The discoveries that individuals had contracted anthrax and that letters containing B. anthracis had been sent by U.S. mail launched a full-scale investigation by the U.S. Centers for Disease Control and Prevention (CDC), the U.S. Postal Inspection Service (USPIS), and the Federal Bureau of Investigation (FBI). Numerous investigative techniques were applied throughout the investigation, as outlined in Table 3-1. This chapter describes in brief the early stages of the investigation, specifically the gathering of evidence, formation of investigative teams, and decisions regarding scientific analyses that led to the FBI findings and conclusions that are summarized and evaluated more fully in Chapters 4 through 6.1 It also introduces the concepts of science, scientific investigation in law enforcement, and the different views of uncertainty—and the manner in which it is described—in science versus law.
3.2 SCIENCE AND SCIENTIFIC INVESTIGATION AS PART OF A LAW ENFORCEMENT INVESTIGATION
In a scientific study, explanations for observable phenomena are sought through the gathering of reliable data and the formulation of testable hypotheses. Scientific observations must be reproducible and scientific hypotheses must be refutable. Science is typically an iterative, collaborative, and open process requiring the ability to pose hypotheses, test them, and pose new questions based on the resulting information. At times, a scientific investigation is a divergent process, in which new results drive research in several directions only
__________
1 Throughout this report, the committee describes its evaluation of the primary reports and data contained in the materials provided to the committee by the FBI. In cases where the committee was not able to review primary data, the committee’s assessment of statements or analyses of data by others is provided.
TABLE 3-1 Timeline of Scientific Events in the Anthrax Mailings Investigation
| Project initiated | Final report | Agency/institution/individual conducting the work | Project | FBI document number |
| 10/4/01 | First case of anthrax reported to CDC. The first lab confirmation of an initial clinical identification of B. anthracis from a victim of the letter attacks (Stevens) was done at the Florida State Laboratory in Jacksonville. | N/A | ||
| 10/12/01 | 11/14/01 | FBI and local law enforcement | Collection of biological evidence: 4 envelopes, 17 clinical samples, 106 environmental samples along mail paths (FL, DC, NJ, NY, CT) | N/A |
| 10/17/01 | 10/19/01 | Battelle Memorial Institute (BMI) | Microbiological analyses of letter material identifies 2 Bacillus species: one non-beta-hemolytic (consistent with B. anthracis) and one beta-hemolytic (not further characterized) | B2M1D1 B2M13D4 |
| 10/01 | Beecher | Environmental sampling of mail bags | ||
| 10/01 | 11/01 | CDC | Clinical isolates from stricken patients identified as B. anthracis | N/A |
| 10/18/01 | 11/27/01 | USAMRIID | Initial characterization of letter material (CFU, EM, visual inspection) | B1M1D2 |
| 10/01 | 11/26/01 | Battelle Memorial Institute | SEM-EDX analysis of letter material | B2M13D3, B2M13D8 |
| 11/01 | Armed Forces Institute of Pathology (AFIP) | SEM-EDX analysis of letter material | AFIP, 2001 | |
| 10/01 | 05/02 | Los Alamos National Laboratory (LANL) | Material analysis for evidence of genetic engineering | B1M4 |
| 10/01 | 9/02 | Northern Arizona University | Identification of USAMRIID samples as Ames strain | B1M3 |
| Project initiated | Final report | Agency/institution/individual conducting the work | Project | FBI document number |
| Fall 2001 | 2/28/02 | BMI | Particle size distribution performed on letter samples and some surrogate samples, but not the Dugway Proving Ground surrogates | B2M13D11 |
| Fall 2001 | CDC | Combined epidemiological analysis of all case related to the anthrax mailings | ||
| 11/07/01 | 12/03/01 | CDC | Using phenotypic substrate utilization and 16S rDNA sequencing, Bacillus contaminant identified as B. subtilis | B2M1D2 |
| 11/09/01 | 11/09/01 | FBI | SEM analysis of envelopes | B1M7D16 |
| Fall 2001 | Consortium of agencies (NSF, NIH, DOE, DOJ, FBI, USDA, DOD, and Intelligence Community) formed to advise investigation and provide support resources | N/A | ||
| Fall 2001 | BMI | Silicon incorporation into spore coat | B2M13D7 | |
| Fall 2001 | Spring 2002 | The Institute for Genomic Research (TIGR) | Completion of genome sequence for B. anthracis Porton and “Ames 2001 Florida strain” (clinical isolate); publication in Science, June 2002 | B1M5D1, B1M5D3 |
| Fall 2001 | Spring 2002 | USAMRIID | Detection of phenotypic variants (“morphotypes”) among colonies derived from letter spores | B1M2D12 |
| 12/01 | 12/06 | Dugway Proving Ground (DPG) | Reverse engineering of spore “powders” | B1M13 B1M14 |
| Project initiated | Final report | Agency/institution/individual conducting the work | Project | FBI document number |
| 2/02 | 10/14/02 | Lawrence Livermore National Laboratory (LLNL), National Ocean Sciences Accelerator Mass Spectrometry Facility National Ocean Sciences Accelerator Mass Spectrometry Facility (NOSAMS), Woods Hole Oceanographic Institute | Carbon dating by Accelerator Mass Spectrometry | B1M8 |
| 2/02 | FBI | Subpoenas of laboratories for samples of B. anthracis Ames | ||
| 2/02 | 10/06 | Sandia National Laboratory (SNL) | Elemental analyses (SEM-EDX) of letter material, envelopes, and DPG surrogates (final report not dated) | B1M7 |
| 2/02 | 10/06 | SNL | Silicon analyses | B1M6 |
| 2/01/02 | 8/13/05 | University of Maryland (UMD) | Agar analysis | B1M10 |
| 3/07/02 | 2/01/06 | DPG | Analytical chemistry analysis of spore powders | B1M13 |
| 3/22/02 | 4/14/02 | FBI | Volatile organic compound analysis in evidentiary material | B1M7D2 |
| Early | 7/6/05 | FBI | ICP-OES: elemental | B1M6 |
| 2002 | composition of letter material, culture media, envelope types | B1M7 | ||
| 12/02 | 6/1/04 | TIGR | Whole genome sequencing of Morphs A, B, C, D | B1M5 |
| 8/02 | 2/22/04 | UMD | Heme analysis | B1M10 |
| 8/02 | 8/05 | Edgewood Chemical Biological Center (Army) ECBC | Agar and heme analysis | B1M10 |
| 8/02 | 9/02 | BMI | Agar and heme analysis | B1M10 |
| Project initiated | Final report | Agency/institution/individual conducting the work | Project | FBI document number |
| 10/02 | 2/04 | Commonwealth Biotechnologies, Inc. (CBI) | Contract to develop Morph A assays; only A1 and A3 were validated | B2M5 |
| 3/03 | 5/05 | University of Utah | Stable isotope signatures | B1M9 |
| 3/14/03 | 10/10/03 | Applied Biosystems (AB) | 16S rDNA sequencing of Brokaw letter B. subtilis contaminants | B2M1D4 |
| 7/31/03 | 8/11/03 | Novozymes Biotech, Inc. | B. subtilis contaminant compared to B. licheniformis | B2M1D3 |
| 10/03 | 5/14/06 | TIGR | Multiple locus PCR-based assay for direct comparison of B. subtilis strains to Post B. subtilis | B1M5D2 |
| 10/03 | 6/4/05 | TIGR | Whole genome sequencing of Morph E and B. subtilis contaminant (Post and Leahy) | B1M5 |
| 10/10/03 | 6/30/05 | AB | Genome sequencing of B. subtilis “H2122” (not identified elsewhere) | |
| 3/04 | 2/06 | CBI | Repository (1104 samples) screening for A1 and A3; second screening (300 samples) 7/07-10/07) | B2M5D8 |
| 6/25/04 | 7/05 | CBI | Contract to develop assays for Morphs B and D: both rejected | B2M6 |
| 7/13/04 | 9/14/04 | USAMRIID | Screening of selected samples of FBIR for presence of Morphotypes | B1M2D13 |
| 7/23/04 | 6/7/05 | Midwest Research Institute (MRI) | Contract to develop assay for Morphs B and D; Morph B assay rejected; Morph D assay accepted | B2M8 |
| 11/19/04 | 4/05 | IIT Research Institute (IITRI) | Contract to develop assays for Morphs B and D; Morph B assay rejected; Morph D assay accepted | B2M7 |
| 12/01/05 | 10/10/07 | MRI | Repository screening for Morph D | B2M8 |
| Project initiated | Final report | Agency/institution/individual conducting the work | Project | FBI document number |
| 5/05 | 4/24/07 | IITRI | Repository screening for Morph D | B2M7 |
| 12/05 | 1/07 | TIGR | Contract to develop assay for Morph E | B2M9 |
| 10/06 | 7/07 | Pacific Northwest National Laboratory (PNNL) | Agar and heme analysis | B1M11 |
| 11/06 | 12/07 | NBFAC | Repository screening for B. subtilis contaminant | B2M4D2 |
| 2/15/07 | 12/04/07 | CBSU (FBI) National Bioforensic Analysis Center (NBFAC) | B. subtilis analysis by real-time PCR: screening of repository and other samples | B2M4 |
| 6/25/07 | 8/25/07 | TIGR | Repository screening for Morph E | B2M9 |
| 8/04/07 | 11/30/07 | National Center for Agricultural Utilization Research (NCAUR) | Genetic diversity and phylogenetic analysis of B. subtilis samples | B2M3 |
| 8/29/07 | 8/29/08 | FBI | Analysis of meglumine and diatrizoate in RMR-1029, letter material, other samples | B1M12 |
| 10/05/07 | 10/08/08 | TIGR | Finalization of B. subtilis genome sequence | B2M2D2 |
| 1/09/08 | 6/08/08 | TIGR | Screening of unidentified B. subtilis isolates for presence of sequence specific to Post/Brokaw contaminant | B2M2D3, B2M2D4 |
| 3/27/08 | 9/30/08 | University of Cincinnati | Statistical analysis of FBIR screening data | B2M10 |
to converge again when more information informs decisions about which directions to pursue. This divergence and convergence make a scientific investigation different from a law enforcement investigation, in which the drive toward convergence dominates to a greater degree. In addition, the approach used to gather, process, and analyze evidence can differ between a purely scientific investigation and a law enforcement investigation. Both types of approaches are necessary in a bioterrorism investigation, which also requires attention to public health risks and safety needs (see Box 3-1).
An important feature of science is that observations are made in a manner that is independent of the observer and on the assumption that other observers can and would make the same observations. Science relies on validated methods for gathering observations and making quantitative measurements systematically and reproducibly. Standards must be set for collecting data under controlled and well-specified conditions, assessing possible sources of error, establishing causality (and acknowledging that a relationship is only a correlation when causality cannot be inferred and supported), and applying empirical findings to validate or refute particular hypotheses. New scientific methods must be assessed for their accuracy and reliability, their limitations, and the range of circumstances under which they can be appropriately applied.
The Qualifiers of Certainty in the Biological Sciences
A key question in this study was “Based on the available data, how strong is the apparent association between the letter evidentiary material and a particular source or sample (e.g., flask RMR-1029)?” Some of the committee’s most important findings focus on the strength of a given association and on the conclusions that one should draw from the available scientific data about the nature of the association. Thus, it is important to review briefly the use of terminology to describe the strength of an association.
Quantifying an association, as well as the degree of certainty (or uncertainty) in that association, involves statistical methods (see Chapter 6). Common language involves qualifiers, rather than quantifiable measures, of this association and the degree of confidence in it, which can cause confusion among practitioners from different fields that use the terms. Since the interpretation of these qualifiers and the ways in which they are used differ across disciplines (e.g., statistics, science, law, common language), their use by the committee is clarified here. In the chapters that follow, the committee uses the following four qualifiers of association, listed in order of increasing certainty (decreasing uncertainty):
- consistent with an association
- suggest an association
- indicate an association
- demonstrate an association
BOX 3-1
Bioterrorism Investigations
A crime scene typically is a place where the victims and perpetrators meet in time and space. The traditional crime scene, which may have multiple locations, is the logical place to search for physical evidence leading to the identity of the perpetrator(s). Identifying, collecting, and preserving probative evidence combined with investigative detective work is the usual approach to a successful prosecution. Solving the crime is the ultimate goal of the scene investigation, but there also are other reasons to investigate the crime scene, including: 1) developing investigative leads for detectives; 2) developing specific information in the form of evidence or investigative logic to enable a successful prosecution; 3) locating, collecting, and preserving probative physical evidence that can provide evidence of innocence or guilt; 4) developing information and physical evidence that provides an accurate reconstruction of the events of the crime; and 5) linking multiple crimes through the evidence collected across sites (USDOJ, 2000; Fisher, 2005).
All crime scene investigations require the integration of multiple forensic disciplines through the juxtaposition of science and scene investigative skills. Scientific criminal investigations require an amalgamation of capabilities including scene experience, attention to detail, a skeptical perspective, powers of observation, and the application of logic (Gardner, 2005).
Although bioterrorism event scenes have elements in common with other crime scenes, such as the identification, collection, and preservation of the forensic evidence, they can also differ because of the inherent risks to investigators and to the public. Also, unlike traditional crimes, they may not always involve a location where the participants meet in time and space, as shown by the 2001 B. anthracis mailings in which the dissemination of B. anthracis occurred by means of the United States Postal Service (Jernigan et al., 2002). The perpetrator(s) presumably worked at a distance, so the criminal investigation spanned several locations, including those of the envelopes, post offices, and street postal boxes that might have held contaminated envelopes, and the location(s) where the B. anthracis might have been manufactured. Each scene required a comprehensive and coordinated investigation to find, collect, and preserve the B. anthracis spores.
Future bioterrorism events may differ in the nature of the biological agent or toxin and in the mode of delivery. In general, bioterrorism incidents can be expected to be handled differently than the typical homicide scene investigation because such events require both traditional scene management skills and the special requirements of scenes involving bioagents. Bioterrorism investigators must consider issues such as public safety, operational planning, sampling strategy, packaging, transport, and storage. The immediate imperative to consider public health needs requires finding and collecting the biological agent so that it can be identified expeditiously, as well as defining and containing the environmental risk to those not yet exposed. The investigation and containment must be accomplished while ensuring the safety of investigators and the public during the investigation and while remediating the scene. Sampling strategies must combine the collection and preservation of bioagents with the collection of usual forensic evidence (Budowle, 2006). In some future scenarios, delivery of the biological agent might occur through natural routes of biological transmission, and thus the “crime scene” may be limited to the site at which the biological agent was prepared or delivered.
The expression “consistent with” is frequently used in this report and conveys the weakest level of certainty (greatest amount of uncertainty). In general, when the term “consistent with” is used, it means that an association may or may not be present; the available data can neither rule out nor confirm an association. The term “suggests” denotes a greater level of certainty for an association than “consistent with,” but even here the normal use of the word in science denotes a weaker level of certainty than is implied by the word in everyday parlance. That is, the potential for an association is stronger, and the evidence for the absence of an association is weaker, but both are still possible. In contrast, the terms “indicate” and “demonstrate” denote higher degrees of certainty and these are usually reserved for strong scientific conclusions (i.e., less uncertainty, or less likelihood of an absence of an association). All four levels could potentially be quantified with measures of “statistical significance,” but the committee does not assign such measures in most instances because the data at hand are generally not appropriate for such precise quantification of the degree of uncertainty.
In summary, the reader is cautioned to consider carefully the terminology in this report in light of the fact that the qualifiers of certainty used here are those used most commonly in the scientific literature and that these words can carry different weight in common language and in the courtroom.
3.3 THE FEDERAL COORDINATED RESPONSE AND ASSIGNMENT OF LABORATORY WORK
Oversight and coordination of a complex scientific study are critical. Large teams of scientists have been successful at complex studies (e.g., in particle physics and genomic research) because they have a clear leadership structure for the coordination and planning of their efforts (International Human Genome Sequencing Consortium, 2001; Venter et al. 2001; NRC, 2003). With pressures of time and expense in any study, someone or some group in the research team must make decisions about which avenues to pursue and which to abandon.
In 2001, the FBI had a science laboratory at Quantico, the Hazardous Materials Response Unit, and another team of weapons of mass destruction (WMD) experts, but it did not have the capabilities to handle all of the types of scientific experiments that would be required to examine the evidence in the investigation of the B. anthracis mailings. FBI investigators quickly realized the need to turn to outside laboratories and experts for help. The Bureau immediately formed an internal group that had members from the scientific team, investigative team, and terrorism team. Recognizing the importance of parallel criminal and scientific investigations, the FBI embedded high-level DOJ staff from the internal team, who then advised them throughout the investigation.
At that time, the FBI did not have the organizational structure needed to oversee such a complex, multifaceted, and involved scientific investigation.
In 2003, the agency remedied this organizational limitation by forming a new unit focused on investigations involving chemical, biological, radiological, and nuclear sciences, called the Chemical, Biological, Radiological, and Nuclear (CBRN) Sciences Unit, or the Chemical Biological Science Unit (CBSU).
The committee was told that the team working on the scientific investigation met weekly with the law enforcement team for information sharing, strategy, and coordination. Reports and notes from some of these meetings were shared with the committee late in the process of finalizing this report. The reports indicate the complexity of the parallel tracks of the investigation and document progress of each aspect of the scientific efforts and the decisions to proceed with or abandon particular lines of the investigation.
In the early stages of the investigation, the FBI sought the advice of outside experts to assist in characterizing the properties of the B. anthracis evidence. The engagement of these experts was aided by the creation of an advisory group led by the director of the National Science Foundation, the director of the National Institute of Allergy and Infectious Diseases of the National Institutes of Health, and federal officials from numerous other science agencies. This group met regularly in classified sessions with FBI leaders to hear about the investigation and to provide advice and the names of potential subject matter experts the FBI could engage for assistance.
In addition, several Technical Review Panels were formed consisting of scientists from Department of Energy National Laboratories, academic laboratories, and members of the National Academy of Sciences. Panels were constituted to review the analytical plan (that is, what tests should be done and by whom), the progress of the investigation, and the chemistry and biology techniques used. We reviewed several reports from meetings held in late 2001 and thereafter (FBI Documents, B1M1, B3D1-7). According to these materials and the DOJ report, “At the outset of the investigation, three panels comprised of 33 of the nation’s leading authorities in bioweapons development from the former offensive bioweapons program, microbiology, chemistry, and microscopy were convened to assist the FBI in developing a comprehensive analytical framework to evaluate the anthrax powders recovered from the envelopes and the contamination found in the AMI Building” (USDOJ, 2010, p. 13).
The FBI benefitted from these early informal and regular meetings of senior leadership from other science agencies (FBI, 2009; Colwell, 2009). The FBI also received input from the Department of Defense (DOD), the Intelligence Community, DOJ, CDC, and Armed Forces Institute of Pathology (AFIP) regarding the scientific investigation (FBI/USDOJ, 2011).
As is shown in Table 3-1, the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID) played a central role in the scientific investigation. The facility had provided analytical services to the FBI Laboratory since 1998. During October and November 2001, scientists at USAMRIID were included in the team performing on-site testing at the American Media,
Inc. (AMI) building in Florida, and they conducted the initial examinations of the letter spore preparations for physical characteristics (using microscopy and electron microscopy) and spore viability (see Chapters 4 and 5). USAMRIID scientists also conducted microbiological analyses and identified the dominant and variant morphological colony types that appeared in the evidentiary material (see Chapter 5).
The FBI also sought the help of dozens of outside laboratories. In total, thousands of samples were processed and analyzed by 29 academic, government, and private BSL-3 laboratories across the country (Piggee, 2008; USDOJ, 2010). The work commissioned by the FBI was highly compartmentalized. Most of the laboratories conducting analyses were not aware of other analyses under way (Keim, 2009; Michael, 2009; Weber, 2009). Scientists at these laboratories responded rapidly and provided the bulk of the scientific studies on which the FBI relied in its investigation. The committee read reports prepared by outside scientists responding to specific requests from the FBI and we received reports of periodic reviews of contracted work by panels of experts (B3D1-7). The committee also reviewed reports of work carried out in parallel at the AFIP although it is not clear how closely AFIP and the FBI investigative and scientific teams worked together or coordinated their efforts.
As the scientific investigation proceeded, several laboratories conducted sequential and parallel scientific analyses on the evidentiary material gathered from the letters, environmental samples, and clinical samples (see Table 3-2). These analyses first focused on identifying the nature of the letter and environmental materials, their similarities and differences, their biological, chemical, and physical properties, and, eventually, their similarity to other samples of Ames strain B. anthracis in laboratories around the world. According to the affidavit in support of a search warrant submitted by Postal Inspector Thomas F. Dellafera (Case number O7-524-M-01, October 31, 2007), 16 domestic laboratories and three foreign laboratories (in Canada, Sweden, and the United Kingdom) were identified as having the Ames strain in their inventories prior to the attacks; two additional domestic laboratories were subjected to consent searches, and one domestic laboratory was subjected to a search warrant. A subpoena prepared by the FBI in early 2002 for sample submission specified that only Ames strain samples be submitted; the subpoena protocol for these submissions is described in Chapter 6. In the end, the FBI assembled a repository of over 1,070 Ames strain samples from 20 laboratories, of which 1,059 were viable. Attributes of the samples in the repository were compared against the characteristics of the evidentiary samples (as discussed in Chapters 5 and 6).
According to FBI officials, the focus of these analyses was to provide FBI investigators with scientific leads that could be used to assist in its criminal investigation (FBI, 2009). Chapters 4 through 6 provide in-depth descriptions of the analyses conducted, conclusions reached, and this committee’s findings and lessons learned for the future.
TABLE 3-2 Analytical Techniques Used on the Evidentiary Material
| Technique | Florida environmental and clinical samples | DC, NY, CT, NJ clinical samples | New York Post letter | Brokaw letter | Daschle letter | Leahy letter |
| MLVA (genetic analysis of strain) | X (B1M3D1) | X | X | X | X | X |
| Sequencing of pagA genes | X (B1M4D2) | X | X | X | ? | |
| Complete genome sequencing of wild-type B. anthracis | X | X | X | |||
| Sequencing and PCR analysis to determine whether genetic engineering occurred | X | X | X | X | ? | |
| Partial genome sequencing of B. subtilis | X | X | No B. subtilis | No B. subtilis | ||
| Whole genome sequencing of B. subtilis | X | Not sequenced | No B. subtilis | No B. subtilis | ||
| Phenotypic screens of variants (morphotypes) | X (B1M2D14) | X | X | X | ||
| Whole genome sequencing of morphotype A | X | X | ||||
| Whole genome sequencing of morphotype B | X | X | ||||
| Whole genome sequencing of morphotype C | X | |||||
| Whole genome sequencing of morphotype D | X | |||||
| Whole genome sequencing of morphotype E | X | X | ||||
| Technique | Florida environmental and clinical samples | DC, NY, CT, NJ clinical samples | New York Post letter | Brokaw letter | Daschle letter | Leahy letter |
| SEM analysis to assess size and shape of spores | X | X | X | |||
| Assessment of bulk silicon content | X | X | ||||
| SEM-EDX bulk analysis of silicon | X | X | X | |||
| SEM-EDX to assess silicon in spore coat | X | X | X | |||
| SEM-EDX bulk elemental analysis | X | X | X | |||
| SEM-EDX for spatial resolution and sensitivity | X | X | ||||
| Assays for presence of agar | X | X | ||||
| Assays for presence of meglumine diatrizoate | X | X | ||||
| Headspace GC-MS and infrared spectroscopy to detect VOCs | X | |||||
| Radiocarbon analysis for dating | X | |||||
| Stable isotope analysis to determine growth medium | X | |||||
| Envelope analysis | X | X | X | |||
| Assays of genotypic variants (A, B, C/D, E) | X | X | ||||
GC-MS = gas chromatography-mass spectroscopy; MLVA = multiple-locus VNTR [variable-number tandem repeat] analysis; PCR = polymerase chain reaction; SEM = scanning electron microscopy; SEM-EDX = scanning electron microscope with energy-dispersive X-ray analysis; VOC = volatile organic compounds
3.4 COLLECTION AND ANALYSIS OF CLINICAL AND ENVIRONMENTAL SAMPLES AND CROSS CONTAMINATION
Beginning in October 2001, investigators collected biological evidence from a variety of sources and locations. By the end of the collection phase, B. anthracis isolates were gathered from 4 powder-containing envelopes, 17 clinical specimens obtained from infected patients, and 106 locations along the mail path of the implicated envelopes from Florida, the National Capital Region (Washington, D.C.), New Jersey, New York, and Connecticut, and overseas (Jernigan, 2002).
3.4.1 Clinical and Epidemiological Samples
As noted in Chapter 2, anthrax is generally a rare disease that most often occurs in people who have contact either with infected livestock or with contaminated animal skins or other animal products. Inhalational anthrax is particularly rare (Bush et al., 2001), and the few cases reported in the United States have been due to occupational exposures. Before 2001, the last case of inhalational anthrax in the United States was reported in 1976 (Jernigan et al., 2002). Thus, the occurrence of inhalational anthrax in Robert Stevens, an employee of AMI, was not immediately recognized as such.
Larry Bush, a physician with training in microbiology, was the first to surmise that the Gram-positive bacilli in the patient’s cerebrospinal fluid (CSF) were B. anthracis (Cole, 2009). Phil Lee, at the Florida State Laboratory, provided confirmation based on various tests, including lysis by a B.anthracis-specific virus. Researchers in CDC laboratories used standard microbiological techniques to verify the putative agent as B. anthracis (Jernigan et al., 2002).
Bush and colleagues (2001) point out that in the Stevens case, although there was no typical “occupational exposure” to infected animals or contaminated animal products, the victim was apparently infected at work. No spore-containing letter was ever found, but coworkers reported that on September 19, 2001, Stevens had closely examined a suspicious letter containing powder. Anthrax spores were later found on the victim’s computer keyboard, at other locations in AMI, in asymptomatic coworkers, in the AMI mailroom, and at regional and local postal centers that served the AMI worksite. Three days after his hospital admission on October 2, 2001, Robert Stevens died, becoming the index case for this 2001 bioterrorism event (Bush et al., 2001).
In the following week, as illustrated in Table 1-1 (Chapter 1), cases of cutaneous anthrax were reported in New York City, subsequently linked to exposure to letters containing suspicious powder (Greene et al., 2002). Ultimately, four envelopes containing B. anthracis spores were recovered from media outlets in New York and from government offices in Washington, D.C., or the containment facility created to store potentially contaminated mail. All four envelopes were postmarked by the United States Postal Service (USPS) Trenton Processing and Distribution Center in New Jersey. On October 18,
a case of cutaneous anthrax was confirmed in a postal worker at the Trenton Center, which was then closed. An investigation by CDC and the New Jersey Department of Health was initiated (Greene et al., 2002). Subsequently, CDC and other agencies conducted a combined epidemiological analysis of all cases related to the anthrax mailings (Jernigan et al., 2002). This analysis was a coordinated effort among medical and laboratory facilities and local, state, and federal public health and law enforcement agencies. CDC’s Emergency Operations Center supported local, state, and federal public health investigators in Florida, New York City, New Jersey, the District of Columbia, and Connecticut. Teams also coordinated with the USPIS, the Department of Defense, the FBI, and other federal organizations.
Jernigan and colleagues (2002) summarized the early overall epidemiologic findings. As illustrated in Figure 3-1 from the CDC, a total of 22 cases of bioterrorism-related anthrax were identified—11 inhalational cases and 11 cutaneous cases. Victims were identified in seven states along the east coast: Florida (2 cases), Maryland (3), New Jersey (5), New York (8, including one New Jersey resident exposed in New York), Connecticut (1), Pennsylvania (1), and Virginia (2). Five of the inhalational anthrax patients died, resulting in a fatality rate for inhalational anthrax of 45 percent. Eight of the 11 cases of inhalational anthrax were confirmed as B. anthracis from analysis of clinical specimens. In most cases, the exposures likely occurred in the victim’s place of work. More than half the victims (12 of 22) were mail handlers at the USPS or in government and media industry mailrooms, eight of whom developed inhalational disease and four cutaneous disease. Of the remaining 10 victims, 6 were other media company employees working at locations where mail containing powder was presumed to have been received (AMI, CBS, NBC, and the New York Post). A seventh victim, a 7-month-old child had visited ABC’s offices in New York.
Three other victims—a 61-year-old Manhattan hospital supply room worker, a 51-year-old bookkeeper from New Jersey, and a 94-year-old Connecticut resident—appeared to have no workplace associations. Since postal processing facilities were widely contaminated with B. anthracis, the FBI suggested that cross-contaminated mail was the source of the exposures for these three patients (Jernigan et al., 2002; Greene et al., 2002). Jernigan and colleagues (2002) noted that “The possibility of B. anthracis exposure from envelopes secondarily contaminated from implicated postal facilities greatly extended the group of potentially exposed persons in our investigation.”
The epidemiological investigations found two distinct case clusters separated in time. The first case cluster was related to the two envelopes postmarked September 18, which were apparently mailed in or around Trenton, New Jersey, and delivered in New York City. As shown in Figure 3-1, 11 cases in all were classified as part of the cluster related to the September 18 mailings. Seven cases of cutaneous anthrax occurred in media company employees in New York (5) and postal workers in New Jersey (2). The two cases of inhalational anthrax
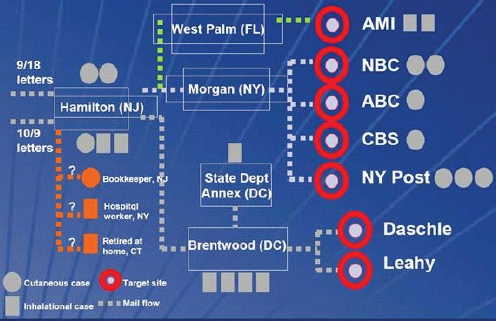
FIGURE 3-1 Trajectory and outcomes of anthrax mailings.
SOURCE: CDC. This image is a work of the Centers for Disease Control and Prevention, taken or made during the course of an employee’s official duties. As a work of the U.S. federal government, the image is in the public domain.
in Florida were also assigned to this cluster, based on the hypothesis that an unrecovered mailing had been sent there. B. anthracis was isolated from environmental samples at six postal facilities along the path followed by the mail to AMI as well as at the AMI building itself. The dates of onset of illness in the two AMI employees in Florida were also consistent with exposure to envelopes mailed in mid-September. Victims in the cluster that followed the September 18 mailings were more likely to have cutaneous disease and to have been exposed at news media sites (Jernigan et al., 2002).
The second case cluster involved the two recovered envelopes sent to the offices of Senators Tom Daschle and Patrick Leahy, also mailed in or around Trenton and postmarked October 9, 2001. All five cases from the D.C. metropolitan area were part of this cluster and all five of these victims contracted inhalational anthrax and worked in postal facilities contaminated by the October 9 letters. Two additional cases of inhalational anthrax occurred in postal employees in New Jersey. In general, the October 9 mailings were associated with more severe illness. Victims in the second cluster were more likely to have been exposed at mail handling facilities along the path to the Senate offices.
Chapter 4 examines the differences in the physical appearance and consistency of the powders between the September and October mailings as reported

FIGURE 3-2 New York Post letter powder.
SOURCE: FBI. This image is a work of the Federal Bureau of Investigation, taken or made during the course of an employee’s official duties. As a work of the U.S. federal government, the image is in the public domain.

FIGURE 3-3 Leahy letter powder.
SOURCE: FBI. This mage is a work of the Federal Bureau of Investigation, taken or made during the course of an employee’s official duties. As a work of the U.S. federal government, the image is in the public domain.
by the FBI (see Figures 3-2 and 3-3). In brief, the powders from the New York Post and Brokaw letters were multicolored and granular in consistency. The envelopes in the second wave of mailings contained a powder of uniform color and smaller particle size, which would facilitate more efficient airborne transmission of the B. anthracis spores and could account for the prevalence of inhalation cases in the second cluster (Hassell, 2009). In addition, despite the fact that the envelopes in the October mailings were unopened during their passage through the postal centers, the use of high-speed processing and sorting machines may have contributed to dispersal of the spores and exposure of postal workers. Greene and colleagues (2002) reported that, although the envelopes containing the B. anthracis were handled in only a small area of the Trenton facility, environmental sampling found evidence of spores throughout the facility.
The B. anthracis isolates cultivated from the clinical specimens of patients, the four recovered powder-containing envelopes, and over 100 environmental samples collected along the suspected path traveled by the contaminated mail were subtyped by the CDC using multiple-locus variable-number tandem repeat analysis and sequencing of the protective antigen gene (pagA). In addition, pagA was amplified and sequenced directly from some clinical specimens. All of the results indicated the presence of the B. anthracis Ames strain (Hoffmaster et al., 2002).
Jernigan and colleagues (2002) noted that their epidemiological investigation had several limitations. Because identification of case patients involved numerous local, state, and federal officials, data collection methods were not uniform. The widespread use of postexposure prophylaxis and the difficulty of obtaining information about potentially exposed persons prevented accurate estimates of anthrax exposure rates. Some cases may have been overlooked because patients might have been administered antimicrobials after being mistakenly diagnosed with other types of infectious diseases, without physicians recognizing the disease to be anthrax. The lack of prior experience with bioterrorism also forced the investigators to refine methods and redefine interventions on a continuing basis.
3.4.2 Crime Scene Environmental Samples
The FBI hazardous materials (HAZMAT) team, with the assistance of scientists from USAMRIID, CDC, USPIS, Environmental Protection Agency (EPA), and contractors, performed environmental investigations to assess the presence and extent of B. anthracis contamination and to guide decontamination and environmental remediation (Jernigan et al., 2002). Environmental samples were collected at contaminated worksites and mailboxes by public health, law enforcement, and other government and contract staff (CDC, 2001a; Sanderson, 2001, 2002). At the time, the FBI did not have biosafety level 3 (BSL-3) laboratory facilities capable of handling B. anthracis, so samples
were tested at laboratories participating in the local, state, and federal investigation efforts as described in Chapters 4 through 6.
Crime scene environmental samples were collected by surface sampling, mostly with RODAC (replicate organism detection and counting) contact plates. Swabs, wipes, high-efficiency particulate air (HEPA) vacuum filtration, and air sampling also were used (Dull et al., 2002; Jernigan et al., 2002; Teshale et al., 2002; Beecher, 2006). Based on the documents provided to the committee, it appears that the FBI and CDC relied on culture-based techniques to detect B. anthracis in the environmental samples (Jernigan et al., 2002; Hoffmaster et al., 2002). In much of the work on samples collected from the environment, colonies propagated on agar plates were presumptively identified as B. anthracis based on morphology (Beecher, 2006). Selected colonies were definitively identified as B. anthracis using standard confirmatory tests. At least one environmental swab sample from AMI was sent to Patricia Worsham at USAMRIID (in June 2005) for detection and identification of B. anthracis variant colony morphotypes. Material from this swab was used to inoculate sheep blood agar. The report of this work by Worsham (2009; FBI Documents, B1M2D14) states that B. anthracis variant morphotypes A, B, and C/D were found, but not morphotype E, in addition to the wild-type colony morphotype. Furthermore, Worsham states that a Bacillus strain was recovered that resembled the B. subtilis found in the New York Post letter. The report states that in October 2006, cell suspensions from 34 colonies that exhibited a variant morphotype, as well as from the B. subtilis-like isolate, were sent to the National Bioforensic Analysis Center (NBFAC) for DNA extraction, and that the DNA from the variant colony morphotypes were to be sent to the Institute for Genomic Research (TIGR) for sequencing of the morphotype A, B, C/D, and E genomic regions. However, according to statements by the FBI to this committee (FBI/USDOJ, 2011), the U.S. Attorney’s Office advised that this sequencing and further characterization of these colony morphotypes from AMI would not be undertaken.
The FBI did not use molecular assays for detecting B. anthracis DNA directly in environmental samples (in the absence of a cultivated isolate). Molecular assays targeting B. anthracis-specific genetic markers had been developed prior to 2001 for detecting this organism in food (Yamada et al., 1999) and in environmental samples (Beyer et al., 1995). An on-site polymerase chain reaction (PCR)-based device was used by the CDC at the Brentwood postal facility in 2001 for preliminary assessment or adjunct analysis of B. anthracis DNA in environmental samples, but this approach had not been validated by them at that time for these types of samples. However, the Biological Aerosol Sentry and Information System was deployed in 2001 for environmental monitoring and incorporated PCR-based detection methods for a variety of biological agents (Fitch et al., 2003).
The committee recognizes that the FBI may have hesitated to apply a newly emerging method to the assessment of forensic evidence before it had been widely
adopted or validated for this purpose; however, under exigent circumstances, it can be appropriate to proceed without completing full validation. The FBI stated to the committee (FBI/USDOJ, 2011) that laboratories in the CDC Laboratory Response Network that were equipped for PCR analysis were overwhelmed with samples early in the investigation, and that the FBI obtained more timely results on the presence of B. anthracis in environmental samples by relying instead on RODAC plate isolation techniques. In any case, by 2004, PCR had been validated for various bacterial agents in environmental samples (Malorny et al., 2004) and could have been performed on the environmental samples from 2001.
In addition to the environmental sampling, nasal swab specimens were collected from potentially exposed individuals to help delineate the area of exposure to aerosolized spores and to determine where persons with inhalational anthrax might have been exposed based on where they worked. According to Jernigan and colleagues (2002), because the sensitivity of nasal swab cultures diminishes with time following human exposure, attempts were made to obtain cultures within seven days of exposure. The presence of B. anthracis from nasal swab cultures was determined by Gram stain and colony characteristics as well as through confirmatory testing by laboratories participating with the local, state, and federal efforts (Jernigan et al., 2002).
Finally, in the new materials provided to the committee it is noted that PCR analysis was performed on human remains from United flight 93 on 9/11/2001 that were identified as those of the hijackers (B3D1). Analysis was performed at USAMRIID and at AFIP for sequences diagnostic of B. anthracis. One assay at USAMRIID gave positive results, but these results were believed by the FBI to be due to laboratory contamination. All other results were negative. As the committee learned at the January 2011 meeting, there were no tests done on remains from any of the other September 11, 2001 hijackers.
3.4.3 Samples from an Overseas Site Identified by Intelligence
In December 2010-January 2011, the FBI first made available to the Committee “AMX Weekly Science Updates” and a newly de-classified document that described the collection and analysis of environmental samples from an undisclosed site outside the continental United States (OCONUS) for the presence of B. anthracis Ames (FBI/USDOJ, 2011, FBI Documents, WFO Report). This work was performed as part of the anthrax letters investigation. Few details were made available to the committee.
At least three sample collection missions were conducted by the FBI and/or partners from the intelligence community at an overseas site because of information about efforts by Al Qaeda to develop an “anthrax program” (FBI/USDOJ, 2011). In May 2004, the FBI and partners from the intelligence community visited an overseas location at which they had been told an anthrax program had been operating, and brought back swab and swipe samples to the United
States. None of the samples grew B. anthracis after incubation in culture media. However, three swab samples were reported as positive for B. anthracis and for B. anthracis Ames-specific sequences by PCR, including swabbings from the outside of an unopened medicine dropper package, a sink, and a sink drain hose. Repeat testing of these three positive samples as part of a group of 15 blinded samples, including soil samples, water blanks and non-Ames Bacillus species, again yielded positive results for two of the three same samples (and for none of the other samples). However, not all replicates of the DNA extracts from the positive samples gave positive results. Apparently, an earlier collection mission to this site, prior to May 2004, by others in the intelligence community had also yielded samples with positive PCR results for B. anthracis DNA and negative culture results. As a result of these findings, a third collection mission was conducted in November 2004 and this time large portions of the site were returned intact to the United States, including the entire sink, drain, and associated plumbing that had been the source of the positive March 2004 samples. These items were extensively sampled, and again tested for both viable B. anthracis and for B. anthracis DNA. This time, according to the June 2008 declassified document, all the tests were negative (FBI Documents, WFO Report).
The committee was provided only fragmentary information about and limited primary data from this work and received them very late in our study. We consider these data to be inconclusive regarding the possible presence of B. anthracis Ames at this undisclosed overseas site. Several scientific and technical issues should be explored in more detail, such as the performance characteristics of the assays, whether or not the assays were validated for use with these sample types, the degree to which samples or sample locations gave repeatedly positive results, interpretation of inconsistent positive results, whether or not the Ames genetic mutations in the anthrax letters were detected in any of these overseas samples, and the natural distribution of B. anthracis strain types in this overseas geographic region.
3.4.4 Letter Material and Cross Contamination
Material was collected directly from the Daschle, Leahy, New York Post, and Brokaw letters, but in varying quantities. As indicated above, no letter was found at the AMI building in Florida, thus only environmental and clinical samples were available for analysis. The decision by the U.S. Attorney’s Office not to pursue molecular analysis on the AMI crime scene samples (FBI, 2011) limits the ability to definitively connect this attack to the material in the recovered letters from New York and Washington, D.C.
The letter addressed to Tom Brokaw at NBC was found after Erin O’Connor, an assistant to Brokaw, developed cutaneous anthrax after opening a letter containing a white powder (Cole, 2009). As mentioned before, the FBI laboratory was not equipped to handle B. anthracis, so all of the New York
samples were sent to the New York Department of Health in Albany or elsewhere. The New York City Health Department officials who tested the Brokaw letter accidentally lost most of the sample and contaminated the laboratory, rendering the space unusable for a critical period of time after the B. anthracis was discovered and before the full scope of the problem was known. As a result, little material from the Brokaw letter remained available for further analyses. A biopsy obtained from a black eschar lesion that formed on O’Connor and was sent to CDC tested positive by immunohistochemical staining for the cell wall antigen of B. anthracis.
The New York Post letter had been thrown into a bin for hate mail and was found after employee Johanna Huden became ill with cutaneous anthrax. The letter had been unopened and the enclosed material was available for future analyses.
On October 15, when staff in Senator Daschle’s office opened an envelope containing a white powder, police quarantined the office and surrounding rooms, shut down the Capitol’s mail system, and suspended public tours. Law enforcement officials in protective biohazard suits took over Senator Daschle’s office. Unlike previous samples, which were sent to the CDC for analysis, the FBI sent the Daschle letter to USAMRIID because of its biocontainment facilities (FBI, 2009). Since much of the material was dispersed when the letter was opened, limited material was available for future analyses.
On November 16, 2001, FBI and EPA HAZMAT personnel found the letter addressed to Senator Leahy in one of 280 barrels of unopened mail collected from Capitol Hill after the discovery of the anthrax-contaminated letter sent to Senator Daschle (FBI, 2008a). The search that resulted in retrieval of the Leahy letter was conducted by teams of HAZMAT workers from the FBI and EPA Criminal Investigative Division (FBI, 2008a). They developed a sampling protocol intended to eliminate the need for HAZMAT teams to sift through each piece of mail in 642 trash bags to find contaminated mail (FBI, 2008a).
The mail was sampled and sorted in a containment facility constructed in a large warehouse and maintained with negative air pressure. The intake and exhaust air was passed through HEPA filters, which trap essentially all particles the size of anthrax spores. Air samplers were used to monitor the air inside and outside the containment area for the presence and quantity of airborne spores. Investigative workers were monitored for exposure by sampling their clothing. In general, the only investigators who were contaminated were those who handled a “hot” bag containing the letter addressed to Leahy laden with spores (Beecher, 2006).
Each bag was shaken in an attempt to distribute any spores that could be present. A swab was then inserted into a small hole in each bag and wiped around the inside. After the swab was withdrawn, the hole was sealed with duct tape and the swab was used to inoculate a Petri dish, which was sent to the Naval Medical Research Center (NMRC) for analysis (FBI, 2010b).
Trace contamination was detected in 62 bags, likely due to a high level of shedding from cross-contaminated mail (Beecher, 2006). Five bags produced more than 100 bacterial colonies from a swab and were considered “hot.” Innovative air sampling was used to maximize the recovery of the target organism (Beecher, 2010). One bag, the one containing the Leahy letter, was orders of magnitude more contaminated than the others. This bag produced between 19,000 and 23,000 spores (or 760 to 920 colony-forming units per liter of air sampled) (Beecher, 2006).
Beecher (2006) examined some of the issues related to cross contamination of mail, mail bags, and the local environment by the Leahy letter. He documented extensive contamination of personnel especially during physical handling of the spore-laden letter. In the conduct of this work, the investigators followed thoughtful and appropriate practices and procedures for the purpose of minimizing artifactual cross contamination. Of note, they identified a correlation between the degree of letter contamination and the previous handling of the letter by sorting machines in the Trenton and Washington, D.C., postal processing and distribution centers. The insights from this work proved useful in developing a more detailed understanding of the route taken by the anthrax letters from the point of deposit through the U.S. Postal System to the site of delivery. Although suggestive of a mechanism and scenario by which Ottilie Lundgren, a resident of Oxford, Connecticut, might have developed inhalational anthrax (cross contamination of mail en route to her home), the committee lacked sufficient information (e.g., other possible exposures, unusual susceptibility to low numbers of anthrax spores) with which to assess the plausibility and likelihood of this mechanism and this particular scenario.
On September 1, 2001, Defence Research Establishment Suffield (DRES) in Canada released the results of a study (FBI Documents, B2M11D1) that had been designed to measure and better understand the dispersion of spores that might occur after the opening of an envelope containing B. anthracis spores. This study involved a series of experiments in which envelopes containing either 0.1 or 1.0 gram of B. globigii spores at a concentration of ~1 × 1011 cfu/g (as a surrogate for B. anthracis spores) were opened in a DRES aerosol test chamber that was configured to represent a mail room or office. The chamber measured 18 × 10 × 10 ft (i.e., with a volume of 1,800 cu ft) and had a recirculating air handling system operating at 1,050 cu ft/min. The presence of spores at various sites in the chamber was assessed using culture-based approaches, not molecular detection methods. The results showed that the act of handling or opening these envelopes was “far more effective than initially suspected” in causing dispersion of spores in the chamber. Particles of respirable size were released quickly and spread throughout the chamber, such that after the opening of a 0.1 g spore envelope, 10 minutes of exposure to the air in the chamber would have provided a dose 480-fold greater than the amount needed to kill a human with 50 percent probability. The investigators noted that envelopes were
more likely to cause cross contamination of the local environment, including the envelope handler, if the open corners of the envelope were not deliberately sealed by the preparer. The investigators at DRES did not seal the corners of the envelopes they used in these experiments. However, the corners of the envelopes mailed through the U.S. Postal System with B. anthracis spores in September and October 2001 apparently were sealed. This study was valuable in revealing the potential speed, magnitude, and spatial distribution of environmental contamination by spores subsequent to the handling or opening of a spore-laden envelope.
3.5 COMMITTEE FINDINGS AND RECOMMENDATIONS
The events of autumn 2001 unfolded rapidly, with CDC’s initial public health response quickly extended to a major criminal investigation under the control of the FBI. A multipronged investigative strategy emerged for the scientific investigation, with one set of activities focused on understanding the characteristics of the material in the letters and another on developing and conducting a comparative analysis of these evidentiary materials against samples collected from the scientific community. The science and technology that formed the basis for these analyses evolved rapidly and had a major impact on the field of microbial forensics. Although the public health crisis largely subsided after the last victim died in November 2001, the scientific investigation continued until 2008 and the criminal investigation continued until the case was closed in February 2010 (see Table 3-1).
Finding 3.1: Over the course of the investigation, the FBI found and engaged highly qualified experts in some areas. It benefited from the unprecedented guidance of a high-level group of agency directors and leading scientists. The members of this group had top secret national security clearances, met regularly over several years in a secure facility, and dealt with classified materials. The NRC committee authoring this report, in keeping with a commitment to make this report available to the public, did not see these materials.
In a complex investigation during a period of extreme national urgency such as this one, it is imperative to recruit and make wise use of the best and most relevant expertise. The Bureau regularly briefed its high-level advisory group in a secure setting where classified material was reviewed. They also relied on subject matter experts and expertise from other government agencies. The committee recognizes, in agreement with the FBI (FBI/USDOJ, Jan 2011) that the unique skills and expertise needed to conduct microbial forensic examinations might, in some cases such as this one, require reliance on the same set of scientific personnel, who also might be considered potential suspects. Thus it is important to have in place an external oversight structure with the
capacity to recognize where relevant expertise and conflicts might reside and to provide guidance and coordination for the overall scientific investigation. In addition, the small number of laboratories with expertise in a given pathogen or technique requires that a certain level of independent oversight be maintained.
Finding 3.2: A clear organizational structure and process to oversee the entire scientific investigation was not in place in 2001. In 2003, the FBI created a new organizational unit (the Chemical, Biological, Radiological, and Nuclear [CBRN] Sciences Unit, sometimes referred to as the Chemical Biological Science Unit, or CBSU) devoted to the investigation of chemical, biological, radiological, and nuclear attacks. The formation of this new unit with clearer lines of authority is commendable.
Weekly meetings occurred between the science team and those leading the criminal investigation. The results of these meetings and the way the scientific, criminal and legal aspects of the investigation interplayed will be beneficial in preparing for future attacks (B3D1). In addition, the development of a new U.S. Government microbial forensics infrastructure and research strategy will be important steps toward enhancing future capabilities for attribution in the event of a biological attack (Pesenti, 2010).
Finding 3.3: Investigators used reasonable approaches in the early phase of the investigation to collect clinical and environmental samples and to apply traditional microbiological methods to their analyses. Yet during subsequent years, the investigators did not fully exploit molecular methods to identify and characterize B. anthracis directly in crime scene environmental samples (without cultivation). Molecular methods offer greater sensitivity and breadth of microbial detection and more precise identification of microbial species and strains than do culture-based methods.
The committee recognizes that the circumstances of 2001 created an abundance of samples and associated work for the LRN (laboratory response network) resulting in a decision to use RODAC plates for analysis of the environmental samples. Thus, the FBI did not confirm the presence of the letters-associated genetic mutations in the environmental samples. In the nine years since that time, dramatic advances in high throughput sequencing technology have greatly improved the ability to detect and characterize rare strains and species in complex environmental samples2 (for further, related discussion,
__________
2 Multiple studies in each of a number of diverse environmental settings have demonstrated the feasibility and reliability of resolving strain-specific fine genomic structure (at the level of SNPs and local structural rearrangements) in highly complex biological samples using next-generation sequencing technology and metagenomic approaches. Examples include strain resolution and gene
see Finding 6.8 in Chapter 6). In thinking of future incidents, it is imperative, based on lessons learned from this investigation, to anticipate the types of situations and circumstances that might affect evidence collection, preservation, and documentation, and to employ protocols and procedures that ensure the best possible outcomes.
Finding 3.4: There was inconsistent evidence of B. anthracis Ames DNA in environmental samples that were collected from an overseas site.
At the end of this study, the NRC committee was provided limited information for the first time about the analysis of environmental samples for B. anthracis Ames from an undisclosed overseas site at which a terrorist group’s anthrax program was allegedly located. This site was investigated by the FBI and other federal partners as part of the anthrax letters investigation. The information indicates that there was inconsistent evidence of Ames strain DNA in some of these samples, but no culturable B. anthracis. The committee believes that the complete set of data and conclusions concerning these samples, including all relevant classified documents, deserves a more thorough scientific review.
Finding 3.5: As was done in the anthrax investigation, at the outset of any future investigation the responsible agencies will be aided by a scientific plan and decision tree that takes into account the breadth of available physical and chemical analytical methods. The plan will also need to allow for possible modification of existing methods and for the development and validation of new methods (see Chapter 4, Section 12).
The scientific investigation of any future biological attack would greatly benefit fromrobust independent oversight and ongoing review. To accomplish this, the government should maintain a standing body of scientific experts with proper security clearances who are fully briefed on matters of importance for preparedness and response to a biological attack. When an investigation is launched, members of this group could help guide the scientific investigation.
In preparing for future investigations, all relevant U.S. government agencies and departments will need to work together to ensure that independent,
__________
family structure in ocean surface water (see multiple publications from the Sorcerer II Global Ocean Sampling Expedition), evidence for rare strain ecotypes in acid mine drainage (see multiple publications from the Banfield group at UC Berkeley), and gene family sequence microheterogeneity in prophage genomes that have been reconstructed from shotgun sequencing of virus-like particles in human feces (e.g., see supplemental data from Nature 466:334, 2010). This large and growing body of work strongly suggests that the application of these same techniques to some of the environmental samples from the anthrax letters case might provide additional clarity about B. anthracis genome sequence variants and the relationships among the strains in the samples and the letters.
high-quality, external science advice is available from individuals with expertise in critical scientific areas likely to be relevant given anticipated scenarios for scientific investigations. By identifying a core set of external experts and convening this group ahead of time, productive working relationships could be established between this group and members of the government’s bioforensics community. When a new investigation is launched, this core set of external experts could assist in recruiting others with more specific expertise relevant to the investigation under way. For example, the FBI included some expert scientists early in the anthrax letters investigation, but it does not seem to have sought formal expertise in statistics until the investigation was nearly completed. Because many inferences depend on the design and analysis of datasets that may be complex, as in this case, for any similar investigation in the future, it will be important that the FBI consult with expert statisticians throughout the processes of experimental design and planning, sample collection, sample analysis, and data interpretation. The committee recognizes that much has been done by the government over the past several years to build and enhance this important infrastructure and set of resources, although the scope of this study did not include assessment of the current infrastructure.
When an investigation is launched, the panel of external experts could recommend and review strategies, protocols, and procedures; help with the development of new methods and scientific approaches; provide advice on the selection of contract scientists and additional outside experts; assist in data interpretation; and help generate alternative hypotheses. Members of the panel should not be directly involved in conducting the scientific investigation itself. It will also be important that the panel have an ongoing role throughout the course of the investigation and be briefed on all of the science that is contemplated and pursued. Consistent documentation of recommendations and input, and the subsequent responses, will be of great value.
In the future relevant agencies should review and periodically update appropriate protocols and experimental designs to use best strategies for preserving evidence, exploiting samples for scientific information, and meeting subsequent legal challenges. Protocols should ensure that clinical and environmental evidence is properly collected, preserved, documented, and analyzed to maximize the utility of the samples collected. Furthermore, state-of-the-art molecular methods, such as next-generation nucleic acid sequencing techniques, offer great potential for characterizing clinical and environmental samples (see Finding 6.8 in Chapter 6).
Recommendation 3.1: A review should be conducted of the classified materials that are relevant to the FBI’s investigation of the 2001 Bacillus anthracis mailings, including all of the data and material pertaining to the overseas environmental sample collections.
The committee did not receive nor review classified material. In November 2010 discussions with FBI and DOJ leadership regarding this report, we were made aware of additional information that would require review of classified material. Due to the lateness of this revelation and the importance we placed on issuing a timely report, and the agreement between the NRC and the FBI that all material we considered be publicly available, the committee did not undertake this additional review of classified material.
Recommendation 3.2: The goals of forensic science and realistic expectations and limitations regarding its use in the investigation of a biological attack must be communicated to the public and policymakers with as much clarity and detail as possible before, during, and after the investigation.
Communicating with the public and policymakers is extremely important in order to ensure that accurate information is available and to minimize unrealistic expectations. Special attention will need to be paid to communicating scientific information to these groups in an accurate and credible manner, especially if the information will play a critical role in the investigation.
When presenting to the public the findings of an investigation that involve scientific evidence, especially one as important as the anthrax letters investigation, officials will need to make every effort to have scientists verify the accuracy of the scientific information they report. The inaccurate reporting of facts or the overstatement of scientific evidence is a disservice to the public. In the anthrax letters investigation, there were repeated claims that all of the attack letters contained all of the genotypic variants (see Chapter 6 and Finding 6.7) that implicated flask RMR-1029 as the source of the anthrax spores, when in fact not all of the letters were checked for these variants. Of even greater concern, because it suggested possible deception by the suspect, the strength of the evidence was overstated that a disputed sample submitted by the suspect had not come from the proper source (see Chapter 6 and Finding 6.4). Similar mistakes can be avoided in the future by involving the relevant scientists in fact checking of the reports before they are released.




























