4
Engaging Patient and Population Needs
INTRODUCTION
In order to truly improve health and health care, the digital infrastructure will require appreciation, support, enthusiasm, and active involvement from patients, providers, and the population as a whole. Papers included in this chapter highlight strategies for engaging of stakeholders and facilitating higher value care, including putting more relevant information under their control, leveraging health information to better coordinate care of chronically ill patients, and combating health disparities.
Mark McClellan from the Brookings Institution details the essential components of a digital infrastructure that can more closely align quality measurement and improvement in order to achieve high-value health care. He notes that patient-centered measures, repurposing data already being used to coordinate care for performance measurement, and alignment of these processes with other reform efforts—namely, value incentives—will be necessary to improve care and lower costs. Dr. McClellan uses diabetes care coordination to highlight ways in which information could be used to help providers improve care in a timely way, help patients obtain better care, and serve as the basis for driving value-based reforms. He notes that pilots such as accountable care organizations and Office of the National Coordinator for Health Information Technology (ONC)-funded Beacon Communities will be instrumental in helping identify best practices and aligning processes and incentives for systemwide change.
Addressing the issue of engaging individuals in population health monitoring, Kenneth Mandl from Children’s Hospital Boston asserts that harness-
ing the knowledge possessed by populations through longitudinal studies of large, distributed, consenting populations, will be the focus of work in population health over the next decade. Based on his experience developing Indivo—a patient-centered health record that places patients in control of their own health information—coupled with federal incentive initiatives, he predicts a shift in the health information economy from institutional to individual or patient control. This shift will likely change population health research in a way already being seen through forums such as PatientsLikeMe. Finally, Dr. Mandl suggests that a critical outstanding research question is how to achieve sustained engagement of patients in research.
Sophia Chang from the California HealthCare Foundation states that a digital infrastructure provides important opportunities for informing and improving the care of patients with chronic disease. She discusses the potential to actively engage patients in management of their conditions, but notes that, currently, the locus of control lies solely with the healthcare providers and not the patient. Additionally, Dr. Chang points to the lack of common nomenclature, data formats, and protocols for incorporating patient-generated information as barriers to aggregating and translating health data into useful decision support. Pointing to Kaiser Permanente and the Veterans Health Administration as examples of institutions that have successfully used electronic health records (EHRs) for population health management, she notes that smaller institutions or individual physicians might have less opportunity for exposure, and therefore be less aware of their value. In order to maximize the value of EHRs, she asserts, research paradigms must shift to real-time knowledge development and feedback. Finally, Dr. Chang highlights several steps to move toward the goals of recentering the system around the patient, such as providing useful support for chronic disease management, aligning EHR data elements with patient priorities, and developing better paradigms for learning from patient data.
M. Christopher Gibbons of the Johns Hopkins Urban Health Institute discusses opportunities for using a digitally supported learning health system to better comprehend and combat health disparities. Noting that understanding and treating health disparities requires integration of knowledge spanning many sources and disciplines, he points to several demographic trends that make this challenge ever more pressing—rising prevalence of chronic disease, an aging population, and the growing racial and ethnic diversity of the U.S. population. Dr. Gibbons introduces the terms “populomics” and “populovigilance” to describe the integrative, systems-oriented, and informatics-intensive approaches to understanding and monitoring the complex causes and manifestations of diseases and disparities. He suggests that as more and more data from diverse sources are collected and available for analysis, it will be important to adopt these new perspectives in order to enable advances in treatment, public health, and healthcare disparities.
ELECTRONIC HEALTH DATA FOR HIGH-VALUE HEALTH CARE
Mark McClellan, M.D., Ph.D.
The Brookings Institution
Achieving real healthcare reform requires aligning all five elements of the quality enterprise: measurement and data collection, payment reform, benefit reform, better evidence, and quality improvement support. Electronic data exchange is an essential tool to support these strategies. While all five of these elements are important, this paper will focus on the role of the data collection and reporting infrastructure in improving quality and lowering costs.
The essential features of the data collection infrastructure are implementing technically sound strategies to obtain patient-centered measures, utilizing electronic data that are already being used to coordinate care for performance measurement, and ensuring that this process is consistent with the implementation of other related reform efforts. The overarching goal for an improved data collection infrastructure is to be able to measure performance with the same data being collected during the routine delivery of care.
Improving the Data Collection Infrastructure
The focus of the improvements to the data collection infrastructure is on identifying scalable methods to coordinate the flow of information from existing sources. This approach is aligned with ongoing health information technology (HIT) reform efforts in the areas of meaningful use, value-based purchasing, pay-for-reporting, and others. It is also important to incorporate efforts from a wide array of stakeholders to facilitate coordination across the broad spectrum of initiatives in the public and private sectors.
Work in the area of improved data collection should build on existing progress and account for alternative sources in exchanging data and generating functionally equivalent performance information. Effort should also be made to identify nationally consistent methods and approaches through pilot projects prior to nationwide implementation. Finally, the resulting systems should effectively cover the vast majority of providers and patients in a timely fashion.
Healthcare reform is a complex undertaking, but at its core, reform is about how we can provide better care and value for patients. Patient care takes place within a complicated ecosystem composed of many stakeholders, from physicians and hospitals to labs and pharmacies. A tremendous amount of valuable health information is generated at each patient encounter along this continuum. With funding from the American Reinvest-
ment and Recovery Act of 2009 to encourage the adoption and meaningful use of HIT, we can expect that more of this health information will be collected electronically and that it will be more easily exchanged to better coordinate care and to provide valuable feedback to healthcare providers. For example, registry functions, decision support tools, and medication alerts all have the potential to help healthcare providers deliver better, safer, high-value health care (Figure 4-1).
The contribution of claims data from public and private payers further enhances the value of the feedback and enables consistent performance measurement. Payment and service delivery are inextricably linked. The fragmented care that we currently have is, in large part, because of the fragmented way in which we pay for care. What this means for testing healthcare innovations is that we can expect changes in the payment to drive changes in service delivery. Those changes can be compared and evaluated to determine which payment models produce comparatively better quality at lower cost.
On the demand side of health care, performance information can also be provided to patients to enable them to select high-value healthcare providers, reinforcing broader health reform efforts that attempt to foster greater accountability for care. Patients can also make valuable contributions to healthcare information. Data they provide on their experience of care and their demographic backgrounds will help us improve patient satisfaction and track and reduce healthcare disparities. Additionally, patient
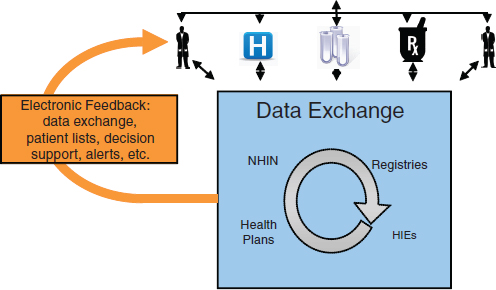
FIGURE 4-1 Basic data exchange infrastructure in a learning health system.
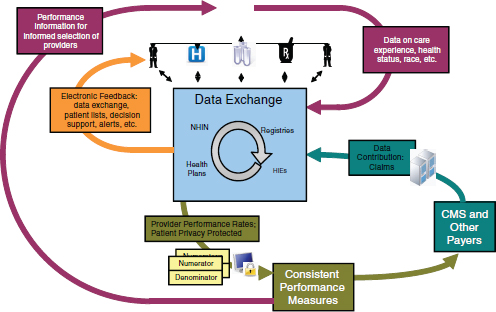
FIGURE 4-2 Electronic health data as the center of a model for better care at lower cost.
data on the quality of their healthcare experience can be used by other consumers in making future choices about healthcare providers.
Incorporating all of these elements—coordinating data exchange among healthcare providers, using payment models as levers to reform service delivery to improve quality and reduce costs, and involving the consumer in both data collection and use—can form the basis of a reform model to improve care and lower costs (Figure 4-2).
Better Diabetes Care at Lower Cost
Diabetes, one of the leading chronic diseases in the nation, highlights many ways in which improvements in electronic data exchange can enhance care and reduce costs (Figure 4-3). There is already electronic information exchange in such areas as filling prescriptions and managing laboratory results that is used in the delivery of care. With feedback from these electronic sources of information, it becomes more feasible to support improvements in care for patients with diabetes by driving evidence-based treatments for a defined population of patients from a registry who may be at risk from certain diabetic complications and also to measure costs. This does not require pooling all information. As long as the systems from different providers
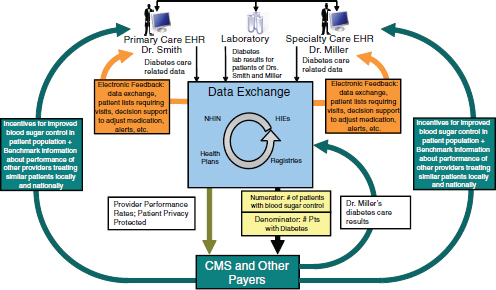
FIGURE 4-3 Schematic in Figure 4-2 applied to diabetes. The result: better care at lower cost.
and organizations are using consistent methodologies for constructing the measures, it is possible to get a more comprehensive picture of quality of care. The information can then be utilized in several areas: by providers to help them improve care in a timely way, by patients to help them identify ways to obtain better care, and as the basis for payment and benefit reform.
Accountable Care Organizations
Many of the lessons learned from how to utilize existing data to improve quality and reduce costs are being applied on launching performance measurement programs for five pilot accountable care organizations (ACOs) supported by the Brookings-Dartmouth Learning Network for Accountable Care Organizations. These pilot sites and their payer partners are attempting to implement changes in payments around value at the organizational level. These efforts typically begin with the information that they already have available in existing systems—such as claims—which in many cases, are the only data available.
Efforts to implement learning networks at ACOs could involve three stages. In the basic phase, ACOs utilize existing data from medical, pharmacy, and laboratory claims from payers. In the intermediate phase, ACOs incorporate specific clinical data, such as electronic laboratory results, as well as limited survey data. In the advanced phase, ACOs add more com-
plete clinical data, such as electronic records and registries, and robust patient-generated data, such as health risk appraisals and functional status.
These incremental improvements in the utilization of available health data can have simultaneous impacts in several key areas including care effectiveness (in individual and public health), safety, patient engagement, and efficiency (reducing overuse). More detailed description of the measures within these categories is provided in Table 4-1.
Advances in HIT and quality measurement, along with coordination between efforts will ultimately lead to improved care and better quality and cost information. In the coming years, as the various aspects of quality measurement and HIT are each improved on their own, additional attention is needed to ensure that these elements work together to produce increasingly sophisticated, patient-centered information about quality, costs, and care experience (Figure 4-4).
The Path Forward
The focus for the next 3 years will be on identifying and expanding best practices, as well as developing and aligning incentives that support the quality reporting infrastructure. Recent successes that can be expanded include pilot projects to demonstrate the feasibility of integrating additional data to support more clinically sophisticated, person-centered measures over time.
Incentives will continue to play a central role in quality improvement efforts and it will be increasingly important to align measures between public and private sector payers to further promote effective incentives. Also required will be a clear plan for implementation of increasingly sophisticated patient-centered measures to encourage continuing progress in coordinating care.
Since different data sources and data collection methods may be used by different organizations, one specific area for work is on producing functionally equivalent performance results. Finally, the use of incentives will contribute to quality improvement efforts such as e-prescribing and HIT payments, quality reporting payments, shared savings, and other performance-based payments.
Using Pilot Programs to Improve Quality Reporting
Pilot programs, such as those funded by the Office of the National Coordinator (ONC) for Health Information Technology’s Beacon Community Program, have the opportunity to drive movement toward nationally consistent methods in quality reporting. Some areas to be tested with future pilot programs include the use of consistent summary reporting methods,
TABLE 4-1 Incorporating Advanced Measures Through Accountable Care Organizations
| Basic Phase—Claims-Based Measures ACOs have access to medical, pharmacy, and laboratory claims from payers |
|
| ACO Impact | Quality Improvement Measure |
| Care effectiveness/population health | Cancer care acreenings Diabetes care (LDL and A1c tests, eye exams, etc.) Coronary artery disease care (LDL test) |
| Safety | High-risk medication for the elderly Appropriate testing for patients using high-risk medications |
| Overuse/efficiency | Imaging for low back pain (in absence of “red flags”) during first 30 days Inappropriate antibiotic prescribing Utilization rates of select services (e.g., C-section) |
| Intermediate Phase—Limited Clinical and Survey Measures ACOs use specific clinical data (e.g., electronic laboratory results) and limited survey data |
|
| ACO Impact | Quality Improvement Measure |
| Care effectiveness/population health | Immunization rates for children and adolescents Patients with diabetes whose blood sugar (A1c) are in control Patients with diabetes or ischemic vascular disease whose lipids are in control Patients with hypertension whose blood pressure is in control |
| Safety | “Never events” in hospitals |
| Patient engagement | Physician instructions understood (Consumer Assessment of Health Providers and Systems [CAHPS]) Care received when needed (CAHPS) |
| Overuse/efficiency | Episode-based resource use—linked to quality measures for common medical (e.g., diabetes, acute myocardial infarction [AMI]) and common surgical conditions (e.g., hip replacement) |
| Advanced Phase—Comprehensive Patient-Focused Measures More complete clinical and robust patient-generated data |
|
| ACO Impact | Quality Improvement Measure |
| Care effectiveness/population health | Comprehensive health risk summary score (body mass index, blood pressure, cholesterol, smoking, exercise, alcohol) Stage-specific quality of life and functional outcomes for common cancers Quality of life and functional outcomes for common conditions (e.g., AMI, hip replacement, diabetes) |
| Safety | Hospital infection and risk-adjusted mortality rates Outpatient medication errors |
| Patient engagement | Care plans—patient activation and engagement in chronic/other conditions Preference-sensitive conditions—level of information communicated regarding patient choice (e.g., knee surgery) Patient preferences—adherence to design and execution of care plan (e.g., advanced directives) |
| Overuse/efficiency | Episode-based resource use—linked to quality of life, functional and patient engagement measures for common medical (e.g., diabetes, AMI) and surgical conditions (e.g., hip replacement) |
methods to ensure complete reporting (representing all patients without double counting), mechanisms to generate provider feedback, and the ability to capture and use information on race, ethnicity, language, and other valuable patient data. Successful pilot programs in these and other areas will provide links to measurable improvements in patient outcomes, error reductions, and administrative burdens. They will also provide better evidence to identify best practices going forward. The overarching goal of such efforts is to assemble the electronic infrastructure with currently available data sources, while promoting pilot programs to improve methods.
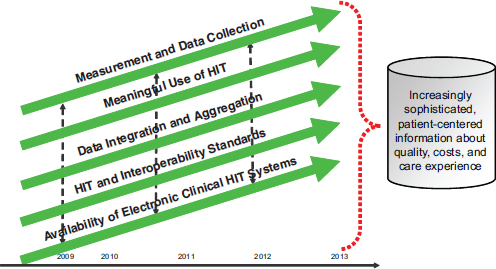
FIGURE 4-4 All aspects of quality improvement must work in concert to improve care.
Key Next Steps
Quality measurement and improvement will be crucial to healthcare delivery reform. All five elements—measurement and data collection, payment reform, benefit reform, better evidence, and quality improvement support—are important, but the focus in this paper has been on the data collection and reporting infrastructure and its role in improving quality and lowering costs. Some of the crucial areas for work in the near term are to identify best practices for quality reporting and payment reform, to expand successful pilots projects for measurement and payment reform, and to develop and pilot more advanced patient-focused measures. Ultimately, momentum in all of these areas will combine synergistically to yield more advanced patient-focused measures and more sophisticated delivery of care.
ENGAGING INDIVIDUALS IN POPULATION HEALTH MONITORING
Kenneth D. Mandl, M.D., M.P.H.
Children’s Hospital Boston
The next major step in the evolution of population research is to engage large, distributed consenting populations in longitudinal study. By treating individuals as collaborators and not just subjects in research or cases in public health we will mine a largely untapped source of knowledge about health and disease—the patient. Emerging patient-oriented health information technologies will transform the research enterprise, helping to establish a learning health system.
Following are four near-term predictions all informed by our experiences in developing, evaluating, and diffusing the Indivo personally controlled health record (PCHR) to engage the patient in medicine, research, and public health (Mandl et al., 2007).
- Individuals will share their own data and observations for the public good.
- As data begin to flow into patient-controlled mechanisms (such as PCHRs), populations of individuals will control datasets that are larger and more complete than those traditionally used in population research.
- Present-day online social networks are a rudimentary version of a major pillar in an emerging health information technology (HIT) infrastructure.
- Engaging populations will require development of incentives and enticements as well as blurring the boundaries between clinical care and research.
The Personally Controlled Health Record
In 1998, structural and sociopolitical concerns motivated us to define a new approach to managing electronic health information. Though most health information at the time was stored on paper, we anticipated a time when it would be stored electronically. That time has been slow to arrive. Studies have shown that, even now, very few outpatient or inpatient settings have a complete electronic health record (EHR) (DesRoches et al., 2008; Jha et al., 2009). In the 1990s, the main problem was that the information stored in EHR systems was—and for the most part still is—generally stored locally at the site where it was recorded. As a result, EHR data were often unavailable at the point of care (Bourgeois et al., 2010). In response, we developed the PCHR—a subset of personal health records (PHRs)—which inverts the standard model and instead allows a patient to assemble, maintain, and manage a secure copy of their medical data. Originally called the Personal Internetworked Notary and Guardian, and created under funding from the National Library of Medicine, Indivo is an open source, open standards PCHR platform. Indivo, much like Quicken and MINT.com function for financial data, is a tool that enables patients to collect copies of their data longitudinally across sites of care. Similar to the iPhone and Apple’s App Store, Indivo exposes those data (under patient control) to third-party applications across an open application programming interface (Mandl and Kohane, 2009). Hence, Indivo is designed to spawn an ecosystem of apps, providing functionality and promoting innovation (Figure 4-5). Reflecting on the promise of the PCHR model, Harvard Professor Clayton Christensen observes: “We cannot overstate how important PHRs are to the efficient functioning of a low-cost, high quality health-care system.…We think that the Indivo system, or something like it is a good place to start” (Christensen et al., 2009).
After diffusion of the PCHR model at two Harvard Medical School invitational conferences,1 Indivo has become the reference model for subsequent PCHRs: Microsoft’s HealthVault used Indivo software code; Google-Health implemented the model on its own servers with its own code; and the Dossia consortium contracted with the Indivo creators to create a version for deployment to populations of employees from organizations such as Wal-Mart, AT&T, and Intel.
The Next Stage
Uptake of PCHRs has been gradual primarily because PCHRs work best when they can readily obtain a copy of data from EHRs, and EHR vendors have been slow to allow data liquidity. Yet there is reason for
___________________
1 See www.pchri2006.org and www.pchri2007.org (accessed February 24, 2011).
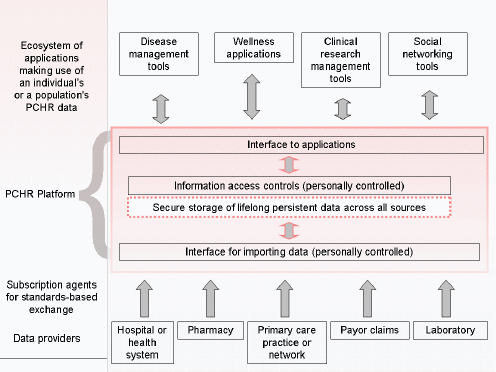
FIGURE 4-5 The personally controlled health record (PCHR) architecture.
SOURCE: Adapted from (Mandl and Kohane, 2008).
optimism. The Health Information Technology for Economic and Clinical Health Act provides that for covered entities using or maintaining an EHR, “the individual shall have a right to obtain from such covered entity a copy of such information in an electronic format.” By 2013, this feature of EHRs will be required under the final rule for “meaningful use” of certified EHR technology. In preparation, early-stage efforts have arisen to promote this data liquidity, including the very well marketed “Blue Button” initiative (Chopra et al., 2010). Hence we can expect that even before 2013 a “tectonic shift in the health information economy” will begin, mediated by a change in the locus of control of health information from institutions to individuals (Mandl and Kohane, 2008). For this shift to happen, we must learn to entice populations of patients to share data for research and public health by engaging them on their own terms. The PCHR is a technology designed to do just that. PCHRs enable the patient to authorize access to information—views or even copies of the record—to intelligent software agents (“apps”) or individuals including clinical providers, family members, healthcare proxies, and researchers.
While this shift will be largely driven by a need to improve clinical care processes, it will also have a deep impact on population health research. The ability to reach out to populations directly will produce very large cohorts of individuals who can share EHR data and provide detailed self-reported information about their care and health status.
Evidence suggests that patients are faithful reporters about their health. In fact, parents report more accurately on the past medical history of their infants than do physicians in the chart (Porter et al., 2000). When patients self-report their emergency department chief complaints using a brief survey, the accuracy of real-time disease surveillance systems greatly improves (Bourgeois et al., 2007). Furthermore, patient reports about adverse events are timelier and more concordant with their actual health status than those reported by clinicians (Basch, 2010).
There is also evidence that patients are willing to share data with researchers and public health professionals. In a population of patients that had used an early version of a PCHR for over a year, only 9% were unwilling to allow researchers or public health professionals access to an anonymized copy of those data. Patients were most willing to share when they were guaranteed anonymity and that the data would be used primarily for research. While altruism was clearly a motivator, willingness to share was increased if the subjects were offered compensation (Weitzman et al., 2010).
Recent studies also suggest that returning information about health, even in aggregate, to an online community is highly gratifying and promotes information altruism. The for-profit online social networking community PatientsLikeMe has demonstrated that individuals with a severe chronic disease are highly willing—even without compensation—to contribute data and observations to a patient community in order to accelerate learning about their disease (Frost and Massagli, 2008). Similarly, we recently ran a “data donation drive” in a nonprofit online social network of patients with diabetes,2 rapidly recruiting a cohort of nearly 2,000 individuals sharing data under an implied consent model. What was returned to the community was a “riskscape” picture of glycemic control displayed to the community in aggregate form on maps and graphs. In this experiment, funded by the Centers for Disease Control and Prevention, over 95% of participating network members were willing to be recontacted about opportunities to participate in research.
Engaging Patients
A critical research question is how to engage individuals in sustained participation in research cohorts. Across multiple domains, the data col-
___________________
2 See www.tudiabetes.org (accessed February 24, 2011).
lected in research are becoming increasingly relevant in clinical settings. For example, as genomic data acquired for research become more clinically pertinent to individuals, inventing creative mechanisms to manage this communication will become an imperative. There is a mounting consensus that participants in genomic research deserve to learn of findings pertinent to their health and well-being (Fabsitz et al., 2010). At Children’s Hospital Boston’s Gene Partnership Project,3 we have begun to engage our patients as collaborators in research, developing ethically sound approaches to returning actionable results directly to patients (Kohane et al., 2007).
Focused study is needed to determine whether, and under what circumstances, it is research results, aggregate community-level views, information about “patients like me,” financial incentives, pure altruism, or something else that most motivates individuals to share information in a sustained manner. Unlocking the knowledge possessed by populations of individuals is the work of the next decade.
OPTIMIzING CHRONIC DISEASE CARE AND CONTROL
Sophia W. Chang, M.D., M.P.H.
California HealthCare Foundation
Chronic disease care provides important opportunities for the use of electronic health information to inform care delivery. The prevalence of chronic disease in the United States continues to increase and 20% of the population currently accounts for 80% of healthcare costs (Anderson and Wilson, 2006). The opportunity for better quality and more cost-effective care lies in identifying and delivering useful interventions in a timely and nonduplicative manner. The prevalence of comorbid conditions (over 50% of those with a chronic condition) also raises important questions about better care management approaches (Anderson and Wilson, 2006). There is already indication that overemphasis on the close management of a single condition may cause harm—for example, in the setting of diabetes and cardiovascular disease (Skyler et al., 2009). Beyond the knowledge gained from randomized controlled trials, systematic review of population data may allow us to reap timely information about optimal therapies and approaches from clinical observation, experience, and documented patient outcomes.
Leveraging Patient Health Data
As the most powerful actors in care, actively engaging patients in the management of their chronic conditions is going to be vital to any system-
___________________
3 See www.geneparnership.org (accessed February 24, 2011).
wide improvements in healthcare delivery. The need for increased and more timely use of electronic health data provides an opportunity to involve patients in providing, validating, and using their own health information, priming them to reap significant benefits in both health and quality of life. This requires a paradigm and culture shift from our present systems where shared data infrastructures are solely controlled by healthcare providers.
The existing healthcare data infrastructure has a number of limitations that make it difficult for healthcare professionals and patients to effectively use patient health data. Currently, there is a lack of commonly accepted standardized nomenclatures and data formats—and only limited use of what does exist—for items consistently included in medical documentation (e.g., chief complaints or symptoms, laboratory results, radiology and pathology interpretations). This lack of standardization limits the ability to aggregate meaningful information across providers and even within large institutions. These same limitations also prevent widespread decision rules and tools from being used effectively to support the consistent practice of evidence-based care (i.e., robust clinical decision support). Furthermore, opportunities to include quality of life, functional status, and self-management measures—all of which would be predominantly provided by patients—are also hindered by the lack of standardization and system functionality. Current patient portal approaches to sharing EHR information generally lack the ability for patients to extract and use their own structured electronic data. In addition, these records do not include the concrete information that often is most valuable for patients managing a chronic condition: how to make dietary changes, adhere to medications, manage side effects, incorporate exercise into their schedules, etc.
While we progress toward the paradigm of engaging patients in the management of their own electronic health data, an important stepping stone is the clear understanding that data within the record belong to the patient and not to either the provider or the institution (Ralston et al., 2010; Walker et al., 2009). Using EHR data as a mode of communication between patients and providers simultaneously validates data quality and engenders trust in its use for clinical care decisions. Although to date no organization has a truly interactive and shared patient record system, some have developed successful patient portals that may move in that direction.
Large enterprises like Kaiser Permanente and the Veterans Health Administration (VHA) have maintained population management systems in parallel to their transactional EHR systems (High Value Health Care Project, 2010). The VHA recently published their experience in managing a national data warehouse (disease registry) for the high-cost chronic conditions of HIV/AIDS and Hepatitis C. Focusing on data use for quality management, the VHA’s approach uses a system that pulls data nationally from its EHR system and includes local clinicians at each medical center
who systematically confirm patients in each population. A key enabler of adoption was providing local clinical support and query tools to help clinicians validate data elements and manage patient populations locally. Adoption and use are further supported through national-level reporting of quality measures, which are in turn tied to financial bonuses and incentives (Backus et al., 2009).
A similar example exists in the Health Maintenance Organization (HMO) Research Network (HMORN), an organization of HMO research programs. HMORN creates virtual data warehouses pulled from EHR systems across 16 provider organizations. It should be noted that each HMO in the network has been using its EHR system in the context of a robust quality improvement culture and infrastructure. That is, the review and use of EHR data for clinical management play a significant role in providing validated data for research purposes.
Challenges and Next Steps
Small physician offices not affiliated with a large system have, to date, had less opportunity to receive rapid-cycle quality improvement feedback and participate in clinical research. Recognizing the need for and value of aggregated population-level data, a growing number of regional health information exchanges are offering (or plan to offer) population reports to support improvements in disease management. Furthermore, it is hoped that federal investments in regional extension centers will support smaller practices in EHR adoption. Experience to date, however, has demonstrated that the EHR adoption effort is highly time- and resource-intensive and requires ongoing local management and feedback in order to reap improvements in clinical care (Nutting et al., 2009).
To maximize the value of EHRs—for both improved quality of care and increased patient satisfaction—existing research paradigms need to shift toward real-time feedback and knowledge development. Electronic data hold the promise of being able to provide more timely and extensive comparative effectiveness data. Especially in settings of comorbid conditions, it will be increasingly difficult to have a single evidence-based best practice. More likely, we will have a range of options, with associated potential risks and benefits, to support shared decision making between clinicians and patients on which chronic disease management course to take. The important next step is the ability to collect and aggregate those treatment regimens and outcomes to better inform current and future practice. To bring the patient back into the center, data about care processes, quality of life, and side-effect experiences will be increasingly valued, collected, and shared directly with patients.
To move toward these goals, some next steps should include
- Wider adoption of standardized core data elements. Current implementation efforts are time-consuming and expensive, and may be redundant without national requirements.
- Better align what is important to patients with the data elements collected by EHRs. This requires a move beyond portals toward new paradigms for patient-entered data and for data sharing, with sharing directed by patients to a range of providers and others who support them in the management of their health.
- Learn how to incorporate patient experience into our knowledge base and data systems. If we spend too much time, effort, and expense on the provider side, we are missing the biggest opportunities to improve chronic disease. The care experience goes well beyond what is documented in a health record, and a “learning system” must be able to understand, document, and improve care processes outside the clinical encounter.
- Develop a better paradigm for learning from patient data. Our expanding Health Insurance Portability and Accountability Act privacy framework is a start, but a clearer continuum for data use—quality improvement, system improvement, clinical effectiveness, and health services research—is needed. There is a blurring of the lines between real-time data feedback, measuring to improve care processes, and improving clinical outcomes when EHR data can be used to meet all these needs.
- Better translate what we already “know” about population health into actionable information. The potential to mine aggregated data to improve the health of populations gives us incredible power. Given the value of these potential data, how do we ensure that new knowledge is put into practice when we do a poor job of doing so already?
In the end, we must be wary of arguments about “primary” vs. “secondary” use of clinical data, and keep in mind that it is the patients who own their clinical information and live with their chronic conditions. It is incumbent upon us to ensure that the investments made in automating this vital information are brought to the service of patients who, in the end, will at some point be every single one of us.
TARGETING POPULATION HEALTH DISPARITIES
M. Christopher Gibbons, M.D., M.P.H.
Johns Hopkins Urban Health Institute
Traditionally, modern medicine has sought to understand health and disease largely through elucidating molecular, physiological, or psycho-
logical mechanisms and determinants. While this approach has yielded significant gains in individual longevity, achieving sustained population-wide gains has been much more elusive (NRC, 2001). This reality is perhaps most convincingly illustrated by the existence of healthcare disparities. Systematically reducing healthcare disparities is a vexing challenge that, to date, has yet to be accomplished. Part of the challenge lies in our understanding of the causes of these disparities. Disease causation in general, and health disparities in particular, results from complex interactions of many factors that simultaneously and often cooperatively act longitudinally across more than one level of influence (Gibbons et al., 2007). As such, a comprehensive understanding of disparities requires the integration of knowledge derived from the bench with that from sociobehavioral and population sciences. In a similar fashion, treating disease at the bedside and addressing healthcare disparities in the population will require an integrating health and social care systems with a focus on clinical, behavioral, and environmental determinants of health.
Demographic Trends and Challenges
Several national trends suggest the pressing need for this type of integrated approach to population health and health disparities. First, the high prevalence of chronic disease in our society represents a challenge for a healthcare system largely oriented to acute care episodes. Chronic diseases are the leading cause of illness, disability, and death with over 15% of the U.S. population suffering from activity limitations resulting from chronic diseases (IOM, 2001). Because individuals suffering from chronic disease often have these diseases for many years, yet only need acute clinical services for relatively short periods of time, most of the “care” they will receive will be provided by relatives and friends in the home or community (IOM, 2001). To complicate matters further, the United States is experiencing a burgeoning of the senior population. In 2000, 35.0 million people (12%) were over the age of 65 (Meyer, 2001). The proportion of seniors in the U.S. population (age ≥ 65) is expected to increase to approximately 20% in 2030 (71 million seniors). The number of persons age > 80 years is expected to increase from about 9 million in 2000 to 19 million in 2030. With 80% of all seniors burdened by a chronic condition, and 50% having two or more, these trends will challenge our ability to provide chronic disease care (CDC, 2003).
The United States is also becoming more racially and ethnically diverse. Between 2000 and 2050, the number of Asians is expected to increase by 22.7 million (213%), while the number in the “all other races” (which includes American Indians and Alaska Natives, Native Hawaiians and other Pacific Islanders, and individuals who identify with two or more races)
category will increase by 15.3 million (217%). The population of Hispanic or Latino origin is projected to steadily increase as a percentage of the total U.S. population through 2050, rising from 12.6% in 2000 to 24.4% in 2050 (Shrestha, 2006). This increase is occurring even as the U.S. population overall is also expected to grow and reach approximately 420 million persons by year 2050.
These population-level increases come at a time when the physician and nurse workforces are rapidly aging (HRSA, 2003). This will inevitably result in greater patient reliance on family and informal caregivers to help meet healthcare management needs. The actions of these caregivers and patients will be influenced by cultural norms, attitudes, beliefs, and practices that could influence healthcare decision making and health outcomes. Taken together, these trends suggest a growing importance of “nonclinical” factors in the genesis and treatment of disease as well as the reduction of healthcare disparities.
Populomics
Elucidation of mechanisms of action and understanding disease pathogenesis in an integrated way will require the generation and synthesis of large, complex, and diverse datasets. In addition, treating disease from this perspective will require real-time synthesis and analysis of multilevel data at the point of care. As such, advances in health information technology (HIT), electronic health records (EHRs), and health information exchanges offer significant promise in bringing these needs into reality. The term populomics has emerged from the synthesis of the population sciences, medicine, and informatics to describe this integrative, systems-oriented perspective (Abrams, 2006; Gibbons, 2005). Populomics is focused on transdisciplinary, integrative disease/risk characterization, interdiction, and mitigation and relies heavily on innovations in computer and information technologies to characterize the interplay of sociobehavioral pathways, and biophysiological/molecular mechanisms that work across levels of existence to impact health at the individual and population levels (Gibbons, 2008).
Research paradigms like the sociobiologic integrative model provide a conceptual framework for populomics-oriented research and analysis (Gibbons et al., 2007). This model posits that individuals are constantly being exposed to many health-impacting factors in the environments in which they live. These are collectively called “inputs.” Some of these inputs may be modified to increase or attenuate their effects via “other” environmental factors. These other factors are called indirect environmental inputs. Once an individual or population is exposed to a given factor or set of factors, these direct and indirect inputs are, in turn, acted upon by metabolic, digestive, and/or detoxification systems within the body. If
inputs or their metabolic products overwhelm bodily defense or regulatory mechanisms, illness and disease will occur (Gibbons et al., 2007). Because inputs, biological processes, and outcomes exist on several levels, the model is conceived as operating on the cellular, individual, and population levels—temporally proceeding from input (exposure) to cellular-, individual-, or population-level outcomes. Within this context, disease will only occur if the magnitude of impact produced by inputs and metabolic processes is sufficient to overwhelm bodily reparative, restorative, or compensatory mechanisms and cause genotypic, phenotypic, or psychological abnormalities that ultimately result in a disease state or health deficit (Gibbons et al., 2007). If the resulting deficits manifest only at the cellular level, it may be detectable as a change in susceptibility or predisposition. If they manifest at the individual level, it would result in a disease or illness state. If they occur in a large number of people in a given population, it could be detected as an epidemic, pandemic, or disparity. Science must organize and define the inputs, biological processes, and outcomes that exist and the relationships between them that undergird disease at each level of exposure (Gibbons et al., 2007).
Populovigilance
Recent advances in information technology and computer science are making the capture, organization, and synthesis of large amounts of data possible. With the evolution of EHRs, personal health records, consumer health informatics, and social media, we are entering an era when this synthesis and analysis are possible at the bedside in real-time. In the future, working from this integrative perspective, we may find that understanding single etiologies or factors (bacteria, viruses, poverty, race, ethnicity) might be less important than knowing that a given group of factors work together, across levels of analysis (cellular, individual, and population), to collectively influence discreet biomolecular mechanisms and result in a given outcome. These groups of individual- and population-level factors that predictably coexist and act cooperatively to influence discrete health outcomes could then form the basis of so-called sociobehavioral disparities phenotypes. Further, scientists may be able to usher in a new generation of genome-wide association studies (GWAS) that actually start at the population level. Here, scientists would seek to define one or more sociobehavioral phenotypes across a given population of consumers or patients and then link them with underlying biophysiological, psychological, and molecular mechanisms, constructing “causal profiles.”
Across a group of patients with a given disease or disparities, one or more causal profiles may exist. This suggests that across a population of patients, with a given disease (breast cancer) or disparity (elevated prostate
cancer rates among African Americans), there exist multiple pathways to a given outcome. Because this form of analysis starts at the population level—by first elucidating those sociobehavioral phenotypes that actually exist across that population—this “PheGe” (phenotypic-genotypic) analysis may be more cost-effective than typical GWAS that attempt to first identify molecular pathways and then determine the prevalence of the identified pathway in a defined population. For similar reasons, these causal profiles could ultimately prove to have more predictive value than commonly used constructs like race, ethnicity, or any other single factor thought to be a “fundamental” cause of disparities.
Rather than debating “fundamental causes” as the only credible starting point for disparities research, it may be possible to think in terms of a disparities-oriented “populovigilance” where scientists work to collect, monitor, and evaluate data from defined populations, on the adverse effects of disparate care, environmental hazards, behavior, and policies. Implemented effectively, this could identify hazards and/or sentinel events associated with the existence of healthcare disparities as well as prevent harm to patients and individuals among the target subpopulations (disparities harm reduction research).
Clearly, many challenges must be overcome prior to accomplishing these tasks and realizing these goals. While the future is unknown, the potential of HIT to yield novel insights and enable new advances in treatment, public health, and healthcare disparities is certainly significant.
REFERENCES
Abrams, D. B. 2006. Applying transdisciplinary research strategies to understanding and eliminating health disparities. Health Education and Behavior 33(4):515-531.
Anderson, G. F., and K. B. Wilson. 2006. Chronic disease in California: Facts and figures. http://www.chcf.org/publications/2006/10/chronic-disease-in-california-facts-and-figures (accessed September 10, 2010).
Backus, L. I., S. Gavrilov, T. P. Loomis, J. P. Halloran, B. R. Phillips, P. S. Belperio, and L. A. Mole. 2009. Clinical case registries: Simultaneous local and national disease registries for population quality management. Journal of the American Medical Informatics Association 16(6):775-783.
Basch, E. 2010. The missing voice of patients in drug-safety reporting. New England Journal of Medicine 362(10):865-869.
Bourgeois, F. C., K. L. Olson, and K. D. Mandl. 2010. Patients treated at multiple acute health care facilities: Quantifying information fragmentation. Archives of Internal Medicine 170(22):1989-1995.
Bourgeois, F. T., S. C. Porter, C. Valim, T. Jackson, E. F. Cook, and K. D. Mandl. 2007. The value of patient self-report for disease surveillance. Journal of the American Medical Informatics Association 14(6):765-771.
CDC (Centers for Disease Control and Prevention). 2003. Trends in aging—United States and worldwide. Morbidity & Mortality Weekly Report 52(6):101.
Chopra, A., T. Park, and P. Levin. 2010. “Blue Button” provides access to downloadable personal health data. http://www.whitehouse.gov/blog/2010/10/07/blue-button-providesaccess-downloadable-personal-health-data (accessed March 11, 2011).
Christensen, C., J. Grossman, and J. Hwang. 2009. The innovator’s prescription: A disruptive solution for health care. New York: McGraw-Hill.
DesRoches, C. M., E. G. Campbell, S. R. Rao, K. Donelan, T. G. Ferris, A. Jha, R. Kaushal, D. E. Levy, S. Rosenbaum, A. E. Shields, and D. Blumenthal. 2008. Electronic health records in ambulatory care—a national survey of physicians. New England Journal of Medicine 359(1):50-60.
Fabsitz, R. R., A. McGuire, R. R. Sharp, M. Puggal, L. M. Beskow, L. G. Biesecker, E. Bookman, W. Burke, E. G. Burchard, G. Church, E. W. Clayton, J. H. Eckfeldt, C. V. Fernandez, R. Fisher, S. M. Fullerton, S. Gabriel, F. Gachupin, C. James, G. P. Jarvik, R. Kittles, J. R. Leib, C. O’Donnell, P. P. O’Rourke, L. L. Rodriguez, S. D. Schully, A. R. Shuldiner, R. K. Sze, J. V. Thakuria, S. M. Wolf, and G. L. Burke. 2010. Ethical and practical guidelines for reporting genetic research results to study participants: Updated guidelines from a national heart, lung, and blood institute working group. Circulation: Cardiovascular Genetics 3(6):574-580.
Frost, J. H., and M. P. Massagli. 2008. Social uses of personal health information within PatientsLikeMe, an online patient community: What can happen when patients have access to one another’s data. Journal of Medical Internet Research 10(3):e15.
Gibbons, M. C. 2005. A historical overview of health disparities and the potential of eHealth solutions. Journal of Medical Internet Research 7(5):e50.
———. 2008. Populomics. Studies in Health Technology and Informatics 137:265-268.
Gibbons, M. C., M. Brock, A. J. Alberg, T. Glass, T. A. LaVeist, S. Baylin, D. Levine, and C. E. Fox. 2007. The sociobiologic integrative model (SBIM): Enhancing the integration of sociobehavioral, environmental, and biomolecular knowledge in urban health and disparities research. Journal of Urban Health 84(2):198-211.
High Value Health Care Project. 2010. How registries can help performance measurement improve care. http://www.rwjf.org/files/research/65448.pdf (accessed September 10, 2010).
HRSA (Health Resources and Services Administration). 2003. Changing demographics: Implications for physicians, nurses, and other health workers ftp://ftp.hrsa.gov/bhpr/nationalcenter/changedemo.pdf (accessed January 18, 2011).
IOM (Institute of Medicine). 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
Jha, A. K., C. M. DesRoches, E. G. Campbell, K. Donelan, S. R. Rao, T. G. Ferris, A. Shields, S. Rosenbaum, and D. Blumenthal. 2009. Use of electronic health records in U.S. hospitals. New England Journal of Medicine 360(16):1628-1638.
Kohane, I. S., K. D. Mandl, P. L. Taylor, I. A. Holm, D. J. Nigrin, and L. M. Kunkel. 2007. Medicine. Reestablishing the researcher-patient compact. Science 316(5826):836-837.
Mandl, K. D., and I. S. Kohane. 2008. Tectonic shifts in the health information economy. New England Journal of Medicine 358(16):1732-1737.
———. 2009. No small change for the health information economy. New England Journal of Medicine 360(13):1278-1281.
Mandl, K. D., W. W. Simons, W. C. Crawford, and J. M. Abbett. 2007. Indivo: A personally controlled health record for health information exchange and communication. BMC Medical Informatics and Decision Making 7:25.
Meyer, J. 2001. Age 2000. Washington, DC: U.S. Census Bureau.
NRC (National Research Council). 2001. New horizons in health: An integrative approach. Washington, DC: National Academy Press.
Nutting, P. A., W. L. Miller, B. F. Crabtree, C. R. Jaen, E. E. Stewart, and K. C. Stange. 2009. Initial lessons from the first national demonstration project on practice transformation to a patient-centered medical home. Annals of Family Medicine 7(3):254-260.
Porter, S. C., M. T. Silvia, G. R. Fleisher, I. S. Kohane, C. J. Homer, and K. D. Mandl. 2000. Parents as direct contributors to the medical record: Validation of their electronic input. Annals of Emergency Medicine 35(4):346-352.
Ralston, J. D., K. Coleman, R. J. Reid, M. R. Handley, and E. B. Larson. 2010. Patient experience should be part of meaningful-use criteria. Health Affairs 29:607-613.
Shrestha, L. B. 2006. The changing demographic profile of the United States. http://digital.library.unt.edu/ark:/67531/metacrs9276/ (accessed January 18, 2011).
Skyler, J. S., R. Bergenstal, R. O. Bonow, J. Buse, P. Deedwania, E. A. Gale, B. V. Howard, M. S. Kirkman, M. Kosiborod, P. Reaven, and R. S. Sherwin. 2009. Intensive glycemic control and the prevention of cardiovascular events: Implications of the ACCORD, ADVANCE, and VA diabetes trials: A position statement of the American Diabetes Association and a scientific statement of the American College of Cardiology Foundation and the American Heart Association. Circulation 119(2):351-357.
Walker, J., D. K. Ahern, L. X. Le, and T. Delbanco. 2009. Insights for internists: “I want the computer to know who I am.” Journal of General Internal Medicine 24(6):727-732.
Weitzman, E. R., L. Kaci, and K. D. Mandl. 2010. Sharing medical data for health research: The early personal health record experience. Journal of Medical Internet Research 12(2):e14.
























