4
FATE AND TRANSPORT OF HERBICIDES USED IN VIETNAM
The potential for exposure of Blue Water Navy personnel to Agent Orange and its contaminant 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) is related to the fate of these chemicals in the Vietnamese environment. This chapter lays out the committee’s understanding of the fate of Agent Orange after its environmental release as described in Chapter 3, with special attention to TCDD transport to and fate in coastal waters and atmospheric drift, and to modeling efforts to estimate environmental concentrations of TCDD. Because the relative importance of fate pathways is chemical specific, the discussion is restricted to TCDD that reached the environment through spraying and other releases of Agent Orange. The committee’s rationale for the focus on TCDD is its understanding that TCDD is the toxic agent that has been associated with adverse health effects in Vietnam veterans (IOM, 1994).
The chapter takes a multimedia approach to discussing fate by looking at the air–vegetation–soil–water–sediment system of coastal Vietnam. Coastal fish are also included in the discussion. A multimedia approach considers all environmental compartments (such as air and soil) simultaneously, so intercompartmental transfers of TCDD are considered at the same time as the movement of TCDD within a single compartment. An example of a multimedia approach is consideration of TCDD evaporation from the surfaces of leaves to the air at the same time keeping in mind the movement of TCDD by means of air currents.
The committee did not identify any information on environmental concentrations of TCDD at the time of the Vietnam War that met current criteria for analytic quality assessment and quality control. The committee next considered using a mathematical model to estimate likely concentrations on the basis of TCDD that entered the environment. All models require a host of input parameter values, including standard physical and chemical properties of TCDD, characteristics of the
Vietnamese environment, and such chemical- and environment-specific parameter values as rates of TCDD degradation in Vietnamese coastal sediments. The committee found that, as is often the case for multimedia models and dioxins, input data and especially data with which to evaluate model performance were not available. Furthermore, large uncertainty would accompany any attempt to model overall TCDD fate by modeling emissions from individual spray paths on which data are available and scaling them up to hundreds of miles of coastline and coastal waters.
The committee explored the variability and uncertainty of several of the many parameters that could be used for assessing the fate and transport of TCDD in the environment, as described in the sections below. The goal of the exercise was a qualitative assessment of the fate of TCDD released during the Vietnam War. The discussion acknowledges the importance of location-specific factors that mediate chemical fate and transport, notably the tropical climate and the geography of Vietnam, which include episodic monsoons and the Mekong Delta, respectively. Most of the literature on the chemical fate of dioxins originates in temperate areas of the globe, such as Europe and North America, that differ substantially from the Vietnamese environment, and the committee was mindful of the differences as it developed its understanding of the fate of Agent Orange–associated TCDD in Vietnam.
THE VIETNAMESE ENVIRONMENT
The Republic of Vietnam is in the southern portion of the Indochina Peninsula in Southeast Asia. Its long, narrow shape extends nearly 1,100 km from north to south (about 8° to 17°N latitude) with a maximum width, in an east–west direction, of about 210 km and an area of nearly 170,000 km2. About 1,400 km of the nearly 1,660 km of Vietnam’s coastline is on the South China Sea, and the remaining (roughly 260 km) short stretch of coast on the southern end of the country faces the Gulf of Thailand. The inland portion of the northern two-thirds of southern Vietnam consists of high plateaus with coastal valleys and alluvial plains and streams that bring materials to the coastal zone. The coastline is irregular, with numerous headlands and bays.
The transport and fate of contaminants, such as TCDD, in the environment cannot be discussed without an understanding of the predominant hydrologic conditions of the area. The southern portion of
Vietnam is dominated by complex alluvial deltas, such as that of the Mekong River, and smaller river systems, such as that of the Bien Hoa. This portion of the country is mostly low lying and subject to frequent inundations and floods from monsoon rains; outside the monsoon season, river flows near the coast are controlled mainly by tidal forces. The coastline is mostly marshy and is lined with mangrove swamps distributed along much of the coastline in the Mekong Delta. It should be noted that the low-lying swamps at the interface between land and water were targets for heavy spraying of Agent Orange. Offshore of the swamps, the continental shelf off Vietnam slopes gently from the shoreline to the continental break at depths of about 100–200 m; from that point on, the slope is steeper, reaching depths of over 3,000 m in the South China Basin.
Climate of Vietnam
The Republic of Vietnam has a tropical monsoon climate with prevailing winds from the south to southeast during the summer monsoon and from the north to northeast during the winter monsoon. Air temperature and rainfall vary with the monsoon season. The average temperature and rainfall along the coast range from 24° to 30°C and from about 100 cm (40 in.) to over 300 cm (118 in.). The plateaus and higher foothills are cooler and have localized rainfall in excess of 400 cm (160 in.). The characteristics of the monsoon seasons (for example, winds, rainfall, and duration) vary throughout the country in that the winter monsoon (October–January) is more pronounced over the northern two-thirds of the country and the summer monsoon (May–August) tends to affect mostly the southern delta region.
The direction of predominant monsoonal winds follows the axis of the South China Sea, which gives the wind a maximum fetch that produces more powerful currents and larger waves during the fully developed monsoon season. At the beginning of the northeast monsoon (October), water in the South China Sea begins to flow to the southwest along the coast of Vietnam; it reaches its maximum, often exceeding 1 m/s, when the monsoon is fully developed (December). By April, the southerly flow intensity decreases with the decrease in monsoon intensity. The southwest or summer monsoon reverses the flow direction, and the surface water moves to the northeast, reaching its maximum flow in August.
Mekong River and Its Delta
The alluvial Mekong River is the dominant river system in southern Vietnam. Its estimated length is 4,350 km from its sources in southern China, it drains an area of 795,000 km2, and it discharges 475 km3 of water annually to the South China Sea. Hordoir et al. (2006) investigated river-coastal water flows of the Mekong Delta by developing a local coastal ocean model based on the Princeton Ocean Model of Blumberg and Mellor (1987). They ran the model for a year (1997) and evaluated it by comparing model simulations with conductivity–temperature–depth measurements made along the coast in March 1997 (the dry season) and at the beginning and end of October 1997 (the end of the monsoon season and the beginning of the dry season, respectively).
The results of Hordoir et al. (2006) indicated that although the flow from the Mekong Delta is very small compared with ocean currents, it influences ocean dynamics and physics in coastal regions as a result of the river’s input of low-density fresh water into the higher-density saline water of the ocean. During winter, when the monsoons affect the northern part of the country, the total Mekong flow is about 2,500 m3/s with the wind blowing predominantly from the northeast. That wind creates a compressed freshwater plume flowing southwest toward the Gulf of Thailand. The offshore plume, from the upper (Dai and Ham Luong) and lower (Dinh An, Tranh De, Hau, and Chieu) mouths of the Mekong Delta, would be well mixed vertically as a result of turbulence caused by the current’s interaction with the shallow coastal shelf.
Hordoir et al. (2006) estimated that the offshore plume would be 5–10 m deep. The width of the plume would be 20–50 nautical miles (37–93 km) and be greatest off the southernmost point of the delta. During summer, when monsoons affect the south, the Mekong freshwater flow reaches a maximum of about 35,000 m3/s with the wind changing radically from the northeast to the southwest. At that time, the winds cause the freshwater plume to head offshore, with only a weak current heading southeast. As a consequence, the freshwater influence would extend far offshore from the mouth of the delta. Hordoir et al. did their final simulation at the end of October, when the winds changed to the northeast and the river flow, although it decreased from early October, was still higher than that in winter; this resulted in a broader plume that hugged the coast and again flowed in a southeast direction.
FATE AND TRANSPORT OF AGENT ORANGE–ASSOCIATED TCDD IN THE VIETNAMESE ENVIRONMENT
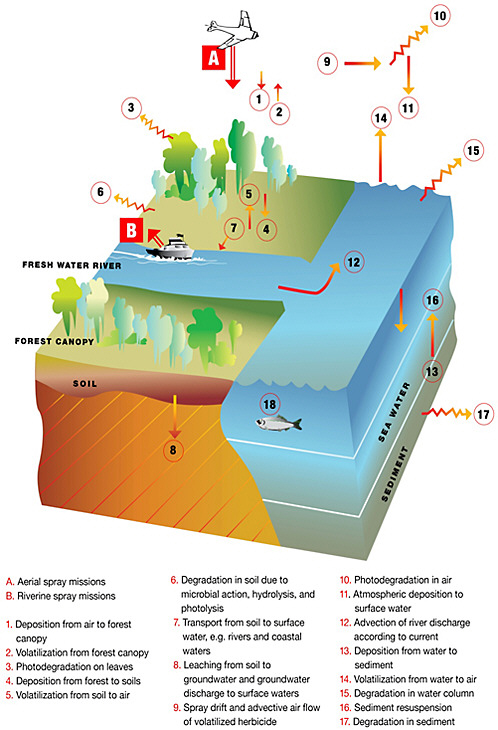
As discussed in Chapter 3, 95% of the tactical herbicide used during the Vietnam War was applied by aerial spraying as part of Operation Ranch Hand (Stellman et al., 2003). Figure 4-1 illustrates the entry, transport, and fate pathways of Agent Orange–associated TCDD in the Vietnamese environment. The amount of TCDD that was sprayed in Vietnam during the Operation Ranch Hand missions has been variously estimated at 221–336 kg (Stellman et al., 2003) and 105–119 kg (Young, 2009). As described in Chapter 3, aerial applications of Agent Orange were conducted to deliver the maximum amount of herbicide to the forest canopy and to minimize spray drift.
An unknown fraction of aerial spraying occurred close to or over water—either rivers or coastal zones. Rivers were targeted because they served as transportation pathways. III Corps Area, north of the Mekong Delta, was the most heavily sprayed area of Vietnam (IOM, 1994). That area includes mangrove swamps and wetland soils, which, as described earlier, are organically rich and subject to seasonal inundation. Agent Orange could enter water courses by runoff from soil and vegetation from the sprayed areas. Agent Orange could also enter water courses from the spraying of river banks by the Brown Water Navy. The amount of Agent Orange applied to the river banks is uncertain, but it was less than 5% of the total amount used in Vietnam (Young, 2009). The committee assumed that most herbicide was released in air by aerial spraying (Figure 4-1[A]) and entered the water either by direct spraying over waterways or deposition of spray drift (Figure 4-1[9]) or indirectly by runoff from soil or vegetation (Figure 4-1[7]).
Modeling Fate and Transport of TCDD
The goal of assessing the fate and transport of Agent Orange–associated TCDD in the environment was to determine concentrations of TCDD that would probably have been found in each environmental compartment. On the basis of the presence or absence of TCDD, it might be possible to determine whether military personnel in Vietnam, particularly Blue Water Navy veterans, had the opportunity for exposure to TCDD and at what concentrations.
Mathematical models of chemical fate are widely used to reconstruct historical, current, and future conditions after an emission into a specified environment. A variety of models have been used to estimate the fate of polychlorinated dibenzodioxins and polychlorinated dibenzofurans (PCDDs and PCDFs), of which TCDD is one. Atmospheric dispersion models—such as TREND (van Jaarlsveld and Schutter, 1993), CMAQ (Zhang et al., 2009), and HYSPLIT (Cohen et al., 2002)—have been used to track PCDD and PCDF emissions over North America and Europe, where emissions originate mainly from combustion processes, such as municipal solid-waste incineration. There are three major constraints in using such models in the context of Agent Orange application in Vietnam. First, the models are extremely data intensive in that they require detailed information of time-dependent or statistically averaged meteorologic conditions (that is, historical weather data) at the time of chemical release. Such data were not available to the committee for simulating Vietnamese meteorologic conditions during the Vietnam War.
Second, the models have been applied to simulate PCDD and PCDF emissions from combustion in temperate regions. Translating the model to spray emissions in Vietnam requires detailed information on chemical partitioning and reaction rates in tropical regions. As explained below, there is a paucity of information on Vietnamese environmental conditions.
Third, the models must be evaluated with measured chemical concentrations, which could be another set of related compounds, such as polycyclic aromatic hydrocarbons. Model evaluation is crucial for gaining confidence that the model is providing reasonable representations of reality. Data on model evaluation specific to Vietnam are not available. This is a critical need because to the committee’s knowledge such models have not been used to estimate the fate of Agent Orange in Vietnam, although there was some simulation of Agent Orange fate in tropical conditions at Elgin Air Force Base in Florida during the Vietnam War (Young, 2009).
Multimedia box models have also been used to estimate “average” TCDD fate and concentrations in the environmental compartments of air, water, soil, sediment, and fish. The models emphasize chemical transport among environmental compartments but simplify movement within each compartment, such as air and water. Box refers to the assumption that each compartment is a well-mixed box rather than having detailed spatial
resolution, as in the air-dispersion models discussed above. Examples of the application of multimedia models to dioxins are the single box model of Dalla Valle et al. (2003) and the multibox models of Persson et al. (2006) and Sommerfreund et al. (2010). These models have the same constraints as the air-dispersion models but do not require the same level of detailed knowledge of time-dependent meteorology and hydrodynamics.
Models like AgDISP and AgDRIFT have been developed to estimate near-field (within about 1 km) deposition and drift of pesticides applied by aerial spraying. AgDISP and AgDRIFT consider climate (for example, temperature and relative humidity) and spray conditions but not other facets of fate, such as atmospheric degradation or multimedia transfer (Bilanin et al., 1989; Bird et al., 2002; Teske et al., 2002).
The committee concluded that using models to estimate likely concentrations of TCDD in the Vietnamese environment to which Blue Water Navy personnel would be exposed is not possible because of the lack of data with which to assign values to model parameters. Even if parameter values could be assigned, data are not available to evaluate model performance. In the absence of model evaluation, the uncertainty of model results would be too large for the committee to be confident that they were reasonable. Thus, the committee turned to understanding the fate of TCDD on a qualitative or descriptive basis.
Transport of TCDD in the Environment1
The committee considered the fate pathways of Agent Orange and TCDD after its environmental release from aerial spraying (Figure 4-1[A]) and riverbank spraying (Figure 4-1[B]). Although the committee believes that all important fate processes are discussed, the fraction of Agent Orange subject to one or another is highly uncertain. The following discussion is therefore qualitative, not quantitative, and does not include any estimate of likely environmental concentrations of TCDD that prevailed during the Vietnam War. Rather, the purpose of the following discussion is to evaluate qualitatively the likelihood that TCDD concentrations in the Vietnamese air and water after Agent Orange spraying would exceed the TCDD concentrations that would be expected in the absence of Agent Orange spraying.
|
1 |
The discussion in this section refers to the letters and numbers in Figure 4-1. |
TCDD Fate in the Atmosphere
Spray drift
Spray sorties were timed to coincide with calm days and occurred very early in the morning to maximize the contact of herbicide with the forest and to minimize spray drift. Young (2009) summarized calculations of the fate of Agent Orange sprayed in Operation Ranch Hand sorties. Young assumed an atmospheric deposition velocity of 30 cm/s2 for Agent Orange on the basis of a minimum droplet size of <100 µm in the spray emitted from the spray nozzles on the C-123 aircraft. He reported that that would mean that 87% of the Agent Orange sprayed would reach the forest canopy and the remaining 13% would be subject to spray drift with an exponential dropoff in drift as a function of perpendicular distance from the line of spraying.
The Institute of Medicine (IOM) Committee on Making Best Use of the Agent Orange Exposure Reconstruction Model (IOM, 2008) reported various distances that Agent Orange spray could drift, from 2 km (Darrow, 1969) to more than 10 km (Department of the Army, 1971). Ginevan et al. (2009a), however, stated that less than 2% drift would have occurred on the basis of simulations of the AgDRIFT pesticide-spray model. Ross and Ginevan (2007) arrived at an area subject to herbicide deposition of less than 1/40 that of the Stellman model (Stellman and Stellman, 2004).
At issue with the varied estimates of the distance over which spray drift would have occurred is the amount of herbicide deposited on land or water (Figure 4-1[1, 11]). All assume that deposition would drop off exponentially with distance from the spray path, that is, deposition would be greatest at the edge of the spray path. The committee asked the US Department of Agriculture’s Agricultural Research Service to run simulations of the AgDISP pesticide-spray model (Clint Hoffman, USDA ARS, personal communication, December 27, 2010) to evaluate that assumption. With a range of input values intended to reproduce conditions during Agent Orange spraying and drift over open water—for example, 46–76 m release height, 1–6.2 m/s wind speed, 25–30°C, 90% relative humidity, stable atmospheric conditions, ASAE medium–coarse droplet distribution, and 25 L/ha spray volume rate—the AGDISP model estimated that more than 90% of the Agent Orange sprayed was deposited within 100–200 m of the spray path when it was sprayed from a height of 46 and 76 m, respectively. However, a wind of 6.2 m/s, or about 12 knots, the maximum at which spray missions were conducted, could increase the spray drift so that 90–95% of the amount sprayed
would be deposited within 800 m of the spray path (Clint Hoffman, US Department of Agriculture, Agricultural Research Service, personal communication, December 30, 2010). According to runs of AgDISP over a forest canopy versus water, the committee expects that drift would be greater over water than over land because the smoothness of the water surface maximizes drift.
Conditions favoring spray drift from coast to land would occur during the day, particularly during spring and early summer, when the land temperature would exceed that of the coastal waters. That phenomenon can be explained by the land heating faster than water, which causes the air over the land to become warmer and less dense. As the air rises, it is replaced by cooler, denser air flowing in from over the water. That causes an onshore wind, called a sea breeze. At night, the land cools faster than water, as does the corresponding air; the warmer air over the water rises and is replaced by the cooler, denser air from the land, creating an offshore wind called a land breeze. Most sorties were flown early in the morning, when a sea breeze was expected. Thus, the daily timing of the sorties favored sea-breeze conditions, which would minimize spray drift over coastal waters.
Atmospheric fate
The 13% of the Agent Orange not deposited on the forest canopy would be subject to spray drift (Figure 4-1[9]) (Young 2009). The committee considered direct aerial spraying over water. While airborne in Agent Orange spray, TCDD would partition between gas and particle phases. The fraction in the gas phase in temperate urban environments relative to total TCDD has been estimated to vary from 20% to nearly 90% (for example, 10–80% would be sorbed by particles) (Bidleman, 1988; Eitzer and Hites 1989a,b; Hippelein et al., 1996). Gas-phase TCDD in air would degrade by reaction with hydroxyl radicals and to a lesser extent photodegradation (degradation of a molecule due to ultraviolet radiation) (Figure 4-1[10]) (Atkinson et al., 1987).
Degradation of particle-sorbed TCDD is negligible (Koester and Hites, 1992). Podoll et al. (1986) calculated an upper limit for a photolytic half-life of 58 minutes for TCDD in the vapor phase at 40°N latitude. Atkinson et al. (1987) estimated, through an analysis of literature values and by chemical analogy, that the half-life of gas-phase TCDD would be about 3 days (assuming 12 h of sunlight and average global hydroxyl radical concentration of 1.5 × 106 molecules/cm3). Cohen et al. (2002) mathematically derived a half-life of TCDD due to reaction with OH radicals of 5 days, on the basis of the same
assumptions as those of Atkinson et al. Cohen et al. assumed a photodegradation half-life of 2 days on the basis of a review of the literature. Katritzky et al. (2010) calculated a photodegradation half-life for TCDD of 55.5–72.3 h, or 2.3–3 days. These half-lives are long enough to minimize the importance of atmospheric degradation relative to spray drift and deposition.
Deposition on the intentionally targeted vegetation would occur, along with some on soil and surface water, as discussed below (Figure 4-1[11]). Because most spraying occurred during calm conditions with no precipitation, it is assumed that most atmospheric deposition would be dry deposition. Other mechanisms that would deliver TCDD to the coastal surface water would be gas-phase absorption due to diffusion, assuming that the concentration (or more correctly fugacity or activity) gradient was from air to water. Wet deposition or washout due to gas-phase and particle-phase washout is considered the least likely of the deposition mechanisms because most spraying occurred during dry conditions; however, washout would be an efficient mechanism for removing TCDD and other compounds from air. Thus, the committee concluded that with atmospheric deposition, there was an opportunity for herbicides applied by aerial spraying in Vietnam to enter inland waters of Vietnam and the coastal waters that received the discharge from those waters in addition to herbicide that was applied directly to water.
Deposition of TCDD from Air onto Forest Canopy
Young et al. (2004) conducted a detailed analysis of the fate of Agent Orange, and TCDD in particular, sprayed over the Vietnamese forests during the war. They reported results of tests at Elgin Air Force Base in Florida that were designed to simulate environmental conditions during Operation Ranch Hand. They estimated that 87% of the herbicide would have reached the tree canopy within 1 min within or near the spray swath (Figure 4-1[1]). That estimate assumed a leaf area index (LAI) of 5; that is, the area of leaves was 5 times greater than the area of the ground surface below. They further stated that 99% of the sprayed Agent Orange would affect the forest if sorption by tree branches and trunks and the vegetative understory were included. No other studies were found that estimated the amount of TCDD deposited on target spray areas versus the amount subject to atmospheric drift under conditions relevant to the Vietnamese situation.
At the leaf surface, TCDD would contact and sorb to the waxy cuticle of leaves. McCrady and Maggard (1993) measured an initial
uptake rate constant of 1,750 h−1 for TCDD, or an uptake half-life of 1.4 s. The committee was unable to assess the validity of those results, which were reported as an average of several experiments. It is possible that the uptake rate was inflated (that is, too fast) because of uptake by walls of the experimental chamber, which would reduce the concentration of TCDD in chamber air. Young et al. (2004) commented that TCDD sorbed by the leaf cuticle “could not be physically dislodged.” However, other studies have shown that the finger-like protrusions of the cuticle, with sorbed contaminants, can be eroded from leaf surfaces; this can lead to transport of sorbed contaminants from the leaf and possibly to deposition on soil (Sauter et al., 1987; Turunen and Hattunen, 1991).
Loss of TCDD from Vegetation
A fraction of TCDD that came into contact with the leaf would be expected to evaporate (volatilize) as air temperatures increased during the day after spraying (Figure 4-1[2]). A low but measurable rate of volatilization of TCDD from foliage would be expected because of its low vapor pressure (2.0 × 10−7 Pa at 25°C). Temperature-dependent volatilization of persistent semivolatile chemicals from vegetation has been documented in temperate zones (Hornbuckle and Eisenreich, 1996; Su et al., 2007), but that phenomenon has not been reliably measured for dioxins (Lohmann and Jones, 1998). Bacci et al. (1992) found that 1,2,3,4-TCDD, which has partitioning properties similar to that of 2,3,7,8-TCDD, has a strong leaf/air equilibrium bioconcentration factor of 1.03 × 108, suggesting that TCDD will move from the vapor phase in air to leaf surfaces. TCDD on leaf surfaces could also be lost via photodegradation (Figure 4-1[3]) with estimated half-lives around 3–6 h (Crosby and Wong, 1977; Choudhry and Webster, 1989; Schuler et al., 1998). Crobsy and Wong (1977) measured the rate of photodegradation of Agent Orange that contained TCDD at 15 ppm applied to excised leaves of a rubber plant when exposed to sunlight for a half life of about 2 h. They measured a slower photodegradation rate (half life of about 5–6 h) when Agent Orange was applied to glass plates; dark controls were unaffected. This suggests that photochemical dechlorination is a primary removal process for TCDD on leaf surfaces. McCrady and Maggard (1993) reported a much longer TCDD half-life of 44 days via photodegradation measured during a sophisticated chamber experiment in which grass was exposed to gas-phase radiolabeled TCDD and then illuminated by lamps intended to simulate natural sunlight. The
temperatures in the chamber ranged from 18° to 28°C. The authors found that the volatilization half-life was greater than 128 h, which led them to conclude that photodegradation was the main mechanism of loss of TCDD from grass. They commented that the shorter photodegradation half-life measured by Crosby and Wong could be attributable to the promotion of photodegradation by the other organic solvents in Agent Orange in contrast with the pure TCDD used in their experiment. On the basis of those studies, the committee concludes that the photodegradation half-life of TCDD delivered to foliage in Agent Orange would be hours to days, or even weeks, if leaves did not receive direct sunlight. It is also important to note that most experiments and calculations of photodegradation are conducted to simulate environmental conditions at about 40°N latitude; at this latitude, ultraviolet radiation from the sun is less intense than that at 16°N latitude, where Vietnam is.
Deposition of TCDD from Canopy to Soil
TCDD could deposit on soil directly from Agent Orange spraying, by the falling of sprayed foliage, by dry deposition of eroded leaf waxes, and by leaf washoff during precipitation (Figure 4-1[4]). Ginevan et al. (2009a) cited results from the AgDRIFT model that indicated that 1–2% of Agent Orange sprayed aerially would reach the ground. Several researchers (Horstmann et al., 1997; Brorstrom-Lunden and Lofgren, 1998; Wania and McLachlan, 2001) have documented higher concentrations of dioxins in forest than in pasture soils as a result of leaf accumulation of atmospheric dioxins at ambient concentrations and later transfer from plant to soil via the deposition mechanisms mentioned above. In other words, the tree canopy is efficient in collecting atmospherically deposited dioxin and transferring it to soil in cases in which dioxins or other herbicides have not caused leaf dieback. Dioxin transfer to soils from the tree canopy has not been assessed where TCDD was a component of a herbicide.
TCDD Fate in Soil
The organic content of most Vietnamese soils ranges between 1.8% and 5% (Ton, 1996). Because of TCDD’s hydrophobicity (a chemical’s tendency to partition to water and seek nonpolar media, such as soil organic matter), those soils would be efficient sinks for TCDD. Dioxins, in general, are known to be highly persistent in soils (Wania and McLachlan, 2000), as confirmed by Freeman and Schroy (1986), who
found TCDD in soils some 12 years after a field experiment at Elgin Air Force Base that involved applying Agent Orange to soils at a depth of 10 cm. In general, the small fraction of TCDD not sorbed to soil would be lost through volatilization (Figure 4-1[5]). In an experiment at Times Beach, Missouri, where TCDD-contaminated oil had been applied to roads in the early 1970s, Freeman and Schroy (1989) found that volatilization of TCDD from the soil column was very slow with a flux of 6 ×10−8 kg/d m2. In 1984, the observed loss of TCDD from the top 3 mm of soil was 50%; 0.1% of the TCDD in the top 1 mm of soil was estimated to be lost by volatilization, with the remaining TCDD lost by photodegradation. Virtually no TCDD was lost from the soil below this depth. Muir et al. (1985) concluded that the loss of radiolabeled 1,3,6,8-TCDD applied to field soil resulted in a half-life of 131–321 days and could have been due primarily to volatilization. The half-life in sediments was greater than 600 days. Although not large, rates of volatilization would be expected to increase after defoliation when more sunlight would reach soils and soil temperatures would increase.
TCDD in soils may be degraded by microbial activity, hydrolysis, or sunlight (Figure 4-1[6]). Defoliation would also promote photodegradation in soils. Podoll et al. (1986) calculated that—based on TCDD’s low Henry’s constant of 12 torr M−1 and high octanol/water partition coefficient (Kow) of approximately 3 × 108—in wet soil (1% organic carbon and 30% water by volume), 99.99% of the TCDD will be sorbed on soil and would not volatilize. However, in dry soil, TCDD would be subject to some volatilization but substantial TCDD would remain sorbed to the soil. Miller et al. (1989) found that direct photodegradation was important only at soil depths of less than 0.2 mm, so it would account for little of the total TCDD loss from soil. Young (1983) reported that 99.9% of the TCDD applied in herbicides at Elgin Air Force Base in Florida was photodegraded during and immediately after application. Crosby and Wong (1977) found slow but measurable loss of TCDD from soil surfaces exposed to direct sunlight.
Microbial degradation is expected to occur but over time scales of decades or even a century (Sinkkonen and Paasivirta, 2000), although these estimates pertain to temperate regions where microbial degradation is much slower than in warmer regions, such as Vietnam. Karch et al. (2004) note that microbial degradation of TCDD may be enhanced by the presence of herbicide which stimulates microbial activity.
Although the most likely fate of TCDD in soils is to remain in place for decades, a small amount may also be lost as a result of wind or rain
erosion (Muir et al., 1985) and leaching to lower soil horizons (Freeman and Schroy, 1986) (Figure 4-1[7]). A very small amount of TCDD may leach from soil to shallow groundwater (Figure 4-1[8]). The most mobile fraction of TCDD would be associated with colloidal humic or organic material (also referred to as colloidal material in the literature). In contrast to the relatively immobile soil particles, colloids can sometimes travel and facilitate the transport of hydrophobic contaminants. However, colloid concentrations are usually extremely low because aquifers filter out colloids efficiently. Thus, it is highly unlikely that TCDD will have been relocated via groundwater colloids to the river water.
Especially during the flooding of coastal areas, erosion and leaching rather than volatilization followed by redeposition would be expected to deliver the greatest fraction of TCDD from soils to surface waters (Quinh et al., 1989). Erosion could be wind driven or water driven, and erosion rates would increase as defoliation proceeded.
The proximity of soils to waterways and the frequent flooding of the soils, particularly in the heavily sprayed III Corps Area, would increase the probability that soil-bound and humic-material-bound herbicides would reach water courses. The monsoon rains would be expected to transport the humic-bound and soil-bound herbicides to surface waters. Although it is logical to conclude that some fraction of TCDD and other herbicides applied in heavily sprayed areas would reach inland and coastal waters from soils, particularly as the forest is denuded, the amount of TCDD that entered water courses would be subject to enormous dilution from river flows. Quinh et al. (1989) report that the annual discharges of the Tien Giang and Hau Giang rivers in the Mekong Delta, are 92,000 m3/s and 90,000 m3/s, respectively, or “76% and 79% of their flows during the wet season.”
Fate of TCDD in River Waters
If Agent Orange reached water, TCDD would be subject to photolysis. Podoll et al. (1986) estimated that the half-life of TCDD in near surface waters at 40° latitude in sunlight during the summer would be 21 hours compared with 118 hours in the winter. Photolysis would be hindered by the presence of sediment; because of its low water solubility, 0.0193 µg/L, TCDD would tend to sorb to suspended particles and humic material in the receiving waters (Hsieh et al., 1994) and hydrophobic nature. TCDD sorption to suspended particles and humic material would be highly likely in the turbid and humic-rich waters of the rivers in Vietnam. River-water quality is controlled largely by land-based runoff
that, in turn, is controlled by precipitation. The committee was unable to obtain data related to water quality of rivers in Vietnam. It was presumed that the rivers carried organic matter, including humic material, from a terrestrial origin, possibly with sorbed TCDD. On reaching estuaries at river mouths along the coast, particles, colloidal organic matter, and sorbed TCDD would be subject to flocculation and agglomeration as colloids bind to one another and to particles. That creates a zone of high turbidity (a maximum-turbidity zone) that can act as a partial filter for the removal or immobilization of sorbed TCDD from the water column. The mechanism will be less efficient in removing dissolved TCDD, which would be expected to occur at minuscule concentrations (Geyer et al., 2004). The efficiency of the maximum-turbidity zone depends on the concentration of suspended sediment, organic carbon content, and the estuarine chemistry and flow regime. Away from the estuarine and coastal regions, marine water becomes more homogeneous over a wider area, although patchiness due to a lack of mixing occurs. Thus, the committee concluded that whereas most particle-bound and colloid-bound TCDD would deposit in the estuaries of river deltas, some small fraction of total TCDD in the dissolved phase or bound to colloids and particles would make its way out to coastal waters.
Fate of TCDD in Marine Coastal Waters
As TCDD entered the coastal waters of Vietnam as a result of spray drift and atmospheric deposition (Figure 4-1[A]), direct spraying on and along rivers (Figure 4-1[B]), and river discharge, it would be in three phases: truly dissolved, sorbed to humic material (colloidal), and particle bound. Humic-material-bound and particle-bound TCDD would have a high probability of settling to the sediments in estuaries as the fresh river water meets the saline coastal water, as discussed above. A fraction of TCDD that reached coastal waters would be transported by the prevailing coastal zone currents, as described by Hordoir et al. (2006) (Figure 4-1[12]). Although the committee cannot estimate the concentration, the TCDD that entered the water (that is, the TCCD load) would be expected to be greatly diluted at this stage, and dilution would increase with distance from shore.
TCDD could be lost from the top 1–5 m of coastal waters by photodegradation (Figure 4-1[15]). Photodegradation could be important in this top layer of water, presuming that the waters were not excessively turbid; humic material and particles discharged to the coastal marine waters from rivers reduce photodegradation as a result of shading.
Reported half-life values for TCDD in surface water range from 4–5 days (Dulin et al., 1986) in the summer at 40°N latitude and 25°C to up to 600 days (Ward and Matsumura, 1978) at 24°C using water from Lake Mendota and Lake Wingra in Wisconsin. It is expected that the half-life of TCCD in Vietnamese coastal waters, if they were relatively clear, would be shorter than that because of the higher intensity of ultraviolet radiation hitting Vietnamese water than that at 40°N. Hydrolysis is not expected to be an important degradation process for TCDD (Sinkkonen and Paasivirta, 2000).
In most waters, the greatest fraction of TCDD would bind to particles such as algae, detritus, and mineral matter, because these particles are typically most abundant, followed by binding to humic material; finally, a very small fraction of TCDD would be truly dissolved in water, as mentioned above. The partitioning of TCDD among those three would be a function of the concentrations of particles and humic material in coastal waters. The committee was unable to identify information on the concentrations of particles and humic material in Vietnamese coastal waters. Particle-sorbed TCDD would be lost from the water column to bottom sediment by deposition (Figure 4-1[13]). The loss rate would be determined by the concentration of particles and their settling rate. Humic-bound TCDD would be transported by water movement and diffusive exchange with sediment but not deposition, because humic colloids are not subject to settling. Diffusive exchange of colloidal TCDD is not expected to be an important loss process (Sommerfreund et al., 2010). Volatilization of truly dissolved TCDD (Figure 4-1[14]) is also expected to be minimal because of the negligible fraction of TCDD that would be in this phase and the low vapor pressure of TCDD.
TCDD sorbed to coastal sediment particles could return to the water column as a result of sediment resuspension (Figure 4-1[16]). A smaller amount could return to the water column as a result of sediment-to-water diffusion of colloidal TCDD; however, this is usually negligible (Sommerfreund et al., 2010). Resuspension of sediment-sorbed chemicals can be an important process in shallow waters, such as the coastal zone off the Mekong Delta, where the sea bottom drops from a depth of 5 m extending about 5 km offshore to 20 m about 40 km offshore (Hordoir et al., 2006). Resuspension events in shallow waters are expected when winds are high, at times of very high river discharge (for example, during the summer monsoon season) when turbulent flows could entrain sediment particles, and possibly as a result of a ship
dropping or pulling up an anchor. Although resuspension can bring particle-bound chemicals back into the water column, they will redeposit as the turbulence dissipates. Thus, sediment-bound chemicals can cycle between sediment and the water column.
The final loss processes, which ultimately remove TCDD from the aquatic system, are microbial degradation in sediment and burial in deeper sediment (Figure 4-1[17]). As is the case with soils, degradation by anaerobic microorganisms in sediment is expected over time scales of 10s to 100s of years (Sinkkonen and Paasivirta, 2000).
LIMITATIONS OF AND UNCERTAINTIES ON FATE AND TRANSPORT OF TCDD
No studies of the fate of Agent Orange or TCDD in the Vietnamese environment at the time of the Vietnam War were found. As noted in the discussion above, the committee’s conclusions are based on a theoretical understanding of fate processes of TCDD. Field studies of TCDD have been undertaken in temperate zones or with systems set up to simulate conditions that prevailed during the Vietnam War, such as the experiments conducted at Elgin Air Force Base in Florida. Modeling results cited here, such as those of AgDRIFT and AgDISP, are based on conditions similar to those in the Vietnam War.
The several studies below have reported measurable TCDD in environmental samples taken in Vietnam and Laos from the 1980s onward that are attributed to the use of Agent Orange during the Vietnam War. Those studies indicate that TCDD accumulated at low concentrations in aquatic systems (e.g., Pavlov et al., 2004) or soils, and this suggests transport from sprayed regions or substantial TCDD degradation since Operation Ranch Hand. The results of the earlier studies are confounded by issues of TCDD detection; however, analytic methods have improved dramatically over the last few decades. TCDD contamination has been found around areas known to have been used as Agent Orange depots. Schecter et al. (1989a,b) analyzed five samples of river silt taken from the Dong Nai River (upstream of Ho Chi Minh City [Saigon] and near a village that had been heavily sprayed with Agent Orange) and one sample from the Saigon River in the Ho Chi Minh City area in 1985 and 1986 using a high-resolution analytic method. They found that TCDD in sediment samples from the Dong Nai River were below the limit of detection (21 pg/g dry weight), however, TCDD was
present at 210 pg/g in sediment (limit of detection, 6 pg/g dry weight) from the Saigon River in Ho Chi Minh City. The latter finding is for only one sample. In 1999, Schecter et al. (2001) measured concentrations of TCDD in soil and sediment samples taken from Bien Hoa Air Base and its vicinity in southern Vietnam and from Hanoi. Concentrations were 1,164,699 pg/g (ppt) dry matter in soil from the air base where Agent Orange was known to be used, stored, and spilled; 0.8–117 ng/kg (ppt) in sediments of Bien Hung Lake near the air base; and 0.8–1.5 ng/kg in the Dong Nai River that flows from the lake; no TCDD was detected in the sediments of the Hanoi River. More recently, Piazza et al. (2007) reported low concentrations of total dioxins and furans, particularly TCDD, in the sediment of Vietnamese coastal lagoons and some inland waters. That observation led them to conclude that degradation took place shortly after Agent Orange was sprayed. Dwernychuk et al. (2002) found increased concentrations of TCDD in soil and food samples systematically collected in 1996–1999 from areas of the Aluoi Valley in Vietnam known to have been heavily sprayed with Agent Orange compared with areas that received less spraying. The highest concentrations were found in soil and animal fat collected in a Vietnamese village on a former US Army base where Agent Orange had been stored during 1963–1966. Nhu et al. (2009) also found that TCDD concentrations in soil and breast-milk samples taken from Agent Orange-sprayed areas of Vietnam in 2002–2003 were higher than those in samples from nonsprayed areas. In addition, Quinh et al. (1989) suggested that the relatively high concentrations of TCDD found in adipose tissue of some Vietnamese residents of areas that had not been heavily sprayed with Agent Orange might be caused by the runoff of TCDD-contaminated soils from sprayed areas into rivers during the monsoon season with subsequent flooding carrying the contaminated soil into fields.
CONCLUSIONS
The committee reviewed the fate of Agent Orange and TCDD in particular in the Vietnamese environment as a result of spray applications during the Vietnam War. The discussion is entirely qualitative because the committee was not able to quantify any component of the fate processes discussed. Furthermore, insufficient data were available even
to attempt a rudimentary calculation of the fate of TCDD emitted into the Vietnamese environment during the war.
On the basis of a review of the literature and knowledge of fate and transport processes in the context of Vietnam, the committee concludes that most Agent Orange and TCDD sprayed as part of Operation Ranch Hand intercepted target vegetation. Undoubtedly, some fraction entered soil either immediately after spraying or as the vegetation died back because of Agent Orange’s herbicidal activity. Most TCDD deposited on soil would be expected to persist, as has been confirmed by some more recent studies of contemporary dioxin concentrations in soils (Dwernychuk et al., 2002) but not other earlier studies (Schecter et al., 1989). The committee concludes that Agent Orange and TCDD could have entered rivers from spraying along riverbanks (although this was a small fraction of the total Agent Orange applied in Vietnam) or from soil runoff, particularly in heavily sprayed areas that experienced frequent flooding. River loading would be highly diluted by river flows. A substantial fraction of particles and humic material to which TCDD would be bound would settle in river mouths and estuaries because of estuarine dynamics and chemistry, although it is puzzling that recent studies have found minimal residual TCDD in sediment dated back to Agent Orange spraying during the war.
TCDD would enter coastal marine water from river discharge (albeit a very small load because of the mechanisms discussed) and from spray drift. The committee concludes that TCDD loading due to spray drift could have occurred but would have been minimal. Atmospheric deposition of TCDD on coastal waters from spray drift would have been greatly diluted in these waters. Given the paucity of information and the variability and uncertainty in the available information, the committee concludes that it is not possible to estimate the likely concentrations of TCDD in marine waters and air at the time of the Vietnam War.
REFERENCES
Atkinson, R. 1987. Estimation of OH radical reaction rate constants and atmospheric lifetimes for polychlorobiphenyls, dibenzo-p-dioxins, and dibenzofurans. Environmental Science and Technology 21:305-307.
Bacci, E., M. J. Cerejeira, C. Gaggi, G. Chemello, D. Calamari, and M. Vighi. 1992. Chlorinated dioxins: Volatilization from soils and bioconcentration in plant leaves. Bulletin of Environmental Contamination and Toxicology 48:401-408.
Bidleman, T. F. 1988. Atmospheric processes: Wet and dry deposition of organic compounds are controlled by their vapor-particle partitioning. Environmental Science and Technology 22:361-367.
Bilanin, A. J., M. E. Teske, B. W. Barry, and R. B. Ekbald.1989. AGDISP: The aircraft spray dispersion model, code development, and experimental validation. Transactions of the ASAE 32(1):327-334.
Bird, S. L., S. G. Perry, S. L. Ray, and M. E. Teske. 2002. Evaluation of the AGDISP aerial spray algorithms in the AgDRIFT model. Environmental Toxicology and Chemistry 21(3):672-681.
Blumberg, A. F., and G. L. Mellor. 1987. A description of a three-dimensional coastal ocean circulation model. In Three-Dimensional Coastal Ocean Models, Vol. 4, edited by N. Heaps. Washington, DC: American Geophysical Union.
Brorstrom-Lunden, E., and C. Lofgren. 1998. Atmospheric fluxes of persistent semivolatile organic pollutants to a forest ecological system at the Swedish west coast and accumulation in spruce needles. Environmental Pollution 102(139-149):277-291.
Choudhry, G. G., and G. R. B. Webster. 1989. Environmental photochemistry of PCDDs quantum yields of the direct phototransformation of 1,2,3,7-tetra-, 1,3,6,8-tetra, 1,2,3,4,6,7,8-hepta, and 1,2,3,4,6,7,8,9-octachlorodibenzo-p-dioxin in aqueous acetonitrile and their sunlight half-lives. Journal of Agricultural and Food Chemistry 37(1):254-261.
Cohen, M., et al. 2002. Modeling the Atmospheric Transport and Deposition of PCDD/F to the Great Lakes. Journal of Environmental Science and Technology 36:4831-4845.
Crosby, D. G., and A. S. Wong. 1977. Environmental degradation of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). Science 195:1337-1338.
Dalla Valle, A. 2004. The skew-normal distribution. In Skew Elliptical Distributions and Their Applications, edited by M.G. Genton. Boca Raton, FL: CRC Press. Pp. 2-24.
Darrow, R. A. 1969. Report of trip to Republic of Vietnam, 15 August-2 September 1969. Department of the Army.
Department of the Army. 1971. Field manual: Tactical employment of herbicides. Washington, DC: Headquarters.
Dulin, D., H. Drossman, and T. Mill. 1986. Products and quantum yields for photolysis of chromoaromatics in water. Environmental Science and Technology 20:72-77.
Dwernychuk, L. W., H. D. Cau, C. T Hatfield, T. G. Boivin, T. M. Hung, P. T. Dung, and N. D. Thai. 2002. Dioxin reservoirs in southern Viet Nam—A legacy of Agent Orange. Chemosphere 47:117-137.
Eitzer, B. D., and R. A. Hites. 1989a. Atmospheric transport and deposition of polychlorinated dibenzo p-dioxins and dibenzofurans. Environmental Science and Technology 1989:1396-1401.
Eitzer, B. D., and R. A. Hites. 1989b. Polychlorinated dibenzo-p-dioxins and dibenzofurans in the ambient atmosphere of Bloomington, Indiana. Environmental Science and Technology 23:1389-1395.
EPA (Environmental Protection Agency). 1993. Interim report on data and methods for assessment of 2,3,7,8-tetrachlorodibenzo-p-dioxin risks to aquatic life and associated wildlife. Washington, DC: Office of Water.
Freeman, R. A., and J. M. Schroy. 1986. Modeling the transport of 2,3,7,8-TCDD and other low volatility chemicals in soils. Environmental Progress 5(1).
Geyer, W. R., P. S. Hill, and G. C. Kineke. 2004. The transport, transformation and dispersal of sediment by buoyant coastal flows. Continental Shelf Research 24(7-8):927-949.
Ginevan, M. E., J. H. Ross, and D. K. Watkins. 2009. Assessing exposure to allied ground troops in the Vietnam War: A comparison of AgDRIFT and Exposure Opportunity Index models. Journal of Exposure Science and Environmental Epidemiology 19(2):187-200.
Hippelein, M., H. Kaupp, G. Dorr, M. S. McLauchan, and O. Hutzinger. 1996. Baseline contamination assessment for a new resource recovery facility in Germany. Part II: Atmospheric concentration of PCDD/F. Chemosphere 32:1605-1616.
Hordoir, R., K. D. Nguyen, and J. Polcher. 2006. Simulating tropical river plumes, a set of parameterizations based on macroscale data: A test case in the Mekong Delta region. Journal of Geophysical Research-Oceans 111(C9).
Hornbuckle, K. C., and S. J. Eisenreich. 1996. Dynamics of gaseous semivolatile organic compounds in a terrestrial ecosystem—Effects of diurnal and seasonal climate variations. Atmospheric Environment 30:3935-3945.
Hsieh, D. P. H., F. F. Chiao, R. C. Currie, and T. E. McKone. 1994. Intermedia transfer factors for contaminants found at hazardous waste sites: 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). Final draft report. Davis, CA: University of California, Department of Environmental Toxicology.
IOM (Institute of Medicine). 1994. Veterans and Agent Orange: Health effects of herbicides used in Vietnam. Washington, DC: National Academy Press.
IOM. 2008. The utility of proximity-based herbicide exposure assessment in epidemiologic studies of Vietnam veterans. Washington, DC: The National Academies Press.
Karch, N. J., D. K. Watkins, A. L. Young, and M. E. Ginevan. 2004. Environmental fate of TCDD and Agent Orange and bioavailability to troops in Vietnam. Organohalogen Compounds 66:3689-3694.
Katritzky, A. R., S. H. Slavov, I. B. Stoyanova-Slavova, and M. Karelson. 2010. Correlation of the photolysis half-lives of polychlorinated dibenzo-p-dioxins and dibenzofurans with molecular structure. Journal of Physical Chemistry 114:2684-2688.
Koester, C. J., and R. A. Hites. 1992. Photodegradation of polychlorinated dioxins and dibenzofurans adsorbed to fly-ash. Environmental Science & Technology 26(3):502-507.
Lohmann, R., and K. C. Jones. 1998. Dioxins and furans in air and deposition: A review of levels, behaviour and processes. Science of the Total Environment 219(1):53-81.
McGrady, J. K., and S. P. Maggard. 1993. Uptake and photodegradation of 2,3,7,8-tetrachlorodibenzo-p-dioxin sorbed to grass foliage. Environmental Science and Technology 27:343-350.
Miller, G. C., V. R. Hebert, M. J. Miille, R. Mitzel, and R. G. Zepp, R.G. 1989. Photolysis of octachlorodibenzo-p-dioxin on soils: production of 2,3,7,8-TCDD. Chemosphere 18:1265-1274.
Muir, D. C. G., A. L. Yarechewski, R. L. Corbet, G. R. B. Webster, and A. E. Smith. 1985. Laboratory and field studies on the fate of 1,3,6,8-tetrachlorodibenzo-p-dioxin in soil and sediments. Journal of Agricultural and Food Chemistry 33:518-523.
Nhu, D. D., T. Kido, R. Naganuma, N. Swano, K. Tawara, M. Nishijo, H. Nakagawa, N. N. Hung, and L. T. H. Thom. 2009. A GIS study of dioxin contamination in a Vietnamese region sprayed with herbicide. Environmental Health and Preventive Medicine 14:353-360.
Pavlov, D. S., A. V. Smurov, L. V. Il’yash, D. N. Matorin, N. A. Kluyev, S. V. Kotelevtsev, V. S. Rumak, and T. G. Smurova. 2004. Present-day state of coral reefs in Nha Trang Bay (Southern Vietnam) and possible reasons for the disturbance of habitats of scleractinian coral. Russian Journal of Marine Biology 30(1):43-50.
Persson, N. J., L. T. Cousins, J. Molvaer, D. Broman, and K. Naes. 2006. Modeling the long-term fate of polychlorinated dibenzo-p-dioxins and furans (PCDD/Fs) in the Grenland Fjords, Norway. Science of the Total Environment 369:188-202.
Piazza, R., M. Sprovieri, M. L. Feo, R. Zangrando, M. Vecchiato, L. G. Bellucci, S. Guiliani, M. Frignani, N. H. Cu, and E. Marsella. 2007. PCDD/F hydrocarbons and pesticides in sediments of the Tam Giang-Cau Hai Lagoon, Central Vietnam. Journal of Marine Science and Technology (Vietnam) 1:102-109.
Podoll, R. T., H. M. Jaber, et al. (1986). Tetrachlorodibenzodioxin—Rates of volatilization and photolysis in the environment. Environmental Science & Technology 20(5):490-492.
Quinh, H. T., L. C. Dai, and L. T. H. Thom. 1989. Effects of geographical conditions, soil movement and other variables on the distribution of 2,3,7,8-TCDD levels in adipose tissues from Vietnam—Preliminary observations. Chemosphere 18(1-6):967-974.
Ross, J., and M. Ginevan. 2007. Points for the committee to consider when evaluating the Stellman model. Presentation to the IOM Committee on Making the Best Use of the Agent Orange Exposure Reconstruction Model. May 1.
Sauter, J. J., H. Kammerbauer, L. Pambor, and B. Hock. 1987. Evidence for the accelerated micromorphological degradation of epistomatal waxes in Norway spruce by motor vehicle emissions. European Journal of Forest Pathology 17(7):444-448.
Schecter, A., B. D. Eitzer, and R. A. Hites. 1989a. Chlorinated dioxin and dibenzofuran levels in sediments collected from rivers in Vietnam, 1984-6. Chemosphere 18(1-6):831-834.
Schecter, A., H. Y. Tong, S. J. Monson, and M. L. Gross. 1989b. Levels of 2,3,7,8-TCDD in silt samples collected between 1985-86 from rivers in the north and south of Vietnam. Chemosphere 19(1-6):547-550.
Schuler, F., P. Schmid, and C. Schlatter. 1998. Photodegradation of polychlorinated dibenzo-p-dioxins and dibenzofurans in cuticular waxes of laurel cherry (Prunus laurocerasus). Chemosphere 36(1):21-34.
Sinkkonen, S., and J. Paasivirta. 2000. Degradation half-life times of PCDDs, PCDFs and PCBs for environmental fate modeling. Chemosphere 40(9-11):943-949.
Sommerfreund, J. K., N. Gandhi, M. L. Diamond, C. Mugnai, M. Frignani, G. Capodaglio, M. Gerino, L. G. Bellucci, and S. Giuliani. 2010. Contaminant fate and transport in the Venice Lagoon: Results from a multi-segment multimedia model. Ecotoxicology and Environmental Safety 73(3):222-230.
Stellman, S. D., and J. M. Stellman. 2004. Exposure opportunity models for Agent Orange, dioxin, and other military herbicides used in Vietnam, 1961-1971. Journal of Exposure Analysis and Environmental Epidemiology 14(4):354-362.
Stellman, J. M., S. D. Stellman, R. Christian, T. Weber, and C. Tomasallo. 2003. The extent and patterns of usage of Agent Orange and other herbicides in Vietnam. Nature 422(6933):681-687.
Su, Y., F. Wania, Y. D. Lei, T. Harner, and M. Shoeib. 2007. Temperature dependence of the air concentrations of polychlorinated biphenyls and polybrominated diphenyl ethers in a forest and a clearing. Environmental Science & Technology 41(13):4655-4661.
Teske, M. E., S. L. Bird, D. M. Esterly, T. B. Curbishley, S. L. Ray, and S. G. Perry. 2002. AgDRIFT (R): A model for estimating near-field spray drift from aerial applications. Environmental Toxicology and Chemistry 21(3):659-671.
Ton, T. C. 1996. Vietnam soil. Hanoi, Vietnam: Agricultural Publishing House. Turunen, M., and S. Hattunen. 1991. Effects of simulated acid rain on the epicuticular wax of Scots pine needles under northerly conditions. Canadian Journal of Botany 69:412-419.
van Jaarlsveld, J. A., and M. A. A. Schutter. 1993. Modeling the long-range transport and deposition of dioxins; first results for NW Europe. Chemosphere 27:131-139.
Wania, F., and M. S. McLachlan. 2001. Estimating the influence of forests on the overall fate of semivolatile organic compounds using a multimedia fate model. Environmental Science & Technology 35(3):582-590.
Ward, C. T., and F. Matsumura. 1978. Fate of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) in a model aquatic environment. Archives of Environmental Contamination and Toxicology 7:349-357.
Young, A. L. 1983. Long-term studies on the persistence and movement of TCDD in a natural ecosystem. Environmental Science Research 26:173-190.
Young, A. L. 2009. The history, use, disposition and environmental fate of Agent Orange. New York, NY: Springer.
Zhang, H. J., J. P. Chen, Y. W. Ni, Q. Zhang, and L. Zhao. 2009. Uptake by roots and translocation to shoots of polychlorinated dibenzo-p-dioxins and dibenzofurans in typical crop plants. Chemosphere 76(6):740-746.