1
Introduction
Health effects from exposure to formaldehyde have been a topic of research for decades. Past concerns arose from exposures in indoor environments and studies of workers showing increased risk of nasopharyngeal cancer. In recent years, people who were displaced by Hurricane Katrina and Hurricane Rita and lived in trailers provided by the Federal Emergency Management Agency have reported adverse health effects attributed to formaldehyde exposure. Published research has also indicated a possible link between leukemia and formaldehyde exposure.
The U.S. Environmental Protection Agency (EPA) has been working to update its health assessment of formaldehyde for its Integrated Risk Information System (IRIS) for a number of years. The large amount of new research and data on formaldehyde since its original assessment in the early 1990s has made the task challenging. Given the complex nature of the assessment and the knowledge that the assessment will be used as the basis of regulatory decisions, EPA asked the National Research Council (NRC) to conduct an independent scientific review of the draft IRIS assessment and answer questions related specifically to its derivation of reference concentrations (RfCs) for noncancer effects and of its unit risk estimates for cancer. In response to EPA’s request, NRC convened the Committee to Review EPA’s Draft IRIS Assessment of Formaldehyde, which prepared this report.
FORMALDEHYDE AND THE DRAFT ASSESSMENT
Formaldehyde, which has the chemical structure shown in Figure 1-1, is a chemical building block of numerous compounds that are used in a wide array of products (see Gerberich and Seaman 1994; ATSDR 1999; IARC 2006). One main use is to make resins that are used as adhesives in the production of particle board, fiberboard, plywood, and other wood products. The resins are also used to make molding and insulating materials and are used in a variety of other industries, including the textile, rubber, and cement industries.
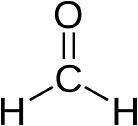
FIGURE 1-1 Formaldehyde chemical structure. Formaldehyde is described as a colorless gas at room temperature with a pungent, suffocating odor.
Formaldehyde is a common environmental chemical that is found in ambient and indoor air. It is also present naturally in some foods and is a metabolic intermediate in the human body. For ambient air, major emission sources include power plants, incinerators, refineries, manufacturing facilities, and automobiles (ATSDR 1999; IARC 2006). Formaldehyde is also produced by vegetative decay, animal wastes, forest fires, and photochemical oxidation of hydrocarbons in the lower atmosphere (ATSDR 1999; IARC 2006). The most recent EPA data on ambient-air concentrations indicate that the annual means at monitoring sites range from 0.56 to 36.31 ppb, and the overall mean is 2.77 ppb (EPA 2010). If the data are categorized by land use, agricultural locations have the lowest mean, 1.68 ppb, and locations affected primarily by mobile sources have the highest mean, 5.52 ppb.
Indoor air typically has higher formaldehyde concentrations than ambient air (ATSDR 1999; IARC 2006; EPA 2010). Major indoor emission sources include building materials, consumer products, gas and wood stoves, kerosene heaters, and cigarettes. Indoor-air concentrations depend on the age and type of construction. Older conventional homes have lower formaldehyde concentrations than newer constructions, and conventional homes have lower formaldehyde concentrations than mobile homes. Formaldehyde concentrations in indoor air have been decreasing since the 1980s, when restrictions on formaldehyde emissions from building materials were tightened (ATSDR 1999; EPA 2010; Salthammer et al. 2010). However, on the basis of a review of international studies, Salthammer et al. (2010) estimated the average formaldehyde exposure of the general population to be 16-32 ppb in air. Figure 1-2 provides ranges of formaldehyde air concentrations in various environments.
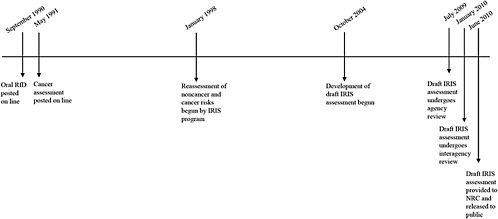
Given the pervasive exposure of the general population to some concentration of formaldehyde, federal agencies tasked with protecting public health are concerned about the health effects of formaldehyde exposure. EPA is re-evaluating regulations on the emissions of formaldehyde from composite wood products and, as part of that effort, is re-evaluating its assessment of noncancer and cancer risks associated with formaldehyde. Figure 1-3 provides a timeline of EPA’s activity since its original assessments of noncancer and cancer risks were released in 1990 and 1991, respectively.
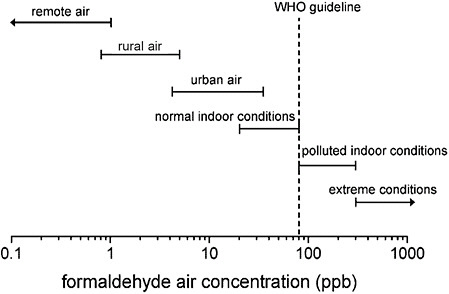
FIGURE 1-2 Formaldehyde concentration in various environments. Abbreviation: WHO, World Health Organization. Source: Salthammer et al. 2010. Reprinted with permission; copyright 2010, American Chemical Society.
Since 1991, numerous studies of the toxicity and carcinogenic potential of formaldehyde have been published. In 2006, the International Agency on Cancer Research (IARC) revised its formaldehyde classification from probably carcinogenic to humans (Group 2A) to carcinogenic to humans (Group 1). The revision was based on what IARC concluded to be sufficient evidence of nasopharyngeal cancer in humans, strong but not sufficient evidence of leukemia in humans, and limited evidence of sinonasal cancer in humans (IARC 2006). In 2009, IARC reaffirmed its classification of formaldehyde but concluded that there was sufficient evidence of leukemia in humans (Baan et al. 2009). Furthermore, in 2010, an expert National Toxicology Program (NTP) panel on formaldehyde recommended that formaldehyde be listed as a known human carcinogen in its Report on Carcinogens (McMartin et al. 2009). That recommendation was a change from the previous edition, which listed formaldehyde as “reasonably anticipated to be a human carcinogen” (NTP 2005). Some scientists do not agree with the recent conclusions from IARC and NTP and have published new studies that they claim cast doubt on them.
Given the complex nature of assessing the health effects of formaldehyde and the knowledge that the IRIS assessment will be used as a basis of new regulations, EPA asked NRC to convene a committee to review its draft IRIS assessment.
THE COMMITTEE’S TASK AND APPROACH
The committee convened as a result of EPA’s request included experts in epidemiology, exposure assessment, leukemogenesis, mechanisms of carcinogenicity, inhalation toxicology, neurotoxicology, reproductive and developmental toxicology, statistics, physiologically based pharmacokinetic modeling, and risk assessment (see Appendix A for biographic information on the committee). The committee was asked to review EPA’s draft IRIS assessment and to answer questions concerning the identification of potential noncancer health effects, the selection of the points of departure for those health effects, and the basis of the determination of uncertainty factors used to derive the RfCs. The committee was also asked specifically to comment on the scientific rationale provided for the cancer assessment and the quantified estimates derived. The verbatim statement of task is provided in Box 1-1.
|
BOX 1-1 Statement of Task A committee of the National Research Council (NRC) will conduct an independent scientific review of the U.S. Environmental Protection Agency (EPA) draft human health assessment of formaldehyde for the Integrated Risk Information System (IRIS). The committee will provide a brief report that comments on EPA's identification of potential adverse noncancer health effects, assessment of carcinogenic potential, exposure-response analysis for identified end points, quantitative risk assessment methods, and evaluation of sources of uncertainty in the health assessment. Specifically, the committee will address tasks such as the following: Inhalation Reference Concentration for Formaldehyde
|
Carcinogenicity of Formaldehyde
|
To accomplish its task, the committee held four meetings from June 2010 to December 2010. The first two meetings included public sessions during which the committee heard primarily from the sponsor on the development of the draft IRIS assessment and approaches used to derive the estimates presented in it. During each public session, interested parties addressed the committee. The committee reviewed the draft assessment, numerous scientific publications, and all materials submitted to it by outside parties.
The committee was tasked with conducting an “independent scientific review” of the draft IRIS assessment, not with conducting its own assessment. Therefore, the committee did not conduct its own literature search, review all relevant evidence, systematically formulate its own conclusions regarding causality, or recommend values for the RfC and unit risk. The committee reviewed
the draft IRIS assessment and its methods and key literature and determined whether EPA’s conclusions were supported on the basis of that assessment and literature. Thus, the present report contains the committee’s conclusions and recommendations resulting from its review of the draft assessment. The committee notes that it does not provide a comprehensive discussion of any particular topic or health outcome, although it does provide brief descriptions where necessary to give the reader some context as to what it is recommending. Furthermore, the committee discussed the various health outcomes using the categories presented in the draft IRIS assessment. Some overlap among the categories was noted; for example, asthma—a disease with an immunologic basis—was handled separately from immunologic effects.
Because the committee evaluated what EPA did, there is some inherent variability in the depth of the committee’s review given the varied discussions in the draft IRIS assessment. For example, the draft assessment presents discussions on mode of action that vary in level of detail, analysis, and referencing. In some cases, mode-of-action data—which would support EPA’s conclusion—are available, but they are not presented in the draft assessment. In those cases, the committee recommends that those data be reviewed and evaluated. In other cases, mode of action is highly speculative, and the speculations are discussed at length in the draft assessment. In those cases, the committee recommends that the discussion be truncated given the speculative nature of the hypotheses. The committee notes that a well-established mode of action is not required to make causal inferences, but mode-of-action data should be discussed when those data support EPA’s conclusions.
ORGANIZATION OF REPORT
The committee organized its report by separating the overarching elements of its charge from the more specific ones. Specifically, Chapter 2 addresses the general methods to develop the draft IRIS assessment because the committee has concerns about the methods used in its development. Chapter 3 reviews the toxicokinetics of formaldehyde, which has general relevance for effects at the portal of entry and elsewhere, and therefore this review precedes the other chapters. The remaining chapters were structured to address the specific elements of the charge related to the RfCs and unit risk. Accordingly, Chapters 4 and 5 discuss the weight of evidence for hazard identification and study selection for portal-of-entry and systemic effects, respectively, and Chapter 6 addresses the derivation of the RfC and unit risk. Chapter 7 provides general recommendations for revisions of the draft assessment and, on the basis of the findings in Chapters 2-6, comments on the IRIS process used to generate the present assessment.
REFERENCES
ATSDR (Agency for Toxic Substances and Disease Registry). 1999. Toxicological Profile for Formaldehyde. U.S. Department of Health and Human Services, Public Health Services, Agency for Toxic Substances and Disease Registry, Atlanta, GA. July 1999 [online]. Available: http://www.atsdr.cdc.gov/ToxProfiles/tp111.pdf [accessed Jan. 5, 2011].
Baan, R., Y. Grosse, K. Straif, B. Secretan, F. El Ghissassi, V. Bouvard, L. Benbrahim-Tallaa, N. Guha, C. Freeman, L. Galichet, and V. Cogliano. 2009. A review of human carcinogens—Part F: Chemical agents and related occupations. Lancet Oncol. 10(12):1143-1144.
EPA (U.S. Environmental Protection Agency). 2010. Toxicological Review of Formaldehyde (CAS No. 50-00-0) – Inhalation Assessment: In Support of Summary Information on the Integrated Risk Information System (IRIS). External Review Draft. EPA/635/R-10/002A. U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://cfpub.epa.gov/ncea/iris_drafts/recordisplay.cfm?deid=223614 [accessed Nov. 22, 2010].
Gerberich, H.R., and G.C. Seaman. 1994. Formaldehyde. Pp. 929-951 in Kirk-Othmer Encyclopedia of Chemical Technology, Vol. 11, 4th Ed., J.I. Kroschwitz, and M. Howe-Grant, eds. New York: Wiley.
IARC (International Agency for Research on Cancer). 2006. Formaldehyde. Pp. 39-325 in Formaldehyde, 2-Butoxyethanol and 1-tert-Butoxypropan-2-ol. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Vol. 88. Lyon, France: International Agency for Research on Cancer.
McMartin, K.E., F. Akbar-Khanzadeh, G.A. Boorman, A. DeRoos, P. Demers, L. Peterson, S. Rappaport, D.B. Richardson, W.T. Sanderson, M.S. Sandy, L.B. Freeman, M. DeVito, S.A. Elmore, and L. Zhang. 2009. Part B—Recommendations for the Listing Status for Formaldehyde and Scientific Justification for the Recommendation. Formaldehyde Expert Panel Report Part B [online]. Available: http://ntp.niehs.nih.gov/ntp/roc/twelfth/2009/November/FA_PartB.pdf [accessed Jan. 5, 2011].
NTP (National Toxicology Program). 2005. Formaldehyde (Gas): CAS No. 50-00-0. Substance Profiles. Report on Carcinogens, 11 Ed. U.S. Department of health and Human Services, Public Health Service, National Toxicology Program [online]. Available: http://ntp.niehs.nih.gov/ntp/roc/eleventh/profiles/s089form.pdf [accessed Jan. 5, 2011].
Salthammer, T., S. Mentese, and R. Marutzky. 2010. Formaldehyde in the indoor environment. Chem. Rev. 110(4):2536-2572.