Implications of Contamination Remaining in Place
Despite the ability of some remedial technologies to remove substantial amounts of mass, at most complex sites contamination will remain in place at levels above those allowing for unlimited use and unrestricted exposure (see Chapter 4). This chapter discusses the potential technical, legal, economic, and other practical implications of this finding.
First, contamination from these sources must be contained on-site, by using either hydraulic or physical containment systems combined with institutional controls. Indeed, 65 percent of source control RODs from FY 1998–2008 included containment, and institutional controls are used at the vast majority of CERCLA source control remedial actions to enhance and ensure their effectiveness and protectiveness (EPA, 2010a). Because the failure of these systems could create new exposures, potentially responsible parties (PRPs) should weigh the robustness and potential for failure during remedy selection and implementation. Second, our understanding of the risk posed by contaminated groundwater is inherently dynamic. For example, toxicity information is regularly updated, and contaminants that were previously unregulated may become so, changing the drivers for risk assessment and cleanup decisions. In addition, pathways of exposure that were not previously under consideration can be found to be important, such as has happened with the vapor intrusion pathway over the past decade. Consideration of these new factors can change the overall protectiveness of a remedy that leaves contamination in place. Third, residual contamination necessarily reduces the amount of groundwater available for unrestricted use. Treating groundwater for drinking water purposes is very costly and, for some contaminants (e.g., 1,4-dioxane), technically challenging. Finally,
leaving contamination in the subsurface may expose the landowner, property manager, or original disposer to complications that would not exist in the absence of the contamination. PRPs may be sued for natural resource damages by the resource trustee (if the underlying groundwater is no longer potable without treatment because of remaining contamination) or for personal injury and/or property damages pursuant to common law by local residents or others (if the contamination crosses property boundaries and causes injury or property damage).
At any given site, the risks and the technical, economic, and legal complications associated with residual contamination need to be compared to the time, cost, and feasibility involved in removing contamination outright. As a practical matter, the Committee did not seek to estimate the relative scope of the nontechnical impacts of leaving contamination in place, and it is probably not feasible to do so. Whether these potential consequences are likely to occur is site specific, and some implications may not materialize at some sites.
POTENTIAL FOR FAILURE OF REMEDIES AND ENGINEERED CONTROLS
The long-term management strategies for many complex sites include leaving significant amounts of contamination in place. At such sites the achievement of risk-based goals is based on a reduction of the contaminant flux (e.g., reduction in source strength) between the zone of residual contamination and the point(s) of compliance. Such flux reduction is generally accomplished by one of four approaches, possibly coupled with partial removal of source zone contamination: (1) hydraulic containment, (2) physical containment, (3) reduction of contaminant concentrations through natural processes (monitored natural attenuation), and/or (4) reduction of contaminant concentrations through an engineered reaction zone, most commonly in the form of a downgradient permeable reactive barrier (PRB) (see Chapter 4 for descriptions of these technologies). This section summarizes key concepts and tools for assessing the likelihood and consequences of failure for these approaches.
Each of the remedial strategies listed above is well established and is unlikely to exhibit “complete” failure in any meaningful sense. Rather, some degree of contaminant flux reduction is likely to be realized, even if the overall magnitude and/or spatial extent of the reduction is less than expected from design calculations. The consequences of such “partial failure” would depend both on the measures used to monitor performance and the corrective actions that are triggered by inadequate performance. There are few reports in the peer-reviewed literature that document both the failure of a long-term remedial strategy and the resulting response (although these
issues should be addressed in the five-year review process for sites regulated under CERCLA). This lack of focused literature on the failure of remediation systems designed for long-term management may be due, in part, to the likelihood that system failure would generate incremental, rather than sharp, increases in operation costs, as discussed below for each of the four strategies.
Hydraulic Containment
Pump and treat (P&T) has increasingly been implemented as a long-term management strategy, with the primary goal of hydraulic containment to prevent further spreading of contamination. In a general sense, “failure” of hydraulic containment occurs when groundwater that originates from within the target capture zone is not completely captured by extraction wells, but instead is allowed to migrate downgradient beyond property boundaries and toward a receptor. Such failure could occur as a direct consequence of inadequate well placement and/or underspecified pumping rate(s) due to a misunderstanding of the governing hydrogeology (e.g., an incorrect or incomplete groundwater model). Even for a properly designed extraction system, containment failure could occur after startup because of temporal changes in hydrologic conditions such as recharge or regional flow conditions.
To assist with identifying potential P&T failure, the U.S. Environmental Protection Agency (EPA) has recently developed a six-step procedure for evaluating the hydraulic containment of target capture zones, with an emphasis on comparing measured water levels and concentrations against model predictions (EPA, 2008a). While establishing a formal comparison between measured and predicted capture zones still requires considerable site-specific judgment, the availability of established guidance (and an ongoing process to refine it and expand its applicability) is an important development.
As discussed in Chapter 4, EPA has applied the Remedial System Evaluation (RSE) process to more than 60 operating P&T systems at Superfund facilities. At many of these, field observations were unable to establish the success of hydraulic containment at the desired level of confidence. In some cases, additional monitoring was recommended to clarify the evaluation. However, for other sites, adjustment to the locations and/or operation of extraction wells was recommended. Although such midcourse corrections typically increase the cost of P&T system operation, pumping rates, monitoring programs, or even extraction wells can also be reduced if the system is overdesigned for current conditions. In general, actions to improve P&T performance are straightforward to implement and normally generate an incremental, rather than drastic change in the life-cycle cost of site manage-
ment. In this regard, hydraulic containment may be regarded as an adaptive strategy that can readily be updated in response to new information about the site.
Physical Containment
Barriers are frequently used to influence groundwater flow in combined remedies that also use extraction wells and/or engineered reaction zones. From a containment standpoint, the overall remediation goal is similar to hydraulic containment: maintain control of groundwater within a target capture zone. Thus, similar monitoring and analytical approaches might be used to assess performance. Failures in physical containment may occur due to incorrect design or construction of barriers, poor seals between sections (in the case of sheet pile barriers or geomembranes), holes/defects in materials, physical or chemical damage, poor connection between a vertical barrier and underlying confining bed, and lack of control of recharge inside the contained area. These and similar expressions of “failure” are likely to occur locally at small defects in vertical walls, rather than across the full extent of the barrier system. Because the flow influence of a barrier irregularity will likely be distributed across a large area, detecting such local failure through routine groundwater monitoring is likely to be difficult.
A recent NRC review concluded, in part, that available field data are insufficient to provide a robust assessment of the potential for or actual occurrence of failure in vertical barriers (NRC, 2007), particularly over long decision horizons. However, reports from site-specific remedial system evaluations and CERCLA five-year reviews have identified instances where hydraulic monitoring indicated that physical containment systems may have “failed” (e.g., EPA, 1999; Northgate Environmental Management, 2008), although specific mechanisms are typically not identified. Even if the precise location of a barrier defect could be identified through field monitoring, effective measures for the direct repair of a flawed or cracked vertical barrier have not been developed. Instead, adjustments to other aspects of the remedial system would likely be needed. For both of the above CERCLA examples, the vertical barriers functioned as components of combined remedies that also included extraction wells, which resulted in straightforward adjustments to system operation that maintained a high degree of confidence in successful hydraulic containment.
In additional to the possibility of hydraulic failure, earthen barriers can also release contaminants by molecular diffusion. Because chemicals in most barrier materials have diffusion coefficients that are similar to those in aquifer material, and the diffusion path length is relatively short (typically one meter or less), the time for a solute to diffuse across the barrier could be relatively short, on the order of years rather than decades (e.g., Mott and
Weber, 1991; Khandelwal et al., 1998; Krol and Rowe, 2004). Although the potential for diffusion across slurry walls has been long recognized by scholars, field studies to assess this scenario have not been performed. However, even if elevated contaminant concentrations are present in the immediate vicinity of a vertical barrier, diffusive contaminant fluxes are typically several orders of magnitude less than advective fluxes, and it is plausible that molecular diffusion would constitute a significant concern at only a very small number sites (e.g., sites with both large concentrations within a containment zone and a receptor located in close proximity to a vertical barrier).
Permeable Reactive Barriers
To function successfully, a permeable reactive barrier (PRB) must provide hydraulic control of the upgradient target capture zone, such that all contaminated water flows through the PRB rather than around or below it. In addition the PRB must have sufficient reaction capacity to sustain the necessary reduction in contaminant concentrations over the appropriate design time frame. Failure to achieve either or both objectives can occur because of inadequate design (e.g., improper wall placement or reaction zone thickness) or because of changes within the PRB that occur over time (loss of permeability and/or reactivity). In addition, if a PRB was placed downgradient of a source zone but within a region that previously contained dissolved contamination, it is possible that measurable downgradient concentrations will persist due to back-diffusion, even if the PRB is functioning as designed (Sale and Newell, 2010).
The vast majority of installed PRBs are constructed of zero-valent iron, which produces redox conditions and results in pH changes that are likely to promote precipitation of groundwater minerals. This phenomenon has long been recognized as a potential problem, and numerous laboratory and modeling studies have explored the potential consequences of these processes for PRB longevity (e.g., Yabusaki et al., 2001; Kohn et al., 2005; Johnson et al., 2008; Wilkin and Puls, 2003; Sass et al., 2002; Phillips et al., 2010). However, as noted by ITRC (2005a, 2011), no PRB has “failed” due to loss of permeability or reactivity. In the most detailed published evaluation of iron-based PRB performance (Henderson and Demond, 2007), a handful of active PRB projects reported situations where improper design (insufficient depth or width) resulted in incomplete hydraulic capture. Of the 40 projects, only three exhibited post-installation performance degradation involving the loss of permeability due to precipitation and/or deceased reactivity.
As with low-permeability barrier systems, the failure of a PRB system is likely to occur locally rather than across the entire plane of interest, and it is
plausible that repair, rather than replacement, could be the appropriate response action. At the time of this writing, reports where installed PRBs were repaired or replaced were not located in the literature. As with the other long-term management strategies, the operating history of PRB technology is simply too short to support a robust assessment of the potential long-term management costs. However, concerns related to back-diffusion could potentially limit the application of PRB systems to sites where substantial contamination is not initially present downgradient of the installed PRB.
Monitored Natural Attenuation
Monitored natural attenuation (MNA) is most often used in conjunction with other active or engineering remedial components and is seldom employed as a stand-alone measure (EPA, 2010a). The success of natural attenuation as a remedy depends on the site-specific ability to predict the evolution of complex biogeochemical processes over an extended period of time. Because of uncertainties in long-term predictions, natural attenuation requires confirmatory monitoring, such that MNA remedies are accompanied by a detailed program of monitoring (e.g., NRC, 2000; EPA, 2004a).
Numerous protocols exist for evaluating MNA performance including a recently proposed decision framework for evaluating MNA for inorganic or radionuclide contamination (e.g., ITRC, 2010). Although focused on inorganic contaminants, the ITRC protocol contains many elements appropriate for sites with organic contaminants. In particular, the need for a contingency plan was emphasized, which provides a cleanup approach that will be implemented if “the selected remedy fails to perform as anticipated” (EPA, 2007). For MNA remedies, a suitable contingency plan might include optimization of source or plume treatments, implementation of an enhanced attenuation (EA) technology, pursuit of a technical impracticability waiver, or the use of institutional controls.
MNA systems could fail for many reasons, including temporal changes in site-specific hydrologic or geochemical conditions, the depletion of natural sources of nutrients or electron acceptors/donors, and lower-than-anticipated transformation rates. Further, the regulators may believe that there is insufficient evidence that MNA is occurring in the intended fashion. For example, it may be difficult to verify that the presence of daughter products is due to parent compound degradation and not co-contamination.
It is difficult to generalize regarding the potential cost of MNA failure, which will depend on site-specific conditions, the nature of the contingency actions, and the degree of conservatism built into the monitoring program. A properly designed monitoring program should provide “early detection” that allows for the implementation of a contingency plan prior to the point when a migrating plume would present elevated risks to receptors.
However, if contaminant migration and/or plume expansion occurs prior to the detection of failure, additional costs may be incurred. In certain circumstances, the combined cost of failed MNA and implementation of an additional remedy may exceed the cost that would have accrued had the remedy originally been put in place instead of MNA. To avoid such occurrences, the monitoring program should be directed at providing confirmation of the assumptions used to extrapolate the performance of MNA, in an adaptive management mode.
***
Common to all the remedies discussed above are unplanned and catastrophic events that may lead to failure of the proposed containment/treatment techniques, potentially for long periods of time. For example, natural disasters (e.g., earthquakes, floods, or other events) could cause changes in local hydrology, damage the remediation/containment system, or cause a loss of power to an active containment process. Flooding or other events could spread contamination to new areas and/or create new exposure pathways (e.g., vapor intrusion). Because contaminant migration from source zones or the plume is often slow, none of these events is likely to lead to catastrophic failure of the remedial system, but such events could lead to contaminant releases from the target capture zone if the failure is not identified and remedied. In summary, at sites where contamination remains in place, an evaluation of potential events that could lead to a failure of the long-term management approach should be performed and contingency plans developed.
IMPLICATIONS OF THE LONG-TERM NEED FOR INSTITUTIONAL CONTROLS
At every site where contaminants will be left in place (for any significant length of time), institutional controls are necessary to prevent the exposure of local residents to chemicals in groundwater and soil. At groundwater sites, institutional controls play three roles. First, they can restrict the use of contaminated groundwater. Second, they can protect the occupants of overlying buildings (or proposed buildings) from exposure to chemicals from contaminated groundwater through vapor intrusion (e.g., by requiring systems and barriers to prevent vapor from entering buildings). And third, they can prevent activities that might compromise remedies, such as penetration of landfill caps where the landfill is a source of groundwater contamination or pumping that is likely to spread contamination. If properly implemented and enforced, institutional controls allow a groundwater remedy to be protective in cases where residual contamination
remains above unrestricted use level. From 1986 to 1996, 3 to 20 percent of groundwater remedies at Superfund facilities had institutional controls. However, by 2008, 93 percent of the groundwater remedies selected that year included institutional controls (EPA, 2010a) and current guidance is likely to require such controls at every groundwater contamination site.
Types of Institutional Controls
Institutional controls (ICs) are administrative and/or legal controls that minimize the potential for human exposure to contamination and/or protect the integrity of a remedy, generally by attempting to modify human behavior. For example, proprietary controls represent a private agreement between the current property owner and, in this situation, EPA, a state, or a federal agency that has transferred or plans to transfer property that has use restrictions. The control is generally authorized by state law. An easement or restrictive covenant prohibiting the extraction of groundwater for drinking water on property containing the contaminated groundwater plume is an example of this type of instrument.
There are also direct governmental controls on the use of property, such as zoning laws, building codes, or state, tribal, or local groundwater use regulations. Federal agencies such as the Army may possess the authority to enforce institutional controls on their property, e.g., in Base Master Plans, facility construction review processes, facility digging permit systems, and/or the facility well permitting systems.
The third category of institutional controls are components of enforcement instruments or permits issued by federal or state regulators to private or federal PRPs (e.g., administrative orders, permits, Federal Facility Agreements, and judicial consent decrees). These legally enforceable instruments may limit site activities or require the performance of specific activities like the monitoring of IC effectiveness.
Finally, there are informational devices such as recording site cleanup documents in property records and providing advisories to local communities, tourists, recreational users, or other interested persons that residual contamination remains on-site. Although informational devices are not enforceable, they may be required by an enforceable consent decree or other enforceable instrument.
Each type of institutional control has advantages and disadvantages, which revolve around, for example, how the control enables or restricts future economic development, whether the control is enforceable, and at which level of government it is enforced (e.g., zoning is traditionally a function of local government and generally, EPA and federal agencies have little or no direct role in local zoning). Different institutional controls differ with respect to who pays to maintain and enforce the control. At CERCLA-
funded cleanups, EPA does not pay for monitoring or enforcing institutional controls because the statute requires states to ensure the payment of all future routine operations and monitoring following CERCLA-financed remedial actions. However, at sites where private companies or other federal agencies perform the cleanup, they, not the states, pay for monitoring or enforcing institutional controls (see discussion below). The degree to which the public is involved in establishing, monitoring, and ensuring that institutional controls are enforced differs by type (EPA, 2010b), as does the length of time over which the institutional control must be maintained.
Past Experience with Institutional Controls
Not surprisingly, past experience suggests that institutional controls have been effective at some sites and have failed other sites (ELI, 1999, and see Box 5-1 for three prominent failures). Institutional controls “rely heavily on humans to implement, oversee, and administer them” and it is human “to ignore tasks that no one else seems to care about or where the purpose is not readily apparent and indeed is often buried underground” (ELI, 1999). A specific problem is the fact that zoning requirements can be modified by political bodies (ELI, 1999; Spina, 2008). Furthermore, environmental regulatory agencies may not be able to enforce restrictions on subsequent property owners (Spina, 2008; Probst, 2006), although increasingly states have adopted statutes that allow enforcement of land use restrictions on subsequent owners. Finally, where EPA does not regularly consult with local authorities about institutional controls, remedies may be selected, including a specific institutional control, without determining whether it can be implemented by the local government (ELI, 1999; Probst, 2006).
The New Emphasis and Direction on Institutional Controls
EPA has substantially improved its process of developing, implementing, and enforcing institutional controls. Each Superfund facility is supposed to have an Institutional Control Implementation and Assurance Plan (ICIAP) “prior to, or at the same time as, the remedial design phase under CERCLA and finalize it with the completion of the response action” (EPA, 2010b), and coordination between states, tribes, and local land use planning jurisdictions is required. Institutional controls at “construction-completion” sites have begun to be recorded within the Superfund Enterprise Management System to help ensure the long-term effectiveness of the controls (EPA, 2011a).
EPA has clarified that institutional control documents and instruments should clearly articulate the substantive restrictions that are needed at a
BOX 5-1
Examples of the Failure of Institutional Controls
At Love Canal (one the first hazardous waste sites of general public concern), the City of Niagara Falls built a school on a landfill in 1954, even though there was a 1953 deed from a chemical company to the city (i.e., both a proprietary and informational IC) disclosing that chemical production waste was buried on the property and disclaiming responsibility for any injuries that might result (Technical Review Committee, 1988). In the mid-1970s after residential housing was built around the landfill, heavy rain caused the groundwater to mobilize and release the chemicals onto residential properties and into local storm sewers, resulting in the first Presidential Declaration of a man-made national disaster (Technical Review Committee, 1988).
At the Cannons Engineering Corporation Superfund facility in Bridgewater, MA, the ROD required that a Declaration of Restrictions (i.e., a proprietary IC) be recorded with the deeds to the affected properties, along with zoning ordinances (a direct governmental control) and public education programs (an informational IC) (ELI, 1999). In 1998, a company, without prior approval of the environmental agencies, excavated soil below the water table, dewatered the excavation, and discharged the water on the property while erecting a telecommunications relay tower, in violation of the Declaration of Restrictions (EPA, 2010c). EPA issued a written notice of violation of the deed restriction to the property owner, lessee, and the Town of Bridgewater. In response, the leases and subleases have been modified (EPA, 2010c). In addition, the Town of Bridgewater has incorporated the deed restriction and the requirement to notify EPA prior to work at the tower into its site plan approval process. The deed restriction currently remains in place and there have been no additional violations. The education program apparently was never carried out because of lack of public interest (ELI, 1999).
At the Sharon Steel Superfund facility in Midvale, Utah, the ICs included (1) regulations governing excavations on private property within a residential area where some contaminated soils were left in place (a governmental control) and (2) education programs (ELI, 1999). The education programs were not successful, in large part due to lack of cooperation between the city, state, and EPA (ELI, 1999). As a result, one property owner who did not know about the ordinance began unpermitted construction of a new sewer line, another property owner removed his patio exposing unremediated soils for a day and half until the City learned of the activity, and another property owner and the state Department of Transportation failed to coordinate with state environmental regulators concerning the excavation of a city right of way (ELI, 1999).
property to ensure that the land use assumptions that were made as part of the remedy decision continue to remain accurate (EPA, 2011b). Where residential properties are located over a contaminated groundwater plume and the properties are not the source of contamination, well drilling restrictions
may be put in place to limit the use of groundwater rather than negotiating covenants or easements with a large number of parties (EPA, 2010b). EPA (2010b, 2011b) requires that each institutional control instrument be reviewed annually to consider such things as their long-term effectiveness and enforceability, and whether the property owner/lessee is aware of and complying with the institutional controls when they change land uses, perform new construction, or transfer the property.
Costs
EPA recognizes that institutional controls, maintenance, and enforcement costs “may extend beyond the 30-year period traditionally used in many response cost calculations,” and that these continuing costs should be acknowledged when developing response cost estimates because they “can be important in evaluating long-term effectiveness” (EPA, 2010b). Indeed, the IC development process should begin with estimating the cost for monitoring and reporting activities over the full life cycle of the control.
At Superfund-financed sites (i.e., those without viable PRPs), EPA does not pay for monitoring or enforcing institutional controls because CERCLA Section 104(c)(3) requires states to ensure the payment of all future routine operations and monitoring following remedial actions. At sites where there are viable PRPs or federal RPs, EPA has long negotiated settlement agreements or consent orders with such parties, and where necessary obtained a court order, to require a PRP to perform work necessary to achieve and maintain performance standards or the effectiveness of the remedy (e.g., five-year review, additional remedy work, and/or new information or unknown condition reopener consent decree) (EPA, 2006). Recent EPA guidance explicitly directs EPA staff to have the settling parties in such settlement agreements or consent orders gather and submit data and analyses about institutional controls in conjunction with requests for monitoring data (EPA, 2011b). Additionally, EPA now recommends the use of direct payments from PRPs, settling party trust funds, surety bonds, letters of credit, insurance, and settlement proceeds to fund site-specific accounts for institutional controls (EPA, 2010b). Federal agencies, including DoD, generally pay for long-term monitoring and perform oversight of institutional controls at their sites (DoD, 2001).
***
EPA has improved its institutional control program so that it encourages cooperation among federal, state, and local governments; incorporates independent oversight of the entities that implement institutional controls; includes redundancy; mandates monitoring; and increases the amount of
information available to the public about controls that are in place and the public’s involvement in oversight. Because this guidance is new, there is limited information about the effectiveness of current institutional control efforts and their costs, and there is no documentation of injury being caused by the failure of institutional controls. Conceptually one can predict that the cost of institutional controls will increase substantially under the new guidance, particularly as settling and responsible parties pay local or state governments to oversee and enforce institutional controls.
The primary risk from a failure to establish or enforce an adequate set of institutional controls are that the public (residents, workers, and visitors) may be exposed unknowingly to contaminated groundwater or vapor intrusion at levels above those allowing unlimited use and unrestricted exposure. For example, individuals may drill wells for potable use in contaminated aquifers. Developers may perform construction in contaminated soil or water that results in worker exposure. The failure of institutional controls may lead to property damage or personal injury lawsuits. Finally, each failure undermines the credibility of the regulators and local officials with the public, which is likely to make long-term management of the site more difficult and expensive—costs that could be avoided if contaminants are not left in place. It is for this reason that DoD guidance explicitly requires that “where a use restriction will be imposed through the environmental restoration process” the DoD must “ensure that the evaluation of response alternatives includes an analysis of an alternative with a use restriction, as well as an analysis … of a response not requiring a use restriction” (DERP, 2001).
EMERGENCE OF UNREGULATED AND UNANTICIPATED CONTAMINANTS
Leaving contamination in place can become problematic when the contamination includes compounds that were previously unregulated or unanticipated or when the toxicity information for known contaminants changes. Conventional contaminants of concern have been known for some time, are widespread at many sites, are generally detected at elevated concentrations, and can be readily detected and measured using standard laboratory instruments. These include many organics (e.g., volatile organic compounds [VOCs], semi-volatile organic compounds [SVOCs]), inorganics (e.g., metals, explosives, propellants), and radionuclides. Contaminants of emerging concern are chemical substances that have been identified recently or that have been known for some time, but for which it has previously been thought that action was not necessary. It could be that they are now detected because of advances in analytical techniques (e.g., detectable to parts per trillion, nanograms per liter [ng/L]) or that a
newly discovered adverse impact on humans or the environment has been identified. An example is perchlorate, where after 1997 the lowering of the detection limit led to the recognition that this rocket fuel oxidizer had been broadly released to the environment at sites of aerospace manufacture and testing, both initially in California and thereafter throughout the nation. Subsequent studies documented the presence of perchlorate associated with blasting and fertilizer use, as well as naturally high background concentrations (ITRC, 2005b). Contaminants of emerging concern in groundwater, discussed individually in Box 5-2, may present challenges not only from a risk assessment perspective but also from the perspective of remedial technologies. Existing remedial technologies may be inadequate for these compounds, necessitating either adaptation of existing methods or development of new methods.
Under the Safe Drinking Water Act (SDWA), EPA develops a list of contaminants of potential human health concern from drinking water exposure called the Contaminant Candidate List (CCL). The most recent list (EPA, 2009b) contains 104 chemicals or chemical groups and 12 microbiological contaminants including pesticides, antibiotics, and other pharmaceuticals or their degradation products, disinfection by-products, industrial chemicals, and waterborne pathogens that are not currently regulated under national primary drinking water standards. Not all contaminants on the list are likely regulatory targets for drinking water limits; i.e., MCLs are expected to be developed for only a small subset of the contaminants on the CCL. Identification of candidates for which an MCL may be appropriate is based on a number of factors including prevalence of the contaminant in drinking water supplies (including both groundwater and surface water sources), the magnitude of risk presented by the contaminant, and the potential for meaningful risk reduction through additional regulation (NRC, 2001). It should also be emphasized that EPA’s decision not to develop an MCL for a chemical does not indicate that a particular chemical will not be of toxicological interest at any particular site. Nonetheless, the CCL can provide a roadmap for site managers with respect to potential site reopeners.
New toxicity information can change the way a contaminant is regulated. In particular, if the cancer potency or non-cancer toxicity value for a chemical changes (even if the MCL does not), sites regulated under CERCLA will have to be evaluated during the five-year review process to make sure the remedy is still protective. One of the most important developments for groundwater remediation is the increase in the cancer potency, and the issuance of a non-cancer reference dose, for tricholorethene or TCE (EPA, 2011h). It is not clear whether these changes will alter groundwater remediation goals for TCE or the MCL of 5 μg/L, but it is certain that EPA will evaluate what, if any, modifications may be appropriate. The decision on changing the MCL will depend on many factors, including the chosen
BOX 5-2
Contaminants of Emerging Concern in Groundwater
By definition, contaminants of emerging concern have the potential to present a risk not previously known. The examples provided below highlight compounds that could potentially be found at subsurface hazardous waste sites and may soon come under more intense scrutiny.
1,4-Dioxane. 1,4-Dioxane, a probable human carcinogen, is a stabilizer added to some chlorinated solvents, and thus is a co-contaminant at sites contaminated with these compounds (primarily 1,1,1-trichloroethane). 1,4-Dioxane is miscible with water and has low volatility, and thus is poorly retained by the solid media in groundwater systems. This makes air stripping and sorption to activated carbon used with pump-and-treat systems ineffective as treatment techniques, although advanced oxidation techniques are effective. Recent work has shown that while 1,4-dioxane is degraded via co-metabolism under specialized conditions, sustained biodegradation using enrichment cultures or aquifer materials under a variety of redox conditions were not successful (Vainberg et al., 2006; Steffan, 2007). Phytoremediation is able to remove 1,4-dioxane, but this would be limited to shallow water tables or wetland systems (Dietz and Schnoor, 2001; Aitchison et al., 2000). Oxidation of 1,4-dioxane using permanganate (Waldemer and Tratnyek, 2006) and in advanced oxidation processes (Bowman et al., 2003; Suh and Mohseni, 2004) effectively destroys 1,4-dioxane, but there is a clearly a need for the development of additional in situ treatment techniques.
A comprehensive review of the environmental issues surrounding this compound is available (Mohr, 2010). Recent increases in the frequency of detection of this chemical in municipal wells in California suggest that 1,4-dioxane is a chemical that may lead to many reopeners at hazardous waste sites where 1,1,1-TCA was released.
Naphthalene. Naphthalene, a polycyclic aromatic hydrocarbon consisting of two benzene rings, has been found in groundwater, particularly associated with coal tar production and distillation from former manufactured gas plan (MGP) operations. Although naphthalene is rarely found in drinking water supplies, at some MGP sites concentrations of naphthalene in the thousands of µg/L have been found (ATSDR, 2005), creating the potential for vapor intrusion given the high volatility of naphthalene. There is currently no MCL for naphthalene.
Although an EPA assessment in 2004 concluded that naphthalene was likely to be carcinogenic in humans via the inhalation pathway (EPA, 2004b), that assessment is no longer considered up to date and there are ongoing analyses within the agency and by outside scientists (e.g., Rhomberg et al., 2011). Nonetheless, the California EPA has designated naphthalene a carcinogen via the inhalation pathway and concluded that, at least at one site, cancer risks from naphthalene were comparable to those from benzene (Christopher et al., 2005). The recent toxicology findings for naphthalene, combined with increasing interest in the vapor intrusion pathway, provide an example of how emerging contaminants of concern may arise.
N-nitrosodimethylamine (NDMA). NDMA is a potential carcinogen and known to cause liver damage in humans and animals (ATSDR, 1989). NDMA pollution of groundwater may arise from two sources: manufacture of rocket propellants and
recharge of disinfected wastewater. While numerous studies have explored advanced oxidation processes to treat NDMA in drinking water (raw or finished) and wastewater, only limited studies have been performed to assess means to treat NDMA in contaminated groundwater. Zero-valent iron is able to reduce NDMA, albeit slowly, and the reaction is accelerated with a nickel-catalyst (Gui et al., 2000; Odziemkowski et al., 2000). There is also evidence of biodegradation at a water recharge site (Zhou et al., 2009). A recent SERDP report found that under oxic conditions, NDMA is mineralized via a co-metabolic processes, and that under anaerobic conditions (either natural or generated via in situ redox manipulation), abiotic reduction of NDMA occurred that was mediated by ferrous iron adsorbed to the aquifer material (Szecsody et al., 2009). Depending on the scope of the NDMA contamination problem, additional work is necessary to identify potential treatment techniques for NDMA-contaminated sites.
Perchlorate. Perchlorate (ClO4–) contamination of groundwater is a result of its use as an oxidizer in solid propellants, explosives, and pyrotechnics. Compared to the other contaminants in this section, there has been substantially more research on the treatment of perchlorate contaminated groundwater. Both laboratory studies and a field demonstration (Naval Surface Warfare Center at Indian Head, MD) have shown that in situ biostimulation using low-cost carbon amendments (e.g., lactate, acetate, molasses, canola oil) is an effective means to treat perchlorate (GeoSyntec Consultants, 2002; Cramer et al., 2004). The bacteria responsible have been characterized in terms of their biochemistry and genetics (Coates and Achenbach, 2004, 2006). Biostimulation by plant-produced electron donors has also been demonstrated (Shrout et al., 2006), and a permeable barrier comprised of wood particles designed to allow bacteria to create reducing conditions also lead to removal of perchlorate (Robertson et al., 2007).
The potential of zero-valent iron to reduce perchlorate has been demonstrated in laboratory studies (Huang and Sorial, 2007), but the presence of other anions slows or inhibits perchlorate removal. Results from systems containing both bacteria and zero-valent iron have suggested that the combination leads to improved removal (Son et al., 2006; Yu et al., 2006), while another study suggests that iron metal inhibits bacterial removal (Shrout et al., 2005).
EPA has decided to regulate perchlorate pursuant to the Safe Drinking Water Act due to its effect on thyroid function (EPA, 2011c).
Perfluoroalkyl substances (PFAS). PFAS are a family of organic compounds that consist of a perfluorocarbon chain (typically 4 to 14 in length) and an anionic head group (Simcik, 2005; Lau et al., 2007; Ranye and Forest, 2009). They have been synthesized and used in a broad range of industrial and commercial applications for over 50 years, but their environmental risk was not highlighted until a decade ago (Simcik, 2005; Fujii et al., 2007; Richardson and Ternes, 2005). Up until 2002, the total historical production of PFAS reached approximately 100,000 tons, of which the majority will ultimately gain their entry into the environment (Pistochi and Loos, 2009). PFAS are also components of aqueous film-forming foams (AFFFs) that are used in fire fighting. Training with AFFFs has been conducted for more than 30 years, resulting in releases in fire fighting training areas, including military bases. A recent survey of the Department of Defense Knowledge Based Corporate Reporting System found 594 fire and crash training sites operated or managed by military organizations. It was found that 353 of these sites were
operated by the Air Force, the Navy operated 132, and the Army had 94 (Rak and Vogel, 2009). The rest were under the purview of the Formerly Used Defense Sites. Any military site where AFFFs have been used to fight fires or to train in firefighting may have a dissolved plume containing PFAS.
In January 2009, the EPA announced Provisional Health Advisory values for perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) of 0.4 μg/L in drinking water to protect humans from a variety of noncarcinogenic health effects (EPA, 2009a). To date, only pump-and-treat systems using activated carbon as a sorbent are effective at removing these compounds. The stability of the C-F bond makes chemical and biological treatment difficult. There is clearly a need to develop new techniques to treat groundwater contaminated with this class of chemicals.
Pharmaceuticals and Personal Care Products (PPCP). This class of compounds includes prescription and non-prescription medications, antibiotics, hormones, and others. Recently 35 different PPCP compounds were detected in groundwater samples (Barnes et al., 2008). Possible pathways to groundwater include landfill leachate (Buszka et al., 2009); indirect or direct aquifer recharge with wastewater effluent (Kreuzinger et al., 2004; Rabiet, et al., 2006); irrigation with wastewater effluent; leaking sewer pipes (Nakada et al., 2008); application of biosolids to soils; leakage from septic tanks (Standley et al., 2008; Carrara et al., 2008); or leakage from animal waste lagoons at confined animal feeding operations—suggesting that the detection of these compounds at hazardous waste sites is unlikely.
There is evidence of compound attenuation in aquifer recharge systems (Drewes et al., 2003; Snyder et al., 2004; Greskowiak et al., 2006; Soliman et al., 2007) and also of compound persistence (Drewes et al., 2003; Kreuzinger et al., 2004). Further studies are needed to determine if the environmental levels of pharmaceuticals and personal care products present a significant human or environmental risk. Any evaluation of the risks presented by such releases to the environment should consider the advantages and disadvantages of (1) limiting their entrance into groundwater systems through improved treatment prior to dis
acceptable risk level, whether the cancer or non-cancer risk is considered, the detection limit, costs, and technical feasibility. The present MCL of 5 μg/L is now associated with a 10–5 risk over a 70-year exposure, which falls within EPA’s acceptable risk range of 10–6 to 10–4. The non-cancer reference dose has led to a regional screening level for TCE in drinking water of 2.6 μg/L1 (although it should be noted that the screening level is not a cleanup goal, and site-specific factors could result in a non-cancer TCE cleanup goal that exceeds 2.6 μg/L).
__________________
1http://www.epa.gov/reg3hwmd/risk/human/rb-concentration_table/.
charge; (2) verifying that natural attenuation processes in surface soils degrade them to acceptable levels; or (3) verifying removal during drinking water treatment.
Nanomaterials. Nanomaterials are between 1 and 100 nm in size and are used widely in personal care products, clothing, and electronics. However, the issue of importance to this report is the use, at more than 36 sites, of nanomaterials to remediate environmental contaminants such as organohalides, trinitrotoluene, and phenols (EPA, 2011d,e; Obare and Meyer, 2005). For example, nanomaterials can serve as semiconductors in the photocatalysis of chlorinated phenols, and sorbents developed from nanomaterials can be used to remove arsenic and cadmium from water (Vaseashta et al., 2006; Husain, 2008). However, little is known about the human or environmental health effects of nanomaterials, although they are increasingly used in consumer applications.
EPA regulates new chemicals prior to widespread commercial use by requiring a pre-manufacturing review pursuant to the Toxic Substance Control Act. Regulation is based on a finding that the chemical may present an “unreasonable risk.” Historically EPA did “not consider a nanoscale version of a chemical to be a different chemical substance from the macroscale version,” i.e., it was not a new chemical subject to pre-manufacturing review. This interpretation, however, has changed, and EPA issued significant new use notices requiring reporting and testing of siloxane-modified silica nanoparticles, carbon nanotubes, and other nanomoterials (EPA 2008b,c, 2011e,f,g,). These actions include requiring testing to assess inhalation risks, ecotoxicity, and aquatic toxicity. EPA also is regulating nanomaterials in pesticides through the Federal Insecticide, Fungicide, and Rodenticide Act.
Siloxanes. Siloxanes are organosilicon chemicals (containing an R2SiO unit, where R can be hydrogen or a hydrocarbon group). Chemicals with multiple R2SiO units are called polysiloxanes. Siloxanes have high vapor pressure, low water solubility, and high Henry’s law constant (McBean, 2008), and are commonly found in personal care products such as shampoo and antiperspirant. Thus, they are unlikely to be found at hazardous waste sites. The toxicity of siloxanes is variable.
The impact of the new TCE toxicity information on the vapor intrusion pathway is important because of the potential longer exposure periods, although again the nature of this impact is still uncertain. The new cancer potency value yields a 10–4 risk at 20 μg/m3, a 10–5 risk at 2 μg/m3, and a 10–6 risk at 0.2 μg/m3 for a lifetime-year exposure2. The new non-cancer reference concentration (RfC) is 2 μg/m3. Thus, indoor air concentrations
__________________
2 For a 30-year exposure, as is typically used in Superfund, the corresponding concentrations are 41, 4.1, and 0.41 ìg/m3. http://www.epa.gov/reg3hwmd/risk/human/rb-concentration_table/Generic_Tables/pdf/master_sl_table_run_MAY2012.pdf.
of TCE arising from vapor intrusion may fall within the acceptable cancer risk range, yet exceed the non-cancer criterion.
Adding further complexity to the entire TCE situation is the question of the appropriate averaging time for non-cancer risk from TCE. Typically the exposure concentration would be an average over the period of exposure. However, the endpoints on which the RfC is based are developmental. For some developmental toxicants, EPA may find it appropriate to average over a shorter duration, because of concern that a single exposure during a critical developmental period may produce an adverse effect (EPA, 1991a). While there may be scientific debate about the appropriate averaging time, at least one EPA region is considering an averaging time for TCE over a shorter exposure duration (e.g., Lee, 2012) which, if implemented, could have significant ramifications for determining compliance. At a given site, alternative averaging periods should be determined based on specific exposure elements and chemical-specific properties.
Going forward, any review of the protectiveness of a TCE-related remedy at a Superfund facility during a five-year review is likely to consider both cancer and non-cancer risks using these new TCE toxicity criteria. EPA may conclude that some remedies meeting the current MCL are no longer protective (e.g., if the long-term concentration is above the non-cancer screening level of 2.6 μg/L). At sites where the cleanup level has not yet been set, EPA may choose a goal more stringent than the current MCL of 5 μg/L because the non-cancer level (2.6 μg/L) is lower. Because of its prevalence in contaminated groundwater, the potential reduction in the MCL for TCE has been estimated by some to have a major impact on the costs of remedial actions at DoD sites, on the order of $15 billion (Yonkers, 2012). However, there is much debate about the likely costs, and the Committee has insufficient information to reach a conclusion concerning the magnitude of the impact.
Hexavalent chromium Cr (VI) has been detected in groundwater at sites associated with waste disposal. For example, ATSDR (2008) describes several examples of sites in the United States with elevated groundwater concentrations of Cr (VI), with some concentrations exceeding 10,000 μg/L; the sources of the contamination were variable and included disposal of chromate used as a corrosion inhibitor and wastes from aircraft plants. Although inhaled Cr (VI) is clearly carcinogenic, studies of ingested Cr (VI) in humans have been conflicting, and overall, do not confirm Cr (VI) as a human carcinogen (e.g., Beaumont et al., 2008; Kerger et al., 2009). However, recent studies of high concentrations of Cr (VI) in drinking water in rodents provide evidence of carcinogenicity in the oral cavity and gastrointestinal tract (NTP, 2008). The significance of such findings for humans exposed to Cr (VI) in drinking water, albeit at much lower concentrations than in the rodent studies, is being considered. California and New Jersey have
already set advisory levels, and EPA is assessing whether to set an MCL for Cr (VI). EPA has decided to require monitoring for Cr (VI) in drinking water, even in the absence of an MCL (LaRoss, 2012). [Interestingly, recent mode-of-action studies suggest that the dose-response relationship may be nonlinear (Thompson et al., 2012), which could have important ramifications for setting an MCL.]
New quantitative dose-response analyses have elevated the importance of inorganic arsenic. In 2001 the drinking water limit for inorganic arsenic was lowered from 50 μg/L to 10 μg/L based on evidence that inorganic arsenic could cause internal cancers in addition to skin cancer. The MCL analysis employed a dose-response assessment in which the potency was effectively equivalent to 3.7 mg/kg/d based on bladder cancer, versus the prior limit of 1.5 mg/kg/d based on skin cancer. The reduction in the arsenic MCL from 50 μg/L to 10 μg/L has already resulted in a remedy reopener at the Arsenic Trioxide Site in North Dakota, as a result of the determination that the drinking water limit of 50 ppb was no longer protective of public health.3 Sampling determined that 84 percent of the well water users in the 26 townships were drinking water containing arsenic concentration at or over the arsenic MCL of 10 ppb (EPA, 2010d). A number of new actions were taken, including supplying some residents with bottled water, connecting other residents to a rural water supply system, and enhancing the water treatment system. EPA is continuing to evaluate the carcinogenicity of inorganic arsenic, which could have further implications for the MCL and determinations of the protectiveness of remedies.
It should be noted that, at times, new understandings of chemical toxicity can indicate lower risk values than had previously been established. This is the case with tetrachloroethene (PCE), where the recent cancer toxicity criteria are well below (i.e., indicating less potency) the prior values.4 Specifically, the oral cancer slope factor is more than 200-fold lower and the inhalation cancer risk value is more than 20-fold lower than prior values. Assuming a 30-year exposure duration, the air concentration for 10–6 risk would be 9.4 μg/m3, and the corresponding tap water concentration (considering ingestion, inhalation, and dermal exposure) would be 9.7 μg/L (above the MCL of 5 μg/L). While it is highly unlikely that the MCL would be increased, these findings could lead to reduced focus on vapor intrusion of PCE.
Thus, contaminants of emerging concern may be new, previously unrecognized contaminants, or previously recognized contaminants where new toxicity understanding or new exposure limits have developed, leading to concerns at lower concentrations. Contaminants of emerging concern
__________________
3 See http://www.epa.gov/region8/superfund/nd/arsenic/index.html.
present challenges for a number of reasons, including the lack of generally accepted toxicological criteria that can be used for risk assessment purposes. The absence of such criteria complicates the determination of health-protective limits and can result in different limits in different regions. Such contaminants also add uncertainty to risk management decisions, leaving open the possibility of future site reopeners. The Department of Defense’s program for contaminants of emerging concern, called “Scan-Watch-Action” (Murnyak et al., 2011) employs a multi-tier process to identify contaminants and weigh them in terms of their likelihood of having an impact on multiple DoD functional areas, such as acquisition, training and readiness, and remediation. For the subset of contaminants of emerging concern that could have an impact on these key functional areas, risk management options are developed.
In addition to identification of new constituents of potential toxicological concern as well as development of new toxicity information for “old” chemicals, new exposure pathways may be identified or recognized as more significant than had previously been considered. Even when the existence of a pathway is recognized, there may be insufficient information as to the parameters that influence that pathway and hence the lack of clearly articulated guidance for how to incorporate the pathway in a risk assessment. These deficiencies can result in failure to adequately incorporate the pathway into site-specific risk analyses or result in significant uncertainty in calculated risk results. New pathways raise institutional and public health concerns that need to be considered in remedial decision making.
Vapor Intrusion
Although not “new” in that it has been increasingly recognized over the last ten tears, the vapor intrusion pathway is of particular interest with respect to subsurface contamination. The sources of these vapors may be free phase NAPLs entrapped in the vadose zone (from a spill and/or exposed during water table fluctuations) or dissolved constituents that partition from the groundwater plume to soil gas. Vapors emitted from these contaminant sources disperse into air-filled void spaces within the soil and migrate below surface structures, leading to the intrusion of contaminant vapors into indoor air. The exposure can be substantial even at low indoor air contaminant concentrations because the average American spends more than 21 hours per day indoors (Olson and Corosi, 2002). Note that there is the potential for volatile organic compounds from coal- and oil-derived DNAPL sites to present a risk via the vapor intrusion pathway (see Singh et
al., 2011, and Doroski et al., 2011). However because such investigations are ongoing with no definitive results available, the following discussion focuses on chlorinated solvents.
It is unknown how many chlorinated solvent sites in the United States have vapor intrusion levels considered by federal or state policies to be unsafe. Thousands of dry cleaners in the United States, many of which have long been out of business, may have generated plumes of PCE in the subsurface, but it is unknown what percentage of such sites is contaminated because no federal or state regulatory program requires their investigation. Several documented cases of vapor intrusion involving chlorinated solvents and their breakdown products, such as TCE and DCE, have received national attention. For example, the Redfield and Colorado Department of Transportation sites in Colorado were two of the first major sites to bring the vapor intrusion issue to the regulators’ and public attention in the late 1990s. At the Redfield site, the indoor air in about half of the 800 single-family homes overlying a dissolved groundwater plume of VOCs exceeded 0.49 μg/m3 (Folkes et al., 2009). Hill Air Force Base, Utah, was one of the first DoD sites to address the vapor intrusion pathway (with details provided by Erik Dettenmaier, Hill AFB, personal communication). There are approximately 3,000 residences overlying dilute chlorinated solvent groundwater plumes outside the base, and about half agreed to participate in an indoor air monitoring program. Roughly 4,500 indoor air samples have been collected, with TCE being detected in 235 homes. 123 homes have indoor air concentrations in excess of 2.3 μg/m3, which correlates with a cancer risk of approximately 5 × 10–6 over 30 years (EPA, 2012a). Of the homes with concentrations above 2.3 μg/m3, about 25 percent are suspected to be affected by indoor sources of TCE (such as gun cleaner, craft glue, and cleaning products). Sub-slab depressurization systems have been installed at homes where vapor intrusion is known or suspected to be occurring. As a result of this site, the DoD has gained considerable experience over the past decade with designing indoor air monitoring programs, managing community relations, and interpreting and communicating data on vapor intrusion. Box 5-3 describes how EPA and the states have altered cleanup programs to consider the vapor intrusion pathway.
Vapor intrusion investigations are generally required anywhere chlorinated solvents are detected in shallow aquifers or the vadose zone. Multiple lines of evidence, such as sampling of indoor air, outdoor air, and soil gas, are needed to determine the extent of indoor air contamination and its source. Some jurisdictions, such as New York State, require action based solely upon high levels of contamination in the soil gas under a building, recognizing the potential for intrusion should a crack or hole develop in the building’s slab; others base responses upon current indoor air levels.
Although the recognition of vapor intrusion by EPA is at least ten years
BOX 5-3
Consideration of Vapor Intrusion in Federal and State Regulatory Programs
Contaminated sites were rarely evaluated for vapor intrusion until 2002, when the Denver Post published a landmark series of articles on the subject (Obmascik, 2002) documenting the Redfield site, the Colorado Department of Transportation site, and other sites in Colorado. At that time, EPA’s RCRA program had issued a draft vapor intrusion pathway assessment guidance (EPA, 2001a) as part of its Environmental Indicator program, but awareness of vapor intrusion was only just growing. In the wake of the Post series, EPA’s Office of Solid Waste and Emergency Response issued Draft Guidance for Evaluation the Vapor Intrusion to Indoor Air Pathway from Groundwater and Soils (EPA, 2002a), and several states followed suit. The EPA Region 9 Superfund division systematically reviewed their portfolio of sites for potential vapor intrusion. The New York Department of Environmental Conservation conducted a desktop review of 421 inactive sites with pre-2003 remedial decisions where disposal of chlorinated hydrocarbons occurred (NYS DEC, 2005), reopening numerous “closed sites” for investigation of vapor intrusion. However, in other jurisdictions (such as Texas, Michigan, and EPA Regions 4 and 6), regulators have been reluctant to reopen sites or broaden investigations to assess the vapor intrusion pathway. Finally, certain other jurisdictions have dealt with the new exposure pathway by issuing action levels (target concentration levels) that resulted in no further action.
There are a substantial number of sites where interim remediation and ongoing investigation have been under way, but where the vapor intrusion pathway was not considered. For example, the Motorola 52nd Street semiconductor factory in Phoenix, Arizona, was placed on the NPL in 1984. There is continuing investigation of the high levels of TCE in the deep bedrock under the neighborhood immediately downgradient from the plant, but Arizona’s Department of Environmental Quality—lead agency for that operable unit—has not evaluated potential vapor intrusion under the moderate income, predominantly Latino neighborhood, despite repeated requests from community leaders. Only in 2010 did EPA Region 9 announce a vapor intrusion investigation and form a Community Information Group to oversee it (EPA Region 9, 2010).
Just recently EPA has initiated a process whereby vapor intrusion may become a component of the NPL listing process (EPA, 2011i).
old, the magnitude of vapor intrusion as a national issue remains poorly understood. This is partly because of significant uncertainty in understanding the vapor intrusion pathway. That is, contaminant concentrations in indoor air are variously determined by concentrations in the subsurface, the subsurface geology, environmental conditions (such as wind speed and temperature), and building characteristics (e.g., presence or absence of a basement, foundation type and condition, heating/cooling). In addition,
background sources such as ambient (outdoor) air and consumer products may contribute to the indoor air levels of contaminants of concern. Chapter 6 discusses both the current limits in our understanding of vapor intrusion as well as advances in real-time sensor technologies that will make it easier to detect indoor air contamination from subsurface sources and determine the best mitigation strategies.
EPA has compiled an empirical data set for hazardous waste sites with paired groundwater, soil gas, and indoor air data on chlorinated solvents (EPA, 2012b). The raw data demonstrate that there are sites where there is reasonable potential for exceeding indoor air screening levels and, depending upon regulators’ final risk management decisions, the non-cancer risk management level in particular. Furthermore, detailed field studies under way in residences overlying dilute plumes suggest that the contaminant intrusion rate may be highly variable with time and the most significant contributions to longer-term exposure might happen through infrequent and short-duration periods of high vapor intrusion activity (Johnson et al., 2012). Thus, there are two choices for dealing with a possible vapor intrusion pathway at a given site: (1) invest in sampling and analyses to confirm whether or not the potential exposure is of concern, or (2) install a vapor mitigation system. The former is problematic because the number of samples likely needed to be confident in pathway assessment evaluation may not be practicable, and costs could easily exceed $10,000 per building when considering sampling spread out over multiple seasons and years (DoD, 2009). The latter also is challenging because installation costs are likely to be $2,000 or greater per residential mitigation system with annual electrical costs of $100 or more (EPA, 2008d; ITRC, 2007), and there are uncertainties surrounding continued operation and performance validation of mitigation systems. These issues are magnified when considering larger plumes with tens to hundreds of overlying buildings.
The current methods for disruption of the vapor intrusion pathway are based on substructure depressurization systems originally developed for residential radon mitigation (EPA, 2012c). In fact, one of the benefits of vapor mitigation is that it also prevents radon exposure (which, in certain regions of the United States, can pose a greater cancer risk than the levels of solvent vapors that require mitigation). Some local governments, such as Mountain View, California, require the installation of vapor mitigation systems in developments on or near solvent contamination plumes (Woodhouse, 2009). New Jersey requires Radon Resistant Construction for all homes and schools in high-radon areas.5 In general, though, state and local governments rarely require that VOC mitigation systems be installed in existing buildings in the absence of an environmental investigation.
__________________
5 See http://www.state.nj.us/dca/divisions/codes/codreg/pdf_regs/njac_5_23_10.pdf.
Mitigation techniques usually consist of perforated plastic pipes installed in the subsurface that are linked to venting systems propelled by electric blower fans. These must be operated, maintained, and monitored to ensure that they are working properly over the long term. Installing vapor mitigation systems is easiest and most inexpensive during initial building construction. To retrofit existing buildings, it is usually necessary to drill holes through floors and run vent pipes through rooms, while for new construction, horizontal pipes and plastic or rubber vapor barriers can be laid beneath foundations, with venting systems hidden, for a fraction of the cost of a retrofit. Some regulatory agencies suggest installing systems without fans, and then testing the indoor air after construction but before occupancy, at least where the threat of vapor intrusion is uncertain. If sampling shows elevated levels of VOCs indoors, fans are then installed and operated (e.g., see CalEPA, 2011). There are also commercially available passive barriers that mitigate vapor intrusion, including concrete sealants for industrial structures.
At many vapor intrusion sites involving residential property owners, some homeowners refuse to allow access for sampling or they reject mitigation (EPA, 2012c). There are a number of reasons for this, including owners’ fear that vapor intrusion will stigmatize their property and drive down its value, or a distrust of the agencies and companies that are conducting the response. Regulators do not insist that non-cooperating homeowners take part in investigations in the belief that they have a right to refuse protection. This presents challenges when property is sold or leased because buyers or occupants may not have the opportunity to have mitigation or even sampling conducted. They may be unaware that a vapor intrusion response has taken place in the area. EPA (2012c) addresses steps to be taken if the property owner refuses access, ranging from providing information on the consequences to requiring access “in the interest of protecting the occupants, for evaluating the need for response, choosing a response action, taking a response action, or otherwise enforcing CERCLA.”
It is difficult to enforce proper maintenance of the mitigation system over the life of the contamination and building. Building occupants may not understand why or how the systems work or otherwise fail to maintain them over the long run; some building occupants simply unplug them because of the energy cost or noise. Furthermore, there is often no protection for new occupants of buildings previously owned or occupied by people who chose not to cooperate with the vapor intrusion response. Therefore, regulatory agencies with active vapor intrusion programs consider mitigation an interim response (NJ DEP, 2005).
Leaving residual contamination in place above unrestricted groundwater and land use levels has several important legal ramifications, as discussed in detail below. The Committee recognizes that litigation is contentious and, by definition, there are at least two diametrically opposed versions of the facts and views of the law. The purpose of this section is not to argue that litigation will occur in every case, but rather that the potential is likely to exist as long as chemicals remain in the groundwater above drinking water levels. The reader should not consider the examples and illustrations given to be a comprehensive review.
Environmental Liability Disclosure Requirements
Leaving contaminants in place may trigger a variety of public notification and/or disclosure requirements, some of which are illustrated below. The Securities and Exchange Commission (SEC) requires companies to disclose, among other things, (1) administrative or judicial environmental protection proceedings arising under any federal, state, or local law, including hazardous waste cleanup laws; (2) when it is probable that an asset is impaired; and (3) trends in cleanup liabilities that are reasonably likely to affect its liquidity or its capital expenditures (SEC, 1982, 2011). Similarly, the Financial Accounting Standards Board has published requirements on reporting about environmental remediation liabilities in financial statements (Coate et al., 2012). These requirements exist to satisfy U.S. securities laws and general accounting practices, which are based on the principle that investors should have sufficient information about the financial status of companies to make an informed decision on whether to invest in company A versus company B (Rogers, 2007). The determination of exactly what environmental cleanup liabilities are required to be reported is based on the specific facts, e.g., the size of the company and the uncertainty associated with the cost and liability. The DoD regularly reports its environmental liabilities to Congress in order to provide reliable financial and budgetary information, among other purposes (Army, 2011).
A variety of evaluations have suggested that not all companies were reporting cleanup liabilities (NRC, 1997; SEC, 2003; Cox and Douthett, 2009; EPA, 2008e). SEC (2003) claimed that “many companies did not provide adequate disclosure relating to” environmental liabilities and “urged companies with material contingent liabilities to carefully review their disclosures and ensure that they include all required information.” There have been few SEC actions enforcing these requirements in the last two decades (EPA, 2008e).
In summary, companies are required to report long-term hazardous
substance cleanup liabilities under certain circumstances. Some believe that such reporting may provide an incentive for private and governmental PRPs to minimize their long-term liabilities by reducing the extent of residual contamination at their sites (NRC, 1997; EPA, 2008e).
Potential Personal Injury and Property Damage Liability
If chemicals remain in soil or groundwater in concentrations above unrestricted use levels, there is a potential that the active remedy, engineering controls, and institutional controls may fail and result in lawsuits alleging personal injury and/or property damage. In fact, even if the remedy does not fail and the government regulators determine the remedy is protective, private citizens may still file a lawsuit seeking alleged damages. [The reader is referred to Federal Judicial Center and NRC (2011) for a discussion of causes of action and burdens of proof.]
Each personal injury or property damage case depends upon the unique facts of the site and state law. Most contaminated groundwater sites do not result in litigation, although in the Committee’s opinion there appears to be a trend toward increasing litigation. There is no central repository of toxic tort cases and their outcomes, as most cases are likely to settle out of court rather than be litigated to a verdict, and many settlements are confidential or not readily accessible. Box 5-4 briefly illustrates a few of the major groundwater cases.
There are many defenses to such personal injury and property damage suits, not the least of which is a lack of causation. As a result, there are many cases that are either not brought or where the defendants prevail. Nonetheless, there is a possibility of personal injury or property damage litigation if residual contamination remains (or even if no contamination remains since personal injury lawsuits can be filed for past exposure). The cost of such litigation is highly uncertain, but the transaction costs (even if the defendant prevails) can be substantial. The uncertainty arises from variable transaction costs, difficulties measuring the decrease in property value and the extent of personal injury, and limited information about when groundwater contamination (and hence the damages) began or when it is considered “over.” Thus, it is not clear how responsible parties should factor the potential risk of litigation into a particular decision concerning remedy selection at a site.
Potential Liability for Natural Resource Damage Claims
At large or complex sites where chemicals are left above unrestricted use levels, there is the potential for natural resource damage (NRD) claims. CERCLA provides the United States, states, or, in certain circumstances, In-
dian Tribes (when they are acting as “trustees” of the resources in question) a statutory cause of action for damages, which refers to injury to, destruction of, or loss of natural resources [CERCLA § 107(f)(1)]. The natural resources covered are “land, fish, wildlife, biota, air, water, ground water, drinking water supplies, and other such resources belonging to, managed by, held in trust by, appertaining to, or otherwise controlled by the United States … any State, local government, or any foreign government, any Indian tribe …” [CERCLA § 101(16)]. Damage caused to such natural resources may result in a federal or state lawsuit to restore the resource or receive compensation for such damage. The legal basis of NRD lawsuits varies from state to state and has evolved over time (Israel, 2009).
NRDs include (a) the reasonable costs of assessing the extent of the damage (e.g., the field studies and reports); (b) “direct and indirect injury, destruction, or loss;” and (c) interim loss of use from the time of the release until the time of restoration (taking into consideration “replacement value, use value, and ability of the ecosystem or resource to recover”) [CERCLA § 107(a)(4)(C)]. One of the losses recoverable in such lawsuits is the amount of money required to compensate the public for the loss of use of the resource. This includes both commercial uses of the resource (e.g., supplying drinking water, supplying fish, or a wetland providing flood control) as well as nonuse damages (i.e., a value from the mere existence of a resource even in the absence of use) [43 CFR § 11.83(c)(1)].6 Indeed, the nonuse damages are explicitly provided for in Department of Interior and National Oceanic and Atmospheric Administration NRD assessment rules, which have been upheld from challenge (Ohio v. United States Department of the Interior, 880 F.2d 432, D.C. Cir. 1987). Most direct use values of groundwater can be approximated based on water prices (NRC, 1994); however, there is controversy over whether one can reliably estimate nonuse values.
Many of the larger NRD lawsuits involve contaminated sediment (Commencement Bay in Washington) or oil discharges to surface waters (such as the Exxon-Valdez and Gulf Oil spills) (Hutton and Edwards, 2011) and therefore are not relevant to contaminated groundwater sites. NRD settlements for mining sites, large oil spills, and contaminated sediment tend to be higher than other NRD settlements. Table 5-1 shows several large groundwater NRD settlements or actions to illustrate the types of historic claims that have been made. In summarizing 32 groundwater sites, Ando
__________________
6 The clearest example is New Jersey NRD guidance, which states that the damage calculation may be based on the current water rate derived from the NJ Board of Public Utilities (that is, how much it costs a home owner to buy water) times the volume of water (areal extent and thickness) times the duration that the water cannot be used (i.e., the time that it would take for the remedy to reduce the concentration to below the NJ Groundwater Quality standards set for the site).
BOX 5-4
Select Examples of Litigation Involving Contaminated Groundwater Sites
Love Canal. Hooker Chemical Company disposed of approximately 21,000 tons of a wide variety of chlorinated chemicals (including 2,3,7,8-tetrachlorodibenzo-p-dioxin) in an excavation in an undeveloped area of Niagara Falls (called Love Canal) from the 1940s to 1953 (EPA, 1991b). The City required Hooker to transfer the property to the School Board in order to construct a school, which soon acted to attract additional residents to the area. In the early 1970s, unusually heavy rainfall resulted in the migration of chemicals from the original disposal. President Carter declared the area a national disaster area in 1976. In 1979, a group of local citizens sued Hooker Chemical, the city, the county, and the Board of Education seeking $11 billion in damages (University of Buffalo, 2011). 1,336 Love Canal residents settled their personal injury suit for approximately $20 million in one settlement, and in another settlement 899 residents settled for $6.75 million (Center for Justice & Democracy, 2007).
Woburn, MA. A class action lawsuit was brought by 19 residents of Woburn, Massachusetts against two industrial companies for contaminating groundwater with chlorinated compounds (including TCE) alleging that exposure to groundwater caused injury to the local residents because the city used the groundwater as a source of drinking water. The case was settled, without an admission of liability, for $8 million (Center for Justice & Democracy, 2007).
Hinkey, California (the Erin Brockavitch case). Hexavalent chromium that was used by a utility in cooling towers for corrosion control contaminated the groundwater in Hinkley, California, prompting approximately 650 area residents to sue the utility. The utility ultimately agreed, without admitting liability, to pay $333 million to the residents, to clean up the groundwater, and to cease use of hexavalent chromium (Center for Justice & Democracy, 2007). Interestingly, in 2011 the California Desert Sierra Cancer Surveillance Program found “cancer occurrence in the Hinkley Census Tract that is slightly, but not significantly, below the number of new cases expected for an average risk population having the same demographic characteristics as the Hinkley Census Tract population” (Morgan, 2011).
et al. (2004) found the average groundwater NRD settlement to be $15 million as of 2004 (or on average, a half a million dollars per site).
New Jersey is unique in the large volume of NRD cases that have been filed—approximately 123 lawsuits involving over 1,500 sites (NJAG, 2011). The settlements resulted in payments of approximately $72.9 million ($45,617 per site) and thousands of acres of open space preserved as wildlife habitat and groundwater recharge areas (NJ DEP, 2007, 2008), although some of the settlements involved larger payments and the cost of
MTBE Groundwater Class Action. Public drinking water providers filed product liability lawsuits against a number of oil companies concerning contamination of groundwater by methyl tert-butyl ether (MTBE), which leaked from underground storage tanks. MTBE is an oxygenate added to gasoline to meet the requirements of the Clean Air Act. In 2006, a district court held that injury to the water suppliers did not require an exceedence of state and federal drinking water standards (BNA, 2006). Most of these cases have been settled without admitting liability. For example, in May 2008, a dozen defendants agreed to pay $422 million to 153 water suppliers and to pay 70 percent of the future treatment costs that arise over the next 30 years (BNA, 2010). On August 4, 2010, 48 defendants agreed to a $42 million settlement with 27 water districts and towns on New York’s Long Island and in Florida (BNA, 2010). One case was tried and resulted in a $105 million jury verdict in favor of New York City, although the case is being appealed.
Sunburst, Montana. In a 4-3 vote, the Montana Supreme Court upheld a jury award of $15 million (more than seven times the pre-contamination value of the property) in compensatory damages for restoration of the groundwater contaminated by benzene releases from an oil refinery. The defendant had proposed, and the Montana Department of Environmental Quality (MDEQ) approved, a monitored natural attenuation remedy with a 20- to 100-year time horizon to attain the benzene drinking water standard (Tanner, 2009, summarizing the facts in Sunburst Sch. Dist. 2 v. Texaco, Inc., 165 P.3d 1079, 1090). MDEQ based its approval of MNA on the fact there was no exposure pathway and, therefore, no risk. However, the trial court had found (and the Montana Supreme Court assumed) that the defendant had omitted data and provided other data “to create the misleading impression that the benzene level in groundwater had been declining.”
Tuscon Airport TCE Plume. Approximately 1,618 area residents living within or near a TCE groundwater plume emanating from the Tucson Airport brought a lawsuit against the City of Tucson and the Tucson Airport Authority claiming that the TCE used to clean airplanes in the area from the mid-1940s through the early 1950s caused the plume and resulted in bodily injury. After years of litigation, insurance companies for the City and Airport authority agreed, without admitting liability, to pay $35 million to resolve the personal injury lawsuits (Claims Journal, 2004).
the land preservation exceed the payments in some cases. The State of Massachusetts has recovered a total of $46.4 million in natural resources damages at ten sites, i.e., an average of $464,000 per site (MAOEEA, 2012).
In summary, the potential for NRD claims to be brought is site specific, and an NRD payment is not obtained in every case. The remedy and at least a portion of the NRD claim are interrelated, such that if more contamination is removed during the remedy, it is likely to reduce the damages calculated.
TABLE 5-1 Select Natural Resource Damage Settlements
| Site | Impacts | NRD Action and Amount |
| Kennecott Utah Copper mining site in West Jordan Valley, UT | Surface and groundwater southwest of Salt Lake Valley contaminated by metals from mining |
1. Kennecott paid $37 million ($9 million plus letter of credit for up to $28 million). 2. Extracted and treated high metals concentration portion of groundwater plume (i.e., performed more remediation than might be required by a CERCLA remedial action). 3. Conveyed 617 acres of land to The Nature Conservancy to resolve its liability for NRD (331 acres and an additional ~286 acres of land as a donation) and paid TNC $175,000 for the purpose of preserving and managing such land. 4. The final restoration plan is to construct two drinking water treatment plants using reverse-osmosis technology, provide a minimum of 8,235 acre feet per year of drinking water, contain the spread of contamination in the deep aquifer and remediate it within the “affected area,” and integrate the CERCLA remedial response with the NRD Consent Decree provisions. |
| Ensign-Bickford Trojan Facility, Utah | Explosives manufacturing allegedly created a groundwater plume extending approximately three miles from the plant | Ensign-Bickford paid Utah $2.58 million to replace the water that would have been available had the groundwater not been contaminated. |
| Six landfills near Bloomington, Indiana | Groundwater contamination | CBS Corp. paid the Interior Department $1.88 million to restore natural resources that have been injured by ongoing releases of PCBs from the six sites. |
| South Valley Superfund facility, Albuquerque, NM | Groundwater contamination | New Mexico sought nearly $1.2 billion in damages from General Electric, but the 10th circuit court of appeals dismissed the state’s claim in part because they had difficulty envisioning any significant loss-of-use damage. |
| Site | Impacts | NRD Action and Amount |
| Industri-Plex Superfund facility in Woburn, MA | Landfill/waste disposal site contaminated the groundwater | Bayer Crop Science Inc. paid $4.25 million to federal and state natural resource trustees to resolve claims for damages. |
| Mass. Military Reservation | Munitions testing contaminated a portion of the Cape Cod sole source aquifer | Textron Systems Corp., a contractor to the Army, agreed to a $1 million NRD settlement (http://www.mass.gov/eea/land-use-habitats/antural-resource-damages/mass-nrd-cases/textron-nrd-settlement.html). |
CONSEQUENCES FOR WATER UTILITIES
Over 50 percent of potable water in the United States is derived from underground sources (USGS, 2003). In general, groundwater offers a relatively clean water resource that is low in organic content (i.e., natural organic matter) and often suitable for drinking purposes with minimal treatment. Exceptions to this are sources that are affected by high levels of natural organic matter (e.g., some Florida aquifers), naturally occurring elevated levels of dissolved inorganics (e.g., some Arizona aquifers), and leached nitrogen from agricultural and animal raising activities (e.g., many shallow aquifers across the country). In addition, releases of hazardous substances to the subsurface have resulted in deleterious impacts to groundwater resources used as a source of potable water, as discussed in Chapter 2. The magnitude of the impacts to water utilities from these sources is not well defined. However, there are many examples of significant impacts to sole source aquifers from hazardous waste sites where treatment is required if the groundwater is to be used as a potable supply. In a number of cases, wellhead treatment is an integral part of the remedial action approved under CERCLA (e.g., San Gabriel Valley, San Fernando Valley, and Bethpage Navy Plant). Box 5-5 discusses two of these cases, along with two others from Southern California, that illustrate the range of situations in which drinking water utilities find themselves with respect to a contaminated groundwater source.
The U.S. Bureau of the Census projects that the U.S. population will grow from 310 million in 2010 to 439 million in 2050 (Vincent and Velkoff, 2010). This growth may force many potable water providers to use impaired water sources, including aquifers contaminated with anthropogenic hazardous chemicals. The added treatment costs are often fully or partially offset by the parties responsible for the contamination, when they can be identified.
BOX 5-5
Comparison of Southern California Groundwater Contamination Cases
Four large-scale groundwater contamination cases in Southern California illustrate the diversity of issues that are faced by water utilities drawing from contaminated aquifers. This box compares the San Fernando Valley, the San Gabriel Valley, the Orange County, and the Santa Monica cases in terms of water supply and demand; the extent of contamination; the choice of remedy and cost; and the rationale and factors affecting the selected remedy. Litigation is ongoing in some of these cases, which have not reached resolution. The summaries are drawn from four presentations given at the second meeting of the Committee in May 2010, and do not reflect more recent happenings.
The common features of the four basins include their geographic location (all are within a 50-mile radius); the production of water from unconsolidated sand, silt, and clay strata; and the servicing of millions of people from groundwater resources. Also, VOCs have been the principal contaminants of concern among these cases, with multiple PRPs named in each case. The contamination discovery dates in all four cases were in the late 1970s and early 1980s, except for the Santa Monica case which was in the mid 1990s. The entities responsible for delivering water to the public have been faced with the urgency of the task on the one hand and with the slow progress in carrying out the investigation and the design, construction, and operation of the groundwater remediation systems on the other hand, especially when the pace of the required activities depends on funding and regulatory requirements.
In the San Gabriel Valley case, it was alleged that numerous companies were PRPs and the site was placed on the NPL. While EPA was working with the California Regional Water Quality Control Board to carry out the CERCLA process, the water supply problem needed to be solved. Consequently, wellhead treatment was instituted for treating VOCs in the groundwater. When other contaminants such as perchlorate and nitrates were detected, many systems were shut down and the San Gabriel Watermaster took charge of the activities to avoid interruption in water delivery to the public. The Watermaster was faced with challenges including how to obtain funding from the PRPs to pay for the project (at an approximate cost of $200 million) and receiving approval from the California Department of Public Health under Policy Memo 97-005, which is required after the treatment system is built. Progress has been made at the San Gabriel Valley Operable Units including treatment facilities being built and operated and ongoing cost recovery.
In the Santa Monica case, 45 companies were tentatively identified as PRPs
The systems used by water utilities to treat anthropogenic chemicals in groundwater are required to meet all applicable drinking water standards mandated by the SDWA as well as any additional requirements from the primacy agency (i.e., the state or local authority). As discussed earlier in the chapter, drinking water standards continue to evolve as more chemicals
including several oil companies that were named as the principal contributors of the MTBE and tert-butyl alcohol (TBA) detected in the water supply. After extensive investigations, the City attempted to fund the project by bringing the PRPs into the process voluntarily and working through engineering committees. When this process failed, the City sued and settled with the major PRPs for $231 million to pay for water replacement, monitoring, and the design, construction, and operation of the groundwater treatment system. The GAC treatment system became operational in December 2010 with the objective of treating MTBE to less than 3 μg/L and TBA to less than 1 μg/L.
In Orange County, when VOC contamination was detected in the shallow aquifers, the Orange County Water District (OCWD) became involved in the investigation and development of remedial approaches because of its ultimate responsibility in delivering water to the public. The OCWD’s approach relied on containing the plumes to prevent migration laterally and to the deeper drinking water aquifers by extracting 5,000 to 6,000 AF; treating the water by GAC, advanced oxidation, and ion exchange; and discharging in upgradient areas—all at a capital cost of $40 million and annual O&M cost of $6 million. The OCWD passes this cost to its customers but will attempt to recover the cost from PRPs through litigation. The OCWD has not gone through the Policy Memo 97-005 process because it is trying to capture the contaminants before they enter the production wells.
Finally, in the San Fernando Valley Basin where the Los Angeles Department of Public Works is responsible for delivering water to 4 million people, numerous potential sources of groundwater contamination and the emergence of new chemicals of concern such as hexavalent chromium and 1,4-dioxane have been identified. Although only approximately 11 percent of the drinking water is supplied from groundwater, the majority of this amount is produced from the San Fernando Valley Ground Water Basin. The investigations (according to the RI/FS) have shown that uncontaminated zones for drinking water extraction are scarce and under threat of impact in the next six to ten years. Thus, wellhead treatment is planned at a capital cost of approximately $704 million and an annual O&M cost of $53 million (which although high, is less than the cost of imported water). The 97-005 Policy process will need to be followed once the treatment system is built.
In the Southern California cases, the urgency of water delivery to the public combined with funding and regulatory requirements has driven the projects toward the implementation of wellhead treatment. The Orange County case is an exception because contaminant plumes have not reportedly penetrated into the deep aquifers. In the other three cases, however, there is no choice other than wellhead treatment to recover the use of the groundwater, as most of the producing aquifers are already contaminated.
are detected due to the improvement of analytical methods, as occurrence surveys indicate the presence of these compounds in potable sources (e.g., USGS studies), and as toxicological research uncovers acute or chronic human health effects at lower concentrations. Although occurring at a relatively slow pace (only a few new chemicals are regulated under the SDWA
every five years), this continuous evolution of drinking water standards poses a challenge for wellhead treatment system configuration. For example, at sites in the San Fernando Valley, treatment systems were initially designed to remove TCE and PCE, using either air stripping or granular activated carbon (GAC). However, subsequent testing revealed the presence of less volatile and less adsorbable chemicals at the Glendale NPL site (e.g., 1,2,3-trichoropropane), which required modifications to the existing groundwater treatment systems and caused an increase in operating costs.
In addition to future changes in the drinking water standards for individual chemicals, EPA is also considering alternative regulatory strategies that may affect both remedial objectives as well as well-head treatment standards. For example, in early 2010 EPA administrator Lisa Jackson announced a new drinking water strategy to expand public health protection more cost effectively by addressing contaminants with similar modes of action in groups (EPA, 2010e). The first group proposed for regulation under this strategy focuses on carcinogenic VOCs, including eight that have been previously regulated by EPA and up to eight more VOCs that were on the CCL. The most recent information from EPA indicates that the drinking water standards for the previously regulated compounds may be made more strict within the new regulatory strategy to recognize improved analytical methods (e.g., the TCE standard as discussed earlier) (EPA, 2011j).
State Policies for Wellhead Treatment for Vulnerable or Impacted Water Supply Aquifers
In the context of groundwater contamination from a wide range of anthropogenic sources, various states have developed policies to establish appropriate methods to protect aquifers so they can continue to be used as water supplies. For example, in 1997 California developed an effective policy for use of groundwater resources considered to be “extremely impaired,” defined as having levels of chemical contaminants several times higher than established drinking water standards (California Policy Memo 97-005, Policy Guidance for Direct Domestic Use of Extremely Impaired Sources). The general philosophy of this guidance is that the best quality sources of water reasonably available to a water utility should be used for drinking, and wherever possible lower quality water source waters should be used for non-consumptive uses that pose lower health risks such as irrigation, recreation, or industrial uses. If a permit for the use of a severely impaired source is to be approved by the California Department of Public Health, the source of the contamination must be controlled to prevent contaminant concentrations from rising, and dependence upon treatment should be minimized. Water treatment facilities should be designed and
operated to meet the MCLG (when lower than the MCL) where this can be achieved in a cost-effective manner.
Shortly after this policy was created, California adopted a program to assess and protect sources of drinking water (called Policy Memo 97-005, CDHS, 1997). In the drinking water source assessment, an area around a drinking water source is delineated through which contaminants could move and reach the drinking water supply. In addition, the drinking water source assessment includes an inventory within the delineated area of activities that could lead to the release of contaminants. In California, less than a dozen permits have been approved since the initiation of the 97-005 policy, and the approval process can be lengthy (Collins, 2011). It should be noted that California recently (2012) declared that the 97-005 policy is no longer considered obligatory, and is considered as guidance only, since the policy was not subjected to legislative review and approval. Nonetheless, the risk analysis components of the 97-005 process provide the state with some assurance that the well-head treatment system will consistently remove all chemicals of concern to appropriate drinking water standards or below, and that the approved treatment system is capable of dealing with chemicals likely to be present in the aquifer, in addition to the chemicals designated for removal in the permit.
Impacts of Groundwater Contamination on Aqueduct Storage and Recovery or Conjunctive Use Programs
In the more arid areas of the country, groundwater aquifers are increasingly being considered as potential sites for storage of excess water that may become available during wet periods or transportable from wetter areas for subsequent use during periods of shortage (so-called water banking or conjunctive use). Traditionally, water agencies have built percolation ponds to divert spring stream runoff for groundwater recharge. More recently municipal wastewater that has been treated sufficiently can be used to recharge aquifers, although the increased mineral concentrations of wastewater can limit the future use of the groundwater. Although expensive and energy intensive, aquifer storage and recovery (ASR) is an increasingly attractive strategy for many areas. In 2005 there were over 70 active ASR projects in 15 states (NRC, 2008). Various forms of ASR are being practiced in the Kern, San Gabriel, and San Fernando basins of California; Texas and Arizona have similar programs.
Conjunctive use can expose the stored water to potential contamination from anthropogenic chemicals that are present in the unsaturated zone above the aquifer, posing additional treatment challenges and disputes on liability for cleanup when the water is withdrawn from the aquifer by the utility. Extraction wells must have wellhead treatment if the groundwater
is contaminated, and such treatment strategies must be considered at the time a basin is planned for such use. Even if the water extracted is used for non-potable purposes, current state and federal laws support the objective of cleanup to drinking water standards.
It is highly likely that a growing number of groundwater basins throughout the United States with detectable amounts of contaminants will be considered for ASR. It is in the public interest to develop an approach to managing these aquifers that can establish the human health risk across the whole range of potential uses of the groundwater.
Wellhead Treatment
The objective of wellhead treatment is to remove natural and anthropogenic contaminants to levels consistent with standards established for the intended use. If the intended use is human consumption, the water must be rendered safe as defined by the SDWA (i.e., meet all federal MCLs or other state drinking water standards). The permitting process for the treatment system by the local authorities is expected to ensure that the treatment system is capable of complying with all the applicable regulations under SDWA. In this type of application of wellhead treatment, the water also needs to be acceptable to the consuming public and thereby may require the removal of chemicals to levels below MCLs. The burden is on the treatment system owner to obtain such regulatory approval for the design, construction and operation of a groundwater treatment system.
The Current State of Technologies for Groundwater Treatment
Above-ground treatment of contaminated groundwater is often carried out at the well head or at a location where several wells are pumped into a central treatment facility. The latter option, often utilized to achieve economies of scale, is more typical for treatment systems where the intended end use is human consumption. Many different treatment technologies are available depending on the target contaminant. Removal of organic pollutants is often accomplished through volatilization, adsorption, or oxidation whereas the removal of inorganic contaminants is accomplished through ion exchange, adsorption, or precipitation followed by solids separation. The following is a list of commonly applied treatment technologies used for cleanup of groundwater impacted with organic chemicals and toxic metals:
• Packed Tower Aeration
• Diffused Bubble Aeration
• Tray Aeration
• Granular Activated Carbon Adsorption
• Ion Exchange
• Advanced Oxidation using Ozone
• Advanced Oxidation using Ultraviolet Light
Table 5-2 lists commonly applied groundwater treatment technologies along with some limitations to their use. Among the listed technologies, packed tower aeration and granular activated carbon are most often used for ex situ groundwater treatment for the most common chlorinated solvents, including TCE and PCE. Anion exchange, advanced oxidation, and biological processes are becoming more common as the treatment objectives for groundwater are being expanded to include multiple contaminants as well as due to the reduction in drinking water standards for treated water.
TABLE 5-2 List of Commonly Used Groundwater Treatment Technologies
| Technology | Relative Cost | Technology Applicability | Technology Limitations |
| Packed Tower Aeration | Low | Moderately to highly volatile compounds characterized with high Henry’s Law Constant (e.g., TCE and PCE) |
• Tower height usually limited to 20 or 30 ft • Air-to-water ratio usually limited to 150:1 • Scaling control (acid addition) needed for highly alkaline and hard waters |
| Multi Stage Bubble Aeration | Low | Highly volatile compounds only | • High degree of removal is more difficult to achieve |
| Tray Aeration | Low | Highly volatile compounds only |
• High degree of removal is more difficult to achieve |
| Granular Activated Carbon Adsorption (GAC) | High | Moderately to strongly adsorbable compounds characterized with favorable Freunlich Constant (e.g., PCE) |
• Natural organic matter reduces removal efficiency • Cost is high • High degree of removal may not be achieved at low influent concentrations |
| Technology | Relative Cost | Technology Applicability | Technology Limitations |
| Advanced Oxidation with Ozone (O3-AOP) | High | Smaller organic molecules (e.g., 1,4-dioxane) |
• Presence of bromide in the water results in bromate formation • Excess peroxide must be quenched |
| Advanced Oxidation with Ultra Violet Light (UV-AOP) | High | Smaller organic molecules (e.g., 1,4-dioxane) |
• Higher dosages required compared to UV disinfection • Excess peroxide must be quenched |
| Biological Treatment | Low to Moderate | Anaerobic processes can be used to remove perchlorate |
• Complex treatment process • Requires substrate (e.g., ethanol) addition • Requires polishing treatment |
| Strong Base Anion Exchange | Moderate to High | Charged anions (e.g., chromate, arsenate, or perchlorate) |
• Generates concentrated brine stream that may be hazardous • Interference from other anions (e.g., sulfate) |
| Weak Base Anion Exchange | Moderate to High | Charged anions (e.g., chromate, arsenate, or perchlorate) |
• May require pH adjustment • Generates concentrated brine stream that may be hazardous • Exhausted resins in single pass applications may be hazardous |
| Reduction Coagulation Filtration | High | Chromate |
• Significant residual stream |
| Electrodialysis/ Electrodialysis Reversal | High | Moderate degree of removal for a large variety of contaminants |
• Significant concentrate stream (up to 25%) • May require pH adjustment |
Treatment facilities targeted for the production of potable water from a contaminated groundwater source are generally designed with technologies capable of removing contaminants well below drinking water standards and are thus designed conservatively. For example, some water utilities target non-detect levels of TCE in the treated water even though the applicable federal standard in 2012 is 5 μg/L; current analytical methods are routinely capable of measuring concentrations below 1.0 μg/L. In some instances, water utilities will attempt to the remove contaminants that are not currently regulated, but where state standards or compliance levels have been established or are likely in the future. For example, at a groundwater treatment facility located near the Tucson Airport where TCE-contaminated groundwater has been treated for several years using packed tower aeration, operators are installing a UV-AOP treatment process for removal of 1,4-dioxane, which has been recently detected at low levels. Similarly, the groundwater treatment facilities located in the San Fernando Valley in California (Box 5-5) may be upgraded to include treatment for perchlorate and hexavalent chromium.
Challenges for Wellhead Treatment
Wellhead treatment for removal of chemicals typically present at hazardous waste sites is not a new challenge for water utilities. Treatment systems have been in place for more than 30 years in locations where VOCs impact groundwater sources (e.g., New Jersey, California, Arizona). Despite a long history of using specific treatment technologies (such as packed tower aeration or GAC) for removing organic chemicals from groundwater, the presence of multiple organic chemicals in groundwater resources poses a number of treatment challenges. First, the design of water treatment systems must be based on estimates of the number of chemicals of concern present and their likely maximum concentrations. Accurate estimates of these design values are sometimes difficult to come by, given limited site characterization data or limited knowledge of the nature and extent of the contamination present in the aquifer and within the capture zone of the water supply well. Groundwater fate and transport modeling is inherently limited in terms of accurate predictions, but is currently the best tool available to estimate these design parameters.
Each of the individual treatment systems now in use are subject to operational upsets. For example, an air stripping tower may be effective at removing TCE but should a less volatile organic chemical occur, such as 1,2-DCA, the removal efficiency of the system may not be sufficient to meet the treatment objectives. For GAC, the appearance of compounds less strongly adsorbed may result in more rapid breakthrough of the chemical
and thus increase the usage rate for GAC, leading to more frequent replacement and increased costs.
More recently, some organic chemicals have been found in groundwater that are not efficiently removed by either air stripping or GAC. For example, 1,4-dioxane has become a very prevalent contaminant due to its widespread use as a solvent stabilizer (Mohr, 2010). For those utilities who have already installed GAC, this chemical will not be removed effectively, and an additional treatment process is needed. The most widely adopted technology for removing 1,4-dioxane is advanced oxidation, using either the UV/hydrogen peroxide or the ozone/hydrogen peroxide technology. AOP is a more recent addition to the suite of wellhead treatment options for organics, and limited operational experience is available. An additional issue with the use of AOP technologies is the formation of oxidation byproducts that can be problematic. For example, the ozone/hydrogen peroxide technology will convert bromine ion to bromate, which has a 10 μg/L MCL. Other byproducts could also be problematic. Finally, the excess hydrogen peroxide must be destroyed prior to transfer to the water distribution system.
The reliability of any wellhead treatment system to meet treatment goals (usually below MCLs) on a consistent basis is uncertain. Water treatment plants are usually monitored at intervals that reflect the relative constant influent water quality. Often, for VOCs and other regulated contaminants, a running average based on quarterly sampling frequency is used to assess performance. Given the potential variability in influent water quality characteristics, greater frequency of sampling may be appropriate until the reliability of the treatment systems can be confirmed.
***
The decision of whether to perform wellhead treatment versus conducting more conventional groundwater remediation goes beyond the ability of technologies to remove contaminants at the point of use. Indeed, there are significant economic considerations, as wellhead treatment can be roughly as expensive as source or plume remediation, depending on the contaminants involved and the site hydrogeology. Second, it is not yet clear that wellhead treatment is a reliable, long-term strategy for accomplishing exposure cutoff (and it certainly cannot prevent exposure via vapor intrusion). These tensions are illustrated at the Northrop-Grumman/Navy facility in Bethpage, Long Island, New York (see Appendix B). In this case, wellhead treatment of a groundwater plume used as a primary water supply was deemed necessary because full containment of the groundwater plume was thought to be technically infeasible and not cost effective. Nonetheless, affected water districts have lobbied for plume remediation because the plume
is spreading and there is the chance that MCLs for the contaminants of concern will be lowered in the near future. This will make wellhead treatment, currently estimated by one water utility to be $128 million, significantly more expensive than plume remediation (estimated to be about $91 million, by the same utility) (Massapequa Water District, 2011). State regulatory authorities have come up with different cost estimates and continue to recommend wellhead treatment over plume remediation because some wells are already contaminated and will have to be treated in perpetuity. It is too soon to tell how the dispute over the proper remediation strategy at the Navy/Northrop-Grumman plume will be resolved.
Sites with subsurface contamination by hazardous materials present economic challenges to responsible parties and people and businesses affected by the contamination. When contamination remains in place above unrestricted use levels, there are direct, out-of-pocket costs of controlling contamination. There are also opportunity costs such as forgone revenue from not being able to use the contaminated site and nearby areas for other endeavors.
Figure 5-1 provides a stylized example of costs associated with a contaminated groundwater site. “Services under baseline” represents water quality in an aquifer absent of contamination. Suppose a release of a hazardous substance occurs at time T0. In this example, it is assumed that 25 percent of the groundwater remains accessible for use with contaminant levels below MCLs, i.e., pumping can continue in some parts of the aquifer. Once contamination occurs, the Recovery Path represents improvements in groundwater quality due to natural attenuation of the hazardous substance.
A treatment program will enhance recovery and is indicated by the Recovery Path with Remedial Action in Figure 5-1. For simplicity, the recovery path begins at time T0, although this would only be the case where a contaminant release was instantaneously known and remediation efforts were initiated immediately to control damages. Losses in groundwater services after remediation are shown by the shaded area and can only be fully revealed over time. Potentially responsible parties can compensate affected parties for residual losses (shaded area) or they can provide equivalent services known as compensatory restoration (dotted area). Compensatory restoration is typically designed so that the benefits of the compensatory restoration just offset the loss (the dotted area just equals the shaded area). Note, the hatched area constitutes losses that occurred prior to the implementation of CERCLA; some trustees submit claims for these losses and others just submit claims for losses subsequent to the implementation of CERCLA.
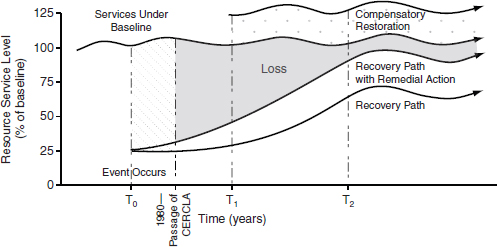
FIGURE 5-1 Stylized diagram of economic losses.
A key issue at sites that have recalcitrant and/or poorly accessible contaminants that prevent site closure is that costs to the potentially responsible party and affected parties can continue indefinitely, indicated by the arrows on the right side of Figure 5-1. Even with a remedial action plan, groundwater quality may not be returned to baseline conditions for a very long time. This residual and recalcitrant contamination increases the cost uncertainties faced by all parties associated with such a site.
Potentially Responsible Party Costs
The costs faced by a potentially responsible party are the costs of remedial actions and litigation expenses (discussed in a previous section). Remedial action costs are influenced by available cleanup technologies, the extent of contamination, and the health effect pathways of the contaminants. The cost of conducting remediation is closely tied to the cost of energy and to the cost of landfill space. As the cost of energy rises, more energy-intensive remediation methods like excavation and thermal treatment will become less competitive with methods that use or enhance natural processes.
The time it takes from the initiation of the first cleanup action until construction completion has varied since the initiation of Superfund in 1980, but was typically about nine years for NPL facilities in the late 1990s (EPA, 2002b), while the average economic cycle in the United States from 1945 to 2001 has been about 67 months or about 5.6 years (NBER, 2010). Thus, a typical cleanup at a Superfund facility can bridge two or three cycles of the national economy, during which annual cleanup and monitoring costs can increase or decrease. One factor that could induce such variation is energy
costs. For example, energy costs to operate a pump-and-treat system could have started out high, then dropped, then increased substantially toward the end of the treatment project (EPA, 2001b).
New information about the site can arise after a ROD has been approved, which can affect cleanup costs. This new information can take a number of forms, such as a reduction in the MCL in the face of new toxicity values, the identification of an additional contaminant due to enhanced detection technology, or a new arm of the contamination plume could be discovered. These occurrences could increase cleanup and management costs beyond what the PRP expected at the time a ROD is approved.
Costs are only one of the nine criteria of the NCP, and the cost criterion does not require any consideration of incremental or marginal costs for additional cleanup or risk reduction. Thus, at complex sites where UU/UE levels cannot be attained, costs may increase over time with little or no reduction in risks. It is proposed in Chapter 7 that when a site reaches the point where the marginal cost of additional cleanup actions increases but there is no corresponding reduction in contaminant concentrations and hence risk, then the cleanup expenditures could be better spent elsewhere in the economy (such as investing in developing more effective remediation technologies or to improve conditions and reduce risks at other sites).
Measurement of the costs discussed above is hampered by limitations in available data. Barth and McNichols (1994) note that “actual cleanup costs generally are not known; at most sites cleanup is incomplete and there is no central tracking of cleanup costs” (p. 177). Hamilton and Viscusi (1999) reported cleanup costs for a sample of 19 NPL facilities and found the average cost per site with soil and or groundwater contamination was $25.7 million (in 1993 dollars). These cleanup costs may be lower-bound costs because Hamilton and Viscusi (1999) did not include legal and management expenses incurred by the responsible parties and regulators in securing acceptance of the ROD. For comparison, in 2009 GAO reported that, based on EPA data, the average expenditure for Superfund facilities through 2007 was $9.9 million per ROD (note that there can be multiple RODs at one Superfund facility) (GAO, 2009). These are total costs and not the key cost information—marginal costs—which indicate how costs vary with the amount of remediation accomplished.
In addition to the paucity of peer-reviewed literature on the cost of cleaning up NPL sites, firm-specific and site-specific cleanup costs are difficult to obtain. Barth and McNichols (1994) reported that 44 percent of Superfund facilities have more than one potentially responsible party and 52 percent of potentially responsible parties have more than one site where they have responsibility. These conditions make it difficult to obtain site-specific cost data for individual firms from public records and it is also difficult to aggregate cost data across firms for specific sites.
Despite the limited number of empirical cost studies, the available literature does present a few insights. First, Hamilton and Viscusi (1999) found that 20 percent of the sites account for 50 percent of the cleanup costs. Gupta et al. (1996) suggest that the higher cost sites may be associated with greater risks to human health and the environment. Overall, these results suggest cost uncertainties may be more important for complex sites that are likely to incur large cleanup costs or with large risks to human health and the environment. The number of studies, however, is too few to draw any firm conclusions.
Affected Parties
Costs to affected parties, be they businesses or individuals, can arise during cleanup activities and from residual contamination that is left after completion of active cleanup activities. These costs include claims for health effects, property damage, and lost business income. Furthermore, because complete protection from the contaminants might not occur instantaneously and compensation paid by responsible parties may not occur for years, affected parties can be faced with interim, out-of-pocket costs of providing self-protection from the contamination.
Costs for dealing with health effects can, if the concentrations of a contaminant are of sufficient magnitude, potentially be incurred where firms or households use groundwater that is contaminated by a site and/or if the subsurface contamination results in vapor intrusion into nearby buildings where people work or live. Immediate costs may include protective actions such as buying bottled water or installing ventilation systems in buildings, while long-term costs may arise from health care expenses as a consequence of past exposure to contaminants.
Property costs occur because nearby property values may be diminished by the contamination at a site. Note that if health risks are even a perceived concern, this might serve to diminish property values. Conversely, if the potentially responsible parties’ protection efforts are effective, or effective self-protection was installed in a property, this may remove some of the property value diminution (Boyle et al., 2010; Kiel, 1995). However, property values still may be diminished due to a price stigma from the nearby contamination site (McCluskey and Rausser, 2003; Messer et al., 2006). This price diminution due to the stigma can be in addition to any associated health costs.
As with the costs to potentially responsible parties, the costs of residual contamination to affected parties described above are challenging to address. The goal of any damage claim is to measure these costs as accurately as possible and to seek compensation as quickly as possible. However, accurate measurement is a topic of considerable academic debate.
Site Development Impacts
The link between future economic development on a site and residual groundwater and soil contamination was dramatically illustrated when the local banks refused to lend money to Love Canal home owners and commercial entities (Niagara Gazette, 1979). Similarly, in 1991 widespread contamination of groundwater by chlorinated solvents was discovered in Wichita, Kansas, encompassing approximately 5½ square miles and 8,000 parcels of land and affecting more than 550 businesses and hundreds of residential properties (ICMA, 2006). This contamination undercut the City’s tax base, and banks were reluctant to finance transactions within the site (Weinfield, 2007). At El Toro Marine Corps Air Station in Irvine, CA, the Navy was able to commence transferring the closing base to the public only after carving out the more contaminated areas and precluding them from sale.
Some of these risks have been lessened by federal legislation. For example, CERCLA was explicitly amended to exempt banks that hold mortgages on property as secured lenders from CERCLA cleanup liability, if they do not exercise decision-making control over a property’s environmental compliance (EPA, 2009c). In addition, the Small Business Liability Relief and Brownfields Revitalization Act exempts from liability bona fide prospective purchasers who purchase property with knowledge of existing contamination if they (1) are not responsible parties; (2) acquire the property after January 11, 2002; (3) establish that all disposal of hazardous substances occurred before they acquired the facility; (4) make all appropriate inquiries into previous ownership and prior uses of the property; and (5) are not affiliated with a responsible party (EPA, 2009c).
It should be noted that environmental due diligence prior to property acquisition is now standard practice in everyday transactions and when mergers and acquisitions are contemplated (ASTM, 2006). Typically, the acquiring company and lenders seek certainty concerning the magnitude of future liabilities from the selling company. As a result, mergers and acquisitions may trigger action to obtain regulatory certainty concerning cleanup efforts, the adoption of indemnification allocating future liability among the purchaser and seller, renegotiation of the sales price, or set asides of funds for cleanup or other activities. Furthermore, in 2011 six federal financial institution regulatory agencies proposed credit risk retention rules for commercial real estate property where environmental hazards, such as groundwater contamination, may potentially jeopardize the value of commercial real estate as well as the borrower’s ability to repay a loan (OCC et al., 2011). Thus, even though there is no longer the extreme avoidance of lending for contaminated properties that existed when hazardous waste issues first arose, the continuing presence of chemicals above unrestricted
use levels is considered in transactions and may impact future economic development.
Fortunately, the liabilities discussed above that arise from having residual contamination in place can be offset by benefits that may result from redevelopment of contaminated land (EPA, 2011k). Regulators tend to use residential land use assumptions at sites because the goal of groundwater cleanup is often to return groundwater to beneficial uses (EPA, 1990, 2009d). Reuse of contaminated properties (so-called Brownfields redevelopment) or for some other use that does not require residential land use is becoming more common and is supported by federal policies (EPA, 2011L; ASTM, 2010). At Brownfields, regulatory policies and financial grants are often complemented by state and local incentives for property use or even employment.7 Indeed, some states promote Brownfields redevelopment even in the absence of significant environmental problems.
CONCLUSIONS AND RECOMMENDATIONS
This chapter has focused on the implications of leaving contamination in place at hazardous waste sites at levels above those allowing for unlimited use and unrestricted exposure. All of the risks discussed in this chapter should be considered carefully before choosing a remedy that leaves contamination in place.
Research is needed to build a more robust understanding of the potential for failure in hydraulic and physical containment systems. Two types of studies are recommended: (1) a “data mining” study in which site-specific evaluations such as RSEs and five-year reviews are analyzed to provide a general characterization of the frequency, mechanisms, responses, and costs associated with the failure of hydraulic and physical containment, permeable reactive barriers, and MNA, and (2) consistent with previous NRC recommendations (2007), establish field experimental stations at operating waste sites to assess the long-term performance of physical containment systems.
Implementing institutional controls at complex sites is likely to be difficult because it requires long-term monitoring of site conditions and potential exposures and maintenance of land use restrictions that may be in tension with the development preferences of local authorities. Although EPA has developed a number of measures to improve the reliability, enforceability, and funding of institutional controls, their long-term efficacy has yet to be definitely determined. The public who live and work near a
__________________
given site have a direct and enduring interest in ensuring that institutional controls are followed and are more likely than others to know whether institutional controls are working.
Regulators and federal responsible parties should incorporate a more significant role for local citizens in the long-term oversight (i.e., monitoring and enforcement) of institutional controls. At a minimum, those who live, work, study, play, or otherwise occupy residences, commercial buildings, or industrial facilities near hazardous waste sites should be notified of institutional controls, including the specific reasons for them. A national, searchable, geo-referenced institutional control database covering as many regulatory programs as practical as well as all federal sites would ensure that the public is notified of institutional controls. Methods can also be developed to encourage citizens to report to regulators any developments that might undermine the protectiveness of such institutional controls and thus improve oversight (see Chapter 7).
New toxicological understanding and revisions to dose-response relationships will continue to be developed for existing chemicals, such as TCE and PCE, and for new chemicals of concern, such as perchlorate and perfluorinated compounds. The implications of such evolving understanding are multi-fold, including identification of new or revised MCLs (either more or less restrictive than existing ones), potentially leading to a determination that the existing remedy at some hazardous waste sites is no longer protective of human health and the environment.
Modification of EPA’s existing CERCLA five-year review guidance would allow for more expeditious assessment of the protectiveness of the remedy based on any changes in EPA toxicity factors, drinking water standards, or other risk-based standards. Although EPA’s existing five-year review guidance provides that cleanup levels will be evaluated when a new ARAR for drinking water has been developed and/or new or revised toxicological information is available, the existing process is slow. Furthermore, as EPA accelerates its national toxicity assessment process, there will be a large number of chemicals at hazardous waste sites whose protectiveness will need to be reassessed. EPA should ensure that it has adequate resources dedicated to this process.
Careful consideration of the vapor intrusion pathway is needed at all sites where VOCs are present in the soil or groundwater aquifer. Although it has been recognized for more than a decade that vapor intrusion of volatile chemicals is a potential exposure pathway of concern, a full understanding of the risks over time and appropriate methods for characterizing them are still evolving.
Mitigation strategies such as subslab depressurization can prevent va-
por intrusion exposure. In most cases, the cost of building mitigation into new construction is significantly less than the cost of repetitive sampling. Furthermore, such systems reduce exposure to naturally occurring radon. As a precautionary measure, vapor mitigation could be built into all new construction on or near known VOC groundwater plumes; this could be imposed proactively as part of local or state building codes or other requirements or imposed as institutional controls at regulated sites. In either situation, vapor mitigation systems require monitoring over the long-term to ensure that they are operating properly.
As populations increase and industrial demands for high-quality water also increase, the demands placed on groundwater supplies will increase. Contaminated aquifers have been and may well be used more extensively in the future to augment supplies of uncontaminated water. Wellhead treatment may be an optimal remedy for low concentrations of contaminants in potential water supplies. Current wellhead treatment technologies are materials intensive and are not energy efficient. Improved and efficient water treatment technologies should be developed both for more cost-effectively destroying VOCs and recalcitrant organic compounds as well as for removing toxic metals. An emphasis should be placed on technologies that treat a broad spectrum of chemicals.
Aitchison, E. W., S. L. Kelley, J. J. Pedro-Alvarez, and J. L. Schnoor. 2000. Phytoremediation of 1,4-dioxane by hybrid poplar trees. Water Environment Research 72(3):313-321.
Ando, A. W., M. Khanna, A. Wildermuth, and S. Vig. 2004. Natural Resource Damage Assessment: Methods and Cases. Waste Management and Resource Center, Illinois Department of Natural Resources. http://www.istc.illinois.edu/info/library_docs/RR/RR-108.pdf.
Army. 2011. Environmental Liabilities. http://aec.army.mil/usaec/cleanup/e100.html.
ASTM (American Society for Testing and Materials). 2006. ASTM Standards on Environmental Site Assessments For Commercial Real Estate, 5th Edition, which includes ASTM E 1527–00, Standard Practice for Environmental Site Assessments: Phase I Environmental Site Assessment Process, ASTM E1903-11 Standard Practice for Environmental Site Assessments: Phase II Environmental Site Assessment Process, and ASTM E 1528 Transaction Screen Process.
ASTM. 2010. ASTM Standard Classification of Environmental Condition of Property Area Types for Defense Base Closure and Realignment Facilities.
ATSDR (Agency for Toxic Substances and Disease Registry). 1989. Public Health Statement for n-Nitrosodimethylamine. CAS 62-75-0. Altanta, GA: ATSDR.
ATSDR. 2005. Toxicological Profile for Naphthalene, 1-Methylnaphthalene, and 2-Methylnaphthalene. http://www.atsdr.cdc.gov/toxprofiles/tp67.pdf.
ATSDR. 2008. Draft Toxicological Profile for Chromium. http://www.atsdr.cdc.gov/toxprofiles/tp7.pdf.
Barnes, K. K., D. W. Kolpin, E. T. Furlong, S. D. Zaugg, M. T. Meyer, and L. B. Barber. 2008. A national reconnaissance of pharmaceuticals and other organic wastewater contaminants in the United States - (I) Groundwater. Science of the Total Environment 402(2-3):192-200.
Barth, M. E., and M. F. McNichols. 1994. Estimation and market valuation of environmental liabilities relating to superfund sites. Journal of Accounting Research 32(Sup.):177-209.
Beaumont, J. J., R. M. Sedman, S. D. Reynolds, C. D. Sherman, L. H. Li, R. A. Howd, M. S. Sandy, L. Zeise, and G. V. Alexeeff. 2008. Cancer mortality in a Chinese population exposed to hexavalent chromium in drinking water. Epidemiology 19(1):12-23.
BNA. 2006. Daily Environment Report. News 11/15/2006. Drinking Water: Judge Trying Federal MTBE Litigation Rejects Regulatory Standards as Baseline for Injury. http://news.bna.com/deln/display/batch_print_display.adpsearchid=15581702.
BNA. 2010. Daily Environment Report. News 08/06/2010. Drinking Water: Oil Companies Agree to Settle Claims Over MTBE Contamination for $42 Million. http://news.bna.com/deln/display/batch_print_display.adp?searchid=15581702.
Bowman, R. H., P. Miller, M. Purchase, and R. Schoellerman. 2003. Ozone-peroxide advanced oxidation water treatment system for treatment of chlorinated solvents and 1,4-dioxane. Contaminated Soils 8:1-12.
Boyle, K. J., N. Kuminoff, C. Zhang, M. Devanney, and K. P. Bell. 2010. Does a property-specific environmental health risk create a ‘neighborhood’ housing-price stigma? Arsenic in private well water. Water Resources Research 46:W03507.
Buszka, P. M., D. J. Yeskis, D. W. Kolpin, E. T. Furlong, S. D. Zaugg, and M. T. Meyer. 2009. Waste-indicator and pharmaceutical compounds in landfill-leachate-affected ground water near Elkhart, Indiana, 2000-2002. Bulletin of Environmental Contamination and Toxicology 82(6):653-659.
CalEPA. 2011. California’s Vapor Intrusion Mitigation Advisory. Sacramento, CA: CalEPA Department of Toxic Substances Control.
Carrara, C., C. J. Ptacek, W. D. Robertson, D. W. Blowes, M. C. Moncur, E. Sverko, and S. Backus. 2008. Fate of pharmaceutical and trace organic compounds in three septic system plumes, Ontario, Canada. Environmental Science & Technology 42(8):2805-2811.
CDHS (California Department of Health Services). 1997. Policy Memo 97-005 Policy Guidance for Direct Domestic Use of Extremely Impaired Sources. www.cdph.ca.gov/certlic/drinkingwater/Documents/DWdocuments/memo97-005.pdf.
Center for Justice & Democracy. 2007. Fact Sheet: Environmental Tort Lawsuits: Holding Polluters Accountable. January 23, 2007. http://centerjd.org/content/fact-sheet-environmental-tort-lawsuits-holding-polluters-accountable.
Christopher, J. P., B. K. Davis, J. M. Polisini, and M. J. Wade. 2005. Designation of Naphthalene as a Carcinogen: Risk Assessment for Inhalation Exposure Pathways at Hazardous Waste Sites. http://www.dtsc.ca.gov/AssessingRisk/upload/Naphthalene_Handout.pdf.
Claims Journal. 2004. Tucson-Area Water Contamination Victims Triumph in Ariz. Appeals Court Ruling Against Insurer, October 1, 2004. http://www.claimsjournal.com/news/west/2004/10/01/46444.htm.
Coate, C. J., K. Frey, and N. Sakuvich. 2012. Environmental Accounting and Reporting 101. September 5, 2012. http://www.newaccountantusa.com/newsfeat/ip/ip_environmental.html.
Coates, J. D., and L. A. Achenbach. 2004. Microbial perchlorate reduction: Rocket-fuelled metabolism. Nature Reviews Microbiology 2:569-580.
Coates, J. D., and L. A. Achenbach. 2006. In situ bioreduction and removal of ammonium perchlorate. Final report for the Strategic Environmental Research and Development Program.
Collins, H. 2011. Water Quality Regulations and Permitting Challenges for Groundwater Treatment Systems: California Policy 97-005. Presented at the AWWA Water Quality Technology Conference, November 2011.
Cox, C., and E. Douthett. 2009. Further evidence on the factors and valuation associated with the level of environmental liability disclosures. July 1, 2009. http://www.freepatentsonline.com/article/Academy-Accounting-Financial-Studies-Journal/209238515.html.
Cramer, R. J., C. Yates, P. Hatzinger, and J. Diebold. 2004. Field demonstration of in situ perchlorate bioremediation at building 1419. Final report for the Strategic Environmental Research and Development Program. CU-1163-TR-01.
DERP. 2001. Management Guidance for the Defense Environmental Restoration Program. September 21, 2001. http://aec.army.mil/usaec/cleanup/derpguidance0411.pdf.
Dietz, A. C., and J. L. Schnoor. 2001. Advances in phytoremediation. Environmental Health Perspectives Supplements 109(1):163-168.
DoD (Department of Defense). 2001. Policy on Land Use Controls Associated with Environmental Restoration Activities. Memorandum for Assistant Secretaries of the Army, Navy, Air Force, and Director, Defense Logistics Agency. January 17, 2001.
DoD. 2009. DoD Vapor Intrusion Handbook. Washington, DC: DoD Tri-Service Environmental Risk Assessment Workgroup. http://www.clu-in.org/download/char/dodvihdbk200901.pdf.
Doroski, M., K. Anders, and G. Heitzman. 2011. Environmental Energy Alliance of New York State / New York State MGP Soil Vapor Intrusion Database. 27th Annual Conference on Soil, Sediments, Water, and Energy, Amherst, MA, October 17-21.
Drewes, J. E., T. Heberer, T. Rouch, and K. Reddersen. 2003. Fate of pharmaceuticals during groundwater recharge. Ground Water Monitoring & Remediation 23(3):64-72.
ELI. 1999. Environmental Law Institute, Protecting Public Health of Superfund Sites: Can Institutional Controls Meet the Challenge? http://www.elistore.org/reports_popup.aspdid=543&file=/Data/products/d10.01.pdf.
EPA (U.S. Environmental Protection Agency). 1990. NCP Preamble. Federal Register 55:8732.
EPA. 1991a. Guidelines for Developmental Toxicity Risk Assessment. Federal Register 56(234):63798-63826. http://www.epa.gov/raf/publications/pdfs/DEVTOX.PDF.
EPA. 1991b. EPA Superfund Record of Decision: Love Canal OU 9. http://www.epa.gov/superfund/sites/rods/fulltext/r0291165.pdf.
EPA. 1999. Second five-year review: Delaware Sand and Gravel Site, New Castle Delaware. Philadelphia, PA: EPA Region 3.
EPA. 2001a. RCRA Draft Supplemental Guidance for Evaluating the Vapor Intrusion to Indoor Air Pathway. EPA/600/SR-93-140. http://www.epa.gov/wastes/hazard/correctiveaction/eis/vapor/vapor.pdf.
EPA. 2001b. Cost Analyses for Selected Groundwater Cleanup Projects: Pump and Treat Systems and Permeable Reactive Barriers. EPA 542-R-00-013. http://www.epa.gov/tio/download/remed/542r00013.pdf.
EPA. 2002a. Draft Guidance for Evaluation the Vapor Intrusion to Indoor Air Pathway from Groundwater and Soils. Washington, DC: EPA OSWER.
EPA. 2002b. The Superfund process, from site listing to cleanup construction, on average has taken roughly 8 to 10 years. Statement of Marianne Lamont Horinko, Assistant Administrator, Office of Solid Waste And Emergency Response, U.S. Environmental Protection Agency, before the Subcommittee on Superfund, Toxics, Risk And Waste Management. United States Senate (July 31, 2002). http://epw.senate.gov/107th/Horinko_073102.htm.
EPA. 2004a. Performance monitoring of MNA remedies for VOCs in groundwater. EPA/600/R-04/027. Washington, DC: EPA Office of Research and Development.
EPA. 2004b. IRIS Toxicological Review of Naphthalene. http://cfpub.epa.gov/ncea/iris_drafts/recordisplay.cfm?deid=84403.
EPA. 2006. Enforcement First to Ensure Effective Institutional Controls at Superfund Sites at 3-4, OSWER 9208.2.
EPA. 2007. Monitored Natural Attenuation of Inorganic Contaminants in Groundwater, Vol. 1: Technical Basis for Assessment. EPA/600/R-07/139. Ada, OK: National Risk Management Research Laboratory, Groundwater and Ecosystems Restoration Division.
EPA. 2008a. A systematic approach for evaluation of capture zones at pump and treat systems. EPA 600/R-08/003. http://www.epa.gov/nrmrl/pubs/600R08003/600R08003.pdf.
EPA. 2008b. Significant New Use Rules on Certain Chemical Substances. 73 Fed. Reg. 65743-65,752 (November 5, 2008).
EPA. 2008c. Toxic Substances Control Act Inventory Status of Carbon Nanotubes Manufacturers. 73 Fed. Reg. 64,946-64,947 (October 31, 2008).
EPA. 2008d. EPA Engineering Issue: Indoor Air Vapor Intrusion Mitigation Approaches. www.epa.gov/nrmrl/pubs/600r08115/600r08115.html.
EPA. 2008e. Preliminary Summary of Financial Accounting Standards for Environmental Liabilities: Intangible Assets and Climate Change Risk. DRAFT Report April 28, 2008. http://www.epa.gov/osem/financial/e-disclosure.pdf.
EPA. 2009a. Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonate (PFOS). Provisional Health Advisory information. http://www.epa.gov/waterscience/criteria/drinking/pha-PFOA_PFOS.pdf.
EPA. 2009b. Contaminant Candidate List. Federal Register 74(194):51850.
EPA. 2009c. EPA, CERCLA, Brownfields, and Lender Liability Fact Sheet. http://www.epa.gov/brownfields/aai/lenders_factsheet.pdf.
EPA. 2009d. Memorandum from James E. Woolford, Director, Office of Superfund Remediation and Technology Innovation and John E. Reeder, Director a Federal Facilities Restoration and Reuse Office to Superfund National Policy Managers, Regions 1-10. Subject: Summary of Key Existing EPA CERCLA Policies for Groundwater Restoration at 3 (June 26, 2009). http://www.epa.gov/superfund/health/conmedia/gwdocs/pdfs/9283_1-33.pdf, citing the 1990 NCP.
EPA. 2010a. Superfund Remedy Report, Thirteenth Edition, at 10 Figure 6. EPA-542-R-10-004. Washington, DC: EPA OSWER.
EPA. 2010b. Institutional Controls: A Guide to Planning, Implementing, Maintaining, and Enforcing Institutional Controls at Contaminated Sites. Washington, DC: EPA OSWER. http://www.epa.gov/superfund/policy/ic/pdfs/PIME-IC-Guidance-Interim.pdf.
EPA. 2010c. Fourth Five-Year Review Report for Cannons Engineering Bridgewater Superfund Site Town of Bridgewater Plymouth County, Massachusetts, September 2010. http://www.epa.gov/region1/superfund/sites/cannon/470176.pdf.
EPA. 2010d. February 2010 Update: Arsenic Trioxide Superfund Site, Richland and Sargent Counties, North Dakota. http://www.epa.gov/region8/superfund/nd/arsenic/AT_5YrUpdate2010.pdf.
EPA. 2010e. A New Approach to Protecting Drinking Water and Public Health. EPA 815F10001. Washington, DC: EPA Office of Water. http://water.epa.gov/lawsregs/rulesregs/sdwa/dwstrategy/upload/Drinking_Water_Strategyfs.pdf.
EPA. 2011a. Integrated Cleanup Initiative at 18. http://www.epa.gov/oswer/docs/ici_implementation_plan.pdf.
EPA. 2011b. Recommended Evaluation of Institutional Controls: Supplement to the Comprehensive Five Year Review Guidance. Washington, DC: EPA OSWER. www.epa.gov/superfund/cleanup/postconstruction/641333.pdf.
EPA. 2011c. Regulatory Determination on Perchlorate. February 2, 2011. https://www.federalregister.gov/articles/2011/02/11/2011-2603/drinking-water-regulatory-determination-on-perchlorate2011j.
EPA. 2011d. Nanotechnology: Applications for Environmental Remediation CLU-IN Technology Focus Area Fact Sheet. Washington, DC: EPA OSWER.
EPA. 2011e. Selected Sites Using or Testing Nanoparticles for Remediation. www.clu-in.org/download/remed/nano-site-list.pdf.
EPA. 2011f. Proposed Significant New Use Rules on Certain Chemical Substances. 76 Federal Register 81,447. http://www.gpo.gov/fdsys/pkg/FR-2011-12-28/pdf/2011-33261.pdf.
EPA. 2011g. EPA Needs to Manage Nanomaterial Risks More Effectively. Report No. 12-P-0162 December 29, 2011. Washington, DC: EPA Office of the Inspector General.
EPA. 2011h. Integrated Risk Information System (IRIS), Trichloroethlyene (September 28, 2011). http://www.epa.gov/IRIS/subst/0199.htm, which is based on EPA’s Toxicological Review of Trichloroethylene and related Excel spreadsheet.
EPA. 2011i. Potential Addition of Vapor Intrusion Component to the Hazard Ranking System. Federal Register 76(20):5370–5374.
EPA. 2011j. Basic Questions and Answers for the Drinking Water Strategy Contaminant Groups Effort. http://water.epa.gov/lawsregs/rulesregs/sdwa/dwstrategy/upload/FactSheet_DrinkingWaterStrategy_VOCs.pdf.
EPA. 2011k. EPA Handbook on the Benefits, Costs and Impacts of Land Cleanup and Reuse. http://yosemite.epa.gov/ee/epa/eerm.nsf/vwAN/EE-0569-02.pdf/$file/EE-0569-02.pdf.
EPA. 2011L. Revitalizing Contaminated Sites: Addressing Liability Concerns (2011 Edition). http://www.epa.gov/compliance/resources/publications/cleanup/brownfields/handbook/index.html.
EPA. 2012a. Regional Screening Level (RSL) Summary Table April 2012. http://www.epa.gov/reg3hwmd/risk/human/rb-concentration_table/Generic_Tables/pdf/master_sl_table_run_MAY2012.pdf.
EPA. 2012b. EPA’s Vapor Intrusion Database: Evaluation and Characterization of Attenuation Factors for Chlorinated Volatile Organic Compounds and Residential Buildings. EPA 530-R-10-002. Washington, DC: Office of Solid Waste and Emergency Response.
EPA. 2012c. Superfund Vapor Intrusion FAQs. http://www.epa.gov/superfund/sites/npl/Vapor_Intrusion_FAQs_Feb2012.pdf.
EPA Region 9. 2010. Formation of a Community Informational Group (CIG) for the Motorola 52nd St. Superfund Site. May 2010 Important Announcements Fact Sheet.
Federal Judicial Center and NRC. 2011. Third Edition of the Reference Manual on Scientific Evidence. Washington, DC: The National Academies Press.
Folkes, D., W. Wertz, J. Kurtz, and T. Kuehster. 2009. Observed spatial and temporal distributions of CVOCs at Colorado and New York vapor intrusion sites. Ground Water Monitoring & Remediation 29(1):70-80.
Fujii, S., C. Polprasert, S. Tanaka, N. P. H. Lien, and Y. Qiu. 2007. New POPs in the water environment: Distribution, bioaccumulation and treatment of perfluorinated compounds—a review paper. Journal of Water Supply Research and Technology-Aqua 56(5):313-326.
GAO (Government Accountability Office). 2009. Superfund Litigation Has Decreased and EPA Needs Better Information on Site Cleanup and Cost Issues to Estimate Future Program Funding Requirements, Table 11. GAO-09-656. Washington, DC: GAO. http://www.gao.gov/assets/300/292299.pdf.
GeoSyntec Consultants. 2002. In situ bioremediation of perchlorate impacted groundwater. Final report for the Strategic Environmental Research and Development Program. CU-1164-FR-01.
Greskowiak, J., H. Prommer, G. Massmann, and G. Nuetzmann. 2006. Modeling seasonal redox dynamics and the corresponding fate of the pharmaceutical residue phenazone during artificial recharge of groundwater. Environmental Science & Technology 40(21):6615-6621.
Gui, L., R. W. Gillham, and M. S. Odziemkowski. 2000. Reduction of N-nitrosodimethylamine with granular iron and nickel-enhanced iron. 1. Pathways and kinetics. Environmental Science & Technology 34:3489-3494.
Gupta, S., G. Van Houtven, and M. Cropper. 1996. Paying for performance: An economic analysis of EPA’s cleanup decisions at superfund sites. Rand Journal of Economics 27:563-582.
Hamilton, J. T., and W. K. Viscusi. 1999. How Costly is “Clean”? An analysis of the benefits and costs of superfund site remediations. Journal of Policy Analysis and Management 18(1):2-27.
Henderson, A. D., and A. H. Demond. 2007. Long-term performance of zero-valent iron permeable reactive barriers: A critical review. Environmental Engineering Science 24(4):401-423.
Huang, H., and G. A. Sorial. 2007. Perchlorate remediation in aquatic systems by zero valent iron. Environmental Engineering Science 24(7):917-926.
Husain, S. 2008. Literature overview: Emerging organic contaminants in water and their remediation. Remediation 18:91-105.
Hutton, D., and A. Edwards. 2011. Recent developments in Natural Resource Damages claims. Trends 42(6) July/August 2011. http://www.winston.com/siteFiles/Publications/HuttonEdwardsarticleTrends42_6July_August2011.pdf.
ICMA. 2006. International City/County Management Association ICMA Program Excellence Award Nomination, GILBERT-MOSLEY Setting a Precedent in Environmental Cleanup. http://icma.org/Documents/Document/Document/5796.
Israel, B. D. 2009. State-by-State Guide to NRD Programs in All 50 States. http://www.mass.gov/Eoeea/docs/eea/nrd/nrd_state_by_state.pdf.
ITRC (Interstate Technology & Regulatory Council). 2005a. Permeable Reactive Barriers: Lessons Learned/New Directions. PRB-4. Washington, DC: ITRC Permeable Reactive Barriers Team.
ITRC. 2005b. Perchlorate: Overview of Issues, Status, and Remedial Options. http://www.itrcweb.org/Documents/PERC-1.pdf.
ITRC. 2007. Vapor Intrusion Pathway: A Practical Guideline. Washington, DC: ITRC Vapor Intrusion Team.
ITRC. 2010. A Decision Framework for Applying Monitored Natural Attenuation Processes to Metals and Radionuclides in Groundwater. APMR-1. Washington, DC: ITRC Attenuation Processes for Metals and Radionuclides Team.
ITRC. 2011. Permeable Reactive Barrier: Technology Update. PRB-5. Washington, DC: ITRC PRB: Technology Update Team.
Johnson, P. C., H. Luo, C. Holton, P. Dahlen, and Y. Guo. 2012. Vapor Intrusion above a Dilute CHC Plume: Lessons-Learned from Two Years of Monitoring. EPA-AEHS Workshop, Recent Advances to VI Application & Implementation 20 March 2012, San Diego. https://iavi.rti.org/WorkshopsAndConferences.cfm.
Johnson, R. L., R. B. Thoms, R. O’Brien Johnson, and T. Krug. 2008. Field evidence for flow reduction through a zero-valent iron permeable reactive barrier. Ground Water Monitoring & Remediation 28(3):47-55.
Kerger, B. D., W. J. Butler, D. J. Paustenbach, J. Zhang, and S. Li. 2009. Cancer mortality in Chinese populations surrounding an alloy plant with chromium smelting operations. Journal of Toxicology and Environmental Health A 72(5):329-344.
Khandelwal, A., A. J. Rabideau, and P. Shen. 1998. Analysis of diffusion and sorption of volatile organic contaminants in soil/bentonite barrier materials. Environmental Science & Technology 32(9):1333-1339.
Kiel, K. A. 1995. Measuring the impact of the discovery and cleaning of identified hazardous waste sites on house values. Land Economics 71(4):428-435.
Kohn T., K. J. T. Livi, A. L. Roberts, and P. J. Vikesland. 2005. Longevity of granular iron in groundwater treatment processes: Corrosion product development. Environmental Science & Technology 39(8):2867-2879.
Kreuzinger, N., M. Clara, B. Strenn, and B. Vogel. 2004. Investigation on the behavior of selected pharmaceuticals in the groundwater after infiltration of treated wastewater. Water Science and Technology 50(2):221-228.
Krol, M. M., and R. K. Rowe. 2004. Diffusion of TCE through soil-bentonite slurry walls. Soil and Sediment Contamination 13:81-101.
LaRoss, D. 2012. EPA Requires Cr6 Monitoring in New List of Drinking Water Contaminants. Inside EPA, May 1, 2012.
Lau, C., K. Anitole, C. Hodes, D. Lai, A. Pfahles-Hutchens, and J. Seed. 2007. Perfluoroalkyl acids: A review of monitoring and toxicological findings. Toxicological Sciences 99(2):366-394.
Lee, A. 2012. EPA’s Comments on January 3, 2012 revision to Air Monitoring and Soil Sampling Plan for Google-Fairchild Plumbing Excavation Activities, 313 and 323 Fairchild Drive, Mountain View, CA; Middlefield-Ellis-Whisman (MEW) Superfund Study Area.
MAOEEA (Massachusetts Office of Energy and Environmental Affairs). 2012. Massachusetts NRD Case Settlements. http://www.mass.gov/eea/land-use-habitats/antural-resource-damages/mass-nrd-cases/nrd-settlements.html [accessed December 17, 2011].
Massapequa Water District. 2011. Massapequa Water District Case in Opposition to NYSDEC Navy ROD OU-2. http://www.massapequawater.com.
McBean, E. A. 2008. Siloxanes in biogas from landfills and wastewater digesters. Canadian Journal of Civil Engineering 35(4):431-436.
McCluskey, J. J., and G. C. Rausser. 2003. Stigmatized asset value: Is it temporary or long-term? Review of Economics and Statistics 85(2):276-285.
Messer, K. D., W. D. Schulze, K. F. Hackett, T. A. Cameron, and G. H. McClelland. 2006. Can stigma explain large property value losses? The psychology and economics of superfund. Environmental and Resource Economics 33:299-324.
Mohr, T. 2010. Environmental Investigation and Remediation: 1,4-Dioxane and Other Solvent Stabilizers. London: CRC Press.
Morgan, J. W. 2011. Update on Hinkley Cancer Incidence. California Cancer Registry. The Criterion 4(1). http://www.ccrcal.org/DSQC_Pubs/Criterion/2011_03_Criterion.htm.
Mott, H. V., and W. J. Weber. 1991. Factors influencing organic contaminant diffusivities in soil-bentonite cutoff barriers. Environmental Science & Technology 25(10):1708–1715.
Murnyak, G., J. Vandenberg, P. J. Yaroschak, L. Williams, K. Prabhakaran, and J. Hinz. 2011. Emerging contaminants: Presentations at the 2009 Toxicology and Risk Assessment Conference. Toxicology and Applied Pharmacology 254(2):167-169.
Nakada, N., K. Kiri, H. Shinohara, A. Harada, K. Kuroda, S. Takizawa, and H. Takada. 2008. Evaluation of pharmaceuticals and personal care products as water-soluble molecular markers of sewage. Environmental Science & Technology 42(17):6347-6353.
NBER (National Bureau of Economic Research). 2010. U.S. Business Cycle Expansions and Contractions. http://www.nber.org/cycles.html, accessed April 22, 2010.
Niagara Gazette. 1979. Glynn, Don, Banks halt FHA, VA mortgage loans. Niagara Gazette (11/16/1979).
NJAG. 2011. Natural Resource Damage (NRD) Lawsuits. The State of New Jersey Department of Law & Public Safety, Office of the Attorney General. http://www.nj.gov/oag/newsreleases07/NRD-lawsuits-07/ [accessed November 8, 2011].
NJ DEP (New Jersey Department of Environmental Protection). 2005. Vapor Intrusion Guidance. Pp. 135.
NJ DEP. 2007. State Files Lawsuits Seeking Compensation for Damages Polluters Caused to Natural Resources. State of New Jersey Department of Environmental Protection. http://www.state.nj.us/dep/newsrel/2007/07_0037.htm [accessed November 8, 2011].
NJ DEP. 2008. State Secures $4 Million in Natural-Resource Damage Settlements, Preserves more than 700 Acres. State of New Jersey Department of Environmental Protection. http://www.state.nj.us/dep/newsrel/2008/08_0038.htm [accessed November 8, 2011].
Northgate Environmental Management Inc. 2008. Fairchild Buildings Slurry Wall System Efficiency Study Report, Middlefield-Ellis-Whisman Study Area Mountain View, California, Oakland CA.
NRC (National Research Council). 1994. Alternatives for Ground Water Cleanup at 245. Washington, DC: National Academy Press.
NRC. 1997. Innovations in Soil and Ground Water Cleanup. Washington, DC: National Academy Press.
NRC. 2000. Natural Attenuation for Groundwater Remediation. Washington, DC: The National Academy Press.
NRC. 2001. Classifying Drinking Water Contaminants for Regulatory Consideration. Washington, DC: National Academy Press.
NRC. 2007. Assessment of the Performance of Engineered Waste Containment Barriers. Washington, DC: The National Academies Press.
NRC. 2008. Prospects for Managed Underground Storage of Recoverable Water. Washington, DC: The National Academies Press.
NTP (National Toxicology Program). 2008. Technical Report on the Toxicology and Carcinogenesis Studies of Sodium Dichromate Dihydrate (CAS No. 7789-12-0) in F344/N Rats and B6C3F1 Mice (Drinking Water Studies). NIH Publication 08-5887; NTP TR-546. http://ntp.niehs.nih.gov/files/546_WEB_FINAL.pdf.
NYS DEC. 2005. List of Inactive Hazardous Waste Sites with Pre-2003 Remedial Decisions Where Disposal of Chlorinated Hydrocarbons Occurred. Albany, NY: New York State Department of Environmental Conservation. http://www.dec.ny.gov/regulations/51715.html and http://www.dec.ny.gov/environmentdec/52789.html.
Obare, S. O., and G. J. Meyer. 2004. Nanostructured materials for environmental remediation of organic contaminants in water. Journal of Environmental Science and Health, Part A: Toxic/Hazardous Substances and Environmental Engineering 39(10):2549-2582.
Obmascik, M. 2002. Home Deadly Home: Toxins in Air. Denver Post, January 6, 2002.
OCC (Office of the Comptroller of the Currency, Treasury), Board of Governors of the Federal Reserve System, Federal Deposit Insurance Corporation, U.S. Securities and Exchange Commission, Federal Housing Finance Agency, and Department of Housing and Urban Development. 2011. Proposed Credit Risk Retention Rule. http://www.sec.gov/rules/proposed/2011/34-64148.pdf.
Odziemkowski, M. S., L. Gui, and R. H. Gillham. 2000. Reduction of N-nitrosodimethylamine with granular iron and nickel-enhanced iron. 2. Mechanistic Studies. Environmental Science & Technology 34:3495-3500.
Olson, D. A., and R. L. Corsi. 2002. Fate and transport of contaminants in indoor air. Soil and Sediment Contamination 11(4):583-601.
Phillips, D. H., T. V. Nooten, M. I. Russell, K. Dickson, S. Plant, J. M. E. Ahad, T. Newton, T. Elliot, and R. M. Kalin. 2010. Ten year performance evaluation of a field-scale zero-valent iron permeable reactive barrier installed to remediate trichloroethene contaminated groundwater. Environmental Science & Technology 44(10):3861-3869.
Pistocchi, A., and R. Loos. 2009. A map of European emissions and concentrations of PFOS and PFOA. Environmental Science & Technology 43(24):9237-9244.
Probst, K. 2006. Critical Issues Facing the Superfund Program at 9. Washington, DC: Resources for the Future. http://www.rff.org/RFF/Documents/RFF-CTst_06-Probst.pdf.
Rabiet, M., A. Togola, F. Brissaud, J. L. Seidel, H. Budzinski, and F. Elbaz-Poulichet. 2006. Consequences of treated water recycling as regards pharmaceuticals and drugs in surface and ground waters of a medium-sized Mediterranean catchment. Environmental Science & Technology 40(17):5282-5288.
Rak, A., and C. M. Vogel. 2009. Increasing Regulation of Perfluorinated Compounds and the Potential Impacts at Air Force Installation. Poster Presentation. AFCEE EMDQ. http://www.noblis.org/MissionAreas/ees/Documents/AFCEE%20EMDQ%20Poster_2009_?FINAL.pdf.
Rayne, S., and K. Forest. 2009. Perfluoroalkyl sulfonic and carboxylic acids: A critical review of physicochemical properties, levels and patterns in waters and wastewaters, and treatment methods. Journal of Environmental Science and Health Part A: Toxic/Hazardous Substances & Environmental Engineering 44(12):1145-1199.
Rhomberg, L. R., L. A. Bailey, and J. E. Goodman. 2010. Hypothesis-based weight of evidence: A tool for evaluating and communicating uncertainties and inconsistencies in the large body of evidence in proposing a carcinogenic mode of action—Naphthalene as an example. Critical Reviews in Toxicology 40:671-696.
Richardson, S. D., and T. A. Ternes. 2005. Water analysis: Emerging contaminants and current issues. Analytical Chemistry 77(12):3807-3838.
Robertson, W. D., C. J. Ptacek, and S. J. Brown. 2007. Geochemical and hydrogeological impacts of a wood particle barrier treating nitrate and perchlorate in ground water. Ground Water Monitoring & Remediation 27(2):85-95.
Rogers, C. 2007. Taking the Next Step in Environmental Due Diligence at 3, American Bar Association, Section of Environment, Energy, and Resources, 36th Conference on Environmental Law, Keystone, CO. www.advancedenvironmentaldimensions.com/documents/ED3.pdf.
Sale, T., and C. Newell. 2010. Guide for Selecting Remedies for Subsurface Releases of Chlorinated Solvents. Draft report, ESTCP Project ER-0530.
Sass, B., A. Gavaskar, W. Yoon, C. Reeter, and E. Drescher. 2002. Geochemical Factors Affecting Performance and Longevity of Permeable Reactive Barriers. Proceedings, Third International Conference on Remediation of Chlorinated and Recalcitrant Compounds, Monterey, California. Columbus, Ohio: Battelle Press.
SEC (Securities and Exchange Commission). 1982. Regulation S-K, 17 C.F.R. § 229.103 (Item 103) and SEC Regulation S-B, Item 103 and Item 103, Instructions 1-4, 17 C.F.R. § 228.103. 47 Fed. Reg. 11401, Mar. 16, 1982.
SEC. 2003. Summary by the Division of Corporation Finance of Significant Issues Addressed in the Review of the Periodic Reports of the Fortune 500 Companies: Monitor of the Fortune 500 by the Division of Corporation Finance. http://www.sec.gov/divisions/corpfin/fortune500rep.htm.
SEC. 2011. Staff Accounting Bulletin 114. Release No. SAB 114 at 133-134. http://www.sec.gov/interps/account/sab114.pdf.
Shrout, J. D., A. G. Williams, M. M. Scherer, and G. F. Parkin. 2005. Inhibition of bacterial perchlorate reduction by zero-valent iron. Biodegradation 16(1):23-32.
Shrout, J. D., G. C. Struckhoff, G. F. Parkin, and J. L. Schnoor. 2006. Stimulation and molecular characterization of bacterial perchlorate degradation by plant-produced electron donors. Environmental Science & Technology 40(1):310-317.
Simcik, M. F. 2005. Global transport and fate of perfluorochemicals. Journal of Environmental Monitoring 7(8):759-763.
Singh, A., E. Neuhauser, and A. J. Rabideau. 2011. Dealing with censored vapor concentration data, Case study of former Manufactured Gas Plant sites. 27th Annual Conference on Soil, Sediments, Water, and Energy, Amherst, MA, October 17-21.
Snyder, S. A., J. Leising, P. Westerhoff, Y. Yoon, H. Mash, and B. Vanderford. 2004. Biological and physical attenuation of endocrine disruptors and pharmaceuticals: implications for water reuse. Ground Water Monitoring & Remediation 24(2):108-118.
Soliman, M. A., J. A. Pedersen, H. Park, A. Castaneda-Jimenez, M. K. Stenstrom, and I. H. Suffet. 2007. Human pharmaceuticals, antioxidants, and plasticizers in wastewater treatment plant and water reclamation plant effluents. Water Environment Research 79(2):156-167.
Son, A., J. Lee, P. C. Chiu, B. J. Kim, and D. K. Cha. 2006. Microbial reduction of perchlorate with zero-valent iron. Water Research 40(10):2027-2032.
Spina, P. 2008. State Environmental Easement Statutes as ARARs: A New Option for Enforcing Institutional Controls at Superfund Sites. Columbia Journal of Environmental Law 33:122-129. www.columbiaenvironmentallaw.org/assets/pdfs/33.1/Spina.33.1.pdf.
Standley, L. J., R. A. Rudel, C. H. Swartz, K. R. Attfield, J. Christian, M. Erickson, and J. G. Brody. 2008. Wastewater-contaminated groundwater as a source of endogenous hormones and pharmaceuticals to surface water ecosystems. Environmental Toxicology and Chemistry 27(12):2457-2468.
Steffan, R. J. 2007. Biodegradation of 1,4-Dioxane. Final report for the Strategic Environmental Research and Development Program. ER-1422.
Suh, J. H., and M. Mohseni. 2004. A study on the relationship between biodegradability enhancement and oxidation of 1,4-dioxane using ozone and hydrogen peroxide. Water Research 38(10):2596-2604.
Szecsody, J. E., J. P. McKinley, F. H. Crocker, A. T. Breshears, B. J. Devary, H. L. Fredrickson, and K. T. Thompson. 2009. Abiotic and Biotic Mechanisms Controlling In Situ Remediation of NDMA, Final Report. SERDP Project ER-1421. SERDP ER-1421. Richland, WA: Pacific Northwest National Laboratory.
Tanner, R. 2009. Sunburst Sch. Dist. No. 2 V. Texaco, Inc.: Rethinking Restoration Damages as a Remedy for Tortious Injury to Land and Property. http://www.montanalawreview.com/sitebuildercontent/sitebuilderfiles/publication-ready_casenote.pdf.
Technical Review Committee. 1988. Love Canal Emergency Declaration Area Habitability Study, Final Report, Volume I: Introduction and Decision-Making Documentation at 1-2. http://nepis.epa.gov/EPA/html/DLwait.htm?url=/Exe/ZyNET.exe/9100HT40.PDF?ZyActionP=PDF&Client=EPA&Index=1986 Thru 1990& File=D%3A%5CZYFILES%5CINDEX%20DATA%5C86THRU90%5CTXT%5C00000021%5C9100HT40.txt&Query=902488002A%20or%20Love%20or%20Canal%20or%20Emergency%20or%20Declaration%20or%20Area%20or%20%E2%80%93Remediation%20or%20EDA%20and%20%E2%80%93%20or%20Final%20or%20Study%20or%20Report&SearchMethod=1&FuzzyDegree=0&User=?ANONYMOUS&Password=anonymous&QField=pubnumber%5E%22902488002A%22&UseQField=pubnumber&IntQFieldOp=1&ExtQFieldOp=1&Docs=.
Thompson, C. M., D. M. Proctor, M. Suh, L. C. Haws, C. D. Hébert, J. F. Mann, H. G. Shertzer, J. G. Hixon, and M. A. Harris. 2012. Comparison of the effects of hexavalent chromium in the alimentary canal of F344 rats and B6C3F1 mice following exposure in drinking water: Implications for carcinogenic modes of action. Toxicological Sciences 125(1):79–90.
University of Buffalo. 2011. Love Canal Collection. http://library.buffalo.edu/special?collections/lovecanal/about/chronology.php.
USGS. 2003. Ground-Water Depletion Across the Nation. U.S. Geological Survey Fact Sheet 103-03.
Vainberg, S., K. McClay, H. Masuda, D. Root, C. Condee, G. Zylstra, and R. J. Steffan. 2006. Biodegradation of ether pollutants by Pseudonocardia sp. strain ENV478. Applied and Environmental Microbiology 72(8):5218-5224.
Vaseashta, A., M. Vaclavikova, S. Vaseasht, G. Gallios, P. Roy, and O. Pummakarnchana. 2007. Nanostructures in environmental pollution detection, monitoring, and remediation. Science and Technology of Advanced Materials 8:47-59.
Vincent, G. K., and V. Velkoff. 2010. The Next Four Decades: The Older Population in the United States: 2010 to 2050. Washington, DC: U.S. Census Bureau.
Waldemer, R. H., and P. G. Tratnyek. 2006. Kinetics of contaminant degradation by permanganate. Environmental Science & Technology 40(3):1055-1061.
Weinfield, N. H. 2007. A Brownfield model collapses under the weight of litigation: City of Wichita v. Trustees of the Apco Oil Corporation Liquidating Trust. Proceedings of the Annual International Conference on Soils, Sediments, Water and Energy: Vol. 12, Article 37. http://scholarworks.umass.edu/soilsproceedings/vol12/iss1/37.
Wilkin, R., and R. Puls. 2003. Capstone Report on the Application, Monitoring, and Performance of Permeable Reactive Barriers for Groundwater Remediation: Volume 1—Performance Evaluation at Two Sites. EPA/600/R-03/045a. Washington, DC: EPA.
Woodhouse, K. S. 2009. Comments on July 2009 Proposed Plan for the VI Pathway. http://www.cpeo.org/pubs/MV-VIPolicy.pdf.
Yabusaki, S., K. Cantrell, B. Sass, and C. Steefel. 2001. Multicomponent reactive transport in an in situ zero-valent iron cell. Environmental Science & Technology 35(7):1493–1503.
Yonkers, T. 2012. Army Urges Hill To Block EPA Policy Changes Midway Through Cleanups. Inside EPA Daily News. March 12, 2012. Yu, X., C. Amrhein, M. A. Deshusses, and M. R. Matsumoto. 2006. Perchlorate Reduction by Autotrophic Bacteria in the Presence of Zero-Valent Iron. Environmental Science & Technology 40(4):1328-1334.
Zhou, Q., S. McCraven, J. Garcia, M. Gasca, T. A. Johnson, and W. E. Motzer. 2009. Field evidence of biodegradation of N-nitrosodimethylamine (NDMA) in groundwater with incidental and active recycled water recharge. Water Research 43(3):793-805.


























































