1
Corrosion—Its Influence and Control
INTRODUCTION
With much of the world’s population living in close proximity to water and humidity, corrosion of metallic materials has been an inevitable part of the human experience. While the oxidation of iron (rust) is the most easily identified form of corrosion, this oxidation process represents only a fraction (albeit substantial) of material losses. Today, the impact of corrosion on society and the associated degradation of materials are far reaching owing in part to the increased complexity and diversity of materials systems, which include not only metallic materials but also ceramics, polymers, and composites, which are subject as well to environmental extremes. While legacy corrosion concerns remain, advancing technology and the need for global sustainability bring with them new and emerging corrosion issues whose negative impacts must be minimized through appropriate materials selection, mitigation and monitoring, and new materials development. See Figure 1.1 for an example of multiple simple mitigation efforts.
The impacts of corrosion are often described in economic terms. Financial losses have been assessed in several studies which concluded that premature materials degradation costs industrialized nations approximately 3 percent of their gross domestic product (GDP).1 In the United States it is estimated that between

FIGURE 1.1 A mooring ring, shackle, and thimble with rope illustrate three different techniques for combatting the effects of materials degradation,. Originally, the ring and eye bolt were painted, the shackle and thimble were galvanized (zinc coated), and the mooring line was made of nylon. Courtesy of Erik Svedberg.
$2 trillion and $4 trillion are lost to corrosion each decade—on a relative scale, this amount equates to the cost of repairing the infrastructure damage of three or four hurricanes as large as Hurricane Katrina, which caused massive damage in New Orleans, southern Mississippi, and Alabama.
However, the true costs of corrosion to society are even more pervasive and, in practice, difficult to compile. Several studies, including a recent National Research Council (NRC) report on corrosion education,2 have described both the economic impacts of corrosion and the less measurable impacts such as loss of readiness—that is, the nation’s ability to respond militarily or otherwise to emergencies or other situations involving national security. For example, while the maintenance and replacement costs associated with the corrosion of military systems can be
|
2 |
National Research Council, Assessment of Corrosion Education, The National Academies Press, Washington, D.C., 2009, available at http://www.nap.edu/catalog.php?record_id=12560. |
readily estimated, the dollar costs associated with the military’s inability to respond promptly to a national emergency are difficult to capture directly. Similarly, while the costs of replacing deteriorating bridges and highway infrastructure can be estimated—including the impacts on national productivity and security brought about by failures and traffic congestion during repairs—such estimates require assumptions that are subject to considerable judgment.
Corrosion can affect public health, the environment, and global sustainability in ways that cannot be quantified simply in terms of GDP loss. The deterioration of an early generation of medical devices and implants resulting from interactions with human body fluids, the leaching of corrosion products into the environment, and the weakening of the nation’s energy and transportation infrastructures all have impacts that greatly exceed those that are purely financial. The NRC report Assessment of Corrosion Education3 discusses the broader impact of corrosion and educational challenges in greater detail (Figure 1.2).
Interestingly, the physical processes that cause materials to degrade may be harnessed for society’s benefit. For example, the fabrication of semiconductor devices relies on a variety of etching, deposition, and oxidation processes often operating at the nanometer level. The ability to precisely control the rates and extent of these processes is critical to that fabrication. Corrosion-associated processes are also relevant to other technologies, both in terms of routes by which to synthesize materials and as a means to understand their performance from a mechanistic standpoint. Examples of materials issues include biodegradability and recycling, battery design and development, nanoporous metals for catalysis and sensing, and fuel cells and gas separation membranes. State-of-the-art corrosion research therefore has the potential not only to contribute significantly to many other fields of science and engineering but also to enable them.
TYPES OF CORROSION
Corrosion has historically been defined as the destructive oxidation of metallic materials. More recent definitions have described corrosion as the degradation of any material and its attendant loss of function by exposure to and interaction with its environment. The committee, mindful of the increased application of nonmetals in important structural applications, chose to define corrosion in the following broader context: Corrosion is the environmentally induced degradation of a material that involves a chemical reaction. Mechanical degradation mechanisms, such as creep, wear, and fatigue, are not considered to be corrosion, although corrosion processes may accelerate these degradation modes. Worth mentioning at this point
|
3 |
National Research Council, Assessment of Corrosion Education, The National Academies Press, Washington, D.C., 2009, available at http://www.nap.edu/catalog.php?record_id=12560. |
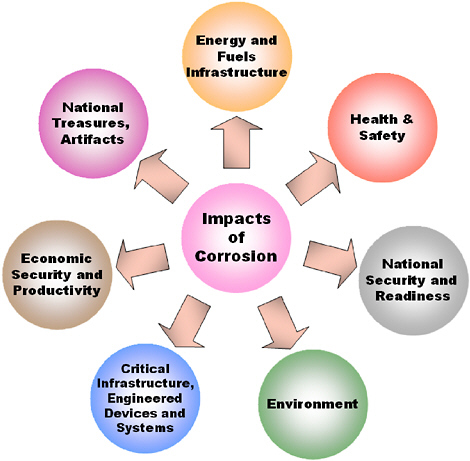
FIGURE 1.2 Corrosion affects nearly every aspect of modern society. In many of these areas, however, its impact is difficult to quantify.
is hydrogen embrittlement, the process by which various metals and alloys become brittle and crack following exposure to hydrogen. Hydrogen embrittlement or hydrogen cracking is often the result of the unintentional introduction of hydrogen into susceptible metals and alloys during formation or finishing operations. The leading types of corrosion are outlined in Box 1.1.
It should be pointed out here that corrosion processes often involve multiple conjoint effects. Rarely does a single mechanism or event drive corrosion; rather, a number of events combine to produce severe effects. Thus, we must keep in mind that corrosion processes usually occur in the context of other factors (loads, wear, crevices, temporally and spatially varying environments, etc.). One such combination of factors can lead to mechanically assisted corrosion in total hip replacements:
|
BOX 1.1 Types of Corrosion Metallic Corrosion Uniform, or “general,” corrosion Dealloying Pitting Crevice related Intergranular Filiform Corrosion by high-temperature gases (oxidation,1 sulfidation, chlorination, etc.) Deposit-induced corrosion, which includes “hot corrosion” Galvanic mechanically assisted corrosion Stress corrosion cracking Corrosion fatigue Fretting corrosion Tribocorrosion Erosion corrosion Hydrogen embrittlement, hydrogen-induced cracking, and hydrogen attack Nonmetallic Corrosion Recession of ceramics in the presence of water vapor or volatile compounds Diffusion-controlled leaching (ion exchange) in glass Glass network hydrolytic dissolution Ultraviolet degradation of organic coatings and polymers Hydrolysis Radiolysis Autocatalytic (acid-driven) degeneration Metal-ion-induced oxidation |
-
Wear (typically fretting),
-
Passive oxide abrasion,
-
Repassivation,
-
Hydrogen ion generation, and
-
Crevice environments.
It is the biological inflammatory response to these processes that generates aggressive oxidative environments in vivo (generation of reactive oxygen species, etc.) that combine to lead to severe attacks on medical alloys. Ti alloys can become subjected to pitting in vivo, and Co-Cr-Mo alloys can undergo penetrating intergranular corrosion as well, leading to implant fatigue and fracture.
Another kind of corrosion sustained by medical implants is the metal-ion oxidation sustained by polyurethane pacemaker leads. This failure mode is highly complex and involves water transport across the insulation of the lead, allowing contact with the Co-Cr alloy, which then corrodes by a fretting mechanism. The metal ions, in particular Co2+, then penetrate the polyurethane and oxidize it in a catalytic fashion. This leads ultimately to an electrical breach in the lead and failure of the pacemaker, often resulting in the death of the patient.
EXAMPLES OF CORROSION MITIGATION CHALLENGES
Corrosion may be inevitable, but there are ways to retard it—that is, to slow the kinetics of deterioration. The mitigation strategy for a material must be tailored to the environment and to the composition and structure of the material. Some materials are inherently slow to corrode, especially in the absence of oxygen; others corrode slowly by forming layers of protective corrosion product (Box 1.2).
Materials with different properties will require different mitigation strategies:
-
Metals and alloys without intrinsic corrosion resistance. Such materials can corrode in otherwise innocuous waters or atmospheres, when dissolved oxygen is present or in which water can be reduced to generate free hydrogen. These metals and alloys usually need to be actively protected. Alloys such as low-carbon steels can be used in thick sections to accommodate the loss of material.
-
Passive metals and alloys. Usually alloys such as stainless steel or nickel-chromium can be used unprotected in innocuous environments and in a certain range of aggressive environments such as seawater or mild acids, depending on the content of alloying elements. Superpassive metals—such as tantalum, which resists strong hydrochloric acid—also exist but are considerably more expensive. The main issue with passive metals is their propensity for localized—rather than uniform—corrosion.
-
Copper-based materials. Owing to the thermodynamic immunity of copper, corrosion is normally slow or absent unless oxygen or another strong oxidant is present. Aqueous sulfide solutions are an exception. When oxygen is present or in the presence of acid rain, these materials may react like other nonpassive metals (see Figure 1.3).
-
Certain high-strength alloys. These alloys can survive at very high temperatures because they form surface layers that protect against oxygen in the application environment or because they are given metallic coatings that perform this function.
-
Glass and ceramics. Such materials are affected not by electrochemical processes but mainly by simple dissolution of the material. One way of protecting against the corrosion of glass is to add lime: added to soda-glass it reduces solubility in water.
|
BOX 1.2 Survivor In his 2007 book The World Without Us,1 Alan Weisman suggests that copper and its alloys are the structural materials most likely to survive for thousands of years in a world suddenly depopulated of human beings. This conclusion, based on a strictly thermodynamic criterion (copper being the most noble structural metal and sometimes found uncombined in nature), may not be correct, however, given that some invented materials, like stainless steel, will also endure for thousands of years because their surface is protected by passivating oxide films. |

FIGURE 1.3 Bronze statue with a protective layer of patina created by slow chemical alteration of the copper content, producing a basic carbonate. The statue has been exposed to the coastal weather outside the city hall of Stockholm. Courtesy of Erik Svedberg.
-
Polymers. Degradation is due to a wide array of physiochemical processes. One common problem is swelling, where small molecules infiltrate the structure, reducing strength and stiffness and causing a volume change. Conversely, many polymers are intentionally swelled with plasticizers, which can be leached out of the structure, causing brittleness or other undesirable changes. The most common form of degradation, however, is a decrease in polymer chain length. The mechanisms that break polymer chains are ionizing radiation (most commonly ultraviolet light), free radicals, and oxidizers such as oxygen, ozone, and chlorine. Additives as simple as a UV-absorbing pigment (i.e., titanium dioxide or carbon black) can slow these processes very effectively.
Mitigation techniques can be roughly classified as either active or passive. Examples of active mitigation techniques include inhibitors, external cathodic protection, with or without coatings, and sacrificial anodes. Passive techniques include material selection, organic and inorganic coatings, and metallic coatings (including both barrier and sacrificial coatings). One other example of mitigation is control of the environment (oxygen, ions, etc.) such as is sometimes done in boilers when oxygen and ions are removed while inhibitors (amines) are added. The decision to use one technique or a combination of techniques depends on the type of corrosion that is expected, the tolerance for risk, the cost of the technique, the material, the environment, and other factors related to the design of a structure, such as accessibility and size.
The committee assembled and distributed a questionnaire focused on corrosion mitigation in order to better understand the current concerns and problems. The questionnaire, which was made available to key personnel at DOD, MTI, and LMI; the NACE Technical and Research Activities Committees; and directly through the ROCSE Web site, gathered information on the respondents’ backgrounds; types of corrosion that were of greatest concern; types, costs, and efficacies of mitigation systems employed; and idealized mitigation systems. Respondents were also given the opportunity to suggest scientific advances that could lead to new and/or better mitigation technologies. Almost 200 people from a wide variety of industry sectors responded to the questionnaire. The majority of the respondents had been involved with corrosion mitigation for more than 15 years managing assets valued at more than $10 million each. A variety of corrosion mechanisms were cited as being troublesome to the respondents, with pitting corrosion the biggest single problem. What most concerned the respondents was safety. Many corrosion mitigation strategies were relied on by the respondents, the most frequent being material selection, monitoring/inspections, external cathodic protection with coatings, organic coatings, and inhibitors. A majority of respondents reported having spent more than $200,000 per year on corrosion mitigation.
Overall, the respondents were at least moderately satisfied with their choice of mitigation technique. In responses to questions about difficulties encountered with the different techniques, some areas of concern became apparent, including sensors for measuring localized corrosion, remote sensing, protection under disbonded coatings, surface preparation requirements for coatings, lack of training, and lack of reliable real-time models to predict lifetimes and damage mechanisms. When asked where future mitigation research should be focused, most questionnaire respondents suggested monitoring and modeling (especially remote monitoring or monitoring of localized corrosion and modeling for lifetime prediction and for new alloy performance), coatings (especially for high-temperature applications or to reduce the need for surface preparation), and development of active systems.
The responses are given in more detail in Appendix B, “Results of the Commitee’s Corrosion Mitigation Questionnaire.”
SUCCESS STORIES FROM CORROSION RESEARCH
The success stories that follow are not exhaustive. Rather, they were selected to illustrate the impact of advances in science and engineering—emerging tools, analytical approaches, and new materials design, synthesis, and processing—on strategies for material and operational solutions to environmentally induced degradation. These advances have historically helped the community develop an underlying scientific understanding as well as the technological means to mitigate corrosion. The committee is confident that the community will continue to pursue these goals and felt that it was not necessary to arbitrarily limit (or to underestimate) the imagination of researchers by specifying which developments and techniques should be pursued, particularly given the vast range of related phenomena. Rather, historical examples are contained in many of these success stories, which, additionally, offer a glimpse into why future advances hold promise for greater progress in combating corrosion and also illustrate the other factors (such as societal needs, technological drivers, policy, multidisciplinary approaches, and thrusts of critical size) that contribute to successful endeavors to deal with corrosion. The examples begin with general materials development and then move to application-specific success stories.
Corrosion- and Heat-Resistant Alloys
Despite the fact that it is not thermodynamically possible to develop alloys that are totally immune to corrosion, there have been extraordinary developments with respect to heat- and corrosion-resistant alloys in the last century, and—as a result of research efforts—these developments have accelerated over the last few decades. Among the many success stories that are associated with these new mate-
rials, one of the most important has been the development of the modern family of austenitic stainless steels.
As early as 1821, Berthier, based on the work of Faraday and Stodart, produced stainless alloys of iron and chromium.4 However these alloys were extremely brittle and had no structural usefulness. The first useful stainless steels were developed in the beginning of the twentieth century by Monnartz in Germany and Brearley in England,5 but it was not until the 1912-1914 period that the commercial success of these austenitic steels—primarily based on the addition of 18 percent chromium and 8 percent nickel—was first recognized.6
In the 1970s—when strength considerations became an issue for stainless steels—duplex versions were developed, which also increased resistance to chloride stress corrosion cracking. In the 1980s stainless steels with higher molybdenum were formulated to solve problems with localized corrosion encountered in aggressive environments.
This abbreviated history of stainless steels illustrates the successes in the development of iron-based, corrosion-resistant alloys. Developments were accompanied by use of increasingly sophisticated experimental and characterization techniques from advances in allied fields. However, it was also a labor-intensive effort, and the time period—from recognition of the problem, to an understanding of its origin, to the development of the most resistant alloys—was on the order of 80 years. This was hardly an efficient process.
Even with advances in alloy development technologies, it still took almost a quarter of a century to improve the nickel-containing alloys of the 600 family of stainless steels to the more corrosion-resistant versions (e.g., alloys 690, 22, 59, and 2000) that are enabling many applications in extremely aggressive environments found in the chemical industries, nuclear reactors, steam generators, and sour oil and gas production.
The development of heat-resistant alloys was similarly lengthy and inefficient. In the 1970s the protective oxide layers on nickel-based alloys were much improved by alloying them with elements that resulted in a more stable and tenacious layers of alumina versus chromia. It is only now that the concept of alumina protective layers is being applied to more cost-effective and high-temperature iron-based heat-resistant alloys, effectively producing a new class of stainless steels based on the principles of selective oxidation and advanced microstructural control of precipitates for strengthening.7
|
4 |
Louis Kuslan, “Berthier, Pierre,” pp. 72-73 in Dictionary of Scientific Biography, Charles Scribner’s Sons, New York, 1970-1980. |
|
5 |
See “Harry Brearley, 1871–1948,” Tilt Hammer Web site at http://www.tilthammer.com/bio/brear.html. |
|
6 |
New York Times, January 31, 1915. |
|
7 |
Y. Yamamoto, M.P. Brady, Z.P. Lu, P.J. Maziasz, C.T. Liu, B.A. Pint, K.L. More, H.M. Meyer, and E.A. Payzant, Creep-resistant, Al2O3-forming austenitic stainless steels, Science 316:433-436, 2007. |
In the last few decades, amorphous and nonequilibrium alloys processed using heretofore exotic methods (e.g., spun cooled, sprayed, sputter deposited, and laser surface melted) presented the potential for extraordinary advances in the development of corrosion-resistant alloys. Iron- and nickel-based metallic glasses have been developed whose corrosion resistance rivals that of the best conventional nickel-based superalloys in the low-temperature regime.8 This advance was enabled by certain glass-forming elements that allow for the addition of large amounts of traditional corrosion-beneficial alloying elements without detrimental effects; the emergence of metallic glass composites; and the benefits of selected minor alloying elements. In addition, multifunctional amorphous and semiamorphous alloys9 that offer tunable barrier, sacrificial, and chemical-inhibiting capabilities have also been produced. While these materials had long been considered impractical, laser surface treating used by heavy equipment manufacturers has enabled mass production of coatings and bulk metallic glasses and demonstrated routes to practical processing technologies that can produce significant improvements in corrosion protection.
What’s Next for Corrosion- and Heat-Resistant Materials?
The emergence of the metallic glasses and alumina-forming stainless steels as potentially highly corrosion-resistant materials is only one example of the progress that is continuing to be made. Both show ways in which lessons learned, new materials developments, the incorporation of modern tools into research activities, and growing understanding of the relationships between structure, materials behavior, and component design can speed the development of such alloys. Key challenges remain, however, in the design or specification of materials for targeted lifetimes in particularly aggressive environments.
Conclusion (Corrosion- and Heat-Resistant Materials)
The most impressive corrosion-resistant alloys in the last half century began to be developed by metals producers. Work on amorphous metals and advanced surface treatments have been funded by university-led efforts or consortia of universities and companies. One area where industry has taken the lead recently is corrosion-resistant rebar materials where cost-effective stainless grades are being
sought. Unfortunately this work is often not at a fundamental level but instead is aimed at achieving engineering performance without also understanding the scientific underpinning.
The development of corrosion-and heat-resistant alloys over the past century has had huge economic, environmental, and safety impacts. However, as ever greater demands are imposed on materials performance, it will be necessary to come up with new materials at an even faster pace. Given recent and continuing advances in material types, characterization techniques, alloy modeling, and an understanding of fundamental corrosion and kinetic processes, the committee believes it will be possible to rapidly evolve new materials with improved corrosion and heat resistance that are more closely integrated into design for specified lifetimes in particular environments.
Motor Vehicles
Some decades ago, it was common for automobile bodies to rust through within a few years of manufacture, especially where roads were heavily salted. Paint systems failed, pinholes in chrome plating led to the destructive corrosion of fenders, and exhaust systems had to be replaced regularly. Newer cars now come with an extended anticorrosion warranty, and the useful lifetime of a car is more often limited by the mechanical and electrical components than by the external body. Driven in part by competition between manufacturers and regulations against corrosion perforation by the Canadian government, this change has been facilitated by new protective coatings and coating application processes, more corrosion-resistant structural materials, and the incorporation of best design practices. The first company to implement zinc coatings in the automotive industry was Chevrolet, which used it on rocker panels. At the same time Porsche introduced zinc coatings on the steel frames of its cars, and other automotive companies soon followed. Today, these state-of-the-art, multilayer coating systems are not only long lasting but also more environmentally friendly and have resulted in a huge savings to consumers.
Car body panels are now routinely fabricated from two-sided galvanized steel, which provides considerable protection against corrosion. The paint primer layer, universally applied by the cathodic electrodeposition process—in combination with advanced metal pretreatments and galvanizing—results in an almost defect-free, highly protective coating system. Similarly, exhaust systems are made from relatively inexpensive but long-lasting stainless steel, while chrome trim has either been eliminated or galvanically isolated.
Polymeric materials have replaced metals in car bumpers and fenders for both corrosion resistance and weight reduction. To accomplish this, ultraviolet (UV) radiation- and heat-resistant polymers were required. Enhanced resistance to UV
degradation also has been incorporated into the protective coatings on the whole car body.
What’s Next for Motor Vehicles?
Corrosion science accomplishments continue to impact automotive design. For instance, altered oxide semiconducting properties in new zinc-magnesium alloys with lower self-corrosion rates promise improved lifetimes for the sacrificial galvanic layer. Even extremely corrodible metals such as magnesium are being used for weight savings with no detrimental consequences. In a DOE-supported collaboration with organizations abroad, auto manufacturers in the United States are now designing a car with a front end made completely of magnesium alloy, which is possible owing to advanced surface treatments and an understanding of galvanic isolation. There is also intense work on aluminum/magnesium engine blocks. Corrosion mitigation for these materials will certainly need the attention of the corrosion community.
Conclusion (Motor Vehicles)
Automobiles are an unambiguous example of the successful application of corrosion science and engineering to increase useful service life of an everyday item. Progress has been driven by competition, consumer demand, and regulation, supported by advances in materials and coatings. The trend to more efficient vehicles and the need for lighter materials is once more challenging the community to develop materials that resist corrosion.
Aging Aircraft Airframes
The problem of aging aircraft came to the forefront in the mid-1990s, largely as the result of the report of an Air Force Blue Ribbon Panel.10 The Air Force had a number of aircraft that were young in terms of fatigue cycles—the typical measure of aircraft age—but old in terms of years since construction. As such, tremendous cost and effort have been required to maintain these airplanes.11
Related to this problem was the reliance on the environmentally undesirable, chromate-based corrosion inhibitors that were incorporated into the coatings and
|
10 |
National Research Council, Aging of U.S. Air Force Aircraft: Final Report, National Academy Press, Washington, D.C., 1997, available at http://www.nap.edu/catalog.php?record_id=5917. |
|
11 |
For U.S. Air Force aircraft, see National Research Council, Aging of U.S. Air Force Aircraft: Final Report, National Academy Press, Washington, D.C., 1997, available at http://www.nap.edu/catalog.php?record_id=5917. |
primers used to protect the corrosion-susceptible, high-strength aluminum-copper alloys used in many aerospace applications. Furthermore, few of the fundamental corrosion characteristics of these alloys were known. For these reasons, the Air Force panel identified the need to simultaneously reduce maintenance corrosion costs and replace chromate with a more effective, environment-friendly inhibitor.
To address these issues, the Air Force Office of Scientific Research funded a number of research programs at universities. Multidisciplinary university research initiatives were initiated to study chromate inhibition mechanisms and nondestructive testing. This funding motivated other researchers and funding agencies to get involved in the field, and the number of people working in corrosion-related investigations increased substantially within a few years. At the same time, a special office integrated corrosion management into the Air Force’s structural integrity program, so that the impact of corrosion on aircraft was addressed in considerable quantitative detail.
This field was active for about 8 years, from the late 1990s into the current decade, and led to considerable new understanding of the mechanisms of high-strength aluminum alloy corrosion and inhibition. However, many of these suggested improvements have yet to be implemented. Implementation of the new technologies and science developed in these studies has been delayed because of inadequate or overly time-consuming qualification test methods, most of which are qualitative rather than quantitative.
Conclusion (Aging Aircraft Airframes)
Understanding of aluminum alloy corrosion and mitigation was advanced by large multi-institutional efforts funded by government agencies and a need to address specific technological problems. However, changes in government priorities prevented the research successes from being fully developed into practical solutions.
Pipelines
More than 2 million miles of pipeline in the United States are used to transport natural gas, crude oil, petroleum products, and other petrochemicals economically and efficiently over long distances. These steel pipelines can be subjected to corrosive conditions both internally (from the aggressive fluids being transported) and externally (from the aggressive soil or subsea environments). The catastrophic failure of an oil or gas pipeline can result in loss of life and environmental disasters. In United States, more incidents/accidents in pipeline systems are due to human intervention, followed by external and internal corrosion.12 The U.S. Depart-
|
12 |
Available at http://www.phmsa.dot.gov/. |
ment of Transportation’s Pipelines and Hazardous Materials Safety Administration (PMHSA) has been implementing regulations requiring new inspection methods and specific inspection intervals. These inspections, in conjunction with better coatings and cathodic protection systems, have been instrumental in preventing failures due to external corrosion. The PMHSA reported that serious incidents on pipelines have been reduced by more than 50 percent since 1989.
The demonstrated reliability of these pipelines has required the use of several different corrosion protection schemes. To reduce attacks on the outer surfaces of pipelines, improved cathodic protection systems and coating materials (fusion-bonded epoxy, which replaced coal tar systems) have been used along with better inspection programs.
For high-pressure natural gas transmission lines, the most feared hazards are two forms of external stress corrosion cracking: One is associated with failure of cathodic protection to penetrate to the base of a coating defect, and the other is associated with near-neutral water that has equilibrated with the soil. Decades of research using ever more sophisticated tools have pinpointed the stress and environment criteria for both kinds of stress corrosion cracking, leading to more efficient monitoring, protection, prediction, and, ultimately, mitigation.
What’s Next for Pipelines?
Although significant strides have been made in the pipeline industry, future challenges do exist. For example, the corrosion science and engineering related to the transport and storage of biofuels (such as ethanol) are largely unknown. Another potentially important material system is supercritical carbon dioxide (SCCO2) for transport in dedicated pipelines. This system will be significant if carbon capture and sequestration processes begin to replace fossil fuel for use in power plants, and it may present challenges for corrosion protection as the cost of purification/separation processes for SCCO2 grows.13 Although successful, the current approaches used in protection systems may not be sufficiently robust to support newer fuels, increasingly aggressive environments, and/or longer pipelines, and better schemes or new material compositions with fundamentally better corrosion resistance will likely be required. Internal corrosion due to hydrogen sulfide, carbon dioxide, and biological factors is still poorly understood. More recently, important spills that threatened the pristine environment in Alaska and the Gulf of Mexico are believed to have been caused by internal corrosion. Localized internal corrosion is difficult to evaluate by the use of “smart pigs,” but no accurate predictive models to estimate pipeline life have yet been formulated.
Technology for carbon capture, transportation (in pipelines), and storage in various types of underground geologic reservoirs is also viewed as an important for mitigating CO2 emissions. Long-term containment of buried CO2 is essential to the successful mitigation of CO2 emissions. Supercritical CO2 is injected into a reservoir, where it is contained by natural formations that are essentially impermeable to CO2. However, leakage pathways could allow the escape of CO2 into subsurface aquifers or to the atmosphere. Because dissolved CO2 creates acidic conditions, drinking water systems could become contaminated by the leaching of species from host rock. Such leakage could occur principally from the injection well or from other wells that intersect the formation at various depths as the result of earlier oil and gas drilling. Injection wells are reasonably well engineered, but the earlier boreholes are not always in good condition. At present, there are hundreds of thousands of boreholes in the United States in various stages of disrepair. Predicting the long-term (hundreds to thousands of years) performance of these boreholes requires a combination of fundamental electrochemical models and experimental data. Such data in supercritical CO2 or in multiphase liquid CO2-aqueous environments are not easily available. Furthermore, emissions from various industries will introduce additional contaminants, even after the CO2 has been purified in the capture processes. Some data show that these impurities can cause corrosion. The experience gained by the corrosion community in nuclear waste container life prediction could be utilized in CO2 containment, but unique challenges remain for mitigating corrosion in high-pressure CO2-water systems.
Conclusion (Pipelines)
Environmental protection and safety regulations have driven the need to assure pipeline integrity and have led to the introduction of new materials and coatings—including nonmetallics—and the application of corrosion protection principles. A fundamental understanding of corrosion and cathodic protection effects has had and will have an important part in assuring pipeline integrity under aggressive conditions, particularly as innovative material systems (perhaps adapted from other application areas) are used more widely and advances in tools for modeling and simulation are realized. Naturally, there are many other pipeline and storage issues to address in the future, such as water mains, sprinkler valves, gas tanks, double-walled tanks for underground gas storage, and hydrogen pipelines.
Medical Devices
In the early days of the medical device industry, there were significant problems with corrosion and the general degradation of materials in the aggressive
environment of the human body. However, by leveraging materials advances in other industries—such as the corrosion-resistant materials used for aerospace—developers have been able to make good progress in this field.
For example, in pacemakers, the electronics and the power source—two of the most vulnerable components of the device—are protected inside a laser-welded, hermetically sealed titanium enclosure, which isolates them from the hostile environment. Electrical connections outside the device are made via corrosion-resistant glass or ceramic feedthroughs that are further isolated from the body by protective layers of well-adhering materials (e.g., special epoxies). This sophisticated sealing technology did not exist in the early days of the industry, when corrosion resulted when water entered the epoxy potted devices. Other implantable devices such as stents, orthopedic joints, and spinal appliances were originally made of stainless steels. Sometimes corrosion was observed in such implants, especially if the materials were not processed optimally. As with pacemakers, better materials, like super-alloys, were borrowed from other industries. The use of materials and technologies designed for other applications allowed the implantation of medical devices to extend and enhance the quality of life for the individuals who received them. This trend continues as new technologies like ion implantation are borrowed and used in more wear-resistant materials such as those for orthopedic applications.
Recently, the concept of controlled corrosion has been proposed as a way to provide implantable devices that can perform a function (e.g., a stent or drug delivery vehicle) and then erode away over time and leave no permanent trace in the body. Some polymeric materials based on this concept have been turned into products, and some magnesium alloys are currently being investigated.
What’s Next for Medical Devices?
The need for better materials for medical device implants is driven by a combination of factors, including (1) increased longevity and expectations of reliability, (2) the increasing complexity of demands on those materials as devices become more complicated, and (3) the need for safety in devices to be used in the human body. Regulatory agencies such as the Food and Drug Administration require that device companies demonstrate the functionality and biocompatibility of their materials and devices (see Figure 1.4). Such demonstration calls for both testing and modeling, but the human body is a complex system and can be difficult to model. Moreover, many of the fundamental questions related to accelerated testing, biokinetic modeling, and acceptance criteria have not been answered. However, if we can improve modeling and testing, and can elicit responses in a controlled and predictable manner, society will greatly benefit.
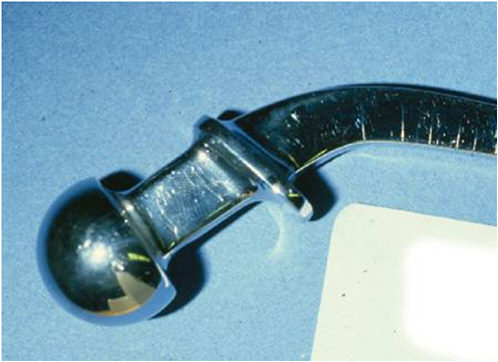
FIGURE 1.4 Femoral replacement part (T28) with stress corrosion cracking. Courtesy of Stanley A. Brown, U.S. Food and Drug Administration.
Conclusion (Medical Devices)
As advances in the understanding of medicine and technology allow for innovative devices and therapies, new and better materials will have to be developed for use in the human body. Up to now most of the materials used in implantable medical devices were developed for other applications and later applied for medical uses because they had some of the desired requirements (strength, flexibility, fatigue resistance, electrical conductance, corrosion resistance). However, the cost of developing and qualifying materials for implantation is often not very attractive for the many companies. Government funding for R&D in implantable medical devices is especially desirable in this area. Current understanding of how biology and materials interact is still quite primitive. To truly design new medical devices from first principles requires a far better understanding of the interface of materials and biology.
Nuclear Reactor Systems
Commercial and military nuclear reactors have experienced a wide range of corrosion problems over the past 55 years, and—starting in the 1960s—corrosion research has been put to good use to mitigate and solve those problems. All operating plants in the United States use ordinary light water with few impurities but have nevertheless been surprisingly susceptible to corrosion.
In boiling water reactor systems, the dominant problem had been intergranular stress corrosion cracking (SCC) in 304 austenitic stainless steel sensitized by welding, which was causing crippling levels of plant outage. This situation was transformed by the recognition that the cracking could be controlled by modifying the water chemistry and the surface condition of the wetted surfaces of the plant.
First, the impurity content of the water was reduced; then, hydrogen was introduced, at undesirably high levels, to lower the corrosion potential of the steel. Then, in a classic application of corrosion science, it was shown that the wet deposition of noble metals on the plant surfaces could achieve a similar level of protection with much lower hydrogen levels. All this practical mitigation was supported by extensive corrosion science research, including the development of in situ probes for monitoring hydrogen content, electrode potential, and crack growth rate.
In pressurized water reactors, corrosion problems have been associated mostly with steam generators, where nickel-based alloy 600 was originally used for the tubing that separated the primary reactor coolant water from the water that is boiled to drive the turbines. This proved to be a bad choice because the material was susceptible to SCC from both sides. However, remedial measures—including heat-treating the tubing material, retubing with a new alloy, and reducing the deposition of sludge arising from impurities in the feed water—prolonged the life of the steam generators.
In the process of this mitigation, superb research in support of critical issues was done in metallurgy, chemical engineering corrosion science, and even geochemistry. Now, the industry is in the position that it can—with fair confidence—predict extremely long life for its new plants as well as exceed the life extension targets for refurbished plants.
Both kinds of nuclear plants have experienced SCC problems of neutron-irradiated material in the core, and some of the most ambitious corrosion research of the past two decades has dealt with the resulting blend of material property alteration, microstructure, and SCC behavior using advances in modeling and characterization. As a result of experimentation and basic research, material life can be predicted more accurately, and recommendations exist for new alloys with enhanced resistance to this specialized form of SCC. The ultimate goal is quantitative prediction of life once the corrosion and degradation mechanisms have been fully understood.
Models and real-time information measured at the plants allow the plant life to be extended while still operating within technical specifications. This real-time information provides a deeper understanding of plant behavior and leads to improved plant performance. This multitiered approach of using modeling, sensors, and plant information allowed researchers to solve the problem of corrosion-triggered activity transport around the primary cooling circuit, leading to a buildup of radiation fields outside the core. Several models, in conjunction with plant data, are used around the world to predict the accumulation of those unwanted radiation fields.14 The models take into account that the water chemistry of pressurized water reactors is affected by water radiolysis, chemical reactions, and convection of the injected chemicals like hydrogen gas, boron, and lithium (to maintain the proper percent hydrogen). Particle deposition is modeled around the entire primary coolant loop. Such modeling and monitoring are used to assure that exposure of plant personnel to radiation can be kept at levels that meet regulatory standards.
The “chemistry control” of primary and secondary coolant fluid has become a mature, integral part of nuclear reactor corrosion control, because water chemistry and the oxidizing power of the fluids are key factors in corrosion initiation phenomena and propagation rates. Water chemistry, potential, and redox electrode sensors have all been developed that enable the verification of key water chemistry variables and their manipulation in various ways to reduce corrosion. Moreover, damage sensors have been developed to help verify model predictions and confirm the beneficial effects of water chemistry “housekeeping” on rates if stress corrosion propagation. These advances in sensor and measurement technology have been instrumental in programs to extend reactor service life and to reduce reactor downtime. These measurements and others are crucial to the ability to predict the lifetime of existing and new nuclear power reactors and corrosion in both.
What’s Next for Nuclear Reactor Systems?
New designs and new types of advanced nuclear power plants (e.g., supercritical steam) present the same need for in-depth understanding of corrosion processes and the associated means to control corrosion’s detrimental effects. Indeed, for new plant concepts, knowledge gained from current R&D should be used prescriptively (rather than remedially and reactively, as in earlier generations of plants) in the design of components and control systems to avoid operational problems from
corrosion and environmental effects. Indeed, as water-cooled reactors are driven to higher efficiencies and burn-ups and stricter limits on safety performance, corrosion challenges will only multiply and the environments to which the materials will be subjected become more extreme. Then there are the formidable corrosion challenges in advanced (generation IV and beyond) reactor systems. Supercritical water poses extreme risks of stress corrosion failure, especially in pressure vessel designs (as opposed to the CANDU-like design), and water radiolysis in such conditions is not well enough understood to provide a baseline for definition of the environment. Neutron irradiation complicates such assessments even further. Developments in high-temperature gas-cooled systems will be plagued by corrosion issues due to impurities in the helium gas, and other advanced designs using liquid metals will be subject to various forms of degradation, including corrosion.15,16
Conclusion (Nuclear Reactor Systems)
In response to regulatory and economic pressures, some of the best and most critical successes in corrosion science and engineering have come about by solving a host of initially unanticipated problems related to the continued safe operation of nuclear plants.
Radioactive Waste
Disposal of high-level nuclear waste is a worldwide issue. The United States alone has 77,000 tons of high-level nuclear waste, equivalent to one half pound for every citizen. The Federal Nuclear Waste repository was established to isolate this waste for at least 10,000 years without radionuclide release, which will be dependent on the successful choice, fabrication, and total performance of an engineered waste barrier that is resistant, but not immune, to corrosion.
The total system relies on defense in depth through vitrification and immobilization of the nuclear waste within glass, the storage of borated stainless steel baskets, and enclosure in a modern corrosion-resistant alloy. An extraordinary effort has been under way around the world to verify acceptable performance over what had heretofore been unthinkable lifetimes. This has brought about some revolutionary new lines of thinking in the corrosion field—on, for instance, the theory, experiment, and modeling of extremely low corrosion rates; the revisitation of such issues as long-term passivity of alloys in complex environments; and
on critical partnerships with experts in other fields who are able to bring insights from, and advances in, their fields to bear on the problem.
Conclusion (Radioactive Waste)
Unprecedented coupling between corrosion scientists, geochemists, and risk assessment experts can serve as a useful model for any high-priority thrust in corrosion. Our understanding of corrosion, welding, phase stability, and nickel-chromium-molybdenum superalloys has been advanced to levels equal to any other alloy system. While translation to practice awaits policy and political decisions, this research has led to a vast improvement in corrosion science and to technological confidence that an engineered waste barrier can be perfected to contain nuclear waste.
Protective Coatings for High-Temperature Combustion Turbines
Coatings are used in a variety of applications to protect alloys that exhibit attractive properties at high temperatures but would be too reactive in the service environment. For example, aluminum-oxide-forming bond coats provide super-alloys used in the hot sections of gas turbines with oxidation resistance as part of the ongoing drive toward higher efficiencies and better performance characteristics for air-breathing propulsion and land-based power systems.
Alternatively, materials that already exhibit good high-temperature properties and corrosion resistance at the requisite temperatures are desirable. As such, ceramics and ceramic-composite materials such as silicon carbide have been the subject of study and development for a number of years. However, when such composites were targeted for use in combustion turbines, the presence of water vapor at elevated pressures revealed that silicon dioxide is unstable owing to its susceptibility to volatilization. Work at the National Aeronautics and Spaces Administration (NASA) Glenn Research Center and elsewhere conclusively demonstrated the impact of water vapor on silicon carbide recession and produced a robust model that definitively described the recession rates in terms of the important environmental parameters.17 This work also indicated candidate materials that could be used for environmental barrier coatings18 to protect these composites (Figure 1.5) and enabled the development of reliable accelerated testing of can-

FIGURE 1.5 Combustor liner made from SiC-fiber-reinforced SiC (SiC/SiC) composite for the Solar Turbines, Inc., Centaur 50 gas turbine. An environmental barrier coating was applied to the inside circumference of the outer ring and to the outside circumference of the inner ring (the surfaces facing the combustion gas). Inset: Cross section of the coating; BSAS, barium-strontium aluminosilicate; mullite, aluminosilcate; underlying the Si layer is the SiC/SiC composite. Courtesy of Solar Turbines, Inc., and Oak Ridge National Laboratory.
didate coatings.19 Collaborations between NASA, the national laboratories, and industrial partners have led to the development of robust processes for applying state-of-the-art coatings to SiC matrix composites.
What’s Next for Protective Coatings?
The need for even higher engine efficiencies and performance requires materials that show extraordinary stability and resistance to degradation at environmental extremes of temperature, pressures, and reactive species.20 Increasingly robust environmental barrier coatings will be needed, or failures as those described in Box 1.3 will continue to occur. These coatings will require the development of even more advanced mechanistic knowledge and models.
Conclusion (Protective Coatings)
Through government and industry collaborations, a fundamental understanding of the degradation mechanisms associated with high-temperature volatilization of SiC matrix composites, combined with the development of highly reliable coating processes, has paved the way for use of the high-temperature SiC matrix composites in aero propulsion and land-based combustion turbine applications. The ability to use SiC matrix composites in oxidizing environments containing water vapor and other impurities has pushed by 200°C the envelope for the temperature at which this structural material can be used, making it one of the most robust commercial materials for very high temperature applications.
SUMMARY OBSERVATIONS
There has been significant progress in the development of corrosion-resistant materials, understanding of basic mechanisms by which corrosion occurs, and the implementation of mitigation techniques. This progress has allowed for the current state of materials usage in both common and harsh environments. The primary drivers for corrosion research have been industrial and government needs. Addressing these needs has led to advances in science, as well as further practical improvements, which then provide for further scientific advances. Although there is always a possibility for doing more, the government has played a vital role in the field of corrosion research—including challenging industry with critical problems (e.g., related to access to space, nuclear power, reduced maintenance of roads and bridges, and performance of military equipment), funding and conducting research, establishing standards, and prescribing regulatory requirements for safety and health. Nevertheless, the industrial research and development base that led to many of the advances in new materials and understanding of corrosion behavior has eroded, and as such, the government agencies that have responsibilities dependent on the
|
20 |
U.S. Department of Energy, Office of Basic Energy Sciences, Basic Research Needs for Materials under Extreme Environments, report on the Workshop on Materials under Extreme Environments, June 11-13, 2007, available at http://www.er.doe.gov/bes/reports/files/MUEE_rpt.pdf. |
|
BOX 1.3 Corrosion Challenges with Infrastructure Systems Infrastructure systems are meant to safely serve society for long periods. Design codes provide guidance and requirements to limit the corrosion of these infrastructure systems with the objective of ensuring these longer service lives. It is generally not the objective of these codes to prevent all corrosion, but in conjunction with proper inspections to limit the risk of failure resulting from corrosion. However, these guidelines and requirements have not always resulted in long-lasting infrastructure systems. Corrosion in our infrastructure systems results in reduced structural capacities, which can lead to structural failures. Figure 1.3.1 shows the underside of a steel bridge member exhibiting significant corrosion—in this case, good inspection practices revealed the damage, thereby preventing failure. However, corrosion has led to failures (in 1967, corrosion and fatigue of an I-beam on the Silver Bridge resulted in its collapse,1 killing 26 people who were using the bridge. More recently (2000), a pedestrian bridge in Charlotte, North Carolina, failed due to corrosion of the steel reinforcement, injuring 107 people.2 Underground infrastructure systems also exhibit corrosion. Figure 1.3.2 shows a failed cast iron pipe. Water pipe failures throughout the United States have led to closures of subways, roads, and businesses, often temporarily crippling the local economy. In fact, the failure of large-diameter water mains has led to significant flooding and millions of dollars in  FIGURE 1.3.1 The underside of a steel bridge member exhibiting significant corrosion. Courtesy of the Texas Department of Transportation, © 2007. All rights reserved. |
 FIGURE 1.3.2 A failed cast iron pipe. Courtesy of Mark Lewis, East Bay Municipal Utility District. repair costs. The Pipeline and Hazardous Materials Safety Administration (PHMSA) regulates the inspection and incident reporting of pipelines carrying hazardous materials. Increased inspection requirements have led to a 40 percent reduction in serious incidents.3 Although corrosion has resulted in and will continue to result in significant loss of economy, the implementation of sound inspection requirements have proven to reduce serious incidents and failures. With better science and engineering, a more economical solution to corrosion prevention can likely be discovered.
|
materials used will need to consider new options. In addition, despite major differences in corrosion problems as well as solutions from industry to industry, the issue is always there. To ensure that corrosion-related issues are identified and solutions implemented, industry must be integrally involved.
Advances in corrosion research have also been facilitated over the years by developments from other fields. Major advances in materials characterization and computation and modeling in recent years make this an opportune time to address previously unsolvable corrosion problems.
New materials developments have been significant, and recent developments based on computational methodologies allow the process to occur at a much faster pace. However, corrosion resistance is not yet among the properties being optimized (see the section on integrated computational materials science and engineering in Chapter 3).
With some exceptions (see, for example, the nuclear reactor systems success story outlined above), a common element lacking in materials development and selection is the ability to predict the effects of corrosion on useful system lifetime and to use condition monitoring to detect end of life. Methodologies for accelerated testing and predictive modeling are often lacking but are nonetheless critical to the design and management of complex systems. Recent advances in data analysis and engineering practice are aimed at predicting and managing lifetime using advanced techniques of informatics, which can be of great value to knowledge discovery regarding corrosion processes and development of mitigation strategies but has, for the most part, been lacking in corrosion efforts. Hence, corrosion is not one of the properties generally considered in this new prognosis strategy. The impact of corrosion on society is very significant and affects nearly every aspect of daily life in an industrialized country. In monetary terms, degradation by corrosion causes a loss of several percent of the gross domestic product of industrialized nations every year. Other major impacts of corrosion may be challenging to estimate, such as loss of productivity, but are no less significant to society than pure monetary loss.
Although corrosion processes are inevitable, their rate of progression can be significantly curtailed by appropriate mitigation strategies. Many examples exist of how mitigation was successfully achieved after corrosion problems were observed and often became the limiting factor in the engineering lifetime of an application.
Corrosion research has been inspired by application needs, seeking to understand the specific details of the mechanisms of corrosion. Mitigation, which is a corrosion engineering activity, has been most successful when it has been guided by an understanding of the forces driving corrosion processes.




























