Reference Guide on Exposure Science
Joseph V. Rodricks, Ph.D., is Principal at Environ, Arlington, Virginia.
CONTENTS
A. What Do Exposure Scientists Do?
B. Who Qualifies as an Expert in Exposure Assessment?
C. Organization of the Reference Guide
III. Contexts for the Application of Exposure Science
B. Environmental and Product Contaminants
C. Chemicals in Workplace Environments
D. Claims of Disease Causation
A. Organic and Inorganic Chemicals
V. Human Exposures to Chemicals
A. Exposure Sources—An Overview
B. The Goal of Exposure Assessment
E. Summary of the Descriptive Process
VI. Quantification of Exposure
B. Doses from Indirect Exposure Pathways
C. Direct Measurement: Analytical Science
E. Integrated Exposure/Dose Assessment
B. Monitoring the Body (Biomonitoring)
VIII. Evaluating the Scientific Quality of an Exposure Assessment
The sciences of epidemiology1 and toxicology2 are devoted to understanding the hazardous properties (the toxicity) of chemical substances. Moreover, epidemiological and toxicological studies provide information on how the seriousness and rate of occurrence of the hazard in a population (its risk) change as exposure to a particular chemical changes. To evaluate whether individuals or populations exposed to a chemical are at risk of harm,3 or have actually been harmed, the information that arises from epidemiological and toxicological studies is needed, as is the information on the exposures incurred by those individuals or populations.
Epidemiologists and toxicologists can tell us, for example, how the magnitude of risk of benzene-induced leukemia changes as exposure to benzene changes. Thus, if there is a need to understand the magnitude of the leukemia risk in populations residing near a petroleum refinery, it becomes necessary to understand the magnitude of the exposure of those populations to benzene. Likewise, if an individual with leukemia claims that benzene exposure was the cause, it becomes necessary to evaluate the history of that individual’s exposure to benzene.4
Understanding exposure is essential to understanding whether the toxic properties of chemicals have been or will be expressed. Thus, claims of toxic tort or product liability generally require expert testimony not only in medicine and in the sciences of epidemiology and toxicology, but also testimony concerning the nature and magnitude of the exposures incurred by those alleging harm. Similarly, litigation involving the regulation of chemicals said to pose excessive risks to health also requires litigants to present evidence regarding exposure. The need to understand exposure is a central topic in the reference guides in this publication on epidemiology and toxicology. This reference guide provides a view of how the magnitude of exposure comes to be understood.5
1. See Michael D. Green et al., Reference Guide on Epidemiology, in this manual.
2. See Bernard D. Goldstein & Mary Sue Henifin, Reference Guide on Toxicology, in this manual.
3. See, e.g., Rhodes v. E.I. du Pont de Nemours & Co., 253 F.R.D. 365 (S.D. W. Va. 2008) (suit for medical monitoring costs because exposure to perfluoroctanoic acid in drinking water allegedly caused an increased risk of developing certain diseases in the future); In re Welding Fume Prods. Liab. Litig., 245 F.R.D. 279 (N.D. Ohio 2007) (exposure to manganese fumes allegedly increased the risk of later developing brain damage).
4. See, e.g., Lambert v. B.P. Products North America, Inc., 2006 WL 924988 (S.D. Ill. 2006), 2006 U.S. Dist. LEXIS 16756 (plaintiff diagnosed with chronic lymphocytic leukemia was exposed to jet fuel allegedly containing excessive levels of benzene).
5. This chapter focuses on measuring exposure to toxic substances as a specific developing area of scientific investigation. This topic is distinct from the legal concept of “exposure,” which is an element of a claim in toxic tort litigation. The legal concept of exposure relies on the evolving scientific understanding of the manner and extent to which individuals come into contact with toxic substances. However, the legal concept also reflects substantive legal principles and interpretations that vary across jurisdictions. Compare Parker v. Mobil Oil Corp., 793 N.Y.S.2d 434 (2005) (requiring findings of specific levels of exposure to benzene by plaintiff who claimed that his leukemia was the result of his
Not all questions concerning human exposures to potentially harmful substances require expert testimony. In those circumstances in which the magnitude of exposure is not relevant, or is clearly evident (e.g., because a plaintiff was observed to take the prescribed amount of a prescription medicine), expert testimony is not indicated. But if the magnitude of exposure is an important component of the needed evidence, and if that magnitude is not a simple question of fact, then expert testimony will be important.
Exposure science is not yet a distinct academic discipline. Although some schools of public health may offer courses in exposure assessment, there are no academic degrees offered in exposure science. When regulatory and public health agencies began in the 1970s to examine toxicological risks in a quantitative way, it became apparent that quantitative exposure assessments would become necessary. Initially, exposure assessment was typically practiced by toxicologists and epidemiologists. As the breadth and complexity of the subject began to be recognized, it became apparent that scientists and engineers with a better grasp of the properties of chemicals (which affect how they behave and undergo change in different environments), and of the methods available to identify and measure chemicals in products and in the environment, would be necessary to provide scientifically defensible assessments. As the importance of exposure assessment grew and began to present significant scientific challenges, its practice drew increasing numbers of scientists and engineers, and some began to refer to their work as exposure science. Not surprisingly, most of the early expositions of exposure assessment came from government agencies that recognized the need to develop and refine the practice to meet their risk assessment needs. Indeed, various documents and reports used by the U.S. Environmental Protection Agency (EPA) remain essential sources for the practice of exposure assessment.6 Academics and practitioners have written chapters on exposure science for major multiauthor reference works
17-year occupational exposure to gasoline containing benzene) with Westberry v. Gislaved Gummi AB, 178 F.3d 257 (4th Cir. 1999) (evidence of specific exposure level not required where evidence of talc in the workplace indicated that the worker was covered in talc and left footprints on the floor) and Allen v. Martin Surfacing, 263 F.R.D. 47 (D. Mass. 2009) (admissible expert testimony may be based on symptom accounts by those exposed rather than direct measurements of solvent concentrations). This chapter takes no position regarding exposure as a substantive legal concept.
6. U.S. Environmental Protection Agency, Exposure Assessment Tools and Models (2009), available at http://www.epa.gov/oppt/exposure/ (last visited June 6, 2011); National Exposure Research Laboratory, U.S. Environmental Protection Agency, Scientific and Ethical Approaches for Observational Exposure Studies, Doc. No. EPA 600/R-08/062 (2008), available at http://www.epa.gov/nerl/sots/index.html (last visited July 14, 2010); U.S. Environmental Protection Agency. Exposure Factors Handbook (1997).
on toxicology,7 but most of the work in this area is still found in the primary reference works.
Although exposure science is not yet a distinct academic discipline, in this reference guide the phrase is retained and used to refer to the work of scientists and engineers (“exposure scientists”) working in one or more aspects of exposure assessment.
A. What Do Exposure Scientists Do?
Human beings are exposed to natural and industrial chemicals from conception to death, and because almost all chemicals can become harmful if exposures exceed certain levels, understanding the magnitude and duration of exposures to chemicals is critical to understanding their health impacts. Exposure science is the study of how people can come into contact with (are exposed to)8 chemicals that may be present in various environmental media (air, water, food, soil, consumer products of all types) and of the amounts of those chemicals that enter the body as a result of these contacts.9 Exposure scientists also study whether and how those amounts change over time. The goal of exposure science is to quantify those amounts and time periods. The quantitative expression of those amounts is referred to as dose. Ultimately the dose incurred by populations or individuals is the measure needed by health experts to quantify risk of toxicity. Exposure science does not typically deal with the health consequences of those exposures.
The dose entering the body (through inhalation or ingestion, through the skin, and through other routes) is often referred to as the “exposure dose,” to distinguish it from the dose that enters the bloodstream and reaches various organs of the body. The latter is typically only a fraction of the exposure dose and is identified through studies that can trace the fate of a chemical after it enters the body. The term “dose” as used in this reference guide is synonymous with “exposure dose,” and doses reaching blood or various organs within the body are referred to as “target site doses” or “systemic doses,”
Exposure assessments can be directed at past, present, or even future exposures and can be narrowly focused (one chemical, one environmental medium, one population group) or very broad in scope (many chemicals, several environ-
7. P.J. Lioy, Exposure Analysis and Its Assessment, in Comprehensive Toxicology (I.G. Sipes et al. eds., 1997); D.J. Paustenbach & A. Madl, The Practice of Exposure Assessment, in Principles and Methods of Toxicology (Wallace Hayes ed., 5th ed. 2008).
8. See, e.g., Kitzmiller v. Jefferson, 2006 WL 2473399, 2006 U.S. Dist. LEXIS 61109 (N.D. W. Va. 2006) (defendants offered expert’s testimony that plaintiff’s use of liquid cleaning agents containing benzalkonium chloride failed to show that she was exposed to benzalkonium chloride in the air); Hawkins v. Nicholson, 2006 WL 954654, 2006 U.S. App. Vet. Claims LEXIS 197, 21 Vet. App. 64 (Vet. App. 2006) (noting that “a veteran who served on active duty in Vietnam between January 9, 1962, and May 7, 1975, is entitled to a rebuttable presumption of exposure to Agent Orange”).
9. The term “enter the body” also includes entering the external surface of the body.
mental media, several different population groups). This reference guide explores the various contexts in which exposure assessments are conducted and how their scope is determined.
B. Who Qualifies as an Expert in Exposure Assessment?
As noted, it is unlikely that any expert can present evidence of having an academic degree in exposure science. An expert’s qualifications thus have to be tested by examining the expert’s experience,10 including his or her knowledge of and reliance on authoritative reference works.11 Experts generally will have strong academic credentials in environmental science and engineering, chemistry, chemical engineering, statistics and mathematical model building, industrial hygiene, or other hard sciences related to the behavior of chemicals in the environment.
To the extent exposure assessments deal with the amounts and behaviors of chemicals in the body, individuals can qualify as experts if they can offer academic credentials or substantial experience in toxicology and in the measurement of chemicals in blood or in biological tissues. Certainly, toxicology, epidemiology, or medical credentials are needed if experts are to offer testimony on the health consequences associated with particular exposures.
Not all exposure assessments are complex; indeed, some, as will be seen, are relatively simple. Most toxicologists and epidemiologists have considerable training and experience assessing dose from medicines and other consumer products—and even from food. But if exposures result from chemicals moving from sources through one or more environmental media, it is unlikely that toxicologists or epidemiologists will be able to offer appropriate qualifications, because modeling or other forms of indirect measurement are needed to assess exposures. Further details on the qualifications of experts are offered in the closing sections of the reference guide.
C. Organization of the Reference Guide
The reference guide begins with a discussion of the various contexts in which exposure science is applied (Section III). Following that discussion is a section on chemicals and their various sources. Three broad categories of chemicals are discussed: (1) those that are produced for specific uses; (2) those that are byproducts of chemical production, use, and disposal and that enter the environment as contaminants; and (3) those that are created and released by the combustion of all types of organic substances (including tobacco) and of fuels used for energy
10. See, e.g., Best v. Lowe’s Home Ctrs, 2009 WL 3488367, 2009 U.S. Dist. LEXIS 97700 (E.D. Tenn. 2009) (a medical doctor with extensive industrial toxicology and product safety experience opined that the plaintiff could not have been exposed to the chemical at issue as alleged).
11. Most of the EPA’s guidance documents on exposure assessment have been issued after extensive peer review and thus are considered authoritative.
production. Each of these categories can be thought of as a source for chemical exposure. Next, there is a discussion of the pathways chemicals follow from their sources to the environmental media to which humans are or could be in contact. Such contact is said to create an exposure. Chemicals can then move from these media of human contact and enter the body by different routes of exposure—by ingestion (in food or water, for example), by inhalation, or by direct skin contact (the dermal route). The section on exposure routes includes a discussion of how chemicals contact and enter the body and of how they behave within it. This last topic comprises the interface between exposure science and the sciences of epidemiology and toxicology. Traditionally, exposure scientists have described their work as ending with the description of dose to the body (exposure dose). As will be seen, some practitioners are focusing on the amounts of chemicals present in blood or various tissues of the body as a result of exposure. Unlike the toxicologist, the exposure scientist is not qualified to evaluate the health consequences of these so-called biomarkers of exposure.
This reference guide first presents all of the above material in nonquantitative terms—to describe and illustrate the various processes through which human exposures to chemicals are created (Sections III–V). The guide then focuses on the quantitative aspects (Sections VI and VII). Without some quantitative understanding of the magnitude of exposure, and of the duration of time over which exposure occurs, it becomes difficult to reach meaningful conclusions about health risks. Thus, the remaining sections are devoted to a critical quantitative concept in exposure science—that of dose—and are intended to integrate all of the earlier descriptive material. The reference guide ends with a review of the qualifications of exposure science experts and how they can be assessed.
III. Contexts for the Application of Exposure Science
There are perhaps four major contexts in which exposure science is applied: (1) consumer products, (2) contaminants in the environment and in consumer products, (3) chemicals in the workplace, and (4) disease causation.
Many intentional uses of chemical substances lead to human exposures, and the health risks that are associated with those exposures need to be understood.12 In some cases, laws and regulations require that health risks be understood in
12. See, e.g., In re Stand ’n Seal, 623 F. Supp. 2d 1355 (N.D. Ga. 2009) (consumer use of spray-on product allegedly resulted in inhalation exposure to toxic substances, causing respiratory injuries).
advance of the marketing of such chemicals or products containing them. Thus, intentionally introduced food additives, pesticides, and certain industrial chemicals must have regulatory approvals before they are marketed, and manufacturers of such substances are required to demonstrate the absence of significant health risks (i.e., their safety) based on toxicology studies and careful assessments of expected exposures. Pharmaceuticals and other medical products must undergo similar premarket evaluations. The safety and efficacy of such products must be demonstrated through clinical studies (which are undertaken after animal toxicology studies have been done and have demonstrated the safety of such products for individuals who are involved in clinical trials). Human exposure assessments are central to the regulatory approval of these products.13
Many other consumer products require risk assessments, but premarket approvals are not generally required under our current laws. The list of such products is very long, and not all substances included in these products have been subjected to exposure and risk assessments, but regulatory initiatives in the United States and abroad are creating new requirements for more complete assessments of consumer safety.
B. Environmental and Product Contaminants
Byproducts of many industrial processes, including those created by combustion, have led to much environmental contamination (see Section IV for a discussion of the sources of such contamination).14 Technically speaking, contamination refers to the presence of chemical substances in environmental media (including consumer products) in which such substances would not ordinarily be found. The term also may be used to refer to their presence in greater amounts than is usual.15 The assessment of health risks from such contaminants depends upon an understanding of the magnitude and duration of exposure to them. Exposures may occur through the presence of contaminants in air, drinking water, foods, consumer products, or soils and dusts; in many cases, exposures may occur simultaneously through more than one of these media.
The results from exposure and risk assessments (which incorporate information regarding the toxic properties of the contaminants) are typically used by regulators and public health officials to determine whether exposed populations are at significant risk of harm. If regulators decide that the risks are excessive, they
13. B.D. Beck et al., The Use of Toxicology in the Regulatory Process, in Principles and Methods of Toxicology (A. Wallace Hayes ed., 5th ed. 2008).
14. See, e.g., Orchard View Farms, Inc. v. Martin Marietta Aluminum, Inc., 500 F. Supp. 984, 1008 (D. Or. 1980) (failure to monitor fluoride emissions that harmed nearby orchards supported award of punitive damages).
15. For example, lead is naturally present in soils. It could be said that a sample of soil is contaminated with lead only if it were clear that the amounts present exceeded natural levels. The issue is complicated by the fact that natural levels are highly variable.
will take steps to reduce them, typically by using interventions that will reduce exposures (because the inherent toxic properties of the chemicals involved cannot be altered). Exposure scientists are called upon to assess the magnitude of exposure reduction (and therefore risk reduction) achieved through a given intervention.16
C. Chemicals in Workplace Environments
Workers in almost all industrial sectors are exposed to chemicals.17 Exposures are created in industries involved in the extraction of the many raw materials used to manufacture chemical products (the mining, agricultural,18 and petroleum industries). Raw materials are refined and otherwise processed in thousands of different ways and are eventually turned into manufactured chemical products that number in the tens of thousands. These products enter many channels of distribution and are incorporated into many other products (so-called downstream uses). Occupational exposures can occur at all of these various steps of manufacturing and use. Exposure also can occur from disposal of wastes. Exposure assessments in all of these various occupational settings are important to understand whether health risks are excessive and therefore require reduction.19
D. Claims of Disease Causation
In the above three situations, the exposures of interest are those that are currently occurring or that are likely to occur in the future. In those situations the exposure assessments are used to ascertain whether risks of harm are excessive (and thus require reduction) or to document safety (when risks are negligible). There are, however, many circumstances in which individuals claim they actually have been harmed by chemicals. Specifically, they allege that some existing medical condition has been caused by exposures occurring in the past, whether in the workplace, the environment, or through the use of various consumer products.20
16. National Research Council, Air Quality Management in the United States (2004).
17. See, e.g., Kennecott Greens Creek Min. Co. v. Mine Safety & Health Admin., 476 F.3d 946 (D.C. Cir. 2007) (suit over regulations addressing miners’ exposure to diesel particulate matter).
18. The term “agriculture” is applied here very broadly and includes the production of a wide variety of raw materials that have industrial and consumer product uses (including flavors, fragrances, fibers of many types, and some medicinal products). See, e.g., Association of Irritated Residents v. Fred Schakel Dairy, 634 F. Supp. 2d 1081, 1083 (E.D. Cal. 2008) (methanol emissions from dairy allegedly resulted in exposure sufficient to create human health risks).
19. Office of Pesticide Programs, U.S. Environmental Protection Agency, General Principles for Performing Aggregate Exposure and Risk Assessments, available at http://www.epa.gov/pesticides/trac/science/aggregate.pdf (last visited July 14, 2010).
20. See Michael D. Green et al., supra note 1, in this manual, for a discussion on disease causation. Regulations and public health actions are usually driven by findings of excessive risk of harm (although sometimes evidence of actual harm).
Exposure science comes into play in these cases because the likelihood that any given disease or injury was induced because of exposure to one or more chemicals depends in large part on the size of that exposure.21 Thus, with the advent of large numbers of so-called toxic tort claims has come the need to assess past exposures. Exposure scientists have responded to this need by adapting the methods of exposure assessment to reconstruct the past—that is, to produce a profile of individuals’ past exposures.22
A plaintiff with a medical condition known from epidemiological studies to be caused by a specific chemical may not be able to substantiate his or her claim without evidence of exposure to that chemical of a sufficient magnitude.23 Exposure experts are needed to quantify the exposures incurred; causation experts are then called upon to offer testimony on whether those exposures are of a magnitude sufficient to cause the plaintiff’s condition. Chemicals known to cause diseases under certain exposure conditions will not do so under all exposure conditions.
Exposure reconstruction has a history of use by epidemiologists who are studying disease rates in populations that may be associated with past exposures.24 Epidemiologists have paved the way for the use of exposure assessment methods to reconstruct the past. Although the methods for evaluating current and past exposures are essentially identical, the data needed to quantify past exposures are often more limited and yield less certain results than the data needed to evaluate current exposures. Assessment of past exposures is especially difficult when considering diseases with very long latency periods.25 By the time disease occurs, documentary proof of exposure and magnitude may have disappeared. But courts regularly deal with evidence reconstructing the past, and assessment of toxic exposure is another application of this common practice.26
21. See supra notes 1 & 2. Causation may sometimes be established even if quantification of the exposure is not possible. See, e.g., Best v. Lowe’s Home Ctrs, Inc., 563 F.3d 171 (6th Cir. 2009) (doctor permitted to testify as to causation based on differential diagnosis).
22. Confounding factors must be carefully addressed. See, e.g., Allgood v. General Motors Corp., 2006 WL 2669337, at *11 (S.D. Ind. 2006) (selection bias rendered expert testimony inadmissible); American Farm Bureau Fed’n v. EPA, 559 F.3d 512 (2009) (in setting particulate matter standards addressing visibility, the data relied on should avoid the confounding effects of humidity); Avila v. Willits Envtl. Remediation Trust, 2009 WL 1813125, 2009 U.S. Dist. LEXIS 67981 (N.D. Cal. 2009) (failure to rule out confounding factors of other sources of exposure or other causes of disease rendered expert’s opinion inadmissible); Adams v. Cooper Indus. Inc., 2007 WL 2219212, 2007 U.S. Dist. LEXIS 55131 (E.D. Ky. 2007) (differential diagnosis includes ruling out confounding causes of plaintiffs’ disease).
23. See Michael D. Green et al., Reference Guide on Epidemiology, in this manual.
24. Id.
25. W.T. Sanderson et al., Estimating Historical Exposures of Workers in a Beryllium Manufacturing Plant, 39 Am. J. Indus. Med. 145–57 (2001).
26. Courts have accepted indirect evidence of exposure. For example, differential diagnosis may support an expert’s opinion that the exposure caused the harm. Best v. Lowe’s Home Ctrs., Inc., 563 F.3d 171 (6th Cir. 2009). On occasion, qualitative evidence of exposure is admitted as evidence
Before embarking on a description of the elements of exposure science, it is useful to provide a brief primer on some of the characteristics of chemicals that influence their behavior and that therefore affect the ways in which humans can be exposed to them. The primer also introduces some technical terms that frequently arise in exposure science.
A. Organic and Inorganic Chemicals
For both historical and scientific reasons, chemists divide the universe of chemicals into organic and inorganic compounds. The original basis for classifying chemicals as organic was the hypothesis, known since the mid-nineteenth century to be false, that organic chemicals could be produced only by living organisms. Modern scientists classify chemicals as organic if they contain the element carbon.27 Carbon has the remarkable and nearly unique property that its atoms can combine with each other in many different ways, and, together with a few other elements—including hydrogen, oxygen, nitrogen, sulfur, chlorine, bromine—can create a huge number of different molecular arrangements. Each such arrangement is a unique chemical. Several million distinct organic chemicals are already known to chemists, and there are many more that will no doubt be found to occur naturally or that will be created by laboratory synthesis. All of life—at least on Earth—depends on carbon compounds and probably could not have evolved if carbon did not have its unique and extraordinary bonding properties.
All other chemicals are called inorganic. There are 90 elements in addition to carbon in nature (and several more that have been created in laboratories), and because these elements do not have the special properties of carbon, the number of different possible combinations of them is smaller than can occur with carbon.
Living organisms contain or produce organic chemicals by the millions. One of the most abundant organic chemicals on Earth is cellulose—a giant molecule containing thousands of atoms of carbon, hydrogen, and oxygen. Cellulose is produced by all plants and is their essential structural component. Chemically, cellulose
that the magnitude was great enough to cause harm. See, e.g., Westberry v. Gislaved Gummi AB, 178 F.3d 257 (4th Cir. 1999) (no quantitative measurement required where evidence showed plaintiff was covered in talc and left footprints); Allen v. Martin Surfacing, 263 F.R.D. 47 (D. Mass. 2009) (symptom accounts at the time of exposure formed the basis for expert’s opinion that exposure was high enough to cause harm). And courts have accepted the government’s reconstruction of exposure to radiation. Hayward v. U.S. Dep’t of Labor, 536 F.3d 376 (5th Cir. 2008); Hannis v. Shinseki, 2009 WL 3157546 (Vet. App. 2009) (no direct measure of veteran’s exposure to radiation was possible but VA’s dose estimate was not clearly erroneous).
27. There are a few compounds of carbon that chemists still consider inorganic: These are typically simple molecules such as carbon monoxide (CO) and carbon dioxide (CO 2) and the mineral limestone, which is calcium carbonate (CaCO3).
is a carbohydrate (one that is not digested by humans), a group that together with proteins, fats, and nucleic acids are the primary components of life. But living organisms also produce huge numbers of other types of organic molecules. The colors of plants and animals and their odors and tastes are a result of the presence of organic chemicals. The numbers and structural varieties of naturally occurring chemicals are enormous.
Other important natural sources of organic chemicals are the so-called fossil fuels—natural gas, petroleum, and coal—all deposited in the Earth from the decay of plant and animal remains and containing thousands of degradation products. Most of these are simple compounds containing only carbon and hydrogen (technically known as hydrocarbons). The organic chemical industry depends upon these and just a few other natural products for everything it manufactures; the fraction of fossil fuels not used directly for energy generation is used as feedstock for the chemical industry. There are also inorganic chemicals—the minerals—present in living organisms, many essential to life. But the principal natural source of inorganic chemicals is the nonliving part of the Earth that humans have learned how to mine.
The modern chemical industry had its origins in the late nineteenth century when chemists, mostly European, discovered that it was possible to create in the laboratory chemicals that had previously been found only in nature. Most remarkably, scientists also discovered they could synthesize compounds not found in nature—substances never previously present on Earth. In other words, they found ways to alter through chemical reactions the bonds present in one compound so that a new compound was formed. The first compound synthesized in this way was a dye called aniline purple by the British chemist, William Henry Perkin, who discovered it.28 The work of chemical synthesis grew out of the development of so-called structural theory in the nineteenth century and remains central to the science today. This theory explains that the number and type of chemical elements present, and the ways in which those elements are bonded to each other, are unique for each chemical compound and therefore distinguish one chemical from another.
In the late nineteenth century and up to World War II, coal was the major starting material for the organic chemical industry. When coal is heated in the absence of oxygen, coke and volatile byproducts called coal tars are created. All sorts of organic chemicals can be isolated from coal tar—benzene, toluene, xylenes, ethylbenzene, naphthalene, creosotes, and many others. The organic
28. This compound and others related to it became the bases for the first chemical industry, that devoted to dye production. Perkins’ dye was later called “mauve” and its wide use led to what came to be called the Mauve Decade (1890s).
chemical industry also uses other natural products, such as animal fats, vegetable oils, and wood byproducts.
The move to petroleum as a raw materials source for the organic chemical industry began during the 1940s. Petrochemicals, as they are called, are now used to create thousands of useful industrial chemicals. The rate of commercial introduction of new chemicals shot up rapidly after World War II.
Among the thousands of products produced by the organic chemical industry and by related industries are medicines (most of which are organic chemicals of considerable complexity), dyes, agricultural chemicals, including substances used to eliminate pests (insecticides, fungicides, herbicides, rodenticides, and other “cides”), soaps and detergents, synthetic fibers and rubbers, paper chemicals, plastics and resins of great variety, adhesives, food additives, additives for drinking water, refrigerants, explosives, cleaning and polishing materials, cosmetics, and textile chemicals. Because of past disposal practices, chemicals primarily used as solvents (for many purposes) are among the most widespread environmental contaminants.
The history of human efforts to tap the inorganic earth for useful materials is complex and involves a blend of chemical, mining, and materials technologies. Included here is everything from the various silicaceous materials derived from stone (glasses, ceramics, clays, asbestos) to the vast number of metals derived from ores that have been mined and processed (iron, copper, nickel, cadmium, molybdenum, mercury, lead, silver, gold, platinum, tin, aluminum, uranium, cobalt, chromium, germanium, iridium, cerium, palladium, manganese, zinc, and many more). Other nonmetallic materials, such as chlorine and bromine, salt (sodium chloride), limestone (calcium carbonate), sulfuric acid, and phosphates, and various compounds of the metals, have hundreds of different uses, as strictly industrial chemicals and as consumer products. These inorganic substances reach, enter, and move about our environment, and we come into contact with them, sometimes intentionally, sometimes inadvertently. The number of organic and inorganic chemicals in commercial production exceeds 70,000, and the number of uses and products created from them far exceeds this number.
There are important health questions related to what is generally referred to as particulate matter (PM). Small particulates in the air usually arise from combustion of almost any organic material. The chemical composition of such particulates can vary depending upon source, but it is possible that their health effects depend more upon their physical size than their chemical composition. This issue is currently unresolved, but it is important to include PMs of all types as a class of chemical contaminants.
Finally, it is important to note that, in addition to PM, many chemicals are produced when fuels or other organic materials are burned. Organic chemicals take on oxygen atoms during combustion and yield large numbers of substances not present in the materials that are burned. Combustion also produces simple inorganic oxides of carbon, nitrogen, and sulfur, which are major air pollutants.
Burning tobacco introduces 4000 to 5000 chemicals into the lungs. Combustion products are another important source of environmental contamination.29
V. Human Exposures to Chemicals
As noted earlier, this section is entirely descriptive, rather than quantitative. It describes all the various physical processes that lead to human exposures to chemicals and introduces the terms that exposure scientists apply to those processes. Section VI illustrates how these various processes can be quantified and the types of data that are required to do so.
A. Exposure Sources—An Overview
Figure 1 provides a broad overview of most of the major sources of exposure. As shown, sources can be intended or unintended. Thus, many chemicals are intentionally used in ways that will lead to human exposures. Substances added to food and indeed food itself,30 cosmetics, personal care products, fibers and the colorants added to them, and medical products of many types are included in this broad category. Direct ingestion of, or other types of direct contact with (on the skin or through inhalation), such products obviously creates exposures. Nicotine and tobacco combustion products might also be classified as intended exposures. Generally, these exposures are more readily quantifiable than those associated with unintended exposures.
Although the term is somewhat ambiguous, unintended exposures may be said to fall into two broad categories. There are deliberate uses of certain chemicals that, although not intended to lead to human exposures, will inevitably do so. Pesticides applied to food crops, some components of food packaging materials that may migrate into food, and many types of household products are not intended for direct human ingestion or contact, but exposures will nonetheless occur indirectly. Occupational exposures, although unintended, are similarly unavoidable. Also, many exposures to a very broad range of environmental contaminants are unintended (see Figure 1).
In all of these cases, such exposures are not described as intentional, in the sense that the term is applied to a pharmaceutical ingredient or a cosmetic, but most are not completely avoidable. Unintended exposures are generally more
29. National Research Council, Human Exposure Assessment for Airborne Pollutants: Advances and Opportunities (1991); J. Samet & S. Wang, Environmental Tobacco Smoke, in Environmental Toxicants (M. Lippmann ed., 2d ed. 2000).
30. The natural constituents of food include not only substances that have nutritional value, but also hundreds of thousands of other natural chemicals.
Figure 1. Opportunities for exposure: Sources of chemical releases.
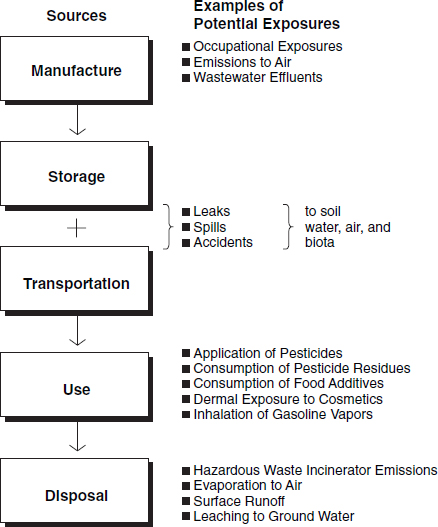
difficult to identify and quantify than are intended exposures.31 In the case of the intended exposures, the pathway from source to humans is direct; in the case of unintended exposures, the pathway is indirect, sometimes highly so. Thus, the most important distinction for purposes of exposure assessment concerns the directness of the pathway from source to people.
31. There are significant differences in the laws regarding the regulation of substances that have been grouped as creating intended or unintended exposures.
B. The Goal of Exposure Assessment
Exposure assessment is generally intended to answer the following questions:
- Who has been, or could become exposed to a specific chemical(s) arising from one or more specific sources? Is it the entire general population, or is it a specific subpopulation (e.g., those residing near a certain manufacturing or hazardous waste facility, or infants and children), or is it workers?32
- What specific chemicals comprise the exposures?
- What are the pathways from the source of the chemical to the exposed population? Pathways include direct product use, or those (so-called indirect pathways) in which the chemical moves through one or more environmental media to reach the media to which people are exposed (air, water, foods, soils, and dusts). Understanding pathways is necessary to understanding exposure routes (below) and quantifying exposures.
- By what routes are people exposed? Routes include ingestion, inhalation, and dermal contact.33 Identifying exposure routes is important because those routes affect the magnitude of ultimate exposures and because they often affect health outcomes.
- What is the magnitude and duration of exposure incurred by the population of interest? Dose is the technical term used for magnitude, and it is the amount of chemical entering the body or contacting the surface of the body, usually over some specified period of time (often over 1 day34). Duration refers to the number of days over which exposure occurs. Note that exposures can be intermittent or continuous and can be highly variable, especially for some air contaminants.
The ultimate goal of exposure assessment is to identify dose and duration. The concept of dose is further developed in Section VI. After a chemical enters or contacts the body, it can be absorbed (into the bloodstream), distributed to many organs of the body, metabolized (chemically altered by certain enzymes in cells of the liver and other organs), and then excreted. Understanding these processes is important to determining whether and how a chemical may cause adverse health effects. These processes mark the interface between exposure science and toxicology, epidemiology, and medicine. Understanding the dose is the necessary first step in understanding these processes; for purposes of this reference guide, the boundary of exposure science is set at understanding dose. However, some
32. See, e.g., Hackensack Riverkeeper, Inc. v. Del. Ostego, 450 F. Supp. 2d 467 (D.N.J. 2006) (river and bay users alleged that hazardous waste runoff and emissions polluted the water).
33. Additional routes of exposure are relevant for some pharmaceuticals, diagnostics, and medical devices.
34. Shorter periods of time are used when the concern is very short-term exposures to chemicals that have extremely high toxicity—so-called acutely poisonous materials.
discussion of how it is possible to gain more direct measures of exposure (target site doses) by examining human blood and urine is included.
The completion of an exposure assessment provides the information needed (the dose and duration of exposure) by epidemiologists and toxicologists, who will have information on the adverse health effects of the chemicals involved and on the relationships between those effects and the dose and duration of exposure.35 Recall that exposure assessments can be directed at exposures that occurred in the past, those that are currently occurring, or those that will occur in the future should certain actions be taken (e.g., the entry of a new product into the consumer market or the installation of new air pollution controls).
The discussion of each of these elements of exposure assessment is expanded in the following section, beginning with pathways.
Assuming that the chemical of interest and its sources have been identified, exposure assessment focuses on the pathway the chemical follows to reach the population of interest.36
To ensure thoroughness in the assessment, all conceivable pathways should be explicitly identified, with the understanding that ultimately some pathways will be found to contribute negligibly to the overall exposure. Identifying pathways is also important to understanding exposure routes.
As noted earlier, the simplest pathways are those described as direct. Thus, a substance, such as a noncaloric sweetener or an emulsifier, once added to food, follows a simple and direct pathway to the people who ingest the food. The same can be said for pharmaceuticals, cosmetics, and other personal care products.
35. See reference guides on epidemiology and toxicology in this manual. See also, e.g., White v. Dow Chem. Co., 321 Fed. App’x. 266, 2009 WL 931703 (4th Cir. 2009) (plaintiff must show more than possible exposure; must show concentration and duration); Anderson v. Dow Chem. Co., 255 Fed. Appx. 1, 2007 WL 1879170 (5th Cir. 2007) (lawsuit dismissed because uncontested data showed that magnitude and duration of exposure was insufficient to cause adverse health effects); Finestone v. Florida Power & Light Co., 272 Fed. App’x. 761, 2008 WL 931703 (4th Cir. 2009) (experts’ testimony was properly excluded where their conclusions relied on unsupported assumptions).
36. SPPI-Somersville, Inc. v. TRC Cos., 2009 WL 2612227, at *16 (N.D. Cal. 2009) (groundwater contamination claim was dismissed because there was no current pathway to exposure); United States v. W.R. Grace Co., 504 F.3d 745 (9th Cir. 2007) (affirming exclusion of report, but not expert testimony based on the report, identifying which pathways of asbestos exposure were most associated with lung abnormalities); Grace Christian Fellowship v. KJG Investments Inc., 2009 WL 2460990, at *12 (E.D. Wis. 2009) (preliminary injunction was denied because the plaintiff did not establish that a complete pathway currently existed for toxins to enter the building); National Exposure Research Laboratory, U.S. Environmental Protection Agency, Scientific and Ethical Approaches for Observational Exposure Studies, Doc. No. EPA 600/R-08/062 (2008), available at http://www.epa.gov/nerl/sots/index.html (last visited July 14, 2010); U.S. Environmental Protection Agency. Exposure Factors Handbook (1997).
calculating doses for such substances, as shown in Section VI, is generally a straightforward process. Even in such cases, however, complexities can arise. Thus, in the case of certain personal care products that are applied to the skin, there is a possibility of inhalation exposures to any substance in those products that can readily volatilize at room temperatures. One physical characteristic of chemicals that exposure scientists need to understand is their capacity to move from a liquid to a gaseous state (to volatilize). Not all chemicals are readily volatile (and almost all inorganic, metal-based substances are close to nonvolatile), but inhalation routes can be significant for those that are volatile, regardless of their sources.37
Indirect pathways of exposure can range from the relatively simple to the highly complex. Many packaging materials are polymeric chemicals—very large molecules synthesized by causing very small molecules to chemically bind to each other (or to other small molecules) to make very long chemical chains. These polymers (polyethylene, polyvinyl chloride, polycarbonates, and others) tend to be physically very stable and chemically quite inert (meaning they have very low toxicity potential). But it is generally not possible to synthesize polymers without very small amounts of the starting chemicals (those small molecules, usually called monomers) remaining in the polymers. The small molecules can often migrate from the polymer into materials with which the polymer comes into contact. If those materials are foods or consumer products, people consuming those foods or otherwise using those products will be exposed.
Some amount of the pesticides applied to food crops may remain behind in treated foods and be consumed by people.38 This last pathway can become more complicated when treated crops are used as feed for animals that humans consume (meat and poultry and farm-raised fish) or from which humans obtain food (milk and eggs). Exposure scientists who study these subjects thus need to understand what paths pesticides follow when they are ingested by farm animals used as food. The same complex indirect pathways arise for some veterinary drugs used in animals from which humans obtain food.39
In the realm of environmental contamination, pathways can multiply and the problem of exposure assessment can become even more complex. Sources of environmental contamination include air emissions from manufacturing facilities and from numerous sources associated with the combustion of fuels and other
37. Inhalation exposures to nonvolatile chemicals can occur if they are caused to move into the air as dusts. See National Research Council, Human Exposure Assessment for Airborne Pollutants: Advances and Opportunities (1991).
38. Other pathways for pesticide exposure include spraying homes or fields. Kerner v. Terminix Int’l Co., 2008 WL 341363 (S.D. Ohio 2008) (pesticides allegedly misapplied inside home); Brittingham v. Collins, 2008 WL 678013 (D. Md. Feb. 26, 2008) (crop-dusting plane sprayed plaintiff’s decedent); Haas v. Peake, 525 F.3d 1168 (Fed. Cir. 2008) (veteran claimed exposure to Agent Orange).
39. P. Frank & J.H. Schafer, Animal Health Products, in Regulatory Toxicology (S.C. Gad, ed., 2d ed. 2001).
organic materials.40 Similar emissions to water supplies, including ground water used for drinking or for raising plants and animals, can result in human exposures through drinking water and food.41 Contaminants of drinking water that are volatile can enter the air when water is used for bathing, showering, and cooking. A recent problem of much concern is the contamination of air in homes and other buildings because of the presence of volatile chemical contaminants in the water beneath those structures.42
Wastes from industrial processes and many kinds of consumer wastes can similarly result in releases to air and water.43 In some cases, emissions to air can lead to the deposition of contaminants in soils and household dusts; this type of contamination is usually associated with nonvolatile substances. Some such substances may remain in soils for very long periods; others may migrate from their sites of deposition and contaminate ground water; whereas others may degrade relatively quickly.
All of these issues regarding the movement of chemicals from their sources and through the environment to reach human populations come under the heading of chemical fate and transport.44 Transport concerns the processes that cause chemicals to follow certain pathways from their sources through the environment, and fate concerns their ultimate disposition—that is, the medium in which they finally reside and the length of time that they might reside there. Fate-and-transport scientists have models available to estimate the amount of chemical that will be present in that final environmental medium.45 Some discussion of the nature of these models is offered in Section VI.
One final feature of pathways analysis that should be noted concerns the fact that some chemicals degrade rapidly when they enter the environment, others slowly, and some not at all or only exceedingly slowly. The study of environmental persistence of different chemicals is a significant feature of exposure science; its goal is to understand the chemical nature of the degradation products and the duration of time the chemical and its degradation products persist in any
40. See, e.g., Natural Resources Defense Council, Inc. v. EPA, 489 F.3d 1250 (D.C. Cir. 2007) (vacating EPA rule for solid waste incinerators); Kurth v. ArcelorMittal USA, Inc., 2009 WL 3346588 (N.D. Ind. 2009) (defendant manufacturers allegedly emitted toxic chemicals, endangering schoolchildren); American Industrial Hygiene Association, Guideline on Occupational Exposure Reconstruction (S.M. Viet et al. eds., 2008).
41. United States v. Sensient Colors, Inc., 580 F. Supp. 2d 369, 373 (D.N.J. 2008) (leaching lead threatened to contaminate ground water used for drinking).
42. Interstate Technology & Regulatory Council (ITRC), Vapor Intrusion Pathway: A Practical Guideline. (Jan. 2007), available at http://www.itrcweb.org/Documents/VI-1.pdf.
43. American Farm Bureau Fed’n. v. EPA, 559 F.3d 512 (D.C. Cir. 2009) (EPA outdoor air pollution standards).
44. The common phrase used by exposure scientists is “fate and transport.” In fact, transport takes place and has to be understood before fate is known.
45. In the context of exposure science, the term “final” refers to the medium through which people become exposed. A chemical may in fact continue to move to other media after that human exposure has occurred.
given environmental medium. Most inorganic chemicals are highly persistent; metals that become contaminants may change their chemical forms in small ways (lead sulfide may convert to lead oxide), but the metal persists forever (although it may migrate from one medium to another). Most organic chemicals degrade in the environment as a result of their exposure to light, to microorganisms present in soils and sediments, and to other environmental substances. But a few organic substances (e.g., polychlorinated biphenyls (PCBs) and the chlorinated dioxins, certain chlorinated pesticides such as DDT that were once widely used) are quite resistant to degradation and may persist for unexpectedly long periods (although even these ultimately degrade).46
Exposure scientists also need to be aware of the possibility that the degradation products of certain chemicals may be as or more toxic than the chemicals themselves. The once widely used solvents trichloroethylene and perchloroethylene (tetrachloroethylene) are commonly found in ground water. Under certain conditions, these compounds degrade by processes that lead to the replacement of some chlorine atoms by hydrogen atoms; one product of their degradation is the more dangerous chemical called vinyl chloride (monochloroethylene). The presence of such a degradation product in drinking water should not be ignored.
A description of pathways is the critical first step in exposure assessment and, especially for environmental contaminants, must be done with thoroughness. Are all conceivable pathways accounted for? Have some pathways been eliminated from consideration, and if so, why? Are any environmental degradation products of concern? Only with adequate description can adequate quantification (Section VI) be accomplished.
A graphical description of pathways is offered in Figure 2.
Pathways analysis leads to the identification of the environmental media in which the chemical of interest comes to be present and with which human contact can occur—the media of human exposure.
The inhalation of air containing the chemical of interest is one route of exposure.47 The physical form of the chemical in air, which should be known from the pathways analysis, will influence what happens to the chemical during inhalation. Chemicals that are in the vapor phase will remain in that physical
46. K.W. Fried & K.K. Rozman, Persistent Polyhalogenated Aromatic Hydrocarbons. in Toxicology and Risk Assessment: A Comprehensive Introduction H. Greim & R. Snyder eds., 2009).
47. See, e.g., Byers v. Lincoln Elec. Co., 607 F. Supp. 2d 840 (N.D. Ohio 2009) (welder inhaled toxic manganese fumes); O’Connor v. Boeing North American, Inc., 2005 WL 6035256 (C.D. Cal. 2005) (alleged failure to monitor ambient air emissions of radioactive particles); In re FEMA Trailer Formaldehyde Prod. Liab. Litig., 2009 WL 2382773 (E.D. La. 2009) (trailer residents exposed to formaldehyde).
Figure 2. Description of the many possible environmental pathways that chemicals may follow after releases from different sources.
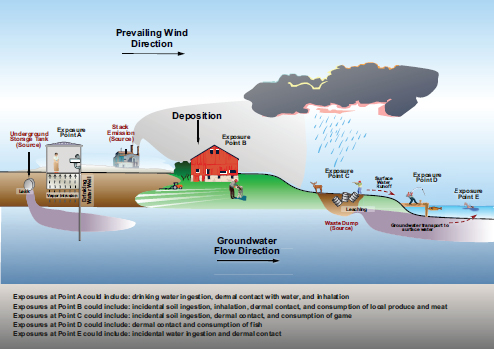
Source: Graphic created by Jason Miller.
state and will move to the lungs, where a certain fraction will pass through the lungs and enter the bloodstream. The extent to which different chemical substances pass through the lungs is dependent in large part upon their physical properties, particularly solubilities in both fatlike materials and water. Passage through cell membranes (of the cells lining the lungs) requires that substances have a degree of both fat solubility and water solubility. Predicting the extent of absorption through the lungs (or the gastrointestinal tract or skin, discussed below) cannot be accomplished with accuracy; knowledge in this area can be gathered only through measurement.
Certain fibrous materials (including but not limited to asbestos) and particulate matter and dusts may move through the airways and may reach the lungs, but some of these kinds of materials may be trapped in the nose and excreted. Generally, only very fine particles reach the lower lung area. Some particles may be deposited in the upper regions of the respiratory tract and then carried by certain physical processes to the pharynx and then be coughed up or swallowed. Thus, inhaled chemicals and particulates can enter the body through the gastrointestinal
(GI) tract or the respiratory tract.48 Understanding risk requires information about these characteristics of the chemicals involved.
Ingestion is the second major route of exposure to substances in environmental media.49 Chemicals that comprise or come to be present in foods, in drinking water, in soils and dusts,50 and many of those that serve as medicines are all ingested. They are swallowed, enter the GI tract, and to greater or lesser degrees are absorbed into the bloodstream at various locations along that tract. This is often referred to as the oral route of exposure.
The largest organ of the body, the skin, is the third route of exposure for chemicals in products and the environment.51 As with the GI tract and the lungs, chemicals are absorbed through the skin to greater or lesser degrees, depending on their physical and chemical characteristics. In some cases, toxic harm can occur directly within the respiratory or GI tracts or on the skin before absorption occurs.52
The pathways analysis allows the identification of all the routes by which chemicals from a given source may enter the body, because it identifies the media of human contact into which the chemicals migrate from their sources. Once the media of human contact are identified, the possible exposure routes are known.
E. Summary of the Descriptive Process
Once the exposure question to be examined has been defined, the exposure scientist sets out to identify all the relevant sources of exposure to the chemicals of interest. All the pathways the chemicals can follow from those sources to reach the population of interest are then described, with careful attention to the possibility that chemical degradation (to more or less toxic substances) can occur. The pathways analysis concludes with a description of what chemicals will be present in the various environmental media with which the exposed populations were, are, or could become exposed (air, water, foods, soils and dusts, consumer products). At this point, it becomes possible to identify the routes by which the chemicals can enter the body.
48. J.V. Rodricks, From Exposure to Dose, in Calculated Risks: The Toxicity and Human Health Risks of Chemicals in Our Environment (2d ed. 2007).
49. See, e.g., Foster v. Legal Sea Foods, Inc., 2008 WL 2945561 (D. Md. 2008) (hepatitis A allegedly contracted from eating undercooked mussels); Winnicki v. Bennigan’s, 2006 WL 319298 (D.N.J. 2006) (alleged foodborne illness contracted from defendant’s restaurant led to renal failure and death); Palmer v. Asarco Inc., 2007 WL 2298422 (N.D. Okla. 2007) (children allegedly ingested dust and soil contaminated with lead).
50. Inadvertent exposures to these and other nonfood items are known to occur and can be especially common in children.
51. See, e.g., United States v. Chamness, 435 F.3d 724 (7th Cir. 2006) (evidence that methamphetamine and the ingredients used in its manufacture are toxic to the eyes, mucous membranes, and skin supported sentencing enhancement for danger to human life).
52. J.V. Rodricks, From Exposure to Dose, in Calculated Risks: The Toxicity and Human Health Risks of Chemicals in Our Environment (2d ed. 2007).
Description by itself, however, often is inadequate. Attempts have to be made to quantify exposure, to arrive at estimates of the dose received by the exposed population, and to determine the duration of time over which that dose is received.
VI. Quantification of Exposure
The simplest dose calculations relate to situations in which direct exposures occur.53 Thus, for example, consider the case of a substance directly added to food (and approved by the U.S. Food and Drug Administration (FDA) for such addition). Suppose the chemical is of well-established identity and is approved for use in nonalcoholic beverages at a concentration of 10 milligrams of additive for each liter of beverage (10 mg/L).54 To understand the amount (weight) of the additive ingested each day, it is necessary to know how much of the beverage people consume each day. Data are available on rates of food consumption in the general population. Typically, those data reflect average consumption rates and also rates at the high end of consumption. To make sure that the additive is safe for use, FDA seeks to ensure the absence of risk for individuals who may consume at the high end, perhaps at the 95th percentile of consumption rates.55 Surveys of intake levels for the beverage in our example reveal that the 95th percentile intake is 1.2 L per day for adults.
The weight of additive ingested by individuals at the 95th percentile of beverage consumption rate is thus obtained as follows:
10 mg/L × 1.2 L/day = 12 mg/day.
For a number of reasons, toxicologists express dose as weight of chemical per unit of body weight. For adults having a body weight (bw) of, on average, 70 kilograms (kg), the dose of additive is
12 mg/day ÷ 70 kg bw = 0.17 mg/kg bw per day.56
53. See, e.g., McLaughlin v. Sec’y of Dep’t of Health & Human Servs., 2008 WL 4444142 (Fed. Cl. 2008) (plaintiff exposed to known dose of thimerosol in vaccine; study using four times that dose was not reliable evidence that exposure caused his autistic symptoms).
54. See Appendix A for a discussion of units used in exposure science.
55. J.V. Rodricks & V. Frankos, Food Additives and Nutrition Supplements, in Regulatory Toxicology 51–82 (C.P. Chengeliss et al. eds., 2d ed. 2001).
56. To gain approval for such an additive, FDA would require that no toxic effects are observable in long-term animal studies at doses of at least 17 mg/kg bw per day (100 times the high-end human intake).
Doses from other ingested products containing specified amounts of chemicals are calculated in much the same way. It generally would be assumed that the duration of exposure for a substance added to a food or beverage would be continuous and would cover a large fraction of a lifetime. For other products, particularly pharmaceuticals, exposure durations will vary widely; dose calculations would be the same, regardless of duration, but the potential for harm requires consideration of exposure duration.
It will be useful, before proceeding further, to illustrate dose calculations for exposures occurring by the inhalation and dermal routes.57 Consider a hypothetical workplace setting in which a solvent is present in the air. Measurement by an industrial hygienist reveals its presence at a weight of 2 mg in each cubic meter (m3) of air. Data on breathing rates reveal that a typical worker breathes in 10 m3 of air each 8-hour workday.58 Thus, the worker dose will be
2 mg/m3 × 10 m3/day = 20 mg/day
20 mg/day ÷ 70 kg = 0.28 mg/kg bw per day.
As noted earlier, it is likely that only a fraction of this dose will reach and pass through the lungs and enter the bloodstream. As also noted earlier, if the chemical is a fiber or other particle, its dynamics in the respiratory tract will be different than that of a vapor, with a portion of the inhaled dose entering the GI tract.
Dose from skin exposure often is expressed as the weight of chemical per some unit of skin surface area (e.g., per m2 of skin). The body surface area of an average (70 kg) adult is 1.8 m2. Thus, consider a body lotion containing a chemical of interest. If the lotion is applied over the entire body, then it is necessary to know the total amount of lotion applied and then the total amount of chemical present in that amount of lotion. That last amount will then be divided by 1.8 to yield the skin dose in units of milligrams per square meter. If the chemical causes toxicity directly to the skin, that toxicity dose information also will be expressed in milligrams per square meter. Then risk is evaluated by examining the quantitative relationship between the toxic dose (milligrams per square meter) and the (presumably much lower) human dose expressed in the same units. If the chemical can penetrate the skin and produce toxicity within the body, then the dose determination must include an examination of the amount absorbed into the human body.59
57. See, e.g., Henricksen v. ConocoPhillips Co., 605 F. Supp. 2d 1142, 1164 (E.D. Wash. 2009) (benzene exposure on skin and by inhalation); Bland v. Verizon Wireless (VAW) LLC, 2007 WL 5681791, at *9 (S.D. Iowa 2007) (inhalation exposure to Freon in “canned air” sprayed into water bottle). For a discussion of the importance of assessment of dose as a measure of exposure, see Bernard D. Goldstein & Mary Sue Henifin, Reference Guide on Toxicology, Section I.A.1.c, in this manual.
58. The 24-hour inhalation rate outside the workplace setting is ca. 20 m3. The lack of direct proportion to time reflects the fact that breathing rates increase under exertion.
59. Rates of absorption of chemicals into the body, through the GI tract, the lungs, or the skin, usually must be obtained by measurement; they are not readily predicted.
One final matter concerning dose estimation concerns the importance of body size, in particular that of the infant and the growing child. In matters such as food and water intake, and breathing rates, small children are known to take in these media at higher rates per unit of their body weights than do adults.60 Thus, when a small child is exposed to a food contaminant, that child will often receive a greater dose of the contaminant than will an adult consuming food with the same level of contaminant. Children also tend to ingest greater amounts of nonfood items, such as soils and dusts, than do adults. In some cases, nursing mothers excrete chemicals in their milk. The exposure scientist generally conducts separate assessments for children that take into account the possibility of periods of increased exposure during the developmental period.61
B. Doses from Indirect Exposure Pathways
Recall that the goal of exposure assessment is to identify the media through which people will be exposed to chemicals of interest that are emitted from sources of interest. As will be seen, the assessment, when completed, will reveal the amount of the chemical of interest in a certain weight or volume of each of the media with which people come into contact. Once this is known, dose calculations can proceed in the manner described in the preceding section.
In the preceding section, firm and readily available knowledge was available about the amount of chemical present in a given weight of food or consumer product (the body lotion example) or in a given volume (cubic meters) of air. These measures are called concentrations of the chemicals in the media of exposure (see Appendix A). When a chemical must move from one or more sources, and then through one or more environmental media, before it comes to be present in the media with which people have contact (the media of exposure), determining the concentrations of the chemical in the media of exposure becomes difficult.62 Such a situation is clearly different from that in which a specific amount of an additive is directly added to a specific amount of food. The challenge faced by exposure scientists when the chemical comes to be present in the medium of human exposure not by direct and intentional addition, but by indirect means, through movement from source through the environment, is to find a reliable
60. See, e.g., Northwest Coalition for Alternatives to Pesticides (NCAP) v. EPA, 544 F.3d 1043 (9th Cir. 2008) (dispute over how much lower allowable pesticide levels should be to account for children’s greater susceptibility).
61. For some substances, susceptibility to toxicity is also enhanced during the same periods. See Section VII.B.
62. See, e.g., Hannis v. Shinseki, 2009 WL 3157546 (Vet. App. 2009) (no direct measure of veteran’s exposure to radiation was possible but VA’s dose estimate was not clearly erroneous); Fisher v. Ciba Specialty Chem. Corp., 2007 WL 2302470 (S.D. Ala. 2007) (allowing expert’s qualitative account of DDT and its metabolites spreading from defendant’s plant to plaintiffs’ property, because quantification would necessarily rely on speculative data).
way to estimate concentrations in the medium of human exposure.63 Once concentrations are known, dose is readily calculated (as in Section VI.A), but reliably estimating concentrations can be difficult.
Two methods typically are used to estimate those concentrations. One involves direct measurement using the tools of analytical chemistry. The second involves the use of models that are intended to quantify the concentrations resulting from the movement of chemicals from the source to the media of human exposure.
C. Direct Measurement: Analytical Science
Once the media that could be subject to contamination have been identified through pathways analysis (Section V.C), one available choice for determining the concentrations of contaminants involves sampling those media and subjecting the samples taken to chemical analysis. The analysis will not only reveal the concentrations of chemicals in the media of concern, but should also confirm their identities. Environmental sampling and analysis is under way all over the world, at and near contaminated waste sites, in the vicinity of facilities emitting chemicals to air and water, and in many other circumstances.64
One purpose of such sampling and analysis is to determine whether products and environmental media contain substances at concentrations that meet existing regulatory requirements. In many circumstances, regulators have established limits on the concentrations of certain chemicals in foods, other products, water, air, and even soils. These limits generally are based on assessments of health risk and calculations of concentrations that are associated with what the regulators believe to be negligibly small risks. The calculations are made after first identifying the total dose of a chemical that is safe (poses a negligible risk) and then determining the concentration of that chemical in the medium of concern that should not be exceeded if exposed individuals (typically those at the high end of media contact) are not to incur a dose greater than the safe one. The most common concentration limits are regulatory tolerances for pesticide residues in food, Maximum Con-
63. See, e.g., Knight v. Kirby Inland Marine Inc., 482 F.3d 347, 352–53 (5th Cir. 2007) (study of people with much longer exposure to organic solvents could not support conclusion that plaintiff’s injuries were caused by such solvents); Kennecott Greens Creek Mining Co. v. Mine Safety & Health Admin., 476 F.3d 946, 950 (D.C. Cir. 2007) (because diesel particulate matter was difficult to monitor, MSHA’s surrogate limits on total carbon and elemental carbon were reasonable).
64. See, e.g., Genereux v. American Beryllia Corp., 577 F.3d 350, 366–67 (1st Cir. 2009) (“all beryllium operations should be periodically air-sampled, and a workspace may be dangerous to human health even though no dust is visible”); Allen v. Martin Surfacing, 2009 WL 3461145 (D. Mass. 2009) (where air sampling was not done, expert resorted to modeling plaintiff’s decedent’s exposure); Jowers v. BOC Group, Inc., 608 F. Supp. 2d 724, 738 (S.D. Miss. 2009) (OSHA measurements showed that 30% of welders experienced manganese fumes at higher than allowable concentrations); In re FEMA Trailer Formaldehyde Prod. Liab. Litig., 583 F. Supp. 2d at 776 (air sampling revealed formaldehyde levels higher than allowable).
taminant Levels (MCLs) for drinking water contaminants, National Ambient Air Quality Standards (NAAQS), and, for workplace exposure, Permissible Exposure Limits (PELs) or Threshold Limit Values (TLVs).65 Much environmental sampling and analysis is done, by both government agencies and private organizations, for the purpose of ascertaining compliance with existing concentration limits (sometimes referred to as standards).
But sampling and analysis also are undertaken to investigate newly identified contamination or to ascertain exposures (and risks) in situations involving noncompliance with existing standards. As described earlier, information on concentrations in the media through which people are exposed is the necessary first step in estimating doses.
Although at first glance it might seem that direct measurements of concentrations would provide the most reliable data, there are limits to what can be gained through this approach.
- How can we be sure that the samples taken are actually representative of the media sampled?
-
Standard methods are available to design sampling plans that have specified probabilities of being representative, but they can never provide complete assurance. Generally, when contamination is likely to be highly homogeneous, there is a greater chance of achieving a reasonably representative sample than is the case when it is highly heterogeneous. In the latter circumstance, obtaining a representative sample, even when very large numbers of samples are taken, may be unachievable.
- How can we be sure that the samples taken represent contamination over long periods?
-
Sampling events may provide a good snapshot of current conditions, but in circumstances in which concentrations could be changing over time, and where the health concerns involve long-term exposures, snapshots could be highly misleading. This type of problem may be especially severe when attempts are being made to reconstruct past exposures, based on snapshots taken in the present.
- How can we be sure that the analytical work was done properly?
-
Most major laboratories that routinely engage in this type of analysis have developed standard operating procedures and quality control
65. PELs are official standards promulgated by the Occupational Safety and Health Administration. TLVs are guidance values offered by an organization called the American Conference of Governmental Industrial Hygienists. See, e.g., In re Howard, 570 F.3d 752, 754 (6th Cir. 2009) (challenging PELs for coal mine dust); Jowers v. BOC Group, Inc., 608 F. Supp. 2d 724, 735–36 (S.D. Miss. 2009) (PELs and TLVs for welders’ manganese fume exposure); International Brominated Solvents Ass’n v. American Conf. of Gov. Indus. Hygienists, Inc., 625 F. Supp. 2d 1310 (M.D. Ga. 2008) (challenging TLVs for several chemicals); Miami-Dade County v. EPA, 529 F.3d 1049 (11th Cir. 2008) (MCLs for public drinking water).
-
procedures. Laboratory certification programs of many types also exist to document performance. When analytical work is performed in certified, highly experienced laboratories, there is a reasonably high likelihood that the analytical results are reliable. But it is very difficult to confirm reliability when analytical work is done in laboratories or by individuals who cannot provide evidence of certification or of longstanding quality control procedures.
- How are data showing the absence of contamination to be interpreted?
-
In most circumstances involving possible contamination of environmental media, the analysis of some (and sometimes many) of the samples will fail to find the contaminant. The analytical chemist will often report “ND” (for nondetect) for such samples. But an ND should never be considered evidence that the concentration of the contaminant is zero. In fact, most chemists will (and should) report that the contaminant is “BDL” (below detection limit). Every analytical method has a nonzero detection limit; the method is not sensitive to and cannot measure concentrations below that limit. Thus, for each sample reported as BDL, all that can be known is that the concentration of contaminant is somewhere below that limit. If there is clear evidence that the contaminant is present in some of the samples (its concentration exceeds the method’s BDL), then it is usually assumed that all the samples of the same medium reported as BDL will actually contain some level of contaminant, often and for reliable reasons assumed to be one-half the BDL. Practices for dealing with BDL findings vary, but assuming that the BDL is actually zero is not one of the acceptable practices.
Sampling and measurement are no doubt useful, but are nonetheless limited in important ways. The alternative involves modeling. In fact, a combination of both approaches—one acting as a check on the other—is often the most useful and reliable.
A model is an attempt to provide a mathematical description of how some feature of the physical world operates. In the matters at hand, a model refers to a mathematical description of the quantitative relationship between the amount of a chemical emitted from some source, usually over a specified period of time, to the concentrations of that chemical in the media of human exposure, again over some specified time period.66
66. See, e.g., NCAP v. EPA, 544 F.3d 1043 (9th Cir. 2008) (EPA was permitted to rely on modeling in developing allowable pesticide residual levels); O’Neill v. Sherwin-Williams Co., 2009 WL 2997026, at *5 (C.D. Cal. 2009) (exposure model was inappropriate because it was based on a different type of paint than plaintiff was exposed to); Hayward v. U.S. Dep’t of Labor, 536 F.3d 376
Models are idealized mathematical expressions of the relationship between two or more variables. They are usually derived from basic physical and chemical principles that are well established under idealized circumstances, but may not be validated under actual field conditions. Models thus cannot generate completely accurate predictions of chemical concentrations in the environment. In some cases, however, they are the only method available for estimating exposure—for example, in assessing the impacts of a facility before it is built or after it has ceased to operate. In such circumstances, they are necessary elements of exposure assessments and have been used extensively. Models are necessary if projections are to be made backward or forward in time or to other locations where no measurements have been made.
Typically, a model is developed by first constructing a flow diagram to illustrate the theoretical pathways of environmental contamination, as shown in Figure 2 and for a hazardous waste site in Appendix B. These models can be used to estimate concentrations in the relevant media based on several factors related to the nature of the site and the chemicals of interest. Model variables include the following:
- The total amount of chemical present in or emitted from the media that are its sources;
- The solubility of the chemical in water;
- The chemical’s vapor pressure (a measure of volatility);
- The degree to which a chemical accumulates in fish, livestock, or crops (bioconcentration or bioaccumulation factor);
- The nature of the soil present at the site; and
- The volumes and movement of water around and beneath the site.
Some of this information derives from laboratory studies on the chemical (the first four points) and some from an investigation at the site (the remaining two points). The development of the data and modeling of the site often require the combined skills of chemists, environmental engineers, and hydrogeologists. In addition to the information listed above, time projection models also require information on the stability of the chemical of interest. As noted earlier, some chemicals degrade in the environment very quickly (in a matter of minutes), whereas others are exceedingly resistant to degradation. Quantitative information on rates of degradation is often available from laboratory and field studies.
Models that assess the exposures associated with air emissions consider the fact that the opportunity for people to be exposed to chemicals depends upon their activities and locations.67 These models account for the activity patterns of
(5th Cir. 2008) (a model was used to reconstruct the dose of radiation that the employee was exposed to); Rodricks & Frankos, supra note 55.
67. See, e.g., Palmer v. Asarco Inc., 2007 WL 2298422 (N.D. Okla. 2007) (children allegedly were exposed to lead by “hand-to-mouth activity ingestion of soil/house dust”); Henricksen
potentially exposed populations and provide estimates of the cumulative exposure over specified periods.
Perhaps the most widely used models are those that track the fate and transport pathways followed by substances emitted into the air. Knowledge of the amounts emitted per unit of time (usually obtainable by measurement) from a given location (a stack of a certain height, for example) provides the basic model input. Information on wind directions and velocities, the nature of the physical terrain surrounding the source, and other factors needs to be incorporated into the modeling. Some substances will remain in the vapor phase after emission, but chemical degradation (e.g., because of the action of sunlight) could affect media concentrations. Some models provide for estimating the distributions of soil concentrations for those substances (particulates of a certain size) that may fall during dispersion. Much effort has been put into developing and validating air dispersion models.68 Similar models are available to track the movement of contaminants in both surface and ground waters.
The fate and transport modeling issue becomes more complex when attempts are made to follow a chemical’s movement from air, water, and soils into the food chain and to estimate concentrations in the edible portions of plants and animals.69 Most of the effort in this area involves the use of empirical data (e.g., What does the scientific literature tell us about the quantitative relationships between the concentration of cadmium in soil and its concentration in the edible portions of plants grown in that soil?). This type of empirical information, together with general data on chemical absorption into, distribution in, and excretion from living systems, is the usual approach to ascertain concentrations in these food media.70
Many models for environmental fate and transport analysis are available. It is not possible to specify easily which models have established validity and which have not; rather, some are preferred for some purposes and others are preferred for different purposes.
Perhaps the best that can be done to scrutinize the work of an expert in this area is to
- Require that the expert describe in full the basis for model selection;
- Ask the expert to describe the standing of the model with authoritative bodies such as EPA;
- Require the expert to state why other possible models are not suitable;
v. ConocoPhillips Co., 605 F. Supp. 2d 1142, 1164 (E.D. Wash. 2009) (expert calculated plaintiff’s benzene exposure by adjusting study results to account for plaintiff’s activities); Junk v. Terminix Int’l Co., 2008 WL 6808423 (S.D. Iowa 2008) (study measured chlorpyrifos exposure of inhabitants of houses sprayed indoors); In re W.R. Grace & Co., 355 B.R. 462 (Bankr. D. Del. 2006) (asbestos in attic insulation released by normal activity).
68. National Research Council, Models in Environmental Regulatory Decision Making (2007).
69. Ecologists also use modeling results to evaluate risks to wildlife, plants, and ecosystems.
70. National Research Council, supra note 68.
- Require that the expert describe the scientific basis and underlying assumptions of the model, and the ways in which the model has been verified;71 and
- Require the expert to describe the likely size of error associated with model results.
Other issues pertaining to the sources and reliability of the data used in the application of a model can be similarly pursued.
Results from modeling are concentrations in media of concern over time. If sampling and analysis data are available for the same media, they can be compared with the modeling result, and efforts can be made to reconcile the two and arrive at the most likely values (or range of likely values).
E. Integrated Exposure/Dose Assessment
We have shown the various methods used to determine the concentrations of chemicals in products and in various environmental media and also the methods used to determine doses from each of the relevant media. Dose estimation as described in Section VI.A applies to each of the relevant routes of exposure.
In many cases, the dose issue concerns one chemical in one product and only one route of exposure. But numerous variations on this basic scenario are possible: one chemical in several products or environmental media, many chemicals in one product or environmental medium, or many chemicals in many environmental media. Even though some exposure situations can be complex and involve multiple chemicals through both direct and indirect pathways, the exposure assessment methods and principles described here can be applied. Exposures occurring by different routes can be added together, or they can be reported separately. The decisions on the final dose estimates and their form of presentation can be made only after discussions with the users of that information—typically the toxicologists and epidemiologists involved in the risk assessment.72 The dose metrics emerging from the exposure assessment need to match the dose metrics that are used to describe toxicity risks.
One additional point should be highlighted. The principle that exposure to chemicals through foods and consumer products typically focuses on high-end consumers of those foods or products also applies in environmental settings. Thus,
71. This point is to ensure that the expert truly understands the model and its limits and that he or she is not simply using some “black box” computer software.
72. See, e.g., American Farm Bureau Fed’n v. EPA, 559 F.3d 512 (D.C. Cir. 2009) (challenging EPA’s risk assessment for fine PM); Miami-Dade County v. EPA, 529 F.3d 1049 (11th Cir. 2008) (assessment of risk of wastewater disposal methods to drinking water); Kennecott Greens Creek Min. Co. v. Mine Safety & Health Admin., 476 F.3d 946 (D.C. Cir. 2007) (risk assessment of diesel particulate matter to miners); Rowe v. E.I. du Pont de Nemours & Co., 2008 WL 5412912, 12 (D.N.J 2008) (risk assessment for proposed class).
for example, it is possible to assert with relatively high confidence that almost no one consumes more than 3.5 L of water a day and that almost everyone consumes less. If the dose calculation assumes a water consumption rate of 3.5 L/day, then the risk estimated for that dose is almost certainly an upper limit on the population risk, and regulatory actions based on that risk will almost certainly be highly protective. For regulatory and public health decisionmaking, such a precautionary approach has a great deal of precedent, although care must be taken to ensure adherence to scientific data and principles.73
This approach becomes problematic, however, if applied to assessments of exposures that may have been incurred in the past by individuals claiming to have been harmed by them. In such cases, it would seem that there is no basis for a precautionary approach; an approach based on attempts to accurately describe the individual’s exposure would seem to be necessary. Whatever the case, the exposure scientist must be careful to ensure accurate description of the exposure concentration (and resulting dose), so that the users of the information can understand whether upper limits or more typical exposures and doses have been provided.
Section V described how chemicals in the environment contact the three major portals of entry into the body—the respiratory tract, the GI tract, and the skin. For some chemicals, the dose contacting one or more of those portals may be sufficient to cause harm before those chemicals are absorbed into the body; that is, they may cause one or more forms of toxicity to the respiratory system, to the GI tract, or to the skin. Although these forms of contact toxicity can be important, it is also important to consider the many forms of systemic toxicity. The latter refers to a large number of toxic manifestations that can affect any of the organs or organ systems of the body after a chemical is absorbed into the bloodstream and distributed within the body. Recall also that most chemicals are acted upon by certain large protein molecules, called enzymes, contained in cells, particularly those of the liver, the skin, and the lungs, and are converted to new compounds, called metabolites (the process leading to these changes is called metabolism). Metabolite formation
73. National Research Council, Evolution and Use of Risk Assessment in the Environmental Protection Agency: Current Practice and Future Prospects, in Science and Decisions: Advancing Risk Assessment (2008). Those who must comply with regulations that were developed based on a high degree of caution often protest that more accurate assessments should be used as their basis. For several reasons, truly accurate prediction of risk is difficult to achieve (see Bernard D. Goldstein & Mary Sue Henifin, Reference Guide on Toxicology, in this manual), while predicting an upper bound on the risk is not. At the same time, unless carefully done and described, upper-bound estimates may be so remote from reality that decisions based on them should be avoided.
is one of the body’s mechanisms for creating compounds that are easily removed from the body by one or more excretion processes. Unfortunately, metabolism sometimes creates new compounds that are more toxic than the original (so-called parent) molecule, and, if the internal dose of toxic metabolite exceeds a so-called threshold, toxic harm may occur. Of course, not all toxicity is produced by metabolites; in some cases harm may be caused directly by the parent compound.74
As in the other areas of exposure science that have been discussed, it usually becomes important to move from description to quantification. Exposure scientists seek to understand the amount of chemical absorbed into the body after contact (i.e., the fraction of the dose that is absorbed), the amount of chemical reaching and distributed within the body (the blood concentration being the most easily measurable), and the rate of loss of the chemical from the body. The science devoted to understanding these important phenomena is called pharmacokinetics (drug rates). That name came to be used because most of the developmental work in this area related to the behavior of pharmaceuticals in the body, but the tools of pharmacokinetics have been extended to study all types of chemicals.
Pharmacokinetics is important because it reveals where in the body a chemical is most likely to cause harm (where the greatest concentrations, or target site doses, are reached for the longest period of time) and also the concentration—duration level necessary to cause harm. To understand these relationships, pharmacokinetic studies typically are carried out in conjunction with toxicity studies in animals, and their results are used to assess possible toxic risk in humans.75
Pharmacokineticists do not ordinarily characterize themselves as exposure scientists; more often they are toxicologists or pharmacologists. But they are in fact extending the usual work of exposure scientists into the body, and it is here that we see the interface between exposure science and toxicology and epidemiology.
B. Monitoring the Body (Biomonitoring)
As long as we live in a world of chemicals, we will be exposed to them. If analytical chemists developed sufficiently sensitive measuring techniques, it would not be far-fetched to say that we could find within the human body, at some level and for some period, virtually any of the tens of thousands of chemicals, natural and synthetic, with which it comes into contact. Some would be found only occasionally, some continuously; some would be found to persist for days, weeks,
74. J.V. Rodricks, From Exposure to Dose, in Calculated Risks: The Toxicity and Human Health Risks of Chemicals in Our Environment (2d ed. 2007)
75. See Bernard D. Goldstein & Mary Sue Henifin, Reference Guide on Toxicology, in this manual. See also, e.g., In re Fosamax Prod. Liab. Litig., 645 F. Supp. 2d 164, 186 (S.D.N.Y. 2009) (rat and dog studies showing a bisphosphonate caused jaw necrosis relevant to whether Fosamax, another type of bisphosphonate, could cause jaw necrosis in humans); Rose v. Matrixx Initiatives, Inc., 2009 WL 902311, at *14 (W.D. Tenn. 2009) (studies in animals of nasal spray effects could not be extrapolated to humans because olfactory physiology was too different).
or even longer, whereas others would persist for only minutes or hours. The concentrations in blood would likely vary over many orders of magnitude. Currently, we can measure only a few thousand chemicals in the body, a large share of them pharmaceuticals, nutrients, and substances of abuse. Some standards for occupational exposures are expressed as allowable blood or urine concentrations, and their measurement is a useful supplement to air monitoring.76
The environmental chemical that has perhaps received the most attention in this area of exposure science is lead (chemical symbol Pb). Indeed, lead may be the most studied of all environmental substances. After it was learned in the 1950s that the concentration of lead in blood could be easily measured, it became common to sample and test the blood level of lead (BPb) in individuals who had suffered one or more forms of this metal’s toxicity. Some epidemiological studies of lead began to include BPb as the measure of exposure, and since the 1970s, hundreds of such studies involving lead have reported results using this measure.77
BPb is particularly useful for substances such as lead that have (or did have) a relatively large number of environmental sources.78 The simple measure of BPb provides a single, integrated measure of exposures through multiple sources, pathways, and routes (although this measure reflects relatively recent and not long-term exposure).79 This is perhaps the best example of the use of target site dose in risk assessment.
The Centers for Disease Control and Prevention (CDC) began, in the late 1970s, to take blood samples from a relatively large number of children as part of its National Health and Nutrition Examination Survey (NHANES). Children were selected because it was known that they take up more lead from their
76. See, e.g., Haas v. Peake, 525 F.3d 1168, 1177 (Fed. Cir. 2008) (presumption of dioxin exposure instituted because of the difficulty of measuring dioxin in the body); Young v. Burton, 567 F. Supp. 2d 121 (D.D.C. 2008) (hormone and enzyme levels allegedly altered by exposure to bio-toxins in mold); Hazlehurst v. Sec’y of Dep’t of Health & Human Servs., 2009 WL 332306, at *62 (Fed. Cl. 2009) (study measuring porphyrin in urine as a marker for mercury in the body); United States v. Bentham, 414 F. Supp. 2d 472 (S.D.N.Y. 2006) (cocaine use monitored by a “sweatpatch” on the skin).
77. National Center for Environmental Health, Centers for Disease Control and Prevention, Fourth National Report on Human Exposure to Environmental Chemicals (2009), available at http://www.cdc.gov/exposurereport/pdf/FourthReport.pdf (last visited July 1, 2010).
78. See, e.g., Potter v. EnerSys, Inc., 2009 WL 3764031 (E.D. Ky. 2009) (alleged lead exposure from working on battery manufacturing site); City of North Chicago v. Hanovnikian, 2006 WL 1519578 (N.D. Ill. 2006) (alleged lead contamination of soil); Perry ex rel. Perry v. Frederick Inv. Corp., 509 F. Supp. 2d 11 (D.D.C. 2007) (residential lead paint exposure); Goodstein v. Continental Cas. Co., 509 F.3d 1042 (9th Cir. 2007) (environmental contamination from lead waste site); Evansville Greenway & Remediation Trust v. Southern Indiana Gas & Elec., 661 F. Supp. 2d 989 (S.D. Ind. 2009) (contamination of battery recycling site).
79. BPb usually is reported in units of micrograms (1 one-millionth of 1 gram) in each deciliter (one-tenth of a liter) of blood (µg/dL). More recently, noninvasive methods to measure lead levels in teeth and bones have become available; such measures reflect cumulative exposures over long periods, but their relationships to health are less clear than those based on BPb.
environments (air,80 water, food, paint, soils and dusts, emissions from lead and other metal smelters, consumer products, and more) than do adults; they are also, especially during early periods of development, more vulnerable to the adverse effects of lead than are adults. Nationwide, childhood BPb levels averaged 15–20 µg/dL during the 1970s, with substantial numbers of children having BPb levels well in excess of what was at the time thought to be the minimum BPb associated with adverse health effects (40 µg/dL). The most recent NHANES surveys reveal that average childhood levels are in the range of 2 µg/dL, although there remain substantial numbers of children with levels greater than the current CDC health guideline of 10 µg/dL.81
Lead is not the only chemical now being studied under the NHANES bio-monitoring program. The most recent surveys involve nationwide sampling of blood and urine from close to 8000 children and adults for more than 100 different chemicals.82 The program focuses on commonly used pesticides and consumer products and certain ubiquitous environmental contaminants, particularly those that persist in the body for long periods. Not surprisingly, most of these chemicals have been detected in some individuals. The NHANES program will continue, and similar programs are under way in government and research centers around the world.
The presence of a chemical in the body is not evidence that it is causing harm. And in some cases—those that involve chemicals, such as the metals and some organic compounds that occur naturally—the NHANES findings may simply reflect natural background levels.83 In any case, data such as these provide far more direct measures of dose (often referred to as body burden), and in those cases (which are increasing in number) in which epidemiologists and toxicologists are able to relate disease rates to body burdens (instead of to external dose, as is the usual case), far more accurate measures of human risk should become available.
VIII. Evaluating the Scientific Quality of an Exposure Assessment
Exposure scientists may offer expert testimony regarding exposures to chemicals incurred by individuals or populations. Their assessments typically will include
80. At the time of the first NHANES lead survey, leaded gasoline, which emitted lead to air and to soil, was in wide use. That use, at least in the United States, came to an end in the 1980s. For a discussion of the routes of exposure to toxic substances, see Bernard D. Goldstein & Mary Sue Henifin, Reference Guide on Toxicology, Section III.A, in this manual.
81. There is developing evidence of IQ deficits in children at levels below 10 µg/dL.
82. National Center for Environmental Health, supra note 77.
83. Natural background levels of certain metals may, in some geological regions, be quite high and may even be associated with excess disease.
a description of how and when exposures have or could occur, the identities of the chemicals involved, the routes of exposure, the doses incurred, and the durations of exposure. In some cases, testimony will include a description and quantification of body burdens. If the exposure scientist is also an epidemiologist or toxicologist,84 he or she may offer additional testimony on the health risks associated with those exposures or even regarding the question of whether such exposures have actually caused disease.
For purposes of this reference guide, it is assumed that questions regarding disease risk and causation are beyond the bounds of exposure science. Below is offered a set of questions that exposure scientists should be able to answer, with appropriate documentation and scientific reasoning, to support any given exposure assessment:
- Is the purpose of the assessment clear? Is the exposed population specified?
- What is the source(s) of exposure?
- When did the exposures occur: past? present? If they are occurring now, will they continue to occur?
- What is the assumed duration of exposure, and what is its basis?
- What are the pathways from the source to the exposed individuals? How has it been established that those pathways exist (past? present? future?).
- What is the concentration of the chemical in the media with which the exposed population comes into contact (past? present? future?). What is the basis for this answer: direct measurement? modeling?
- If the concentration is based on direct measurement, what procedures were followed in obtaining that measurement? Was media sampling sufficient to ensure that it was representative? If not, why is representativeness not important? Were validated analytical methods used by an accredited laboratory? If not, how can one be assured that the analytical results are reliable?
- If models were used, what is their reliability (see Section VI.D)? What is the variability over time in concentrations in the media of concern? How has the variability been determined?
- What is the variability among members of the population in their exposure to the chemical of concern? How is this known?
- What is known or assumed about the nature and extent of media contact by members of the exposed population? How has this been ascertained?
- What dose, over what period of time, by which routes, has been incurred? What calculations support this determination?
84. See Section IX, which deals with the question of the qualifications of exposure scientists. In many cases, the work of exposure experts is turned over to the health experts to incorporate into their evaluation of risk and disease causation. In some cases, usually the less complex ones, exposure assessments may be undertaken by the health experts.
- What is the likely error in the exposure estimates?
- What uncertainties are associated with the dose/duration findings? Is it a “most likely” estimate, or is it an “upper limit”? To what fraction of the population is the “upper limit” likely to apply?
- What has been omitted from the exposure assessment, and why?
These questions are perhaps the minimum that an expert should be able to address when offering testimony. Obviously, most such questions can be answered fully only if the expert can support the answers with documentation.
As noted in Section III.D, the evaluation of whether a current medical condition is causally related to exposures occurring in the past (prior to the onset or diagnosis of the medical condition) requires a retrospective examination of the conditions that led to those exposures. Thus, for example, a plaintiff suffering from leukemia and who alleges that benzene exposure in his or her workplace caused the disease may easily demonstrate the fact of benzene exposure. But ordinarily an estimation of the quantitative magnitude and duration of the incurred benzene exposure is necessary to evaluate the plausibility of the causation claim.85 The methodological tools necessary to “reconstruct” the plaintiff’s past exposure are identical to those used to estimate current exposures, but the availability of the data necessary to apply those methods may be limited or, in some cases, nonexistent.
Reconstruction of occupational exposures has been a relatively successful pursuit, because often historical industrial hygiene data are available involving the measurement of workplace air levels of chemicals. If it is possible, through the examination of employment records, to reconstruct an individual’s job history, it may be possible to ascertain that individual’s exposure history.86 Guidelines for occupational exposure reconstruction have been published by the American Industrial Hygiene Association.87 Clearly, experts presenting testimony regarding exposure reconstruction must be queried heavily on the sources of data used in their applications of exposure methods.
IX. Qualifications of Exposure Scientists
Exposure science is not yet a true academic discipline. Rather, scientists and engineers from diverse backgrounds have, over the past several decades, come together to give shape and substance and scientific rigor to what is clearly a criti-
85. See Michael D. Green et al., Reference Guide on Epidemiology, Section VII, in this manual.
86. T.W. Armstrong, Exposure Reconstruction, in Mathematical Models for Estimating Occupational Exposures to Chemicals (Charles B. Keil et al. eds., 2d ed. 2009).
87. American Industrial Hygiene Association, Guideline on Occupational Exposure Reconstruction (S.M. Viet et al. eds., 2008).
cal element in understanding toxicity risks and disease causation. Typically, those who have contributed to this developing field have come from backgrounds in industrial hygiene, environmental and analytical chemistry, chemical engineering, hydrogeology, and even behavioral sciences (pertaining to those aspects of human behavior that affect exposures).88 Most toxicologists and epidemiologists have considerable experience in exposure science, as do pharmacologists who study drug kinetics and disposition. Many exposure assessments involve collaborative efforts among members of these various disciplines.
There are currently no certification programs available for exposure scientists, but increasingly exposure science research appears in publications such as Environmental Health Perspectives, Risk Analysis, and the Journal of Exposure Science and Environmental Epidemiology.
Certification programs do exist in occupational exposure science. Qualified industrial hygienists will almost always be certified (CIH). The American Industrial Hygiene Association Journal includes much scholarly work related to exposure science.
88. See, e.g., Allen v. Martin Surfacing, 2009 WL 3461145, 2008 U.S. Dist. LEXIS 111658, 263 F.R.D. 47 (D. Mass. 2008) (industrial hygienist qualified to testify regarding concentration and duration of plaintiffs’ decedent’s exposure to toluene and other chemicals); Buzzerd v. Flagship Carwash of Port St. Lucie, Inc., 669 F. Supp. 2d 514 (M.D. Pa. 2009) (industrial hygienist qualified to opine on carbon monoxide exposure, but his conclusions were not based on reliable methodology).
Appendix A: Presentation of Data—Concentration Units
Choosing the proper units to express concentrations of chemicals in environmental media is crucial for precisely defining exposure. Chemical concentrations in environmental media usually are reported in one of two forms: as numeric ratios, such as parts per million or billion (ppm and ppb, respectively), or as unit weight of the chemical per weight or volume of environmental media, such as milligrams per kilogram (mg/kg) or milligrams per cubic meter (mg/m3). Although concentrations expressed as parts per million or parts per billion are easier for some people to conceptualize, their use assumes that media are always sampled at standard temperature and pressure (25°C and 760 torr, respectively). Consequently, scientists prefer to express chemical concentrations as weight of chemical per unit weight or volume of media. This method also makes conversions to dose equivalents, usually expressed in terms of weight of chemical per unit body weight (mg/kg bw), more convenient.
To permit the presentation of results without excessive zeroes before or after the decimal point, appropriate units are needed. The choice of units depends on both the medium in which the chemical resides and the amount of chemical measured. For example, if 50 nanograms of chemical were found in 1 L of water, the appropriate units would be ng/L, rather than 0.00005 mg/L. If 50 grams were found instead, the appropriate units would be 50,000 mg/L, because milligrams are generally the largest units used to express the mass of a chemical in media (Table 1).
Table 1. Weight of Chemical per Unit Weight of Medium
| Preferred Unit | Alternative Unit |
| mg/kg | ppm (parts per million) |
| µg/kg | ppb (parts per billion) |
| ng/kg | ppt (parts per trillion) |
| pg/kg | ppq (parts per quadrillion) |
In water or food, concentration expressed by the preferred unit equals concentration expressed by alternative unit; thus, 2 mg/kg = 2 ppm. One mg (10−3 g) per kg (103 g) equals 1 part per million (10−3/103 = 10−6). Similarly, 1 µg (10−6 g) per kilogram (103 g) equals 1 part per billion (10−6/103 = 10−9), and so on (Table 2).
Note that in air, parts per million and parts per billion have different meanings than they do in water or food; to avoid confusion, it is always preferrable to express air concentrations in weight of chemical per unit volume (rather than weight) of air (usually cubic meters, m3).
Appendix B: Hazardous Waste Site Exposure Assessment
Several principles of exposure assessment can be illustrated by examining the steps taken to evaluate a hazardous waste disposal site. From 1964 to 1972, more than 300,000 55-gallon drums of solid and liquid pesticide production wastes were buried in shallow trenches at a hazardous waste disposal site in Hardeman County, Tennessee. As early as 1965, county engineers had raised concerns that these operations might have affected the aquifer supplying drinking water to the City of Memphis, Tennessee. The State of Tennessee ordered the landfill to stop accepting hazardous waste in 1972; all operations were reported to have ceased by 1975. Testing in 1978 confirmed the presence of toxic chemicals in domestic wells, and by January 1979 all uses of the contaminated well water had been discontinued.
Among the chemicals of concern detected in the ground water were benzene, carbon tetrachloride, chlordane, chlorobenzene, chloroform, and several other pesticides or chemicals associated with pesticide production. As is often the case for ground water polluted by landfills, the observed concentrations fluctuated over a relatively wide range. For example, in a domestic well approximately 1500 feet north of the landfill, carbon tetrachloride concentrations ranged from 10 ppm to 20 ppm between November 1978 and November 1979; from May 1981 to June 1982, carbon tetrachloride levels varied from 18 ppm to 164 ppm.
The chemicals of greatest concern detected during ground-water monitoring near the Hardeman site included carbon tetrachloride, chloroform, and tetrachloroethylene. For each of these three chemicals, the concentrations detected in well water were significantly elevated over levels typically found in potable water. Health surveys conducted in 1978 and 1982 suggested that these chemicals might be causing a variety of health problems in nearby residents.
To confirm the cause-and-effect relationship suggested by the health surveys, an exposure assessment was conducted so that the findings of the health surveys could be compared to adverse health impacts predicted from exposure estimates and toxicological data from laboratory experiments. The exposure assessment for the Hardeman site focused on carbon tetrachloride, because of the high concentrations of this chemical found in the ground water and the severity of the potential health effects associated with exposure to it.
To estimate the range of possible exposures, the Hardeman site assessment considered exposures of both an adult and an infant. The exposure assessor then needed to identify the pathways of exposure that might be important. For the infant, the following exposure pathways were examined:
- Consumption of formula made using well water,
- Dermal absorption during bathing in contaminated water, and
- In-utero exposure of the fetus through exposure of the mother during pregnancy.
Adult exposures were evaluated for two pathways:
- Consumption of contaminated drinking water and
- Inhalation of carbon tetrachloride emanating from water during showers.
Because measurements of concentrations of carbon tetrachloride in the ground water were scant before 1978, estimates were modeled for these years; measured concentrations were used for 1978, the last year residents utilized ground water for drinking. Standard assumptions regarding the ingestion of water by adults (2 L/day) were used; water consumption by a child was assumed to be 0.5 L/day for 3 months following birth. Dermal absorption by infants was estimated by assuming that the child bathed in 30 L/day of well water, that 50% of this volume contacted the skin, and that 10% of the contaminant was absorbed through the skin. Three baths per week were assumed for the first 3 months after birth. In-utero exposure was estimated assuming equal concentrations of carbon tetrachloride in fetal and maternal blood. The concentration of carbon tetrachloride in air during showering was calculated assuming that it would quickly reach equilibrium with carbon tetrachloride in the shower water.
In Table 3, carbon tetrachloride exposure estimates for the infant and adult are compared with the minimum daily exposure producing liver damage in guinea pigs and the lifetime cumulative exposure producing liver cancer in mice. Daily exposure rates were based on a predicted yearly average exposure during the highest year of exposure. Monitoring data indicate that the concentration of carbon tetrachloride in the ground water may have varied by a factor of 10 around the mean. The maximum daily exposure rate may have been considerably higher than the estimates presented in the table, whereas the long-term averages may have been lower.
Table 3. Carbon Tetrachloride Exposure Estimates for Infants and Adults Compared with Minimum Daily Exposure Producing Liver Damage in Guinea Pigs and Lifetime Cumulative Exposure Producing Liver Cancer in Mice
| Daily Dose Rate (mg/kg/day) | |
| Liver damage in guinea pigs | 1.5 |
| Estimated infant exposure | 1.8 |
| Estimated adult exposure | 0.3 |
| Cumulative Dose (mg/kg) | |
| 40% Liver tumors in mice | 1200 |
| Estimated adult exposure | 284 |
absorbed dose. The amount of a substance that actually enters the body following absorption.
absorption. The penetration of a substance through a barrier (e.g., the skin, the gut, or the lungs).
acute exposure. An exposure of short duration and/or rapid onset. An acute toxic effect is one that develops during or shortly after an acute exposure to a toxic substance.
average daily dose (ADD). The average dose received on any given day during a period of exposure, expressed in mg/kg body weight per day. Ordinarily used in assessing noncancer risks.
bioavailability. The rate and extent to which a chemical or chemical breakdown product enters the general circulation, thereby permitting access to the site of toxic action.
body burden. The total amount of a chemical present or stored in the body. In humans, body burden is an important measure of exposure to chemicals that tend to accumulate in fat cells, such as DDT, PCBs, or dioxins.
chronic exposure. A persistent, recurring, or long-term exposure, as distinguished from an acute exposure. Chronic exposure may result in health effects (such as cancer) that are delayed in onset, occurring long after exposure has ceased.
direct exposure. Exposure of a subject who comes into contact with a chemical via the medium in which it was initially released to the environment. Examples include exposures mediated by cosmetics, other consumer products, some food and beverage additives, medical devices, over-the-counter drugs, and single-medium environmental exposures.
dose. The amount of a substance entering a person, usually expressed for chemicals in the form of weight of the substance (generally in milligrams (mg) or micrograms (µg)) per unit of body weight (generally in kilograms (kg)). It is necessary to specify whether the dose referred to is applied or absorbed. The time over which it is received must also be specified. The time of interest is typically 1 day. If the duration of exposure is specified, dose is actually a dose rate and is expressed as mg or µg/kg per day.
dose–response assessment. An analysis of the relationship between the dose administered to a group and the frequency or magnitude of the biological effect (response).
duration of exposure. Toxicologically, there are three categories describing duration of exposure: acute (one time), subchronic (repeated, for a fraction of a lifetime), and chronic (repeated, for nearly a lifetime).
environmental media. Air, water, soils, and food; consumer products may also be considered media. Chemicals may be directly and intentionally introduced into certain media. Others may move from their sources through one or more media before they reach the media with which people have contact.
exposure. The opportunity to receive a dose through direct contact with a chemical or medium containing a chemical. See also direct esposure; indirect exposure.
exposure assessment. The process of describing, for a population at risk, the amounts of chemicals to which individuals are exposed, or the distribution of exposures within a population, or the average exposure over an entire population.
frequency of exposure. The number of times an exposure occurs in a given period; exposure may be continuous, discontinuous but regular (e.g., once daily), or intermittent (e.g., less than daily, with no standard quantitative definition).
indirect exposure. Often defined as an exposure involving multimedia transport of chemicals from source to exposed individual. Examples include exposures to chemicals deposited onto soils from the air, chemicals released into the ground water beneath a hazardous waste site, or consumption of fruits or vegetables with pesticide residues.
intake. The amount of contact with a medium containing a chemical; used for estimating the dose received from a particular medium.
levels. An alternative term for expressing chemical concentration in environmental media. Usually expressed as mass per unit volume or unit weight in the medium of interest.
lifetime average daily dose (LADD). Total dose received over a lifetime multiplied by the fraction of lifetime during which exposure occurs, expressed in mg/kg body weight per day. Ordinarily used for assessing cancer risk.
models. Idealized mathematical expressions of the relationship between two or more factors (variables).
pathway. The connected media that transport a chemical from source to populations.
point-of-contact exposures. Exposure expressed as the product of the concentration of the chemical in the medium of exposure and the duration and surface area of contact with the body surface, for example, mg/cm2-hours. Some chemicals do not need to be absorbed into the body but rather produce toxicity directly at the point of contact, for example, the skin, mouth, GI tract, nose, bronchial tubes, or lungs. In such cases, the absorbed dose is not the relevant measure of exposure; rather, it is the amount of toxic chemical coming directly into contact with the body surface.
population at risk. A group of subjects with the opportunity to be exposed to a chemical.
risk. The nature and probability of occurrence of an unwanted, adverse effect on human life or health or on the environment.
risk assessment. Characterization of the potential adverse effects on human life or health or on the environment. According to the National Research Council’s Committee on the Institutional Means for Assessment of Health Risk, human health risk assessment includes the following: description of the potential adverse health effects based on an evaluation of results of epidemiologic, clinical, toxicological, and environmental research (hazard identification); extrapolation from those results to predict the type and estimate the extent of health effects in humans under given conditions of exposure (dose–response assessment); judgments regarding the number and characteristics of persons exposed at various intensities and durations (exposure assessment); summary judgments on the existence and overall magnitude of the public-health problem; and characterization of the uncertainties inherent in the process of inferring risk (risk characterization).
route of exposure. The way a chemical enters the body after exposure, that is, by ingestion, inhalation, or dermal absorption.
setting. The place or situation in which a person is exposed to the chemical. Setting is often modified by the activity a person is undertaking, for example, occupational or in-home exposures.
source. The activity or entity from which the chemical is released for potential human exposure.
subchronic exposure. An exposure of intermediate duration between acute and chronic.
subject. An exposed individual, whether a human or an exposed animal or organism in the environment. An exposed individual is sometimes also called a receptor.
systemic dose. A dose of a chemical within the body—that is, not localized at the point of contact. Thus, skin irritation caused by contact with a chemical is not a systemic effect, but liver damage due to absorption of the chemical through the skin is. Often referred to as target site dose.
total dose. The doses received by more than one route of exposure are added to yield the total dose.
D.B. Barr, Expanding the Role of Exposure Science in Environmental Health, 16 J. Exposure Sci. Envtl. Epidemiol. 473 (2006).
Exposure Assessment in Occupational and Environmental Epidemiology (M.J. Nieuwenhuiysen ed., 2003).
S. Gad, Regulatory Toxicology (2d ed. 2001). Includes much discussion of pharmaceuticals, food ingredients, and other consumer products.
P. Lioy, Exposure Science: A View of the Past and Milestones for the Future, 118 Envtl. Health Persp. 1081–90 (2010).
U.S. Environmental Protection Agency, Guidelines for Exposure Assessment, Doc. No. EPA/600/Z-92/001 (1992), available at http://cfpub.epa.gov/ncea/cfm/recordisplay.cfm?deid=15263.














































