7
Preventing Disability Related to Sensory Loss in the Older Adult
Senses link individuals to their environment. Sight and hearing allow for communication; taste, olfaction, proprioception, and temperature perception provide the information necessary not only to traverse safely but to enjoy the world. Diminution, to some degree, of any or all of these senses is a recognized consequence of aging in many people.2,42 Although the loss of any of the senses is potentially disabling, vision and hearing will be the focus of this paper—for several reasons. Losses in these two areas are among the most prevalent conditions among elderly Americans.35,38 In addition, decreased vision and hearing have been linked to limitations in physical, emotional, and social functioning,8,12,42,49,53 which therefore makes the spectrum of potential disability and handicap very broad. From a practical point of view, impairments in these two areas are potentially preventable as are the associated disabilities and handicaps.6,33 Limiting this chapter to sight and hearing also allows for development of an agenda for preventing sensory impairment and associated disability or handicap that reflects current knowledge. It is not intended to suggest that other sensory losses do not deserve careful attention.
DEFINITION AND PREVALENCE
Both visual and hearing impairments rank among the 15 most prevalent chronic conditions in Americans aged 65 and older.37 Yet
varying definitions of visual impairment affect the magnitude of its incidence and prevalence. The National Health Interview Survey defines visual impairment as the ability to read newsprint. According to this measure, preliminary data indicate that approximately 13 percent of noninstitutionalized Americans aged 65 and older have some form of visual impairment.23 Of this group, 8 percent suffer with a severe impairment, which is defined as the inability to read newsprint even with glasses.39 The severely impaired therefore also include those who are blind in both eyes.39 The survey reports that 3.2 percent of respondents reported blindness in one eye, and 1 percent reported blindness in both eyes.23 Legal blindness, on the other hand, is defined as 20/200 vision or worse. (An eye with 20/200 vision sees at 20 feet that which an eye with 20/20 vision can see at 200 feet.) Confident estimates of the prevalence of legal blindness, however, are not available.
If one defines poor visual acuity as anything equal to or worse than 20/50 vision, the incidence of visual loss increases by 13 percent between ages 60 and 69 and 32 percent among those 70 to 80 years old.42 Using the same definition (20/50 or worse), 11 percent of persons aged 65 to 73 who wear glasses are impaired; 26 percent of this age group who do not wear glasses are also impaired.38 By 80 years of age, only 10 to 20 percent of this group will have a visual acuity of 20/20.42 Performance on tests of stereoscopic acuity decrease from nearly 100 percent at age 30 to approximately 60 percent at age 80.42 Performance on red-green color match tests decrease from 60 percent at age 30 to less than 10 percent at age 70.42
Hearing loss is also measured using different definitions and therefore yields varying prevalence estimates as well. Using a threshold of 40 decibels (dB) (speech is generally at the 50-dB level—see Figure 7-1) for any two measured frequencies, the prevalence of hearing impairment is 45 per 100,000 for those 17 to 44 years of age, 119 per 100,000 for those 45 to 64 years of age, and 282 per 1,000 for those over 64 years of age.6 Preliminary data from the National Health Interview Survey show a similar distribution in the population aged 64 and older; they also indicate that hearing impairment (defined as ''the reported presence of deafness in one or both ears or any other trouble hearing"23) varies with age and sex. Twenty-three percent of individuals aged 65 to 74, 32.7 percent of those 75 to 84, and 48.4 percent of those 85 and older show hearing impairment. Men had a greater percentage of hearing impairments in each age range. For example, among those 65 to 74 years old, 30 percent of the men were impaired as compared with 17.5 percent of the women.23 Other studies that focus on sensory loss among specific populations (e.g., nursing home residents) have found even higher prevalence rates.3,19,47 For example,
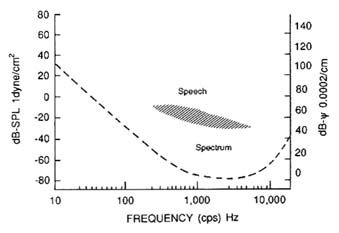
FIGURE 7-1 Human audiogram with absolute threshold of hearing expressed in reference to dB-SPL and the psychophysical thresholds dB-ψ, at various frequencies, with the minimum audible pressure (MAP) at the eardrum. Shaded area represents audible frequency spectrum for human speech.
Source: J. M. Ordy and K. R. Brizee, Sensory Systems and Communication in the Elderly, Raven Press, New York, 1979.
the Framingham Heart Study Cohort reported a prevalence of hearing impairments of 83 percent among men and women aged 57 to 89 years of age.35 It should be noted, however, that this study used a threshold of 20 dB, which is considerably lower than the 40 dB used in many other studies.6,35 When a higher threshold (40 dB) was used, the prevalence of hearing impairments was 48 percent. Two studies reporting hearing impairment prevalences among nursing home residents found rates of 94 percent and 82 percent.3,47
In some cases these prevalence data are drawn from population-based surveys, but the data are limited in ways that affect the ability to estimate the true burden of disease in all other adult populations. For example, the lack of detailed racial and ethnic identifiers limits an understanding of prevalence among Hispanic and other minority populations. Existing national survey data comparing blacks and whites suggest that there may be differences in hearing loss between these groups, particularly for the higher frequencies.
ETIOLOGIC BREAKDOWN
Visual Impairments
The leading causes of visual impairment among the elderly are cataracts, glaucoma, macular degeneration, and diabetic retinopathy.6,16,42,50,53
The incidence of cataracts increases steadily with age and is responsible for the largest number of new cases of blindness.50 Between the ages of 50 and 80 there is a ninefold increase from 4 to 46 percent in the percentage of the population with visual acuity of less than 20/30 as a result of cataracts.50 Although it is beyond the scope of this chapter to discuss the details of cataract pathology and pathophysiology, the type and location of the cataract have implications for the type of visual impairment and therefore the type of disability and handicap experienced. For example, nuclear cataracts result in poor distance acuity, whereas posterior cataracts result in poor near vision. In advanced stages, however, the visual impairment resulting from cataracts is usually global. Visual impairment from cataracts is frequently progressive, but the rate of visual impairment can vary considerably (occurring over a space of months to one of years).
Glaucoma is associated with progressive visual field defects that can eventually become extensive enough to cause serious visual impairment, even blindness.22 Open-angle glaucoma is the second most common cause of blindness in the elderly. The congressional Office of Technology Assessment has estimated that approximately 4,600 elderly people go blind from glaucoma each year and that 0.5 percent of 65-year-olds and 1.1 percent of 75-year-olds will develop glaucoma within five years.43 Blacks, diabetics, and people with a family history of glaucoma seem to be at increased risk.31 Similarly, blindness is more likely to occur in blacks with glaucoma and to occur at an early age.26 The most significant predictor of the development of glaucoma is elevated intraocular pressure, but it is by no means a perfect predictor, which complicates screening for glaucoma in the elderly. Thirty percent of those with very high intraocular pressures (>30 mmHg) will develop glaucoma over a 17- to 20-year period. Overall, people with significantly increased ocular pressure are seven times more likely to develop glaucoma than those with normal intraocular pressure.43
Accurate figures for the prevalence of macular degeneration (loss of central vision in both eyes, frequently characterized by spots of pigmentation and other abnormalities) as a cause of blindness are not available; however, data pooled from the National Health and Nutrition Examination Survey and the Framingham Eye Study indicate that it is a leading cause of blindness among older adults. The incidence of macular degeneration increases with age especially after the age of 50.16 Prevalence estimates in the 65- to 75-year-old age group are 6.9 percent in white females, 11.4 percent in black females, 9.6 percent in white males, and 9.3 percent in black males. Data on other racial and ethnic groups are not readily available. Exudative matter (from
blood vessels, often including serum, fibrin, and white blood cells) is the more common cause of blindness. Hyperopia (a condition in which images come into focus behind the retina) is the most frequently reported risk factor for macular degeneration; other factors including a family history of hyperopia, decreased handgrip, and systemic high blood pressure have been reported in case-control studies.15 Generally, visual loss is caused by an ingrowth of choroidal neovascularization and the formation of round or oval macular scars. Unlike visual impairment that results from cataracts and glaucoma, visual impairment that results from macular degeneration usually occurs suddenly.
Diabetic retinopathy is the next most common cause of blindness among older adults. One-third of all diabetic patients have some retinopathy, which carries a high risk of sudden visual loss as a result of a vitreous hemorrhage or retinal detachment, or both.
There are two reasons why it is important to keep in mind the specific etiologies of visual loss. First, the natural history of each of these causes of visual impairment is very different; therefore, the extent and timing of the associated disabilities are likely to be very different in both individuals and populations.22,43,50 Second, the availability and effectiveness of treatment for each of these conditions vary. Consequently, the ability to prevent not only the associated disability but the visual impairment itself will vary across conditions. Some of the committee's recommendations related to visual impairment will necessarily be linked to specific etiologies of visual impairment.
Hearing Impairments
The most common cause of bilateral hearing impairment among the elderly, presbycusis, is characterized by increased thresholds for high-frequency tones, recruitment, and difficulty with speech comprehension (see Figure 7-2).17 There is a 25 percent reduction in speech comprehension that occurs between the ages of 50 and 80; the reduction is particularly noticeable with complex, rapid patterns of speech 6,17
Pathologically, there are four types of presbycusis: sensory, mechanical, strial, and neuronal.17 The exact nature of the hearing impairment as recorded on an audiogram differs with each of these. The etiology of presbycusis is poorly understood, but the pattern of hearing loss is very similar to that associated with noise exposure.17 Indeed, age-related hearing impairment is much less common among populations living in relatively noise-free environments.27,45
Although presbycusis is the most common cause of hearing
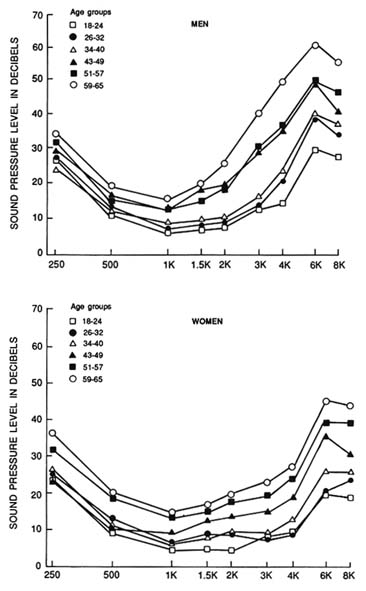
FIGURE 7-2 Mean threshold values for hearing pure tones as a function of frequency for men and women in six different age groups. The subjects in this study were carefully screened for excessive noise exposure and any history of diseases known to affect hearing. Age groups in years 18–24 ( - ); 26–32 ( - ); 34–40 ( - ); 43–49 ( - ); 51–57 ( - ); and 59–65( - ). Source: J. M. Ordy and K. R. Brizee, Sensory Systems and Communication in the Elderly, Raven Press, New York, 1979.
impairment among the elderly, there are other causes of hearing loss in the older population as well. Today's cohort of older adults whose childhoods precede the antibiotic era may suffer the sequelae of untreated otitis media (an infection causing inflammation of the middle ear) to a greater extent than will future post-antibiotic era cohorts. Vascular disease, thyroid disease, and arthritis are other common causes of hearing impairment because of the relatively high prevalence of these conditions among older people. Thus, as with visual loss, the specific etiology of hearing loss is important in structuring preventive strategies.
DISABILITIES ASSOCIATED WITH SENSORY LOSS
The burden imposed on the elderly by the two specific sensory impairments discussed in this chapter has been inadequately studied. The prevalence of sensory impairment among various age groups is known, but nuances in prevalence among specific racial and ethnic subgroups are not. Sensory impairment is common among older adults because many common diseases and conditions can result in sensory impairment. A true understanding of this burden requires an understanding of the impact of sensory impairment. Unfortunately, in this area there are more questions than answers. As confusing as the data are for individual senses, they are still better than the virtually nonexistent data regarding the impact of multiple sensory impairments. More work is urgently needed to clarify the disabilities and handicaps associated with individual and multiple sensory impairments.
Although hearing and visual losses are each the result of a separate set of heterogeneous diseases, disabilities, or impairments,6,16,17,36,42,43,50 both types of losses can substantially decrease an individual's ability to interact with the environment and communicate with others.42 These sensory impairments, whose effects on an individual are seen as being similar, often lead to disability and handicaps. As a result, an exploration of the nature of the disabilities and handicaps associated with hearing and vision losses, and the evidence supporting their existence, seems the most logical place to begin. Social isolation, depression, cognitive loss, and psychoses, as well as common physical limitations, are often associated with hearing or vision deficits.4,8,10,12,29,48,49,51,53,54,57,58 However, the scientific evidence supporting the relationship of these handicaps to hearing and visual impairments is not strong enough to be considered conclusive.
Social isolation has been described among older adults with
sensory impairments and disabilities,58 but discerning the degree to which it can be attributed to either visual or hearing impairment is challenging. Because of the small sample sizes and variable definitions employed in studies of social isolation and sensory impairment, the strength and nature of their relationship are uncertain at best.24,29,58 The determinants of social isolation are multifactorial and might include age, educational level, living situation, and external factors such as the availability of transportation, in addition to sensory impairment. Two studies that consider social isolation among elderly individuals with either visual or hearing impairments conclude that these sensory deficits play a role in such isolation.24,58 The finding is strengthened by the observation that more severe hearing impairment is associated with greater degrees of social isolation.58 (In this study social isolation was measured with the Subjective Isolation Scale and the Objective Isolation Scale of the Comprehensive Assessment and Referral Evaluation.21 The measurement criteria for social isolation of the other study24 are available in the text.) Neither study, however, measures social isolation in a control group, although both use multiple regression to control for potentially confounding variables such as age and type of activity. Both studies report on clinic-based populations with sample sizes of less than 100. Given the small samples, limited generalizability, and study designs of this research, the relationship between social isolation and visual or hearing impairment must still be considered questionable.
Much of the work linking sensory impairment to psychiatric illnesses, such as paranoid psychosis, depression, or other affective disorders, lacks adequate control groups or thorough statistical analysis. A. F. Cooper and colleagues have published several studies reporting a high prevalence of long-standing hearing impairment among elderly patients with paranoid psychoses and affective disorders.7,8,9 (The mean age of paranoid patients studied was 67.3 years, and the mean age of those with affective disorders was 69 years.) In general, Cooper found that patients with paranoid psychoses had a higher prevalence of hearing impairment and disability than those with affective disorders. Patients with paranoid psychoses also tended to have more severe hearing impairment over all of the frequencies tested.9 Cooper also reported an increased incidence of psychoses in elderly patients with decreased visual acuity.10
Whereas Cooper's early work lacked controls, other such studies have not. For example, a study conducted at Johns Hopkins Hearing and Speech Clinic involving 174 outpatients found that 47 percent of those with some hearing impairment scored high on a screening test for psychiatric illness, as compared with 30 percent of those with
normal hearing.49 Similarly, a population-based study involving 365 patients over the age of 70 living in a London borough found a significant relationship between depression and deafness that persisted even after controlling for age and socioeconomic status.18 There was no relationship, however, between the severity of the hearing impairment or disability and the severity of the depression.
In short, much of the work in this area is descriptive. The basic assumption made in all these studies—that poor hearing actually preceded the psychiatric disease—is not confirmed; this must be tested through a prospective study. A case-control study that controls for most potential confounders also has yet to be performed. Although later studies are suggestive, they are not definitive, and because many studies are done abroad, their results are not generalizable to all American populations. These issues are further obfuscated by a contradictory cohort study among elderly hospitalized psychiatric patients in England that did not show the expected association between paranoid and affective disorders and sensory deficits.34 This study has methodological limitations, however, similar to those already mentioned. Thus, the relationship between sensory impairments and serious psychiatric illness is only suggestive at best. Furthermore, the small samples of most studies do not allow comment on the relationship of an additional variable—that is, age.
Until very recently, the relationship between sensory impairments and cognitive deficits has been fraught with similar methodologic problems. Several studies have concluded that a relationship exists between either visual or hearing impairment and dementia.41,49,53,54,58 Other studies have not been able to document such a relationship.18,51 The lack of adequate controls, limited sample sizes, and heterogeneous definitions of both sensory impairment and cognitive deficits that characterize most of these studies makes it difficult to interpret their findings and come to any definitive conclusions. Recently, Uhlman conducted a case-control study involving 100 cases of Alzheimer's type dementia and 100 nondemented controls matched by age, sex, and education.54 This study reported a higher prevalence of significant hearing impairment (odds ratio, 2) among Alzheimer's cases. Even though the findings of the available studies do not allow definitive conclusions to be drawn with regard to the relationship between hearing impairment and cognitive decline, the Uhlman study adds weight to the suggestion that hearing loss is associated with cognitive dysfunction. A similar statement cannot as yet be made with regard to visual impairment and cognitive loss.49
Sensory loss disabilities have yet to be fully characterized, although the literature suggests several potential types. Surveys
conducted by the National Center for Health Statistics indicate that older adults consider visual impairment but not hearing impairment to be among the six leading contributors to their perceived disability.37 A recent article reported a correlation between visual impairment and hip fracture.14
When six categories of leisure activities were evaluated in a group of low-vision elderly, the participants reported a significant decrease in active and sedentary crafts, such as gardening and auto maintenance or sewing and knitting, respectively.24 There were no significant changes in travel, sports, social activities, the use of radio, or television.24 Data gathered in the second and third waves of the Massachusetts Health Care Panel Study indicate that older noninstitutionalized individuals who report visual decline are more likely to need assistance in the so-called instrumental activities of daily living* than those who report continuing excellent or good vision. There were no significant differences, however, in the performance of the activities of daily living* or in bed days between the two groups. The Massachusetts study also indicated that older individuals with declining vision tended to have other disabilities (e.g., difficulties related to stair climbing or heavy housework) and handicaps in social function related to transportation. In addition, mental health and self-perceptions of health were likely to be problematic among the visually impaired.4
Finally, losing the ability to drive in the United States can be a serious handicap, particularly in areas where public transportation is limited. The clinical determinants of driving skills have yet to be established; however, evidence is mounting that age-associated loss of peripheral vision may be a significant factor in driving performance.30,44 To ensure both the independence of older people and the public safety, driver retraining courses for older people should be directed toward improving some age-related declines in sensory function and in learning to compensate for others.
In summary, many of the associations postulated between sensory losses and various emotional or physical disabilities are potentially
devastating and as such have tremendous implications for an older adult's quality of life. Yet the vast majority of the studies describing these associations have serious methodological limitations—for example, study designs that do not include control groups or a population-based sampling procedure. The lack of standards for determining which age groups to study and the lack of criteria to define sensory impairments and the nature and severity of the resulting disabilities make comparison across studies difficult. Therefore, although the relationship between sensory impairment and physical, emotional, or cognitive disabilities is intuitively obvious, it is impossible to make definitive statements regarding that relationship at this time.
APPROACHES TO PREVENTION
Preventing sensory loss logically involves interventions at primary, secondary, and tertiary points in the etiologic chain. Approaches that involve the actual prevention of sensory loss have yet to be developed. Such primary preventive strategies will necessarily be related to specific etiologies of sensory loss and are likely to involve screening for early sensory losses that create impairments and disability. This review uncovered no literature that addressed the potential benefits of primary prevention for sensory impairment in populations over the age of 50.
Most of the literature focuses on secondary preventive practices, in particular, screening, or the timing of therapeutic interventions to prevent disability. The recommendations for screening vary with the particular disease being addressed. Screening for visual impairment resulting from glaucoma, for example, requires a very different approach than screening for visual impairment from cataracts.
The issues related to glaucoma screening were recently reviewed by the Office of Technology Assessment.43 Once a person is screened for glaucoma and found to have the disease, treatment aimed at reducing intraocular pressure is the standard of care, even though such treatment has not been shown definitively to prevent blindness. The data are strongly suggestive, and there are randomized controlled trials currently in progress, but conclusions regarding the utility of early detection and treatment of glaucoma in the prevention of blindness cannot be made at this time.43 There is inconclusive evidence that early detection and treatment can prevent visual loss owing to macular degeneration.
There are several ways to screen for hearing impairment. For example, there are a number of physical measures and scales that seem to correlate reasonably well with pure tone audiometry, the
current ''gold standard."32,52 Pure tone audiometry can be made more or less sensitive and specific depending on the frequencies tested and the decibel threshold used to define hearing loss. Third, there are hearing handicap scales.11,25 Studies correlating perceived handicaps with pure tone audiometry indicate that the perception of a handicap correlates best with pure tone losses above 40 dB among the elderly. Perceived disability may be related to educational level, as well as economic status and participation in activities.56 Preventing disability or handicap from sensory impairment in vision or hearing therefore cannot rely solely on screening strategies.6,43
Theoretically, disability can be prevented by restoring the lost sense, which is the guiding principle of the strategy in senile cataracts and presbycusis. However, restoring a sense does not always prevent disability or restore function. For example, in the absence of underlying retinal disease, cataract removal and lens implantation has been shown in most cases to be effective in restoring reasonable vision.50 The crucial issue is the timing of the surgery and determining whether surgical intervention will improve the disability. Studies have shown that functional status improves in some patients following cataract surgery and that the improvement persists at least to the one-year follow-up point;1,13 the aspects and extent of disability and handicap will vary according to the demands and expectations placed on individuals. Factors other than the technical outcome of surgery, however, are also important in improving functional status—for example, mental status, level of functioning, measured visual acuity, and age.1
Very little work has been done to evaluate the effectiveness of aural rehabilitation in diminishing disability arising from hearing impairment. A small study involving 18 subjects demonstrated that disability (as measured by the Hearing Handicap Inventory for the Elderly) decreased after treatment with a hearing aid.46 However, this sample is too small to allow any generalization of its results to other populations. There have been no studies to demonstrate improvement in cognitive function, decreased depression, or greater social participation after aural rehabilitation.40,46 Although the literature suggests that a hearing aid is only one component of a rehabilitation program, there has been no systematic evaluation of other possible components that might be appropriate for older adults (e.g., lip reading, telephone amplification).40
Evaluating the degree to which disability can be prevented by developing strategies to minimize specific functional deficits related to sensory impairment is impossible based on current knowledge. As the first section of this chapter notes, there is no literature that
convincingly ascribes specific functional deficits, be they physical, emotional, or social, to sensory losses. For now, general conclusions about the ability to prevent disability and handicap by addressing sensory loss in older adults must await further study.
RECOMMENDATIONS
Policymakers and Government Agencies
In 1979 Robert Butler and Barbara Gastel presented a description of research challenges related to hearing and age to the National Institute on Aging.5 Many of the same issues were raised in 1986 when an international work group of hearing experts gathered to discuss hearing problems in the elderly.6 Unfortunately, most of the recommendations made in 1979 and 1986 are still applicable today. These two reports highlight a characteristic feature of the entire sensory impairment literature: an inadequate understanding of the nature of the disabilities and handicaps associated with sensory impairment and therefore an inadequate understanding of how to approach prevention. The specific recommendations presented below are designed to fill some of the gaps in information in these areas.
-
Services or devices that appear to help prevent disability or handicap owing to sensory impairment should be evaluated for availability and accessibility. Many of these services or devices are expensive, and their costs are not reimbursed by public or private insurance plans in this country. Comparisons with other nations that cover these services should be helpful.
-
Sensory impairment should remain a high priority for research. Projects designed to improve our understanding of the nature of the disabilities associated with sensory impairment are a crucial first step. It is important that individual sensory impairments be examined, but it is equally important to examine multiple sensory impairments to understand their interactive effects on disability and handicap.
-
The development of uniform criteria for the definition of individual sensory impairments (e.g., hearing impairment, visual impairment) should be encouraged.
-
Funding should be increased for programs aimed at educating older adults about sensory impairments and their prevention, treatment, and sequelae.
-
Researchers should be encouraged to incorporate investigations of sensory impairment into longitudinal studies. The natural
-
history of age-related sensory impairment is poorly understood, and longitudinal studies are the best way to address this deficit.
-
The sections of regularly conducted national surveys that address sensory impairment should be expanded.
-
Studies of sensory impairment in minority populations should be increased, especially in instances in which current data suggest there is increased risk among some populations.
Professional Societies
Professional societies and other such organizations are in the best position to coordinate efforts toward standardization, encourage cooperative study, establish guidelines, and educate their members as well as the public. Especially in the case of hearing impairment, there is a particular need to appreciate the problems faced by older adults.20,28
-
Uniform definitions of sensory impairment that can be used by all concerned disciplines should be developed and promoted.
-
Uniform measurements of disability as well as handicap related to individual and multiple sensory impairments should be developed. These efforts should build on well-validated measures of functional status (physical, mental, emotional, and social).
-
The implementation of multicenter, randomized controlled trials should be promoted to investigate screening and treatment practices.
-
Educational programs should be developed for the elderly person who may already be suffering from untreated or inadequately treated sensory losses. Programs should also be designed for the homebound or illiterate elderly.
-
Interdisciplinary bodies should be established to address the issues related to multiple sensory losses, including prevalence, interactions among sensory losses, and the impact of multiple sensory deficits.
-
Existing guidelines and regulations covering the dispensing of devices for the treatment of sensory losses, particularly hearing loss, should be reviewed.
-
Creative mechanisms should be developed to reduce the financial barriers that prevent older adults from receiving devices and services.
Clinicians
-
Sensory evaluations should always be included in routine, periodic evaluations. Although referrals may not always be necessary,
-
it is appropriate to question the patient regarding any sensory deficits or diminishment he or she may be experiencing.
-
Special attention should be given to medications and conditions that might contribute to sensory impairment.
-
Patients with perceived decreased hearing should be referred for screening by an audiologist.
-
Diabetic patients should be referred to an ophthalmologist annually.
-
Biannual screening should be recommended to all patients at high risk for glaucoma. Tonometry alone is not adequate screening for glaucoma.
-
Because many older patients are poorly informed about sensory loss, clinicians should develop a library of patient information on sensory impairment, its screening, disabilities, and treatments.
-
Patients should be taught the signs and symptoms of sensory impairment—particularly those related to hearing and visual impairment.
-
Patients should be taught how to minimize the safety risks associated with sensory impairment.
-
Clinicians should discuss the importance of adequate glare-free lighting with their visually impaired patients.
-
Magnifying glasses should be prescribed for patients with macular degeneration that is still at an early stage.
-
Clinicians should provide information on large-print books and newspapers to visually impaired patients.
-
All patients should be questioned routinely to uncover hearing impairment.
-
Elderly patients should be taught how to position themselves in a room to minimize visual and hearing deficits.
REFERENCES
1. Applegate, W. B., Miller, S. T., and Elam, J. T. Impact of cataract surgery with lens implantation on vision and physical function in elderly patients. Journal of the American Medical Association 1987; 257:1064-1066.
2. Bartoshuk, L. M., Rifkin, B., and Marks, L. E. Taste and aging. Journal of Gerontology 1986; 41:51-57.
3. Bingea, R. L., Raffin, M. J. M., Aune, K. J., et al. Incidence of hearing loss among geriatric nursing home residents. Journal of Auditory Research 1982; 22:275-283.
4. Branch, L. G., Horowitz, A., and Can, C. The implications for everyday life of incident self-reported visual decline among people over age 65 living in the community. The Gerontologist 1989; 29(3):359-365.
5. Butler, R. N., and Gastel, B. Hearing and Age: Research challenges and the National Institute on Aging. Annals of Otology 1979; 88:676-683.
6. Christiansen, J., Eric, J., Kennedy, T. E., et al. Hearing problems and the elderly. Danish Medical Bulletin 1986; 33:1-22.
7. Cooper, A. F. Deafness and psychiatric illness. British Journal of Psychiatry 1976; 129:216-226.
8. Cooper, A. F., and Curry, A. R. The pathology of deafness in the paranoid and affective psychoses. Journal of Psychosomatic Research 1976; 20(2):97-105.
9. Cooper, A. F., Curry, A. R., Kay, D. W. K., et al. Hearing loss in paranoid and affective psychoses of the elderly. Lancet 1974; 2(7885):851-854.
10. Cooper, A. F., and Porter, R. Visual acuity and ocular pathology in the paranoid and affective psychoses of later life. Journal of Psychosomatic Research 1976; 20:107-114.
11. Corbin, S., Reed, M., Nobbs, H., et al. Hearing assessment in homes for the aged: A comparison of audiometric and self-report methods. Journal of the American Geriatrics Society 1984; 32:396-400.
12. Eastwood, M. R., Corbin, S. L., Reed, M., et al. Acquired hearing loss and psychiatric illness: An estimate of prevalence and co-morbidity in a geriatric setting. British Journal of Psychiatry 1985; 147:552-556.
13. Elam, J. T., Graney, M. J., and Applegate, W. B. Functional outcome one year following cataract surgery in elderly persons. Journal of Gerontology 1988; 43:M122-M126.
14. Felson, D., Anderson, J., Hannan, M., et al. Impaired vision and hip fracture: The Framingham Study. Journal of the American Geriatrics Society 1989; 37:495-500.
15. Ferris, F. L. Senile macular degeneration: Review of epidemiologic features. American Journal of Epidemiology 1983; 118:132-149.
16. Ghafour, M. M., Allan, D., and Foulds, W. S. Common causes of blindness and visual handicap in the rest of Scotland. British Journal of Ophthalmology 1983; 67:209-213.
17. Gilad, O., and Glorig, A. Presbycusis: The aging ear, Part I. Journal of the American Auditory Society 1979; 4:195-217.
18. Gilhome-Herbst, K., and Humphrey, C. Hearing impairment and mental state in the elderly living at home. British Medical Journal 1980; 281:903-905.
19. Gilhome-Herbst, K., and Humphrey, C. Prevalence of hearing impairment in the elderly living at home. Journal of the Royal College of General Practitioners 1981; 31:155-160.
20. Gluck, M. E., Wagner, J. L., and Duffy, B. M. The Use of Preventive Services by the Elderly. Washington, D.C.: Health Program, Office of Technology Assessment, Congress of the United States, January 1989.
21. Gurland, B., Kuriansky, J., Sharpe, L., Simon, R., Stiller, P., and Birkett, P. The Comprehensive Assessment and Referral Evaluation (CARE)—Rationale, development and reliability. International Journal of Aging and Human Development 1977-1978; 8(1)9-42.
22. Hart, W. M., and Becker, B. The onset of evolution of glaucomatous visual field defects. Ophthalmology 1982; 89:268-279.
23. Havlik, R. J. Aging in the eighties: Impaired senses for sound and light in persons age 65 years and over. Preliminary data from the supplement on aging to the National Health Interview Survey: United States, January-June 1984. DHHS Publ. No. (PHS)86-1250. Advance data from Vital and Health Statistics of the National Center for Health Statistics, No. 125, September 19, 1986.
24. Heinemann, A. W., Colorez, A., Frank, S., et al. Leisure activity participation of elderly individuals with low vision. The Gerontologist 1988; 28:181-184.
25. High, W. S., Fairbanks, G., and Glorig, A. Scale for self-assessment of hearing handicap. Journal of Speech and Hearing Disorders 1964; 29:215-230.
26. Hiller, R., and Kahn, H. A. Blindness from glaucoma. American Journal of Ophthalmology 1975; 80:62-67.
27. Hinchcliffe, R. Hearing levels of elderly in Jamaica: A pilot survey. Annals of Otology, Rhinology and Laryngology 1964; 73:1012-1019.
28. Humphrey, C., Gilhome-Herbst, K., and Faurqi, S. Some characteristics of the hearing-impaired elderly who do not present themselves for rehabilitation. British Journal of Audiology 1981; 15:25-30.
29. Jones, D. A., Victor, C. R., and Vetter, N. J. Hearing difficulty and its psychological implications for the elderly. Journal of Epidemiology and Community Health 1984; 38:75-78.
30. Keltner, J. L., and Johnson, C. A. Visual function, driving safety, and the elderly. Ophthalmology 1987; 94(9):1180-1188.
31. Leske, M. C. The epidemiology of open-angle glaucoma: A review. American Journal of Epidemiology 1983; 118(2):166-191.
32. Lichtenstein, M. J., Bess, F. H., and Logan, S. A. Validity of screening tools for identifying hearing-impaired elderly in primary care. Journal of the American Medical Association 1988; 259:2875-2878.
33. Mitchell, P. Prevention of blindness in Australia. Australian Family Physician 1985; 14:757-765.
34. Moore, N. C. Is paranoid illness associated with sensory defects in the elderly? Journal of Psychosomatic Research 1981; 25:69-74.
35. Moscicki, E. K., Elkins, E. F., Baum, H. M., et al. Hearing loss in the elderly: An epidemiologic study of the Framingham Heart Study Cohort. Ear and Hearing 1985; 6:184-190.
36. Murphy, R. P. Age-related macular degeneration. Ophthalmology 1986; 93:969-971.
37. National Center for Health Statistics (Moss, A. J., and Parsons, V.). Estimates from the National Health Interview Survey, U.S., 1985. DHHS Publ. No. (PHS) 86-1588. Washington, D.C.: National Center for Health Statistics, 1986.
38. National Center for Health Statistics. Refraction status and motility defects of persons 4-74 years. United States 1971-1972. Publ. No. (PHS) 86-1250. Vital and Health Statistics, Series 11, No. 206, 1978, pp. 89-93.
39. Nelson, K. A. Visual impairment among elderly Americans: Statistics in transition . Journal of Visual Impairment and Blindness 1987; 81:331-334.
40. Newman, C. W., and Weinstein, B. E. The hearing handicap inventory for the elderly as a measure of hearing aid benefit. Ear and Hearing 1988; 9:81-85.
41. Ohta, R. J., Carlin, M. F., and Harmon, B. M. Auditory acuity and performance on the mental status questionnaire in the elderly. Journal of the American Geriatrics Society 1981; 29:476-478.
42. Ordy, J. M., and Brizee, K. R. Sensory Systems and Communication in the Elderly. New York: Raven Press, 1979.
43. Power, E. J., Wagner, J. L., and Duffy, B. M. Screening for Open-Angle Glaucoma in the Elderly. Washington, D.C.: Health Program, Office of Technology Assessment, Congress of the United States, October 1988.
44. Retchin, S. M., Cox J., and Fox, I. L. Performance based measurements
among elderly drivers and nondrivers. Journal of the American Geriatrics Society 1988; 36(9):813-819.
45. Rosen, S., Bergman, M., Plester, D., et al. Presbycusis study of a relatively noise-free population in the Sudan. Annals of Otology, Rhinology and Laryngology 1962; 71:727-743.
46. Rousseau, P. Hearing loss in the elderly. American Family Physician 1987; 36(3):107-113.
47. Schow, R. L., and Nerbonne, M. A. Hearing levels among elderly nursing home residents. Journal of Speech and Hearing Disorders 1980; 45:124-132.
48. Singerman, B., Riedner, E., and Folstein, M. Emotional disturbance in hearing clinic patients. British Journal of Psychiatry 1980; 137:58-62.
49. Snyder, L. H., Pyrek, J., and Smith, K. C. Vision and mental function of the elderly. The Gerontologist 1976; 16:491-495.
50. Straatsma, B. R., Foos, R. Y., and Horowitz, J. Aging-related cataract: Laboratory investigation and clinical management . Annals of Internal Medicine 1985; 102:82-92.
51. Thomas, P. D., Hunt, W. C., and Garry, P. J. Hearing acuity in a healthy elderly population: Effects on emotional, cognitive, and social status. Journal of Gerontology 1983; 38:321-325.
52. Uhlmann, R. F., Rees, T. S., Pasty, B. M., et al. Validity and reliability of auditory screening tests in demented and non-demented older adults. Journal of General Internal Medicine 1986; 4(2):90-96.
53. Uhlmann, R. F., Larson, E. B., and Koepsell, T. D. Hearing impairment and cognitive decline in senile dementia of the Alzheimer's type. Journal of the American Geriatrics Society 1989; 34:207-210.
54. Uhlman, R. F., Larson, E. B., Rees, T. S., et al. Relationship of hearing impairment to dementia and cognitive dysfunction in older adults. Journal of the American Medical Association 1989; 261:1916-1919.
55. Ventry, I. M., and Weinstein, B. E. Identification of elderly people with hearing problems. ASHA 1983; 25(7):37-42.
56 Weinstein, B. E. Validity of a screening protocol for identifying elderly people with hearing problems. ASHA 1986; 28(5):41-45.
57. Weinstein, B. E., and Amsel, L. Hearing loss and senile dementia in the institutionalized elderly. Clinical Gerontologist 1986; 4:3-15.
58. Weinstein, B. E., and Ventry, I. M. Hearing impairment and social isolation in the elderly. Journal of Speech and Hearing Research 1982; 25:593-599.


















