___________
Policy Foundations and Criteria for the EHB
The Institute of Medicine committee was tasked with developing a framework for the Secretary to use when explicitly defining and revising the essential health benefits (EHB). Upon integrating its research and public input from its workshop and online public comment form, the committee established four policy foundations to guide its work: economics, ethics, evidence-based practice, and population health. Related criteria are outlined for three purposes: (1) assessment of the aggregate EHB package, (2) evaluation of individual services to be included in the EHB package, and (3) guidance on fair processes.
The Secretary asked the Institute of Medicine (IOM) committee to develop an explicit framework for considering the EHB package that would serve the Department of Health and Human Services (HHS) now and in the future (Glied, 2011). This chapter outlines the resulting framework, including the specific policy foundations and criteria that the committee used to guide its own work. The committee later recommends that the Secretary use this framework when defining the EHB package and updating it in the years to come (see Chapters 5 and 9).
The committee specifically queried the public via its online public comment form about what principles, criteria, and processes the Secretary might use for defining and revising the EHB. The many suggestions made, as well as information gleaned from the committee’s research, could be classified into four main policy foundation domains: economics, ethics, evidence-based medical practice, and population health. What follows is a general discussion of these domains, principles associated with these domains, and a list of criteria that emerged common to these policy areas to direct evaluation of the aggregate EHB package, to assess individual technologies for inclusion, and to establish appropriate characteristics of processes. The committee’s framework of policy foundations and related criteria is consistent with the goals of the Patient Protection and Affordable Care Act (ACA) and those initiatives contained within it, such as HHS’s National Quality Strategy1—to expand access to health insurance, to improve the quality of care, to improve the health of individuals and communities, and to reduce the cost of care. It also reflects previous IOM work related to improving the quality of care, such as Crossing the Quality Chasm’s six aims2 (IOM, 2001).
1 Patient Protection and Affordable Care Act of 2010 as amended. § 3011, 111th Cong., 2d sess. The current version of the National Quality Strategy is available at http://www.healthcare.gov/center/reports/quality03212011a.html#na (accessed June 27, 2011); it adopts the Institute for Healthcare Improvement’s “Triple Aim” (Berwick et al., 2008).
2 The IOM’s six quality aims are safe, effective, patient-centered, timely, efficient, and equitable (IOM, 2001).
The committee finds that no single policy lens is sufficient or comprehensive enough for explicitly framing decisions about the EHB. Figure 3-1 graphically illustrates the four domains and principles associated with those domains. Each of these distinct perspectives—complementary in some cases, overlapping in others, conflicting at times—influences how we think about what health insurance should cover and how it should be implemented.
Economics
A benefit design framework rooted in economics primarily conceives of coverage as insurance— protecting individuals and their families against the risk of unforeseen health care needs, particularly those associated with large expenses (Santerre and Neun, 2010). An economics approach uses markets to promote value and efficiency, relying heavily on these markets to find the equilibrium between price and demand. With respect to the ACA, new markets are being developed for health insurance products that will include the EHB. Decisions about the scope of coverage in the EHB package and product benefit design will affect its success in the market—both whether it can be sustained with private and public funds over time and whether a sufficient number of insurers will participate in the exchange, or otherwise opt to exclusively sell plans not subject to the EHB (e.g., those with a grandfathered status, or self-insured in the large employer market). Insufficient participation of willing purchasers or willing suppliers of insurance products may affect competitiveness as well as require government intervention in the content of packages offered or their price. Ideally, insurers will compete for the estimated 68 million EHB-related purchasers and will do so in a way that provides multiple insurance options at competitive prices.
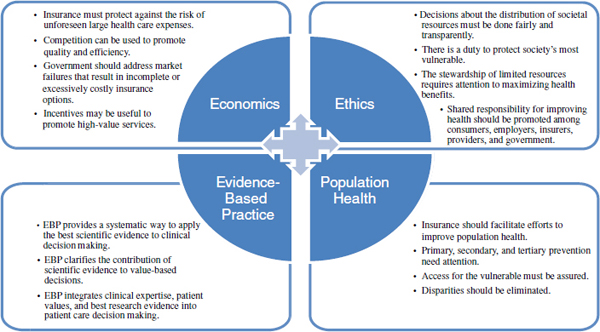
NOTE: EBP = evidence-based practice.
While markets reflect the preferences of purchasers, economists recognize the risks of moral hazard and adverse selection. Moral hazard refers to the situation in which consumers alter their behavior when provided with health insurance (Santerre and Neun, 2010) such as in the case of insurance-induced use in which individuals with insurance seek more care—both appropriate and inappropriate—than they otherwise would (Pauly, 1968). An insurance market is hard to sustain when induced use exceeds premium income, particularly when the benefits do not justify the costs. To remedy this, insurers have implemented cost sharing, but this often reduces appropriate and inappropriate use equally (Siu et al., 1986), or they may opt for managed care arrangements. Yet if the moral hazard is too great, and the cost of mitigating it too high for a given service, private insurers may drop the service from coverage because customers do not want to pay for the moral hazard in a higher premium.
Nonrandom enrollment can result from both adverse selection and risk selection. Benefit choice and design elements by insurers can result in these consequences; consumer choice to enroll or not enroll similarly affects the pool of purchasers and the potential to spread financial risk across a broader-based population. For example, adverse selection occurs “when individuals at greater risk of high health spending are more likely to need and seek coverage, while low-risk individuals are more likely to opt out of coverage” (AAA, 2011, p. 2). A premium spiral typically ensues as the average insured risk increases and results in higher premiums, which, in turn, “may lead to more low-risk individuals opting out of coverage, which would result in even higher premiums. Avoiding such spirals requires minimizing adverse selection and instead attracting a broad base of low-risk individuals, over which the costs of high-risk individuals can be spread” (AAA, 2011, p. 2). The individual mandate was envisioned as the most effective means to maximally broaden the insured population base to help address the current state of cross-subsidization of the uninsured by the insured and minimize adverse selection, while also gaining insurance industry acceptance for the insurance reforms contained in the ACA (Chandra et al., 2011; McGlynn et al., 2010). Furthermore, the ACA also provides three risk-adjustment programs to help address risk selection:3
• Temporary reinsurance,4 which reimburses insurers for the cost of individuals who have unusually high claims;
• Temporary risk corridors,5 which “mitigate the pricing risk that insurers face, when their data on health spending for potential enrollees are limited,” by providing “a government subsidy if insurer losses exceed a certain threshold”—set at 3 percent in this case (conversely, excess gains are also limited with the requirement for plans to pay the government if their gains exceed this threshold); and
• A permanent risk-adjustment program that requires a transfer of funds between health plans that disproportionately enroll low-risk individuals to those that enroll a greater percentage of high-risk individuals, thus eliminating any incentive for competing plans to avoid enrolling these (AAA, 2011, pp. 3-4).
Notably, while risk adjustment reduces the effects of adverse selection between plans based on the relative risk of their enrollees, “it cannot, however, mitigate the effects of adverse selection against the market as a whole if a disproportionate share of low-risk individuals choose not to purchase coverage from any health plan” (AAA, 2011, p. 3). Without a large pool of enrollees, all health costs cannot be fairly distributed or amortized across an entire population.
Risk selection (aka “cherry picking” or “cream skimming”), in contrast to patient-driven adverse selection, is insurer-driven and refers to the use of benefit package design (or other tools) to disproportionately attract healthy individuals. This can result in greater profits for insurers and the potential for insufficient access for the sick in the non-guaranteed issue market; the ACA addresses this potential by incorporating consumer protections
3 § 1343.
4 Beginning in 2014 and ending in 2016 (§ 1341).
5 Beginning in 2014 and ending in 2016 (§ 1342).
(such as, no preexisting condition exclusions,6 guaranteed issue,7 and limiting the medical loss ratio8) and prohibiting the Secretary from making any coverage decisions or designing benefits for the EHB in a way that would discriminate against individuals because of their age, disability, or expected length of life. Implementation bears watching to ensure that benefit design at the plan level does not inadvertently breach these nondiscrimination provisions or create barriers to accessing care.
A variety of benefit design approaches and value-based incentives are being used, as noted in Chapter 2. Cautions have been raised to ensure that incentives have their intended consequences. For example, cost sharing is a means for insurers to involve enrollees in the cost burden of their care and seeks to align incentives with purchase price. Network tiering is a method that encourages enrollees to seek care from designated “high-value” providers, with a lower corresponding co-payment than for “out-of-network” care. Network tiering can increase health disparities if the number of designated high-value providers cannot accommodate the patient load (i.e., they do not accept new patients) or if lack of proximity (in rural areas especially) creates an unreasonable travel burden (Tackett et al., 2011). A similar mechanism applying to pharmaceutical use is formulary tiering. A three-tier plan, for example, could have three co-payment levels; the lowest for generic drugs, the next for formulary (preferred) drugs, and the highest for nonformulary brand name drugs (Joyce et al., 2002). However, if the tiers are based on price alone, it is not a value-based design. In a variant pharmaceutical benefit design called reference pricing, insurers cover “only the prices of low-cost, benchmark drugs in therapeutic clusters that are deemed to be close substitutes for one another in treating specific illnesses. Patients who desire a higher-price substitute in a cluster must then pay the full difference between the retail price of that drug and the reference price covered by the insurer” (Kanavos and Reinhardt, 2003, p. 16). Similarly, such a pricing model is being considered to put the burden on offerers of new technologies to convey why any price increases would be justified over currently available technologies in terms of better outcomes for patients.
Porter remarks, “If value improves, patients, payers, providers, and suppliers can all benefit while the economic sustainability of the health care system increases….Cost reduction without regard to the outcomes achieved is dangerous and self-defeating, leading to false ‘savings’ and potentially limiting effective care” (Porter, 2010, p. 2477). The committee defines value as outcomes relative to costs. In Chapter 2, there is a brief overview of cost drivers for health care spending and insurance premium growth, as well as discussion of unwarranted preference and supply-sensitive variations in care. Market pressures can lead to unnecessary utilization of high-cost services and items without a commensurate benefit—for example, marketing by vendors directly to patients of profitable services that have low health gains over alternatives (e.g., virtual physical exams, high-cost pharmaceuticals). Similarly, interest groups can be potent in trying to force through benefits that evidence does not support. Comparative effectiveness and cost-effectiveness analyses are tools that can be used to compare relative outcomes.
Ethics
A framework rooted in ethics recognizes that no set of metrics can provide a guarantee of ethical actions, but promotes theoretical reflection and conceptual analysis in support of adherence to “ethical norms.” Public examination of clinical effectiveness and priority setting among benefits, combined with the use of ethically guided benefit management processes, will contribute to preserving access to benefits that are the safest and most effective, all while eliminating interventions that have been proven to be ineffective or even harmful. Thus, an ethical framework requires consideration of stewardship of shared population-wide resources and, at the same time, fidelity to the needs of the individual (Daniels and Sabin, 1997, 2008). Stewardship is not just a matter of living within a budget, but of having a broader obligation for the judicious use of resources so that they are available when people who contribute to the resource pool most need them.
The American Medical Assocation’s (AMA’s) Ethical Force Program proposed five content areas directly related to the fairness of a health benefits design and subsequent administration, stating that health care coverage
6 § 1201, amending § 2704 of the Public Health Service Act.
7 § 1201, amending § 2702 of the Public Health Service Act.
8 The medical loss ratio informs consumers and regulators about the percentage of the premium being spent on fees, administration, and profits. The ACA requires insurance companies to spend at least 80 percent or 85 percent of premium dollars on medical care depending on market segment. § 2718 of the Public Health Service Act [42 U.S.C. 300gg–18].
decisions should be (1) transparent, (2) participatory, (3) equitable and consistent, (4) sensitive to value, and (5) compassionate (Box 3-1). Of note, these are not listed in order of importance, but rather in the logical order in which they may be considered in the decision-making process (AMA, 2004).
The five areas suggested are to be advocated for because
1. Transparency is necessary for market accountability;
2. Participatory processes ensure that public concerns are understood and considered, foster a heightened sense of fairness and legitimacy among stakeholders, and additionally promote quality improvement by drawing attention to grievances;
3. Equity and consistency safeguard against inappropriate discrimination (both for legal reasons in setting and enforcing precedents and for promoting public acceptability of a system);
4. Sensitivity to value is rooted in the consequentialist approach of promoting the greatest good; and
5. Compassion is consistent with the health insurance function of protecting against an imbalance in individual risk, requiring that health care resource allocation transcend the formulaic by incorporating flexibility and responsiveness to extraordinary individual circumstances and informing itself on such individual variations (AMA, 2004).
The five areas identified by the AMA represent aspirational targets. They are not easy to achieve in full measure. With transparency, it is not possible to convey all information (particularly with regard to additions or revisions) to all people; participatory processes are often skewed to those who are already empowered, so attention must be placed on minorities (such as those with rare diseases) who may have been heard less often. A focus on consistency requires meticulous record-keeping, which can be challenging and should not hinder progress in incorporating developments when previous decisions are determined to be incorrect or new circumstances are encountered. Sensitivity to value requires a modality for assessment that must ensure the intended effect of the service is considered. Finally, beyond the difficulty of gauging compassion lies the challenge of balancing individual need with stewardship of societal resources, while avoiding inequity in the distribution of those resources (AMA, 2004). The IOM committee concluded that in a large, pluralistic society like the United States, there are no shared principles by which to assign fixed weights to the values the AMA identified. For that reason, as discussed in Chapter 6, the committee posited a central role for a fair process to guide EHB design and administration, and described the role of public deliberation and “accountability for reasonableness” (Daniels and Sabin, 2008) in creating that process.
Transparency: The processes for designing and administering health benefits should be fully transparent to those affected by these processes.
Participatory: Organizationsa should purposefully and meaningfully involve all stakeholders in creating and overseeing the processes for designing and administering health benefits.
Equity and consistency: Processes for designing and administering health benefits should result in similar decisions under similar circumstances.
Sensitivity to value: Processes for designing and administering health benefits should take into account the net health outcomes of services or technologies under consideration and the resources required to achieve these outcomes.
Compassion: The design and administration of health benefits should be flexible, responsive to individual values and priorities, and attentive to the most vulnerable individuals and those with critical needs.
_________
a For example, health insurance exchanges.
SOURCE: AMA, 2004.
Evidence-Based Practice
Evidence drives the evolution of medical practice—from its origins rooted in mythology, to observation, randomized trial, systematic review, and large-scale personalized modeling. In directing this move from the “usual and customary” and “standard of practice” to mixed models of evidence-or expert-based practice, evidence-based medical practice improves the quality of clinical decision making. In addition to positively impacting the health of the population, there is also the potential to “reduce the use of marginal services and control some of the variation in utilization among providers” (Santa and Gibson, 2006, p. 3). Yet current incentives in the health care system—whether related to physician behavior, patient behavior, hospital behavior, or manufacturer behavior—are not necessarily aligned with evidence-based practice (Jacques, 2011), contributing to unexplained and/or unintended variation in medical and health care quality.
Evidence-based practice (EBP) could be fostered through EHB design. In practice, “evidence based medicine is the conscientious, explicit, and judicious use of current best evidence in making decisions about the care of individual patients” (Sackett et al., 1996, p. 71). The use of evidence helps both to define coverage and to determine medical necessity for individual patients. The IOM has published a number of studies stressing the “imperative for change” to improve the quality of health care through the use of evidence and incentives to support its use in practice—noting unsustainable rates of increase in costs, unwarranted variation in the use of services, shortcomings in the health care system’s ability to translate knowledge into practice, and too frequent lapses in applying new technology safely and appropriately (IOM, 2001, 2008, 2011a,b). Numerous studies support findings that unnecessary care is delivered (NEHI, 2008; Schuster et al., 2005), recommended care is not (McGlynn et al., 2003), and that care when delivered inappropriately can be unsafe (Brenner and Hall, 2007; Rosen, 2010; Simpson, 2010).
Recognizable gaps in the evidence base and in the strength of evidence mean that uncertainty remains around decision making for numerous services. Opportunities for coverage with evidence development, as Medicare does (e.g., pancreatic islet cell transplantation), can help if there is a mechanism to collect rigorous treatment and outcomes data. The expected technological explosion from multiple fields may fuel growth in what is known as personalized medicine, and this will add further challenges to applying EBP as the tension between studies based on populations and treatment decisions at the individual level increases. As important as evidence is, care must be taken to ensure that the absence of evidence (different from negative evidence) does not have more impact on specific groups. For example, given the directive to “take into account the health care needs of diverse segments of the population,” if evidence gaps selectively disadvantage particular groups, such as children, disabled persons, minorities or women, special effort should be taken to avoid discrimination.
EBP is understood by clinicians, but has variable acceptability among the general public on a conceptual level, with one study finding “many of these consumers’ beliefs, values, and knowledge to be at odds with what policy makers prescribe as evidence-based health care” (Carman et al., 2010, p. 1400). Furthermore, “When science or the evidence flies in the face of people’s desires or their personal health beliefs,” there can be a backlash from both medical professionals and patients (Good, 2010). However, evidence should help clarify decisions in a more transparent way to the public; in the context of making tradeoffs among benefits, when the choice is living within evidence-based guidelines vs. paying more, or giving up a covered category, people accept the guidelines (Ginsburg et al., 2006). Furthermore, aligning coverage with scientific evidence can be a safeguard against inappropriate discrimination (Rosenbaum, 2011) in the practice of individualized, condition-specific care.
Given the broad range of EBP’s potential applications, it is important to consider how evidence is currently used for decision making within health care (Table 3-1). As noted by Clancy and Cronin (2005), “Increasing the relevance of scientific evidence to clinical and policy decisions relies on both a transparent approach to evaluating
the quality of scientific studies and a broad debate about the interpretation of scientific findings and their optimal application” (Clancy and Cronin, 2005, p. 152). One research area of considerable interest is the question of “appropriate use”—because it is possible to establish that a service has credible evidence for efficacy, with conditions of coverage (e.g., higher vs. lower cost sharing, participation in research to generate evidence) as opposed to considering benefits independently, absolutely, and unconditionally covered.
TABLE 3-1 Uses of Evidence for Decision Making
|
|
||
| Type of Decision | Decision Maker | Role of Evidence |
|
|
||
| Product approval | FDA | Level I |
| Product purchasing, for example, formulary selection | Health plans, PBMs | Level II |
| Clinical decisions | ||
| Practice guidelines | Clinicians | Level II |
| Shared decisions | Clinicians, patients | Level III |
| Assess and improve health care quality | ||
| Internal improvement | Health care organizations | Level II |
| Public reporting | Payers/purchasers; states | Level II |
| Pay for performancea | Payers/purchasers | Level II |
| Choice of plans or providers | Consumers; employers | Level III |
| Select benefits and coverage | Insurers; employers | Levels II-III |
| Organizational and management decisions | Health care organization leaders | Level IV |
| Care optionsb | Individuals; patient and disease groups | Levels III-IV |
|
|
||
NOTES: Level I: rigorous evidence required (absolute requirement); level II: evidence is predominant input when available, supplemented by expert judgment; level III: available evidence is one but not the only input to decisions; level IV: available evidence is limited, other considerations are important. FDA = U.S. Food and Drug Administration; PBM = pharmacy benefit manager.
a Pilot programs.
a Emerging focus.
SOURCE: Clancy and Cronin, 2005. Copyrighted and published by ProjectHOPE/Health Affairs as Clancy and Cronin, Evidence-based decision making: Global evidence, local decisions. Health Aff (Millwood) 1(24):151-162.
Obstacles to increased use of evidence in health benefits design include a perceived lack of sufficient evidence, inability to communicate—with credibility and transparency—to consumers the rationale behind perceived limitations on coverage, inadequate benefit design description, financial and administrative considerations, and resistance to change among providers, vendors, and consumers (Bernstein, 2010; Santa and Gibson, 2006). Among these, lack of adequate information—either because the issue has not been studied or because no positive results have been found—is the most significant challenge encountered in EBP. Evidence-based medical practice is an ever-evolving effort that requires analysis—which can be costly—to develop standards that will guide clinical decision making—often through one or a number of entities charged with reviewing and analyzing evidence (e.g., U.S. Preventive Services Task Force, Cochrane, Agency for Healthcare Research and Quality [AHRQ]).
A related difficulty that arises is in the interpretation of the information available, particularly with regard to cases where there is conflicting evidence, treatment heterogeneity (variable response based on some other factor9), or no consensus about relative effectiveness among services. Indeed, the quality of all evidence must be evaluated—for risk of bias, consistency, precision, directness, and reporting bias. In 2011, the IOM published Clinical Practice Guidelines We Can Trust and Finding What Works in Health Care: Standards for Systematic Reviews, which set standards to improve the quality of clinical practice guidelines (CPG) and systematic reviews (SR), respectively. When the Secretary of HHS and insurers use evidence to make benefit design decisions, they will be working in an evidence environment that is far from ideal, but they should use the best evidence available (IOM, 2011a,b).
Population Health
The IOM has long highlighted the need for a greater focus on population health alongside individual health (IOM, 1988, 2002). The committee agrees that it is necessary to consider the needs of both the individual and the
9 For example, co-morbid condition, gender, or metabolic ability.
overall population when setting priorities (Sabin, 1998) and acknowledges the tension this places on policy makers when making coverage decisions. Population health focuses on improving the overall health status of a community, thus departing from the predominant late 20th century medical care model of focusing interventions only on the individual (Kindig, 2007). The function of health insurance, in this framework, is to encourage access to health-promoting care services, through primary and secondary prevention (e.g., immunizations to reduce transmission of communicable diseases; screenings for conditions such as high blood pressure, type 2 diabetes mellitus, or certain cancers, in which a delay in the initiation of treatment is associated with increased mortality).
The ACA changes the paradigm for preventive health services, because preventive and wellness services (including chronic disease management) are 1 of the 10 categories of care required for the EHB. Furthermore, services that have been rated highly effective by the U.S. Preventive Services Task Force must be offered without patient cost sharing10 (currently 45 preventive services have received a high evidence grade of A or B) (USPSTF, 2010; HHS, 2010).
In 2006, in anticipation of efforts to develop an essential benefits package, Oregon’s Health Services Commission changed its prioritized list’s ranking methodology (discussed in Chapter 2) to incorporate a population focus, moving certain preventive services higher on the list (MedPAC, 2011). Additionally, one study by Thornton and Rice (2008) suggests that population health can be improved when health insurance coverage is extended to the uninsured (a 10 percent increase in insurance coverage of a state’s population was estimated to reduce mortality by 1.69-1.92 percent). Such an effect may not be seen in national statistics if the proportion of the population becoming newly insured is small or the extension of life is minimal, but the expansion of health coverage under the ACA and the insureds’ response to care provide an opportunity to study these effects. Finally, efforts to identify and then eliminate the disparities experienced while accessing health care must accompany any population health approach.
A few states also explicitly assess the impact on the health of their overall population when considering whether certain health benefits should be mandated for insurance packages. Among these, California, by law, undertakes a public health impact analysis of proposed mandated health benefits by collecting data from state registries, state-specific population-based surveys, and national surveys to determine disease prevalence and incidence, impact of benefit coverage on health outcomes (including morbidity, mortality, disability, and quality of life), health care utilization, and how it will reduce premature health and economic loss (CHBRP, 2011; McMenamin et al., 2006). Furthermore, a focus on and improvement of population health is considered to positively influence economic growth (Bloom and Canning, 2000; Mirvis and Bloom, 2008), while having the potential to save money in the long term (Murphy and Topel, 2003).
After considering these policy foundations for explicitly designing coverage, the committee derived criteria for determination of the EHB as the Secretary of HHS requested. Individual criteria go across multiple policy domains—for example, the concept of stewardship is as much of an ethical as an economic criterion. A need for distinction among criteria for three purposes became apparent: first, criteria for assessing the EHB package’s content in the aggregate; second, criteria for assessing individual services for possible inclusion or exclusion in the package; finally, separate criteria for the methods employed in deliberations over composition, whether employed by the Secretary, states, any advisory body, or the public. These criteria promote responsible oversight of the EHB.
Guidance on Content
Figure 3-2 lists the criteria for the aggregate package and individual service assessments for the EHB. These criteria are part of the process, defined by the committee, to define the initial EHB package (Recommendation 1 in Chapter 5) and updating the package (Recommendation 5 in Chapter 9).
With respect to the application of criteria for the overall package, the committee acknowledges that the expected effects for each of these criteria can be along a continuum and multiple metrics could be applied to
10 Only when these services are delivered by a network provider.
measure that effect. For example, estimates by the Congressional Budget Office (CBO), RAND, and others give guidance on expected enrollment numbers, reductions in the uninsured, and changes in average premium prices (CBO, 2011a,b; Eibner et al., 2010; KFF, 2011), which, along with actuarial estimates of specific benefit categories, can be applied in estimating the impact of the contents of various benefit package options. The committee was acutely aware that the affordability of the EHB package would affect market response and has more to say in Chapter 5 on how a projected national average premium can guide EHB package development. The committee recognizes the intent of the ACA to maximize the number of people who are meaningfully insured and thereby the implicit goal embedded in the CBO scoring of the health reform bill for the potential reduction in the number of uninsured and take up of coverage through private means. Monitoring of implementation will inform these and other metrics. The committee recommends development of a monitoring and research agenda in Chapter 7, which should include identification of metrics to monitor the criteria.
Criteria to Guide Content of the Aggregate EHB Package
In the aggregate, the EHB must:
• Be affordable for consumers, employers, and taxpayers.
• Maximize the number of people with insurance coverage.
• Protect the most vulnerable by addressing the particular needs of those patients and populations.
• Encourage better care practices by promoting the right care to the right patient in the right setting at the right time.
• Advance stewardship of resources by focusing on high value services and reducing use of low value services. Value is defined as outcomes relative to cost.
• Address the medical concerns of greatest importance to enrollees in EHB-related plans, as identified through a public deliberative process.
• Protect against the greatest financial risks due to catastrophic events or illnesses.
Criteria to Guide EHB Content on Specific Components
The individual service, device, or drug for the EHB must:
• Be safe—expected benefits should be greater than expected harms.
• Be medically effective and supported by a sufficient evidence base, or in the absence of evidence on effectiveness, a credible standard of care is used.
• Demonstrate meaningful improvement in outcomes over current effective services/treatments.
• Be a medical service, not serving primarily a social or educational function.
• Be cost effective, so that the health gain for individual and population health is sufficient to justify the additional cost to taxpayers and consumers.
• Caveats:
Failure to meet any of the criteria should result in exclusion or significant limits on coverage.
Each component would still be subject to the criteria for assembling the aggregate EHB package.
Inclusion does not mean that it is appropriate for every person to receive every component.
The ACA and HHS’s National Quality Strategy underline the need for better care practices. The committee believes that decisions on covered benefits and benefit design choices can support better care practices. The committee supports an evidence-based and value-based approach to coverage of health services as desirable to maximize the health gains of such services as well as provide patients with the best choice of safe and effective treatments. This argues for development of a shared EHB evidence base, monitoring of access to designated EHB coverage so that benefits supported by evidence are available to those who need them, and being encouraging to new modes of delivery or insurance design that foster value. How one evaluates an entire package or even 1 of the 10 categories of care as being fully evidence-based will be challenging but also argues for more detail in benefit specification (see Chapters 5 and 9) and improvement in the quality of evidence over time. Currently, each insurer, whether public or private, is making decisions on effectiveness of services separately, at times duplicating efforts that might be better directed in cooperative evaluation of research and establishment of clinical policies.
Insurance is a method of pooling the risk of financial loss across a group or a population. This prompts the question, “Which medical services should be paid for using a limited pool of shared funds?” Insurance policies exclude certain benefits; HHS will certainly have to exclude benefits that might be important to certain stakeholders, and these may even be services that have an evidence base that shows at some level they are effective. Most of us will be paying into insurance pools, directly through purchasing insurance and/or indirectly through taxes. Thus, we all depend on the possibility of spending from these shared pools and should want financial protection against catastrophic illnesses and conditions and assurances that care paid for out of shared resources is medically necessary. In contrast to other policy foundations, an insurance/economic frame emphasizes mitigation of short-term risk. Thus, relying on this frame alone is insufficient for establishing the EHB; coverage of prevention services, which are often relatively low cost and whose use can be anticipated, would tend not to be covered under an insurance frame solely.
The ACA puts an emphasis on prevention, and it will be necessary to invest in effective prevention and treatment practices for leading causes of morbidity and mortality to advance population health. In setting that as a goal, however, there must always be, as the ACA requires, attention to diverse segments of society and a spectrum of needs throughout the lifecycle and across a variety of conditions to prevent discrimination in the choice of benefits.
The committee criteria on evaluating individual components reflect current practice of evaluating such services as discussed in Chapter 2. There are many existing groups that evaluate specific services (e.g., BlueCross BlueShield Association’s Technology Evaluation Center, AHRQ Evidence-Based Practice Centers) for effectiveness, although not always using cost and comparative effectiveness as the committee endorses. The committee also emphasizes that the EHB package should focus on medical services (Chapter 4), but that some nonmedical services may add value (i.e., promote health gain for the cost) and are supported by an evidence base of effectiveness. Several caveats are added to this list of criteria; services that fail to meet all of the criteria might be subject to restriction if they are included, for example, through the setting of limits on their duration and scope. Services that meet all of the criteria must still be subject to review as part of a package of benefits. And even when included as a covered benefit, if it is not medically necessary for an individual, then it should not be covered (Chapter 5).
Criteria to Guide the Methods
Repeatedly emphasized to the committee was the importance of having a trustworthy method for defining and updating the EHB. In developing these criteria, the committee identifies the following as key (Figure 3-3)—that the methods be transparent, participatory, equitable and consistent, sensitive to value, responsive to new information, attentive to stewardship, encouraging to innovation, and data-driven. These attributes of methods can apply at many levels, not only the national definition of the EHB, but also how states may make decisions about benefits in their states (Chapter 8 on innovation) and insurer decisions on medical necessity and any subsequent appeals.
The transparency of the EHB process at the national level extends to making the rationale behind choices made for the EHB package public (Chapter 5). Participation can take many forms, but in the context of making tradeoffs among benefits, the committee recommends going beyond usual stakeholder public comment to have formalized deliberation on tradeoffs (Chapters 5, 6, and 9). Equity and consistency are particularly important to ensure that medical necessity decisions are conducted fairly and with transparency, at a minimum, in external appeals (Chapters 5 and 7).
Criteria to Guide Methods for Defining and Updating the EHB
Methods for defining, updating, and prioritizing must be:
• Transparent. The rationale for all decisions about benefits, benefit design, and changes is made publicly available.
• Participatory. Current and future enrollees have a role in helping define the priorities for coverage.
• Equitable and consistent. Enrollees should feel confident that benefits will be developed and administered fairly.
• Sensitive to value. To be accountable to taxpayers and plan members, the covered service must provide a meaningful health benefit.
• Responsive to new information. The EHB will change over time as new scientific information becomes available.
• Attentive to stewardship. For judicious use of pooled resources, budgetary constraints are necessary to keep the EHB affordable.
• Encouraging to innovation. The EHB should allow for innovation in covered services, service delivery, medical management, and new payment models to improve value.
• Data-driven. An evaluation of the care included in the EHB is based on objective clinical evidence and actuarial reviews.
FIGURE 3-3 Criteria to guide methods for defining and updating the essential health benefits (EHB).
Together, these policy foundations, principles, and criteria comprise the committee’s framework for defining and revising the EHB package, in a manner consistent with the ACA.
AAA (American Academy of Actuaries). 2011. Risk adjustment and other risk-sharing provisions in the Affordable Care Act. http://www.actuary.org/pdf/Risk_Adjustment_IB_FINAL_060811.pdf (accessed July 8, 2011).
AMA (American Medical Association). 2004. Ensuring fairness in health care coverage decisions: A consensus report on the ethical design and administration of health care benefit packages. Chicago, IL: American Medical Association.
Bernstein, J. 2010. Using evidence to design benefits. Washington, DC: Academy Health.
Berwick, D. M., T. W. Nolan, and J. Whittington. 2008. The triple aim: Care, health, and cost. Health Affairs 27(3):759-769.
Bloom, D. E., and D. Canning. 2000. The health and wealth of nations. Science 287(5456):1207-1209.
Brenner, D. J., and E. J. Hall. 2007. Computed tomography—an increasing source of radiation exposure. New England Journal of Medicine 357(22):2277-2284.
Carman, K., M. Maurer, J. M. Yegian, P. Dardess, J. McGee, M. Evers, and K. Marlo. 2010. Evidence that consumers are skeptical about evidence-based health care. Health Affairs 29(7):1400-1406.
CBO (Congressional Budget Office). 2011a. Additional information about CBO’s baseline projections of federal subsidies for health insurance provided through exchanges. Washington, DC: Congressional Budget Office.
______. 2011b. CBO’s March 2011 baseline: Health insurance exchanges. http://www.cbo.gov/budget/factsheets/2011b/HealthInsuranceExchanges.pdf (accessed June 30, 2011).
Chandra, A., J. Gruber, and R. McKnight. 2011. The importance of the individual mandate—evidence from Massachusetts. New England Journal of Medicine 364(4):293-295.
CHBRP (California Health Benefits Review Program). 2011. Public health impact analysis. http://chbrp.org/pubhealth.html (accessed September 22, 2011).
Clancy, C. M., and K. Cronin. 2005. Evidence-based decision making: Global evidence, local decisions. Health Affairs 1(24):151-162.
Daniels, N., and J. Sabin. 1997. Limits to health care: Fair procedures, democratic deliberation, and the legitimacy problem for insurers. Philosophy & Public Affairs 26(4):303-350.
______. 2008. Setting limits fairly: Learning to share resources for health. 2nd ed. New York: Oxford University Press.
Eibner, C., F. Girosi, C. C. Price, A. Cordova, P. S. Hussey, A. Beckman, and E. A. McGlynn. 2010. Establishing state health insurance exchanges: Implications for health insurance enrollment, spending, and small businesses. Santa Monica, CA: RAND Corporation.
Ginsburg, M., S. D. Goold, and M. Danis. 2006. (De)constructing basic benefits: Citizens define the limits of coverage. Health Affairs 25(6):1648-1655.
Glied, S. 2011. Testimony to the IOM Committee on the Determination of Essential Health Benefits by Sherry Glied, Assistant Secretary for Planning and Evaluation, HHS, Washington, DC, January 13.
Good, L. B. 2010. Breast cancer screening USPSTF update: An interview with Miriam Alexander, MD, MPH, ACPM President-elect. http://www.medscape.com/viewarticle/714497 (accessed September 22, 2011).
HHS (Department of Health and Human Services). 2010. Preventive services covered under the Affordable Care Act. http://www.healthcare.gov/law/about/provisions/services/lists.html (accessed June 14, 2011).
IOM (Institute of Medicine). 1988. The future of public health. Washington, DC: National Academy Press.
______. 2001. Crossing the quality chasm: A new health system for the 21st century. Washington, DC: National Academy Press.
______. 2002. The future of the public’s health in the 21st century. Washington, DC: The National Academy Press.
______. 2008. Knowing what works in health care: A roadmap for the nation. Washington, DC: The National Academies Press.
______. 2011a. Clinical practice guidelines we can trust. Washington, DC: The National Academies Press.
______. 2011b. Finding what works in health care: Standards for systematic reviews. Washington, DC: The National Academies Press.
Jacques, L. 2011. Coverage policy: Aligning an insurance benefit with evolving evidence. PowerPoint Presentation to the IOM Committee on the Determination of Essential Health Benefits by Louis Jacques, Director of Coverage and Analysis Group, Centers for Medicare & Medicaid Services, Washington, DC, January 13.
Joyce, G. F., J. J. Escarce, M. D. Solomon, and D. P. Goldman. 2002. Employer drug benefit plans and spending on prescription drugs. Journal of the American Medical Association 288(14):1733-1739.
Kanavos, P., and U. Reinhardt. 2003. Reference pricing for drugs: Is it compatible with U.S. health care? Health Affairs 22(3):16-30.
KFF (Kaiser Family Foundation). 2011. A profile of health insurance exchange enrollees. Washington, DC: Kaiser Family Foundation.
Kindig, D. A. 2007. Understanding population health terminology. Milbank Quarterly 85(1):139-161.
McGlynn, E. A., S. M. Asch, J. Adams, J. Keesey, J. Hicks, A. DeCristofaro, and E. A. Kerr. 2003. The quality of health care delivered to adults in the United States. New England Journal of Medicine 348(26):2635-2645.
McGlynn, E. A., A. Cordova, J. Wasserman, and F. Girosi. 2010. Could we have covered more people at less cost? Technically, yes; politically, probably not. Health Affairs 29(6):1142-1146.
McMenamin, S. B., H. A. Halpin, and T. G. Ganiats. 2006. Assessing the public health impact of state health benefit mandates. Health Services Research 41(3p2):1045-1060.
MedPAC (Medicare Payment Advisory Commission). 2011. June 2011 report to the Congress: Medicare and the health care delivery system. Washington, DC: Medicare Payment Advisory Commission.
Mirvis, D. M., and D. E. Bloom. 2008. Population health and economic development in the United States. Journal of the American Medical Association 300(1):93-95.
Murphy, K., and R. Topel. 2003. Diminishing returns? The costs and benefits of improving health. Perspectives in Biology and Medicine 46(3 Suppl): S108-128.
NEHI (New England Healthcare Institute). 2008. How many more studies will it take? Cambridge, MA: New England Healthcare Institute.
Pauly, M. V. 1968. The economics of moral hazard: Comment. The American Economic Review 58(3):531-537.
Porter, M. E. 2010. What is value in health care? New England Journal of Medicine 363(26):2477-2481.
Rosen, A. K. 2010. Are we getting better at measuring patient safety? http://www.webmm.ahrq.gov/perspective.aspx?perspectiveID=94 (accessed June 26, 2011).
Rosenbaum, S. 2011. Statement to the IOM Committee on the Determination of Essential Health Benefits by Sara Rosenbaum, Hirsh Professor and Chair, Department of Health Policy and Health Services, School of Public Health, The George Washington University, Washington, DC, January 13.
Sabin, J. E. 1998. The second phase of priority setting. Fairness as a problem of love and the heart: A clinician’s perspective on priority setting. British Medical Journal 317(7164):1002-1004.
Sackett, D. L., W. M. C. Rosenberg, J. A. M. Gray, R. B. Haynes, and W. S. Richardson. 1996. Evidence based medicine: What it is and what it isn’t. British Medical Journal 312(7023):71-72.
Santa, J., and M. Gibson. 2006. Designing benefits with evidence in mind. Washington, DC: Employee Benefit Research Institute.
Santerre, R. E., and S. P. Neun. 2010. The demand for medical insurance: Traditional and managed care coverage. In Health economics: Theory, insights, and industry studies. Mason, OH: South-Western, Cengage Learning.
Schuster, M. A., E. A. McGlynn, and R. H. Brook. 2005. How good is the quality of health care in the United States? Milbank Quarterly 83(4):843-895.
Simpson, K. R. 2010. Reconsideration of the costs of convenience: Quality, operational, and fiscal strategies to minimize elective labor induction. Journal of Perinatal & Neonatal Nursing 24(1):43-52.
Siu, A. L., F. A. Sonnenberg, W. G. Manning, G. A. Goldberg, E. S. Bloomfield, J. P. Newhouse, and R. H. Brook. 1986. Inappropriate use of hospitals in a randomized trial of health insurance plans. New England Journal of Medicine 315(20):1259-1266.
Tackett, S., C. Stelzner, E. McGlynn, and A. Mehrotra. 2011. The impact of health plan physician-tiering on access to care. Journal of General Internal Medicine 26(4):440-445.
Thornton, J. A., and J. L. Rice. 2008. Does extending health insurance coverage to the uninsured improve population health outcomes? Applied Health Economics and Health Policy 6(4):217-230.
USPSTF (U.S. Preventive Services Task Force). 2010. USPSTF A and B Recommendations. http://www.uspreventiveservicestaskforce.org/uspstf/uspsabrecs.htm (accessed June 14, 2011).












