9
Homology and Homoplasy of Swimming Behaviors and Neural Circuits in the Nudipleura (Mollusca, Gastropoda, Opisthobranchia)
![]()
JAMES M. NEWCOMB,*AKIRA SAKURAI,† JOSHUA L. LILLVIS,† CHARUNI A. GUNARATNE,† AND PAUL S. KATZ†‡
How neural circuit evolution relates to behavioral evolution is not well understood. Here the relationship between neural circuits and behavior is explored with respect to the swimming behaviors of the Nudipleura (Mollusca, Gastropoda, Opithobranchia). Nudipleura is a diverse monophyletic clade of sea slugs among which only a small percentage of species can swim. Swimming falls into a limited number of categories, the most prevalent of which are rhythmic left–right body flexions (LR) and rhythmic dorsal–ventral body flexions (DV). The phylogenetic distribution of these behaviors suggests a high degree of homoplasy. The central pattern generator (CPG) underlying DV swimming has been well characterized in Tritonia diomedea and in Pleurobranchaea californica. The CPG for LR swimming has been elucidated in Melibe leonina and Dendronotus iris, which are more closely related. The CPGs for the categorically distinct DV and LR swimming behaviors consist of nonoverlapping sets of homologous identified neurons, whereas the categorically similar behaviors share some homologous identified neurons, although the exact composition of neurons and synapses in the neural circuits differ. The roles played by homologous identified neurons in categorically distinct behaviors differ. However, homologous identified neurons also play different roles even in the swim CPGs of the two LR swimming species.
__________
*Department of Biology, New England College, Henniker, NH 03242; and †Neuroscience Institute, Georgia State University, Atlanta, GA 30302. ‡To whom correspondence should be addressed. E-mail: pkatz@gsu.edu.
Individual neurons can be multifunctional within a species. Some of those functions are shared across species, whereas others are not. The pattern of use and reuse of homologous neurons in various forms of swimming and other behaviors further demonstrates that the composition of neural circuits influences the evolution of behaviors.
Behavior and neural mechanisms can be considered to represent two different levels of biological organization (Lauder, 1986, 1994; Striedter and Northcutt, 1991; Rendall and Di Fiore, 2007). Nevertheless, the evolution of behavior and the evolution of neural circuits underlying behavior are intertwined. For example, it has been suggested that the properties of neural circuits affect the evolvability of behavior); the evolution of particular behaviors could be constrained or promoted by the organization of neural circuits (Airey et al., 2000; Bendesky and Bargmann, 2011; Carlson et al., 2011; Katz, 2011; Yamamoto and Vernier, 2011). Darwin and the early ethologists recognized that behaviors, like anatomical features, are heritable characters that are amenable to a phylogenetic approach (Darwin, 1876; Whitman, 1899; Heinroth, 1911; Lorenz, 1981). The use of behavioral traits to determine phylogenies has been validated several times (Wenzel, 1992; De Queiroz and Wimberger, 1993; Proctor, 1996; Stuart et al., 2002), and the historical debates about homology and homoplasy of behavior have been thoroughly reviewed (Lauder, 1986, 1994; Wenzel, 1992; Foster et al., 1996; Proctor, 1996; Rendall and Di Fiore, 2007). Examining the neural bases for independently evolved (i.e., homoplastic) behaviors within a clade could provide insight into fundamental aspects of neural circuit organization. However, it is difficult enough to determine the neural basis for behavior in one species. Doing this in several species with quantifiable behaviors is even more challenging.
Studies of the neural bases of swimming behaviors in the Nudipleura (Mollusca, Gastropoda, Opisthobranchia) offer such a possibility. These sea slugs exhibit well differentiated categories of swimming behaviors, and their nervous systems have large individually identifiable neurons, allowing the neural circuitry underlying the swimming behaviors to be determined with cellular precision.
Here we will summarize what is known about the phylogeny of Nudipleura, their swimming behaviors, and the neural circuits underlying swimming. We will also provide data comparing the roles of homologous neurons. We find that neural circuits underlying the behaviors of the same category are composed of overlapping sets of neurons even if they most likely evolved independently. In contrast, neural circuits underlying categorically distinct behaviors use nonoverlapping sets of neurons. Furthermore,
homologous neurons can have different functions in different behaviors and even in similar behaviors.
PHYLOGENY OF NUDIPLEURA
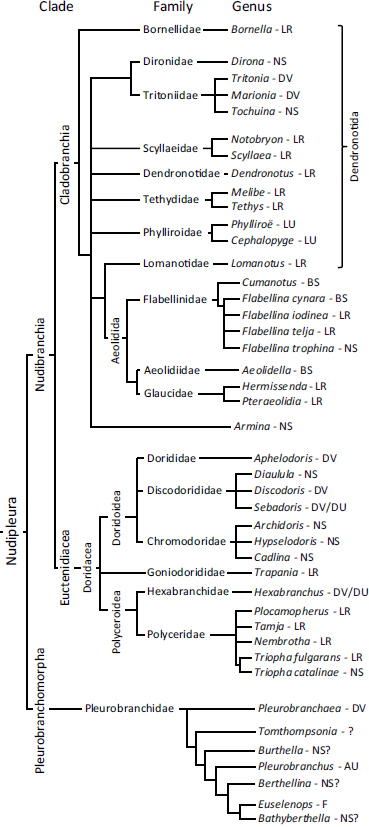
The Nudipleura form a monophyletic clade within Opisthobranchia (Gastropoda) that contains two sister clades: Pleurobranchomorpha and Nudibranchia (Waegele and Willan, 2000; Wollscheid-Lengeling et al., 2001; Göbbeler and Klussmann-Kolb, 2010) (Fig. 9.1). Molecular evidence suggests that the two sister groups separated approximately 125 Mya (Göbbeler and Klussmann-Kolb, 2010). Nudibranchia (or, informally, nudibranchs), which are shell-less and have a slug-shaped appearance with “naked gills,” were traditionally classified as their own order. The most recently agreed-upon taxonomic classification system for nudibranchs uses unranked clades instead of orders, suborders, and superfamilies (Bouchet and Rocroi, 2005). There are at least 2,000 to 3,000 identified nudibranch species (Behrens, 2005). Studies that used morphological and molecular data support the monophyly of Nudibranchia (Waegele and Willan, 2000; Wollscheid-Lengeling et al., 2001; Vonnemann et al., 2005; Dinapoli and Klussmann-Kolb, 2010; Göbbeler and Klussmann-Kolb, 2010; Pola and Gosliner, 2010).
Within Nudibranchia, there are two monophyletic clades (Waegele and Willan, 2000): Euctenidiacea (Anthobranchia) (Thollesson, 1999; Valdes, 2003) and Cladobranchia (Pola and Gosliner, 2010). Euctenidiacea includes Doridacea, which is larger than Cladobranchia, subdividing into 25 families (Thollesson, 1999). Within Cladobranchia, Bornellidae forms a sister group to the other subclades (Pola and Gosliner, 2010). Aeolidida is a monophyletic clade with Lomanotidae as a sister group (Pola and Gosliner, 2010). What was traditionally called Dendronotida forms a paraphyletic grouping. A recent study was unable to include the nudibranch Melibe in Cladobranchia because of a 12-bp deletion in its genome (Pola and Gosliner, 2010). However, its natural affinity with Tethys in terms of shared derived characteristics strongly suggests that it belongs in Cladobranchia, as we have indicated in Fig. 9.1. There are several additional unresolved relations in Nudibranchia, most notably in Dendronotida and Doridacea. Consideration of locomotor behavior and neural circuits may help resolve these relations.
CATEGORIES OF LOCOMOTOR BEHAVIOR
Crawling is the primary form of locomotion for all Nudipleura (Audesirk, 1978; Audesirk et al., 1979; Chase, 2002). The majority of species crawl via mucociliary locomotion; cilia on the bottom of the foot beat
and propel the animal over a surface of secreted mucus. The speed of crawling is affected by efferent serotonergic and peptidergic neurons that control the ciliary beat frequency (Audesirk, 1978; Audesirk et al., 1979; Willows et al., 1997). Some species also use muscular crawling, which relies on waves of contraction or extension and contraction of the foot. Crawling is a trait shared with most Opisthobranchia and is therefore plesiomorphic to the Nudipleura. Only three nudibranch species do not crawl because they are truly pelagic: Phylliroë atlantica, Phylliroë bucephala, and Cephalopyge trematoides (Lalli and Gilmer, 1989). This is also true for gastropods in general; there are ~40,000 marine gastropod species but only approximately 150 are pelagic (Lalli and Gilmer, 1989).
In addition to crawling, a limited number of benthic species can also swim (Farmer, 1970). We classify swimming in the Nudipleura into seven general categories: (i) left–right flexion (LR), (ii) dorsal–ventral flexion (DV), (iii) left–right undulation (LU), (iv) dorsal–ventral undulation (DU), (v) asymmetric undulation (AU), (vi) breaststroke (BS), and (vii) flapping (F) (Table 9.1).
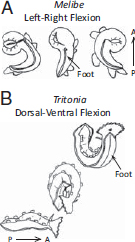
LR swimming is characterized by the flattening of the body in the sagittal plane and repeated left–right bending near the midpoint of the body axis with the head and tail coming together laterally (Fig. 9.2A). This movement propels the animal through the water. Some animals, such as Melibe leonina, exhibit foot-first directionality, presumably because the dorsal cerata create drag. Other animals, such as Tambja eliora, proceed headfirst, with the tail lagging slightly, causing the body to take on an “S” form (Farmer, 1970). Animals in the genus Plocamopherus typically have a dorsal crest at the posterior end of the body that may act as a paddle and cause the head to proceed the tail (Rudman and Darvell, 1990).
FIGURE 9.1 An abbreviated phylogeny of the Nudipleura with reference to their behavior. Only the genera of the species listed in Table 9.1 are shown here unless species differences exist within the genus. The phylogenic relationships are based on Thollesson (1999), Waegele and Willan (2000), Wollscheid-Lengeling et al. (2001), Vonnemann et al. (2005), Göbbeler and Klussmann-Kolb (2010), and Pola and Gosliner (2010). The references for the behavior are listed in Table 9.1. Note that this figure represents all the known swimming species and only a tiny fraction of the more than 2,000 species that are not capable of swimming or for which there are no published reports of swimming. LR, left–right flexion; NS, nonswimmer; DV, dorsal–ventral flexion; LU, left–right undulation; BS, breaststroke; DU, dorsal–ventral undulation; AU, asymmetric undulation; F, flapping.
TABLE 9.1 Abbreviated Nudipleura Taxonomy with Reference to Swimming
| Taxonomy | Swim Type | References |
| Nudibranchia | ||
| Cladobranchia | ||
| Aeolidida | ||
| Aeolidioidea | ||
| Aeolidiidae | ||
| Aeolidiella alba | BS | Pruvot-Fol (1954), Farmer (1970) |
| Glaucidae | ||
| Hermissenda crassicornis | LR | Lillvis et al. (2012) |
| Flabellinoidea | ||
| Flabellinidae | ||
| Flabellina cynara | BS | Marcus and Marcus (1967), Farmer (1970) |
| Flabellina iodinea | LR | MacFarland (1966), Farmer (1970) |
| Flabellina telja | LR | Marcus and Marcus (1967), Farmer (1970), Ferreira and Bertsch (1972) |
| Flabellina trophina | NS | a |
| Cumanotus beaumonti | BS | Picton and Morrow (1994) |
| Cumanotus cuenoti | BS | Tardy and Gantes (1980) |
| Arminoidea | ||
| Armina californica | NS | a |
| Dendronotidab | ||
| Bornellidae | ||
| Bornella anguilla | LU | Johnson (1984) |
| Bornella calcarata | LR | Thompson (1980) |
| Bornella stellifer | LR | Risbec (1953), Farmer (1970), Willan and Coleman (1984) |
| Dendronotidae | ||
| Dendronotus albopunctatus | LR | Robilliard (1972) |
| Dendronotus albus | LR | Farmer (1970), Robilliard (1970) |
| Dendronotus dalli | LR | Robilliard (1970) |
| Dendronotus diversicolor | LR | Robilliard (1970) |
| Dendronotus frondosus | LR | Farmer (1970), Robilliard (1970) |
| Dendronotus iris | LR | Kjerschow-Agersborg (1922), Haefelfinger and Kress (1967), Marcus and Marcus (1967), Farmer (1970), Robilliard (1970) |
| Taxonomy | Swim Type | References |
| Dendronotus nanus | LR | Marcus and Marcus (1967), Farmer (1970), Robilliard (1972) |
| Dendronotus rufus | LR | Robilliard (1970) |
| Dendronotus subramosus | LR | Farmer (1970), Robilliard (1970) |
| Lomanotidae | ||
| Lomanotus genei | LR | Garstang (1890), Thompson and Brown (1984) |
| Phylliroidae | ||
| Phylliroë atlantica | LU | Lalli and Gilmer (1989) |
| Phylliroë bucephala | LU | Lalli and Gilmer (1989) |
| Cephalopyge trematoides | LU | Steinberg (1956), Lance (1968) |
| Scyllaeidae | ||
| Notobryon wardi | LR | Thompson and Brown (1981) |
| Scyllaea pelagica | LR | Collingwood (1879), Pruvot-Fol (1954), Farmer (1970) |
| Tethydidae | ||
| Melibe bucephala | LR | Schuhmacher (1973) |
| Melibe engeli | LR | Risbec (1937) |
| Melibe fimbriata | LR | Thompson and Crampton (1984) |
| Melibe japonica | LR | Willan and Coleman (1984) |
| Melibe leonina | LR | Kjerschow-Agersborg (1921), Hurst (1968), Farmer (1970), Lawrence and Watson (2002) |
| Melibe megaceras | LR | Gosliner (1987b) |
| Melibe pilosa | LR | Pease (1860), Farmer (1970), Ostergaard (1955) |
| Tethys fimbria | LR | Pruvot-Fol (1954), Farmer (1970) |
| Dironidae | ||
| Dirona picta | NS | a |
| Dirona albolineata | NS | a |
| Tritoniidae | ||
| Marionia blainvillea | DVc | Pontes (2002) |
| Marionia tethydes | DVc | Haefelfinger and Kress (1967) |
| Tritonia diomedea | DV | Willows (1967), Hume et al. (1982) |
| Tritonia festiva | DV | Birkeland (1974) |
| Tritonia hombergii | DV | Willows and Dorsett (1975) |
| Taxonomy | Swim Type | References |
| Euctenidiacea | ||
| Doridacea | ||
| Doridoidea | ||
| Dorididae | ||
| Aphelodoris antillensis | DV | Quiroga et al. (2004) |
| Aphelodoris brunnea | DV | Gosliner (1987a) |
| Aphelodoris gigas | DV | Wilson (2003) |
| Aphelodoris karpa | DV | Wilson (2003) |
| Aphelodoris varia | NS | Wilson (2003) |
| Discodorididae | ||
| Diaulula sandiegensis | NS | a |
| Discodoris evelinae | DV | Marcus (1955), Marcus and Marcus(1967) |
| Discodoris pusae | DV | Marcus (1955) |
| Sebadoris nubilosa | DV/DUd | Marcus and Marcus (1967), Farmer (1970) |
| Chromodoridae | ||
| Archidoris odhneri | NS | a |
| Archidoris montereyensis | NS | a |
| Hypselodoris picta | NS | a |
| Cadlina luteomarginata | NS | a |
| Onchidoridoidea | ||
| Goniodorididae | ||
| Trapania velox | LRf | Cockerell (1901), Farmer (1970) |
| Polyceroidea | ||
| Hexabranchidae | ||
| Hexabranchus aureomarginatus | DV/DUd | Neu (1932), Ostergaard (1955), Farmer (1970) |
| Hexabranchus morsomus | DV/DUd | Risbec (1928), Marcus and Marcus (1962) |
| Hexabranchus sanguineus | DV/DUd | Risbec (1928), Gohar and Soliman (1963), Vincente (1963), Edmunds (1968), Farmer (1970) |
| Hexabranchus tinkeri | DV/DUd | Ostergaard (1955), Farmer (1970) |
| Polyceridae | ||
| Nembrotha megalocera | LR | Yonow (1990) |
| Plocamopherus ceylonicus | LR | Willan and Coleman (1984), Rudman and Darvell (1990) |
| Taxonomy | Swim Type | References |
| Plocamopherus imperialis | LR | Willan and Coleman (1984), Ellis (1999a), Marshall and Willan (1999) |
| Plocamopherus maculatus | LR | Pease (1860) |
| Plocamopherus maderae | LR | Lowe (1842) |
| Plocaompherus tilesii | LR | Rudman and Darvell (1990), Ellis (1999b) |
| Tambja blackii | LR | Pola et al. (2006) |
| Tambja eliora | LR | Lance (1968), Farmer (1970) |
| Tambja morose | LR | Marshall and Willan (1999) |
| Triopha fulgurans | LR | Risbec (1925), Farmer (1970) |
| Triopha catalinae | NS | a |
| Pleurobranchomorpha | ||
| Pleurobranchidae | ||
| Euselenops luniceps | Fe | Pace (1901), Farmer (1970) |
| Pleurobranchaea californica | DV | Gillette et al. (1991), Davis and Mpitsos (1971) |
| Pleurobranchus membranaceus | AU | Thompson and Slinn (1959), Farmer (1970) |
NOTE: This taxonomy is based upon that of Bouchet and Rocroi (2005). Abbreviations: AU = asymmetric undulation; BS = breaststroke; DU = dorsal–ventral undulation; DV = dorsal–ventral flexion; F = flapping; LR = left–right flexion; LU = left–right undulation; NS = nonswimmer.
aTested with mechanical and salt stimuli in our laboratories.
bA paraphyletic group (Pola and Gosliner, 2010).
cFarmer (1970) reported that Marionia swim via left–right flexions and cited a German reference (Haefelfinger and Kress, 1967). However, a translation of this reference into English, by P. Katz, indicates that Haefelfinger and Kress reported dorsal–ventral flexions.
dFarmer (1970) categorized swimming in Sebadoris and Hexabranchus as “flapping.” However, swimming in these species appears to include dorsal–ventral flexions of the body, in addition to undulations of the mantle.
eVideo observation.
fFarmer (1970) classified Trapania velox as an LR swimmer. However, see text for additional discussion.
Plocamopherus ceylonicus (Rudman and Darvell, 1990; Marshall and Willan, 1999) and Plocamopherus maderae (Lowe, 1842) swim with LR flexions when dislodged from a substrate or disturbed in some way. Tambja appears to use LR swimming as an escape response; contact with the predacious nudibranch Roboastra will elicit swimming in Tambja (Farmer, 1970; Pola et al., 2006). LR swimming in Melibe and Dendronotus iris can be initiated in response to loss of contact with the substrate or in response to the touch of a predatory sea star (Lawrence and Watson, 2002; Sakurai et al., 2011).
FIGURE 9.2 Two examples of swimming behaviors. (A) LR swimming exhibited by M. leonina. The ventral side of the animal is shown with the mouth at the top of the image. During swimming, the foot is narrowed to a strip and the animal rhythmically flexes its body leftward and rightward, bending at a point midway along the body axis. (B) DV swimming exhibited by T. diomedea. The animal starts on the substrate, shown at the bottom with its head to the right. It launches with a ventral flexion, where the head and tail meet under the foot. Then, it flexes so that the head and tail meet above the dorsal body surface. The foot is flattened and expanded to the width of the body. A, anterior; P, posterior.
Melibe may also swim seasonally to disperse (Mills, 1994). The flexion cycle period for Melibe and Dendronotus is approximately 3 s, and swim bouts can last many minutes (Lawrence and Watson, 2002; Sakurai et al., 2011).
As its name suggests, Bornella anguilla swims with an eel-like movement caused by waves of muscular contraction (Johnson, 1984). Therefore, unlike other members of its genus, it is classified as an LU swimmer. LU swimming, which otherwise is found mostly in pelagic species, may be a further refinement of LR swimming.
DV swimming involves the animal flattening its body in the horizontal plane and repeatedly bending such that the tail and head meet in alternation above and below the midpoint of the body (Fig. 9.2B). Tritonia diomedea and Pleurobranchaea californica are two examples of DV swimmers that have been extensively studied (Willows, 1967; Davis and Mpitsos, 1971; Gillette and Jing, 2001; Katz, 2009). Swim bouts for Tritonia and Pleurobranchaea last less than 1 min and are triggered by contact with a predatory sea star or in the laboratory by high salt solutions or electric shock (Katz, 2010). The flexion cycle period under natural conditions is 5 to 10 s in Tritonia (Hume et al., 1982) and 3 to 6 s in Pleurobranchaea (Jing and Gillette, 1995).
DU swimming, like DV swimming, involves movement in dorsal and ventral directions, but here there are progressive symmetric waves of body wall or mantle muscular contraction. The Spanish dancer, Hexabranchus sanguineus, and other members of that genus are famous for their flamboyant swimming behavior (Gohar and Soliman, 1963; Edmunds, 1968; Farmer, 1970). Hexabranchus swimming differs in several ways from the DV swimming of Tritonia and Pleurobranchaea; in addition to the symmetrical undulation of the lateral fringes of the mantle, it has a shorter flexion
cycle period (2–4 s), swim bouts occur spontaneously, and swimming can last for long periods of time.
F swimming is similar to DV swimming in that the movement is bilaterally symmetric and dorsal–ventral in orientation, but instead of the head and tail meeting, the lateral edges of the mantle or foot rise and fall. F swimming is much more common in Opisthobranchia outside of the Nudipleura, such as Clione limacina (Arshavsky et al., 1986) and many species of Aplysia (Bebbington and Hughes, 1973; Donovan et al., 2006).
AU and BS are less common forms of locomotion. AU is characteristic of Pleurobranchus membranaceus (Thompson and Slinn, 1959) in which the animal swims upside down using its mantle as a passive keel while producing alternating muscular waves along its foot. BS involves the use of appendages including cerata and tentacles to stroke the water in a manner similar to a human swimmer’s movements. Only four nudibranch species have been described as exhibiting this type of behavior (Table 9.1).
PHYLOGENETIC DISTRIBUTION OF SWIMMING BEHAVIORS
As noted earlier, we have been unable to find reports of swimming by about 97% of nudibranch species and approximately half the major subfamilies in the Pleurobranchomorpha clade. However, this does not mean they are not capable of swimming. Some species swim only as a high threshold escape response. Still, it is highly probable that the vast majority of the Nudipleura cannot and do not swim. This discussion is limited to species for which the type of swimming has been reported or for which swimming has been explicitly tested and shown not to occur.
LR swimming is by far the most prevalent of the six modes of swimming exhibited by nudibranchs: of the 60 nudibranch species documented to swim in the scientific literature, 40 species use LR or LU (Table 9.1). These 40 species are phylogenetically disparate, encompassing species in Doridacea and Cladobranchia (Fig. 9.1). Within the latter, there are LR swimmers in Aeolidoidea and Dendronotoidea. In Doridacea, all but one of the LR swimmers are in the family Polyceridae. There are no LR swimmers in the Pleurobranchomorpha or, to our knowledge, in any other Opisthobranch clade. This suggests that LR swimming is a derived characteristic of the nudibranch clade.
Unlike LR swimming, DV swimming is found in Nudibranchia and in Pleurobranchomorpha (Fig. 9.1). DV swimming is not present outside of Nudipleura and is therefore likely to be a synapomorphy of this clade. However, it is not widely displayed within Nudibranchia, appearing in just one family of Dendronotida (Tritoniidae) and in three families of Doridacea (Discodorididae, Dorididae, and Hexibranchidae). Discodorididae and Hexibranchidae also exhibit dorsal–ventral undulations (i.e., DU).
EVOLUTION OF SWIMMING BEHAVIORS
There are a number of possible scenarios that could account for the phylogenetic distribution of swimming behaviors among the Nudipleura. Considering the extreme rarity of swimming, it is possible, maybe even likely, that swimming evolved on multiple occasions from nonswimming species. The repeated gain of a function such as rhythmic movement could suggest that there is a predisposition toward these behaviors. The repeated appearance of LR and DV swimming may simply indicate that these two basic movements are the most likely to occur in a slug-shaped body with few appendages. When appendages such as moveable cerata are present, they have been repeatedly used for BS swimming. In the absence of such appendages, the only means of swimming are with LR-like or DV-like movements.
Given the presence of swimming across the phylogeny, it is possible that, rather than evolving independently many times from nonswimmers, swimming behaviors were repeatedly lost. Although this may lead to more transformations, it may be easier to lose a character than to gain one, as has been seen in other systems (Whiting et al., 2003; Moczek et al., 2006; Wiens et al., 2007; Harshman et al., 2008; Duboué et al., 2011).
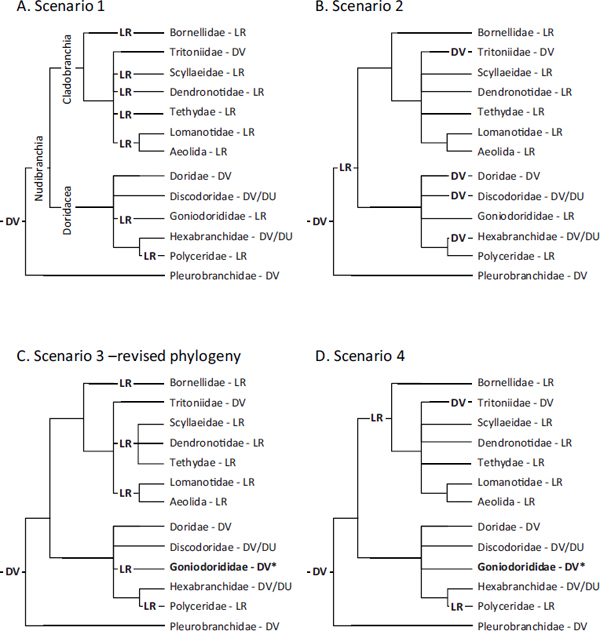
For the moment, we will only consider the possible evolutionary scenarios that include transformations from one swimming state to another and ignore nonswimmers. It is generally the case that members of the same genus and often the same family exhibit the same form of swimming (Table 9.1), allowing us to group them together (Fig. 9.3). Here we will consider potential scenarios involving just the evolution of DV and LR swimming. It is possible that the ancestral species was able to swim using either DV or LR movements. However, this seems unlikely because there are no extant species that exhibit both of these behaviors. It is also unlikely that the ancestral state was LR swimming because of its absence in Pleurobranchomorpha.
Consider scenario 1 (Fig. 9.3A) in which DV swimming arose once at the base of the Nudipleura and LR swimming evolved independently several times. In this scenario, DV swimming behaviors in Pleurobranchomorpha, Doridacea, and Cladobranchia are homologous because they are shared by a common ancestor. Scenario 1 would also suggest that LR swimming evolved independently as many as seven times. Because of the unresolved branches in the phylogeny, there may be fewer switches in phenotype than this. In scenario 2 (Fig. 9.3B), LR swimming evolved once in the Nudibranchia, and DV swimming reevolved independently as many as four times. Again, the number of homoplastic events could be lower if the bifurcations in the phylogeny were better resolved.
The phylogenetic distribution of the swimming behavior suggests a resolution to the Dendronotida phylogeny, with Tritoniidae branching
FIGURE 9.3 Possible evolutionary scenarios explaining the phylogenetic distribution of swimming behaviors. Just the families of the DV and LR swimming animals are shown. (A) In scenario 1, DV swimming is a synapomorphy of the Nudipleura that was lost and replaced six times by LR swimming. (B) In scenario 2, LR swimming is a synapomorphy of the Nudibranchia. DV swimming then reappears four times in different nudibranch lineages. (C) For scenario 3, the phylogenetic tree of Dendronotida is altered to group LR swimmers together. Goniodorididae (asterisk), which includes T. velox, is switched from LR to DV (as discussed in the text). This reduces the number of transitions to LR from six in scenario 1 to four. (D) Scenario 4 is similar to scenario 2, with Goniodorididae (asterisk) switched to DV. This represents the most parsimonious explanation if DV swimming is ancestral, with just three transitions from the basal DV state.
off separately from the LR swimmers. This would reduce the number of homoplastic events in Cladobranchia according to scenario 1 from five to three (scenario 3; Fig. 9.3C).
The phylogenetic distribution of the behavior also calls into question the accuracy of a report about the behavior of Trapania velox. Outside of the family Polyceridae, T. velox (family: Goniodorididae) is the only doridacean reported to swim with left–right flexions. Farmer (1970) categorized T. velox as an LR swimmer based on a previous report by Cockerell (1901), who described T. velox as being, “very active when swimming with an undulating motion on the surface of the water.” However, there is no indication as to the plane of movement. Farmer (1970) reported working with this rare species and being unsuccessful at making it swim, and was thus unable to provide any additional information. We were unable to find any other reports of its behavior. If T. velox is reclassified as a DV swimmer, it would further decrease the number of homoplastic events in scenario 1 from seven to four (Fig. 9.3C). Thus, examining the phylogenetic distribution of behavior makes a prediction about the behavior of this rare species.
Redefining T. velox as a DV swimmer also suggests a fourth scenario (Fig. 9.3D), whereby LR swimming arose independently in Cladobranchia and Polyceridae. This would also involve reevolution of DV swimming in Tritoniidae. Scenario 4 would therefore be the most parsimonious explanation for the phylogenetic distribution of swimming behaviors if one does not take into account the hundreds of nonswimming species.
NEURAL CIRCUITS UNDERLYING SWIMMING
With our potential scenarios about the homology and homoplasy of swimming behaviors, it is now of interest to compare the neural mechanisms for these behaviors. The neural activity that underlies rhythmic DV and LR movements originates from central pattern generator (CPG) circuits (Delcomyn, 1980). These swim CPGs are composed of neurons whose anatomical and physiological properties allow them to be individually identifiable from animal to animal within a species. The same sets of characteristics can be used to identify homologous neurons in other species (Croll, 1987). This allows the composition of neural circuits and the roles of homologous neurons to be compared across species. The neural circuits underlying swimming have been determined in two DV swimmers [T. diomedea (Katz, 2009) and P. californica (Gillette and Jing, 2001; Jing and Gillette, 1999)] and two LR swimmers [M. leonina (Sakurai et al., 2011; Thompson and Watson, 2005) and D. iris (Sakurai et al., 2011)]. We can now begin to compare neural circuits underlying behaviors of animals to address phylogenetic and functional hypotheses.
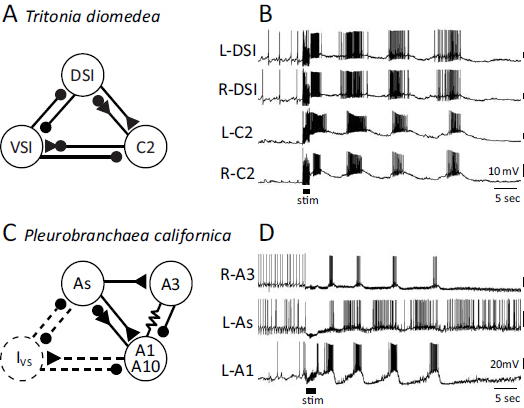
The neural basis for DV swimming was first studied in T. diomedea (Willows, 1967; Dorsett et al., 1969; Getting et al., 1980; Getting, 1981, 1983). The swim CPG consists of just three neuron types (Fig. 9.4A). On each side of the brain, there are three dorsal swim interneurons (DSIs), one ventral swim interneuron (VSI), and one cerebral interneuron 2 (C2), for a total of 10 neurons (Katz, 2009, 2010). The DSIs initiate the dorsal flexion cycle in which C2 participates. C2 then excites VSI, which inhibits DSI and C2 and elicits the ventral phase of the movement. As would be expected for a DV swimmer, the contralateral counterparts for each neuron fire in relative synchrony (Fig. 9.4B).
The neurons comprising the CPG for DV swimming in P. californica include DSI and C2 homologues called As and A1, respectively (Jing and Gillette, 1995, 1999). The connectivity and activity of these homologues is similar in both species (Fig. 9.4C and D). The homologue of the Tritonia VSI has not been identified in Pleurobranchaea, although there is synaptic input to As and A1 during the ventral phase of the motor pattern that may arise from such a neuron (i.e., Ivs neuron) (Jing and Gillette, 1999). Alternatively, ventral-phase synaptic input may arise from a neuron that is not homologous to VSI, but serves a similar role.
There are also Pleurobranchaea swim CPG neurons (A3 and A10) that have not been identified in Tritonia. Despite more than 40 years of electro-physiological study concentrated in the area where the A3 and A10 somata would be, no neurons with equivalent synaptic connectivity or activity have been found in Tritonia. Thus, either these neurons do not exist in Tritonia or they cannot be recognized with electrophysiological criteria.
With the information available about the swim CPGs in Tritonia and Pleurobranchaea, we can currently say that some homologous neurons are used for similar functions in distantly related species. This result is compatible with any of the phylogenetic scenarios (Fig. 9.3). If DV swimming is homologous (scenarios 1 or 3; Fig. 9.3A and C), the similarities in the DV swim CPGs in Tritonia and Pleurobranchaea could be a result of their homology and the potential differences in the swim CPGs could represent divergence of the circuit architecture. The differences in the swim CPGs may just as readily reflect independent evolutionary paths (scenarios 2 or 4; Fig. 9.3B and D), which might suggest a predisposition to use certain neurons to produce these behaviors.
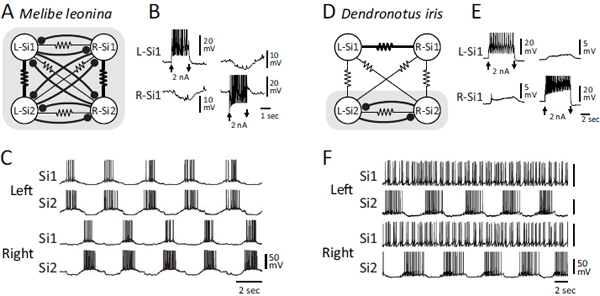
LR Swim CPGs
The LR swim CPG was first described in M. leonina (Watson et al., 2001; Thompson and Watson, 2005). The published circuit consists of a pair of bilaterally represented neurons: swim interneuron 1 (Si1) and swim
FIGURE 9.4 Neural circuits and swim motor patterns for the DV swimmers Tritonia and Pleurobranchaea. (A) The Tritonia swim CPG consists of three neuron types: DSI, C2, and VSI. (B) Simultaneous intracellular microelectrode recordings show that two contralateral DSIs fire bursts of action potentials in phase with each other and slightly ahead of the two C2s. VSI (not recorded here) fires action potentials in the interburst interval. The motor pattern is initiated by electrical stimulation of a body wall nerve (stim). (C) The Pleurobranchaea swim CPG contains five types of neurons (Jing and Gillette, 1999). The As neurons are homologues of the DSIs. A1 is homologous to C2. A10 is strongly electrically coupled to A1 and, for simplicity, is shown together with it. A3 is not found in Tritonia. The Ivs neuron has not been found, but has been postulated to exist based on recordings of inhibitory postsynaptic potentials in other neurons. (D) Simultaneous intracellular recordings from an A3, As, and A1. The As neuron leads the A1 neuron just as DSI leads C2. The swim motor pattern is initiated by electrical stimulation of a body wall nerve (stim). In A and C, the small filled circles represent inhibitory synapses, the triangles are excitatory synapses, and combinations are mixed inhibition and excitation. The resistor symbol represents electrical synapses.
interneuron 2 (Si2; Fig. 9.5A). Based on their anatomy and neurochemistry, these neurons are not homologous to any of the Tritonia or Pleurobranchaea swim CPG neurons.
In the Melibe swim CPG, each neuron reciprocally inhibits the two contralateral counterparts (Fig. 9.5B). There is also strong electrical coupling between the ipsilateral Si1 and Si2, causing them to fire in phase with each other and 180° out of phase with the contralateral pair (Fig. 9.5C). This bursting pattern drives the left–right alternations of the swimming behavior (Watson et al., 2002).
Homologues of the Melibe Si1 and Si2 were identified in D. iris based on anatomical, neurochemical, and electrophysiological features (Sakurai et al., 2011). However, there are important differences in the neural circuit formed by these neurons (Fig. 9.5D). Although the contralateral Si2 neurons reciprocally inhibit each other, Si1 does not inhibit or receive inhibition from either contralateral neuron. Instead, Si1 exhibits strong electrical coupling to its contralateral counterpart (Fig. 9.5E). During a swim motor pattern, the contralateral Si2 neurons fire bursts of action potentials in alternation, but the Si1 pair fire irregularly (Fig. 9.5F). Thus, whereas both Si1 and Si2 are members of the LR swim CPG in Melibe, only Si2 is in Dendronotus.
If LR swimming in Melibe and Dendronotus is homologous, as would be expected from scenarios 2, 3, or 4 (Fig. 9.3B–D), this would be an example in which the neural mechanisms diverged while the behavior stayed the same. However, it could be the case that the differences in neural mechanism reflect a different evolutionary origin for LR swimming in Melibe and Dendronotus as in scenario 1 (Fig. 9.3A).
FUNCTIONS OF DV SWIM CPG NEURONS IN OTHER SPECIES
DSI and C2 homologues can be recognized by using neuroanatomical and neurochemical criteria, allowing them to be identified in species that are not DV swimmers (Table 9.2). The DSIs are serotonergic (Katz et al., 1994; McClellan et al., 1994) and have a characteristic axon projection pattern (Getting et al., 1980). They have been identified in 10 different genera, including two opisthobranchs outside of the Nudipleura (Newcomb and Katz, 2007). Electrophysiological traits of the DSI homologues show little correlation with the type of behavior produced by the species (Newcomb and Katz, 2007). C2 has been identified based on peptide immuno-reactivity and characteristic morphology in five genera within the Nudipleura (Lillvis et al., 2012). These DV swim CPG neurons are present regardless of the animal’s mode of locomotion. This suggests that the swimming CPGs were built upon previously existing neural circuits, coopting existing neurons for new functions.
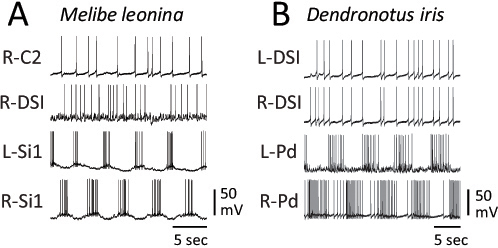
The DV swim CPG neurons are not members of the LR swim CPGs. The DSI and C2 homologs in Melibe are not rhythmically active in phase with the motor pattern (Fig. 9.6A), nor are the DSI homologues rhythmically active during the Dendronotus swim motor pattern (Fig. 9.6B). Thus,
FIGURE 9.5 Neural circuitry and swim motor pattern for the LR swimmers Melibe and Dendronotus. (A) In the Melibe swim CPG (Thompson and Watson, 2005), there are two bilaterally represented neurons Si1 and Si2 that are mutually inhibitory across the midline and exhibit strong electrical coupling ipsilaterally (as indicated by thicker resistor symbol). (B) Depolarization of one Si1 by injecting 2 nA of current into it hyperpolarizes the contralateral counterpart. (C) The Melibe swim motor pattern consists of ipsilateral synchrony and alternation with the contralateral side. (D) In Dendronotus, the inhibitory connections to and from Si1 are absent, and the electrical coupling between the contralateral Si1 pair dominates (Sakurai et al., 2011). (E) Depolarization of an Si1 with 2-nA current injection depolarizes the contralateral counterpart. (F) In the Dendronotus swim motor pattern, the left and right Si2 fire alternating bursts of action potentials, but the Si1s fire irregularly. In A and D, the shaded boxes represent the functional CPGs.
TABLE 9.2 Homologous Neurons Identified in Different Species with Different Behaviors
| Nudipleura | Other Opisthobranchia | |||
| Neuron | DV swimmers | LR swimmers | Nonswimmers | |
| DSI | Tritonia (Getting, 1977) | Melibe (Newcomb and Katz, 2007) | Armina (Newcomb and Katz, 2007) | Aplysia (Mackey et al., 1989; Wright et al., 1995; Xin et al., 2001; Jing et al., 2008) |
| Pleurobranchaea (Jing and Gillette, 1999) | Dendronotus (Newcomb and Katz, 2007) | Triopha (Newcomb and Katz, 2007) | Clione (Panchin et al., 1995; Satterlie and Norekian, 1995) | |
| Hermissenda (Tian et al., 2006) | Tochina (Newcomb and Katz, 2007) | |||
| C2 | Tritonia (Getting, 1977; Taghert and Willows, 1978) | Melibe (Lillvis et al., 2012) | ||
| Pleurobranchaea (Jing and Gillette, 1995) | Hermissenda (Lillvis et al., 2012) | |||
| Flabellina (Lillvis et al., 2012) | ||||
categorically distinct behaviors are produced by CPGs containing nonoverlapping sets of neurons.
It was shown that the DSI homologues in Melibe do have an effect on the production of the swim motor pattern; they can initiate a motor pattern in a quiescent preparation, and hyperpolarization can temporarily halt an ongoing motor pattern (Newcomb and Katz, 2009). In contrast to Tritonia, in which the DSIs are an integral part of the DV swim CPG, in Melibe, they act as extrinsic modulators. Thus, the functions of homologous neurons differ in species with different behaviors.
The DSIs are not dedicated to one function even within a species. In Pleurobranchaea, the DSI homologues synapse onto serotonergic neurons that increase ciliary beating and thereby increase the speed of crawling (Jing and Gillette, 2000). In Tritonia, DSI accelerates crawling through synapses onto the efferent peptidergic pedal neuron Pd5, which in turn increases cilia beat frequency (Popescu and Frost, 2002). DSI homologues in the nonswimming Tochuina tetraquetra and Triopha catalinae also monosynaptically excite homologues of Pd5 and presumably increase the speed of crawling (Newcomb and Katz, 2007). In Hermissenda, which produces LR flexions, the DSI homologues do not increase ciliary beating, but
FIGURE 9.6 Homologues of the Tritonia DV swim CPG neurons are not rhythmically active during LR swim motor patterns. (A) In Melibe, the C2 and DSI homologues do not display any rhythmic bursting in phase with the swim motor pattern reflected in the alternating firing pattern of the left and right Si. (B) In Dendronotus, a contralateral pair of DSI homologues exhibit synchronous irregular spiking that shows no relation to the ongoing LR swim motor pattern displayed by two contralateral pedal motor neurons (L-Pd and R-Pd).
instead excite motor neurons that cause contraction of the anterior foot (Tian et al., 2006). In the more distantly related opisthobranch, Aplysia californica, DSI homologues also initiate muscular crawling (Jing et al., 2008). Whereas, in the pelagic opisthobranch, C. limacina, the DSI homologues increase the frequency of parapodial “wing” flapping and excite motor neurons that innervate the wings (Arshavsky et al., 1992; Satterlie and Norekian, 1995). Thus, the DSI homologues share common functions in controlling the foot and/or locomotion.
The C2 and DSI homologues have additional roles outside of locomotion. In Pleurobranchaea, the C2 homologue (A1) suppresses feeding through its connections to feeding-related interneurons (Jing and Gillette, 1995). In contrast, the DSI homologues (As) have the opposite effect by exciting a number of feeding interneurons (Jing and Gillette, 2000). This is a shared function with other opisthobranchs such as A. californica, in which the DSI homologues (CC9-10) help excite one of the same feeding interneurons as in Pleurobranchaea, the metacerebral cell (Jing et al., 2008). Thus, individual neurons are multifunctional. Some functions are shared across species, whereas other functions are particular to some species.
CONCLUSIONS
A phylogenetic analysis of the neural basis for swimming in the Nudipleura has revealed several interesting aspects about the evolution
of behavior. First, the basic building blocks of neural circuits, namely the neurons, are shared across diverse species. For example, DSI homologues are found across Opisthobranchia. Second, neurons, which are multifunctional within a species, appear to take on additional functions over the course of evolution. For instance, the DSI homologues are involved in several behaviors in various species, including generating DV swimming or enhancing other types of locomotion such as enhancing LR swimming or wing flapping. They also accelerate crawling and promote feeding. It is reasonable to expect that highly interconnected interneurons would not be dedicated to a single function, but would dynamically interact with many neurons involved in a variety of different behaviors.
This comparative analysis has also revealed that species with categorically similar behaviors such as the two DV swimmers, Tritonia and Pleurobranchaea, or the two LR swimmers, Melibe and Dendronotus, have overlapping sets of neurons in the swim CPG circuits. In contrast, the CPGs underlying categorically distinct behaviors consist of nonoverlapping sets of neurons. However, even in species that exhibit similar behaviors such as Melibe and Dendronotus, the CPG circuits can differ in neuronal and synaptic composition. Thus, although behavior itself is not a predictor of its underlying neural mechanism, it is a good first approximation.
We do not understand why the circuits in Melibe and Dendronotus differ. There could be functional reasons; perhaps Si1, which is not rhythmically active in Dendronotus, has an additional function that is incompatible with swimming in that species. There may also be phylogenetic reasons; perhaps Melibe and Dendronotus independently evolved swim CPGs and came up with different circuit organizations. Whatever the reason, the results show that analogous behaviors can be generated by circuits with different circuit architectures. Recent work in invertebrates has shown that there can be variability in neural circuits that is not reflected in the performance of the behavior even across individuals within a species (Goaillard et al., 2009; Roffman et al., 2011).
There is a great degree of behavioral homoplasy. Although scenario 4 (Fig. 9.3D) may be the most parsimonious explanation for the phylogenetic distribution of the swimming behaviors, it should be kept in mind that only approximately 2% to 3% of nudibranch species have been reported to swim. Therefore, there is probably even more behavioral homoplasy than any of the scenarios in Fig. 9.3 indicate. It is conceivable that swimming arose independently in each family where it is found, 16 times in all (Fig. 9.1 and Table 9.1).
Given that Tritonia and Pleurobranchaea are very distantly related within the Nudipleura clade, it is even more likely that they independently evolved DV swim CPGs. If so, the incorporation of DSI and C2 homologues into such a circuit represents parallel evolution, whereby
homologous structures independently came to have similar functions (Sanderson and Hufford, 1996; Hoekstra and Price, 2004; Scotland, 2011; Wake et al., 2011). This has been suggested for other systems as well. For example, homologous brain nuclei appear to be involved in vocal learning in lineages of birds that evolved song independently (Feenders et al., 2008; Hara et al., 2012). Similarly, interaural coincidence detection circuits arose independently in the brainstem nuclei of birds and mammals (Schnupp and Carr, 2009). Finally, the appearance of similar cortical areas are correlates with the independent evolution of precision hand control in primates (Padberg et al., 2007), suggesting that constraints in cortical organization led to the evolution of similar neural mechanisms underlying dexterity (Krubitzer, 2009).
If homologous neurons are repeatedly incorporated into neural circuits for analogous behaviors, it suggests that these neurons may be part of a more readily achievable state for swimming. Thus, the nervous system may affect the evolvability of behavior because some configurations of existing neurons could be more robust than others. The concept of evolvability first arose from genetics (Kirschner and Gerhart, 1998; Masel and Trotter, 2010), but has since been applied to nervous systems (Airey et al., 2000; Bendesky and Bargmann, 2011; Katz, 2011; Yamamoto and Vernier, 2011). Exploring the aspects of neural organization that lead to repeated evolution of particular behaviors will point to the factors that are most important for behavioral output.
ACKNOWLEDGMENTS
We thank Arianna Tamvacakis for feedback on the manuscript. This work was supported by National Science Foundation Integrative Organismal Systems Grants 0814411, 1120950, and 1011476.