5
Patient-Driven Rapid Learning Systems
Patients are increasingly playing an active role in driving the development of a cancer RLHS, reflecting the greater participation of patients in their healthcare decision-making processes. “I have been in practice long enough to have gone from being trained as an oracle that tells people what to do, to being asked to help patients understand what the information they have collected means to them. Multiple patients demonstrated to me that they knew more about their conditions didactically than I did,” noted Dr. Wallace. “As we are thinking about generating knowledge, it is really prudent for us to think about the knowledge needed by the patient, and what the patient can help us learn,” he added.
Wide-scale use of the Internet and social networking sites and tools by patients has also fostered the rapid gathering and spread of information about various conditions, explained Susannah Fox, associate director of digital strategy for the Pew Internet and American Life Project. A 2009 survey done by this project found that 8 out of 10 adults in the United States have access to the Internet and two-thirds have broadband at home. Information seeking and reporting are also more mobile and rapid, Fox noted, as 56 percent of American adults regularly go online wirelessly with their phones, laptops, or other portable devices, the survey revealed. “There are rapid learning systems that are ad hoc that are going on right now, and you should be aware of them because patients are using them,” Fox said, adding that “even though there are only about 80 of us in this room today, there are
thousands of people who can follow what we are doing,” via “tweets” she had simultaneously posted on Twitter. “The Internet is moving from being a stationary, slow, desktop-based information vending machine into what is now a fast, mobile, communications appliance,” Fox added.
This information technology is enabling many different ways for people not only to consume information about health and health care, but also to contribute information, Fox said. The Pew survey revealed that 52 percent of Internet users watch videos online, more than one-third share online photos, including X-rays and other medical graphics, and one out of five Internet users with cancer uses social network sites. “The Internet is really changing people’s expectations of what should be available to them, and there is a broad uptake for social media in health,” said Fox. “We are seeing that patients are looking for stories about people who are just ahead of them on the path, and they are learning from each other. Patients are doing the data collection that you crave, and they are ready to participate and be seen as your colleagues.”
Fox pointed out that between 2006 and 2008, Pew surveys found that the proportion of American adults who responded that they, or someone they knew, had been helped by medical advice they found online went from one-quarter to nearly one-half. Only 3 percent responded in the most recent survey that they had been harmed from medical advice they found online. Fox concluded by saying, “Researchers and clinicians can take advantage of what patients are already doing outside the system and welcome them into the system.”
An online leader in gathering and disseminating information about cancer to those affected and their caregivers is the international Association of Cancer Online Resources (ACOR), the largest online social network for cancer patients. Composed of close to 200 separate online support groups and social networks for individuals with cancer, ACOR has served more than a half-million cancer patients and caregivers and currently has 60,000 members. ACOR delivers an average of 1.5 million e-mails weekly to its members, including a tremendous amount of the latest scientific information about their disorder and clinical trials in which they can participate, reported Gilles Frydman, who founded ACOR in 1995. “ACOR is not a chat room,” he said, although it does provide members with the opportunity to share their personal stories with an emphasis on medically or quality-of-life significant events. One of the goals of the organization is patient empowerment and activation, and accelerating access to relevant
information with unmatched currency, bypassing what Frydman calls the “built-in lethal lag time” of professional research publishing.
ACOR also fosters data collection from within its online cancer communities. Such data collection includes postmarketing self-reporting, as well as group-wide data collection with an emphasis on adverse event reporting. For example, the International Myeloma Foundation used ACOR’s myeloma and breast cancer listserv to conduct a survey, whose results it used for a ground-breaking postmarketing study on bisphosphonates and necrosis of the jaw. This study was published in the New England Journal of Medicine and led Novartis to change information about the drug on its label. Similarly, reports of kidney toxicity on the ACOR listserv by myeloma patients taking Zometa® led to experts’ calling for slowing the infusion time of the drug. Myeloma patients were quickly notified of this change via the listserv, and kidney problems were rapidly and dramatically reduced, Frydman reported. “We see daily conversations about side effects that are not found in FDA publications,” he said.
ACOR has also literally saved many lives and led to record-breaking clinical trial accruals, according to Frydman. For example, ACOR rapidly informed its members with gastrointestinal stromal tumors (GISTs) as soon as word got out that Gleevec® was likely to be effective for these tumors, leading to remarkably short accrual times for a clinical trial of Gleevec for GISTs. “Novartis told me they expected their accrual time for the trial to be three years, and it was done in just over eight months, with 18 percent of the accrued patients coming through our system,” said Frydman.
He summed up his presentation by stressing that “activated patients are the most underutilized healthcare resource. Social networks of patients suffering from a rare disease may often be the best resource for high-quality information. Network patients are inventing and shaping a better healthcare model where their input is constant and central.” However, he added that patient input requires patients’ having access to all their medical data and the option to participate in the decision-making process.
New technologies are empowering patients and patient-driven research organizations,” said Dr. Simone Sommer, president of the Chordoma Foundation, co-founded with her son Josh, who was diagnosed with a chordoma in 2005. She expressed impatience with “the slow wheels of science. Healthcare organizations and patient advocacy groups like ours are going to play a much more active role as we have the highest stakes and the greatest motivation to change the system, and we cannot wait.” Dr. Sommer added
that the speed and quality of patient care are proportional to the flow of information and urged that there be open access to medical literature, and that venues and systems be created that unite disparate researchers, patients, and physicians to accelerate the process of searching for treatments for cancers, especially rare cancers such as chordoma, where both researchers and patients are few and far between.
The need for rapid learning and global networking is most pressing for patients with rare, life-threatening diseases, such as chordoma. Dr. Sommer and her son Josh spoke about their efforts to spur such learning and networking so as to more quickly find and deliver effective treatment for chordoma. There are no approved drugs for this condition, which is generally resistant to chemotherapy. With only 300 new diagnoses per year in the United States, and 20 isolated researchers studying the condition, the main barrier the Sommers found to making progress on the disease was a lack of communication, collaboration, and coordination among stakeholders, including patients, researchers, physicians, scientists, and industry, resulting in scant evidence of progress against this rare condition.
There are few clinical studies on chordoma because of the rarity of the condition. Yet because of its fatal nature, many patients with chordoma are subjected to experimental treatments, such as off-label use of cancer drugs that are approved for other malignancies. “One oncologist described it as throwing drugs at the disease until something sticks. When there is no other option, oncologists become resourceful and, in essence, they do experiments on their patients,” Dr. Sommer said. She described this process as “learning chaotically” and added, “Every day all over the world, chordoma patients and patients with all sorts of rare cancers are being experimented on, and isn’t it a shame that those experiments are not adding to our knowledge about the disease and informing care of future patients?” She called for setting up a system in which science can use such data that are being generated but not collected. Patient networks can quickly disperse any anecdotal information about experimental treatments to their members. Dr. Sommer noted that patients learn fast but there are several shortcomings in such patient-to-patient learning. These shortcomings include survivor bias that distorts the findings. “The dataset is enriched with patients who have responded to a particular treatment because you do not hear from the patients who did not respond,” Dr. Sommer pointed out. In addition, anecdotes are not generalizable, especially for chordoma, which is a heterogeneous disease. “While these social networks and online tools are very valuable for patients to learn about new potential therapies, there is a
risk in patients’ relying on these anecdotal reports and thinking that what happens to someone else should guide their own therapy, because in reality, their tumors are very different. We need to capitalize on patients’ ability to talk and communicate with one another, but do it in a scientific manner, and not in a patient-to-patient manner,” she said.
To further research, the Chordoma Foundation is creating a systems approach featuring a centralized chordoma biospecimen bank and patient registry that links ongoing, prospectively collected patient data to biospecimens that researchers can use in their studies. The Sommers are in the process of finalizing a contract with Ohio State University to house the biobank, which will operate on the caBIG platform (see Figure 5-1).
As Josh Sommer pointed out, chordoma patients often have several surgeries at different institutions, and no one hospital has all the tumor specimens or complete records on patients, so having a centralized repository is a valuable asset that will spur research on chordoma.
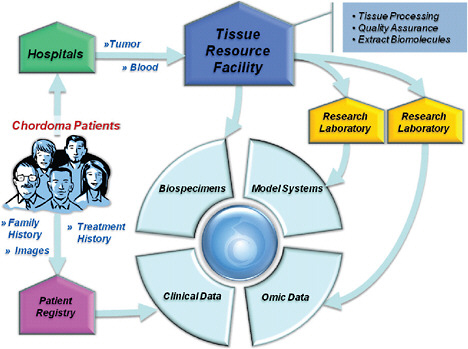
FIGURE 5-1 An example of a patient-driven rapid learning system for a rare cancer, chordoma.
SOURCE: Reprinted, with permission, from The Chordoma Association 2010. Copyright 2010 by the Chordoma Association.
Dr. Sommer ended her presentation by pointing out that patients are eager to contribute to research, but rightfully expect to benefit from results. Physicians and researchers need to respect patient privacy, as well as figure out how to quickly, safely, and ethically feed results back to patients, she said. “We need to look forward in a way that we make sure that the new frontiers include patients and their personalized health care,” Dr. Sommer said. Dr. Sommer also stressed the need for portable, longitudinal records and biospecimens tied to the patient and not to the locale and hospital, given that medical care is often done at multiple locations.
Like chordoma, amyotrophic lateral sclerosis (ALS) is a rare and fatal disease. Having his brother diagnosed with the disease spurred Jamie Heywood to create a patient online social networking site called PatientsLikeMeTM (PatientsLikeMe, 2009). The goal of this organization is to enable people to share their experiences and learn from and connect with others like them. The website provides a platform for collecting and sharing real-world, outcome-based patient data and is establishing data-sharing partnerships with doctors, pharmaceutical and medical device companies, research organizations, and nonprofits. “PatientsLikeMe is a new social contract that says if you share everything about yourself, including your DNA, your blood, your tumor, your symptoms, every drug you take, and your side effects, we will use that information in an open medical network that really begins to transform what is possible in a new way,” said Heywood.
The basic premise of PatientsLikeMe is that if enough patient-reported anecdotal information is collected, it can be turned into useful data if the right questions are asked and properly structured analyses are done on the data. PatientsLikeMe can use the information it collects to provide patients with basic predictions, such as their likely timetables of disease progression. PatientsLikeMe has created new computer analysis tools that can provide personalized predictions, based on the wealth of multivariate patient data collected on its site that are matched to individual data entered into the system by the patient requesting such predictions. These analyses strive to control patient selection bias in their careful matching process, which compares, for example, ALS patients taking the experimental treatment lithium with those ALS patients on the site who opt not to take the drug, yet have the same disease onset time, degree of disease progression, age, and so forth, as the treated group. “We integrate across every variable we know about the patients over the entire history of time we know about them, and match them to controls,” said Heywood.
A PatientsLikeMe analysis revealed that lithium was not effectively
improving symptoms or delaying disease progression in patients who took the drug, refuting preliminary evidence in the medical literature published 18 months earlier from a small group of patients suggesting it might be an effective treatment for ALS. Heywood claimed that his study had four times the power of this original preliminary study, using only patient volunteers from the “real world.” “Patient data can up-power your double-blind study because we know how to integrate the prior information about the patient’s data into the equation,” Heywood said. Random controls are not a “patient like me.” PatientsLikeMe discovered and disseminated its findings about lithium to its ALS members one year before a large-scale clinical trial testing lithium as a treatment for ALS was stopped because of futility. “They would not have released that data, via a patient press release, if we had not put our data out ahead of time,” claimed Heywood. “So not only have we begun to deliver meaningful answers ahead of time, but we have changed the expectation for the trial community to deliver data to the patient community,” he added, noting that the negative clinical trial study results were never published.
PatientsLikeMe is doing similar studies of patients with other disorders such as leukemia, Parkinson’s disease, and multiple sclerosis, and Heywood urged the medical community to fund the infrastructure needed for these observational research endeavors. “I think that we have, in medicine, failed to deliver the platform that allows the innovators that will cure disease to do it effectively,” he said, claiming that these platforms will continue to be developed by and for patients and that it would behoove the established medical research community to collaborate with such projects.
Later during discussion, Dr. John Mendelsohn, from the M.D. Anderson Cancer Center, pointed out that his center has an open records Internet site called “My MD Anderson” in which physicians and patients share information. “The website is not complete yet, but it is something that I think will improve our ability to give care,” he said. He then asked Heywood how PatientsLikeMe decided what data to collect on patients and whether such decisions were patient driven. Heywood responded that the questions asked patients are usually modified from a standard set. “We adapt existing questions because when we test them on the patients, we generally find the existing measures are not describing what the patients feel is important,” he said.
Dr. Hal Sox noted that the larger and more representative a patient population one has to work with, the more likely progress is to be made, and asked Heywood what proportions of people with the diseases tracked
by PatientsLikeMe use the site. Heywood responded that 20 percent of the ALS patients who were diagnosed in the United States within the last six months joined PatientsLikeMe. “We have about 11 percent of the U.S. ALS population in the website right now, and of those, about 60 percent are giving us what we call a clinical-trial-a-year’s worth of data.” Josh Sommer added that about one-fifth to one-quarter of all U.S. chordoma patients are represented in the Chordoma Foundation database. Heywood noted that “there is a minimum scale of about 500 patients that we find needed to get an effective dialogue going, and there appears to be a ceiling of about 11,000 to 12,000 patients within a single community that we need to figure out how to break through with our current information architecture.” Heywood implied that the high degree of dialogue that occurs on some sites limits the number of people that can effectively participate in them. Dr. Sommer also noted generational differences, with older individuals perhaps not as willing to be open and share everything online as the younger generation (e.g., on Facebook).








