6
Process for Regularly Updating the Recommendations
In this report, the Committee on Preventive Services for Women identifies a supplemental set of preventive health care services for women that should be considered by the U.S. Department of Health and Human Services (HHS). This task meets the first portion of the committee’s charge, which was to identify services and screenings that could fill the identified gaps in women’s preventive care not otherwise included in existing preventive services covered under the Patient Protection and Affordable Care Act of 2010 (ACA).
The second part of the committee’s charge was to provide guidance on a process for updating the preventive services and screenings to be considered. Developing and maintaining a comprehensive list of covered preventive services for women is not currently under the specific purview of any advisory group, task force, committee, or agency within HHS. Thus, the committee believes that it will be necessary to develop structures, accountability, and processes to ensure that preventive services meeting evidence standards are considered for coverage in the context of the general approach taken to identify and update preventive services for women. Here, the committee recommends a process supported by guiding principles that separates assessment and coverage decisions. The co-mingling of evidence reviews and coverage decision making in one body could result in skewing scientific results and a decrease in transparency in the rationale for the coverage decision. Components for a comprehensive structure are discussed below.
GUIDING PRINCIPLES AND RECOMMENDATIONS
Recommendation 6.1: The committee recommends that the process for updating the preventive services for women covered under the ACA be:
- Independent;
- Free of conflict of interest;
- Evidence-based;
- Gender specific;
- Life-course oriented;
- Transparent;
- Informed by systematic surveillance and monitoring;
- Cognizant of the need to integrate clinical preventive services with effective interventions in public health, the community, the workplace, and the environment; and
- Appropriately resourced to meet its mandate.
A PREVENTIVE SERVICES COVERAGE COMMISSION
The committee notes that coverage decisions must take into consideration a more extensive list of factors—including medicolegal considerations, ethical considerations, patient and provider preferences, cost, and cost-effectiveness—and that these decisions must be made in the context of the coverage decisions made in other clinical domains. Existing evidence review bodies (such as the United States Preventive Services Task Force [USPSTF]) focus on clinical evidence; and other bodies that develop clinical guidelines (professional organizations) do not have the methods, the expertise, or the independence to make coverage recommendations. The committee believes that the review of the evidence and decision making about coverage are two separate activities and that there is value in preserving the separation. Thus, the committee does not recommend adding coverage decision making to the scope of work of existing evidence review bodies or bodies that develop clinical guidelines.
Recommendation 6.2: The committee recommends that the Secretary of HHS establish a commission to recommend coverage of new preventive services for women to be covered under the ACA.
In carrying out its work, the commission should:
- Be independent from bodies conducting evidence reviews, free of conflict of interest, and transparent;
- Set goals for prevention (it may use available HHS reports and products or commission its own at its discretion);
- Design and implement a methodology for making coverage decisions that considers information from bodies that review the available clinical evidence (and other bodies that establish clinical guidelines) and coverage factors (e.g., cost, cost-effectiveness, and legal and ethical factors);
- Conduct horizon scanning or examine priority goals and/or persistent trends relating to women’s health and well-being to identify new information on significant health conditions; preventive interventions; and new evidence on efficacy, effectiveness, periodicity, and safety;
- Focus on the general population but also search for conditions that may differentially affect women and high-risk subpopulations of women;
- Assign topics and set priorities for evidence-based reviews for the bodies reviewing clinical effectiveness;
- Set timetables and processes for updating clinical practice guidelines and coverage recommendations; and
- Submit its coverage recommendations to the Secretary of HHS.
As noted in the guiding principles, suggested priorities are systematic surveillance and monitoring, as well as horizon scanning for new information on significant health conditions, preventive interventions, and new evidence on efficacy, effectiveness, periodicity, and safety. Similarly, setting agendas, timetables, and resources for developing the evidence reviews and guidelines will need to be recommended to the Secretary of HHS. A commission would not conduct its own systematic reviews of clinical effectiveness, relying instead on reviews completed by evidence review bodies under its direction. Recommendations will also need to be made by the commission regarding updates of evidence reviews and coverage decisions. Five years is a common benchmark for reevaluation of clinical practice guidelines and is the benchmark used by the National Guidelines Clearinghouse, but the committee notes that the process of scanning for new developments often uncovers issues that may require updates at other times.
ROLE OF EVIDENCE-BASED REVIEW BODIES
The committee believes that bodies that review the evidence, such as USPSTF, Bright Futures, and the Advisory Committee on Immunization Practices (ACIP), should continue to focus on evidence of efficacy and effectiveness. These bodies have an important role to perform and to contribute to this process in responding to direction from the Secretary of HHS and addressing topics requested. If necessary, systematic reviews will be commissioned, meeting established standards (e.g., the standards outlined in
Finding What Works in Health Care: Standards for Systematic Reviews [IOM, 2011b]). The evidence-review bodies should review the evidence with a primary focus on efficacy and effectiveness and develop clinical practice guidelines meeting established standards (e.g., the standards outlined in Clinical Practice Guidelines We Can Trust [IOM, 2011a]).
If the Secretary of HHS determines that existing evidence-review bodies cannot support these activities, new bodies that review the evidence should be created. Such bodies would best be populated with experts from within and outside government who are free of conflicts of interest and who represent a wide range of health and related disciplines. These experts should use standard, transparent, and accountable approaches to identify, assess, and synthesize the relevant evidence.
Recommendation 6.3. The committee recommends that the Secretary of HHS identify existing bodies or appoint new ones as needed to review the evidence and develop clinical practice guidelines to be reviewed by a preventive services coverage commission.
Bringing coverage for clinical preventive health care services into rational alignment with coverage for other health care services provided under the ACA will be a major task. The committee notes that many of the individual components are already managed within HHS but currently lack effective coordination for the purposes outlined in the ACA and that some functions are entirely new. The structure might be effectively built over time by using some current bodies and adding new ones as resources permit. The committee does not believe that it has enough information to specifically recommend which unit in HHS should implement the recommendations. Figure 6-1 illustrates the committee’s suggested structure for updating preventive services under the ACA.
Additionally, the 2011 Institute of Medicine (IOM) study Finding What Works in Health Care: Standards for Systematic Reviews examines different grading systems in use. One review mentioned in the study found that there were more than 50 evidence-grading systems and 230 quality assessment instruments in current use. The variation, complexity, and lack of transparency in existing systems were identified (IOM, 2011b). In light of this, the Preventive Services for Women Committee chose not to identify a recommendation for HHS to consider for use in grading evidence. However, many of these models may warrant consideration.
The committee is aware that the IOM Determination of Essential Health Benefits Committee is developing recommendations regarding the criteria and methods for determining and updating the essential health
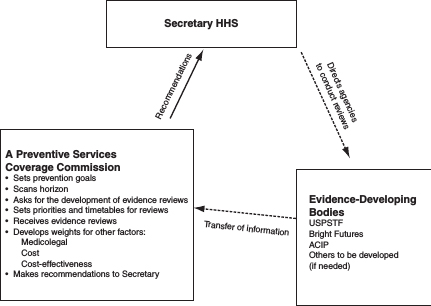
FIGURE 6-1 Suggested structure for updating preventive services under the ACA.
benefits package. That committee is reviewing how insurers determine covered benefits and medical necessity and will provide guidance on the policy principles and criteria for the Secretary to take into account when examining qualified health plans for appropriate balance among categories of care and limits on patient cost sharing. The committee’s recommendations are forthcoming.
Although the ACA’s preventive coverage rules are clearly directed at clinical services, the committee recognizes that in view of the critical importance of community-based preventive services and the public health system in achieving clinical aims, the committee thus encourages the Secretary to consider widening the scope of authority to include public health efforts to more comprehensively address prevention (e.g., as discussed in Healthy People 2020: Topics & Objectives [HHS, 2011]). It will be critical for the proposed preventive services coverage commission to coordinate with the new and existing bodies that are involved with other elements of the ACA.
Finally, the committee notes that it would make the most sense to consider preventive services for women, men, children, and adolescents in the same way. Thus, although the committee’s recommendations presented here address women’s preventive services, the process could be equally useful for
determining preventive services for men, children, and male adolescents that should be covered by the ACA.
HHS (U.S. Department of Health and Human Services). 2011. Healthy People 2020: Topics & objectives. http://www.healthypeople.gov/2020/topicsobjectives2020/default.aspx (accessed April 19, 2011).
IOM. 2011a. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
IOM. 2011b. Finding what works in health care: Standards for systematic reviews. Washington, DC: The National Academies Press.






