increase in prevalence of GDM of 122 percent for the U.S. population as a whole and 260 percent among African American women (Getahun et al., 2008).
Most women with normal glucose tolerance develop elevated blood ketones with ketonuria at various times during pregnancy (Chez and Curcio, 1987). Pregnant women with diabetes, on the other hand, are more likely to develop sustained elevated blood ketones and ketonuria during pregnancy. Gin et al. (2006), who measured capillary blood ketones and beta-hydroxybutyrate in women with normal glucose tolerance (controls) and those with GDM three times a day between 25 and 37 weeks’ gestation, found that fasting ketonuria was strongly correlated with ketonemia in controls but not in women with GDM. Maternal ketonuria or acetonuria during pregnancy is a concern because it can result in neonatal or childhood neurocognitive dysfunction (see discussion in Chapters 3 and 6).
The IOM (1990) report did not include information on the relationship between GWG and abnormal glucose metabolism. The literature since 1990, as described in the AHRQ review (Viswanathan et al., 2008), includes 11 published articles that together provide weak evidence in support of an association between GWG and development of abnormal glucose metabolism (either GDM or impaired glucose tolerance). Four of the studies reported that GWG above the range recommended in the IOM (1990) report was positively associated with abnormal glucose tolerance (Edwards et al., 1996; Kieffer et al., 2001; Kabiru and Raynor, 2004; Saldana et al., 2006). Three studies reported that women whose GWG was below the recommended range had a higher likelihood of GDM (Thorsdottir et al., 2002; Brennand et al., 2005; Kieffer et al., 2006), and four studies found no significant association between GWG and glucose tolerance (Bianco et al., 1998; Murakami et al., 2005; Seghieri et al., 2005; Hackmon et al., 2007). A methodological limitation in all but one study (Saldana et al., 2006) was the use of total GWG as the exposure variable rather than weight gain until the time of diagnosis. This is problematic because management of GDM includes dietary counseling and efforts to control weight gain.
Outside the AHRQ review, Catalano et al. (1993) reported that weight gain in women with GDM was less than in a normal glucose tolerance group primarily because of greater pregravid weight. However, when GWG was assessed separately for early, mid- and late gestation, there was a significant decreased rate of weight gain in overweight women with GDM only from 30 weeks’ gestation until delivery. There is biologic plausibility for an effect of GWG on the development of glucose tolerance: higher GWG could result in greater fat deposition, which could then influence insulin sensitivity. The body of evidence to date, however, is weak in support of such an association.
Hypertensive Disorders
Hypertensive disorders during pregnancy include pregnancy-induced hypertension, preeclampsia, and eclampsia. The risk for pregnancy-induced hypertension is greater among women who enter pregnancy overweight or obese. For example, Thadhani et al. (1999) examined the relationship between pregravid BMI, elevated cholesterol level, and the development of hypertensive disorders of pregnancy among 15,262 women and found that the age-adjusted relative risk for developing gestational hypertension was 1.7 and 2.2 for women with BMI values of 25-29.9 and ≥ 30 kg/m2, respectively, compared to women with BMI values < 21 kg/m2. Likewise, preeclampsia is about twice as prevalent among overweight and about three times as prevalent among obese women (Sibai et al., 1997; Catalano, 2007), and severity of the disease increases as BMI increases (Bodnar et al., 2007).
Although the relationship between these hypertensive disorders and the BMI of women entering pregnancy is fairly well established, the association between these conditions and increased GWG is less clear. This was true two decades ago when the IOM (1990) report described the relationship between GWG and hypertensive conditions as being unclear due to limited and inconclusive data; and it remains true today. The rationale, for example, is that preeclampsia is a condition noted for a decrease in the normal (50-60 percent) expansion in maternal intravascular (plasma) volume. The condition may also affect weight gain in early gestation. In addition, increased vascular permeability and decreased plasma oncotic pressure, caused by preeclampsia, can lead to increased edema and excessive weight gain in late gestation. Hence placental dysfunction in early gestation may effect both early and late weight gain—albeit in opposite directions. These physiologic parameters preclude the use of total weight as a measure of GWG in preeclampsia.
Since the IOM (1990) report was published and as described in the AHRQ review, 12 published studies examined the association between
GWG and hypertensive disorders. Five of these studies (two rated fair and the rest rated poor) examined pregnancy-induced hypertension (Edwards et al., 1996; Bianco et al., 1998; Thorsdottir et al., 2002; Brennand et al., 2005; Jensen et al., 2005). Only two of the studies reported an association between higher GWG and pregnancy-induced hypertension (Thorsdottir et al., 2002; Jensen et al., 2005). The five studies differed in their control for confounding. Thorsdottir et al. (2002) adjusted for age, parity, height, and gestational age. Jensen et al. (2005) adjusted for 2-hour oral glucose tolerance test (OGTT) result, maternal age, prepregnancy BMI, gestational age (continuous variables), parity, smoking, and ethnic background.
The outcome of preeclampsia has been examined in a total of 10 studies (Edwards et al., 1996; Ogunyemi et al., 1998; Thorsdottir et al., 2002; Kabiru and Raynor, 2004; Brennand et al., 2005; Murakami et al., 2005; Cedergen, 2006; Wataba et al., 2006; DeVader et al., 2007; Kiel et al., 2007), of which 7 were rated fair and the rest were rated of poor quality. Overall, an association between higher total GWG and higher risk of preeclampsia was found in six of these studies (Edwards et al., 1996; Ogunyemi et al., 1998; Brennand et al., 2005; Cedergen, 2006; DeVader et al., 2007; Kiel et al., 2007). Lower total weight gains were found to be protective in four studies (Brennand et al., 2005; Cedergren, 2006; DeVader et al., 2007; Kiel et al., 2007). Those studies that did not find an association for high total GWG (Thorsdottir et al., 2002; Kabiru and Raynor, 2004; Murakami et al., 2005; Wataba et al., 2006) were primarily conducted in women who were not overweight or obese (two were conducted in Japan). It is difficult to compare these studies because of the lack of a consistent definition for preeclampsia across them.
Since the AHRQ review, two studies using birth certificate data from the state of Missouri (DeVader et al., 2007; Kiel et al., 2007) showed similar results, namely that GWG above the recommended range leads to higher risk of preeclampsia among overweight women (Langford et al., 2008). These studies were also limited by methodological problems associated with the use of total weight gain as the exposure as opposed to a weight gain before the diagnosis of preeclampsia.
Other Quality of Life Measures
Although the influence of psychosocial status on GWG (see Chapter 4) has been examined in several studies, none has examined the reverse: the possible effects of GWG on maternal mental health during pregnancy. The IOM (1990) report contained no information on any quality-of-life measures during pregnancy. There are eight studies covered in AHRQ review (Viswanathan et al., 2008) on other antepartum outcomes, including a composite outcome for discomfort in general (Rodriguez et al., 2001), physical
energy and fatigue (Tulman et al., 1998), stretch marks (Madlon-Kay, 1993; Atwal et al., 2006), heartburn (Marrero et al., 1992), gallstones (Lindseth and Bird-Baker, 2004; Ko, 2006), and hyperemesis (Dodds et al., 2006). Three of these studies were rated as fair (Tulman et al., 1998; Rodriguez et al., 2001; Ko, 2006) and five as poor quality (Marrero et al., 1992; Madlon-Kay, 1993; Lindseth and Bird-Baker, 2004; Atwal et al., 2006; Dodds et al., 2006). Overall, there was no association between higher GWG and the outcomes of interest except for the two studies in which stretch marks were examined (Madlon-Kay, 1993; Atwal et al., 2006). This association was weak because of the small sample size, study design (one was a cross-sectional study), and the lack of adjustment for confounding factors. In the one study in which hyperemesis was examined, women who gained a total of < 7 kg had an increased likelihood of more antenatal admissions for this outcome (Dodds et al., 2006). For this outcome in particular, GWG was not a causal factor but was more likely the result of having had hyperemesis during the pregnancy.
CONSEQUENCES AT DELIVERY
The IOM (1990) report examined the link between GWG and complications during labor and delivery but only because such complications were viewed as being consequences of the delivery of a large-for-gestational age (LGA) infant. That report concluded that the contribution of GWG to delivery outcomes was quite small. Since then, the literature has grown and the outcomes related to delivery have been subdivided to better understand the process of labor. The discussion below addresses recent evidence for an association between GWG and each of these delivery outcomes. In summary, current evidence supports a strong association between GWG above recommended ranges and increased risk of cesarean delivery. There is no evidence, however, to support an association of GWG with maternal mortality in countries where women have ready access to obstetric care.
Induction of Labor
The AHRQ review (Viswanathan et al., 2008) included five studies related to an association between GWG and induction of labor (Ekblad and Grenman, 1992; Kabiru and Raynor, 2004; Jensen et al., 2005; Graves et al., 2006; DeVader et al., 2007). The strength of the evidence from these studies was rated weak for an association between high GWG and labor induction or failure of labor induction. Although statistically significant increases in the outcomes associated with high GWG were reported in all five studies, comparisons across studies were not meaningful because of
differences in the definition of high GWG and a lack of control for confounding factors.
Length of Labor
Three studies in the AHRQ review (Viswanathan et al., 2008) examined associations between GWG and length of labor (Ekblad and Grenman, 1992; Johnson et al., 1992; Purfield and Morin, 1995). Although two of the three studies found a significant increase in the length of labor with higher weight gains, both lacked control for confounding factors (Ekblad and Grenman, 1992; Purfield and Morin, 1995). As a result, the evidence was rated as weak for an association between higher GWG and longer duration of labor.
Mode of Delivery
Substantial research has been conducted since the IOM (1990) report on the association between GWG and mode of delivery, with the AHRQ review (Viswanathan et al., 2008) examining a total of 21 studies using GWG as a continuous or categorical variable unrelated to the IOM (1990) guidelines (Ekbald and Grenman, 1992; Johnson et al., 1992; Purfield and Morin, 1995; Witter et al., 1995; Bianco et al., 1998; Shepard et al., 1998; Young and Woodmansee, 2002; Joseph et al., 2003; Chen et al., 2004; Kabiru and Raynor, 2004; Brennand et al., 2005; Jensen et al., 2005; Murakami et al., 2005; Rosenberg et al., 2005; Cedergren, 2006; Graves et al., 2006; Wataba et al., 2006; DeVader et al., 2007; Jain et al., 2007; Kiel et al., 2007; Sherrard et al., 2007). Overall, these studies provided moderate evidence for an association between high GWG and cesarean delivery; only four studies failed to find an association (Bianco et al., 1998; Brennand et al., 2005; Murakami et al., 2005; Graves et al., 2006). An important factor to consider in this literature is the route of previous delivery for multiparous women. Only half of the studies reviewed adjusted for this; among those that did, five also adjusted for co-morbidities (e.g., GDM and preeclampsia) that could also have contributed to the route of delivery (Witter et al., 1995; Shepard et al., 1998; Joseph et al., 2003; Rosenberg et al., 2005; Sherrard et al., 2007). Higher weight gains were associated with instrumental deliveries in three (Purfield and Morin, 1995; Kabiru and Raynor, 2004; Cedergren, 2006) studies but not in two others (Ekblad and Grenman, 1992; DeVader et al., 2007).
When GWG was categorized according to the ranges recommended in the IOM (1990) report, the body of research provided moderate evidence that weight gain above the recommended ranges was associated with cesarean delivery among normal- and underweight women. In contrast, the evi-
dence among obese and morbidly obese women was rated as weak (Parker and Abrams, 1992; Edwards et al., 1996; Bianco et al., 1998; Thorsdottir et al., 2002; Stotland et al., 2004; Hilakivi-Clarke et al., 2005; DeVader et al., 2007; Kiel et al., 2007).
Of all of the studies on an association between GWG and mode of delivery, 10 were consistent in noting that overweight or obese women prior to pregnancy were at higher risk of cesarean delivery compared to women who entered pregnancy at a lower BMI (Johnson et al., 1992; Witter et al., 1995; Shepard et al., 1998; Joseph et al., 2003; Chen et al., 2004; Murakami et al., 2005; Rosenberg et al., 2005; Graves et al., 2006; Jain et al., 2007; Sherrard et al., 2007).
Maternal Mortality
Both the IOM (1990) report and the AHRQ review (Viswanathan et al., 2008) found no information on the relationship between GWG and maternal mortality. From a theoretical perspective, if GWG above recommended ranges is associated with LGA infants and shoulder dystocia in settings that do not allow for immediate cesarean delivery or attendance by a trained clinician, the mother could die during childbirth. In such an event, the immediate cause of death would be attributed to the size of the infant and associated labor and delivery complications. This impedes the study of consequences of GWG on maternal mortality.
POSTPARTUM CONSEQUENCES
The discussion below summarizes the committee’s evaluation of the evidence on associations between GWG and three postpartum consequences for the mother: lactation, postpartum weight retention, and postpartum depression. Overall, the evidence suggests that GWG below the levels recommended in IOM (1990) is moderately associated with initiation of breastfeeding and that there is a strong association between higher GWG and postpartum weight retention (3 months to 3 years). The outcome of mental health is understudied and worthy of exploration.
Lactation
The IOM (1990) report reviewed only one study examining the relationship between GWG and lactation (Butte et al., 1984). That study did not show any relationship between GWG and either milk quality or quantity. The AHRQ review (Viswanathan et al., 2008) included only four studies on the association of GWG, categorized according to the recommendations of IOM (1990), with lactation performance (Rasmussen et al.,
2002; Li et al., 2003; Hilson et al., 2006; Baker et al., 2007). Although three of the studies showed that obese women had a shorter duration of breastfeeding (both exclusive and any breastfeeding) regardless of GWG (Rasmussen et al., 2002; Hilson et al., 2006; Baker et al., 2007), the evidence for any association between GWG and duration of exclusive or any breastfeeding was rated weak; evidence that low weight gain is associated with decreased initiation of breastfeeding was rated moderate. Since the AHRQ review, the committee identified one other study, a cross-sectional study done in Greece reporting that women with higher prepregnancy BMI were less likely to initiate breastfeeding and that GWG had no effect on either initiation or duration of breastfeeding (Manios et al., 2008).
Postpartum Weight Retention
Postpartum weight is a woman’s weight immediately after delivery of the fetus, placenta, and amniotic fluid. In the subsequent days to weeks, the increase in the woman’s extracellular and extravascular water that occurred during pregnancy is lost and her plasma volume returns to prepregnancy values. Postpartum weight retention is the amount of weight that remains at this later time minus the woman’s pregravid weight; it includes the weight of any increased breast tissue being used for lactation as well as any remaining fat mass gained during pregnancy.
The IOM (1990) report stated that women with GWG well beyond the recommended ranges are more likely to retain weight postpartum and are at increased risk for subsequent obesity. Because the focus of that report was on optimizing birth weight, however, the emphasis of the IOM (1990) guidelines was on infant outcomes rather than maternal postpartum weight retention.
The AHRQ review included only studies that directly examined associations between GWG and postpartum weight retention and did not include those that used parity or childbearing as a proxy for GWG (i.e., Rosenberg et al., 2003; Gunderson et al., 2004). These later studies provide information that is consistent with the AHRQ report conclusions. The report found only two studies that examined differences in the amount of fat retained in the postpartum period for GWG according to IOM (1990) categories (Lederman et al., 1997; Butte et al., 2003). In the first, Butte et al. (2003) examined a convenience sample of nonsmoking women aged 18-40 from Houston (17 underweight, 34 normal weight, 12 overweight/obese). Body composition was measured using dual-energy x-ray absorptiometry (DXA) before and after pregnancy and weight was obtained before pregnancy, during pregnancy, and after pregnancy. Results showed that maternal fat retention was significantly higher among women who gained above (5.3 kg) compared to those who gained within (2.3 kg) or below (−0.5 kg) the IOM (1990) guidelines.
In the second, Lederman et al. (1997) studied 196 nonsmoking women aged 18-36 years, recruited from 3 prenatal clinics in New York City. Women who gained below the IOM (1990) recommendations had the lowest fat gain from 14 to 37 weeks of gestation compared to those with an intermediate and those with the highest fat gain. In addition the study found that, among obese women who gained within the IOM (1990) guidelines, the amount of body fat change (−0.6 kg) was significantly lower than among women in the other BMI groups who also gained within the recommendations (6.0 for underweight, 3.8 for normal, and 2.8 kg for overweight women). Unfortunately no test of significance was conducted. These data suggest, however, that higher GWG results in higher maternal fat gains, although the evidence for this is weak because of the limited number of studies and small sample sizes.
The AHRQ review (Viswanathan et al., 2008) separated the studies on postpartum weight retention into three categories according to when postpartum weight retention was assessed: short-term (less than 11 weeks), intermediate (3 months to 3 years), and long-term (greater than 3 years). Within the short-term (≤ 11 weeks) studies, there was weak evidence for a relationship between GWG as a continuous variable and postpartum weight retention (Muscati et al., 1996). However, when GWG was categorized according to the IOM (1990), there was a moderate, consistent relationship. Four studies showed that GWG exceeding the IOM (1990) guidelines was associated with higher postpartum weight retention (Stevens-Simon and McAnarney, 1992; Scholl et al., 1995; Luke et al., 1996; Walker et al., 2004). This observation was consistent for women irrespective of age.
In the intermediate term (3 months to 3 years), one study rated good (Harris et al., 1999), three studies rated fair (Ohlin and Rossner, 1990; Soltani and Fraser, 2000; Walker et al., 2004), and one study rated poor (Parham et al., 1990) provided moderate evidence for a relationship between GWG above recommended ranges and greater postpartum weight retention. Likewise, the strength of the evidence for subjects who gained within the guidelines was also moderate, based on five studies rated fair (Scholl et al., 1995; Walker, 1996; Rooney and Schauberger, 2002; Olson et al., 2003; Amorim et al., 2007) and one study rated poor (Keppel and Taffel, 1993). Thus, overall, higher GWG is associated with greater postpartum weight retention measured at 3 to 36 months postpartum. The authors noted, however, that the data should be interpreted with caution because of a lack of consistent adjustment for covariates such as nutrition and exercise. In interpreting these data, it is important to note that the relationship between GWG and postpartum weight retention depends not only on dietary intake and physical exercise but also on breastfeeding behavior. In the only available study that considered prepregnancy BMI, GWG, and breastfeeding simultaneously, Baker et al. (2008) showed that women from the Danish National Birth Cohort with reasonable weight gains (e.g.,
~12 kg) and who exclusively breastfed for 6 months as currently recommended had no weight retention at 6 months postpartum. For racial/ethnic groups, only one study was available. Keppel and Taffel (1993) used a nationally representative database to show that black women retained more weight than white women regardless of GWG.
In the long term (> 3 years), the evidence is less conclusive for a relationship between GWG and postpartum weight retention. One study rated good (Callaway et al., 2007) found a weak association between GWG and weight of the mother 21 years after the pregnancy, while another study rated fair (Linne et al., 2003) found that women who became overweight after 15 years had higher GWG in the index pregnancy compared to women who remained within a normal weight range (although no adjustment was made for confounding). Linne et al. (2004) also concluded that women who began pregnancy at a higher BMI tended to stay on the same weight trajectory later in life. Three studies (rated as fair) in the AHRQ review provided moderate evidence in support of a relationship between gaining above the IOM (1990) guidelines and greater postpartum weight retention (Rooney and Schauberger, 2002; Rooney et al., 2005; Amorim et al., 2007); however, the amount of weight retained was small.
Outside the AHRQ review, studies by Gunderson et al. (2004) and Rosenberg et al. (2003), provide information that is consistent with the conclusions of the AHRQ review. The work of Nohr et al. (2008) also largely corroborates these earlier findings and strengthens the evidence for an association between GWG and postpartum weight retention in the intermediate period. Nohr et al. (2008) gathered data from 60,892 women with term pregnancies in the Danish National Birth Cohort. They linked these data to birth and hospital-discharge registers. After adjustment for multiple confounding factors, they reported that women who gained 16-19 kg or ≥ 20 kg were at 2.3- and 6.2-fold higher odds of retaining ≥ 5 kg at 6 months postpartum than women who gained only 10-15 kg.
A major concern with postpartum weight retention is movement into a higher BMI category, which is associated with a greater risk of pregnancy complications and adverse birth outcomes in a subsequent pregnancy. For example, Scholl et al. (1995) calculated that women (12-29 years old) had a 2.8-fold higher risk of becoming overweight at 6 months postpartum if their rate of weight gain during pregnancy was > 0.68 kg per week than women with lower gains. Gunderson et al. (2000) reported similar results based on calculating the risk of becoming overweight at the start of the second pregnancy with weight gains above the IOM (1990) recommendations in the first. Nohr et al. (2008) also showed that with GWG between 16-19 kg, 12 to 14 percent of women with pregravid BMIs > 18.5 kg/m2 move up one category of weight status at 6 months postpartum and that this increases to 25 percent with weight gains > 20 kg.
Postpartum Depression
As with depression during pregnancy, there were no data on the relationship of GWG and postpartum depression in the IOM (1990) report. The AHRQ review (Viswanathan et al., 2008) does not include data on this relationship and the committee was unable to identify new data on this possible relationship.
LONG-TERM CONSEQUENCES
The IOM (1990) report was focused more on infant outcomes and did not address long-term maternal outcomes of GWG. Excess postpartum weight retention could exacerbate these problems (see discussion above) and contribute to the development of chronic conditions, including diabetes, hypertension, and other cardiovascular risk factors (Arendas et al., 2008). The following discussion includes studies that focused on the relationship between GWG and postpartum type 2 diabetes and metabolic disorders, mental health, and cancer. In summary, there is insufficient evidence to link GWG to long-term health consequences of the mother as a result of the lack of studies in this area.
Type 2 Diabetes/Metabolic Disorders
The committee was unable to identify any published studies examining the possible association between GWG and the development of metabolic disorders later in a woman’s life. Such an association is biologically plausible because of the link between GWG and postpartum weight retention. Although they did not collect GWG data, Gunderson et al. (2008) showed that childbearing was associated with increased visceral fat postpartum; and Lim et al. (2007) identified a relationship between abnormal glucose tolerance at 1 year postpartum and increased visceral fat in women who had GDM that was independent of maternal age and BMI. Berg and Scherer (2005) reviewed evidence on the role of adipose tissue in systemic inflammation and determined that both the distribution and amount of fat are important. Visceral fat was more strongly associated with insulin resistance in obese subjects than in lean subjects.
Cardiovascular Disorders
The committee was also unable to identify any published studies that examined a direct association between GWG and the development of cardiovascular disorders later in life. However, obesity, preeclampsia, and toxemia of pregnancy are linked to long-term sequelae that include cardiovascular disease (Bellamy et al., 2007; Zhang et al., 2008).
Other Adverse Health Outcomes
Mental Health
As previously discussed, the topic of mental health of the mother is not addressed in the AHRQ review (Viswanathan et al., 2008). Two small studies (Jenkin and Tiggemann, 1997; Walker, 1997) provide weak evidence for a connection between postpartum weight retention up to 1 year post-delivery and self-esteem/depression. These studies did not control for prepregnancy BMI.
Cancer
The committee found weak evidence for an association of GWG and risk of breast cancer. Specifically, a retrospective cohort study of 2,089 Finnish women showed a positive relationship between weight gain in the upper tertile (> 15 kg) and post-menopausal breast cancer risk, after adjustment for prepregnancy BMI (RR = 1.62, 95% CI: 1.03-2.53) (Kinnunen et al., 2004). In a nested case-control study of 65 cases of breast cancer in this cohort, the BMI at the time of diagnosis did not change the findings. Among premenopausal women in the population, weight gains of > 16 kg during pregnancy and an increase in BMI of greater than 7 kg/m2 after age 20 were associated with a reduced risk of pre-menopausal breast cancer. However, the question of why BMI and GWG affect pre-menopausal and post-menopausal breast cancer risk differently remains unanswered (Hilakivi-Clarke et al., 2005).
CONCLUDING REMARKS
Overall, the consequences for the mother of GWG above recommended ranges appear to be well-substantiated for outcomes such as cesarean delivery and postpartum weight retention. The studies that have examined glucose abnormalities and hypertensive disorders of pregnancy have been methodologically flawed and thus do not provide sufficient evidence to support or refute a possible association. For GWG below recommended ranges, the only outcome for which there is any substantial evidence is initiation of breastfeeding. There are no available studies of a relationship between low GWG and increased maternal mortality among American women.
There is a general lack of research that relates GWG to maternal outcomes beyond the first year postpartum other than for postpartum weight retention and subsequent obesity. This is understandable because most of the outcomes that are of the greatest interest, such as cardiovascular disease, cancer, and depression take longer to study because they occur later in the
woman’s life. It is well established, however, that obesity is associated with increased morbidity and mortality (i.e., from hypertension, dyslipidemia, diabetes mellitus, cholelithiasis, coronary heart disease, osteoarthritis, sleep apnea, stroke, and certain cancers) (Must et al., 1992; Troiano et al., 1996; Allison et al., 1999; Calle et al., 2003; Gregg et al., 2005). Furthermore, for subsequent pregnancies, maternal overweight and obesity are associated with higher rates of cesarean delivery, GDM, preeclampsia, and pregnancy-induced hypertension as well as postpartum anemia (Bodnar et al., 2007).
FINDINGS AND RECOMMENDATIONS
Findings
-
The literature related to GWG and maternal outcomes does not allow inference of causality since it is based solely on observational studies.
-
Evidence for an association between GWG and pregnancy complications such as glucose abnormalities and gestational hypertension disorders is inconclusive and problematic due to methodological flaws, and the outcome of mental health during pregnancy is understudied.
-
There is a strong association between higher GWG and increased risk of cesarean delivery.
-
There is no research on the effect of GWG on maternal mortality from which the committee could make any conclusions.
-
Low GWG is moderately associated with failure to initiate breastfeeding.
-
There is a strong association between higher GWG and postpartum weight retention in the immediate postpartum period (3 months to 3 years).
-
The outcome of mental health is understudied.
-
There is insufficient evidence to link GWG to long-term health consequences of the mother due to the lack of studies in this area.
-
Maternal prepregnancy weight status is an important independent predictor of maternal short- and long-term outcomes.
Recommendations for Action and Research
Action Recommendation 5-1: The committee recommends that appropriate federal, state, and local agencies as well as health care providers inform women of the importance of conceiving at a normal BMI and that all those who provide health care or related services to women of childbearing age include pre-conceptional counseling in their care.
Research Recommendation 5-1: The committee recommends that the National Institutes of Health and other relevant agencies should provide support to researchers to conduct observational and experimental studies on the association between GWG and (a) glucose abnormalities and gestational hypertensive disorders that take into account the temporality of the diagnosis of the outcome and (b) the development of glucose intolerance, hypertension, and other cardiovascular disease risk factors as well as mental health and cancer later in a woman’s life.
Research Recommendation 5-2: The committee recommends that the National Institutes of Health and other relevant agencies should provide support to researchers to conduct studies that (a) explore mechanisms, including epigenetic mechanisms, that underlie effects of GWG on maternal and child outcomes and (b) address the extent to which optimal GWG differs not only by maternal prepregnancy BMI but also by other factors such as age (especially among adolescents), parity, racial/ethnic group, socioeconomic status, co-morbidities, and maternal/paternal/fetal genotype.
Areas for Additional Investigation
The committee identified the following areas for further investigation to aid in future revisions of GWG recommendations. The research community should conduct studies on:
-
Associations between gestational weight gain and maternal mortality.
-
Effects of GWG on maternal mental status during pregnancy, in the postpartum period, and in the long term.
-
The causal nature of how gestational weight gain leads to short-and long-term maternal outcomes.
REFERENCES
Allison D. B., K. R. Fontaine, J. E. Manson, J. Stevens and T. B. VanItallie. 1999. Annual deaths attributable to obesity in the United States. JAMA 282(16): 1530-1538.
Amorim A. R., S. Rossner, M. Neovius, P. M. Lourenco and Y. Linne. 2007. Does excess pregnancy weight gain constitute a major risk for increasing long-term BMI? Obesity (Silver Spring) 15(5): 1278-1286.
Arendas K., Q. Qiu and A. Gruslin. 2008. Obesity in pregnancy: pre-conceptional to postpartum consequences. Journal of Obstetrics and Gynaecology Canada 30(6): 477-488.
Atwal G. S., L. K. Manku, C. E. Griffiths and D. W. Polson. 2006. Striae gravidarum in primiparae. British Journal of Dermatology 155(5): 965-969.
Baker J. L., K. F. Michaelsen, T. I. Sorensen and K. M. Rasmussen. 2007. High prepregnant body mass index is associated with early termination of full and any breastfeeding in Danish women. American Journal of Clinical Nutrition 86(2): 404-411.
Baker J. L., M. Gamborg, B. L. Heitmann, L. Lissner, T. I. Sorensen and K. M. Rasmussen. 2008. Breastfeeding reduces postpartum weight retention. American Journal of Clinical Nutrition 88(6): 1543-1551.
Bellamy L., J. P. Casas, A. D. Hingorani and D. J. Williams. 2007. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: systematic review and meta-analysis. British Medical Journal 335(7627): 974.
Berg A. H. and P. E. Scherer. 2005. Adipose tissue, inflammation, and cardiovascular disease. Circulation Research 96(9): 939-949.
Bianco A. T., S. W. Smilen, Y. Davis, S. Lopez, R. Lapinski and C. J. Lockwood. 1998. Pregnancy outcome and weight gain recommendations for the morbidly obese woman. Obstetrics and Gynecology 91(1): 97-102.
Bodnar L. M., J. M. Catov, M. A. Klebanoff, R. B. Ness and J. M. Roberts. 2007. Prepregnancy body mass index and the occurrence of severe hypertensive disorders of pregnancy. Epidemiology 18(2): 234-239.
Brennand E. A., D. Dannenbaum and N. D. Willows. 2005. Pregnancy outcomes of First Nations women in relation to pregravid weight and pregnancy weight gain. Journal of Obstetrics and Gynaecology Canada 27(10): 936-944.
Butte N. F., C. Garza, J. E. Stuff, E. O. Smith and B. L. Nichols. 1984. Effect of maternal diet and body composition on lactational performance. American Journal of Clinical Nutrition 39(2): 296-306.
Butte N. F., K. J. Ellis, W. W. Wong, J. M. Hopkinson and E. O. Smith. 2003. Composition of gestational weight gain impacts maternal fat retention and infant birth weight. American Journal of Obstetrics and Gynecology 189(5): 1423-1432.
Callaway L. K., H. D. McIntyre, M. O’Callaghan, G. M. Williams, J. M. Najman and D. A. Lawlor. 2007. The association of hypertensive disorders of pregnancy with weight gain over the subsequent 21 years: findings from a prospective cohort study. American Journal of Epidemiology 166(4): 421-428.
Calle E. E., C. Rodriguez, K. Walker-Thurmond and M. J. Thun. 2003. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. New England Journal of Medicine 348(17): 1625-1638.
Catalano P. M. 2007. Increasing maternal obesity and weight gain during pregnancy: the obstetric problems of plentitude. Obstetrics and Gynecology 110(4): 743-744.
Catalano P. M., N. M. Roman, E. D. Tyzbir, A. O. Merritt, P. Driscoll and S. B. Amini. 1993. Weight gain in women with gestational diabetes. Obstetrics and Gynecology 81(4): 523-528.
Cedergren M. 2006. Effects of gestational weight gain and body mass index on obstetric outcome in Sweden. International Journal of Gynaecology and Obstetrics 93(3): 269-274.
Chen G., S. Uryasev and T. K. Young. 2004. On prediction of the cesarean delivery risk in a large private practice. American Journal of Obstetrics and Gynecology 191(2): 616-624; discussion 616-624.
Chez R. A. and F. D. Curcio, 3rd. 1987. Ketonuria in normal pregnancy. Obstetrics and Gynecology 69(2): 272-274.
Chu S. Y., W. M. Callaghan, S. Y. Kim, C. H. Schmid, J. Lau, L. J. England and P. M. Dietz. 2007. Maternal obesity and risk of gestational diabetes mellitus. Diabetes Care 30(8): 2070-2076.
Dahlgren J. 2006. Pregnancy and insulin resistance. Metabolic Syndrome and Related Disorders 4(2): 149-152.
Deeks J. J., J. Dinnes, R. D’Amico, A. J. Sowden, C. Sakarovitch, F. Song, M. Petticrew and D. G. Altman. 2003. Evaluating non-randomised intervention studies. Health Technology Assessment 7(27): iii-x, 1-173.
DeVader S. R., H. L. Neeley, T. D. Myles and T. L. Leet. 2007. Evaluation of gestational weight gain guidelines for women with normal prepregnancy body mass index. Obstetrics and Gynecology 110(4): 745-751.
Dodds L., D. B. Fell, K. S. Joseph, V. M. Allen and B. Butler. 2006. Outcomes of pregnancies complicated by hyperemesis gravidarum. Obstetrics and Gynecology 107(2 Pt 1): 285-292.
Downs S. H. and N. Black. 1998. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. Journal of Epidemiology and Community Health 52(6): 377-384.
Edwards L. E., W. L. Hellerstedt, I. R. Alton, M. Story and J. H. Himes. 1996. Pregnancy complications and birth outcomes in obese and normal-weight women: effects of gestational weight change. Obstetrics and Gynecology 87(3): 389-394.
Ekblad U. and S. Grenman. 1992. Maternal weight, weight gain during pregnancy and pregnancy outcome. International Journal of Gynaecology and Obstetrics 39(4): 277-283.
Getahun D., C. Nath, C. V. Ananth, M. R. Chavez and J. C. Smulian. 2008. Gestational diabetes in the United States: temporal trends 1989 through 2004. American Journal of Obstetrics and Gynecology 198(5): 525e521-e525.
Gin H., A. Vambergue, C. Vasseur, V. Rigalleau, P. Dufour, A. Roques, M. Romon, D. Millet, P. Hincker and P. Fontaine. 2006. Blood ketone monitoring: a comparison between gestational diabetes and non-diabetic pregnant women. Diabetes and Metabolism 32(6): 592-597.
Graves B. W., S. A. DeJoy, A. Heath and P. Pekow. 2006. Maternal body mass index, delivery route, and induction of labor in a midwifery caseload. Journal of Midwifery and Women’s Health 51(4): 254-259.
Gregg E. W., Y. J. Cheng, B. L. Cadwell, G. Imperatore, D. E. Williams, K. M. Flegal, K. M. Narayan and D. F. Williamson. 2005. Secular trends in cardiovascular disease risk factors according to body mass index in US adults. Journal of the American Medical Association 293(15): 1868-1874.
Gunderson E. P., M. A. Murtaugh, C. E. Lewis, C. P. Quesenberry, D. S. West and S. Sidney. 2004. Excess gains in weight and waist circumference associated with childbearing: The Coronary Artery Risk Development in Young Adults Study (CARDIA). International Journal of Obesity and Related Metabolic Disorders 28(4): 525-535.
Gunderson E. P., B. Abrams and S. Selvin. 2000. The relative importance of gestational gain and maternal characteristics associated with the risk of becoming overweight after pregnancy. International Journal of Obesity and Related Metabolic Disorders 24(12): 1660-1668.
Gunderson E. P., B. Sternfeld, M. F. Wellons, R. A. Whitmer, V. Chiang, C. P. Quesenberry, Jr., C. E. Lewis and S. Sidney. 2008. Childbearing may increase visceral adipose tissue independent of overall increase in body fat. Obesity (Silver Spring) 16(5): 1078-1084.
Hackmon R., R. James, C. O’Reilly Green, A. Ferber, Y. Barnhard and M. Divon. 2007. The impact of maternal age, body mass index and maternal weight gain on the glucose challenge test in pregnancy. Journal of Maternal-Fetal & Neonatal Medicine 20(3): 253-257.
Harris H. E., G. T. Ellison and S. Clement. 1999. Relative importance of heritable characteristics and lifestyle in the development of maternal obesity. Journal of Epidemiology and Community Health 53(2): 66-74.
Hilakivi-Clarke L., R. Luoto, T. Huttunen and M. Koskenvuo. 2005. Pregnancy weight gain and premenopausal breast cancer risk. Journal of Reproductive Medicine 50(11): 811-816.
Hilson J. A., K. M. Rasmussen and C. L. Kjolhede. 2006. Excessive weight gain during pregnancy is associated with earlier termination of breast-feeding among White women. Journal of Nutrition 136(1): 140-146.
IOM (Institute of Medicine). 1990. Nutrition During Pregnancy. Washington, DC: National Academy Press.
Jain N. J., C. E. Denk, L. K. Kruse and V. Dandolu. 2007. Maternal obesity: can pregnancy weight gain modify risk of selected adverse pregnancy outcomes? American Journal of Perinatology 24(5): 291-298.
Jenkin W. and M. Tiggemann. 1997. Psychological effects of weight retained after pregnancy. Women & Health 25(1): 89-98.
Jensen D. M., P. Ovesen, H. Beck-Nielsen, L. Molsted-Pedersen, B. Sorensen, C. Vinter and P. Damm. 2005. Gestational weight gain and pregnancy outcomes in 481 obese glucose-tolerant women. Diabetes Care 28(9): 2118-2122.
Johnson J. W., J. A. Longmate and B. Frentzen. 1992. Excessive maternal weight and pregnancy outcome. American Journal of Obstetrics and Gynecology 167(2): 353-370; discussion 370-352.
Joseph K. S., D. C. Young, L. Dodds, C. M. O’Connell, V. M. Allen, S. Chandra and A. C. Allen. 2003. Changes in maternal characteristics and obstetric practice and recent increases in primary cesarean delivery. Obstetrics and Gynecology 102(4): 791-800.
Kabiru W. and B. D. Raynor. 2004. Obstetric outcomes associated with increase in BMI category during pregnancy. American Journal of Obstetrics and Gynecology 191(3): 928-932.
Keppel K. G. and S. M. Taffel. 1993. Pregnancy-related weight gain and retention: implications of the 1990 Institute of Medicine guidelines. American Journal of Public Health 83(8): 1100-1103.
Kieffer E. C., W. J. Carman, B. W. Gillespie, G. H. Nolan, S. E. Worley and J. R. Guzman. 2001. Obesity and gestational diabetes among African-American women and Latinas in Detroit: implications for disparities in women’s health. Journal of the American Medical Women’s Association 56(4): 181-187, 196.
Kieffer E. C., B. P. Tabaei, W. J. Carman, G. H. Nolan, J. R. Guzman and W. H. Herman. 2006. The influence of maternal weight and glucose tolerance on infant birthweight in Latino mother-infant pairs. American Journal of Public Health 96(12): 2201-2208.
Kiel D. W., E. A. Dodson, R. Artal, T. K. Boehmer and T. L. Leet. 2007. Gestational weight gain and pregnancy outcomes in obese women: how much is enough? Obstetrics and Gynecology 110(4): 752-758.
Kinnunen T. I., R. Luoto, M. Gissler, E. Hemminki and L. Hilakivi-Clarke. 2004. Pregnancy weight gain and breast cancer risk. BMC Women’s Health 4(1): 7.
Ko C. W. 2006. Risk factors for gallstone-related hospitalization during pregnancy and the postpartum. American Journal of Gastroenterology 101(10): 2263-2268.
Langford A., C. Joshu, J. J. Chang, T. Myles and T. Leet. 2008. Does gestational weight gain affect the risk of adverse maternal and infant outcomes in overweight women? Maternal and Child Health Journal. Epub ahead of print.
Lederman S. A., A. Paxton, S. B. Heymsfield, J. Wang, J. Thornton and R. N. Pierson, Jr. 1997. Body fat and water changes during pregnancy in women with different body weight and weight gain. Obstetrics and Gynecology 90(4 Pt 1): 483-488.
Li R., S. Jewell and L. Grummer-Strawn. 2003. Maternal obesity and breast-feeding practices. American Journal of Clinical Nutrition 77(4): 931-936.
Lim S., S. H. Choi, Y. J. Park, K. S. Park, H. K. Lee, H. C. Jang, N. H. Cho and B. E. Metzger. 2007. Visceral fatness and insulin sensitivity in women with a previous history of gestational diabetes mellitus. Diabetes Care 30(2): 348-353.
Lindseth G. and M. Y. Bird-Baker. 2004. Risk factors for cholelithiasis in pregnancy. Research in Nursing and Health 27(6): 382-391.
Linne Y., L. Dye, B. Barkeling and S. Rossner. 2003. Weight development over time in parous women—the SPAWN study—15 years follow-up. International Journal of Obesity and Related Metabolic Disorders 27(12): 1516-1522.
Linne Y., L. Dye, B. Barkeling and S. Rossner. 2004. Long-term weight development in women: a 15-year follow-up of the effects of pregnancy. Obesity Research 12(7): 1166-1178.
Luke B., M. L. Hediger and T. O. Scholl. 1996. Point of diminishing returns: when does gestational weight gain cease benefiting birthweight and begin adding to maternal obesity? Journal of Maternal-Fetal Medicine 5(4): 168-173.
Madlon-Kay D. J. 1993. Striae gravidarum. Folklore and fact. Archives of Family Medicine 2(5): 507-511.
Manios Y., E. Grammatikaki, K. Kondaki, E. Ioannou, A. Anastasiadou and M. Birbilis. 2008. The effect of maternal obesity on initiation and duration of breast-feeding in Greece: the GENESIS study. Public Health Nutrition 1-8.
Marrero J. M., P. M. Goggin, J. S. de Caestecker, J. M. Pearce and J. D. Maxwell. 1992. Determinants of pregnancy heartburn. British Journal of Obstetrics and Gynaecology 99(9): 731-734.
Murakami M., M. Ohmichi, T. Takahashi, A. Shibata, A. Fukao, N. Morisaki and H. Kurachi. 2005. Prepregnancy body mass index as an important predictor of perinatal outcomes in Japanese. Archives of Gynecology and Obstetrics 271(4): 311-315.
Muscati S. K., K. Gray-Donald and K. G. Koski. 1996. Timing of weight gain during pregnancy: promoting fetal growth and minimizing maternal weight retention. International Journal of Obesity and Related Metabolic Disorders 20(6): 526-532.
Must A., P. F. Jacques, G. E. Dallal, C. J. Bajema and W. H. Dietz. 1992. Long-term morbidity and mortality of overweight adolescents. A follow-up of the Harvard Growth Study of 1922 to 1935. New England Journal of Medicine 327(19): 1350-1355.
Nohr E. A., M. Vaeth, J. L. Baker, T. Sorensen, J. Olsen and K. M. Rasmussen. 2008. Combined associations of prepregnancy body mass index and gestational weight gain with the outcome of pregnancy. American Journal of Clinical Nutrition 87(6): 1750-1759.
Ogunyemi D., S. Hullett, J. Leeper and A. Risk. 1998. Prepregnancy body mass index, weight gain during pregnancy, and perinatal outcome in a rural black population. Journal of Maternal-Fetal Medicine 7(4): 190-193.
Ohlin A. and S. Rossner. 1990. Maternal body weight development after pregnancy. International Journal of Obesity 14(2): 159-173.
Olson C. M., M. S. Strawderman, P. S. Hinton and T. A. Pearson. 2003. Gestational weight gain and postpartum behaviors associated with weight change from early pregnancy to 1 y postpartum. International Journal of Obesity and Related Metabolic Disorders 27(1): 117-127.
Parham E. S., M. F. Astrom and S. H. King. 1990. The association of pregnancy weight gain with the mother’s postpartum weight. Journal of the American Dietetic Association 90(4): 550-554.
Parker J. D. and B. Abrams. 1992. Prenatal weight gain advice: an examination of the recent prenatal weight gain recommendations of the Institute of Medicine. Obstetrics and Gynecology 79(5 Pt 1): 664-669.
Purfield P. and K. Morin. 1995. Excessive weight gain in primigravidas with low-risk pregnancy: selected obstetric consequences. Journal of Obstetric, Gynecologic, and Neonatal Nursing 24(5): 434-439.
Rasmussen K. M., J. A. Hilson and C. L. Kjolhede. 2002. Obesity as a risk factor for failure to initiate and sustain lactation. Advances in Experimental Medicine and Biology 503: 217-222.
Rodriguez A., G. Bohlin and G. Lindmark. 2001. Symptoms across pregnancy in relation to psychosocial and biomedical factors. Acta Obstetricia et Gynecologica Scandinavica 80(3): 213-223.
Rooney B. L. and C. W. Schauberger. 2002. Excess pregnancy weight gain and long-term obesity: one decade later. Obstetrics and Gynecology 100(2): 245-252.
Rooney B. L., C. W. Schauberger and M. A. Mathiason. 2005. Impact of perinatal weight change on long-term obesity and obesity-related illnesses. Obstetrics and Gynecology 106(6): 1349-1356.
Rosenberg L., J. R. Palmer, L. A. Wise, N. J. Horton, S. K. Kumanyika and L. L. Adams-Campbell. 2003. A prospective study of the effect of childbearing on weight gain in African-American women. Obesity Research 11(12): 1526-1535.
Rosenberg T. J., S. Garbers, H. Lipkind and M. A. Chiasson. 2005. Maternal obesity and diabetes as risk factors for adverse pregnancy outcomes: differences among 4 racial/ethnic groups. American Journal of Public Health 95(9): 1545-1551.
Saldana T. M., A. M. Siega-Riz, L. S. Adair and C. Suchindran. 2006. The relationship between pregnancy weight gain and glucose tolerance status among black and white women in central North Carolina. American Journal of Obstetrics and Gynecology 195(6): 1629-1635.
Scholl T. O., M. L. Hediger, J. I. Schall, I. G. Ances and W. K. Smith. 1995. Gestational weight gain, pregnancy outcome, and postpartum weight retention. Obstetrics and Gynecology 86(3): 423-427.
Seghieri G., A. De Bellis, R. Anichini, L. Alviggi, F. Franconi and M. C. Breschi. 2005. Does parity increase insulin resistance during pregnancy? Diabetic Medicine 22(11): 1574-1580.
Shepard M. J., A. F. Saftlas, L. Leo-Summers and M. B. Bracken. 1998. Maternal anthropometric factors and risk of primary cesarean delivery. American Journal of Public Health 88(10): 1534-1538.
Sherrard A., R. W. Platt, D. Vallerand, R. H. Usher, X. Zhang and M. S. Kramer. 2007. Maternal anthropometric risk factors for caesarean delivery before or after onset of labour. British Journal of Obstetrics and Gynaecology 114(9): 1088-1096.
Sibai B. M., M. Ewell, R. J. Levine, M. A. Klebanoff, J. Esterlitz, P. M. Catalano, R. L. Goldenberg and G. Joffe. 1997. Risk factors associated with preeclampsia in healthy nulliparous women. The Calcium for Preeclampsia Prevention (CPEP) Study Group. American Journal of Obstetrics and Gynecology 177(5): 1003-1010.
Soltani H. and R. B. Fraser. 2000. A longitudinal study of maternal anthropometric changes in normal weight, overweight and obese women during pregnancy and postpartum. British Journal of Nutrition 84(1): 95-101.
Stevens-Simon C. and E. R. McAnarney. 1992. Adolescent pregnancy. Gestational weight gain and maternal and infant outcomes. American Journal of Diseases of Children 146(11): 1359-1364.
Stotland N. E., L. M. Hopkins and A. B. Caughey. 2004. Gestational weight gain, macrosomia, and risk of cesarean birth in nondiabetic nulliparas. Obstetrics and Gynecology 104(4): 671-677.
Thadhani R., M. J. Stampfer, D. J. Hunter, J. E. Manson, C. G. Solomon and G. C. Curhan. 1999. High body mass index and hypercholesterolemia: risk of hypertensive disorders of pregnancy. Obstetrics and Gynecology 94(4): 543-550.
Thorsdottir I., J. E. Torfadottir, B. E. Birgisdottir and R. T. Geirsson. 2002. Weight gain in women of normal weight before pregnancy: complications in pregnancy or delivery and birth outcome. Obstetrics and Gynecology 99(5 Pt 1): 799-806.
Troiano R. P., E. A. Frongillo, Jr., J. Sobal and D. A. Levitsky. 1996. The relationship between body weight and mortality: a quantitative analysis of combined information from existing studies. International Journal of Obesity and Related Metabolic Disorders 20(1): 63-75.
Tulman L., K. H. Morin and J. Fawcett. 1998. Prepregnant weight and weight gain during pregnancy: relationship to functional status, symptoms, and energy. Journal of Obstetric, Gynecologic, and Neonatal Nursing 27(6): 629-634.
Viswanathan M., A. M. Siega-Riz, M.-K. Moos, A. Deierlein, S. Mumford, J. Knaack, P. Thieda, L. J. Lux and K. N. Lohr. 2008. Outcomes of Maternal Weight Gain, Evidence Report/Technology Assessment No. 168. (Prepared by RTI International-University of North Carolina Evidence-based Practice Center under contract No. 290-02-0016.) AHRQ Publication No. 08-E-09. Rockville, MD: Agency for Healthcare Research and Quality.
Walker L. O. 1996. Predictors of weight gain at 6 and 18 months after childbirth: a pilot study. Journal of Obstetric, Gynecologic, and Neonatal Nursing 25(1): 39-48.
Walker L. O. 1997. Weight and weight-related distress after childbirth: relationships to stress, social support, and depressive symptoms. Journal of Holistic Nursing 15(4): 389-405.
Walker L., J. H. Freeland-Graves, T. Milani, G. George, H. Hanss-Nuss, M. Kim, B. S. Sterling, G. M. Timmerman, S. Wilkinson, K. L. Arheart and A. Stuifbergen. 2004. Weight and behavioral and psychosocial factors among ethnically diverse, low-income women after childbirth: II. Trends and correlates. Women & Health 40(2): 19-34.
Wataba K., T. Mizutani, K. Wasada, M. Morine, T. Sugiyama and N. Suehara. 2006. Impact of prepregnant body mass index and maternal weight gain on the risk of pregnancy complications in Japanese women. Acta Obstetricia et Gynecologica Scandinavica 85(3): 269-276.
Witter F. R., L. E. Caulfield and R. J. Stoltzfus. 1995. Influence of maternal anthropometric status and birth weight on the risk of cesarean delivery. Obstetrics and Gynecology 85(6): 947-951.
Young T. K. and B. Woodmansee. 2002. Factors that are associated with cesarean delivery in a large private practice: the importance of prepregnancy body mass index and weight gain. American Journal of Obstetrics and Gynecology 187(2): 312-318; discussion 318-320.
Zhang C., K. M. Rexrode, R. M. van Dam, T. Y. Li and F. B. Hu. 2008. Abdominal obesity and the risk of all-cause, cardiovascular, and cancer mortality: sixteen years of follow-up in US women. Circulation 117(13): 1658-1667.






















