2
Current Concepts in HIV/AIDS
EVOLUTION OF THE EPIDEMIC
The first cases of AIDS were reported in the United States in 1981 (CDC, 2001). At that time the average life expectancy for a person diagnosed with AIDS was 6 months (Satriano et al., 2005). Subsequent advances in treatment, particularly the use of combination antiretroviral therapy, have transformed HIV/AIDS into a chronic, manageable condition. A 35-year-old person diagnosed with HIV infection has an estimated life expectancy of 32 years, depending on nadir CD4 (Hogg et al., 2008). In the setting of these successes, however, patients with HIV infection still face many challenges.
As treated patients live longer, illnesses typically associated with an aging population, such as cardiovascular disease, osteoporosis, and diabetes, are being seen at increased frequency. Some of these conditions may be related to treatment, and others may be related to the chronic inflammatory state associated with HIV infection. Furthermore, self-management and adherence to the sometimes complex treatment regimens required by HIV/AIDS represent an ongoing and lifelong challenge for many patients. Finally, the remarkable advances in combination antiretroviral therapy are of little value to those who are unable to access state-of-the-art care, including treatments for conditions associated with HIV infection.
In 2008, the Centers for Disease Control and Prevention (CDC) estimated that more than 56,000 incident HIV cases occurred in the United States in 2006 (the most recent year data are available) (Hall et al., 2008). As mortality rates have declined due to improvements in treatment, the
cumulative number of people living with HIV/AIDS has steadily increased. In 2006, the CDC estimated approximately 1.1 million people were living with HIV/AIDS in the United States (CDC, 2008). Over the past decade, the prevalence of HIV/AIDS in the United States has risen disproportionately among some racial and ethnic groups. Slightly more than 50 percent of HIV-positive individuals in the United States are African Americans, and 18 percent are Hispanic/Latino (CDC, 2007). In addition, adolescents and young adults account for an increasing proportion of all new cases of HIV infection. While 29 percent of people living with HIV/AIDS in the United States are between the ages of 13 and 29, this age group accounts for 34 percent of all new cases of HIV (Hall et al., 2008).
Pathogenesis of HIV
HIV damages the immune system by (1) depleting a critical element of the body’s immune system, the CD4+ T-cell pool (CD4 cells), and (2) causing a state of generalized activation and inflammation of the immune system. As a consequence of these two changes, patients with HIV infection are at an increased risk of other infectious diseases and virus-associated cancers as well as an array of noninfectious diseases. The diagnoses and clinical conditions associated with HIV infection are listed in Table C-1 (see Appendix C). In addition to the secondary complications of HIV infection, the virus is capable of directly infecting the nervous system, leading to neurocognitive dysfunction (Grant, 2008).
Comorbidities of HIV
Although the list of opportunistic infections and neoplasms associated with HIV infection is well characterized and has been relatively stable since the beginning of the epidemic, a new set of serious complications of HIV infection has emerged in recent years (Table C-1). These conditions have replaced opportunistic infections as the leading cause of death in patients with HIV infection (Buchacz et al., 2010). It is believed that the ongoing immune activation and inflammation characteristic of HIV infection in patients with or without treatment and manifest by elevated levels of interleukin-6 and D-dimer is responsible, at least in part, for these problems. It has been suggested that HIV infection leads to an accelerated senescence of multiple organ systems, or premature aging. Complicating this picture is the fact that many of these same problems are seen as side effects of some medications used to treat HIV infection.
The presence of comorbidities such as major depressive disorder, other mental disorders, or substance abuse may affect neurocognition in HIV-positive individuals. Cognitive limitations may impair many patients and
range from marginal to severe (Valcour et al., 2004; Vance and Struzick, 2007). Cognitive dysfunction can have a major impact on a patient’s ability to maintain activities of daily living, remain employed, or engage in other regular activities such as driving.
Common comorbidities among HIV-infected populations with and without neurocognitive disorder include psychiatric and behavioral disorders, which can lead to functional impairment and potential disability. Within psychiatric disorders, major depression, delirium, and anxiety are seen in 25 to 50 percent of individuals living with HIV infection (Pence et al., 2006). Studies have suggested the risky behaviors associated with bipolar disorder or schizophrenia may lead to higher prevalence rates of HIV infection among affected populations. These disorders are somewhat less common than major depression, but if untreated may lead to impairment and disability (Vlassova et al., 2009). Behavioral disorders include untreated drug dependence, which can lead to impairment and disability. Injection drug use can lead to immune suppression and increased HIV transmission (Fama et al., 2009; Vlassova et al., 2009). Cocaine, amphetamine, and alcohol abuse can also lead to increased immune suppression (Fama et al., 2009), decreased adherence, and poor virologic outcomes (Vlassova et al., 2009). Psychological reactions to negative life experiences can lead to posttraumatic stress disorder, seen in 13 to 20 percent of individuals living with HIV infection. Posttraumatic stress disorder cooccurs with major depression and substance use disorders in 25 to 50 percent of individuals living with HIV infection and is associated with lower CD4 counts and incompletely suppressed viral load (Vranceanu et al., 2008).
HIV and Aging
A growing number of older adults are affected by HIV/AIDS, including those who have aged with the disease due to advances in treatment as well as individuals who have been infected later in life. In 2007, the largest number of new infections occurred among those ages 40 to 44. Furthermore, according to CDC estimates for 2007, 28 percent of HIV-infected adults were over age 45. New aspects of chronic HIV infection will likely become apparent as the HIV/AIDS population ages.
Older age is associated with more rapid progression of HIV infection. Research has linked age as an independent prognostic factor for patients with HIV (Egger et al., 2002). Patients older than age 50 who began combination antiretroviral therapy showed higher clinical progression of disease, including higher rates of mortality, compared to younger patients beginning treatment (Kirk and Goetz, 2009). Furthermore, untreated older adults progress to AIDS and death much faster than younger individuals. In addition, older adults may experience longer-term effects from the virus,
treatment, and comorbidities compared to younger people. Researchers and treating clinicians may need to consider differentiating between those recently infected at an older age and those who were infected at a younger age and have grown older with the disease due to improvements in therapy (Stoff et al., 2004). Older adults may be diagnosed later in disease progression, as many age-related illnesses can mimic HIV-associated symptoms (Kirk and Goetz, 2009).
MANAGEMENT OF HIV/AIDS
In the beginning of the epidemic, researchers and clinicians struggled to understand the nature of the immune defect and the identity of the causative agent. With the discovery of HIV as the causative agent of AIDS, work proceeded rapidly to identify and develop inhibitors of critical steps in the viral life cycle (see Figure 2-1). In 1987, monotherapy with the nucleoside analogue zidovudine (ZDV, also known as azidothymidine [AZT]) became the first licensed treatment for patients with AIDS. Although this treatment saved lives, its effects were relatively short lived because the virus was able to develop resistance to the single agent. The licensure of ZDV was rapidly followed by the development of a series of additional nucleoside analogues, followed by the introduction of nonnucleoside reverse transcriptase inhibitors, protease inhibitors, fusion inhibitors, entry inhibitors, and integrase inhibitors.
Used in combination, these drugs lead to substantial suppression of HIV replication for extended periods of time. Despite this success, these drugs are not able to eradicate the virus and patients currently must remain on therapy for the remainder of their lives. Even brief lapses in therapy can lead to increases in viral replication and subsequent immune system and other end organ damage.
The overall goal of combination antiretroviral therapy (also known as highly active antiretroviral therapy or HAART) is to limit the ability of the virus to replicate and thus limit its ability to damage the host. Following the widespread use of combination antiretroviral therapy in the United States in the mid-1990s, the mortality rate due to AIDS declined steeply (see Figure 2-2). Continued success in the future will depend to a large extent on the ability to engage HIV-infected persons in care early in the course of their infection and to support them in adhering to treatment regimens. Successful treatment regimens begin with early diagnosis of HIV infection. Research has shown that patients have better outcomes when they begin combination antiretroviral therapy regimens before their CD4 counts decline to fewer than 350 cells/mm3, compared to patients who begin treatment at a more advanced stage of disease (Egger et al., 2002). A broader discussion of CD4 count and prognosis is included in Chapter 3.
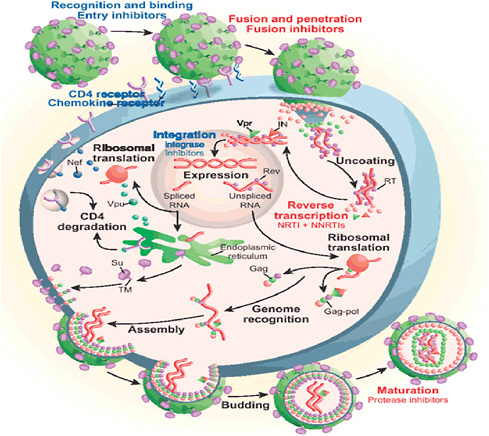
FIGURE 2-1 Replication cycle of HIV with stages to initiate treatment.
NOTE: NNRTI = nonnucleoside reverse transcriptase inhibitor; NRTI = nucleoside reverse transcriptase inhibitor.
SOURCE: Reprinted by permission from Macmillan Publishers Ltd. Bennett and Rose, 2010.
The high rate of viral production (109 particles/day) coupled with the error rate of the critical viral enzyme reverse transcriptase (1/genome) means that an enormous number of viral variants are produced each day in an infected individual. Although many of these variants are likely to be defective, only a single virus with a mutation conferring resistance to a specific antiretroviral agent can lead to a population of resistant virus. The possibility that a population of resistant virus will be established is minimized by the use of a combination of antiretroviral agents. In the setting of multiple drugs, a series of mutations is required for resistance to emerge. Resistance was a major problem in treating patients with HIV
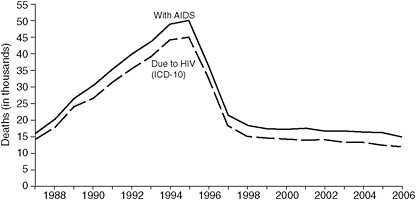
FIGURE 2-2 Estimated numbers of U.S. AIDS case reports and death certificates in which HIV disease was selected as the underlying cause of death, 1987–2006.
SOURCE: Centers for Disease and Control and Prevention, 2007.
infection when single-drug or dual-drug therapy was used. Now it is less of a problem with the use of potent combination regimens (Bangsberg et al., 2000, 2003, 2004). Although only some viruses are resistant to all current antiretroviral drugs, it is possible that such strains will become more prevalent in the future. CD4 count and viral load are currently the leading laboratory indicators of the progression of HIV infection (see Chapter 3). Other independent indicators include hemoglobin and direct markers of inflammation and coagulation.
Treatment Side Effects
Although they save lives, antiretrovirals have a variety of side effects, some of which can be permanently disabling. Further complicating the picture is the fact that some side effects from treatment resemble manifestations of HIV/AIDS; as a result, clinicians may have trouble determining whether the root cause of the symptom is the antiretroviral or the underlying disease. Given the array of antiretrovirals currently available (see Table C-2 for a list of current HIV/AIDS antiretrovirals and potential side effects), one is often able to switch to an alternative regimen to attempt to distinguish a side effect from a disease manifestation. Some of the formerly common side effects from treatment are now relatively rare. For example, pancreatitis and kidney stones are no longer common complications of HIV/AIDS therapy.
Under certain circumstances, patients continue to be disabled by HIV/ AIDS, despite advances in treatment and the opportunity to choose alternate medications (Klimas et al., 2008). Although currently the overall out-
look is much brighter for patients with HIV infection than it was in 1981, not all patients respond to medication, including those:
-
Who do not have access to state-of-the-art treatment;
-
Who are infected with multidrug-resistant viruses;
-
Who are unable to adhere to treatment regimens; or
-
Who have a relatively small number of usually irreversible complications, such as progressive multifocal leukoencephalopathy.
Furthermore, patients with comorbidities, such as depression, diabetes, and cardiovascular disease, must manage multiple medical therapies (Stoff et al., 2004). The resulting use of multiple medications may lead to further complications and drug interactions. Finally, individuals continue to enter medical care with advanced HIV disease (indicated by low CD4 counts) and suffer from an array of HIV-associated conditions that will likely result in disability for more than one year.
Medication Adherence
Adherence to antiretroviral therapy is widely accepted as crucial to successful outcomes. The level of adherence is reflected by both the suppression of plasma levels of HIV (Aloisi et al., 2002; Lanièce et al., 2003; Nieuwkerk and Oort, 2005) and the individual’s overall quality of life (Mannheimer et al., 2005). Nonadherence to combination antiretroviral therapy regimens, which is common among HIV-positive individuals, compromises clinical effectiveness and is an antecedent to the development of viral resistance (Abel and Painter, 2003; Aloisi et al., 2002; Chesney et al., 2000). It is also associated with increased risk of mortality (Lima et al., 2009). A systematic review of 72 developed-country studies and 12 developing-country studies identified barriers to adherence common to both settings, including fear of disclosure, substance abuse, forgetfulness, suspicion of treatment, complicated regimens, number of pills, quality of life, work and family responsibilities, falling asleep, and lack of access to medications (Mills et al., 2006).
IMPROVEMENTS IN FUNCTIONING AND CAPACITY TO RETURN TO WORK
In most cases, HIV/AIDS is no longer a near-term fatal disease. In these situations, following initiation and stabilization on therapy, patients with HIV infection may engage in a full range of activities, including employment (Goldman and Bao, 2004). Return to work is an important social and economic milestone for HIV-infected individuals and their families, and a
contribution to society as a whole (Hergenrather et al., 2005). Although no national employment statistics are available for people living with HIV/ AIDS in the United States, nearly 70 percent are between ages 25 and 49, the average working ages (CDC, 2008). In 2000, slightly more than 60 percent of working-age Americans with disabilities were employed full- or part-time, compared to nearly 80 percent of nondisabled Americans (U.S. Census Bureau, 2003).
Moving from disability status to employment leads to increased, if not complete, independence in the community. Individuals who maintain part- or full-time employment achieve improved physical and mental health due to their improved economic status. Psychological well-being may also improve due to individuals’ ability to engage in a standard activity deemed valuable by society (Braveman and Keilhofner, 2006).
For individuals with HIV/AIDS, the decision to work may be influenced by a number of factors. These factors may include personal beliefs that shape behaviors, either motivating return to work or impeding employment. The opinions of various groups, such as family, friends, or the treating physician, may also positively or negatively influence a person’s decision to work. Empirical research has not shown that any one of these groups’ opinions has a greater effect on the individual’s decision to work than another. Due to the myriad factors to consider when deciding to work, individuals welcome guidance from trusted social networks (Hergenrather et al., 2004).
In studies examining return-to-work patterns for people with HIV/ AIDS, the most often cited motivation to consider employment was financial need (Arns et al., 2004; Martin et al., 2006; Rabkin et al., 2004). Sociodemographic indicators are also strongly associated with increased likelihood of returning to work. For example, men are more likely to work than women, and younger individuals are more likely to work than older adults (Brooks et al., 2004; Burns et al., 2006). Other motivators leading individuals to consider returning to the workplace include positive support from family and friends, and, in some cases, explicit encouragement to seek employment (Hergenrather et al., 2004; Martin et al., 2004). Common barriers to employment include lower socioeconomic status, less education, and limited work experience (Braveman and Keilhofner, 2006). Perceived lack of flexible schedules to accommodate adherence to prescribed treatment or lack of sensitive workplace environments may also impede an individual’s decision to return to work (Hergenrather et al., 2004).
Not everyone with HIV/AIDS who is capable of employment actually returns to work. The overall patterns by which patients continue, resume, or suspend employment after learning their HIV status vary. Often patients stop working for a brief time when they first learn of their diagnosis. This is frequently in the setting of a secondary infection that prompted diagnos-
tic testing. Once patients are successfully on a stable therapeutic regimen, they may achieve a balanced health status and desire to resume their work activities.
However, treatment-related complications and the challenges of managing their health condition may limit full participation in daily activities for some individuals living with HIV/AIDS. Therefore, many individuals remain on Social Security disability despite physical indicators of positive health status such as higher CD4 count, low viral load, or the absence of AIDS-defining illnesses or other infections (Burns et al., 2006). In particular, neurocognitive impairment associated with HIV infection has been shown to have a significant association with the rate of unemployment among affected individuals, despite the absence of physical symptoms due to HIV (Heaton et al., 2010).
REFERENCES
Abel, E., and L. Painter. 2003. Factors that influence adherence to HIV medications: Perceptions of women and health care providers. Journal of the Association of Nurses in AIDS Care 14(4):61–69.
Aloisi, M. S., C. Arici, R. Balzano, P. Noto, R. Piscopo, G. Filice, F. Menichetti, A. Monforte, G. Ippolito, and E. Girardi. 2002. Behavioral correlates of adherence to antiretroviral therapy. Journal of Acquired Immune Deficiency Syndromes 31(Suppl 3):S145–S148.
Arns, P., D. Martin, and R. Chernoff. 2004. Psychosocial needs of HIV-positive individuals seeking workforce re-entry. AIDS Care 16(3):377–386.
Bangsberg, D. R., F. M. Hecht, E. D. Charlebois, A. R. Zolopa, M. Holodniy, L. Sheiner, J. D. Bamberger, M. A. Chesney, and A. Moss. 2000. Adherence to protease inhibitors, HIV-1 viral load, and development of drug resistance in an indigent population. AIDS 14(4):357–366.
Bangsberg, D. R., E. D. Charlebois, R. M. Grant, M. Holodniy, S. G. Deeks, S. Perry, K. N. Conroy, R. Clark, D. Guzman, A. Zolopa, and A. Moss. 2003. High levels of adherence do not prevent accumulation of HIV drug resistance mutations. AIDS 17(13):1925–1932.
Bangsberg, D. R., A. R. Moss, and S. G. Deeks. 2004. Paradoxes of adherence and drug resistance to HIV antiretroviral therapy. Journal of Antimicrobial Chemotherapy 53(5): 696–699.
Bennett, N. J., and F. B. Rose. 2010. HIV disease. emedicine.com, http://emedicine.medscape.com/article/211316-overview (accessed June 18, 2010).
Braveman, B., and G. Keilhofner. 2006. HIV/AIDS and employment: The continuing challenge. Work 27(3):205–207.
Brooks, R. A., D. J. Martin, D. J. Ortiz, and R. C. Veniegas. 2004. Perceived barriers to employment among persons living with HIV/AIDS. AIDS Care 16(6):756–766.
Buchacz, K., R. K. Baker, F. J. Palella Jr., J. S. Chmiel, K. A. Lichtenstein, R. M. Novak, K. C. Wood, J. T. Brooks, and the HOPS Investigators. 2010. AIDS-defining opportunistic illnesses in U.S. patients, 1994–2007: A cohort study. AIDS 24(10):1549–1559.
Burns, S. M., L. R. L. Young, and S. Maniss. 2006. Predictors of employment and disability among people living with HIV/AIDS. Rehabilitation Psychology 51(2):127–134.
CDC (Centers for Disease Control and Prevention). 2001. First report of AIDS. Atlanta, GA: Centers for Disease Control and Prevention, U.S. Department of Health and Human Services.
CDC. 2007. HIV/AIDS surveillance report: Cases of HIV infection and AIDS in the United States and Dependent Areas, 2007. Atlanta, GA: Centers for Disease Control and Prevention, U.S. Department of Health and Human Services.
CDC. 2008. HIV prevalence estimates—United States, 2006. Morbidity and Mortality Weekly Report 57(39):1073–1076.
Chesney, M. A., J. R. Ickovics, D. B. Chambers, A. L. Gifford, J. Neidig, B. Zwickl, and A. W. Wu. 2000. Self-reported adherence to antiretroviral medications among participants in HIV clinical trials: The AACTG adherence instruments. AIDS Care 12(3):255–266.
Egger, M., M. May, G. Chêne, A. N. Phillips, B. Ledergerber, F. Dabis, D. Costagliola, A. D’Arminio Monforte, F. de Wolf, P. Reiss, J. D. Lundgren, A. C. Justice, S. Staszewski, C. Leport, R. S. Hogg, C. A. Sabin, M. J. Gill, B. Salzberger, and J. A. C. Sterne. 2002. Prognosis of HIV-1-infected patients starting highly active antiretroviral therapy: A collaborative analysis of prospective studies. The Lancet 360(9327):119–129.
Fama, R., M. J. Rosenbloom, B. N. Nichols, A. Pfefferbaum, and E. V. Sullivan. 2009. Working and episodic memory in HIV infection, alcoholism, and their comorbidity: Baseline and 1-year follow-up examinations. Alcoholism: Clinical and Experimental Research 33(10):1815–1824.
Goldman, D. P., and Y. Bao. 2004. Effective HIV treatment and the employment of HIV(+) adults. Health Services Research 39:1691–1712.
Grant, I. 2008. Neurocognitive disturbances in HIV. International Review of Psychiatry 20(1):33–47.
Hall, H., R. Song, P. Rhodes, J. Prejean, Q. An, L. M. Lee, J. Karon, R. Brookmeyer, E. H. Kaplan, M. T. McKenna, and R. S. Janssen. 2008. Estimation of HIV incidence in the United States. Journal of the American Medical Association 300(5):520–529.
Heaton, R., D. Clifford, D. Franklin Jr., S. Woods, C. Ake, F. Vaida, R. Ellis, S. Letendre, T. Marcotte, J. Atkinson, M. Rivera-Mindt, O. Vigil, M. Taylor, A. Collier, C. Marra, B. Gelman, J. McArthur, S. Morgello, D. Simpson, J. McCutchan, I. Abramson, A. Gamst, C. Fennema-Notestine, T. Jernigan, J. Wong, and I. Grant for The CHARTER Study Group. 2010 (in press). HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy. Neurology.
Hergenrather, K. C., S. D. Rhodes, and G. Clark. 2004. Employment-seeking behavior of persons with HIV/AIDS: A theory-based approach. Journal of Rehabilitation 70(4):22–32.
Hergenrather, K. C., S. D. Rhodes, and G. Clark. 2005. The employment perspectives study: Identifying factors influencing the job-seeking behavior of persons living with HIV/AIDS. AIDS Education & Prevention 17(2):131–142.
Hogg, R., J. A. C. Sterne, V. Lima, S. Grabar, M. Battegay, M. Bonarek, A. D’Arminio Monforte, A. Esteve, M. J. Gill, A. Justice, A. Hayden, F. Lampe, A. Mocroft, M. J. Mugavero, S. Staszewski, J. C. Wasmuth, A. van Sighem, M. Kitahata, J. Guest, M. Egger, and M. May. 2008. Life expectancy of individuals on combination antiretroviral therapy in high-income countries: A collaborative analysis of 14 cohort studies. The Lancet 372:293–299.
Kirk, J. B., and M. B. Goetz. 2009. Human immunodeficiency virus in an aging population, a complication of success. Journal of the American Geriatrics Society 57(11):2129–2138.
Klimas, N., A. O’Brien Koneru, and M. A. Fletcher. 2008. Overview of HIV. Psychosomatic Medicine 70(5):523–530.
Lanièce, I., M. Ciss, A. Desclaux, K. Diop, F. Mbodj, B. Ndiaye, O. Sylla, E. Delaporte, and I. Ndoye. 2003. Adherence to HAART and its principal determinants in a cohort of Senegalese adults. AIDS 17(Suppl 3):S103–S108.
Lima, V. D., R. Harrigan, D. R. Bangsberg, R. S. Hogg, R. Gross, B. Yip, and J. Montaner. 2009. The combined effect of modern highly active antiretroviral therapy regimens and adherence on mortality over time. Journal of Acquired Immune Deficiency Syndromes 50(5):529–536.
Mannheimer, S., J. Matts, E. Telzak, M. Chesney, C. Child, A. Wu, and G. Friedland. 2005. Quality of life in HIV-infected individuals receiving antiretroviral therapy is related to adherence. AIDS Care 17(1):10–22.
Martin, D. J., P. G. Arns, R. A. Chernoff, and M. Steckart. 2004. Working with HIV/AIDS: Who attempts workforce reentry following disability? Journal of Applied Rehabilitation Counseling 35(3):28–38.
Martin, D. J., M. J. Steckart, and P. G. Arns. 2006. Returning to work with HIV/AIDS: A qualitative study. Work 27(3):209–219.
Mills, E. J., J. B. Nachega, I. Buchan, J. Orbinski, A. Attaran, S. Singh, B. Rachlis, P. Wu, C. Cooper, L. Thabane, K. Wilson, G. H. Guyatt, and D. R. Bangsberg. 2006. Adherence to antiretroviral therapy in Sub-Saharan Africa and North America: A meta-analysis. Journal of the American Medical Association 296(6):679–690.
Nieuwkerk, P. T., and F. J. Oort. 2005. Self-reported adherence to antiretroviral therapy for HIV-1 infection and virologic treatment response: A meta analysis. Journal of Acquired Immune Deficiency Syndromes 38(4):445–448.
Pence, B. W., W. C. Miller, K. Whetten, J. J. Eron, and B. N. Gaynes. 2006. Prevalence of DSM-IV-defined mood, anxiety, and substance use disorders in an HIV clinic in the Southeastern United States. Journal of Acquired Immune Deficiency Syndromes 42(3):298–306.
Rabkin, J. G., M. McElhiney, S. J. Ferrando, W. Van Gorp, and S. Hsing. 2004. Predictors of employment of men with HIV/AIDS: A longitudinal study. Psychosomatic Medicine 66(1):72–78.
Satriano, J., A. Berkman, and R. H. Remien. 2005. Acquired immune deficiency syndrome and human immunodeficiency virus. In Medical aspects of disability, 3rd ed. A handbook for the rehabilitation professional, edited by H. H. Zaretsky, E. F. Richter III, and M. G. Eisenberg. New York: Springer Publishing Co.
Stoff, D. M., L. Mitnick, and S. Kalichman. 2004. Research issues in the multiple diagnoses of HIV/AIDS, mental illness and substance abuse. AIDS Care 16(Suppl 1):S1–S5.
U.S. Census Bureau. 2003. Disability status: 2000. Census 2000 brief. Washington, DC: U.S. Census Bureau.
Valcour, V. G., C. M. Shikuma, M. R. Watters, and N. C. Sacktor. 2004. Cognitive impairment in older HIV-1-seropositive individuals: Prevalence and potential mechanisms. AIDS 18(Suppl 1):S79–S86.
Vance, D. E., and T. C. Struzick. 2007. Addressing risk factors of cognitive impairment in adults aging with HIV: A social work model. Journal of Gerontological Social Work 49(4):51–77.
Vlassova, N., A. Angelino, and G. Treisman. 2009. Update on mental health issues in patients with HIV infection. Current Infectious Disease Reports 11(2):163–169.
Vranceanu, A. M., S. A. Safren, M. Lu, W. M. Coady, P. R. Skolnik, W. H. Rogers, and I. B. Wilson. 2008. The relationship of post-traumatic stress disorder and depression to antiretroviral medication adherence in persons with HIV. AIDS Patient Care and STDs 22(4):313–321.












