3
Research and Development of New Cancer Nanomedicines—Challenges and Solutions
Although much progress has been made in applying nanotechnology to medicine, in order to effectively design, develop, test, and regulate nanoproducts, more needs to be understood about nanomaterials, including their stability, what biological barriers they are able to cross, how best to predict and track their biodistribution, how best to assess their toxicity, and how effective and reliable is their cancer targeting.
Much needs to be done to improve the design and development of nanomedicines, including designing nanomedicines that are more clinically relevant and translatable, improving the scale up and quality control of nanomaterials, developing inexpensive molecular probes that can be manufactured more reliably than antibodies, and developing prevention nanotherapies that can be administered orally. This chapter discusses challenges in basic biology (including biomarker discovery), strategies for improving nanoparticle targeting effectiveness and efficiency, design complexity of nanomaterials for medical applications, the transition from laboratory to manufacturing, and bridging multiple disciplines.
BASIC BIOLOGY
Improving our understanding of nanotechnology and how it can be applied to oncology rests, in part, on improving our understanding of basic biology and the pathogenesis of cancer, as well as biomarkers for cancer, some speakers noted. The dearth of good biomarker targets for prevention, diagnosis, prognosis, treatment, and monitoring is currently
a limitation on development of nanomedicine for oncology and more generally. For nanomedicines for cancer prevention, for example, there needs to be a better understanding of what a precancerous lesion is, Dr. Hawk noted, and improved sensitivity and specificity of the technologies used to detect preinvasive neoplasia or early-stage cancer. Also needed are ways to identify populations at risk for prevention studies, and to identify meaningful endpoints for cancer prevention trials. “There is great promise in measuring multiple biomarkers for early detection, but that promise remains a promise. It is not yet demonstrated, certainly, in the context of prevention,” Dr. Hawk said.
Dr. Barker added that “If I look at least at where we are in regulatory science today, we’re thinking about biomarkers, and how you qualify them, how you use them in trials to directly or indirectly measure what you are trying to measure. This may be one of the most powerful avenues that nanotechnology has to bring to the table in terms of what biomarkers could be, and how you might use them.” The Institute of Medicine recently released a report on the challenges of biomarker evaluation (IOM, 2010).
In some cases, the biomarker knowledge needed to move the field forward is already developed, but not applied due to lack of validation and clinical adoption, Dr. Rogério Sá Gaspar, full professor in pharmaceutics at the University of Lisbon, pointed out. “Currently we have scientific knowledge and technology to genetically profile every single patient, and we know that we have 120 drugs on the market that will behave differently according to their genetic profiling, but we’re not doing that. So it’s not only about the technology and the regulation, it’s also about healthcare and medical practice, and how we do integrate the different components of the sector,” Dr. Gaspar said. Dr. Gaspar called for understanding underlying basic molecular mechanisms and integrating anatomy and physiology issues with pathological disease state and disease progression when designing nanomedicines, which Dr. Duncan echoed by asking that the disease, and not materials science, should be the driver of nanomedicine design and development. “I don’t care whether it’s a small molecule, a liposome, or a PEGylated liposome—the disease should drive it,” she said, and Dr. Grodzinski, director of the NCI Office of Cancer Nanotechnology Research, added that “we need to listen to oncologists about where the most relevant potential need for nanotechnology exists, and how best to apply what technology has developed there.” He suggested picking problems to address with nanotechnology that conventional technology currently can’t solve.
Dr. Duncan added, “We really need to look at the clinical background, pathology, and the biology. People need to look outside their nano journals and into some of the founding literature of biology. … I’m not
knocking the materials science. But, for example, we know from the whole antibody field that antibodies and proteins have problems when we inject them into people and cause immune reactions. So decorating the surface of particles with some of those for targeting is not such a good idea. Just sitting down as a group around the table and deciding what we want to make will really run us forward in keeping it simple and working. ” She later claimed that “ninety-five percent of the experts that we see are not prepared to look outside their box. The biggest challenge for all of us is harnessing this multidisciplinarity. We have to join together the fields and know what we did—what worked and what didn’t work—and then we’ll go forward along the road more quickly.”
STRATEGIES FOR IMPROVING NANOPARTICLE TARGETING EFFECTIVENESS AND EFFICIENCY
Workshop speakers and participants discussed the need for and strategies for improving targeting capabilities of nanotherapeutics and imaging agents. One key question that needs to be addressed is whether cancer-targeting molecules are reliably attached to the nanoparticles, Dr. Curley and Dr. Li pointed out. “We are faced with tracking every component of our particles, and we can’t assess every single component just by putting an imaging agent on one component,” Dr. Li said. He added that one has to separate the effect of passive versus active targeting, which is difficult to do. “You see localization at your target, but how do you know it is passive targeting, and how much of it is achieved through active targeting?” Dr. Li asked. He noted that researchers at the University of California, San Francisco, showed that when they tested the liposomes they developed that were covered with antibodies for the HER2 receptor, the actual localization to the tumor was the same whether they had active targeting with the attached antibodies or just passive targeting with naked liposomes. “It is just an internalization into the cancer cell that was facilitated, so it was more pharmacodynamics that they affected with the active targeting but not pharmacokinetics,” Dr. Li said.
Dr. Desai added that “If these nanoconstructs have been developed to target certain specific locations, we need to establish those tests that define the targeting and show the mechanism of action or mechanism of transport, so we can then design target-specific studies to establish efficacy.” For example, to show that Abraxane was entering endothelial cells via active caveolar gp60-type transport, he put in an inhibitor to the caveolar process and showed that when that inhibitor was used, the level of Abraxane detected in tumor tissue dropped down to that of conventional non-albumin coated Taxol, which only has passive transport into tumor cells (see Figure 6).
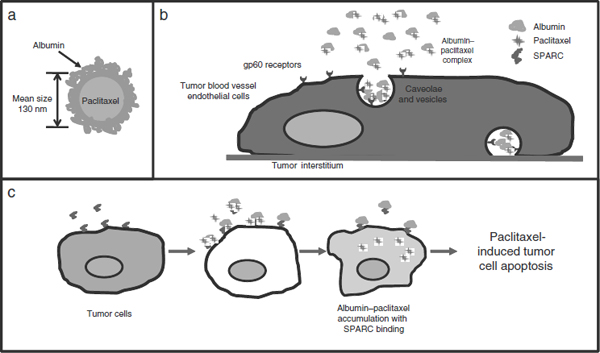
FIGURE 6 The nab technology platform: harnessing endogenous albumin pathways. (a) Particles are injected into circulation, whereupon (b) they dissociate into individual albumin-bound paclitaxel complexes at concentration below threshold. Following dissocation, active receptor-mediated transport the albumin–paclitaxel complexes across the cells via transcytosis mediated by two postulated mechanisms of action: gp60 and caveolae. (c) Active binding of albumin-drug complex by SPARC in tumor, leading to paclitaxel-induced tumor cell apoptosis.
NOTE: gp60 = endothelial cell surface receptor that mediates transcytosis; nab = nanoparticles albumin bound; SPARC = secreted protein, acidic and rich in cysteine
SOURCE: Desai presentation (July 12, 2010).
Both Dr. McNeil and Dr. Duncan pointed out that researchers and regulators have to be aware that changes in the surface of a nanomaterial, such as PEGylation1 to avoid excretion or platination for imaging purposes, can dramatically alter biodistribution and other performance characteristics. “How we process these nanomaterials is critical. A platinated surface is very different from an empty surface and as soon as you start putting a drug in them or functionalizing them, their performance changes a lot,” said Dr. Duncan. Dr. McNeil added “If you change anything on the surface, it becomes a new nanoparticle.”
______________
1 PEGylation is when polyethylene glycol molecules of a given length or length range and given functional groups are attached to a particle in order to confer beneficial characteristics.
DESIGN COMPLEXITY OF NANOMATERIALS FOR MEDICAL APPLICATIONS
There was much discussion about how simple or complex nanomedicines should be, with some speakers cautioning against overengineering nanomedicines with combination constructs and sophisticated structures that may not be relevant or necessary. “The big problem of therapeutics is not so much targeting—it is a great thing if you can get in front of the magic door with the magic key—but to get to that point, you need to make it across so many different sequential biological barriers and the addition of targeting molecules to nanoparticles makes it so much harder to get them to the target lesion, because of the additional transport complexities that are brought in by the target moiety,” Dr. Ferrari said. “Make your nanoparticles as simple as possible, but not any simpler because we have a complex problem to solve.”
Others suggested limiting the complexity of nanoparticles to simplify the testing that will have to be done to show their safety and effectiveness. “If we can make things as simple as possible, that will be better for the regulatory process, approval, and moving to the clinical environment,” Dr. Grodzinski said. But Dr. Gaspar countered, “we cannot make simpler what is complex,” which Dr. Ferrari echoed by saying that within oncology “the low-hanging fruits have been taken care of, and unless we come up with a true paradigm change, it is not going to be that simple. So don’t keep it simple.” Dr. Grodzinski then clarified his previous statement by saying “Limit complexity to a dominion that allows you to cure cancer.”
Dr. Barker added, “I don’t actually think that anything we’re going to be doing in the future is going to be very simple. We’re going to sort out these molecular pathways that lead to cancer, and there are going to be a finite number of them, but every individual is going to have a series of changes along those pathways that are going to be a little different. Nanotechnology will allow us to actually functionalize [nanomaterials] in multiple ways that we could never do if we were doing this in serial fashion along the paradigms we currently have. So embrace complexity, because that’s what you’ve got to deal with.” Related to the complexity versus simplicity issue, Dr. Josephson pointed out the difficulties in assessing what research will be truly clinically translatable. “How do we decide what is clinically translatable and what isn’t, and when to abandon things?” he asked.
TRANSITION FROM THE LABORATORY TO MANUFACTURING
Several speakers addressed the need to improve the scale up and quality control of nanomedicines. “You have to scale up [production of]
the particle that works in the small mouse so it also works for people and meets quality control requirements,” Dr. Ferrari said. He suggested that photolithography methods address both scale-up and quality control issues. (See Box 3.)
Dr. Curley voiced concern that proteins may be reliably attached to nanomaterials in the lab, but it is unclear whether the attachment methods used will scale up for production of larger quantities of the nanomaterials. Dr. Li added that quality control is more cumbersome for multicomponent nanomedicines because one has to do quality control for each component first before they are combined, and then do quality control of the combination. Dr. Zhao pointed out that many nanoparticles adsorb proteins found in the body, and this causes aggregation that changes their metabolism and their biological behavior in the body.
Dr. Desai noted that one key hurdle is being able to reproducibly manufacture complex nanomaterials, and he recommended testing them with orthogonal tests that assess the same thing but use different instrumentation, “because you can get artifacts if you just stick to one technique,” he said. He stressed the importance of fully characterizing and defining nanotechnology products. But characterization of nanomedicines is just one of the first steps that must be taken to translate them into the clinic. Another major challenge is ensuring that nanomedicines are consistently and reproducibly manufactured, Dr. Desai pointed out. “We had many years of headache and heartache trying to get to that level of good consistency and reproducibility. You have to put in appropriate in-process controls and the finished product tests to define the product and to define your manufacturing process. Good engineering and manufacturing skills are essential,” Dr. Desai said. Dr. Libutti added that one major concern FDA raised at his IND meeting for the nanoTNF medicine he created was that the particles should be uniform in size and substance when his lab characterized them.
Dr. Heath noted that antibodies that are frequently used as probes in nanodiagnostics, such as in barcode technology, are highly variable from batch to batch. “If you buy a new batch of antibody, it changes your entire calibration for this thing—it is an absolute killer. So even though you know you can do this, it makes it an academic exercise until you get around this antibody problem. I don’t know a solution to that, other than get rid of the antibodies,” he said. “The solution is finding non-biological capture agents, things where you make them once, you make them the next time, and the next time, and they are the same every single time, and you can guarantee a calibration, your training set, etc.”
Dr. McNeil added that this was a major problem for many types of nanomaterials. “You have to tightly control batch-to-batch variability in your manufacturing and development process. You have to make sure
that you know which parameters influence biocompatibility for your particular nanoparticle concept and be able to control that with each subsequent batch that you manufacture,” he said. He noted that some nanomaterials, such as liposomes, are well-suited for mass production and have an ease of scale up, but it may take months to years to fine-tune their formulation so their drug contents are stable and are not released too early.
Dr. Duncan stressed the importance of knowing the impurities that might be linked to nanomedicines. “We have to know what the weaknesses are so that we can make a calibration that we can validate, and we can show a regulatory agency that we know what we have in the bottle every time we make it, and that the impurities are going to be safe in that context,” she said.
Dr. Duncan added that it is important to fully characterize and test each type of formulation of a nanomedicine. “We rarely give a drug to a patient on its own. So we do all of this preclinical work and then we are asked for a tablet formulation or an injection formulation that will be stable on the shelf for a year. So now we have all these other bits that the nanomedicine can interact with, and that can change its safety, efficacy and pharmacokinetics. We need to really reflect upon those things and look down the road while we’re still at the beginning,” she said. Dr. Hawk pointed out that most nanomedicines for prevention currently being developed must be administered intravenously, and urged more development of those that can be taken orally, on a regular, long-term basis, as that is needed for a cancer preventative. “IV administration may be acceptable in those at very high risk if done on an intermittent basis, but is not applicable to the majority of the population at risk, looking for preventive strategies.” he said.
BRIDGING MULTIPLE DISCIPLINES
Several speakers echoed the sentiment that there need to be more bridges between the multiple disciplines needed to bring nanomedicine to fruition. As Dr. Ferrari pointed out, nanomedicine is highly interdisciplinary, requiring the expertise of clinicians, materials scientists, mathematicians, biologists, molecular biologists, physicists, and chemists. “In my group I have about 150 people right now, and I cannot think of two that have the same background,” he said. Dr. Ferrari added that it is hard to find institutional setups that enable such multidisciplinary, team research, while at the same time enabling researchers to pursue their own individual careers. Dr. Curley added that editors were having trouble finding reviewers for his journal articles because they cover physics, chemistry, biology, and cancer, and no one is an expert in all those fields.
BOX 3
Manufacturing Nanomaterials
To make the manufacturing of nanomedicines more efficient, reliable, and less expensive, researchers are applying some of the approaches used to manufacture semiconductors to the production of nanomedicines, Dr. DeSimone, Chancellor’s Eminent Professor of Chemistry at the University of North Carolina Chapel Hill and William R. Kenan Jr. Distinguished Professor of Chemical Engineering at North Carolina State University, reported. This involves mass-producing nanostructures by removing or adding material to a surface using microscopic lithography. For example, with the Particle Replication In Non-wetting Templates (PRINT) system that Dr. DeSimone developed, silicon wafer surfaces are coated with fluoropolymers and then etched with a photochemical process to create a mold for nanomaterials. To scale up and produce large quantities, he then uses a “roll to roll” process similar to what is used in the film industry. With this process, the mold, in sheet form, is matched to a delivery sheet that is used to form the actual nanomedicines, which are peeled away from the mold sheet using a harvesting film with an adhesive on it (see figure below).
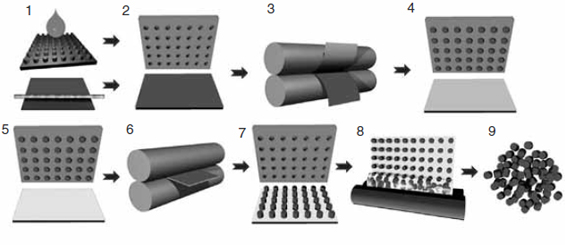
FIGURE B3-1 Diagram showing the roll-to-roll transfer templating technique. (1) The sheet shown with raised posts is the mold used to create the delivery sheet with cylindrical indentations. (2) Liquids are then spread on another sheet. (3) The sheets are sandwiched together and run through a roll-to-roll process. (4) Because of the unique surface characteristics of the flouropolymers used, the cavities are filled using capillary force without wetting the surface area between the cavities. (5) The filled cavities are brought into contact with another film that has an adhesive on it using (6) a roll-to-roll technique, and (7) the structures are transferred onto the harvesting film. (8)–(9) Structures can then be removed from the films.
NOTE: PRINT = particle replication in non-wetting templates
SOURCE: DeSimone presentation (July 12, 2010).
“One of the key things for this nanomanufacturing method is that it is a templated manufacturing process,” Dr. DeSimone said. “The particles we generate are derived from the features of these master templates, so we have a way of fabricating particles of uniform size and structure.” The etching process, he noted,
ensures such uniformity. “One of the key things in nanomanufacturing is reproducible manufacturing. This is where we think we have some significant advantages with a templated approach,” Dr. DeSimone said.
PRINT can be used to control the size, shape, and surface chemistry of nanoparticles, which can be made from organic materials. Many of the particles have a hydrogel as their base, to which different drugs are added, including traditional proteins and steroids as well as innovative siRNAs. Targeting moieties can also be attached with the molding process. “We can mold a wide range of chemistries,” Dr. DeSimone said, adding that the molding process enables them to form “pseudoconjugates” without the complex chemistry that is usually required to create polysaccharide–protein or other conjugated molecules.
PRINT’s molding process also has more efficiency than the standard chemical self-assembly process for encapsulating a drug in a matrix, according to Dr. DeSimone. He has been able to use PRINT to encapsulate docetaxel with PLGA (polylactic-co-glycolic acid) that has an encapsulation efficiency of 40 percent, he said. “In liposomes, very often you have very little latitude in controlling the amount of drugs you can put into these self-assembled structures. With our approach, we can vary the amount of drug directly within these particles, which can have a profound effect on the viability and the potency of these particles. We are getting to the point where a single particle can induce cell death, so one has to be careful [about drug concentration] now that these particles have become so potent,” he said.
Dr. DeSimone has found that particle size and shape plays a big role in how various drugs are delivered directly into the airway using a dry powder inhaler. “We can start engineering the particle size and the aerodynamic characteristics of these particles as we vary the size and shape,” he said, thereby determining whether the drug extends to the mid or deep regions of the lung or whether it mostly settles into the trachea.
Dr. DeSimone used his PRINT (Particle Replication In Non-wetting Templates) nanomanufacturing process to create a thousand doses for vaccines that are Good Manufacturing Practice (GMP)–compliant that will be tested in a phase II clinical trial. “We looked at this as [setting the stage for future] products moving forward in cancer. Our vaccine program is driving PRINT forward and allowing our other therapeutics to come in behind it,” said Dr. DeSimone. He noted for his inhalation drug program, he was able to use PRINT to make hundreds of grams of a drug in a two-day run. “We believe we can be in a kilogram quantity in an afternoon with this templated approach, and that is the direction we are going,” he said.
One aspect of his research involves modifying the deformability of the nanoparticles he engineers because there is evidence that the deformability of metastatic cancer cells enables them to metastasize (Suresh, 2007). He recently used PRINT to create extremely deformable nanoparticles, whose biodistribution he is currently testing in an animal model. “We think the ability to control size, shape, and deformation is a key component to nanomanufacturing, where calibration quality particles are intrinsically derived from a template approach using this roll-to-roll process,” he concluded.
“It is a team sport,” said Dr. Grodzinski. “We have to learn a common language and work under one roof in a multidisciplinary environment.” Dr. McNeil added, “It’s really been a privilege working at the NCL where we’ve got physicists, immunologists, toxicologists, chemists, biotechnicians, and cell biologists working together. When all of us came together, we were able to offer new solutions that each of us in our own disparate fields was not able to tackle. As intimidating as it may seem at first, I would just encourage you as soon as possible, if you’re a materials scientist, to have lunch with a biologist, and vice versa.”
Dr. Jonathan Sackner-Bernstein, associate director for post market operations at the Food and Drug Administration’s Center for Devices and Radiologic Health, pointed out that technological advances often stem from the synergy between multiple disciplines, and both Dr. Duncan and Dr. Barker pointed out that what is known in one discipline, such as polymer science, is news to another discipline. “I’d suggest that we not reinvent the wheel in terms of polymer science, but take advantage of what we know already,” Dr. Barker said.
But she added, “It is still difficult in this environment, where we focus so heavily on the individual investigator that we are really making it difficult to do team science. We have got to change that.” Dr. Gaspar also called for more collaborative efforts with diagnostic companies, imaging companies, and drug companies working together to develop nanoproducts for the clinic.
NCI Alliance for Nanotechnology in Cancer
Launched in 2004, the NCI Alliance for Nanotechnology in Cancer’s mission is to harness the power of nanotechnology to change the way cancer is diagnosed, treated, and prevented. Through its programs and initiatives, the Alliance is committed to building a community of researchers dedicated to using nanotechnology to advance the fight against cancer.
The Alliance is focused on team science and has multiple interagency collaborations, including those with the National Institute of Standards and Technology, the Food and Drug Administration, the National Institute of Environmental Health Sciences and the Environmental Protection Agency.
A major goal for the NCI Alliance for Nanotechnology in Cancer is to quicken the pace of nanotechnology discovery and development efforts, and to lower the barriers to commercialize these advances for the benefit of cancer patients. “The Alliance is an applications-driven activity. We are not interested in just fundamental science, but about changing the lives of patients,” said Dr. Anna Barker, former deputy director of the National Cancer Institute. “We built this Alliance to commercialize technology, and
we have over 50 companies, either created by or associated with the Alliance, and over 200 patents and disclosures have been filed.”
The Alliance has four major programs, which are described in the following sections.
Centers of Cancer Nanotechnology Excellence (CCNEs)
The CCNE network designs and tests nanomaterials and nanodevices, and translates their use into clinical research. The CCNEs will bridge gaps in the development pipeline from materials discovery to testing in clinical trials.
By balancing structured directives with investigator-initiated research, the CCNEs bring together the interdisciplinary teams from existing NCI resources and provide the infrastructure necessary to develop and translate nanotechnology advances to the clinic.
Multidisciplinary Research Training and Team Development
The Alliance supports training and career development initiatives to establish integrated teams of cancer researchers, epidemiologists, engineers, and others to approach the fundamental challenges of cancer using cancer biology, physical science skills, and the knowledge base of nanotechnology. The NCI is initially using existing training and career development mechanisms to direct talent to this area as quickly as possible. The NCI also encourages program development with interfaces to the training programs of other federal agencies.
Nanotechnology Platforms for Cancer Research
The NCI has identified specific technology requirements and cancer biology problems that constitute critical nanotechnology platform needs for cancer. These directed research programs are funding technology development projects through both grants and contracts overseen by project specialists.
These projects are aimed at deployment for clinical application in cancer research, and applicants are required to team with the NCI to develop a dissemination plan for the technology. Examples of these platform needs include, but are not limited to
- Molecular Imaging and Early Detection,
- In Vivo Nanotechnology Imagining Systems,
- Reporters of Efficacy,
- Multifunctional Therapeutics,
- Prevention and Control and
- Reserch Enablers.
Nanotechnology Characterization Laboratory (NCL)
Through a collaboration with the National Cancer Institute, the Food and Drug Administration, and the National Institute of Standards and Technology (NIST), the Nanotechnology Characterization Laboratory is developing data that will facilitate standards for nanoscale devices, and facilitate regulatory review of these products prior to market release.
Using standardized methods, the NCL characterizes the physical and chemical parameters of nanoparticles and conducts structure–activity relationships studies that aid assessments of biocompatibility. NCL also aids preclinical scale up and development, including assessing the quality, purity, and stability of nanomaterials, and conducting in vitro studies that assess sterility, cell uptake and distribution, blood contact properties and toxicity, and in vivo studies that focus on biodistribution, dose-related toxicities, and to a limited degree confirm efficacy (see Table 3).
“An investigator may have a proof of concept with a few milligrams of material, and we help them get into clinical trials over about the next year or year and a half, in some cases,” said Dr. Scott McNeil, director of the NCL.
To characterize nanomaterials, NCL conducts a number of tests and assays, many of which differ from those that are commonly done to characterize small molecules (see Table 4). “We’re still interested in the same physical and chemical properties assessed in small molecules, but we use a different portfolio of instrumentation to get at those,” Dr. McNeil said.
The NCL’s services are available to academia, industry, and government users under its application process, and its services are provided at no cost to the users. The NCL is facilitating collaborations among the NCI, academia, and the private sector to accelerate the translation of nanotechnology research into clinical advances. It also interfaces with the FDA regularly to explore issues of regulation and policy concerning nanomaterials for medical applications, and recently began collaborating with the National Center for Toxicological Research and the National Institute of Environmental Health Sciences.
“We felt that to get ahead of this field, we had to be able to characterize these materials to accelerate the translation of these agents into the clinic,” said Dr. Barker. “This has turned out to be a common source of information for all the government agencies.”
TABLE 3 The Nanotechnology Characterization Laboratory Conducts Physical, Chemical, and Structure–Activity Assessments of Nanomaterials
| In vitro | In vivo |
|
Sterility Cell uptake and distribution Blood contact properties Toxicity |
Initial disposition study Dose-range finding toxicity Efficacy |
SOURCE: McNeil presentation (July 12, 2010).
Chinese Academy of Sciences Key Lab for Nanosafety
Recently established, China’s Key Lab for Nanosafety has more than 100 researchers, students, and administrators dedicated to assessing nanoparticle properties and the hazards to humans and the environment that nanomaterials may pose. The Nanosafety Lab also makes recommendations regarding regulation of research and industrial activities on nanotechnology, fosters international nanotechnology collaborations and standards, and aids safety assessment for nanotechnology industry by developing assessment methods and procedures, and identifying toxic classes of nanomaterials.
The Nanosafety Lab engages 16 institutions and universities in China in the study of nanosafety issues, and receives its support from the Chinese government, the National Natural Science Foundation of China (NSFC) and the Chinese Academy of Sciences.
TABLE 4 Tests and Assays Used by the Nanotechnology Characterization Laboratory to Determine the Physicochemical Parameters of Nanomaterials
| Small molecules | Physicochemical parameters | Nanomaterial |
| Elemental analysis Mass spectrometry NMR UV-Vis IR HPLC GC Polarimetry |
Composition Physical properties Chemical properties Identification Quality Purity Stability |
Microscopy (AFM, TEM, SEM) Light scattering (static, dynamic) SEC, FFF Electrophoresis (CE, PAGE) Zeta sizer Fluorimetry |
NOTES: In many cases, different instrumentation is required to analyze nanomaterials as compared to small molecules. Nonetheless, both sets of techniques (left and right columns) are used to probe the same fundamental properties (center column). AFM = atomic force microscopy; CE = capillary electrophoresis; FFF = field flow fractionation; GC = gas chromatography; HPLC = high-performance liquid chromatography; IR = infrared spectroscopy; NMR = nuclear magnetic resonance spectroscopy; PAGE = polyacrylamide gel electrophoresis; SEC = size exclusion chromatography; SEM = scanning electron microscopy; TEM = transmission electron microscopy; UV-Vis = ultraviolet–visible spectroscopy.
SOURCE: McNeil presentation (July 13, 2010).
Center for Biological and Environmental Nanotechnology (CBEN) and International Council on Nanotechnology (ICON)
Center for Biological and Environmental Nanotechnology (CBEN)
Established in 2001, the CBEN is a federally funded National Science Foundation research center whose mission is to discover and develop nanomaterials that enable new medical and environmental technologies.
The Center’s research activities explore the interface between nanomaterials and aqueous systems at multiple length scales, including interactions with solvents, biomolecules, cells, whole organisms, and the environment. These explorations form the basis for understanding the natural interactions that nanomaterials will experience outside the laboratory, and also serve as foundational knowledge for designing biomolecule–nanomaterial interactions, solving bioengineering problems with nanoscale materials, and constructing nanoscale materials useful in solving environmental engineering problems.
Though unified intellectually by the wet/dry interface, the Center’s research programs are oriented toward tangible technological outcomes, or engineered systems. These are
- Nanoparticles that detect and treat disease, including those that can be used for drug delivery, photothermal cancer treatments, and imaging contrast agents; and
- Effective, high performance water purification systems that use nanoscale materials to both remove and remediate waste.
International Council on Nanotechnology (ICON)
ICON (http://icon.rice.edu) is an international, multi-stakeholder organization whose mission is to develop and communicate information regarding potential environmental and health risks of nanotechnology. ICON was founded in 2004 as an extension of the US National Science Foundation Center for Biological and Environmental Nanotechnology (CBEN) at Rice University in Houston, Texas. Composed of individuals from academia, industry, government, and non-governmental organizations from France, Japan, the Netherlands, Switzerland, Taiwan, the United Kingdom, and the United States, ICON is a technically-driven organization that does not engage in advocacy or commercial activities.
“ICON is a one-stop shop for all information related to the the environmental health and safety implications of nanomaterials,” said Dr. Kristen Kulinowski, senior faculty fellow in the Department of Chemistry at Rice University and Director for External Affairs for the Center for Biological and Environmental Nanotechnology.
This page intentionally left blank.
















