To Flock or Fight: Neurochemical Signatures of Divergent Life Histories in Sparrows
![]()
JAMES L. GOODSON,* LEAH C. WILSON, AND SARA E. SCHROCK
Many bird species exhibit dramatic seasonal switches between territoriality and flocking, but whereas neuroendocrine mechanisms of territorial aggression have been extensively studied, those of seasonal flocking are unknown. We collected brains in spring and winter from male field sparrows (Spizella pusilla), which seasonally flock, and male song sparrows (Melospiza melodia), which are territorial year-round in much of their range. Spring collections were preceded by field-based assessments of aggression. Tissue series were immunofluorescently multilabeled for vasotocin, mesotocin (MT), corticotropin-releasing hormone (CRH), vasoactive intestinal polypeptide, tyrosine hydroxylase, and aromatase, and labeling densities were measured in many socially relevant brain areas. Extensive seasonal differences are shared by both species. Many measures correlate significantly with both individual and species differences in aggression, likely reflecting evolved mechanisms that differentiate the less aggressive field sparrow from the more aggressive song sparrow. Winter-specific species differences include a substantial increase of MT and CRH immunoreactivity in the dorsal lateral septum (LS) and medial amygdala of field sparrows, but not song sparrows. These species differences likely relate to flocking rather than the suppression of winter aggression in field sparrows, because similar winter differences were found for two other emberizids that are not territorial in winter—
____________
Department of Biology, Indiana University, Bloomington IN 47405. *To whom correspondence should be addressed: E-mail: jlgoodso@indiana.edu.
dark-eyed juncos (Junco hyemalis), which seasonally flock, and eastern towhees (Pipilo erythropthalmus), which do not flock. MT signaling in the dorsal LS is also associated with year-round species differences in grouping in estrildid finches, suggesting that common mechanisms are targeted during the evolution of different life histories.
At the termination of the breeding season, many bird species leave their exclusive territories and join flocks that range from small parties to thousands of individuals. This dramatic seasonal shift in behavioral phenotype undoubtedly has profound fitness implications, but to our knowledge, no studies have addressed the neural or endocrine mechanisms that promote seasonal flocking. In contrast, mechanistic studies of avian territorial aggression are relatively extensive and have inarguably revolutionized the field of behavioral endocrinology (Wingfield, 2005; Soma, 2006). However, few of these studies explore the brain mechanisms of territoriality (Soma, 2006; Maney and Goodson, 2011). Using four emberizid songbird species that have evolved divergent life-history strategies, we here examine seasonal variation and evolutionary diversity in six neurochemical systems and demonstrate links of those systems to both winter flocking and territorial aggression.
On the basis of the immediate early gene responses of (i) male rodents to resident–intruder encounters, and (ii) male song sparrows (Melospiza melodia) to simulated territorial intrusion (playback of song and presentation of a caged male decoy), it seems that the neural substrates of territorial aggression are extensively comparable in birds and mammals. Thus, in both taxa significant activation is observed in the medial bed nucleus of the stria terminalis (BSTm), lateral septum (LS), paraventricular nucleus of the hypothalamus (PVN), anterior hypothalamus (AH), lateral portion of the ventromedial hypothalamus (VMH), and midbrain central gray [Kollack-Walker et al. (1997), Maney and Ball (2003), Goodson and Evans (2004), Goodson et al. (2005); also see Kingsbury et al. (2011)]. For the year-round territorial song sparrow, immediate early gene results are largely comparable in winter and summer (Goodson and Evans, 2004; Goodson et al., 2005), although microarray data suggest that hypothalamic responses to simulated intrusion are very different in winter and summer, perhaps reflecting the fact that luteinizing hormone is released during territorial challenges only in the breeding season (Mukai et al., 2009). Conversely, neurons that produce steroidogenic enzymes such as aromatase (ARO) may show greater activity in winter, given that territoriality in song sparrows shifts from reliance on gonadal steroids during the breeding season to nongonadal hormone production during the fall and winter (Wingfield, 2005; Soma, 2006).
Remarkably, neural mechanisms that influence group-size decisions have received very little attention, although recent studies have begun to address this topic using five estrildid finch species that exhibit relatively stable group sizes year-round. These studies show that multiple neurochemical systems have evolved in relation to grouping behavior, particularly within the LS and associated subnuclei of the posterior septum. Receptor densities for vasotocin (VT; homolog of the mammalian nonapeptide vasopressin), mesotocin (MT; homolog of the mammalian nonapeptide oxytocin), corticotropin-releasing hormone (CRH), and vasoactive intestinal polypeptide (VIP) all exhibit patterns of parallel and divergent evolution that closely track species-typical group size (Goodson et al., 2006, 2009b). Furthermore, VT neurons in the BSTm that project to the LS are sensitive to social valence and exhibit differential Fos responses in territorial and flocking species (Goodson and Wang, 2006). Antisense knockdown of VT production in those cells potently reduces gregariousness in the highly social zebra finch (Taeniopygia guttata) (Kelly et al., 2011), and antagonism of V1a-like and oxytocic receptors in the septum likewise reduces preferred group sizes (Goodson et al., 2009b; Kelly et al., 2011). The relative distribution of nonapeptide receptors across LS subnuclei may also be relevant to species differences in grouping, because flocking species have proportionally higher receptor binding in the dorsal (pallial) LS, whereas territorial species exhibit proportionally more binding in the subpallial LS (Goodson et al., 2006, 2009b). Consistent with these findings, septal VT infusions reduce territorial aggression in emberizid sparrows and estrildid finches (Goodson, 1998a,b). Finally, dopamine circuits are likely also relevant to grouping behavior, as gregarious finch species exhibit significantly more tyrosine hydroxylase-immunoreactive (TH-ir) neurons in the caudal ventral tegmental area (VTA) than do territorial species (Goodson et al., 2009a). The activity of these neurons is tightly coupled to courtship behavior, and perhaps to other aspects of affiliation as well (Goodson et al., 2009a).
These prior studies of avian sociality have focused exclusively on species that exhibit stable, year-round variation in species-typical group sizes (Goodson and Kingsbury, 2011). We hypothesize that the same neurochemical systems have evolved to mediate seasonal transitions between territoriality and flocking, but this remains to be determined. As a first approach to this hypothesis, we here quantify the neurochemical innervation of numerous brain areas in emberizid species that (i) alternate between gregarious and territorial phenotypes (field sparrow, Spizella pusilla, and dark-eyed junco, Junco hyemalis) (Carey et al., 1994; Nolan et al., 2002), (ii) are territorial year-round in much of their range (song sparrow) (Arcese et al., 2002), or (iii) switch from breeding territoriality to loose distributions in fall and winter, without flocking (eastern towhee, Pipilo erythropthalmus)
(Greenlaw, 1996). The four clades giving rise to these species diverged at approximately the same time, relatively early in emberizid phylogeny (Carson and Spicer, 2003). Our focus is on males, given that breeding territoriality is typically most intense in males. Complete data-sets from spring and winter birds are reported for song and field sparrows, including correlations with spring aggression. Winter differences that may reflect flocking in field sparrows were further explored in comparisons of winter juncos and towhees. Given that winter differences in neurochemistry between field and song sparrows potentially reflect differences in either winter aggression or winter flocking, the junco-towhee comparison is particularly useful. Specifically, we hypothesize that if winter differences between field and song sparrows reflect flocking, then juncos and towhees should exhibit a comparable winter difference. If winter differences between field and song sparrows reflect a lack of aggression in field sparrows, then juncos and towhees should not differ, because neither is territorial in winter.
We hypothesized that flocking-related changes in neurochemistry would be evidenced in one of two ways. Most obvious would be a winter increase in field sparrows (which flock in winter) that is not exhibited by song sparrows (which are territorial year-round). Alternatively, given that neurochemical circuits that promote winter flocking may also be involved in other affiliation behaviors that are expressed in the breeding season, such as pair bonding and caring for young, we hypothesized that field sparrows may maintain some neuroendocrine systems year-round that show a winter collapse in song sparrows. Both patterns are observed and are strongly supported by follow-up comparisons of juncos and towhees.
Finally, all of the substances examined here are made in multiple cell groups in the brain and may be relevant to a wide variety of behaviors, including both flocking and territoriality, dependent upon the brain area. For instance, whereas VT neurons in the BSTm respond primarily to affiliation-related social stimuli, those in the PVN are responsive to a diversity of stressors (Goodson and Kingsbury, 2011). TH cell groups likewise show great variation in response profiles (Charlier et al., 2005; Bharati and Goodson, 2006; Goodson et al., 2009a). We therefore do not combine analyses across all brain areas for each neurochemical, given that each neurochemical is not a unitary “system.”
RESULTS
General Approach
Tissue from field and song sparrows (n= 6 males per species and season; 24 total) was immunofluorescently multilabeled for VT, VIP, and
TH (series 1), and MT, CRH, and ARO (series 2). We were not uniformly satisfied with the quality of TH labeling in series 1, and therefore labeled a third series for TH using an antibody that yielded robust labeling in all subjects (Methods; a third series was not available for two spring subjects, one field and one song sparrow, because of earlier processing errors). We followed up on significant winter differences by labeling a single series of junco and towhee tissue for MT, CRH, and TH; and labeled a limited amount of tissue from a second junco–towhee series for VT and VIP. Note that for logistical purposes related to antibody lineups, most antigens were labeled using different fluorophores in the field–song and junco–towhee datasets, and thus labeling densities can only be compared within each species pair, not across.
Optical densities (ODs) of immunolabeling were measured in the medial preoptic nucleus, several hypothalamic areas (PVN, AH, and lateral and medial divisions of the VMH); anterior and posterior medial amygdala (MeA); BSTm; lateral BST; central gray; nucleus intercollicularis; rostral and caudal VTA; and nucleus accumbens. In addition, we quantified labeling in subnuclei of the septal complex that are differentiated on the basis of chemoarchitecture, peptide receptor distributions, and/or transcriptional responses to social stimuli (Goodson and Evans, 2004; Goodson et al., 2004, 2006, 2009b; Leung et al., 2011). These are the nucleus of the pallial commissure; caudocentral septum (CcS); rostral LS subdivision (LSr); and both pallial and subpallial portions of the caudal LS subdivision, which are denoted here as LSc.d and subpallial LSc (includes both ventral and ventrolateral subnuclei). The LSc.d and subpallial LSc were analyzed at rostral and caudal levels. In addition to OD, we conducted counts of TH-ir) cells in the VTA (A10 cell group), central gray (A11), dorsolateral tuberomammillary area (external mammillary nucleus; A12), and subparaventricular area (A14). VIP-ir cells were counted in the tuberal hypothalamus, and CRH, VT, and MT cells were counted in the PVN. Alpha values after Benjamini-Hochberg corrections for the false discovery rate (Benjamini and Hochberg, 1995) are reported in the figure captions and tables for the field and song sparrows, for which we collected full datasets (Methods). Results of Species × Season ANOVAs and within-species regressions with aggression are reported in the SI Appendix of Goodson et al. (2012b).
Neurochemical Signatures of Seasonal Flocking
As described in the Introduction, we hypothesized that flocking-related changes in neurochemistry would take the form of either (i) a winter increase in flocking field sparrows that is not exhibited by song sparrows, or (ii) the maintenance of some neuroendocrine systems year-round in field sparrows that show a winter collapse in song sparrows.
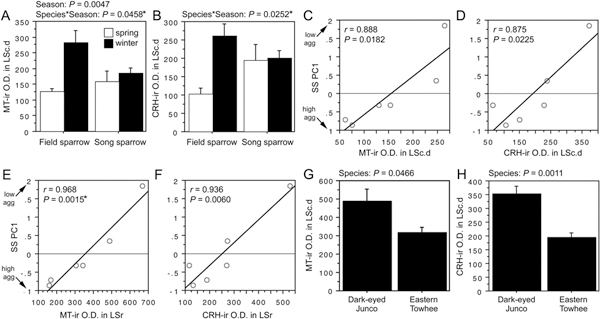
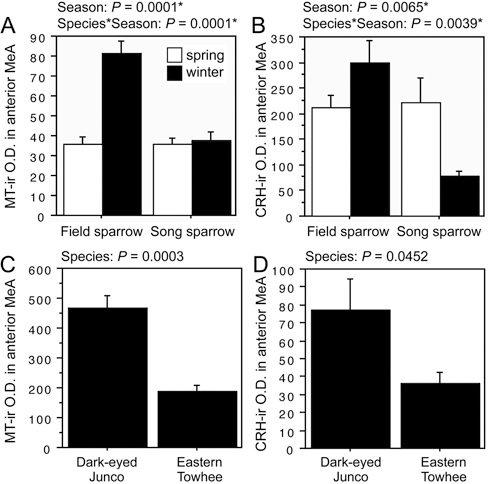
The first pattern is observed for both MT-ir and CRH-ir fiber densities in the anterior and posterior MeA (“nucleus taeniae”), and the rostral LSc.d [SI Appendix, Tables S1 and S2, of Goodson et al. (2012b)]. CRH is additionally increased in the LSr. The LS innervation consists of extremely fine-caliber processes that arborize most extensively in the pallial LS. In winter field sparrows, MT-ir processes form numerous light pericellular baskets. Similarly fine processes are observed in the MeA.
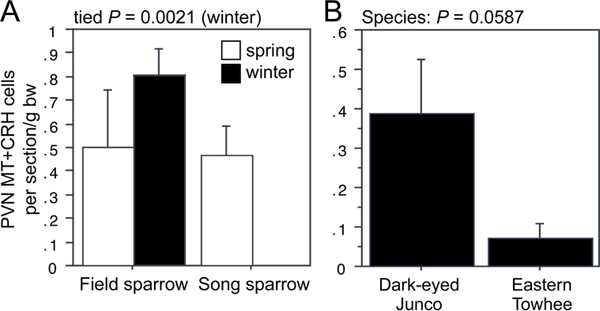
ANOVA results for the LSc.d are shown in Fig. 11.1A and B. Importantly, both MT-ir and CRH-ir fiber densities in the rostral LSc.d and LSr correlate negatively with multiple measures of aggression (Fig. 11.1C–F), and thus the increased densities in winter field sparrows may serve to suppress aggression rather than promote flocking. To address this issue, we quantified MT and CRH immunolabeling in wintering dark-eyed juncos, which flock, and eastern towhees, which loosely distribute in winter and do not flock. This comparison reveals significantly higher MT-ir and CRH-ir fiber densities in the rostral LSc.d of juncos relative to towhees (Fig. 11.1G and H) but no differences in CRH OD in the LSr (P= 0.07). A parallel set of results is obtained for MT and CRH OD in the anterior MeA (field > song; junco > towhee; Fig. 11.2), but juncos and towhees do not differ in the posterior MeA (MT, P= 0.28; CRH, P= 0.71). Notably, colocalization of CRH and MT in PVN neurons is significantly greater in winter field sparrows than song sparrows (Fig. 11.3A), and winter juncos likewise tend to show more colocalization than towhees (P < 0.06; Fig. 11.3B). Double-labeling does not correlate with measures of aggression (all P> 0.10).
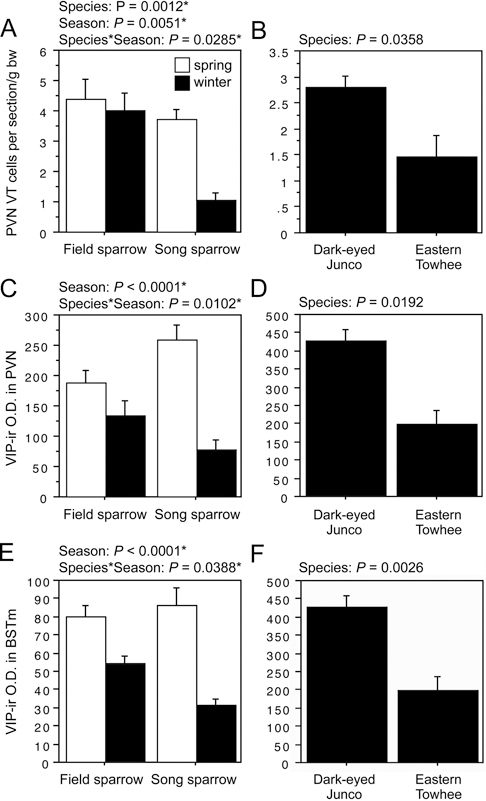
The second pattern described above, in which field sparrows maintain circuitry year-round that collapses during winter in song sparrows, is observed for VT-ir cell number in the PVN; and VIP OD in the PVN, AH, rostral subpallial LSc, CcS, and BSTm (in some cases field sparrows maintain relatively more but show a slight decline from spring). As shown in Fig. 11.4A and B, the field–song difference in VT neurons is matched by a similar difference between winter juncos and towhees, indicating a relationship to flocking. However, with the exception of VIP OD in the BSTm, the Species × Season effects for VIP are complex, with species differences in both winter and spring, but in different directions. That is, spring VIP OD measures in the PVN, AH, and septal areas are actually higher in song than in field sparrows. Furthermore, as described in the following section, AH and CcS measures correlate positively with spring aggression, which we did not anticipate for variables that promote flocking. Despite these complexities, we conducted follow-up comparisons in juncos and towhees, and although no differences are observed for VIP OD in the AH (P = 0.14) or CcS (P = 0.85; areas where VIP immunolabeling correlates positively with aggression), juncos do show greater VIP OD in the PVN and BSTm,
FIGURE 11.1 OD (in arbitrary units) of (A) MT-ir fibers and (B) CRH-ir fibers in the LSc.d of field and song sparrows collected in spring and winter, showing increased innervation density in winter field sparrows. (C–F) MT-ir and CRH-ir fiber densities correlate negatively with song sparrow aggression (SS PC1) in both the LSc.d (C and D) and LSr (E and F), suggesting that the increased innervation in winter field sparrows may suppress aggression rather than promote flocking. (G and H) However, comparisons of two species that are not territorial in winter show that MT-ir and CRH-ir fiber densities are greater in the flocking species (dark-eyed junco) than in the nonflocking species (eastern towhee). Data are shown as means ± SEM. *Significant after Benjamini-Hochberg corrections (sparrows).
FIGURE 11.2 OD (in arbitrary units) of (A) MT-ir fibers and (B) CRH-ir fibers in the anterior MeA of field and song sparrows collected in spring and winter, showing increased innervation density in winter field sparrows. (C and D) MT-ir and CRH-ir fiber densities are greater in the flocking dark-eyed junco than in the nonflocking eastern towhee. Data are shown as means ± SEM. *Significant after Benjamini-Hochberg corrections (sparrows).
following the pattern of higher fiber density in winter field sparrows relative to song sparrows. Relevant data are shown in Fig. 11.4C–F.
In addition to the patterns described above, one other finding initially suggested a possible relationship to flocking. This is a main effect of Species for TH immunolabeling in the rostral and caudal VTA, where field sparrows exhibit significantly higher TH-ir cell numbers and OD year-round relative to song sparrows [SI Appendix, Table S3, of Goodson et al. (2012b)]. Cell numbers also correlate negatively with aggression (next section). However, comparable differences are not exhibited by winter juncos and towhees, suggesting that the year-round difference between field and song sparrows reflects their year-round differences in aggression, as presented below.
Finally, no winter differences are exhibited for VT OD in the BSTm (as would be predicted from estrildids), although VT-ir fiber density in spring is significantly higher in field sparrows than in song sparrows [SI
FIGURE 11.3 (A) Number of PVN neurons double-labeled for MT and CRH in field and song sparrows. Because of a lack of variance in winter song sparrows, winter data were analyzed using Mann-Whitney tests. (B) A similar trend is observed for winter juncos and towhees.
Appendix, Fig. S1, of Goodson et al. (2012b)]. Again, as described in the next section, this is associated with species differences in aggression.
Neurochemical Signatures of Species-Specific Territorial Behavior
Before collections in the breeding season, we took three measures of territorial behavior during 3 min of song playback: latency to respond (by song, fly-by, or flyover), flights (defined as close fly-bys and flyovers), and songs. We then erected a mist net, began another round of playback, and took a second measure of response latency. Many measures of neurochemistry correlate significantly with these behavioral measures on a within-species level (next section). However, relevant to our focus on divergent life histories, we were particularly interested in determining whether measures of neurochemistry predicted species differences in aggression, given that that field sparrows are substantially less aggressive during the breeding season than are song sparrows.
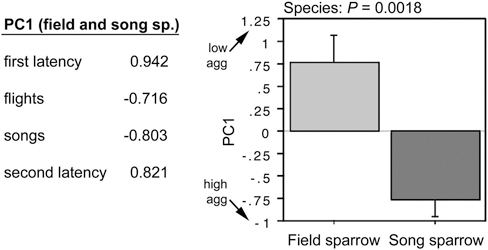
To quantify the species differences in aggression, we conducted a principal component (PC) analysis of the four behavioral measures, combining data for both species (P = 0.0029). This yields a single component (PC1) that strongly loads all four measures (Fig. 11.5) and explains 68% of the behavioral variance. A t test of PC scores confirms that song sparrows are more aggressive than field sparrows during the breeding season (Fig. 11.5), and more striking, PC scores for the two species are nonoverlapping.
FIGURE 11.4 (A–F) Left panels show VT-ir cell number in the PVN, VIP-ir OD (in arbitrary units) in the PVN, and VIP-ir OD in the BSTm of field and song sparrows. Right panels show corresponding data for juncos and towhees. Data are shown as means ± SEM. *Significant after Benjamini-Hochberg corrections (sparrows).
Thus, neurochemical measures that correlate with PC1 are strong candidates as mechanisms underlying evolutionary divergence in territoriality (although experience of aggression may also be a factor; see Discussion).
FIGURE 11.5 PC loadings from a combined analysis of field and song sparrow aggression (Left) and a comparison of PC scores by species (Right). PC1 explains 68% of the variance and yields non-overlapping values for field and song sparrows. Data are shown as means ± SEM.
Note that because of the strong loadings of latency measures, the direction of PC1 values is counterintuitive (i.e., higher PC scores reflect lower aggression). The PC1 score for one of the field sparrows was 2.8 standard deviations above the mean and thus this subject was excluded from the regressions.
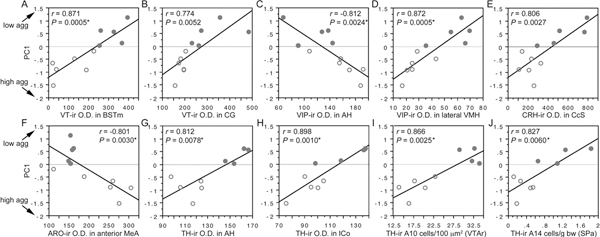
Regression analyses reveal significant negative correlations with PC1 (and thus positive correlations with aggression) for VIP OD in the AH and CcS; ARO OD in the posterior MeA (with a strong trend in the anterior MeA, as well); CRH OD in the posterior MeA and nucleus accumbens; and MT OD in the caudal subpallial LSc. In contrast, regression analyses reveal positive correlations with PC1 (and thus negative correlations with aggression) for VIP OD in the medial and lateral VMH; VT OD in the BSTm, central gray, and nucleus intercollicularis; CRH OD in the CcS; and TH OD in the medial preoptic nucleus, AH, LSr, and nucleus intercollicularis. In addition, TH-ir cell numbers in the rostral VTA, tuberomammillary hypothalamus, and subparaventricular area correlate positively with PC1. Ten of the strongest correlations are shown in Fig. 11.6. Note that significance is not obtained solely on the basis of large species differences, because data points within each species tend to follow the overall slope.
Individual Differences in Aggression
As just described, many neurochemical measures correlate with both individual and species differences in aggression. However, neurochemical variables may relate to individual differences within a given species without also relating to differences in aggression across species. We
FIGURE 11.6 (A–J) Regressions of neurochemical measures (OD, A–H; cell counts, I and J) and an index of aggression (PC1; Fig. 11.5) in field and song sparrows (closed and open circles, respectively). See x-axes for neurochemical variable and brain area. *Significant after Benjamini-Hochberg corrections (sparrows). CG, central gray; ICo, nucleus intercollicularis; SPa, subparaventricular area.
therefore conducted behavioral PC analyses for field and song sparrows independently. However, whereas a significant matrix is obtained for song sparrows (P= 0.0318), this is not the case for field sparrows (P= 0.60), likely because the field sparrows displayed few flights and songs, and little variation in those measures. Thus, we conducted regressions for field sparrows based on the average of their two latency measures, and for song sparrows based on a single-species PC (SS PC1), that explains 64% of the variance and exhibits strong loadings for flights (-0.913) and both latencies (0.901 and 0.928, respectively), but a weak loading for songs (-0.234). Results of these analyses are reported in the SI Appendix, Tables S7–S12, of Goodson et al. (2012b).
DISCUSSION
Although neuroendocrine mechanisms of seasonal territoriality have been extensively described (Wingfield, 2005; Soma, 2006; Maney and Goodson, 2011), those of seasonal flocking have not, and brain mechanisms that evolve in relation to species differences in the intensity of territorial aggression are likewise unknown. We now show that in emberizid songbirds, several neurochemical variables reflect seasonal shifts from territoriality to flocking, whereas numerous other variables correlate with both individual and species differences in territorial aggression. Given that the relevant neurochemical systems may be influenced by social interactions (e.g., via altered hormone levels), we must be cautious in our interpretations, because neurochemical variation may be the product of species differences in behavior rather than the drivers of it. However, as expounded upon in the following sections, other relevant findings suggest that many of the species differences are indeed products of evolution and mechanistic drivers of behavioral variation. Finally, our results reveal a remarkable degree of seasonal, neurochemical plasticity within socially relevant brain areas that is far more extensive than previously appreciated.
Neurochemical Profiles of Seasonal Flockers
Estrildid finches that are gregarious year-round exhibit nonapeptide binding sites in the rostral LSc.d (pallial LS) at much higher densities than do territorial estrildids (Goodson et al., 2006, 2009b). The relevance of these binding sites to flocking is supported by the demonstrations that intraventricular and intraseptal infusions of nonapeptide receptor antagonists (V1a and oxytocin receptor antagonists) reduce preferences for larger groups in the highly gregarious zebra finch (Goodson et al., 2009b; Kelly et al., 2011), as does antisense knockdown of VT-ir neurons in the BSTm (Kelly et al., 2011)—neurons that seem to provide the majority of VT-ir innervation to
the LS (De Vries and Buijs, 1983; De Vries and Panzica, 2006). Conversely, preferences for larger groups are facilitated by intraventricular infusions of MT (Goodson et al., 2009b). The present findings are strongly consistent with those in estrildids: field sparrows show a significant increase in MT-ir fiber density in the LSc.d during winter, when they form flocks, whereas the year-round territorial song sparrow does not. Flocking dark-eyed juncos likewise show a higher MT-ir fiber density in the LSc.d during winter than do nonflocking, nonterritorial eastern towhees. This pattern of MT results is replicated in the anterior MeA, and a very similar pattern of CRH innervation is observed in both the rostral LSc.d and anterior MeA.
Social affiliation in rodents is also linked to nonapeptide signaling in the LS. For instance, nonapeptide receptor densities in the LS increase in response to communal rearing (Curley et al., 2009), promote pair bonding (Liu Y et al., 2001), and correlate positively with both social investigation (Ophir et al., 2009) and maternal behaviors [and in the pallial LS specifically (Curley et al., 2012)]. Although the specific significance of peptide action in the pallial LS remains to be directly demonstrated, recent findings in mice demonstrate that the pallial LS plays an important role in linking contextual stimulus information to the activation of the mesolimbic dopamine system, which influences incentive motivational processes and reward (Luo et al., 2011). The functional properties of the anterior MeA are relatively less clear. In mammals, the posterior subnuclei have been far more extensively studied, although Newman (1999) has suggested that the anterior MeA exerts broad effects on social arousal. Homology of MeA subnuclei in birds and mammals remains to be demonstrated.
The finding that CRH innervation paralleled the MT innervation was unexpected, but is consistent with the fact that these two peptides are produced in many of the same neurons in the PVN and that colocalization is greater in winter flockers (Fig. 11.3). CRH is generally linked to anxiety-like processes and stress (Lovejoy and Balment, 1999), which may be the connection to flocking, given that thermoregulatory and foraging challenges lead to facultative grouping in many vertebrate species (Davies, 1976; Gilbert et al., 2010). Thus, we might hypothesize that winter flockers are in some sense hyperresponsive to the challenges of winter. This hypothesis also fits well with the observation that flocking birds exhibit significantly greater numbers of VT-ir PVN neurons in the winter than do nonflocking birds. Given that VT-ir fiber density collapses during winter in almost every brain area that we examined, it seems likely that these “extra” PVN neurons in flocking species project to the anterior pituitary, where VT acts as a secretagogue for adrenocorticopin hormone (Goodson and Bass, 2001) and thereby contribute to a higher glucocorticoid tone.
Finally, we observed complex patterns of VIP-ir fiber densities, some of which correlate positively with aggression (next section). However,
winter flocking (and not aggression) is associated with higher densities of VIP-ir fibers in the PVN and BSTm. Similarly, gregarious finch species exhibit higher densities of VIP binding sites in the BSTm than do territorial species (Goodson et al., 2006), providing additional evidence that VIP signaling in the BSTm promotes grouping.
Species Differences in Territorial Aggression
As shown here, field sparrows are significantly less aggressive than are song sparrows. Thus, the present dataset allows us to identify neurochemical mechanisms that may have evolved in relation to territorial behavior, because we are able to correlate measures of neurochemistry with aggressive behavior across both individuals and species. As a caveat to this approach, we observed widespread winter decreases in immunolabeling, suggesting the likelihood of positive relationships between gonadal hormones and labeling density. Thus, because male–male interactions typically elevate levels of testosterone (Wingfield, 2005), we must consider that any positive correlations between neurochemistry and behavior may be the product of male–male interactions and not the cause of it. For instance, ARO gene expression correlates positively with both aggression and plasma T in juncos (Rosvall et al., 2012). Nonetheless, most of the strongest relationships described here for neurochemistry and aggression are negative.
For instance, VT-ir fiber density in the BSTm collapses in winter, yet we also see that it correlates negatively with individual and species differences in aggression. This observation is consistent with the findings that (i) gregarious estrildids exhibit relatively more VT-ir neurons in the BSTm than do territorial species (Goodson and Wang, 2006), (ii) those neurons respond selectively to affiliation-related stimuli (Goodson and Wang, 2006), and (iii) infusions of VT into the septum (a major recipient of BSTm VT projections) reduce overt territorial aggression in both field sparrows and territorial finches (Goodson, 1998a,b).
Similarly, VIP immunolabeling correlates negatively with sparrow aggression in the lateral VMH and tuberal hypothalamus, but also positively in the AH and caudal septum. These results are strongly consistent with a variety of findings in territorial finches. For instance, intraseptal VIP infusions facilitate offensive aggression (Goodson, 1998b), whereas antisense knockdown of VIP production in the AH virtually abolishes it (Goodson et al., 2012a) (note that VIP-ir cells in the AH are only detectable after colchicine pretreatment and were thus not examined here). VIP-ir cell numbers in the AH of control finches correlate positively with aggression, but consistent with our present findings, VIP-ir cell numbers relate negatively to aggression in the tuberal hypothalamus [SI Appendix
in Goodson et al. (2012a)]. These finch data were obtained from birds in nonbreeding condition, suggesting that the positive relationship between AH VIP and aggression is not dependent upon gonadal steroids. Hence, VIP circuitries in the AH-CcS and mediobasal hypothalamus, which bear positive and negative relationships to aggression, respectively, are likely both relevant to behavioral evolution in sparrows.
We observed many other correlations across species that cannot be as readily interpreted because of a lack of direct functional data, but those findings nonetheless provide the basis for many hypothesis-driven experiments on the evolution of aggression.
Widespread Seasonal Plasticity
Although the present study was designed to focus on aggression and flocking, the analyses in field and song sparrows reveal a remarkable and unanticipated amount of seasonal plasticity, including all six neurochemical systems and 21 brain areas that we examined. Most remarkable are CRH and VIP. Seasonal plasticity has been shown for VIP within the septum and infundibulum (Kosonsiriluk et al., 2008; Wacker et al., 2008), but to our knowledge no such plasticity has been shown for the CRH innervation of the brain. However, we observed significant seasonal variation in 13 of the sampling areas for CRH, and 11 of the sampling areas for VIP. Seasonal plasticity for both peptides is exhibited in the MeA, BST, septal complex, medial preoptic nucleus, hypothalamic nuclei, and midbrain. Even in the case of VT, for which extensive seasonal and hormone-mediated plasticity is already known (as with VP in mammals) (Goodson and Bass, 2001; De Vries and Panzica, 2006), the extent of seasonal remodeling came as a surprise. Interestingly, the most extensive plasticity known for mammals comes from jerboas (Jaculus orientalis) that were collected in the field (Lakhdar-Ghazal et al., 1995), as were the animals in the present study, suggesting that exposure to a full range of seasonal cues is necessary to reveal the natural extent of seasonal plasticity.
CONCLUSIONS
We here hypothesized that flocking-related changes in neurochemistry take the form of either (i) a winter increase in flockers that is not exhibited by nonflocking species, or (ii) the maintenance of some neuroendocrine systems year-round in flockers that show a winter collapse in nonflockers. The first pattern is exhibited in the MT and CRH innervation of the pallial LS and anterior MeA, and in the colocalization of MT and CRH in the PVN. The second pattern is observed for VT-ir cell numbers in the PVN, and VIP innervation of the PVN and BSTm. A much larger number of neurochemical
variables seem to evolve in relation to territorial aggression, and all neurochemicals and brain areas examined here exhibit remarkable seasonal plasticity.
METHODS
Animals
Spring field and song sparrows were caught April thru May 2009 in the vicinity of Bloomington, IN. Wintering sparrows were caught in the vicinity of Bloomington, IN, and in Davidson County, TN, between December 2008 and February 2009. Juncos and towhees were collected in the vicinity of Bloomington, IN, in January 2010. Collections were made under applicable state and federal permits, and all procedures were in accordance with guidelines established by the National Institutes of Health for the ethical treatment of animals.
Tissue Processing and Image Analysis
Subjects were euthanized within 30 min of capture. Perfusions, tissue processing, and immunofluorescent labeling followed standard protocols (Goodson et al., 2004, 2009a; Kabelik et al., 2010). All Alexa Fluor (A.F.) conjugates were purchased from Invitrogen. Secondaries were raised in donkey. Sparrow series 1 was labeled using sheep anti-TH (Novus Biologicals), guinea pig anti-VP (Bachem), and rabbit anti-VIP (Bachem), with A.F. 488, biotin followed with streptavidin-A.F. 594, and A.F. 680 secondaries, respectively. Sparrow series 2 was labeled using custom sheep anti-ARO, rabbit anti-MT (VA10; a kind gift of H. Gainer, National Institute of Neurological Disorders and Stroke, Bethesda, MD), and guinea pig anti-CRH (Bachem), using A.F. 488, 594, and 680 secondaries, respectively. Sparrow series 3 was labeled using mouse anti-TH (Immunostar) and A.F. 594 secondary. The specificity of all antibodies has been addressed [Goodson et al. (2004), Kabelik et al. (2010); see company datasheets for TH]. Each processing run contained a mixture of species and seasons. Junco and towhee series 1 was labeled using rabbit anti-MT, mouse anti-TH, and guinea pig anti-CRH, with A.F. 488, 594, and 680 secondaries, respectively. Additional junco and towhee tissue was labeled using guinea pig anti-VP and rabbit anti-VIP, with A.F. 594 and 680 secondaries, respectively.
Although some larger areas with robust labeling were captured at 5×, most photomicrographs were obtained at 10× using a Zeiss AxioImager microscope outfitted with a Z-drive and optical dissector (Apotome; Carl Zeiss). OD of label and background was measured in Adobe Photoshop CS5 (Adobe Systems, Seattle, WA) from monochrome images, and background
values were subtracted for statistical analysis. Cell counts were conducted as previously described (Goodson and Wang, 2006; Goodson et al., 2009a). All cells were counted in each relevant section for smaller cell groups and are represented as number of cells per section/gram body weight. TH-ir cells in the VTA were counted within a standardized box and are represented as number of cells per 100 μm2.
Statistics
All ANOVAs, regressions, and PC analyses described in the Results were conducted using Statview 5.0 for Macintosh. Given the large number of analyses, some concern arises with regard to type I error, although all brain areas and neurochemicals examined here are known a priori to be relevant to social behavior (although not in all possible combinations). Corrections for multiple comparisons in such instances are usually too conservative and not appropriate (Rothman, 1990), and we therefore do not emphasize them in our interpretations. However, they may still provide a useful metric for evaluation; thus each of our data tables and figure panels provides information on significance relative to Benjamini-Hochberg corrections for the false discovery rate (Benjamini and Hochberg, 1995). Corrections were applied to each set of ANOVAs (e.g., for VT measures across all brain areas) and to each corresponding set of regressions. Again, though not emphasized in the Results, the robustness of our findings is notable; for example, 73 of 78 ANOVAs that yield P values < 0.05 were significant following corrections. Note that although the Benjamini-Hochberg correction initially applies a Bonferroni criterion, it adjusts α in a stepwise manner for remaining tests as long as P values continue to be significant at each step.
ACKNOWLEDGMENTS
We thank Francisco Ayala, John Avise, and Georg Striedter for inviting this contribution; Jacob Callis, Brian Gress, Alexis Howard, Aubrey Kelly, Melissa Knisley, and Brittany Welsh for assistance with immunocytochemistry and/or cell counts; Ellen Ketterson, Dawn O’Neal, and Ryan Kiley for assistance with collections; and Drew King and Meredith West for property access; Harold Gainer for the donation of antiserum. Support for this study was provided by Indiana University.
From Chemotaxis to the Cognitive Map: The Function of Olfaction
![]()
A paradox of vertebrate brain evolution is the unexplained variability in the size of the olfactory bulb (OB), in contrast to other brain regions, which scale predictably with brain size. Such variability appears to be the result of selection for olfactory function, yet there is no obvious concordance that would predict the causal relationship between OB size and behavior. This discordance may derive from assuming the primary function of olfaction is odorant discrimination and acuity. If instead the primary function of olfaction is navigation, that is, predicting odorant distributions in time and space, variability in absolute OB size could be ascribed and explained by variability in navigational demand. This olfactory spatial hypothesis offers a single functional explanation to account for patterns of olfactory system scaling in vertebrates, the primacy of olfaction in spatial navigation, even in visual specialists, and proposes an evolutionary scenario to account for the convergence in olfactory structure and function across protostomes and deuterostomes. In addition, the unique percepts of olfaction may organize odorant information in a parallel map structure. This could have served as a scaffold for the evolution of the parallel map structure of the mammalian hippocampus, and possibly the arthropod mushroom body, and offers an explanation for similar flexible spatial navigation strategies in arthropods and vertebrates.
____________
Department of Psychology, University of California, Berkeley, CA 94720. E-mail: jacobs@berkeley.edu.