Clean Water for People and Ecosystems
Most living things—humans and ecosystems—depend on availability of clean water. The quality of the planet’s waters is changing on time scales of minutes to centuries in ways that are only partially understood. Ensuring clean water for the future requires an ability to understand, predict, and manage changes in water quality.
As the “lifeblood” of the planet and the universal solvent, water transports vast quantities of dissolved chemicals and suspended matter through the biosphere. The concentration of these constituents defines the quality of water and is controlled by naturally occurring and anthropogenic phenomena. Natural variations of the water quality of groundwater, streams, and lakes occur because of geological, climatic, and biological influences. Rain and snowmelt percolate through organic material near Earth’s surface, then through the mineral soil, and through the rocks making up aquifers. Surface streams are fed in part by groundwater discharge and the waters also interact with the streambed. In all of these environments, materials dissolve into, or are removed from, the water through reactions that often are mediated by microbes. Water flowing over the land surface also mobilizes sediment, which is then transported in streams and rivers. In this way the hydrologic cycle is intimately linked with all of Earth’s element cycles.
Human activities have changed the natural element cycles in many ways. Massive alterations in land use and water allocation, as well as the use of synthetic chemicals, have set in motion a transformation of water quality in many hydrologic basins that, at best, will lead to enhanced water treatment effort or, at worst, to the deterioration of water quality with potentially adverse impacts on human and ecosystem health. In the United States, most of these transformations have been under way since the mid-20th century. In other countries this transformation is just beginning. Yet for many groundwater systems and surface water bodies, the reversal of
damages will require a time scale of decades to centuries. For example, groundwater in many parts of the nation is highly enriched in nitrogen, leaving a legacy of nitrogen pollution that, even without new inputs, will take many decades to be diluted by groundwater recharge. Thus, the current quality of any water resource reflects the past as well as ongoing contamination, and water, particularly groundwater, may not become clean even if future sources of contamination are eliminated.
As the global human population grows through this century the demand for clean water will increase. Humans already appropriate more than half of the annual renewable freshwater supply globally, and there are few untapped sources of clean freshwater in the places on Earth where most people live (Postel et al., 1996). Supplying clean water to the growing human population will require much greater water reuse and water treatment than in the past. As societies attempt to increase their standard of living, economic growth will likely exacerbate problems related to water quality and availability, because cleaning water requires energy and energy generation consumes and may contaminate water. The agricultural intensification necessary to feed the growing population and an increasingly urban global population are trends that are likely to further concentrate human and livestock wastes and place additional stress on water treatment systems. The decisions societies make about how to acquire, clean, and dispose of water have enormous impacts on the aquatic ecosystems that are the source of water and the recipient of wastes. The degradation of aquatic ecosystems and the loss of sensitive aquatic taxa can lead to a reduced capacity for natural wetlands, streams, and lakes to trap, store, and transform contaminants. This provides a positive feedback that can further exacerbate water quality problems. Finally, numerous opportunities exist for climate change to impact water quality.
Access to clean water is a political and social problem, but decision makers who are tasked with resolving the problem should be informed by the results of hydrologic research. Science will ensure that the knowledge base necessary to address the challenges of maintaining good water quality where it exists and restoring it when it has been degraded will be available in the future. Research opportunities related to water quality stem largely from a need to know the processes that control the evolution of water quality in both relatively pristine and in heavily impacted environments. The requisite research also spans spatial scales from local to global, and time scales from minutes to decades. Understanding will come through research on the transport and fate of the constituents that dictate water quality of surface and groundwater. Similar to the surge in analytical techniques enabling detection of minute amounts of contaminants in water, the committee anticipates a surge of discoveries from the hydrologic sciences
to advance the scientific understanding needed to promote clean water for the planet.
Many broad research questions fall within the scope of promoting clean water for the planet and challenge hydrologists and their collaborators in biogeochemistry, environmental engineering, and chemistry. Thus, hydrologic research should pursue more linkages with these disciplines than it has in the past. Feedbacks among the fluxes of water and a variety of elements and compounds are at the core of phenomena that should be understood to advance the Earth sciences. This report presents research opportunities in three broad topic areas. The first area relates to element fluxes in a dynamically variable, highly heterogeneous, connected Earth. The second specifically involves contaminant hydrology and the vexing problems that should be addressed to understand the evolution of water quality. The third focuses on the three important, large-scale drivers of water quality: climate change, energy needs, and agriculture.
4.1. Chemical Fluxes through Complex Environments
Geological materials and surfaces are heterogeneous, which confounds the description of fundamentally important hydrologic processes.
The world is complex and composed of diverse and dissimilar parts, a concept that is referred to as heterogeneity. Heterogeneity presents challenges that scientists grapple with in different ways. Geologists identify and classify rocks by the heterogeneity of their matrix, for example, by grain size or mineralogy. Chemists work with mixtures of substances and characterize compounds by their solubility in one phase of a mixture versus another. Hydrologists grapple with the flow of water through a heterogeneous landscape, both on Earth’s surface and in the soils and rocks of the subsurface.
Earth’s heterogeneity can be observed at different scales, from pore size in the subsurface, to the irregularity of a river channel, to patchiness of ecosystems—all of which impact the flow of water and, in turn, the movement and concentration of constituents. The flow of water and dissolved or suspended mass fluxes can vary many orders of magnitude over spatial scales ranging from centimeters to kilometers. It is unrealistic to expect that every detail of heterogeneity can be measured and cataloged to further understanding; useful scientific theories, after all, are ones that capture the essence of phenomena simply by setting aside unnecessary
complications. Yet, the effects of heterogeneity on hydrological transport and biogeochemical transformation of substances should be generalized to some basic principles so effective water management strategies can be devised. Significant scientific effort has directed toward achieving this goal, yet challenges still remain. With current capabilities, cleanup of contaminated groundwater sites is difficult and not likely to be achieved in a desirable timeframe. This difficulty is, in large part, due to the heterogeneity of the subsurface, which confounds treatment technologies. Finally, heterogeneity leads to the observation that discrete units of landscape are connected by water-mediated transport of matter, energy, and organisms. Understanding the connectivity is one of the goals of hydrologic research related to heterogeneity. Opportunities exist to further understand hydrologic connectivity and its relationship to water quality.
What is the role of subsurface heterogeneity and connectivity in mass transport, and how can it be characterized?
Much of the subsurface contains well-connected pathways of relatively high permeability that constitute only a small portion by volume of porous or fractured media. It is also quite common for most of that portion of the subsurface, referred to as aquifers, to contain substantial volumes of nonaquifer materials, such as silts and clays or unfractured rock, which are not conduits for the flow of water. In the actual three-dimensional world, one need only have a relatively small volume fraction of high-permeability material for that material to fully connect or “percolate” (Harter, 2005). The well-connected, high-conductivity paths cause earlier arrival of contaminants at wells or other receptors than conventional analyses would indicate. Slow mass transfer between these fast paths and the presence of this lower-conductivity media lead to broad dispersion of dissolved constituents. This is consistent with field observations, including the ubiquitous observation that contaminant cleanup by well extraction methods is difficult and often requires decades of pumping.
Thus, progress has been made in understanding the impact of connectivity on the dispersion of chemical contaminants, but it reveals information about connectivity that is of concern. Contaminants are moving faster and are dispersed more widely in the subsurface than was originally thought. Given the importance of groundwater as a source of potable water and in recharging surface water systems, continued improvements in understanding fluxes and transformations in such connected networks should be pursued and will require much new work. Oftentimes, this understanding is derived from research conducted at experiment sites (Box 4-1). Yet the overarching question exists: Can connected networks in the subsurface be
characterized to yield a simplified theory, thereby overcoming the heterogeneity problem?
Opportunities exist to develop tools to overcome the heterogeneity challenge. One example involves methods to determine groundwater residence time, the elapsed time from when a parcel of water enters the groundwater system to when it reaches a down-gradient location such as a well or spring. Because groundwater consists of a collection of water and solute molecules that typically have taken very different pathways to the destination, the same groundwater sample can potentially contain water molecules with residence times ranging from years to centuries or millennia. Groundwater age therefore has an age distribution that represents, on the one extreme, the fast flow paths through the high-conductivity, well-connected portions of the system, and on the other extreme, the slow flow paths through the often nonaquifer portions of the system. The age distribution is rich with information about the complexity of the surrounding flow regime, the presence of preferential flow paths, different scales of heterogeneity, and slow mass exchange between the fast and slow parts of the system.
Methods for determining groundwater ages are based on interpreting the presence of dissolved materials and/or associated water molecules at the time they enter the groundwater. The most widely used methods include those based on measuring concentrations of tritium-helium (3H/3He), chlorofluorocarbon (CFC), and carbon-14 (14C). The key obstacle to measuring the groundwater age distribution is the lack of environmental tracer methods for dating groundwater in the 50- to 3,000-year range (Figure 4-2). What are the new methods that will allow enhanced tracking of water quality by age in this “dating gap window?” Can multiple chemical species, including a variety of isotopes that cover the full range of likely groundwater ages, be used in tandem with an appropriate reactive transport model to infer the residence time distribution in three dimensions for an aquifer system?
Pursuit of these research opportunities will advance understanding of fluxes and transformations in connected, heterogeneous, subsurface networks through field characterization and experimentation as well as new theories. How can water and solute residence time distributions in subsurface hydrologic systems be measured? How can these measurements promote new theories to understand the impact of heterogeneity and connectivity on contaminant distribution? What are the most appropriate models to represent solute transport in aquifers containing different degrees of heterogeneity?
BOX 4-1
Revelations in the Complexity of Flow
and Transport Processes
The MADE tracer experiment site at Columbus Air Force Base, Mississippi, is a contaminated site paired with a highly complex, underlying heterogeneous aquifer. This heavily studied, and as a result heavily instrumented, site has provided insights into heterogeneity and connectivity issues at the plume scale over the past 25 years. Research activities have revealed an extreme complexity of flow and transport processes in highly heterogeneous porous media. Existing theories and models have been shown to be incorrect or inadequate, while new ideas and improved approaches are continuously being proposed and developed (Zheng et al., 2011; Figure 4-1).
The key questions and hypotheses emerging from these research activities include the following: Are small-scale preferential flow paths resulting from variations in hydraulic conductivity (K) the primary cause of the observed “nonideal” behaviors in tracer plumes at the MADE site and elsewhere? What are the nature, geometry, and scale of preferential flow channel networks in a highly heterogeneous fluvial aquifer such as that at the MADE site? How are such flow channel networks (and flow barriers) related to the texture, structure, grain size, and facies distribution of fluvial sediments? What is the most appropriate upscaled model to represent solute transport in aquifers containing small-scale, connected, high-K networks without an explicit definition of these networks? How can the parameters for the upscaled model be obtained from readily available field data?
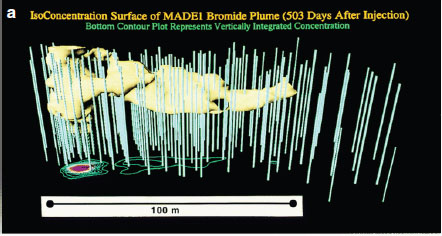
FIGURE 4-1 Observed conservative tracer plumes at the MADE site: (a) bromide plume (concentration in mg/L) at 503 days after 2-day injection from the MADE-1 test; (b) observed MADE-2 tritium plume (concentration in picocurie per milliliter [pCi/ml]) at 328 days after 2-day injection from the MADE-2 test; and (c) observed MADE-3 bromide plume at 152 days after
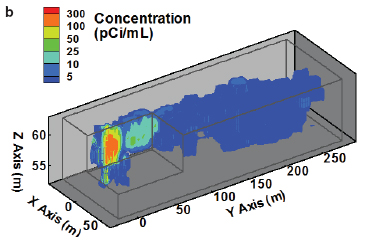
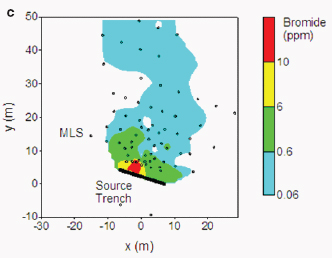
trench source release. (The two-dimensional contour map is constructed from the peak concentrations at each multilevel sampler location from the MADE-3 test.) All plumes exhibit a highly complex (“non-Gaussian”) pattern that cannot be readily described by classical models. SOURCE: Reprinted, with permission, from Zheng et al. (2011). © 2011 by John Wiley and Sons.
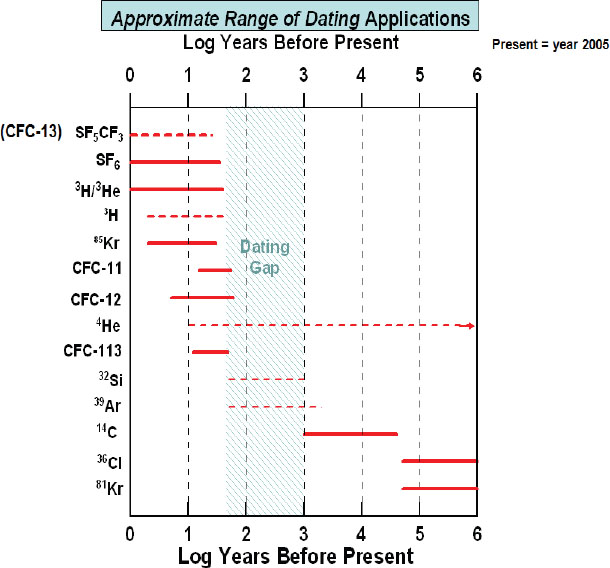
FIGURE 4-2 List of available environmental tracers for estimating water residence time in geologic systems and the “dating gap” for which no reliable methods yet exist for estimating age. SOURCE: Courtesy of N. Plummer, U.S. Geological Survey.
How much of the spatiotemporal heterogeneity in linked hydrological-biogeochemical cycling must be understood to estimate, model, and predict net watershed solute export?
The increasing ability to monitor chemical and biological conditions in real time is revealing previously unknown temporal trends that in turn push the boundaries of process understanding. The more finely watersheds are subdivided and the more frequently hydrologic and biological processes are measured, the more dynamic ecosystem hydrology and biogeochemistry are discovered to be. As hydrologists move into the era of high-resolution data sets, a critical challenge for hydrologists is to determine the data density and distribution necessary to address critical questions about ecosystem-scale biogeochemical behavior. How much detail is needed?
For regional fluxes of water, hydrologic models are typically able to provide reasonable approximations and predictions without highly resolving properties and processes in space and time. For biogeochemically active solutes and elements with significant gaseous forms, coupled hydrological-biogeochemical models often fail to match observations. This is because (1) hydrologic models can match the flux of water without having to accurately approximate pathways and mass exchange between regions of fast and slow flow and (2) representing biogeochemical reaction rates requires reasonable approximations of reactant supply and environmental conditions (temperature, pH, and oxidation reduction potential) along flow paths as well as accurate flow path routing of water. Processes that occur homogeneously across the ecosystem (e.g., organic matter decomposition) will be easier to model and upscale than processes occurring under a more limited set of conditions (e.g., denitrification, which occurs only under conditions of low oxygen, high carbon, and high nitrate). For heterogeneously distributed biogeochemical processes, measures of whole ecosystem rates provide little information about where and when the process occurs.
There can be great spatial and temporal heterogeneity in the availability of nutrients and the extent of hydrologic connectivity within ecosystems. Because of this variation, there are places and times within ecosystems that can play disproportionately large roles in driving whole-ecosystem biogeochemistry. For example, riparian zones that receive materials from the surrounding uplands and that are more highly hydrologically connected to streams than the rest of the catchment tend to have heightened rates of nutrient transformations. Although scientists conceptually recognize the likely importance of these rare moments and locations for enhanced biological activity, in practice it has been challenging to define, locate, and monitor these ecosystem control points. The increasing ability to map and monitor hydrology, chemical concentrations, and biological activity in real time is revealing previously unknown spatiotemporal hydrologic and biogeochemical dynamics (Box 4-2) that in turn push the boundaries of process understanding. What variables control the occurrence and magnitude of hot spots in terrestrial, riparian, and hyporheic zones? Can knowledge about the joint distribution of organic carbon and fast flow paths be used to infer the major control points for ecosystem biogeochemical reactions? How do human interventions on the landscape change hydrological connectivity and therefore the spatial distribution of hot spots?
BOX 4-2
Understanding of the Correlated Variation in Hydrologic Science and Biogeochemistry through Technological Innovations
Technological innovations are providing an unprecedented understanding of the spatial and temporal covariation in hydrology and biogeochemistry. At landscape scales, remote sensing technologies effectively capture hydrologic variation in space and time. For example, synthetic aperture radar derived vegetative cover and inundation for a reach of the Solimões River in the central Amazon Basin, Brazil, is shown in Figure 4-3. On a much smaller scale, microelectrode profiles illustrate the variability of oxygen, iron, and manganese concentrations in co-located sediment profiles (Figure 4-4). Measurement techniques have provided new insights into soil stream interactions, as well. For example, dissolved carbon dioxide (CO2) concentrations for a peatland stream vary over the course of a storm hydrograph (Figure 4-5). Finally, high-frequency in situ sensor arrays in Ichetucknee River, Florida show the close relationship between nutrient cycling and metabolic activity (Figure 4-6).
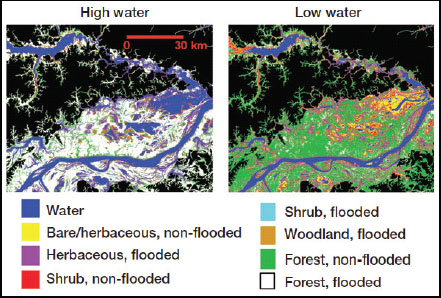
FIGURE 4-3 Landscape-scale remote sensing technology showing vegetative cover and inundation for a reach of the Solimões River in the central Amazon Basin, Brazil. SOURCE: Reprinted, with permission, from Hess et al. (2003). © 2003 by Elsevier.
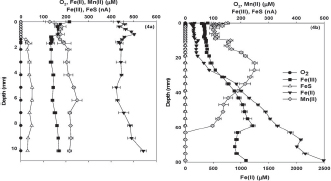
FIGURE 4-4 Voltammetric profiles indicate significant variability in speciation in wetland soils 2 cm apart in sediment cores collected on the same day in Jug Bay, Maryland. SOURCE: Reprinted, with permission, from Ma et al. (2008). © 2008 John Wiley and Sons.
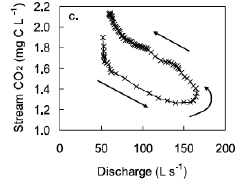
FIGURE 4-5 Variability of dissolved CO2 concentrations (mg C L–1) for a peatland stream over the course of a storm hydrograph (discharge in L s–1). SOURCE: Reprinted, with permission, from Dinsmore and Billett (2008). © 2008 by the American Geophysical Union.
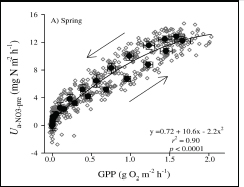
FIGURE 4-6 Diel covariation in gross primary production (GPP in g O2 m–2 h–1) and nitrate uptake (autotrophic N assimilation or uptake as Ua-NO3-pre in mg N m–2 h–1) rates in the highly autotrophic Itchetucknee River, Florida. The line is best-fit least-mean-square regression on individual observations (see Figure 1-4 for the in situ observations used to probe this coupling). SOURCE: Reprinted, with permission, from Heffernan and Cohen (2010). © 2010 by the American Society of Limnology and Oceanography.
How do river networks interact within the channel and upland areas to organize sediment composition and mass flux, and in turn, river biogeochemistry and water quality?
Sediment produced from hillslopes travels into and through the river network both spatially and through time, swept in pulses by floods, sorted hydraulically, and changed in size and composition by abrasion and other mechanisms. Enhanced sediment input into aquatic systems can impact light penetration and hence photosynthesis and the health of biological habitats. Sediment carries with it constituents that are dispersed in the landscape, rivers and floodplains, and affect water quality and biogeochemistry. Recent research has highlighted that bedload sediment composition and mass flux in a river basin might be set by not only the channel network structure, but also, to a large degree, the sediment size and composition supplied from upstream hillslopes. This dynamic connectivity between channels and their surroundings is not unique to erosional systems; in fact, it is even more pronounced in net depositional systems such as deltaic networks. For example, the hypsometric, or height measurement, analysis of the land-building field site at Wax Lake Delta, Louisiana, displays the intricate connection between land, water, and plant communities that dynamically changes under natural and human influences (Figure 4-7).
Understanding the dynamic co-evolution of channels and their surroundings is key to improving prediction of stream biogeochemistry, water quality, and ecosystem integrity in a landscape. Such understanding is also crucial in developing minimal complexity predictive models, which capture the essential components of the system and can be used as management tools to explore system response to perturbations such as climatic change (e.g., storm intensification or sea level rise) or anthropogenic stresses (e.g., land-use changes). New observations, such as high resolution topography and vegetation cover from Light Detection and Ranging (LiDAR), as well as new geophysical monitoring techniques for subsurface flow characterization and water and sediment dating offer new opportunities for advancing understanding of water-mediated transport of environmental fluxes through landscapes.
Many open research questions exist, and the committee provides here only a few examples. What is the relative role of nonfluvial (hillslope or subsurface) versus fluvial (channel network) transport pathways in determining water quality, biogeochemistry, and river ecosystem health? Are there places in the landscape where accelerated changes or amplifications occur and where observations and focused understanding would do most good? Under what conditions do systematic changes in hydrologic regimes (altered flood cycles, prolonged droughts) or land use changes (agricultural drainage, fires, etc.) bring the coupled water-sediment-biota system to new
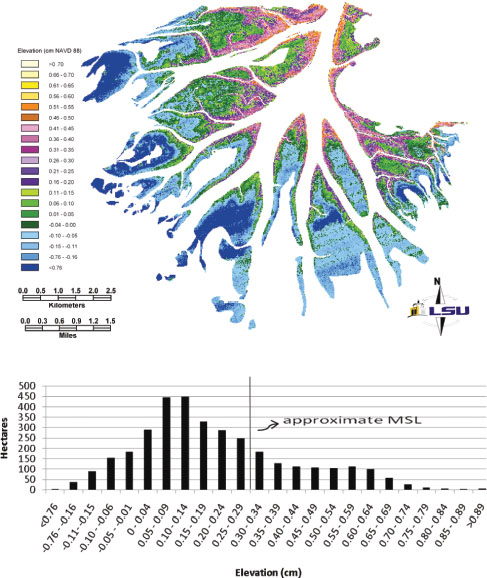
FIGURE 4-7 The terrain complexity in a deltaic system of channels modulates complex interactions between water, earth, and biota for which predictive models are lacking. This picture shows the elevation analysis of LiDAR data from the Wax Lake Delta, Louisiana, with the distribution of total area (hectares) within 5-cm elevation classes. The elevation is corrected to cm NAVD 88 and paired with an estimate of mean sea level (MSL). Analysis of plant community types overlaid on this elevation data is needed to advance predictive understanding of delta island plant community structure. SOURCE: Image courtesy of Elaine Evers, Louisiana State University.
equilibrium states or to states that are unstable and thus prone to large shifts even under small perturbations?
How can transport and transformation of constituents through complex media be better described mathematically?
The broad range of space and time scales of transport through a heterogeneous world challenge existing predictive models and has prompted development of new classes of predictive models through generalization of standard models that have been used for decades but have essentiallyignored heterogeneity. For transport in surface waters, new ideas and theories have recently been put forward to characterize a range of processes: from particle transport in a single stream, to sediment and water transport on hillslopes, to dynamic transport on erosional and depositional river networks.
Research has shown that mineral heterogeneity is important to controlling field-scale reactive transport under many field conditions of interest (Allen-King et al., 2002). Development of models that include processes at all scales relevant to field-scale reactive transport, including statistically quantifying observed mineral heterogeneity for these models, remains a challenge. This challenge motivates a variety of research needs including appropriately quantifying basin-scale mass transfer among different geologic media, increasing computational efficiency, and dealing with details of biogeochemical reactions (Fogg and LaBolle, 2006). Quantifying the uncertainty of these models represents an additional major hurdle. Specific research questions of interest include the following: Do physically based models of transport give rise to the emergent patterns of space-time flux variability observed in nature? How can these new classes of models be tested from observations, and how can their parameters be estimated? What experiments are needed to verify and further advance these new theories of transport?
4.2. Earth’s Evolving Water Quality Profile
The water quality profile of the planet is evolving as new contaminants are introduced to the water cycle and old contaminant use continues.
As contaminants move through an irregular but interconnected world, Earth’s water quality profile evolves in space and time. Discharge of contaminants has disturbed the planet’s water chemical composition and raises the question: How widespread and severe is the deterioration in the planet’s water quality? The idea that clean water is accessed only upstream from
human activity has hardly ever proven to be a useful concept and is now particularly questionable given that “there is no upstream anymore.” Some contaminants have spread globally through the water cycle. Even after years of concern about a variety of contaminants, some problems continue to worsen, and this trend is expected to continue in the future.
The existence of “classic” contaminants—arsenic, lead, chloride, and many others—and knowledge about their behavior in and impact on the environment is relatively well established, although there are open research questions related to the bioavailability and transport of these contaminants. The content of nitrogen and phosphorus in rivers and streams worldwide has increased as a consequence of human activities (Nilsson and Renöfält, 2008). It is now estimated that as many as 77 million people in Bangladesh—approximately half the country’s population—and perhaps many more in other portions of southeast Asia living on river valleys draining the Himalaya are afflicted with arsenic poisoning from drinking shallow groundwater containing naturally high concentrations of arsenic from the aquifer materials (Figure 4-8). Although the situation in Bangladesh has
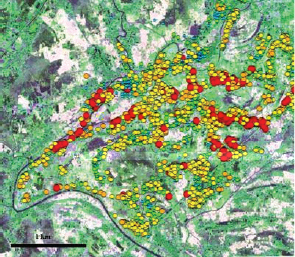
FIGURE 4-8 Arsenic contamination in the groundwater of the floodplains of central Bangladesh. Symbols are graduated in size and color to indicate higher arsenic concentration. Arsenic concentration varies spatially because of complex interrelationships among hydrologic flow paths, sediment structure, organic carbon, and pumping. SOURCE: Image courtesy of Beth Weinman, Vanderbilt University. Available online at http://sitemason.vanderbilt.edu/site/kFvWDK/Research [accessed August 6, 2012].
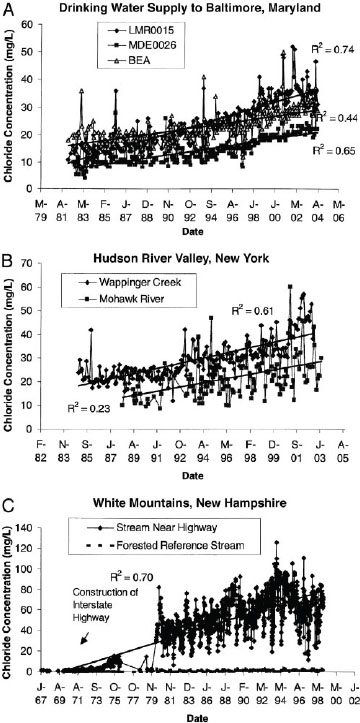
received the most attention because of the scale of this human tragedy, arsenic contamination is a global problem that affects water supplies in the United States as well as other locations. Opportunities in the Hydrologic Sciences showed the dramatic increase in chloride flux in Europe’s Rhine River from 1890 to 1990, a phenomenon closely tied to the use of sodium chloride as road deicing salt. This increase in chloride flux continues today, for example, in the northeastern United States where in some cases stream water concentrations of chloride have increased significantly since the 1960s (Figure 4-9). Extrapolations suggest that baseline values of chloride in some streams will exceed the accepted level for human consumption and the toxicity level to many freshwater organisms in the future.
The recent advancements in analytical techniques mentioned in Chapter 1 have allowed for identification of a suite of other contaminants in an alarming number of freshwater bodies about which much less is known. These contaminants of emerging concern include pharmaceuticals, engineered nanoparticles, and additives in personal care products, to name a few. Their presence in the water cycle can be traced to numerous point and nonpoint sources such as wastewater treatment systems, septic systems, regulated and unregulated industrial discharges, manufacturing processes, large-scale animal feeding operations, combined sewage overflow, and urban runoff. The chemical properties of these contaminants often promote transport (e.g., high solubility) and chemical transformation (e.g., susceptibility to oxidation) in the aqueous environment. The list of contaminants of emerging concern known to be present in natural waters is quite large, ranging from hormonally active agents to caffeine. With development of new chemicals and their subsequent introduction to the water cycle, this list continues to grow.
Much of the fundamental work mentioned in the section above on Chemical Fluxes Through Complex Environments is critical for understanding the evolution of the water quality profile. Yet pursuing answers to other questions will promote scientific understanding of how the water quality profile evolves in space and time and how contaminants interact with hydrologic processes and impact stream ecosystems.
How do hydrologic processes impact contaminant fate and, in turn, affect the evolution of water quality?
In the United States, much of the groundwater used for the public drinking water supply is from deeper portions of the aquifer systems where the groundwater is too old, decades to centuries to millennia, to contain significant anthropogenic contamination. However, shallow, poorer-quality groundwater is typically moving downward through largely unknown pathways and increasingly contaminating deeper groundwater at largely
unknown rates. Thus, problems with contaminants in groundwater may occur years or decades in the future making it difficult to muster public or political concern. The groundwater quality in some basins may be non-sustainable; if left unchecked over decades to centuries contamination can theoretically lead to regional degradation of entire groundwater basins, the quality of which is linked to surface water. Yet, work to understand, monitor, and model the relevant phenomena to predict long-term sustainability of groundwater quality mostly lies ahead (Fogg and LaBolle, 2006). How is the contaminant signature in deeper groundwater changing over time?
It is hypothesized that the conversion of grasslands and forest to row crop agriculture has decreased evapotranspiration and therefore increased recharge in these landscapes, which has led to increased base flow contributions to the streams and rivers in the basin (Zhang and Schilling, 2006). Along with this increase in groundwater has been an increase in the flux of chemical weathering products such as bicarbonate, the dominant form of inorganic dissolved carbon in most rivers (Figure 4-10). It has been suggested that approximately half of the increase in bicarbonate concentration in the lower Mississippi River over the past 50 years is due to the alteration of the hydrologic system brought about by conversion to agriculture (Raymond et al., 2008). How do anthropogenic changes in land use alter water flow paths that in turn impact other elemental fluxes and water quality?
On a much smaller scale, some progress has been made on tracking the chemical transformation and fate of contaminants of emerging concern because of, in large part, advances in analytical techniques. It is known that some contaminants of emerging concern transform by light-driven chemical reactions in the upper portions of the water column, some contaminants are hydrophobic and immobilized in the surrounding substrate, and some are transformed by microbial processes. Some chemical reactions are often enhanced by naturally present constituents like iron or dissolved organic matter, accelerating transformation of these compounds. These chemical transformations generate an entire suite of “second-generation” contaminants of emerging concern, the scope and impact of which are largely unknown. For example, dioxins, one of the most toxic synthetic organic compound classes known, have been detected in Mississippi River sediment. This was attributed to the existence of a chemical precursor triclosan in the natural environment, perhaps most widely used in the formation of liquid hand soaps, which is known to form dioxins when irradiated by sunlight (Buth et al., 2010).
Relevant questions ripe for interdisciplinary research between hydrologists, chemists, and environmental engineers include the following: What is the chemical “end game” for a given contaminant depending on flow path? What is the mobility (i.e., transformation and fate) of unstudied
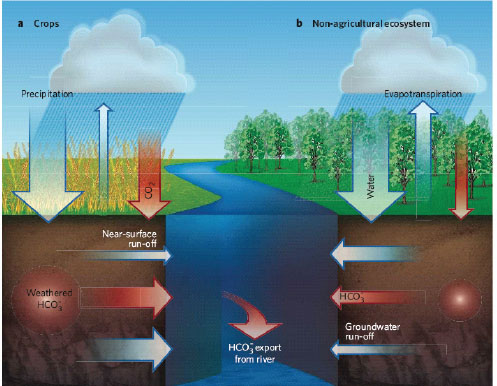
FIGURE 4-10 Even with a similar precipitation rate, soil type, and soil condition, certain agricultural practices result in greater groundwater runoff and less evapotranspiration in comparison to nonagricultural ecosystems. The increase in groundwater runoff leads to larger bicarbonate (HCO3–) exports to rivers. SOURCE: Reprinted, with permission, from Mayorga (2008). © 2008 by the Nature Publishing Group.
contaminants of concern? Which contaminants of emerging concern are bioavailable and which may bioaccumulate, and how do these processes interact with hydrologic processes to affect the evolution of water quality? What are the typical suites of contaminants to which humans and ecosystems are commonly exposed? How will the evolution of the water quality profile differ in urban and rural environments with different hydrologic and biogeochemical drivers?
What are the relationships between contaminants and human and ecosystem health and how can deleterious effects be avoided in the future?
Alterations in water supply rarely occur without concomitant changes in water chemistry and water temperature. The “replumbing” of the water
cycle has disturbed the link between flow regime and water quality, causing a shift in water quality beyond natural variability that has become especially profound since the industrial era (Nilsson and Renöfält, 2008). The timing of contaminant delivery to surface water and groundwater and the combined effects of flow alteration and contaminant loading have not been adequately addressed by the hydrologic community. Better understanding and prediction of the links between contaminants and hydrologic alteration and the impacts on water quality (and in turn on humans and aquatic ecosystems) are needed. This knowledge will inform changes in policy that will be needed to protect water quality in the future. How do alterations in water flow paths affect temperature and contaminant regimes of freshwater ecosystems? What maintains thermal and chemical refugia in freshwaters, and how can they can be protected, preserved, and restored?
Exploration of diel (24-hour) signals in natural waters yields insight into the intricate linkages among hydrological, biological, and geochemical processes (Figure 4-11). For example, variations in dissolved oxygen concentrations and pH in streams and rivers are driven by the biological processes of photosynthesis and respiration. These diel variations in stream metabolism may in turn play significant roles in controlling the dissolved metal concentrations in solution. Research in a wide variety of streams and rivers has discovered substantial diel variation in potentially toxic metals, where dissolved cationic species, such as zinc, cadmium or manganese, have their highest dissolved concentrations shortly before sunrise, while anionic species, such as arsenic, have their highest dissolved concentrations in the late afternoon (Figure 4-11). The timing of the maximum and minimum dissolved concentrations with light and dark cycles has significant implications for the determination of fluxes as well as potential biological uptake. What biogeochemical processes, such as variations in photosynthesis and respiration, affect other potential contaminants?
Hand in hand with the issue of contaminants of emerging concern is the increasing role that wastewater effluent plays in streamflow in arid regions and even nonarid regions during periods of low base flow. Under these circumstances, wastewater effluent in urban environments constitutes the dominant source of streamflow (Brooks et al., 2006). Thus, in such streams the chemical composition can be very different from that of natural waters. How do these differences translate into effects on aquatic ecosystems? For example, the occurrence of intersex fish has increased in surface waters throughout the United States (Hinck et al., 2009), and the presence of human estrogenic compounds (as well as other anthropogenic substances) have been implicated for this phenomenon. Thus, streams that receive significant amounts of domestic wastewater effluent with trace levels of human estrogenic substances could have long lasting deleterious effects on aquatic ecosystems.
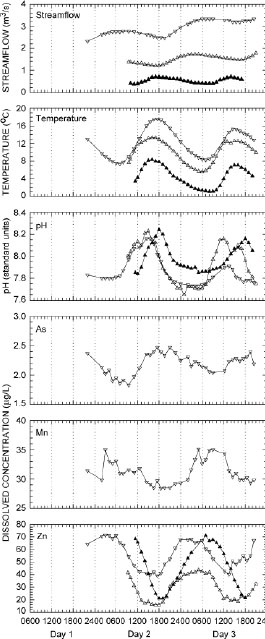
FIGURE 4-11 Diel (24-hour) cycles in streamflow, temperature, and pH, and dissolved arsenic (As), manganese (Mn), and zinc (Zn) concentrations during snowmelt runoff episodes. Episodes were sampled during an early snowmelt-runoff (March 22-23, 2001; closed triangles) and a late snowmelt-runoff (April 26-27, 2001; open triangles and May 23-25, 2011; open inverted triangles) at Prickly Peak Creek, Montana. SOURCE: Reprinted, with permission, from Nimick et al. (2005). © 2005 by Springer Science + Business Media.
A more detailed understanding of water quality continues to emerge from the increasing sophistication of chemical analytical instrumentation and includes detecting contaminants at increasingly lower concentrations as well as new contaminants. Yet, when is the risk from the presence of these constituents so small that it is negligible for all practical purposes? Merging information about the relationship between hydrologic flux and contaminant concentration with risk assessment is critical. What are the contaminant dilution ratios resulting from hydrologic flux, and how should the findings inform risk analysis?
4.3. The Future of Water Quality in a Hot, Flat,1 and Crowded World
As Earth’s human population moves toward 9 billion, as resource use intensifies, and as climate changes, maintaining adequate water quality will rely on new knowledge.
The final layer of complexity in the water quality picture involves the large-scale drivers of water quality. Much conversation revolves around Earth’s growing population and demographic change. Intellectual leaders debate questions about how the world should deal with the corresponding intensification of resource use and the impact of climate change. The amount of food required to feed the projected population of 9 billion is staggering—and currently many people go hungry. Similarly, the energy required for transportation, to generate electricity, or to provide clean water for 9 billion people is enormous. And all of these demands should be considered against the backdrop of a changing climate, providing yet another challenge to maintaining adequate water quality for humans and ecosystems.
The layers of complexity in a hot, flat, crowded world are daunting. The world will need scientific knowledge to even begin to deal with these issues. The hydrologic research community has an obligation to tackle the water quality questions embedded within these large-scale drivers.
How do changing flow paths as a result of urbanization correspond to changes in water quality?
Currently, about half of the global population lives in urban areas. As global urban migration continues, this is expected to increase to 60 percent
____________
1 The term “flat,” coined by the author Thomas Friedman in his books The World is Flat (2005) and Hot, Flat, and Crowded: Why We Need a Green Revolution—and How It Can Renew America (2008), is used to describe a new era of globalization that allows people and entities around the world to compete, connect, and collaborate.
by 2030 (UN, 2006), which will have impacts on the environment. For example, the increase in impervious surface area associated with urbanization slows evaporative cooling, allowing surface temperatures to rise higher than in rural areas. Impacts on the urban hydrologic cycle also exist; it has long been recognized that the increase in impervious surface area leads to a decrease in infiltration and an increase in runoff, thereby changing the hydrologic response of streams.
The recognition of water quality problems associated with urban development has influenced engineering design in the 21st century. Incorporation of “green” infrastructure into the engineering of cities has become prevalent, whereby effects of stormwater and impervious surfaces are mitigated through designs that attempt to mimic the natural hydrologic cycle and management of stormwater on a watershed scale (NRC, 2008). Practices include promoting infiltration of stormwater such as removing hardened surfaces and regreening urban lands; incorporating grass swales, rain gardens, and green roofs into landscape design; using pervious paving materials; and installing subsurface storage where infiltration practices are not possible. An opportunity exists to probe the geochemical phenomena associated with changing urban flow paths, i.e., stormwater.
Knowledge exists in this area. For example, a smaller percentage of atmospherically introduced contaminants are retained in urban landscapes compared to forested ones. As a result, chemical concentrations of nitrogen and metals are higher in urban streams (Figure 4-12). This lack of retention of contaminants in urban drainages can have effects on downstream aquatic ecosystems and potable water supplies. Future increased water demand in urban systems will exacerbate this problem. What are the impacts of urbanization on hydrologic connectivity and, therefore, water quality? How do urban management practices, for example, urban stormwater management, impact hydrologic connectivity, water quality, and ecosystem health?
What new hydrologic knowledge is needed to enable agriculture and silviculture to be sustainable with respect to water quality?
The role of agriculture in water quality continues to be a major global problem. As the global population increases, the need for food and fiber will increase, leading to the need to convert more marginal land to agriculture and to increase agricultural production per unit land area. In the latter case, increased fertilizer usage will increase nutrient fluxes, especially fixed nitrogen compounds, into both groundwater and surface water systems. These nonpoint sources enhance eutrophication and nuisance algal blooms in water bodies locally, regionally, and even far afield as demonstrated by increased hypoxia in the Gulf of Mexico due to input of nutrients from
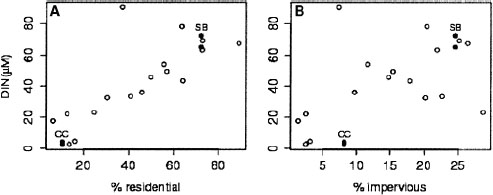
FIGURE 4-12 Increasing annual flow weighted dissolved organic nitrogen concentration (DIN, μM) with increasing residential area (A) and impervious surface (B). SB and CC represent two catchments of contrasting land use (heavily residential and heavily forested). SOURCE: Modified, with permission, from Wollheim et al. (2005). © 2005 by Springer Science + Business Media.
Mississippi River waters derived primarily from row-crop agriculture in the Midwest.
In addition to introducing contaminants into the environment on a landscape scale, irrigated agriculture also has a large demand for water, which in turn often leads to groundwater overdraft. Under predevelopment conditions, the groundwater systems flowed from recharge areas to natural discharge areas, providing an exit for dissolved salt. However, when overdraft occurs and the pumped groundwater is reapplied to the land for irrigation, a closed hydrologic basin is potentially created whereby shallow groundwater of lesser quality (for example, elevated salinity) eventually makes its way to the pumping wells, only to be withdrawn and reapplied to the land, resulting in further concentration of salts and nutrients. Such a closed system in agricultural basins such as the San Joaquin Valley of California may lead to long-term salinization of groundwater, which will eventually render the groundwater unsuitable for irrigation and most other uses. In certain environments, conversion of natural landscapes to agriculture also can impact groundwater due to groundwater rise. If the native vegetation is replaced with less water consumptive crops, then the subsequent rise in the water table can solubilize previously accumulated salts, increasing groundwater salinity, mobilizing nitrate, and salinizing soil.
Efforts to ensure global food security will lead to changes in practices that will need to be assessed, which underscores the importance of addressing critical research questions. What are the hydrologic and water quality implications of changes in crop patterns and rotations? What are
the possibly nonlinear feedbacks between irrigation water quality and plant productivity, and how are these affected by management of the hydrologic regime? As the approach to “peak phosphorus” leads to resource restrictions, it will become necessary to pervasively employ nutrient recycling (for example, recovery and use of nutrients from municipal and other waste products). What are the water quality implications related to the use of different fertilizer forms?
How can the hydrologic sciences inform solutions to the “water-energy nexus”?
Water and energy are mutually dependent resources. Increased energy consumption will lead to increased water consumption and water availability challenges throughout the globe (explored in Chapter 2). Yet, the “water-energy nexus” has the potential to impact water quality, as well. Water is needed in the production, refinement, or distribution of a variety of energy sources, and when water is used in this manner there is an impact on water quality. For example, 90 percent of all of the electricity used in the United States is generated by steam, which must then be cooled to condense and reuse or returned to waterways (Averyt et al., 2011). Power plant cooling uses approximately 30 percent more water than is used in irrigation in the United States, and although the water use is mostly nonconsumptive, return flows are warmer than initially extracted, impacting aquatic ecosystems (Averyt et al., 2011). The scope of the water-energy nexus is broad indeed given the need for water and energy for 9 billion people; changes in the thermal regime of aquatic ecosystems is just the beginning. How will the increasing need for energy impact the planet’s water quality?
Extraction techniques to obtain a variety of carbon-based fuels have a long history of causing adverse water quality impacts. For example, drainage of acidic waters from coal mines (acid mine drainage) has long plagued coal-producing regions. Many open questions remain related to the impacts of energy production on water quality. One current example relates to the development of natural gas from deep shale beds. Hydraulic fracturing, a technique that produces natural gas otherwise locked in organic-rich shales, involves injecting water and chemical additives into shales at high pressure. The potential environmental impact of hydrofracturing has been a contentious topic. Accidental releases of contaminants into aquifers have been documented, likely because of improper well construction. Disposal of flowback water from wells in surface disposal ponds also has been implicated in water contamination. What research on hydrogeological implications of hydrofracturing and on the chemical composition of injection fluids and their interaction with natural minerals is needed to properly inform the public debate?
Marine and hydrokinetic energy—energy from waves, tides, and currents in oceans and so forth—generates power by harnessing the natural flow of water without using a dam, a diversionary structure, or an impoundment. While in its infancy, this energy source has potential; marine and hydrokinetic energy could provide almost 10 percent of the U.S. electricity demands, a step toward the 2020 mandated requirement of 20 percent renewable energy by electric utilities in the United States (American Clean Energy and Security Act of 2009). Yet, the environmental effects of marine and hydrokinetic energy are far from understood. It is known that this technique will reduce water velocities in the vicinity of the project, as well as increase water surface elevations and decrease flood conveyance capacity at larger scales (Bryden et al., 2004). As this technology develops (Figure 4-13), research efforts to provide the scientific understanding of how harnessing the natural power of water disrupts the water cycle and alters water quality will become a priority. What are the related changes in
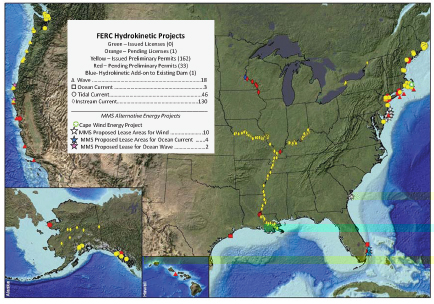
FIGURE 4-13 Federal Energy Regulatory Commission (FERC) marine and hydrokinetic alternative energy projects in the United States. The pressure for advancing marine and hydrokinetic energy is running faster than the knowledge needed to ensure that these projects are not adversely affecting the environment in the near or long term. SOURCE: NOAA Fisheries Office of Habitat Conservation, February 11, 2009.
natural flow paths, which could in turn lead to altered water quality and effects on the aquatic food web? What are the long-term effects in estuary and lagoon inlets of, for example, changing wave height?
Solutions to offset the buildup of carbon from carbon-based fuels also are being pursued. Carbon sequestration, processes to remove CO2 from the atmosphere, is increasingly looked to as a means of mitigating the impact of global warming. Carbon can be stored in various forms: liquid in the oceans, gas in deep geologic formations, or solid using chemical reactions to produce stable carbonates. Carbon sequestration processes also can take a variety of forms, from injection of carbon into underground reservoirs to development of tree plantations. But like with any retooling of the natural environment, the consequences of these efforts can be surprising. For example, semipermanent, biological carbon sequestration through extensive tree plantations will reduce streamflow and impact water quality. Carbon sequestration in deep geologic formations has the potential to affect groundwater quality. What are the water quality tradeoffs associated with various carbon sequestration strategies? What data are necessary to inform risk analysis models assessing the impact of carbon sequestration techniques on water quality?
What might be the effects of climate change on freshwater quality?
Understanding the impact of climate change on water availability has received particular attention. Far less attention has been paid to the impact of climate change on water quality and, as a result, far less is known. Numerous opportunities exist for climate change to produce new regimes that impact water quality, from increasing sediment, chemical, and pathogen loading in runoff to saltwater intrusion threatening the quality of coastal groundwater supplies. One of the most obvious is a change in the thermal regime of water bodies. How does a changing temperature regime impact flow dynamics and biogeochemical processes in streams, lakes, and reservoirs? How do thermal and hydrologic changes interact to affect freshwater ecosystems through, for example, increased eutrophication?
Management of urban and agricultural catchments for rapid water conveyance (i.e., replumbing of the water cycle) has led to increasing proportions of contaminant loads moving into and through freshwaters during peak flows. Climate change also is expected to exacerbate this phenomenon. For example, increased precipitation could increase sediment yield and contaminant runoff in agricultural dominant as well as urban- and suburban-dominant watersheds. A potential increase in contaminant loading has important consequences for downstream reservoirs, lakes, and coastal environments. How does climate change impact river flows and the flushing of constituents into down-gradient areas? Increased flooding
has the potential not only to flush contaminants downstream, but also to remobilize contaminants through increased erosion. Will increased fluxes remobilize contaminants, and, if so, which contaminants are more susceptible to this remobilization?
Similarly, water extraction from surface and groundwaters has led to prolonged periods of low flows, which can aggravate contamination problems. Again, climate change is expected to exacerbate this phenomenon. In some areas there could be a decrease in precipitation leading to decreases in base flow and evapoconcentration of water, increasing salinity. In others, decreased frequency of rainfall events could introduce concentrations of non-point-source contaminants that have accumulated on the landscape over longer time periods, leading to enhanced pulses to the aquatic ecosystem. Changes in flow regimes due to changes in precipitation patterns or changes in percentages of snow versus rain might also impact water quality.
As contaminant loads are added to declining water volumes, chemical signals are amplified. Low flows in surface waters can also further exacerbate problems of low oxygen and alter the toxicity of contaminants. This concept has a variety of consequences that expand into the realm of Water and Life, because low flow shrinks habitat for aquatic species and increases the contaminant concentration and, in turn, exposure. What will be the impact of climate change induced low flows on contaminant concentrations? In the spirit of the previous chapter how does this translate to ecosystem impact?
The committee presented research questions designed to further understand Earth’s water quality in relatively pristine and impacted environments. Similar to the approach to address the challenges and opportunities outlined in Chapters 2 and 3, field studies, whenever feasible and appropriate, are important to answering the questions proposed in this chapter. Answering these questions also will require work by hydrologic scientists and engineers as well as a focused effort from those in related subdisciplines such as aquatic geochemistry and biogeochemistry. In the previous chapters the committee presented research questions probing the dynamic relationship between water and physical process and life, questions that also will require work at disciplinary interfaces. Thus, a common theme extends through the range of research questions noted in this report, that is, the need for interdisciplinary research that takes advantage of cutting-edge technological capabilities to grapple with the complex water-related challenges of today and tomorrow. Examples in the current context include research on groundwater age linking physical hydrologists using Darcy’s Law and chemical hydrologists perfecting age-dating techniques. Or research
using advances in chemical analytical techniques to help disentangle the coupled hydrological biogeochemcial processes that control the evolution of Earth’s water quality profile.
Many of the research questions discussed here and in the previous chapters have relevance and applicability to the multitude of stakeholders who are concerned with water resources. For example, environmental management or stewardship requires an understanding of cause and effect as well as the ability to predict effects of different management practices on the environment. Such a predictive analysis requires not only long-term monitoring and understanding changes in water quality but also development of sophisticated models capable of representing the effects of heterogeneity, connectivity, and biogeochemical reactions on water quality at the regional scale. Thus, while the hydrologic sciences are critical to the resolution of the issues discussed in this report, it will be interdisciplinary teams of researchers, including not only physical, chemical, and biological scientists, but also social scientists, that produce useful scientific results. Ongoing consultations with those who build and maintain infrastructure are essential. To ensure that society, in additional to science, benefits from research results, interactions with policy makers and other decision makers will have to follow.
The next and final chapter presents findings for the hydrologic community to consider with respect to both advancing fundamental science and to contributing to solutions of the complex water issues of today.
Allen-King, R. M., P. Grathwohl, and W. P. Ball. 2002. New modeling paradigms for the sorption of hydrophobic organic chemicals to heterogeneous carbonaceous matter in soils, sediments, and rocks. Advances in Water Resources 25(8-12):985-1016.
Averyt, K., J. Fisher, A. Huber-Lee, A. Lewis, J. Macknick, N. Madden, J. Rogers, and S. Tellinghuisen. 2011. Freshwater Use by U.S. Power Plants: Electricity’s Thirst for a Precious Resource, A report of the Energy and Water in a Warming World Initiative. Cambridge, MA: Union of Concerned Scientists.
Brooks, B. W., T. M. Riley, and R. D. Taylor. 2006. Water quality of effluent-dominated ecosystems: Ecotoxicological, hydrological, and management considerations. Hydrobiologia 556(1):365-379. doi: 10.1007/s10750-004-0189-7.
Bryden, I. G., T. Grinstead, and G. T. Melville. 2004. Assessing the potential of a simple tidal channel to deliver useful energy. Applied Ocean Research 26(5):198-204.
Buth, J. M., P. O. Steen, C. Sueper, D. Blumentritt, P. J. Vikesland, W. A. Arnold, and K. McNeill. 2010. Dioxin photoproducts of triclosan and its chlorinated derivatives in sediment cores. Environmental Science and Technology 44(12):4545-4551. doi: 10.1021/es1001105.
Dinsmore, K. J., and M. F. Billett. 2008. Continuous measurement and modeling of CO2 losses from a peatland during stormflow events. Water Resources Research 44:W12417. doi: 10.1029/2008WR007284.
Fogg, G. E., and E. M. LaBolle. 2006. Motivation of synthesis, with an example on groundwater quality sustainability. Water Resources Research 42:W03S05. doi: 10.1029/2005WR004372.
Harter, T. 2005. Finite-size scaling analysis of percolation in three-dimensional correlated binary Markov chain random fields. Physical Review E 72(2):026120. doi: 10.1103/PhysRevE.72.026120.
Heffernan, J. B., and M. J. Cohen. 2010. Direct and indirect coupling of primary production and diel nitrate dynamics in a subtropical spring-fed river. Limnology and Oceanography 55(2):677-688.
Hess, L. L., J. M. Melack, E. M. L. M. Novo, C. C. F. Barbosa, and M. Gastil. 2003. Dual-season mapping of wetland inundation and vegetation for the central Amazon basin. Remote Sensing of Environment 87(4):404-428.
Hinck, J., V. Blazer, C. Schmitt, D. Papoulias, and D. Tillitt. 2009. Widespread occurrence of intersex in black basses (Micropterus spp.) from U.S. rivers, 1995-2004. Aquatic Toxicology 95(1):60-70. doi: 10.1016/j.aquatox.2009.08.001.
Kaushal, S. S., P. M. Groffman, G. E. Likens, K. T. Belt, W. P. Stack, V. R. Kelly, L. E. Band, and G. T. Fisher. 2005. Increased salinization of freshwater in the northeastern United States. Proceedings of the National Academy of Sciences of the United States of America 102(38):13517-13520. doi: 10.1073/pnas.0506414102.
Ma, S., G. W. Luther, J. Keller, A. S. Madison, E. Metzger, D. Emerson, and J. P. Megonigal. 2008. Solid state Au/Hg microelectrode for the investigation of Fe and Mn cycling in a freshwater wetland: Implications for methane production. Electroanalysis 20(3):233-239. doi: 10.1002/elan.200704048.
Mayorga, E. 2008. Carbon cycle: Harvest of the century. Nature 451 (7177):405-406. doi: 10.1038/451405a.
Nilsson, C., and B. M. Renöfält. 2008. Linking flow regime and water quality in rivers: A challenge to adaptive catchment management. Ecology and Society 13(2):18. Available online at http://www.ecologyandsociety.org/vol13/iss2/art18/ [accessed December 6, 2011].
Nimick, D. A., T. E. Cleasby, and R. B. McCleskey. 2005. Seasonality of diel cycles of dissolved trace metal concentrations in a Rocky Mountain stream. Environmental Geology 47(5):603-614. doi: 10.1007/s00254-004-1178-x.
NRC (National Research Council). 2008. Urban Stormwater Management in the United States. Washington, DC: The National Academies Press.
Postel, S. L., G. C. Daily, and P. R. Ehrlich. 1996. Human appropriation of renewable fresh water. Science 271(5250):785-788. doi: 10.1126/science.271.5250.785.
Raymond, P. A., N.-H. Oh, R. E. Turner, and W. Broussard. 2008. Anthropogenically enhanced fluxes of water and carbon from the Mississippi River. Nature 451:449-452. doi: 10.1038/Nature06505.
UN (United Nations). 2006. World Urbanization Prospects: The 2005 Revision. United Nations Department of Economic and Social Affairs, Population Division. Available online at http://www.un.org/esa/population/publications/WUP2005/2005WUPHighlights_Final_Report.pdf [accessed December 6, 2011].
Wollheim, W. M., B. A. Pellerin, C. J. Vörösmarty, and C. S. Hopkinson. 2005. N retention in urbanizing headwater catchments. Ecosystems 8(8):871-884. doi: 10.1007/s10021-005-0178-3.
Zhang, Y.-K., and K. E. Schilling. 2006. Increasing streamflow and baseflow in Mississippi River since the 1940s: Effect of land use change. Journal of Hydrology 324(1-4):412-422.
Zheng, C., M. Bianchi, and S. M. Gorelick. 2011. Lessons learned from 25 years of research at the MADE site. Ground Water 49(5):649-662. doi: 10.1111/j.1745-6584.2010.00753.x.
Cordella, D., J. O. Drangerta, and S. White. 2009. The story of phosphorus: Global food security and food for thought. Global Environmental Change 19(2):292-305. doi: 10.1016/j.gloenvcha.2008.10.009.
Foufoula-Georgiou, E., and C. Stark. 2010. Rethinking geomorphic transport: Stochastic theories, broad scales of motion and non-locality. Journal of Geophysical Research 115:F00A01. doi: 10.1029/2010JF001661.
Kolpin, D. W., E. T. Furlong, M. T. Meyer, E. M. Thurman, S. D. Zaugg, L. B. Barber, and H. T. Buxton. 2002. Pharmaceuticals, hormones, and other organic wastewater contaminants in U.S. streams, 1999-2000: A national reconnaissance. Environmental Science & Technology 36(6):1202-1211. doi: 10.1021/es011055j.
NRC. 2005. Contaminants in the Subsurface. Washington, DC: The National Academies Press.
Plummer, L. N. 2005. Dating of young groundwater. Pp. 193-220 in Isotopes in the Water Cycle: Past, Present, and Future of a Developing Science. P. Radeep, K. Aggarwal, J. R. Gat, and K. F. O. Froehlich (eds). Dordrecht, The Netherlands: Springer.
Schoups, G., J. W. Hopmans, C. A.Young, J. A.Vrugt, W. W. Wallender, and K. K. Tanji. 2005. Sustainability of irrigated agriculture in the San Joaquin Valley, California. Proceedings of the National Academy of Sciences of the United States of America 102(43):15352-15356. doi: 10.1073/pnas.0507723102.
Sklar, L. S., W. E. Dietrich, E. Foufoula-Georgiou, B. Lashermes, and D. Bellugi. 2006. Do gravel bed river size distributions record channel network structure? Water Resources Research 42:W06D18. doi: 10.1029/2006WR005035.
Whitehead, P. G., R. L. Wilby, R. W. Battarbee, M. Kernan, and A. J. Wade. 2009. A review of the potential impacts of climate change on surface water quality. Hydrological Sciences Journal 54(1):101-123. doi: 10.1623/hysj.54.1.101.