Biological Control of Undesirable Plants
This chapter provides background on biological control of undesirable plants and covers general issues related to the development, use, and assessment of mycoherbicides. The concept of biological control is introduced, and examples of mycoherbicides that have been approved and registered in the United States and other countries are provided. The chapter considers key attributes of effective mycoherbicides that are in use and important challenges to their development and large-scale use. It provides a brief overview of pesticide regulation in the United States and international considerations for testing mycoherbicides. Attention also is given to a conceptual framework for considering risks to animals and humans posed by biological control agents. Information on specific mycoherbicides against illicit drug crops is provided in Chapters 4 and 5.
Biological control pertains to the deliberate use of living agents (natural enemies) to reduce populations of undesirable species (weeds and insect pests) with minimal nontarget effects. There are two main approaches to biological control of undesirable plants: the classical and bioherbicide approaches. Table 2-1 summarizes the distinguishing features of the two approaches.
The classical (or inoculative) approach is generally used against nonnative invasive species and involves the introduction of an agent from a foreign country into a population of the undesirable species to control its growth and spread. This approach does not provide instantaneous control; rather, control is achieved over months or even years after the introduction of the pathogen (Yandoc Ables et al. 2007). Under consideration since the 1960s (Inman 1971), the classical approach has been in use since the 1970s, when two rust fungi were used: one to control blackberry (Rubus spp.) in Chile (Oehrens 1977) and the other to control rush skeletonweed (Chondrilla juncea) in Australia. The latter case, control of rush skeletonweed by the rust fungus Puccinia chondrillina, is widely recognized as an early example of success in classical biological control
with fungi (Watson 1991; Hajek 2004). Rush skeletonweed is native to Mediterranean Europe and was a problematic weed in Australia until the deliberate introduction of the rust fungus as a control agent. Introduction of the fungus was followed by a rust disease epidemic, which caused widespread reductions in rush skeletonweed plant height, flower numbers, and viable seed production. Rush skeletonweed mortality was over 90% in areas of Australia where the climate was most favorable for infection (Cullen et al. 1973; Cullen 1985). Another successful example of the classical approach is the introduction of Uromycladium tepperianum, a rust fungus from Australia, into South Africa to control the invasive tree Acacia saligna (Morris et al. 1999). This fungal pathogen, which causes severe gall formation and tree death, has reduced A. saligna populations by 90-95% throughout the plant’s range in South Africa; successful results were evident within 8 years after the release of the fungus (Morris 1997; Wood and Morris 2007).
In the United States, three rust fungi have been released during the last 4 decades to control invasive weeds: Puccinia chondrillina to control rush skeletonweed, P. carduorum for musk thistle (Carduus thoermeri), and P. jaceae var. solstitialis for yellow starthistle (Centaurea solstitialis) (Baudoin et al. 1993; O’Brien et al. 2010). P. jaceae var. solstitialis was found to be ineffective in controlling its target weed. P. chondrillina and P. carduorum were found to have caused damage to their target species (reduced plant density and reduced seed production), but these observations were made in field sites where insect species had also been introduced (Supkoff et al. 1988; Baudoin et al. 1993; O’Brien et al. 2010).
TABLE 2-1 Classical vs Bioherbicide Approach
| Feature | Classical (Inoculative) | Bioherbicide (Inundative) |
| Target plants | Exotic, aggressive, widespread, generally perennials | Native or naturalized, generally annuals |
| Biological control agents | Exotic arthropods and pathogens | Locally occurring pathogens (a few insects) |
| Habitats | Marginal lands (such as rangelands) and natural systems | Intensive croplands |
| Host specificity | Very narrow (monospecific) | May be broader |
| Delivery | Small (inoculative) population released | Mass-reared and large inundative applications (releases) |
| Establishment | Several years to become established | Immediate effect |
| Control objective | Sustained long-term control | Transient nonsustainable control |
| Approach | Ecological | Technological |
The bioherbicide (or inundative) approach involves the use of a pathogen that naturally occurs (that is, is indigenous) in a population of a native or naturalized undesirable plant species (target plant). The pathogen is grown in the laboratory, and its inoculum (infective propagules in the form of mycelium or spores of various types) is mass-produced, formulated, made into a standardized product, and applied in a large dose to inundate the target plant in a manner similar to the application of chemical herbicides. For this approach to be successful in controlling an undesirable plant species in commercial crops, a high level of control of the target plant is required within a short period to reduce economic losses (Charudattan 1990; Weston 1999). The approach might require annual application of the pathogen to cause disease or damage in the succeeding populations of the target plant. Plant pathogens or products containing the pathogens that are used in this approach are called bioherbicides. When fungal pathogens are used in this approach, they are referred to as mycoherbicides. The earliest examples of attempts to control undesirable plants with the bioherbicide approach include the unsuccessful use of Fusarium oxysporum on a large scale to control the white form of the prickly pear (Opuntia megacantha) in Hawaii in the 1940s (Fullaway 1954; Wilson 1969) and the successful use of Acremonium diospyri on weedy persimmon (Diospyros virginiana) in the United States in the 1960s (Wilson 1965).
The first mycoherbicide to be registered in the United States by the U.S. Environmental Protection Agency (EPA) was DeVine in 1981, which is composed of Phytophthora palmivora strain MWV. It was developed in the 1960s and 1970s by researchers working for the Florida Department of Agriculture and Consumer Services Division of Plant Industry to control milkweed vine, or stranglervine (Morrenia odorata), a major nonnative weed in citrus groves (Charudattan 1991). P. palmivora causes root rot, wilting of the whole plant, and plant death. In field trials, a single application of DeVine was reported to reduce milkweed vine population by over 90% within 1-2 years (Woodhead 1981).
In 1982, another mycoherbicide, Collego (Colletotrichum gloeosporioides f.sp. aeschynomene), was registered in the United States. Collego was developed by the Upjohn Company in collaboration with researchers at the University of Arkansas and the U.S. Department of Agriculture Agricultural Research Service for the control of northern jointvetch (Aeschynomene virginica) in rice and irrigated soybean in Arkansas and neighboring rice-growing states (TeBeest 1985). The mycoherbicide was reregistered by EPA in 1992 and again in 2006, the latter time under the label Lockdown (Yandoc Ables et al. 2007).
Bioherbicides can constitute an alternative to mechanical methods and chemical herbicides in the control of undesirable plants. They can help to mitigate the adverse effects of chemical herbicides, such as ground and water contamination, nontarget effects, and development of herbicide resistance. They could be a cheaper alternative to expensive mechanical controls and help to reduce some undesirable side effects, such as acceleration of soil erosion from frequent cultivation. The cancellation of registration of several chemical herbicides, the lack of registered herbicides for some nonagricultural and nonnative
invasive weeds, the high cost of controlling undesirable plants in natural areas, and the need for weed-control methods for organic production systems are other reasons for developing bioherbicides (Yandoc Ables et al. 2007).
In the United States, overt interest in using mycoherbicides to control illicit-drug crops dates from the late 1970s, when Hildebrand and McCain (1978) suggested using Fusarium oxysporum f.sp. cannabis to control cannabis. In the 1990s, the feasibility of using three fungi—two specialized forms of F. oxysporum and Crivellia papaveracea (formerly called Pleospora papaveracea)—as mycoherbicides for the control of illicit narcotic crops (coca, cannabis, and opium poppy) was further investigated by scientists in the United States, Kazakhstan, and Uzbekistan (see Chapters 4 and 5 for details). The use of those pathogens was regarded as an option for eradicating illicit-drug crops because of their purported ability to cause serious damage to the crops, their specificity and narrow host range (they would have minimal nontarget effects), and the likelihood that they would survive in the soil for an extended period and cause disease in succeeding crops of the target plant species (Hildebrand and McCain 1978; Tiourebaev et al. 2001). Mycoherbicides were also promoted as a safer alternative to chemical herbicides (UNODC 2002). Another reason for considering the use of biological control is the need to replace the commonly used eradication methods for illicit crops (mechanical and manual removal and aerial and ground application of chemical herbicides) or to augment the level of control achieved with these methods.
KEY ATTRIBUTES OF EFFECTIVE MYCOHERBICIDES IN USE
Registered mycoherbicides, their current status, and the reasons for discontinued production and use are presented in Table 2-2. Although 12 plant pathogens were registered worldwide as mycoherbicides during the last 3 decades, only four—Chontrol, Collego (now Lockdown), DeVine, and Sarritor—have been used with measurable success. An important reason for that small number is mainly economic (economically nonsustainability in the marketplace), although lack of consistent efficacy is also a major reason. Five of 62 bioherbicide programs (involving fungi, bacteria, and a virus) have produced successful products that are used to a certain degree (Charudattan 2005). The remainder, about 92% of the projects, were abandoned because of lack of efficacy, because of inconsistency of performance, or because they were not profitable. Synergy between research and industry has been instrumental in the successful development of registered mycoherbicides. As pointed out by Bailey and Falk (in press), the registered mycoherbicides are the result of a linking of research developments to a series of key management decisions based on a “stage and gate” process used in business models. To be successful, the proposed mycoherbicides for drug crops should follow a similar research-industry partnership.
TABLE 2-2 Status of Registered Mycoherbicides (2010)
| Pathogen | Target Plants | Trade Name | Status | Reference |
| Alter naria destruens strain 059 | Cuscuta spp., such as dodder, swamp dodder, large-seed dodder, field dodder, and small-seed dodder | Smolder | Registered to Sylvan Bioseiences in 2005 (United States) | EPA 2005a Personal communication, Mark Wach, Sylvan Biosciences, Kittanning, PA (August 16, 2010) |
| Not currently registered and marketed by the company | ||||
| Chondrostereum purpureum strain PFC2139 | Sitka, red alder, and other hardwood species in rights-of-way and forests (used as a stump treatment) | ChontrolTM | Registered in 2004 (Canada); 2004 (United States) | Health Canada 2011a; EPA2004a. b |
| Chondrostereum purpureum strain HQ1 | Birch, piii-cherry, poplar/aspen, maples, and speckled alder growing in rights-of-way, wood lots, and conifer plantations (stump treatment) | MycoTechTM Paste | Registered in 2002 (Canada); canceled in 2008 | EPA 2005b,c |
| MycoTech strain HQ1 Concentrate | Registered in 2005 (United States) Conditional registration in 2005 (United States) | |||
| Colletotrichum acutaturn | Silky hakea (Hakea sericea) | Hakatak | Registered in 1990 (South Africa); registration was allowed to lapse in 1991 because of budget cuts by the main user and the limited market for the product | Morris et al. 1999 |
| Currently, the Agricultural Research Council Plant Protection Research Institute (South Africa) is supplying a small number of farmers and biocontrol implementation officers with dried spore preparation of the fungus | Personal communication, Andries Fourie, ARC-PPRI, Stellenbosch. South Africa (June 2010) | |||
| Pathogen | Target Plants | Trade Name | Status | Reference |
| Colletotrichum gloeosporio ides f.sp. aschynomene ATCC 20358 | Northern jointveteh (A eschynom en e virgin icd) in rice | Collego(1982) Lockdown (2006) | Registered in 1982; 1993 data call-in required additional product data; reregistered as Lockdown m 2006 (United States) | EPA 1997 |
| Colletotrichum gloeosporio ides f.sp. cuscutae | Dodder (Cuscuta spp.) | Luboa | Registered 1111963 (China); not available commercially in China suice 1980 | Personal communication. Sheng Qiang. Nanjing Agricultural University, Nanjing, China (July 7, 2010) |
| Colletotrichum gloeosporio ides f.sp. malvae | Rpund-leaved mallow (Malva pusiUd) in field crops | BioMal | Registered iii 1992 (Canada); registration is currently inactive because it is not bemg manufactured and sold | Personal communication. Karen Bailey, Agriculture and Agri-Food Canada, Saskatchewan, Canada (July 2010) |
| Commercialization plans for BioMal were discontinued because of unfavorable market conditions and high production costs | Bioline International 1995 | |||
| Cylindrobasidiom laeve | Black wattle (Acacia mearnsi): golden wattle (A. pycnantha) | Stumpout | Registered ui 1997 (South Africa) as a fungal inoculant; registration allowed to lapse in 2005 because of problems with annual registration process; inoculum is still produced and supplied on demand (a public-good service) | ARC 2010 Personal communication. Alan Wood, ARC- PPRI. Stellenbosch, South Africa (June 2010) |
| Phytophthora palmivora MWV | Strangler vine or milkweed (Morrenia odorata) in citrus | DeVine | First mycoherbicide to be registered and marketed in United States (1981); still registered as of 2006 to Valent Biosciences Corporation | EPA 2006a,b |
| No longer available, because of small market niche | Ridings 1986 | |||
| Not included in the [Valent Biosciences Corp. online agricultural products list | Valent 2010 | |||
| Puccini a canaliculate | Yellow nutsedge (Cyperus escutentus) | Dr. BioSedge | First rust fungus to be registered as a bioherbieide although it cannot be mass-produced on nonliving, artificial substrates | Phatak et al. 1983 |
| The product was not offered for sale within 1-2 years after registration, because of marketing considerations | Personal communication, Sharad Phatak, University of Georgia (to R. Charudattan) | |||
| The product is not available commercially, because of difficulty in mass production | Hajek 2004 | |||
| Puccinia thlaspeos strain woad | Dyer’s woad (Isatis tinctoria) | Woad Warrior | Registered in 2002 (United States) | EPA 2002 |
| Sclerotinia minor IMI 3144141 | Dandelion (Taraxacum officinale) in turf | Sarritor | Registered in 2007 (Canada) | Health Canada 2011c |
| U.S. registration pending | ||||
| Currently manufactured by Sylvan Biosciences | Sylvan Bio 2009 | |||
A brief review of a successful mycoherbicide, Collego (Lockdown), sheds some light on the attributes of a successful mycoherbicide and provides lessons to guide the debate on mycoherbicides for drug crops. The Collego fungus (Colletotrichum gloeosporioides f.sp. aeschynomene) causes a disease characterized by lesions on stems, petioles, and leaves of northern jointvetch, an annual plant and a weed of rice and irrigated soybean in Arkansas, Mississippi, and Louisiana. Lesions expand rapidly, and the fungus colonizes tissues beneath the lesions and grows laterally and vertically on the hollow stems. The coalescence of lesions results in the girdling of the stem and the death of plant parts above the lesions (TeBeest et al. 1978). In the field, Collego is applied shortly after northern jointvetch emerges above the crop canopy, when it is applied from a fixedwing, crop-sprayer aircraft. The product is formulated to permit appliers to simply add each unit of the dry product to an appropriate number of gallons of water (10 gal/acre). The spray suspension is adjusted to provide 2 million spores per milliliter (94 billion spores/acre). The final product consists of 15% fungus and 85% inert diluents. On the average, the amount of material required for an application is often only 20-40 g/acre (TeBeest and Templeton 1985). The application is intended not to control (kill) northern jointvetch but to establish the pathogen on the weed so that it can produce a new crop of several million spores (secondary inoculum) in each lesion. Pathogens that lack the ability to multiply from an initial application are generally not suitable as mycoherbicides.
C. gloeosporioides f.sp. aeschynomene can persist from year to year on infected northern jointvetch stubble (TeBeest 1982). It infects seeds, and seed infections can give rise to seedling infections (TeBeest and Brumley 1978). However, it does not persist as spores in soil or rice irrigation water in the absence of the host plant for more than several months, so annual applications of Collego are necessary to provide acceptable levels of northern jointvetch control.
An initial study suggested that the host range of C. gloeosporioides f.sp. aeschynomene was limited to northern jointvetch and Indian jointvetch (Aeschynomene indica), of which the latter was highly resistant to infection by the fungus (Daniel et al. 1973). However, later tests showed that seven genera of Fabaceae were susceptible (TeBeest 1988; Weidemann et al. 1988; Weidemann and TeBeest 1990). Because those genera were not found in the area or fields to be inoculated, the newly recognized broader host range did not affect registration or its requirements.
Studies show that the commercial strain (ATCC 20358) of C. gloeosporioides f.sp. aeschynomene is not necessarily competitive with other strains of the fungus that occur naturally in the field (Yang and TeBeest 1995; Luo and TeBeest 1997, 1998, 1999). Another virulent isolate of C. gloeosporioides f.sp. aeschynomene easily replaces the commercial strain on northern jointvetch after coinoculation (Ditmore et al. 2008); the replacement occurs within several weeks of inoculation and might account for the inability to find the commercial strain in the field after discontinuation of its use.
Thus, the success of Collego can be attributed to a combination of host specificity, the virulence and aggressiveness of the strain and the susceptibility
of its host, the conducive environment in which it is used, the prodigious amounts of secondary inoculum produced on lesions that result from primary infections, increase and the greater dispersal in the host population relative to other strains, the ease of formulation and reasonable application requirements, and the rapid decline from the environment after the host is destroyed or Collego use is discontinued.
CHALLENGES TO AND CONSTRAINTS IN THE DEVELOPMENT AND LARGE-SCALE USE OF MYCOHERBICIDES
Collego exemplifies a highly successful mycoherbicide, but an analysis of the examples of failed mycoherbicides reveals a number of issues in the development and use of mycoherbicides, including the cost and feasibility of mass production, shelf-life, and feasibility of application of the mycoherbicide (Weaver et al. 2007). Many market considerations—such as the cost of the product, competing products, market size, and user preferences—that impinge on the commercialization of mycoherbicides might not apply if policy rather than market considerations dictated the development and use of mycoherbicides for illicit-drug crops.
Moreover, under the existing regulatory guidelines, scientific data in support of efficacy, safety, and statement of formula claimed in the application for EPA registration should be gathered on a strain of the pathogen identified as the mycoherbicide active ingredient. It is also necessary to characterize this strain for environmental-monitoring purposes and to deposit a voucher specimen in a leading culture collection, such as the American Type Culture Collection. For those reasons, the process of gathering data for EPA registration begins with a designated isolate or strain and typically requires years of intensive research and development efforts after the initial demonstration of the potential of a candidate pathogen. For example, Collego underwent 13 years of precommercial studies of proof of concept, small-scale efficacy assessment, and host-range determination in addition to 3-5 years of industrial research and development, trials of efficacy on a large scale for an experimental-use permit, gathering of registration data, and registration.
One constraint on the development of mycoherbicides for large-scale use is the need to find strains that will reliably infect the target plant under a variety of moisture and temperature conditions. Most fungal plant pathogens, including those used as mycoherbicides, require a combination of adequate moisture on plant surfaces (humidity and free water) and appropriate temperature (TeBeest and Templeton 1985; Greaves et al. 1989; Heale et al. 1989) for germination, infection, and disease development. Several fungal pathogens evaluated as potential mycoherbicides have been found to require at least 10 hours of moisture (generally from overnight dew formation) to cause disease at magnitudes that sufficiently damage the target plant (Boyette and Walker 1985; Makowski 1993; McRae and Auld 1988; Wymore et al. 1988; Morin et al. 1990; Zhang et al.
2002; Peng et al. 2004; Stewart-Wade and Boland 2004). Inadequate moisture and high temperature can prevent fungi from infecting plants; many mycoherbicide agents are ineffective when used in areas that have infrequent or minimal rainfall and low relative humidity. Studies done in controlled environments have shown that mycoherbicides can be less effective at some temperatures. C. gloeosporioides f.sp. aeschynomene causes rapid infection on northern jointvetch at 20-32°C and optimally near 28°C; disease development is restricted at 36°C, and this indicates the crucial role of temperature in disease development (Te-Beest 1991). Solar radiation can also affect the efficacy of mycoherbicides. A study by Stojanovic et al. (1999) showed that germination and germ-tube length of C. gloeosporioides were reduced by exposure to the sun. Ghajar et al. (2006) reported that UV-B exposure at some levels can deactivate spores or delay spore germination of the fungi Colletotrichum orbiculare and Plectosporium alismatis. The adverse effects of low moisture, suboptimal temperature, and solar radiation are generally mitigated by using formulations (e.g., liquid concentrates, emulsions, pellets).
Another important consideration in the development of a mycoherbicide is the possible constraint imposed by the target plant. Over time, the use of a biological control agent might select for plant genotypes that are resistant to the agent. The genotypes may be present in the existing plant population, they may emerge through mutation of existing plant genotypes, or they may be imported by agriculturists who are invested in the cultivation of the plants. There has been no report of a plant exhibiting resistance to a mycoherbicide over time, as happens with chemical herbicides, but it is known that naturally resistant plant varieties or biotypes exist. For example, three leaf forms of rush skeletonweed (C. juncea) occur in Australia, each with a different susceptibility to the classical biological control agent P. chondrillina. After the introduction of the biocontrol fungus, the prevalence of the most susceptible form A was reduced, but the more tolerant forms B and C became more widespread (Cullen and Groves 1977; Cullen 1978; Burdon et al. 1981). P. chondrillina was also released in western U.S. states to control C. juncea (Adams and Line 1984; Emge et al. 1981; Lee 1986; Supkoff et al 1988); as in Australia, the differences in the susceptibility of C. juncea biotypes required the introduction of more than one strain of the rust pathogen (Emge et al. 1981; Lee 1986).
Other challenges to effective control by a mycoherbicide are the effects of plant age and vigor on disease development. In some plant species, the seedling stage is the most susceptible to pathogen attack; in others, the mature plant is more susceptible (Graham et al. 2007; Holcomb 1982; Rosskopf et al. 2005; Watson and Wymore 1990). A vigorous target plant may recover quickly from nonlethal foliar infection (Auld and Morin 1995); two weeds targeted for mycoherbicidal control, velvetleaf (Abutilon theophrasti) and round-leafed mallow (Malva pusilla), reportedly overcame infection on the leaves and upper stem by growing axillary shoots (Makowski 1993; Wymore et al. 1988).
Finally, producers of illicit-drug crops have an incentive to prevent damage to their crop yields and should be expected to develop countermeasures that
reduce the efficacy of mycoherbicides. Such countermeasures could include the use of fungicides or soil fumigants to kill the mycoherbicide strains directly or the cultivation of plant varieties that are resistant to the mycoherbicides.
PESTICIDE REGULATION IN THE UNITED STATES
In the United States, the regulation of pesticides and their use is administered by EPA under the authority of the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA) and the Federal Food, Drug, and Cosmetic Act (FFDCA). EPA registers mycoherbicides as biopesticides, and the proposed mycoherbicides for illicit-drug crops would fall under the EPA registration requirement. By EPA’s definition, “[b]iopesticides include naturally occurring substances that control pests (biochemical pesticides), microorganisms that control pests (microbial pesticides), and pesticidal substances produced by plants that contain added genetic material (plant-incorporated protectants)” (EPA 2011). Microbial pesticides consist of microorganisms (including fungi) that are somewhat specific to their target pests.
EPA may require an experimental-use permit for field-testing of microbial pest-control agents, including ones that are genetically altered or nonindigenous. Before field trials of such an agent are initiated, the research organization, company, or individual developing it must submit a notification to EPA so that the agency can determine whether an experimental-use permit is required.
The U.S. Department of Agriculture’s Animal and Plant Health Inspection Service (APHIS) regulates the import of organisms that are genetically engineered or are considered as plant pests. The latter category includes nonnative fungi that are intended for development as mycoherbicides. An APHIS permit is required to introduce such regulated organisms into the United States and for their interstate transport.
FIFRA authorizes EPA to review and register pesticides for specific uses and to suspend or cancel registrations when a use would pose an unreasonable risk. FFDCA authorizes EPA to establish tolerances, the maximum pesticideresidue concentrations allowed to remain in or on foods or animal feeds. Under the Food Quality Protection Act as amended in 1996, the evaluation of risk includes provisions for protecting infants and children to ensure reasonable certainty of no harm. Consideration is given to aggregate exposure from multiple routes, cumulative risk from related pesticides, and the potential for endocrinedisruption effects.
Data required for the registration of biochemical and microbial pesticides by EPA are published in the Code of Federal Regulations (40 CFR 158, Subpart V). EPA’s requirements and guidelines for testing (Series 885) are provided in Appendix B of the present report. The requirements include:
• Product analysis, including product chemistry and composition, analysis and certified limits, and physical and chemical characteristics.
• Pesticide-residue analysis, including chemical identity, residue nature and analytic methods in plants and animals, storage stability, and magnitudes in plants, food, and water.
• Toxicity, which involves three tiers of testing (see Box 2-1).
• Toxicity and pathogenicity to nontarget organisms and environmental fate, which involves four tiers of testing (see Box 2-1).
In compliance with the Endangered Species Act of 1973, EPA must also assess potential harm to endangered or threatened species from pesticides that it registers.
Toxicity Testing:
• Tier I: includes acute oral and pulmonary toxicity and pathogenicity tests, cell culture for the technical grade of the active ingredient, five types of acute toxicity tests (oral, dermal, and inhalation toxicity and dermal and eye irritation), and hypersensitivity tests.
• Tier II: consists of acute toxicity and subchronic toxicity-pathogenicity tests; these are required on the basis of the substantial route-specific toxicity, infectivity, or unusual persistence found in Tier I.
• Tier III: consists of tests for reproductive fertility, carcinogenicity, immunotoxicity, and infectivity-pathogenicity analysis; tests are conditionally required on the basis of findings in Tier I and Tier II.
Toxicity and Pathogenicity Testing:
• Tier I: generally includes studies of effects on birds, freshwater fish, invertebrates, and honeybees; other tests might be required on the basis of potential exposure and use; nontarget-plant testing is required if the microbial pesticide is taxonomically related to a known plant pathogen; nontarget-insect testing is not required unless the mechanism of pesticidal action on the target insect pest is through infectivity; depending on the results of Tier I, Tiers II-IV might be required.
• Tier II: includes environmental expression.
• Tier III: includes avian chronic pathogenicity and reproduction tests, aquatic-invertebrate range and ecosystem tests, and fish life-cycle tests.
• Tier IV: includes terrestrial wildlife and aquatic field tests and simulated or field tests for birds, mammals, aquatic animals, insect predators, parasites, insect pollinators, and plants; Tier IV testing might be required either for registration or for postregistration monitoring.
See Appendix B for more details.
Although the EPA requirements for pesticide registration (under which the requirements for biopesticides fall) do not include product-performance data (efficacy data), EPA reserves the right to request performance data case by case during the registration process (Part 158 – Data Requirements for Pesticides, Subpart E – Product Performance). In other countries, such as Canada and the United Kingdom, agencies that regulate pesticides (including biopesticides) require submission of product-performance data as part of the registration process.
Pesticides must be registered by both EPA and states before use. States may place more restrictive requirements on pesticides than EPA. Most states conduct a review of pesticide labels to ensure compliance with federal labeling requirements and any additional state restrictions on use. States that have adopted and are implementing pesticide-use regulations may be delegated primary enforcement responsibility for use violations. Enforcement may also be delegated to states under a cooperative agreement with EPA in connection with specific pesticides.
In the United States, the National Environmental Policy Act (NEPA) requires all federal agencies to consider the environmental impact of any proposed agency action before taking such action and to prepare an environmental-impact statement for any proposed action that is expected to affect the environment substantially (42 USC § 4332(2)(C)). Although NEPA applies to domestic federal activities, it also has extraterritorial applications in some instances, through Executive Order 12114, related to environmental effects abroad of major federal actions (44 FR 1957, 3 CFR, 1979 Comp., p. 356). Executive Order 12114 requires that responsible officials of federal agencies be informed of environmental considerations and take those considerations into account when making decisions on major federal actions that could have environmental impacts anywhere beyond the borders of the United States. NEPA case law has reinforced the need to analyze environmental impacts of federal actions, including the decision-making process, regardless of geographic boundaries (Eccleston 2008).
CONSIDERATIONS FOR INTERNATIONAL
TESTING AND USE OF MYCOHERBICIDES
The International Organization for Biological Control of Noxious Animals and Plants (IOBC) is a professional organization that promotes the development of biological control and its application in integrated pest management. IOBC serves as a clearinghouse for information on biological control, organizes conferences and symposia, and publishes a journal. It has worked with various organizations in developing standards for testing of pesticides and guidelines for transport and release of biological control agents.
At the international level, applicable guidelines and requirements for testing, approval, and application of mycoherbicides against illicit crops fall under the International Plant Protection Convention (IPPC), the International Standards for Phytosanitary Measures (ISPM), and possibly the Biological Weapons
Convention of 1972 (BWC). The IPPC (ratified by the United States in 1972 and amended in 1987) creates an international framework to prevent the spread and introduction of plant and plant-product pests. It is based on exchange of phytosanitary certificates between importing and exporting countries’ national plant protection offices (NPPOs). The provisions of the IPPC extend to any organism capable of harboring or spreading plant pests, particularly where international transportation is involved (Article I of the IPPC, 1997). NPPOs established according to the IPPC have authority in relation to quarantine control, risk analysis, and other measures to prevent the establishment and spread of invasive alien species that, directly or indirectly, are pests of plants. The IPPC further provides that NPPOs have authority to prohibit or restrict the movement of biological control agents and other organisms that might pose a threat to plants.
Specific guidelines are found in the ISPM, promulgated by the UN Food and Agriculture Organization. ISPM No. 3 “provides guidelines for risk management related to the export, shipment, import and release of biological control agents and other beneficial organisms…. The standard addresses biological control agents capable of self-replication (including … pathogens such as fungi …) and includes those packaged or formulated as commercial products. Provisions are also included for import for research in quarantine facilities of nonindigenous biological control agents and other beneficial organisms” (FAO 2006a, p. 25). However, the standard does not address genetically modified organisms or issues specifically related to biopesticide registration.
Responsibilities relating to ISPM No. 3 (2005) are held by contracting parties, NPPOs, or other responsible authorities and by importers and exporters. The standard delineates the responsibilities of the NPPOs of the exporting and importing countries to implement and document adherence to the standard. Those responsibilities include carrying out pest risk analysis of biological control agents before import or release; ensuring compliance with phytosanitary import requirements; overseeing appropriate documentation relevant to the export, shipment, import, or release of biocontrol agents; ensuring that biocontrol agents are transported appropriately; and encouraging the monitoring of released agents (Vapner and Manzella 2007).
The 2005 ISPM revision also provides recommended guidelines for safe use of biological control agents and other beneficial organisms. In addition to phyosanitary concerns, the standard includes guidelines concerning potential effects on nontarget organisms and possible consequences to habitats or ecosystems (FAO 2006a). ISPM No. 3 refers to other standards on pest risk analysis (ISPM No. 2, Guidelines for Pest Risk Analysis; and ISPM No. 11, Pest Risk Analysis for Quarantine Pests Including Analysis of Environmental Risks and Living Modified Organisms) (FAO 2006b, c), which provide the fundamental processes for carrying out the relevant pest risk assessments, including determination of environmental risks.
The 1972 BWC, as ratified by the United States in 1975, provides that state parties to the convention undertake “never … to develop, produce, stockpile or otherwise acquire or retain: (1) Microbial or other biological agents …
of types and in quantities that have no justification for … protective or other peaceful purposes; (2) Weapons, equipment or means of delivery designed to use such agents … for hostile purposes or in armed conflict.” However, the BWC does allow for “international exchange of bacteriological (biological) agents and toxins and equipment for the processing, use or production of bacteriological (biological) agents and toxins for peaceful purposes.” That provision has been interpreted as excluding biological control agents used in agriculture, presumably even mycoherbicides used by consenting governments against illicit crops.
However, possible legal implications of using mycoherbicides in border regions, where the potential for drift into nonconsenting countries exists, need to be considered. For example, Ecuador recently filed a lawsuit against Colombia in the International Court of Justice in The Hague, Netherlands, seeking to end Colombia’s application of chemical herbicide against coca crops growing along the border between the two countries (Ecuador v Colombia, pending). Ecuador claimed that herbicide drift had killed legal crops in Ecuador and resulted in illness of Ecuadoreans living near the border. It is likely that use of mycoherbicides in similar circumstances could provoke a similar suit. There are potential international legal implications, which should be explored, about whether intentional application of a mycoherbicide that drifts into an unwilling country would still fall under the “peaceful purposes” exemption of the BWC.
RISK TO NONTARGET PLANTS AND ORGANISMS
This section reviews some aspects of the scientific literature regarding potential exposure pathways and risks to nontarget plants and organisms (ecological receptors) posed by the introduction of biological control agents and provides a conceptual model to describe the pathways at a specific site (insofar as that is possible given current data constraints).
Review of Ecological Pathways and Risks
Evaluations of potential ecological effects of biological control agents usually recognize that information on host specificity alone is inadequate for identifying and addressing risks because the risks are often propagated through the food chain or express themselves in other indirect effects (such as changes in competition or displacement of prey). Although much of this work addresses risk from classical control approaches (e.g., Wright et al. 2005; Seymour and Veldtman 2010), many of the conclusions are applicable to the bioherbicide approach. Some information is specific to mycoherbicide application and includes assessment of its effects on community levels of organization, such as changes in fungal community structure in the soil as a result of the introduction of a fungal biocontrol agent (e.g., Schwarzenbach 2008).
Some general observations on the potential for ecological exposures and risks posed by the application of biological control agents to control weeds or plant pathogens or pests are emerging:
• The potential for indirect risks to nontarget species even if the biological control agent is highly host-specific. Pearson and Calloway (2005) reviewed case studies demonstrating the nontarget effects of biological control agents and evaluated them in the context of theoretical work in community ecology. Their analysis suggests that the magnitude of the indirect effects on nontarget organisms is indirectly proportional to the abundance of the biological control agent; this suggests that the more efficacious agents are less likely to cause indirect effects. They conclude that “[b]iological control agents that greatly reduce their target species while remaining host-specific will reduce their own populations through density-dependent feedbacks that minimize risks to nontarget species” (Pearson and Calloway 2005, p. 288).
• The classical biological-control literature suggests that a high level of sophistication is available for assessing risks. The methods include population models (e.g., Johnson 1994) and the application of probabilistic techniques (e.g., Wright et al. 2005). Such techniques have not been explicitly used to assess the risks associated with the introduction of mycoherbicides, but the principles derived from the classical control literature could be transferred to such assessments. The application of such techniques requires a detailed understanding of the toxicology, environmental fate, and biological interactions of the proposed strains of mycoherbicides. For example, using a probabilistic risk assessment of the application of mycoherbicides would require detailed statistical knowledge of the range and type of distribution of measurements that described the biology and ecological interactions of the mycoherbicides (for example, the range and distribution of values that describe toxic responses of nontarget organisms, temporal persistence, and environmental dispersal).
• The construction of food webs helps in assessing the safety of biological control agents. Willis and Memmott (2005, p. 299), working with examples of classical biological control, recommend “that constructing and analyzing food webs may be a valuable addition to standard biological control research techniques, as they offer a means of assessing the post-release safety of control agents…. [F]ood webs can be used to generate testable hypotheses regarding indirect interactions between introduced agents and non-target species.”
• Establishment and dispersal of a biological control agent are critical properties that contribute to the potential for indirect ecological effects. Van Lenteren et al. (2003), in their development of an environmental risk assessment of inundative biological control, note that the critical issues are the probability of attack on nontarget organisms, the dispersal of the biological control agent, and the establishment capacity of the agent. They state that the time scales for establishment are critical and might range from one generation (no reproduction) to seasonal or long-term survival and reproduction. Evaluation of the potential
for establishment requires knowledge of the abiotic and biotic requirements of the control agent’s life cycle and knowledge of the capacity of the area of release to meet the requirements. They recognize that it might be necessary to conduct laboratory and field tests to determine and prove whether a control agent is capable of establishing itself in a particular area. They also describe the range of potential indirect effects of changes in competition and energy flow due to the introduction of a biological control agent.
• Modeling and field studies provide site-specific methods for assessing risk, especially risk of increased load of mycoherbicide inoculum, to nontarget areas. De Jong et al. (1990) describes a risk-analysis method for biocontrol with mycoherbicides that incorporates field studies, surveys, and air-dispersal model simulations for estimating the dispersal of spores to nontarget areas and some preliminary assessment of the relative inoculum load to these areas. The method was applied to assess the risk of a mycoherbicide application to downwind market garden crops by calculating safety zones on the basis of the ratio of added to naturally present spores (De Jong et al. 1999, 2002). Bourdôt et al. (2000) described field experiments to assess the survival of the mycoheribicde in soils in such downwind areas. The studies demonstrate the feasibility and the level of effort necessary for analyzing risks associated with the application of a particular mycoherbicide in a specific area and risks to specific, local nontarget organisms.
• Recent reviews identify and categorize the information necessary for conducting risk assessments of biological control agents. McCoy and Frank (2010) address the question of how risks associated with biological control agents should be estimated (largely on the basis of the introduction of such agents into Florida). They concluded that the ability to assess risks associated with the introduction of biological control agents is related primarily to the understanding of how an agent will behave after application. They note that this understanding is often based on inadequate knowledge derived from inadequate testing of the specificity of the biological control agent, inadequate appreciation of basic ecological relationships, and a rush to solve acute pest problems without proper consideration of the consequences (for example, the consequences of using only moderately effective agents or of releasing highly mobile agents).
Conceptual Model
Risk assessments generally involve four major steps: hazard identification, dose-response assessment, exposure assessment, and risk characterization. Hazard identification is an assessment of whether an agent causes an adverse effect and is typically based on laboratory and field observations of adverse effects and exposures to the agent. Dose-response assessment involves evaluating the relationship between the dose of the agent and the incidence of the adverse effect in the population of concern. Exposure assessment involves the prediction of route and level of exposure that might be experienced under different conditions. The
final step of risk characterization involves the integration and synthesis of the information and analysis in the first three steps to describe the estimated incidence of an adverse effect in a given population (NRC 1994).
This section focuses on exposure assessment. Exposures to stressors (such as chemical agents, pathogens, introduction of invasive species, and physical stresses) are generally defined by a conceptual model that describes which ecological and human receptors have the potential to be exposed. It typically includes a narrative description and diagram of:
• General ecological conditions and potential human exposure scenarios.
• The stressor in question.
• Potential sources of the stressor, transport pathways, and affected media.
• Ecological exposure routes between affected media and representative receptors.
• Identification of representative ecological receptors.
• Human exposure routes between affected media and likely human receptors. Conceptual exposure models are integral to assessing exposure and risk within various federal and state agency frameworks in the United States. Guidance on the development of conceptual models is available from several agencies and departments, such as EPA (1995, 2005d), the U.S. Army Corps of Engineers (Cura et al. 1999), and the U.S. Navy (DeGrandchamps and Barron 2005). The conceptual model gives or allows for the following:
• Format for summarizing exposure pathways that are familiar to regulators and scientific reviewers.
• Discussion of data gaps and recommended further work (field work and risk assessment approach).
• An explicit understanding of the site-specific fate and transport mechanisms upon which regulators and stakeholders can reach agreement.
• Framed recommendations of potential exposures that are site specific.
Figure 2-1 is an example of a conceptual model that might be used for the proposed mycoherbicides. It allows one to trace the physical exposure pathways (independent of geography) of toxins, spores, and vegetative bodies from a particular application method through potential physical transport mechanisms to potentially affected environments. It generally defines the exposure routes and the general classes of ecological and human receptors. The dotted lines and question marks in the figure show where there is particular uncertainty about a pathway. As will be shown in later chapters, there is little information on the proposed mycoherbicides and the specifics of their behavior in the environment
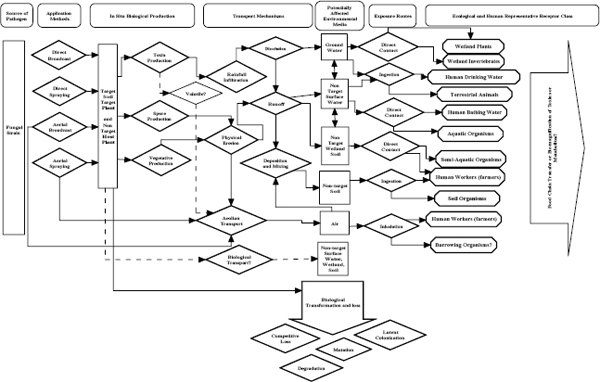
FIGURE 2-1 An example of a generalized conceptual model for mycoherbicide exposure that traces the potential physical exposure pathways of toxins, spores, and vegetative bodies from a particular application method to the general classes of ecological and human receptors. Dotted lines and question marks show where there is likely to be a particular lack of information about a pathway. There is a lack of strain-specific information to provide detailed description of the potential exposures, but the figure provides a guide to help in identifying the type of information needed to characterize the potential exposures.
that would be needed to describe the potential exposures beyond generalizations. If a concerted effort to develop the mycoherbicides is embarked on, Figure 2-1 could be used as a guide to identify where the data gaps are and the types of information that are needed to characterize potential exposures.
Classical or Bioherbicide Approach
The registered mycoherbicides discussed earlier (Table 2-2) were all developed from pathogens indigenous to the regions where they were proposed to be used. Unless a decision is made to develop only strains indigenous to the relevant crop-producing areas, a mycoherbicide based on a nonindigenous strain may require more comprehensive testing to assess the risk to nontarget plant species than would a mycoherbicide based on indigenous strains. The higher level of testing would be comparable with the current testing requirement for the release of a classical weed-biocontrol agent. Such extensive testing may not be required in regions where the mycoherbicide pathogen is indigenous and has never been found to pose a risk to nontarget plant species. It is presumed that the proposed mycoherbicide strains would come from the population of fungi found in the geographic area where the target crop is being grown. If the mycoherbicide were released in areas where the pathogen is not native, estimating the potential for disease in nontarget crops caused by the released mycoherbicide strain or strains descended from it would be far more complicated.




















