3
Applications of Exposure Science
Exposure science plays a fundamental role in the development and application of epidemiology, toxicology, and risk assessment. It provides critical information for protecting human and ecosystem health. Exposure science also has the ability to play an effective role in other fields, including environmental regulation, urban and ecosystem planning, and disaster management; in many cases these are untapped opportunities. Exposure science links human and ecologic behavior to environmental processes in such a way that the information generated can be used to mitigate or prevent future adverse exposures. This chapter discusses current and future opportunities for and challenges to applying exposure science to those fields and frames some of the needs for further development of exposure science. Text boxes are intended to illustrate specific examples of the role of exposure science.
Exposure assessment is a major component of environmental epidemiology; it is equal in importance to outcome assessment. Historically, the main focus of many environmental epidemiology studies has been on single chemical, biologic, and physical stressors (for example, individual pesticides, air pollutants, ionizing radiation, or water contaminants). But human populations are exposed to a multitude of potentially hazardous stressors simultaneously, often with highly correlated patterns of spatial and temporal variation, and are affected by human activities and behaviors, so it is difficult to pinpoint the specific stressor associated with a particular outcome. New high-throughput genomic and biomonitoring technologies (discussed in Chapter 5) are providing for a greater number of potential biomarkers that can be used to assess multiple exposures simultaneously. In addition to chemical, biologic, or physical stressors, epidemiologists may be concerned with psychosocial stressors that influence
disease risks directly or modify a person’s susceptibility to the effects of other agents (Shankardass et al. 2009). The direct effects of psychosocial risk factors on health outcomes are beyond the scope of this report, but their role as modifiers of exposures is within the committee’s charge. Figure 3-1 provides a general schema for thinking about the components of exposure assessment in environmental epidemiology. Each of the components shown in solid boxes represent areas where exposure science can contribute, providing data on determinants of people’s exposures (the intensity of the spatio-temporal exposure fields they are moving through and their personal time/activity patterns in that field) and direct measurements of individuals’ external exposures or internal doses, along with relevant modifying factors. The hypothesized (but not directly observable) history of doses to the relevant target organs are then used by epidemiologists to relate to the health outcomes.
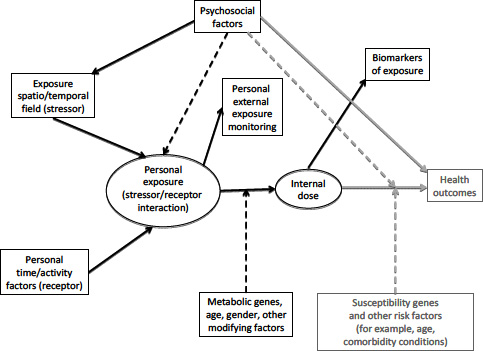
FIGURE 3-1 General schema of exposure assessment in environmental epidemiology. Items in gray, related to health outcomes and their determinants other than environmental exposures, are included to place exposure assessment in context but are outside the charge of this committee. Boxes represent measurable quantities, and ovals denote hypothetical intermediate variables that can be assessed only indirectly. Solid arrows denote direct effects, and dashed arrows indicate modifying effects. For example, the oval marked “personal exposure” represents the entire history of an individual’s true biologically-relevant exposure, which is not directly observable. The boxes labeled “personal external exposure monitoring” and “biomarkers of exposure” represent the data that are potentially observable.
Traditional exposure assessment for epidemiology combines the use of measurements and models to characterize the spatio-temporal field of environmental concentrations of a stressor with individual data on interactions of receptors (people) with their environment (for example, derived from questionnaires on time—activity patterns) to estimate personal exposures. In some studies, direct measurements of personal exposures (for example, film-badge measurements of occupational radiation exposures) and novel methods of tracking individual activities (for example, Global Positioning System (GPS) monitoring of locations and accelerometers for physical activity rates and videotaping of activities) have been used, either on an entire study population or on some sample for calibration or validation of model predictions. The emerging field of molecular epidemiology, based on the use of biomarkers of exposure (as well as of susceptibility or early signs of disease), offers potentially transformative advances in exposure science, particularly if combined with novel genomic, transcriptomic, metabolomic, and other “—omic” technologies and bioinformatic tools for organizing and integrating the massive, often disparate, data sets (see Chapter 5). Box 3-1 illustrates some of the complexities of exposure assessment for the National Children’s Study, with longitudinal measurements of a broad array of environmental and personal (external and internal) exposures and health outcomes.
Historically, comprehensive measurement of environmental exposures has not been possible, requiring statistical models to interpolate among relatively sparse measurements. The models can be purely statistical, such as geostatistical models for air pollution, or can be based on mathematical models for tracking agents from sources through intake by receptors (see NCRP 2010a for a discussion of the general principles of environmental dose reconstruction for radiation exposures and NCRP 2008, 2010b for recommended approaches to uncertainty analysis for external and internal exposures respectively). Box 3-2 provides an example of environmental pathway analysis applied to evaluate radionuclide exposures from the Hanford nuclear plant in Hanford, WA, and illustrates the value of involving the affected communities in all stages of the planning of an epidemiology project.
As novel sensing technologies, such as satellite imaging, become more widely available and more accurate, the need for models will remain, but the focus will shift from interpolation to exploitation of massive datasets. A key function of models is not just to provide point estimates of individual exposures but to quantify the uncertainty in exposure estimates, to understand measurement error in health analyses.
Environmental exposures typically occur over extended periods of time at varying intensities, requiring a shift in thinking from simple exposure-response to exposure-time-response relationships (Thomas 1988). These can be quite complex, involving modifying effects of age-at-exposure (for example, at particularly sensitive developmental stages), time-since-exposure, duration-of-exposure, or other time-related factors. In addition, for most conditions, little is known about whether short intense exposures have larger or smaller effects than
BOX 3-1 Case Study of Exposure Assessment for the National Children’s Study
The National Children’s Study (NCS) is a nationwide cohort study of pregnancy outcomes and child development through the age of 21 years. The study aims to investigate the separate and combined effects of environmental exposures on pregnancy outcomes, child health and development, and origins of adult disease. Environmental exposures in the NCS are broadly defined to include chemical, biologic, physical, psychosocial, and genomic factors. The study also aims to examine determinants of child, maternal, and developmental health disparities, such as prenatal exposures, geography, social status, race, ethnicity, neighborhood characteristics, and quality of social networks, and the impact of various disparities on health outcomes. The current study has about 100 study locations, but the number, size, and selection process for these study locations may evolve (Hirschfeld et al. 2011).
A complex exposure assessment is planned, entailing a combination of techniques—questionnaires on diet and product use; environmental sampling of house dusts; and collection, storage, and assays of biologic specimens (for example, blood, urine, saliva, hair, breast milk, and adipose tissue) (Barr et al. 2005; Needham et al. 2005). Depending on the particular agent, exposure assessments will be conducted at various life stages, for example, in utero, postnatal, and peripubertal (Bradman and Whyatt 2005; Wang et al. 2005; Landrigan et al. 2006). In addition, data from national and state agencies are being used to profile areas within a county and locations of the study participants (Lioy et al. 2009; Downs et al. 2010); a test case is being conducted in Queens County, NY (Lioy et al. 2009). Sophisticated modeling will be needed to combine the various data sources.
Given the size, scope, and complexity of the NCS, there have been challenges to identify exposure assessment approaches and methods that are feasible, acceptable, and limited in both cost and participant burden. Validation sub-studies have been investigated (Strauss et al. 2010), extant data will be relied on, and choices will have to be made about which chemicals to measure in stored environmental or biologic samples (Özkaynak et al. 2005; Gilliland et al. 2005; NRC/IOM 2008).
The development of the exposure assessment component of the NCS study highlights the challenges for exposure science to meet the demands for exposure information across time scales for large populations.
In 2010, EPA, NIEHS, and the NCS organized a workshop to engage scientists from the exposure, epidemiology, and health effects disciplines with the goal of identifying the most promising and practical exposure metrics to use in a study the size and scope of the NCS (Tulve et al. 2010). For the most part, the workshop participants agreed that questionnaires and diaries provide inadequate and unreliable exposure information; that more effective exposure metrics are needed that will provide better information on exposures and their inter- and intra-individual variabilities, and that high quality samples should be collected (in particular in the perinatal period) and archived for future analyses as new analytic methods are developed.
BOX 3-2 Case Study of the Hanford Environmental Dose-Reconstruction Project
As part of the Manhattan Project and continuing into the 1960s, all the plutonium for the U.S. nuclear-weapons program was produced at the Hanford Nuclear Reservation in the southeastern part of Washington State. During the early years, considerable quantities of radionuclides were released into the environment, notably iodine-131, which tends to accumulate in the thyroid gland of exposed people and can lead to thyroid cancers and other thyroid abnormalities. To address concerns of the downwind population, two projects were launched by the Centers for Disease Control and Prevention: the Hanford Environmental Dose Reconstruction (HEDR) Project and the Hanford Thyroid Disease Study (HTDS). The latter was an epidemiologic cohort study that made use of dose estimates produced by the HEDR Project. The HEDR Project is an important illustration of how exposure science informs epidemiologic research.
The HEDR Project began by reviewing over 38,000 pages of environmental-monitoring documents. Technical panels of experts in nuclear engineering, radiation dosimetry, environmental transport, meteorology, hydrology, statistics, and other fields developed an environmental-pathway model. The model reconstructed the releases from the plant and modeled their transport through air, soil, and water contamination; uptake by vegetation; intake by dairy cows and goats; milk production and distribution and ingestion by individual study participants and their mothers while pregnant; and ultimately dose delivery to the thyroid gland. Some parts of the complex model were informed by measurements, others by expert judgment; each step entailed careful consideration of the relevant uncertainties. The entire model was incarnated in a Monte Carlo computer program that yielded multiple realizations of possible doses to each individual, with variability among the realizations reflecting the uncertainties in the final dose assignments (Shipler et al. 1996). Although much concern had been expressed about contamination of the Columbia River, the analysis found the contribution to human exposures by that pathway to be negligible. The distribution of final dose estimates used by the HTDS had a range that was shown to provide adequate power to test for dose—response relationships with various thyroid outcomes (Kopecky et al. 2004). The null results for all types of thyroid abnormalities provided evidence that most cases in the region were unlikely to be due to radioactive releases from the Hanford plant (Kopecky et al. 2005), although this interpretation remains somewhat controversial (Hoffman et al. 2007).
A major lesson yielded by the HEDR Project and HTDS was the importance of community involvement, particularly in light of a concurrent class action lawsuit and suspicions that the U.S. Department of Energy (the owner and operator of the site) was influencing the study. Public, state, and American Indian representation in the independent Technical Steering Panel (TSP) that oversaw the project was viewed as essential from the beginning (Shipler 1995). The TSP adopted a commendable policy of openness in all aspects of
the project, including definition of scope, budget, and priorities. The investigators commented that the open approach required more time and energy but concluded that “if these issues and concerns are addressed early and if scoping of the project with an advisory panel is completed before major work is initiated, cost-effective planning and management can be achieved and ‘science in a fishbowl’ can be successful” (Shipler 1995, p. 108).
long low-intensity exposures or other patterns of temporal variation. While such issues are amenable to epidemiologic analysis, this is possible only if detailed histories of exposure are available, requiring exposure scientists to develop novel ways of reconstructing the entire history of past exposures or for monitoring time-varying exposures prospectively for extended periods.
The “exposome” concept (see Chapter 1) may provide a framework for representing a person’s lifetime of exposure to all potentially hazardous or beneficial agents. Although the concept is generally interpreted as relating to the totality of biologically-relevant exposures of either external or endogenous origins, the current enthusiasm is driven largely by the rapid advances of various—omics technologies (reviewed in Chapter 5) that permit agnostic assessment of a broad swath of internal biomarkers of exposure. For example, two recent publications illustrate its potential utility with “Environment-Wide Association Studies (EWAS),” agnostic scans for associations using a panel of a few hundred metabolite measurements (Box 3-3). These illustrations are analogous—albeit on a smaller scale—to genome-wide association studies that test the association of a disease or trait with hundreds of thousands to millions of genetic variants, but provide a “proof of concept” for an approach that could in principle be extended to a much broader range of exposures, monitored longitudinally. Novel approaches are needed to mine such data (Thomas 2010), together with internal and external markers of exposure to improve assessment of exposure-response relationships and, more importantly, to find ways to intervene before an adverse outcome is observed in an individual or population. That is a long-term goal that will require new approaches for conducting research, including capitalizing on future advances in individualized medicine and understanding the effects of changes in lifestyle and human behaviors.
Exposure assessment is usually constrained by cost or other feasibility considerations. It is seldom possible to measure everything that one would like to measure over the lifetime of an entire epidemiologic cohort. Hence, various sub-study designs are needed to devise a cost-efficient method of exposure assessment. That typically entails statistical modeling to combine the substudy data with the main-study data (Breslow et al. 2009). The study-design challenge involves trying to optimize the various tradeoffs—for example, between numbers of subjects and locations and breadth and duration of measurements—to yield the most precise estimates of the exposure—response relationship of inter-
est. Key to the optimization challenge is the measurement-error distribution expected with alternative designs and the use of statistical methods for adjusting for measurement errors (Carroll et al. 2006). Another type of hybrid design entails combining individual and group measurements. For example, one might correlate disease rates in large populations with estimates of the joint distribution of exposure, confounders, and modifiers obtained from sample surveys within each population (Sheppard et al. 1996). Box 3-4 illustrates the value of improved exposure estimates for epidemiologic studies.
The availability of various population-wide outcome databases—such as databases of mortality, hospitalization, and cancer incidence—is unfortunately not matched by population-wide exposure databases. The availability of a national dose registry for radiation workers in Canada, however, has made it possible to use record-linkage techniques to conduct large-scale studies of dose—response relationships for cancer (Zablotska et al. 2004). Establishing such registries and extending them to include medical-radiation doses, perhaps in the form of an electronic personal dose history, would be a boon for the field of radiation epidemiology. Ultimately, it would be desirable to have some life-course exposure registry for the entire population, or a periodic census of a large sample of the population that would inquire about a broad spectrum of environmental exposures for research purposes.
BOX 3-3 Environment-Wide Association Study (EWAS)
Patel et al. (2010) conducted an agnostic scan for associations of many measurements of internal exposures with type 2 diabetes; this was similar in spirit to Genome-Wide Association Studies (GWAS) that test the association of a disease or trait with hundreds of thousands to millions of genetic variants. Rather than using traditional methods of characterizing external exposures, the investigators used the “exposome” concept to assess potentially biologically effective exposures with a panel of 266 metabolite measurements obtained from the National Health and Nutrition Examination Survey of 503-3,318 people. They found statistically significant associations (after adjusting for multiple comparisons) with heptachlor epoxide (a pesticide derived metabolite), vitamin γ tocopherol, and some PCBs and found protective effects of β carotenes. A similar EWAS (Patel and Butte 2010) looked at associations with gene expression levels. Because it was not a longitudinal study, there is the potential for “reverse causation” bias, a tendency for disease or its treatment to affect biologic measurements rather than for the exposure to be a cause of the disease. Cohort studies would avoid that difficulty by relating biomarker measurements in unaffected people to their later onset of new disease. The Patel et al. study should be considered as a “proof of concept” for an approach that shows great promise for application of far more extensive panels of biomarkers from the various biobanks being assembled or already in existence that have stored biologic specimens from hundreds of thousands or millions of subjects with followup for disease incidence.
BOX 3-4 Value of Improved Exposure Estimates for Epidemiologic Studies
Reducing exposure error is critical for epidemiologic investigations. Previous air-pollution health-effects studies have underlined the importance of capturing spatial variability, particularly in urban areas (Logue et al. 2010; Bell et al. 2011). Accurate assessment of human exposures to atmospheric pollution requires knowledge of the spatial distribution of pollutants over cities on scales of 1-100 m (Chow et al. 2002). The improved resolution is expected not only to reduce exposure-assessment error but generally to result in larger health-effects estimates. For example, Jerrett et al. (2005) applied kriging techniques to study the association between within-city PM2.5 exposure gradients and mortality and found a substantially larger effect than previously reported with the city-average exposures (Liu et al. 2009, p. 886). The Women’s Health Initiative found a larger pollution effect on mortality when within-city exposure estimates were used (Miller et al. 2007). Nevertheless, although it is generally true that reductions in exposure measurement error can lead to improvements in health-effect estimates, it is not always true, and it depends on the specifics of the measurement error and the true exposure distributions (Szpiro et al. 2011). (See discussion in Chapter 5.)
The goal of a truly population-based exposure registry may be less feasible in the United States than in countries that have national health systems and population registries, at least in the foreseeable future. However, health-maintenance organizations (HMOs) may provide unique opportunities to build large-scale databases that, when combined with biomarkers of exposure assayed from routinely collected biospecimens and systematically collected exposure information (from clinic visits or questionnaires), could form the basis of long-term cohort studies. Outcome data reflecting clinic visits, hospitalizations, diagnoses, medication prescriptions, and mortality would be routinely available through followup data collection. Although not strictly random, the coverage of the larger HMOs is extensive enough to represent a broad spectrum of the population. Exposure science could also take advantage of data obtained on individuals and populations through market-based and product-use research to improve questions on exposures in epidemiologic studies.
Toxicology, whether focused on mechanisms or hazards, has historically been conducted outside the context of actual human exposures. Two major hazard evaluation programs, the U.S. Environmental Protection Agency (EPA) ToxCast and the National Institute of Environmental Health Sciences National Toxicology Program select chemicals and other materials (such as nanomaterials) for evaluation and use exposure as one qualitative selection criterion (Dix et al. 2007). But exposure context is for more than selection of chemicals for test-
ing. The biology of systems perturbed by exposures to stressors is highly sensitive to the magnitude of exposure (Slikker et al. 2004; Andersen et al. 2010). Mechanistic studies and hazard assessments conducted at concentrations that far exceed actual human exposures may produce results that are misleading because the observed effects are not likely to occur at lower doses or because low-dose effects may be masked by more overt toxicity at high doses. The availability of more exposure data could guide dose concentrations in toxicity studies.
The interdependence of toxicology and exposure science is recognized in these two communities, but exposure science is typically underemphasized as a principle in toxicology. The committee responsible for Toxicity Testing in the 21st Century: A Vision and A Strategy (NRC 2007) recognized the need for better integration and use of exposure science in toxicity assessment and called for its greater use in each step of the vision. The report spurred the rapid development of toxicity testing, in particular in in vitro, high-throughput methods, but its use of exposure science has seen little growth over the same period. That is unfortunate in light of the fundamental interdependence of the two fields, including the importance of exposure information in the design and interpretation of toxicity testing (Cohen Hubal et al. 2010; 2011). In place of current practice, the present committee envisions a shift toward a toxicologic assessment program that has an interface with exposure science and is influenced by and responsive to human and environmental exposure data. Such a program would strengthen the current toxicology-driven paradigm by focusing on the four activities described below.
• Select and set priorities among chemicals for toxicity testing. As EPA implements the recommendations of the 2007 National Research Council report, exposure science will become even more important for priority-setting (Cohen Hubal et al. 2010). As described in Chapter 2, that report envisioned a process for screening chemicals in commerce for hazard potential with rapid toxicity-pathway screens informed by and with priorities set through screening-level exposure assessments. The EPA ToxCast program is one example of such an implementation effort. In addition, efforts to reform the Toxic Substances Control Act (TSCA) will probably rely on priority-setting strategies that consider both exposure and toxicity potential. For those reasons, the present committee’s vision of enhancing the publicly available information on chemicals in commerce, improving screening for chemicals in the environment and in people (via biomonitoring and microsensor networks), and improving exposure modeling will form a solid foundation for priority-setting in relation to toxicity-pathway screening studies.
• Provide internal and external exposure information to inform selection of relevant concentrations of stressors for high-throughput toxicity testing. Exposure science needs to develop strategies to provide the information required to enable testing of stressors in animals. However, statistical-power considerations may make it infeasible or inadvisable to use only environmentally relevant doses in whole-animal studies. Newer in vitro toxicity-pathway studies can be con-
ducted at a wide range of doses, and these studies can be informed by exposure-related information, especially as it relates to internal dose. Reaching that goal will require a shift in exposure science toward collection of internal measures of exposure, as discussed in Chapter 5.
• Provide quantitative pharmacokinetic data (on absorption, distribution, metabolism, and excretion) derived from human-exposure studies. Targeted exposure studies need to include collection of exposure information to allow inference about human pharmacokinetic measures, such as the time course of exposure, for some high-priority chemicals. Exposure-characterization protocols should include measurements of external and internal markers of exposure to assess bioavailability, especially when exposures are predominantly via a single route. Greater use of longitudinal internal exposure studies that include periods of high and low or no exposures (such as those in occupational environments during and after work) could provide concentration time-course data similar to repeated-dose pharmacokinetic studies. For example, absorption rates, half-lives, and other pharmacokinetic measures and their variability within and between individuals could be derived from those data and would provide a wealth of critical human pharmacokinetic data for setting exposure concentrations for toxicity testing and for use in risk assessment (Teeguarden et al. 2011).
• Link exposure data with in vivo data on perturbations of toxicity pathways in human or wildlife populations to identify exposure-response relationships directly. The conventional hazard-assessment paradigm uses cell-culture systems or animal models to identify hazards. In the future, the present committee expects collection of higher-resolution and larger quantities of exposure data in a broader swath of the population to allow epidemiologists to identify potential hazards in human populations or ecosystems. The characterization of the hazards could then be explored by using more focused, efficient toxicologic studies at relevant exposure concentrations and durations or measurements of perturbations of toxicity pathways (that is, as seen in genetic or other biomarkers of effect) in exposed human or wildlife populations. Although these studies might be expected initially to focus on individual stressors, they would evolve with advances in exposure technologies to identify and characterize combinations of stressors.
There are also opportunities for epidemiology and toxicology to be more closely tied to the process of exposure assessment. For example, cell lines derived from epidemiologic study subjects could be exposed in culture to mixtures derived from samples from the specific environments to which the subjects were exposed, to identify measures of biologic activity of these complex mixtures. The measures of biologic activity would then represent more relevant, more specific measures of response for use as variables in subsequent epidemiologic studies. Exposure of experimental animal models to environmental mixtures associated with specific epidemiologic studies would provide additional information on exposure-response relationships, the time-course of development of disease, and the role of genetics as modifiers of exposure and response. In vivo
challenge studies in humans or experimental animal models can also provide much information on intermediate biologic responses to agents (for example, diesel-exhaust particles or second-hand smoke) and on genetic modifiers of these responses to exposures (Gilliland et al. 2004). That information could inform exposure—response or gene—environment interaction analyses of epidemiologic data. Finally, epidemiology and toxicology would benefit from a more sophisticated approach to modeling that takes advantage of the source-to-effect continuum and reduces uncertainties in study design and misinterpretation of results for use in mitigation and prevention (Georgopoulos et al. 2009).
Risk Assessment
Exposure assessment is one of the core components of regulatory quantitative risk assessment; therefore, the quality of exposure information and the state of exposure science are paramount in the quality and utility of risk assessment. The National Research Council and EPA have previously described the major steps in risk assessment, including hazard identification, dose—response assessment, exposure assessment, and risk characterization (NRC 1983, 1994, 2009). Although exposure assessment is often described as perhaps the most challenging component of risk assessment, prior National Research Council reports on risk assessment have made limited recommendations for improving the quality of exposure data or the utility of exposure assessment for quantitative risk assessment. Although those steps are important, a strategy for improving the quality and quantity of exposure data is needed to reduce uncertainties stemming from the exposure-assessment component of risk assessment.
A recent EPA Science Advisory Board panel provided nearly 100 recommendations for improving guidance in ecologic risk assessment (EPA SAB 2007; Dale et al. 2008) and suggested that further consideration was needed for assessing simultaneous exposure to multiple stressors, assessing spatial and temporal variation in exposures, and addressing uncertainties in exposure models.
Exposure assessment poses numerous challenges for risk assessment. Exposures change, so a risk assessment that uses data that are available today may no longer be valid months or years from now; this is especially true for chemicals newly entering the market, for which use and exposure patterns have not yet fully emerged. Important exposure pathways may be missed, and this can lead to underestimation of overall exposure or neglect of highly exposed populations. Risk assessments and exposure assessments tend to focus on one chemical at a time and potentially miss interactive effects that could influence both exposure and risk. Data needed for exposure assessment, such as data on chemical sales and product ingredients, may be proprietary and not publicly available because of trade-secrecy protections.
To be useful for risk assessment, an exposure assessment needs to be capable of identifying and quantifying the exposure of the populations that are most highly exposed and populations that are most vulnerable. The assessment should strive to include all relevant exposure pathways and allow the pathways to be identified and defined individually (for example, to allow for quantification of exposure from water or from food as well as total exposure). Exposure assessment also needs to consider background exposures to chemical or nonchemical hazards that could influence cumulative exposure or risk within a population. A mechanism for tracking and updating exposure estimates is needed to ensure that they continue to reflect real-world conditions. Finally, uncertainty, which is inherent in an exposure assessment, needs to be quantified to determine the level of confidence in the overall risk assessment.
The 2009 National Research Council report Science and Decisions: Advancing Risk Assessment, which focused on improving human-health risk assessment but also considered ecologic risk assessment, contains several recommendations related to exposure assessment. It describes major challenges that risk assessment is facing and concludes that regulatory risk assessment has become bogged down and that many assessments take a decade or longer to complete. Strategies that the present committee believes may improve the efficiency, quality, and utility of the exposure-assessment component of risk assessment include the following:
• Determine in advance the level of detail and type of exposure information needed to address the risk-management question at hand. Risk assessment should be viewed as a method for evaluating the relative merits of various options for managing risk rather than as an end in itself (NRC 2009). It is important to pose the policy question first and then gather the information needed to answer it. In the case of exposure assessment, screening-level information may be adequate to address some questions, targeted data may be useful for others, and extensive data may be needed in some circumstances, particularly for developing scientifically sound policies and regulations.
• Use health-protective exposure default assumptions when adequate data are not available and define the criteria needed to depart from them. Default assumptions are typically used in risk assessments when measured or modeled data are unavailable or to allow extrapolation from existing data. Defaults are intended for a risk assessment to move forward in a timely fashion despite inevitable data gaps and to be health-protective. For example, one standard default assumption in exposure assessment is the choice of a cutoff for estimating exposure in a population (for example, the 90th or 99th percentile) (EPA 1992; NRC 2009). An exposure assessment used for risk assessment should select a cutoff that is as health-protective as the data allow and should explain the choice of the cutoff in the context of EPA’s default assumptions and the existing data.
• Quantify population vulnerability better. An important purpose of exposure assessment is to identify populations that are at greater risk than the ma-
jority of the population because of their exposure patterns or susceptibility to health effects. For example, for a risk assessment of methylmercury, it is essential to gather data on people who fish for subsistence, because these people are expected to face the highest exposures (EPA 2000). In the example above, instead of selecting a population cutoff (such as the 99th percentile), a better approach would involve identifying the subpopulation in the upper end of the exposure distribution, measuring exposure in that subpopulation, and identifying strategies to reduce the upper-end exposures.
• Assess and quantify cumulative and aggregate exposures. Exposures may involve aggregate exposures to a given chemical via a variety of exposure pathways or may involve cumulative exposures to various chemical or nonchemical stressors that influence human exposure and health risk by influencing metabolism or excretion of a chemical and acting on the same biologic pathway or affecting the same organ system. There is a need to include multiple chemical, physical, or biologic stressors but also to consider other vulnerability and susceptibility factors that influence the effects of these stressors, such as nutritional and psychosocial status, including stress. Exposure assessments may therefore need to collect information about exposure to a variety of chemical and nonchemical stressors that may interact to influence health risk.
• Improve stakeholder involvement and make the process more accessible to the general public. Exposure information used as the basis of a risk assessment needs to be made available to the public and presented to enhance understanding, particularly regarding the assumptions used and the uncertainties and limitations of the data. Efforts should be made to make all models used to estimate human exposure for risk-assessment purposes nonproprietary and open to public scrutiny so that the basis of modeled data is clear and transparent. In addition, an open-source strategy for modeling software and algorithms could ensure an ongoing process of peer review and improvements in model specifications while ensuring transparency (for example, PLOTS 2012). Information on chemical use and product ingredients also needs to be made publicly available to the greatest extent possible to allow the public to understand exposure pathways and to ensure that important exposure information is not hidden by trade-secrecy provisions.
Box 3-5 illustrates the role that exposure data and risk assessment have played in the establishment of a drinking-water standard for perchlorate.
Risk Management
Exposure science can inform public policy in a broad array of scenarios. Exposure information, even with minimal or no data on chemical hazard, can provide important information about trends, disparities in population, geographic “hotspots,” cumulative exposures, and predictors of vulnerability. Fur-
BOX 3-5 Case Study of Perchlorate in Drinking Water
In 1997, perchlorate, a chemical component of rocket fuel was detected in several drinking water wells in Sacramento County, CA, near a large industrial facility. In response, the California Department of Public Health laboratory developed a new analytic method for detecting perchlorate at lower concentrations than was previously feasible—down to 4 ppb. The method was later used to conduct a survey of multiple drinking water and agricultural wells in California, including in densely populated Los Angeles, Riverside, and San Bernardino Counties, where perchlorate was detected in numerous wells and nearby pollution sources were identified. Sampling also showed perchlorate at low concentrations (5-9 ppb) in Colorado River water, an important source of drinking water and water for agriculture in southern California (CDPH 2010a).
The widespread detection of perchlorate in drinking water catapulted it to public attention and drove action in numerous states to develop enforceable drinking water standards. Further discoveries of the chemical in agricultural wells spurred extensive investigations of perchlorate uptake into food by the Food and Drug Administration and by independent scientists, and the resulting information was used in exposure assessments by the California Environmental Protection Agency and others to derive risk based standards. Development of a more sensitive analytic method for detecting perchlorate in water supplies ultimately drove the cleanup of numerous industrial and military sites and the creation of a National Research Council committee that reviewed the U.S. EPA’s risk assessment for perchlorate. EPA cited the widespread exposure to this chemical as a major justification for their recent decision to promulgate a national drinking water standard for perchlorate.
It is apparent from this case study that environmental monitoring of exposure sources can identify significant public health issues, inform science and risk assessment, and drive risk management.
thermore, the very existence of exposure data creates an imperative to generate information about hazard so that the importance of the exposure can be better understood. Several important existing and potential applications of exposure science in public policy are described below.
Exposure-based prevention. A screening-level exposure assessment can inform decisions before new chemicals enter the market. Currently, that is done in the context of a formal risk assessment of pesticides. For new industrial chemicals on which exposure data are not available, EPA considers projected use and predicted chemical properties in making a decision about whether to request testing under TSCA, although in reality this is rarely done (GAO 2009). Screening exposure assessments could be used much more aggressively. For example, consideration could be given to keeping chemicals that are highly likely to persist and bioaccumulate off the market, so as to avoid the potential to contaminate the environment at increasing concentrations over time and poten-
tially result in human and environmental hazard. Another example of exposure-based prevention would involve identification of chemicals that are predicted to contaminate water because of their persistence and solubility and restriction of uses that could threaten surface water or groundwater. Finally, testing of consumer products in premarket controlled studies can be used to prevent toxic materials from reaching humans, and causing harmful exposures.
Exposure-based control. Post-marketing surveillance of chemical exposures can be used to ensure safety with respect to chemicals used or distributed in large quantities. About 200 chemicals were included in the most recent National Report on Human Exposure to Environmental Chemicals (CDC 2011). Other chemicals are studied by individual researchers or in state programs. There is no requirement that companies conduct post-marketing surveillance, and the Centers for Disease Control and Prevention does not have the resources to screen routinely for the thousands of chemicals that are used and distributed in large quantities or to which people may be exposed. Broad exposure surveillance of a longer list of high-priority chemicals would provide important information about trends, exposure variability, and magnitudes of exposure. Such surveillance would help evaluate whether regulatory actions designed to control exposure are effective, would allow priority-setting among chemical exposures to which are increasing, and would target at-risk populations for exposure reduction. For chemicals that can be biomonitored, surveillance could initially focus on worker populations and ecosystems near manufacturing or processing facilities. For widely dispersed chemicals (for example, those in consumer products), exposure of the general public or specific populations could be monitored. For chemicals that cannot be biomonitored (for example, because they have short biologic half-lives), near-source environmental monitoring or modeling of exposure could be possible. If exposure of any population is above a screening level of concern or if exposures are shown to be increasing, policy action could reduce exposure to prevent potential health problems from developing. Examining product-use patterns and the release of contaminant residual from products can keep highly toxic contaminants from coming into contact with consumers or can help to reduce the effects of such exposures.
Exposure justice and equity. The reduction of disparities in exposure to chemical and nonchemical stressors is an important goal of public policy. That principle is reflected in Presidential Executive Order 12898 (February 11, 1994), which calls on federal agencies to “improve research and data collection relating to the health of and environment of minority populations and low-income populations (p. 2)”, including “multiple and cumulative exposures” (p. 3). The Executive Order expects, furthermore, that “each Federal agency shall make achieving environmental justice part of its mission” (p. 1).
For agents on which there are population exposure data, exposure disparities can be tracked by evaluating the distribution of the exposure concentrations
over time (for example, the median, mean, standard deviation, or range).1 It is important that exposure science focus on identifying the segments of the population that are in the top end of the exposure range for any given stressor. These “high-end” exposed populations need to be explicitly assessed so that the exposure pathways can be understood and addressed. Policy actions could appropriately include engagement of the affected communities in a collaborative process to identify and reduce the sources of exposure (for example, Brown et al. 2012). Community participation can help ensure that the research questions that are being asked are relevant to the needs and concerns of both the researchers and the affected community, increasing the likelihood that the project will contribute to improving public health (O’Fallon and Dearry 2002). Progress toward that goal will be seen in a reduction in the skewness of the population exposure data or through followup assessment in targeted populations.
In addition to population-based surveillance to reduce exposure disparities, exposure science has an important role in performing rapid on-site assessments of the relationship between potential exposure and potential health outcomes in communities. Such assessments have been done around hazardous waste sites and in other settings where contaminants have already been introduced into the environment, but can also be used more consistently as a rapid exposure screening tool before contaminants are inadvertently introduced into communities or the personal environment.
Box 3-6 illustrates the role that exposure science played in managing potential risks posed by polybrominated diphenyl ethers (PBDEs), including spurring the creation of the California biomonitoring program and encouraging a broad strategy for identifying emerging chemicals of concern in consumer products.
Compensation Policy
Exposure science plays an important role in policy decisions regarding compensation of people who may have been harmed by exposure to hazardous substances. Many government programs have been established to provide compensation to veterans exposed to various deployment-related or combat-related exposures (for example, Agent Orange exposure of Vietnam veterans), to workers (for example, uranium miners), or to members of the general public (for example, people downwind of the Nevada nuclear test site). In addition, state workers’ compensation programs often have substantial difficulty addressing questions of causation of potential occupational illness due—at least in significant part—to a lack of exposure data for workers.
_______________________
1Extremely skewed data can be identified easily because the mean is significantly higher than the median, and the standard deviation is large. Although most exposure data do exhibit a log-normal (skewed) pattern, the degree of skewness varies.
BOX 3-6 Case Study of Chemicals in Breast Milk: Policy Action Based on Exposure Data
In 1999, researchers in Sweden discovered a previously unknown group of chemicals in women’s breast milk. When the researchers tested stored breast-milk samples from the 1970s and 1980s, they discovered that the chemicals had increased dramatically over the decades, doubling every 5 years (Meironyte et al. 1999). Soon after the Swedish discovery, U.S. scientists reported the same chemicals in breast fat and breast milk in Indiana, Texas, and California (Schecter et al. 2003). The U.S. levels were much higher than those reported in Europe; the California samples had concentrations 40 times higher than those in Sweden. The chemicals, polybrominated diphenylethers (PBDEs), were flame retardants commonly used in consumer products. At that time, the PBDEs were essentially untested for toxicity to humans or ecosystems, but they were in widespread use in foam cushions, fabrics, and electronics. PBDEs are structurally similar to polychlorinated biphenyls and dioxins, and they share environmental chemical characteristics of persistence and bioaccumulation.
Those discoveries led to numerous policy actions: PBDEs immediately had high environmental-health research priority, sparking numerous exposure, toxicology, and epidemiology studies; the European Union banned most of the PBDEs in 2004; California and other states passed legislation to phase out some of or all the PBDEs; and EPA reached an agreement with the U.S. manufacturer to cease production of the most bioaccumulative PBDE voluntarily, and other PBDEs were later phased out of U.S. production. Although these chemicals are still being produced in China and other countries. The California statewide biomonitoring program was created by the legislature in large part because of the discovery of PBDEs in breast milk and breast tissue. The legislation identifies one of the priorities of the program as “the need to assess the efficacy of public health actions to reduce exposure to a chemical” (CDPH 2010b, p. 32). The California Scientific Guidance Panel added an additional criterion that was based on the need for early identification of emerging chemicals of potential concern (CDPH 2010b). One of the first actions of the program was to list as priority chemicals “chlorinated and brominated organic chemicals used as flame retardants” (CDPH 2010b, p. 6). The broad language of the listing was designed to address the chemicals of concern that were increasing in market share to replace the PBDEs.
Some instances of determination of compensation, such as “presumptive disability policies” for some classes of veterans intended to give the veterans the “benefit of the doubt”, require no individual exposure assessment; the mere fact of having developed a particular disease deemed potentially service-connected and having had the potential for exposure is sufficient. The need for presumptions often arises because of a lack of exposure data (IOM 2008). In other situations, extensive “dose-reconstruction” investigations may be required for a person, such as an “atomic veteran,” to qualify for compensation. For radiation-related exposures, Congress enacted the Radiation Exposure Compensation Act
after a provision in the Orphan Drug Act that directed the National Institutes of Health to establish a set of “radio-epidemiologic tables” of estimates of probability of causation (PC), the probability that a given cancer was caused by a particular radiation dose (NIH 1985).
A key feature of that and many other compensation programs and toxic tort litigations is the uncertainty about a person’s contact with the toxicant of concern that resulted in exposure. For radiation in particular, determining exposure has been facilitated by a Monte Carlo program from the National Institute for Occupational Safety and Health, the Integrated RadioEpidemiologic Program (NIOSH 2011), which provides a nuclear worker with not only a best estimate of his or her radiation dose but also an estimate of its uncertainty and carries this uncertainty through to the calculation of PC. Although the validity of the PC concept has been called into question (NRC 1984; Greenland and Robins 2000), there is no doubt that exposure information and its uncertainty—at either the individual or the population level—are central to establishing fair compensation policies. For example, IOM (2008) pointed out that policies aimed at erring on the side of avoiding false-negative decisions (failing to give compensation when it is due) in favor of false-positive decisions (excessively liberal policies that compensate people who do not deserve it), such as certain presumptive—disability provisions for veterans, would have the effect of rewarding ignorance—people with highly uncertain exposures (or with rare diseases for which the dose—response relationship is highly uncertain) are more likely to be compensated than those with more precise information. Such policies need further consideration to ensure an appropriate balance between fairness and societal burden. Overall, better assessment and characterization of worker exposures are needed to develop more accurate and protective exposure limits for the workplace. Better monitoring of workers can help to assure that current exposures are not harmful and improved exposure data can help to clarify compensation issues when people do become ill.
Exposure assessment has contributed to urban and environmental planning, informing our understanding of how different patterns of land use can change the magnitudes of emissions and exposures of humans and ecosystems. Exposure science is increasingly used in health impact assessments.2Box 3-7 illustrates the application of exposure science to health impact assessment in San Francisco.
_______________________
2A 2011 National Research Council report defined health impact assessment as a structured process that uses scientific data, professional expertise, and stakeholder input to identify and evaluate public-health consequences of proposals and suggests actions that could be taken to minimize adverse health impacts and optimize beneficial ones (NRC 2011).
BOX 3-7 Health Impact Assessment of Mobile Sources in San Francisco
Several health impact assessments conducted in 2004–2006 on land-use plans in Oakland and San Francisco identified conflicts between roadway proximity and sensitive land use. The conflicts resulted from placement of new high-density infill developments on land parcels along major highways (Bhatia 2007). Such conflicts were not addressed by federal, state, or local environmental standards, nor by local planning codes. The California Air Resources Board recommended not building housing within 500 ft of highways, but land-use planners typically did not follow this guidance.
In 2007, the San Francisco Department of Public Health (SFDPH), in its role as the environmental-health authority for the city, participated in an environmental review—required under the California Environmental Quality Act (CEQA)—of four neighborhood rezoning plans (Bhatia and Wernham 2008). The plans proposed large-scale industrial-to-residential rezoning in areas of the city near highways and busy arterial roads. SFDPH identified several important environmental impacts (air and noise pollution and traffic collisions) and proposed mitigations. To create a mitigation program compatible with the needs of local planners and CEQA regarding environmental impact assessment, SFDPH needed to identify an assessment method for each development project, a quantitative action level that triggered mitigation, and alternative engineering approaches for mitigation. To meet those needs, SFDPH proposed using a threshold concentration of fine particulate matter (PM2.5) of 0.2 μg/m3 from nearby roadway sources within 500 ft of a sensitive receptor as a proxy for important roadway hazards. The CALINE dispersion model (Benson 1989) was used to predict traffic-attributable PM2.5, and a series of ventilation and filtration standards were proposed for new buildings within the 500-ft zone of roadways as a performance-based mitigation measure (Bhatia and Rivard 2008).
The approach was accepted by planners and the development community and adopted as mitigation for the four neighborhood plans (Bhatia and Wernham 2008). Later, SFDPH and the Department of Planning successfully sought the adoption of the approach as a public-health law applicable to all projects citywide. It remains the only local law in the United States that requires the assessment and mitigation of air pollution hazards from nearby roadways.
The current statute applies only to new residential development, but initiatives are underway to address existing development and a wider array of impacts, including noise pollution and dangers to pedestrians and bicyclists from traffic collisions. A key strategy for addressing hazards from nearby roadways is to develop metrics and modeling tools that can account for refined spatial characterizations of exposure and allow for assessments of attributable disease burden or other health-effect estimates (Wier et al. 2009a, 2009b; Bhatia and Seto 2011). The new law and associated policies rely heavily on information generated by exposure science and environmental epidemiology.
Urban Planning
Major economic and urban transformation over the last 30 years has led to a shift from large stationary emitters to larger concentrations of mobile sources and small manufacturing facilities, and more complex commuting and mobility patterns among workers. Those changes have accentuated small-area variations in pollution due to traffic and localized manufacturing. The restructuring has generally increased the amount of traffic and created spatial mismatches in residential and employment locations, lengthening commuting times and in-transit exposures of many workers (HEI 2010). As a consequence, environmental exposures affecting health have become more spatially heterogeneous and more dependent on human mobility and activities within and between places.
Exposures associated with urban transportation systems display variation over small areas that is difficult to quantify because of its spatial heterogeneity. Those exposures are pervasive in that they affect large populations. Recent studies by the Health Effects Institute (HEI 2010; Jerrett et al. 2011) have documented the proportion of the population that experiences high exposures from traffic, resulting in increased risks of accidents and exposures to air pollution and noise. In Toronto and Los Angeles, 43-45% of the population were in the “high-exposure zones” (that is, 500 meters from a major highway and 100 meters from a major road) (HEI 2010, p. 3-13). In the denser cities of Asia, 55-77% of the population were “highly exposed” (Jerrett et al. 2011, p. 35). Exposure science in urban areas faces the challenge of assessing spatially heterogeneous exposures that are pervasive throughout the urban structure. There is an increasing urgency to link exposure science to urban planning because of increasing population densities in cities, particularly through infill development, intended to reduce pressure from vehicle miles traveled to achieve climate change mitigation and other environmental goals. Often such “densification” efforts puts residents in greater proximity to transportation corridors, leading to increased exposures from traffic emissions and other related risks (for example, noise pollution) (Melia et al. 2011; Hankey et al. 2012). At the same time there will be continual stress on the water supply and the quality of tap water available.
Exposure science helps to inform urban planning, often providing insights into inherent trade-offs (Künzli et al. 2003). In many countries, for example, physical inactivity is a major threat to public health, and some of the inactivity is due to an urban environment that discourages active travel by foot or bicycle. However, active travel involves other exposures and potential health risks, including air pollution, accidents, and noise (de Nazelle et al. 2011). Exposure studies are being conducted to understand the effects of active travel. They require integration of ambient-pollutant concentration data with information on physical activity to assess potential inhalation and dose of air pollutants.3
_______________________
3Information on ambient-pollutant concentrations combined with data on physical activity can inform potential dose, inasmuch as physically active people have inhalation
de Hartog et al. (2010) conducted an integrated health impact assessment that compared the likely effects of physical activity in bicycling with the risks posed by increased air-pollution exposure. The benefits of increased exercise far outweighed the increased risks from air pollution, but ideally urban planners can use exposure science to devise bicycling and walking routes that minimize the risks faced by active commuters, and exposure science can have a critical role in identifying the environments that are associated with the highest exposures (Marshall et al. 2010; de Nazelle et al. 2011).
Salutogenic (beneficial) exposures may generate health benefits. Many of the attributes (for example, parks) that promote human health also support ecosystem health and ecologic services. For example, urban forests contribute to carbon sequestration and protect water and air quality (Dwyer et al. 1992, McPherson et al. 1997) while encouraging behaviors positively associated with mental and physical health (Maas et al. 2006; Maller et al. 2006). Access to green space, parks, and recreational programming has been associated with increased physical activity or reduced obesity in large epidemiologic studies (Wolch et al. 2011). Studies in England have shown that in areas with greater access to green space, the socioeconomic group differences in cardiovascular risk is reduced (Mitchell and Popham 2008). In a study conducted in southern California, children equipped with GPSs and accelerometers were shown to have significant increases in physical activity while exposed to green space (Almanza et al. 2011). Recent studies also suggest that access to such resources may follow socioeconomic group differences, in that minority groups and socio-economically disadvantaged groups have fewer options to access these resources (Dahmann et al. 2009). Also parks in poor and minority-group neighborhoods tend to be more polluted than those in neighborhoods of wealthier people and higher proportions of white people (Su et al. 2011). Exposure science can help urban planners to identify areas that lack salutogenic exposures that may buffer adverse exposures or produce direct health benefits. Salutogenic exposures also illustrate the link between healthy ecosystems and human health.
Ecosystem Planning
Although exposure assessments can include a diverse suite of ecologic receptors (for example, fish, aquatic plants, amphibians, birds), integrating ecologic information in a way that captures risk for the whole ecosystem has been difficult. In practice, a true ecosystem perspective may be restricted to the initial development of site conceptual models that identify potential exposure pathways, after which the focus may be on specific and limited receptors. Exposure science and risk assessment could benefit from adopting a holistic, systems-level perspective, similar to that embodied in ecosystem planning and management. Already, exposure assessments are beginning to integrate and apply information
_______________________
rates that are 2-5 times greater than those of persons in public transit or in private vehicles, and that also may be higher because of the proximity to car traffic.
from the ecologic, socioeconomic, and political realms as does ecosystem planning (Grumbine 1994; Brussard et al. 1998; Szaro et al. 1998). Like ecosystem management (Grumbine 1994), exposure assessments also consider the hierarchic context of the process or problem, work collaboratively across ecologic boundaries, use diverse data from research and monitoring efforts, and, to some extent, recognize that humans are part of the ecosystem. However, one piece of ecosystem planning and management that is largely absent from current exposure assessments is the emphasis on adaptive management, whereby managers and planners treat management as a learning process (an experiment) and adjust actions as understanding improves. (Box 3-8 illustrates how exposure science has contributed to our understanding of the environmental impacts of stressors on Lake Tahoe.)
An ecosystem-based focus on land-use practices might be especially useful for exposure science. For example, regional changes in land-use practices can result in high levels of agricultural runoff and untreated sewage effluent that lead to eutrophication, in which aquatic ecosystems accumulate high concentrations of nutrients (such as phosphates and nitrates) that promote plant growth. Algal growth can become excessive and sometimes lead to harmful algal blooms (Paerl 1997; Cloern 2001; Anderson et al. 2002; Kemp et al. 2005). Likewise, land uses that reduce vegetative cover, especially trees, may result in changes in temperature that can directly prompt health-threatening exposures (for example, extreme heat events) or indirectly influence exposure to other substances. In aquatic ecosystems, degraded riparian zones, loss of forest cover, runoff from impervious surfaces, and discharges from industry can lead to rising water temperatures and increased toxicity. Above-normal temperatures compromise the function and integrity of aquatic ecosystems. They can also increase the sensitivity of aquatic animals to heavy metals and pesticides, including cadmium (Cherkasov et al. 2006, 2007), copper (Gupta et al. 1981, Khan et al. 2006), diazinon (Osterauer and Köhler 2008), and trichlorfon (Brecken-Folse et al. 1994).
An ecosystem approach to planning requires recognition that exposures may result from a series of diffuse and indirect interactions among humans and nonhuman species. Exposure to emerging infectious diseases, many of which originate in wildlife hosts or reservoirs, is an excellent illustration of this complexity. Human activities, particularly those related to habitat modification and resource subsidization, not only influence ecologic health but mediate wildlife—pathogen interactions through changes in host density, behavior, and spatial distribution (Dobson and Foufopoulos 2001; Gehrt 2010). Urban development, for instance, promotes high densities of some wildlife species (such as raccoon, Procyon lotor) that serve as hosts of many pathogens of public-health importance (such as rabies and leptospirosis: Bradley and Altizer 2006; Gehrt 2010) and can profoundly alter trophic interactions in ways that can result in loss of biodiversity (Faeth et al. 2005). Socioeconomic factors can exacerbate exposure if people lack the financial and educational resources that support preventive behaviors, such as vaccinating pets, excluding or removing wildlife from homes, and securing refuse and other potential wildlife attractants.
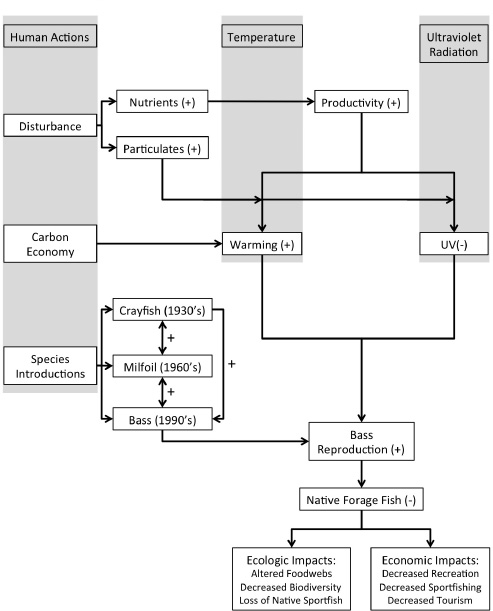
BOX 3-8 Exposure to Multiple Stressors in a Large Lake Ecosystem
Lake Tahoe is a large (19 × 35 km) and deep (501 m), ultraoligotrophic, montane lake in the Sierra Nevada of California and Nevada. It is known for its cobalt blue waters that are due, in part, to its small watershed area in comparison with its large lake volume. The economy of Lake Tahoe is intimately linked with its ecology—the economic value of the lake and surrounding basin is closely related to the perceived scenic and pristine character of the region’s water, air, and forest resources (for example, Kearney et al. 2008). Recreation is the main industry of the region, with an economic impact from visitors of about $320 million per year, which supports nearly 10,000 jobs (Eiswerth et al. 2000; Dean Runyan Associates 2009). The intense pressure from local recreational activities, atmospheric deposition of nutrients and toxicants, intentional and unintentional introduction of nonnative species (for example, Eiswerth et al. 2000; Kamerath et al. 2008; Chandra et al. 2009), and climate change result in the simultaneous exposure of the ecosystem to multiple stressors (see Figure 3-2). Assessment of multiple stressor exposure in large, complex ecosystems is not easy, and the outcomes of the exposures are often unpredictable. However, Lake Tahoe presents a good case study of the ecologic and potential economic impacts of multiple stressor exposures.
Within the last 40 years, scientists have documented a steady decline in lake clarity and an increase in primary production (Jassby et al. 1996; Goldman 2000). That is especially the case with respect to ultraviolet (UV) radiation attenuation in the nearshore areas of the lake that are close to human developments and activities (Rose et al. 2009). Several factors play a role in the decrease in clarity, including the introduction of fine inorganic particles from the watershed; resuspension of fine particles due to wind, currents, and boats; and the settling of particles on the lake bottom (Jassby et al. 1996; Swift et al. 2006). Those changes have been accompanied by an increase in water temperature facilitated by increased absorption of solar radiation in turbid waters and due to climate change (Tucker et al. 2010). The combined and interactive effects of exposure to those stressors have had substantial adverse effects on the ecology of the watershed.
The role of interaction of water temperature, UV radiation, and resident nonnative species in the establishment of novel nonnative species, altered food-web structure, and increased exposure to toxic pollutants illustrates the effects of multiple stressor exposures. Nonnative crayfish became established in the lake in the 1930s (Chandra et al. 2009) and are widespread throughout the lake, Eurasian watermilfoil (an aquatic plant) was first discovered in the southern portion of the lake in the 1960s (Eiswerth et al. 2000), and warm-water centrachid fishes (largemouth bass and bluegill) were introduced by anglers in the middle to late 1990s (Kamerath et al. 2008; Chandra et al. 2009). By the middle 1990s, milfoil had spread to nearly all low-energy nearshore areas; by 2007, bass and bluegill were observed in marinas and nearshore areas containing milfoil and crayfish around the lake (Kamerath et al. 2008). Bass and bluegill are aggressive and can have predator-related and
competition-related effects on the native fishes in the nearshore areas (Gevertz et al. 2012). Their establishment has been possible only through the combined effects of increased water temperature, decreased UV radiation and water clarity, habitat alteration (milfoil), and abundant prey (crayfish and native fishes) (Kamerath et al. 2008; Tucker et al. 2010; Gevertz et al. 2012). Recently, water-quality thresholds for temperature, UV radiation, and water clarity required to prevent the establishment and spread of bass and bluegill have been suggested (Tucker et al. in press).
Ecologic and physiologic research has predicted that exposure to multiple stressors would facilitate the establishment of warm-water fish in Lake Tahoe, but an unpredicted outcome of the interactions may be an increase in exposure to toxic pollutants in the ecosystem. Mercury is an important toxic pollutant globally and in Lake Tahoe (Drevnick et al. 2010). Methylmercury (MeHg) biomagnifies in food webs, with the highest rates of transfer occurring in lower trophic levels (for example, water to algae) (Hammerschmidt and Fitzgerald 2006) and with top-predator fish and birds accumulating the most (Wiener et al. 2002). Crayfish can accumulate large amounts of MeHg as a benthic organism eating primarily periphyton and macrophytes (Momot et al. 1978) and largemouth bass commonly supplement their diets with crayfish (García-Berthou 2002). In addition to its primarily plant-based diet, crayfish are facultative omnivores and will feed on fish carcasses (Minckley and Craddock 1961). The biomagnification of persistent bioaccumulative toxicants, such as MeHg, may be increased by a trophic feedback cycle in which bass eat crayfish, which eat bass, and so on. Laboratory experiments and modeling demonstrate that such a trophic feedback cycle substantially increases MeHg accumulation in bass and crayfish (Bowling et al. 2011). The results are supported by anecdotal evidence that another, native species (brown trout) has higher tissue concentrations of MeHg in lakes that have crayfish than in lakes that do not (Oris et al. 2004). Thus, it is possible that exposure to multiple stressors that have facilitated the establishment of largemouth bass in Lake Tahoe can result in large increases in MeHg exposure of both native and nonnative species and affect the ecologic resources of the region. These native and non-native species are also direct vectors of MeHg exposure of humans and could decrease the recreational value of the lake.
Changes in species composition due to human activities may affect exposure through alterations in species interactions. For example, human-facilitated invasion of Amur honeysuckle (Lonicera maackii), a nonnative shrub, increases human risk of ehrlichiosis, an emerging infectious disease transmitted by ticks (Allan et al. 2010). The high risk results from a preference of a key tick and pathogen reservoir, white-tailed deer (Odocoileus virginianus), for areas with dense honeysuckle. In aquatic systems, the invasive round goby (Neogobius melanostomus) is thought to facilitate mobilization of contaminants in food
webs and to increase exposure of humans because its persistence in contaminated environments draws predatory fish, which also are popular game species, into polluted habitats (Marentette et al. 2010). Thus, understanding human exposure to pathogens requires a holistic ecosystem perspective.
An ecosystem approach extends beyond “natural” systems to include the built environment. Cities are coupled human—nature systems that can be understood only through their linked ecologic, physical, and socioeconomic components (Pickett et al. 2001; Alberti et al. 2003). Spatially explicit approaches common to ecosystem planning fit urban ecosystems well in that they can be viewed as mosaics of risk and protection (Fitzpatrick and LaGory 2011). Characterizing the heterogeneity of social and environmental attributes in urban ecosystems better is especially useful in a climate of diminishing resources. For example, recent studies demonstrate that bioavailability of contaminants in soils is strongly mediated by environmental heterogeneity (Filser et al. 2008), and this environmental heterogeneity can be characterized with sophisticated mapping techniques to understand exposure risk (Lahr and Kooistra 2010). By understanding heterogeneity, planners and managers can identify the most critical areas for remediation, saving resources and sometimes avoiding ecologically damaging consequences of unnecessary efforts (such as removing topsoil from large areas). Understanding complex socio-ecologic processes that are operating in urban ecosystems is a prerequisite for protecting ecologic and human health.
Response to a natural, accidental, or terrorist event or to an act of war often requires understanding of exposure to a stressor or multiple stressors. Instrumentation is needed to detect biologic, chemical, and physical stressors accurately and cost effectively, and, depending on the evolving situation, both community and occupational exposure monitoring may be required. Defined strategies for rapidly assessing exposures are critical for applying predictive and measurement tools. Active exposure assessment is an important need that has been discussed in previous National Research Council reports in connection with the deployed military (NRC 1999; 2000a,b; IOM 2000), in analyses of exposure in the aftermath of the World Trade Center (WTC) attack (Lioy 2010a), and in general disaster discussions and analyses (Dominici et al. 2005; Bongers et al. 2008; Rodes et al. 2008).
Several National Research Council reports (NRC 1999; 2000a,b; IOM 2000) have discussed the need for exposure assessment after military activities, during combat, or where troops are deployed to guard a facility or area associated with identified or suspected hazards, including oil fields, chemical plants, contraband destruction (incinerator) sites, and power plants. Exposure assessments are intended as a means of reducing personnel injuries and as a form of preventive surveillance as information on exposures can be used by physicians at a later time to treat exposed individuals if they develop symptoms. In such situations, there is a need to provide information on the personal protective equipment required and to enforce administrative controls to guard sites. Monitoring needs to be flexible and surveys need to be conducted to characterize the exposures that may be encountered before establishing permanent security or
conducting similar activities. Biologic monitoring and surveillance may need to be formalized for deployed troops.
In the aftermath of the WTC disaster, the nature of the exposures and the populations exposed changed at Ground Zero (Lioy and Gochfeld 2002; EPA 2002, 2003; GAO 2007; Lorber et al. 2007). The evolving situation resulted in exposure of responders and others under different conditions. Box 3-9 discusses issues surrounding emergency management after the attack on the WTC.
It is important to develop and test a series of protocols to ensure that community and occupational monitoring and analytic capabilities are field-ready in the event of an emergency situation. Exposure-monitoring platforms may need to include personal sensors and portable monitors mounted on vehicles and be portable and robust enough to be airlifted to remote sites. A variety of samplers can be tested, including ones that measure physical, chemical, and biologic materials. Surveillance of workers and the community with biologic monitoring is essential for understanding continuing exposures or the cessation of exposures at least through rescue, re-entry, and recovery. Similarly, a field laboratory to process biomonitoring samples would be important. Strategies to minimize continuing community exposure are essential, for example, the use of exclusion zones for various periods. Effective personal protective devices can be made available to those in harm’s way to minimize injury or disease, and leaders could define and enforce the use of this equipment.
During disasters, there need to be clear and effective lines of communication, members of a team that can interpret data, and officers who understand the meaning of the exposures that are determined. Most important, guidance and recommendations from professionals on how to proceed are needed.
Exposure science has made and continues to make contributions to many fields. Increased emphasis on integration and new applications of the principles of exposure science can contribute major benefits to the areas of epidemiology, toxicology, environmental regulation, ecologic planning, and disaster management.
Epidemiology
Exposure science should aim to provide epidemiologists with novel tools to improve exposure assessment, particularly comprehensive assessment of cumulative exposures to the ensemble of relevant stressors. That will require use of new remote-sensing and other high-volume techniques for measuring the external environment and new—omics technologies for the internal environment. Population-wide exposure databases (such as a national dose registry for radiation exposures) that could be routinely linked with population-wide outcome databases (such as cancer registries or the National Death Index) would facilitate rapid discovery of novel hazards.
BOX 3-9 Emergency Management After the Attack on the World Trade Center
The collapse of the WTC towers was a horrific aftermath of the 9/11 terrorist attacks. The generation of dust due to the disintegration of building materials and the ensuing fires created demands for exposure information to understand the associated potential health effects on workers and the community—in the immediate aftermath of the disaster, in the days immediately afterwards when the dust was resuspended, and during cleanup in the weeks and months after the disaster. The need for such data was not anticipated before the event (Lioy and Gochfeld 2002; Lioy 2010a).
The WTC collapse illustrates the need for an integrated and timely approach to data collection involving complex mixtures (EPA 2003). Data on the presence of radiation and other hazardous agents were initially collected by several agencies but with little coordination. Inventories of potential sources of known hazardous chemicals (such as pesticides) were documented. Biologic samples were collected from firefighters to examine the concentrations of gases and other contaminants that may have been inhaled at Ground Zero (Edelman et al. 2003). However, the composition of WTC dust remained unknown, and this crippled the initial assessments of the potential hazards (Lioy 2010a). The failure to characterize exposures adequately stemmed from a lack of real-time portable devices to measure particles and gaseous materials in dusty environments and from a lack of understanding that the immediate concerns were short-term exposures (Lioy 2010a). The mindset at the time was focused on low-level exposures (for example, to asbestos and fine particles) associated with long-term disease outcomes.
The exposure-assessment issues stemming from the WTC disaster led to discussions of how to characterize exposure during rescue, reentry, recovery, restoration, and rehabitation after an event (Rodes et al. 2008; Lioy 2010b). An important aspect of the WTC disaster was the recognition that the combination of collapse of the buildings with intense fires resulted in emissions of complex mixtures because of the nature of the construction materials, furnishings, and equipment (EPA 2002, 2003; GAO 2007). Training of emergency and law-enforcement agency personnel regarding potential exposures and potential hazards to health is important, including rapid exposure characterization to minimize additional injuries to workers and the public.
Risk assessment, risk management, and risk communication are valuable evaluation tools, but their effectiveness depends on the accuracy of information and on the speed with which it can be interpreted and conveyed. A lesson for exposure science from the disaster is that it is important to assume a worst-case scenario and efforts would need to be made in advance to implement exposure-characterization strategies, including prepositioning of appropriate measuring devices. The application of exposure-science principles during acute events can augment activities associated with a national response plan.
Toxicology
Exposure science needs to interface with toxicology to inform selection of and priority-setting among chemicals at environmentally relevant concentrations for toxicity testing, to develop human pharmacokinetic data, and to improve understanding of the exposure—response relationships involved in perturbations of toxicity pathways in exposed humans or wildlife. Closer interactions between toxicologists and epidemiologists to characterize the biologic effectiveness of exposures could provide better exposure metrics.
Environmental Regulation
Exposure science needs to play a greater role in the process of risk assessment, risk management, and compensation policy to improve characterization of population-wide exposure distributions, their uncertainties, aggregate and cumulative exposures, and high-risk populations. More powerful incentives for data generation—such as conservative default values—are needed to address critical data gaps. A more risk-preventive approach to introduction of new chemicals into the environment and better post-marketing surveillance of those already present is needed, particularly with the goal of reducing disparities in the population. An improved role of exposure science in the regulations will advance priority-setting, help measure the efficacy of regulations, and reduce the likelihood of widespread human or environmental exposure to substances that are likely to be hazardous.
Environmental Planning
Exposure science should be an increasingly integral component of environmental planning because of its ability to inform decisions during urban projects that may affect public health (for example, through exposure to increased air-pollutant emissions or potential increases in exposure to green space) and because of its ability to recognize the complex interactions among humans and ecosystems that are critical for protecting human and ecosystem health.
Disaster Management
A great challenge in the management of natural or human-made disasters is to anticipate the needs for exposure science before a disaster occurs. To that end, field-ready protocols need to be established and tested, including the necessary equipment (such as drones) and expert personnel, and the equipment needs to be pre-positioned for rapid deployment to conduct community and occupational monitoring. The availability of personal protective devices is an important element in these preparations. Biologic monitoring for surveillance of workers
and the community is essential. Ideally, it would be conducted routinely on first-responder personnel, in advance of a disaster, to measure toxicants associated with their jobs and thus provide a baseline measure. However, biomonitoring cannot take the place of environmental sensors for real-time measurement of hazardous agents.
Alberti, M., J. M. Marzluff, E. Shulenberger, G. Bradley, C. Ryan, and C. Zumbrunnen. 2003. Integrating humans into ecology: Opportunities and challenges for studying urban ecosystems. Bioscience 53(12):1169-1179.
Allan, B.F., H.P. Dutra, L.S. Goessling, K. Barnett, J.M. Chase, R.J. Marquis, G. Pang, G.A. Storch, R.E. Thach, and J.L. Orrock. 2010. Invasive honeysuckle eradication reduces tick-borne disease risk by altering host dynamics. Proc. Natl. Acad. Sci. USA 107(43):18523-18527.
Almanza, E., M. Jerrett, G. Dunton, E. Seto, and M. Pentz. 2011. Green Spaces in Healthy Places: Objective Data Demonstrates an Association between Greenness and Momentary Measures of Physical Activity in Children. Presentation at Active Living Research (ALR) Conference, February 22-24 2011, San Diego, CA [online]. Available: http://www.activelivingresearch.org/files/2011_GPS_Almanza.pdf [accessed Dec. 15, 2011].
Andersen, M.E., H.J. Clewell, III, E. Bermudez, D.E. Dodd, G.A. Wilson, J.L. Campbell, and R.S. Thomas. 2010. Formaldehyde: Integrating dosimetry, cytotoxicity, and genomics to understand dose-dependent transitions for an endogenous compound. Toxicol. Sci. 118(2):716-731.
Anderson, D.M., P.M. Glibert, and J.M. Burkholder. 2002. Harmful algal blooms and eutrophication: Nutrient sources, composition, and consequences. Estuaries 25(4b):704-726.
Barr, D.B., R.Y. Wang, and L.L. Needham. 2005. Biologic monitoring of exposure to environmental chemicals throughout the life stages: Requirements and issues for consideration for the National Children’s Study. Environ. Health Perspect. 13(8):1083-1091.
Bell, M.L., K. Ebisu, and R.D. Peng. 2011. Community-level spatial heterogeneity of chemical constituent levels of fine particulates and implications for epidemiological research. J. Expo. Sci. Environ. Epidemiol. 21(4):372-384.
Benson, P. 1989. Caline4 - A Dispersion Model for Predicting Air Pollution Concentration Near Roadways. Report No. FHWA/CA/TL-84/15. California Department of Transportation, Sacramento, CA [online]. Available: http://www.weblakes.com/products/calroads/resources/docs/CALINE4.pdf [accessed May 9, 2012].
Bhatia, R. 2007. Protecting health using an environmental impact assessment: A case study of San Francisco land use decision-making. Am. J. Public Health 97(3):406-413.
Bhatia, R., and T. Rivard. 2008. Assessment and Mitigation of Air Pollutant Health Effects from Intra-urban Roadways: Guidance for Land Use Planning and Environmental Review. San Francisco, CA: San Francisco Department of Public Health [online]. Available: http://www.sfphes.org/publications/Mitigating_Roadway_AQLU_Conflicts.pdf [accessed Dec. 14, 2011].
Bhatia, R., and E. Seto. 2011. Quantitative estimation in health impact assessment: Opportunities and challenges. Environ. Impact Assess. Rev. 31(3):301-309.
Bhatia, R., and A. Wernham. 2008. Integrating human health into environmental impact assessment: An unrealized opportunity for environmental health and justice. Environ. Health Perspect. 116(8):991-1000.
Bongers, S., N.A.H. Janssen, B. Reiss, L. Grievink, E. Lebret, and H. Kromhout. 2008. Challenges of exposure assessment for health studies in the aftermath of chemical incidents and disasters. J. Expo. Sci. Environ. Epidemiol. 18(4):341-359.
Bowling, A.M., C.R. Hammerschmidt, and J.T. Oris. 2011. Necrophagy by a benthic omnivore influences biomagnification of methylmercury in fish. Aquat. Toxicol. 102(3-4):134-141.
Bradley, C.A., and S. Altizer. 2006. Urbanization and the ecology of wildlife diseases. Trends Ecol. Evol. 22(2):95-102.
Bradman, A., and R.M. Whyatt. 2005. Characterizing exposures to nonpersistent pesticides during pregnancy and early childhood in the National Children’s Study: A review of monitoring and measurement methodologies. Environ. Health Perspect. 113(8):1092-1099.
Brecken-Folse, J.A., F.L. Mayer, L.E. Pedigo, and L.L. Marking. 1994. Acute toxicity of 4-nitrophenol, 2,4-dinitrophenol, terbufos and trichlorfon to grass shrimp (Palaemonetes Spp) and sheepshead minnows (Cyprinodon variegatus) as affected by salinity and temperature. Environ. Toxicol. Chem. 13(1):67-77.
Breslow, N.E., T. Lumley, C.M. Ballantyne, L.E. Chambless, and M. Kulich. 2009. Using the whole cohort in the analysis of case-cohort data. Am. J. Epidemiol. 169(11):1398-1405.
Brown, P., J.G. Brody, R. Morello-Frosch, J. Tovar, A.R. Zota, and R.A. Rudel. 2012. Measuring the success of community science: The Northern California Household Exposure Study. Environ. Health Perspect. 120(3):326-331.
Brussard, P.F, M.J. Reed, and T.C. Richard. 1998. Ecosystem management: What is it really? Landscape Urban Plan. 40:9-20.
Carroll, R.J., D. Ruppert, L.A. Stefanski, and C. Crainiceanu. 2006. Measurement Error in Nonlinear Models: A Modern Perspective, 2nd Ed. Boca Raton, FL: Chapman & Hall/CRC.
CDC (Centers for Disease Control and Prevention). 2011. Fourth National Report on Human Exposure to Environmental Chemicals. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, Atlanta, GA [online]. Available: http://www.cdc.gov/exposurereport/ [accessed Dec. 12, 2011].
CDPH (California Department of Public Health). 2010a. Public Drinking Water System [online]. Available: http://www.cdph.ca.gov/certlic/drinkingwater/Pages/default.aspx [accessed Jan. 3, 2012].
CDPH (California Department of Public Health). 2010b. Implementation of the California Environmental Contaminant Biomonitoring Program. Report to the California Legislature. California Department of Public Health in Collaboration with California Environmental Protection Agency’s Office of Health Hazard Assessment and Department of Toxic Substances Control, January 2010 [online]. Available: http://oehha.ca.gov/multimedia/biomon/pdf/CECBPLegReport.pdf [accessed Dec. 6, 2011].
Chandra, S., H. Segale, and S. Adler. 2009. Lake Tahoe Species Introduction Timeline. Tahoe Environmental Research Center [online]. Available: http://terc.ucdavis.
edu/education_outreach/educationprograms/TIC_InvasiveSpeciesTimeline.pdf [accessed 17 May, 2012].
Cherkasov, A.S., P.K. Biswas, D.M. Ridings, A.H. Ringwood, and I.M. Sokolova. 2006. Effects of acclimation temperature and cadmium exposure on cellular energy budgets in the marine mollusk Crassostrea virginica: Linking cellular and mitochondrial responses. J. Exp. Biol. 209(7):1274-1284.
Cherkasov, A.S., S. Grewal, and I.M. Sokolova. 2007. Combined effects of temperature and cadmium exposure on haemocyte apoptosis and cadmium accumulation in the eastern oyster Crassostrea virginica (Gmelin). J. Therm. Biol. 32(3):162-170.
Chow, J.C., J.P. Engelbrecht, N.C. Freeman, J.H. Hashim, M. Jantunen, J.P. Michaud, S. Saenz de Tejada, J.G. Watson, F. Wei, W.E. Wilson, M. Yasuno, and T. Zhu. 2002. Chapter one: Exposure measurements. Chemosphere 49(9):873-901.
Cloern, J.E. 2001. Our evolving conceptual model of the coastal eutrophication problem. Mar. Ecol. Prog. Ser. 210:223-253.
Cohen Hubal, E.A., A.M. Richard, I. Shah, J. Gallagher, R. Kavlock, J. Blancato, and S.W. Edwards. 2010. Exposure science and the U.S. EPA National Center for Computational Toxicology. J. Expo. Sci. Environ. Epidemiol. 20(3):231-236.
Cohen Hubal, E.A., D.B. Barr, H.M. Koch, and T. Bahadori. 2011. The promise of exposure science. J. Expo. Sci. Environ. Epidemiol. 21(2):121-122.
Dahmann, N., J. Wolch, P. Joassart-Marcelli, K. Reynolds, and M. Jerrett. 2009. The active city? Disparities in provision of urban public recreation resources. Health Place 16(3):431-445.
Dale, V., G.R. Biddinger, M.C. Newman, J.T. Oris, G.W. Suter, T. Thompson, T.M. Armitage, J.L. Meyer, R.M. Allen-King, G.A. Burton, P.M. Chapman, L.L. Conquest, I.J. Fernandez, W.G. Landis, L.L. Master, W.J. Mitsch, T.C. Mueller, C.F. Rabeni, A.D. Rodewald, J.G. Sanders, and I.L. van Heerden. 2008. Enhancing the ecological risk assessment process. Integr. Environ. Assess. Manag. 4(3):306-313.
Dean Runyan Associates. 2009. The Economic Significance of Travel the North Lake Tahoe Area: 2003-2008 Detailed Visitor Impact Estimates. Prepared for North Lake Tahoe Resort Association, Tahoe City, CA, by Dean Runyan Associates, Portland, OR [online]. Available: http://www.deanrunyan.com/doc_library/FinalReportCA.pdf [accessed May 21, 2012].
de Hartog, J.J., H. Boogaard, H. Nijland, and G. Hoek. 2010. Do the health benefits of cycling outweigh the risks? Environ. Health Perspect. 118(8):1109-1116.
de Nazelle, A., M.J. Nieuwenhuijsen, J.M. Anto, M. Brauer, D. Briggs, C. Braun-Fahrlander, N. Cavill, A.R. Cooper, H. Desqueyroux, S. Fruin, G. Hoek, L.I. Panis, N. Janssen, M. Jerrett, M. Joffe, Z. Jovanovic Andersen, E. van Kempen, S. Kingham, N. Kubesch;, K.M. Leyden, J.D. Marshall, J. Matamala, G. Mellios, M. Mendez, H. Nassif, D. Oglivie, R. Peiro, K. Perez, A. Rabl, M. Ragettli, D. Rodriguez, D. Rojas, P. Ruiz, J.F. Sallis, J. Terwoert, J.F. Toussaint, J. Tuomisto, M. Zuurbier, and E. Lebret. 2011. Improving health through policies that promote active travel: A review of evidence to support integrated health impact assessment. Environ. Int. 37(4):766-777.
Dix, D.J., K.A. Houck, M.T. Martin, A.M. Richard, R.W. Setzer, and R.J. Kavlock. 2007. The ToxCast program for prioritizing toxicity testing of environmental chemicals. Toxicol. Sci. 95(1):5-12.
Dobson, A., and J. Foufopoulos. 2001. Emerging infectious pathogens of wildlife. Philos. Trans. R. Soc. London B 356(1411):1001-1012.
Dominici, F., J.I. Levy, and T.A. Louis. 2005. Methodological challenges and contributions in disaster epidemiology. Epidemiol. Rev. 27(1):9-12.
Downs, T.J., Y. Ogneva-Himmelberger, O. Aupont, Y. Wang, A. Raj, P. Zimmerman, R. Goble, O. Taylor, L. Churchill, C. Lemay, T. McLaughlin, and M. Felice. 2010. Vulnerability-based spatial sampling stratification for the National Children’s Study, Worcester County, Massachusetts: Capturing health-relevant environmental and sociodemographic variability. Environ. Health Perspect. 118(9):1318-1325.
Drevnick, P.E., A. Shinneman, C. Lamborg, D.R. Engstrom, M. Bothner, and J.T. Oris. 2010. Mercury flux to sediments of Lake Tahoe, California-Nevada. Water Air Soil Pollut. 210 (1-4):399-407.
Dwyer, J.F., E.G. McPherson, H.W. Schroeder, and A. Rowntree. 1992. Assessing the benefits and costs of the urban forest. J. Arborculture 18(5):227-234.
Edelman, P., J. Osterloh, J. Pirkle, S.P. Caudill, J. Grainger, R. Jones, B. Blount, A. Calafat, W. Turner, D. Feldman, S. Baron, B. Bernard, D.B. Lushniak, K. Kelly, and D. Prezant. 2003. Biomonitoring of chemical exposure among New York City firefighters responding to the World Trade Center fire and collapse. Environ. Health Perspect. 111(16):1906-1911.
Eiswerth, M.E., S.G. Donaldson, and W.S. Johnson. 2000. Potential environmental impacts and economic damages of Eurasian watermilfoil (Myriophyllum spicatum) in western Nevada and northeastern California. Weed Technol. 14(3):511-518.
EPA (U.S. Environmental Protection Agency). 1992. Guidelines for Exposure Assessment. EPA600Z-92/001. Risk Assessment Forum, U.S. Environmental Protection Agency, Washington, DC.
EPA (U.S. Environmental Protection Agency). 2000. Guidance for Assessing Chemical Contaminant Data for Use in Fish Advisories. Volume 2: Risk Assessment and Fish Consumption Limits -Third Edition. EPA 823-B-00-008. Office of Science and Technology, Office of Water, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://water.epa.gov/scitech/swguidance/fishshellfish/techguidance/risk/upload/2009_04_23_fish_advice_volume2_v2cover.pdf [accessed May 9, 2012].
EPA (U.S. Environmental Protection Agency). 2002. Exposure and Human Health Evaluation of Airborne Pollution from the World Trade Center Disaster. External Review Draft. EPA/600/P-2/002A. National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://nycosh.org/uploads/listit/id21/WTC_FINAL5.pdf [accessed Jan. 4, 2012].
EPA (U.S. Environmental Protection Agency). 2003. EPA’s Response to the World Trade Center Collapse: Challenges, Successes, and Areas for Improvement. Report No. 2003-P-00012. Office of Inspector General, U.S. Environmental Protection Agency, Washington, DC. August 21, 2003 [online]. Available: http://www.epa.gov/oig/reports/2003/WTC_report_20030821.pdf [accessed Dec. 12, 2011].
EPASAB (U.S. Environmental Protection Agency Science Advisory Board). 2007. Advice to EPA on Advancing the Science and Application of Ecological Risk Assessment in Environmental Decision Making. EPA-SAB-08-002. Science Advisory Board, U.S. Environmental Protection Agency, Washington, DC [online]. Available: http://yosemite.epa.gov/sab/sabproduct.nsf/7140DC0E56EB148A8525737900043063/$File/sab-08-002.pdf [accessed Dec. 14, 2011].
Faeth, S.H., P.S. Warren, E. Shochat, and W.A. Marussich. 2005. Trophic dynamics in urban communities. Bioscience 55(5):399-407.
Filser, J., H. Koehler, A. Ruf, J. Rombke, A. Prinzing, and M. Schaefer. 2008. Ecological theory meets soil ecotoxicology: Challenge and chance. Basic Appl. Ecol. 9(4):346-355.
Fitzpatrick, K., and M. LaGory. 2011. Unhealthy Cities: Poverty, Race, and Place in America, 2nd Ed. New York: Routledge.
GAO (U.S. Government Accountability Office). 2007. World Trade Center: EPA’s Most Recent Test and Clean Program Raises Concerns That Need to Be Addressed to Better Prepare for Indoor Contamination Following Disasters. GAO 07-1091. Washington, DC: U.S. Government Accountability Office. September 2007 [online]. Available: http://www.gao.gov/new.items/d071091.pdf [accessed Dec. 12, 2011].
GAO (General Accountability Office). 2009. Chemical Regulation: Options for Enhancing the Effectiveness of the Toxic Substances Control Act. GAO-09-428T. Washington, DC: U.S. Government Accountability Office [online]. Available: http://www.gao.gov/new.items/d09428t.pdf [accessed May 9, 2011].
García-Berthou, E. 2002. Ontogenetic diet shifts and interrupted piscivory in introduced largemouth bass (Micropterus salmoides). Int. Rev. Hydrobiol. 87(4):353-363.
Gehrt, S.D. 2010. The urban ecosystem. Pp. 3-11 in Urban Carnivores: Ecology, Conflict, and Conservation, S.D. Gehrt, S.P.D. Riley, and B.L. Cypher, eds. Baltimore, MD: The Johns Hopkins University Press.
Georgopoulos, P.G., A.F. Sasso, S.S. Isukapalli, P.J. Lioy, D.A. Vallero, M. Okino, and L. Reiter. 2009. Reconstructing population exposures to environmental chemicals from biomarkers: Challenges and opportunities. J. Expo. Sci. Environ. Epidemiol. 19(2):149-171.
Gevertz, A.K., A.J. Tucker, A.M. Bowling, C.E. Williamson, and J.T. Oris. 2012. Differential tolerance of native and non-native fish exposed to ultraviolet radiation and fluoranthene in Lake Tahoe (CA/NV). Environ. Toxicol. Chem. 31(5): 1129-1135.
Gilliland, F.D., Y.F. Li, A. Saxon, and D. Diaz-Sanchez. 2004. Effect of glutathione-S-transferase M1 and P1 genotypes on xenobiotic enhancement of allergic responses: Randomised, placebo-controlled crossover study. Lancet 363(9403): 119-125.
Gilliland, F., E. Avol, P. Kinney, M. Jerrett, T. Dvonch, F. Lurmann, T. Buckley, P. Breysse, G. Keeler, T. de Villiers, and R. McConnell. 2005. Air pollution exposure assessment for epidemiologic studies of pregnant women and children: Lessons learned from the Centers for Children’s Environmental Health and Disease Prevention Research. Environ. Health Perspect. 113(10):1447-1454.
Goldman, C.R. 2000. Four decades of change in two subalpine lakes. Verh. Internat. Verein. Limnol. 27(Pt. 1):7-26.
Greenland, S., and J.M. Robins. 2000. Epidemiology, justice, and the probability of causation. Jurimetrics 40:321-340.
Grumbine, R.E. 1994. What is ecosystem management? Conserv. Biol. 8(1):27-38.
Gupta, P.K., B.S. Khangarot, and V.S. Durve. 1981. The temperature-dependence of the acute toxicity of copper to a fresh-water pond snail, Viviparus bengalensis L. Hydrobiologia 83(3):461-464.
Hammerschmidt, C., and W. Fitzgerald. 2006. Bioaccumulation and trophic transfer of methylmercury in the Long Island Sound. Arch. Environ. Contam. Toxicol. 51(3):416-424.
Hankey, S., J.D. Marshall, and M. Brauer. 2012. Health impacts of the built environment: Within-urban variability in physical inactivity, air pollution, and ischemic heart disease mortality. Environ. Health Perspect. 120(2):247-253.
HEI (Health Effects Institute). 2010. Traffic-Related Air Pollution: A Critical Review of the Literature on Emissions, Exposure, and Health Effects. Special Report 17. Boston, MA: Health Effects Institute [online]. Available: http://pubs.healtheffects.org/view.php?id=334 [accessed Dec. 12, 2011].
Hirschfeld, S., D. Songco, B.S. Kramer, and A.E. Guttmacher. 2011. National Children’s Study: Update in 2010. Mt Sinai J. Med. 78(1):119-125.
Hoffman, F.O., A.J. Ruttenber, A.I. Apostoaei, R.J. Carroll, and S. Greenland. 2007. The Hanford Thyroid Disease Study: An alternative view of the findings. Health Phys. 92(2):99-111.
IOM (Institute of Medicine). 2000. Protecting Those Who Serve: Strategies to Protect the Health of Deployed U.S. Forces. Washington, DC: National Academy Press.
IOM (Institute of Medicine). 2008. Improving the Presumptive Disability Decision-Making Process for Veterans, J.M. Samet, and C.C. Bodurow, eds. Washington, DC: National Academies Press.
Jassby, A.D., J.E. Reuter, and C.R. Goldman. 1996. Determining long-term water quality in the presence of climate variability: Lake Tahoe (USA). Can. J. Fish. Sci. 60(12):1452-1461.
Jerrett, M., R.T. Burnett, R. Ma, C.A. Pope, D. Krewski, K.B. Newbold, G. Thurston, Y. Shi, N. Finkelstein, E.E. Calle, and M.J. Thun. 2005. Spatial analysis of air pollution and mortality in Los Angeles. Epidemiology 16(6):727-736.
Jerrett, M., J. Su, J. Apte, and B. Beckerman. 2011. Estimates of population exposure to traffic-related air pollution in Beijing, China and New Delhi, India: Extending exposure analyses report in HEI Special Report 17, Traffic-Related Air Pollution: Critical Review of the Literature on Emissions, Exposure, and Health Effects, November 5, 2010. Boston, MA: Health Effects Institute [online]. Available: http://www.healtheffects.org/International/Jerrett_Asia_Traffic_Exposure.pdf [accessed Dec. 10, 2011].
Kamerath, M., S. Chandra, and B.C. Allen. 2008. Distribution and impacts of war water invasive fish in Lake Tahoe, USA. Aquat. Invasions 3(1):35-41.
Kearney, A.R., G.A. Bradley, C.H. Petrich, R. Kaplan, S. Kaplan, and D. Simpson-Colbank. 2008. Public perception as support for scenic quality regulation in a nationally treasured landscape. Landscape Urban Plan. 87(2):117-128.
Kemp, W.M., W.R. Boynton, J.E. Adolf, D.F. Boesch, W.C. Boicourt, G. Brush, J.C. Cornwell, T.R. Fisher, P.M. Glibert, J.D. Hagy, L.W. Harding, E.D. Houde, D.G. Kimmel, W.D. Miller, R.I.E. Newell, M.R. Roman, E.M. Smith, and J.C. Stevenson. 2005. Eutrophication of Chesapeake Bay: Historical trends and ecological interactions. Mar. Ecol. Prog. Ser. 303:1-29.
Khan, M.A., S.A. Ahmed, B. Catalin, A. Khodadoust, O. Ajayi, and M. Vaughn. 2006. Effect of temperature on heavy metal toxicity to juvenile crayfish, Orconectes immunis (Hagen). Environ. Toxicol. 21(5):513-520.
Kopecky, K.J., S. Davis, T.E. Hamilton, M.S. Saporito, and L.E. Onstad. 2004. Estimation of thyroid radiation doses for the Hanford thyroid disease study: Results and implications for statistical power of the epidemiological analyses. Health Phys. 87(1):15-32.
Kopecky, K.J., L. Onstad, T.E. Hamilton, and S. Davis. 2005. Thyroid ultrasound abnormalities in persons exposed during childhood to 131I from the Hanford nuclear site. Thyroid 15(6):604-613.
Künzli, N., R. McConnell, D. Bates, T. Bastain, A. Hricko, F. Lurmann, E. Avol, F. Gilliland, and J. Peters. 2003. Breathless in Los Angeles: The exhausting search for clean air. Am. J. Public Health 93(9):1494-1499.
Lahr, J., and L. Kooistra. 2010. Environmental risk mapping of pollutants: State of the art and communication aspects. Sci. Total Environ. 408(18):3899-3907.
Landrigan, P.J., L. Trasande, L.E. Thorpe, C. Gwynn, P.J. Lioy, M.E. D’Alton, H.S. Lipkind, J. Swanson, P.D. Wadhwa, E.B. Clark, V.A. Rauh, F.P. Perera, and E. Susser. 2006. The National Children’s Study: A 21-year prospective study of 100,000 American children. Pediatrics 118(5):2173-2186.
Lioy, P.J. 2010a. Dust: The Inside Story of its Role in the Septemer 11th Aftermath. Lanham, MD: Rowman & Littlefield Publishers.
Lioy, P.J. 2010b. Exposure science: A view of the past and milestones for the future. Environ Health Perspect. 118(8):1081-1090.
Lioy, P.J., and M. Gochfeld. 2002. Lessons learned on environmental, occupational, and residential exposures from the attack on the World Trade Center. Am. J. Ind. Med. 42(6):560-565.
Lioy, P.J., S.S. Isukapalli, L. Trasande, L. Thorpe, M. Dellarco, C. Weisel, P.G. Georgopoulos, C. Yung, M. Brown, S. Alimokhtari, and P.J. Landrigan. 2009. Using national and local extant data to characterize environmental exposures in the National Children’s Study: Queens County, New York. Environ Health Perspect. 117(10):1494-1504.
Liu, Y., C.J. Paciorek, and P. Koutrakis. 2009. Estimating regional spatial and temporal variability of PM(2.5) concentrations using satellite data, meteorology, and land use information. Environ. Health Perspect. 117(6):886-892.
Logue, J.M., M.J. Small, D. Stern, J. Maranche, and A.L. Robinson. 2010. Spatial variation in ambient air toxics concentrations and health risks between industrial-influenced, urban, and rural sites. J. Air Waste Manag. Assoc. 60(3): 271-286.
Lorber, M., H. Gibb, L. Grant, J. Pinto, J. Pleil, and D. Cleverly. 2007. Assessment of inhalation exposures and potential health risks to the general population that resulted from the collapse of the World Trade Center towers. Risk Anal. 27(5):1203-1221.
Maas, J., R.A. Verheij, P.P. Groenewegen, S. de Vries, and P. Spreeuwenberg. 2006. Green space, urbanity, and health: How strong is the relation? J. Epidemiol. Community Health 60(7):587-592.
Maller, C., M. Townsend, A. Pryor, P. Brown, and L. St Leger. 2006. Healthy nature healthy people: ‘Contact with nature’ as an upstream health promotion intervention for populations. Health Promot. Int. 21(1):45-54.
Marentette, J.R., K.L. Gooderham, M.E. McMaster, T. Ng, J.L. Parrott, J.Y. Wilson, C.M. Wood, and M. Balshine. 2010. Signatures of contamination in invasive round gobies (Neogobius melanostomus): A double strike for ecosystem health? Ecotoxicol. Environ. Saf. 73(7):1755-1764.
Marshall, J.D., R.D. Wilson, K.L. Meyer, S.K. Rajangam, N.C. McDonald, and E.J. Wilson. 2010. Vehicle emissions during children’s school commuting: Impacts of education policy. Environ. Sci. Technol. 44(5):1537-1543.
McPherson, E.G., D. Nowak, G. Heisler, S. Grimmond, C. Souch, R. Grant, and R. Rowntree. 1997. Quantifying urban forest structure, function, and value: The Chicago Urban Forest Climate Project. Urban Ecosystems 1(1):49-61.
Meironyte, D., K. Noren, and A. Bergman. 1999. Analysis of polybrominated diphenyl ethers in Swedish human milk. A time-related trend study, 1972-1997. J. Toxicol. Environ. Health 58(6):329-341.
Melia, S., G. Parkhurst, and H. Barton. 2011. The paradox of intensification. Transport. Policy 18(1):46-52.
Miller, K.A., D.S. Siscovick, L. Sheppard, K. Shepherd, J.H. Sullivan, G.L. Anderson, and J.D. Kaufman. 2007. Long-term exposure to air pollution and incidence of cardiovascular events in women. N. Engl. J. Med. 356(5):447-458.
Minckley, W.L., and J.E. Craddock. 1961. Active predation of crayfish on fishes. Prog. Fish Cult. 23(3):120-123.
Mitchell, R., and F. Popham. 2008. Effect of exposure to natural environment on health inequalities: An observational population study. Lancet 372(9650):1655-1660.
Momot, W.T., H. Gowing, and P.D. Jones. 1978. The dynamics of crayfish and their role in ecosystems. Am. Midl. Nat. 99(1):10-35.
NCRP (National Council on Radiation Protection and Measurements). 2008. Uncertainties in the Measurement and Dosimetry of External Radiation. Report No. 158. Bethesda, MD: National Council on Radiation Protection and Measurements.
NCRP (National Council on Radiation Protection and Measurements). 2010a. Radiation Dose Reconstruction: Principles and Practices. Report No. 163. Bethesda, MD: National Council on Radiation Protection and Measurements.
NCRP (National Council on Radiation Protection and Measurements). 2010b. Uncertainties in Internal Radiation Dose Assessment. Report No. 164. Bethesda, MD: National Council on Radiation Protection and Measurements.
Needham, L.L., H. Ozkaynak, R.M. Whyatt, D.B. Barr, R.Y. Wang, L. Naeher, G. Akland, T. Bahadori, A. Bradman, R. Fortmann, L.J. Liu, M. Morandi, M.K. O’Rourke, K. Thomas, J. Quackenboss, P.B. Ryan, and V. Zartarian. 2005. Exposure assessment in the National Children’s Study: Introduction. Environ. Health Perspect. 113(8):1076-1082.
NIH (National Institutes of Health). 1985. Report of the National Institutes of Health Ad Hoc Working Group to Develop Radioepidemiologic Tables. NIH Publication No. 85-2748. U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health, Washington, DC [online]. Available: http://www.cdc.gov/niosh/ocas/pdfs/42cfr81/71.pdf [accessed Dec. 13, 2011].
NIOSH (National Institute for Occupational Safety and Health). 2011. Interactive RadioEpidemiological Program. NIOSH-IREP v.5.6 [online]. Available: https://www.niosh-irep.com/irep_niosh [accessed Oct. 20, 2011].
NRC (National Research Council). 1983. Risk Assessment in the Federal Government: Managing the Process. Washington, DC: National Academy Press.
NRC (National Research Council). 1984. Assigned Share for Radiation as a Cause of Cancer: Review of Radioepidemiological Tables Assigning Probabilities of Causation: Final Report. Washington, DC: National Academy Press.
NRC (National Research Council). 1994. Science and Judgment in Risk Assessment. Washington, DC: National Academy Press.
NRC (National Research Council). 1999. Strategies to Protect the Health of Deployed U.S. Forces: Force Protection and Decontamination. Washington, DC: National Academy Press.
NRC (National Research Council). 2000a. Strategies to Protect the Health of Deployed U.S. Forces: Analytical Framework for Assessing Risks. Washington, DC: National Academy Press.
NRC (National Research Council). 2000b. Strategies to Protect the Health of Deployed U.S. Forces: Detecting, Characterizing and Documenting Exposures. Washington, DC: National Academy Press.
NRC (National Research Council). 2007. Toxicity Testing in the 21st Century: A Vision and A Strategy. Washington, DC: National Academies Press.
NRC (National Research Council). 2009. Science and Decisions: Advancing Risk Assessment. Washington, DC: National Academies Press.
NRC (National Research Council). 2011. Improving Health in the United States: The Role of Health Impact Assessment. Washington, DC: National Academies Press.
NRC/IOM (National Research Council and Institute of Medicine). 2008. The National Children’s Study Research Plan: A Review. Washington, DC: National Academies Press.
O’Fallon, L.R., and A. Dearry. 2002. Community-based participatory research as a tool to advance environmental health sciences. Environ. Health Perspect. 110(suppl. 2):155-159.
Oris, J.T., S. Guttman, A.J. Bailer, J. Reuter, and G. Miller. 2004. Multi-Level Indicators of Ecosystem Integrity in Alpine Lakes of the Sierra Nevada (EPA No. R827643). U.S. EPA Final Project Report, Ecological Indicators Program, 99-NCERQA-E1[online]. Available: http://zoology.muohio.edu/oris/tahoe/SierraFinalReport.htm [accessed May 21, 2012].
Osterauer, R., and H. Koehler. 2008. Temperature-dependent effects of the pesticides thiacloprid and diazinon on the embryonic development of zebrafish (Danio rerio). Aquat. Toxicol. 86(4):485-494.
Özkaynak, H., R.M. Whyatt, L.L. Needham, G. Akland, and J. Quackenboss. 2005. Exposure assessment implications for the design and implementation of the National Children’s Study. Environ. Health Perspect. 113(8):1108-1115.
Paerl, H.W. 1997. Coastal eutrophication and harmful algal blooms: Importance of atmospheric deposition and groundwater as “new” nitrogen and other nutrient sources. Limnol. Oceanogr. 42(5):1154-1165.
Patel, C.J., and A.J. Butte. 2010. Predicting environmental chemical factors associated with disease-related gene expression data. BMC Med Genomics. 3:17.
Patel, C.J., J. Bhattacharya, and A.J. Butte. 2010. An environment-wide association study (EWAS) on Type 2 Diabetes Mellitus. PLoS ONE 5(5):e10746.
Pickett, S.T.A., M.L. Cadenasso, J.M. Grove, C.H. Nilon, R.V. Pouyat, W.C. Zipperer, and R. Constanza. 2001. Urban ecological systems: Linking terrestrial ecological, physical, and socioeconomic components of metropolitan areas. Annu. Rev. Ecol. Syst. 32:127-157.
PLOTS (Public Laboratory for Open Source Technology). 2012. About PLOTS [online]. Available: http://publiclaboratory.org/about [accessed March 29, 2012].
Rodes, C.E., E.D. Pellizzari, M.J. Dellarco, M.D. Erickson, D.A. Vallero, D.B. Reissman, P.J. Lioy, M. Lippmann, T.A. Burke, and B.D. Goldstein. 2008. ISEA2007 panel: Integration of better exposure characterizations into disaster preparedness for responders and the public. J. Expo. Sci. Environ. Epidemiol. 18(6):541-550.
Rose, K.C., C.E. Williamson, S.G. Schladow, M. Winder, and J.T. Oris. 2009. Patterns of spatial and temporal variability of UV transparency in Lake Tahoe, California-Nevada. J. Geophys. Res. Biogeosci. 114:G00D03.
Schecter, A., M. Pavuk, O. Päpke, J.J. Ryan, L. Birnbaum, and R. Rosen. 2003. Polybrominated diphenyl ethers (PBDEs) in U.S. mother’s milk. Environ. Health Perspect. 111(14):1723-1729.
Shankardass, K., R. McConnell, M. Jerrett, J. Milam, J. Richardson, and K. Berhane. 2009. Parental stress increases the effect of traffic-related air pollution on childhood asthma incidence. Proc. Natl. Acad. Sci. USA 106(30):12406-12411.
Sheppard, L., R.L. Prentice, and M.A. Rossing. 1996. Design considerations for estimation of exposure effects on disease risk, using aggregate data studies. Stat. Med. 15(17-18):1849-1858.
Shipler, D.B. 1995. Science in a fishbowl: Public involvement in the Hanford Environmental Dose Reconstruction Project. Fedl. Fac. Environ. J. 6(3):97-108.
Shipler, D.B., B.A. Napier, W.T. Farris, and M.D. Freshley. 1996. Hanford Environmental Dose Reconstruction Project—an overview. Health Phys. 71(4):532-544.
Slikker, W., Jr., M.E. Andersen, M.S. Bogdanffy, J.S. Bus, S.D. Cohen, R.B. Conolly, R.M. David, N.G. Doerrer, D.C. Dorman, D.W. Gaylor, D. Hattis, J.M. Rogers, R. Woodrow Setzer, J.A. Swenberg, and K. Wallace. 2004. Dose-dependent transitions in mechanisms of toxicity. Toxicol. Appl. Pharmacol. 201(3):203-225.
Strauss, W.J., L. Ryan, M. Morara, N. Iroz-Elardo, M. Davis, M. Cupp, M.G. Nishioka, J. Quackenboss, W. Galke, H. Ozkaynak, and P. Scheidt. 2010. Improving cost-effectiveness of epidemiological studies via designed missingness strategies. Stat Med. 29(13):1377-1387.
Su, J.G., M. Jerrett, A. deNazelle, and J. Wolch. 2011. Does exposure to air pollution in urban park shave socioeconomic, racial or ethnic gradients? Environ. Res. 11(3):319-328.
Swift, T.J., J. Perez-Losada, S.G. Schladow, J.E. Reuter, A.D. Jassby, and C.R. Goldman. 2006. A mechanistic clarity model of lake waters: Linking suspended matter characteristics to clarity. Aquat. Sci. 68:1-15.
Szaro, R.C., W.T. Sexton, and C.R. Malone. 1998. The emergence of ecosystem management as a tool for meeting people’s needs and sustaining ecosystems. Landscape Urban Plan. 40(1-3):1-7.
Szpiro, A.A., C.J. Paciorek, and L. Sheppard. 2011. Does more accurate exposure prediction necessarily improve health effect estimates? Epidemiology 22(5):680-685.
Teeguarden, J.G., A.M. Calafat, X. Ye, D.R. Doerge, M.I. Churchwell, R. Gunawan, and M.K. Graham. 2011. Twenty-four hour human urine and serum profiles of bisphenol A during high-dietary exposure. Toxicol. Sci. 123(1):48-57.
Thomas, D.C. 1988. Models for exposure-time-response relationships with applications to cancer epidemiology. Annu. Rev. Publ. Health 9:451-482.
Thomas, D. 2010. Gene-environment-wide association studies: Emerging approaches. Nat. Rev. Genet. 11(4):259-272.
Tucker, A.J., C.E. Williamson, K.C. Rose, J.T. Oris, S. Connelly, M.H. Olson, and D.L. Mitchell. 2010. Ultraviolet radiation affects invasibility of lake ecosystems by warmwater fish. Ecology 91(3): 882-890.
Tucker, A.J., C.E. Williamson, and J.T. Oris. in press. Development and application of a UV attainment threshold for the prevention of warmwater aquatic invasive species. Biol. Invasions in press.
Tulve, N.S., L.S. Sheldon, and R.C. Fortmann. 2010. Workshop on Optimizing Exposure Metrics for the National Children’s Study: Summary of Workgroup Discussions and Recommendations. EPA/600/R-10/064. Office of Research and Development, U.S. Environmental Protection Agency, Research Triangle Park, NC. June 2010 [online]. Available: http://oaspub.epa.gov/eims/eimscomm.getfile?p_download_id=497487 [accessed Jan. 2, 2012].
Wang, R. Y., L.L. Needham, and D.B. Barr. 2005. Effects of environmental agents on the attainment of puberty: Considerations when assessing exposure to environmental chemicals in the National Children’s Study. Environ. Health Perspect. 113(8):1100-1107.
Wiener, J.G., D.P. Krabbenhoft, G.H. Heinz, and A.M. Scheuhammer. 2002. Ecotoxicology of mercury. Pp. 409-463 in Handbook of Ecotoxicology, 2nd Ed., D.J. Hoffman, B.A. Rattner, G.A. Burton, and J. Cairns, eds. Boca Raton: Lewis.
Wier, M., J. Weintraub, E. Humphreys, E. Seto, and R. Bhatia. 2009a. An area-level model of vehicle-pedestrian injury collisions with implications for land use and transportation planning. Accid. Anal. Prev. 41(1):137-145.
Wier, M., C. Sciammas, E. Seto, R. Bhatia, and T. Rivard. 2009b. Health, traffic, and environmental justice: Collaborative research and community action in San Francisco, California. Am. J. Public Health 99(suppl. 3):S499-S504.
Wolch, J., M. Jerrett, K. Reynolds, R. McConnell, R. Chang, N. Dahmann, K. Brady, F. Gilliland, J.G. Su, and K. Berhane. 2011. Childhood obesity and proximity to urban parks and recreational resources: A longitudinal cohort study. Health Place 17(1):207-214.
Zablotska, L.B., J.P. Ashmore, and G.R. Howe. 2004. Analysis of mortality among Canadian nuclear power industry workers after chronic low-dose exposure to ionizing radiation. Radiat. Res. 161(6):633-641.








































