Insurer Decisions of Benefit Coverage and Medical Necessity
The Institute of Medicine (IOM) committee is tasked with considering how both public and private insurers choose benefits, develop clinical policies, and make medical necessity determinations. Dr. Louis Jacques began the panel discussion by providing lessons gleaned from Medicare’s process for deciding what is covered. Dr. Jeffrey Kang then described how CIGNA develops a benefit plan and how the benefits offered by a typical CIGNA employer plan compare to the categories of care listed in Section 1302 in the Patient Protection and Affordable Care Act (ACA). He then proposed an approach to developing essential health benefits (EHB). Dr. Virginia Calega, speaking on behalf of the Blue Cross and Blue Shield Association (BCBSA), expanded on the factors insurers take into account in choosing covered benefits and promoting evidence-based practices. Dr. Robert McDonough addressed Aetna’s process for evaluating medical technologies and defining clinical policies. Ms. Carmella Bocchino of America’s Health Insurance Plans (AHIP) concluded the panel by emphasizing the need to balance the scope of benefits with the affordability of premiums and to offer consumer choice among a variety of health plan options.
PRESENTATION BY DR. LOUIS JACQUES, CENTERS FOR MEDICARE & MEDICAID SERVICES (CMS)
Dr. Jacques, director of the Coverage & Analysis Group at CMS, called the task of determining the scope of the EHB “extraordinarily cumbersome,” and advised the committee that it will “be beat on by everybody.” Presuming public health is important in designing the EHB package, he said, the committee’s high-level priorities “aren’t going to be remarkably different from” the priorities used by federal agencies or private plans. To assist the committee in its process, he began by providing a set of considerations that Medicare uses when designing its benefit plan:
Evidence-based care. Current incentives in the health care system—whether related to physician, patient, hospital, or manufacturer behavior—are not necessarily aligned with evidence-based practice, Dr. Jacques said. To what extent will science matter? How much evidence do you need? What kind of evidence is needed? Dr. Jacques advised the committee that whatever it decides regarding these questions, “it is better to be forthright, upfront, with whatever you design.”
Innovation. Do you want to incent medical technology innovation and support beneficiary participation in clinical trials? The realities of insurance mean that plans “pay for whatever physicians and other providers
choose to submit bills for.” This is very different, Dr. Jacques said, from prioritizing health care technologies that are of the most public benefit.
Precedents. Do you want to rely on the rulings or regulations of federal agencies as benchmarks for EHB coverage policy? The committee could, for instance, say that EHB are directly tied to U.S. Food and Drug Administration (FDA) approval, or the withdrawal of FDA approval, of a particular drug. He cautioned the committee, though, to consider that because the Department of Defense (DOD) TRICARE, U.S. Deparment of Veterans Affairs (VA), and Medicare serve special populations (DOD, 2011; VA, 2011), specific covered benefits under their benefit plans may not necessarily align with the needs of the population insured under an EHB package.
Reactivity vs. pro-activity. Will your coverage policies be used for medical review or audit? The committee’s definition of medical necessity, he noted, will impact the ability of individuals to appeal for “individual consideration” of a specific benefit.
Administrative agility vs. formalized consistency. Developing policies that try to anticipate every eventuality may not be feasible, he said. Rather, the system might need to naturally evolve. The committee will need to consider, he said, whether the U.S. Department of Health and Human Services (HHS) wants “pre-written policies established to enhance transparency” or if it wants the capacity to provide individual consideration for coverage decisions. Whatever decisions are made will require balance among different features as outlined in the grid across the four dimensions of being reactive versus proactive and having formalized consistency versus administrative agility (see Figure 6-1).
Dr. Jacques implored the IOM and ultimately HHS to “design a system that keeps in mind a more long-term view.” All of the early decisions will be precedents for what happens later, he said. Therefore, decisions about EHB
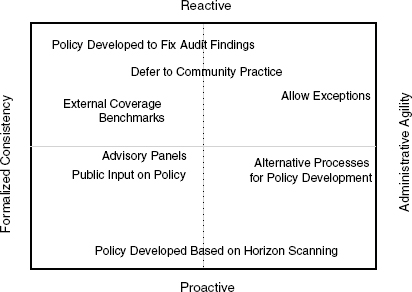
FIGURE 6-1 Defining benefit plans requires balancing administrative agility or consistency with the need to proactively or reactively define benefits. SOURCE: Jacques, 2011.
“must be built on integrity, credibility, and consistency.” While stakeholder groups are influential, he cautioned the committee that they all have their “own particular interests at heart” and that there is a tension between engaging these stakeholders and “abdicating responsibility” to them. It is important, he said, to build common ground around agreed upon principles.
When prompted by committee chair Dr. Ball to explain how CMS accounts for cost in its coverage decisions, Dr. Jacques noted that the standard response of CMS officials is that “Medicare does not consider cost in actually making these decisions.” And this remains true, he said. Hays v. Sebelius,1 for instance, overruled attempts by Medicare or its contractors to implement least costly alternative policies. In Hays, the court ruled that CMS could not reduce the payment amount, even for identical drugs that were packaged differently.
PRESENTATION BY DR. JEFFREY KANG, CIGNA CORPORATION
Dr. Kang, the chief medical officer for CIGNA Corporation discussed how CIGNA “constructs” a benefit plan, how covered benefits interact with medical necessity, and which issues the committee might consider as it debates what is “essential” and what defines a “typical” employer.
Inclusions and Exclusions in Benefit Coverage
Benefit design is a contractual agreement between a plan and a customer that identifies excluded and included services; in the case of included services, each service may be subject to a medical necessity determination to assess appropriateness for an individual patient. The purpose of the medical necessity determination, Dr. Kang said, is not cost, but rather, “to ensure that the services delivered are reasonable, necessary, safe, and effective.” The contractually outlined excluded services may be ruled out regardless of medical need and the availability of a strong supporting scientific evidence base of effectiveness of treatment. An oft-cited example of an exclusion is that Medicare fee-for-service did not cover oral prescription drugs until Medicare Part D was implemented in 2006. This was not because prescription drugs were not medically necessary. Rather, Medicare made a policy decision based on affordability.
The distinction between included and excluded services has implications, Dr. Kang said, for determining the EHB. Employers can purchase buy-ups or riders if they wish to include “typically excluded services.” In the exchanges, individuals or employers could purchase supplemental policies to meet individual lifestyle needs. For example, acupuncture, cosmetic surgery, dental and vision care, infertility care, and experimental or investigational treatments are often excluded services, but could be added through individually purchased riders.
Typically, Dr. Kang said, plan documents also reference cost sharing arrangements (deductibles, co-pays, co-insurance) for each benefit category, and included items might have limits for specific services (e.g., physical therapy might be covered for up to 30 visits per year). Dr. Kang noted that sometimes, certain services are not explicitly included or excluded in the plan document. If a requested service can be reasonably construed to fall within an included benefit category, then coverage determination is based on medical necessity. For example, kidney dialysis is an example of a service that may not be explicitly listed as a covered service within the plan document but falls within a general benefit category, so it is covered if medically necessary.
Benefit limits, Dr. Kang emphasized, are not just “arbitrary numbers”; the limits are based on the plan’s evaluation of population needs. When an employer requests a rehabilitation benefit, for example, CIGNA uses its data to determine the appropriate number of physical therapy visits that are adequate for a restorative benefit. As 90 percent of enrollees “accomplished their restoration” within 30 visits, the number of physical therapy visits is set at 30. If an employer wants a broader benefit that includes maintenance or improvement of function over a person’s baseline, CIGNA works with the employers to price the benefit accordingly.
In response to a query from committee member Dr. Santa, Dr. Kang suggested that denials based on medical necessity are rare within CIGNA. Last year, he said, CIGNA paid for 91 million claims in the United States and approximately 99 percent of those were paid without a denial or required pre-authorization. Approximately .006 percent of total eligible claims were not ultimately approved. Of the initial denials (7,974 cases), 32 percent
____________________
1Hays v. Sebelius, Case number 08-5508, DC Circuit Court of Appeals.
TABLE 6-1 Scope of Included Benefits: ACA vs. CIGNA’s Standard Employer Plan
|
|
|||
| Included in | |||
| Item or Service | ACA-Section 1302(b)(1) | CIGNA Standard Medical Plan | |
|
|
|||
| Ambulatory Patient Services | Included | Included | |
| Emergency Services | Included | Included | |
| Hospitalizations | Included | Included | |
| Maternity and Newborn Care | Included | Included | |
| Mental Health and Substance Use Disorder Services, including | Included | Included | |
| behavioral health treatment | |||
| Prescription Drugs | Included | Included | |
| Rehabilitative Services and Devices | Included | Included | |
| And Habilitative Services and Devices | Included | Excluded | |
| Laboratory Services | Included | Included | |
| Preventive Services | Included | Included | |
| And Wellness Services (Needs Definition) | Included | Buy-Up (separate programs) | |
| And Chronic Disease Management | Included | Included | |
| Pediatric Services (Medical) | Included | Included | |
| Pediatric Services (Oral and Vision Care) | Included | Buy-Up (separate coverage) | |
|
|
|||
| SOURCE: Kang, 2011. | |||
were overturned mostly because of new or additional clinical information made available upon appeal. Less than 0.1 percent were initially denied and after the appeals process was exhausted, about .006 percent of total claims were completely denied.2
Comparing a Standard Employer Plan to ACA Categories of Care
Dr. Kang compared the 10 categories of care listed in Section 1302 of ACA with the CIGNA Standard Medical Plan; to facilitate comparison, he disaggregated the 10 categories into 14 more discrete categories (Table 6-1). Typically, he said, large group (50 or more employees) medical policies exclude habilitative services and devices, without options for buy-up. Pediatric oral and vision care, and what he called the ambiguously defined “wellness services” (perhaps, smoking cessation, weight management programs) are typically excluded from the standard plan but available as buy-ups and purchased as separate products. Dr. Kang noted that the committee will have to “sort through” the conflict between the 10 categories of care required by the ACA and the requirement that the EHB package is similar to a typical employer plan. Approximately 99 percent of CIGNA’s medical coverage business is in the large group market, which Dr. Kang defined as 50 employees or greater. If the benchmark for “typical” is more precisely defined as large employer group plans, the benefits offered under these plans would be “fairly comprehensive and not terribly far apart from the categories” included in Section 1302.
A Proposed Framework for a Tiered Benefit Design
Dr. Kang suggested that the committee consider a theoretical framework that adds more benefits at each actuarial tier.3 For each category of care, Dr. Kang proposed placing services on a continuum from the most essential services (e.g., life preserving services such as hospitalization to treat a heart attack) at the bronze level to services that may be life-enhancing but not essential (e.g., treatment for varicose veins, infertility, prescriptions for Viagra) at the platinum level. Most carriers would put preventive services, for instance, on the life preserving side of the continuum and offer “first dollar coverage” for these preventive services. The middle of the continuum would be
____________________
2 Personal communication with Jeffrey Kang, CIGNA Corporation, February 17, 2011.
3 Section 1302(d)(1)(A)-(D) outlines different levels of coverage that are actuarially differentiated: bronze is actuarially equivalent to 60 percent of the full actuarial value; silver 70 percent, gold 80 percent, platinum 90 percent.
comprised of chronic care management services, among others. For example, a healthy 20-year-old might define the EHB as life-preserving services that offer protection from catastrophic events,4 and would therefore prefer a bronze plan with a high deductible, a limited scope of services, and a lower premium. A chronically ill, disabled individual may, on the other hand, define the EHB as those that span the continuum from life-preserving to life-enhancing; this person, Dr. Kang said, might be willing to pay a higher premium. If each tier has to include the exact same scope of services, individuals would make decisions based solely on the tradeoff between the amount of cost sharing and the premium. For this reason, Dr. Kang argued in favor of including differing scopes for differing tiers.5
The reason, he said, for additional refinement is that the definition of what constitutes essential care currently “depends on the eye of the beholder.” Committee member Ms. Ginsburg asked Dr. Kang who should make the determination of where a particular service is placed on the continuum. This framework, he said, was not based on scientific analyses, but rather, is an illustrative concept based on multiple conversations with clients (employers) and customers (individuals) on what services might be considered essential. When Ms. Ginsburg also noted that including a differential scope of services in the tiered plans could segment the market and “destroy the risk pool,” and did not seem permissible under ACA, Dr. Kang responded that some employers are beginning to offer tiered plans with different premium contributions, and insurers usually offer high/low options. Furthermore, in the individual market, individuals will already self-segment; you are unlikely to find many young people, he said, who will buy the platinum plan. Dr. Kang cautioned the committee to keep in mind that interpretation of the term essential in the individual market is going to vary based on an individual’s circumstances.
Dr. Santa asked whether CIGNA’s current plans offer any benefit categories beyond those listed in Section 1302. Dr. Kang stated that the services included in Section 1302 are open to interpretation but that for larger groups serving more than 50 employees, services that might not be included in the ACA categories include (1) home care—while it could be considered under ambulatory care services, in CIGNA’s benefit language, it is a separate category; (2) skilled nursing facility care—while it could be considered under rehabilitation service, in CIGNA’s benefit language, it is a separate category; and (3) hospice is also a separate category.
Given Dr. Kang’s previous role working for CMS, Dr. Ball asked Dr. Kang to contrast the coverage policy decision making within CMS and CIGNA. Dr. Kang replied that there is actually “very little difference” in how coverage policy is determined, but that there are differences in how the policies get implemented. On the commercial side, for instance, insurers will use prior authorization, but this does not “exist” in Medicare fee-for-service; instead, Medicare uses a post-payment approach for review of appropriateness. Another difference is that because Medicare has a scope of benefits that are legislated, the issue, Dr. Kang said, becomes what services are “reasonable and necessary.” Neither CIGNA nor Medicare uses cost as part of the decision-making process for medical necessity; these determinations, he said, are strictly based on evidence and whether the services have been proven safe and effective. The enforcement of these policies, though, “is completely different.”
PRESENTATION BY DR. VIRGINIA CALEGA, BLUE CROSS AND BLUE SHIELD ASSOCIATION (BCBSA)
Dr. Calega, the Vice President for Medical Management and Policy at Highmark Blue Cross Blue Shield (Highmark), spoke on behalf of the BCBSA. She began by noting that the definition of EHB will “primarily impact individual consumers, small businesses, and the self-employed,” as these are the individuals who are most likely to purchase insurance through the exchanges. Highmark and BCBSA, she said, recommend that the definition of EHB “preserve an insurers’ ability to utilize medical management tools, including medical necessity evaluation.” She advocated for the initial scope of the EHB package to reflect a small business’ typical plan; extensive research has shown that the individual and small group markets are especially sensitive to price as individuals in such plans bear much of the premium costs (Feldman et al., 1997; Gruber and Lettau, 2004; Hadley and Reschovsky, 2002).
____________________
4 There is an option in Section 1302(e) for a catastrophic plan for people under 30 years of age and for those over the age of 30 if they cannot find affordable coverage in the exchange.
5 The law specifies, however, that the essential health benefits must be offered at each metal level, and insurers may offer additional benefits.
For example, research on demand for individual health insurance in California found that consumers were sensitive to price and that this sensitivity is generally higher for younger and lower income individuals (Marquis et al., 2004). Benefit design, Dr. Calega noted, influences and is influenced by the size of the premium. Consequently, Dr. Calega cautioned that the committee should keep in mind that an overly inclusive definition of EHB and one that does not require a sound evidence base could negatively impact affordability for consumers and small employers.
Industry Practices for Benefit Design
Benefit design needs to be “an iterative process” that includes input from various sources to ensure meaningful coverage is available at a variety of price points and that premiums match market demand across multiple market segments. If insurers do not offer a plan and a benefit structure at a price that consumers want, “consumers are not going to pick what is offered,” she said. Highmark, she said, uses focus groups and satisfaction surveys to ensure it knows what consumers want. Insurance brokers and sales representatives also provide critical information on market demand. Highmark and other BCBSA plans have “very strong ties with their communities” as part of their social mission, and to build credibility, they make their policies and decisions transparent and regularly partner with the employers they insure.
In addition to collecting consumer insights, Dr. Calega noted that Highmark partners with health care providers in the development of medical policies to reflect the standards of care. Furthermore, medical management staff keeps abreast of the medical literature to identify new treatments or changes in medical protocols that may necessitate a change in benefits. Plans also conduct internal performance reviews of their plan portfolios; these reviews consider sales data, medical trends by geographic areas, product types, and benefit designs to ensure meaningful coverage at a variety of price points across different markets.
Using Plan Policies to Encourage Evidence-Based Decisions
Dr. Calega observed that plans have clinical policies in place to help guide medical necessity determinations. The goal of these determinations, she said, is to ensure that the patient receives the most appropriate care at the right time in the right setting. As evidenced by the Dartmouth Atlas,6 there is a wide degree of variation in the delivery of care; adherence to evidence-based medicine, she said, will help reduce unnecessary variation and inappropriate care. For example, upon discovering a 25 to 35 percent annual increase in utilization of advanced imaging tests (including variations in prescribing and duplicative tests with the potential exposure of patients to unnecessary radiation), Highmark established an advanced imaging program to better manage the appropriate utilization of these tests. First, a privileging program required providers to meet quality and safety standards in addition to being accredited and licensed. Next, a prior authorization program was implemented with the aim of reducing duplicate tests and enhancing adherence to safety standards (Highmark Blue Cross Blue Shield, 2011). Furthermore, Highmark uses clinical decision-support products (e.g., InterQual®)7 in conjunction with its medical policies; these evidence-based tools guide patients and providers to appropriate treatments.
Committee member Dr. Selby commented that while the “primacy of rigorous evidence is something everyone agrees on,” for some medical care, scientifically validated evidence does not exist. In Dr. Alan Garber’s presentation (see Chapter 5), for instance, Dr. Garber highlighted that the Stanford definition of medical necessity recognized a need for varying levels of evidence for existing vs. new technologies. Dr. Calega acknowledged there are often gaps in available evidence but reiterated that Highmark uses the best available evidence. In the absence of this evidence, “we work with the published clinical literature that we have,” but it is “very difficult” to remove coverage unless the evidence clearly indicates a service is no longer of value. In the case of bone marrow transplant for stage 4 breast cancer, it took 10 years, she said, for evidence to prove it was not of value. Once this evidence was
____________________
6 The Dartmouth Institute for Health Policy and Clinical Practice. 2011. The Dartmouth Atlas of Health Care. http://www.dartmouthatlas.org/ (accessed February 9, 2011).
7 Dr. Calega reports that InterQual® clinical decision support products from McKesson are used by many private insurers, CMS, and military health systems.
available, insurers “pulled back” coverage. “It is critically important,” she said, that the nation devote funding to develop more evidence (e.g., through evidence-based practice centers).
Utilization Management Tools
Dr. Calega observed that Congress explicitly preserved the right of group health plans to employ commonly used management techniques like medical necessity.8 BCBSA and Highmark recommend, she said, that the IOM and HHS do not limit the use of medical necessity or other commonly used medical management tools as part of the administration of EHB. The key reasons for the use of these tools by employers and insurers, including the Federal Employees Health Benefits Program (FEHBP) and Medicare, are to keep coverage affordable while ensuring consumers receive the right care. Medical necessity determination, she emphasized, is a tool that is used after a benefit package is designed.
Dr. Calega gave examples of other utilization management tools that, in addition to medical necessity, should continue to be permissible: (1) coverage of benefits only through licensed providers and facilities within the scope of their license or certification; (2) use of provider networks and cost sharing to direct consumers to those providers that deliver the best value and quality; (3) precertification and prior approval for inpatient hospital admissions except in cases of a medical emergency; (4) precertification for certain outpatient surgeries such as bariatric surgery; and (5) general exclusions for services not medically necessary or appropriate under accepted standards of insurance for medical practice such as for cosmetic services or custodial care.
PRESENTATION BY DR. ROBERT MCDONOUGH, AETNA
Dr. McDonough, the Head of Clinical Policy Research and Development at Aetna addressed the development of clinical policies and patients’ rights to appeal medical necessity determinations. He began by noting that while Aetna had once been unique in making its clinical policies publicly available, most other insurance companies are now doing the same.
Clinical Policy Development Process
Aetna’s clinical policy unit evaluates technologies to determine whether they are experimental and what the medically necessary indications are for the technology. These evaluations and the policies that they influence are publicly available on Aetna’s website.9 The goal of the policies, Dr. McDonough said, is to “develop objective, clinically supported, and defensible determinations.”
Dr. Selby observed that the insurance industry receives considerable criticism and asked how insurers might strengthen their reputation for integrity and credibility; literature and experience suggest that one factor is transparency about processes. Dr. McDonough responded that Aetna has received recognition because of its transparent clinical policies. In the late 1990s, for instance, it was the first commercial insurer to make its policies publicly available on the Internet. Furthermore, legitimacy, he said, can be derived from having outside experts review insurers’ policies for reasonableness.
Criteria and Process for Evaluating Technologies
There are a lot of contextual considerations, Dr. McDonough said, in evaluating a technology, including whether the technology relates to a rare condition, whether it is a last resort treatment, or whether there are other established treatments for the condition. If the technology is of minimal cost, there may not even be an evaluation. The “vast majority of technologies,” Dr. McDonough said, is not selected for evaluation and is not subject to
____________________
8 Patient Protection and Affordable Care Act of 2010 as amended. Public Law 111-148 § 1563(d)(1), 111th Cong., 2d sess.
9Aetna clinical policy bulletins. http://www.aetna.com/healthcare-professionals/policies-guidelines/cpb_alpha.html (accessed February 9, 2011).
BOX 6-1
The Blue Cross and Blue Shield Association’s Technology Evaluation Center
Clinical Coverage Criteria
The following criteria are considered in evaluating a medical technology:
- The technology must have final approval from the appropriate governmental regulatory bodies [when required]
- The scientific evidence must permit conclusions concerning the effect of the technology on health outcomes
- The technology must improve net health outcome
- The technology must be as beneficial as any established alternatives
- The improvement must be attainable outside the investigational settings
SOURCE: BCBSA, 2011.
utilization management (e.g., pre-authorization, limits on visits). His team selects technologies for review based on quantity of use and importance of questions that have arisen regarding the specific technology; the potential impact of the technology on Aetna and its members; the availability of evidence in the peer-reviewed literature, guidelines and consensus statements; changes in regulatory status; or other information that is material to the status of the medical technology.
To evaluate the technology, Aetna has a process in which it looks at evidence in the peer-reviewed literature, the regulatory status, and any relevant clinical practice guidelines and technology assessments. Aetna’s clinical coverage criteria are derived from those of the Blue Cross and Blue Shield Association’s Technology Evaluation Center (Box 6-1). In addition to these criteria, Aetna considers indications in major drug compendia recognized by CMS, the approval status of technologies from relevant government regulatory bodies (e.g., CMS or FDA), and technology assessments from other reliable sources of information such as the California Technology Assessment Forum and the Blue Cross and Blue Shield Association’s Technology Evaluation Center. These assessments, among others, are indexed by Health Technology Assessment International (HTAi).
Each clinical policy bulletin goes through a review and approval process that involves physician advisors, Aetna medical policy and legal departments, and the chief medical officer, who is ultimately responsible for signing off on any of these policies. All policies are reviewed for updating at least annually. Aetna posts its review schedule on the Internet so that the public can comment. Furthermore, Aetna solicits provider input through quality advisory committees, a specialty society liaison group, and a physician advisory group mailbox. Aetna reviews all of the external input to see if patterns might indicate a need to change its policies. After updating, the implementation phase ensures coding practices are aligned; that claims systems are updated to indicate what is covered, not covered, or conditionally covered; and that providers are notified of material changes.10
____________________
10 Aetna has agreed to provide 90-day prior notification to its participating providers of all policy changes that will have a material adverse impact on providers. In addition, some states have requirements for prior notification to providers of certain policy changes. These requirements vary from state to state. Personal communication with Robert McDonough, Aetna, May 10, 2011.
Appeals Process
If an individual disagrees with a coverage determination, the member has access to internal and external appeal and grievance procedures. Dr. McDonough noted that all clinical denials include information about how to file an appeal. All medical necessity, experimental, and cosmetic appeals are reviewed by clinicians, with adverse determinations being reviewed by the medical director. Aetna’s appeals and grievances processes, Dr. McDonough noted, have to comply with U.S. Department of Labor (DOL) regulations (DOL, 2011), National Committee for Quality Assurance (NCQA) standards (NCQA, 2011), and now, an ACA requirement that members have access to an independent external review after exhausting the internal appeals process.
PRESENTATION BY MS. CARMELLA BOCCHINO, AMERICA’S HEALTH INSURANCE PLANS (AHIP)
Ms. Bocchino, Executive Vice President of Clinical Affairs and Strategic Planning at AHIP, began by building on the comments of the previous panelists and explaining that in a commercial market, employers and plans work together to determine which benefits will be offered. While the categories of services listed in the ACA are similar to the care offered by large employer plans (as described by Dr. Kang and shown in Table 6-1), the ACA expanded benefits to include categories that some consumers have purposely decided to forego in the past. In the small group and individual market, for instance, plans are available that do not offer maternity benefits, prescription drugs, or mental health coverage. Consumers, Ms. Bocchino said, “choose to buy products without those services because: a) they do not feel the services meet their individual needs, and b) it helps to keep the premium down.” The ACA has added categories to a minimum standard benefit package and this “appears to be inconsistent with the statement” that the ACA would allow people to “keep the health insurance that they currently have.”
Dr. Selby noted that Representative James Dunnigan testified that most enrollees in Utah opt for something more rich than the minimum benefit plan and suggested that a “too basic” minimum benefit plan or a tiered benefit structure might be particularly disadvantageous to low-income and sick people (see Chapter 4). Ms. Bocchino responded that in the commercial market, plans offer a “range of products with different categories of care and different limitations on those services,” and individuals decide, on an annual basis, the best fit for them.
Ms. Bocchino advised the committee to “be cognizant” of the fact that “the imposition of richer benefit packages will have the effect of raising group employers’ premiums.” She emphasized that the “coupling” of the elimination of lifetime benefits and the inclusion of out-of-pocket maximums indicates “congressional intent was to ensure an adequate level of coverage, that consumers have a range of choices, and that these products are affordable.”
Medical Necessity
Medical necessity reviews, Ms. Bocchino pointed out, are not conducted on most routine services. These reviews “come into play” if questions arise regarding a lack of evidence for such treatment, its clinical effectiveness or potential for harm, or if the patient did not meet the subpopulation characteristics for which such an intervention might be prescribed. In response to an inquiry from the committee about the degree to which different definitions of medical necessity result in differences in coverage, Ms. Bocchino briefly referenced a Connecticut court settlement that defined a medical necessity determination framework (see Chapter 5 for further discussion of medical necessity) (Kaminski, 2007). This framework, she said, is used throughout the industry. Furthermore, medical necessity is subject to oversight from state regulators and employers; and, in the case of the plans participating in the FEHBP, the U.S. Office of Personnel Management (OPM) reviews and approves the medical necessity provisions used by plans. Dr. Calega supported Ms. Bocchino by noting that Congress did not call for a definition of medical necessity in the ACA.
State Mandates
The ACA contains a provision allowing states to require a qualified health plan in the exchange to offer benefits beyond the defined set of essential health benefits if the state is willing to assume the associated costs.11 Ms. Bocchino asserted that most state mandates have been enacted without an assessment of scientific evidence. It will be “almost impossible,” to include a large number of mandates in the EHB package or require individuals, small businesses, or states that do not currently have these mandates to incur the added cost.
Few states have rigorous reviews like the California Health Benefits Review Program (CHBRP), which evaluates benefit changes proposed by the California legislature before they are mandated. CHBRP assesses the existing scientific evidence related to the proposed benefit and prepares an independent analysis of its medical, financial, and public health impact (Note: this process is further detailed in Chapter 12).
Appeal Processes
Health plans, Ms. Bocchino said, “fully support a fair, robust, and timely process for consumers to appeal benefit denials through external review administered by independent third-party review organizations” as required under the ACA. She urged the committee to review the interim final rule (U.S. Department of the Treasury et al., 2010). The committee’s work, she said, should stay consistent with the direction of that regulation.
Principles for Defining and Updating the EHB
Ms. Bocchino outlined principles she believes should drive the definition of the EHB:
- Provide access for consumers to the appropriate care at the right time and in the right setting.
- Ensure that the processes, principles, or criteria are rigorously evidence-based and free from political influence.
- Consider cost-effectiveness, quality, and appropriateness. The process should consider the findings of comparative effectiveness research (CER), including the work of the Patient-Centered Outcomes Research Institute (PCORI).
- Focus definition on the categories of service already established in the ACA, rather than being “too specific” about covered services, which could risk undermining affordability.
- Establish a consistent decision-making process that includes independent analysis and explicit guidelines that consider medical efficacy, and social and financial impacts.
- Balance the comprehensiveness of benefits against ensuring that coverage is affordable. Consumers should have a choice of products that meet their individual needs or desires.
- Minimum thresholds should allow for innovation in benefit design and meaningful differentiation to provide consumer choice across the bronze, silver, gold, and platinum plans.
- Survey the marketplace to further understand what is equal in scope under a typical employer plan (beyond the ACA requirement that the DOL survey employer plans). Initially, this survey should focus on the scope of benefits offered by smaller companies to more accurately represent the needs of the exchange-based market.
- Evaluate all benefits on a de novo basis rather than with the frequency and process used to evaluate state benefit mandates.
The process for defining EHB, Ms. Bocchino said, should focus on the degree of specificity included in the Massachusetts exchange and FEHBP program. These benefit packages only specify general categories of service and not the number or frequency of services covered. FEHBP provides guidance relative to the categories of services, but allows individual plans, when they bid competitively, to further define how frequently those services
____________________
11 Patient Protection and Affordable Care Act of 2010 as amended. Public Law 111-148 § 1311(d)(3)(B)(ii), 111th Cong., 2d sess.
will be provided and if there are any limitations on those services. This flexibility, Ms. Bocchino argued, allows for a more competitive market for consumers to make decisions.
BCBSA (Blue Cross and Blue Shield Association). 2011. Technology Evaluation Center criteria. http://www.bcbs.com/blueresources/tec/tec-criteria.html (accessed June 15, 2011).
DOD (Department of Defense). 2011. TRICARE: Covered services. http://www.tricare.mil/mybenefit/ProfileFilter.do;jsessionid=N3fchlV4wPSRTlrf6slc4zFpNXLXpf90q8gxnllF8fK6y3dKBGgJ!-1918827786?p=Covered_Services&puri=%2Fhome%2FMedical%2FIsItCovered.jsp (accessed June 15, 2011).
DOL (Department of Labor). 2011. Compliance assistance group health and disability plans benefit claims procedure regulation (29 CFR 2560.503-1). http://www.dol.gov/ebsa/pdf/caghdp.pdf (accessed May 11, 2011).
Feldman, R., B. Dowd, S. Leitz, and L. A. Blewett. 1997. The effect of premiums on the small firm’s decision to offer health insurance. Journal of Human Resources 32:635-658.
Gruber, J., and M. Lettau. 2004. How elastic is the firm’s demand for health insurance. Journal of Public Economics 88:1273-1293.
Hadley, J., and J. D. Reschovsky. 2002. Small firms’ demand for health insurance: The decision to offer insurance. Inquiry 39(2):118-137.
Highmark Blue Cross Blue Shield. 2011. Provider Resource Center: Highmark Radiology Management Program. http://prc.highmarkblueshield.com/rscprc/faces/prcMainPage.jsp (accessed April 19, 2011).
Jacques, L. 2011. Coverage policy: Aligning an insurance benefit with evolving evidence. PowerPoint Presentation to the IOM Committee on the Determination of Essential Health Benefits by Louis Jacques, Director of Coverage and Analysis Group, Centers for Medicare & Medicaid Services, January 13, 2011, Washington, DC.
Kaminiski, J. L. 2007. Defining medical necessity. http://www.cga.ct.gov/2007/rpt/2007-r-0055.htm (accessed April 20, 2011).
Kang, J. 2011. Statement by Jeffrey Kang, Chief Medical Officer, CIGNA to the IOM Committee on the Determination of Essential Health Benefits, Washington, DC, January 13.
Marquis, M. S., M. B. Buntin, J. J. Escarce, K. Kapur, and J. M. Yegian. 2004. Subsidies and the demand for individual health insurance in California. Health Services Research 39(5):1547-1570.
NCQA (National Committee for Quality Assurance). 2011. Consumer protections in health plan accreditation. http://www.ncqa.org/Portals/0/Public%20Policy/Strong%20consumer%20protection%20is%20a%20cornerstone%20of%20NCQA.pdf (accessed May 11, 2011).
U.S. Department of the Treasury, DOL, and HHS (Department of Health and Human Services). 2010. Interim final rules for group health plans and health insurance issuers relating to internal claims and appeals and external review processes under the Patient Protection and Affordable Care Act; interim final rule. Federal Register 75(141):43330-43364.
VA (Department of Veterans Affairs). 2011. VA health care eligibility & enrollment: medical benefits package (standard benefits). http://www.va.gov/healtheligibility/coveredservices/standardbenefits.asp (accessed June 14, 2011).












