Many pathogens and allergens are profoundly affected by environmental conditions. Their survival may be directly influenced by temperature, humidity, or moisture, or their availability may depend on the distribution, abundance, or behavior of their hosts or vectors. A changing climate will thus affect human exposure to these agents.
This chapter addresses indoor environmental quality concerns associated with the infectious agents and other pests that research suggests may be influenced by climate-change–induced alterations in the indoor environment. The chapter also touches on exposure to chemicals used to control pest infestations. Exposures that are directly related to dampness are the subject of Chapter 5.
Two earlier National Academies reports have addressed issues relevant to the material discussed in this chapter. The 2001 National Research Council report Under the Weather: Climate, Ecosystems, and Infectious Disease (NRC, 2001) and the 2008 Institute of Medicine workshop summary Global Climate Change and Extreme Weather Events (IOM, 2008a) take on the larger question of the linkages among climate, ecosystems, and infectious disease. A white paper commissioned by the US Environmental Protection Agency (EPA) in conjunction with the present effort discusses the potential effects of climate change on microbial air quality in the built environment (Morey, 2010).
Infectious diseases have been major drivers of evolution and of human evolution in particular. The vast majority of infections are acquired from the environment or transmitted from humans or other animals. Therefore, factors that affect the physical environment, how we build in it, and how we share it with other humans are critical determinants of the infections to which we are exposed and how we perpetuate the exposures. Seasonal variation—a complex summing of multiple influences ranging from sunlight to moisture to wind speed and varying by region—has also been recognized as a critical influence on infectious-disease epidemiology dating back to Hippocrates (Naumova, 2006). Thus, climate change in general and indoor-air exposure in particular are major elements in the spread or interruption of infectious diseases in humans. Despite the extensive knowledge base on the effects of climate change on environmental growth of microorganisms and their vectors and hence infections, however, data on the effects of climate change on indoor air and infectious diseases are incomplete.
This section briefly reviews some of the most pertinent model systems that highlight elements of the knowledge in direct effects of climate on infectious disease. It explores them by category of infection, inasmuch as each kingdom (for example, bacteria, fungi, and viruses) has distinct features and is involved in different processes and exposures. One critical factor is that air and moisture, and therefore water, are inextricably linked. Most microorganisms are exquisitely sensitive to moisture, either requiring it or avoiding it. Therefore, the study of indoor air is closely linked to the state of indoor water, its aerosols, and the magnitude of humidity. Furthermore, pipes and other water-delivery systems are prone to development of biofilms, thin, removal-resistant layers of metabolically inaccessible bacteria that are constantly available for release into water and indoor air through taps, showers, humidifiers, and the like.
Respiratory Viruses
Experience dating back thousands of years has taught that infectious diseases can be affected by seasonal changes; this suggests that environment plays a critical role in the modulation of disease load, spread, and susceptibility. Obvious and recurring examples are provided by the respiratory viruses, most notably influenza viruses, respiratory syncytial virus (RSV), and the rhinoviruses. Mechanisms of spread are varied and include aerosol, fomite,1 and direct contact. Direct contact, such as hand-to-hand transfer, is the most easily modified and is a major contributor to the spread of respira-
_____________________
1 Fomites are inanimate objects or substances—a door knob, for example—that function to transfer infectious organisms from one individual to another.
tory viruses. Fomite spread is affected by ambient humidity, which can in turn be affected by indoor air.
Influenza Viruses
Influenza viruses continue to account for substantial annual morbidity and mortality interspersed with periods of increased activity. The 2009–2010 H1N1 influenza epidemic is estimated to have involved around 61 million infections, 274,000 hospitalizations, and more than 12,000 deaths in the United States (CDC, 2010b).
Although there has been prolonged controversy over the environmental correlates of influenza epidemic spread, it appears that absolute humidity—the amount of water vapor in a given volume of air—is a critical determinant (Shaman and Kohn, 2009; Shaman et al., 2010a,b). In contrast, relative humidity—the amount of water vapor in a given volume of air at a given temperature expressed as the percentage of the maximum possible for that temperature—is well regulated in the indoor environment and appears not to be as important a determinant of influenza transmission and spread. However, studies by Myatt et al. (2010) show that increased absolute humidity and relative humidity, achieved by the use of indoor air humidification, can lead to substantial reductions in viable influenza virus.2 Overall, the effects of humidity on influenza virus outbreaks and peak epidemic periods are greater in temperate than in tropical environments. In some tropical and subtropical settings, relative humidity has been more closely associated with influenza epidemics (Tang et al., 2010a,b). Because periods of high relative humidity corresponded to periods of increased indoor time and air conditioning, the population-based correlations are confounded. However, because indoor air conditioning affects indoor temperature and humidity, these require more investigation to determine whether the critical aspects of influenza spread are determined by the indoor or outdoor environmental conditions. The different results in temperate and tropical zones may reflect differences in viral and human biology in those regions. However, comparative studies for tropical and subtropical regions for respiratory transmission have not been completed in the United States.
Respiratory Syncytial Virus
RSV is the greatest cause of bronchiolitis and pneumonia in infants worldwide and causes up to about 125,000 hospitalizations in US in-
_____________________
2 As discussed later in this chapter, though, increased humidity may create a more hospitable environment for mold growth and accelerate the degradation and subsequent off-gassing of building materials and furnishings.
fants each year. In the US elderly population, it accounts for an estimated 177,000 hospitalizations and 14,000 deaths (CDC, 2010). RSV appears to contribute to invasive pneumococcal disease more than influenza viruses do (Murdoch and Jennings, 2009; Talbot et al., 2005; Watson et al., 2006).
Like influenza virus activity, RSV activity is highest in temperate climates during fall and winter months and into spring. However, there can be variability in the time of onset and duration, at least in more subtropical regions (CDC, 2010a). Unlike influenza virus, RSV is stabilized by higher humidity, and transmission in some studies correlates with relative humidity, lower temperature, and increased cloud cover (Meerhoff et al., 2009). Whether the mechanisms of these factors are due to direct effects on the virus or to indirect effects in driving people indoors into crowded environments is an open question. In some settings, such as Indonesia, RSV activity correlated strongly with rainfall and temperature (Omer et al., 2008). However, the apparently differing epidemiology in temperate and tropical climates remains incompletely explained (Welliver, 2009). In Spain, RSV admissions of infants with severe disease were strongly associated with lower temperature and lower absolute humidity (Lapeña et al., 2005).
Rhinovirus
Human rhinovirus (HRV) is a common and relatively mild pathogen, but one that by its very ubiquity and frequency has a major impact on human health, especially in the setting of pre-existing airway disease like asthma. Adults may have up to four bouts per year, typically in the fall through spring, accounting for up to 62 million cases in the United States annually (Sloan et al., 2011). In addition, because HRV is highly transmissible, settings that favor human-to-human and fomite transmission tend to result in relatively high rates of HRV during certain times of the year. Less research has been conducted on HRV than on influenza and RSV, in part because these latter organisms’ morbidity and mortality are much higher and their etiologies somewhat less complex.
Human rhinoviruses are comprised of three main groups—A, B, and C—which replicate in the epithelial cells of the upper and lower respiratory tracts, leading to cough, wheeze, and rhinorrhea (Dulek and Peebles, 2011). Allergic triggers act along with HRV to fuel the exacerbation of asthma. Extensive work has shown that HRV is one of the most prevalent cofactors in asthma exacerbations, making their role in overall medical care critical to understand and interrupt.
A few studies address the determinants of HRV transmission and prevalence in indoor environments. Myatt et al. (2004) showed that the amount of HRV recovered from building air handling filters varied with the amount of outside air entrained, suggesting that HRV transmission might be influ-
enced by the number of air exchanges in the work environment. Singleton and colleagues (2010) found that HRV was recovered from 44% of Alaskan native children hospitalized with a respiratory infection, but this rate was quite close to that in control children who were not hospitalized. Tovey and Rawlinson (2011) note that the rates of asthma rise precipitously two to three weeks after the start of school, indicating that some new exposure in the classroom is responsible. The authors hypothesize that these factors include HRV as well as numerous other costimulators of asthma such as endotoxin, proteins, and allergens. du Prel and colleagues (2009) found that HRV rates are associated with higher humidity levels, which might become more common as a result of climate change.
Gram-Negative Bacteria
The gram-negative bacteria present special issues in climate-associated infectious-disease epidemiology. They are not dependent on human-to-human spread, are not dependent on human inhabitation for survival, and have the ability to form biofilms—slippery, poorly penetrable slimes that cover the inside of water conduits. Given their close ties to the environment and their access to humans through water consumption, aerosol generation, heating, and cooling, the epidemiology of gram-negative rod infections is a window into infectious diseases in the setting of climate change.
Legionella
From its initial recognition as a cause of human respiratory disease, Legionella infection has been closely tied to water-droplet exposure in hotels and hospitals (Stout and Yu, 1997). However, the modes of transmission clearly can involve both aerosol spread (by water misters in grocery stores, for example) and aspiration. Spread from potting soil has also been well documented (de Jong and Zucs, 2010).
Regardless of the exposures or the modes of transmission, it is clear that legionellae are relatively common in some water supplies and has seasonal variation. In a case-crossover study in the greater Philadelphia area, Fisman and colleagues identified summertime occurrence of reported Legionella pneumonia to correlate with rainfall and increased relative humidity in the preceding week or so, rather than temperature (Fisman et al., 2005). Whether that reflects increased recruitment of legionellae into the water supply through rainfall, increased survival in higher humidity, indoor transmission, or outdoor transmission remains to be concretely determined. However, it is clear that in many instances, such as in hospitals, Legionella transmission is presaged by high levels of bacterial or bacterial DNA recovery from ambient water sources, such as faucets (Feazel et al., 2009). This
dynamic reservoir of organisms probably serves as the source of aerosol generation, the source of bacteria that can be aspired by predisposed hosts, or both. Thus, indoor water clearly influences Legionella transmission and is itself influenced by regional environmental factors.
Pseudomonas aeruginosa
Stapleton et al. (2007) studied the incidence and causes of keratitis in contact-lens wearers in Australia. They found that Pseudomonas aeruginosa accounted for a plurality of cases and that it varied with higher mean minimum temperature but not humidity. Conducting their study in a country with well-characterized tropical and more temperate zones, they found that although P. aeruginosa was most common in the tropical regions, gram-positive organisms, such as Staphylococcus aureus, predominated in more temperate regions (Stapleton et al., 2007). Perencevich et al. (2008) studied the effects of seasonal temperature on nosocomial infection rates at the University of Maryland Medical Center. On review of almost 218,594 cases and 26,624 unique cultures, they found that rates of some gram-negative bacillary infections, including P. aeruginosa infections, were higher during warmer months and that rates of P. aeruginosa infection increased in relation to temperature rise. Gram-negative organisms that showed similar seasonal variation included Acinetobacter baumanii, Enterobacter cloacae, and Escherichia coli. Rates of gram-positive bacteria, such as S. aureus and Enterococcus spp., were not increased over the same periods and did not show similar relationships to temperature. Those infections occurred in hospitals, so they are reflections of effects of indoor environment, but they presumably reflect some changes in the outdoor environment as well. That other nosocomial pathogens, such as S. aureus, did not vary in the same pattern excludes simple effects of climate on human practices and suggests a more intrinsic effect of climate on gram-negative nosocomial pathogens.
As mentioned above, Perencevich et al. (2008) showed that gram-negative nosocomial infections increased with increasing temperature in Baltimore. In a national survey, McDonald et al. (1999) also found seasonal variation in Acinetobacter baumanii nosocomial infections but not in P. aeruginosa infections. They also noted marked differences in regional rates of A. baumanii infections, with higher rates in the eastern than western parts of the United States.
Mycobacteria
The Mycobacteriaceae are typically environmentally hardy gram-positive rods that include the high-grade primate pathogen Mycobacterium tuberculosis, the more numerous environmental or nontuberculous myco-
bacteria, and M. leprae, the agent of leprosy. Some of these organisms have emerged as agents of lung infection in patients who have underlying lung diseases that lead to impaired clearance of respiratory secretions. Those diseases are best exemplified by cystic fibrosis, a genetic disease in which impairment and dysfunction of the airway-lining cilia lead to the airway-widening condition known as bronchiectasis. Bronchiectasis is a common feature of the other syndromes in which nontuberculous mycobacterial infections occur, including primary ciliary dyskinesia, alpha-1 antitrypsin deficiency, and hyper-IgE recurrent-infection syndrome (Zoumot and Wilson, 2010).
The role of environmental exposure, including exposure to the indoor environment, in nontuberculous mycobacterial infection has recently received intense interest. The nontuberculous mycobacteria live in temperate and tropical waters and soils throughout the world. Unlike M. tuberculosis and M. leprae, which depend almost exclusively on human-to-human spread for their propagation, the nontuberculous mycobacteria are environmental opportunists that live in biofilms and can survive otherwise hostile environments because of their waxy cell walls (Falkinham, 2010). Feazel et al. (2009) showed recovery of M. avium complex genetic signatures from biofilms collected from inside showerheads in homes. Other organisms were also detected, including legionellae. Falkinham et al. (2008) reported a case of pulmonary infection with a particular species of M. avium complex that was recovered from the home water supply; this suggested spread from the household water to the patient. That potential mechanism of spread has been expanded on by Chan and Iseman (2010). The occurrence of pulmonary nontuberculous mycobacterial infection is highest in cystic fibrosis patients who have the mildest forms of disease, especially in women (Rodman et al., 2005).
Fomites
Increasing relative humidity and temperature outdoors will probably lead to increased indoor dampness and dampness-related health effects. As is the case with many infectious-disease vectors, the effects of temperature and relative humidity may increase or decrease the survival of viruses and bacteria and facilitate the persistence of infectious fomites (Boone and Gerba, 2007). Increases in environmental temperature decrease the survival of many viruses. For example, the H5N1 avian influenza virus persisted on duck feathers and on surfaces for long times but only at lower temperatures (Wood et al., 2010; Yamamoto et al., 2010). The combination of a stable indoor environment and increased dampness may actually decrease the transmission of some respiratory viruses and increase the survival of other pathogens on fomites, such as the ones that harbor bacteria and mold
(Boone and Gerba, 2007; Gubler et al., 2001). Increased dampness indoors, possibly exacerbated by building deterioration, may exacerbate or increase the risk of developing select respiratory diseases caused by mold and bacterial exposure (IOM, 2004; WHO, 2007).
Fungi
Fungi pose a special set of problems because they are ubiquitous, grow easily in the environment, and cause human diseases. However, the language surrounding fungal interactions with humans is fraught with imprecision, which leads to confusion. In addition, there are several distinct types of fungi, including yeasts, molds, and dimorphic yeasts (fungi that live as yeasts under one set of circumstances but can act like molds in other circumstances) (Holland and Vinh, 2009). The distinctions are important because the dimorphic yeasts are able to live both in the environment and in humans and cause some degree of invasive disease even in healthy humans. Examples include Histoplasma capsulatum, Coccidioides immitis, Blastomyces dermatitidis, Sporothrix schenkii, and Paracoccidioides brasiliensis. Those agents are relatively regional in their distribution and are therefore often referred to as endemic fungi. In healthy hosts, they can cause usually self-limited respiratory illnesses, such as valley fever due to C. immitis. They are organisms that live in the upper layer of soil outdoors and are rarely associated with indoor exposures and have rarely associated with indoor exposures to date. However, a white paper commissioned by EPA (Morey, 2010) suggests a mechanism by which this could change. It indicates that the upper layer of soil is prone to disturbance by dust storms, which may become more common in geographic areas that experience drought because of shifts in climatic conditions. This may in turn lead to greater indoor penetration of pathogenic fungi contained in soil and to higher indoor exposures in the absence of enhanced HVAC filtration or settled dust removal.
Invasive fungal infections are quite rare in humans and occur almost exclusively in the setting of immunocompromise, either inborn, such as some primary immunodeficiencies, or acquired, such as that acquired through transplantation or chemotherapy. However, with the advent of more drugs that affect immunity, such as tumor-necrosis factor alpha–(TNF-α)-blocking agents used for rheumatoid arthritis and inflammatory bowel disease, the number of people at risk for the development of fungal infection is increasing (Tsiodras et al., 2008). In susceptible persons Aspergillus fumigatus, a thermotolerant filamentous mold, can cause invasive disease that is usually spread by inhalation. Pneumonias that occur in the setting of immunocompromise carry high morbidity and mortality.
In the nonimmunocompromised host, the most important fungal disease in the respiratory tract is allergic bronchopulmonary aspergillosis
(ABPA), a syndrome of allergic response to fungi that is most common in those who are atopic, those who have cystic fibrosis, and those who have asthma (Patterson and Strek, 2010). Allergic fungal sinusitis is similar in that eosinophil-rich secretions become dense and involved with fungi without tissue invasion; in this case, the organisms involved include the dematiaceous (dark-walled) molds Bipolaris spicifera and Curvularia lunata or Aspergillus fumigatus, A. niger, and A. flavus (Schubert, 2009). The syndromes of chronic fungal rhinosinusitis are regionally concentrated around the South and Southwest of the United States. These allergic respiratory syndromes straddle the lines between infection, colonization, and allergy.
Synthesis
Climate change has many effects on infectious diseases, some malign and some ameliorative. How we adapt the indoor environment to the continuing changes in the outdoor environment will be critical determinants of how we affect the occurrence and spread of infectious diseases. In particular, effects on moisture, temperature, and the organisms trafficked into our homes, places of work, hospitals, and schools in water will determine the rates of viral, bacterial, mycobacterial, fungal, and allergic diseases.
Indoor environments contain a number of unwelcome insects, other arthropods, and invasive animals. All of these are at some level sensitive to environmental conditions, but some are more susceptible to the conditions associated with climate change. This section summarizes the available literature on the characteristics of these pests; the health effects of exposure to the allergens and microbial agents that they produce, host, or carry; and how climate-change–induced alterations in the indoor environment—including changes in occupant behavior—may affect adverse exposures associated with them.
House Dust Mites
House dust mites are microscopic arthropods that are ubiquitous in indoor environments. They are among the most important sources of allergens in house dust and of allergic disease in the United States (IOM, 2000).
Exposure
Voorhorst and colleagues were the first to show that dust mites of the genus Dermatophagoides were the source of “house dust” allergens
(Voorhorst et al., 1969). Dermatophagoides pteronyssinus (de Boer and Kuller, 1997; van Strien et al., 1994; Voorhorst et al., 1969) and D. farinae (Antens et al., 2006) are commonly recovered in home settings. D. farinae is the hardier of the two (Arlian, 1975; Arlian and Veselica, 1981). An intervention study showed that the major allergen from D. pteronyssinus may have been decreased by an extremely dry (and cold) winter during the study period rather than by the home interventions themselves (Brunekreef et al., 2005; Gehring et al., 2005).
Dust mite viability is highly influenced by environmental conditions. There may be some inferences that as the climate warms, dust mites will thrive (Ayres et al., 2009). That is not entirely true. As noted in Chapter 2, although some regions of the country will experience warmer climates, they will not necessarily experience higher humidity. The critical factor for dust mites is water activity (Aw), which is relative humidity at a surface. Dust mites do not have lungs that can condition the air; rather, they conduct transpiration through their exoskeletons. A decrease in ambient relative humidity (which is paralleled by a drop in Aw) can affect dust mites not only in laboratory settings (Arlian, 1975; Arlian and Veselica, 1981) but in the home (Arlian et al., 2001; Cabrera et al., 1995; Harving et al., 1994) and at a community level (Acosta et al., 2008; Chew et al., 1999).
New York and Boston are coastal cities, but many of their homes can be dry in winter, and this factor eradicates the dust mite population. Studies indicate that increased indoor temperature in those communities has not been accompanied by an observed increase in the dust mite population; rather, dust mites decreased (Acosta et al., 2008; Chew et al., 1999). The homes where overheating was measured in these studies were multifamily apartment buildings whose residents had little control over their heating. The heating was turned on (building wide) early in fall and turned off late in spring. Figures 6-1 and 6-2 illustrate how overheated apartments compared with single-family homes whose residents had more control over their heating.
A change in climate could also affect the ecologic niches of some types of dust mites in such a way that the geographic patterns of endemic dust mites could change. As discussed earlier, some dust mites are more sensitive to humidity than others. The Dutch intervention study described earlier (Brunekreef et al., 2005) showed not only that dust mite levels decreased in this coastal country but that the profile of dust mite taxa had changed. Although it was not highlighted in the study, careful examination of one of the figures shows that between the beginning of the study (1996) and eight years later, Der f 1 (the major allergen from D. farinae) apparently became the most highly concentrated allergen in house dust (Antens et al., 2006). Even if humidity does not change substantially, warmer climate patterns are predicted, and this (in the absence of any adaptation measures, such as
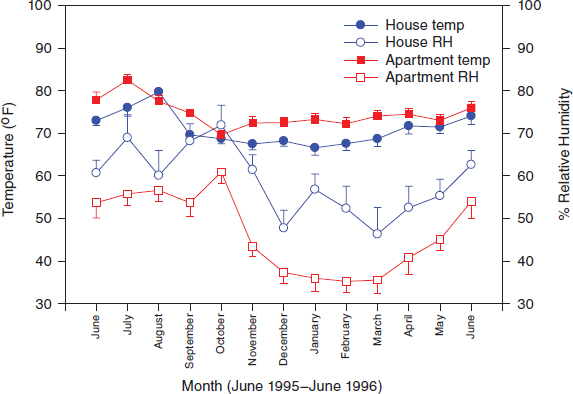
FIGURE 6-1 Variations in indoor temperature and relative humidity as functions of housing type and time of year in a sample of urban residences. (Derived from data presented in Chew et al., 1999.)
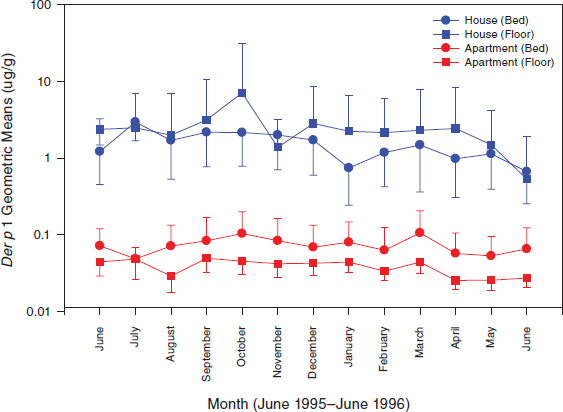
FIGURE 6-2 Variations in Der p 1 allergen levels as a function of housing type and location and time of year in a sample of urban residences. (Derived from data presented in Chew et al., 1999.)
increased use of air conditioning and dehumidifiers) could lead to a shift in the types of dust mites that are found in northern latitudes. The long-term outlook would be for increased numbers of dust mites and a wider variety of dust mite species in northern climates. However, that is speculative and does not consider adaptation measures.
Adaptation
More air-conditioning and dehumidifier use in summer In Ohio, Arilan et al. (2001) found that among three groups—those with only opening windows for cooling purposes, those with only air conditioning, and those with air conditioning and a high-efficiency dehumidifier—dust mite allergen concentrations in the homes with both air conditioning and dehumidifiers were less than 10% of concentrations in the other homes. Given that the monthly outdoor temperature in the Ohio summer months averaged 20.5–25.9°C (69–79°F), it is not clear how often the air conditioning was used in the air conditioning–only group. For that reason alone, the dehumidification (with only air conditioning) might not have been as much as could be expected in a scenario in which Ohio summers would be longer and warmer. Nonetheless, in a study that covered a wider geographic area of the United States, air conditioning was independently associated with lower dust mite allergen levels (Lintner and Brame, 1993). Furthermore, Swedish researchers found that mechanical exhaust and adequate supply ventilation in energy-efficient housing can decrease mite allergen concentrations substantially (Sundell et al., 1995). Air-conditioning use in the United States has steadily increased over the past several years, and it is likely that this adaptation measure will affect dust mite populations.
More humidifier use in summer Humidifier use is an adaptation strategy for those in arid environments. A humidifier would need to run quite often to keep the humidity high enough to sustain a dust mite population, and there is some evidence that they are in some circumstances. Prasad et al. (2009) showed that evaporative coolers can increase indoor relative humidity to a point where the prevalence of patients who have asthma or allergic rhinitis with sensitization to dust mites increases. The researchers posit that it is an increase in dust mite allergen in the environment that is tied with the increase in sensitization, but they collected no allergen measurements. Nonetheless, it remains an interesting research question that warrants further investigation.
Less heating in winter (because winters are milder) If winters become milder, some residents might use their heat less in the winter, and this could give rise to more dust mites and possibly a shift in the types of dust mites
that can thrive in a particular climate. For example, if the northeast United States becomes warmer and more humid, such mites as Blomia tropicalis—which is found in Puerto Rico—might extend their range northward in the United States (Acosta, 2008; Montealegre et al., 1997).
Epidemiology
Current dust mite sensitization patterns in the United States Most of what is known about current dust mite sensitization patterns in the United States is from the National Health and Nutrition Examination Survey (NHANES) population-based sample of the general population and from childhood-asthma studies (Table 6-1). The NHANES data did not focus on dust mite sensitization more widely than the regional level. However, the National Cooperative Inner-City Asthma Study (NCICAS), Childhood Asthma Management Program (CAMP), and Inner-City Asthma Study (ICAS) show that, among children who have asthma, dust mite sensitization varied by city.
How allergen avoidance could affect sensitization patterns Early-life environmental exposures are key in the development of allergic sensitization (Illi et al., 2006; Lau et al., 2000). It is controversial which factors are most important, such as early-life pet ownership and early-life endotoxin exposure (Holt and Thomas, 2005), but it is clear that those with a genetic predisposition to inhalant allergies tend to become sensitized to what is in their environment (Chew et al., 2008; Eldeirawi et al., 2005; Huss et al., 2001; Ingram et al., 1995; Montealegre et al., 2004; Phipatanakul et al., 2000a,b). The CAMP showed that of children who had asthma in Albuquerque, Baltimore, Boston, Denver, St. Louis, San Diego, Seattle, and Toronto, those in Denver and Albuquerque had the lowest concentrations of dust mites in their homes and the lowest prevalence of dust mite sensitization (33.1% and 21.5%, respectively) (Huss et al., 2001). If climate change leads to longer heat waves, areas with climates similar to that of Albuquerque could expand, and residents in those areas would experience not only decreased allergen exposure (which would lead to fewer symptoms) but changes in the pattern of their allergic sensitization.
How asthma incidence patterns could be affected Generally speaking—that is, without regard to the sensitizing agent(s)—asthma mortality and morbidity rates exhibit geographic, regional, and seasonal differences in the United States. Rates are elevated in urban areas when compared to rural areas (Carr et al., 1992; Grant et al., 1999; Weiss and Wagner, 1990), with New York City, Chicago, and Phoenix having consistently high mortality and hospitalization rates (Carr et al., 1992; Lang and Polansky, 1994;
TABLE 6-1 Prevalence of Dust Mite Sensitization
| Study | Type of Study | Population of Interest | Sample Size | Fraction Sensitized |
| NHANES II and III (Arbes et al., 2005) |
Population-based survey of general population | Children and adults (6–59 years old) | 17,738 | 4.2% (D. farinae) |
| NHANES III (Stevenson et al., 2001) |
Population-based survey of general population | Children (6–16 years old) | 4,164 | 25% (D. farinae) |
| CAMP (Huss et al., 2001) |
Multisite asthma study | Children (5–12 years old) | 1,041 | 48.5% (D. pteronyssinus |
| NCICAS (Kattan et al., 1997) |
Multisite asthma study | Children (4–9 years old) | 1,286 | or D. farinae) 24% (D. farinae), 31% (D. pteronyssinus) |
| ICAS (Gruchalla et al., 2005) |
Multisite asthma study | Children (5–11 years old) | 937 | 61.8% (D. pteronyssinus or D. farinae) |
| Tucson Children’s Respiratory Study (Stern et al., 2004) |
Birth cohort (Tucson) | Children (11 years old) | 626 | 14.9% (D. farinae) |
| Epidemiology of Home Allergens and Asthma Study (TePas et al., 2006) |
Birth cohort (Boston) | Children (7 years old) | 131 | 34% (D. pteronyssinus or D. farinae) |
NOTE: CAMP = Childhood Asthma Management Program, ICAS = Inner-City Asthma Study, NCICAS = National Cooperative Inner-City Asthma Study, NHANES =National Health and Nutrition Examination Survey.
Marder et al., 1992). Asthma mortality rates were consistently higher in the West and Midwest US Census regions than in the Northeast or South over the years 1980–1998 (Mannino, 2002). Akinbami (2006) notes that asthma prevalence rates among children 0–17 years of age are generally higher in the Northeast region than elsewhere but cautions:
While it is tempting to attribute prevalence patterns to climate or air quality, many factors affect prevalence and may also vary by region. Some examples include the likelihood that symptomatic children are diagnosed accurately with asthma and population composition. For example, the Puerto Rican population, in which asthma prevalence is highest, tends to be concentrated in the Northeast region of the country.
Exacerbations follow the seasonal patterns exhibited by asthma comorbidities, including rhinovirus and other respiratory viral infections, and triggers like pollen and mold (Johnston and Sears, 2006). Peak exacerbation occurs during the fall, although its magnitude varies depending on the age of the subject (younger asthmatics are more sensitive to seasonal changes).
Studies suggest that climate change will take place over a long period, and “allergen avoidance” might not be as extreme as that in some study interventions. Boner et al. (2002) found that dust mite–sensitized children who had asthma and were moved temporarily to the Italian Alps had reduced morbidity, and this was attributed to the absence of dust mites. Morgan et al. (2004) found that a targeted allergen-avoidance strategy for children reduced their asthma symptoms and emergency-department visits over a period of two years. What is more likely is that with the changing pattern of dust mite–endemic areas and the change in allergic-sensitization patterns, people who have allergic asthma will mount immune responses to elements of their environment, such as cockroaches, cats, and mice (Gruchalla et al., 2005). In contrast, if the northern states experience milder winters and an increase in humidity, dust mites might become the dominant allergen and surpass cockroaches and pets as the allergen most associated with increased asthma morbidity (Chew et al., 2009).
Other Pests
Little information is available on the potential effects of climate change on indoor exposure to other pests. Research has noted that the presence of increased mammalian pests in the indoor environment can spread disease and exacerbate allergies (IOM, 2008b), and increased outdoor temperatures are thought to have brought rodents indoors and led, for example, to disease from exposure to hanta virus in mouse droppings (Gubler et al., 2001). It is plausible that climate change will engender other indoor expo-
sures to pests, but research on the question is lacking. It should be noted, however, that data collected as part of the National Survey of Lead and Allergens in Housing (Cohn et al., 2004) and National Cooperative Inner-City Asthma Study (Phipatanakul et al., 2000a,b; Platts-Mills et al., 2007; Pongracic et al., 2008) do not indicate that there are appreciable geographic differences in mouse allergen levels measured indoors. It is thus unclear whether climate variations may have an effect on rodent infestations.
Pest Controls
Several factors may cause pesticide exposure to increase under conditions of climate change. Higher temperatures may lead to increased numbers of structural, agricultural, and forest insect pests (Boxall et al., 2009; Quarles, 2007). Water and storm damage may expand opportunities for pests to invade the indoor environment (Brennan, 2010). And damage caused by flooding and an increase in available water or moisture could create environments that are more hospitable to pests and increase the capacity of buildings to support infestations. All those circumstances are likely to lead to greater residential and agricultural use of pesticides to control increasing populations of insects, rodents, and other disease vectors and thus to a greater risk of exposures of populations.
Vulnerable Populations
Some communities appear to be at greater risk for indoor pesticide exposure than others. In the United States, urban communities are particularly at risk. In New York state, a study found that the heaviest application of pesticides occurred not in agricultural counties but in the New York City boroughs of Manhattan and Brooklyn (Thier et al., 1998). Some 93% of residents in public housing in New York City reported applying pesticides in their homes, and more than half said that they did so once a week (Surgan et al., 2002). Bradman et al. (2005) found that rodent infestation in homes increased in the presence of peeling paint, water damage, and high residential density, and the use of pesticides is common in communities that have adverse housing conditions.
Extensive work in the past decade has demonstrated the presence of pesticides in urban house dust and addressed the risk of exposure to these chemicals in vulnerable groups such as pregnant women and children. Such factors as multifamily dwellings, leaky buildings that allow pests to come into the indoor environment, and home ownership influence the likelihood of pesticide use (Julien et al., 2008). Although regular application of chemical pesticides, some of which include banned or restricted products, may be used to combat severe infestation, the methods are often deemed ineffective.
Once introduced into the home environment, pesticide residues may persist for years beyond the time of application, as demonstrated by evidence of banned compounds, such as DDT, in residential dust samples (Julien et al., 2008; Stout et al., 2009).
Integrated pest management (IPM) is a reasonable adaptation to increasing populations of pests, but many families lack the education or resources to implement the changes that will be needed. Residents of multifamily urban dwellings where pesticides are commonly used may have little control over the pesticides used in their buildings.
Changes in Vector Distribution
Changes in patterns of infestation in the outdoor environment may affect indoor air quality. Milder and shorter winters are expected to increase the geographic distribution of pests, such as mosquitoes and insects that attack agricultural crops (Quarles, 2007). Outbreaks of West Nile virus, carried by mosquitoes, in 2001–2005 correlated with increasing temperature and rainfall, and this leads to the expectation that such outbreaks will increase with climate change. Others have drawn attention to how outbreaks of disease, such as dengue fever and possibly malaria, could result from climate change (Girman, 2010; Hales et al., 2002; Randolph and Rogers, 2000).
The change in vector distribution and increasing threats to the agriculture industry are expected to result in increased use of agricultural chemicals (Boxall et al., 2009). As outdoor pesticide applications increase in an attempt to control increasing distribution of pests, indoor levels of these contaminants could rise. Pesticides applied in the outdoor environment do not remain outdoors but can find their way indoors through air exchange or can be brought in on clothing, skin, and especially shoes. People who live close to agricultural operations that increase their use of chemicals to control insect infestation may be at particularly high risk (Ward et al., 2006). In orchard-producing areas of Washington and Oregon, pesticide levels in house dust have been associated with distance from agricultural fields (Lu et al., 2000; McCauley et al., 2001), and pesticide metabolites in urine increased with proximity to the fields and during the pesticide-application season (Lu et al., 2000). Levels of pesticides metabolites have been higher in the urine of agricultural children than in the urine of children who reside in nonagricultural communities (Lambert et al., 2005; Lu et al., 2000).
In countries where malaria is endemic, the residential ban on applications of DDT is being lifted. In 2006, the World Health Organization and the US Agency for International Development endorsed indoor DDT spraying to control malaria (WHO, 2009). The increasing distribution of pests associated with climate change will result in increased measures to control
outbreaks and potentially increase the risk of exposure to pesticides and of associated health effects, particularly in vulnerable populations.
Health Concerns
Biologic monitoring in the United States indicates widespread exposure to organochlorine and organophosphate pesticides in the general population (National Exposure Research Library, 2005). Prospective cohort studies of mothers and newborns have documented considerable pesticide exposure during pregnancy in urban populations, with insecticides detected in air samples and in blood samples from women and newborns at delivery (Berkowitz et al., 2003; Whyatt et al., 2002, 2003). Those findings raised concern about potential health effects of residential exposure to pesticides. Many pesticides are developed to degrade quickly in outdoor environments but sequester in indoor environments in the absence of sunlight and rain. Pesticides can pass through the blood–brain barrier and penetrate the placenta. In addition, young children may receive greater exposure than adults because they eat, drink, and breathe more per unit of body weight (NRC, 1993). Children are also particularly vulnerable because they play in the dirt and on the floor (Fenske et al., 1990; Zwiener and Ginsburg, 1988).
Numerous animal studies have demonstrated that in utero or early exposure to organophosphate pesticides affects neurodevelopment (Eskenazi et al., 1999). Fetuses and young children may be more susceptible to neurotoxic effects of pesticides and have lower than adult levels of enzymes that are needed to detoxify organophosphate pesticides (Furlong et al., 2006). An emerging literature provides evidence of neurobehavioural consequences of relatively small exposure to organochlorine and organophosphate pesticides in infants and children (Eskenazi et al., 2008). Recent studies have found that the levels of organophosphate pesticides in dwellings may be great enough to cause neurodevelopmental effects (Eskenazi et al., 2007; Perera et al., 2003).
The increasing use of DDT to control malaria poses important questions about potential health effects. DDT is extremely persistent in the environment, so the potential association with indoor air quality is of concern. In a recent review of health effects associated with DDT exposure, a consensus group concluded that indoor residual spraying can result in substantial exposure to DDT and that DDT exposure may pose a risk to human populations (Eskenazi et al., 2009). There is a growing body of evidence that exposure to DDT and its breakdown product DDE may be associated with adverse health outcomes, such as breast cancer, diabetes, decreased semen quality, spontaneous abortion, and impaired neurodevelopment in children. However, few studies have measured body burdens of both DDE and DDT, and studies have rarely investigated the effects of DDT or DDE
exposure at levels observed in populations exposed through indoor residual spraying or populations exposed through drift of outdoor applications.
Rosas and Eskenazi (2008), in a review of the association between pesticides and neurodevelopment, concluded that although there are some inconsistencies among studies that may arise from differences in exposure and in methods of exposure assessment, there is surprising consistency in the few studies that have been conducted. The studies suggest that there is reason to be cautious about exposure of pregnant women to DDT, DDE, and organophosphates because of the potential effect on the neurodevelopment of their children.
Integrated Pest Management
If climate change causes the spread of diseases now considered to be tropical diseases into what are now more temperate climates, the use of pesticides could increase and have the potential to degrade indoor air quality. Alternatively, the concern about rodent and insect vectors could be used to promote wider use of Integrated Pest Management (IPM). IPM is an effective and environmentally sensitive approach to pest management that relies on a combination of common-sense practices. IPM programs use current comprehensive information on the life cycles of pests and their interactions with the environment. That information, in combination with available pest-control methods, is used to manage pest damage by the most economical means and with the least possible hazard to people, property, and the environment (EPA, 2010). IPM integrates common principles in the approach that should be taken to control pests in the environment. First is setting a threshold for action. For indoor environments, the potential of harming building integrity or human health is considered before any action. Families need to consider alternatives to chemical pesticides and avoid using the wrong kind of pesticide for a problem. Many intervention programs have been implemented to help families to recognize how to manage indoor space to prevent pests from posing a threat. The extent to which a family has the resources or control to modify its home environment will determine the success of its IPM interventions. If chemicals are needed, IPM assesses the proper control method for both effectiveness and risk minimization. Researchers are beginning to study the effectiveness of IPM interventions in high-risk populations. Williams et al. (2006) reported on an IPM intervention study of pregnant New York City black and Latina women; the study used education, sealing of pest entry points, and application of low-toxicity pesticides. They were able to show decreased cockroach infestation; lower indoor air concentrations of piperonyl butoxide, which is a synergist commonly added to pyrethroid insecticides that were applied; and lower levels of insecticides in maternal blood samples at delivery.
Synthesis
Generally speaking, alterations in outdoor environmental conditions may affect indoor exposures to pests by changing the habitable range of creatures known to invade indoor environments or by changing indoor environmental conditions or behavior in ways that drive them indoors. Buildings and building-maintenance practices that work well for one set of environmental conditions may not protect against infestations under other conditions. Termite infestations, for example, are less common in northern parts of the United States, and buildings and building codes there do not always require termite-prevention measures (Peterson, 2010). If termite ranges move northward, it may lead both to increased property damage and to occupant exposure to pesticides unless anticipatory maintenance and regulatory changes are made.
Several of the key findings of the 2001 National Research Council report Under the Weather: Climate, Ecosystems, and Infectious Diseases remain pertinent and bear repeating. They are excerpted and quoted below; additional explanatory detail is available in that report.
Key Findings Regarding Linkages Between Climate and Infectious Diseases from the Report Under the Weather: Climate, Ecosystems, and Infectious Diseases
- Weather fluctuations and seasonal-to-interannual climate variability influence many infectious diseases.
- Observational and modeling studies showing an association between climatic variations and disease incidence must be interpreted cautiously.
- Climate change may affect the evolution and emergence of infectious diseases.
- The relationships between climate and infectious disease are often highly dependent upon local-scale parameters and there are potential pitfalls in extrapolating climate and disease relationships from one spatial/temporal scale to another.
- The potential disease impacts of global climate change remain highly uncertain.
Research Needs and Surveillance Regarding Climate and Infectious Diseases from the Report Under the Weather: Climate, Ecosystems, and Infectious Diseases
- Research on the linkages between climate and infectious diseases must be strengthened.
- Further development of disease transmission models is needed to assess the risks posed by climatic and ecological changes.
- Epidemiological surveillance programs should be strengthened.
- Observational, experimental, and modeling activities are all highly interdependent and must progress in a coordinated fashion.
- Research on climate and infectious disease linkages inherently requires interdisciplinary collaboration.
Other Conclusions
In addition, on the basis of its review of the papers, reports, and other information presented in this chapter, the present committee has reached the following conclusions regarding infectious agents and pests:
- More investigation is needed to determine the extent to which the critical aspects of influenza spread are determined by indoor vs outdoor environmental conditions. It should consider air conditioning, which affects indoor temperature and humidity, and geographic location because there may be salient differences among regions in viral and human biology.
- The ecologic niches for house dust mites will change in response to climate change. Locations that are hotter and drier and that have increased use of air conditioning will tend to have fewer dust mite infestations. Decreased use of heating systems in winter because of milder conditions may result in increased dust mite populations.
- Decreases in dust mite populations in some locations may lower the incidence of allergic reactions to dust mites, but the overall incidence of allergic disease may not go down, because those who are predisposed to allergies may become sensitized to other air contaminants.
- Climate change may also lead to shifting patterns of indoor exposure to pesticides as occupants and building owners respond to infestations of pests whose ranges have changed.
Acosta LM, Acevedo-Garcia D, Perzanowski MS, Mellins R, Rosenfeld L, Cortes D, Gelman A, Fagan JK, Bracero LA, Correa JC, Reardon AM, Chew GL. 2008. The New York City Puerto Rican asthma project: Study design, methods, and baseline results. Journal of Asthma 45(1):51–57.
Akinbami LJ. 2006. The state of childhood asthma, United States, 1980–2005. Advance data from vital and health statistics No. 381. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics.
Antens CJ, Oldenwening M, Wolse A, Gehring U, Smit HA, Aalberse RC, Kerkhof M, Gerritsen J, de Jongste JC, Brunekreef B. 2006. Repeated measurements of mite and pet allergen levels in house dust over a time period of 8 years. Clinical and Experimental Allergy 36(12):1525-1531.
Arbes SJ Jr, Gergen PJ, Elliott L, Zeldin DC. 2005. Prevalences of positive skin test responses to 10 common allergens in the US population: Results from the third National Health and Nutrition Examination Survey. Journal of Allergy and Clinical Immunology 116(2):377-383.
Arlian LG. 1975. Dehydration and survival of the European house dust mite, Dermatophagoides pteronyssinus. Journal of Medical Entomology 12(4):437-442.
Arlian LG, Veselica MM. 1981. Re-evaluation of the humidity requirements of the house dust mite Dermatophagoides farinae (Acari: Pyroglyphidae). Journal of Medical Entomology 18:351-352.
Arlian LG, Neal JS, Morgan MS, Vyszenski-Moher DL, Rapp CM, Alexander AK. 2001. Reducing relative humidity is a practical way to control dust mites and their allergens in homes in temperature climates. Journal of Allergy and Clinical Immunology 107(1):99-104.
Ayres JG, Forsberg B, Annesi-Maesano I, Dey R, Ebi KL, Helms PJ, et al. 2009. Climate change and respiratory disease: European Respiratory Society position statement. The European Respiratory Journal 34(2):295-302
Berkowitz GS, Obel J, Deych E, Lapinski R, Godbold J, Liu Z, et al. 2003. Exposure to indoor pesticides during pregnancy in a multiethnic, urban cohort. Environmental Health Perspectives 111(1):79-84.
Boner A, Pescollderungg L, Silverman M. 2002. The role of house dust mite elimination in the management of childhood asthma: An unresolved issue. Allergy 57(Suppl 74):23-31.
Boone SA, Gerba CP. 2007. Significance of fomites in the spread of respiratory and enteric viral disease. Applied and Environmental Microbiology 73(6):1687-1696.
Boxall AB, Hardy A, Beulke S, Boucard T, Burgin L, Falloon PD, et al. 2009. Impacts of climate change on indirect human exposure to pathogens and chemicals from agriculture. Environmental Health Perspectives 117(4):508-514.
Bradman A, Chevrier J, Tager I, Lipsett M, Sedgwick J, Macher J, et al. 2005. Association of housing disrepair indicators with cockroach and rodent infestations in a cohort of pregnant Latina women and their children. Environmental Health Perspective 113(12):1795-1801.
Brennon T. 2010. Adaption and mitigation strategies for buildings in a changed climate. Presentation at the National Academy of Sciences Workshop on Climate Change and Indoor Environment: Washington, DC.
Brunekreef B, van Strien R, Pronk A, Oldenwening M, de Jongst JC, Wijga A, Kerhof M, Aalberse RC. 2005. La mano di DIOS … was the PIAMA intervention study intervened upon? Allergy 60(8):1083-1086.
Carbrera P, Julià-Serdà G, Rodrgíuez de Castro F, Caminero J, Barber D, Carrillo T. 1995. Reduction of house dust mite allergens after dehumidifier use. Journal of Allergy and Clinical Immunology 95(2):635-636.
Carr W, Zeitel L, Weiss K. 1992. Variations in asthma hospitalizations and deaths in New York City. American Journal of Public Health 82:59-65.
CDC (Centers for Disease Control and Prevention). 2010a. Respiratory syncytial virus activity—United States, July 2008—December 2009. MMWR 59(8):230-233.
CDC. 2010b. Updated CDC estimates of 2009 H1N influenza cases, hospitalization and deaths in the United States, April 2009–April 10, 2010. http://www.cdc.gov/h1n1flu/estimates_2009_h1n1.htm (accessed February 2, 2011).
Chan ED, Iseman MD. 2010. Slender, older women appear to be more susceptible to nontuberculous mycobacterial lung disease. Gender Medicine 7(1):5-18.
Chew GL, Higgins KM, Gold DR, Muilenberg ML, Burge HA. 1999. Monthly measurements of indoor allergens and the influence of housing type in a northeastern US city. Allergy 54(10):1058-1066.
Chew GL, Perzanowski MS, Canfield SM, Goldstein IF, Mellins RB, Hoepner LA, et al. 2008. Cockroach allergen levels and associations with cockroach-specific IgE. Journal of Allergy and Clinical Immunology 121(1):240-245.
Chew GL, Reardon AM, Correa JC, Young M, Acosta L, Mellins R, et al. 2009. Mite sensitization among Latina women in New York, where dust-mite allergen levels are typically low. Indoor Air 19(3):193-197.
Cohn RD, Arbes SJ Jr, Yin M, Jaramillo R, Zeldin DC. 2004. National prevalence and exposure risk for mouse allergen in US households. Journal of Allergy and Clinical Immunology 113(6):1167-1171.
de Boer R, Kuller K. 1997. Mattresses as a winter refuge for house-dust mite populations. Allergy 52(3):299-305.
De Jong B, Zucs P. 2010. Legionella, springtime and potting soils. Eurosurveillance 15(8):2-3. du Prel JB, Puppe W, Gröndahl B, Knuf M, Weigl JA, Schaaff F, Schmitt HJ. 2009. Are meteorological parameters associated with acute respiratory tract infections? Clinical Infectious Diseases 49(6):861-868.
Dulek DE, Peebles RS Jr. 2011. Viruses and asthma. Biochimica et Biophysica Acta doi:10.1016/j.bbagen.2011.01.012.
Eldeirawi K, McConnell R, Freels S, Persky VW. 2005. Associations of place of birth with asthma and wheezing in Mexican American children. Journal of Allergy and Clinical Immunology 116(1):42-48.
EPA (Environmental Protection Agency). 2010. Integrated pest management (IPM) strategies. http://www.epa.gov/pesticides/factsheets/ipm.htm (accessed February 3, 2011).
Eskenazi B, Bradman A, Castorina R. 1999. Exposures of children to organophosphate pesticides and their potential adverse health effects. Environmental Health Perspectives 107(Suppl 3):409-419.
Eskenazi B, Marks AR, Bradman A, Harley K, Barr DB, Johnson C. 2007. Organophosphate pesticide exposure and neurodevelopment in young Mexican-American children. Environmental Health Perspectives 115(5):792-798.
Eskenazi B, Rosas LG, Marks AR, Bradman A, Harley K, Holland N, et al. 2008. Pesticide toxicity and the developing brain. Basic & Clinical Pharmacology & Toxicology 102(2):228-236.
Eskenazi B, Chevrier J, Rosas LG, Anderson HA, Bornman MS, Bouwman H, et al. 2009. The Pine River statement: Human health consequences of DDT use. Environmental Health Perspectives 117(9):1359-1367.
Falkinham JO III, Iseman MD, de Haas P, van Soolingen D. 2008. Mycobacterium avium in a shower linked to pulmonary disease. Journal of Water Health 6(2):209-213.
Falkinham JO. 2010. Impact of human activities on the ecology of nontuberculous mycobacteria. Future Microbiology 5(6):951-960.
Feazel LM, Baumgartner LK, Peterson KL, Frank DN, Harris JK, Pace NR. 2009. Opportunistic pathogens enriched in showerhead biofilms. Proceedings of the National Academy of Sciences of the United States of America 106(38):16393-16399.
Fenske RA, Black KG, Elkner KP, Lee CL, Methner MM, Soto R. 1990. Potential exposure and health risks of infants following indoor residential pesticide applications. American Journal of Public Health 80(6):689-696.
Fisman DN, Lim S, Wellenius GA, Johnson C, Britz P, Gaskins M, Maher J, Mittleman MA, Spain CV, Haas CN, Newbern C. 2005. It’s not the heat, it’s the humidity: Wet weather increases legionellosis risk in the greater Philadelphia metropolitan area. Journal of Infectious Diseases 192(12):2066-2067.
Furlong CE, Holland N, Richter RJ, Bradman A, Ho A, Eskenazi B. 2006. PON1 status of farmworker mothers and children as a predictor of organophosphate sensitivity. Pharmacogenetics and Genetics 16(3):180-190.
Girman J. 2010. Research needed to address the impacts of climate change on indoor air quality. Washington DC: US Environmental Protection Agency.
Gehring U, Brunekreek B, Fahlbusch B, Wichmann HE, Heinrich J, INGA Study Group. 2005. Are house dust mite allergen levels influenced by cold winter weather? Allergy 60(8):1079-1082.
Grant EN, Wagener R, Weiss KB. 1999. Observations on emerging patterns of asthma in our society. Journal of Allergy and Clinical Immunology 104:S1-S9.
Gruchalla RS, Pongracic J, Plaut M, Evans R 3rd, Visness CM, Walter M, et al. 2005. Inner City Asthma Study: Relationships among sensitivity, allergen exposure, and asthma morbidity. Journal of Allergy and Clinical Immunology 115(3):478-485.
Gubler DJ, Reiter P, Ebi KL, Yap W, Nasci R, Patz JA. 2001. Climate variability and change in the United States: Potential impacts on vector- and rodent-borne diseases. Environmental Health Perspectives 109(Suppl 2):223-233.
Hales S, de Wet N, Maindonald J, Woodward A. 2002. Potential effect of population and climate changes on global distribution of dengue fever: An empirical model. Lancet 360(9336):830-834.
Harving H, Korsgaard J, Dahl R. 1994. Clinical efficacy of reduction in house-dust mite exposure in specially designed, mechanically ventilated “healthy” homes. Allergy 49(10): 866-870.
Holland SM, Vinh DC. 2009. Yeast infections–human genetics on the rise. New England Journal of Medicine 361(18):1798-1801.
Holt PG, Thomas WR. 2005. Sensitization to airborne environmental allergens: Unresolved issues. Nature Immunology 6(10):957-960.
Huss K, Adkinson NF Jr, Eggleston PA, Dawson C, Van Natta ML, Hamilton RG. 2001. House dust mite and cockroach exposure are strong risk factors for positive allergy skin test responses in the Childhood Asthma Management Program. Journal of Allergy and Clinical Immunology 107(1):48-54.
Ingram JM, Sporik R, Rose G, Honsinger R, Chapman MD, Platts-Mills TA. 1995. Quantitative assessment of exposure to dog (Can f 1) and cat (Fel d 1) allergens: Relation to sensitization and asthma among children living in Los Alamos, New Mexico. Journal of Allergy and Clinical Immunology 96(4):449-456.
Illi S, von Mutius E, Lau S, Niggemann B, Grüber C, Wahn U, et al. 2006. Perennial allergen sensitisation early in life and chronic asthma in children: A birth cohort study. Lancet 368(9537):763-770.
IOM (Institute of Medicine). 2000. Clearing the air—Asthma and indoor air exposures. Washington, DC: National Academy Press.
IOM. 2004. Damp indoor spaces and health. Washington, DC: The National Academies Press.
IOM. 2008a. Global climate change and extreme weather events: Understanding the contributions to infectious disease emergence: Workshop summary. Washington, DC: The National Academies Press.
IOM. 2008b. Vector-borne diseases: Understanding the environmental, human health, and ecological connections: Workshop summary. Washington, DC: The National Academies Press.
Johnston N, Sears M. 2006. Asthma exacerbations. 1: Epidemiology. Thorax 61:722-728.
Julien R, Levy JI, Adamkiewicz G, Hauser R, Spengler JD, Canales RA, et al. 2008. Pesticides in urban multiunit dwellings: Hazard identification using classification and regression tree (CART) analysis. Journal of the Air and Waste Management Association 58(10): 1297-1302.
Kattan M, Mitchell H, Eggleston P, Gergen P, Crain E, Redline S, et al. 1997. Characteristics of inner-city children with asthma: The National Cooperative Inner-City Asthma Study. Pediatric Pulmonology 24(4):253-262.
Lambert WE, Lasarev M, Muniz J, Scherer J, Rothlein J, Santana J, McCauley L. 2005. Variation in organophosphate pesticide metabolites in urine of children living in agricultural communities. Environmental Health Perspectives 113(4):504-508.
Lang DM, Polansky M. 1994. Patterns of asthma mortality in Philadelphia from 1969 to 1991. New England Journal of Medicine 331:1542-1546.
Lapeña S, Robles MB, Castañón L, Martínez JP, Reguero S, Alonso MP, Fernández I. 2005. Climatic factors and lower respiratory tract infection due to respiratory syncytial virus in hospitalised infants in northern Spain. European Journal of Epidemiology 20(3):271-276.
Lau S, Illi S, Sommerfeld C, Niggemann B, Bergmann R, von Mutius E, Wahn U. 2000. Early exposure to house-dust mite cat allergens and development of childhood asthma: A cohort study. Multicentre Allergy Study Group. Lancet 356(9239):1392-1397.
Lintner TJ, Brame KA. 1993. The effects of season, climate, and air-conditioning on the prevalence of Dermatophagoides mite allergens in household dust. Journal of Allergy and Clinical Immunology 91(4):862-867.
Lu C, Fenske RA, Simcox NJ, Kalman D. 2000. Pesticide exposure of children in an agricultural community: Evidence of household proximity to farmland and take home exposure pathways. Environmental Health Research 84(3):290-302.
Mannino DM, Homa DM, Akinbami LJ, Moorman JE, Gwynn C, Redd SC. 2002. Surveillance for asthma—United States, 1980–1999. Morbidity and Mortality Weekly Report 51(SS01):1-13.
Marder D, Targonski P, Orris P, Persky V, Addington W. 1992. Effect of racial and socioeconomic factors on asthma mortality in Chicago. Chest 101:4265-4295.
McCauley LA, Lasarey MR, Higgins G, Rothlein J, Muniz J, Ebbert C, Phillips J. 2001. Work characteristics and pesticide exposures among migrant agricultural families: A community-based research approach. Environmental Health Perspectives 109(5):533-538.
McDonald LC, Banerjee SN, Jarvis WR. 1999. Seasonal variation of Acinetobacter infections: 1987-1996. Nosocomial Infections Surveillance System. Clinical Infectious Diseases 29(5):1133-1137.
Meerhoff TJ, Paget JW, Kimpen JL, Schellevis F. 2009. Variation of respiratory syncytial virus and the relation with meteorological factors in different winter seasons. The Pediatric Infectious Diseases Journal 28(10):860-866.
Montealegre F, Sepulveda A, Bayona M, Quiñones C, Fernádez-Caldas E. 1997. Identification of the domestic mite fauna of Puerto Rico. Puerto Rico Health Sciences Journal 16(2):109-116.
Montealegre F, Fernández B, Delgado A, Fernández L, Román A, Chardón D, Rodríguez-Santana J, Medina V, Zavala D, Bayona M. 2004. Exposure levels of asthmatic children to allergens, endotoxins, and serine proteases in a tropical environment. The Journal of Asthma 41(4):485-496.
Morey PR. 2010. Climate change and potential effects on microbial air quality in the built environment. http://www.epa.gov/iaq/pdfs/climate_and_microbial_iaq.pdf (accessed February 3, 2011).
Morgan WJ, Crain EF, Gruchalla RS, O’Connor GT, Kattan M, Evans R 3rd, et al. 2004. Results of a home-based environmental intervention among urban children with asthma. New England Journal of Medicine 351(11):1068-1080.
Murdoch DR, Jennings LC. 2009. Association of respiratory virus activity and environmental factors with the incidence of invasive pneumococcal disease. The Journal of Infection 58(1):37-46.
Myatt TA, Johnston SL, Zuo Z, Wand M, Kebadze T, Rudnick S, Milton DK. 2004. Detection of airborne rhinovirus and its relation to outdoor air supply in office environments. American Journal of Respiratory & Critical Care Medicine 169(11):1187-1189.
Myatt TA, Kaufman MH, Allen JG, MacIntosh DL, Fabian MP, McDevitt JJ. 2010. Modeling the airborne survival of influenza virus in a residential setting: The impacts of home humidification. Environmental Health 9:55.
National Exposure Research Laboratory. 2005. Compendium of NERL-sponsored children’s exposure data and tools for assessing aggregate exposure to residential-use pesticides in support of the August 2006 reassessment. FY2005 Annual Performance Measure 33. Research Triangle Park, North Carolina.
Naumova EN. 2006. Mystery of seasonality: Getting the rhythm of nature. Journal of Public Health Policy 27(1):2-12.
NRC (National Research Council). 1993. Pesticides in the diets of infants and children. Washington, DC: National Academy Press.
NRC. 2001. Under the weather: Climate, ecosystems, and infectious diseases. Washington, DC: National Academy Press.
Omer SB, Sutanto A, Sarwo H, Linehan M, Djelantik IG, Mercer D, Moniaga V, Moulton LH, Widjaya A, Muljati P, Gessner BD, Steinhoff MC. 2008. Climatic, temporal, and geographic characteristics of respiratory syncytial virus disease in a tropical island population. Epidemiology and Infection 136(10):1319-1327.
Patterson K, Strek ME. 2010. Allergic bronchopulmonary aspergillosis. The Proceedings of the American Thoracic Society 7(3):237-244.
Perencevich EN, McGregor JC, Shardell M, Furuno JP, Harris AD, Morris JG Jr, Fisman DN, Johnson JA. 2008. Summer peaks in the incidences of gram-negative bacterial infection among hospitalized patients. Infection Control and Hospital Epidemiology 29(12):1124-1131.
Perera FP, Rauh V, Tsai WY, Kinney P, Camann D, Barr D, et al. 2003. Effects of transplacental exposure to environmental pollutants on birth outcomes in a multiethnic population. Environmental Health Perspectives 111(2):201-205.
Peterson CJ. 2010. Termites and climate change: Here, there and everywhere? EARTH Magazine (January 2010):46-53.
Phipatanakul W, Eggleston PA, Wright EC, Wood RA. 2000a. Mouse allergen. I. The prevalence of mouse allergen in inner-city homes. The National Cooperative Inner-City Asthma Study. Journal of Allergy and Clinical Immunology 106(6):1070-1074.
Phipatanakul W, Eggleston PA, Wright EC, Wood RA; National Cooperative Inner-City Asthma Study. 2000b. Mouse allergen. II. The relationship of mouse allergen exposure to mouse sensitization and asthma morbidity in inner-city children with asthma. Journal of Allergy and Clinical Immunology 106(6):1075-1080.
Platts-Mills TA, Satinover SM, Naccara L, Litonjua AA, Phipatanakul W, Carter MC, Heymann PW, Woodfolk JA, Peters EJ, Gold DR. 2007. Prevalence and titer of IgE antibodies to mouse allergens. Journal of Allergy and Clinical Immunology 120(5):1058-1064.
Pongracic JA, Visness CM, Gruchalla RS, Evans R, Mitchell HE. 2008. Effect of mouse allergen and rodent environmental intervention on asthma in inner-city children. Annals of Allergy, Asthma and Immunology 101(1):35-41.
Prasad C, Hogan MB, Peele K, Wilson NW. 2009. Effect of evaporative coolers on skin test reactivity to dust mites and molds in a desert environment. Allergy and Asthma Proceedings 30(6):624-627.
Quarles W. 2007. Global warming means more pests. The IPM Practitioner XXIX(9/10):1-8.
Randolph SE, Rogers DJ. 2000. Fragile transmission cycles of tick-borne encephalitis virus may be disrupted by predicted climate change. Proceedings. Biology Sciences/The Royal Society 267(1454):1741-1444.
Rodman DM, Polis JM, Heltshe SL, Sontag MK, Chacon C, Rodman RV, Brayshaw SJ, Huitt GA, Iseman MD, Saavedra MT, Taussig LM, Wagener JS, Accurso FJ, Nick JA. 2005. Late diagnosis defines a unique population of long-term survivors of cystic fibrosis. American Journal of Respiratory and Critical Care Medicine 171(6):621-626.
Rosas LG, Eskenazi B. 2008. Pesticides and child neurodevelopment. Current Opinions in Pediatrics 20(2):191-197.
Schubert MS. 2009. Allergic fungal sinusitis: Pathophysiology, diagnosis and management. Medical Mycology 47(Suppl 1):S324-S330.
Shaman J, Kohn M. 2009. Absolute humidity modulates influenza survival, transmission, and seasonality. Proceedings of the National Academy of Sciences of the United States of America 106(9):3243-3248.
Shaman J, Goldstein E, Lipsitch M. 2010a. Absolute humidity and pandemic versus epidemic influenza. American Journal of Epidemiology 173(2):127-135.
Shaman J, Pitzer VE, Viboud C, Grenfell BT, Lipsitch M. 2010b. Absolute humidity and the seasonal onset of influenza in the continental United States. PLoS Biology 8(2):e100031.
Singleton RJ, Bulkow LR, Miernyk K, DeByle C, Pruitt L, Hummel KB, Bruden D, Englund JA, Anderson LJ, Lucher L, Holman RC, Hennessy TW. 2010. Viral respiratory infections in hospitalized and community control children in Alaska. Journal of Medical Virology 82(7):1282-1290.
Sloan C, Moore ML, Hartert T. 2011. Impact of pollution, climate, and sociodemographic factors on spatiotemporal dynamics of seasonal respiratory viruses. Clinical and Translational Science 4(1):48-54.
Stapleton F, Keay LJ, Sanfilippo PG, Katiyar S, Edwards KP, Naduvilath T. 2007. Relationship between climate, disease severity, and causative organism for contact lens-associated microbial keratitis in Australia. American Journal of Ophthalmology 144(5):690-698.
Stern DA, Lohman IC, Wright AL, Taussig LM, Martinez FD, Halonen M. 2004. Dynamic changes in sensitization to specific aeroallergens in children raised in a desert environment. Clinical and Experimental Allergy 34(10):1563-1669.
Stevenson LA, Gergen PJ, Hoover DR, Rosenstreich D, Mannino DM, Matte TD. 2001. Sociodemographic correlates of indoor allergen sensitivity among United States children. Journal of Allergy and Clinical Immunology 108(5):747-752.
Stout JE, Yu VL. 1997. Legionellosis. New England Journal of Medicine 337(10):682-687.
Stout DM 2nd, Bradham KD, Egeghy PP, Jones PA, Croghan CW, Ashley PA, et al. 2009 American Healthy Homes Survey: A national study of residential pesticides measured from floor wipes. Environmental Science and Technology 43(12):4294-4300.
Sundell J, Wickman M, Pershagen G, Nordvall SL. 1995. Ventilation in homes infested by house-dust mites. Allergy 50(2):106-112.
Surgan MH, Congdon T, Primi C, Lamster S, Louis-Jacques J. 2002. Pest control in public housing, schools and parks: Urban children at risk. LAW 180-4 PESP 202-7643. Albany, NY: New York State Department of Law, Environmental Protection Bureau.
Talbot TR, Pehling KA, Hartert TV, Arbogast PG, Halasa NB, Edwards KM, et al. 2005. Seasonality of invasive pneumococcal disease: Temporal relation to documented influenza and respiratory syncytial viral cirvulation. American Journal of Medicine 118(3):285-291.
Tang JW, Lai FY, Nymadawa P, Deng YM, Ratnamohan M, Petric M, Loh TP, Tee NW, Dwyer DE, Barr IG, Wong FY. 2010a. Comparison of the incidence of influenza in relation to climate factors during 2000-2007 in five countries. Journal of Medical Virology 82(11):1958-1965.
Tang JW, Lai FY, Wong F, Hon KL. 2010b. Incidence of common respiratory viral infections related to climate factors in hospitalized children in Hong Kong. Epidemiology and Infection 138(2):226-235.
Tepas EC, Litonjua AA, Celedón JC, Sredl D, Gold DR. 2006. Sensitization to aeroallergens and airway hyperresponsiveness at 7 years of age. Chest 129(6):1500-1508.
Thier A, Enck J, Klossner C. 1998. Plagued by pesticides: An analysis of New York State’s 1997 pesticide use and sales data. Albany, NY: Environmental advocates.
Tovey ER, Rawlinson WD. 2011. A modern miasma hypothesis and back-to-school asthma exacerbations. Medical Hypotheses 76(1):113-116.
Tsiodras S, Samonis G, Boumpas DT, Kontoyiannis DP. 2008. Fungal infections complicating tumor necrosis factor alpha blockade therapy. Mayo Clinic Proceedings 83(2):181-194.
Van Strien RT, Verhoeff AP, Brunekreef B, Van Wijnen JH. 1994. Mite antigen in house dust: Relationship with different housing characteristics in The Netherlands. Clinical and Experimental Allergy 24(9):843-853.
Voorhorst R, Spieksma FThM, Varekamp N. 1969. House dust mite atopy and the house dust mite Dermatophagoides pteronyssinus (Troussart, 1897). Leiden: Stafleu’s Scientific Publishing Co.
Ward MH, Lubin J, Giglierano J, Colt JS, Wolter C, Bekiroglu N, et al. 2006. Proximity to crops and residential exposure to agricultural herbicides in Iowa. Environmental Health Perspectives 114(6):893-897.
Watson M, Gilmour R, Menzies R, Ferson M, McIntyre P; New South Wales Pneumococcal Network. 2006. The association of respiratory viruses, temperature, and other climatic parameters with the incidence of invasive pneumococcal disease in Sydney, Australia. Clinical Infectious Diseases 42(2):211-215.
Weiss KB, Wagner DK. 1990. Changing patterns in US asthma mortality: Identifying populations at high risk. Journal of the American Medical Association 264:1683-1687.
Welliver R. 2009. The relationship of meteorological conditions to the epidemic activity of respiratory syncytial virus. Pediatric Respiratory Reviews 10(Suppl 1):6-8.
Williams MK, Barr DB, Camann DE, Cruz LA, Carlton EJ, Borjas M, Reyes A, Evans D, Kinney PL, Whitehead RD, Jr., Perera FP, Matsoanne S, Whyatt RM. 2006 An intervention to reduce residential insecticide exposure during pregnancy among an inner-city cohort. Environmental Health Perspectives 114(11):1684-1689.
Wood JP, Choi YW, Chappie DJ, Rogers JV, Kaye JZ. 2010. Environmental persistence of a highly pathogenic avian influenza (H5N1) virus. Environmental Science & Technology 44(19):7515-7520.
WHO (World Health Organization). 2007. WHO guidelines for indoor air quality: Dampness and mould. Denmark: WHO Regional Office for Europe.
WHO. 2009. WHO and DDT for malaria control—June 2009: WHO position statement. Geneva: WHO Press.
Whyatt RM, Camann DE, Kinney PL, Reyes A, Ramirez J, Dietrich J, et al. 2002. Residential pesticide use during pregnancy among a cohort of urban minority women. Environmental Health Perspectives 110(5):507-514.
Whyatt RM, Barr DB, Camann DE, Kinney PL, Barr JR, Andrews HF, et al. 2003. Contemporary-use pesticides in personal air samples during pregnancy and blood samples at delivery among urban minority mothers and newborns. Environmental Health Perspectives 111(5):749-756.
Yamamoto Y, Nakamura K, Yamada M, Mase M. 2010. Persistence of avian influenza virus (H5N1) in feathers detached from bodies of infected domestic ducks. Applied and Environmental Microbiology 76(16):5496-5499.
Zoumot Z, Wilson R. 2010. Respiratory infection in noncystic fibrosis bronchiectasis. Current Opinion in Infectious Diseases 23(2):165-170.
Zwiener RJ, Ginsburg CM. 1988. Organophosphate and carbamate poisoning in infants and children. Pediatrics 81(1):121-126.






























