3
Overview of Neuroscience Research: A Closer Look at the Neural Hierarchy
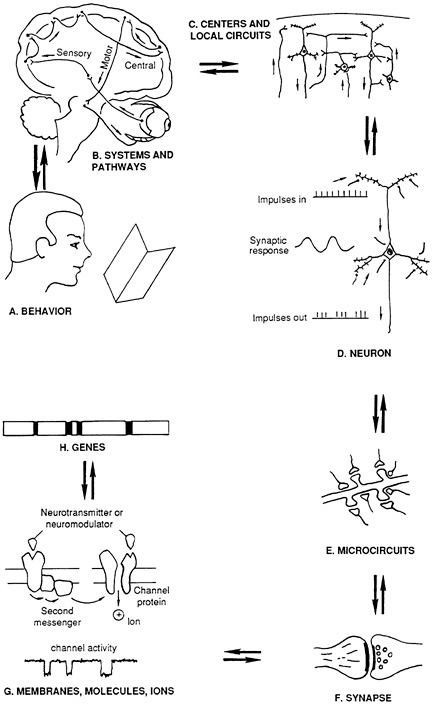
This chapter is intended as a primer or simplified overview of some aspects of neuroscience for those readers not familiar with the field. As such, the chapter describes some of the experiments that are done at each level of a vertical hierarchy of neural functioning, from behavior to genetic mechanisms (Figure 3-1 ). Researchers have developed hundreds of techniques and formulated elaborate repertoires of experimental strategies to answer fundamental questions about brain functions. When combined, these techniques provide information at all levels of the vertical hierarchy. Although apparently distinct, these levels cannot be separated because information that exists at one horizontal plane must be considered in concert with information from all other planes to generate a coherent picture of brain functioning. In addition, at each vertical level, the results of many different experimental approaches need to be integrated.
Behavior and emotion are manifestations of brain activity
Behavior encompasses many degrees of complexity, from the most rudimentary knee-jerk reflex to the subtlety and sophistication of movement exhibited by a ballet dancer or chess grand master. The study of behavior can be subdivided into several broad facets: sensory processing (vision, hearing, touch, and smell leading to conscious perception), motor processing (reflexes and coordinated movements), and cognitive processing (learning, thinking, and planning). Behavior also includes psychological processes, such as personality and mood.
Animals as simple as fruit flies and snails exhibit behaviors that are neurally driven. As organisms become more complex, their behaviors become correspondingly more intricate. Human behavior represents the ultimate in rich and elaborate repertoires that are affected by a range of factors from “hard-wired” genetic codes to minute-by-minute environmental perturbations. Behavior and its neural underpinnings must also be understood across different developmental stages and different abnormal or pathological conditions. The challenge in neuroscience is to understand the contributions of different factors to specific behavioral and emotional functions. Because important factors exist at all levels of the hierarchy, from organized brain systems to individual cells and even genes, this formidable challenge can be met only by invoking a diverse and powerful arsenal of experimental tools.
Many behavioral studies in animals employ conditioning techniques first developed by Pavlov in the late 1920s. By pairing a bell with the presentation of food to a dog, Pavlov was able to “condition” the dog to respond physiologically with increased gastric secretion when only the bell was sounded and no food was present. (In other words, a bell/food association was established, and this mental association produced a physiological response in the animal's body.) Later research efforts developed the concept of operant conditioning: animals can be taught, using associations of food and sensory stimuli such as sound or light, to press bars, enter doors, or perform some other activity. Moreover, once an animal is trained to perform a specific activity, new associations can be taught; how well these activities are learned, as well as the disruption of well-learned activities, can then be assessed using a variety of experimental manipulations. For example, many psychotropic drugs, including morphine and nicotine, have been tested by operant conditioning in studies aimed at finding replacement drugs that are perceived by the user to be similar to the real drug but that have no addictive properties. Finally, because operant conditioning models involve learning, behavioral experiments with rats born of alcohol-ingesting mothers or rats exposed to lead poisoning have contributed to an understanding of some of the learning deficits that result from prenatal or environmental exposure to toxic substances.
Other behavioral research has involved the selective destruction or electrical stimulation of discrete brain regions and the subsequent observation of behavioral changes. Such experiments helped to associate a particular part of the hypothalamus with the regulation of eating behavior, certain limbic system nuclei with aggressive behavior, specific nuclei of the diencephalon with a variety of movement disor-
ders, and the hippocampus with memory deficits. As more is learned about the presence, function, and location of brain neurotransmitters and neuromodulators, these kinds of investigations are expanding to measure the effects of loss and replacement of specific neurochemicals.
Learning and memory can also be measured in nonhuman animals. Simple organisms, such as the fruit fly, can be classically conditioned, and, amazingly, certain specific genetic mutations in the fruit fly are known to compromise severely this type of learning (Dudai, 1985). Because there are important similarities, or homologies, between the genes of fruit flies and those of higher animals, identification of the protein encoded by the defective gene may provide a clue to the genetic basis of learning in higher animals.
Animals such as monkeys are capable of complex learning tasks— for example, long chains of discrete movements, association of symbols with specific movements, association between different objects, or manipulation of spatial relationships between objects. These tasks can be designed in such a way as to distinguish short-term memory from long-term memory and to allow precise measurement of learning acquisition. Behaviors such as these can then be used as benchmarks for a range of manipulations of brain structure and chemistry. Electrical recordings of brain activity made during specific tasks can help localize the part of the brain that mediates the information processing and control of movements necessary for the task. These tasks are also useful for assessing the deficits caused by experimental brain injury or stroke.
The aim of behavioral research in animals is to discover neural processes that can be correlated with human brain functions. Yet the techniques applied to human behavior have also sought to go beyond such correlations and acquire a window into cognitive and emotional function. For a long time, behavioral research in humans was severely limited by the paucity of techniques appropriate for human experimentation. Now, a number of new techniques permit the study of human brain function. One important step was the advent of noninvasive recording methods such as electroencephalographic (EEG) recordings, which made it possible to associate certain behavioral states with changes in the electrical activity of the cerebral cortex. (The recordings exhibit different patterns during different states of consciousness, including quiet wakefulness, alert wakefulness, and sleep.) Following its development, the EEG technique was immediately applied to the study of many human neurological and mental illnesses.
More recently, it has become possible to discriminate between different kinds of brain electrical activity by amplifying particular com-
ponents of the EEG recordings during stimulation of specific visual, auditory, or sensory systems. These so-called event-related potential (ERP) recordings, which are essentially more finely tuned EEGs, contain specific waveforms that can be mapped, albeit with limited resolution, to specific structures of the brain. ERPs have been used to diagnose brain injury locations, describe abnormalities in schizophrenic patients, and trace the pathways underlying certain types of information processing. In the past five years, techniques have begun to be developed to record the magnetic rather than electrical fields that are generated by cortical activity. Magnetoencephalography, or neuromagnetic imaging, has been applied to the study of epileptic seizure activity and may in the future add greater resolution to the study of human brain activity.
Another exciting development is the ability to measure simultaneously the activity of most of the discrete brain regions in humans, during a variety of situations, with positron emission tomography. In PET scanning, short-lived radioactive markers are attached to specific molecules and injected into the bloodstream. These tagged molecules can then be visualized by detectors outside the brain and the resulting images stored in a computer (Plate 3-1 ). In some cases, the tagged molecules trace the blood flow to or the uptake of glucose by the brain. Tracing these patterns in the nervous system helps to identify brain regions with higher activity levels—they generally exhibit increased blood flow and increased uptake of glucose. Likewise, decreased blood flow and glucose uptake are associated with those brain regions that are not as active or that are being inhibited. Researchers have also used PET to visualize the binding of various drugs and neurotransmitter substances to receptors in the brain during discrete behavioral tasks and in certain emotional states.
Because PET studies can be repeated several times, they can be used to evaluate clinical treatments for behavioral and emotional disorders, in addition to their use in diagnosis or basic research. PET screening permits the reassessment of individual subjects after the administration of therapeutic agents or therapies and thus may show a return to or approximation of normal brain activity following successful therapies. Clearly, the use of PET in normal subjects also has great potential for increasing basic understanding of the brain and its processes in addition to providing a baseline from which to judge experimental therapies. So far, PET has provided the best and most complete information on humans with which to correlate the mass of data from studies of nonhuman animals, and its use will facilitate a more rapid transfer of basic science research to human beings.
Magnetic resonance imaging is another technology that has proved
to be useful in visualizing brain functions. MRI uses external magnetic fields to visualize the three-dimensional structure of the brain at a resolution much better than that provided by x-rays or computer-assisted tomography (CT). Even though MR and CT images are essentially views of static structures and do not reflect dynamic changes, their improved resolution has had an immediate impact on the localization of brain damage from stroke, Alzheimer's disease, epilepsy, and brain cancer. Furthermore, numerous efforts are under way to achieve the precise overlay, or registration, of MR images with PET images so that the anatomical localization of PET data can be improved. The future should see the improvement of MRI techniques to allow visualization of particular molecules, such as phosphorus, to reveal dynamic changes in neurochemical activity over time.
The systems of the brain are connected by elaborate pathways and serve many functions
Although behavioral research has associated many functions with specific parts of the brain, resolution of precisely how neural computation occurs depends on knowledge of how various brain regions are connected to each other and how activity in one region affects other regions. This level of the hierarchy is concerned with organized groups of neurons—“areas” of the cerebral cortex and “nuclei” of subcortical regions. Groups of nuclei and areas make up the major systems of the brain, and vast networks of connections link these components. At the turn of the century, the development and use of histochemical stains revealed the rich variety of neuronal types and allowed researchers to map and identify many of the interconnections among major systems of the brain.
For many years this early information guided initial studies of factors crucial to the development of certain brain regions. For example, research showed that the neurons in the visual part of the cerebral cortex in kittens, who were prevented from seeing by birth defects or experimental closing of the eyelids at birth, did not develop properly. This finding provided the important knowledge that proper development depended on the ability of the system to function (also see Box 2-2 ).
More recent developments include neuroanatomical tract tracing techniques to analyze the circuitry of the brain. These techniques take advantage of the fact that neurons absorb certain substances, which subsequently move from the axonal endings to the cell body or from the cell body to the axonal endings. These tracer substances can be made visible in a number of ways, and their presence or ab-
sence in specific regions of the brain indicates whether a region has connections to some other region. In other words, the substances allow investigators to demonstrate a connection between the region of the brain in which the substance was placed and the region to which it was transported. Sometimes it is possible to visualize the tracers, through high magnification with an electron microscope, in axonal endings that contact one specific neuron. Such high-resolution studies have determined that these connections are often to a specific part of a neuron and occur in a highly predictable pattern. In addition, by using more than one of these tracing substances at a time, it is possible to map multiple pathways of connections among neurons and neuron groups. Complementing the anatomical research have been electro-physiological studies of brain pathways and systems. Such efforts not only establish circuitry information but often shed light on the functional relationships between certain regions (because physiological responses to stimulation, such as excitation and inhibition, can also be measured).
Much of the present understanding of the networks of the brain has come from anatomical and physiological studies such as those described above. This work has generated conceptual frameworks that have greatly advanced our thinking about the brain and that have been borrowed by computer scientists who are attempting to build “thinking” computers. A key framework from these studies is the concept of parallel distributed processing of information. Rarely is there just one brain pathway for a certain type of information; instead, many pathways, involving tens of millions of neurons, run parallel to each other. Nevertheless, information is selectively distributed to specific regions of the brain by functionally distinct processing streams. This organization underlies the brain's ability to store, process, and mediate hundreds of kinds of information simultaneously and reliably.
Overlying the anatomically defined neural systems are the chemical systems of the brain. It is common for particular neurotransmitter substances to predominate in specific brain nuclei, and thus knowledge about the actions of these neurotransmitter substances must be added to knowledge of the known anatomical connections of a particular brain region. The functional importance of the interaction of anatomical and chemical systems is particularly well illustrated in a region of the brain known as the substantia nigra. This region contains many neurons that contain the neurotransmitter dopamine, and the loss of these neurons is responsible for most of the effects noted in Parkinson's disease (Plate 3-2 ).
Techniques for defining these chemical systems began to be developed in biochemical studies, which isolated bioactive neurochemicals
from specific brain regions. As these neurochemicals were characterized, their effects in discrete nuclei were assessed by a variety of techniques —for example, injecting tiny amounts of the neurochemical into the brain and recording the physiological response, or injecting the substance and measuring changes in an animal's behavior. A major breakthrough in the study of neurochemical systems was the development of antibody labeling techniques, or immunocytochemistry, in which antibodies are obtained that recognize and bind to a specific neurochemical. The antibodies can be tagged in various ways and used to visualize the neurochemical's specific locations in the brain. Very soon after this discovery, researchers had developed specific antibodies that recognized a number of neurochemicals (or their synthetic enzymes), and detailed maps of the locations of these neurotransmitters in the brain began to emerge. Throughout the past 10 years, detailed maps of the locations of scores of neurotransmitters and neuromodulators have been generated. Furthermore, techniques have been developed that combine tract tracing methods with immunocytochemistry and thus reveal simultaneously the connections and neurochemistry of discrete brain regions. Finally, the changes in these maps during development and following injury have been and continue to be actively examined.
Neurons come in many shapes and sizes, and contain different chemicals
Neurons are the signaling cells or basic functional units of the brain (Box 3-1 ). Each neuron has three major parts: cell body, dendrites, and axon. The cell body is the neuron's powerhouse and contains the biosynthetic machinery for making proteins and other molecules needed by the cell. Extensions of the cell body (processes), called dendrites, normally form a multitude of branching patterns, accounting in part for the many different shapes neurons exhibit. Most of the surface area of neurons is in the dendrites, which are the primary receiving areas for signals from other neurons. Neurons have only one axon extending from their cell bodies; the axon transmits neural signals from the cell body to other cells. All neurons are electrically charged (like a battery) as a result of different ion concentrations inside and outside the cell. When a neuron is excited, ions flow through the cell's membrane, and the voltage difference between inside and outside is briefly reversed. This voltage reversal, called an action potential, is propagated down the axon to its endings, where it causes the release of the neurotransmitters stored within these endings.
Many different kinds of neurochemicals are transported back and
|
BOX 3-1 NOT ALL NEUROSCIENCE RESEARCH IS CONCERNED WITH NEURONS Glia are the supporting cells of the brain and are not involved in synaptic transmission. More numerous than neurons, glial cells perform a variety of functions. For example, they constitute the insulating myelin sheaths that cover many axons. Microglia ingest debris from brain injury or normal cell death. Another type of glial cell guides the migration of developing neurons. Following brain injuries, glia proliferate and form scars. As researchers develop new methods to encourage injured axons to regrow, such scars could present a serious barrier. The blood-brain barrier is a physical barrier between the blood and brain tissue, formed by special junctions between the cells lining the cerebral blood vessels. The brain is exquisitely sensitive to changes in acidity, extracellular protein content, and other factors. The blood-brain barrier protects the brain from these changes by preventing many chemicals from entering the brain's extracellular spaces. However, the barrier also prevents many therapeutic agents, including anticancer and antibiotic drugs, from reaching the site of tumors or infections. Research has revealed some mechanisms for bypassing the blood-brain barrier and has identified conditions under which the barrier is disrupted. |
forth to the cell body in axons, sometimes along specialized protein structures (microtubules) that are like railroads along which little packets (e.g., vesicles containing neurotransmitters) of material can travel. (These same packets are sometimes responsible for transporting the pathway-tracing substances mentioned in the previous section.) Axons are important in development because directed growth of the axon from the developing neuron to its intended targets is critical to proper development of a whole brain. Although such growth is affected by a host of chemical cues and mechanisms, the transport of certain “trophic” factors by the axon is a key aspect of the growth process.
In addition to the more than 100 major neuronal types defined by size and shape, neurons differ in the kind of neurotransmitters and neuromodulators they produce. More than a hundred of these neurochemicals have been identified, and the list continues to grow. Some act to excite or to inhibit other neurons, and some act in subtle ways to enhance or depress these effects. Investigation of the regional location of specific neurotransmitters has indicated a role for the neurotransmitter acetylcholine in Alzheimer's disease. Analysis with immunocytochemistry in many regions has further revealed that
many neurons contain and release more than one neurochemical at a time. Moreover, the balance of functionally important neurochemicals may change during development. Sometimes one cell type can be the locus for significant neurological dysfunctions—as mentioned earlier, the specific population of cells that are damaged in Parkinson's disease. All of these factors must be studied to map the structural and functional organization of each region of the brain and spinal cord.
Researchers also employ electrophysiological techniques at the cellular level of analysis, which often involves the penetration of individual cells with microscopically small electrodes to study the activity of a single cell. A recently developed physiological technique that shows great promise is the use of voltage- and ion-sensitive dyes (Plate 3-3 ). These dyes are fluorescent substances that are taken up by neurons and that change colors depending on the voltage state of the cell or the ion content of the cytoplasm inside the neuron. Special microscopes can record these color changes (in response to a variety of experimental manipulations) and provide images of the changes over time and in discrete regions of the neuron. Researchers have applied this technique to the study of regional activation of specific neuronal types, activity of specific neurotransmitters, and activity of developing neurons.
Another recently developed technique, in situ hybridization, employs specially synthesized pieces of nucleic acids that have a sequence complementary to the RNA that codes for specific proteins within neurons. These complementary pieces, which can be tagged with radioactive labels, bind to the RNA of interest; exposing brain tissue to these probes, the presence and quantity of RNA coding for specific proteins can be visualized and measured (Plate 3-4 ). This technique is particularly valuable because it can measure, through the changes in RNA synthesis, the effect of many variables, including neural activity, on genetic regulation of protein synthesis.
Considerable overlap occurs between investigations at the cellular level, such as those described here, and investigations at the systems, synaptic, and molecular levels. In many respects, the individual cells are the nodal points where the various molecular and genetic processes combine to give rise to the structural and gross functional organization of the nervous system. Thus, investigators at many levels of the neural hierarchy must consider the information their efforts generate within the context of the neuronal types to which their data may apply.
Synapses and microcircuits filter vast numbers of messages
Synapses are highly specialized regions of neurons at which communication between cells takes place. Structurally, most synapses
are polarized, in that the sending part of one neuron (an axonal ending) makes contact with the receiving part of another neuron (usually a cell body or dendrite). Between these two components is a small space, called the synaptic cleft. When an action potential occurs in the sending, or presynaptic, neuron, it causes the release of neurotransmitter substances into the synaptic cleft. These substances diffuse across the space and bind to specific receptor molecules on the receiving, or postsynaptic, neuron. Because each neuron is studded with literally thousands of synapses, any one synapse contributes only a tiny amount to excitation or inhibition of a cell. An action potential is produced if the excitatory inputs to the cell outweigh the inhibitory inputs (Figure 3-2 ). Underlying this process are complex biophysical mechanisms related to the properties of the neuronal membrane and to the precise location and distribution of different kinds of inputs.
The structural aspects of synapses and synapse formation are most often studied using an electron microscope. Such investigations have delineated many of the cellular specializations that occur at the synapse and have described the function of these specializations. One of the most interesting phenomena documented by electron microscopic studies is the retraction of presynaptic axonal endings, which results from various types of damage to the presynaptic cell. Such retraction can be transient or permanent and, depending on the number of lost synaptic inputs, can fundamentally change the balance of inputs to the postsynaptic cell. The loss of synapses may also cause structural changes in the postsynaptic cells, which are often visible in regular microscopic analysis. For example, in many large neurons of the cerebral cortex, dendrites have tiny projections called dendritic spines. The density of these spines, which provide even more surface area for synapses, has been shown to decrease with advanced age, an effect that can also occur in certain neurological disease states (Plate 3-5 ).
The physiology of synaptic transmission has been investigated using a variety of electrophysiological techniques in a wide range of animal species. These experiments, combined with chemical analysis, provide a rich picture of the many events that occur in the synaptic endings and in the postsynaptic neurons: ionic changes, the release of precise amounts of neurotransmitters, and actions, in the synaptic cleft, of enzymes that sometimes degrade neurotransmitters and thus end their effects. For example, researchers know that the amount of transmitter released depends on changes in the intracellular concentration of calcium in the axonal ending. This characteristic provides a mechanism by which synaptic transmission can be modified or reg-
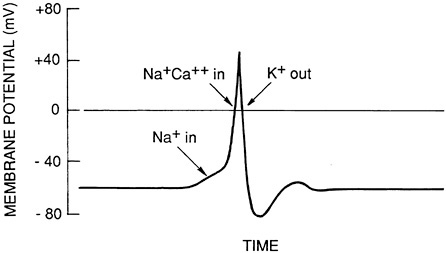
FIGURE 3-2 An action potential tracing. The action potential, the basis of neuronal signaling, is an electrical impulse generated by a change in the permeability of the cell membrane to sodium, calcium, potassium, and other ions. Adapted from Kandel and Schwartz, 1985.
ulated (varying the amount of transmitter that is released changes the likelihood of action potential generation in the postsynaptic cell). This ability to change is called plasticity and is another hallmark of neural information processing that computer scientists would like to reproduce in “thinking” computers.
Regulation of synaptic transmission has a number of interesting effects. It has been known for a long time that when a neuron is repeatedly stimulated, it often stops firing, despite-continued stimulation. This process is known as habituation and is an adaptive response that helps filter out stimuli. Researchers have shown that repeated stimulation of a presynaptic ending results eventually in a decrease in the calcium entering the ending. The synapse then “loses its strength, ” so that presynaptic action potentials result in decreased release of neurotransmitter. A reverse mechanism, called sensitization, helps strengthen particular inputs.
By balancing inputs and filtering out unnecessary information, these mechanisms are probably critical to the microcircuitry involved in learning and memory. Many different brain regions are likely to be involved in various aspects of memory and learning, but a common theme for all of them is the strengthening of certain inputs over others on the cellular and synaptic levels. Many investigators in the field of computational neuroscience are concerned with defining the
mathematical constraints of these processes. Their goal is to develop a deeper understanding of the nature of information storage in the brain and how it differs from information storage in conventional computers.
Messages can change molecular states and genetic expression
Neural signaling comprises much more than stimulus-response patterns. The molecular chemistry involved in transducing neural signals is an area of intense scientific interest and often serves as the point at which pharmacological treatments intervene. Excitation and inhibition of a neuron are mediated by ion channels that selectively regulate the flow of small ions, especially sodium, calcium, potassium, or chloride ions. Each ion channel is a complex molecule that is embedded in the cell membrane and contains a pore through which particular ions can pass. Most ion channels are either open or closed; these states are often “gated,” or determined, by specific external influences. Some channels are gated by the action of neurotransmitters, others by the voltage difference across the membrane, and still others by mechanical stimuli, as occurs in peripheral nerve endings to signal touch and pressure on the skin. The regulation of gated channels gives rise to action potentials and involves many different types of biochemical reactions that have implications for all kinds of excitable membranes in the body, including skeletal and cardiac muscle cells, egg cells of the ovary, and certain immune system cells.
The first step in synaptic transmission is the binding of a neurotransmitter to a receptor—similar to a key in a lock. Some receptors actually form an ion channel when activated, and neurotransmitter binding to the receptor changes the structure of the pore so as to facilitate or inhibit ions from crossing the membrane. A specific type of acetylcholine receptor, which mediates muscle movement, is part of an ion channel, as are certain receptor subtypes for gamma aminobutyric acid (GABA), the receptor at which certain tranquilizers, such as Valium®, act. Other neurotransmitter-receptor complexes, however, initiate a complicated cascade of biochemical reactions that culminate in the structural modification of distinct ion channels. Such cascades involve so-called second messenger molecules, which function to activate other molecules.
Cyclic adenosine monophosphate (cAMP), a nucleotide molecule, is the best understood of these second messengers. cAMP is formed after many neurotransmitters attach to their receptors—including the type of receptor for norepinephrine that mediates increases in heart rate and is blocked by the beta blocker drugs that many heart pa-
tients take. At this receptor, the binding of transmitters causes the linking of the receptor to a protein, called a G protein, which transduces the signal by activating an enzyme (adenylate cyclase) that in turn causes the production of cAMP (Figure 3-3 ). cAMP then activates other enzymes (protein kinases) that cause phosphate groups to be added to other molecules, some of which may interact with the ion channel proteins. To make matters even more complex, some G proteins are inhibitory and some excitatory to this cascade. Other
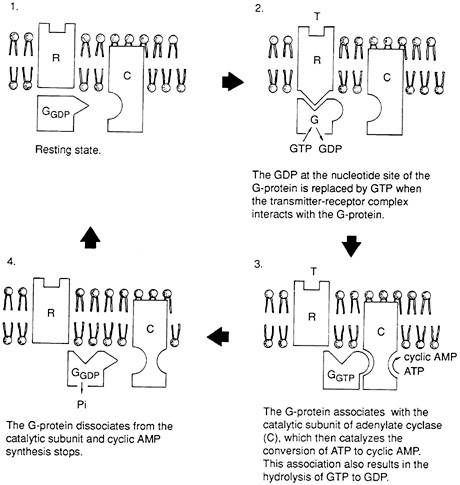
FIGURE 3-3 The synthesis of cyclic adenosine monophosphate (cAMP) that results when transmitter (T) binds to the beta-adrenergic receptor (R) is mediated through a transducer, or G-protein (G). GTP = guanosine triphosphate; GDP = guanosine diphosphate. Adapted from Kandel and Schwartz, 1985.
second messengers are lipids such as diacylglycerol, which activates protein kinase C in a cascade of chemical reactions usually distinct from that involving cAMP. Calcium, sometimes bound to proteins such as calmodulin, is yet a third second messenger that functions to activate a number of different enzymes. It is important to note that the chemical reaction cascades involving the three second messengers described here often intersect at key molecular points, allowing the different messenger systems to interact with each other.
Second messenger systems are a good example of the concept that the effects of synaptic transmission are not limited to the regulation of ion channels and the resulting excitation or inhibition of action potentials. Synaptic transmission also influences, to a greater or lesser degree, all of the biosynthetic processes of the cell, from the synthesis of neurotransmitters and receptors to the synthesis of proteins necessary to maintain the cell's structure. Such effects are the late consequences of cell activation and involve the regulation of genetic expression.
Certain genes found in human cells, including brain cells, are similar to viral genes implicated in cancer. These so-called proto-oncogenes have been found in brain cells, and it is thought that these genes function as third messengers because they code for proteins that in turn regulate gene expression. Soon after nerve injury, the expression of one of these oncogenes, called cfos, increases. The functional importance of this increase is not known but, among other possibilities, may well have important consequences for cellular repair. As mentioned previously, neurons synthesize different amounts of neurotransmitters and other neurochemicals depending on the developmental stage of the organism and other factors. The genetic regulation that controls the balance of these biosynthetic processes is another area with critical implications for normal and pathological brain functioning as well as for the response of the brain to injury.
Neuroscientists are also investigating the genes contained in ceils of the nervous system through gene mapping, gene and protein sequencing techniques, and antibody techniques. For example, anti-bodies can be used to isolate a substance—a complex protein, for example—whose molecular structure is not known but whose presence is critical to some aspect of neural function. Once the molecular structure is known, it is then possible to find the gene that codes for that protein. Conversely, one can also use the genes as starting points to isolate and characterize a specific protein. Application of techniques that permit the isolation and sequencing of proteins has elucidated the structure of the acetylcholine receptor mentioned earlier (Figure 3-4). The receptor is composed of four large, related molecules (called sub-units, and one of these is repeated so that five molecules make
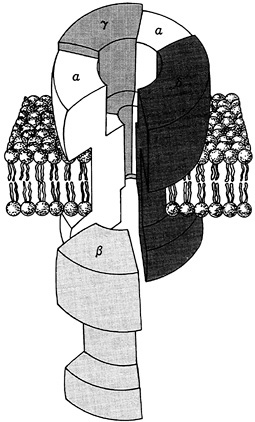
FIGURE 3-4 A three-dimensional model of the “spool-shaped” acetylcholine receptor. Adapted from Kandel and Schwartz, 1985.
up the receptor) that are arranged in a shape that resembles a thick spool. The center of the spool is open and provides a channel through which ions can pass. Each subunit of the receptor is coded by a separate messenger RNA (mRNA), which have now been sequenced, leading to a greater understanding of the structures of the genes themselves. Changes in gene expression, measured by assay of the mRNAs for each subunit, have shown that, in the developing junction between nerve and muscle, release of specific peptides from the growing axons causes activation of the acetylcholine receptor genes and stimulation of receptor synthesis. Such experimental strategies can now be applied to the study of the molecular and genetic effects of neurotransmitters, neuromodulators, and hormones that affect brain cells and, ultimately, behavior.
The application of molecular biological and genetic techniques in
|
BOX 3-2 THE GENETICS OF COLOR VISION On the deepest surface of the retina lie the primary receptors of the human eye, called rods and cones. Rod cells, responsible for black-and-white vision in dim light, are far more numerous than cone cells, which enable us to perceive colors. Although Isaac Newton discovered the color spectrum some 300 years ago, it was not until the late eighteenth century that scientists began to understand the basis of color vision: light-absorbing proteins called pigments that have overlapping but distinct sensitivities to particular wavelengths of the visible spectrum. There are three classes of cone cells: those containing red pigment, those containing green pigment, and those containing blue. Scientists now know that colorblindness, or the inablilty to discriminate red and green, results from alterations in the genes that encode these color pigments in the cone cells. That deficiencies in red-green discrimination are more common in males than in females is explained by the fact that the gene responsible for the variation is located in the X chromosome. Males will be colorblind if the single X chromosome they inherit from their mother carries the trait; females will be affected only if both their X chromosomes are variant. Aberrations in blue sensitivity, which affect males and females equally, are rooted in a gene on another, non-sex chromosome. In hopes of supplementing the findings of these classic genetic studies, Jeremy Nathans and his colleagues at the Johns Hopkins University School of Medicine applied modern scientific techniques to the puzzle of abnormal color vision (Nathans, 1989). Using DNA hybridization, they isolated the genes that code for the color pigments of the cone cells and compared their structures in people with normal and variant color vision. Interestingly, they discovered that although people have only one copy of the gene that codes for red pigment, they have two or three copies of the gene that codes for green pigment. When multiple copies of a gene exist next to each other in this way, the copies have a tendency to recombine in peculiar patterns when cell division occurs, a phenomenon called unequal homologous recombination. Of the 25 subjects studied by Nathans with abnormal red-green discrimination, all but one had DNA that had been shuffled as a result of unequal homologous recombination, confirming the hypothesis that when green pigment genes rearrange themselves “unequally” or irregularly, deficiencies in color vision result. Although much remains to be learned about the roles of cones and pigments in color vision, the work of Nathans and his colleagues yields interesting clues to the genetics behind what we see and what we cannot see. |
neuroscience is very recent and quite promising. Recent landmark successes in neurogenetics have direct application to human health. As scientists identify and characterize ion channels, proteins, receptors, and neurotransmitters that play a role in disease, they gain clues to the location of the genes that control the production of these elements (Box 3-2 ). Already some neurological diseases are yielding to this approach. The genetic locus for Huntington's disease was discovered by an interdisciplinary group of researchers and has led to a test that predicts with remarkable accuracy whether a person at risk for Huntington's will develop the disease. The abnormal (mutated) genes for Duchenne's muscular dystrophy and neurofibromatosis have also been identified. In the case of muscular dystrophy, identification of the defective gene led to isolation of the protein dystrophin, for which the gene codes. Information about dystrophin may well permit scientists to design treatment strategies to replace the defective protein. Identification of the gene for neurofibromatosis is also beginning to lead researchers to important clues about the mechanism of that disease (see Box 2-1 ). Advances such as these are the necessary first steps in disease prevention, but further progress depends on basic scientific investigation into the regulation and expression of neural genes and the functional consequences of that expression in the brain.
References and Bibliography
Dudai, Y. 1985. Genes, enzymes, and learning in Drosophila.Trends in Neuroscience 8 : 18–21.
German, D. C. , K. Manaye , W. K. Smith , D. J. Woodward , and C. B. Saper. 1989. Midbrain dopaminergic cell loss in Parkinson's disease: Computer visualization. Annals of Neurology 26 : 507–514.
Kandel, E. R. , and J. H. Schwartz , eds. 1985. Principles of Neural Science. New York : Elsevier.
Nathans, J. 1989.The genes for color vision. Scientific American 260(2) : 42–49.
Pavlov, I. P. 1927. Conditioned Reflexes: An Investigation of the Physiological Activity of the Cerebral Cortex, G. V. Anrep, trans. London : Oxford University Press.
Shepherd, G. M. 1988. Neurobiology , 2nd ed. New York : Oxford University Press.
Tank, D. W. , M. Sugimori , J. A. Connor , and R. R. Llinas. 1988. Spatially resolved calcium dynamics of mammalian purkinje cells in cerebellar slice. Science 242 : 773–777.
The following sources were also used as background for preparation of this chapter but are not cited specifically in the text.
Darnell, J. , H. Lodish , and D. Baltimore. 1986. Molecular Cell Biology. New York : Scientific American Books, Inc.
Kelner, K. L. , and D. E. Koshland , eds. 1989. Molecules to Models: Advances in Neuroscience. Papers from Science 1986–1989. AAAS Pub. No. 89–17S. Washington, D.C. : American Association for the Advancement of Science.
Kuffler, S. W. , and J. G. Nichols. 1977. From Neuron to Brain: A Cellular Approach to the Function of the Nervous System. Sunderland, Mass. : Sinauer Assoc., Inc.
Ottoson, D. , and W. Rostène , eds. 1989. Visualization of Brain Functions. New York : Stockton Press.