4
Sharing the Responsibility for a Risk-Based System: Models of Governance and Oversight
The safety of the U.S. domestic and imported food system is a responsibility shared by suppliers, farmers, food handlers, processors, wholesalers and retailers, food service companies, consumers, third-party organizations, and government (federal and state) agencies in the United States and abroad. Given the size and scope of the system, it is unrealistic to expect the U.S. Food and Drug Administration (FDA), or any agency at the federal level, to be everywhere and to do everything necessary to ensure food safety through surveillance and inspection without the help of those who share this responsibility.
The design of approaches to governance to achieve society’s goals has been the subject of much debate and experimentation in a wide range of areas, from the financial system to public safety. The published literature on the subject addresses the pros and cons of various approaches to sharing responsibility, factors to be considered, and lessons learned from the implementation of these approaches. Models of governance that deviate from the traditional enforcement of rules through the imposition of penalties include voluntary approaches whereby regulators work with industry to develop codes of practice, third-party audits, management-based systems in which firms are responsible for adhering to plans that limit harms, and performance-based approaches that emphasize results rather than the use of specific technologies or actions. These alternatives may serve to distribute accountability across all parties that might affect outcomes.
Innovative governance approaches have already been applied to the environment, building safety, consumer product safety, nuclear power plant safety, transportation safety, and health care, among many other areas, with
|
BOX 4-1 Examples of the Use of Alternative Governance Approaches Nuclear Power Safety Before the Nuclear Regulatory Commission (NRC) focused attention on issues essential to protecting public health, the Atomic Energy Commission was often criticized for its dual role in protecting public health while also avoiding imposing requirements that would inhibit the growth of the industry. With respect to nuclear reactors, the NRC took the traditional approach of creating standards and requirements to protect public health, eventually giving operators the sense that accidents would be prevented as long as compliance with these standards and requirements was verified by an inspector. This traditional prescriptive approach, however, was criticized as being unable to promote uniform levels of safety. The NRC then moved toward a risk-based system, whereby accountability is placed on the operator’s side. However, the Government Accountability Office has noted major challenges to the success of this system, including the need to encourage a shift to a culture of safety, significant human capital needs and costs, and methodological challenges (GAO, 2006). British Railway System Potential limitations of implementing novel governance approaches in the health and safety arena may be evident in the experience of the British railway system. Hutter (2001) suggests that such a move may have led to breaches in public safety. Often, a self-regulatory regime is seen as a superior governance model in that it relies not only on government accountability, but also on the capacity of corporations to regulate themselves and develop systems tailored to their specific operations. Innovation is encouraged, and companies are more likely to follow their own rules than rules imposed on them. Hutter argues that in the case of the railway industry in Britain, enforced self-regulation was not appropriately monitored and ended up being itself the source of risk. In fact, the self-regulation was more procedural than substantive; although rules were in place, they were not well understood. Lack of communication was a major explanation for the failure of the system in a company that was fragmented functionally and geographically. |
both failure and success. Examples are presented in Box 4-1. These examples illustrate that developing criteria for selecting a governance approach, making the selection, and evaluating performance outcomes are essential activities for regulatory agencies. These two examples are but a small sampling of the many models of regulation and oversight that exist, and the
selection of the most appropriate model for specific circumstances is a subject of active debate. Even within the area of food safety, several different models of governance are evolving worldwide (Batz et al., 2005; Garcia-Martinez et al., 2007; Treasury Board of Canada Secretariat, 2007).
Chapter 3 describes the elements that are essential to the operation of a risk-based food safety system, as concluded by the committee. A governance model for the FDA must articulate criteria for deciding who is responsible for overseeing the various elements, for choosing and implementing policy interventions, and for evaluating the performance of the system. Defining the nature and range of shared responsibility is central to implementing several of these elements. This need for clearly reasoned models for shared responsibility and oversight is the subject of this chapter. The chapter reviews approaches to making governance decisions and developing a regulatory philosophy, as well as choosing policy interventions and assigning responsibility. The discussion includes the committee’s observations on how the FDA selects models of governance.
OVERALL APPROACH TO MAKING GOVERNANCE DECISIONS AND DEVELOPING A REGULATORY PHILOSOPHY
The Food Protection Plan (FPP), written in 2007 under the leadership of the Office of Food Protection, contains the FDA’s general philosophy with respect to food safety and focuses on what the agency considers to be the core elements of food safety: prevention, intervention, and response (see Box 4-2). The FPP also outlines the following four cross-cutting principles for a comprehensive food protection approach: (1) focus on risks over a product’s life cycle from production to consumption, (2) target resources to achieve maximum risk reduction, (3) address both unintentional and deliberate contamination, and (4) use science and modern technology systems. To operationalize these elements and principles and to strengthen its ability to protect Americans from foodborne illnesses, the FDA proposes internal administrative changes and recognizes the need to make legislative changes (Box 4-3). The FPP is a platform for initiating a transformation at the FDA, whereby policy decisions are based on prevention and risk. However, it does not provide detail on how the principles it outlines will be achieved. The committee supports the findings of the U.S. Government Accountability Office (GAO) (GAO, 2008a,b) that the plan does not offer specific strategies for many of the actions proposed. For example, although it refers to risk-based inspections, detail on analytical risk models or even factors that will be considered in developing such models is absent. The terms “risk” and “risk-based approaches” are understood in different ways, underlining the importance of detailed articulation of such factors. Indeed, Chapter 3 explains the importance of a regulatory agency’s delineating in detail a broad strategic
|
BOX 4-2 Three Core Elements of Food Safety in the U.S. Food and Drug Administration’s Food Protection Plan Prevent foodborne contamination:
Intervene at critical points in the food supply chain:
Respond rapidly to minimize harm:
|
approach that explains its philosophy, that is, the factors it will weigh in making decisions about prioritization of efforts, allocation of resources, and selection of interventions. The committee concluded that the FPP should be supported by the kind of detailed strategic planning (both broad and specific) outlined in Chapter 3. To illustrate this shortcoming, this section describes the committee’s understanding of the FPP’s vision for the responsibilities of different parties involved in food safety and how it could be improved.
As part of the strategic planning process (Step 1 in the risk-based system described in Chapter 3), the responsibilities of all parties in achieving the desired level of food safety must be articulated. Because these responsibilities will vary with the situation, and new situations are always arising, there must also be a road map for assigning responsibilities based on a defined set of factors. These elements of a risk-based system constitute an agency’s regulatory philosophy.
The FPP makes several statements about the responsibilities of different parties in the food safety system. A major plank of its prevention strategy is a call for promoting increased corporate responsibility to prevent food-borne illness. The plan notes that examples of enhanced corporate responsibility might include “evaluating safety and security vulnerabilities and possible impacts; when appropriate, implementing preventive measures—
|
BOX 4-3 Additional Protections That Involve Legislative Changes to the U.S. Food and Drug Administration’s (FDA’s) Authority Prevent foodborne contamination:
Intervene at critical points in the food supply chain:
Respond rapidly to minimize harm:
|
both required and voluntary—to ensure that food is produced safely and securely; and developing a contingency plan to aid in a response in the event of contamination” (FDA, 2007a, p. 14). The plan states that an increased emphasis on prevention “will require close interaction with growers, manufacturers, distributors, retailers and food service providers, and importers. These partners have the ability to implement preventive approaches and to require them of their suppliers” (p. 11).
The FPP also states that:
[t]hose with the biggest stake in food safety, after the consumers who eat the food, are the people and companies who grow, process, and sell
food. Their livelihood depends entirely on the confidence of their customers. A poor reputation for proper food handling can drive a company to bankruptcy. Promoting increased corporate responsibility is key in shifting FDA’s food protection effort to a proactive rather than a reactive one. The FDA will seek partnerships with industry to enhance consumer confidence. FDA will continue to work with industry in a) developing food protection plans that address safety and defense vulnerabilities, b) implementing prevention steps, and c) developing contingency plans to improve response to an outbreak of foodborne illness. (p. 15)
In addition, the FPP supports exploring new roles for third-party certification as part of the overall system of food safety assurance. As to working with other responsible parties, the plan states:
FDA will continue to work with industry, state, local, and foreign governments to further develop the tools and science needed to identify vulnerabilities and determine the most effective approaches. With regard to imports, FDA will also work with foreign governments, which have a greater ability to oversee manufacturers within their borders to ensure compliance with safety standards. (p. 11)
Finally, concerning consumer responsibility, the plan notes, “Consumers protect themselves and their families from foodborne illness by responding promptly to FDA alerts” (p. 23).
The above statements indicate that the FDA is focusing on the need for shared responsibility in designing its food safety program. In several exchanges with FDA staff, however, the committee did not find that the FDA has a well-thought-out approach to defining food safety responsibilities beyond these general statements. On several occasions, for example, the committee invited FDA officials to further articulate what the agency sees as the substance and consequences of the FPP’s call for placing more responsibility on the corporate sector. The officials were unable to do so, nor did their answers recognize the need for a systematic approach (a road map) to making these decisions. The agency’s approach appears to be ad hoc and its regulatory philosophy unclear.
Describing the role of each responsible party is an important activity for a regulatory agency and an essential element of its strategic plan. A model for choosing modes of governance is integral to the subsequent choice of interventions and their design and implementation (Steps 4 and 5 of a risk-based system). This model should account for a range of factors that will differ across risks, such as the sources and controllability of risks and the structure of the supply chain, and will affect what mix of shared responsibility will address the risks most effectively.
A generic list of governance options is a useful starting point for think-
ing about shared responsibility for food safety. An example of such as a list is shown in Figure 4-1 (adapted from Garcia-Martinez et al., 2007). On one end of the spectrum, food safety is entirely an individual, private responsibility, and there is no intervention by public agencies. On the other end of the spectrum is direct regulation, whereby public agencies prescribe what companies must or must not do in ensuring food safety, for example, with respect to production practices, product standards, or labeling. This end of the spectrum is frequently referred to as a “command and control” approach. Between these extremes is a range of public–private mixes. Self-regulation involves the use of industry voluntary codes of practice and farm assurance schemes with self- or third-party certification. Information and education entails the government’s generating and communicating information for the use of private parties. Coregulation denotes programs in which responsibility is shared in a public–private partnership, for example, when statutes incorporate industry codes of practice. Finally, incentive-based
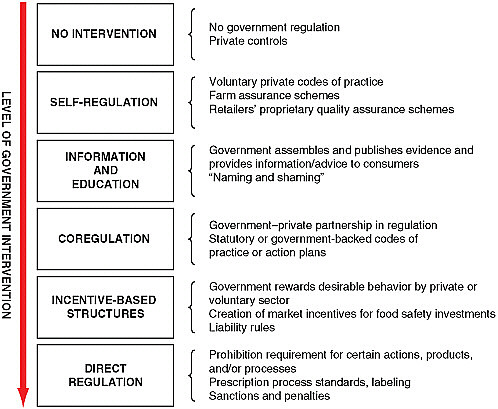
FIGURE 4-1 Options for assigning private–public responsibility to ensure food safety.
SOURCE: Adapted from Garcia-Martinez et al. (2007).
structures vary the amount and type of regulatory oversight based on how well a company performs; this is frequently referred to as a performance approach and also includes the setting of liability rules and related concepts, such as due diligence.
The nature of shared responsibility for the management of food safety risks will evolve over time as legislation is passed, new circumstances arise, knowledge grows, stakeholders express different priorities, and constraints
|
BOX 4-4 Managing the Safety of Produce: An Example of Evolving Shared Responsibility An example of how the U.S. Food and Drug Administration (FDA) has modified its governance philosophy over time is the case of produce safety. While the FDA has jurisdiction over produce, in the past it did not exercise this authority through direct regulation. This lack of direct oversight occurred in part because the FDA gave priority to its efforts to control contaminants in foods known to present such problems, and at the time fresh produce was not recognized as an important vehicle for pathogens. Until recently, there were no guidelines, codes of practice, or regulations directed toward ensuring the safety of fresh produce during production and processing. The FDA started to pay more attention to produce safety when various produce items were identified as vehicles for foodborne illness outbreaks. Recent examples of FDA attempts to manage the safety of fresh produce include the Tomato Safety Initiative (FDA, 2007b), the Lettuce Initiative (FDA, 2009a), and Produce Safety from Production to Consumption (FDA, 2004). Important efforts common to all these initiatives were continuing to reach out to the produce industry, facilitating and promoting research, and working with federal, state, and local public health officials in illness detection and outbreak response. These efforts are examples of an information and education approach to intervention (see Figure 4-1). The FDA first developed Guidelines for Agricultural Practices in 1998. They were followed by guidelines for minimizing or eliminating microbial contamination in commodities that appear to present the greatest risks: tomatoes, leafy greens, and melons. As guidelines, however, none of these documents are enforceable. To encourage the farm community to accept and adopt them, the FDA has engaged in information and education programs, for example, through dedicated efforts by cooperative extension offices. The lack of strong regulatory action by the FDA drove some states to |
shift. Based on outcomes, the mix of responsibility chosen initially may prove to be too reliant on voluntary action, at one end of the spectrum, or too focused on prescriptive government regulations, at the other end. A salient example of this evolution is the FDA’s regulatory approach to the safety of produce (see Box 4-4). The committee notes that an evolving approach makes sense, but found that the FDA’s approach frequently
|
implement stricter measures. For example, the Tomato Good Agricultural Practices (Florida Department of Agriculture and Consumer Services, 2007) are now included in a rule aimed at enhancing the safety of fresh tomatoes produced or handled in Florida; this is an example of direct regulation. Although voluntary, the California Leafy Greens Marketing Agreement (LGMA, 2010), overseen by the California Department of Food and Agriculture, is a mechanism for verifying that participating growers (99 percent of leafy green vegetable production volume) follow specific food safety practices. A similar program implemented in Arizona covers approximately 75 percent of leafy green vegetables produced in the state (AZLGMA, 2008). These efforts are a form of coregulation. In fall 2009, the FDA announced that the agency will issue regulations setting enforceable standards for fresh produce safety at the farm and packing house, based on prevention-oriented public health principles and on current knowledge and guides (HHS/FDA, 2009). The FDA’s proposed rule would establish standards for the implementation of preventive controls, emphasizing the importance of environmental assessments and recognizing the need to tailor preventive controls to particular hazards and operations. This shift in the FDA’s governance approach to produce safety from an educational model to direct regulation could be due to many factors, including new research findings, an increased rate of foodborne illness that suggests higher risk attributed to produce, a low rate of implementation or effectiveness of FDA guidelines, or a change in general philosophy about the management of food safety within the agency. In fact, communications from the FDA about what is expected of industry and regulatory approaches taken over the years have not clearly articulated the rationale for changes or provided a road map that would enable stakeholders to participate in and anticipate such changes. |
cannot be tied systematically to an underlying regulatory philosophy and related road map for making these decisions.
Regardless of the governance models selected or policy interventions used to achieve them, food safety will always be the responsibility of many partners. Thus cooperation and collaboration are key not only in the collection, analysis, and sharing of information and data but also in the enforcement and oversight of policies. A lack of cooperation and collaboration among the many entities with responsibility for food safety results in an inefficient food safety system. To be credible, the development of governance models must be done with transparency and stakeholder involvement.
CHOOSING POLICY INTERVENTIONS AND ASSIGNING/SHARING RESPONSIBILITY
A risk-based approach entails identifying important risks to target and stating the means that will be used to control them. Many factors enter into the selection and design of policy interventions (Steps 4 and 5 in a risk-based system). Given the complexity involved (multiple risks, multiple candidate interventions, uncertainty of information), regulatory agencies benefit from having a risk-based road map for identifying and selecting interventions. Once policy interventions have been selected, assigning responsibility to different parties in the system is an important aspect of their implementation.
The Policy Interventions Tool Kit
Governments and their regulatory agencies can chose from a broad range of possible interventions to influence the performance of markets. It is useful to think of these interventions as a tool kit offering multiple options, depending on the job at hand. In its main document on intervention choice, the Treasury Board of Canada Secretariat (2007) uses the term “policy instruments” to refer to this set of interventions, defined as follows:
Instruments for government action are the means a government has at its disposal to achieve public policy outcomes—to govern. While several definitions of “instruments for government action” exist, this document uses a broad interpretation, defining them as the “means by which policy objectives are pursued” [emphasis in original]. Instruments for government action set up relationships between the state and its citizens. In some cases, such as criminal law, the relationship is of a coercive nature. In other cases, such as legal agreements, the relationship is reciprocal. (p. 3)
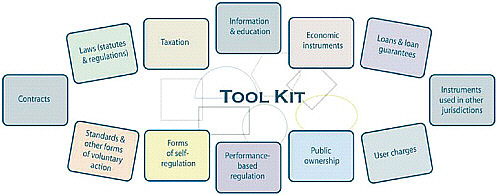
FIGURE 4-2 The interventions tool kit.
SOURCE: Treasury Board of Canada Secretariat, 2007.
Figure 4-2 shows the classes of interventions outlined in the document. These tools are frequently used in combination to achieve the desired performance outcomes.
Road Maps for Choosing Policy Interventions
Deciding what policy interventions to use in different situations and determining the associated assignment of responsibilities is facilitated by having a road map of factors to consider in the selection process. It is common for multiple interventions to be in place simultaneously. For example, processing standards may ensure food safety, while consumer labeling educates about safe use. Referring to Figure 4-1, explicitly thinking about which level of intervention or mix of levels to use and why can lead to choices that enhance the effectiveness of the food safety system.
As mentioned above, the committee asked FDA officials to explain the FDA’s thought process in selecting interventions. From these discussions and a review of FDA documents, the committee concluded that the FDA does not have a systematic method for making these decisions at Step 4 of the risk-based approach. Several countries have developed road maps of the type suggested by the committee. For example, the United Kingdom’s Food Standards Agency has in place a regulatory framework1 (FSA, 2006), and a set of detailed impact assessments has been completed (FSA, 2010).2 Box 4-5 presents an example of a road map for choosing interventions, developed by the Treasury Board of Canada Secretariat (2007).
|
1 |
See http://www.food.gov.uk/foodindustry/regulation/betregs/regframe (accessed February 12, 2010). |
|
2 |
See http://www.food.gov.uk/foodindustry/regulation/betregs/ria (accessed February 12, 2010). |
|
BOX 4-5 Example Analytical Framework for Selecting Policy Instruments The Treasury Board of Canada Secretariat (2007) has developed a framework (see the figure below) for selecting policy instruments (its term for what this report calls interventions) for use by all departments and agencies, which may use the framework as is or as a template for developing their own framework for their respective areas of responsibility. The framework is intended to facilitate a disciplined approach to assessing, selecting, and implementing instruments. According to the Secretariat, the framework establishes a sequence of enquiry, specifies a methodological foundation, and provides guidance for each step in the instrument choice process. The benefits identified as flowing from the use of this framework are 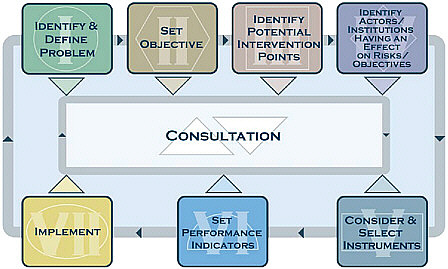 |
Assigning/Sharing Responsibility
The FDA or any agency charged with managing food safety must have mechanisms for overseeing food safety both domestically and internationally (for imported foods). Different intervention choices incorporate different assignments of responsibility for ensuring that the desired level of food safety assurance is achieved. The key parties to whom different levels of responsibility for food safety may be assigned include the private sec-
The Secretariat states that the framework is based on two overarching rationales:
The framework is not intended to be a sequential road map of where and how officials should assess instruments to achieve public policy objectives. The process is inherently iterative in that the accumulation of information and knowledge concerning a problem or situation and the objectives the government is aiming to achieve will require officials to revisit each of the steps in the framework repeatedly. The framework document presents simple but complete approaches to each step of the instrument choice process. SOURCE: Treasury Board of Canada Secretariat, 2007. |
tor, third-party or other accrediting organizations, governments of other countries, and the states.
The Private Sector
A regulatory agency needs to set clear food safety standards and enforce those standards. At the same time, industry has, and must have, the primary
responsibility for ensuring food safety because it is the sector that actually makes or grows the products and is in closest touch with problems as they occur. Industry has broad roles to play; for example, it conducts research on mitigation strategies to produce solutions for food safety practices. Another role of industry is to innovate and explore management approaches. For example, systems analogous to the Hazard Analysis and Critical Control Points (HACCP) system were already in existence and had been applied in some food processing operations prior to being considered by the government as a preventive approach (IOM/NRC, 2003).
In HACCP-based systems, industry formulates control plans that the regulatory agency oversees. When reviewing a HACCP plan, the agency can determine whether the technologies proposed are adequate for food safety protection and are being used appropriately.
Private-sector responsibility is carried out within the range of intervention strategies outlined above. For example, industry responsibility may vary if the government has no intervention strategy. It may take the form of complying with information interventions, for example, with the new requirement of a reportable food registry. Tort law, tax incentives, subsidies, other incentive-based interventions, and direct regulation are other strategies for producing the desired level of food safety. As noted above, the preferred choice of interventions and related assignment of responsibility evolve over time. Box 4-6 describes a current example of this evolution in the area of traceability.
Third-Party Certification
Interest has grown in the use of quality assurance by accreditation bodies (third-party certification) to ensure food safety rather than (or in addition to) relying on government agencies. These assurance/accreditation bodies may be an industry group (the self-regulation option of Figure 4-1) or a third party that is independent of individual firms or the government. They can develop and accredit standards, providing assurance to buyers in the supply chain and/or to consumers. In coregulation interventions, such bodies can partner with the government to ensure food safety. In incentive-based interventions, they can be used as a means of credibly signaling lower risk to the government and may lead to streamlined oversight (e.g., to a fast track for imports).
The use of third-party accreditation as an aspect of government interventions is controversial and, at this time, is more accepted in some countries than in others. Significant questions arise as to how and by whom the accreditation bodies themselves are audited, how transparent they are, to what extent they solicit and use stakeholder input, and whether the audits are reliable (Albersmeier et al., 2009). In the context of a risk-based food safety
|
BOX 4-6 Example of the Evolution of Shared Responsibility for Traceability The term “food traceability” can be defined generally as the ability to identify where a food comes from. In the area of food safety, traceability refers to the ability to identify a food product’s history (e.g., processes, locations, manufacturers). Past experience with foodborne illness or contamination investigations has demonstrated that determining the history of a food product from production to consumption can be a daunting, time- and resource-consuming effort in the United States, but one that is absolutely necessary to making decisions during and after a crisis. With the idea of providing food agencies with prompt, necessary information, Section 306 of the Public Health Security and Bioterrorism Preparedness and Response Act of 2002 requires the establishment and maintenance of records that allow for identification of the immediate previous sources and subsequent recipients of food.a This information, along with labels that identify the contents of the package and the name and address of the manufacturer, packer, or distributor must be made available to the U.S. Food and Drug Administration (FDA) within 24 hours when food contamination is suspected. However, the lack of guidance for best practices and the fact that companies already follow their own traceability procedures for other purposes (e.g., safety, quality, marketing) have resulted in a diverse system with limited value for the FDA. Food traceability for safety purposes is an example of a situation in which simply letting individual food companies establish procedures with little guidance, coordination, or leadership has not led to a well-functioning system. For traceability to be useful during a crisis, procedures need to be seamless and effective. Setting standards is essential. Because both industry and government play distinctive roles (i.e., the system needs to be feasible and practical for industry but also needs to be usable by government investigators), it may be necessary for them to set the standards in collaboration, with a clear definition of the roles of the partners. As this report is being written, the FDA and the U.S. Department of Agriculture have engaged in a joint dialogue with industry to address past inefficiencies by developing procedures that will be useful during investigations (Federal Register, 2009). Collaboration of this type can move systems. forward to meet both societal and industry needs. |
system, a key question is whether these systems meet standards for being risk based and, in particular, how well they address public health issues.
The rapid growth of auditing platforms (e.g., those of the Global Food Safety Initiative, the International Organization for Standardization, Safe Quality Food [SQF], the British Research Consortium [BRC], the Global Partnership for Good Agricultural Practice [GlobalGAP]) shows that supply chains see value in these systems. Interest in leveraging these systems in government regulation and oversight stems from a desire to gain possible efficiencies in the production of food safety through the elimination of duplication of effort. Increased reliance on these systems, however, requires regulatory agencies to institute a system for auditing the auditors and setting standard criteria for these operations.
The FDA has been exploring this issue. For example, as noted earlier, the FPP calls for new legislative authority to authorize the agency to accredit highly qualified third parties for voluntary food inspections. This legislation would authorize the FDA to accredit independent third parties (or to recognize accrediting bodies) to evaluate compliance with FDA requirements, allowing the agency to allocate inspection resources more effectively. According to the FPP,
FDA would use information from these accredited third-party organizations in its decision making but not be bound by such information in determining compliance with FDA requirements. Use of accredited third parties would be voluntary and might offer more in-depth review and possibly faster review times and expedited entry for imported goods manufactured in facilities inspected by accredited third parties. Use of accredited third parties may also be taken into consideration by the FDA when setting inspection and surveillance priorities. (p. 18)
The FDA proposes to oversee these independent third parties by auditing their work to ensure that FDA requirements are consistently assessed, reviewing their inspection reports, and providing ongoing training criteria to ensure that they maintain their skills and knowledge (FDA, 2007a). It should be noted the FPP defines third parties much more broadly than is the case in this report. Included in its definition are other federal departments and agencies, state and local government agencies, foreign government agencies, and private entities without financial conflicts of interest (FDA, 2007a). The committee believes the FDA’s definition is too broad (see Chapter 7).
The FDA’s 2009 Guidance for Industry on Voluntary Third-Party Certification Programs for Foods and Feeds3 describes the agency’s views on the
|
3 |
See http://www.fda.gov/RegulatoryInformation/Guidances/ucm125431.htm (accessed February 16, 2010). |
general attributes of a third-party certification program. The FDA regards this guidance as one of the steps in its future recognition of voluntary third-party certification programs for particular product types, and it has stated that it will recognize a certification program only if it has sufficient confidence in the certification body (FDA, 2009b). The FDA also has explored the use of third-party certification for imported foods, as discussed in the following section. It should be noted that, although GAO reports on food safety programs recommend exploring the development of a third-party certification program, they also recommend taking lessons from the FDA’s medical device program, in which the lack of incentives resulted in weak participation and few inspections (GAO, 2008a,b).
The potential value and legitimacy of third-party certification is a topic of debate internationally as well. For example, private standards have been on the agenda of the World Trade Organization’s (WTO’s) Sanitary and Phytosanitary (SPS) committee since 2005, and their role in the process of public standards setting is under discussion at the Codex Alimentarius Commission, where a position on the matter has not yet been taken. During its last meeting in 2009, Codex decided to conduct an in-depth evaluation of the role and impact of such standards, based on comments about the negative impact of private standards on economies and questions about the science and transparency of the process (Henson and Humphrey, 2009). There is published evidence of the comparatively higher costs of meeting private standards versus European Union (EU) standards (Plunkett and DeWaal, 2008). Overall, however, third-party efforts are clearly an important part of a risk-based system of shared responsibility for food safety.
Governments of Other Countries
There have been only limited, initial efforts to compare food safety performance across countries (Charlebois and Yost, 2008); therefore, no evidence exists to support the idea that vulnerability will increase with the growth of international trade in food and agricultural products and the import share of food consumption. The enforcement of food safety regulations in foreign countries is challenging.
Importing countries ensure import safety through a combination of controls in place in the exporting countries and border inspections. It is unrealistic to expect the FDA to have an effective inspectional presence in countries around the world as border inspection is a difficult, expensive, and sometimes ineffectual means of monitoring food safety. Inspectors cannot check every grape, or even every box of grapes. In this situation, a U.S. regulatory agency may leverage its efforts by verifying and then relying on the safety control systems of other countries. This approach has the added advantage of responding to the call of WTO’s Agreement on the
Application of SPS Measures for recognition of equivalent systems across countries.
An additional challenge in the oversight of imported foods is the inability of a government to interfere with a foreign country’s laws. Exporting countries are outside the FDA’s jurisdiction, and therefore enforcing U.S. food laws with respect to their products is problematic. For example, inspecting foods and facilities in situ not only is impractical but also might not be welcomed or allowed by the exporting country. A regulatory agency needs oversight mechanisms that can overcome these barriers while remaining in line with WTO trade agreements. The current system by which the FDA manages the safety of food imports (mainly inspections at the border) is ineffective (only 1.28 percent of shipments were inspected in 2007) and could use additional tools (GAO, 2009).
As Appendix E describes, with the expansion of the global market for foods and the signing of the WTO agreements, preventive mechanisms have been instituted to ensure the safety of imported foods. Those mechanisms include monitoring and directed sampling (Canada); third-party audits, and equivalency agreements, and limited entry posts for high-risk products (EU countries); inspection based on food risk categories (Australia and New Zealand); and import certificates (New Zealand). Presentations made to the committee and its own investigation further support the existence of a broad range of approaches to allocation of responsibility and coordination with other countries to ensure food safety (Appendix E). Designing a coherent approach to working with other countries to ensure the safety of imports is clearly important.
The committee found that the FPP does not articulate a clear approach to the roles of private parties and other governments in ensuring food safety for products imported into the United States. As discussed in Appendix E, the United States maintains that its approach to imported foods stands on the same general principles as its approach to domestic foods. It is the responsibility of companies and importers to know the U.S. food laws and regulations and to comply with them. However, the U.S. government will ultimately be held accountable for a safe supply of both domestic and imported foods.
The States
Given the size, complexity, and growth of the food industry in the United States (more than 156,008 domestic food facilities [FDA, 2010], more than 1 million food establishments [including restaurants and retail stores4], and more than 2 million farms), it is unrealistic to expect that the
FDA could have enough resources to provide adequate surveillance and inspection of the entire U.S. food supply (Mavity, 2009). The regulations and programs of state and local (including tribal and territorial) governments have been a strong component of the U.S. food safety system for the past century. State surveys conducted in 2001 and 2009 indicate the broad scope of food safety activities conducted by the states, from collecting data on food contamination and outbreak surveillance, to performing food and feed inspections, to enforcing the laws and issuing recalls (AFDO, 2001, 2009). In fact, the FDA’s food safety knowledge (and therefore management) could be enhanced by leveraging data collected by state and local authorities on food safety inspections, disease outbreak investigations, product safety, consumer perspectives, and enforcement actions. Doing so, however, would require that programs be standardized and harmonized; for example, standards for training of inspectors and data collection would need to be in place. In the absence of truly harmonized programs at the state and local levels, the FDA has instituted some mechanisms that facilitate cooperation, such as the signing of confidentiality agreements, contracts, or memorandums of understanding. Although these mechanisms facilitate shared effort, they also have limitations in that funding is not always available, and they are not always utilized.
As with the assignment of responsibility to industry, third parties, and other countries, the FDA needs an overall strategic vision for when it is desirable to rely on or partner with the states to ensure food safety as well as what allocation of appropriate areas and levels of responsibility is optimal. The committee found that the FDA lacks an overall regulatory philosophy or road map for these choices. With a clear approach, the agency might be able to expand its collaborations with state and local food safety programs so these programs would be better recognized and utilized in the national food safety system (see Chapter 7).
Examples of Mixes of Public and Private Responsibility
Clearly options for the choice and design of policy interventions (Steps 4 and 5 in a risk-based system) are broad, cutting across different mixes of public and private responsibility for ensuring food safety. Researchers have begun to analyze these diverse models for shared responsibility, particularly as several countries have expressed their interest in newer, hybrid forms of governance as a means of ensuring food safety more efficiently. As yet, there have been no comprehensive comparisons of the effectiveness of these alternative models, but several studies shed some light on the options currently in use.
The structure of private standards for food safety management has been developing particularly rapidly in the last decade (Henson, 2008). Histori-
cally, a no-intervention approach characterized by private standards set on a business-to-business basis was predominant. These approaches were either national (e.g., Nature’s Choice by Tesco in the United Kingdom, Field-to-Forks by Marks and Spencer in the United Kingdom, Filière Agriculture-Raisonnée by Auchan France) or international (e.g., Wal-Mart and Nestlé). Recently, a self-regulatory approach characterized by joint standards used by a group of suppliers or retailers, frequently with third-party certification, has been gaining ground. Examples of these joint standards include, at the national level, the Dutch HACCP, the BRC Global Standard, Assured Food Standards, Qualität und Sicherheit (the “QS system”), and Integrate Keten Beheersing. At the international level, they include the International Food Standards, SQF 1000/2000/3000, and GlobalGAP (formerly EuroGAP) (Henson, 2008).
A study conducted for the Food Standards Agency in the United Kingdom documents a mix of private and public responsibility in use across the United Kingdom, the United States, Canada, and Australia (Fearne et al., 2006; Garcia-Martinez et al., 2007). The United Kingdom has been active in thinking about and experimenting with different mixes of responsibility. An example is the Zoonoses Action Plan Salmonella Programme for pigs. In this case, standards setting was private (voluntary), with funding from the government and a multistakeholder group advising on ongoing developments. Implementation was private, with funding and facilitation from the government. Enforcement and monitoring were private as well (as part of farm assurance scheme requirements), with the public sector providing on-farm support and advice to high-risk producers. A further example of exploration of alternative public–private mixes is the voluntary HACCP Advantage program in Ontario, Canada. Here, the standards setting was public–private, the system was introduced through educational programs led by the government, and enforcement and monitoring were conducted through private, third-party audits.
An example of exploration of different mixes of public and private responsibility from the United States is a series of efforts the FDA has conducted to assess the value of third-party certification systems as a tool to verify the compliance of foreign food companies with U.S. food laws. Such exploratory efforts are recommended in the Action Plan for Import Safety: A Roadmap for Continual Improvement (Interagency Working Group on Import Safety, 2007), as well as in the 2007 FPP. The FDA conducted a pilot study to evaluate voluntary third-party certification programs for imported aquaculture shrimp. The FDA envisioned that such a program could help the agency make decisions about the safety of imported foods, such as prioritizing inspections and sampling. The pilot program was conducted in two phases. During Phase I, participants were paper audited and selected on the basis of a set of criteria, with six certification bodies selected. Phase II,
involving onsite audits and targeted sampling, was scheduled to be completed and evaluated in July 2009. The committee was not given any results of this pilot program and was unable to evaluate it as an approach to shared responsibility for food safety.
KEY CONCLUSIONS AND RECOMMENDATIONS
Food safety in the United States is the responsibility of suppliers, farmers, food handlers, processors, wholesalers and retailers, food service companies, consumers, third-party organizations, and government (federal and state) agencies in both the United States and abroad. It is, therefore, unrealistic to expect the FDA, or any government agency, to have sufficient resources to manage food safety without the help of others who share this responsibility.
A risk-based approach to the choice and design of interventions (Steps 4 and 5 in a risk-based system) requires a comprehensive understanding of the policy intervention tool kit and a road map for choosing and designing interventions. Further, developing an approach to defining the roles of other responsible parties is a component of strategic planning in a risk-based food safety system. In essence, this road map should also serve to assign shared responsibility among the federal government, the private sector, third parties, the governments of other countries, the states, and consumers. The design of novel approaches to governance to achieve food safety is currently the subject of experimentation by other governments and debate by scholars.
The committee found that the FDA has made ad hoc efforts in this direction but does not have a clear regulatory philosophy for assigning responsibility for food safety or a comprehensive strategy for choosing the level and intensity of interventions as part of strategic planning in a risk-based approach. The committee offers the following recommendations to address these shortcomings.
Recommendation 4-1: To ensure food safety, the FDA should develop a plan for defining the extent of and form for sharing responsibilities with the states, the private sector, third parties (e.g., independent auditors), and other countries’ governments.
Recommendation 4-2: The FDA should develop a comprehensive strategy for choosing the level and intensity of policy interventions needed for different food safety risks. Criteria for choosing the level and intensity of policy interventions and a plan for evaluating the selected interventions should be developed with transparency, stakeholder participation, and clear lines of communication.
REFERENCES
AFDO (Association of Food and Drug Officials). 2001. State Resource Survey. York, PA: AFDO.
AFDO. 2009. State Food Safety Resource Survey. York, PA: AFDO.
Albersmeier, F., H. Schulze, G. Jahn, and A. Spiller. 2009. The reliability of third-party certification in the food chain: From checklists to risk-oriented auditing. Food Control 20:927–935.
AZLGMA (Arizona Leafy Green Products Shipper Marketing Agreement). 2008. About AZ LGMA 2008. http://www.azlgma.gov/about_azlgma/default.asp (accessed February 12, 2010).
Batz, M. B., M. P. Doyle, G. Morris, Jr., J. Painter, R. Singh, R. V. Tauxe, M. R. Taylor, and D. M. Lo Fo Wong. 2005. Attributing illness to food. Emerging Infectious Diseases 11(7):993–999.
Charlebois, S., and C. Yost. 2008. Food Safety Performance World Ranking 2008. Saskatchewan, Canada: Research Network in Food Systems, University of Regina. http://www.ontraceagrifood.com/admincp/uploadedfiles/Food%20Safety%20Performance%20World%20Ranking%202008.pdf (accessed March 13, 2010).
FDA (U.S. Food and Drug Administration). 2004. Produce Safety from Production to Consumption: 2004 Action Plan to Minimize Foodborne Illness Associated with Fresh Produce Consumption. Washington, DC: FDA. http://www.fda.gov/Food/FoodSafety/Product-SpecificInformation/FruitsVegetablesJuices/FDAProduceSafetyActivities/ProduceSafetyActionPlan/ucm129487.htm (accessed February 4, 2010).
FDA. 2007a. Food Protection Plan: An Integrated Strategy for Protecting the Nation’s Food Supply. Rockville, MD: FDA.
FDA. 2007b. Tomato Safety Initiative. Washington, DC: FDA. http://www.fda.gov/Food/FoodSafety/Product-SpecificInformation/FruitsVegetablesJuices/FDAProduceSafetyActivities/ucm115334.htm (accessed February 4, 2010).
FDA. 2009a. Lettuce Safety Initiative. Washington, DC: FDA. http://www.fda.gov/Food/FoodSafety/Product-SpecificInformation/FruitsVegetablesJuices/FDAProduceSafetyActivities/ucm115906.htm (accessed February 4, 2010).
FDA. 2009b. Guidance for Industry: Voluntary Third-Party Certification Programs for Foods and Feeds. http://www.fda.gov/RegulatoryInformation/Guidances/ucm125431.htm (accessed February 16, 2010).
FDA. 2010. Food and Drug Administration FY 2011 Congressional Budget Request: Narrative by Activity—Foods Program FY2011. http://www.fda.gov/downloads/AboutFDA/ReportsManualsForms/Reports/BudgetReports/UCM205367.pdf (accessed June 2, 2010).
Fearne, A., M. Garcia, J. Caswell, S. Henson, and Y. Khatri. 2006. Exploring Alternative Approaches to Traditional Modes of Food Safety. London, UK: Imperial College/Food Standards Agency. http://www.foodbase.org.uk//admintools/reportdocuments/202-1-337_D03004.pdf (accessed February 16, 2010).
Federal Register. 2009. Product tracing systems for food. 74(211):56843–56855.
Florida Department of Agriculture and Consumer Services. 2007. Tomato Best Practices Manual: DACS Tomato Best Practices Manual. Tallahassee: Florida Department of Agriculture and Consumer Services. www.doacs.state.fl.us/fs/TomatoBestPractices.pdf (accessed March 30, 2010).
FSA (Food Standards Agency). 2006. FSA Regulatory Framework. http://www.food.gov.uk/foodindustry/regulation/betregs/regframe (accessed February 12, 2010).
FSA. 2010. Impact Assessments 2009. http://www.food.gov.uk/foodindustry/regulation/betregs/ria/ (accessed February 12, 2010).
Garcia-Martinez, M., A. Fearne, J. A. Caswell, and S. Henson. 2007. Co-regulation as a possible model for food safety governance: Opportunities for public–private partnerships. Food Policy 32(3):16.
GAO (U.S. Government Accountability Office). 2006. Nuclear Regulatory Commission: Oversight of Nuclear Power Plant Safety Has Improved, but Refinements Are Needed. Washington, DC: GAO.
GAO. 2008a. Federal Oversight of Food Safety: FDA Has Provided Few Details on the Resources and Strategies Needed to Implement Its Food Protection Plan. Washington, DC: GAO.
GAO. 2008b. Federal Oversight of Food Safety: FDA’s Food Protection Plan Proposes Positive First Steps, but Capacity to Carry Them Out Is Critical. Washington, DC: GAO.
GAO. 2009. Agencies Need to Address Gaps in Enforcement and Collaboration to Enhance Safety of Imported Food. Washington, DC: GAO.
Henson, S. 2008. The role of public and private standards in regulating international food markets. Journal of International Agricultural Trade and Development 4(1):63–81.
Henson, S., and J. Humphrey. 2009. The Impacts of Private Food Safety Standards on Food Chain and on Public Standard-Setting Processes. Paper presented at Report to the Codex Alimentarius Commission, 32nd Session, Rome, Italy, June 29–July 4, 2009.
HHS/FDA (U.S. Department of Health and Human Services/U.S. Food and Drug Administration). 2009. Produce Safety Regulation, RIN #: 0910-AG35. http://www.reginfo.gov/public/do/eAgendaViewRule?pubId=200910&RIN=0910-AG35 (accessed May 13, 2010).
Hutter, B. M. 2001. Is enforced self-regulation a form of risk taking? The case of railway health and safety. International Journal of the Sociology of Law 29(4):379–400.
Interagency Working Group on Import Safety. 2007. Action Plan for Import Safety: A Roadmap for Continual Improvement. http://www.importsafety.gov/ (accessed April 22, 2010).
IOM/NRC (Institute of Medicine/National Research Council). 2003. Scientific Criteria to Ensure Safe Food. Washington, DC: The National Academies Press.
LGMA (California Leafy Green Products Handler Marketing Agreement). 2010. About LGMA 2007. http://www.caleafygreens.ca.gov/about/lgma.asp (accessed January 28 2010).
Mavity, S. 2009. Safety of Imported Foods: A Perspective from a Company in the Seafood Industry. Paper presented at Institute of Medicine/National Research Council Committee on Review of the FDA’s Role in Ensuring Safe Food Meeting, Washington, DC, May 28, 2009.
Plunkett, D., and C. S. DeWaal. 2008. Who is responsible for the safety of food in a global market? Government certification v. importer accountability as models for assuring the safety of internationally traded foods. Food and Drug Law Journal 63(3):657–664.
Treasury Board of Canada Secretariat. 2007. Assessing, Selecting, and Implementing Instruments for Government Action. Ottawa: Treasury Board of Canada Secretariat.
























