Exposure science is essential for the protection of public health and the environment. However, the challenges and opportunities for exposure science are considerable. The ability to address them will influence advances in human health and ecosystem protection. Exposure science also will play a role in decision-making in other arenas, including consumer-product safety, environmental planning, climate-change mitigation, and energy development. This report provides a roadmap to navigate the future of exposure science to achieve greater integration and maximize its utility in the environmental and occupational health sciences, environmental-systems science, risk assessment, sustainability science, and industrial ecology.
Exposure science addresses the contact of humans and other organisms with chemical, physical, or biologic (CBP) stressors1 (EPA 2003; EPA 2011b) over space and time and the fate of these stressors within the ecosystem and organisms—including humans. Although methods of assessment will depend on the situation, exposure science has two primary goals: to understand how stressors affect human and ecosystem health and to prevent or reduce contact with harmful stressors or to promote contact with beneficial stressors to improve public and ecosystem health. The impact of environmental stressors on human and ecologic health is enormous.
For example, the World Health Organization estimates that 24-40% of global disease burden (healthy life-years lost) can be attributed to environmental factors (Smith et al. 1999; WHO 2004; Prüss-Üstün and Corvalán 2006). However, it is not possible to be exact in such calculations, partly because what is “environmental” is not defined consistently (see Box 1-1 for use of the term environmental in this report). In a burden-of-disease context, environmental
_______________________
1The Environmental Protection Agency defines a stressor as “any physical, chemical, or biological entity that can induce an adverse response” (EPA 2011a).
factors play a role in nearly all diseases, even ones that are not caused directly by environmental risk factors, by altering the course of disease initiated by other causes. In addition, if the total burden of disease is simply decomposed into “nature” or “nurture”, it fails to account directly for the possibly large proportion that could be due to the interplay between the two (gene—environment interactions). Improving our understanding of environmental factors and their relationships with disease is critical for preventing illness and death.
With respect to ecosystems, the 1999 National Research Council report Our Common Journey: A Transition Toward Sustainability (NRC1999) reported that the rising losses of wild nature, species number, species diversity, and ecosystem integrity were associated with exposures to environmental stressors, including those related to urban and agricultural land conversion and climate change.
Figure 1-1 illustrates the relationship of exposure to other key elements along the environmental-health continuum from the source of a stressor to an outcome. This figure has evolved from previous diagrams (for example, Smith 1988a; Lioy 1990; NRC 1998; EPA 2009a). For more than 20 years, this framework has demonstrated the central role of exposure science in environmental health science in that exposure sits midway between the sources of pollution (and other stressors) on the left—elements that typically can be controlled—and adverse health outcomes on the right, which need to be prevented. Exposure is strategically located upstream of dose and yet provides information and metrics that inform source control and health risk.
BOX 1-1 Definition and Scope of Exposure Science
Exposure science is defined by this committee as the collection and analysis of quantitative and qualitative information needed to understand the nature of contact between receptors (such as people or ecosystems) and physical, chemical, or biologic stressors. Exposure science strives to create a narrative that captures the spatial and temporal dimensions of exposure events with respect to acute and long-term effects on human populations and ecosystems.
For the purposes of this report, the committee focuses on environmental risk factors and excludes behavioral or lifestyle factors—such as diet, alcohol, and smoking—although it includes contaminants in food, water, and environmental tobacco smoke. It also excludes social risk factors (for example, crime and child abuse) but does consider them as modifying influences on exposures to stressors (Smith et al. 1999). The influence of social factors on environmental exposures is an area of active research. Natural hazards (for example, weather and arsenic contamination) are included here.
A central theme of this report is the interplay between the external and internal environments and the opportunity for exposure science to exploit novel technologies for assessing biologically active internal exposures from external sources.
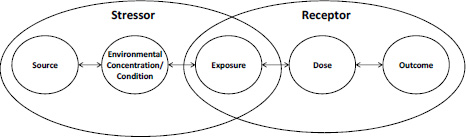
FIGURE 1-1 The classic environmental-health continuum. Figure 1-2 illustrates the revised version discussed in the present report. Source: Adapted from EPA 2009a.
There are many notable examples of the roles that exposure science can play in protecting public health. Consider how measurements of childhood blood lead concentrations since the 1970s reveal the dramatic efficacy of lead removal from gasoline in reducing exposure to this neurotoxicant in children (Muntner et al. 2005; Jones et al. 2009). Population-scale measurements of cotinine in urine document the reduction of exposure to second-hand tobacco smoke that resulted from control of tobacco-smoking in the workplace and public areas (EPA SAB 1992). Exposure modeling from the U.S. Environmental Protection Agency’s (EPA) National-Scale Air Toxics Assessment program has provided valuable information for communities on their exposure sources, concentrations, and risks and has helped to shed light on exposure disparities and environmental-justice issues (for example, Pastor et al. 2005).
Exposure science has played a critical role in understanding the influence of stressors on ecologic systems. For example, extensive exposure assessments of polycyclic aromatic hydrocarbons (PAHs) have been linked to liver damage in bottom-dwelling fish in Puget Sound, and field studies have demonstrated that containment of PAH sources has led to declines in PAH concentrations and a resulting decline in liver damage in fish (Myers et al. 2003).
Exposure science has applications in industrial, military, commercial, and global contexts. It is central to tracking chemicals and other agents that are introduced into global commerce at increasing rates, often with little information on their hazard potential (GAO 2005). Increasingly, exposure science is used for homeland security and the protection of deployed soldiers. Rapid detection of potentially harmful radiation or toxic chemicals is essential for protecting troops and the general public (IOM 2000). The ability to detect chemical contaminants in drinking water at low, biologically relevant concentrations quickly can help to identify emerging health threats, and monitoring of harmful algal blooms and airborne pollen can help to identify health-relevant effects of a changing climate.
As described in more detail in Chapter 3, applications of exposure science are critical for toxicology, epidemiology, risk assessment, and risk management. For example, toxicology provides information about how different chemical concentrations may affect public or ecologic health in laboratory studies or
computer models, but the value of information is greatly increased when it is combined with comprehensive and reliable exposure information. Similarly, epidemiology requires exposure information to compare outcomes in populations that have different exposures. Collection of better exposure data can also provide more precise information regarding alternative control or regulatory measures and lead to more efficient and cost-effective protection of public and ecologic health.
In addition to its applications to other fields, exposure science data can be used independently to define trends, assess spatial or population variability, provide information on prevention and intervention, identify populations or ecosystems that have disproportionate exposures, and evaluate regulatory effectiveness.2
Exposure science is also poised to play a critical role in improving the ability to understand and address increasingly important human health and ecologic challenges and to support the development of sustainable industrial, agricultural, and energy technologies. Recognizing the need for a prospective examination of exposure science, EPA and the National Institute of Environmental Health Sciences asked the National Research Council to develop a long-range vision for exposure science and a strategy for implementing the vision over the next 20 years (see Appendix B for statement of task). In response to this request, the National Research Council convened the Committee on Human and Environmental Exposure Science in the 21st Century. The committee—which comprised experts in monitoring, modeling, environmental transport and transformation, geographic information science and related technologies, measurement and analytic techniques, risk assessment and risk management, epidemiology, occupational health, risk communication, ethics, informatics, and ecologic services—prepared this report.
DEFINING THE SCOPE OF EXPOSURE SCIENCE
Exposure science—sometimes defined as the study of the contact between receptors (such as humans or ecosystems) and physical, chemical, or biologic stressors—can be thought of most simply as the study of stressors, receptors, and their contact, including the roles of space and time. For example, ecosystems are receptors for such stressors as mercury, which may cascade from the ecosystem to populations to individuals within the ecosystem because of concentration and accumulation in the food web, which leads to exposure of humans and other species. As the stressor (mercury in this case) is absorbed into
_______________________
2In 2011, the International Society of Exposure Science and the Journal of Exposure Science and Environmental Epidemiology published a compendium of digests (Graham 2011) that illustrate situations in which application of exposure science resulted in substantial health or policy benefits and situations in which lack of exposure information resulted in adverse consequences.
the bodies of individuals, it may come into contact with other receptors, such as tissues and organs.
As the scientific communities generating and using exposure data have evolved, so have the terms and definitions used to characterize exposures. Some refer to dose (exposure dose, target dose, or external dose), others to exposure (for example, external or internal exposure), and yet others to an amalgam (exposure is external, dose is internal). A consistent language for the field of exposure science is important for communicating within the field and among disciplines and for developing exposure-science metrics for source monitoring and exposure prevention and reduction. The evolution of the field over the past 15 years has included a greater emphasis on the use of internal markers of exposure to assist in defining exposure-response relationships. As such, the conceptual basis of the field includes both external and internal exposures, using external measurement and modeling methods and internal markers as tools for characterizing past or current exposures. Appendix C provides more detailed discussion on the application of this terminology.
To reflect the definition of exposure science and to embrace a broader view of the role that exposure science plays in human-health and ecosystem-health protection, the committee developed the conceptual framework in Figure 1-2.
The conceptual framework identifies and links the core elements of exposure science: sources of stressors, environmental intensity (such as pollutant concentrations3), time—activity and behavior, contact of stressors and receptors, and outcomes of contact. Figure 1-2 shows the role of upstream human and natural factors in determining which stressors are mobilized and transported to key receptors. (Examples of factors include choosing to use natural gas vs diesel buses, or choosing to pay more for gasoline to drive a car vs taking the bus, where the choice influences the source and can also influence behavior.) It indicates the role of behavior of receptors and time in modifying the contact that results from environmental intensities that influence exposure. It brings both external and internal environments within exposure but retains the idea that exposure is measured at some boundary between the source and receptor and that dose is the amount of material that passes or otherwise has influence across the boundary to come into contact with the target system, organ, or cell and produces an outcome. For example, a dose in one tissue, such as the blood, can serve as an exposure of another tissue that the blood perfuses. Figure 1-2 recognizes the feedbacks inherent in exposure science. Consider, for example, how behavior changes in a diseased person or organism and influences exposure. The outcome can also affect the source, as when a person who has an environmentally mediated infectious disease becomes a source of pathogens in water supplies (Eisenberg et al. 2005).
_______________________
3Intensity is the preferred term because some stressors, such as temperature excesses, cannot be easily measured as concentrations.
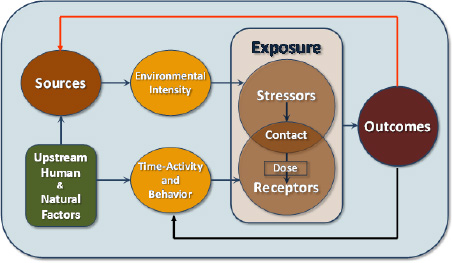
FIGURE 1-2 Core elements of exposure science.
Figure 1-3 frames an exposure narrative that plays out in space and time, and is intended to elucidate the stressor-receptor linkages at different levels of intergation. As a human (or fish, bird, or other organism) has changing contacts with different habitats, the intensity of a stressor changes, as do the number and duration of contacts. Here, exposure amounts to a multidimensional description of the location, time, and intensity of the target—stressor contacts. The exposure narrative covers relationships between receptors and locations and between locations and stressors; it provides a basis for drawing inferences about receptor—outcome relationships. That often requires recognition that any receptor can be associated with multiple environments (locations) and that locations can be associated with multiple stressors. Exposure science can be applied at any level of biologic organization—ecologic, community, or individual—and, within the individual, at the level of external exposure, internal exposure, or dose.
To appreciate the vision for exposure science in the 21st century (discussed in Chapter 2), it is important to understand its historical context. Exposure science arose from such disciplines as industrial hygiene, radiation protection, and environmental toxicology, in which the importance of assessing exposure has been demonstrated. In one of the earliest efforts to address exposure, the ancient Greek physician Hippocrates (about 400 BC) demonstrated in his treatise Air, Water, and Places that the appearance of disease in human populations is influenced by the quality of air, water, and food; the topography of the land; and general living habits (Wasserstein 1982). In the 1500s, the
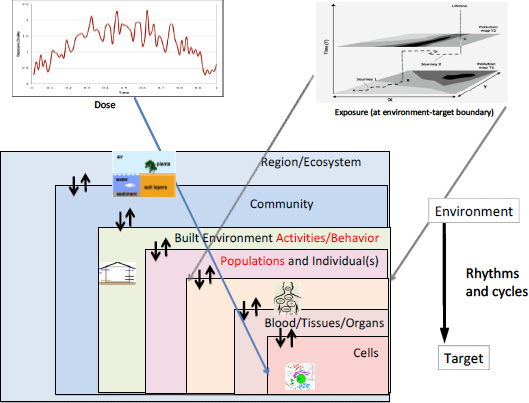
FIGURE 1-3 An illustration of how exposures can be measured or modeled at different levels of integration in space and time, from source to dose, and among different human, biologic, and geographic systems. That is exposure science can be applied at any level of biologic organization—ecologic, community, or individual—and, within the individual, at the level of external exposure, internal exposure, or dose. Source: Inset on exposures in space adapted from Gulliver and Briggs 2005.
physician and alchemist Paracelsus framed the widely cited toxicologic concept that “dose makes the poison” (Binswanger and Smith 2000). Ramazzini, in his 1703 treatise Diseases of the Workers, identified workplace exposures to single and multiple agents and the migration of contaminants into the community environment as causing disease (Ramazzini 1703). Percivall Pott first demonstrated the association between cancer and exposure to soot with his studies of scrotal cancer in chimney sweeps (Pott 1775). John Snow’s study of water-use patterns and their relation to disease in London allowed him to link a source of water contamination to cholera (Snow 1885). The avoidance of potentially harmful exposures through the separation of land use between human residences and industrial facilities was proposed in the latter part of the 19th century (Howard 1898).
Use of exposure assessment in radiation health protection can be traced back to roughly 1900 after the discovery of x rays. During the 1920s, Alice Hamilton established the formal study of industrial medicine in the United States. The metrics for and applications of exposure science to radiation protection have grown in sophistication and reliability over the last century (NRC 2006; ICRP 2007; EPA 2011c). Many of the basic principles for measuring, monitoring, and modeling exposures to airborne contaminants, including the earliest use of exposure biology, come from the field of industrial hygiene. The publication of Silent Spring (Carson 1962) and its focus on the transfer and magnification of persistent pollutants through food webs fostered the growth of environmental toxicology and chemistry, which address chemical fate and transport through multiple media and multiple pathways.
By the middle 1980s, exposure evaluations had evolved into an established scientific discipline that moved beyond single routes, single chemicals, and single pathways toward an understanding of “total” exposure. The 1991 National Research Council report Human Exposure Assessment for Airborne Pollutants (NRC 1991a) laid the foundation for further development of the field by defining the core principles of exposure assessment. Between 1980 and 1985, the Total Exposure Assessment Methodology (TEAM) study was conducted to assess personal exposures of 600 residents in seven US cities to chemical exposures by one or more routes of entry into the body and to estimate the exposures and body burdens of urban populations in several cities (EPA 1987). The TEAM studies established a framework for examining total human exposure covering multiple routes of entry into the body (Wallace 1987).
By promoting the concept that it is important to “measure where the people are” (Wallace 1977), the TEAM studies revealed new source categories and control options to reduce or prevent exposures. For example, application of the concept resulted in increasing attention to exposures indoors, where people spend a substantial portion of their lives (Smith 1988a). Globally, it pointed to the importance of indoor pollution in rural areas of developing countries, where a large portion of the world’s breathing is done but relatively little research or monitoring was being conducted (Smith 1988b).
Control measures revealed by a total-exposure framework include measures to increase the time that doors are closed between a house and its garage in the United States and thereby reduce human exposure to tailpipe emissions in the home, even though this has no effect on vehicle emissions or ambient concentrations in the garage. The exposure control included a simple spring on the door to allow it to stay open a shorter time.
Two major advances that helped to establish the credibility of exposure science as a discipline were the formation of the International Society of Exposure Analysis in 1989 (now the International Society of Exposure Science) and the publication of the Journal of Exposure Science and Environmental Epidemiology in 1990.
A number of important milestones followed. In 1992, EPA published its Guidelines for Exposure Assessment, which served as a companion to its toxicology and risk-assessment guidelines. That was followed in 1993 by the initiation of the National Human Exposure Assessment Survey (NHEXAS), which evaluated human exposure to multiple chemicals on a community and regional scale (EPA 2009b). NHEXAS monitored chemicals in blood and urine; incorporated environmental sampling of air, water, soil, and dust; and conducted personal monitoring of air, food, and beverages (NRC 1991b; EPA 2009b). It brought attention to the role of the proximity of emissions as opposed to the magnitude of emissions in determining overall exposure—low-level emissions near human receptors, such as those from indoor environments, need to receive at least as much attention as outdoor stack emissions (Sexton et al. 1995). In 1997, EPA’s Exposure Factors Handbook was published that presents data and evaluation of allometric and behavioral factors that affect exposures. It became an international resource for risk assessors who use these factors to estimate exposures for various pathways.4
Over the last 20 years, exposure science has evolved as a theoretical and practical science to include the development of mathematical models and other tools for examining how individuals and populations come into contact with environmental stressors of concern. For example, the discovery that airborne lead from gasoline combustion is deposited on soil, is tracked into homes, and enters children via hand-to-mouth activities greatly expanded the focus on multipathway exposure assessments and the development of exposure models that are validated through biomonitoring. Ott and others introduced time—activity models that were applied to air pollutants (Ott 1995). In the 1990s, exposure models addressed multimedia and multipathway exposures, tracking pollutants from multiple sources through air, water, soil, food, and indoor environments (McKone and Daniels 1991).
_______________________
4A 2011 version has been released (EPA 2011b).
Borrowing from the concept of “dose commitment”5 in radiation protection, researchers elaborated the concept of “exposure efficiency” in the 1980s and 1990s (for example, Smith 1993). Early in the 21st century, the term intake fraction was adopted to describe that concept (Bennett et al. 2002). It is defined as the amount of material crossing the body’s barriers per unit emitted and thus is dimensionless. For air pollution, population intake fraction is the amount inhaled by the population divided by the amount emitted per unit activity or time. It directly connects the source and environmental-intensity boxes in Figure 1-2 with the exposure box, effectively incorporating the pathways in between without needing to specify them. A striking characteristic of intake fraction is that it varies by orders of magnitude among standard source categories—for example, in the case of air pollution, from 10-6 for such remote sources as power plants to 10-4 for urban outdoor sources, roughly 5 x 10-3 for such indoor sources as unvented stoves, and 1.0 for active smoking. Not only does “dose make the poison”, therefore, but because proximity makes the dose, ultimately “place makes the poison” (NRC 2003). However, the biologically-relevant time and intensity of contact with an agent for each route of exposure needs to be considered (Lioy 1999).
OPPORTUNITIES AND CHALLENGES: THE NEW MILLENNIUM
Since 2000, a number of activities have benefited from advances in exposure science, and new challenges and opportunities have emerged. The Children’s Health Act of 2000 authorized the establishment of the National Children’s Study, a large-scale multiyear prospective study of children’s health and exposures intended to identify and characterize environmental influences (including physical, chemical, biologic, and psychosocial) on children from birth to adulthood. The study is under way, after the completion of the Vanguard Center pilot programs and the incorporation of new tools and approaches to streamline data collection at the household level and to capitalize on existing data for constructing community exposure baselines (IOM 2008; Trasande et al. 2011).
The increasing collection and evaluation of biomarkers of exposure and effect also is providing growing opportunities for exposure science. The Centers for Disease Control and Prevention’s National Health and Nutrition Examination Survey (NHANES) published the first National Human Exposure Report in 2001, which used a subset of its subjects to assess the US population’s exposure to environmental chemicals on the basis of biomonitoring data. The reports have been updated with publications released in 2003, 2005, and 2009, and annual reports are expected. The NHANES data provide a unique and growing potential
_______________________
5Dose commitment is the dose that will accumulate in an individual or population over a given period (for example, 50 years) from releases of radioactivity from a given source.
for evaluating source—exposure and exposure—disease relationships in a national population-based representative sample. California has started its own biomonitoring program (OEHHA 2007), and other states and cities are working on biomonitoring efforts (CDC 2010). The emerging biomonitoring data sets will allow improved tracking of exposures over time, space, and across populations for an increasingly larger number of chemicals. This information will be essential for evaluating the efficacy of exposure reduction policies, and for prioritizing and assessing chemical risks.
A prime example of the benefits of improved methods of exposure assessment is their use in environmental epidemiology, in which more accurate estimates of the health effects of important stressors have been achieved by reducing exposure misclassification, for example, in air pollution (Jerrett et al. 2005) and ionizing radiation (NRC 2006). There are many opportunities for continued improvements in this arena.
The Exposome
The exposome is defined as the record of all exposures both internal and external that people receive throughout their lifetime (Rappaport and Smith 2010).
Rapid advances in methods of sampling and analysis, genomics, systems biology, bioinformatics, and toxicology have laid the groundwork for major advances in the applications of exposure science. One such development is the concept of the “exposome”, which theoretically can capture the totality of environmental exposures (including lifestyle factors, such as diet, stress, drug use, and infection) from the prenatal period on, using a combination of biomarkers, genomic technologies, and informatics (Wild 2005; Rappaport and Smith 2010). Understanding how exposures from occupation, environment, diet, lifestyle, and the like interact with unique individual characteristics—such as genetics, physiology, and epigenetic makeup resulting in disease—is the fundamental challenge implicit in the exposome. The exposome in concert with the human genome holds promise for elucidating the etiology of chronic diseases (Rappaport and Smith 2010; Wild 2012).
The concept of the exposome offers an intriguing and promising direction for exposure science that will continue to spur developments in the field, especially in biomarkers, data-sharing, and informatic approaches to large datasets. By encompassing many biomarkers and stressors at once, exposome analysis can be the source of important new hypotheses of relationships between internal markers of stress and the external environment. Within the conception of exposure science proposed here (see Figure 1-2), the committee, in Chapter 2, broadens the exposome concept to the “eco-exposome”, that is the extension of exposure science from the point of contact between stressor and receptor inward into the organism and outward to the general environment, including the ecosphere.
Links to Toxicology and Risk Assessment
In recent years, the National Research Council has released two groundbreaking reports—Toxicity Testing in the 21st Century (NRC 2007) and Science and Decisions: Advancing Risk Assessment (NRC 2009)—that substantially advance conceptual and experimental approaches in the companion fields of toxicology and risk assessment. Those reports emphasize the importance of improving the assessment of early biologic markers of effects, individual susceptibility, life-stage and population vulnerability, and cumulative exposures and risks. Toxicity Testing in the 21st Century laid the foundation for a paradigm shift toward the use of new scientific tools to expand in vitro pathway-based toxicity testing. A key component of that report is the generation and use of population-based and individual human exposure data for interpreting test results and using toxicity biomarker data with exposure data for biomonitoring, surveillance, and epidemiologic studies. The focus of the report on systems approaches to understanding human biology, coupled with information about systems-level perturbations resulting from human—environment interactions, is critical for understanding biologically relevant exposures (Cohen Hubal 2009; Farland 2010). By emphasizing early perturbations of biologic pathways that can lead to disease, the report moved the focus of risk assessment along the exposure—disease spectrum toward exposure, especially the role of prior and current exposures in altering vulnerability of individuals and communities to additional environmental exposures. The resulting toxicology focus has essentially been on early biomarkers of effects in the population. At the same time, such concepts as the exposome have moved the focus of exposure science along the exposure—disease spectrum toward the health-effects side, especially biologic perturbations that correlate with exposure and are predictive of disease. The “meeting in the middle” carries promise for closer connections in the fields of exposure science and toxicology and for better linkages between exposure and disease (Cohen Hubal et al. 2010).
Science and Decisions: Advancing Risk Assessment, which examined ways to improve risk assessment, identified the need for better tools to address exposures in cumulative risk assessments. Its themes include the need for more and better exposure data for understanding dose—effect relationships, the need for investment in biomarkers of exposure, the importance of understanding both chemical and nonchemical stressors and their interactions, the need to use appropriate defaults to account for individual susceptibility and population vulnerability when stressor-specific data are not available, and better characterization of exposures in the context of cumulative risk assessment. The focus of Science and Decisions: Advancing Risk Assessment on capturing vulnerability better, on improving dose—response models, and on the observation that vulnerability arises from both prior and concurrent exposures creates important opportunities for exposure science.
Use of Exposure Science
The potential benefits of exposure science have not yet been fully realized. Among the important lags has been the slow incorporation of exposure science into policy and regulation. For example, EPA has focused on control of radon in drinking water whereas population radon exposure is actually dominated by other unregulated sources (NRC 1994; EPA 2008). Another example is the poor monitoring and control of indoor sources (for example, volatile organic compounds) even though air-pollution exposures clearly are dominated by them, as first definitively shown by the TEAM studies in the 1980s (Wallace 1991; Myers and Maynard 2005). Finally, even though occupational settings still dominate exposures to many important stressors in some populations, no effort to integrate them into population exposure-reduction strategies is under way. Political and economic barriers may help to explain those lapses, but they constitute lost opportunities to protect more people at lower cost by using exposure science (Smith 1995; Ott et al. 2007).
Integration of Human and Ecologic Exposure Science
There has been a gap been between the application of exposure science to human health and its application to ecosystem health, which is due in part to the lack of recognition of the connection between human and ecosystem health—in reality, they are inextricably linked. The connection between human health and ecosystem health is explored in the context of ecosystem services; as seen in Figure 1-4, human welfare depends on ecosystem health.
A better integration of ecologic and human exposure science is critical because ecologic conditions strongly mediate exposures and their consequences for humans and ecosystems. Not only do ecosystems contain multiple stressors that can act synergistically but organisms’ environments are seldom optimal and may heighten their sensitivity to stressors. As illustrated by the examples in Box 1-2, degradation of ecosystems due to human activities increases exposure to or consequences of chemical and biologic stressors in both humans and ecosystems. Elucidating relationships between exposure and key abiotic and biotic ecologic factors is necessary if we are to understand risk.
The present report builds on the concepts presented in the National Research Council reports Toxicity Testing in the 21st Century and Science and Decisions: Advancing Risk Assessment to develop a framework for bringing exposure science to a point where it fully complements toxicology and risk assessment and can be used to protect human health and the environment better. The committee also addresses a set of emerging needs, such as the need to
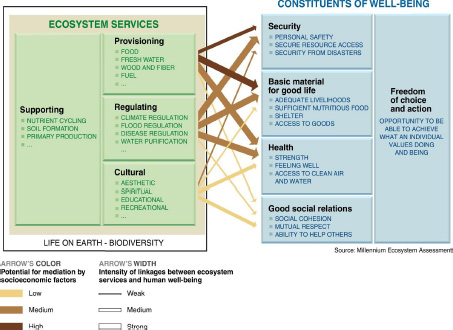
FIGURE 1-4 Connections between ecosystem services and human well-being. The framework of ecosystem services makes explicit the linkages between human and ecologic health. The strength of the linkages and the potential for mediation differ in different ecosystems and regions. Adverse exposures can indirectly affect human health and well-being by influencing a range of services provided by ecosystems. Source: Millenium Ecosystem Assessment 2005. Reprinted with permission; copyright 2005, World Resources Institute.
provide rapid assessment protocols and technologies to respond to natural and human-caused disasters and the needs for community participation and environmental justice. The report describes new technologies and opportunities to make exposure science even more effective in its traditional roles of evaluating environmental control measures, improving understanding of the link between environmental stressors and disease, and designing more cost-effective ways to reduce and prevent health risks. Finally, where possible, the committee offers ideas for integrating the applications of exposure science to human health and ecosystem health.
In Chapter 2, the committee presents a vision for exposure science. Chapter 3 describes the opportunities and challenges for applying exposure science to toxicology, epidemiology, and risk assessment and how exposure science can play a more effective role in other fields, such as environmental regulation, urban planning, ecosystem management, and disaster management. Chapter 4 addresses emerging demands for exposure-science information. Chapter 5 identifies scientific and technologic advances that are shaping the field and that support the committee’s vision. Chapter 6 discusses promoting and sustaining
public trust in exposure science including the management of personal and environmental exposure data. Chapter 7 describes the path forward for exposure science in the 21st century.6
Rising temperatures. Whether caused by shifts in climate or land uses (for example, deforestation, reduced vegetative cover, and urban heat islands), changes in temperature can directly prompt health-threatening exposures (for example, extreme heat events) or indirectly influence exposure to other substances. In aquatic ecosystems, degraded riparian zones, loss of forest cover, runoff from impervious surfaces, and discharges from industry can lead to rising water temperatures and increased toxicity. Above-normal temperatures compromise function and integrity of aquatic ecosystems. In addition, high temperatures can increase sensitivity of aquatic animals to heavy metals, including cadmium (Lannig et al. 2006; Cherkasov et al. 2006, 2007), mercury (Slotsbo et al. 2009), copper (Gupta et al.1981; Boeckman and Bidwell 2006; Khan et al. 2006), and lead (Khan et al. 2006). High temperatures also may amplify effects of pesticides—such as diazinon (Osterauer and Köhler 2008), terbufos, and trichlorfon (Brecken-Folse et al. 1994; Howe et al. 1994)—on fish.
Anthropogenic nutrient enrichment. Agricultural runoff and untreated sewage effluent are two important causes of eutrophication, in which aquatic ecosystems accumulate high concentrations of nutrients (for example, phosphates and nitrates) that promote plant growth. Algal growth can become excessive and sometimes lead to harmful algal blooms (Paerl 1997; Cloern 2001; Anderson et al. 2002; Kemp et al. 2005) and anoxic (low-oxygen) conditions that directly kill organisms and that can increase sensitivity to chemical stressors. For example, low dissolved oxygen prompted higher mortality in daphnids exposed to carbendazim (Ferreira et al. 2008), in crabs exposed to copper (Depledge 1987), and in fish exposed to alkylphenols (Gupta et al. 1983).
Reduced access to water. Human-associated changes in hydrologic regimes—including construction of dams and levees, depletion of groundwater supplies, drainage of wetlands, and removal of vegetation—profoundly affect water availability for humans and ecologic communities alike. Aside from the direct effects on ecosystem goods and services related to water, these an-
_______________________
6Given its terms of reference, the committee addresses primarily exposure-science issues related to the United States and other developed countries. In addition, the committee does not focus on uses of exposure principles in regulation and policy.
thropogenic stressors can promote dehydration, which can increase concentration of toxicants and thereby increase risk of damage. Chemicals also can reduce drought tolerance of organisms by interfering with physiologic adaptations, as has been demonstrated in earthworms exposed to copper (Holmstrup 1997) and in springtails exposed to polyclic aromatic hydrocarbons (Sjursen et al. 2001), lidane (Demon and Eijsackers 1985), and surfactants (Holmstrup 1997; Skovlund et al. 2006). Diminishing access to safe water can increase risk of some diseases as wildlife, livestock, and humans are brought into closer contact.
Invasive species. Biotic invasion is one of the top drivers of biodiversity loss and species endangerment. Invasive species can alter species interactions and disrupt ecologic processes in ways that elicit serious ecologic, economic, and health consequences. Even seemingly benign species can provoke unexpected exposures. For example, a recent experiment suggested that Amur honeysuckle (Lonicera maackii), a widespread invasive shrub in North America, increases human risk of exposure to ehrlichiosis, an emerging infectious disease transmitted by ticks (Allan et al. 2010). The high risk would result from a preference of a key tick and pathogen reservoir, white-tailed deer (Odocoileus virginianus), for areas of dense honeysuckle. In aquatic systems, the invasive round goby (Neogobius melanostomus) is thought to facilitate mobilization of contaminants in food webs and to increase exposure to humans because its persistence in contaminated environments draws predatory fish, which also are popular game species, into polluted habitats (Marentette et al. 2010).
Shifts in species composition. Because species differ in bioaccumulation kinetics, changes in the structure of animal communities can influence bioaccumulation and human exposure. Indeed, mercury accumulation rates differ among bivalve species according to feeding strategies and assimilation efficiencies (Cardoso et al. 2009). At the terrestrial—aquatic interface, spiders had more of the highly bioavailable methylmercury than other invertebrates (such as lepidopterans and orthopterans) and, therefore were thought to be responsible for transporting aquatic mercury into terrestrial food webs (Cristol et al. 2008). The presence of particular species can provide buffers to exposure in some cases; for example, some algal blooms are known to reduce uptake of methylmercury into freshwater food webs (Pickhardt et al. 2002).
Habitat degradation. Habitat degradation can facilitate transport of contaminants into aquatic systems, transmission of diseases by promoting high densities of vectors, and increases in the sensitivity of animals to exposures. Changes in food availability in degraded habitats also can affect nutritional status in ways that can mediate toxicity (Holmstrup et al. 2010). Consequences of habitat degradation can be surprising. For example, erosion of
European saltmarshes dominated by cord grasses has resulted in massive cadmium release from sediments in areas of cord-grass dieback (Hubner et al. 2010); this shows how habitat degradation or sea-level rise, for example, can increase exposure to heavy metals.
Emerging contaminants. A number of new compounds with novel chemistries are rapidly expanding in commerce and are now appearing, or are expected to occur, as widespread environmental contaminants. Important examples include brominated and chlorinated organic compounds used as flame retardants and a large and growing array of diverse organic and metal-based nanomaterials (Lorber 2008; Stapleton et al. 2006, 2009; Wiesner et al. 2009). Little is known about human health or ecologic effects of most of these materials. Similarly, little is known about fates and exposures; it is difficult to measure most of them at environmentally relevant concentrations in media and in organisms. The situation is particularly problematic with nanomaterials. It is likely that as their abundance increases in the environment, they will contribute to the sum of multiple stressors to which humans and ecosystems are exposed. And their fates and effects will be influenced by the environmental variables described above. It is timely for exposure science to be rapidly developing concomitantly with these new chemistries.
Greenhouse-gas emissions. Increased concentrations of atmospheric carbon dioxide stimulate growth of poison ivy (Toxicodendron radicans) and causes plants to produce a more allergenic form of urushiol (Mohan et al., 2006). Anthropogenic changes in the atmosphere thus are expected to lead to more abundant and more toxic poison ivy.
Allan, B.F., H.P. Dutra, L.S. Goessling, K. Barnett, J.M. Chase, R.J. Marquis, G. Pang, G.A. Storch, R.E. Thach, and J.L. Orrock. 2010. Invasive honeysuckle eradication reduces tick-borne disease risk by altering host dynamics. Proc. Natl. Acad. Sci. U. S. A. 107(43):18523-18527.
Anderson, D.M., P.M. Glibert, and J.M. Burkholder. 2002. Harmful algal blooms and eutrophication: Nutrient sources, composition, and consequences. Estuaries 25(4B):704-726.
Bennett, D.H., T.E. McKone, J.S. Evans, W.W. Nazaroff, M.D. Margni, O. Jolliet, and K.R. Smith. 2002. Defining intake fraction. Environ. Sci. Technol. 36(9):206A-211A.
Binswanger, H.C., and K.R. Smith. 2000. Paracelsus and Goethe: Founding fathers of environmental health. Bull. World Health Organ. 78(9):1162-1164.
Boeckman, C.J., and J.R. Bidwell. 2006. The effects of temperature, suspended solids, and organic carbon on copper toxicity to two aquatic invertebrates. Water Air Soil Pollut. 171(1-4):185-202.
Brecken-Folse, J.A., F.L. Mayer, L.E. Pedigo, and L.L. Marking. 1994. Acute toxicity of 4-nitrophenol, 2,4-dinitrophenol, terbufos and trichlorofon to grass shrimp (Palaemonetes Spp.) and sheepshead minnows (Cyprinodon variegatus) as affected by salinity and temperature. Environ. Toxicol. Chem. 13(1):67-77.
Cardoso, P.G., A.I. Lillebo, E. Pereira, A.C. Duarte, and M.A. Pardal. 2009. Different mercurybioaccumulation kinetics by two macrobenthic species: The bivalve Scrobicularia plana and the polychaete Hediste diversicolor. Mar. Environ. Res. 68(1):12-18.
Carson, R. 1962. Silent Spring. Cambridge, MA: Riverside Press.
CDC (Centers for Disease Control and Prevention). 2010. National Biomonitoring Program, State Grant Activities [online]. Available: http://www.cdc.gov/biomonitoring/state_grants.html [accessed Dec. 6, 2011].
Cherkasov, A.S., P.K. Biswas, D.M. Ridings, A.H. Ringwood, and I.M. Sokolova. 2006. Effects of acclimation temperature and cadmium exposure on cellular energy budgets in the marine mollusk Crassostrea virginica: Linking cellular and mitochondrial responses. J. Exp. Biol. 209(7):1274-1284.
Cherkasov, A.S., S. Grewal,and I.M. Sokolova. 2007. Combined effects of temperature and cadmium exposure on haemocyte apoptosis and cadmium accumulation in the eastern oyster Crassostrea virginica (Gmelin). J. Therm. Biol. 32(3):162-170.
Cloern, J.E. 2001. Our evolving conceptual model of the coastal eutrophication problem. Mar. Ecol. Progr. Ser. 210:223-253.
Cohen Hubal, E.A. 2009. Biologically relevant exposure science for 21st century toxicity testing. Toxicol. Sci.111(2):226-232.
Cohen Hubal, E.A., A. Richard, L. Aylward, S. Edwards, J. Gallagher, M.R. Goldsmith, S. Isukapalli, R. Tornero-Velez, E. Weber, and R. Kavlock. 2010. Advancing exposure characterization for chemical evaluation and risk assessment. J. Toxicol. Environ. Health B Crit. Rev. 13(2-4):299-313.
Cristol, D.A., R.L. Brasso, A.M. Condon, R.E. Fovargue, S.L. Friedman, K.K. Hallinger, A.P. Monroe, and A.E. White. 2008. The movement of aquatic mercury through terrestrial food webs. Science 320(5874):335-335.
Demon, A., and H.Eijsackers. 1985. The effects of lindane and azinphosmethyl on survival-time of soil animals, under extreme or fluctuating temperature and moisture conditions. Z. Angew. Entomol. 100(1-5):504-510.
Depledge, M.H. 1987. Enhanced copper toxicity resulting from environmental-stress factor synergies. Comp. Biochem. Physiol. C-Pharmacol. 87(1):15-19.
Eisenberg, J.N., X. Lei, A.H. Hubbard, M.A. Brookhart, and J.M. Colford, Jr. 2005. The role of disease transmission and conferred immunity in outbreaks: Analysis of the 1993 Cryptosporidium outbreak in Milwaukee, Wisconsin. Am. J. Epidemiol. 161(1):62-72.
EPA (U.S. Environmental Protection Agency). 1987. Project Summary: The Total Exposure Assessment Methodology (TEAM) Study. EPA 600/S6-87/002. Office of Acid Deposition, Environmental Monitoring and Quality Assurance, Office of Research and Development, U.S. Environmental Protection Agency Washington, DC. September 1987.
EPA (U.S. Environmental Protection Agency). 2003. Framework for Cumulative Risk Assessment. EPA/630/P-02/001. Risk Assessment Forum, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/raf/publiccations/pdfs/frmwrk_cum_risk_assmnt.pdf [accessed Dec. 29, 2011].
EPA (U.S. Environmental Protection Agency). 2008. Care for Your Air: A Guide to Indoor Air Quality. EPA 402/F-08/008. U.S. Environmental Protection Agency, Sep-
tember 2008 [online]. Available: http://www.epa.gov/iaq/pdfs/careforyourair.pdf [accessed April 9, 2012].
EPA (U.S. Environmental Protection Agency). 2009a. A Conceptual Framework for U.S. EPA’s National Exposure Research Laboratory. EPA/600/R-09/003. National Exposure Research Laboratory, Office of Research and Development, U.S. Environmental Protection Agency [online]. Available: http://www.epa.gov/nerl/documents/nerl_exposure_framework.pdf [accessed Dec. 29, 2011].
EPA (U.S. Environmental Protection Agency). 2009b. Human Exposure Measurements: National Human Exposure Assessment Survey (NHEXAS). U.S. Environmental Protection Agency [online]. Available: http://www.epa.gov/heasd/edrb/nhexas.html [accessed Dec. 29, 2011].
EPA (U.S. Environmental Protection Agency). 2011a. EPA Risk Assessment Basic Information: What is Risk? What is a Stressor? U.S. Environmental Protection Agency [online]. Available: http://www.epa.gov/risk_assessment/basicinformation.htm#risk [accessed Dec. 29, 2011].
EPA (U.S. Environmental Protection Agency). 2011b. Exposure Factors Handbook: 2011 Edition. EPA/600/R-090/052F. National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/ncea/efh/pdfs/efh-complete.pdf [accessed Oct. 27, 2011].
EPA (U.S. Environmental Protection Agency). 2011c. EPA Radiogenic Cancer Risk Models and Projections for the U.S. Population. EPA 402-R-11-001. Office of Radiation and Indoor Air, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://www.epa.gov/rpdweb00/docs/bluebook/402-r-11-01.pdf [accessed Nov. 2, 2011].
EPA SAB (U.S. Environmental Protection Agency, Science Advisory Board). 1992. Respiratory Health Effects of Passive Smoking: Lung Cancer and Other Disorders, SAB Review Draft. EPA /600/6-90/0068. Science Advisory Board, U.S. Environmental Protection Agency, Washington, DC. May 1992.
Farland, W.H. 2010. The promise of exposure science and assessment. J. Expo. Sci. Environ. Epidemiol. 20(3):225.
Ferreira, A.L.G., S. Loureiro, and A.M. Soares. 2008. Toxicity prediction of binary combinations of cadmium, carbendazim and low dissolved oxygen on Daphnia magna. Aquat. Toxicol. 89(1):28-39.
GAO (General Accounting Office). 2005. Chemical regulation: Options Exist to Improve EPA’s Ability to Assess Health Risks and Manage its Chemical Review Program. GAO-05-458. Washington, DC: U.S. General Accounting Office [online]. Available: http://www.gao.gov/new.items/d05458.pdf [accessed Dec. 29, 2011].
Graham, J.A., ed. 2011. Exposure Science Digests: Demonstrating How Exposure Science Protects Us From Chemical, Physical, and Biological Agents. Journal of Exposure Science and Environmental Epidemiology [online]. Available: http://www.nature.com/jes/pdf/JESSE_ESD_booklet.pdf [accessed Dec. 29, 2011].
Gulliver, J., and D.J. Briggs. 2005. Time-space modelling of journey-time exposure to traffic-related air pollution using GIS. Environ. Res. 97(1):10-25.
Gupta, P.K., B.S. Khangarot, and V.S. Durve. 1981. The temperature-dependence of the acute toxicity of copper to a fresh-water pond snail, Viviparus bengalensis L. Hydrobiologia 83(3):461-464.
Gupta, S., R.C. Dalela, and P.K. Saxena. 1983. Influence of dissolved-oxygen levels on acute toxicity of phenolic-compounds to fresh-water teleost, Notopterusnotopterus (Pallas). Water Air Soil Pollut. 19(3):223-228.
Holmstrup, M. 1997. Drought tolerance in Folsomia candida Willem (Collemhola) after exposure to sublethal concentrations of three soil-polluting chemicals. Pedobiologia 41(4):361-368.
Holmstrup, M., A. Bindesbol, G.J. Oostingh, A. Duschl, V. Scheil, H. Köhler, S. Loureiro, A.M. Soares, A.L. Ferreira, C. Kienle, A. Gerhardt, R. Laskowski, P.E. Kramarz, M. Bayley, C. Svendsen, and D.J. Spurgeon. 2010. Interactions between effects of environmental chemicals and natural stressors: A review. Sci. Total Environ. 408(18):3746-3762.
Howard, E. 1898. Tomorrow; A Peaceful Path to Real Reform. London: S. Sonnenschein.
Howe, G.E., L.L. Marking, T.D. Bills, J.J. Rach, and F.L. Mayer, Jr. 1994. Effects of water temperature and pH on toxicity of terbufos, trichlorfon, 4-nitrophenol and 2,4-dinitrophenol to the amphipod Gammarus pseudolimnaeus and rainbow trout (Oncorhynchus mykiss). Environ. Toxicol. Chem. 13(1):51-66.
ICRP (International Commission on Radiological Protection). 2007. The 2007 Recommendations of the International Commission on Radiological Protection. Annals of the ICRP 103 [online]. Available: http://www.icrp.org/docs/ICRP_Publication_103-Annals_of_the_ICRP_37%282-4%29-Free_extract.pdf [accessed Nov. 2, 2011].
Hűbner, R., R.J.H. Herbert, and K.B. Astin. 2010. Cadmium release caused by the dieback of the saltmarsh cord grass Spartina anglica in Poole Harbour (UK). Estuar. Coast. Shelf S. 87(4):553-560.
IOM (Institute of Medicine). 2000. Protecting Those Who Serve: Strategies to Protect the Health of Deployed U.S. Forces. Washington, DC: National Academy Press.
IOM (Institute of Medicine). 2008. The National Children’s Study Research Plan: A Review. Washington, DC: National Academies Press.
Jerrett, M., R.T. Burnett, R. Ma, C.A. Pope, D. Krewski, K.B. Newbold, G. Thurston, Y. Shi, N. Finkelstein, E.E. Calle, and M.J. Thun. 2005. Spatial analysis of air pollution and mortality in Los Angeles. Epidemiology 16(6):727-736.
Jones, R.L., D.M. Homa, P.A. Meyer, D.J. Brody, K.L. Caldwell, J.L. Pirkle, and M.J. Brown. 2009. Trends in blood lead levels and blood lead testing among US children aged 1 to 5 years, 1988-2004. Pediatrics 123(3):e376-e385.
Kemp, W.M., W.R. Boynton, J.E. Adolf, D.F. Boesch, W.C. Boicourt, G. Brush, J.C. Cornwell,, T.R. Fisher, P.M. Glibert, J.D. Hagy, L.W. Harding, E.D. Houde, D.G. Kimmel, W.D. Miller, R.I.E. Newell, M.R. Roman, E.M. Smith, and J.C. Stevenson. 2005. Eutrophication of Chesapeake Bay: Historical trends and ecological interactions. Mar. Ecol. Progr. Ser. 303:1-29.
Khan, M.A., S.A. Ahmed, B. Catalin, A. Khodadoust, O. Ajayi, and M. Vaughn. 2006. Effect of temperature on heavy metal toxicity to juvenile crayfish, Orconectes immunis (Hagen). Environ. Toxicol. 21(5):513-520.
Lannig, G., A.S. Cherkasov, and I.M. Sokolova. 2006. Temperature-dependent effects of cadmium on mitochondrial and whole-organism bioenergetics of oysters (Crassostrea virginica). Mar. Environ. Res. 62(suppl.):S79-S82.
Lioy, P.J. 1990. Assessing total human exposure to contaminants: A multidisciplinary approach. Environ. Sci. Technol. 24(7):938-945.
Lioy, P.J. 1999. The 1998 ISEA Wesolowski Award lecture. Exposure analysis: reflections on its growth and aspirations for its future. J. Expo. Anal. Environ. Epidemiol. 9(4):273-281.
Lorber, M. 2008. Exposure of Americans to polybrominated diphenyl ethers. J. Exp. Sci. Environ. Epidemiol. 18(1):2-19.
Marentette, J.R., K.L. Gooderham, M.E. McMaster, T. Ng, J.L. Parrott, J.Y. Wilson, C.M. Wood, and S. Balshine. 2010. Signatures of contamination in invasive round
gobies (Neogobius melanostomus): A double strike for ecosystem health? Ecotoxicol. Environ. Saf. 73(7):1755-1764.
McKone, T.E., and J.I. Daniels. 1991. Estimating human exposure through multiple pathways from air, water, and soil. Regul. Toxicol. Pharmacol. 13(1):36-61.
Millennium Ecosystem Assessment. 2005. Ecosystems and Human Well-being: Synthesis. Island Press, Washington, DC [online]. Available: http://www.maweb.org/documents/document.356.aspx.pdf [accessed Feb. 8, 2011].
Mohan, J.E., L.H. Ziska, W.H. Schlesinger, R.B. Thomas, R.C. Sicher, K. George, and J.S. Clark. 2006. Biomass and toxicity responses of poison ivy (Toxicodendron radicans) to elevated atmospheric CO2. Proc. Natl. Acad. Sci. U. S. A. 103(24): 9086-9089.
Muntner, P., A. Menke, K.B. DeSalvo, F.A. Rabito, and V. Batuman. 2005. Continued decline in blood lead levels among adults in the United States: The National Health and Nutrition Examination Surveys. Arch. Intern. Med. 165(18):2155-2161.
Myers, I., and R.L. Maynard. 2005. Polluted air—outdoors and indoors. Occup. Med. 55(6):432-438.
Myers, M.S., L.L. Johnson, and T.K. Collier. 2003. Establishing the causal relationship between polycyclic aromatic hydrocarbon (PAH) exposure and hepatic neoplasms and neoplasia-related liver lesions in English sole (Pleuronectes vetulus). Hum. Ecol. Risk Assess. 9(1):67-94.
NRC (National Research Council). 1991a. Human Exposure Assessment for Airborne Pollutants: Advances and Opportunities. Washington, DC: National Academy Press.
NRC (National Research Council). 1991b. Monitoring Human Tissues for Toxic Substances. Washington, DC: National Academy Press.
NRC (National Research Council). 1994. Health Effects of Radon: Time for Reassessment? Washington, DC: The National Academy Press.
NRC (National Research Council). 1998. Research Priorities for Airborne Particulate Matter: I. Immediate Priorities and a Long-Range Research Portfolio. Washington, DC: The National Academy Press.
NRC (National Research Council). 1999. Our Common Journey: A Transition Toward Sustainability. Washington, DC: National Academy Press.
NRC (National Research Council). 2003. Managing Carbon Monoxide Pollution in Meteorological and Topographical Problem Areas. Washington, DC: National Academies Press.
NRC (National Research Council). 2006. Health Risks from Exposure to Low Levels of Ionizing Radiation: BEIR VII Phase 2. Washington, DC: National Academies Press.
NRC (National Research Council). 2007. Toxicity Testing in the 21st Century: A Vision and A Strategy. Washington, DC: The National Academies Press.
NRC (National Research Council). 2009. Science and Decisions: Advancing Risk Assessment. Washington, DC: National Academies Press.
OEHHA (Office of Environmental Health Hazard Assessment). 2007. California Environmental Contaminant Biomonitoring Program. Office of Environmental Health Hazard Assessment, California Environmental Protection Agency [online]. Available: http://oehha.ca.gov/multimedia/biomon/about.html [accessed Dec. 6, 2011].
Osterauer, R., and H. Köhler. 2008. Temperature-dependent effects of the pesticides thiacloprid and diazinon on the embryonic development of zebrafish (Danio rerio). Aquat. Toxicol. 86(4):485-494.
Ott, W.R. 1995. Human exposure assessment: The birth of a new science. J. Expo. Sci. Environ. Epidemiol. 5(4):449-472.
Ott, W., A.C. Steinemann, and L.A. Wallace, eds. 2007. Exposure Analysis. Boca Raton: CRC Press.
Paerl, H.W. 1997. Coastal eutrophication and harmful algal blooms: Importance of atmospheric deposition and groundwater as “new” nitrogen and other nutrient sources. Limnol. Oceanogr. 42(5):1154-1165.
Pastor, M., R. Morello-Frosch, and J.L. Sadd. 2005. The air is always cleaner on the other side: Race, space, and ambient air toxics exposures in California. J. Urban Aff. 27(2):127-148.
Pickhardt, P.C., C.L. Folt, C.Y. Chen, B. Klaue, and J.D. Blum. 2002. Algal blooms reduce the uptake of toxic methylmercury in freshwater food webs. Proc. Natl. Acad. Sci. U. S. A. 99(7):4419-4423.
Pott, P. 1775. Cancer Scroti. Pp. 63-68 in Chirurgical Observations Relative to the Cataract, the Polypus of the Nose, the Cancer of the Scrotum, the Different Kinds of Ruptures and the Mortification of the Toes and Feet. London: T.J. Carnegy.
Prüss-Üstün, A., and C. Corvalán. 2006. Preventing Disease Through Healthy Environments: Towards an Estimate of the Environmental Burden of Disease. Geneva: World Health Organization [online]. Available: http://www.who.int/quantifying_ehimpacts/publications/preventingdisease.pdf [accessed Dec. 29, 2011].
Rappaport, S.M., and M.T. Smith. 2010. Environment and disease risks. Science 30(6003):460-461.
Ramazzini, B. 1703. De morbis artificum diatribe. Ultrajecti: Apud Guilielmum van de Water.
Sexton, K., M.A. Callahan, and E.F. Bryan. 1995. Estimating exposure and dose to characterize health risks: The role of human tissue monitoring in exposure assessment. Environ. Health Perspect. 103(suppl. 3):13-29.
Sjursen, H., L.E. Sverdrup, and P.H. Krogh. 2001. Effects of polycyclic aromatic compounds on the drought tolerance of Folsomia fimetaria (collembola, isotomidae). Environ. Toxicol. Chem. 20(12):2899-2902.
Skovlund, G., C. Damgaard, M. Bayley, and M. Holmstrup. 2006. Does lipophilicity of toxic compounds determine effects on drought tolerance of the soil collembolan Folsomia candida? Environ. Pollut. 144(3):808-815.
Slotsbo, S., L.H. Heckmann, C. Damgaard, D. Roelofs, T. de Boer, and M. Holmstrup. 2009. Exposure to mercury reduces heat tolerance and heat hardening ability of the springtail Folsomia candida. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 150(1):118-123.
Smith, K.R. 1988a. Total exposure assessment: Part 1. Implications for the U.S. Environment 30(8):10-15; 33-38.
Smith, K.R. 1988b. Total exposure assessment: Part 2. Implications for developing countries. Environment 30(10):16-20; 28-35.
Smith, K.R. 1993. Fuel combustion, air pollution exposure, and health: The situation in developing countries. Annu. Rev. Energ. Environ. 18:529-566.
Smith, K.R. 1995. The Potential of Human Exposure Assessment for Air Pollution Regulation. Human Exposure Assessment Series WHO/EHG/95.09. Geneva: World Health Organization.
Smith, K.R., C.F. Corvalán, and T. Kjellström. 1999. How much global ill health is attributable to environmental factors? Epidemiology 10(5):573-584.
Snow, J. 1855. On the Mode of Communication of Cholera, 2nd Ed. London: Churchill.
Stapleton, H.M, N.G. Dodder, J.R. Kucklick, C.M. Reddy, M.M. Schantz, P.R. Becker, F. Gulland, B.J. Porter, and S.A. Wise. 2006. Determination of HBCD, PBDEs and MeO-BDEs in California sea lions (Zalophus californianus) stranded between 1993 and 2003. Mar. Pollut. Bull. 52(5):522-531.
Stapleton, H.M., S. Klosterhaus, S. Eagle, J. Fuh, J.D. Meeker, A. Blum, and T.F. Webster. 2009. Detection of organophosphate flame retardants in furniture foam and U.S. house dust. Environ. Sci. Technol. 43(19):7490-7495.
Trasande, L., H.F. Andrews, C. Goranson, W. Li, E.C. Barrow, S.B. VenderBeek, B. McCrary, S.B. Allen, K.D. Gallagher, A. Rundle, J. Quinn, and B. Brenner. 2011. Early experiences and predictors of recruitment success for the National Children’s Study. Pediatrics 127(2):261-268.
Wallace, L.A. 1977. Personal air quality monitors. Pp. 61-91 in Analytical Studies for the U.S. Environmental Protection Agency, Vol. IVa. Environmental Monitoring Supplement. Washington, DC: National Academy of Sciences.
Wallace, L.A. 1987. The Total Exposure Assessment Methodology (TEAM) Study: Summary and Analysis, Vol. 1. EPA/600/6-87/002a. Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://exposurescience.org/pub/reports/TEAM_Study_book_1987.pdf [accessed Nov. 4, 2011].
Wallace, L.A. 1991. Comparison of risks from outdoor and indoor exposure to toxic chemicals. Environ. Health Perspect. 95:7-13.
Wasserstein, A. 1982. Galen’s Commentary on the Hippocratic Treatise Airs, Waters, Places in the Hebrew Translation of Solomon ha-Me’ati. Proceedings of the Israel Academy of Sciences and Humanities 6(3). Jerusalem: Israel Academy of Sciences and Humanities.
WHO (World Health Organization). 2004. World Health Report-2004: Changing History. Geneva: World Health Organization [online]. Available: http://www.who.int/whr/2004/en/report04_en.pdf [accessed Dec. 29, 2011].
Wiesner, M.R., G.V. Lowry, K.L. Jones, M.F. Hochella, Jr., R.T. Di Giulio, E. Casman, and E.S. Bernhardt. 2009. Decreasing uncertainties in assessing environmental exposure, risk and ecological implications of nanomaterials. Environ. Sci. Technol. 43(17):5458-6462.
Wild, C.P. 2005. Complementing the genome with an “exposome”: The outstanding challenge of environmental exposure measurement in molecular epidemiology. Cancer Epidemiol. Biomarkers Prev. 14(8):1847-1850.
Wild, C.P. 2012. The exposome: From concept to utility. Int. J. Epidemiol. 41(1):24-32.























