___________
Approaches to Determining Covered
Benefits and Benefit Design
This chapter reviews approaches to deciding benefit coverage. Insurance terms such as covered benefit, benefit design, utilization management, and medical management are defined and related to the scope of the committee’s task. A discussion of the shared responsibility for achieving the goals of ensuring that care is safe, effective, and appropriate, while using resources wisely, sets the stage for a brief overview of the main cost drivers for health care spending and insurance premium growth. Various approaches to insurance design (e.g., use of evidence, prioritization, value-based insurance design) are noted with illustrative applications.
This chapter provides background information on how insurers and employers make decisions about benefit coverage and clinical policies, and highlights several illustrative approaches to making decisions. Key to understanding what constitutes the essential health benefits (EHB) package are the complementary concepts of covered benefits and benefit design. These as well as other insurance terms are defined. Goals for coverage determination and medical necessity decisions are for safe, effective, and appropriate care while using pooled resources wisely. The committee comments on the shared responsibility among insurers, providers of care, and patients for the effective stewardship of shared, limited resources. Finally, the committee highlights some specific approaches to decision making: use of evidence, prioritization practices, and innovation through value-based design. This background material helped to inform the committee’s development of policy foundations and criteria in the next chapter.
The committee’s charge is to advise on the definition and updating of the essential health benefits, and the committee noted that this task incorporates three aspects: the content of the covered benefits, the elements of benefit design, and the administration of those benefits. The committee defines those terms among others (see Box 2-1) and, given the legislative guidance for a typical employer plan, examines how employers and insurers approach development of a benefit package.
Choosing a Policy
When employers offer health insurance coverage to their employees, they begin by outlining to an insurer, insurance broker, or benefit consultant the scope of benefits that they would like to offer. Insurers have standard
plans, which they can customize to employer needs and the requirements of specific markets (e.g., state mandates for inclusion of specific benefits). The benefit package has to meet what the employer considers an affordable premium for the company and the employees. The overall premium will reflect which services are included, included but with limits, or excluded; deductible and co-payment levels or co-insurance requirements; the network of providers; the insured group’s risk profile; the degree of medical management in the policy; and health plan administrative expenses, overhead, and profits. A plan with more excluded services, a narrower network of providers, more restrictive visit limits, and higher employee deductible and cost sharing will tend to have a less expensive premium for a comparable set of covered benefits.
BOX 2-1
Understanding Basic Terms Used in This Chapter and Report
Actuarial value. The percentage of charges paid by a health plan, calculated using the medical claims from a standard population, along with a plan’s cost-sharing provisions (McDevitt, 2008).
Benefit administration. The insurers’ application of the benefit design outlined in the subscriber contract for its subscribers. Elements of benefit administration include subscriber enrollment and disenrollment, processing of claims, making medical necessity decisions, processing appeals of coverage determinations, and application of any federal or state mandates.
Benefit design. Rules governing the terms under which medical care items or services obtained by subscribers are considered covered benefits. Examples include the expected amount of enrollee or member payments for deductibles and other co-payments or co-insurance, the network of providers a subscriber may see, and the and extent of medical management (e.g., prior authorization or primary care physician referral requirements).
Categories of care. Ten categories listed in Section 1302 of the Patient Protection and Affordable
Care Act (ACA): ambulatory patient services; emergency services; hospitalization; maternity and newborn care; mental health and substance use disorder services, including behavioral health treatment; prescription drugs; rehabilitative and habilitative services and devices; laboratory services; preventive and wellness services and chronic disease management; and pediatric services, including oral and vision care.
Coverage determination. The decision on whether a medical care item or service obtained by a subscriber is a covered benefit. Coverage determinations may be appealed by the subscriber under rules set forth by state law and ACA.
Covered benefits. The medical care items or services obtained by a subscriber that a health insurance plan agrees to pay for, under certain terms and limitations. Covered benefits and excluded services, and the terms and limitations of coverage, are defined in the health insurance plan’s coverage documents or the subscriber contract.
Deductible. The amount you must pay for covered care before your health insurance begins to pay. Insurers apply and structure deductibles differently.
Exclusions. Lists of specific medical items or services or general circumstance (e.g., not medically necessary) in a subscriber contract that are not covered benefits.
Health insurance. A method of pooling risk of financial loss across a group or population, in which a contract or policy under which a third party (i.e., an insurer) agrees to assume the financial risk for the costs of a set of services defined in the contract in return for a premium.
Inclusions. Lists of specific covered benefits in subscriber contracts.
Medical management systems. Systems designed to ensure that members receive appropriate [covered] health care services. Medical management systems include, but are not limited to, utilization management, quality improvement, case management, and complaint resolution (NCQA, 2007).*
Medical necessity determination. A specific type of coverage determination about whether a medical item or service, which is a covered benefit, is medically necessary for an individual patient’s circumstances, and thus a covered benefit. Typically, this determination is made by the insurer.
Medically necessary. A condition of benefit coverage frequently found in subscriber contracts. Under the terms of most subscriber contracts, the receipt of a medical care item or service does not in and of itself indicate that the item or service was medically necessary (see Chapter 5 for additional discussion).
Out-of-Pocket Cost. Your expenses for medical care that are not reimbursed by insurance. These include: deductibles, co-insurance, and co-payments for covered services in addition to all costs for non-covered services.
Premium. The dollar amount paid for an insurance policy. Premiums can be paid by employers, unions, employees or individuals or shared among different payers.
Qualified health plan. Under the ACA, starting in 2014, an insurance plan that is certified by an exchange, provides essential health benefits, follows established limits on cost sharing (e.g., deductibles, co-payments, out-of-pocket maximum amounts), and meets other requirements. A qualified health plan will have a certification by each exchange in which it is sold (HHS, 2010b).
Subscriber. A person and his or her dependents for which a premium has been paid to a health insurer. Also called an enrollee.
Utilization management. The process of evaluating and determining coverage for and appropriateness of medical care services, as well as providing needed assistance to clinician or patient, in cooperation with other parties, to ensure appropriate use of resources (NCQA, 2007).
Utilization review. A formal evaluation (preservice, concurrent, or post-service) of the coverage, medical necessity, efficiency, or appropriateness of health care services and treatment plans (NCQA, 2007). Terms such as retrospective and prospective review are often used.
___________
* [covered] added for clarification to the National Committee for Quality Assurance (NCQA) definition.
What Does It Mean?
Covered Benefits
The scope of covered benefits is outlined in an insurance contract or explanation of coverage document (alternately, called evidence of coverage or summary of benefits). Documents differ in the level of detail used to describe the services and items that are covered and may include general categories of care, specific items and services, or circumstances under which benefits are included or expressly excluded. One usual contract exclusion is of any service not considered medically necessary by the payer. Section 1302 of the Patient Protection and Affordable Care Act (ACA) guides the Secretary of the Department of Health and Human Services (HHS) to define the EHB to include at least 10 specific categories of care (Box 2-1) and to be equal to the scope of benefits provided under a typical employer plan. Although this Institute of Medicine (IOM) committee is not requested to specify the detailed inclusions and exclusions in the EHB package, the committee examined legislative guidance for different programs and numerous sample plan documents to learn about the level of detail in them and the implications these might hold for secretarial guidance on the EHB (see Chapters 4 and 5).
Benefit Design
Benefit design sets out the parameters by which patients can obtain services and their financial liability for deductibles and co-payments or co-insurance. The ACA eliminates some past benefit design options, specifically the use of annual and lifetime dollar limits; puts in place some boundaries on how high deductibles and cost sharing can be; requires that provider networks be adequate; and requires that the design be nondiscriminatory. The ACA permits the use of other benefit design options, specifically utilization management techniques commonly employed as of the date of passage of the act.1 These practices help hold down premiums, as do choices of higher levels of deductibles and cost sharing.
Research on insurance-induced use shows that covered services will be provided at higher rates than those that are not covered (Card et al., 2008, 2009; Dafny and Gruber, 2005); this can result in improved access to care as well as the potential increased utilization of unnecessary services. As a result, for example, insurers may put prior authorization requirements or limits on the number of visits for certain services (Flynn et al., 2002; Wickizer and Lessler, 2002). By the same token, services that can potentially lead to reduced costs as well as better patient outcomes, such as some preventive care and early interventions, lead some plans to adopt medical management programs to encourage use of these services (e.g., some value-based insurance design plans).
Benefit design will have a significant impact on what can be included in the EHB package at a given premium level, so the committee found that benefit design was not readily separable from the contents of the benefits and considered it within the scope of study. Indeed, Dr. Sherry Glied, Assistant Secretary for Planning and Evaluation (ASPE), in her presentation before the committee said that when the Chief Actuary of the Centers for Medicare & Medicaid Services (CMS) certifies the EHB package, the actuary will look at both the content of benefits and the benefit design “in practice in the world and make estimates on that basis” (Glied, 2011).
Most readers will have some general understanding, through their own experience with health insurance, of the basic terms in Box 2-1, but they are likely less familiar with the terms—actuarial estimate, actuarial value, and actuarial equivalence. Understanding the difference in the meaning of these terms and how they relate to benefit design will become important to understanding some aspects of the committee’s recommendations:
• Actuarial estimates project the expected cost of each individual benefit category or service for a standard population. Knowing these expected costs, an insurer can estimate the impact on premium. Then purchasers—whether individuals, employers, state governments, or the federal process of defining the EHB—can look at the actuarial estimate for certain benefits to determine if they are a priority for inclusion for the premium price that will need to be paid. As noted above, employers might start by looking at an insurer’s standard or typical plan offering and then decide to customize by adding or subtracting benefits, and/or by applying benefit design choices such as requiring more stringent limits rather than cut a benefit category out (e.g., limit to 10 versus 20 physical therapy visits).
• Actuarial value is the percentage of covered expenses that a plan is likely to pay on average for a standard population rather than being paid out-of-pocket by the consumer. In the health insurance exchanges, a range of plans will be offered, and the ACA provides a way to ease comparisons among them—the use of Actuarial Value2 (AV) to provide a sense of the relative protection offered by plans. Four tiers or levels of coverage are differentiated in the ACA based on a specified actuarial value percentage: platinum: 90 percent, gold: 80 percent, silver: 70 percent, and bronze: 60 percent. Plans at each actuarial level will
1 Patient Protection and Affordable Care Act of 2010 as amended. § 1563(d)(1), 111th Cong., 2d sess.
2 § 1302(d)(1)(A)-(D).
contain the EHB, but the plans will be differentiated by benefit design choices. Insurers are not required to offer all four “metal” levels, but are required to offer at least one plan at each of the silver and gold levels of coverage (excluding dental-only plans).3 Under the ACA, premium subsidies for low-income individuals are linked to the second lowest priced silver plan available in their exchange. Thus, the silver plan is likely to be the dominant plan sold. To re-emphasize, the actuarial value is an average. Within any insured population group, there will individuals, in for example the silver plan (on average 70 percent actuarial value) whose percentage of return could be from 0 to 100 percent. For example, a person who never spends more than is required by his or her deductible will have a low percentage return, while a very sick person who uses many services will have a higher return rate.
• Actuarial equivalence is a not a concept for consumers to use in deciding which plan to buy even though “actuarial equivalence calculations provide a means to compare the relative generosity of different benefit packages” (AAA, 2009). Furthermore, plans can be actuarially equivalent and still have different premiums because premiums will take into account the health status and utilization patterns of the local population to be enrolled, payments negotiated with providers, breadth of provider network, various degrees of medical management, administrative costs and company profits. The concept of actuarial equivalence, however, will be useful for the CMS Actuary to apply to determine if the EHB are equal in scope to the typical employer plan, and for the Secretary to determine if any state-specified package is equivalent to the nationally defined EHB package. Actuarial equivalence calculations generally consider covered benefits, cost-sharing requirements (deductibles, co-insurance, co-payments [including by service type], out-of-pocket cost limits, and benefit limits) as applicable to an in-network benefit level, but the committee’s interest is mainly in benefit equivalence when comparing the EHB to the scope of typical employers (Chapter 5) and possible state variations to the national EHB.
The committee offers an example of the type of benefit design data that would inform the monitoring and updating of the EHB as well as provide more detail to inform consumers about their plans. Chapter 7 further discusses the need for data collection such as in Box 2-2.
Benefit design and its subsequent administration can be instrumental in addressing the cost and quality of services and care delivered. Insurers and employers are experimenting with an array of medical management and cost-sharing designs (e.g., value-based insurance design). The committee believes the intent of the ACA was to view utilization management (UM) in a broader medical management context to ensure appropriateness and quality and not simply to limit access to care. The National Committee for Quality Assurance (NCQA) defines UM as a “process of evaluating and determining coverage for and appropriateness of medical care services, as well as providing needed assistance to clinician or patient, in cooperation with other parties, to ensure appropriate use of resources” (NCQA, 2007, p. 364). URAC, another accreditation body, uses a similar definition and cites the continued need for traditional utilization management techniques (e.g., pre-certification, concurrent review) as tools for controlling costs (URAC, 2011).
About 15 years ago, Milstein wrote, “UM and the reduced volume of health care services it typically fosters have struck a nerve” feeling that it “may be jeopardizing patients’ well-being” (Milstein, 1997, p. 87). He called for better evidence-based utilization standards for decision making and accreditation standards and certification for UM programs such as those that URAC and NCQA now provide, among other things. Today, purchasers and insurers, while still using UM often with more sophisticated claims analytics, employ a broader array of techniques called medical management to seek to improve quality and cost of care through designation of high-performing networks, patient and provider supports, and more transparent evidence-based clinical policies (Figure 2-1). However, despite this progress, much work remains to be done to ensure the appropriateness of care.
Benefit Administration
Elements of benefit administration also fall into the committee’s purview because the statement of task specifically calls for the committee to assess implementation-related issues such as medical necessity, safeguards for
3 § 1301(a)(1)(C)(ii).
nondiscrimination (which can entail appeals processes), and making choices that are understandable to consumers. The contents of the EHB and the benefit design limits placed upon them can enhance or impede access to care, as well as encourage or discourage enrollment in a plan. Chapter 7 also speaks to assessment of the impact of the EHB on implementation.
BOX 2-2
Description of Benefit Design
Benefit design includes
1. A description of the covered benefits: services, drugs, devices
a. Identification of those covered services, drugs, and devices that are variably covered (tiering)
b. Identification of those covered services, drugs, and devices that are limited in quantity, frequency, or some other way
2. A description of the cost-sharing process
a. Specific definition of and dollar amounts related to deductibles, co-payment, co-insurance, and out-of-pocket maximum
b. Specific identification of any covered services, drugs, or devices having no cost sharing
c. Specific definition of any covered services, drugs, or devices in which cost sharing does not accrue to the out-of-pocket maximum
d. Specific definition of those services, drugs, or devices whose cost is not included in the out-of-pocket maximum when they are not covered
e. The actuarial value
3. A list of coverage exclusions
4. Definitions of key terms affecting coverage, including whether the definition is consistent with an external standard
a. Definition of medical service
b. Medical necessity
c. Experimental, investigational
d. Cosmetic
e. Dental
5. Identification of benefit design innovations
a. Value-based insurance designs that align cost sharing with value
6. Identification of provider networks, incentives, and care delivery options
a. Incentives and disincentives for providers at individual and organizational levels
b. Network design: types of networks (e.g., narrow networks, tiered or concentric networks, broad networks) and level of care or site of service for specific procedures or conditions within networks
c. Centers of excellence (without any out-of-network coverage for specific conditions)
d. Identification of delivery arrangements that could affect care
i. Medical homes
ii. Disease management
iii. Care coordination
iv. Specialty referral requirements
7. Identification of approaches designed to influence the use of services, including specific services that need to be authorized prior to provision, to be provided in specific sites (such as surgery in an ambulatory surgery center), or to be provided at a specific level of care (such as “skilled” services in a nursing home)
8. Identification of medical policies that could affect coverage including an explicit statement that these policies may apply to all covered services on an individual patient basis
a. Access to specific medical policies affecting coverage
b. A description of the process for administering these policies including complaint, request for review, and appeal processes
9. Medical management and/or utilization management programs (e.g., when prior authorization is required for specific services; site of service, level of care, or preferred providers)
10. Payment policies that affect coverage or cost sharing.
a. Hold-harmless arrangements
b. Pricing arrangements that may affect cost sharing and out-of-pocket maximums
c. Reference pricing for drugs and medical or surgical services
11. Quality and cost transparency reports on variation by provider, condition, procedure, facility, and geography
12. Overall description of how benefits are administered, including description of the complaint, request for review, and appeals processes
Deciding Covered Benefits vs. Medical Necessity Determination
During a presentation to the committee, Dr. Alan Garber distinguished a coverage decision (i.e., what an insurer decides to offer in a plan as a covered general benefit category or specific service for a particular price) from a medical necessity determination (i.e., whether the care is deemed appropriate for a particular person for a particular condition and circumstance) (Garber, 2011). Although physicians may make an initial recommendation of service for the patient, its necessity is subject to review and approval by the payer. Insurers indicated that these
decisions are based on specific established criteria, such as safety, clinical evidence, relative cost of services with comparable outcomes (Kaminiski, 2007; Singer et al., 1999), and compliance with state or federal laws, as applicable. Before the committee, Dr. Garber stated that if payment changes put more financial risk on the shoulders of providers, then providers “will have more of a stake in ensuring that only effective care and necessary care is delivered, so medical necessity decision making may turn out to play a lesser role.” The nation is, however, “years off from the time when medical necessity decisions will be unnecessary or much less prominent in determining which care is delivered” (Garber, 2011).
From the practitioner and patient perspectives, a significant issue has been that these determinations are opaque and knowledge of decisions overturned is not always accessible, and further that these determinations may reflect a clinical policy that the health insurer feels is most appropriate (i.e., the treatment they feel is most appropriate for a given condition) rather than taking into account the individual patient. Their perspectives are that these determinations are not always evidence-driven, but could also be driven by attempts to raise barriers to payment
for covered care while a medical necessity determination should most importantly take into account the individual patient’s condition and needs. This contributes to the problem patients have in comprehending why some things their physician recommends are not reimbursable. The well-documented public backlash against managed care in the 1990s was driven by patient, provider, and public concern that insurers were not applying their conditions fairly. Speakers before the committee related stories about denials of care, with Anthony Wright, executive director of Health Access California, noting consumers’ fear of the fine print of insurance contracts (Wright, 2011).
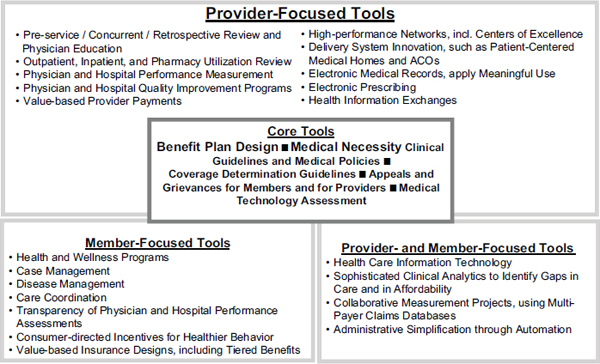
FIGURE 2-1 Illustration of multiple medical management tools used by UnitedHealthcare.
NOTE: ACO = Accountable Care Organization.
SOURCE: Sam Ho, UnitedHealthcare.
The committee concludes that medical management techniques, with appropriate checks and balances, are necessary to ensure that the EHB package can be delivered at the most affordable cost. However, a fair and reasonable appeals process for adverse determinations—including independent medical review will be implemented under the ACA, and the results should be monitored to inform the updating of the EHB.4 The committee reviews insurers’ medical necessity definitions and clinical policy guidelines and advises on possible secretarial guidance in Chapter 5 and the need to monitor appeals in Chapter 7.
UNDERSTANDING CONTRIBUTORS TO COSTS
Goals for coverage decisions and medical necessity determinations are for safe, effective, and appropriate care while using resources wisely. Despite such lofty goals, we know, for example, that unnecessary care is delivered (NEHI, 2008; Schuster et al., 2005), recommended care for adults is delivered just 55 percent of the time (et al., 2003), and some care when delivered inappropriately can actually be unsafe (Brenner and Hall, 2007; Rosen,
4 It is important to note that under the ACA, contested medical necessity determinations can be appealed to an independent external appeals review.
2010; Simpson, 2010). Furthermore, major drivers for health care costs include our growing chronic disease burden and demand for the newest technologies, even if they do not produce greater value than existing less costly technologies. Insurers, providers of care, and patients all have a shared responsibility for appropriate utilization of shared health care resources. Although individuals have the right to spend their own resources as they see fit, spending from a shared pool of funds confers a degree of accountability for effective stewardship of those funds.
Shared Responsibility
Ideally, insurers make their coverage decisions based on evidence, are consistent in the application of evidence, provide meaningful benefits with an acceptable level of risk, and discourage the use of unnecessary or even harmful services. For example, one study suggests that perhaps one-third of computed tomography (CT) scans are unnecessary and could be replaced by alternative approaches or avoided altogether (Brenner and Hall, 2007). CT scans have a radiation dose about 50 times that of conventional X-rays and are implicated in the development of cancers that could have been avoided without this treatment (Brenner and Hall, 2007). On discovering a 25 to 35 percent annual increase in the utilization of advanced imaging tests (including variations in prescribing and duplicative tests with the potential exposure of patients to unnecessary radiation), Highmark Blue Cross Blue Shield established an advanced imaging program to better manage the appropriate utilization of these tests, a privileging program requiring providers to meet quality and safety standards, and a prior authorization program for patients. These were implemented with the aim of reducing duplicate tests and enhancing adherence to safety standards (Highmark Blue Cross Blue Shield, 2011).
Multiple insurers indicated to the committee that they were committed to applying standards of evidence in making benefit coverage decisions and in developing clinical policies (Calega, 2011; Levine, 2011; McDonough, 2011; Nussbaum, 2011). At the same time, they acknowledged that there are often gaps in available evidence. Furthermore, it is “very difficult” to remove coverage unless there is documentation that a service is no longer of value, and there is a need for the nation to devote funding to develop more evidence to support coverage decisions (Calega, 2011). Other examples of inappropriate—not only unnecessary but also potentially harmful—use, cited by others, include inappropriate use of cardiac catheterization (Ko et al., 2010) or single-photon emission computed tomography (SPECT) cardiac scans (Hendel et al., 2010), which themselves not only add unnecessary costs but also can lead to further unnecessary and potentially dangerous surgery for patients; unnecessary diagnostic testing such as $3,000 BRCA-1 genetic tests in patients for whom such testing is not clinically indicated (White et al., 2008); and inappropriate treatment for breast cancer patients (e.g., autologous bone marrow transplant) (Jacobson et al., 2007).
Ideally, physicians also make decisions about necessary care based on evidence; however, there is often significant geographic practice variation (Wennberg, 2011). Unwarranted differences in effective care—that is, “interventions for which the benefits far outweigh the risks”—can reflect underuse of guideline-supported care. Preference-and supply-sensitive care variations reflect differences in professional opinion and response to the capacity of the health care system, resulting in low utilization in some areas and overuse in others. Uncertainty plays a role in variation; when in doubt, there is a tendency to perform a procedure, and the incentives are certainly in place to do so. According to Eddy, “The losers are patients, consumers, and taxpayers—anyone who has to undergo a valueless procedure or pay the bill” (Eddy, 1984, p. 86).
Ideally, patients also make decisions in their own best interests, but this is not always the case. The process for deciding among “preference-sensitive” options must be based on informed understanding of treatment alternatives, an element that is sorely lacking in many situations (Baker et al., 2010). To engage consumers in informed decision making, more insurers and employers are encouraging the use of specialized decisions aids (O’Connor et al., 2007; Stacey et al., 2011).
Evolution of Insurance Coverage and Cost Drivers
The contents of health insurance policies today reflect their historical development, but health insurance is not static and is adapting to today’s health burdens, emerging evidence, and cost drivers in the market. Health insurance grew out of a movement to protect against disability due to accidents or catastrophic illnesses; over time, hospital
pre-payment insurance policies developed and then policies developed to protect against other types of medical expenses (Abraham, 1986; IOM, 1993; Rosenblatt et al, 1997). More recently, coverage for preventive health care services and prescription drugs has emerged—emphasizing ongoing care, not just acute or emergency situations. Indeed, Medicare did not add prescription drug coverage until Part D went into effect in 2006, 41 years after the Medicare program was established.
Major Expenditure Categories
Health care expenditures for persons under 65 years can be categorized into three major expense categories: hospital inpatient, ambulatory care services (e.g., physician office services, hospital outpatient services), and prescription drugs, comprising about 84 percent of health care spending for nonelderly adults and about 70 percent for children (note that this is all spending, not just insurance) (Kashihara and Carper, 2010). Among those under 65 who are privately insured, about 10 percent of the population accounts for 60 percent of all health care spending (Yu and Ezzati-Rice, 2005). The growth in overall inflation-adjusted spending for all ages in these categories over time is shown in Figure 2-2.
Cost Drivers
The major drivers of health care costs are recognized as new technologies, more intensive testing, growing chronic disease burden, increased utilization (growing and aging population), and consumer or provider demand for state-of-the-art care (Table 2-1) (KFF, 2010; PwC Health Research Institute, 2011). Unit price increases for medical care—that is, price per unit of service—are another driver, with one recent estimate attributing 5-19 percent of the annual growth rate between 1960 and 2007 to unit price increases (Smith et al., 2009). Unit prices for a new service vary dramatically by geographic region and volume. Accordingly, all of these cost drivers have implications for the cost of purchasing health insurance as well as benefit design and administration. Health insurance premiums are also affected by federal and state mandates requiring the addition of specific types of benefits; however, the degree of that effect depends on various factors, such as size of the population affected, utilization, and the contents of existing coverage.
Innovation has helped advance medical science and prolong lives, but the Congressional Budget Office (CBO) estimates that the addition of new technologies accounts for about half of the per capita growth in health care spending (Table 2-1) (CBO, 2008). The CBO further notes that some of those advances can be very costly, while others, although relatively inexpensive, can drive up spending because their use becomes widespread, and finally some may reduce aggregate costs (CBO, 2008). A more recent study finds lower estimates of medical technology’s contribution, 27-48 percent of the annual growth rate (average 4.8 percent annually) from 1960 to 2007, and a similar level of 29-43 percent attributable to aggregate income (real per capita gross domestic product [GDP]) (Smith et al., 2009). Emerging technologies such as personalized medicine and biologics will continue to have an impact on spending and insurance premiums. Although there is not a desire to stifle innovation in medical science, rising costs will affect the affordability of coverage in the private sector and in public programs, including subsidization of insurance in the health insurance exchanges. As new technologies become available, important questions are not only whether something is safe and effective, but also whether it will provide benefits beyond comparable treatments and which patients will benefit. Another question to consider will be how best to stimulate the development and dissemination of technologies that may reduce costs.
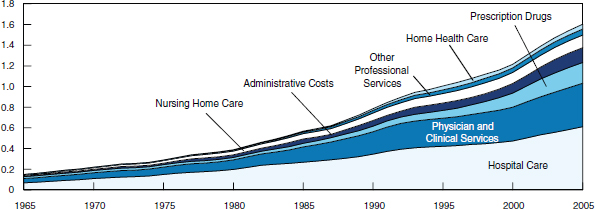
FIGURE 2-2 Real spending on health care in selected categories, 1965-2005.
NOTE: Spending amounts are adjusted for inflation using the gross domestic product implicit price deflator from the Bureau of Economic Analysis.
SOURCE: CBO, 2008.
|
|
|||
| Percentage | Smith et al. (2000)a | Cutler (1995)b | Newhouse (1992)c |
|
|
|||
| Aging of the population | 2 | 2 | 2d |
| Changes in third-party payment | 10 | 13 | 10e |
| Personal income growth | 11-18 | 5 | <23 |
| Prices in the health care sector | 11-22 | 19 | * |
| Administrative costs | 3-10 | 13 | * |
| Defensive medicine and supplier-induced demand | 0 | * | 0 |
| Technology-related changes in medical practice | 38-62 | 49 | >65 |
|
|
|||
NOTE: Amounts in the table represent the estimated percentage share of long term growth that each factor accounts for. < = less than; > = greater than; * = not estimated.
a Congressional Budget Office based on Sheila D. Smith, Stephen K. Heffler, and Mark S. Freeland, 2000, “The Impact of Technological Change on Health Care Cost Increases: An Evaluation of the Literature” (working paper).
b David M. Cutler, 1995, “Technology, Health Costs, and the NIH” (paper prepared for the National Institutes of Health Economics Roundtable on Biomedical Research, September 1995).
c Joseph P. Newhouse, 1992, Medical Care Costs: How Much Welfare Loss? Journal of Economic Perspectives 6(3)Summer:3-21.
d Represents data for 1950 to 1987.
e Represents data for 1950 to 1980.
SOURCE: CBO, 2008.
The huge but often preventable chronic disease burden in this country calls for a greater emphasis on prevention, given that about half of all deaths in the United States are considered attributable to modifiable health behaviors (Mokdad et al., 2004, 2005). The change in handling of preventive health services in the ACA is illustrative of evolving insurance policy, realigning incentives, and making positive use of medical management to promote preventive services and disease management programs to improve care and maintain a healthy workforce (Nussbaum, 2011).
As noted in Table 2-1, administrative costs, including profits for insurers and their shareholders, contribute to premium costs. The ACA has new provisions for the medical-loss ratio, which sets the minimum percentage of premiums that insurers may spend on actual medical care (e.g., 80 percent for individuals and small group insurance and 85 percent for larger group insurance).5
ILLUSTRATIVE APPROACHES TO COVERAGE DECISIONS
Benefit design is “an iterative process,” taking into account what employers and consumers in the marketplace want and are willing to pay for. Thus, what is typical in the market today becomes a base for consideration of the EHB package. Similarly, any insurer benefit package tends to build on what is already covered. In updating packages, insurers told the committee they might conduct focus groups, hold field satisfaction surveys, and receive feedback from insurance brokers on market demand as well as survey advances in medical technology.
5 § 1001, amending § 2718 of the Public Health Service Act [42 U.S.C. 300 gg–18].
The committee heard from numerous stakeholders about ways of making coverage decisions; most of the methods presented deal with assessing whether an individual technology should be included for coverage or is necessary for a specific individual rather than looking at the package of benefits as a whole. The committee three general approaches that reflect a range of current practices: (1) use of evidence for adding individual technologies to the benefit package, (2) prioritization among benefits, and (3) value-based insurance design. Illustrative examples are given.
Evidence-Based Approaches
Examples of the criteria and methods insurers use to develop their evidence base for covered benefit and medical management decisions follow. Insurers seek to apply the best standard of evidence available. In some cases, for example, Medicare allows coverage in conjunction with evidence development in clinical trials, and Washington state has shown some flexibility in cases where a rare condition is less likely to have a sufficiently developed evidence base.
Receiving and Analyzing Multiple Sources of Input
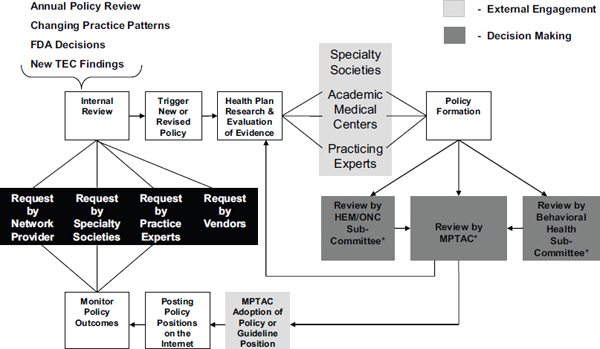
Insurers keep abreast of medical trends, clinical practice guidelines, approvals by CMS and the Food and Drug Administration (FDA) of new technologies and pharmaceuticals, and evidence-based reviews from the BlueCross BlueShield Association’s Technology Evaluation Center (TEC) and others. From these various inputs and internal analyses, insurers examine whether this information should lead to changes in covered benefits and development of clinical policies applied in medical necessity determinations, as illustrated for WellPoint’s process in Figure 2-3.
Aetna similarly selects new and existing technologies for detailed review based on contextual considerations including the quantity of use and the importance of questions that have arisen regarding the specific medical technology; the potential impact of the technology on the company and its members; the availability of evidence in the peer-reviewed literature, guidelines, and consensus statements; changes in regulatory status; whether the technology relates to a rare condition; or other information that is material to the status of the medical technology (McDonough, 2011). Through this process, the need for medical management (e.g., whether the service might best be performed in centers of excellence) is determined. Thus, rather than exclude a specific service, a clinical policy could define under what circumstances it would be covered. Like WellPoint, Aetna’s clinical coverage criteria are derived in part from the BlueCross BlueShield Association’s TEC (Box 2-3). In addition to these criteria, Aetna considers indications in major drug compendia recognized by CMS, the approval status of technologies from relevant government regulatory bodies (e.g., CMS, FDA), and technology assessments from other reliable sources of information such as the California Technology Assessment Forum and Health Technology Assessment International.
Medicare established the Medicare Evidence Development & Coverage Advisory Committee (MEDCAC) in 1998 to provide CMS with independent guidance and expert advice, “based upon the reasoned application of scientific evidence” on specific clinical topics, including issues relevant to coverage policy development (HHS, 2010a). Service-level assessments have included things such as assessing the strength of the evidence for multifactorial, noninvasive, “lifestyle” modifying interventions to treat cardiac disease and clarifying what constitutes the standard of care in wound therapy (HHS, 2010a). Pre-meeting materials considered include external health technology assessment(s) (TAs) conducted by the Agency for Healthcare Research and Quality or an Evidence-Based Practice Center under contract with that agency; any other relevant TAs; an evidence summary prepared by CMS staff; and copies of relevant articles reviewed by CMS.
Applying a Hierarchy of Evidence
UnitedHealthcare uses processes similar to the insurers above, applying a hierarchy of criteria and evidence for coverage determination; a first step is consideration of mandatory requirements from federal or state sources and then application of a hierarchical standard for evidence to potential interventions considered for coverage (Box 2-4).
*Depending on the subject, different review committees will be engaged. This illustration includes the Hematology/Oncology Committee (HEM/ONC), the Medical Policy & Technology Assessment Committee (MPTAC), and the Behavioral Health subcommittee; each of these includes external physician representation.
SOURCE: Nussbaum, 2011.
The following criteria are considered in evaluating a medical technology:
• The technology must have final approval from the appropriate governmental regulatory bodies (when required).
• The scientific evidence must permit conclusions concerning the effect of the technology on health outcomes.
• The technology must improve net health outcome.
• The technology must be as beneficial as any established alternatives.
• The improvement must be attainable outside investigational settings.
SOURCE: BCBSA, 2011.
BOX 2-4
UnitedHealthcare’s Hierarchy of Criteria for Benefit or Coverage Determination
Federal and state mandates (e.g., Centers for Medicare & Medicaid Services [CMS] National Coverage Decisions [NCDs] are the highest criteria for Medicare beneficiaries).
Standards of Evidence:
• Statistically robust, well-designed randomized controlled trials
• Statistically robust, well-designed cohort studies
• Large, multisite observational studies
• Single-site observational studies
• In the absence of incontrovertible scientific evidence, medical policies may be based upon national consensus statements by recognized authorities. The following stratification describes the hierarchy of use of medical policies and clinical guidelines within UnitedHealthcare:
![]() National guidelines and consensus statements (e.g., U.S. Preventive Services Task Force [USPSTF], National Institutes of Health [NIH] clinical statements, Agency for Healthcare Research and Quality [AHRQ] clinical statements)
National guidelines and consensus statements (e.g., U.S. Preventive Services Task Force [USPSTF], National Institutes of Health [NIH] clinical statements, Agency for Healthcare Research and Quality [AHRQ] clinical statements)
![]() Evidence-based nationally recognized clinical guidelines
Evidence-based nationally recognized clinical guidelines
![]() CMS NCDs
CMS NCDs
![]() Clinical position papers of professional specialty societies (e.g., American College of Physicians [ACP], American College of Cardiology [ACC], American College of Chest Physicians [ACCP]) when their statements are based on referenced clinical evidence
Clinical position papers of professional specialty societies (e.g., American College of Physicians [ACP], American College of Cardiology [ACC], American College of Chest Physicians [ACCP]) when their statements are based on referenced clinical evidence
• Expert opinion using Cochrane grading
• Particularly for new or emerging medical technologies, no health service will be deemed unproven solely on the basis of a lack of randomized controlled trials. Similarly, UnitedHealthcare will develop no medical policies based solely on expert opinion.
SOURCE: Personal communication with committee member Sam Ho, UnitedHealthcare, 2011.
Washington State similarly uses a hierarchy of evidence (Box 2-5), embedded in its administrative code,6 to define benefits across the state’s coverage programs (i.e., Medicaid, state employee benefit program, and state basic health plan [a coverage program for lower-income populations not eligible for Medicaid]). The key principles for the design process—with many stakeholders participating including legislators, providers, and beneficiaries—are consistency of decisions, transparency, hierarchy of decision criteria, evidence basis, and focus on patient safety. These benefits use “the best evidence of proven value to the population, while respecting the appropriateness of services and the authority of the treating provider” (Thompson, 2011).
This hierarchy of evidence and its application are used to define covered benefits and to establish the basis for medical necessity decisions. In testimony before the committee, Dr. Jeffery Thompson, chief medical officer, State of Washington Medicaid Program, illustrated changes in coverage and improvements in care and outcomes using this evidence-based approach. Before the introduction of evidence-based benefit design, cardiac rehabilitation was not a covered benefit; but once reviewed, A-level evidence revealed this service contributed to avoiding further surgery, hospitalization, and recurrence of events—so the benefit is now covered (Thompson, 2011). Similarly, bariatric surgery was previously covered for numerous indications, but the mortality rate reached 40 percent at some hospitals. Review of the evidence revealed that surgery is indicated for this program in some instances (e.g., BMI >35 with diabetes and/or joint replacement) but not all. By limiting coverage to specific evidence-based indications and use of centers of excellence, the department self-reports that it has reduced case costs by half ($36,000 to $17,000) and improved outcomes—no bariatric surgery—related death in seven years among individuals enrolled in state-covered plans (Thompson, 2011).
Although the department generally approves benefits supported by A-and B-level evidence, it does not necessarily reject benefits with only C-and D-level evidence. For example, if a provider can prove that a service supported by inconsistent, C-level evidence is “less costly, less risky, and is the next step in reasonable care,” then coverage may be considered. For example, a PET (positron emission tomography) scan for cancer diagnosis may have limited or no outcome studies but in special cases can reduce the costs and risks of a surgical procedure. Additionally, recognizing that certain rare conditions may never have A-level studies, state-covered plans have been willing to cover some experimental D-level treatments.7 Washington state’s evidence hierarchy has also been extended to pharmaceutical benefits in developing a tiered formulary, with the state’s basic health plan having a $10 co-payment for drugs “above the line” (e.g., omeprazole) and a 50 percent co-payment for drugs “below the line” (e.g., Prevacid) (Thompson, 2011).
Prioritization Approach
Individuals and employers have budgets that limit what they are willing to pay, leading to the exclusion of certain benefits and the adoption of multiple benefit design options discussed earlier. Whether private or public funds are spent, questions arise about what people think shared resources should be spent on. To date, insurers and purchasers have made some decisions on which things should be excluded, such as no coverage of medical procedures solely for cosmetic purposes (e.g., surgery or other procedures performed solely for beautification or to improve appearance) (see Appendix F for information on typical exclusions). The State of Oregon has gone a step further to define in much more detail which services are included and excluded, and setting priorities among them so that as budget levels change, it is transparent which things are covered.
Oregon determines a detailed list of prioritized condition-treatment pairs for its Medicaid program, known as the Oregon Health Plan (Box 2-6); these originally totaled more than 700 and are at 679 for 2011 (Oregon Health Services Commission, 2011b). Services necessary to determine a diagnosis are covered. Ancillary services such as prescription drugs and durable medical equipment (DME) are covered for conditions in the funded region. In 2006, the commission revised its methodology to have a population focus. “Greater emphasis is [now] placed on preventive services and chronic disease management, reflecting the fact that providing health care before reaching
6 Washington Administrative Code, 388-501-0165 (1994).
7 Provided the treatment is approved by an internal review board (IRB), the treating physician is in a study, and the patient has provided informed consent.
crisis mode will prevent avoidable morbidity and mortality” (DiPrete and Coffman, 2007, p. 5). The following five impact measures are considered: (1) healthy life-years, (2) suffering, (3) population effects, (4) vulnerability of population, and (5) tertiary prevention, combined with the two factors of effectiveness and need for medical service under the current formula (DiPrete and Coffman, 2007).
BOX 2-5
Hierarchy of Evidence Employed by Washington State
Definition: (a) The hierarchy (in descending order with Type I given the greatest weight) is:
(i) Type I: Meta-analysis done with multiple, well-designed controlled studies;
(ii) Type II: One or more well-designed experimental studies;
(iii) Type III: Well-designed, quasi-experimental studies such as nonrandomized controlled, single group pre-post, cohort, time series, or matched case-controlled studies;
(iv) Type IV: Well-designed, nonexperimental studies, such as comparative and correlation descriptive, and case studies (uncontrolled); and
(v) Type V: Credible evidence submitted by the provider.
Classification: (b) Based on the quality of available evidence, the department determines if the requested service is effective and safe for the client by classifying it as an “A,” “B,” “C,” or “D” level of evidence:
(i) “A” level evidence: Shows the requested service or equipment is a proven benefit to the client’s condition by strong scientific literature and well-designed clinical trials such as Type I evidence or multiple Type II evidence or combinations of Type II, III, or IV evidence with consistent results (an “A” rating cannot be based on Type III or Type IV evidence alone).
(ii) “B” level evidence: Shows the requested service or equipment has some proven benefit supported by:
(A) Multiple Type II or III evidence or combinations of Type II, III, or IV evidence with generally consistent findings of effectiveness and safety (a “B” rating cannot be based on Type IV evidence alone); or
(B) Singular Type II, III, or IV evidence in combination with department-recognized:
(I) Clinical guidelines; or
(II) Treatment pathways; or
(III) Other guidelines that use the hierarchy of evidence in establishing the rationale for existing standards.
(iii) “C” level evidence: Shows only weak and inconclusive evidence regarding safety and/or efficacy such as:
(A) Type II, III, or IV evidence with inconsistent findings; or
(B) Only Type V evidence is available.
(iv) “D” level evidence: Is not supported by any evidence regarding its safety and efficacy, for example that which is considered investigational or experimental.
Application: (c) After classifying the available evidence, the department:
(i) Approves “A” and “B” rated requests if the service or equipment:
(A) Does not place the client at a greater risk of mortality or morbidity than an equally effective alternative treatment; and
(B) Is not more costly than an equally effective alternative treatment.
(ii) Approves a “C” rated request only if the provider shows the requested service is the optimal intervention for meeting the client’s specific condition or treatment needs, and:
(A) Does not place the client at a greater risk of mortality or morbidity than an equally effective alternative treatment; and
(B) Is less costly to the department than an equally effective alternative treatment; and
(C) Is the next reasonable step for the client in a well-documented tried-and-failed attempt at evidence-based care.
(iii) Denies “D” rated requests unless:
(A) The requested service or equipment has a humanitarian device exemption from the Food and Drug Administration (FDA); or
(B) There is a local institutional review board (IRB) protocol addressing issues of efficacy and safety of the requested service that satisfies both the department and the requesting provider.
SOURCE: Washington Administrative Code, 388-501-0165.
The resulting prioritized list is used by the legislature to allocate funding for Medicaid and the State Children’s Health Insurance Program (SCHIP), but the legislature cannot change the priorities set by the independent Health Services Commission (DiPrete and Coffman, 2007). Rather, the legislature “draws a line” on the list beyond which it cannot pay—in 2011 at line 502 (Oregon Health Services Commission, 2011a)—based on the cumulative actuarial estimates of the cost for each condition-treatment pair for the covered population. Approximately 75 percent of the more than 679 condition-treatment pairs are funded (Table 2-2). The list is updated every 6 months to account for changes in medical coding and to make technical corrections, then is reviewed every 2 years for any changes in priority rankings. This process of prioritization is frequently cited as being too time-and labor-intensive for
wider adoption, yet this is done by a staff of four in Oregon.8 Approximately one-third of the state has benefited from the expanded access made possible by setting explicit health service priorities (DiPrete and Coffman, 2007).
Value-Based Insurance Design Approach
Value-based insurance design (VBID) seeks to align patient cost sharing with the value of clinical services, including how they are provided (e.g., most appropriate setting and health care provider). VBID identifies those services whose benefits relative to their costs represent an efficient use of resources for patients, comparing the cost-effectiveness of one intervention to alternatives, including no intervention (Fendrick et al., 2010). The use of these services is then incentivized, traditionally through two mechanisms: (1) minimal cost barriers and (2) financial incentives. Approaches to VBID call for lowering cost sharing for high-value services and raising
8 Personal communication with Mark Gibson, Center for Evidence-Based Policy, Oregon Health and Science University, February 9, 2011.
costs through higher co-payments on services with low value (Choudhry et al., 2010b). Other mechanisms by which VBID programs can impact spending include designating in-and out-of-network providers based on high performance value, not just price, and/or providing incentives for enrollees to adopt more healthful behaviors and/or to achieve better biometric results, such as blood pressure, cholesterol, blood sugar levels, or body mass index (MedPAC, 2011). One challenge is that as one seeks to align coverage with more specific cost-effective services and health outcomes, the more complex benefit design and implementation can become. Although barriers to each VBID approach exist, they all have the potential to improve the efficiency of the health care system. Given the differences across approaches, it is suggested that regulators allow for flexibility when designing such programs (Chernew et al., 2010).
BOX 2-6
Oregon Treatment-Condition Pair Examples
| Line: | 10 | |
| Condition: | Type I diabetes mellitus | |
| Treatment: | medical therapy | |
| ICD-9: | 250.01,250.03,250.11,250.13,250.21,250.23,250.31,250.33,250.51,250.53,250.61,250.63,250.71,250.73,250.91,250.93,251.3,V53.91,V65.46 | |
| CPT: | 49435,49436,90935-90947,90989-90997,92002-2014,92227,95250,95251,96150-96154,97802-97804,98966-8969,99051,99060,99070,99078,99201-99360,99366,99374,99375,99379-99444,99468-99480,99605-99607 | |
| HCPCS: | G0245,G0246,G0406-G0408,G0425-G0427,S0270-S0274,S9145,S9353 | |
|
Line: |
118 |
|
| Condition: | Fracture of ribs and sternum, open | |
| Treatment: | Medical and surgical treatment | |
| ICD-9: | 807.1,807.3,V54.19,V54.29 | |
| CPT: | 11010-11012,21805,21810,21825,97602,98966-98969,99051,99060,99070,99078,99201-99360,99366,99374,99375,99379-99444,99468-99480,99605-99607 | |
| HCPCS: | G0406-G0408,G0425-G0427,S0270-S0274 | |
|
Line: |
573 |
|
| Condition: | Allergic rhinitis and conjunctivitis, chronic rhinitis | |
| Treatment: | Medical therapy | |
| ICD-9: | 372.01-372.06,372.14,372.54,372.56,472,477,995.3,V07.1 | |
| CPT: | 30420,86486,92002-92060,92070-92226,92230-92313,92325-92353,92358-92371,95004,95010,95015-95180,98966-98969,99051,99060,99070,99078,99201-99360,99366,99374,99375,99379-99444,99468-99480,99605-99607 | |
| HCPCS: | G0406-G0408,G0425-G0427,S0270-S0274 |
SOURCE: Oregon Health Services Commission, 2011a.
NOTE: CPT = Current Procedural Terminology; HCPCS = Healthcare Common Procedure Coding
System; ICD = International Statistical Classification of Diseases and Related Health Problems.
TABLE 2-2 The State of Oregon Uses a Prioritized List of Services to Make Coverage Decisions
| Line Number | Examples of Services | Coverage |
| 1 | Malernily care | Covered |
| 101 | Medical treatment of acute lymphocytic leukemia | ↑ |
| 201 | Surgical treatment of brain hemorrhage | |
| 301 | Treatment for rheumatic heart disease | |
| 401 | Laser therapy to prevent retinal tear | |
| 501 | Treatment for noninflammatory vaginal disorders | |
| 551 | Treatment for back pain without neurologic impairment | Not Covered |
| 651 | Treatment for calcium deposits | ↓ |
SOURCE: Oregon Health Services Commission, 2011b.
Consequently, innovative programs to improve health and lower long term costs have been developed and studied to determine the benefits of VBID. For example, one study comparing results in the context of identical disease management programs found that when one employer reduced co-payments for five classes of drugs, adherence increased in four classes and overall nonadherence was reduced by 7 to 14 percent (Chernew et al., 2008). Similarly, Choudhry et al. (2010a) found when a large employer, Pitney Bowes, eliminated co-payments for statins, adherence was 2.8 percent greater than for those still paying co-payments, and when it reduced co-payments for medication inhibiting blood clotting, patient adherence was 4 percent greater. Similarly, Maciejewski et al. (2010) also found that adherence to diabetes, hypertension, hyperlipidemia, and congestive heart failure medications was improved between 2 and 4 percent when employees were offered the medications at a reduced cost (no co-payments for generic and reduced co-payment for brand name medications), compared to employees not offered that option.
Assessing the financial impacts of VBID implementation can be challenging, because many programs are new and “even if a strategy is effective, how that translates into costs or savings may vary from one organization to another” (MedPAC, 2011, p. 88). This lack of generalizability requires an individualized analysis of programs. The state of Minnesota reported a savings of 7 percent after instituting an incentive program in 2002 for enrollees to see efficient providers; primary care clinics are ranked annually on overall claims-based cost and divided into four tiers, with patients facing higher cost sharing when utilizing clinics with the highest overall costs (MedPAC, 2011). Also, actuarial modeling of an Oregon value-based plan suggests “the potential to produce savings of 3 percent to 5 percent initially” when applied in the state’s Oregon Educators Board plan (MedPAC, 2011, p. 85; Smith and Saha, 2011). By these indications, VBID is an approach to control health care spending—with a focus on value, using co-payment rates that are based on the value of clinical services (benefits and costs), rather than solely on the costs of delivering those services (Fendrick et al., 2001).
BOX 2-7
Inclusion Criteria for Oregon’s Value-Based Services (VBS)
• Ambulatory services (i.e., outpatient), including medications, diagnostic tests, procedures, and some office visits
• Primarily offered in the medical home
• Primarily focused on chronic illness management, preventive care, and/or maternity care
• Of clear benefit, strongly supported by evidence
• Cost-effective
• Reduce hospitalizations or emergency department visits, reduce future exacerbations or illness progression, or improve quality of life
• Low cost up front
• High utilization desired
• Low risk of inappropriate utilization
SOURCE: Oregon Health Services Commission, 2011c.
Oregon has a long history of incorporating value into its decisions about health coverage, having rank-ordered its benefits since 1989 as discussed previously. It is now translating lessons learned from its Medicaid program to the private sector. Identifying over time, per inclusion criteria (Box 2-7), a set of 20 value-based services (VBS) applicable to a set of diagnoses, the Health Services Commission determined that these should be promoted and hence offered at no or minimal cost sharing with a waived deductible in the state’s Medicaid program (e.g., the provision of diagnostic spirometry and medications according to the National Institute for Health and Clinical Excellence 2008 stepwise treatment protocol for asthma) (Oregon Health Services Commission, 2010). Finding that many Medicaid recipients already receive these services with little or no cost sharing, the expectation is that this will have a more significant effect when adopted in the private sector. Importantly for the continued role of VBS, the Health Services Commission plans to update this list annually to ensure that the most current high-quality supporting evidence (such as Cochrane systematic reviews of randomized controlled clinical trials and evidence-based guidelines) is used for designing coverage (Oregon Health Services Commission, 2011c).
One additional proposal for a state-specific essential benefits package to cover all Oregonians, since sidelined in anticipation of federal guidance on the EHB, was slated to include among its VBS offered with 0-5 percent cost sharing the following: routine vaccinations, prenatal care, chronic illness management, and smoking cessation treatment. Less effective care and care for minor injury and self-limited illness (e.g., chronic back pain, viral sore throat, seasonal allergies, and acne) would have the highest cost sharing (Saha et al., 2010). Currently, a separate effort through the Oregon Health Leadership Council (OHLC), an organization of business leaders, health plans, and providers seeking to reduce the rate of increase of health care costs, is actively leading efforts to incorporate value-based design in the private sector. With an estimated 8-12 percent premium reduction, the OHLC proposed a benefit package with three tiers of service, in which the middle one—level 2—resembles most traditional plans with a deductible and co-insurance for most services, but the level 1 tier would cover prescription drugs, some lab, imaging, and other ancillary services related to six chronic conditions—coronary disease, congestive heart failure, chronic obstructive pulmonary disease, diabetes, asthma, and depression—with minimal or no cost sharing. On the other hand, several types of surgeries (for example, coronary artery bypass grafting [CABG] and angioplasty) have significant cost sharing. OHLC originally wanted to include primary care visits in the tier without cost sharing; however, administrative barriers, such as the inability of billing systems to distinguish primary care visits from those for a specific chronic condition, may not make it feasible for all insurers (MedPAC, 2011).
As of January 1, 2011, a plan based on the VBS model, which includes no cost sharing for cholesterol and blood pressure medications, is being offered to employees of Evraz, Inc., which operates steel mills in Oregon and Delaware. Furthermore, while some workers have the option of staying in their current plan, the company is waiving the employee premium contribution for individuals who opt for the value-based plan. Finally, a similar plan has been rolled out to the employees of the health insurer ODS, further indicating that value-based insurance design is being recognized as an attractive approach to plan design.
Summary
This chapter provides the committee’s review of considerations in benefit choices, benefit design, cost drivers, and a sample of illustrative approaches to deciding benefit coverage and its application in clinical policies—setting the stage for developing the policy foundations and criteria for designing the EHB package in the next chapter.
AAA (American Academy of Actuaries). 2009. Critical issues in health reform: Actuarial equivalence. http://www.actuary.org/pdf/health/equivalence_may09.pdf (accessed September 22, 2011).
Abraham, K. 1986. Distributing risk: Insurance, legal theory, and public policy. New Haven, CT: Yale University Press.
Baker, N., J. W. Whittington, R. K. Resar, F. A. Griffin, and K. M. Nolan. 2010. Reducing costs through the appropriate use of specialty services. Cambridge, MA: Institute of Healthcare Improvement.
BCBSA (BlueCross BlueShield Association). 2011. Technology Evaluation Center criteria. http://www.bcbs.com/blueresources/tec/tec-criteria.html (accessed June 15, 2011).
Brenner, D. J., and E. J. Hall. 2007. Computed tomography—an increasing source of radiation exposure. New England Journal of Medicine 357(22):2277-2284.
Calega, V. 2011. Comments to the IOM Committee on the Determination of Essential Health Benefits by Virginia Calega, Vice President, Medical Management and Policy, Highmark BlueCross BlueShield, Washington, DC, January 13.
Card, D., C. Dobkin, and N. Maestas. 2008. The impact of nearly universal insurance coverage on health care utilization: Evidence from Medicare. The American Economic Review 98(5):2242-2258.
______. 2009. Does Medicare save lives? The Quarterly Journal of Economics 124(2):597-636.
CBO (Congressional Budget Office). 2008. Technological change and the growth of health care spending. Washington, DC: Congressional Budget Office.
Chernew, M. E., M. R. Shah, A. Wegh, S. N. Rosenberg, I. A. Juster, A. B. Rosen, M. C. Sokol, K. Yu-Isenberg, and A. M. Fendrick. 2008. Impact of decreasing copayments on medication adherence within a disease management environment. Health Affairs 27(1):103-112.
Chernew, M. E., I. A. Juster, M. Shah, A. Wegh, S. Rosenberg, A. B. Rosen, M. C. Sokol, K. Yu-Isenberg, and A. M. Fendrick. 2010. Evidence that value-based insurance can be effective. Health Affairs 29(3):530-536.
Choudhry, N. K., M. A. Fischer, J. Avorn, S. Schneeweiss, D. H. Solomon, C. Berman, S. Jan, J. Liu, J. Lii, M. A. Brookhart, J. J. Mahoney, and W. H. Shrank. 2010a. At Pitney Bowes, value-based insurance design cut copayments and increased drug adherence. Health Affairs 29(11):1995-2001.
Choudhry, N. K., M. B. Rosenthal, and A. Milstein. 2010b. Assessing the evidence for value-based insurance design. Health Affairs 29(11):1988-1994.
Cutler, D. M. 1995. Technology, health costs, and the NIH. Paper prepared for the National Institutes of Health Economics Roundtable on Biomedical Research. http://www.economics.harvard.edu/faculty/cutler/files/Technology,%20Health%20Costs%20and%20the%20NIH.pdf (accessed September 12, 2011).
Dafny, L., and J. Gruber. 2005. Public insurance and child hospitalizations: Access and efficiency effects. Journal of Public Economics 89:109-129.
DiPrete, B., and D. Coffman. 2007. A brief history of health services prioritization in Oregon. http://www.oregon.gov/OHA/OHPR/HSC/docs/PrioritizationHistory.pdf?ga=t (accessed May 19, 2011).
Eddy, D. M. 1984. Variations in physician practice: The role of uncertainty. Health Affairs 3(2):74-89.
Fendrick, A. M., D. G. Smith, M. E. Chernew, and S. N. Shah. 2001. A benefit-based copay for prescription drugs: Patient contribution based on total benefits, not drug acquisition cost. American Journal of Managed Care 7(9):861-867.
Fendrick, A. M., D. G. Smith, and M. E. Chernew. 2010. Applying value-based insurance design to low-value health services. Health Affairs 29(11):2017-2021.
Flynn, K. E., M. A. Smith, and M. K. Davis. 2002. From physician to consumer: The effectiveness of strategies to manage health care utilization. Medical Care Research and Review 59(4):455-481.
Garber, A. M. 2011. Medical necessity, coverage policy, and evidence based medicine. PowerPoint Presentation to the IOM Committee on the Determination of Essential Health Benefits by Alan Garber, Stanford University, Washington, DC, January 13.
Glied, S. 2011. Testimony to the IOM Committee on the Determination of Essential Health Benefits by Sherry Glied, Assistant Secretary for Planning and Evaluation, U.S. Department of Health and Human Services, Washington, DC, January 13.
Hendel, R. C., M. Cerqueira, P. S. Douglas, K. C. Caruth, J. M. Allen, N. C. Jensen, W. Pan, R. Brindis, and M. Wolk. 2010. A multicenter assessment of the use of single-photon emission computed tomography myocardial perfusion imaging with appropriateness criteria. Journal of the American College of Cardiology 55(2):156-162.
HHS (Department of Health and Human Services). 2010a. Charter: Medicare Evidence Development & Coverage Advisory Committee. Washington, DC: U.S. Department of Health and Human Services.
______. 2010b. Glossary. http://www.healthcare.gov/glossary (accessed November 7, 2010).
Highmark Blue Cross Blue Shield. 2011. Provider Resource Center: Highmark Radiology Management Program. https://prc.highmarkblueshield.com/rscprc/faces/prcMainPage.jsp (accessed April 19, 2011).
IOM (Institute of Medicine). 1993. Employment and health benefits. Washington, DC: National Academy Press.
Jacobson, P. D., R. A. Rettig, and W. M. Aubry. 2007. Litigating the science of breast cancer treatment. Journal of Health Politics, Policy and Law 32(5):785-818.
Kaminski, J. L. 2007. Defining medical necessity. http://www.cga.ct.gov/2007/rpt/2007-r-0055.htm (accessed April 20, 2011).
Kashihara, D., and K. Carper. 2010. Statistical brief #301: National health care expenses in the U.S. civilian noninstitutionalized population, 2008. http://www.meps.ahrq.gov/mepsweb/data_files/publications/st301/stat301.shtml (accessed June 26, 2011).
KFF (Kaiser Family Foundation). 2010. U.S. health care costs. http://www.kaiseredu.org/Issue-Modules/US-Health-Care-Costs/Background-Brief.aspx (accessed June 26, 2011).
Ko, D. T., J. S. Ross, Y. Wang, and H. M. Krumholz. 2010. Determinants of cardiac catheterization use in older Medicare patients with acute myocardial infarction. Circulation: Cardiovascular Quality and Outcomes 3(1):54-62.
Levine, S. 2011. PowerPoint Presentation to the IOM Committee on the Determination of Essential Health Benefits by Sharon Levine, Associate Executive Medical Director, The Permanente Medical Group, Costa Mesa, CA, March 2.
Maciejewski, M. L., J. F. Farley, J. Parker, and D. Wansink. 2010. Copayment reductions generate greater medication adherence in targeted patients. Health Affairs 29(11):2002-2008.
McDevitt, R. 2008. Actuarial value: A method for comparing health plan benefits. Oakland, CA: California Healthcare Foundation.
McDonough, R. 2011. Determination of essential health benefits. PowerPoint Presentation to the IOM Committee on the Determination of Essential Health Benefits by Robert McDonough, Director, Clinical Policy Research and Development, Aetna, Washington, DC, January 13.
McGlynn, E. A., S. M. Asch, J. Adams, J. Keesey, J. Hicks, A. DeCristofaro, and E. A. Kerr. 2003. The quality of health care delivered to adults in the United States. New England Journal of Medicine 348(26):2635-2645.
MedPAC (Medicare Payment Advisory Commission). 2011. June 2011 report to the Congress: Medicare and the health care delivery system. Washington, DC: Medicare Payment Advisory Commission.
Milstein, A. 1997. Managing utilization management: A purchaser’s view. Health Affairs 16(3):87-90.
Mokdad, A. H., J. S. Marks, D. F. Stroup, and J. L. Gerberding. 2004. Actual causes of death in the United States, 2000. Journal of the American Medical Association 291(10):1238-1245.
______. 2005. Correction: Actual causes of death in the United States, 2000. Journal of the American Medical Association 293(3):293-294.
NCQA (National Committee for Quality Assurance). 2007. Special needs plan (SNP) phase 1—draft stucture and process requirements glossary. http://www.ncqa.org/portals/0/publiccomment/SNP/SNP_Glossary.pdf (accessed June 26, 2011).
NEHI (New England Healthcare Institute). 2008. How many more studies will it take? Cambridge, MA: New England Healthcare Institute.
Newhouse, J. P. 1992. Medical care costs: How much welfare loss? Journal of Economic Perspectives 6(3):3-21.
Nussbaum, S. 2011. Health insurance plan variance in coverage (inclusions, exclusions, networks) and benefit design for quality improvement. PowerPoint Presentation to the IOM Committee on the Determination of Essential Health Benefits by Sam Nussbaum, Executive Vice-President, Clinical Policy and Chief Medical Officer, Wellpoint, Inc., Costa Mesa, CA, March 2.
Oregon Health Services Commission. 2010. Value-based services: Proposed “barrier-free” services for use within a value-based benefit package. http://www.oregon.gov/OHA/OHPR/HSC/docs/VBS.pdf (accessed August 4, 2011).
______. 2011a. The prioritized list. http://www.oregon.gov/OHA/healthplan/priorlist/main.shtml (accessed June 27, 2011).
______. 2011b. Prioritized list of health services: April 1, 2011. http://www.oregon.gov/OHA/OHPR/HSC/docs/L/Apr11List.pdf (accessed May 10, 2011).
______. 2011c. Value-based services. http://www.oregon.gov/OHA/OHPR/HSC/VBS.shtml (accessed June 26, 2011).
PwC (PricewaterhouseCoopers) Health Research Institute. 2011. Behind the numbers: Medical cost trends for 2012. New York: PricewaterhouseCoopers.
Rosen, A. K. 2010. Are we getting better at measuring patient safety? http://www.webmm.ahrq.gov/perspective.aspx?perspectiveID=94 (accessed June 26, 2011).
Rosenblatt, R., S.A. Law, and S. Rosenbaum. 1997. Law & the American health care system. New York: Foundation Press.
Saha, S., D. D. Coffman, and A. K. Smits. 2010. Giving teeth to comparative-effectiveness research—the Oregon experience. New England Journal of Medicine 362(7):e18.
Schuster, M. A., E. A. McGlynn, and R. H. Brook. 2005. How good is the quality of health care in the United States? Milbank Quarterly 83(4):843-895.
Simpson, K. R. 2010. Reconsideration of the costs of convenience: Quality, operational, and fiscal strategies to minimize elective labor induction. Journal of Perinatal & Neonatal Nursing 24(1):43-52.
Singer, S., L. Bergthold, C. Vorhaus, S. Olson, I. Mutchnick, Y. Y. Goh, S. Zimmerman, and A. Enthoven. 1999. Decreasing variation in medical necessity decision making. Appendix B. Model language developed at the “Decreasing Variation in Medical Necessity Decision Making” Decision Maker Workshop in Sacramento, CA, March 11-13, 1999.
Smith, J., and S. Saha. 2011. Oregon’s value based benefits package. PowerPoint Presentation to the IOM Committee on the Determination of Essential Health Benefits by Jeanene Smith, Administrator, Office for Oregon Health Policy and Research and Somnath Saha, Staff Physician, Portland VA Medical Center and Chair, Oregon Health Services Commission, Costa Mesa, CA, March 2.
Smith, S. D., S. K. Heffler, and M. S. Freeland. 2000. The impact of technological change on health care cost spending: An evaluation of the literature. https://www.cms.gov/NationalHealthExpendData/downloads/tech_2000_0810.pdf (accessed September 12, 2011).
Smith, S., J. P. Newhouse, and M. S. Freeland. 2009. Income, insurance, and technology: Why does health spending outpace economic growth? Health Affairs 28(5):1276-1284.
Stacey, D., C. L. Bennett, M. J. Barry, N. F. Col, K. B. Eden, M. Holmes-Rouner, H. Llewellyn-Thomas, A. Lyddiatt, F. Légaré, and R. 2011. Decision aids for people facing health treatment or screening decisions. Cochrane Database of Systematic Reviews (10): CD001431.
Thompson, J. 2011. Health care that works: Evidence-based Medicaid. PowerPoint Presentation to the IOM Committee on the Determination of Essential Health Benefits by Jeffery Thompson, Chief Medical Officer, Washington Medicaid Program, Washington State Department of Social and Health Services, Costa Mesa, CA, March 2.
URAC. 2011. What is care management? http://www.urac.org/resources/careManagement.aspx (accessed June 26, 2011).
Wennberg, J. E. 2011. Time to tackle unwarranted variations in practice. British Medical Journal 342:687-690.
White, D. B., V. L. Bonham, J. Jenkins, N. Stevens, and C. M. McBride. 2008. Too many referrals of low-risk women for BRCA1/2 genetic services by family physicians. Cancer Epidemiology Biomarkers & Prevention 17(11):2980-2986.
Wickizer, T. M., and D. Lessler. 2002. Utilization management: Issues, effects, and future prospects. Annual Review of Public Health 23:233-254.
Wright, A. 2011. Testimony to the IOM Committee on the Determination of Essential Health Benefits by Anthony Wright, Executive Director, Health Access California, Costa Mesa, CA, March 2.
Yu, W. W., and T. M. Ezzati-Rice. 2005. Statistical brief #81: Concentration of health care expenditures in the U.S. civilian noninstitutionalized population. http://www.meps.ahrq.gov/mepsweb/data_files/publications/st81/stat81.shtml (accessed June 26, 2011).