5
Heart Failure, Cardiomyopathy, and Right Heart Failure
The approach to heart failure adopts the strategy of the current listings to require both a cardiac abnormality diagnostic of the condition and a functional limitation demonstrated by the patient, which recognizes the wide variability of the relationship between anatomic impairment and disability. The cardiac abnormality for heart failure is distinguished as either systolic failure with low ejection fraction or diastolic dysfunction with preserved ejection fraction. The recommendations are cognizant of the safety and utility of exercise testing, particularly if the exercise testing is combined with the determination of peak oxygen consumption. The recommendations would also allow the functional limitation requirement to be met by repeated hospitalizations. Although most cardiomyopathies will be evaluated as heart failure, unique considerations for hypertrophic cardiomyopathy led to the proposal of a separate indication. Although right heart failure is not a unique disease, it can occasionally dominate the clinical picture of left heart failure and other serious cardiac conditions. Therefore, right heart failure has been proposed as another route to meeting a listing.
DESCRIPTION
Heart failure is the inability of the heart to provide sufficient blood flow to meet the needs of the body for oxygenated blood during regular activity or to do so only with elevated pressures within the heart (Arena et al., 2007; Colucci and Braunwald, 1995). Heart failure is not one disease, but
a chronic and progressive condition that can be the final common pathway from a number of structural or functional cardiac disorders that impair the ability of the heart to fill or empty (Arena et al., 2007). Heart failure can occur when the heart muscle is weak (systolic failure) or when it is stiff and unable to relax normally (diastolic failure). Cardiomyopathy, which means “disease of the heart muscle,” is one of many causes of heart failure.
The syndrome of heart failure is often characterized by shortness of breath or fatigue, even during mild exertion. A common feature of heart failure decompensation is retention of excess fluid, which contributes to raise fluid pressures within the heart, lungs, and the rest of the body, which is termed congestion. When congestion occurs on the left side of the heart (left heart failure), it can lead to shortness of breath occurring during modest activity or at rest, particularly while lying down. Elevation of pressures can also occur in the veins draining into the right side of the heart, which can result in peripheral edema and abdominal swelling (ascites). These findings are often termed right heart failure. The type of heart muscle weakness is often described by the pumping strength of the heart, measured by the left ventricular ejection fraction (EF). This is the proportion of blood ejected from the ventricle with each beat, expressed either as a fraction (normally 0.55 to 0.70) or as a percentage of the total volume in the left ventricle (normally 55 to 70 percent).
The primary manifestations of heart failure are fatigue, dyspnea, and peripheral fluid retention, which alone or in combination may limit a patient’s ability to perform activities (Gibbons et al., 2002) and potentially lead to impairment or disability. All three symptoms do not have to manifest for a patient to have heart failure. Although the full-blown chronic syndrome has sometimes been termed “congestive heart failure,” clinical evidence of congestion (as described above) may or may not be present at the time of evaluation (Allen et al., 2008). For example, a patient may experience exercise intolerance from fatigue but have little fluid retention, or may be limited by peripheral swelling without reporting symptoms of fatigue or breathlessness.
EPIDEMIOLOGY
Heart failure is an ongoing public health challenge in the United States. In the adult population over age 20, the prevalence of heart failure is 2.6 percent. It is generally higher in men than in women but highest in black women. The lifetime risk of developing heart failure is one in five at age 40 for both men and women. The prevalence in patients between ages 20 and 39 is less than 0.5 percent of the population. In patients over age 45, the incidence of new cases is estimated at 670,000 annually. From ages 40 to 59, the prevalence is 1.9 per 100 men and 1.4 per 100 women (Lloyd-Jones
et al., 2010). Overall, approximately 5.8 million people have heart failure in the United States, leading to more than 1 million hospitalizations each year and 12 to 15 million patient visits to treating physicians annually.
Heart failure with preserved ejection fraction (sometimes termed HFpEF, or diastolic heart failure) accounts for about half of all U.S. heart failure hospitalizations (Redfield et al., 2003). The majority of these patients are over age 70, and it is particularly common when patients have chronic hypertension, diabetes, and obesity. For the patients under age 65, diastolic heart failure is uncommon, except in association with longstanding severe hypertension, which is particularly prevalent in African-American men. Rare causes of this type of heart failure are amyloidosis and restrictive cardiomyopathy (Redfield et al., 2003). Although hypertrophic cardiomyopathy is present in about 1 in 500 persons, most people with this diagnosis remain highly functional and do not come under consideration for disability (Ho and Seidman, 2006).
Heart failure contributed to 282,754 U.S. deaths in 2006 (Lloyd-Jones et al., 2010). Median life expectancy for a person with heart failure has been described as less than 5 years (Allen et al., 2008), but many of these statistics are derived from the population older than those considered for disability. Some patients under age 65 who are in good general health other than cardiac disease may be considered for cardiac transplantation. Otherwise, mortality within 1 year can be expected for about half of patients in whom severe symptoms due to heart failure limit their daily life despite current medical therapies (Allen et al., 2008).
TYPES OF HEART FAILURE
Systolic Heart Failure
When the syndrome of heart failure occurs with an EF less than 40 percent, the dysfunction is often called “systolic heart failure,” referring to weakened pumping strength. Heart failure with low EF is generally associated with enlargement of the heart (measured by left ventricular diameter on echocardiography, normally less than 5.4 cm in diameter). This is the most common type of heart failure in patients younger than age 65. The most common causes are cardiomyopathy, which means weakness of the heart muscle not resulting from another cardiac diagnosis, and coronary artery disease, which causes irreversible damage to the heart muscle and has usually been preceded by a myocardial infarction (heart attack), although these often occur silently.
Most cases of heart failure not otherwise specified are systolic heart failure. The major symptoms in heart failure, as described above, are the same whether the EF is low (less than 40 percent) or preserved (greater than
40 percent). These relate to fluid retention and congestion, leading to shortness of breath and swelling. However, the reduced pumping strength of the heart in systolic heart failure can also lead to decreased blood flow during periods of increased demand during activity, causing early fatigue and prolonged exhaustion after heavy activity, even without obvious breathlessness. When the disease becomes very advanced, the amount of blood flow to the body (cardiac output) may actually be lower than normal at rest, which is sometimes demonstrated during cardiac catheterization if specific measurements of cardiac output are made. When the reduction is so extreme that it is life threatening, it is called cardiogenic shock.
Diastolic Heart Failure
Heart failure can also occur with “preserved” EF, in which the heart muscle is stiff rather than weak, leading to impaired filling of the chambers of the heart. The strength of the heart muscle and amount of blood flow to the body are usually well maintained and EF is greater than 40 percent, although not always in the normal range of 55 to 70 percent. The major structural abnormality is increased stiffness of the heart muscle, usually with some increase in wall thickness. This stiffness leads to impaired filling of the heart, or diastolic dysfunction. Numerous measurements can be made on echocardiography to determine how well the heart can relax and fill (diastolic function); these vary by laboratory, but are usually summarized in echocardiographic reports as diastolic dysfunction that is “mild,” “moderate,” or “severe.”
Most symptoms of diastolic heart failure relate to chronic shortness of breath during exertion, the same as for systolic heart failure. Symptoms of peripheral swelling may occur earlier in the course of diastolic heart failure than in systolic heart failure. However, the presentation and diagnosis are often dominated by repeated hospitalizations for episodes of sudden decompensation. These episodes are sometimes termed acute or “flash” pulmonary edema (fluid in the lungs with rapid development of severe shortness of breath).
Hypertrophic Cardiomyopathy
Hypertrophic cardiomyopathy is a condition in which the heart muscle is unusually thick, with left ventricular EF that can be above normal. This condition is usually diagnosed by echocardiography, which reveals both abnormal thickness of the heart muscle and abnormal diastolic function. Hypertrophic cardiomyopathy is sometimes considered a type of diastolic heart failure, but it is often considered a separate diagnosis due to unique aspects of therapy and prognosis. The symptoms of hypertrophic cardio-
myopathy are most commonly exertional limitation due to shortness of breath or chest pain. Some patients may develop syncope or near syncope, particularly during or following exertion (Ho and Seidman, 2006).
Most cases are associated with a specific genetic abnormality of the heart muscle cell. Patients often have a family history of this disease, but some cases result from sporadic mutations, with no previously affected family members (Ho and Seidman, 2006).
Right Heart Failure
Right heart failure most commonly accompanies left heart failure, either because it is also affected by the primary cause of heart failure, or because the chronic effects of left heart failure have led to gradual deterioration of the right ventricular function. However, some patients may have a clinical picture dominated by right heart failure. Congenital heart disease with or without prior surgery may be associated with abnormal function of the right side of the heart. This may be due in some cases to congenital underdevelopment of the right side of the heart, but more commonly develops later from chronic excess load on the right ventricle. Other conditions that can lead to the syndrome of right heart failure include tricuspid valve disease, pulmonary hypertension, pericardial disease, and atypical right heart response to left ventricular dysfunction, with less severe abnormality of left ventricular EF or diastolic dysfunction than required to meet criteria for systolic or diastolic heart failure as described above.
DIAGNOSIS OF HEART FAILURE
Careful clinical evaluation is crucial to correctly diagnose and treat patients with symptoms that may be due to heart failure. The term heart failure can lead to confusion, because the heart is usually not truly “failing” to function, in which case death would result. Instead, the heart may provide an adequate blood flow for organs to function at rest but has limited capacity to increase output to meet the needs of the body during sustained activity and exertion. The most common criteria for diagnosing “systolic heart failure” include evidence that the pumping strength of the left ventricle is reduced (measured by the EF, normally 55 to 70 percent). In addition, the size of the heart is often enlarged. Both measurements are usually provided by echocardiography, but they can also be measured through other techniques that image the heart, such as nuclear imaging tests or magnetic resonance imaging (MRI).
Most patients with systolic heart failure do not detect any limitation until the EF is less than 40 percent. Some patients can live virtually normal lives with EFs as low as 20 percent, but most patients with disease this
advanced have major activity limitations (Cohn et al., 1993; Levy et al., 2006; Stevenson et al., 1995). When the pressures in the heart are severely elevated at rest or the blood flow is severely reduced, heart failure is occasionally demonstrated by direct measurement of these pressures and flows during cardiac catheterization.
Diastolic heart failure remains challenging to diagnose from objective tests. Many criteria from echocardiographic assessment relate to how the heart relaxes; current debate remains about which are the most sensitive and specific. Different echocardiographic laboratories will concentrate on different measurements, yet most will describe diastolic dysfunction as mild, moderate, or severe.
Hypertrophic cardiomyopathy is also diagnosed by echocardiography. The unusual thickness of the ventricular muscle is a feature. In addition, about one in three patients may demonstrate some obstruction to flow through the left ventricle (“obstructive cardiomyopathy”).
Currently, the most common and appropriate diagnosis and evaluation of a patient with heart failure or cardiomyopathy includes a comprehensive echocardiogram with Doppler flow studies, because it determines not only severity of disease as measured by ventricular EF, but also how the heart fills and relaxes, which chambers are involved, and whether abnormalities of heart muscle texture, heart valves, or pericardium are present. The comprehensive nature of this test is important because patients with heart failure often present with multiple cardiac impairments (Hunt et al., 2005). Other useful tests to determine the severity of impairment or cardiac abnormality include radionuclide ventriculography or MRI (Hunt et al., 2005). In addition, a blood test showing markedly elevated levels of natriuretic peptides (e.g., BNP, NT-proBNP) supports the diagnosis of heart failure, and these levels are often elevated in patients with symptomatic heart failure, whether it is with systolic heart failure (heart failure with low EF) or diastolic heart failure (heart failure with preserved EF). Normal values of these peptides vary in relation to age, gender, race, and body weight.
TREATMENT
The recommended treatment guidelines for heart failure are designated according to stages of symptoms and apply primarily to patients with systolic heart failure (Hunt et al., 2005). Medical therapy for patients with symptoms of systolic heart failure includes angiotensin-converting enzyme inhibitors or angiotensin II receptor antagonists, beta adrenergic blocking agents, and diuretics as needed to control fluid retention, which may entail multiple changes and use of more than one diuretic. Digoxin, hydralazine, and nitrate combinations or nitrates alone, and aldosterone may also be indicated in patients with symptoms that persist. Anticoagulation is in-
dicated for some patients who are at particular risk for thromboembolic events. Implantable defibrillators may be indicated to decrease the risk of sudden death for some patients. Cardiac resynchronization therapy is a special form of pacing that may improve function in some patients with dysynchrony in the heart contraction pattern. Recurrent tachyarrhythmias may be treated with antiarrhythmic agents.
The recommended regimens for systolic heart failure may improve heart function as measured by increase in the EF and/or decrease in left ventricular enlargement. In general, the maximal impact of these therapies to improve objective cardiac function and exercise capacity may not be realized for 6 months or longer. Evaluation ideally would be deferred until after an adequate time for response to the appropriate medical regimen.
For diastolic heart failure, there are no specific treatment recommendations that are based on evidence from randomized trials. However, therapy should include vigorous efforts to control hypertension, usually with multiple drugs. Fluid retention should be treated aggressively with diuretics, which should be adjusted regularly to maintain fluid balance (Zile and Nappi, 2000).
For hypertrophic cardiomyopathy, therapy is guided primarily by control of the symptoms of exertional dyspnea and chest pain. Beta-blockers and calcium channel blockers are the mainstay of therapy, with occasional use of disopyramide in patients whose symptoms are not controlled with other agents. In patients who have specific risk factors for sudden death with hypertrophic cardiomyopathy, use of implantable defibrillators may be considered (Ho and Seidman, 2006).
The therapy of right heart failure usually includes high doses of diuretics to reduce signs of fluid retention. Therapy with diuretics does not treat the underlying cause of the heart failure, but is a key part of the medical regimen to minimize the impairment and discomfort caused by swelling of the legs or abdomen. The treatment regimen may also include therapies directed when feasible to treat the underlying cause of the right heart failure.
DISABILITY
Assessing Functional Capacity for Disability
Functional capacity is often described using the New York Heart Association Functional Classification System as a measure of symptom severity. Class I indicates no symptoms during vigorous activity. Class II includes symptoms with moderate activity that is greater than required for daily life (e.g., vigorous recreation). Class III symptoms are those occurring with less than the usual daily level of activity. Class IV symptoms occur at rest or
with minimal exertion, such as dressing or walking from room to room. These four classes reflect subjective assessment by health care providers. The major information used for this classification is the description of symptoms by the care provider or patient. These are dynamic classifications, and patients can cross back and forth frequently between different levels of symptoms. When used to describe the severity of disease, the classifications are usually meant to describe the symptom level most of the time, rather than at the best or worst times.
Patients can have a diagnosis of heart failure without being disabled from working. As discussed above, the term heart failure is an unfortunate one because many patients included in this definition have mild or moderate symptoms that do not prevent gainful employment. It is widely recognized that symptoms and functional capacity in heart failure are not easily predicted or explained by the degree of impairment in cardiac function, such as reduction of left ventricular EF or increase in left ventricular size. Although these measurements are important for the diagnosis of cardiac impairment leading to heart failure, they rarely constitute sufficient basis to show disability. In extreme cases, very severe reduction of left ventricular EF, such as 20 percent or less, can be considered a surrogate for limited function and survival, and thus could warrant decision of disability without functional assessment (Cohn et al., 1993; Levy et al., 2006; Stevenson et al., 1995).
The most direct assessment of ability to work is the ability to perform physical exertion. A patient’s ability to perform activities of daily living, work, and vigorous exertion requires the integration of cardiovascular, pulmonary, and circulatory systems (Arena et al., 2007; Fleg et al., 2000; Wilson et al., 1995). Exercise testing has been consistently demonstrated and widely accepted to be practical and safe for most patients with chronic heart failure (Balady et al., 2010). Either treadmill testing or bicycle exercise testing can be performed. The advantage of treadmill testing is that walking is a more familiar activity to many than bicycling; the advantage of bicycle testing is that workload can be controlled more strictly, and patients who have gait instability may feel more comfortable sitting on the bicycle. Treadmill testing is more common, and the protocol and listing criteria will be discussed as for a treadmill test. Bicycle testing may lead to slightly lower estimation of overall exercise capacity, but the difference is not believed to be sufficient to justify having different listing criteria for bicycle and treadmill exercise performance.
The evaluation of exercise capacity recognizes that “the subjective quality of symptoms can detract from objective, reproducible, and sensitive measurements. Personality, mood, culture, and idiosyncratic vacillations as well as cognition, literacy, and socioeconomics are among the factors that impact on these measures” (Balady et al., 2010:200). Measurements based on peak heart rate as a proportion of predicted maximal heart rate are now
“confounded by beta-blockers and other heart-rate limiting medications,” which include antiarrhythmic agents (Balady et al., 2010:200). Thus, “determination of peak VO2 during a symptom-limited treadmill or bicycle cardiopulmonary exercise testing (CPX) is the most objective method to assess exercise capacity in heart failure patients…. It is a useful test to determine the severity of the disease and to help to determine whether heart failure is the cause of exercise limitation” (Balady et al., 2010:201).
The level of peak VO2 less than 15 ml/kg is proposed as the listing criterion for exercise impairment, which is consistent with the level for pulmonary disability, also 15 ml/kg/min. Although there is some consideration of using a percentage of predicted peak oxygen consumption for age and gender, the level of exercise limitation at which no meaningful work can be performed is presumably an absolute rather than a relative number and should be independent of age and gender. It is also consistent with the general level of metabolic equivalents of task (METs), the factor by which resting energy demands are increased to do levels of work as required by most work with modest movement. Disability would be considered adequately documented if patients have objective evidence of limitation that prevent them from reaching oxygen consumption of 15 ml/kg/min by the end of exercise. An objective documentation of circulatory limitation is the respiratory exchange ratio (RER) of at least 1.1 or more, which indicates that anaerobic metabolism has been achieved, and that oxygen delivery has become inadequate to meet metabolic demands during exercise. An RER of 1.1 is the criterion for adequate exercise effort as recommended by the American Heart Association (AHA) 2010 guidelines on CPX in adults (Balady et al., 2010).
For patients performing symptom-limited exercise testing without gas exchange to measure peak VO2, the level is defined by the estimated MET level of the exercise. The estimated MET level selected for patients without peak VO2 measurement has generally been 5 or less. The specific experience with heart failure patients is that the actual peak oxygen consumption is usually significantly lower than that estimated from the MET level of the exercise stage. Thus a measured peak VO2 of 15 ml/kg/min and the estimated MET level of 5 are not inconsistent.
Strengths and Limitations of Current Listing Criteria for Heart Failure
The Social Security Administration (SSA) currently reviews applicants’ disability claims for heart failure based on specific diagnostic and clinical criteria. The current listing (see Box 5-1) incorporates measures of functioning into the evaluation, where the claimant is limited in activities of daily living and unable to perform an exercise test at the specified level. These criteria are required in combination.
|
BOX 5-1 Current Listing for Chronic Heart Failure 4.02 Chronic heart failure while on a regimen of prescribed treatment, with symptoms and signs described in 4.00D2.The required level of severity for this impairment is met when the requirements in both A and B are satisfied.
AND
SOURCE: SSA, 2008. |
The current listing criteria as described in Box 5-1 include and require evidence of each of two aspects of disease: (1) objective measurement of cardiac abnormality and (2) evidence of limited functional capacity. This construct has been useful and remains the basis of recommendations for a revised listing. However, review of the specific criteria for (1) and (2) suggested several areas for which newer definitions may be more applicable to contemporary populations and to current testing and therapies.
The current criteria of EF and left ventricular dimension for evaluation of systolic dysfunction remain highly relevant to assessment. However, diastolic dysfunction is a much more heterogeneous condition in terms of population, assessment, and clinical limitation. The requirement for increased wall thickness is neither sensitive nor specific for diastolic dysfunction. Newer measurements from echocardiography describe the ability of the heart to relax and fill.
Functional assessment in the current listing is severely limited by the previous position that exercise testing posed significant risk to patients with heart failure. The assessment of clinical limitation without exercise testing thus has been highly subjective. Newer experience, as reviewed in the American College of Cardiology (ACC)/AHA guidelines on cardiopulmonary exercise testing, suggests that exercise testing is safe in most patients with chronic heart failure in the absence of acute or recent clinical instability (Balady et al., 2010).
When available, exercise testing is the most helpful assessment. For patients in whom exercise tests can be performed, the current criterion of less than 5 METs is reasonable and can be estimated from the level of exercise performed. However, many reasons for not reaching that level of exercise are highly subjective, relying on patient descriptions of shortness of breath and fatigue. The complaint of palpitations can be replaced by the objective demonstration of arrhythmias, as all standard exercise tests are performed with electrocardiographic monitoring. However, frequent and consecutive premature ventricular contractions (PVCs), even brief runs of up to 10 beats of nonsustained ventricular tachycardia, are common in patients with heart failure and reduced EF; they are not generally considered criteria for exercise limitation except as discussed separately in the arrhythmia disability listing (see Chapter 13). The complaint of chest pain is subjective and should be substantiated by electrocardiographic evidence of myocardial ischemia. (The only population in which chest pain without demonstrable ischemia is generally acceptable as an indication of exercise limitation is hypertrophic cardiomyopathy, which as suggested above might be more conveniently considered separately.) The criterion of a fall in blood pressure is objective and a reasonable documentation of exercise limitation, but unusual in patients with heart failure. The previous listing criteria included exercise limited by signs of inadequate cerebral perfusion such as
ataxic gait and mental confusion; these signs would raise concerns about other diagnoses, as they do not commonly occur during exercise testing, even in severely impaired patients with heart failure.
The current functional criterion of repeated hospitalizations is useful to characterize those patients whose heart failure is of a fluctuating and episodic nature. Although recurrent admissions can result from irregular compliance with medications and outpatient follow-up, they can occur even in patients with optimal management. Patients with severe disease and multiple hospitalizations are also likely to have objective impairment of exercise performance, but either would seem reasonable criteria for disability. Information regarding the frequency with which individual functional limitations are used as criteria is not currently accessible.
CONCLUSIONS AND RECOMMENDATIONS
After review of the current medical literature and related treatment and practice guidelines from ACC and AHA, the committee determined the chronic heart failure listing should be revised to include separate evaluation criteria for the following related conditions: chronic heart failure (both systolic and diastolic heart failure), hypertrophic cardiomyopathy, and right heart failure. The general diagnosis of cardiomyopathy (other than hypertrophic) would not have its own listing criteria, but would be evaluated according to one of these other three conditions.
The conceptual framework of the current listing criteria includes the determination of both (A) a measured anatomic or hemodynamic abnormality of cardiac function and (B) a demonstrated impairment of patient function, either chronically or intermittently. The requirement for both is consistent with the well-documented variability in the relationship between cardiac impairment and physical functioning in heart failure and cardiomyopathy.
RECOMMENDATION 5-1. Retain the current framework of listing 4.02 chronic heart failure, requiring both (A) an objective cardiac abnormality, and (B) a functional limitation. This framework would apply to each of the following: systolic heart failure, diastolic heart failure, hypertrophic cardiomyopathy, and right heart failure.
Systolic Heart Failure (Figure 5-1)
The criteria for systolic heart failure should include:
-
An objective cardiac abnormality demonstrated by a left ventricular ejection fraction of 30 percent or less (or an ejection
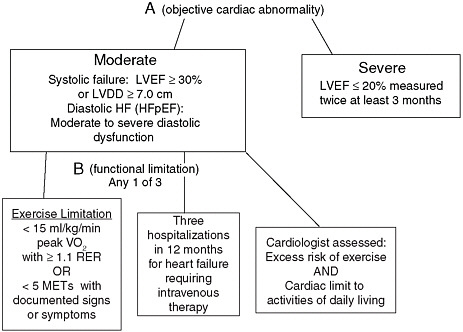
FIGURE 5-1 Recommended listing-level criteria for systolic and diastolic heart failure.
NOTES: HF = heart failure; LVDD = left ventricular diastolic dysfunction; LVEF = left ventricular ejection fraction; METs = metabolic equivalents of task; RER = respiratory exchange ratio.
-
fraction of a systemic ventricle in congenital heart disease without an anatomic, systemic left ventricle), or a left ventricular diameter of 7 cm or greater,
AND
-
A functional abnormality demonstrated by one of three criteria:
-
Exercise testing (see Recommendation 5-2); or
-
Cardiologist-assessed excessive risk of exercise testing and cardiac limit to activities of daily living; or
-
Three hospitalization-equivalent events in the past 12 months.
-
Diastolic Heart Failure (Figure 5-1)
The criteria for diastolic heart failure should include:
-
An objective cardiac abnormality demonstrated by moderate or severe diastolic dysfunction, usually indicated by echocardiography,
AND
-
A functional abnormality can be demonstrated by one of three criteria:
-
Exercise testing (see Recommendation 5-2); or
-
Cardiologist-assessed excessive risk of exercise testing and cardiac limit to activities of daily living; or
-
Three hospitalization-equivalent events in the past 12 months.
-
The criterion of three hospitalizations is anticipated to be met more frequently for diastolic heart failure than it is for the more common systolic heart failure, for which the exercise limitation may be more commonly met.
Hypertrophic Cardiomyopathy (Figure 5-2)
The criteria for hypertrophic cardiomyopathy should include:
-
An objective cardiac abnormality demonstrated by a left ventricular or septal wall thickness greater than 15 mm in the absence of another known cause for left ventricular hypertrophy (e.g., hypertension, aortic valve disease),
AND
-
A functional abnormality demonstrated by one of three criteria:
-
Exercise testing (see Recommendation 5-2); or
-
Cardiologist-assessed excessive risk of exercise testing and cardiac limit to activities of daily living; or
-
Three hospitalization-equivalent events in the past 12 months.
-
Due to the characteristics of their cardiac function or symptoms, some patients with hypertrophic cardiomyopathy may be considered too high
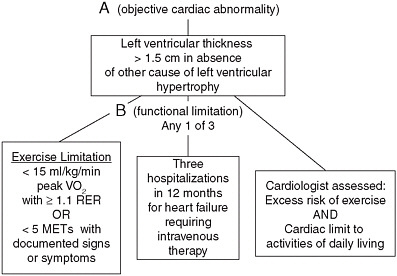
FIGURE 5-2 Recommended listing-level criteria for hyptertrophic cardiomyopathy.
NOTE: METs = metabolic equivalents of task; RER = respiratory exchange ratio.
risk for exercise testing, particularly those with very thick ventricular thicknesses or demonstration of a large pressure gradient (“obstruction”) inside the left ventricle. If these patients are identified by their physicians as having severe symptoms and being at excessive risk for exercise testing, they will meet the functional limitation criteria.
-
Hypertrophic cardiomyopathy excluded from exercise testing with cardiologist documentation of the following:
-
Major risk associated with exercise testing (see Recommendation 5-2), and
-
Symptoms from hypertrophic cardiomyopathy resulting in limitation of ability to independently perform activities of daily living.
-
Right Heart Failure (Figure 5-3)
The criteria for right heart failure should require both an objective cardiac diagnosis implicated as a cause of right heart failure and clinical evidence of functional limitation manifest as severe systemic venous congestion:
-
An objective cardiac abnormality demonstrated by one of the following:
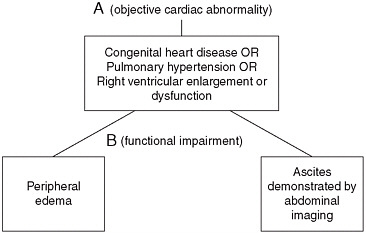
FIGURE 5-3 Recommended listing-level criteria for right heart failure.
-
-
Congenital heart disease; or
-
Pulmonary hypertension; or
-
Right ventricular enlargement or dysfunction.
-
AND
-
Functional abnormalities meeting criteria for right heart failure:
-
Systemic venous congestion despite chronic diuretic therapy, assessed twice with at least 3 months in between, causing either
-
Peripheral edema to the knee or above; or
-
Severe ascites documented by abdominal imaging study.
-
-
Some patients may have primary peripheral edema that can become severe, particularly in the presence of obesity, without an underlying cause of heart failure. They are not to be included here. Intra-abdominal fluid accumulation (ascites) sufficient for listing would have to be documented by abdominal imaging, because it cannot reliably be distinguished from abdominal adiposity.
Exercise Testing
Based on its review of the 2010 ACC/AHA exercise guidelines, the committee concludes that the listing criteria to evaluate claimants for dis-
ability should include objective measures performed during exercise tests. These measures are considered safe and reasonable for most patients with heart failure. The listing criteria accordingly should reflect the updated guidelines for exercise testing.
RECOMMENDATION 5-2. Revise the exercise criteria to reflect the current acceptability of exercise testing as safe in heart failure and the objective measurements that can now be performed during exercise testing. Limitation is defined as a standard treadmill test (or bicycle test) performed at a workload equivalent to one of the following criteria:
-
Less than 15 ml/kg/min peak VO2/(oxygen consumption) on cardiopulmonary exercise test; or
-
Less than 5 metabolic equivalents of task if using standard treadmill test without gas exchange.
The applicant may satisfy the exercise test criteria for disability listing by failing to reach one of these limits due to documented evidence of cardiac limitation. This is easiest with the CPX test, which requires less subjective interpretation.
On cardiopulmonary exercise testing, if the RER is greater than or equal to 1.1, the presence of anaerobic metabolism and inadequate oxygen delivery to the exercising muscles can be considered present, which can indicate a cardiovascular limitation. The reason for including the cardiopulmonary exercise testing is that the simultaneous measurement of gas exchange enables determination of the degree of effort and cardiac limitation. This makes it easier and more objective to exclude intentional underperformance on standardized exercise testing.
If standard exercise testing without gas exchange is the test information provided, objective evidence of true cardiac limitation can be as follows:
-
Signs may include fall in blood pressure, evidence of myocardial ischemia from ST-segment depression or elevation, or ventricular tachycardia. These will rarely be present. Otherwise, termination of a standard treadmill test would need to be validated by subjective evidence from the evaluator that exercise was limited by cardiac causes.
-
Severe symptoms may include angina, dyspnea, and claudication.
Signs and symptoms should be documented as related to heart failure, and not an unrelated condition.
The occurrence of frequent or consecutive PVCs are common at rest
and during exercise in patients with heart failure and should not be considered adequate reason to terminate the exercise test unless accompanied by other evidence of exercise limitation. Prolonged runs of ventricular tachycardia as the basis of termination of exercise testing would be evaluated under the arrhythmia disability criteria in most cases. The occurrence of one such episode, even if specific therapy were required for termination, should trigger specific therapy of the arrhythmia and subsequent repeat exercise testing if a person is seeking disability under the heart failure criteria.
Severe Reduction of Ejection Fraction
An EF of 20 percent or less represents a decrease to less than one-third of normal heart pumping function. This severe level of impairment is generally associated with marked functional limitation and limitation in survival. However, this impairment occasionally can be seen in conjunction with another severe condition that is reversible, such as a severe viral infection or bacterial sepsis. The committee proposed that the EF of 20 percent or less be documented twice with at least 3 months intervening to allow time for improvement of any reversible process. Although little systematic evidence documents patients’ functional status with very low EF, the committee’s expert judgment is that patients with an EF of 20 percent or less would most likely be severely impaired and therefore should be considered for listing-level disability.
The committee recognizes that some individuals, particularly those in their teens or twenties, will be able to work and may even be asymptomatic. This means that a listing requiring only an EF of 20 percent or less will not be 100 percent specific. As an administrative expedient, however, this recommended listing would be specific enough and the number of worthy claimants who would be quickly and easily allowed outweighs the probability that a few individuals who could work would be allowed. Asymptomatic individuals are most likely to be young. They are likely to continue working, even if they are aware they have a very low EF, because the level of benefits they could anticipate receiving would be very low relative to what they could earn by continuing to work. The listing could include a criterion that a claimant must be symptomatic as well as have an EF of 20 percent or less, but this would require adjudicators to determine from the medical records whether the claimant is symptomatic. Adding a second criterion of functional limitation, which is more subjective to document, would com plicate the application of what could be a simple listing.
RECOMMENDATION 5-3. Add an additional listing route in which the objective cardiac abnormality of a left ventricular ejection fraction of 20 percent or less, documented twice with at least 3 months intervening, is sufficiently severe that demonstration of functional limitation is not needed to meet the listing.
REFERENCES
Allen, L. A., J. E. Yager, M. J. Funk, W. C. Levy, J. A. Tulsky, M. T. Bowers, G. C. Dodson, C. M. O’Connor, and G. M. Felker. 2008. Discordance between patient-predicted and model-predicted life expectancy among ambulatory patients with heart failure. Journal of the American Medical Association 299(21):2533–2542.
Arena, R., J. Myers, M. A. Williams, M. Gulati, P. Kligfield, G. J. Balady, E. Collins, and G. Fletcher. 2007. Assessment of functional capacity in clinical and research settings: A scientific statement from the American Heart Association Committee on Exercise, Rehabilitation, and Prevention of the Council on Clinical Cardiology and the Council on Cardiovascular Nursing. Circulation 116:329–343.
Balady, G. J., R. Arena, K. Sietsema, J. Myers, L. Coke, G. F. Fletcher, D. Forman, B. Franklin, M. Guazzi, M. Gulati, S. J. Keteyian, C. J. Lavie, R. Macko, D. Mancini, and R. V. Milani, on behalf of the American Heart Association Exercise, Cardiac Rehabilitation, Prevention Committee of the Council on Clinical Cardiology, Council on Epidemiology Prevention, Council on Peripheral Vascular Disease, and Interdisciplinary Council on Quality of Care Outcomes Research. 2010. Clinician’s guide to cardiopulmonary exercise testing in adults. A scientific statement from the American Heart Association. Circulation 122:191–225.
Cohn, J. N., G. R. Johnson, R. Shabetai, H. Loeb, F. Tristani, T. Rector, R. Smith, and R. Fletcher. 1993. Ejection fraction, peak exercise oxygen consumption, cardiothoracic ratio, ventricular arrhythmias, and plasma norepinephrine as determinants of prognosis in heart failure. The V-HeFT VA Cooperative Studies Group. Circulation 87(6 Suppl): V15–V16.
Colucci, W. S., and E. Braunwald. 1995. Heart failure: Cardiac function and dysfunction. In Atlas of heart diseases, 4th ed., edited by W. S. Colucci. Philadelphia, PA: Blackwell Science. Pp. 128–142.
Fleg, J. L., I. L. Piña, G. J. Balady, B. R. Chaitman, B. Fletcher, C. Lavie, M. C. Limacher, R. A. Stein, M. Williams, and T. Bazzarre. 2000. Assessment of functional capacity in clinical and research applications: An advisory from the Committee on Exercise, Rehabilitation, and Prevention, Council on Clinical Cardiology, American Heart Association. Circulation 102:1591–1597.
Gibbons, R. J., G. J. Balady, J. T. Bricker, B. R. Chaitman, G. F. Fletcher, V. F. Froelicher, D. B. Mark, B. D. McCallister, A. N. Mooss, M. G. O’Reilly, W. L. Winters Jr., E. M. Antman, J. S. Alpert, D. P. Faxon, V. Fuster, G. Gregoratos, L. F. Hiratzka, A. K. Jacobs, R. O. Russell, and S. C. Smith Jr. 2002. ACC/AHA 2002 guideline update for exercise testing. Journal of the American College of Cardiology 40:1531–1540.
Ho, C. Y., and C. E. Seidman. 2006. A contemporary approach to hypertrophic cardiomyopathy. Circulation 113(24):e858–e862.
Hunt, S. A., W. T. Abraham, M. H. Chin, A. M. Feldman, G. S. Francis, T. G. Ganiats, M. Jessup, M. A. Konstam, D. M. Mancini, K. Michl, J. A. Oates, P. S. Rahko, M. A. Silver, L. W. Stevenson, and C. W. Yancy. 2005. Guideline update for the diagnosis and management of chronic heart failure in the adult. Circulation 112:e154–235.
Levy, W. C., D. Mozaffarian, D. T. Linker, S. C. Sutradhar, S. D. Anker A. B. Cropp, I. Anand, A. Maggioni, P. Burton, M. D. Sullivan, B. Pitt, P. A. Poole-Wilson, D. L. Mann, and M. Packer. 2006. The Seattle heart failure model: Prediction of survival in heart failure. Circulation 113(11):1424–1433.
Lloyd-Jones, D., R. J. Adams, T. M. Brown, M. Carnethon, S. Dai, G. De Simone, T. B. Ferguson, E. Ford, K. Furie, C. Gillespie, A. Go, K. Greenlund, N. Haase, S. Hailpern, P. M. Ho, V. Howard, B. Kissela, S. Kittner, D. Lackland, L. Lisabeth, A. Marelli, M. M. McDermott, J. Meigs, D. Mozaffarian, M. Mussolino, G. Nichol, V. L. Roger, W. Rosamond, R. Sacco, P. Sorlie, R. Stafford, T. Thom, S. Wasserthiel-Smoller, N. D. Wong, J. Wylie-Rosett, and on behalf of the American Heart Association Statistics Committee Stroke Statistics Subcommittee. 2010. Heart disease and stroke statistics—2010 update: A report from the American Heart Association. Circulation 121(7):e46–e215.
Redfield, M. M., S. J. Jacobsen, J. C. Burnett Jr., D. W. Mahoney, K. R. Bailey, and R. J. Rodeheffer. 2003. Burden of systolic and diastolic ventricular dysfunction in the community: Appreciating the scope of the heart failure epidemic. Journal of the American Medical Association 289(2):194–202.
SSA (Social Security Administration). 2008. Listing of impairments—Adult listings (Part A). Disability evaluation under Social Security (Blue Book). http://www.socialsecurity.gov/disability/professionals/bluebook/AdultListings.htm (accessed July 22, 2010).
Stevenson, L. W., G. Couper, B. Natterson, G. Fonarow, M. A. Hamilton, M. Woo, and J. W. Creaser. 1995. Target heart failure populations for newer therapies. Circulation 92(9 Suppl):II 174–181.
Wilson, J. R., G. Rayos, T. K. Yeoh, and P. Gothard. 1995. Dissociation between peak exercise oxygen consumption and hemodynamic dysfunction in potential heart transplant candidates. Journal of the American College of Cardiology 26(2):429–435.
Zile, M. R., and J. Nappi. 2000. Diastolic heart failure. Current Treatment Options in Cardiovascular Medicine 2(5):439–450.




















