2
The Effects of Falsified and
Substandard Drugs
A safe medicines supply is fundamental for public health. This chapter describes consequences of falsified and substandard drugs. Drawing on a range of examples from both academic literature and other sources, it discusses the relationships between drug failure and health problems at the individual and population levels. The chapter does not attempt to present an exhaustive analysis of every health consequence of substandard or falsified drugs, but it gives an overview of the kinds of health problems these products cause. The second section of this chapter analyzes the economic and social costs of a substandard drug supply, including loss of confidence in the health care system.
A reliable, good-quality medicine supply is essential for health, but it is often missing in countries with weak regulatory systems (Ratanawijitrasin and Wondemagegnehu, 2002). The fallout of falsified and substandard medicines includes poisoning, untreated disease, early death, and treatment failure.
Some of the most compelling stories of pharmaceutical fraud are those of frank poisoning. Between November 2008 and February 2009, 84 Nigerian children died from acute kidney failure brought on by the industrial solvent diethylene glycol in teething syrup (Akuse et al., 2012;
Key Findings and Conclusions
• Falsified and substandard drugs may contain toxic doses of dangerous ingredients and cause mass poisoning.
• Poor-quality medicines compromise the treatment of chronic and infectious diseases, causing disease progression, drug resistance, and death.
• As chronic diseases increase in low- and middle-income countries, so will the need for reliable medicines.
• Substandard and falsified medicines encourage drug resistance, threatening the health of populations today and in the future.
Polgreen, 2009). The contaminated product, My Pikin, was registered with the Nigerian regulatory authority and made in Lagos, the national manufacturing hub (Akuse et al., 2012). Inspectors traced the problem back to deliberate fraud by a chemical dealer in Lagos, eventually leading to 12 prosecutions (Poisoned teething drug arrests, 2009; Polgreen, 2009).
A similar tragedy unfolded on a larger scale the previous year in Panama when a Chinese chemical manufacturer sold diethylene glycol, the active ingredient in antifreeze, as pharmaceutical-grade glycerin to a European company (Bogdanich and Hooker, 2007). The poison caused acute kidney failure in the people who ingested it, often as the solvent in cough syrup (Bogdanich and Hooker, 2007; CDC, 2009). The Panamanian government counted 219 deaths from kidney failure brought on by diethylene glycol poisoning (Núñez, 2011). Given that more than 60,000 bottles of cough syrup and some lotions were contaminated, the Ministry of Health and the World Health Organization (WHO) assume that these confirmed deaths are probably only a fraction of the total mortality (Rentz et al., 2008).
The 2006 diethylene glycol poisoning was an international tragedy, and 18 of the causalities were Chinese (Bogdanich, 2007). In the early 2000s some sources called China “the world’s largest producer of bogus medicines”; Chinese newspaper accounts contain stories of similar mass poisonings (Fackler, 2002). In 2001 reporters described the death of a southwest China mine owner from a poisoned albumin drip (Fackler, 2002). A decade later, the Chinese State Food and Drug Administration (SFDA) found that 13 percent of capsule manufacturers are making drugs containing unsafe levels of chromium, a toxic metal (Rickman, 2012). The SFDA identified 254 separate companies as sources of the chromium-tainted medicines (Rickman, 2012).
Though poisonous drugs are part of the story, the more insidious
problem is medicine that simply does not work. Ineffective medicines often contain benign ingredients, such as chalk, pollen, or flour, instead of medicinal chemicals. More dangerously, some contain substances intended to mask the illness and feign treatment, such as paracetemol added to fake antimalarials to lower fever. Patients taking ineffective drugs die of apparently natural causes, making these products more difficult to identify.
Untreated Disease, Disease Progression, and Death
Medicine is intended to cure patients, or at least to relieve symptoms or slow the progression of a disease. There is also useful information in treatment failure. When prescribing medicines of known content and potency, the clinician may suspect inadequate dosing, drug resistance, or misdiagnosis if the patient does not respond to treatment as expected. These inferences are central to the practice of medicine. The Partnership for Safe Medicines, an American nonprofit, encourages doctors to suspect counterfeit1 drugs in cases of treatment failure (PSM, n.d.), but there is little published evidence to suggest they do so. Advising physicians to consider the possibility of medicine fraud suggests that they have a way to verify it. In parts of the world where such assays are too costly or too technologically complicated to pursue, this information is usually unknowable. Confirmed accounts of drug failure are only a fraction of the larger, mostly invisible, problem.
Research at the medicine store can help illuminate these problems. A random sample of all known medicine shops in three districts of Ghana found the uterotonic drugs oxytocin and ergometrine to be of uniformly poor quality: 89 percent of the samples tested were below British Pharmacopoeia specifications though only 2 percent were expired (Stanton et al., 2012). Unicef, the United Nations Children’s Fund, estimates the maternal mortality ratio in Ghana to be 350 per 100,000 live births (Unicef, 2003), of which hemorrhage, a condition treated with uterotonic drugs, is the most common cause (Asamoah et al., 2011). Even in Ghanaian hospital studies, where one would expect hemorrhage to be an uncommon cause of death, it accounts for an estimated 17 to 22 percent of maternal deaths (Ganyaglo and Hill, 2012; Lee et al., 2012). Increasing access to emergency obstetric care is a key piece of any strategy to reduce maternal mortality (Campbell and Graham, 2006), one that lies on the assumption that lifesaving uterotonic medicines are of reliable potency. Research suggests they are not, even in a middle-income country like Ghana.
The type of study Stanton and colleagues undertook in Ghana is rare.
They were able to draw conclusions about uterotonic drug quality because their data represented a random sample of drugs from an almost exhaustive sampling frame of known pharmacies, chemical shops, and other dispensaries in their study area (Stanton et al., 2012). The identification of falsified and substandard medicines is more often incidental, found in newspaper accounts or uncovered in research that had a different primary aim.
Medications for Chronic Diseases
In 2009 a southwest China newspaper reported on a substandard version of the diabetes drug glibenclamide (also called glyburide) found to contain six times the pharmacopeial standard dose (Xiang, 2009). The medicine was tested only after killing two people and injuring nine (Cheng, 2009; Xiang, 2009). Like oxytocin and ergometrine, glibenclamide is a WHO essential medicine, as is only one other oral diabetes drug, metformin (WHO, 2011c). Metformin too has been the subject of quality concerns. In a convenience sample of pharmacies in Lagos, Nigeria, researchers found that four of eight popular brands of metformin tablets failed one or more pharmacopeial tests of bioequivalence (Olusola et al., 2012). These are troubling findings, given that an estimated 80 percent of the world’s 347 million diabetics live in low- and middle-income countries, where medicines quality is most variable, and diabetes case-fatality exorbitantly high (Unachukwu et al., 2008; WHO, 2011a). Dora Akunyili, the former director of the Nigerian drug regulatory authority, worked against pharmaceutical fraud, a cause she committed to after her diabetic sister’s death from fake insulin (Cheng, 2009; Lemonick, 2005).
Medication for other chronic diseases has been compromised in developing countries. A Rwandan study on drug stability found that 20 percent of medicines in a sample of Kigali and Butare pharmacies were substandard at the time of purchase (Twagirumukiza et al., 2009). Two studies of the antihypertensive amlodipine’s quality in south Nigeria found problems: one study reported 30 percent of samples failed pharmacopeial tests for content uniformity (Eichie et al., 2011) and, in another, all samples failed (Olajide et al., 2010). The management of diseases such as type 2 diabetes and hypertension depend on maintenance medication and monitoring. The sheer amount of products used to treat these conditions raises the patients’ lifetime risk of encountering a bad product, even in countries with stringent regulatory authorities (see Box 2-1). The need for reliable medicines in low- and middle-income countries will become more pronounced as the burden of chronic disease increases in these countries. Already cardiovascular disease is the main killer of adults in low- and middle-income countries,
BOX 2-1
Defective Glucose Test Strips
In October 2006, the FDA recalled two batches of blood glucose test strips used in LifeScan, Inc. OneTouch Ultra brand blood glucose monitors after LifeScan notified the agency that it had received a number of customer complaints. The strips produced inaccurate blood glucose level readings, the results of which are used by diabetics to monitor their condition and determine medication dosing (Bloomberg News, 2007). Diabetics rely on their blood glucose monitors to manage their self-treatment, and incorrect readings can lead patients to administer the wrong dosage of insulin or induce unnecessary panic. Improper insulin dosing is a potentially fatal error. In the LifeScan recall, the FDA identified the problem strips and instructed consumers to inspect the serial numbers on their boxes and replace any fake or unidentifiable strips (FDA, 2006; WHO, 2006).
Investigation traced the strips back to Halson Pharmaceuticals in Shanghai. The manufacturer sold approximately one million substandard test strips to importers, and from there the strips went through the supply chain to reach U.S. and Canadian pharmacies. Over the course of the next year, the test strips made their way to 8 countries and 35 U.S. states. The Chinese authorities eventually arrested and imprisoned Henry Fu, owner of Halson Pharmaceuticals (Bloomberg News, 2007).
The LifeScan recall is a reminder that substandard medical products can find their way into countries with strong regulatory systems. The United States and Canada have systems in place for prompt recalls, allowing them to mitigate the threat the product poses to public health. Within 2 years the fake test strips were fully recalled in the United States, but between 2009 and 2011 customers and investigators still found them in other countries, including Egypt, India, and Pakistan (Loftus, 2011).
As the prevalence of diabetes increases rapidly in the developing world, new, loosely regulated markets attract potential counterfeiters. India is home to more than 50 million diabetics, more than any other country, and the number is expected to increase dramatically over the coming years (World Diabetes Foundation, 2013). In 2007, not long after the first bad test strips appeared in the United States, there were approximately 40.9 million diabetics in India; by the time they reached the country’s growing diabetic population the number had risen by more than 10 million (Mohan et al., 2007). As the chronic disease burden increases in developing countries, falsified and substandard versions of the expensive products used to treat them pose new risks.
the proper medical treatment of which is often neglected (Gaziano, 2007; Yusuf et al., 2011).
Maintenance medication for cardiovascular disease is a vulnerable target for fraud, but the need for these drugs is still unmet in much of the world (Gaziano, 2007). In developing countries, there has been a greater emphasis on controlling infectious disease, especially the infectious diseases of childhood. Considerable research indicates that the anti-infective drugs used to do this are often compromised in poor countries.
Medications for Infectious Diseases
Since 1999 the WHO has known that antibiotics are commonly falsified or made improperly (Wondemagegnehu, 1999). In the 1990s antibiotics accounted for over 45 percent of the 771 cases of falsified and substandard medicines brought to the WHO’s attention (Wondemagegnehu, 1999). A more recent survey in Egypt, Jordan, Lebanon, and Saudi Arabia found more than half of antibiotics sampled to be substandard (Kyriacos et al., 2008). A similar survey in Burma uncovered substandard drugs in 16 percent of amoxicillin and 13 percent of ampicillin samples (Wondemagegnehu, 1999). More recently, a survey of amoxicillin in the capital of Papua New Guinea found all samples outside of pharmacopeial specifications; 14 percent had undetectable levels of active ingredient (Nair et al., 2011). Chapter 3 describes the depth of the problem of fake antibiotics in more detail.
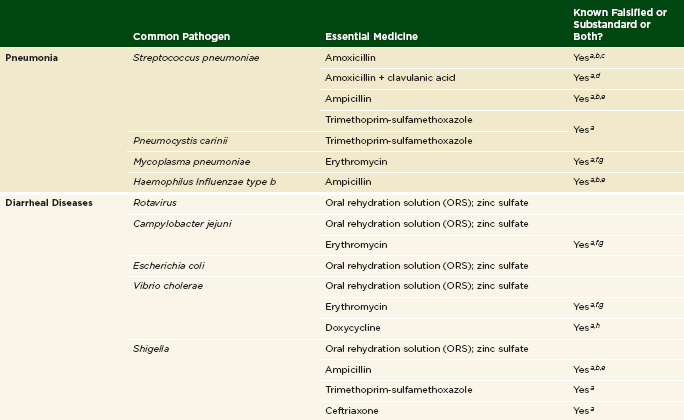
In most low- and middle-income countries β-lactam antibiotics, an inexpensive and widely available class of drugs that includes penicillin and amoxicillin, are the first-line treatment for dozens of bacterial infections, including scarlet fever, pneumonia, and respiratory and urinary tract infections (Byarugaba, 2004). Pneumonia, for example, is the leading cause of death in children under 5 and accounts for 18 percent of all child deaths in the world (Unicef and WHO, 2006; WHO, 2011b). The pathogen Streptococcus pneumoniae causes most of the world’s pneumonia. Alone it accounts for as much as 12 percent of all child deaths worldwide (O’Brien et al., 2009). Unicef’s recommended strategy for preventing pneumonia deaths is recognizing sickness in the child, seeking medical care, and treating with antibiotics (Unicef and WHO, 2006). This will remain the best strategy until the pneumococcal conjugate vaccine becomes more widely available. The treatment of pneumonia and other devastating bacterial infections depends on effective antibiotic supply. No research to date has attempted to quantify the proportion of child deaths attributable to falsified and substandard medicines, but Table 2-1 presents the most common causes of child death and links them to verified reports of substandard medicines.
Vaccines are also important in the control of infectious disease. Chapter 5 describes the medicines supply chain in developing countries; in
general, the vaccine supply chain is simpler, if only because Unicef manages the parts of the chain between the manufacturer and the national port of entry (Kauffmann et al., 2011). Cases of falsified and substandard vaccines are rare, but Box 2-2 describes some.
If antibiotics are some of the oldest and most widely used medicines in the world, antiretrovirals are their opposites: new medicines, prescribed in complicated regimes, to a relatively small segment of the population. An exhaustive WHO survey of antiretroviral drug quality in seven sub-Saharan African countries and a variety of treatment centers found reliable good quality in HIV medications (WHO, 2007). Only 1.8 percent of the drugs tested failed to meet quality specifications, and even those were “[not] serious failures, i.e., no critical deficiencies which would pose a serious risk to patients” (WHO, 2007, p. 19).
BOX 2-2
Deaths from Substandard and Falsified Vaccines
Vaccines are complicated to make, and there are relatively few manufacturers supplying the world market. Vaccines are generally procured in bulk by governments or UN agencies in a supply chain with few intermediaries. Though cases are rare, substandard vaccines can permeate this supply chain. In 1995, during a meningitis epidemic, about 60,000 Nigeriens were injected with water disguised as meningitis vaccine (Cockburn, 2005). The substandard vaccine caused about 2,500 to 3,000 excess deaths (BASCAP, 1996).
More recently, in China, substandard hepatitis B and rabies vaccines killed or sickened about 100 babies (Jia and Carey, 2011). Precise information regarding the event is scarce due to the Chinese government’s denial of a connection between the vaccines and the incidents as well as its control over the Chinese media. According to the Associated Press, the original article in the China Economic Times that exposed the scandal stated that four children who died never had a precise diagnosis, but suffered from fevers and convulsions before their deaths; others who became ill were later diagnosed with encephalitis, among other conditions, and some suffered permanent damage (Associated Press, 2010a). About 200,000 doses of substandard rabies vaccine circulated in Jiangsu province in 2010 before a manufacturer recall (Associated Press, 2010b). These vaccines, like the falsified meningitis vaccine used in Niger, convey no immunity to the patient. When herd immunity is an important result of vaccination, there is no such benefit to society. Assuming the patients survive injection with nonsterile, unidentified liquids, they are still at risk for death from the disease they were not inoculated against.
Some more recent reports suggest that falsified antiretroviral drugs may circulate in African countries. In September 2011, falsified and substandard versions of the triple combination therapy Zidolam-N surfaced in Kenya, many samples molding and crumbling in the packages (Taylor, 2011). A year later, in Tanzania, the regulatory authority uncovered falsified antiretrovirals at a district hospital (Athumani, 2012). These failures put HIV patients at risk for disease progression and favor the selection of resistant virus strains (WHO, 2003). As their viral loads increase, these patients are also more likely to transmit the infection, impeding efforts to control the virus.
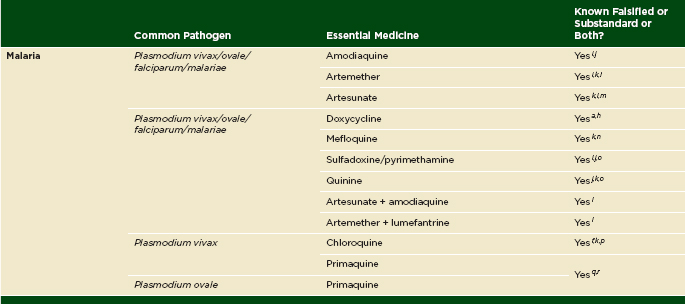
Although data suggesting compromised antiretroviral drug quality are mixed, there is substantial evidence, presented in Chapter 3, that antimalarials are often of poor quality. Substandard and falsified malaria drugs are especially common in malaria-endemic parts of Africa and Asia. In 2003, substandard sulfadoxine-pyrimethamine was used to treat a malaria epidemic in northwest Pakistan refugee camps (Leslie et al., 2009). Researchers concluded that, as the strain of P. falciparum they identified was 90 percent curable when using standard sulfadoxine-pyrimethamine drugs, the substandard medicines, procured from local manufacturers because of drug shortages, were a causal factor in what initially presented as drug-resistant malaria (Leslie et al., 2009). In this example, the effects of the substandard medicine were promptly mitigated. Health workers diagnosed the parasite with microscopy, monitored drug resistance, and checked drug quality using procedures described in the U.S. Pharmacopeia monograph. Good care during initial infection and treatment with an effective second-line drug prevented any deaths, and the onset of cooler weather stopped transmission (Leslie et al., 2009). The prognosis for most people treated with poor-quality antimalarial drugs is worse. Not only will their malaria be untreated, but inadequate treatment favors the selection of resistant parasites, which threaten their entire communities.
Individual patients have much to lose from substandard and falsified medicines. These products also encourage drug resistance and thereby threaten population health today and for future generations. This is a particular concern with substandard products where the dose of active ingredient is low and variable and with falsified products diluted by criminals in an effort to pass screening assays. Drug resistance is common in pathogens with short life cycles: viruses, bacteria, and protozoa. Poor-quality antimicrobial medications, taken frequently and, in poor countries, generally taken without professional supervision, contribute to drug resistance.
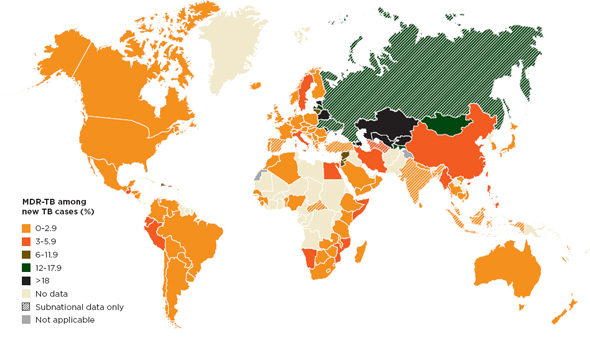
Antibiotics should be used only when indicated, in the appropriate dose, and for the correct length of time. Ensuring the proper treatment with the right combination of drugs is the underlying principle of Directly Observed Treatment–Short Course (DOTS), the internationally accepted method of tuberculosis surveillance and treatment (WHO SEARO, 2006). DOTS also depends on a safe and reliable drug supply. Poor-quality drugs have been cited as a causal factor for the rise of multidrug-resistant tuberculosis (Kelland, 2012). Over time, the bacteria causing tuberculosis have become increasingly drug resistant. Multidrug-resistant tuberculosis precedes extensively drug-resistant tuberculosis, and finally, sometimes, totally drug-resistant tuberculosis (Udwadia, 2012). Extensively drug-resistant strains of tuberculosis account for about 6 percent of incident infections worldwide, but much more in China, India, and the former Soviet Union (Jain and Mondal, 2008). Figure 2-1 shows the increasing incidence of multidrug-resistant tuberculosis around the world.
Drug-resistant bacteria often surface in hospitals, causing infections that are difficult to treat and are an important killer of adults in low-and middle-income countries (Okeke et al., 2005b; WHO, 2012a). It is difficult to estimate the burden of antimicrobial resistance in low- and middle-income countries, in part because of the dearth of data, especially from francophone Africa, the Asian Pacific, and the former Soviet Union (Okeke et al., 2005a). The data that do exist are grim. Multidrug-resistant staphylococcus, an emerging problem in India and sub-Saharan Africa (Parasa et al., 2010; Vincent et al., 2009), accounts for more than half of all nosocomial infections in parts of Latin America (Guzmán-Blanco et al., 2009). (See Figure 2-2.)
In a qualitative study in Orissa, India, doctors, veterinarians, and pharmacists cited poor-quality antibiotics as a cause of drug resistance, but mentioned it only in passing, focusing more on patient and provider behaviors (Sahoo et al., 2010). This is consistent with most public health literature, which gives great deal of attention to the overuse of antibiotics as contributing to the rise of antimicrobial resistance in general (Byarugaba, 2010; Okeke et al., 2005b) and drug-resistant pneumonia in particular (Unicef and WHO, 2006). Comparatively little work, however, discusses the role of drug quality in encouraging bacterial resistance. Antibiotics that contain low doses of active ingredient cause low circulating levels of the drug in the patient. This contributes to treatment failure and selectively favors the growth of drug-resistant organisms (Okeke et al., 2005b). Resistance is most common among the oldest and least expensive families of antibiotics (Okeke et al., 2005b).
According to a recent Tufts University estimate, it costs more than $1.3 billion to bring a new drug to market (Kaitin, 2010). Antibiotics in particular offer pharmaceutical companies a low return on investment; patients take them for only a week or two, in contrast to lifetime regimes of maintenance drugs. There would be even less monetary incentive to develop antibiotic for only the poorest parts of the world. Preserving antibiotics is imperative and depends on maintaining drug quality as much as on encouraging rational use.
Through a conceptually similar mechanism, selectively allowing the growth of drug-resistant parasites by exposing them to subtherapeutic doses of medicines, falsified and substandard drugs favor survival and spread of resistance to antimalarial medicines. Drug-resistant parasites of particular concern are the malaria parasites Plasmodium falciparum and Plasmodium vivax.
Artemisinin combination treatments are effective in treating falciparum malaria (WHO, 2011d). They have been the recommended first-line treatment for falciparum malaria everywhere in the world since 2001 (WWARN, 2012d). In areas where these drugs are available and appropriately used, malaria deaths have dropped dramatically (WHO, 2011d).
Drug resistance could undo the success that artemisinin therapies have won, however (see Box 2-3). A recent review estimates that about 35 percent of the antimalarial medicines in Southeast Asia are substandard, and 36 percent can be classified as falsified (Nayyar et al., 2012). The same researchers found similar patterns in sub-Saharan Africa, where about 35 percent of antimalarials are substandard and 20 percent are falsified (Nayyar et al., 2012). In both regions, underdosing the active ingredients is far more common than overdosing (Nayyar et al., 2012). Already, 8 of the 12 major antimalarial drugs used in the world have been falsified, including products labeled as of mefloquine, but containing sulphadoxine-pyrimethamine and no mefloquine, and a product labeled as artesunate, but containing 6 percent chloroquine and no artesunate (Newton et al., 2006). Poor-quality medicines supply a subtherapeutic dose that selectively encourages the emergence of partially resistant pathogens (Talisuna et al., 2012).
Underdosing with antimalarials causes low concentrations of active drugs in patients and selective pressure to breed resistant parasites (Dondorp et al., 2011; Sengaloundeth et al., 2009; White et al., 2009). In Thailand investigators have observed a progressive lengthening of the time it takes for patients to clear malaria parasites from their bloodstream during treatment, suggesting that the parasites are becoming more resistant to artemisinin (Phyo et al., 2012). Resistance is heritable from one generation
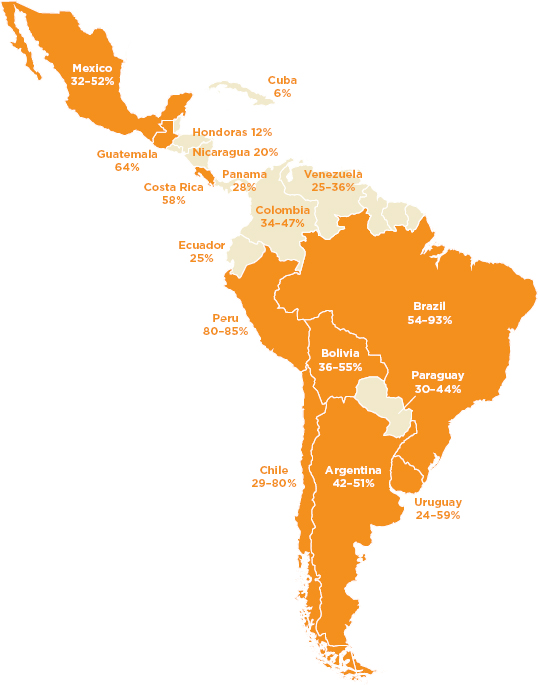
FIGURE 2-2 Percentage of nosocomial Staphylococcus aureus isolates with resistance to meticillin in studies in Latin American countries.
NOTE: Countries in orange are those with >50 percent of nosocomial S. aureus isolates were found to be meticillin-resistant in at least one report.
SOURCE: Guzmán-Blanco et al., 2009. Reprinted with permission from Elsevier.
of parasite to the next; the relatively resistant parasites persist and are transmitted (Anderson et al., 2010). So far, artemisinin resistance has been documented only in Southeast Asia, but its persistence and spread could threaten global malaria control programs. No other antimalarial drugs are available as alternatives. If the current first-line therapy is lost because of resistance, malaria deaths will again increase.
Other Antiparasitic Resistance
Confirming drug resistance in parasites is more complicated than the same assessment in bacteria (Cabaret, 2010). There is good evidence, however, that underdosing with anthelmintic medication has favored survival of resistant worms, and substandard medicines are a noted contributor to anthelmintic resistance in both humans and animals (Geerts and Gryseels, 2001).
Drug resistance threatens efforts to contain other neglected tropical diseases. Visceral leishmaniasis, also called kala azar, is a parasitic disease that affects a half a million people per year, mostly in South Asia, and also in Brazil and Sudan (Sundar, 2001). Untreated kala azar is fatal, but pentavalent antimonial drugs have been a reliable therapy since the 1930s. Pentavalent antimonials are still a first-line treatment today, but drug resistance has diminished the potency of these drugs (Sundar, 2001). A high-osmolarity batch of pentavalent antimonials induced congestive heart failure, killing three kala azar patients and sickening many more at Benares Hindu University hospital in the late 1990s (Sundar et al., 1998). Since then, substandard medicines have been a suspected factor in the increasing resistance of the kala azar parasites to traditional treatment (Sundar, 2001). Newer therapies, such as miltefosine, hold promise for containing the disease, but this promise will not be realized unless the drugs are of reliable quality. As recently as 2012, a convenience sample of miltefosine in Bangladesh found the drugs to be uniformly devoid of any active ingredient (Dorlo et al., 2012).
ECONOMIC AND SOCIAL CONSEQUENCES
Substandard and falsified medicines effect health directly and pose a danger to individual patients and to public health. They also have economic
Key Findings
• Treatment with substandard and falsified drugs wastes time and money, raising drug costs to patients and the health system.
• Drug resistance reduces the effective life of a drug, and society must bear the cost of new drug development.
• A compromised drug supply causes consumers to lose confidence in medicine, health care providers, and national regulatory agencies.
• The sale of falsified medicines funds criminal activities and conveys power to corrupt officials.
BOX 2-3
The WorldWide Antimalarial Resistance Network
Despite the success of malarial control programs starting in 2004, malaria is still a major cause of death, especially in Africa (Murray et al., 2012). WHO estimates that about 655,000 people die from malaria every year, though a recent systematic analysis suggested the true annual mortality is closer to 1.24 million (Murray et al., 2012). Inexpensive oral drugs—chloroquine and sulfadoxine-pyrimethamine—were once common treatment for malaria, but resistance to these treatments is widespread (WHO, 2012c). Since 2001, the WHO has recommended treating malaria with artemisinin combination therapies (WWARN, 2012c). The use of artemisinin combination drugs as first-line therapy is essential to malaria control. As of 2006, however, there is evidence of artemisinin resistance in Southeast Asia (WWARN, 2012c).
The WorldWide Antimalarial Resistance Network (WWARN) is a multidisciplinary, worldwide network of malaria experts run by Oxford University. WWARN is divided into six scientific working groups, including a group that works to encourage research on antimalarial drug quality. To this end, they give step-by-step guidance on field surveys, develop standard reporting and data collection forms, and review chemical assays and packaging analysis protocols (WWARN, 2012a). They also manage online forums to discuss drug quality in English and French (WWARN, 2012a).
WWARN also created a interactive internet database that shows the spread of poor-quality antimalarials over time and space (Tabernero and Newton, 2012). The system maps scientific and lay reports of antimalarial medicine quality. Users can view hotspots as points on a map or in table; they can filter information by medicine, report type, data collection method, medicine source, and date. This tool makes information about antimalarial quality more readily available to regulators and malaria control teams, which in turn improves action against the problem. Future versions of the surveyor will include brand and manufacturer information, as well as graphs of emerging trends and photos of different medicines and packaging (WWARN, 2012b).
and social consequences, including the direct costs of additional treatment and indirect social costs of lost confidence in the health system and the government.
First, the use of falsified and substandard medicines costs the health care system. Providers do not usually suspect that the drugs they prescribe are of poor quality and will respond to a poor therapeutic response by
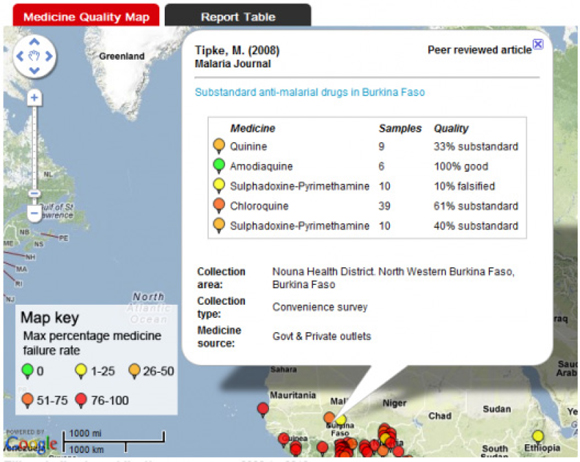
A WWARN online drug quality map.
ordering more tests or by repeating the course of treatment. In poor countries, where medicines rank second only to food as a household expense (Cameron et al., 2008), an increase in the family medicines bill can be a palpable hardship. When government or donors supply medicines, they shoulder the added costs of falsified and substandard drugs.
Chapter 4 describes the pressure on procurement agencies to fill drug orders for the lowest prices, a false frugality that can cause the wasting of an entire medicines budget on drugs with insufficient active ingredients. The costs only grow when expensive drugs are targeted or when they are

Pharmacy in Cambodia.
SOURCE: Hen Sophal in Pharmacide Arts, an exhibit of Southeast Asian artists.
sold in rich countries. It is not yet clear how much patients and insurance companies paid for falsified Avastin during the 2012 crisis, but the Wall Street Journal found that the fake product sold for almost $2,000 per vial (Weaver and Whalen, 2012).
Drug resistance will increase costs to the health system, and not only because of increased clinical attention. Drug resistance reduces the effective life of a drug. Already the cheapest, oldest classes of anti-infective drugs are becoming useless. Society must bear the expense of new drug development, an ever-increasing cost (see Figure 2-3), because resistant pathogens require treatment with more complex drugs. A 2010 estimate put the cost of developing a single drug at $1.3 billion (Kaitin, 2010), and a 2003 study showed that the cost of drug development grew 7.4 percent faster than inflation (DiMasi et al., 2003).
Aside from the direct financial costs of treatment, there are opportunity costs incurred to patients who miss work for additional doctors’ visits or become too sick to work. Chapter 3 will explain that the burden of falsified and substandard medicines is borne mostly by the poor in South and Southeast Asia and sub-Saharan Africa. Transport costs and opportunity costs are a known obstacle to health care for these patients (Whitty et al., 2008). Customers at gray pharmaceutical markets, including flea markets,
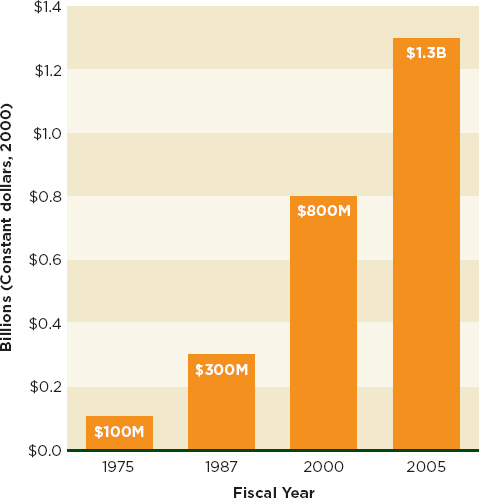
FIGURE 2-3 The increasing costs of drug development.
SOURCE: PhRMA, 2012.
unlicensed medicine shops, and bazaars, are often there because they cannot afford to miss work for a formal consultation (Whitty et al., 2008). For example, participants at the São Paulo site visit for this study explained that although medicines are free through the public health system in Brazil, miners and other daily-wage workers circumvent this system. They continue working and self-treat with medicines of dubious quality from the gray market. In Brazil, as in many parts of the world, falsified and substandard medicines extract the highest costs from those who can afford the least.
Scientists and policy makers in developing countries are aware of the toll falsified and substandard drugs take on their health systems. A 2009 WHO expert working group rated fake medicines as a top priority for research in developing countries (Bates et al., 2009).
Patients may begin to distrust modern pharmacies after experiences with falsified and substandard drugs. In Ugandan villages, the proportion of positive responses to the question “Do you expect that the antimalarial medicines sold by the nearest drug shop are fake?” correlates roughly
with the actual percentage of poor-quality drugs (Björkman-Nyqvist et al., 2012).
As well as having accurate doubts about individual pharmacies, consumers in places where fake drugs circulate have reason to lose faith in the public health system. A recent systematic review suggests that patients across a range of developing countries already have poor perceptions of the health system, especially the technical competence and clinical skills of the staff and the availability of medicines (Berendes et al., 2011). Poor-quality medicines stand to damage the perception of the health system even more. Qualitative research in China suggests that patients view the loosely regulated private health care system poorly, seeing it as rife with “fake doctors” and “fake drugs” (Lim et al., 2004, p. 227).
During a site visit to Brazil, the IOM delegation heard that, although the Brazilian drugs regulatory authority is strong, the public still doubts the quality of many medicines. Participants consistently attributed this poor confidence to unplanned pregnancies following a 1998 lapse in the quality of oral contraceptives (Associated Press, 1998; Goering, 1998). Anvisa, the Brazilian drugs regulatory authority, was created in response to this and other medicine quality problems (Csillag, 1998). Rumors about contraceptive quality linger in Brazil, a kind of urban folklore. They are evidence, however, that fake medicine can do long-term damage to the reputation of the health system.
Social and Developmental Costs
In a larger sense, trade in falsified and substandard medicines undermines not just the health system but all public institutions. Corruption in the health system can cause patients to assume the drug supply is substandard (BBC, 2012). Falsified medicines are often the business of criminal cartels, including the Camorra crime group in Naples, the Russian mafia, and Latin American drug cartels, and terrorist organizations, such as Al-Qaeda and Hezbollah (Findlay, 2011). These organizations run profitable and untaxed businesses. Organized crime flourishes under authoritarian governments and weak rule of law, both common in developing countries (UNODC, 2009). Criminals grow wealthy under either system, eventually wealthy enough that tacit (or active) collaboration becomes necessary for private citizens and politicians to survive (UNODC, 2009). When criminals control politicians, governments cannot be trusted. Donors are then obliged to withhold development aid, as several countries have done in response to corruption in the Zambian health ministry (BBC, 2010; WHO, 2009).
The sale in falsified medicines funds other criminal activities, buys weapons and ammunition, and conveys power and influence to corrupt officials (Findlay, 2011; UNODC, 2009). The United Nations Office on
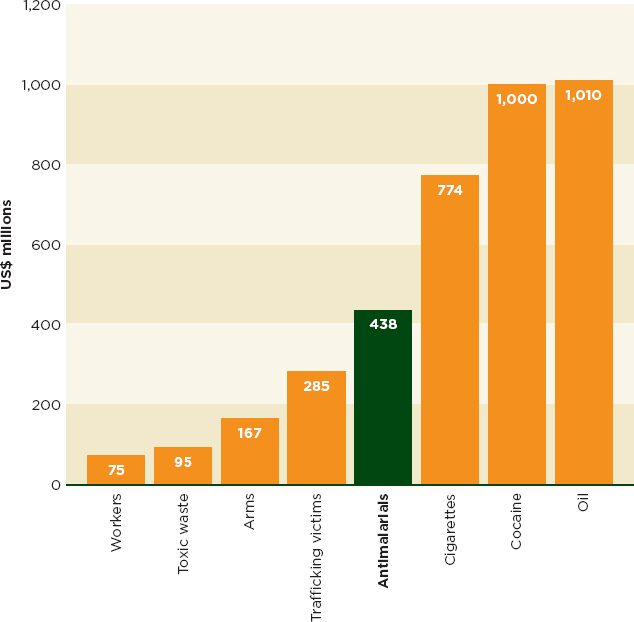
FIGURE 2-4 Comparative values of trafficking flows in West Africa.
SOURCE: UNODC, 2009.
Development and Crime (UNODC) reckons that in West Africa the sale in falsified medicines may be worth as much as the billion-dollar oil and cocaine trafficking industries; their estimate of the worth of trafficked antimalarials alone is more than $400 million (see Figure 2-4).
Chapter 4 describes why medicines fraud is sometimes called the perfect crime. Fake medicines generate income for criminals, and only the weakest evidence, if any, ties them to their crime. Acute cases of medicine poisoning can elicit public outcry, but more often bad drugs go unnoticed, blending in with lawful business. Victims of falsified and substandard drugs usually do not even know they are victims and are therefore deprived of their right to redress. The UNODC described the traffic in fake drugs as both as cause and an effect of political instability, explaining, “Living in a society where
such widespread and serious fraud can occur undermines confidence in government, but the effects are so diffuse and uncertain that they are unlikely to generate an organized political response” (UNODC, 2009, p. 6). In many parts of the world, falsified and substandard medicines further erode the already weak political infrastructure that allows them to circulate, part of a vicious cycle of poverty and crime.
a Delepierre, A., A. Gayot, and A. Carpenter. 2012. Update on counterfeit antibiotics worldwide: Public health risks. Medecine et Maladies Infectieuses 42:247-255.
b Nair, A., S. Strauch, J. Lauwo, R. W. O. Ranke, and J. Dressman. 2011. Are counterfeit or substandard anti-infective products the cause of treatment failure in Papua New Guinea? Journal of Pharmaceutical Sciences 100:5059-5068.
c Taylor, R. B., O. Shakoor, R. H. Behrens et al. 2001. Pharmacopoeial quality of drugs supplied by Nigerian pharmacies. Lancet 357:1933-1936.
d Food and Drug Board of Ghana. 2010. Counterfeit augmentin tablets.
e Sow, P. S., T. S. N. Gueye, E. Sy, L. Toure, C. Ba, and M. Badiane. 2002. Drugs in the parallel market for the treatment of urethral discharge in Dakar: Epidemiologic investigation and physiochemical tests. International Journal of Infectious Disease 6:108-112.
f Bate, R., R. Tren, L. Mooney et al. 2009. Pilot study of essential drug quality in two major cities in India. PLoS ONE, 4(6).
g Hu, C.-Q., W.-B. Zou, W.-S. Hu et al. 2005. Establishment of a Fast Chemical Identification System for screen of counterfeit drugs of macrolide antibiotics. Journal of Pharmaceutical and Biomedical Analysis 40:68-74.
h Prazuck, T., I. Falconi, G. Morineau, V. Bricard-Pacaud, A. Lecomte, and F. Ballereau. 2002. Quality control of antibiotics before the implementation of an STD program in Northern Myanmar. Sexually Transmitted Diseases 29(11):624-627.
i Bate, R., P. Coticelli, R. Tren, and A. Attaran. 2008. Antimalarial drug quality in the most severely malarious parts of Africa—a six country study. PLoS ONE 3(5):e2132.
j Kaur, H., C. Goodman, E. Thompson et al. 2008. A nationwide survey of the quality of antimalarials in retail outlets in Tanzania. PLoS ONE 3(10).
k Lon, C. T., R. Tsuyuoka, S. Phanouvong et al. 2006. Counterfeit and substandard antimalarial drugs in Cambodia. Transactions of the Royal Society of Tropical Medicine and Hygiene 100:1019-1024.
l Newton, P. N., M. D. Green, D. C. Mildenhall et al. 2011. Poor quality vital anti-malarialsin Africa: An urgent neglected public health priority. Malaria Journal 10(352).
m Sengaloundeth, S., M. D. Green, F. M. Fernández et al. 2009. A stratified random survey of the proportion of poor quality oral artesunate sold at medicine outlets in the Lao PDR: Implications for therapeutic failure and drug resistance. Malaria Journal 8(172).
n Dondorp, A. M., P. N. Newton, M. Mayxay et al. 2004. Fake antimalarials in Southeast Asia area major impediment to malaria control: Multinational cross-sectional survey on prevalence of fake antimalarials. Tropical Medicine & International Health 9(12):1241-1246.
o Basco, L. K. 2004. Molecular epidemiology of malaria in Cameroon. XIX. Quality of antimalarial drugs used for self-medication. American Journal of Tropical Medicine and Hygiene 70(3):245-250.
p Syhakhang, L., C. S. Lundborg, B. Lindgren, and G. Tomson. 2004. The quality of drugs in private pharmacies in Lao PDR: A repeat study in 1997 and 1999. Pharmaceutical World Science 26:333-338.
q Kron, M. A. 1996. Substandard primaquine phosphate for US Peace Corps personnel. Lancet 348(9039):1453-1454.
r Petralanda,I. 1995. Quality of antimalarial drugs and resistance to Plasmodium vivax in Amazonian region. Lancet 345(8962):1433.
Akuse, R. M., F. U. Eke, A. D. Ademola, I. B. Fajolu, A. O. Asinobi, H. U. Okafor, S. I. Adeleke, L. I. Audu, A. Otuneye, E. Disu, H. Idis, H. Aikhonbare, A. Yakubu, W. Ogala, O. Ogunrinde, R. Wammanda, A. Orogade, J. Anyiam, E. Eseigbe, L. Umar, H. Musa, R. Onalo, B. West, N. Paul, F. Lesi, T. Ladapo, O. Boyede, R. Okeowo, A. Mustapha, I. Akinola, O. Chima-Oduko, and O. Awobusuyi. 2012. Diagnosing renal failure due to diethylene glycol in children in a resource-constrained setting. Pediatric Nephrology 27(6):1021-1028.
Anderson, T. J. C., S. Nair, S. Nkhoma, J. T. Williams, M. Imwong, P. Yi, D. Socheat, D. Das, K. Chotivanich, N. P. J. Day, N. J. White, and A. M. Dondorp. 2010. High heritability of malaria parasite clearance rate indicates a genetic basis for artemisinin resistance in Western Cambodia. Journal of Infectious Diseases 201(9):1326-1330.
Asamoah, B. O., K. M. Moussa, M. Stafstrom, and G. Musinguzi. 2011. Distribution of causes of maternal mortality among different socio-demographic groups in Ghana: A descriptive study. BMC Public Health 11(159). DOI: 10.1186/1471-2458-11-159.
Associated Press. 1998. Phony birth control pills sold in Brazil. Augusta Chronicle, July 4.
———. 2010a. China probes vaccines after child deaths reported. http://www.boston.com/news/world/asia/articles/2010/03/18/china_probes_vaccines_after_child_deaths_reported (accessed March 5, 2013).
———. 2010b. China rabies vaccine recall prompts changes. http://www.cbc.ca/news/health/story/2010/04/02/china-vaccine.html (accessed March 5, 2013).
Athumani, R. 2012. Tanzania: Factory closed over fake ARVs. Tanzania Daily News, October 11.
BASCAP (Business Action to Stop Counterfeiting and Pharmacy). 1996. Fake vaccine leads to 3,000 deaths in Niger. http://www.icc-ccs.co.uk/bascap/article.php?articleid=363 (accessed February 21, 2013).
Bates, D. W., I. Larizgoitia, N. Prasopa-Plaizier, and A. K. Jha. 2009. Global priorities for patient safety research. British Medical Journal 338. DOI: 10.1136/bmj.b1775.
BBC (British Broadcasting Corporation). 2010. Global Fund freezes Zambia aid over corruption concern. BBC Africa, June 16.
———. 2012. Tanzania investigates fake HIV drugs. BBC News Africa, October 15.
Berendes, S., P. Heywood, S. Oliver, and P. Garner. 2011. Quality of private and public ambulatory health care in low and middle income countries: Systematic review of comparative studies. PLoS Medicine 8(4):e1000433.
Björkman-Nyqvist, M., J. Svensson, and D. Yanagizawa-Drott. 2012. Can good products drive out bad? Evidence from local markets for (fake?) antimalarial medicine in Uganda. Cambridge, MA: Harvard University Center for International Development.
Bloomberg News. 2007. Bogus diabetes test strips traced to Chinese distributor. New York Times, August 17.
Bogdanich, W. 2007. F.D.A. tracked poisoned drugs, but trail went cold in China. New York Times, June 17.
Bogdanich, W., and J. Hooker. 2007. From China to Panama, a trail of poisoned medicine. New York Times, May 6.
Byarugaba, D. K. 2004. Antimicrobial resistance in developing countries and responsible risk factors. International Journal of Antimicrobial Agents 24(2):105-110.
———. 2010. Mechanisms of antimicrobial resistance. In Antimicrobial resistance in developing countries, edited by A. de J. Sosa, D. K. Byarugaba, C. F. Amábile-Cuevas, P.-R. Hsueh, S. Kariuki and I. N. Okeke. New York: Springer. Pp. 15-26.
Cabaret, J. 2010. False resistance to antiparasitic drugs: Causes from shelf availability to patient compliance. Anti-Infective Agents in Medicinal Chemistry 9(3):161-167.
Cameron, A., M. Ewen, D. Ross-Degnan, D. Ball, and R. Laing. 2008. Medicines prices, availability, and affordability in 36 developing and middle-income countries: A secondary analysis. Lancet 373(9659):240-249.
Campbell, O. M. R., and W. J. Graham. 2006. Strategies for reducing maternal mortality: Getting on with what works. Lancet 368(9543):1284-1299.
CDC (Centers for Disease Control and Prevention). 2009. Fatal poisoning among young children from diethylene glycol-contaminated acetaminophen. Morbidity and Mortality Weekly Report 58(48):1345-1347.
Cheng, M. M. 2009. Is the drugstore safe? Counterfeit diabetes products on the shelves. Journal of Diabetes Science and Technology Online 3(6):1516-1520.
Cockburn, R. 2005. Death by dilution. American Prospect, http://prospect.org/article/death-dilution (accessed January 11, 2013).
Csillag, C. 1998. São Paulo: Epidemic of counterfeit drugs causes concern in Brazil. Lancet 352(9127):553.
DiMasi, J., R. Hansen, and H. Grabowski. 2003. The price innovation: New estimates of drug devopment. Journal of Health Economics 22:151-185.
Dondorp, A. M., R. M. Fairhurst, L. Slutsker, J. R. MacArthur, J. G. Breman, P. J. Guerin, T. E. Wellems, P. Ringwald, R. D. Newman, and C. V. Plowe. 2011. The threat of artemisinin-resistant malaria. New England Journal of Medicine 365(12):1073-1075.
Dorlo, T. P. C., T. A. Eggelte, P. J. De Vries, and J. H. Beijnen. 2012. Characterization and identification of suspected counterfeit miltefosine capsules. Analyst 137(5):1265-1274.
Eichie, F. E., M. I. Arhewoh, J. E. Isesele, and K. T. Olatunji. 2011. In vitro assessment of quality control parameters of some commercially available generics of amlodipine besylate in Nigerian drug market. International Journal of Health Research 4(1):57-61.
Fackler, M. 2002. China’s fake drugs kill thousands. San Francisco Examiner, July 31.
FDA (U.S. Food and Drug Administration). 2006. FDA updates its nationwide alert on counterfeit One Touch blood glucose test strips. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2006/ucm108809.htm (accessed June 21, 2012).
Findlay, B. 2011. Counterfeit drugs and national security. Washington, DC: The Stimson Center.
Ganyaglo, G. Y. K., and W. C. Hill. 2012. A 6-year (2004-2009) review of maternal mortality at the eastern regional hospital, Koforidua, Ghana. Seminars in Perinatology 36(1):79-83.
Gaziano, T. A. 2007. Reducing the growing burden of cardiovascular disease in the developing world. Health Affairs 26(1):13-24.
Geerts, S., and B. Gryseels. 2001. Anthelmintic resistance in human helminths: A review. Tropical Medicine and International Health 6(11):915-921.
Goering, L. 1998. Baby boom forces Brazil to confront fake medicines. Chicago Tribune News, July 14.
Guzmán-Blanco, M., C. Mejía, R. Isturiz, C. Alvarez, L. Bavestrello, E. Gotuzzo, J. Labarca, C. M. Luna, E. Rodríguez-Noriega, M. J. C. Salles, J. Zurita, and C. Seas. 2009. Epidemiology of meticillin-resistant Staphylococcus aureus (MRSA) in Latin America. International Journal of Antimicrobial Agents 34(4):304-308.
Jain, A., and R. Mondal. 2008. Extensively drug-resistant tuberculosis: Current challenges and threats. FEMS Immunology & Medical Microbiology 53(2):145-150.
Jia, H., and K. Carey. 2011. Chinese vaccine developers gain WHO imprimatur. Nature Biotechnology 29(6):471-472.
Kaitin, K. 2010. Deconstructing the drug development process: The new face of innovation. Clinical Pharmacology and Therapeutics 87(3):356-361.
Kauffmann, J. R., R. Miller, and J. Cheyne. 2011. Vaccine supply chains need to be better funded and strengthened, or lives will be at risk. Health Affairs 30(6):1113-1121.
Kelland, K. 2012. Drug-resistant “white plague” lurks among rich and poor. Reuters, March 19.
Kyriacos, S., M. Mroueh, R. P. Chahine, and O. Khouzam. 2008. Quality of amoxicillin formulations in some Arab countries. Journal of Clinical Pharmacy and Therapeutics 33(4):375-379.
Lee, Q. Y., A. T. Odoi, H. Opare-Addo, and E. T. Dassah. 2012. Maternal mortality in Ghana: A hospital-based review. Acta Obstetricia et Gynecologica Scandinavica 91(1):87-92.
Lemonick, M. D. 2005. Drug warrior. Time, October 31.
Leslie, T., H. Kaur, N. Mohammed, K. Kolaczinski, R. L. Ord, and M. Rowland. 2009. Epidemic of plasmodium falciparum malaria involving substandard antimalarial drugs, Pakistan, 2003. Emerging Infectious Diseases 15(11):1753-1759.
Lim, M.-K., H. Yang, T. Zhang, W. Feng, and Z. Zhou. 2004. Public perceptions of private health care in socialist China. Health Affairs 23(6):222-234.
Loftus, P. 2011. J&J finds more fake diabetes test strips. Wall Street Journal, April 26.
Mohan, V., R. Sandeep, R. Deepa, B. Shah, and C. Varghese. 2007. Epidemiology of type 2 diabetes. Indian Journal of Medical Research 125:217-230.
Murray, C. J. L., L. C. Rosenfeld, S. S. Lim, K. G. Andrews, K. J. Foreman, D. Haring, N. Fullman, M. Naghavi, R. Lozano, and A. D. Lopez. 2012. Global malaria mortality between 1980 and 2010: A systematic analysis. Lancet 379(9814):413-431.
Nair, A., S. Strauch, J. Lauwo, R. W. O. Jähnke, and J. Dressman. 2011. Are counterfeit or substandard anti-infective products the cause of treatment failure in Papua New Guinea? Journal of Pharmaceutical Sciences 100(11):5059-5068.
Nayyar, G. M. L., J. G. Breman, P. N. Newton, and J. Herrington. 2012. Poor-quality antimalarial drugs in southeast Asia and sub-Saharan Africa. Lancet Infectious Diseases 12(6):488-496.
Newton, P. N., M. D. Green, F. M. Fernández, N. P. J. Day, and N. J. White. 2006. Counterfeit anti-infective drugs. Lancet Infectious Diseases 6(9):602-613.
Núñez, E. 2011. Institute of legal medicine advances in review of pending cases: 219 are killed by syrup. Prensa, June 28.
O’Brien, K. L., L. J. Wolfson, J. P. Watt, E. Henkle, M. Deloria-Knoll, N. McCall, E. Lee, K. Mulholland, O. S. Levine, and T. Cherian. 2009. Burden of disease caused by streptococcus pneumoniae in children younger than 5 years: Global estimates. Lancet 374(9693):893-902.
Okeke, I. N., K. P. Klugman, Z. A. Bhutta, A. G. Duse, P. Jenkins, T. F. O’Brien, A. Pablos-Mendez, and R. Laxminarayan. 2005a. Antimicrobial resistance in developing countries. Part II: Strategies for containment. Lancet Infectious Diseases 5(9):568-580.
Okeke, I. N., R. Laxminarayan, Z. A. Bhutta, A. G. Duse, P. Jenkins, T. F. O’Brien, A. Pablos-Mendez, and K. P. Klugman. 2005b. Antimicrobial resistance in developing countries. Part I: Recent trends and current status. Lancet Infectious Diseases 5(8):481-493.
Olajide, A. S., O. C. Chidinnma, and U. E. Dennis. 2010. Comparative assessment of the quality control measurements of multisource amlodipine tablets marketed in Nigeria. International Journal of Biomedical and Advance Research 1(4). DOI: 10.7439/ijbar.v1i4.10.
Olusola, A. M., A. I. Adekoya, and O. J. Olanrewaju. 2012. Comparative evaluation of physicochemical properties of some commercially available brands of metformin HCL tablets in lagos, Nigeria. Journal of Applied Pharmaceutical Science 2(2):41-44.
Parasa, L. S., L. C. A. Kumar, S. Para, V. S. R. Atluri, K. Santhisree, P. R. Kumar, and C. R. Setty. 2010. Epidemiological survey of mithicillin resistant Staphylococcus aureus in the community and hospital, Gannavaram, Andhra Pradesh, South India. Reviews in Infection 1(2):117-123.
PhRMA (Pharmaceutical Research and Manufacturers of America). 2012. Chart pack: Biopharmaceuticals in perspective. http://www.phrma.org/default/files/159/phrma_chart_pack.pdf (accessed April 10, 2013).
Phyo, A. P., S. Nkhoma, K. Stepniewska, E. A. Ashley, S. Nair, R. McGready, C. ler Moo, S. Al-Saai, A. M. Dondorp, K. M. Lwin, P. Singhasivanon, N. P. J. Day, N. J. White, T. J. C. Anderson, and F. Nosten. 2012. Emergence of artemisinin-resistant malaria on the western border of Thailand: A longitudinal study. Lancet 379(9830):1960-1966.
Poisoned teething drug arrests. 2009. Sky News, February 11.
Polgreen, L. 2009. 84 children are killed by medicine in Nigeria. New York Times, February 6. PSM (Partnership for Safe Medicines). n.d. L.E.A.D.E.R.’S guide for physicians. Vienna, VA: PSM.
Ratanawijitrasin, S., and E. Wondemagegnehu. 2002. Effective drug regulation: A multicountry study. Geneva: WHO.
Rentz, E. D., L. Lewis, O. J. Mujica, D. B. Barr, J. G. Schier, G. Weeraskera, P. Kuklenyik, M. McGeehin, J. Osterloh, J. Wamsley, W. Lum, C. Alleyne, N. Sosa, J. Motta, and C. Rubin. 2008. Outbreak of acute renal failure in Panama in 2006: A case-control study. Bulletin of the World Health Organization 86(10):737-816.
Rickman, J. 2012. China shutters shoddy drugmakes in wake of chromium capsule scare. International Pharmaceutical Regulatory Monitor 40(6).
Sahoo, K. C., A. J. Tamhankar, E. Johansson, and C. S. Lundborg. 2010. Antibiotic use, resistance development and environmental factors: A qualitative study among healthcare professionals in Orissa, India. BMC Public Health 10. DOI: 10.1186/1471-2458-10-629.
Sengaloundeth, S., M. Green, F. Fernandez, O. Manolin, K. Phommavong, V. Insixiengmay, C. Hampton, L. Nyadong, D. Mildenhall, D. Hostetler, L. Khounsaknalath, L. Vongsack, S. Phompida, V. Vanisaveth, L. Syhakhang, and P. Newton. 2009. A stratified random survey of the proportion of poor quality oral artesunate sold at medicine outlets in the Lao PDR—implications for therapeutic failure and drug resistance. Malaria Journal 8(1):172.
Stanton, C., A. Koski, P. Cofie, E. Mirzabagi, B. L. Grady, and S. Brooke. 2012. Uterotonic drug quality: An assessment of the potency of injectable uterotonic drugs purchased by simulated clients in three districts in Ghana. BMJ Open 2(3):1-7.
Sundar, S. 2001. Drug resistance in Indian visceral leishmaniasis. Tropical Medicine and International Health 6(11):849-854.
Sundar, S., P. R. Sinha, N. K. Agrawal, R. Srivastava, P. M. Rainey, J. D. Berman, H. W. Murray, and V. P. Singh. 1998. A cluster of cases of severe cardiotoxicity among kala-azar patients treated with a high-osmolarity lot of sodium antimony gluconate. American Journal of Tropical Medicine and Hygiene 59(1):139-143.
Tabernero, P., and P. N. Newton. 2012. The WWARN antimalarial quality surveyor. Pathogens and Global Health 106(2):77.
Talisuna, A. O., C. Karema, B. Ogutu, E. Juma, J. Logedi, A. Nyandigisi, M. Mulenga, W. F. Mbacham, C. Roper, P. J. Guerin, U. D’Alessandro, and R. W. Snow. 2012. Mitigating the threat of artemisinin resistance in Africa: Improvement of drug-resistance surveillance and response systems. Lancet Infectious Diseases 12(11):888-896.
Taylor, P. 2011. Fake HIV medications uncovered in Kenya. http://www.securingindustry.com/pharmaceuticals/fake-hiv-medications-uncovered-in-kenya/s40/a1095 (accessed January 2, 2013).
Twagirumukiza, M., A. Cosijns, E. Pringels, J. P. Remon, C. Vervaet, and L. Van Bortel. 2009. Influence of tropical climate conditions on the quality of antihypertensive drugs from Rwandan pharmacies. American Journal of Tropical Medicine and Hygiene 81(5): 776-781.
Udwadia, Z. F. 2012. MDR, XDR, TDR tuberculosis: Ominous progression. Thorax 67(4): 286-288.
Unachukwu, C. N., D. I. Uchenna, and E. Young. 2008. Mortality among diabetes in-patients in Port-Harcourt, Nigeria. African Journal of Endocrinology and Metabolism 7(1). http://www.ajol.info/index.php/ajem/article/view/57567/45947 (accessed May 1, 2013).
Unicef (United Nations Children’s Fund). 2003. At a glance: Ghana. http://www.unicef.org/infobycountry/ghana_statistics.html (accessed June 21, 2012).
Unicef and WHO (World Health Organization). 2006. Pneumonia: The forgotten killer of children. Geneva: Unicef and WHO.
UNODC (United Nations Office on Drugs and Crime). 2009. Transnational trafficking and the rule of law in West Africa: A threat assessment. Vienna, Austria: UNODC.
Vincent, J.-L., J. Rello, J. Marshall, E. Silva, A. Anzueto, C. D. Martin, R. Moreno, J. Lipman, C. Gomersall, Y. Sakr, and K. Reinhart. 2009. International study of the prevalence and outcomes of infection in intensive care units. Caring for the Critically Ill Patient 302(21):2323-2329.
Weaver, C., and J. Whalen. 2012. How fake cancer drugs entered U.S. Wall Street Journal, July 20.
White, N., W. Pongtavornpinyo, R. Maude, S. Saralamba, R. Aguas, K. Stepniewska, S. Lee, A. Dondorp, L. White, and N. Day. 2009. Hyperparasitaemia and low dosing are an important source of anti-malarial drug resistance. Malaria Journal 8(1):253.
Whitty, C., C. Chandler, E. Ansah, T. Leslie, and S. Staedke. 2008. Deployment of ACT antimalarials for treatment of malaria: Challenges and opportunities. Malaria Journal 7(Suppl 1):S7.
WHO (World Health Organization). 2003. Alert no. 110: Counterfeit triple antiretroviral combination product (Ginovir 3D) detected in Côte d’Ivoire. Geneva: WHO.
———. 2006. Fact sheet n°275: Counterfeit medicines. http://web.archive.org/web/20061012143704/http://www.who.int/mediacentre/factsheets/fs275/en/index.html (accessed April 19, 2012).
———. 2007. Survey of the quality of antiretroviral medicines circulating in selected African countries. Geneva: WHO.
———. 2009. Fact sheet n°335: Medicines—corruption and pharmaceuticals. http://www.who.int/mediacentre/factsheets/fs335/en/index.html (accessed September 10, 2012).
———. 2011a. Diabetes: Fact sheet n°312. http://www.who.int/mediacentre/factsheets/fs312/en (accessed June 21, 2012).
———. 2011b. Pneumonia: Fact sheet n°331. http://www.who.int/mediacentre/factsheets/fs331/en/index.html (accessed June 21, 2012).
———. 2011c. WHO model list of essential medicines: 17th list. Geneva: WHO.
———. 2011d. World malaria report: 2011. Geneva: WHO.
———. 2012a. Antimicrobial resistance: Fact sheet n°194. http://www.who.int/mediacentre/factsheets/fs194/en (accessed March 5, 2013).
———. 2012b. Global tuberculosis report. Geneva: WHO.
———. 2012c. Malaria: Fact sheet n°94. Geneva: WHO.
WHO SEARO (Regional Office for South-East Asia). 2006. Tuberculosis factsheets: What is DOTS? http://209.61.208/233/en/Section10/Section2097/Section2106_10678.htm (accessed June 21, 2012).
Wondemagegnehu, E. 1999. Counterfeit and substandard drugs in Myanmar and Viet Nam: Report of a study carried out in cooperation with the governments of Myanmar and Viet Nam. Geneva: WHO.
World Diabetes Foundation. 2013. Gestational diabetes among rural and tribal people WDF08-381: Objectives and approaches. http://www.worlddiabetesfoundation.org/projects/karnataka-India-wdf08-381 (accessed April 1, 2013).
WWARN (WorldWide Antimalarial Resistance Network). 2012a. Antimalarial quality. http://www.wwarn.org/about-us/scientific-groups/antimalarial-quality (accessed September 13, 2012).
———. 2012b. Antimalarial quality surveyor. http://www.wwarn.org/resistance/surveyors/antimalarial-quality (accessed September 13, 2012).
———. 2012c. History of resistance. http://www.wwarn.org/resistance/malaria/history (accessed September 13, 2012).
———. 2012d. Malaria drug resistance. http://www.wwarn.org/resistance/malaria (accessed July 30, 2012).
Xiang, Z. 2009. Deadly counterfeit diabetes drugs found outside China’s Xinjiang. China View, February 4.
Yusuf, S., S. Islam, C. K. Chow, S. Rangarajan, G. Dagenais, R. Diaz, R. Gupta, R. Kelishadi, R. Iqbal, A. Avezum, A. Kruger, R. Kutty, F. Lanas, L. Lisheng, L. Wei, P. Lopez-Jaramillo, A. Oguz, O. Rahman, H. Swidan, K. Yusoff, W. Zatonski, A. Rosengren, and K. K. Teo. 2011. Use of secondary prevention drugs for cardiovascular disease in the community in high-income, middle-income, and low-income countries (the PURE study): A prospective epidemiological survey. Lancet 378(9798):1231-1243.
______________________
1 The Partnership for Safe Medicines uses the term counterfeit broadly, the way this report uses falsified. See page 23.