2
Biology and History of Bacillus anthracis
Bacillus anthracis (B. anthracis) is a Gram-positive rod-shaped bacterium that is the causative agent of the disease anthrax. B. anthracis rods typically have dimensions of approximately 1 µm by 4 µm and may occur in chains resembling “boxcars” when observed under a microscope. The word “anthrax” is derived from the Greek word for “coal” or “black” and was applied to this disease because patients with the cutaneous form of the disease often exhibit black skin lesions. The disease has been known since antiquity. In the 19th century, the demonstration by Robert Koch of B. anthracis as the cause of anthrax was a cornerstone of proving the germ theory of disease.
This chapter summarizes historical, biological, and phylogenetic features of B. anthracis, and in particular the Ames strain, as well as clinical features of anthrax, many of which proved highly relevant as the events surrounding the mailing of the B. anthracis spore-laden letters unfolded in fall 2001.
2.2 THE BIOLOGY OF B. ANTHRACIS
B. anthracis spores are the infectious agent for all forms of anthrax. In contrast, B. anthracis vegetative cells are noninfectious in animal models (Mock and Fouet, 2001). The structural characteristics and environmental resistance of the B. anthracis spore are key to its avoidance of the host innate immune response during the initial stages of infection (Cybulski et al., 2009; Mock and Fouet, 2001). Relative to the majority of bacterial vegetative cells, spores like those produced by B. anthracis are highly resistant to a variety of commonly lethal treatments, including dehydration, elevated temperature, UV irradiation, numerous toxic chemicals, and enzymatic digestion by proteases and lysozymes (Setlow, 2006). In the dormant state, spores can retain viability for decades and likely for centuries (Nicholson et al., 2000); however, upon arrival in a hospitable environment, the spores can germinate and resume rapid growth within
hours (Moir, 2006; Setlow, 2003). The stability of spores, with accompanying ease of storage and transport, is also a major factor in their potential utility as a biological weapon.
The resilience of B. anthracis spores derives from their unique physical and structural characteristics. The spore cytoplasm or “core” is relatively dehydrated and contains high concentrations of certain low-molecular-weight solutes (Setlow, 2006). These conditions result in complete metabolic dormancy and extreme protein stability. The low core water content of the spore correlates with a high spore density, a property that is commonly exploited for spore purification. Centrifugation through density gradients of diatrizoate and meglumine is a widely used purification method (Tamir and Gilvarg, 1966; Nicholson and Setlow, 1990).
Surrounding the spore core are a membrane and a specialized peptidoglycan cell wall, the “cortex.” These structures play key roles in limiting movement of water and solutes into and out of the core, maintaining the dehydrated dormant state (Setlow, 2006). The membrane and cortex have not been cited as significant sites of mineral association nor found to play roles in direct spore interactions with external surfaces.
The outermost layers of the spore, the coat and exosporium, are the primary sites of interaction with the host and with other surfaces and are important factors in the resistance of spores to certain enzymatic and chemical treatments (Driks, 2009; Henriques and Moran, 2007; Setlow, 2006). These structures are composed predominantly of protein and glycoprotein. In some spore-forming bacteria, the spore coats have been shown to be sites of association of minerals (Johnstone et al., 1980; Mann et al., 1988; Stewart et al., 1980, 1981; Hirota et al., 2010). These outermost integuments can play major roles in determining spore adherence to surfaces (Bozue et al., 2007; Brahmbhatt et al., 2007) and may affect electrostatic properties and aggregation with other spores or particles, all of which will affect spore dispersal and infectivity. Efforts at spore aerosolization and dispersal have been pursued through modification of the spores’ surface structures and properties (Swartz, 2001).
The fact that B. anthracis can exist in the environment as a dormant, highly stable spore may have evolutionary significance. While a dormant environmental state may help explain observations that populations of B. anthracis have much less genetic variation than many other bacterial species (Van Ert et al., 2007a), other factors may also contribute, including the likely recent origin of this species and the possibility of limited means for horizontal gene transfer.
2.3 CLINICAL ASPECTS OF ANTHRAX
Anthrax is generally a disease of herbivores (e.g., cattle, sheep, horses), which acquire the infection by grazing on contaminated soils. Anthrax spores are highly resistant to environmental insults. These spores allow the bacterium
to survive for long periods of time in soil, its natural reservoir. The disease occurs worldwide, and there are occasional outbreaks of anthrax in livestock in the United States and Canada.
The course and outcome of human anthrax depend on dose and on whether the infection is acquired via the skin, gastrointestinal tract, or inhalation. Most cases of human anthrax involve skin lesions (cutaneous anthrax), whereby the infection is usually acquired as a result of handling infected animal hides or wool, leading to contamination of skin abrasions with B. anthracis. Cutaneous anthrax is the least lethal form of the disease, but still can cause significant mortality of up to 20 percent (Atlas, 2002) if not treated with antimicrobial therapy. Ingestion of food, such as meat contaminated with B. anthracis, can produce gastrointestinal anthrax, which is a serious disease with 25 to 60 percent fatality. This form of the disease is extremely rare in developed countries. The most fulminant and lethal manifestation of anthrax is due to inhalation of B. anthracis spores, causing a highly fatal disease. Inhalational anthrax is generally rare and is observed mainly in individuals who work with animal skins. In the 19th century, it was known as “woolsorter’s disease” and is believed to have been the first documented occupational illness (Leffel and Pitt, 2006). Inhalational anthrax in individuals not likely to have suffered occupational exposure, however, can be a sign of a biological attack with B. anthracis spores.
Inhalational anthrax is a rapidly progressive disease with high mortality and morbidity even when treated with antimicrobial therapy. Anthrax spores germinate when placed in blood or other human or animal tissues that provide a nutrient-rich environment (Inglesby et al., 2002). Upon inhalation, the host’s macrophages (a type of immune system cell) attack and ingest the spores, which are protected from these host cells by the spore coat. The macrophages unwittingly transport the spores to lymph nodes in the respiratory system (Liddington, 2002), where they are released. The lymph nodes provide sufficient nutrients to allow the spores to germinate and begin to proliferate. Proliferation in the lymphatic system in turn allows the bacteria in their “vegetative” state to spread into the blood stream and be disseminated to multiple organs.
The genetic determinants of virulence in B. anthracis reside primarily on two large plasmids, which are extrachromosomal DNA molecules. These plasmids are known as pXO1 and pXO2, and they contain genes that encode for anthrax toxin and a poly-D-glutamate capsule, respectively. Anthrax toxins are composed of a combination of three proteins that work together: protective antigen (PA), edema factor (EF), and lethal factor (LF). When PA combines with EF and LF, toxicity to host cells and a buildup of fluids (edema) in infected tissues (e.g., the lungs) are produced. EF is an adenylate cyclase enzyme that promotes the accumulation of cyclic adenosine monophosphate (AMP), producing a loss of cellular regulation of water and ion metabolism. LF is a metallopeptidase that cleaves proteins of signal transduction pathways, resulting in profound effects that range from cell death to interference with the
cellular functions necessary for mounting appropriate immune responses. The effects of the anthrax toxin can kill an infected patient even after the bacteria in the patient’s body have been killed by antibiotics (Bouzianas, 2009).
In addition to the PA, EF, and LF toxin components, B. anthracis produces other cytotoxic enzymes, such as anthrolysin O, that contribute to pathogenesis. The combination of the protein toxins, which undermine the host’s defenses and interfere with cellular function, and the polymerized amino acid capsule, which protects the bacterium from phagocytic cells, represents a particularly lethal mix. Antibodies that neutralize the toxins or promote phagocytosis of encapsulated cells can confer significant protection to the host. The vaccine currently in use for the prevention of anthrax functions by eliciting neutralizing antibody responses to anthrax toxin. B. anthracis mutants that lack either toxin production or capsules are attenuated for virulence and can be used in vaccine formulations (Friedlander and Little, 2009).
2.4 B. ANTHRACIS AS A BIOLOGICAL WEAPON
Research on B. anthracis as a biological weapon began more than 90 years ago, according to Inglesby and colleagues (2002). When appropriately prepared, B. anthracis spores are premier agents for biological warfare and bioterrorism because they can be produced in prodigious quantities in a form that is readily aerosolized and inhaled. These factors, plus the high mortality associated with inhalational anthrax, make B. anthracis a serious military and bioterrorism threat.
Most offensive biological weapons programs were terminated following the ratification or signing of the Biological Weapons Convention in the early 1970s (Inglesby et al., 2002). The United States’ offensive biological weapons program was terminated before that by President Nixon in 1969. Nevertheless, international state programs to produce and weaponize B. anthracis have remained a concern. For example, in 1995 Iraq acknowledged having an anthrax weapons program to the United Nations.
Estimates of fatalities likely to occur after a major attack on an urban area using aerosolized B. anthracis as a bioweapon range into the millions (Meselson et al., 1994; Inglesby et al., 2002). Even an accidental release of aerosolized B. anthracis at a former Soviet military facility in Sverdlovsk in 1979 resulted in scores of infections and many human and animal deaths (see Box 2-1) (Leffel and Pitt, 2006). No inhalational anthrax fatality had occurred in the United States as the result of an act of war or terror until the 2001 anthrax mailings (Bush et al., 2001).
The events of 2001, when envelopes with B. anthracis spores were distributed through the U.S. mail, showed the potential of this microbe as a biological weapon. The resulting 22 cases of anthrax, including five fatalities, spread great fear and resulted in tremendous disruption and dislocation as mail distribution centers, congressional office buildings, and other sites suffered extensive
BOX 2-1
The Sverdlovsk Outbreak
In 1980, reports appeared in the international press of a widespread outbreak of anthrax in the city of Sverdlovsk (now Ekaterinburg) in the Soviet Union (Gwertzman, 1980). Soviet medical, veterinary, and legal publications reported that an outbreak had taken place in early 1979, involving livestock and humans who ate contaminated meat. In 1988, three Soviet physicians visited the United States to give formal and informal presentations on the outbreak (Meselson et al., 1994). In 1990, more discussion appeared in the Russian press (references in Meselson et al., 1994), leading Soviet President Boris Yeltsin to call for further investigations. Finally, in 1992, Yeltsin was quoted as saying “that the KGB admitted that our military developments were the cause” (references in Meselson et al., 1994).
A group of scientists from the United States carried out an on-site investigation during two visits in 1992 and 1993; their results were published in Science in 1994 (Meselson et al., 1994; Guillemin, 1999). They concluded that “the outbreak resulted from the windborne spread of an aerosol of anthrax pathogen, that the source was at the military microbiology facility, and that the escape of the pathogen occurred during the day on Monday, April 2 [1979].”
The outbreak has been used to model aerosolized agent release and parameters such as plume migration (Hogan et al., 2007), infectious dose (Brookmeyer et al., 2001; Wilkening 2006), incubation period (Brookmeyer et al., 2005; Wilkening, 2008), and the impact of public health intervention (Brookmeyer et al., 2001).A 1998 study by Paul Keim and Paul Jackson (Jackson et al., 1998) used the polymerase chain reaction (PCR) technique to analyze tissue samples from 11 patients from the 1979 Sverdlovsk outbreak. The study found that sequences representing the entire virulence complement (toxins and capsule) were present in all the samples, and also found that strains from at least four of the five known strain categories were distributed among the samples, as determined by variable region sequencing (variable number tandem repeats, or VNTRs). Two subsequent studies were published: one found additional evidence for multiple strains (Price et al., 1999) and the other found no evidence for multiple strains (Okinaka et al., 2008). Whether or not multiple strains were involved in this outbreak and if so the implications of this are not known. A previous analysis of 198 clinical samples from naturally occurring anthrax cases indicated the presence of only one strain in each case (Jackson et al., 1997).
contamination (see Chapter 3). Over 30,000 people were given prophylactic antibiotic treatment and six buildings required decontamination, all at a cost of over $1 billion (CDC, 2001a).
B. anthracis is a member of the large genus Bacillus that includes other common and diverse species, such as B. cereus, B. subtilis, and B. thuringiensis.
Within the genus, B. anthracis represents a separate lineage that apparently evolved from the B. cereus parent species. Some strains of both B. cereus and B. thuringiensis are clearly closely related to B. anthracis (Kolsto et al., 2009). The production of endospores that resist chemicals, heat, UV light, and desiccation when the cells experience starvation or other environmental stresses is a common feature of members of the Bacillus genus.
B. anthracis has often been viewed as a highly monomorphic species, that is, one that shows little genetic variation among isolates. While it is the case that many bacterial species harbor much more extensive genetic diversity, newer molecular methods have allowed researchers to find genetic differences among natural isolates of B. anthracis, as discussed below. Moreover, all populations of bacteria are continually generating new mutations. While most new mutations remain very rare in a population, some of them may rise to high and measurable abundances, especially when they provide an advantage to the bacteria in terms of increasing their growth or survival under certain conditions. As discussed in detail in Chapters 5 and 6, the presence of new mutations among the spores in the attack letters provided an important lead in the anthrax letters investigation.
Modern scientific tools now afford an understanding at the molecular level of the similarities and differences among members of the genus Bacillus. For example, as noted above, the pathogenicity of B. anthracis is related to the presence of the pXO1 and pXO2 plasmids. These were once believed to separate B. anthracis from the other members of the genus, Bacillus. Within the last five years, however, B. cereus strains containing pXO1 or both pXO1 and pXO2 have been discovered, the latter in great apes in Africa. Thus, the presence of these plasmids is no longer believed to be the major factor separating B. anthracis from other members of the genus Bacillus. Rather, a specific mutation in B. anthracis’ global regulator plcR gene, which controls the transcription of virulence factors in B. cereus and B. thuringiensis, now appears to be the key difference. All B. anthracis strains investigated carry a mutation that makes the plcR gene dysfunctional. This feature makes B. anthracis easily distinguishable from its close relatives.
Until the advent of modern molecular approaches, the genetic homogeneity of B. anthracis impeded efforts to reconstruct the evolutionary history of the species (Keim et al., 1999). Substantial progress has recently been made. Van Ert and colleagues (2007a) conducted a large study of 1,033 B. anthracis isolates using canonical SNP (single nucleotide polymorphism) analysis. Based on their results, they divided all B. anthracis isolates into A, B, and C lineages that had been previously recognized and further subdivided these into 12 distinct sublineages or subgroups (see Figure 2-1). Sublineage designation was based on whole-genome sequencing of seven isolates that represent major branches on a phylogenetic tree. The five other subgroups, whose genomes were not sequenced, appear at intermediate positions along these branches.
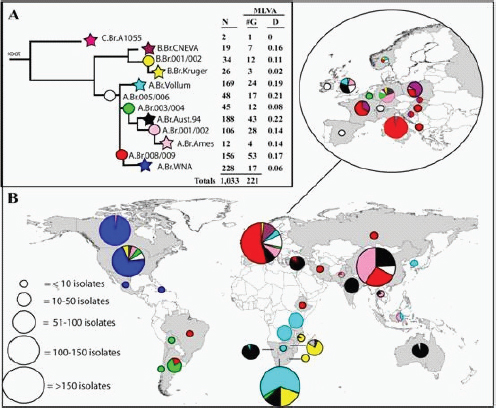
FIGURE 2-1 Worldwide Distribution and Lineages of B. anthracis.
The stars in this dendrogram represent specific lineages defined by one of seven sequenced genomes of B. anthracis. The circles represent branch points along the lineages that contain specific subgroups of isolates (Van Ert et al., 2007a).
Van Ert and colleagues (2007a) point out that dispersal of spores via commodities has distributed anthrax worldwide such that there are foci of the disease on all continents except Antarctica (see Figure 2-1). The more numerous “A” lineage isolates of B. anthracis are the most widely distributed, while there are more restricted distributions of “B” and “C” lineage isolates; for example, the B lineage is found mainly in South Africa and portions of Europe. There are also distinctive genotypes in the Western Hemisphere, with particular North and South American genotype subgroups that are rarely observed outside these regions. Based on molecular clock estimates, Van Ert and colleagues (2007a) note that the radiation of the A lineage seems to have coincided with periods of increased animal domestication and expansion of domestic animal populations. Although North America has a single dominant genetic type, in more recent times, trade in wool, skins, bone meal, and other products appears to have
contributed to the introduction of an assortment of rarer B. anthracis genotypes on the North American continent. One of these, the “Ames” strain, is the strain found in the 2001 anthrax mailings.
2.6 THE EARLY HISTORY OF THE AMES STRAIN OF B. ANTHRACIS
The B. anthracis Ames strain was first isolated from a dead cow in Sarita, Texas, in 1981. Texas A&M University shipped this new isolate to the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID) at Fort Detrick in Frederick, Maryland. Because the box used for this shipment bore an old Ames, Iowa, address, the strain came to be known as the “Ames” strain. As this is a misnomer, at least in terms of the location of the origin of the first sample of the strain, Ravel and colleagues (2009a,b) called this specific isolate from Texas “B. anthracis Ames Ancestor.” Chapter 5 describes the experiments used to determine that the Ames strain was the source of the material in the anthrax mailings.
The Ames strain is uncommon in nature. In the large study by Van Ert and colleagues (2007a), North America was represented by 273 isolates of B. anthracis spanning 44 genotypes but the Ames genotype was found only once, indicating that it is rare. According to Keim (2009), the Ames strain appears to be a recent immigrant to North America, with its closest relatives found in China. In North America, the natural distribution of the Ames strain appears to be limited to a small area of Texas. However, the Ames strain has been widely distributed as a laboratory strain. This fact, coupled with its rarity in nature, “makes it unlikely that the source material utilized in the 2001 bioterrorist attack was acquired directly from nature” (Van Ert et al., 2007).
The bacterium B. anthracis is the causative agent of the disease anthrax. The infectious agent in all forms of the disease is the dormant B. anthracis spore. The spore’s structural characteristics and properties provide the organism with resistance to environmental insults (e.g., dehydration, elevated temperature, UV irradiation) and also enable it to avoid the immune response of an infected person. Spores can remain viable in this dormant state for decades or more. Upon arrival in a hospitable environment (i.e., in a human or animal body) the spores can germinate and resume rapid growth within hours, causing illness.
In humans, the course and outcome of the disease depend on dose and on whether the infection is acquired via the skin, gastrointestinal tract, or inhalation. Most human cases involve skin lesions (cutaneous anthrax), whereby the infection is acquired as a result of handling infected animal hides or wool. Ingestion of food contaminated with B. anthracis can produce gastrointestinal
anthrax, a much more serious disease. But the most lethal manifestation of anthrax is caused by inhalation of spores, causing a severe disease that is often fatal. Inhalational anthrax is generally rare and is mainly observed in individuals who work with animal skins. Inhalational anthrax in individuals not likely to have suffered occupational exposure, however, can be a sign of a biological attack with B. anthracis spores.
When appropriately prepared, anthrax spores are premier agents for biological warfare and bioterrorism because they can be produced in prodigious quantities in a form that can be aerosolized and inhaled. These factors, plus the high mortality associated with inhalational anthrax, make B. anthracis a serious military and bioterrorism threat. For these reasons, the appearance of B. anthracis in the 2001 mailings launched a major public health and criminal investigation.










