2
Visioning Perspectives on the
Digital Health Utility
INTRODUCTION
Building an effective learning health system requires a shared vision among a wide array of stakeholders with sometimes highly varied perspectives. Chapter 2 captures several of these perspectives, including those of the patient, the healthcare team, the quality and safety community, clinical researchers, and the population health community. The included manuscripts explore the current state of the digital infrastructure from their corresponding perspective, articulate their views of the potential for a learning health system supported by an integrated digital infrastructure, and identify sector-specific needs and priorities for progress.
Adam Clark, formerly of the Lance Armstrong Foundation (now FasterCures), shares his vision of a learning health system characterized by bidirectional exchange of health information (individuals are both donors and consumers). He describes the need to develop appropriate interfaces to encourage and facilitate participation in order to support this vision. This includes not only providing the most appropriate information to consumers in a format that is accessible to them, but accommodating the participation of family members and caregivers. Dr. Clark highlights the value of including consumers as information donors in the learning health system, pointing to their ability to contribute types of information—such as accounts of fatigue or depression—and provide a level of context that would otherwise not be captured. He cites data from the Lance Armstrong Foundation indicating that individuals want to share this information as long as their privacy concerns are addressed. Dr. Clark observes that the escalating comp-
lexity of medicine demands new kinds of relationships between patients, clinicians, and researchers, and that the digital infrastructure can serve as a platform for this going forward.
The perspective of the healthcare team is explored by Jim Walker of Geisinger Health System. He defines a learning health system as one of goal-oriented feedforward and feedback loops that create actionable information. Dr. Walker describes his experiences with health information technology (HIT) implementation at Geisinger and highlights the complex, sociotechnical nature of the challenge—requiring as much attention to the social aspects as is currently being given to technical capacity. Citing examples of healthcare system learning needs—such as the proper second-line treatment for diabetes—Walker lays out the potential for a learning system to address these questions and feed that information back to healthcare team members. He concludes by noting that this goal will require fundamental HIT systems redesign in order to support healthcare team decision making.
Janet Corrigan from the National Quality Forum (NQF) observes that little progress had been made to improve quality and safety since the publication of the Quality Chasm report (IOM, 2001), and that value has concurrently decreased. She states that increases in safety, quality, and effectiveness of health care will require investments in a digital infrastructure capable of collecting information across the longitudinal “patient-focused episode,” and feeding back performance results along with clinical decision support for patients and clinicians. Dr. Corrigan describes the framework used by NQF to develop measures for reporting and value-based purchasing, and explores how a digital infrastructure could support capturing the relevant data. Finally, she states that achieving better health outcomes will require collecting information from, and enabling communication with, individuals both within and outside of traditional healthcare settings.
The growing information intensity of modern medicine and biomedical research, coupled with advances in computing capabilities, define the clinical research perspective as articulated by Christopher Chute from the Mayo Clinic. He observes that given these concurrent conditions, the technical requirements for information and knowledge management in health should be high-priority issues. Drawing from examples of “big science” disciplines such as astronomy and physics, he suggests that the future of biology and medicine will be characterized by collaborative efforts and shared data and knowledge. As such, he points to the need for standardization in order to allow for comparability and consistency in health information. Reviewing the historical state of standards uptake and development efforts, he suggests that meaningful use may be a transformative effort that moves health care in this direction.
Martin LaVenture, Sripriya Rajamani, and Jennifer Fritz from the Minnesota State Department of Health share their account of the opportunities
and challenges surrounding a digital platform that supports population health activities. Acknowledging that the learning health system holds great promise for the improvement of health at the population level, they describe the need to improve the capacity and capabilities of population health services in order to realize this potential. The principal challenge, they note, is the lack of an integrated, modernized digital health infrastructure that is used by a trained workforce and stewarded by public health leaders who understand the potential benefits for population health. Accordingly, they articulate the need for a more unified vision of a digital infrastructure for population health, including development of a population health approach to data standards; aggregation and infrastructure; and intelligent, bidirectional messaging for patients and consumers.
INFORMED AND EMPOWERED PATIENTS:
MOVING BEYOND A BYSTANDER IN CARE
Adam M. Clark, Ph.D.
Lance Armstrong Foundation (former)
FasterCures
The concept of a “learning health system” is one in which knowledge generation occurs as a natural outgrowth of healthcare delivery leading to improvements in innovation, quality, safety, and value in care while being inclusive of both patient and provider preferences (IOM, 2007). Fundamental and essential to the success of this concept are the two roles individuals will play in a bidirectional exchange as consumers and donators of health information. As consumers of healthcare information and utilities, a learning health system should provide individuals with information that is understandable, is pertinent to their health at the appropriate time, and is information they can act upon. The semantic content of the information will vary depending on where the individual is in the care continuum and whether the individual is acting as a patient, a caregiver, or a loved one. This will become increasingly important with the shift toward personalized medicine where prevention, screening, treatment, and care decisions become tailored to the individual.
As health information technology (HIT) continues to mature, individuals will increasingly participate in the meaningful exchange of health data. Understanding the needs of individuals as consumers and developing the appropriate interfaces with the individual and patient communities will allow the public to participate in their care and contribute to a research environment that improves both individual and population health. These interfaces could include applications such as personal health management programs, clinical advisory systems, treatment outcomes databases, clinical
trials matching services, caregiver management resources, and molecular profiling tools. Provider interfaces will allow medical information exchange among the various members of the patient’s clinical team and improve coordinated care. Individual interfaces to personal health records will would provide resources for individual health management, and could provide individuals with the control to donate and distribute their medical information as they see fit.
Individuals as Consumers of Health Information
The goal of patient-centered health care is to allow patients to play an active role in their healthcare decision making by working with healthcare providers to identify tools and knowledge appropriate for their health. Supporting the achievement of this goal will be an integrated health informatics infrastructure that allows appropriate information exchange among researchers, clinicians, and patients regarding treatment options, clinical outcomes, research engagement, and continuing care services.
Therefore, in a learning health system, individuals will be able to navigate through vast amounts of information to find that which is relevant to their needs. For example, a testicular cancer diagnosis touches a broad range of issues including finding oncologists in the area who have treated testicular cancer, treatment options, fertility issues, and counseling information to help address anxiety and emotional issues. In parallel, family members and loved ones who go through the cancer experience with the patient may also need information on caring for someone undergoing chemotherapy, emotional coping, appointment scheduling, and managing finances.
As consumers of health care, individuals enter the healthcare ecosystem searching for specific information that is relevant to their particular situation. In many cases the individual entering the healthcare system is not the patient, but still is searching for information related to care, understanding the disease, or identifying resources to help with practical matters. The Lance Armstrong Foundation supports a phone and online navigation program called LIVESTRONG SurvivorCare1 which provides free, confidential, one-on-one support, in English and Spanish, for anyone affected by cancer. LIVESTRONG SurvivorCare provides resources and information on a range of issues including cancer diagnosis and treatment, clinical trials, counseling, financial concerns, insurance and employment concerns, and fertility preservation. Of those individuals contacting SurvivorCare in 2009, approximately half of the individuals were not the patient diagnosed
___________________
1 See http://www.livestrong.org/Get-Help/Get-One-On-One-Support (accessed August 8, 2010).
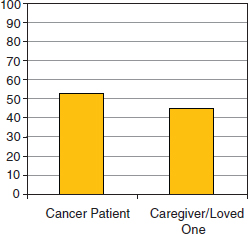
FIGURE 2-1 The Lance Armstrong Foundation’s LIVESTRONG SurvivorCare program offers a navigation resource for anyone affected by cancer. Nearly half of the individuals contacting LIVESTRONG SurvivorCare identify themselves as a caregiver or loved one of someone who has cancer.
with cancer (Figure 2-1). Thus, while individuals may be reaching out for information related to a particular disease, the personal context of their search varies.
A learning health system should account for this context, driving semantic content and resources useful to the individual. By linking patients’ health information with an integrated electronic health information exchange, a knowledge environment can be built to connect clinical care, research, policy, and coverage that supports the best application of medical technologies for an individual patient’s needs.
Individuals as Information Donors
The healthcare ecosystem is composed of a host of interconnected players: patients, doctors, regulatory agencies, insurance companies, and drug developers. In a learning health system, citizens will be equal contributors to building a learning environment, sharing their health data through HIT. In its current state, most information exchange tends to be one-directional, utilized for activities such as recordkeeping, physician reimbursement, and prescription orders. However, this model is shifting toward a bidirectional exchange as individuals adopt tools to help them participate in health management and personal health care.
There is growing evidence on the ability of electronic patient-reported outcomes (e.g., pain, sexual dysfunction, or psychological distress) to in-
TABLE 2-1 Results from the 2010 LIVESTRONG Electronic Health Information Survey
| Agree (%) | Disagree (%) | No Opinion (%) | |
| EHRs should provide patients a way to share their medical information with scientists doing research—as long as the information cannot be linked back to them personally | 86 | 10 | 4 |
| EHRs should allow patients to enter information about their physical health for healthcare providers to review (e.g., pain, fatigue) | 91 | 6 | 3 |
| EHRs should allow patients to enter information about their emotional or mental health needs and concerns for healthcare providers to review (e.g., sadness, worry). | 86 | 10 | 4 |
form clinicians on symptom management and direct medical interventions to improve patient quality of life (Abernethy et al., 2010a). These data are also valuable to researchers, as they provide information regarding the efficacy and/or toxicity of treatments from the perspective of the patient, particularly with respect to quality of life (FDA, 2009; Willke et al., 2004). Individuals can provide a wealth of information by linking clinically annotated data held in an electronic health record (EHR) to personal information such as pain, fatigue, or depression. This health information can be used to populate knowledge environments for analysis in health delivery services, comparative effectiveness research, and population health.
A LIVESTRONG survey2 conducted in the spring of 2010 on electronic health information exchange discovered overwhelming support among the respondents for using electronic exchange to supply personal health information to providers as well as share clinically annotated information from their health records with researchers (Table 2-1). This suggests that individuals want to participate in the research environment, but they want to be in control of when and how they may participate. Additionally, the survey demonstrates that individuals recognize that electronic health exchange
___________________
2 The LIVESTRONG Electronic Health Information Survey was conducted at the Lance Armstrong Foundation by Ruth Rechis, Ph.D., and Stephanie Nutt. Data not published. Survey publicly released April 7, 2010. Survey available at http://www.surveymonkey.com/s/healthinformationsurvey (accessed August 27, 2010).
is an appropriate tool to communicate with their providers on matters of personal health with 86% agreeing that EHRs can help share information about emotional/mental health needs and 91% agreeing EHRs can help share information about physical health needs.
Personalized Medicine and Personalized Care
Advances in biomedical research are revolutionizing our understanding of the molecular underpinnings of diseases as well as the ability to store, share, and compare large volumes of data in real time with integrated informatics platforms. In coming years, the role of the patient in research will expand, becoming a critical component in transforming the research environment. It is the hope that by 2014 the majority of Americans’ health care will be supported through EHRs. In this same time frame, genetic technologies should have advanced to allow individual genome sequencing as a standard clinical analysis. The combination of these approaches will change our approach to diagnosing and treating complex diseases like cancer, drive molecularly informed comparative effectiveness research, aid in developing targeted treatments and personalized medicine, and improve care through federated health information exchanges.
The convergence of electronic personal health information, clinically annotated EHRs, and molecular medicine in an interconnected framework will help to realize the promise of both personalized medicine and personalized care (Abernethy et al., 2010b; Nadler and Downing, 2010). Patients, caregivers, doctors, and researchers will all have a participating role in a system that connects the laboratory bench, the clinical bedside, and the patient’s home. In terms of treatment, as molecular understanding of disease improves, doctors will be able to make informed decisions about targeted drugs and predict patient response, enabling personalized treatment strategies. Similarly, patients will be able to provide valuable information to clinical staff regarding personal health and quality of life, and caregivers will have ready access to information and resources to improve care management.
Expansion and integration of health information exchange efforts can make it possible to aggregate millions of medical encounters in searchable data environments. This will allow for research hypothesis generation and enable researchers and clinicians to model the impact of care interventions. This will provide more detailed profiles to patients and help improve decision making. Additionally, this environment will support information for healthcare policy issues such as electronic information flow, drug/diagnostic approval for patient subpopulations, and reimbursement for targeted therapeutics. This new system relies on a new relationship among patients, doctors, and researchers whereby individuals and patients are all substantive
consumers of HIT. However, in order to succeed, the system must ensure privacy, security, and individual control of personal health information for the patient, while allowing the patient to be both a donor and a recipient of information.
BUILDING A LEARNING HEALTH SYSTEM CLINICIANS WILL USE
James Walker, M.D.
Geisinger Health System
A learning health system will provide all of the healthcare team—patients, caregivers, and all different care providers—with up-to-date, care-process-integrated decision support that is based on the validated benefits and risks of potential interventions. This decision support will be developed through a learning system composed of multiple feedforward and feedback loops, connecting the relevant members of the healthcare team. When we execute this it will lead to marked improvement in population health; at least 100% improvement in delivery of patient-approved, evidence-based care; and at least a 30% reduction in the cost of evidence-based health care delivered (I am not promising decreased overall healthcare costs).
What is a learning system? My definition is a system of goal-directed, feedforward and feedback loops that creates usable and useful—which is to say actionable—information. All of the best data suggest that technology adoption is a function of usability and usefulness. If technology helps users achieve a goal they value and is usable, it will fly off the shelf. If it doesn’t meet those two criteria, it is like most of our health information technology (HIT), and will sit on the shelf. An effective learning health system will need to be useful and usable to all healthcare team members: patients, caregivers, clinicians, public health workers, researchers, and policy makers.
In developing a learning health system, it will be important to consider the sociotechnical context. To systems engineers and increasingly to healthcare designers, it is obvious that any technology intervention is a sociotechnical phenomenon. While technology implementation and optimization are critical (and remarkably difficult), getting the social aspects of a system right is even more important (and more difficult). These social aspects include policies, mutually agreed roles, trust, standardization, resource allocation, mores, and conflict resolution. On the technical side, our existing infrastructure is adequate to support at least an order of magnitude more more shared, actionable learning than we currently achieve. For example, a relatively high-performance electronic health record (EHR) is available to serve well over 80 million Americans. On the social side, however, we miss more opportunities for cooperation than we act on. This lack of action is
one significant reason that it has been difficult to demonstrate benefits of the technical infrastructure.
Building a Learning Health System
The first step in building a learning health system will be to identify the systems learning needs. In terms of clinical decision support, this could be questions like what is the best second-line therapy for type-2 diabetes (rosiglitazone or pioglitazone) or what is the cancer risk associated with antinogensin receptor blockers (ARBs). Other questions include: How are we going to use genomics to improve patient care? Do we need to send every doctor back to medical school? If faced with a public health emergency, can we give clinicians the questions to ask and clinical predictions that will help them to stratify patients for appropriate care? Can we build it into their EHRs? How rapidly?
After identifying the question, the next requirement for a learning system is to identify the information needed to answer the question and the best (most accurate, most efficient, most feasible) way to collect that information. In the case of questions impacting population health, agencies such as the Centers for Disease Control and Prevention and the Food and Drug Administration (FDA) are the logical actors to define the questions and commission user-centered development of the electronic tools that will make data collection efficient enough to be used in everyday care. The EHR infrastructure for collecting and reporting these data from tens of millions of Americans and their clinicians in near-real time is in everyday use today. So for questions like we’re discussing, public health workers will find that if they design their questions to be asked and answered in HIT that clinicians and patients and their caregivers already use—and provide standard-of-care recommendations through that same HIT—they will be able to learn about emerging issues and guide care in days rather than months or years.
One of the most important ways for public health to reward information collection and submission is to feed back relevant information (e.g., trends in ARB adverse effects, patient outcomes on Avandia and other diabetes drugs and drug combinations) to clinicians and the public rapidly. Regarding new drugs for which safety information may emerge over the first years of use, FDA has the potential to make its guidance to care delivery organizations more usable by classifying drugs into one of four groups: (1) drugs that have been proven safe and effective; (2) drugs whose safety is under review and for which an indication for use should be documented and any of the FDA’s standard list of potential adverse effects reported; (3) drugs like Avandia (rosiglitazone) for which significant adverse effects potentially in excess of benefits have been documented (documentation of the indication for use, patient’s formal consent to treatment, and any
adverse effects would be required to be reported; and (4) drugs that have been removed from the market.
In the end, what clinicians get is a set of tools—designed by the appropriate public health agency, developed by the HIT vendor, and implemented by their local HIT team—that would enable them to provide information on the benefits and adverse effects of different interventions and therapies without being distracted from their usual (and critically important) work.
Lessons from Geisinger
What have we at Geisinger learned so far? First, sociotechnical infrastructure development requires highly skilled care-process design teams and technical IT teams. Second, even when those teams work in an organization committed to change, it has taken us over 10 years to make organization-wide changes in 30–40% of our core clinical processes. It may be possible to accelerate this process, but the particularly isolated character of the delivery organizations that need to be integrated going forward make the optimal methods for HIT-supported process redesign a critical topic for research and development as well as careful monitoring. That said, once the infrastructure is in place, the rate at which an organization can make change becomes genuinely breathtaking. Geisinger can now run 5–10 major HIT-supported quality improvement initiatives simultaneously without overtaxing the organization—largely because the infrastructure dramatically decreases the administrative costs of process redesign and management. Finally, existing HIT systems need fundamental redesign to integrate feed-forward and feedback information loops into usable care processes. This is unsurprising, considering how preliminary our understanding of care processes and their information needs still is, but adds significant costs to process redesign and management. For example, Geisinger employs 176 people solely to support the EHR and networked personal health record.
Conclusions
First, we have enough HIT infrastructure in place now to create a much more effective learning health system. Second, our ability to agree among public health professionals, clinicians, HIT developers, patients, and others on the questions that are worth answering and the required information needs substantial development. Third, to optimize the learning system, HIT products and services need fundamental redesign based on actual and potential future needs. Finally, we must consider what will motivate delivery organizations to participate in such a learning system? Providing substantial reimbursement for participation is unlikely to be feasible, and sanctions for failure to participate are unlikely to be feasible or enforceable.
Alternatively, if participation is made optimally easy and enables delivery organizations to meet explicit societal standards of care reliably and cost-effectively, it will likely provide adequate incentive for participation.
IMPROVING QUALITY AND SAFETY
Janet M. Corrigan, Ph.D., M.B.A.
National Quality Forum
It has been 10 years since the Institute of Medicine issued its landmark reports, To Err Is Human and Crossing the Quality Chasm, focusing national attention on the need to improve health care quality and safety (IOM, 2000, 2001). Since that time, there have been some very important accomplishments, but overall, progress has been slow. Per capita expenditures in the United States far exceed those of all other industrialized countries, while quality and safety remain uneven (Fisher et al., 2003; IOM, 2010; Murray and Frenk, 2010).
Although there have been many very successful, localized quality improvement initiatives demonstrating that it is possible to close the quality gap, we have yet to take these innovations to scale. In our current health system, quality measurement and improvement are labor- and time-intensive activities. Measuring quality often involves abstracting information from paper charts or relying on administrative data sources that lack clinical richness. Clinicians may receive performance reports based on data that are a year old or more, and performance results (e.g., mammography rate) may not be accompanied by the necessary information to improve care (e.g., detailed listing of patients who should have received a mammogram but did not).
Our measurement and improvement efforts have also been hampered by the fragmented and siloed nature of the health system. Most quality improvement activities have been focused on aspects of the care process for which some data are captured, namely hospital care and ambulatory visits. Yet many serious safety and quality concerns arise from care transitions (e.g., discharge from the hospital to the community or referral from a primary care provider to a specialist).
In spite of the fact that health care consumes over 16% of U.S. gross domestic product, there is currently no system in place to measure patient outcomes (IOM, 2010). Currently, most available data are recorded by clinicians during health encounters. The health system lacks mechanisms to capture patient-derived data on health functioning, symptoms (e.g., fatigue, pain), health behaviors (e.g., exercise, diet, smoking), and adherence to treatment plans (e.g., medications).
Achieving higher levels of safety, quality, and efficiency requires investment in an electronic data platform capable of capturing the necessary
longitudinal data from clinicians and patients, and providing real-time feedback and clinical decision support to clinicians and patients. With the passage of the Patient Protection and Affordable Care Act (ACA) of 2010, the stakes are now higher than ever before. ACA seeks to encourage and reward higher levels of performance and penalize those who fail to measure up through three key provisions:
- Transparency: expansion of public reporting websites pertaining to virtually all types of providers and clinicians to include safety, quality, and cost information.
- Payment alignment: creation of payment programs tied to performance, including paying more for higher value care and nonpayment for healthcare-acquired conditions.
- Clinically-integrated delivery systems: more favorable payment programs for health care homes and accountable care organizations capable of providing patient-centered, team-based care.
A digital infrastructure that can support robust performance measurement and improvement systems is a necessary prerequisite to succeed in this new environment.
Framework for Performance Measurement
A two-dimensional framework is guiding the development of performance measures and performance measurement requirements for public reporting and value-based purchasing:
- Crosscutting areas: The National Priorities Partnership convened by NQF has identified six crosscutting areas that impact most if not all persons/patients, including population health, safety, care coordination, patient/family engagement, palliative care, and overuse (National Priorities Partnership, 2008). Within each priority area, there are specific aspects of care that will be the focus of intense monitoring and improvement because these areas currently exhibit large quality, safety, and efficiency gaps (Box 2-1).
- Clinical conditions: A limited number of clinical conditions account for a sizable share of healthcare services and health burden (NQF, 2010). By focusing attention on these conditions, it should be possible to positively impact the lives of many patients while also removing waste from the system.
Figure 2-2 provides an example of this two-dimensional performance measurement framework applied to patients with acute myocardial infarc-
BOX 2-1
National Priorities Par tnership
Key Cross-Cutting Areas and Goals
Population health
- Key preventive services
- Healthy lifestyle behaviors
Safety
- Hospital-level mortality rates
- Serious adverse events
- Healthcare-acquired Infections
Care coordination
- Medication reconciliation
- Preventable hospital readmissions
- Preventable emergency department visits
Patient/family engagement
- Informed decision making
- Patient experience of care
- Patient self-management
Palliative care
- Relief of physical symptoms
- Help with psychological, social, and spiritual needs
- Communication on treatment options, prognosis
- Access to palliative care services
Overuse
- Nine major areas
SOURCE: National Priorities Partnership (2008).
tion (AMI). To assess whether the health system is taking appropriate steps to prevent AMIs, information must be captured on the health status and risk behaviors of the entire population and the services provided to mitigate risk (e.g., programs to lower cholesterol levels through diet, exercise, and medication). Once an AMI occurs, information must be captured on the quality and safety of the emergency response system (including community-based and hospital services) and on how well the patient is managed throughout their hospital episode. Upon hospital discharge, the patient will require follow-up care emphasizing secondary and tertiary prevention and to ascertain whether expected outcomes have been achieved. The six cross-cutting areas also are relevant to AMI patients. For example, care coordination (e.g., transfer of treatment plan from hospital to rehabilitation provider with acknowledgement of receipt).
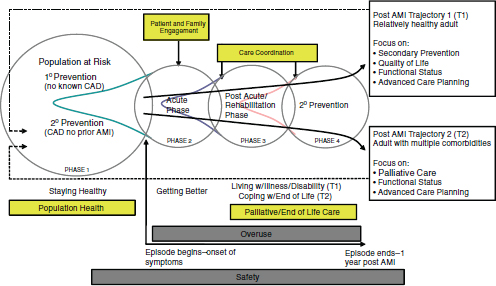
FIGURE 2-2 Two-dimensional measurement framework applied to acute myocardial infarction.
Implications for the Digital Infrastructure
A patient-centered approach to designing the digital infrastructure will be needed to support quality measurement and improvement. The digital infrastructure must be capable of capturing the relevant data from clinicians and patients across the entire longitudinal, “patient-focused episode” to assess both cross-cutting and condition-specific aspects of quality. To achieve the greatest gains in improvement, there should be immediate feedback of performance results accompanied by clinical decision support to both clinicians and patients.
In general, the types of information that must be captured for a patient-focused episode fall into the following domains:
- Patient-level outcomes (better health)
o morbidity and mortality
o functional status
o health-related quality of life
o patient experience of care - Processes of care (better care)
o technical quality of care
o care coordination and transitions
o alignment with patients’ preferences
- Cost and resource use (affordable care)
o total cost of care across the episode
o indirect costs
For some of these data—functional status, alignment with patient preferences—patients and family caregivers are the most reliable sources, so the digital infrastructure must provide for personal health records or other vehicles for incorporating a “patient voice.”
Over time, it will also be important for the digital infrastructure to move beyond the traditional boundaries of the personal healthcare system. Achieving the best outcomes for patients and populations requires the ability to capture information from, and enable communication with, all residents of a community regardless of whether they use healthcare services. It will also be important to capture “context information,” such as race, ethnicity, language, socioeconomic status, and employment, all of which influence adherence to treatment plans and patient outcomes, and are needed to inform policy.
CLINICAL RESEARCH IN THE INFORMATION AGE
Christopher G. Chute, Ph.D., Dr.P.H.
Mayo Clinic College of Medicine
The London Bills of Mortality were initiated by Henry the VIII of England during an onset of the Black Death in an effort to tabulate who was dying of what, mostly so that the nobility could know which areas were plague-infested and avoid them. An unexpected consequence was the publication over a century later of John of Graunt’s seminal work, “Natural and Political Observations…Upon the London Bills of Mortality,” first published in 1662. Through his systematic analyses of these data, public health was transformed into a quantitative science, replete with the introduction of endemic and epidemic patterns, small area analyses of mortality, and the foundation of epidemiological and biostatistical principles (Glass et al., 1963). As with all such seminal work, it was largely ignored for 200 years, but it was probably the first work to show that the systematic collection of mortality statistics could inform the world about much more than where not to travel to avoid the plague.
The Information-Intensive Nature of Modern Health Care
The distinguishing characteristic of modern medicine is the information-intensive nature of its practice. Modern health care comprises two things: managing information and procedural interventions. Virtually ev-
erything done clinically—from gathering a history and a physical, to working through differential diagnoses, managing preventive measures, and most importantly, accessing the wealth of extant medical knowledge—involves information. Humans have been adept at processing clinical information for generations. However, in the midst of the continuously escalating rate of biomedical discovery and knowledge generation, even the most adept among us are overwhelmed. Requirements for computational assistance in the clinical process are apparent.
Equivalently, the formalized improvement of care inevitably involves the definition of numerators and denominators corresponding to clinical phenotypes. Patients are classified into these strata, a process that increasingly cannot rely on human abstraction and judgment. Comparisons among strata are computed, often involving sophisticated numerical or machine learning methods, and inferences made with respect to quality of care, technology assessment, best evidence discovery, or comparative effectiveness. Thus, most of our science associated with care improvement is inexorably linked to information processes.
If we accept that biomedical advancement and clinical practice have become unified as an information-intensive domain, then the technical requirements for information and knowledge management are now high-profile and high-priority issues.
The Transformation of Information Processing
In parallel with the explosion of biomedical knowledge has evolved the transformative change in our capacity to manage and interpret information. Moore’s Law (for Intel founder Gordon Moore), postulated in 1965, asserts that integrated circuit density would double every 2 years. The well-known corollary is that computation power would double every 18 months (the acceleration being due to processor design improvements). Both laws have proven uncannily accurate. Computing capacity has increased on the order of 1012-fold over the past 60 years. The supercomputing power that nations would have sacrificed the lives of spies to secure as recently as 20 years ago are now under Christmas trees as game platforms for children.
However, raw computation power is not the only dimension over which we can meaningfully measure information processing capability. Network performance has experienced dramatic increases in bandwidth from 110-baud teletypes to 100-Gb backbones—an increase of 109 over 50 years. Furthermore, the raw number of high-bandwidth connections around the globe has successfully saturated four generations of IP protocols—another 109 increase. Correspondingly, local memory stores on machines have grown from handfuls of vacuum tubes to 100-Gb RAM configurations not uncommon on intermediate server platforms today—an approximate 1011-fold
increase in capacity. Not to be forgotten are data storage capabilities, exploding from paper-tape holes (where one could reassuringly see those bits) to petabyte drive assemblies—at least a 1015-fold increase.
All these measures of computational capability are gross underestimates in the face of emerging cloud computing and data storage resources, which are arbitrarily scalable to sizes that make these comparisons ultimately irrelevant. Nevertheless, if we add the exponents above—which is really multiplying, since we are in exponential space—humanity has achieved a 1056-fold increase in our capacity to manage and manipulate information during my lifetime. This is a conservative estimate, and a vast, genuinely astronomical number by most measures. Despite its frail arithmetical basis, the conclusion persists that we have experienced a profound increase in our ability to manage information. This must have a profound impact on domains that are information intensive. We are only in the opening chapters of a massive social and cultural transformation of biomedicine. It is without precedent.
These conclusions are concordant with predictions emerging from the genomics community and consistent with our everyday experience with unprecedented access to most anything we could want to know on the Internet. Our Google-aided society can research topics with a speed and depth unimaginable a generation ago. So, too, will our capabilities in biomedical discovery and clinical practice evolve.
The Emergence of “Big Science”
The lessons of astronomy and physics are informative as we consider the future of biology and medicine. These disciplines have evolved into a “big science” paradigm, where the work of collective groups and the amassed knowledge and data resources of the field dominate over the contributions of independent investigators. Gone are the days when a Galileo could gaze into the skies from his porch and make seminal discoveries in astronomy. Similarly, the tabletop experiments of Rutherford, while profound, cannot be matched in the present era in terms of scientific impact or advancement. Both fields today depend on large teams of interdisciplinary scientists, who draw from and contribute to a vast commons of shared data and imputed knowledge.
The duality of biology and medicine having become information-intensive domains, coupled with our vast capacities to manage and manipulate information, make it inevitable that a similar “commons” of biomedical information will form a hub from and to which investigators and practitioners will draw and contribute. A foreshadowing of this reality is already evident in the genomic community, with the myriad of publicly accessible databases that surround the original Genbank suite of resources.
If clinical medicine is to truly become “big science,” we must recognize that this implies that we can learn from the historical experience of patients, study their outcomes, and learn what interventions help or hurt for particular characterizations of patients, diseases, comorbidity, risk factors, genomic traits, and social or patient preferences. While randomized clinical trials remain the gold standard of biomedical evidence, we can and will learn more from empirical studies of patient outcomes. Presently, the meticulous surveys, chart abstractions, quality studies, or comparative effectiveness efforts have yet to coalesce into anything like a scientific commons for large-scale analyses and understanding. Clinical evidence, together with the healthcare delivery infrastructure, remain trapped in a cottage industry–level effort, fraught with noncomparable information and profound barriers to data sharing and access. Medicine, from a knowledge management perspective, remains in a pre-Grauntian state. We are unable to tabulate our fate using 16th century data spreadsheets or other quantitative means for lack of consistent and comparable information about what we do clinically or what happens to patients.
Comparability and Consistency in Healthcare Information
What then would correspond to a present-day London Bills that could sustain the analyses of the intellectual descendents of Graunt and improve our understanding, practice, and outcomes in clinical care? A widely shared vision is the notion of a repository of patient experience, where electronic records were made available under supervised and consented conditions to epidemiologists, health services researchers, biostatisticians, and others to scalably discover best evidence for care, and ultimately a mechanism that would predict best therapies or preventions for specific categories of people. While presently many obstacles—including privacy, confidentiality, and intellectual property concerns—make this vision impractical, one critical path issue remains the reality. Most health information is neither comparable nor consistent among providers, record systems, or researchers. We lack standards for representing patient findings, events, or interventions in a comparable or consistent way. This obviates any scalable analyses without expensive and typically humanly intensive abstraction and harmonization of the data.
The absence of standards is not due to technical obstacles or an absence of specification. Among the cottages of healthcare delivery have emerged what may be characterized as wanton idiosyncrasies. There is no good technical reason why every hospital and clinic feels compelled to create de novo codes and identifiers for clinical laboratory measures; the foundation of the publicly accessible and free-for-use LOINC codes for laboratories could solve this one problem overnight. Furthermore, most electronic medical
record systems developers have been slow to contribute to or adopt clinical information standards; that those who have invested the least in health information technology (HIT) standards development appear to be the most successful in the marketplace suggests there are misaligned incentives operating in the healthcare marketplace—not a new observation, to be sure.
William Farr, a great 19th century English leader of public health, asserted in 1839 that “nomenclature is of as much importance in [medicine], as weights and measures in the physical sciences, and should be settled without delay” (Langmuir, 1976). The metaphor is apt with our big science analogies. How could we conduct astronomy or physics without notions of a meter, second, or gram? We seem as a society not to have heeded Farr’s admonition about nomenclature, since even something as relatively uncontroversial as a serum sodium measure has virtually no adoption of standard nomenclature or code system.
The U.S. Standards Experiments
If we accept that health care is information intensive, that computational capacity has transformed our ability to manage and understand information, that comparable and consistent representation of clinical data using HIT standards is on our critical path to improved healthcare efficacy and efficiency, why have we not fully developed and adopted HIT standards?
There has been no lack of efforts to establish consensus forums in the United States and globally for the specification of HIT standards. The overused quip that “the nice thing about standards is that there are so many to choose from” might apply equivalently to HIT standards bodies and consensus forums. Beginning with the Health Information Standards Planning Panel in the early 1990s, and moving through the American National Standards Institute’s Healthcare Informatics Standards Board, the Health Information Portability and Accountability Act, the Healthcare Information Technology Standards Panel, and the Office of the National Coordinator for HIT (ONC) HIT Standards Committee, there have been significant resources expended on this problem. Few have lasted more than a few years, and most have had minimal impact on clinical practice or biomedical discovery.
“Meaningful use” may be a transformative effort, where the likelihood of broadly based adoption—premised on the suite of incentives and penalties under the Health Information Technology for Economic and Clinical Health Act—may be substantial. If so, then for the first time the United States will have a basis for comparable and consistent representation of clinical data beyond billing codes. The implications of this for future science, enabling the establishment of federated repositories of patient data that can sustain inference and discovery, are profound.
INTEGRATING THE PUBLIC HEALTH PERSPECTIVE
Martin LaVenture, Ph.D., M.P.H., Sripriya Rajamani, Ph.D., M.P.H., and Jennifer Fritz, M.P.H.
Office of Health Information Technology,
Minnesota Department of Health
Achieving the vision for a Digital Infrastructure for the Learning Health System will make profound improvements in the health of individuals, communities, and the entire population. Successfully achieving this vision requires improving the capability and capacity of population health services provided by governmental public health organizations at the local, state, and federal levels; close integration with clinical stakeholders; and fully engaging the general public.
Background
The Health Information Technology for Economic and Clinical Health (HITECH) Act and the Patient Protection and Affordable Care Act (ACA) have provided the nation with an unprecedented opportunity to accelerate the pace for improving healthcare quality, increasing patient safety, reducing healthcare costs, and enabling individuals and communities to make the best possible health decisions. Coordination and training were identified as key issues for the national public health informatics agenda at a meeting of stakeholders almost a decade ago (Yasnoff et al., 2001). These issues are currently being addressed at the national level through initiatives that focus on adoption and use of electronic health records (EHRs) through incentives, technical assistance, training, and support for health information technology (HIT) innovation (Blumenthal, 2010). The extensive policy, governance, and technical foundation established locally to date needs to be leveraged and integrated closely with national efforts facilitated through the Office of the National Coordinator for HIT.
A digital infrastructure for the learning health system can offer immense opportunities for population health improvement in public health surveillance and response, population-based research and policy, coordination and quality improvement, and health education and communication. Challenges to achieving this vision include a lack of a sound electronic public health infrastructure, the need to advance workforce skills, polices that force categorical use of funds and short budget cycles, and uneven understanding among programmatic leaders about public health benefits of HIT. A shared vision and commitment to a clear path are critical, with emphasis on addressing the needs identified above.
Population Health Opportunities of Digital Infrastructure
The meaning of population health varies, but working definitions used by the Minnesota e-Health Initiative are as follows (Minnesota e-Health Initiative, 2010; Westera et al., 2010):
Population health: a conceptual approach to measure the aggregate health of a community or jurisdictional region with a collective goal of improving those measurements and reducing health inequities among population groups. Stepping beyond the individual-level focus of mainstream medicine, population health acknowledges and addresses a broad range of social determinant factors that impact health. Emphasizing environment, social structure, and resource distribution, population health is less focused on the relatively minor impact that medicine and healthcare have on improving health overall. (Koo et al., 2001)
Governmental public health: a core infrastructural entity that is legislatively authorized to protect the public. Public health organizations provide the backbone to the infrastructure for population health improvements. It depends on other sectors (e.g., health care system, academia, business community) to improve the overall health of a community based on population health analysis. (Minnesota e-Health Initiative, 2008)
The digital infrastructure for the learning health system can offer immense opportunities for population health improvement and, more importantly, can serve as a conduit for bringing the domains of population health together. Table 2-2 identifies five areas of population health services and
TABLE 2-2 Types of Population Health Activities and Opportunities for Provider Engagement
| Population Health Area | Opportunity for Provider Engagement |
| Surveillance and response | Identify sentinel events, emerging illness, and injury trends. Access to cross-sectional and longitudinal data to identify patterns, trends, and support response actions |
| Health status/disease measurement | Leverage resources available to optimize health status and outcome measurement |
| Health education/communication | Use new medical information for targeted knowledge/recommendations |
| Population-based health care | Clinic-based profiles of patients informing decision-support programs to assist members in developing/improving self-care skills |
| Population-based research | Applied research to improve care for individuals/the community |
example opportunities for provider engagement in each domain. Achieving a healthier population requires that federal, state, and local organizations be fully engaged. Given the challenges described above, the capacity and capability of public health information systems need to be modernized. Box 2-2 identifies some of the benefits of an integrated, modernized electronic infrastructure that enables secure, authorized bidirectional communications with governmental public health agencies and other organizations providing population health services. All of these activities seek to improve the health of individuals and communities.
Current State of Play
Achieving the population health improvements possible in a learning health system requires significant improvement in the digital infrastructure. This cannot be achieved on a national scale by simply adding some population health fields onto an EHR. We need to achieve a much broader understanding of how we are going to collect, analyze, distribute, and use information to better provide care coordination and other activities at the community level.
Table 2-3 identifies three levels of public health infrastructure in the United States and their general areas of responsibility. Infrastructure varies significantly across these agencies. The systems they employ vary in functional capability as well as capacity. Improvements in individual and organization skills in informatics and information technology are needed. Most agencies are currently experiencing significant budget challenges. Additionally, system capability and capacity as well as workforce informatics skills needs remain barriers to achieving a broader vision.
Figure 2-3 presents an example from Minnesota where plans for HIT incorporate a strategic model that is designed to integrate across the continuum of care, including public health. As a result, the integration of public health into this plan is a core element for achieving a broader population health vision.
Challenges
Many challenges face public health organizations as they seek to modernize and maintain an infrastructure that can support a learning environment. In general, key needs fall into several categories: modernize technical infrastructure, advance the skills of the workforce, commit to development of common business processes across jurisdictions, modify policies that force categorical use of funds and short budget cycles, address the uneven understanding among programmatic leaders about how HIT benefits public health needs; and improve understanding of public health’s role in care coordination.
BOX 2-2
Bidirectional Benefits of Public Health Par ticipation in a
Learning Health System
Public health contributions
- Knowledge and skills on population health measurement
- Intervention expertise to health reform/quality improvement efforts
- Leadership on community-focused population health efforts that increase utilization of primary prevention services
- Improvements in care coordination especially for chronic diseases (e.g., diabetes, asthma, hypertension)
- Can provide data based on select characteristics (summary-level data, epidemiological data)
- Can provide “evidenced-based practice” as well as “practice-based evidence”
- Collaborative efforts to implement clinical decision support systems
- Leadership on efforts to measure and monitor the health of the community by applying data analysis competence
- Capability to execute large population health/community-level changes through recommendations, guidelines, and public policies
- Ability to translate impact of interventions to public health problems
- Optimize systems for disease surveillance, analysis, and alerting
- Coordinate efforts to implement clinical decision support systems that better integrate decision support across multiple diseases/conditions to improve disease management
Benefits to public health
- Ability to use outcomes data from electronic health records and other HIT to supplement existing surveillance methodologies and information
- Ability to optimize systems for disease surveillance, analysis, and alerting based on lessons learned
- Gain new knowledge to improve care coordination and outcomes, especially for chronic diseases
- Quicker translation of insights gained from clinical environment to potential interventions to possible public health recommendations
- Coordination of services and research with academic and learning community
- Creation of a framework where the trend of new and existing acute and chronic conditions are correlated with select population-level metrics (e.g., demographics, socioeconomic status, prevalence of other comorbidities, community characteristics)
SOURCE: Adapted from Improving Population Health and the Minnesota e-Health Initiative fact sheet. http://www.health.state.mn.us/e-health/phphin/index.html (accessed February 22, 2011).
TABLE 2-3 Governmental Public Health Agencies
| Governmental Entity | Number and Scale of the Agencies |
| Cities and counties | ~3,000 health departments |
| State/territory | 50 state health departments, 6 territories |
| Federal | Centers for Disease Control and Prevention as the lead agency Centers for Medicare & Medicaid Services Health Resources and Services Administration Office of the National Coordinator for Health IT Agency for Healthcare Research and Quality |
FIGURE 2-3 Minnesota example of public health infrastructure relative to other systems.
Recommendations
Establish a shared vision and action plan for population health and a clear path to success. The lack of coordination of efforts in the past has proven to be a barrier and must be addressed in order to realize the op-
portunities before us today. The Minnesota e-Health Initiative3 provides an example of one state’s shared vision and how it was incorporated into the statewide plan for e-health to ensure success. The critical components to ensure success include
- A commitment to the development of common business requirements and processes across jurisdictions, starting with local health departments.
- Improved standards, specifications, and certification criteria for interoperability of public health–focused data on individuals and population aggregate information.
- A commitment to modernizing infrastructure using a coordinated and integrated approach.
- A commitment to close the gap in core and advanced informatics skills of the workforce.
- A transition to policies that encourage integrated approaches to programs supporting the larger vision.
- A cohesive message to advance common understanding of how EHRs/HIT benefit public health.
Adopt specific approaches to data standards, aggregation, and/or infrastructure that will help achieve better population health outcomes
- Improve federal and state leadership and coordination on identification and use of standards for interoperability including technical, semantic, and process interoperability.
- Establish the framework for tools that can present population health data in ways that can profile the health status and disease burdens of communities. This should include the ability to analyze patterns of injury and illness in relationship to health status and risk in the community. What gets measured is better understood and often gets done.
- Utilize existing tools to create an informatics profile for public health agencies and expand and adapt the tool to meet evolving needs (Fritz et al., 2009).
- Implement population health dashboard applications that provide community health profile in near-real time. Establishing a population health dashboard will empower individuals and providers with data they need to support the learning health system.
___________________
3 More information on Minnesota’s Statewide Plan for e-Health can be accessed at www.health.state.mn.us/ehealth (accessed September 30, 2010).
- Adopt the standards for the full set of transactions for meaningful use requirements. For example, expand immunization transactions beyond submission to include request, return of history, and the options for forecasting using decision support.
- Adopt standards for all transactions associated with reportable conditions including alerting function capability.
- Certifications of public health software applications are vital. Pursue “orphan” software classification if needed to obtain vendor participation.
- Build upon national standards and large-scale models for implementation strategies. Avoid duplication of population health–only infrastructures.
Implement intelligent, bidirectional public health messaging for providers and consumers. The potential for effective health communication and key messages to the public to modify beliefs and influence behavior has been recognized by the public health community for many years. In order to drive effective messaging, public health agencies and others responsible for population health improvement should fully engage consumers by presenting health information in effective formats that drive improved outcomes and also extend reach through utilization of emerging venues of communication such as social networks and other new media mechanisms. Consumers must be fully engaged and messages based on trusted information sources should
- Articulate the value public health information can bring to them—in terms of quality, cost, and convenience.
- Explain how patient privacy is protected both by law and through the use of appropriate security measures.
Conclusion
A learning health system provides the opportunity to improve the health of the population in profound ways. Significant improvements are needed to modernize information systems, improve needed functional capability, and achieve better trained workforce. “Informatics savvy” organizations are a vital component to achieve the goal of improved population health.
REFERENCES
Abernethy, A. P., A. Ahmad, S. Y. Zafar, J. L. Wheeler, J. B. Reese, and H. K. Lyerly. 2010a. Electronic patient-reported data capture as a foundation of rapid learning cancer care. Medical Care 48(6 Suppl):S32-S38.
Abernethy, A. P., L. M. Etheredge, P. A. Ganz, P. Wallace, R. R. German, C. Neti, P. B. Bach, and S. B. Murphy. 2010b. Rapid-learning system for cancer care. Journal of Clinical Oncology.
Blumenthal, D. 2010. Launching HITECH. New England Journal of Medicine 362(5):382-385.
FDA (Food and Drug Administration). 2009. Guidance for industry patient-reported outcome measures: Use in medical product development to support labeling claims. http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM193282.pdf (accessed September 10, 2010).
Fisher, E., D. Wennberg, T. Stukel, D. Gottlieb, F. Lucas, and Ã. Pinder. 2003. The implications of regional variations in Medicare spending. Part 1: The content, quality, and accessibility of care. Annals of Internal Medicine 138(4):273-287.
Fritz, J. E., P. Rajamani, and M. LaVenture. 2009. Developing a public health informatics profile: A toolkit for state and local health departments to assess their informatics capacity. http://www.phii.org/resources/doc/MN%20PHIP%20Toolkit%20FINAL.pdf (accessed September 30, 2010).
Glass, D. V., M. E. Ogborn, and I. Sutherland. 1963. John Graunt and his natural and political observations [and discussion]. Proceedings of the Royal Society of London. Series B, Biological Sciences 159(974):2-37.
IOM (Institute of Medicine). 2000. To err is human: Building a safer health system. Washington, DC: National Academy Press.
———. 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
———. 2007. The learning healthcare system: Workshop summary. Washington, DC: The National Academies Press.
———. 2010. Healthcare imperative: Lowering costs and improving outcomes: Workshop series summary. Washington, DC: The National Academies Press.
Koo, D., P. O’Carroll, and M. LaVenture. 2001. Public Health 101 for informaticians. Journal of the American Medical Informatics Association 8(6):585-597.
Langmuir, A. D. 1976. William Farr: Founder of modern concepts of surveillance. International Journal of Epidemiology 5(1):13-18.
Minnesota e-Health Initiative. 2010. Minnesota e-health Initiative: Vision. http://www.health.state.mn.us/e-health/abouthome.html (accessed September 30, 2010).
———. 2008. Population health and health information technology framework: Definitions, conceptual model, and principles. http://www.health.state.mn.us/e-health/phphin/phmodel2008.pdf (accessed September 30, 2010).
Murray, C. J. L., and J. Frenk. 2010. Ranking 37th—measuring the performance of the U.S. health care system. New England Journal of Medicine 362(2):98-99.
Nadler, J. J., and G. J. Downing. 2010. Liberating health data for clinical research applications. Science Translational Medicine 2(18):18cm16.
National Priorities Partnership. 2008. National priorities and goals: Aligning our efforts to transform America’s healthcare. Washington, DC: National Quality Forum.
NQF (National Quality Forum). 2010. Prioritization of high-impact Medicare conditions and measure gaps. http://www.qualityforum.org/projects/prioritization.aspx?section=MeasurePrioritizationAdvisoryCommitteeReport2010-05-24 (accessed February 23, 2011).
Westera, B., M. LaVenture, B. Wills, and S. Rajamani. 2010. Case study 14d. Minnesota e-health Initiative. In Nursing and informatics for the 21st century: An international look at practice, education and EHR trends, edited by C. A. Weaver, C. W. Delaney, P. Weber, and R. L. Carr. Chicago, IL: Healthcare Information and Management Systems Society and American Medical Informatics Association. pp. 312-322.
Willke, R. J., L. B. Burke, and P. Erickson. 2004. Measuring treatment impact: A review of patient-reported outcomes and other efficacy endpoints in approved product labels. Controlled Clinical Trials 25(6):535-552.
Yasnoff, W. A., J. M. Overhage, B. L. Humphreys, M. LaVenture, K. W. Goodman, L. Gatewood, D. A. Ross, J. Reid, W. E. Hammond, D. Dwyer, S. M. Huff, I. Gotham, R. Kukafka, J. W. Loonsk, and M. M. Wagner. 2001. A national agenda for public health informatics. Journal of Public Health Management and Practice 7(6):1-21.




























