Physical Activity and Physical Education: Relationship to Growth, Development, and Health
Key Messages
• Regular physical activity promotes growth and development and has multiple benefits for physical, mental, and psychosocial health that undoubtedly contribute to learning.
• Specifically, physical activity reduces the risk for heart disease, diabetes mellitus, osteoporosis, high blood pressure, obesity, and metabolic syndrome; improves various other aspects of health and fitness, including aerobic capacity, muscle and bone strength, flexibility, insulin sensitivity, and lipid profiles; and reduces stress, anxiety, and depression.
• Physical activity can improve mental health by decreasing and preventing conditions such as anxiety and depression, as well as improving mood and other aspects of well-being.
• Physical activity programming specifically designed to do so can improve psychosocial outcomes such as self-concept, social behaviors, goal orientation, and most notably self-efficacy. These attributes in turn are important determinants of current and future participation in physical activity.
• Sedentary behaviors such as sitting and television viewing contribute to health risks both because of and independently of their impact on physical activity.
• Health-related behaviors and disease risk factors track from childhood to adulthood, indicating that early and ongoing opportunities for physical activity are needed for maximum health benefit.
• To be effective, physical activity programming must align with the predictable developmental changes in children’s exercise capacity and motor skills, which affect the activities in which they can successfully engage.
• Frequent bouts of physical activity throughout the day yield short-term benefits for mental and cognitive health while also providing opportunities to practice skills and building confidence that promotes ongoing engagement in physical activity.
• Distinct types of physical activity address unique health concerns and contribute in distinct ways to children’s health, suggesting that a varied regimen including aerobic and resistance exercise, structured and unstructured opportunities, and both longer sessions and shorter bouts will likely confer the greatest benefit.
The behaviors and traits of today’s children, along with their genetics, are determinants of their growth and development; their physical, mental, and psychosocial health; and their physical, cognitive, and academic performance. Technological advances of modern society have contributed to a sedentary lifestyle that has changed the phenotype of children from that of 20 years ago. Children today weigh more and have a higher body mass index (BMI) than their peers of just a generation earlier (Ogden et al., 2012). Behaviorally, most children fail to engage in vigorous- or moderate-intensity physical activity for the recommended 60 minutes or more each day, with as many as one-third reporting no physical activity in the preceding 5 days (CDC, 2012). This lack of participation in physical activity has contributed to a greater prevalence of pediatric obesity, a decrease in fitness (e.g., flexibility, muscular strength, cardiorespiratory capacity), and a greater risk for disease (Boreham and Riddoch, 2001; Eisenmann, 2003; Malina, 2007; Steele et al., 2008). (See Box 3-1 for an overview of the relationship between physical activity and physical fitness.)
BOX 3-1
Physical Activity and Physical Fitness
As noted in Chapter 1 (see the box titled “Key Terms Used in This Report” on p. 17), physical activity, a behavior, is defined as bodily movement that increases energy expenditure, whereas fitness is a physiologic trait, commonly defined in terms of cardiorespiratory capacity (e.g., maximal oxygen consumption), although other components of fitness have been defined (IOM, 2012b). Exercise, a subset of physical activity, is “planned, structured and repetitive” (Carpersen et al., 1985, p. 128) and designed to target a particular outcome, for example, cardiorespiratory capacity or another component of fitness. Physical education provides opportunities for developmentally appropriate physical activity, usually structured to promote motor skill development, fitness, and health.
The relationship between physical activity and physical fitness is complex and bidirectional. Numerous studies have shown a significant relationship between physical activity and cardiorespiratory fitness, which may mean that physical activity improves fitness or that physically fit individuals choose to engage in physical activity more than their less fit peers, or both. Experimental studies have shown that exercise training improves fitness (Malina et al., 2004), although the response is variable and clearly influenced by genetics (Bouchard, 2012), and physical activity and fitness are independently related to health and academic performance (see the figure below).
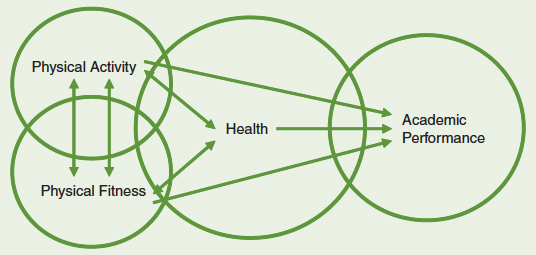
Conceptual framework illustrating relationships among physical activity, physical fitness, health, and academic performance.
While more can always be learned, the evidence for the health benefits of physical activity is irrefutable (HHS, 1996, 2008). Adults engaged in regular physical activity have lower rates of chronic disease (e.g., coronary heart disease, cardiovascular disease, type 2 diabetes, hypertension, osteoporosis, and some cancers) and are less likely to die prematurely (HHS, 1996, 2008; Bauman, 2004). And while the ill effects of chronic disease are manifested mainly in adults, it is increasingly better understood that the development of these conditions starts in childhood and adolescence (Hallal et al., 2006; Cook et al., 2009; Halfon et al., 2012). It appears evident, then, that promotion of health-enhancing behaviors must also start early in life. Indeed, growing evidence points to long-term effects of child and adolescent physical activity on adult morbidity and mortality in addition to its more immediate effects (Hallal et al., 2006) (see Figure 3-1).
Evidence for both direct and indirect health effects of physical activity has been reported (Hallal et al., 2006), and the need for ongoing participation in physical activity to stimulate and maintain the chronic adaptations that underlie those benefits is well documented. To understand the relation-
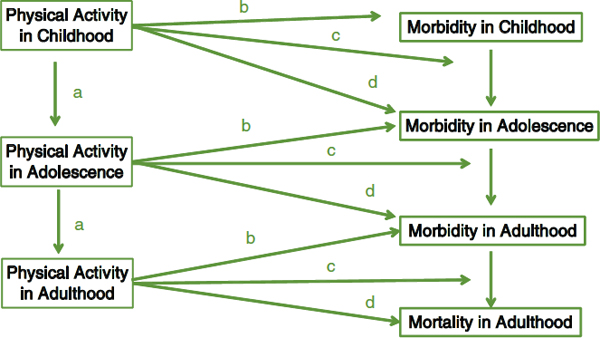
FIGURE 3-1 Conceptual model of how physical activity in childhood and adolescence is beneficial to health. Physical activity has both immediate and long-term health benefits: (a) Physical activity tends to track; early physical activity is associated with physical activity in subsequent life stages. (b) Physical activity reduces morbidity risk in childhood and adolescence. (c) Physical activity may be important in treating and slowing some diseases in children and adolescents. (d) Early physical activity influences future morbidity (e.g., physical activity in childhood and adolescence may reduce fracture risk later in life).
SOURCE: Adapted from Hallal et al., 2006.
ship of physical activity and aerobic fitness to health during childhood, it is important first to recognize the developmental changes that occur throughout maturation. During the early stages of adolescence, for example, participation in physical activity and corresponding physical fitness begin to decline (Duncan et al., 2007). Such differences across stages of development highlight the importance of examining the effects of growth and maturation on physical and cognitive health. Accordingly, this chapter reviews how physical activity may influence developmental processes and other aspects of somatic growth and maturation. A complete review of the effects of physical activity on all tissues and systems is beyond the scope of this report. Rather, the focus is on components of body composition and systems that underlie engagement in physical activity, physical fitness, and chronic disease risk and that in turn influence other aspects of health and academic performance (discussed in Chapter 4). Addressed in turn is the relationship between physical activity and physical, psychosocial, and mental health. Structural and functional brain maturation and how physical activity may influence those developmental processes and cognitive health are also reviewed in Chapter 4.
This section reviews what is known about the relationship between physical activity and (1) somatic growth, development, and function and (2) health- and performance-related fitness.
Somatic Growth, Development, and Function
Growth occurs through a complex, organized process characterized by predictable developmental stages and events. Although all individuals follow the same general course, growth and maturation rates vary widely among individuals. Just as it is unrealistic to expect all children at the same age to achieve the same academic level, it is unrealistic to expect children at the same age to have the same physical development, motor skills, and physical capacity. Regular physical activity does not alter the process of growth and development. Rather, developmental stage is a significant determinant of motor skills, physical capacity, and the adaptation to activity that is reasonable to expect (see Box 3-2).
Developmental Stages
Postnatal growth is commonly divided into three or four age periods. Infancy spans the first year of life. Childhood extends from the end of infancy to the start of adolescence and is often divided into early child-
BOX 3-2
Growth, Development, and Maturation
Growth
Growth is the normal process of increase in size as a result of accretion of tissues characteristic of the organism; growth is the dominant biological activity for most of the first two decades of life. Changes in size are the outcome of an increase in cell number (hyperplasia), an increase in cell size (hypertrophy), and an increase in intercellular substances (accretion).
Development
Encompassing growth and maturation, development denotes a broader concept; when used in a biological context, development refers to differentiation and specialization of stem cells into different cell types, tissues, organs, and functional units. Development continues as different systems become functionally refined. Development also refers to the acquisition and refinement of behavior relating to competence in a variety of interrelated domains, such as motor competence and social, emotional, and cognitive competence.
Maturation
Maturation is the timing and tempo of progress toward the mature state and varies considerably among individuals; variation in progress toward the mature state over time implies variation in the rate of change. Two children may be the same size but at different points on the path to adult size or maturity.
hood, which includes the preschool years, and middle childhood, which includes the elementary school years, into the 5th or 6th grade. Adolescence is more difficult to define because of variation in its onset and termination, although it is commonly defined as between 10 and 18 years of age (WHO, 1986). The rapid growth and development of infancy continue during early childhood, although at a decelerating rate, whereas middle childhood is a period of slower, steady growth and maturation. Differences between boys and girls are relatively small until adolescence, which is marked by accelerated growth and attainment of sexual maturity (Tanner, 1962).
Across developmental stages, neurological development and control of movement advance in cephalocaudal and proximodistal directions; that is, they advance “head to toe” (cephalocaudal) and “midline to periphery” (proximodistal), while predictable changes in body proportions also occur. For example, the head accounts for 25 percent of recumbent length in an infant and only 15 percent of adult height, while the legs account for 38 percent of recumbent length at birth and 50 percent of adult height. These changes in body proportions occur because body parts grow at different rates. From birth to adulthood, as the head doubles in size, the trunk triples in length, and arm and leg lengths quadruple.
Coincident with these changes in body proportions, and in part because of them, the capacity to perform various motor tasks develops in a predictable fashion. For example, running speed increases are consistent with the increase in leg length. Neurological development also determines skill progression. Young children, for example, when thrown a ball, catch it within the midline of the body and do not attempt to catch it outside the midline or to either side of the body. As proximodistal development proceeds, children are better able to perform tasks outside their midline, and by adolescence they are able to maneuver their bodies in a coordinated way to catch objects outside the midline with little effort.
Physically active and inactive children progress through identical stages. Providing opportunities for young children to be physically active is important not to affect the stages but to ensure adequate opportunity for skill development. Sound physical education curricula are based on an understanding of growth patterns and developmental stages and are critical to provide appropriate movement experiences that promote motor skill development (Clark, 2005). The mastery of fundamental motor skills is strongly related to physical activity in children and adolescents (Lubans et al., 2010) and in turn may contribute to physical, social, and cognitive development. Mastering fundamental motor skills also is critical to fostering physical activity because these skills serve as the foundation for more advanced and sport-specific movement (Clark and Metcalfe, 2002; Hands et al., 2009; Robinson and Goodway, 2009; Lubans et al., 2010). Physical activity programs, such as physical education, should be based on developmentally appropriate motor activities to foster self-efficacy and enjoyment and encourage ongoing participation in physical activity.
Biological Maturation
Maturation is the process of attaining the fully adult state. In growth studies, maturity is typically assessed as skeletal, somatic, or sexual. The same hormones regulate skeletal, somatic, and sexual maturation during adolescence, so it is reasonable to expect the effect of physical activity on
these indicators of maturity to be similar. Skeletal maturity is typically assessed from radiographs of the bones in the hand and wrist; it is not influenced by habitual physical activity. Similarly, age at peak height velocity (the most rapid change in height), an indicator of somatic maturity, is not affected by physical activity, nor is the magnitude of peak height velocity, which is well within the usual range in both active and inactive youth. Discussions of the effects of physical activity on sexual maturation more often focus on females than males and, in particular, on age at menarche (first menses). While some data suggest an association between later menarche and habitual physical activity (Merzenich et al., 1993), most of these data come from retrospective studies of athletes (Clapp and Little, 1995). Whether regular sports training at young ages before menarche “delays” menarche (later average age of menarche) remains unclear. While menarche occurs later in females who participate in some sports, the available data do not support a causal relationship between habitual physical activity and later menarche.
Puberty is the developmental period that represents the beginning of sexual maturation. It is marked by the appearance of secondary sex characteristics and their underlying hormonal changes, with accompanying sex differences in linear growth and body mass and composition. The timing of puberty varies, beginning as early as age 8 in girls and age 9 in boys in the United States and as late as ages 13-15 (NRC/IOM, 1999). Recent research suggests that the onset of puberty is occurring earlier in girls today compared with the previous generation, and there is speculation that increased adiposity may be a cause (Bau et al., 2009; Rosenfield et al., 2009). Conversely, some data suggest that excess adiposity in boys contributes to delayed sexual maturation (Lee et al., 2010). Pubescence, the earliest period of adolescence, generally occurs about 2 years in advance of sexual maturity. Typically, individuals are in the secondary school years during this period, which is a time of decline in habitual physical activity, especially in girls. Physical activity trends are influenced by the development of secondary sex characteristics and other physical changes that occur during the adolescent growth spurt, as well as by societal and cultural factors. Research suggests that physical inactivity during adolescence carries over into adulthood (Malina, 2001a,b; CDC, 2006).
It is critical that adolescents be offered appropriate physical activity programs that take into account the physical and sociocultural changes they are experiencing so they will be inspired to engage in physical activity for a lifetime. As discussed below, adequate physical activity during puberty may be especially important for optimal bone development and prevention of excess adiposity, as puberty is a critical developmental period for both the skeleton and the adipose organ.
Adolescence is the transitional period between childhood and adulthood. The adolescent growth spurt, roughly 3 years of rapid growth, occurs early in this period. An accelerated increase in stature is a hallmark, with about 20 percent of adult stature being attained during this period. Along with the rapid increase in height, other changes in body proportions occur that have important implications for sports and other types of activities offered in physical education and physical activity programs. As boys and girls advance through puberty, for example, biacromial breadth (shoulder width) increases more in boys than in girls, while increases in bicristal breadth (hip width) are quite similar. Consequently, hip-shoulder width ratio, which is similar in boys and girls during childhood, decreases in adolescent boys while remaining relatively constant in girls (Malina et al., 2004). Ratios among leg length, trunk length, and stature also change during this period. Prior to adolescence, boys have longer trunks and shorter legs than girls (Haubenstricker and Sapp, 1980). In contrast, adolescent and adult females have shorter legs for the same height than males of equal stature. Body proportions, particularly skeletal dimensions, are unlikely to be influenced by physical activity; rather, body proportions influence performance success, fitness evaluation, and the types of activities in which a person may wish to engage. For example, there is evidence that leg length influences upright balance and speed (Haubenstricker and Sapp, 1980). Individuals who have shorter legs and broader pelvises are better at balancing tasks than those with longer legs and narrower pelvises, and longer legs are associated with faster running times (Dintiman et al., 1997). Also, longer arms and wider shoulders are advantageous in throwing tasks (Haubenstricker and Sapp, 1980), as well as in other activities in which the arms are used as levers. According to Haubenstricker and Sapp (1980), approximately 25 percent of engagement in movement-related activities can be attributed to body size and structure.
Motor Development
Motor development depends on the interaction of experience (e.g., practice, instruction, appropriate equipment) with an individual’s physical, cognitive, and psychosocial status and proceeds in a predictable fashion across developmental periods. Clark and Metcalfe (2002) provide an eloquent metaphor—“the mountain of motor development”—to aid in understanding the global changes seen in movement across the life span. Early movements, critical for an infant’s survival, are reflexive and dominated by biology, although environment contributes and helps shape reflexes. This initial reflexive period is followed quickly by the preadapted period, which begins when an infant’s movement behaviors are no longer reflexive and ends when the infant begins to apply basic movement skills (e.g.,
crawling, rolling, standing, and walking) that generally are accomplished before 12 months of age. The period of fundamental motor patterns occurs approximately between the ages of 1 and 7 years, when children begin to acquire basic fundamental movement skills (e.g., running, hopping, skipping, jumping, leaping, sliding, galloping, throwing, catching, kicking, dribbling, and striking). Practice and instruction are key to learning these skills, and a great deal of time in elementary school physical education is devoted to exploration of movement. Around age 7, during the so-called context-specific period of motor development, children begin to refine basic motor skills and combine them into more specific movement patterns, ultimately reaching what has been called skillfulness. Compensation, the final period of motor development, occurs at varying points across the life span when, as a result of aging, disease, injury, or other changes, it becomes necessary to modify movement.
While all children need not be “expert” in all movement skills, those who do not acquire the fundamental motor skills will likely experience difficulty in transitioning their movement repertoire into specific contexts and engagement in physical activity (Fisher et al., 2005; Barnett et al., 2009; Cliff et al., 2009; Robinson et al., 2012). A full movement repertoire is needed to engage in physical activities within and outside of the school setting. Thus, beyond contributing to levels of physical activity, physical education programs should aim to teach basic fundamental motor skills and their application to games, sports, and other physical activities, especially during the elementary years (i.e., the fundamental motor patterns and context-specific periods). At the same time, it is important to be mindful of the wide interindividual variation in the rate at which children develop motor skills, which is determined by their biological makeup, their rate of physical maturation, the extent and quality of their movement experiences, and their family and community environment.
An increasing amount of evidence suggests that people who feel competent in performing physical skills remain more active throughout their lives (Lubans et al., 2010). Conversely, those who are less skilled may be hesitant to display what they perceive as a shortcoming and so may opt out of activities requiring higher levels of motor competence (Stodden et al., 2008). Children who are less physically skillful tend to be less active than their skillful counterparts (Wrotniak et al., 2006; Williams et al., 2008; Robinson et al., 2012) and thus have a greater risk of overweight and obesity (Graf et al., 2004). Fundamental skills are the building blocks of more complex actions that are completed in sports, physical activities, and exercise settings. For example, throwing is a fundamental skill that is incorporated into the context-specific throw used in activities such as handball, softball, and water polo. Fundamental skills are of primary interest to both physical education teachers and coaches, and physical
education classes should be designed to challenge learners to develop their motor skills.
In 1998 the Centers for Disease Control and Prevention’s (CDC’s) Division of Nutrition and Physical Activity organized a workshop to determine future directions for research on physical activity. The workshop convened 21 experts from a wide range of academic disciplines. One recommendation resulting from the proceedings was for future research to describe the temporal relationship between motor development and physical activity (Fulton et al., 2001), signifying the importance of better understanding of the nature of the relationship between motor competence and physical activity. The assumption of this relationship is implied in multiple models of motor development (Seefeldt, 1980; Clark and Metcalfe, 2002; Stodden et al., 2008), which emphasize the importance of motor competence as a prerequisite for engagement in physical activity throughout the life span.
Two models that are commonly used to examine this relationship are Seefeldt’s (1980) hierarchical order of motor skills development and the dynamic association model of Stodden and colleagues (2008). Seefeldt proposed a hierarchical order of motor skills development that includes four levels: reflexes, fundamental motor skills, transitional motor skills (i.e., fundamental motor skills that are performed in various combinations and with variations and that are required to participate in entry-level organized sports, such as throwing for distance, throwing for accuracy, and/or catching a ball while in motion), and specific sports skills and dances. With improved transitional motor skills, children are able to master complex motor skills (e.g., those required for playing more complex sports such as football or basketball). At the end of this developmental period, children’s vision is fully mature. The progression through each level occurs through developmental stages as a combined result of growth, maturation, and experience. Seefeldt hypothesized the existence of a “proficiency barrier” between the fundamental and transitional levels of motor skills development. If children are able to achieve a level of competence above the proficiency barrier, they are more likely to continue to engage in physical activity throughout the life span that requires the use of fundamental motor skills. Conversely, less skilled children who do not exceed the proficiency barrier will be less likely to continue to engage in physical activity. Thus, it is assumed that “a confident and competent mover will be an active mover” (Clark, 2005, p. 44). For example, to engage successfully in a game of handball, baseball, cricket, or basketball at any age, it is important to reach a minimum level of competence in running, throwing, catching, and striking. The assumption of the existence of a relationship between motor competence and physical activity is at the “heart of our physical education programs” (Clark, 2005, p. 44). A thorough understanding of how this
relationship changes across developmental stages is crucial for curriculum development and delivery and teaching practices.
Lubans and colleagues (2010) recently examined the relationship between motor competence and health outcomes. They reviewed 21 studies identifying relationships between fundamental motor skills and self-worth, perceived physical competence, muscular and cardiorespiratory fitness, weight status, flexibility, physical activity, and sedentary behavior. Overall, the studies found a positive association between fundamental motor skills and physical activity in children and adolescents, as well as a positive relationship between fundamental motor skills and cardiorespiratory fitness. Other research findings support the hypothesis that the most physically active preschool-age (Fisher et al., 2005; Williams et al., 2008; Robinson et al., 2012), elementary school–age (Bouffard et al., 1996; Graf et al., 2004; Wrotniak et al., 2006; Hume et al., 2008; Lopes et al., 2011), and adolescent (Okely et al., 2001) youth are also the most skilled.
An advantage of the “proficiency barrier” hypothesis proposed by Seefeldt (1980) is its recognition that the relationship between motor competence and physical activity may not be linear. Rather, the hypothesis suggests that physical activity is influenced when a certain level of motor competence is not achieved and acknowledges that below the proficiency barrier, there is bound to be substantial variation in children’s motor competence and participation in physical activity. The proficiency barrier is located between the fundamental and transitional motor skills periods. The transition between these two levels of motor competence is expected to occur between the early and middle childhood years. Stodden and colleagues (2008) suggest that the relationship between motor competence and physical activity is dynamic and changes across time. In their model the “development of motor skill competence is a primary underlying mechanism that promotes engagement in physical activity” (p. 290).
The relationship between skills and physical activity is considered reciprocal. It is expected that as motor skills competence increases, physical activity participation also increases and that the increased participation feeds back into motor skills competence. The reciprocal relationship between motor skills competence and physical activity is weak during the early childhood years (ages 2-8) because of a variety of factors, including environmental conditions, parental influences, and previous experience in physical education programs (Stodden et al., 2008). Also, children at this age are less able to distinguish accurately between perceived physical competence and actual motor skills competence (Harter and Pike, 1984; Goodway and Rudisill, 1997; Robinson and Goodway, 2009; Robinson, 2011), and thus motor skills are not expected to strongly influence physical activ-
ity. The literature supports this hypothesis, as indicated by low to moderate correlations between motor skills competence and physical activity
in preschool (Sääkslahti et al., 1999; Williams et al., 2008; Cliff et al., 2009; Robinson and Goodway, 2009; Robinson, 2011) and early elementary school–age (Raudsepp and Päll, 2006; Hume et al., 2008; Morgan et al., 2008; Houwen et al., 2009; Ziviani et al., 2009; Lopes et al., 2011) children.
In older children, perceived competence is more closely related to actual motor skills competence. Older, low-skilled children are aware of their skills level and are more likely to perceive physical activity as difficult and challenging. Older children who are not equipped with the necessary skills to engage in physical activity that requires high levels of motor skills competence may not want to display their low competence publicly. As children transition into adolescence and early adulthood, the relationship between motor skills competence and physical activity may strengthen (Stodden et al., 2008). Investigators report moderate correlations between motor skills competence and physical activity in middle school–age children (Reed et al., 2004; Jaakkola et al., 2009). Okely and colleagues (2001) found that motor skills competence was significantly associated with participation in organized physical activity (i.e., regular and structured experiences related to physical activity) as measured by self-reports. A strength of the model of Stodden and colleagues (2008) is the inclusion of factors related to psychosocial health and development that may influence the relationship between motor skills competence and physical activity, contributing to the development and maintenance of obesity. Other studies have found that perceived competence plays a role in engagement in physical activity (Ferrer-Caja and Weiss, 2000; Sollerhed et al., 2008).
Motor skills competence is an important factor; however, it is only one of many factors that contribute to physical activity. For instance, three studies have reported negative correlations between girls’ motor competence and physical activity (Reed et al., 2004; Cliff et al., 2009; Ziviani et al., 2009), suggesting that sex may be another determining factor. A possible explanation for these findings is that since girls tend to be less active than boys, it may be more difficult to detect differences in physical activity levels between high- and low-skilled girls. It is also possible that out-of-school opportunities for physical activity are more likely to meet the interests of boys, which may at least partially explain sex differences in physical activity levels (Le Masurier et al., 2005). Previous research suggests that in general boys are more motor competent than girls (Graf et al., 2004; Barnett et al., 2009; Lopes et al., 2011) and that this trend, which is less apparent in early childhood, increases through adolescence (Thomas and French, 1985; Thomas and Thomas, 1988; Thomas, 1994), although one study reports that girls are more motor competent than boys (Cliff et al., 2009).
One component of motor competence is the performance of gross motor skills, which are typically classified into object control and
locomotor skills. Consistent evidence suggests that boys are more competent in object control skills, while girls are more competent in locomotor skills (McKenzie et al., 2004; Morgan et al., 2008; Barnett et al., 2009). In light of these sex differences, it is important to examine the relationships of object control and locomotor skills with physical activity separately for boys and girls. For boys, object control skills are more related to physical activity than are locomotor skills (Hume et al., 2008; Morgan et al., 2008; Williams et al., 2008; Cliff et al., 2009), whereas evidence suggests that the reverse is true for girls (McKenzie et al., 2002; Hume et al., 2008; Cliff et al., 2009; Jaakkola et al., 2009). Three studies report a significant relationship between balance and physical activity for girls but not boys (Reed et al., 2004; Ziviani et al., 2009). Cliff and colleagues (2009) suggest that object control and locomotor skills may be more related to boys’ and girls’ physical activity, respectively, because of the activity type in which each sex typically engages.
The relationship between motor competence and physical activity clearly is complex. It is quite likely that the relationship is dynamic and that motor competence increases the likelihood of participating in physical activity while at the same time engaging in physical activity provides opportunities to develop motor competence (Stodden et al., 2008). Despite some uncertainty, the literature does reinforce the important role of physical education in providing developmentally appropriate movement opportunities in the school environment. These opportunities are the only means of engaging a large population of children and youth and providing them with the tools and opportunities that foster health, development, and future physical activity.
Stature
Regular physical activity has no established effect on linear growth rate or ultimate height (Malina, 1994). Although some studies suggest small differences, factors other than physical activity, especially maturity, often are not well controlled. It is important to note that regular physical activity does not have a negative effect on stature, as has sometimes been suggested. Differences in height among children and adolescents participating in various sports are more likely due to the requirements of the sport, selection criteria, and interindividual variation in biological maturity than the effects of participation per se (Malina et al., 2004).
Body Weight
Although physical activity is inversely related to weight, correlations are generally low (~r –0.15), and differences in body weight between active and inactive boys and girls tend to be small (Mirwald and Bailey, 1986;
Saris et al., 1986; Beunen et al., 1992; Lohman et al., 2006;), except in very obese children and adolescents. Similarly, physique, as represented in somatotypes, does not appear to be significantly affected by physical activity during growth (Malina et al., 2004). In contrast, components of weight can be influenced by regular physical activity, especially when the mode and intensity of the activity are tailored to the desired outcome. Much of the available data in children and adolescents is based on BMI, a surrogate for composition, and indirect methods based on the two-compartment model of body composition in which body weight is divided into its fat-free and fat components (Going et al., 2012). While studies generally support that physical activity is associated with greater fat-free mass and lower body fat, distinguishing the effects of physical activity on fat-free mass from expected changes associated with growth and maturation is difficult, especially during adolescence, when both sexes have significant growth in fat-free mass. The application of methods based on the two-compartment model is fraught with errors, especially when the goal is to detect changes in fat-free mass, and no information is available from these methods regarding changes in the major tissue components of fat-free mass—muscle and skeletal tissue.
Muscle Skeletal muscle is the largest tissue mass in the body. It is the main energy-consuming tissue and provides the propulsive force for movement. Muscle represents about 23-25 percent of body weight at birth and about 40 percent in adults, although there is a wide range of “normal” (Malina, 1986, 1996). Postnatal muscle growth is explained largely by increases in cell size (hypertrophy) driving an increase in overall muscle mass. The increase in muscle mass with age is fairly linear from young childhood until puberty, with boys having a small but consistent advantage (Malina, 1969, 1986). The sex difference becomes magnified during and after puberty, driven primarily by gender-related differences in sex steroids. Muscle, as a percentage of body mass, increases from about 42 percent to 54 percent in boys between ages 5 and 11, whereas in girls it increases from about 40 percent to 45 percent between ages 5 and 13 and thereafter declines (Malina et al., 2004). It should be noted that absolute mass does not decline; rather, the relative decline reflects the increase in the percentage of weight that is fat in girls. At least part of the sex difference is due to differences in muscle development for different body regions (Tanner et al., 1981). The growth rate of arm muscle tissue during adolescence in males is approximately twice that in females, whereas the sex difference in the growth of muscle tissue in the leg is much smaller. The sex difference that develops during puberty persists into adulthood and is more apparent for the musculature of the upper extremities.
Sex-related differences in muscular development contribute to differences in physical performance. Muscle strength develops in proportion to the cross-sectional area of muscle, and growth curves for strength are essentially the same as those for muscle (Malina and Roche, 1983). Thus the sex difference in muscle strength is explained largely by differences in skeletal muscle mass rather than muscle quality or composition. Aerobic (endurance) exercise has little effect on enhancing muscle mass but does result in significant improvement in oxygen extraction and aerobic metabolism (Fournier et al., 1982). In contrast, numerous studies have shown that high-intensity resistance exercise induces muscle hypertrophy, with associated increases in muscle strength. In children and adolescents, strength training can increase muscle strength, power, and endurance. Multiple types of resistance training modalities have proven effective and safe (Bernhardt et al., 2001), and resistance exercise is now recommended for enhancing physical health and function (Behringer et al., 2010). These adaptations are due to muscle fiber hypertrophy and neural adaptations, with muscle hypertrophy playing a more important role in adolescents, especially in males. Prior to puberty, before the increase in anabolic sex steroid concentrations, neural adaptations explain much of the improvement in muscle function with exercise in both boys and girls.
Skeleton The skeleton is the permanent supportive framework of the body. It provides protection for vital organs and is the main mineral reservoir. Bone tissue constitutes most of the skeleton, accounting for 14-17 percent of body weight across the life span (Trotter and Peterson, 1970; Trotter and Hixon, 1974). Skeletal strength, which dictates fracture risk, is determined by both the material and structural properties of bone, both of which are dependent on mineral accrual. The relative mineral content of bone does not differ much among infants, children, adolescents, and adults, making up 63-65 percent of the dry, fat-free weight of the skeleton (Malina, 1996). As a fraction of weight, bone mineral (the ash weight of bone) represents about 2 percent of body weight in infants and about 4-5 percent of body weight in adults (Malina, 1996). Bone mineral content increases fairly linearly with age, with no sex difference during childhood. Girls have, on average, a slightly greater bone mineral content than boys in early adolescence, reflecting their earlier adolescent growth spurt. Boys have their growth spurt later than girls, and their bone mineral content continues to increase through late adolescence, ending with greater skeletal dimensions and bone mineral content (Mølgaard et al., 1997). The increase in total body bone mineral is explained by both increases in skeletal length and width and a small increase in bone mineral density (Malina et al., 2004).
Many studies have shown a positive effect of physical activity on intermediate markers of bone health, such as bone mineral content and density.
Active children and adolescents have greater bone mineral content and density than their less active peers, even after controlling for differences in height and muscle mass (Wang et al., 2004; Hind and Burrows, 2007; Tobias et al., 2007). Exercise interventions support the findings from observational studies showing beneficial effects on bone mineral content and density in exercise participants versus controls (Petit et al., 2002; Specker and Binkley, 2003), although the benefit is less than is suggested by cross-sectional studies comparing active versus inactive individuals (Bloomfield et al., 2004). The relationship between greater bone mineral density and bone strength is unclear, as bone strength cannot be measured directly in humans. Thus, whether the effects of physical activity on bone mineral density translate into similar benefits for fracture risk is uncertain (Karlsson, 2007). Animal studies have shown that loading causes small changes in bone mineral content and bone mineral density that result in large increases in bone strength, supporting the notion that physical activity probably affects the skeleton in a way that results in important gains in bone strength (Umemura et al., 1997). The relatively recent application of peripheral quantitative computed tomography for estimating bone strength in youth has also provided some results suggesting an increase in bone strength with greater than usual physical activity (Sardinha et al., 2008; Farr et al., 2011).
The intensity of exercise appears to be a key determinant of the osteogenic response (Turner and Robling, 2003). Bone tissue, like other tissues, accommodates to usual daily activities. Thus, activities such as walking have a modest effect at best, since even relatively inactive individuals take many steps (>1,000) per day. Activities generating greater muscle force on bone, such as resistance exercise, and “impact” activities with greater than ordinary ground reaction forces (e.g., hopping, skipping, jumping, gymnastics) promote increased mineralization and modeling (Bloomfield et al., 2004; Farr et al., 2011). Far fewer randomized controlled trials (RCTs) examining this relationship have been conducted in children than in adults, and there is little evidence on dose response to show how the type of exercise interacts with frequency, intensity, and duration. Taken together, however, the available evidence supports beneficial effects of physical activity in promoting bone development (Bailey et al., 1996; Modlesky and Lewis, 2002).
Physical activity may reduce osteoporosis-related fracture risk by increasing bone mineral accrual during development; by enhancing bone strength; and by reducing the risk of falls by improving muscle strength, flexibility, coordination, and balance (Bloomfield et al., 2004). Early puberty is a key developmental period. Approximately 26 percent of the mineral content in the adult skeleton is accrued during the 2 years around the time of peak height velocity (Bailey et al., 2000). This amount of mineral accrual represents approximately the same amount of bone mineral
that most people will lose in their entire adult lives (Arlot et al., 1997). The increase in mineral contributes to increased bone strength. Mineral is accrued on the periosteal surface of bone, such that the bone grows wider. Increased bone width, independent of the increased mineral mass, also contributes to greater bone strength. Indeed, an increase of as little as 1 mm in the outer surface of bone increases strength substantially. Adding bone to the endosteal surface also increases strength (Parfitt, 1994; Wang et al., 2009). Increases in testosterone may be a greater stimulus of periosteal expansion than estrogen since testosterone contributes to wider and stronger bones in males compared with females. Retrospective studies in tennis players and gymnasts suggest structural adaptations may persist many years later in adulthood and are greatest when “impact” activity is initiated in childhood (Kannus et al., 1995; Bass et al., 1998). RCTs on this issue are few, although the available data are promising (McKay et al., 2000; Fuchs et al., 2001; MacKelvie et al., 2001, 2003; Lindén et al., 2006). Thus, impact exercise begun in childhood may result in lasting structural changes that may contribute to increased bone strength and decreased fracture risk later in life (Turner and Robling, 2003; Ferrari et al., 2006).
Adipose tissue The adipose “organ” is composed of fat cells known as adipocytes (Ailhaud and Hauner, 1998). Adipocytes are distributed throughout the body in various organs and tissues, although they are largely clustered anatomically in structures called fat depots, which include a large number of adipocytes held together by a scaffold-like structure of collagen and other structural molecules. In the traditional view of the adipocyte, the cell provides a storage structure for fatty acids in the form of triacylglycerol molecules, with fatty acids being released when metabolic fuel is needed (Arner and Eckel, 1998). While adipocytes play this critical role, they are also involved in a number of endocrine, autocrine, and paracrine actions and play a key role in regulating other tissues and biological functions, for example, immunity and blood pressure, energy balance, glucose and lipid metabolism, and energy demands of exercise (Ailhaud and Hauner, 1998; Frühbeck et al., 2001). The role of adipocytes in regulation of energy balance and in carbohydrate and lipid metabolism and the potential effects of physical activity on adipocyte function are of particular interest here, given growing concerns related to pediatric and adult obesity (Ogden et al., 2012) and the associated risk of cardiometabolic disease (Weiss et al., 2004; Eisenmann, 2007a,b; Steele et al., 2008). Metabolic differences among various fat depots are now well known (Frühbeck et al., 2001), and there is significant interest in the distribution of adipose tissue, the changes that occur during childhood and adolescence, and their clinical significance.
Adipocytes increase in size (hypertrophy) and number (hyperplasia) from birth through childhood and adolescence and into young adulthood
to accommodate energy storage needs. The number of adipocytes has been estimated to increase from about 5 billion at birth to 30 billion to 50 billion in the nonobese adult, with an increase in average diameter from about 30-40 μm at birth to about 80-100 μm in the young adult (Knittle et al., 1979; Bonnet and Rocour-Brumioul, 1981; Chumlea et al., 1982). In total the adipose organ contains about 0.5 kg of adipocytes at birth in both males and females, increasing to approximately 10 kg in average-weight-for-height males and 14 kg in females (Malina et al., 2004). There is wide interindividual variation, however, and the difficulty of investigating changes in the number and size of adipocytes is obvious given the invasiveness of the required biopsy procedures; understandably, then, data on these topics are scarce in children and adolescents. Also, since only subcutaneous depots are accessible, results must be extrapolated from a few sites.
Based on such information, the average size of adipocytes has been reported to increase two- to threefold in the first year of life, with little increase in nonobese boys and girls until puberty (Malina et al., 2004). A small increase in average adipocyte size at puberty is more obvious in girls than in boys. There is considerable variation in size across various subcutaneous sites and between subcutaneous and internal depots. The number of adipocytes is difficult to estimate. Available data suggest that the cellularity of adipose tissue does not increase significantly in early postnatal life (Malina et al., 2004). Thus, gain in fat mass is the result of an increase in the size of existing adipocytes. From about 1-2 years of age and continuing through early and middle childhood, the number of adipocytes increases gradually two- to threefold. With puberty the number practically doubles, followed by a plateau in late adolescence and early adulthood. The number of adipocytes is similar in boys and girls until puberty, when girls experience a greater increase than boys.
The increases in the number of adipocytes during infancy and puberty are considered critical for enlargement of the adipose tissue organ and for the risk of obesity. Since size and number are linked, the number of adipocytes can potentially increase at any age if fat storage mechanisms are stimulated by chronic energy surfeit (Hager, 1981; Chumlea et al., 1982). Energy expenditure through regular physical activity is a critical element in preventing energy surfeit and excess adiposity. While cellularity undoubtedly is strongly genetically determined, regular physical activity, through its contribution to energy expenditure, can contribute to less adipocyte hyperplasia by limiting hypertrophy.
Fat distribution Fat distribution refers to the location of fat depots on the body. The metabolic activities of fat depots differ, and small variation can have a long-term impact on fat distribution. Differences in metabolic properties across depots also have clinical implications. Visceral adipose tissue
in the abdominal cavity is more metabolically active (reflected by free fatty acid flux) than adipose tissue in other areas (Arner and Eckel, 1998), and higher amounts of visceral adipose tissue are associated with greater risk of metabolic complications, such as type 2 diabetes and cardiovascular disease (Daniels et al., 1999; He et al., 2007; Dencker et al., 2012). In contrast, subcutaneous fat, particularly in the gluteofemoral region, is generally associated with a lower risk of cardiometabolic disease. Age- and sex-associated variations in fat distribution contribute to age- and sex-associated differences in cardiometabolic disease prevalence. Girls have more subcutaneous fat than boys at all ages, although relative fat distribution is similar. After a rapid rise in subcutaneous fat in the first few months of life, both sexes experience a reduction through age 6 or 7 (Malina and Roche, 1983; Malina and Bouchard, 1988; Malina, 1996). Girls then show a linear increase in subcutaneous fat, whereas boys show a small increase between ages 7 and 12 or 13 and then an overall reduction during puberty. The thickness of subcutaneous fat on the trunk is approximately one-half that of subcutaneous fat on the extremities in both boys and girls during childhood. The ratio increases with age in males during adolescence but changes only slightly in girls. In males the increasing ratio of trunk to extremity subcutaneous fat is a consequence of slowly increasing trunk subcutaneous fat and a decrease in subcutaneous fat on the extremities. In girls, trunk and extremity subcutaneous fat increase at a similar rate; thus the ratio is stable (Malina and Bouchard, 1988). As a consequence, the sex difference in the distribution of body fat develops during adolescence. It is important to note that changes in subcutaneous fat pattern do not necessarily represent changes in abdominal visceral adipose tissue.
Tracking of subcutaneous fat has been investigated based on skinfold thicknesses and radiographs of fat widths in males and females across a broad age range (Katzmarzyk et al., 1999; Campbell et al., 2012). Results indicate that subcutaneous fat is labile during early childhood. After age 7 to 8, correlations between subcutaneous fat in later childhood and adolescence and adult subcutaneous fat are significant and moderate. Longitudinal data on tracking of visceral adipose tissue are not available, but percent body fat does appear to track. Thus children and especially adolescents with higher levels of body fat have a higher risk of being overfat at subsequent examinations and in adulthood, although variation is considerable, with some individuals moving away from high fatness categories, while some lean children move into higher fatness categories.
In cross-sectional studies, active children and adolescents tend to have lower skinfold thicknesses and less overall body fat than their less active peers (Loftin et al., 1998; Rowlands et al., 2000; Stevens et al., 2004; Lohman et al., 2006), although the correlations are modest, reflecting variation in body composition at different levels of physical activity, as
well as the difficulty of measuring physical activity. Longitudinal studies indicate small differences in fatness between active and inactive boys and girls. Although some school-based studies of the effects of physical activity on body composition have reported changes in BMI or skinfolds in the desired direction (Gortmaker et al., 1999; McMurray et al., 2002), most have not shown significant effects. High levels of physical activity are most likely needed to modify skinfold thicknesses and percent body fat. In adults, visceral adipose tissue declines with weight loss with exercise. In contrast, in a study of obese children aged 7-11, a 4-month physical activity program resulted in minimal change in abdominal visceral adipose tissue but a significant loss in abdominal subcutaneous adipose tissue (Gutin and Owens, 1999). In adults, decreases in fatness with exercise are due to a reduction in fat cell size, not number (You et al., 2006); whether this is true in children is not certain but appears likely. Given that adipocyte hypertrophy may trigger adipocyte hyperplasia (Ballor et al., 1998), energy expenditure through regular physical activity may be important in preventing excess adipose tissue cellularity. Regular physical activity also affects adipose tissue metabolism so that trained individuals have an increased ability to mobilize and oxidize fat, which is associated with increased levels of lipolysis, an increased respiratory quotient, and a lower risk of obesity (Depres and Lamarche, 2000).
Cardiorespiratory System
The ability to perform sustained activity under predominantly aerobic conditions depends on the capacity of the cardiovascular and pulmonary systems to deliver oxygenated blood to tissues and on the ability of tissues (primarily skeletal muscle) to extract oxygen and oxidize substrate. By age 2 the systems are fully functional, although young children lack the cardiorespiratory capacity of older children and adults because of their small size (Malina et al., 2004). Children’s aerobic capacity and consequently their ability to exercise for longer periods of time increase as they grow. Maximal aerobic power (liters per minute) increases fairly linearly in boys until about age 16, whereas it increases in girls until about age 13 and then plateaus during adolescence (Malina et al., 2004; Eisenmann et al., 2011). Differences between boys and girls are small (~10 percent) during childhood and greater after the adolescent growth spurt, when girls have only about 70 percent of the mean value of boys. Changes with age and sex differences are explained largely by differences in the size of the relevant tissues. Dimensions of the heart and lungs enlarge with age in a manner consistent with the increase in body mass and stature (Malina et al., 2004). The increase in the size of the heart is associated with increases in stroke volume (blood pumped per beat) and cardiac output (product of stroke vol-
ume and heart rate, liters per minute), despite a decline in heart rate during growth. Similarly, increase in lung size (proportional to growth in height) results in greater lung volume and ventilation despite an age-associated decline in breathing frequency. From about age 6 to adulthood, maximal voluntary ventilation approximately doubles (50-100 L/min) (Malina et al., 2004). The general pattern of increase as a function of height is similar in boys and girls. In both, lung function tends to lag behind the increase in height during the adolescent growth spurt. As a result, peak gains in lung function occur about 2 years earlier in girls than in boys.
Blood volume is highly related to body mass and heart size in children and adolescents, and it is also well correlated with maximal oxygen uptake during childhood and adolescence (Malina et al., 2004). Blood volume increases from birth through adolescence, following the general pattern for changes in body mass. Both red blood cells and hemoglobin have a central role in transport of oxygen to tissues. Hematocrit, the percentage of blood volume explained by blood cells, increases progressively throughout childhood and adolescence in boys, but only through childhood in girls. Hemoglobin content, which is related to maximal oxygen uptake, heart volume, and body mass, increases progressively with age into late adolescence. Males have greater hemoglobin concentrations than females, especially relative to blood volume, which has functional implications for oxygen transport during intense exercise.
Growth in maximal aerobic power is influenced by growth in body size, so controlling for changes in body size during growth is essential. Although absolute (liters per minute) aerobic power increases into adolescence relative to body weight, there is a slight decline in both boys and girls, suggesting that body weight increases at a faster rate than maximal oxygen consumption, particularly during and after the adolescent growth spurt (Malina et al., 2004). Changes in maximal oxygen consumption during growth tend to be related more closely to fat-free mass than to body mass. Nevertheless, sex differences in maximal oxygen consumption per unit fat-free mass persist, and maximal oxygen consumption per unit fat-free mass declines with age.
Improvements in cardiorespiratory function—involving structural and functional adaptations in the lungs, heart, blood, and vascular system, as well as the oxidative capacity of skeletal muscle—occur with regular vigorous- and moderate-intensity physical activity (Malina et al., 2004). Concern about the application of invasive techniques limits the available data on adaptations in the oxygen transport system in children. Nevertheless, it is clear that aerobic capacity in youth increases with activity of sufficient intensity and that maximal stroke volume, blood volume, and oxidative enzymes improve after exercise training (Rowland, 1996). Training-induced changes in other components of the oxygen transport system remain to be determined.
Health- and Performance-Related Fitness
Physical fitness is a state of being that reflects a person’s ability to perform specific exercises or functions and is related to present and future health outcomes. Historically, efforts to assess the physical fitness of youth focused on measures designed to evaluate the ability to carry out certain physical tasks or activities, often related to athletic performance. In more recent years, the focus has shifted to greater emphasis on evaluating health-related fitness (IOM, 2012a) and assessing concurrent or future health status. Health- and performance-related fitness, while overlapping, are different constructs. Age- and sex-related changes in the components of both are strongly linked to the developmental changes in tissues and systems that occur during childhood and adolescence. Although genetic factors ultimately limit capacity, environmental and behavioral factors, including physical activity, interact with genes to determine the degree to which an individual’s full capacity is achieved.
Health-Related Fitness
Cardiorespiratory endurance, muscular strength and endurance, flexibility, and body composition are components of health-related fitness historically assessed in school-based fitness assessment programs (IOM, 2012a). These components of health-related fitness are considered important since they can be linked to the risk of cardiometabolic disease and musculoskeletal disability, chronic hypokinetic-related diseases.
Cardiorespiratory endurance Cardiorespiratory (aerobic) endurance reflects the functioning of the pulmonary and cardiovascular systems to deliver oxygen and the ability of tissues (primarily skeletal muscle) to extract oxygen from the blood. Defined clinically as the maximum oxygen consumption during a maximal graded exercise test, in practice it is usually measured indirectly as performance on a field test of endurance, such as 1- or 2-mile run time (IOM, 2012a). During childhood, aerobic capacity approximately doubles in both boys and girls, although girls on average possess a lower capacity. Males continue to improve during adolescence, up to ages 17-18, while aerobic capacity plateaus around age 14 in females (Malina et al., 2004), resulting in an approximately 20 percent difference between males and females (Rowland, 2005).
Favorable associations have been found between aerobic endurance and high-density lipoproteins, systolic blood pressure, diastolic blood pressure, BMI, measures of fatness, arterial stiffness, and measures of insulin sensitivity (Boreham et al., 2004; Imperatore et al., 2006; Hussey et al., 2007; Ondrak et al., 2007). Some evidence suggests a decline in aerobic endurance among U.S. youth in recent decades (Eisenmann, 2003; Carnethon et al.,
2005; Pate et al., 2006), coincident with increased sedentariness and obesity and a greater prevalence of metabolic syndrome in youth. Aerobic exercise has been shown to increase cardiorespiratory endurance by about 5-15 percent in youth (Malina et al., 2004; HHS, 2008). The programs that produce this benefit involve continuous vigorous- or moderate-intensity aerobic activity of various types for 30-45 minutes per session at least 3 days per week over a period of at least 1-3 months (Baquet et al., 2002); improvements are greater with more frequent exercise (Baquet et al., 2003).
Muscle strength and endurance Muscle strength is defined as the highest force generated during a single maximum voluntary contraction, whereas muscle endurance is the ability to perform repeated muscular contraction and force development over a period of time. Muscle strength and endurance are correlated, especially at higher levels of force production. Muscle strength is proportional to the cross-sectional area of skeletal muscle; consequently, strength growth curves parallel growth curves for body weight and skeletal muscle mass (Malina et al., 2004).
Both males and females show impressive increases in muscle strength from childhood to adolescence. Strength in children increases linearly, with boys having a slight advantage over girls. However, these sex differences are magnified during the adolescent years as a result of maturation (Malina and Roche, 1983). Differences in muscle strength between boys and girls become more apparent after puberty, primarily as a result of the production of sex steroid hormones. In boys the increase in strength during adolescence lags behind the growth spurt by at least a year (peak height velocity), which may explain why some boys experience a brief period of clumsiness or awkwardness during puberty, as they have not yet acquired the muscle strength necessary to handle the changes associated with their larger bodies. Muscle strength increases at its greatest rate approximately 1 year after peak height velocity in boys, whereas for girls the strength spurt generally occurs during the same year as peak height velocity (Bar-Or, 1983).
A compelling body of evidence indicates that with resistance training children and adolescents can significantly increase their strength above that expected as a result of normal growth and maturation, provided that the training program is of sufficient intensity, volume, and duration (Committee on Sports Medicine Fitness, 2001). Both boys and girls can benefit, and strength gains in children as young as 5-6 have been reported (Faigenbaum et al., 2009), although most studies are of older children and adolescents. Gains in muscle strength of about 30 percent are typical, although considerably larger gains have been reported. Adolescents make greater gains than preadolescents in absolute strength, whereas reported relative (percent above initial strength) gains in strength during preadolescence and adolescence are similar. A variety of programs and modalities have proved
efficacious (Council on Sports Medicine Fitness, 2008), as long as load (~10-15 repetitions maximum) and duration (~8-20 weeks) are adequate. As in adults, training adaptations in youth are specific to the muscle action or muscle groups that are trained, and gains are transient if training is not maintained (Faigenbaum et al., 2009).
Youth resistance training, as with most physical activities, does carry some degree of risk of musculoskeletal injury, yet the risk is no greater than that associated with other sports and activities in which children and adolescents participate (Council on Sports Medicine Fitness, 2008; Faigenbaum et al., 2009) as long as age-appropriate training guidelines are followed. A traditional area of concern has been the potential for training-induced damage to growth cartilage, which could result in growth disturbances. However, a recent review found no reports of injury to growth cartilage in any prospective study of resistance training in youth and no evidence to suggest that resistance training negatively impacts growth and maturation during childhood and adolescence (Faigenbaum et al., 2009). Injuries typically occur in unsupervised settings and when inappropriate loads and progressions are imposed.
In addition to the obvious goal of gaining strength, resistance training may be undertaken to improve sports performance and prevent injuries, rehabilitate injuries, and enhance health. Appropriately supervised programs emphasizing strengthening of trunk muscles in children theoretically benefit sport-specific skill acquisition and postural control, although these benefits are difficult to study and thus are supported by little empirical evidence (Council on Sports Medicine Fitness, 2008). Similarly, results are inconsistent regarding the translation of increased strength to enhanced athletic performance in youth. Limited evidence suggests that strength-training programs that address common overuse injuries may help reduce injuries in adolescents, but whether the same is true in preadolescents is unclear (Council on Sports Medicine Fitness, 2008). Increasing evidence suggests that strength training, like other forms of physical activity, has a beneficial effect on measurable health indices in youth, such as cardiovascular fitness, body composition, blood lipid profiles and insulin sensitivity (Faigenbaum, 2007; Benson et al., 2008), bone mineral density and bone geometry (Morris et al., 1997; MacKelvie et al., 2004), and mental health (Holloway et al., 1988; Faigenbaum et al., 1997; Annesi et al., 2005; Faigenbaum, 2007). Some work has shown that muscle fitness, reflected in a composite index combining measures of muscle strength and endurance, and cardiorespiratory fitness are independently and negatively associated with clustered metabolic risk (Steene-Johannessen et al., 2009). Moreover, children with low muscle strength may be at increased risk of fracture with exercise (Clark et al., 2011). Finally, muscle hypertrophy, which adds to fat-free mass, contributes to resting metabolic rate and therefore total daily
energy expenditure. Resistance training may be particularly useful for raising metabolic rate in overweight and obese children without the risk associated with higher-impact activities (Watts et al., 2005; Benson et al., 2007).
Flexibility Flexibility has been operationally defined as “the intrinsic property of body tissues, including muscle and connective tissues, that determines the range of motion achievable without injury at a joint or group of joints” (IOM, 2012b, p. 190). At all ages, girls demonstrate greater flexibility than boys, and the difference is greatest during the adolescent growth spurt and sexual maturation. Perhaps the most common field measure of flexibility in children and youth is the sit-and-reach test (IOM, 2012b) of low-back flexibility. Low-back flexibility as measured by this test is stable in girls from age 5 to 11 and increases until late adolescence. In boys, low-back flexibility declines linearly starting at age 5, reaching its nadir at about age 12, and then increases into late adolescence. The unique pattern of age- and sex-associated variation is related to the growth of the lower extremities and the trunk during adolescence. In boys the nadir in low-back flexibility coincides with the adolescent growth spurt in leg length. In both boys and girls, the increase during adolescence coincides with the growth spurt in trunk length and arm length, which influences reach. Flexibility in both males and females tends to decline after age 17, in part as a result of a decline in physical activity and normal aging.
The principal health outcomes hypothesized to be associated with flexibility are prevention of and relief from low-back pain, prevention of musculoskeletal injury, and improved posture. These associations have been studied in adults, with equivocal results (Plowman, 1992). Although flexibility has long been included in national youth fitness tests, it has proven difficult to establish a link between flexibility and health (IOM, 2012a). In contrast to other fitness components that are general or systemic in nature, flexibility is highly specific to each joint of the body. Although appropriate stretching may increase flexibility, establishing a link to improved functional capacity and fitness is difficult. A few studies suggest that improvements in flexibility as measured by the sit-and-reach test may be related to less low-back pain (Jones et al., 2007; Ahlqwist et al., 2008), but the evidence is weak. Consequently, the Institute of Medicine (IOM) Committee on Fitness Measures and Health Outcomes in its recent report elected to forego recommending a flexibility test for a national youth fitness test battery pending further research to confirm the relationship between flexibility and health and to develop national normative data (IOM, 2012a).
Body composition Body composition is the component of health-related fitness that relates to the relative amount of adipose tissue, muscle, bone, and other vital components (e.g., organs, connective tissues, fluid compart-
ments) that make up body weight. Most feasible methods for assessing body composition are based on models that divide the body into fat and fat-free (all nonfat constituents) components (Going et al., 2012). Although fat mass and adipose tissue are not equivalent components, fat mass is easier to estimate than adipose tissue, and it is correlated with performance and disease risk. In settings in which estimation of body fat is difficult, weight-for-height ratios often are used as surrogates for body composition. Indeed, definitions of pediatric overweight and obesity have been based on BMI, calculated as weight in kilograms divided by height squared. Child and adolescent obesity defined by BMI remains at all-time highs. Population surveys indicate that approximately 33 percent of all boys and girls are overweight, and nearly one in five are obese (Ogden and Flegal, 2011). The tendency for excess fatness to persist from childhood and adolescence into adulthood (Daniels et al., 2005), coupled with the strong association between obesity and chronic disease (Weiss and Caprio, 2005; Barlow, 2007), has caused great concern for future obesity levels and the health of youth and adults alike (IOM, 2005, 2012b).
The increase in prevalence of obesity is undoubtedly due to a mismatch between energy intake and expenditure. Population surveys have shown that few children and youth meet recommended levels of daily physical activity (see Chapter 2). Prospective studies have shown a significant and inverse relationship between habitual physical activity and weight gain (Berkey et al., 2003), and in some studies physical activity is a better predictor of weight gain than estimates of calorie or fat intake (Berkey et al., 2000; Janssen et al., 2005). These relationships are better established in adults than in children and youth, although even in preschool children, low levels of physical activity, estimated from doubly labeled water, were found to be indicative of higher body fat content (Davies et al., 1995). While studies of exercise without caloric restriction generally show only small effects on body weight, significant albeit moderate reductions of body fat are generally reported (Eisenmann, 2003). Moreover, even in the absence of significant weight loss, exercise has beneficial effects on risk factors for cardiometabolic disease (Ross and Bradshaw, 2009; Gutin and Owens, 2011).
Body mass index Changes in weight for height with growth and maturation for U.S. boys and girls are described in CDC growth curves (Kuczmarski et al., 2000). Current growth curves were derived from U.S. population surveys conducted before the increase in weight for height that defines today’s pediatric obesity epidemic. In boys and girls, BMI declines during early childhood, reaching its nadir at about ages 5-6, and then increases through adolescence. A gender difference emerges during puberty, with males gaining greater fat-free mass than females. Both the
period of “adiposity rebound” (the increase in BMI in midchildhood following the decline in early childhood) and puberty are times of risk for excess fat gain that correlates with future adiposity (Rolland-Cachera et al., 1984). Physical activity and BMI are inversely correlated in children and adolescents, although the correlations are modest (Lohman et al., 2006), reflecting the difficulty of measuring physical activity, as well as variation in body composition and physical activity at a given weight (Rowlands et al., 2000). Indeed, when studied separately, fat mass index (FMI, or fat mass divided by height squared) and fat-free mass index (FFMI, or fat-free mass divided by height squared) are both inversely related to physical activity. With FMI controlled, however, FFMI is positively related to physical activity, indicating that, for a given level of body fat, individuals with more fat-free mass are more active (Lohman et al., 2006). BMI cut-points for defining overweight and obesity have historically been based on age- and gender-specific population distributions of BMI. Recent work has shown good correspondence between BMI standards and percent fat standards that are referenced to health criteria (Laurson et al., 2011). These new standards should prove useful for identifying children and adolescents at risk for higher levels of cardiometabolic risk factors.
Percent body fat Direct measures of body fat as a percent of weight provide a better index of adiposity and health risk than BMI (Zeng et al., 2012), which is confounded by variation in lean tissue mass relative to height. Recently, percent fat growth curves were established for representative samples of U.S. boys and girls using National Health and Nutrition Examination Survey (NHANES) data (Laurson et al., 2011; Ogden and Flegal, 2011). Median percent fat for boys aged 5-18 ranged from 14 to 19 percent and for girls across the same ages 15 to 28 percent. In both boys and girls, percent fat increases slowly during early childhood, with girls having a consistently greater relative fatness than boys after ages 5-6. In girls, percent fat increases gradually throughout adolescence in the same manner as fat mass. In boys, percent fat increases gradually until the adolescent growth spurt and thereafter gradually declines until about age 16-17, reflecting the rapid growth in fat-free mass relative to fat mass. After age 17, percent fat in males gradually increases again into adulthood.
The increased prevalence of child and adolescent obesity as defined by BMI presumably also reflects increased adiposity, although the degree is not certain as population-based estimates of percent fat have only recently been developed (Laurson et al., 2011). Health-related percent fat standards recently were developed by determining levels of body fat associated with greater occurrence of chronic disease risk factors defined by metabolic syndrome (Going et al., 2011). In boys and girls aged 12-18, body fat above
20-24 percent and above 27-31 percent, respectively, was predictive of metabolic syndrome.
Physical activity is inversely correlated with percent body fat (Rowlands et al., 2000; Lohman et al., 2006), although the correlations are modest, and changes in overall fatness as well as subcutaneous adipose tissue with habitual physical activity are reasonably well documented in children and adolescents (Gutin and Humphries, 1998; Gutin and Owens, 1999; Dionne et al., 2000). In youth, as in adults, the effects of exercise without caloric restriction are modest and are influenced by the initial level of body fat and the duration and regimen of exercise (Going, 1999). Experimental studies have documented reductions in percent body fat with aerobic exercise, especially in children and adolescents who are overweight or obese at the initiation of an exercise program (Davis et al., 2012). Regular physical activity also affects adipose tissue metabolism (Gutin and Owens, 1999). Individuals who engage in aerobic endurance exercise training have an increased ability to mobilize and oxidize fat, which is associated with increased levels of lipolysis (Depres and Lamarche, 2000). Similar information on adipose tissue metabolism in children and youth is lacking, although one can reasonably expect similar adaptations in older adolescents.
Metabolic syndrome The tendency for risk factors for cardiometabolic disease to cluster, now called metabolic syndrome, is well recognized in adults (Alberti and Zimmet, 1998). Similar clustering occurs in older children and especially adolescents (Cook et al., 2003), and interest in metabolic syndrome has increased, driven by the increased prevalence of pediatric obesity and the increasing incidence and earlier onset of type 2 diabetes in youth. There is as yet no accepted definition of metabolic syndrome for use in pediatric populations (Jolliffe and Janssen, 2007). Typically, adult definitions are extrapolated to children and adolescents, with appropriate adjustments of the thresholds for the defining variables. Perhaps the most common approach is to emulate the National Cholesterol Education Program (NCEP), which defines metabolic syndrome as exceeding thresholds on three of five components: waist circumference, blood pressure (systolic or diastolic), blood lipids (high-density lipoprotein [HDL] and triglycerides), and blood glucose levels (NIH, 2001).
The concept of metabolic syndrome is useful as it provides an integrated index of risk, and it recently was used to derive health-related percent-body-fat standards (Laurson et al., 2011). Based on NHANES data, the prevalence of metabolic syndrome varies with the degree of obesity, and it is estimated at 4-6 percent of children and adolescents (Cook et al., 2003; Dubose et al., 2007); among obese youth it may be as high as 30-50 percent (Weiss et al., 2004). Youth with metabolic syndrome have an increased risk of type 2 diabetes and cardiovascular disease. In adults a
loss of 5-10 percent of body weight through calorie restriction and exercise has been shown to reduce the risk of cardiometabolic disease by improving risk factors (Diabetes Prevention Program Research Group, 2002; Ross and Janiszewski, 2008). In particular, weight loss results in reduced visceral adipose tissue, a strong correlate of risk (Knowler et al., 2002), as well as lower blood pressure and blood glucose levels due to improved insulin sensitivity. Even without significant weight loss, exercise can have significant effects in adults by improving glucose metabolism, improving lipid and lipoprotein profiles, and lowering blood pressure, particularly for those who are significantly overweight (Ross and Bradshaw, 2009). Similar benefits have been observed in adolescents.
A growing body of literature addresses the associations of physical activity, physical fitness, and body fatness with the risk of metabolic syndrome and its components in children and especially adolescents (Platat et al., 2006; McMurray et al., 2008; Rubin et al., 2008; Thomas and Williams, 2008; Christodoulos et al., 2012). Studies in adults have shown that higher levels of physical activity predict slower progression toward metabolic syndrome in apparently healthy men and women (Laaksonen et al., 2002; Ekelund et al., 2005), an association that is independent of changes in body fatness and cardiorespiratory fitness (Ekelund et al., 2007). Few population studies have focused on these relationships in children and adolescents, and the use of self-reported activity, which is imprecise in these populations, tends to obscure associations. In a large sample of U.S. adolescents aged 12-19 in the 1999-2002 NHANES, for example, there was a trend for metabolic syndrome to be more common in adolescents with low activity levels than in those with moderate or high activity levels, although the differences among groups were not statistically significant (Pan and Pratt, 2008). Moreover, for each component of metabolic syndrome, prevalence was generally lower with higher physical activity levels, and adolescents with low physical activity levels had the highest rates of all metabolic syndrome components.
The association between cardiorespiratory fitness and metabolic syndrome also was examined in the 1999-2002 NHANES (Lobelo et al., 2010). Cardiorespiratory fitness was measured as estimated peak oxygen consumption using a submaximal treadmill exercise protocol, and metabolic syndrome was represented as a “clustered score” derived from five established risk factors for cardiovascular disease, an adiposity index, insulin resistance, systolic blood pressure, triglycerides, and the ratio of total to HDL cholesterol. Mean clustered risk score decreased across increasing fifths (quintiles) of cardiorespiratory fitness in both males and females. The most significant decline in risk score was observed from the first (lowest) to the second quintile (53.6 percent and 37.5 percent in males and females, respectively), and the association remained significant in both overweight
and normal-weight males and in normal-weight females. Other studies, using the approach of cross-tabulating subjects into distinct fitness and fatness categories, have examined associations of fitness and fatness with metabolic syndrome risk (Eisenmann et al., 2005, 2007a,b; Dubose et al., 2007). Although different measures of fitness, fatness, and metabolic syndrome risk were used, the results taken together across a wide age range (7-18) show that fitness modifies the influence of fatness on metabolic syndrome risk. In both males and females, high-fit/low-fatness subjects have less metabolic syndrome risk than low-fit/high-fatness subjects (Eisenmann, 2007).
That many adult chronic health conditions have their origins in childhood and adolescence is well supported (Kannel and Dawber, 1972; Lauer et al., 1975; Berenson et al., 1998; IOM, 2004). Both biological (e.g., adiposity, lipids) and behavioral (e.g., physical activity) risk factors tend to track from childhood and especially adolescence into adulthood. Childhood BMI is related to adult BMI and adiposity (Guo et al., 1994, 2000; Freedman et al., 2005), and as many as 80 percent of obese adolescents become obese adults (Daniels et al., 2005). Coexistence of cardiometabolic risk factors, even at young ages (Dubose et al., 2007; Ramírez-Vélez et al., 2012), has been noted, and these components of metabolic syndrome also have been shown to track to adulthood (Bao et al., 1994; Katzmarzyk et al., 2001; Huang et al., 2008). Landmark studies from the Bogalusa Heart Study (Berenson et al., 1998; Li et al., 2003) and others (Mahoney et al., 1996; Davis et al., 2001; Morrison et al., 2007, 2008) have demonstrated that cardiometabolic risk factors present in childhood are predictive of adult disease.
The benefits of exercise for prevention and treatment of cardiometabolic disease in adults are well described (Ross et al., 2000; Duncan et al., 2003; Gan et al., 2003; Irwin et al., 2003; Lee et al., 2005; Sigal et al., 2007; Ross et al., 2012). Prospective studies examining the effects of exercise on metabolic syndrome in children and adolescents remain limited, and it is important to refrain from extrapolating intervention effects observed in adults to youth, although one might reasonably assume the benefits in older adolescents to be similar to those in young adults. Indeed, based on the inverse associations of physical activity and physical fitness with metabolic syndrome (Kim and Lee, 2009) and on the available intervention studies, some experts have recommended physical activity as the main therapeutic tool for prevention and treatment of metabolic syndrome in childhood (Brambilla et al., 2010). Comparative studies in adults have shown that the effect of exercise on weight is limited and generally less than that of calorie restriction (Brambilla et al., 2010). Moreover, the relative effectiveness of diet and exercise depends on the degree of excess fatness (Brambilla et al., 2010). Comparative studies in children and youth are few, as behavioral
interventions in overweight children and adolescents commonly combine exercise and dietary restriction, making it difficult to disentangle their independent effects. Nonetheless, diet and exercise have different effects on body composition: While both contribute to fat loss, only exercise increases muscle mass and thus has a direct effect on metabolic health. In children and youth, as in adults, the effect of exercise on cardiometabolic risk factors is greater in overweight/obese youth than in their normal-weight peers (Kang et al., 2002; Lazaar et al., 2007).
Exercise also may have important benefits even without significant modification of body composition (Bell et al., 2007). Experimental studies in overweight and obese youth have shown that exercise leads to reductions in visceral fat (Owens et al., 1999; Gutin et al., 2002; Lee at al., 2005; Barbeau et al., 2007; Kim and Lee, 2009) without a significant change in BMI, as well as improvement in markers of metabolic syndrome, primarily fasting insulin and insulin resistance (Treuth et al., 1998; Ferguson et al., 1999; Carrel et al., 2005; Nassis et al., 2005; Meyer et al., 2006; Shaibi et al., 2006; Bell et al., 2007). Results from experimental studies of the effects of exercise on lipids and lipoproteins (Stoedefalke et al., 2000; Kelley and Kelley, 2008; Janssen and LeBlanc, 2010) are mixed. Although some studies have shown improved lipid and lipoprotein profiles, primarily a decrease in low-density lipoprotein (LDL) cholesterol and triglyceride concentrations and an increase in HDL cholesterol (Ferguson et al., 1999), other studies have shown no improvement in these outcomes (Kelley and Kelley, 2008). In part, such conflicting results are likely due to initial differences in body composition and severity of hyperlipidemia. Well-controlled exercise training studies in obese children (Escalante et al., 2012) and children with adverse blood lipid and lipoprotein profiles have shown positive alterations in their profiles (Stoedefalke et al., 2000), whereas results in normolipid-emic children and adolescents are equivocal. Similarly, exercise has little effect on resting blood pressure in normotensive children and adolescents (Kelley and Kelley, 2008), whereas reductions in resting systolic and sometimes diastolic pressures have been reported in youth with high blood pressure (Hagberg et al., 1983, 1984; Danforth et al., 1990; Ewart et al., 1998; Farpour-Lambert et al., 2009; Janssen and LeBlanc, 2010).
In adults, physical activity is inversely associated with low-grade inflammation (Wärnberg et al., 2010; Ertek and Cicero, 2012), which is now recognized as a significant feature of metabolic syndrome and an independent predictor of cardiometabolic disease (Malina, 2002). In obese children and adolescents, as in their adult counterparts, elevation of inflammatory markers is evident, and observational studies have shown significant relationships among physical activity, physical fitness, and inflammation (Isasi et al., 2003; Platat et al., 2006; Ruiz et al., 2007; Wärnberg et al., 2007; Wärnberg and Marcos, 2008). These relationships are better studied and
stronger in adolescents than in children. In one study of boys and girls aged 10-15, those who were obese and unfit had the highest levels of systemic inflammation, whereas those who were obese yet fit had levels as low as those who were lean and fit (Halle et al., 2004). In another study, low-grade inflammation was negatively associated with muscle strength in overweight adolescents after controlling for cardiorespiratory fitness, suggesting that high levels of muscle strength may counteract some of the negative consequences of higher levels of body fat (Ruiz et al., 2008). Experimental studies of the effects of exercise and markers of low-grade inflammation in children and adolescents are lacking. Improved cardiorespiratory fitness in adults (Church et al., 2002), however, has been shown to be inversely related to concentration of C-reactive protein (CRP), a marker of low-grade inflammation. In a small study of a lifestyle intervention entailing 45 minutes of physical activity 3 times per week for 3 months, a small reduction in body fat and an overall decrease in inflammatory factors (CRP, interleukin [IL]-6) were seen in obese adolescents (Balagopal et al., 2005).
Performance-Related Fitness
Speed, muscle power, agility, and balance (static and dynamic) are aspects of performance-related fitness that change during body development in predictable ways associated with the development of tissues and systems discussed above (Malina et al., 2004). Running speed and muscle power are related, and both depend on full development of the neuromuscular system. Running speed and muscle power are similar for boys and girls during childhood (Haubenstricker and Seefeldt, 1986). After puberty, largely because of differences in muscle mass and muscle strength, males continue to make significant annual gains, while females tend to plateau during the adolescent years. Sociocultural factors and increasing inactivity among girls relative to boys, along with changes in body proportion and a lowering of the center of gravity, may also contribute to gender differences (Malina et al., 2004).
Balance—the ability to maintain equilibrium—generally improves from ages 3 to 18 (Williams, 1983). Research suggests that females outperform males on tests of static and dynamic balance during childhood and that this advantage persists through puberty (Malina et al., 2004).
Motor performance is related in part to muscle strength. Increases in muscle strength as a result of resistance exercise were described above. A question of interest is whether gains in strength transfer to other performance tasks. Available results are variable, giving some indication that gains in strength are associated with improvement in some performance tasks, such as sprinting and vertical jump, although the improvements are generally small, highlighting the difficulty of distinguishing the effects
of training from changes expected with normal growth. Changes in body size, physique, and body composition associated with growth and maturation are important factors that affect strength and motor performance. The relationships vary among performance measures and with age, and these factors often are inadequately controlled in studies of components of performance-related fitness and performance tasks.
Research supports the positive impact of physical activity on the overall psychological health and social engagement of every student. A well-designed physical education curriculum provides students with social and emotional benefits (NASPE, 2001). Simultaneously, exposure to failure experiences, emphasis on competitive sports, and elitism for naturally inclined athletes, along with bullying and teasing of unfit, uncoordinated, and overweight youth, may be important factors discouraging participation in current and future physical activity (Kohl and Hobbs, 1998; Sallis et al., 2000; Allender et al., 2006). School-based physical activity, including physical education and sports, is designed to increase physical activity while also improving motor skills and development, self-efficacy, and general feelings of competency and engaging children socially (Bailey, 2006). The hoped-for psychosocial outcomes of physical education and other physical activity programs in the school setting have been found to be critical for continued physical activity across the life span and are themselves powerful long-term determinants of physical activity (Bauman et al., 2012). Unfortunately, significant gaps exist between the intent and reality of school-based physical education and other activity programs (HHS, 2013).
A large number of psychological and social outcomes have been examined. Specific aspects of psychosocial health showing a beneficial relationship to physical activity include, among others, self-efficacy, self-concept, self-worth (Haugen et al., 2011), social behaviors (Cradock et al., 2009), pro-school attitudes, motivation and goal orientation (Digelidis et al., 2003), relatedness, friendships (de la Haye et al., 2011; Macdonald-Wallis et al., 2011), task orientation, team building, bullying, and racial prejudice (Byrd and Ross, 1991). Most studies are descriptive, finding bidirectional associations between psychosocial outcomes and physical activity. Reviews and meta-analyses confirm a positive association between physical activity and self-esteem, especially for aerobic activities (McAuley, 1994).
Among psychosocial factors, self-efficacy (confidence in one’s ability to be physically active in specific situations) has emerged as an important correlate of physical activity from a large body of work based on the durable and practically useful social learning theory (Bandura and McClelland, 1977; Bandura, 1995). Bandura’s theory compels consideration of the
psychosocial and physical environments, the individual, and in this case the behavior of physical activity. Using this framework, physical activity itself has been shown to be a consistent positive correlate as well as a determinant of physical activity in children and adolescents. A large amount of reviewed research has found that physical education and physical activity experiences can increase children’s confidence in being active and lead to continued participation in physical activity (Bauman et al., 2012). RCTs have shown that both self-efficacy and social interactions leading to perceived social support influence changes in physical activity (Dishman et al., 2009). Skill mastery, confidence building, and group support are well-known strategies for advancing student learning and well-being in many educational domains in the school setting and apply equally to school physical education and other physical activity. Early observational studies of physical, social, and environmental determinants of physical activity at home, school, and recess indicated that prompts to be active (or not) from peers and adults accounted for a significant amount of the variance in directly observed physical activity (Elder et al., 1998). One longitudinal study following the variability and tracking of physical activity in young children showed that most of the variability in both home and recess activity was accounted for by short-term social and physical environmental factors, such as prompts from others and being outdoors (Sallis et al., 1995). Another study, examining activity among preschool children, found that, contrary to common belief, most of the time spent in preschool was sedentary, and correlates of activity were different for preschool boys and girls (Byun et al., 2011). In addition, significant variation in activity by preschool site was noted, indicating that local environmental conditions, including physical environment and equipment, policies, and teacher and administrative quality characteristics, play an important role in promoting physical activity (Brown et al., 2009).
Studies in middle and high school populations have strengthened the evidence base on relationships among self-efficacy, physical activity, and social support (from adults and peers). This research has highlighted the central contribution of self-efficacy and social support in protecting against a decline in activity levels among adolescent girls (Dishman et al., 2009, 2010). Evidence indicates further that these impacts spread to activities outside the school setting (Lytle et al., 2009). Findings of a related study suggest that leisure-time physical activity among middle school students was linked to motivation-related experiences in physical education (Cox et al., 2008).
A recent review of reviews (Bauman et al., 2012) found that population levels of physical activity are low and that consistent individual-level correlates of physical activity are age, sex, health status, self-efficacy, and previous physical activity. Physical activity declines dramatically as children progress from elementary through high school (Nader et al., 2008). Boys are con-
sistently found to be more active than girls from ages 4 to 9. For other age groups of children and adolescents, sex is correlated with but not a determinant of activity (Bauman et al., 2012). These findings suggest the need to tailor physical education and physical activity programs for youth specifically to increase self-efficacy and enjoyment of physical activity among girls (Dishman et al., 2005; Barr-Anderson et al., 2008; Butt et al., 2011).
In summary, a broad range of beneficial psychosocial health outcomes have been associated with physical activity. The promotion of more physical activity and quality physical education in the school setting is likely to result in psychosocially healthier children who are more likely to engage in physical activity as adults. Schools can play an important role in ensuring opportunities for physical activity for a segment of the youth population that otherwise may not have the resources to engage in such activity. It makes sense to assume that, if physical activity experiences and environments were once again structured into the daily school environment of children and adolescents, individuals’ feelings of self-efficacy regarding physical activity would increase in the U.S. population.
Mental illness is a serious public health issue. It has been estimated that by 2010 mental illness will account for 15 percent of the global burden of disease (Biddle and Mutrie, 2008; Biddle and Asare, 2011). Young people are disproportionately affected by depression, anxiety, and other mental health disorders (Viner and Booy, 2005; Biddle and Asare, 2011). Approximately 20 percent of school-age children have a diagnosable mental health disorder (U.S. Public Health Service, 2000), and overweight children are at particular risk (Ahn and Fedewa, 2011). Mental health naturally affects academic performance on many levels (Charvat, 2012). Students suffering from depression, anxiety, mood disorders, and emotional disturbances perform more poorly in school, exhibit more behavioral and disciplinary problems, and have poorer attendance relative to mentally healthy children. Thus it is in schools’ interest to take measures to support mental health among the student population. In addition to other benefits, providing adequate amounts of physical activity in a way that is inviting and safe for children of all ability levels is one simple way in which schools can contribute to students’ mental health.
Impact of Physical Activity on Mental Health
Several recent reviews have concluded that physical activity has a positive effect on mental health and emotional well-being for both adults and children (Peluso and Guerra de Andrade, 2005; Penedo and Dahn, 2005;
Strong et al., 2005; Hallal et al., 2006; Ahn and Fedewa, 2011; Biddle and Asare, 2011). Numerous observational studies have established the association between physical activity and mental health but are inadequate to clarify the direction of that association (Strong et al., 2005). It may be that physical activity improves mental health, or it may be that people are more physically active when they are mentally healthy. Most likely the relationship is bidirectional.
Several longitudinal and intervention studies have clarified that physical activity positively impacts mental health (Penedo and Dahn, 2005; Strong et al., 2005). Physical activity has most often been shown to reduce symptoms of depression and anxiety and improve mood (Penedo and Dahn, 2005; Dishman et al., 2006; Biddle and Asare, 2011). In addition to reducing symptoms of depression and anxiety, studies indicate that regular physical activity may help prevent the onset of these conditions (Penedo and Dahn, 2005). Reductions in depression and anxiety are the commonly measured outcomes (Strong et al., 2005; Ahn and Fedewa, 2011). However, reductions in states of confusion, anger, tension, stress, anxiety sensitivity (a precursor to panic attacks and panic disorders), posttraumatic stress disorder/psychological distress, emotional disturbance, and negative affect have been observed, as well as increases in positive expectations; fewer emotional barriers; general well-being; satisfaction with personal appearance; and improved life satisfaction, self-worth, and quality of life (Heller et al., 2004; Peluso and Guerra de Andrade, 2005; Penedo and Dahn, 2005; Dishman et al., 2006; Hallal et al., 2006; Ahn and Fedewa, 2011; Biddle and Asare, 2011). Among adolescents and young adult females, exercise has been found to be more effective than cognitive-behavioral therapy in reducing the pursuit of thinness and the frequency of bingeing, purging, and laxative abuse (Sundgot-Borgen et al., 2002; Hallal et al., 2006). The favorable effects of physical activity on sleep may also contribute to mental health (Dishman et al., 2006).
The impact of physical activity on these measures of mental health is moderate, with effect sizes generally ranging from 0.4 to 0.7 (Biddle and Asare, 2011). In one meta-analysis of intervention trials, the RCTs had an effect size of 0.3, whereas other trials had an effect size of 0.57.
Ideal Type, Length, and Duration of Physical Activity
Intervention trials that examine the relationship between physical activity and mental health often fail to specify the exact nature of the intervention, making it difficult to determine the ideal frequency, intensity, duration, and type of physical activity involved (Penedo and Dahn, 2005; Ahn and Fedewa, 2011; Biddle and Asare, 2011).
Many different types of physical activity—including aerobic activity, resistance training, yoga, dance, flexibility training, walking programs, and body building—have been shown to improve mood and other mental health indicators. The evidence is strongest for aerobic physical activity, particularly for reduction of anxiety symptoms and stress (Peluso and Guerra de Andrade, 2005; Dishman et al., 2006; Martikainen et al., 2013), because more of these studies have been conducted (Peluso and Guerra de Andrade, 2005). One meta-analysis of RCTs concluded that physical activity interventions focused exclusively on circuit training had the greatest effect on mental health indicators, followed closely by interventions that included various types of physical activity (Ahn and Fedewa, 2011). Among studies other than RCTs, only participation in sports had a significant impact on mental health (Ahn and Fedewa, 2011). The few studies that investigated the impact of vigorous- versus lower-intensity physical activity (Larun et al., 2006; Biddle and Asare, 2011) found no difference, suggesting that perhaps all levels of physical activity may be helpful. Among adults, studies have consistently shown beneficial effects of both aerobic exercise and resistance training. Ahn and Fedewa (2011) concluded that both moderate and intense physical activity have a significant impact on mental health, although when just RCTs were considered, only intense physical activity was significant (Ahn and Fedewa, 2011). While physical activity carries few risks for mental health, it is important to note that excessive physical activity or specialization too early in certain types of competitive physical activity has been associated with negative mental health outcomes and therefore should be avoided (Peluso and Guerra de Andrade, 2005; Hallal et al., 2006). Furthermore, to reach all children, including those that may be at highest risk for inactivity, obesity, and mental health problems, physical activity programming needs to be nonthreatening and geared toward creating a positive experience for children of all skill and fitness levels (Amis et al., 2012).
Various types of physical activity programming have been shown to have a positive influence on mental health outcomes. Higher levels of attendance and participation in physical education are inversely associated with feelings of sadness and risk of considering suicide (Brosnahan et al., 2004). Classroom physical activity is associated with reduced use of medication for attention deficit hyperactivity disorder (Katz et al., 2010). And participation in recess is associated with better student classroom behavior, better focus, and less fidgeting (Pellegrini et al., 1995; Jarrett et al., 1998; Barros et al., 2009).
Strong evidence supports the short-term benefits of physical activity for mental health. Acute effects can be observed after just one episode and can last from a few hours to up to 1 day after. Body building may have a similar effect, which begins a few hours after the end of the exercise. The ideal
length and duration of physical activity for improving mental health remain unclear, however. Regular exercise is associated with improved mood, but results are inconsistent for the association between mood and medium- or long-term exercise (Dua and Hargreaves, 1992; Slaven and Lee, 1997; Dimeo et al., 2001; Dunn et al., 2001; Kritz-Silverstein et al., 2001; Sexton et al., 2001; Leppamaki et al., 2002; Peluso and Guerra de Andrade, 2005). Studies often do not specify the frequency and duration of physical activity episodes; among those that do, interventions ranged from 6 weeks to 2 years in duration. In their meta-analysis, Ahn and Fedewa (2011) found that, comparing interventions entailing a total of more than 33 hours, 20-33 hours, and less than 20 hours, the longer programs were more effective. Overall, the lack of reporting and the variable length and duration of reported interventions make it difficult to draw conclusions regarding dose (Ahn and Fedewa, 2011).
In addition to more structured opportunities, naturally occurring physical activity outside of school time is associated with fewer depressive symptoms among adolescents (Penedo and Dahn, 2005). RCTs have demonstrated that physical activity involving entire classrooms of students is effective in alleviating negative mental health outcomes (Ahn and Fedewa, 2011). Non-RCT studies have shown individualized approaches to be most effective and small-group approaches to be effective to a more limited extent (Ahn and Fedewa, 2011). Interventions have been shown to be effective in improving mental health when delivered by classroom teachers, physical education specialists, or researchers but may be most effective when conducted with a physical education specialist (Ahn and Fedewa, 2011). Many physical activity interventions include elements of social interaction and support; however, studies to date have been unable to distinguish whether the physical activity itself or these other factors account for the observed effects on mental health (Hasselstrom et al., 2002; Hallal et al., 2006). Finally, a few trials (Larun et al., 2006; Biddle and Asare, 2011) have compared the effects of physical activity and psychosocial interventions, finding that physical activity may be equally effective but may not provide any added benefit.
Subgroup Effects
Although studies frequently fail to report the age of participants, data on the effects of physical activity on mental health are strongest for adults participating in high-intensity physical activity (Ahn and Fedewa, 2011). However, evidence relating physical activity to various measures of mental health has shown consistent, significant effects on individuals aged 11-20. A large prospective study found that physical activity was inversely associated with depression in early adolescence (Hasselstrom et al., 2002; Hallal
et al., 2006); fewer studies have been conducted among younger children. Correlation studies have shown that the association of physical activity with depression is not affected by age (Ahn and Fedewa, 2011).
Few studies have examined the influence of other sociodemographic characteristics of participants on the relationship between physical activity and mental health (Ahn and Fedewa, 2011), but studies have been conducted in populations with diverse characteristics. One study of low-income Hispanic children randomized to an aerobic intensity program found that the intervention group was less likely to present with depression but did not report reduced anxiety (Crews et al., 2004; Hallal et al., 2006). A study that included black and white children (aged 7-11) found that a 40-minute daily dose of aerobic exercise significantly reduced depressive symptoms and increased physical appearance self-worth in both black and white children and increased global self-worth in white children compared with controls (Petty et al., 2009). Physical activity also has been positively associated with mental health regardless of weight status (normal versus overweight) or gender (male versus female) (Petty et al., 2009; Ahn and Fedewa, 2011); however, results are stronger for males (Ahn and Fedewa, 2011).
Improvements in mental health as a result of physical activity may be more pronounced among clinically diagnosed populations, especially those with cognitive impairment or posttraumatic stress disorder (Craft and Landers, 1998; Ahn and Fedewa, 2011; Biddle and Asare, 2011). Evidence is less clear for youth with clinical depression (Craft and Landers, 1998; Larun et al., 2006; Biddle and Asare, 2011). Individuals diagnosed with major depression undergoing an intervention entailing aerobic exercise have shown significant improvement in depression and lower relapse rates, comparable to results seen in participants receiving psychotropic treatment (Babyak et al., 2000; Penedo and Dahn, 2005). One program for adults with Down syndrome providing three sessions of exercise and health education per week for 12 weeks resulted in more positive expectations, fewer emotional barriers, and improved life satisfaction (Heller et al., 2004; Penedo and Dahn, 2005). Ahn and Fedewa (2011) found that, compared with nondiagnosed individuals, physical activity had a fivefold greater impact on those diagnosed with cognitive impairment and a twofold greater effect on those diagnosed with emotional disturbance, suggesting that physical activity has the potential to improve the mental health of those most in need.
In sum, although more studies are needed, and there may be some differences in the magnitude and nature of the mental health benefits derived, it appears that physical activity is effective in improving mental health regardless of age, ethnicity, gender, or mental health status.
Sedentary Behavior
Sedentary behavior also influences mental health. Screen viewing in particular and sitting in general are consistently associated with poorer mental health (Biddle and Asare, 2011). Children who watch more television have higher rates of anxiety, depression, and posttraumatic stress and are at higher risk for sleep disturbances and attention problems (Kappos, 2007). Given the cross-sectional nature of these studies, however, the direction of these associations cannot be determined. A single longitudinal study found that television viewing, but not playing computer games, increased the odds of depression after 7-year follow-up (Primack et al., 2009; Biddle and Asare, 2011), suggesting that television viewing may contribute to depression. Because of design limitations of the available studies, it is unclear whether this effect is mediated by physical activity.
Television viewing also is associated with violence, aggressive behaviors, early sexual activity, and substance abuse (Kappos, 2007). These relationships are likely due to the content of the programming and advertising as opposed to the sedentary nature of the activity. Television viewing may affect creativity and involvement in community activities as well; however, the evidence here is very limited (Kappos, 2007). Studies with experimental designs are needed to establish a causal relationship between sedentary behavior and mental health outcomes (Kappos, 2007).
Although the available evidence is not definitive, it does suggest that sedentary activity and television viewing in particular can increase the risk for depression, anxiety, aggression, and other risky behaviors and may also affect cognition and creativity (Kappos, 2007), all of which can affect academic performance. It would therefore appear prudent for schools to reduce these sedentary behaviors during school hours and provide programming that has been shown to be effective in reducing television viewing outside of school (Robinson, 1999; Robinson and Borzekowski, 2006).
Mechanisms
It is not surprising that physical activity improves mental health. Both physiological and psychological mechanisms explain the observed associations. Physiologically, physical activity is known to increase the synaptic transmission of monoamines, an effect similar to that of anti-depressive drugs. Physical activity also stimulates the release of endorphins (endogenous opoids) (Peluso and Guerra de Andrade, 2005), which have an inhibitory effect on the central nervous system, creating a sense of calm and improved mood (Peluso and Guerra de Andrade, 2005; Ahn and Fedewa, 2011). Withdrawal of physical activity may result in irritability, restlessness, nervousness, and frustration as a result of a drop in endorphin
levels. Although more studies are needed to specify the exact neurological pathways that mediate this relationship, it appears that the favorable impact of physical activity on the prevention and treatment of depression may be the result of adaptations in the central nervous system mediated in part by neurotropic factors that facilitate neurogenerative, neuroadaptive, and neuroprotective processes (Dishman et al., 2006). It has been observed, for example, that chronic wheel running in rats results in immunological, neural, and cellular responses that mitigate several harmful consequences of acute exposure to stress (Dishman et al., 2006). A recent study found that children who were more physically active produced less cortisol in response to stress, suggesting that physical activity promotes mental health by regulating the hormonal responses to stress (Martikainen et al., 2013).
Psychological mechanisms that may explain why physical activity improves mental health include (1) distraction from unfavorable stimuli, (2) increase in self-efficacy, and (3) positive social interactions that can result from quality physical activity programming (Peluso and de Andrade, 2005) (see also the discussion of psychosocial health above). The relative contribution of physiological and psychological mechanisms is unknown, but they likely interact. Poor physical health also can impair mood and mental function. Health-related quality of life improves with physical activity that increases physical functioning, thereby enhancing the sense of well-being (McAuley and Rudolph, 1995; HHS, 2008).
Physical activity during childhood and adolescence may not only be important for its immediate benefits for mental health but also have implications for long-term mental health. Studies have shown a consistent effect of physical activity during adolescence on adult physical activity (Hallal et al., 2006). Physical activity habits established in children may persist into adulthood, thereby continuing to confer mental health benefits throughout the life cycle. Furthermore, physical activity in childhood may impact adult mental health regardless of the activity’s persistence (Hallal et al., 2006).
Summary
Physical activity can improve mental health by decreasing and preventing conditions such as anxiety and depression, as well as improving mood and other aspects of well-being. Evidence suggests that the mental health benefits of physical activity can be experienced by all age groups, genders, and ethnicities. Moderate effect sizes have been observed among both youth and adults. Youth with the highest risk of mental illness may experience the most benefit. Although evidence is not adequate to determine the ideal regimen, aerobic and high-intensity physical activity are likely to confer the most benefit. It appears, moreover, that a variety of types of physical activity are effective in improving different aspects of mental health; therefore,
a varied regimen including both aerobic activities and strength training may be the most effective. Frequent episodes of physical activity are optimal given the well-substantiated short-term effects of physical activity on mental health status. Although there are well-substantiated physiological bases for the impact of physical activity on mental health, physical activity programming that effectively enhances social interactions and self-efficacy also may improve mental health through these mechanisms. Quality physical activity programming also is critical to attract and engage youth of all skills level and to effectively reach those at highest risk.
Sedentary activity may increase the risk of poor mental health status independently of, or in addition to, its effect on physical activity. Television viewing in particular may lead to a higher risk of such conditions as depression and anxiety and may also increase violence, aggression, and other high-risk behaviors. These impacts are likely the result of programming and advertising content in addition to the physiological effects of inactivity and electronic stimuli.
In conclusion, frequently scheduled and well-designed opportunities for varied physical activity during the school day and a reduction in sedentary activity have the potential to improve students’ mental health in ways that could improve their academic performance and behaviors in school.
Good health is the foundation of learning and academic performance (see Chapter 4). In children and youth, health is akin to growth. An extensive literature demonstrates that regular physical activity promotes growth and development and has multiple benefits for physical, mental, cognitive, and psychosocial health that undoubtedly contribute to learning. Although much of the evidence comes from cross-sectional studies showing associations between physical activity and various aspects of health, available prospective data support this cross-sectional evidence. Experimental evidence, although more limited for younger children, is sufficient among older children and adolescents to support the notion that children and young adults derive much the same health benefits from physical activity.
Moreover, many adult diseases have their origins in childhood. This finding, together with the finding that health-related behaviors and disease risk factors may track from childhood into adulthood, underscores the need for early and ongoing opportunities for physical activity.
Children’s exercise capacity and the activities in which they can successfully engage change in a predictable way across developmental periods. For example, young children are active in short bursts, and their capacity for continuous activity increases as they grow and mature (see Figure 3-2). In adults and likely also adolescents, intermittent exercise has much the same
FIGURE 3-2 Changes in physical activity needs with increasing age of children and adolescents.
SOURCE: Adapted from Malina, 1991. Reprinted with permission from Human Kinetics Publishers.
benefit as continuous exercise when mode and energy expenditure are held constant. The health benefits of sporadic physical activity at younger ages are not well established. However, the well-documented short-term benefits of physical activity for some aspects of mental and cognitive health suggest that maximum benefit may be attained through frequent bouts of exercise throughout the day.
Children require frequent opportunities for practice to develop the skills and confidence that promote ongoing engagement in physical activity. Physical education curricula are structured to provide developmentally appropriate experiences that build the motor skills and self-efficacy that underlie lifelong participation in health-enhancing physical activity, and trained physical education specialists are uniquely qualified to deliver them (see Chapter 5). However, physical education usually is offered during a single session. Therefore, other opportunities for physical activity can supplement physical education by addressing the need for more frequent exercise during the day (see Chapter 6). In addition to the immediate benefits of short bouts of physical activity for learning and for mental health, developmentally appropriate physical activity during those times, along with the recommended time in physical education, can contribute to daily
energy expenditure and help lessen the risk of excess weight gain and its comorbidities. Specific types of activities address specific health concerns. For example, vertical jumping activities contribute to energy expenditure for obesity prevention and also promote bone development (via the resulting ground reaction forces), potentially contributing to lower fracture risk. Other activities contribute to prevention of chronic disease. Since different types of physical activity contribute to distinct aspects of physical, mental, and psychosocial health, a varied regimen is likely to be most beneficial overall.
The quality of physical activity programming also is critical; psychosocial outcomes and improvements in specific motor skills, for example, are likely the result of programming designed specifically to target these outcomes rather than just a result of increases in physical activity per se. These psychosocial outcomes also are likely to lead to increased levels of physical activity in both the short and long terms, thereby conferring greater health benefits. Unstructured physical activity or free play also confers unique benefits and is an important supplement to more structured opportunities. Quality physical activity programming that makes these activities attractive, accessible, and safe for children and youth of all skill and fitness levels is critical to ensure that all youth participate in these activities and can therefore derive the health benefits.
Sedentary activities, such as screen viewing and excessive time spent sitting, may contribute to health risks both because of and independent of their impact on physical activity. Thus specific efforts in school to reduce sedentary behaviors, such as through classroom and playground design and reduction of television viewing, are warranted.
In sum, a comprehensive physical activity plan with physical education at the core, supplemented by other varied opportunities for and an environment supportive of physical activity throughout the day, would make an important contribution to children’s health and development, thereby enhancing their readiness to learn.
Ahlqwist, A., M. Hagman, G. Kjellby-Wendt, and E. Beckung. 2008. Physical therapy treatment of back complaints on children and adolescents. Spine 33(20):E721-E727.
Ahn, S., and A. L. Fedewa. 2011. A meta-analysis of the relationship between children’s physical activity and mental health. Journal of Pediatric Psychology 36(4):385-397.
Ailhaud, G., and H. Hauner. 1998. Development of white adipose tissue. In Handbook of Obesity, edited by G. A. Bray and W. P. T. James. New York: Marcel Dekker. Pp. 359-378.
Alberti, K., and P. Zimmet. 1998. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus. Provisional report of a WHO consultation. Diabetic Medicine 15(7):539-553.
Allender, S., G. Cowburn, G., and C. Foster. 2006. Understanding participation in sport and physical activity among children and adults: A review of qualitative studies. Health Education Research 21(6):826-835.
Amis, J. M., P. M. Wright, B. Dyson, J. M. Vardaman, and H. Ferry. 2012. Implementing Childhood Obesity Policy in a New Educational Environment: The Cases of Mississippi and Tennessee. American Journal of Public Health 102(7):1406-1413.
Annesi, J. J., W. L. Westcott, A. D. Faigenbaum, and J. L. Unruh. 2005. Effects of a 12-week physical activity protocol delivered by YMCA after-school counselors (Youth Fit for Life) on fitness and self-efficacy changes in 5-12-year-old boys and girls. Research Quarterly for Exercise and Sport 76(4):468-476.
Arlot, M. E., E. Sornay-Rendu, P. Garnero, B. Vey-Marty, and P. D. Delmas. 1997. Apparent pre- and postmenopausal bone loss evaluated by DXA at different skeletal sites in women: The OFELY cohort. Journal of Bone and Mineral Research 12(4):683-690.
Arner, P., and R. H. Eckel. 1998. Adipose tissue as a storage organ. In Handbook of obesity, edited by G. A. Bray, and W. P. T. James. New York: Marcel Dekker. Pp. 379-396.
Babyak, M., J. A. Blumenthal, S. Herman, P. Khatri, M. Doraiswamy, K. Moore, W. E. Craighead, T. T. Baldewicz, and K. R. Krishnan. 2000. Exercise treatment for major depression: Maintenance of therapeutic benefit at 10 months. Psychosomatic Medicine 62(5):633-638.
Bailey, D. A., R. A. Faulkner, and H. A. McKay. 1996. Growth, physical activity, and bone mineral acquisition. Exercise and Sport Science Reviews 24(1):233-266.
Bailey, D. A., A. D. Martin, H. A. McKay, S. Whiting, and R. Mirwald. 2000. Calcium accretion in girls and boys during puberty: A longitudinal analysis. Journal of Bone and Mineral Research 15(11):2245-2250.
Bailey, R. 2006. Physical education and sport in schools: A review of benefits and outcomes. Journal of School Health 76(8):397-401.
Balagopal, P., D. George, N. Patton, H. Yarandi, W. L. Roberts, E. Bayne, and S. Gidding. 2005. Lifestyle-only intervention attenuates the inflammatory state associated with obesity: A randomized controlled study in adolescents. Journal of Pediatrics 146(3):342-348.
Ballor, D., E. Poehlman, and M. Toth. 1998. Exercise as a treatment for obesity. Handbook of obesity. Edited by G. A. Bray, C. Bouchard, and W. P. T. James. New York: Marcel Dekker. Pp. 891-910.
Bandura, A. 1995. Self-efficacy in changing societies. New York: Cambridge University Press.
Bandura, A., and D. C. McClelland. 1977. Social learning theory. Englewood Cliffs, NJ: Prentice-Hall.
Bao, W., S. R. Srinivasan, W. A. Wattigney, and G. S. Berenson. 1994. Persistence of multiple cardiovascular risk clustering related to syndrome X from childhood to young adulthood: The Bogalusa Heart Study. Archives of Internal Medicine 154(16):1842.
Baquet, G., S. Berthoin, G. Dupont, N. Blondel, C. Fabre, and E. Van Praagh. 2002. Effects of high intensity intermittent training on peak VO(2) in prepubertal children. International Journal of Sports Medicine 23(6):439.
Baquet, G., E. Van Praagh, and S. Berthoin. 2003. Endurance training and aerobic fitness in young people. Sports Medicine 33(15):1127-1143.
Barbeau, P., M. H. Johnson, C. A. Howe, J. Allison, C. L. Davis, B. Gutin, and C. R. Lemmon. 2007. Ten months of exercise improves general and visceral adiposity, bone, and fitness in black girls. Obesity 15(8):2077-2085.
Barlow, S. 2007. Expert committee recommendations regarding the prevention, assessment, and treatment of child and adolescent overweight and obesity: Summary report. Pediatrics 120(S4):S164-S192.
Barnett, L. M., E. Van Beurden, P. J. Morgan, L. O. Brooks, and J. R. Beard. 2009. Childhood motor skill proficiency as a predictor of adolescent physical activity. Journal of Adolescent Health 44(3):252-259.
Bar-Or, O. 1983. Pediatric sports medicine for the practitioner. New York: Springer-Verlag.
Barr-Anderson, D. J., D. Neumark-Sztainer, K. H. Schmitz, D. S. Ward, T. L. Conway, C. Pratt, C. D. Baggett, L. Lytle, and R. R. Pate. 2008. But I like PE: Factors associated with enjoyment of physical education class in middle school girls. Research Quarterly for Exercise and Sport 79(1):18-27.
Barros, R. M., E. J. Silver, and R. E. K. Stein. 2009. School recess and group classroom behavior. Pediatrics 123(2):431-436.
Bass, S., G. Pearce, M. Bradney, E. Hendrich, P. D. Delmas, A. Harding, and E. Seeman. 1998. Exercise before puberty may confer residual benefits in bone density in adulthood: Studies in active prepubertal and retired female gymnasts. Journal of Bone and Mineral Research 13(3):500-507.
Bau, A.-M., A. Ernert, L. Schenk, S. Wiegand, P. Martus, A. Grüters, and H. Krude. 2009. Is there a further acceleration in the age at onset of menarche? A cross-sectional study in 1840 school children focusing on age and bodyweight at the onset of menarche. European Journal of Endocrinology 160(1):107-113.
Bauman, A. E. 2004. Updating the evidence that physical activity is good for health: An epidemiological review 2000-2003. Journal of Science and Medicine in Sport 7(1):6-19.
Bauman, A. E., R. S. Reis, J. F. Sallis, J. C. Wells, R. J. Loos, B. W. Martin. Lancet Physical Activity Series Working Group. 2012. Correlates of physical activity: Why are some people physically active and others not? Lancet 380(9838):258-271.
Behringer, M., A. vom Heede, Z. Yue, and J. Mester. 2010. Effects of resistance training in children and adolescents: A meta-analysis. Pediatrics 126(5):e1199-e1210.
Bell, L. M., K. Watts, A. Siafarikas, A. Thompson, N. Ratnam, M. Bulsara, J. Finn, G. O’Driscoll, D. J. Green, and T. W. Jones. 2007. Exercise alone reduces insulin resistance in obese children independently of changes in body composition. Journal of Clinical Endocrinology & Metabolism 92(11):4230-4235.
Benson, A. C., M. E. Torode, and M. A. Fiatarone Singh. 2007. A rationale and method for high-intensity progressive resistance training with children and adolescents. Contemporary Clinical Trials 28(4):442-450.
Benson, A., M. Torode, and M. Fiatarone Singh. 2008. Effects of resistance training on metabolic fitness in children and adolescents: A systematic review. Obesity Reviews 9(1):43-66.
Berenson, G. S., S. R. Srinivasan, W. Bao, W. P. Newman, R. E. Tracy, and W. A. Wattigney. 1998. Association between multiple cardiovascular risk factors and atherosclerosis in children and young adults. New England Journal of Medicine 338(23):1650-1656.
Berkey, C. S., H. R. Rockett, A. E. Field, M. W. Gillman, A. L. Frazier, C. A. Camargo, and G. A. Colditz. 2000. Activity, dietary intake, and weight changes in a longitudinal study of preadolescent and adolescent boys and girls. Pediatrics 105(4):e56.
Berkey, C. S., H. R. Rockett, M. W. Gillman, and G. A. Colditz. 2003. One-year changes in activity and in inactivity among 10- to 15-year-old boys and girls: Relationship to change in body mass index. Pediatrics 111(4):836-843.
Bernhardt, D., J. Gomez, M. Johnson, T. Martin, T. Rowland, E. Small, C. LeBlanc, R. Malina, C. Krein, and J. Young. 2001. Strength training by children and adolescents. Pediatrics 107(6):1470.
Beunen, G. P., R. M. Malina, R. Renson, J. Simons, M. Ostyn, and J. Lefevre. 1992. Physical activity and growth, maturation and performance: A longitudinal study. Medicine and Science in Sports and Exercise 24(5):576.
Biddle, S. J., and M. Asare. 2011. Physical activity and mental health in children and adolescents: A review of reviews. British Journal of Sports Medicine 45(11):886-895.
Biddle, S., and N. Mutrie. 2008. Psychology of physical activity: Determinants, well-being and interventions, 2nd ed. London: Routledge.
Bloomfield, S. A., K. D. Little, M. E. Nelson, and V. R. Yingling. 2004. American College of Sports Medicine® position stand. Medicine & Science in Sports & Exercise 195(9131/04):1985.
Bonnet, F. P., and D. Rocour-Brumioul. 1981. Normal growth of human adipose tissue. In Adipose tissue in childhood, edited by F. Bonnet. Boca Raton, FL: CRC Press. Pp. 81-107.
Boreham, C., and C. Riddoch. 2001. The physical activity, fitness and health of children. Journal of Sports Sciences 19(12):915-929.
Boreham, C. A., I. Ferreira, J. W. Twisk, A. M. Gallagher, M. J. Savage, and L. J. Murray. 2004. Cardiorespiratory fitness, physical activity, and arterial stiffness: The Northern Ireland Young Hearts Project. Hypertension 44(5):721-726.
Bouchard, C. 2012. Genomic predictors of trainability. Experimental Physiology 97(3): 347-352.
Bouffard, M. E., E. J. Watkinson, L. P. Thompson, J. L. C. Dunn, and S. K. E. Romanow. 1996. A test of the activity deficit hypothesis with children with movement difficulties. Adapted Physical Activity Quarterly 13:61-73.
Brambilla, P., G. Pozzobon, and A. Pietrobelli. 2010. Physical activity as the main therapeutic tool for metabolic syndrome in childhood. International Journal of Obesity 35(1):16-28.
Brosnahan, J., L. M. Steffen, L. Lytle, J. Patterson, and A. Boostrom. 2004. The relation between physical activity and mental health among Hispanic and non-Hispanic white adolescents. Archives of Pediatrics and Adolescent Medicine 158(8):818-823.
Brown, W. H., K. A. Pfeiffer, K. L. McIver, M. Dowda, C. L. Addy, and R. R. Pate. 2009. Social and environmental factors associated with preschoolers’ nonsedentary physical activity. Child Development 80(1):45-58.
Butt, J., R. S. Weinberg, J. D. Breckon, and R. P. Claytor. 2011. Adolescent physical activity participation and motivational determinants across gender, age, and race. Journal of Physical Activity and Health 8(8):1074-1083.
Byrd, C. E., and S. M. Ross. 1991. The influence of participation in junior high athletics on students’ attitudes and grades. Physical Educator 48(4):170-176.
Byun, W., M. Dowda, and R. R. Pate. 2011. Correlates of objectively measured sedentary behavior in US preschool children. Pediatrics 128(5):937-945.
Campbell, P. T., P. T. Katzmarzyk, R. M. Malina, D. Rao, L. Pérusse, and C. Bouchard. 2012. Stability of adiposity phenotypes from childhood and adolescence into young adulthood with contribution of parental measures. Obesity Research 9(7):394-400.
Carnethon, M. R., M. Gulati, and P. Greenland. 2005. Prevalence and cardiovascular disease correlates of low cardiorespiratory fitness in adolescents and adults. Journal of the American Medical Association 294(23):2981-2988.
Carrel, A. L., R. R. Clark, S. E. Peterson, B. A. Nemeth, J. Sullivan, and D. B. Allen. 2005. Improvement of fitness, body composition, and insulin sensitivity in overweight children in a school-based exercise program: A randomized, controlled study. Archives of Pediatrics and Adolescent Medicine 159(10):963.
Caspersen, C. J., K. E. Powell, and G. M. Christenson. 1985. Physical activity, exercise, and physical fitness: Definitions and distinctions for health-related research. Public Health Reports 100(2):126.
CDC (Centers for Disease Control and Prevention). 2006. Overweight and obesity: Frequently asked questions. http://www.cdc.gov/obesity/index.html (accessed November 27, 2012).
CDC. 2012. Youth risk behavior surveillance: United States, 2011. Morbidity and Mortality Weekly Report 61(4):1-162.
Charvat, J. 2012. Research on the relationship between mental health and academic achievement. Bethesda, MD: National Association of School Psychologists.
Christodoulos, A. D., H. T. Douda, and S. P. Tokmakidis. 2012. Cardiorespiratory fitness, metabolic risk, and inflammation in children. International Journal of Pediatrics [epub ahead of print].
Chumlea, W., R. Siervogel, A. Roche, D. Mukherjee, and P. Webb. 1982. Changes in adipocyte cellularity in children ten to 18 years of age. International Journal of Obesity 6(4):383-389.
Church, T., C. Barlow, C. P. Earnest, J. Kampert, E. Priest, and S. Blair. 2002. Associations between cardiorespiratory fitness and C-reactive protein in men. Arteriosclerosis, Thrombosis, and Vascular Biology 22(11):1869-1876.
Clapp, J., III, and K. D. Little. 1995. Effect of recreational exercise on pregnancy weight gain and subcutaneous fat deposition. Medicine and Science in Sports and Exercise 27(2):170.
Clark, E. M, J. H. Tobias, L. Murray, and C. Boreham. 2011. Children with low muscle strength are at an increased risk of fracture with exposure to exercise. Journal of Musculoskeletal and Neuronal Interactions 11(2):196-202.
Clark, J. E. 2005. From the beginning: A developmental perspective on movement and mobility. Quest 57:37-45.
Clark, J. E., and J.S. Metcalfe. 2002. The mountain of motor development: A metaphor. In Motor development: Research and review, vol. 2, edited by E. Clark and J. H. Humphrey. Reston, VA: National Association for Sport and Physical Education. Pp. 62-95.
Cliff, D., A. Okely, L. Smith, and K. McKeen. 2009. Relationships between fundamental movement skills and objectively measured physical activity in preschool children. Pediatric Exercise Science 21(4):436.
Committee on Sports Medicine Fitness. 2001. Strength training by children and adolescents. Pediatrics 107(6):1470-1472.
Cook, S., M. Weitzman, P. Auinger, M. Nguyen, and W. H. Dietz. 2003. Prevalence of
a metabolic syndrome phenotype in adolescents: Findings from the Third National
Health and Nutrition Examination Survey, 1988-1994. Archives of Pediatrics and
Adolescent Medicine 157(8):821. Cook, S., P. Auinger, and T. T.-K. Huang. 2009. Growth curves for cardio-metabolic risk factors in children and adolescents. Journal of Pediatrics 155(3):S6.
Council on Sports Medicine Fitness. 2008. Strength training by children and adolescents. Pediatrics 121(4):835-840.
Cox, A. E., A. L. Smith, and L. Williams. 2008. Change in physical education motivation and physical activity behavior during middle school. Journal of Adolescent Health 43(5):506-513.
Cradock, A. L., I. Kawachi, G. A. Colditz, S. L. Gortmaker, and S. L. Buka. 2009. Neighborhood social cohesion and youth participation in physical activity in Chicago. Social Science and Medicine 68(3):427-435.
Craft, L. L., and D. M. Landers. 1998. The effect of exercise on clinical depression and depression resulting from mental illness: A meta-analysis. Journal of Sport and Exercise Psychology 20:339-357.
Crews, D. J., M. R. Lochbaum, and D. M. Landers. 2004. Aerobic physical activity effects on psychological well-being in low-income Hispanic children. Perceptual and Motor Skills 98(1):319-324.
Danforth, J. S., K. D. Allen, J. M. Fitterling, J. A. Danforth, D. Farrar, M. Brown, and R. S. Drabman. 1990. Exercise as a treatment for hypertension in low-socioeconomic-status black children. Journal of Consulting and Clinical Psychology 58(2):237.
Daniels, S. R., J. A. Morrison, D. L. Sprecher, P. Khoury, and T. R. Kimball. 1999. Association of body fat distribution and cardiovascular risk factors in children and adolescents. Circulation 99(4):541-545.
Daniels, S. R., D. K. Arnett, R. H. Eckel, S. S. Gidding, L. L. Hayman, S. Kumanyika, T. N. Robinson, B. J. Scott, S. S. Jeor, and C. L. Williams. 2005. Overweight in children and adolescents: Pathophysiology, consequences, prevention, and treatment. Circulation 111(15):1999-2012.
Davies, P. S., J. Gregory, and A. White. 1995. Physical activity and body fatness in preschool children. International Journal of Obesity and Related Metabolic Disorders 19(1):6.
Davis, C. L., N. K. Pollock, J. L. Waller, J. D. Allison, B. A. Dennis, R. Bassali, A. Meléndez, C. A. Boyle, and B. A. Gower. 2012. Exercise dose and diabetes risk in overweight and obese children: A randomized controlled trial. Journal of the American Medical Association 308(11):1103-1112.
Davis, P. H., J. D. Dawson, W. A. Riley, and R. M. Lauer. 2001. Carotid intimal-medial thickness is related to cardiovascular risk factors measured from childhood through middle age: The Muscatine study. Circulation 104(23):2815-2819.
de la Haye, K., G. Robins, P. Mohr, and C. Wilson. 2011. How physical activity shapes, and is shaped by, adolescent friendships. Social Science and Medicine 73(5):719-728.
Dencker, M., P. Wollmer, M. K. Karlsson, C. Lindén, L. B. Andersen, and O. Thorsson. 2012. Body fat, abdominal fat and body fat distribution related to cardiovascular risk factors in prepubertal children. Acta Paediatrica 101(8):852-857.
Depres, J. P., and B. Lamarche. 2000. Physical activity and the metabolic complications of obesity. In Physical activity and obesity, edited by C. Bouchard. Champaign, IL: Human Kinetics. Pp. 331-354.
Diabetes Prevention Program Research Group. 2002. The Diabetes Prevention Program (DPP): Description of lifestyle intervention. Diabetes Care 25(12):2165-2171.
Digelidis, N., A. Papaioannou, K. Laparidis, and T. Christodoulidis. 2003. A one-year intervention in 7th grade physical education classes aiming to change motivational climate and attitudes towards exercise. Psychology of Sport and Exercise 4(3):195-210.
Dimeo, F., M. Bauer, I. Varahram, G. Proest, and U. Halter. 2001. Benefits from aerobic exercise in patients with major depression: A pilot study. British Journal of Sports Medicine 35(2):114-117.
Dintiman, G., B. Ward, and T. Telez. 1997. Sports speed. Champaign, IL: Human Kinetics.
Dionne, I., N. Almeras, C. Bouchard, and A. Tremblay. 2000. The association between vigorous physical activities and fat deposition in male adolescents. Medicine and Science in Sports and Exercise 32:392-395.
Dishman, R. K., R. W. Motl, R. Saunders, G. Felton, D. S. Ward, M. Dowda, and R. R. Pate. 2005. Enjoyment mediates effects of a school-based physical-activity intervention. Medicine and Science in Sports and Exercise 37(3):478-487.
Dishman, R. K., D. P. Hales, K. A. Pfeiffer, G. A. Felton, R. Saunders, D. S. Ward, M. Dowda, and R. R. Pate. 2006. Physical self-concept and self-esteem mediate crosssectional
relations of physical activity and sport participation with depression symptoms among adolescent girls. Health Psychology 25(3):396-407.
Dishman, R. K., R. P. Saunders, R. W. Motl, M. Dowda, and R. R. Pate. 2009. Self-efficacy moderates the relation between declines in physical activity and perceived social support in high school girls. Journal of Pediatric Psychology 34(4):441-451.
Dishman, R. K., A. L. Dunn, J. F. Sallis, R. J. Vandenberg, and C. A. Pratt. 2010. Social-cognitive correlates of physical activity in a multi-ethnic cohort of middle-school girls: Two-year prospective study. Journal of Pediatric Psychology 35(2):188-198.
Dua, J., and L. Hargreaves. 1992. Effect of aerobic exercise on negative affect, positive affect, stress, and depression. Perceptual and Motor Skills 75(2):355-361.
Dubose, K. D., J. C. Eisenmann, and J. E. Donnelly. 2007. Aerobic fitness attenuates the metabolic syndrome score in normal-weight, at-risk-for-overweight, and overweight children. Pediatrics 120(5):e1262-e1268.
Duncan, G. E., M. G. Perri, D. W. Theriaque, A. D. Hutson, R. H. Eckel, and P. W. Stacpoole. 2003. Exercise training, without weight loss, increases insulin sensitivity and postheparin plasma lipase activity in previously sedentary adults. Diabetes Care 26(3):557-562.
Duncan, S. C., T. E. Duncan, L. A. Strycker, and N. R. Chaumeton. 2007. A cohort-sequential latent growth model of physical activity from ages 12 to 17 years. Annals of Behavioral Medicine 33(1):80-89.
Dunn, A. L., M. H. Trivedi, and H. A. O’Neal. 2001. Physical activity dose-response effects on outcomes of depression and anxiety. Medicine and Science in Sports and Exercise 33(6 Suppl):S587-S597.
Eisenmann, J. C. 2003. Secular trends in variables associated with the metabolic syndrome of North American children and adolescents: A review and synthesis. American Journal of Human Biology 15(6):786-794.
Eisenmann, J. C. 2007. Aerobic fitness, fatness and the metabolic syndrome in children and adolescents. Acta Paediatrica 96(12):1723-1729.
Eisenmann, J., P. Katzmarzyk, L. Perusse, A. Tremblay, J. Despres, and C. Bouchard. 2005. Aerobic fitness, body mass index, and CVD risk factors among adolescents: The Quebec Family Study. International Journal of Obesity 29(9):1077-1083.
Eisenmann, J. C., G. J. Welk, E. E. Wickel, and S. N. Blair. 2007a. Combined influence of cardiorespiratory fitness and body mass index on cardiovascular disease risk factors among 8-18 year old youth: The Aerobics Center Longitudinal Study. International Journal of Pediatric Obesity 2(2):66-72.
Eisenmann, J. C., G. J. Welk, M. Ihmels, and J. Dollman. 2007b. Fatness, fitness, and cardiovascular disease risk factors in children and adolescents. Medicine and Science in Sports and Exercise 39(8):1251.
Eisenmann, J. C., K. R. Laurson, and G. J. Welk. 2011. Aerobic fitness percentiles for US adolescents. American Journal of Preventive Medicine 41(4):S106-S110.
Ekelund, U., S. Brage, P. W. Franks, S. Hennings, S. Emms, and N. J. Wareham. 2005. Physical activity energy expenditure predicts progression toward the metabolic syndrome independently of aerobic fitness in middle-aged healthy Caucasians the Medical Research Council Ely Study. Diabetes Care 28(5):1195-1200.
Ekelund, U., P. W. Franks, S. Sharp, S. Brage, and N. J. Wareham. 2007. Increase in physical activity energy expenditure is associated with reduced metabolic risk independent of change in fatness and fitness. Diabetes Care 30(8):2101-2106.
Elder, J. P., S. L. Broyles, T. L. McKenzie, J. F. Sallis, C. C. Berry, T. B. Davis, P. L. Hoy, and P. R. Nader. 1998. Direct home observations of the prompting of physical activity in sedentary and active Mexican- and Anglo-American children. Journal of Developmental and Behavioral Pediatrics 19(1):26-30.
Ertek, S., and A. Cicero. 2012. Impact of physical activity on inflammation: Effects on cardiovascular disease risk and other inflammatory conditions. Archives of Medical Science 8(5):794.
Escalante, Y., J. M. Saavedra, A. García-Hermoso, and A. M. Domínguez. 2012. Improvement of the lipid profile with exercise in obese children: A systematic review. Preventive Medicine 54(5):293-301.
Ewart, C. K., D. R. Young, and J. M. Hagberg. 1998. Effects of school-based aerobic exercise on blood pressure in adolescent girls at risk for hypertension. American Journal of Public Health 88(6):949-951.
Faigenbaum, A. D. 2007. State of the art reviews: Resistance training for children and adolescents: Are there health outcomes? American Journal of Lifestyle Medicine 1(3):190-200.
Faigenbaum, A., L. Zaichkowsky, W. Westcott, C. Long, R. LaRosa-Loud, L. Micheli, and A. Outerbridge. 1997. Psychological effects of strength training on children. Journal of Sport Behavior 20(2):164-175.
Faigenbaum, A. D., W. J. Kraemer, C. J. Blimkie, I. Jeffreys, L. J. Micheli, M. Nitka, and T. W. Rowland. 2009. Youth resistance training: Updated position statement paper from the National Strength and Conditioning Association. Journal of Strength and Conditioning Research 23(5 Suppl):S60-S79.
Farpour-Lambert, N. J., Y. Aggoun, L. M. Marchand, X. E. Martin, F. R. Herrmann, and M. Beghetti. 2009. Physical activity reduces systemic blood pressure and improves early markers of atherosclerosis in pre-pubertal obese children. Journal of the American College of Cardiology 54(25):2396-2406.
Farr, J. N., V. R. Lee, R. M. Blew, T. G. Lohman, and S. B. Going. 2011. Quantifying bone-relevant activity and its relation to bone strength in girls. Medicine and Science in Sports and Exercise 43(3):476.
Ferguson, M., B. Gutin, N. Le, W. Karp, M. Litaker, M. Humphries, T. Okuyama, S. Riggs, and S. Owens. 1999. Effects of exercise training and its cessation on components of the insulin resistance syndrome in obese children. International Journal of Obesity and Related Metabolic Disorders 23(8):889.
Ferrari, S. L., T. Chevalley, J. P. Bonjour, and R. Rizzoli. 2006. Childhood fractures are associated with decreased bone mass gain during puberty: An early marker of persistent bone fragility? Journal of Bone and Mineral Research 21(4):501-507.
Ferrer-Caja, E., and M. R. Weiss. 2000. Predictors of intrinsic motivation among adolescent students in physical education. Research Quarterly for Exercise and Sport 71(3):267-279.
Fisher, A., J. J. Reilly, L. A. Kelly, C. Montgomery, A. Williamson, J. Y. Paton, and S. Grant. 2005. Fundamental movement skills and habitual physical activity in young children. Medicine and Science in Sports and Exercise 37(4):684.
Fournier, M., J. Ricci, A. W. Taylor, R. J. Ferguson, R. R. Montpetit, and B. R. Chaitman. 1982. Skeletal muscle adaptation in adolescent boys: Sprint and endurance training and detraining. Medicine and Science in Sports and Exercise 14(6):453.
Freedman, D. S., L. Kettel Khan, M. Serdula, W. Dietz, S. Srinivasan, and G. Berenson. 2005. The relation of childhood BMI to adult adiposity: The Bogalusa Heart Study. Pediatrics 115(1):22-27.
Frühbeck, G., J. Gómez-Ambrosi, F. J. Muruzábal, and M. A. Burrell. 2001. The adipocyte: A model for integration of endocrine and metabolic signaling in energy metabolism regulation. American Journal of Physiology-Endocrinology and Metabolism 280(6):E827-E847.
Fuchs, R. K., J. J. Bauer, and C. M. Snow. 2001. Jumping improves hip and lumbar spine bone mass in prepubescent children: A randomized controlled trial. Journal of Bone and Mineral Research 16(1):148-156.
Fulton, J. E., C. R. Burgeson, G. R. Perry, B. Sherry, D. A. Galuska, M. P. Alexander, and C. J. Caspersen. 2001. Assessment of physical activity and sedentary behavior in preschool-age children: Priorities for research. Pediatric Exercise Science 13:113-126.
Gan, S. K., A. D. Kriketos, B. A. Ellis, C. H. Thompson, E. W. Kraegen, and D. J. Chisholm. 2003. Changes in aerobic capacity and visceral fat but not myocyte lipid levels predict increased insulin action after exercise in overweight and obese men. Diabetes Care 26(6):1706-1713.
Going, S. 1999. Body composition alterations with exercise. In Lifestyle medicine, edited by J. M. Rippe. Blackwell Science. Pp. 1089-1097.
Going, S. B., T. G. Lohman, E. C. Cussler, D. P. Williams, J. A. Morrison, and P. S. Horn. 2011. Percent body fat and chronic disease risk factors in US children and youth. American Journal of Preventive Medicine 41(4):S77-S86.
Going, S., M. Hingle, and J. Farr. 2012. Body composition. In Modern nutrition in health and disease, 11th ed., edited by A. C. Ross, B. Caballero, R. J. Cousins, K. L. Tucker, and T. R. Ziegler. Baltimore, MD: Lippincott, Williams & Wilkins. P. 1648.
Goodway, J. D., and M. E. Rudisill. 1997. Perceived physical competence and actual motor skill competence of African American preschool children. Adapted Physical Activity Quarterly 14:314-326.
Gortmaker, S. L., K. Peterson, J. Wiecha, A. M. Sobol, S. Dixit, M. K. Fox, and N. Laird. 1999. Reducing obesity via a school-based interdisciplinary intervention among youth: Planet health. Archives of Pediatrics and Adolescent Medicine 153(4):409-418.
Graf, C., B. Koch, S. Dordel, S. Schindler-Marlow, A. Icks, A. Schüller, B. Bjarnason-Wehrens, W. Tokarski, and H. G. Predel. 2004. Physical activity, leisure habits and obesity in first-grade children. European Journal of Cardiovascular Prevention and Rehabilitation 11(4):284-290.
Guo, S. S., A. F. Roche, W. C. Chumlea, J. D. Gardner, and R. M. Siervogel. 1994. The predictive value of childhood body mass index values for overweight at age 35 y. American Journal of Clinical Nutrition 59(4):810-819.
Guo, S., C. Huang, L. Maynard, E. Demerath, B. Towne, W. C. Chumlea, and R. Siervogel. 2000. Body mass index during childhood, adolescence and young adulthood in relation to adult overweight and adiposity: The Fels Longitudinal Study. International Journal of Obesity and Related Metabolic Disorders 24(12):1628.
Gutin, B., and M. Humphries. 1998. Exercise, body composition, and health in children. In Perspectives in exercise science and sports medicine. Vol. 11, edited by D. R. Lamb and R. Murray. Carmel, IN: Cooper Publishing Group. Pp. 295-347.
Gutin, B., and S. Owens. 1999. Role of exercise intervention in improving body fat distribution and risk profile in children. American Journal of Human Biology 11(2):237-247.
Gutin, B., and S. Owens. 2011. The influence of physical activity on cardiometabolic biomarkers in youths: A review. Pediatric Exercise Science 23(2):169.
Gutin, B., P. Barbeau, S. Owens, C. R. Lemmon, M. Bauman, J. Allison, H.-S. Kang, and M. S. Litaker. 2002. Effects of exercise intensity on cardiovascular fitness, total body composition, and visceral adiposity of obese adolescents. American Journal of Clinical Nutrition 75(5):818-826.
Hagberg, J. M., D. Goldring, A. A. Ehsani, G. W. Heath, A. Hernandez, K. Schechtman, and J. O. Holloszy. 1983. Effect of exercise training on the blood pressure and hemodynamic features of hypertensive adolescents. American Journal of Cardiology 52(7):763-768.
Hagberg, J. M., A. A. Ehsani, D. Goldring, A. Hernandez, D. R. Sinacore, and J. O. Holloszy. 1984. Effect of weight training on blood pressure and hemodynamics in hypertensive adolescents. Journal of Pediatrics 104(1):147-151.
Hager, A. 1981. Adipose tissue cellularity in childhood in relation to the development of obesity. British Medical Bulletin 37(3):287-290.
Halfon, N., P. A. Verhoef, and A. A. Kuo. 2012. Childhood antecedents to adult cardiovascular disease. Pediatrics in Review 33(2):51-61.
Hallal, P. C., C. G. Victora, M. R. Azevedo, and J. C. Wells. 2006. Adolescent physical activity and health: A systematic review. Sports Medicine 36(12):1019-1030.
Halle, M., U. Korsten-Reck, B. Wolfarth, and A. Berg. 2004. Low-grade systemic inflammation in overweight children: Impact of physical fitness. Exercise Immunology Review 10:66-74.
Hands, B., D. Larkin, H. Parker, L. Straker, and M. Perry. 2009. The relationship among physical activity, motor competence and health related fitness in 14-year-old adolescents. Scandinavian Journal of Medicine and Science in Sports 19(5):655-663.
Harter, S., and R. Pike. 1984. The pictorial scale of perceived competence and social acceptance for young children. Child Development 55(6):1969-1982.
Hasselstrom, H., S. Hansen, K. Froberg, and L. B. Andersen. 2002. Physical fitness and physical activity during adolescence as predictors of cardiovascular disease risk in young adulthood. Danish Youth and Sports Study. An eight-year follow-up study. International Journal of Sports Medicine 23(1):27.
Haubenstricker, J., and M. Sapp. 1980. A brief review of the Bruininks-Oseretsky test of motor proficiency. Reston, VA: National Association for Sport and Physical Education.
Haubenstricker, J., and V. Seefeldt. 1986. Acquisition of motor skills during childhood. In Physical activity and well-being, edited by V. Seefeldt. Reston, VA: American Alliance for Health, Physical Education, Recreation and Dance. Pp. 41-92.
Haugen, T., R. Säfvenbom, and Y. Ommundsen. 2011. Physical activity and global self-worth: The role of physical self-esteem indices and gender. Mental Health and Physical Activity 4(2):49-56.
He, Q., X. Zhang, S. He, L. Gong, Y. Sun, S. Heshka, R. J. Deckelbaum, and D. Gallagher. 2007. Higher insulin, triglycerides, and blood pressure with greater trunk fat in Tanner 1 Chinese. Obesity 15(4):1004-1011.
Heller, T., K. Hsieh, and J. H. Rimmer. 2004. Attitudinal and psychosocial outcomes of a fitness and health education program on adults with Down syndrome. American Journal on Mental Retardation 109(2):175-185.
HHS (U.S. Department of Health and Human Services). 1996. Physical activity and health: A report of the Surgeon General. Atlanta, GA: HHS, CDC, National Center for Chronic Disease Prevention and Health Promotion.
HHS. 2008. Physical activity guidelines for Americans. Washington, DC: HHS.
HHS. 2013. Physical activity guidelines for Americans midcourse report: Strategies to increase physical activity among youth. Washington, DC: HHS.
Hind, K., and M. Burrows. 2007. Weight-bearing exercise and bone mineral accrual in children and adolescents: A review of controlled trials. Bone 40(1):14-27.
Holloway, J. B., A. Beuter, and J. L. Duda. 1988. Self-efficacy and training for strength in adolescent girls. Journal of Applied Social Psychology 18(8):699-719.
Houwen, S., E. Hartman, and C. Visscher. 2009. Physical activity and motor skills in children with and without visual impairments. Medicine and Science in Sports and Exercise 41(1):103.
Huang, T. T.-K., T. R. Nansel, A. R. Belsheim, and J. A. Morrison. 2008. Sensitivity, specificity, and predictive values of pediatric metabolic syndrome components in rela-
tion to adult metabolic syndrome: The Princeton LRC follow-up study. Journal of Pediatrics 152(2):185-190.
Hume, C., A. Okely, S. Bagley, A. Telford, M. Booth, D. Crawford, and J. Salmon. 2008. Does weight status influence associations between children’s fundamental movement skills and physical activity? Research Quarterly for Exercise and Sport 79(2):158-166.
Hussey, J., C. Bell, K. Bennett, J. O’Dwyer, and J. Gormley. 2007. Relationship between the intensity of physical activity, inactivity, cardiorespiratory fitness and body composition in 7-10-year-old Dublin children. British Journal of Sports Medicine 41(5):311-316.
Imperatore, G., Y. J. Cheng, D. E. Williams, J. Fulton, and E. W. Gregg. 2006. Physical activity, cardiovascular fitness, and insulin sensitivity among US adolescents: The National Health and Nutrition Examination Survey, 1999-2002. Diabetes Care 29(7):1567-1572.
IOM (Institute of Medicine). 2004. Children’s health, the nation’s wealth. Washington, DC: The National Academies Press.
IOM. 2005. Preventing childhood obesity: Health in the balance. Washington, DC: The National Academies Press.
IOM. 2012a. Accelerating progress in obesity prevention: Solving the weight of the nation. Washington, DC: The National Academies Press.
IOM. 2012b. Fitness measures and health outcomes in youth. Washington, DC: The National Academies Press.
Irwin, M. L., Y. Yasui, C. M. Ulrich, D. Bowen, R. E. Rudolph, R. S. Schwartz, M. Yukawa, E. Aiello, J. D. Potter, and A. McTiernan. 2003. Effect of exercise on total and intra-abdominal body fat in postmenopausal women. Journal of the American Medical Association 289(3):323-330.
Isasi, C. R., R. J. Deckelbaum, R. P. Tracy, T. J. Starc, L. Berglund, and S. Shea. 2003. Physical fitness and C-reactive protein level in children and young adults: The Columbia University Biomarkers Study. Pediatrics 111(2):332-338.
Jaakkola, T., S. Kalaja, J. Liukkonen, A. Jutila, P. Virtanen, and A. Watt. 2009. Relations among physical activity patterns, lifestyle activities, and fundamental movement skills for Finnish students in grade 7. Perceptual and Motor Skills 108(1):97-111.
Janssen, I., and A. G. LeBlanc. 2010. Systematic review of the health benefits of physical activity and fitness in school-aged children and youth. International Journal of Behavioral Nutrition and Physical Activity 7(40):1-16.
Janssen, I., P. Katzmarzyk, W. Boyce, C. Vereecken, C. Mulvihill, C. Roberts, C. Currie, and W. Pickett. 2005. Comparison of overweight and obesity prevalence in school-aged youth from 34 countries and their relationships with physical activity and dietary patterns. Obesity Reviews 6(2):123-132.
Jarrett, O. S., D. M. Maxwell, C. Dickerson, P. Hoge, G. Davies, and A. Yetley. 1998. Impact of recess on classroom behavior: Group effects and individual differences. Journal of Educational Research 92(2):121-126.
Jolliffe, C. J., and I. Janssen. 2007. Development of age-specific adolescent metabolic syndrome criteria that are linked to the Adult Treatment Panel III and International Diabetes Federation criteria. Journal of the American College of Cardiology 49(8):891-898.
Jones, M., G. Stratton, T. Reilly, and V. Unnithan. 2007. The efficacy of exercise as an intervention to treat recurrent nonspecific low back pain in adolescents. Pediatric Exercise Science 19(3):349-359.
Kang, H.-S., B. Gutin, P. Barbeau, S. Owens, C. R. Lemmon, J. Allison, M. S. Litaker, and N.-A. Le. 2002. Physical training improves insulin resistance syndrome markers in obese adolescents. Medicine and Science in Sports and Exercise 34(12):1920.
Kannel, W. B., and T. R. Dawber. 1972. Atherosclerosis as a pediatric problem. Journal of Pediatrics 80(4):544-554.
Kannus, P., H. Haapasalo, M. Sankelo, H. Sievänen, M. Pasanen, A. Heinonen, P. Oja, and I. Vuori. 1995. Effect of starting age of physical activity on bone mass in the dominant arm of tennis and squash players. Annals of Internal Medicine 123(1):27.
Kappos, A. D. 2007. The impact of electronic media on mental and somatic children’s health. International Journal of Hygiene and Environmental Health 210(5):555-562.
Karlsson, M. 2007. Does exercise during growth prevent fractures in later life? Medicine and Sport Science 51:121-136.
Katz, D. L., D. Cushman, J. Reynolds, V. Njike, J. A. Treu, J. Walker, E. Smith, and C. Katz. 2010. Putting physical activity where it fits in the school day: Preliminary results of the ABC (Activity Bursts in the Classroom) for fitness program. Preventing Chronic Disease 7(4):A82.
Katzmarzyk, P. T., L. Pérusse, R. M. Malina, and C. Bouchard. 1999. Seven-year stability of indicators of obesity and adipose tissue distribution in the Canadian population. American Journal of Clinical Nutrition 69(6):1123-1129.
Katzmarzyk, P. T., L. Pérusse, R. M. Malina, J. Bergeron, J.-P. Després, and C. Bouchard. 2001. Stability of indicators of the metabolic syndrome from childhood and adolescence to young adulthood: The Quebec Family Study. Journal of Clinical Epidemiology 54(2):190-195.
Kelley, G. A., and K. S. Kelley. 2008. Effects of aerobic exercise on non-high-density lipoprotein cholesterol in children and adolescents: A meta-analysis of randomized controlled trials. Progress in Cardiovascular Nursing 23(3):128-132.
Kim, Y. K. Y., and S. L. S. Lee. 2009. Physical activity and abdominal obesity in youth. Applied Physiology, Nutrition, and Metabolism 34(4):571-581.
Knittle, J., K. Timmers, F. Ginsberg-Fellner, R. Brown, and D. Katz. 1979. The growth of adipose tissue in children and adolescents. Cross-sectional and longitudinal studies of adipose cell number and size. Journal of Clinical Investigation 63(2):239.
Knowler, W. C., E. Barrett-Connor, S. E. Fowler, R. F. Hamman, J. M. Lachin, E. A. Walker, and D. M. Nathan. 2002. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. New England Journal of Medicine 346(6):393-403.
Kohl, H. W., and K.E. Hobbs. 1998. Development of physical activity behaviors among children and adolescents. Pediatrics 101(Suppl 2):549-554.
Kritz-Silverstein, D., E. Barrett-Connor, and C. Corbeau. 2001. Cross-sectional and prospective study of exercise and depressed mood in the elderly: The Rancho Bernardo Study. American Journal of Epidemiology 153(6):596-603.
Kuczmarski, R. J., C. L. Ogden, L. M. Grummer-Strawn, K. M. Flegal, S. S. Guo, R. Wei, Z. Mei, L. R. Curtin, A. F. Roche, and C. L. Johnson. 2000. CDC growth charts: United States. Advance Data (314):1.
Laaksonen, D. E., H.-M. Lakka, J. T. Salonen, L. K. Niskanen, R. Rauramaa, and T. A.
Lakka. 2002. Low levels of leisure-time physical activity and cardiorespiratory fitness predict development of the metabolic syndrome. Diabetes Care 25(9):1612-1618.
Larun, L., L. Nordheim, E. Ekeland, K. Hagen, and F. Heian. 2006. Exercise in prevention and treatment of anxiety and depression among children and young people. Cochrane Database of Systematic Reviews (3):CD004691.
Lauer, R. M., W. E. Connor, P. E. Leaverton, M. A. Reiter, and W. R. Clarke. 1975. Coronary heart disease risk factors in school children: The Muscatine Study. Journal of Pediatrics 86(5):697-706.
Laurson, K. R., J. C. Eisenmann, and G. J. Welk. 2011. Body fat percentile curves for US children and adolescents. American Journal of Preventive Medicine 41(4):S87-S92.
Lazaar, N., J. Aucouturier, S. Ratel, M. Rance, M. Meyer, and P. Duché. 2007. Effect of physical activity intervention on body composition in young children: Influence of body mass index status and gender. Acta Paediatrica 96(9):1321-1325.
Le Masurier, G., A. Beighle, C. Corbin, P. Darst, C. Morgan, R. Pangrazi, B. Wilde, and S. Vincent. 2005. Pedometer-determined physical activity levels of youth. Journal of Physical Activity and Health 2(2):159-168.
Lee, J. M., N. Kaciroti, D. Appugliese, R. F. Corwyn, R. H. Bradley, and J. C. Lumeng. 2010. Body mass index and timing of pubertal initiation in boys. Archives of Pediatrics and Adolescent Medicine 164(2):139.
Lee, S., J. L. Kuk, L. E. Davidson, R. Hudson, K. Kilpatrick, T. E. Graham, and R. Ross. 2005. Exercise without weight loss is an effective strategy for obesity reduction in obese individuals with and without type 2 diabetes. Journal of Applied Physiology 99(3):1220-1225.
Leppamaki, S., T. T. Partonen, J. Hurme, J. K. Haukka, and J. Lonnqvist. 2002. Randomized trial of the efficacy of bright-light exposure and aerobic exercise on depressive symptoms and serum lipids. Journal of Clinical Psychiatry 63(4):316-321.
Li, S., W. Chen, S. R. Srinivasan, M. G. Bond, R. Tang, E. M. Urbina, and G. S. Berenson. 2003. Childhood cardiovascular risk factors and carotid vascular changes in adulthood. Journal of the American Medical Association 290(17):2271-2276.
Lindén, C., S. Stenevi-Lundgren, P. Gardsell, and M. Karlsson. 2006. A five-year school curriculum-based exercise program in girls during early adolescence is associated with a large bone size and a thick cortical shell—pQCT data from the prospective pediatric osteoporosis prevention study (POP study). Journal of Bone and Mineral Research 21:S38.
Lobelo, F., R. R. Pate, M. Dowda, A. D. Liese, and S. R. Daniels. 2010. Cardiorespiratory fitness and clustered cardiovascular disease risk in US adolescents. Journal of Adolescent Health 47(4):352-359.
Loftin, M., P. K. Strikmiller, B. Warren, L. Myers, L. Schroth, J. Pittman, D. Harsha, and M. Sothern. 1998. Original research comparison and relationship of vo2 peak and physical activity patterns in elementary and high school females. Pediatric Exercise Science 10:153-163.
Lohman, T. G., K. Ring, K. H. Schmitz, M. S. Treuth, M. Loftin, S. Yang, M. Sothern, and S. Going. 2006. Associations of body size and composition with physical activity in adolescent girls. Medicine and Science in Sports and Exercise 38(6):1175.
Lopes, V. P., L. P. Rodrigues, J. A. Maia, and R. M. Malina. 2011. Motor coordination as predictor of physical activity in childhood. Scandinavian Journal of Medicine and Science in Sports 21(5):663-669.
Lubans, D. R., P. J. Morgan, D. P. Cliff, L. M. Barnett, and A. D. Oakley. 2010. Fundamental movement skills in children and adolescents: Review of associated health benefits. Sports Medicine 40(12):1019-1035.
Lytle, L. A., D. M. Murray, K. R. Evenson, J. Moody, C. A. Pratt, L. Metcalfe, and D. Parra-Medina. 2009. Mediators affecting girls’ levels of physical activity outside of school: Findings from the trial of activity in adolescent girls. Annals of Behavioral Medicine 38(2):124-136.
Macdonald-Wallis, K., R. Jago, A. S. Page, R. Brockman, and J. L. Thompson. 2011. School-based friendship networks and children’s physical activity: A spatial analytical approach. Social Science and Medicine 73(1):6-12.
MacKelvie, K. J., K. M. Khan, M. A. Petit, P. A. Janssen, and H. A. McKay. 2003. A school-based exercise intervention elicits substantial bone health benefits: A 2-year randomized controlled trial in girls. Pediatrics 112(6 Pt 1):e447-e452.
MacKelvie, K. J., M. A. Petit, K. M. Khan, T. J. Beck, and H. A. McKay. 2004. Bone mass and structure are enhanced following a 2-year randomized controlled trial of exercise in prepubertal boys. Bone 34(4):755-764.
MacKelvie, K. J., H. A. McKay, K. M. Khan, and P. R. E. Crocker. 2001. A school-based exercise intervention augments bone mineral accrual in early pubertal girls. Journal of Pediatrics 139(4):501-508.
Mahoney, L. T., T. L. Burns, W. Stanford, B. H. Thompson, J. D. Witt, C. A. Rost, and R. M. Lauer. 1996. Coronary risk factors measured in childhood and young adult life are associated with coronary artery calcification in young adults: The Muscatine Study. Journal of the American College of Cardiology 27(2):277-284.
Malina, R. M. 1969. Quantification of fat, muscle and bone in man. Clinical Orthopaedics and Related Research 65:9-38.
Malina, R. M. 1986. Growth of muscle tissue and muscle mass. In Human growth. Vol. 2, edited by F. Falkner and J. M. Tanner. New York: Plenum. Pp. 77-99.
Malina, R. M. 1991. Fitness and performance: Adult health and the culture of youth. In New possibilities, new paradigms? American Academy of Physical Education, No. 24, edited by R. J. Park and H. M. Eckert. Champaign, IL: Human Kinetics Publishers. Pp. 30-38.
Malina, R. M. 1994. Physical growth and biology maturation of young athletes. Exercise and Sports Sciences Review 22:389-433.
Malina, R. M. 1996. Regional body composition: Age, sex, and ethnic variation. In Human body composition, edited by A. F. Roche, S. Heymsfield, and T. G. Lohman. Champaign, IL: Human Kinetics Publishers. Pp. 217-255.
Malina, R. M. 2001a. Adherence to physical activity from childhood to adulthood: A perspective from tracking studies. Quest 53(3):346-355.
Malina, R. M. 2001b. Tracking of physical activity across the lifespan. President’s Council on Physical Fitness and Sports Research Digest 3(14).
Malina, R. M. 2002. 15—exercise and growth: Physical activity as a factor in growth and maturation. In Human growth and development. San Diego: Academic Press. Pp. 321-348.
Malina, R. 2007. Physical fitness of children and adolescents in the United States: Status and secular change. Medicine and Sports Science 50:67-90.
Malina, R. M., and C. Bouchard. 1988. Subcutaneous fat distribution during growth. In Fat distribution during growth and later health outcomes, edited by C. Bouchard and F. E. Johnston. New York: Alan R. Liss. Pp. 63-84.
Malina, R. M., and A. F. Roche. 1983. Manual of physical status and performance in childhood, Vol. 2. New York: Plenum.
Malina, R. M., C. Bouchard, and O. Bar-Or. 2004. Growth, maturation, and physical activity, 2nd ed. Champaign, IL: Human Kinetics Publishers.
Martikainen, S., A.-K. Pesonen, J. Lahti, K. Heinonen, K. Feldt, R. Pyhälä, and T. Tammelin. 2013. Higher levels of physical activity are associated with lower hypothalamic-pituitary-adrenocortical axis reactivity to psychosocial stress in children. Journal of Clinical Endocrinology & Metabolism [epub ahead of print].
McAuley, E. 1994. Physical activity and psychosocial outcomes. In Physical activity, fitness and health, edited by C. Bouchard, R. J. Shepard and T. Stephens. Champaign IL: Human Kinetics Publishers. Pp. 551-568.
McAuley, E., and D. Rudolph. 1995. Physical activity, aging, and psychological well-being. Journal of Aging and Physical Activity 3(1):67-98.
McKay, H. A., M. A. Petit, R. W. Schutz, J. C. Prior, S. I. Barr, and K. M. Khan. 2000. Augmented trochanteric bone mineral density after modified physical education
classes: A randomized school-based exercise intervention study in prepubescent and early pubescent children. Journal of Pediatrics 136(2):156-162.
McKenzie, T. L., J. F. Sallis, S. L. Broyles, M. M. Zive, P. R. Nader, C. C. Berry, and J. J. Brennan. 2002. Childhood movement skills: Predictors of physical activity in Anglo American and Mexican American adolescents? Research Quarterly for Exercise and Sport 73(3):238-244.
McKenzie, T. L., J. J. Prochaska, J. F. Sallis, and K. J. Lamaster. 2004. Coeducational and single-sex physical education in middle schools: Impact on physical activity. Research Quarterly for Exercise and Sport 75(4):446-449.
McMurray, R. G., J. S. Harrell, S. I. Bangdiwala, C. B. Bradley, S. Deng, and A. Levine. 2002. A school-based intervention can reduce body fat and blood pressure in young adolescents. Journal of Adolescent Health 31(2):125-132.
McMurray, R., S. Bangdiwala, J. Harrell, and L. Amorim. 2008. Adolescents with metabolic syndrome have a history of low aerobic fitness and physical activity levels. Dynamic Medicine 7(1):5.
Merzenich, H., H. Boeing, and J. Wahrendorf. 1993. Dietary fat and sports activity as determinants for age at menarche. American Journal of Epidemiology 138(4):217-224.
Meyer, A. A., G. Kundt, U. Lenschow, P. Schuff-Werner, and W. Kienast. 2006. Improvement of early vascular changes and cardiovascular risk factors in obese children after a six-month exercise program. Journal of the American College of Cardiology 48(9):1865-1870.
Mirwald, R., and D. Bailey. 1986. Maximal aerobic power. London, Ontario: Sports Dynamics.
Modlesky, C. M., and R. D. Lewis. 2002. Does exercise during growth have a long-term effect on bone health? Exercise and Sport Sciences Reviews 30(4):171-176.
Mølgaard, C., B. L. Thomsen, A. Prentice, T. J. Cole, and K. F. Michaelsen. 1997. Whole body bone mineral content in healthy children and adolescents. Archives of Disease in Childhood 76(1):9-15.
Morgan, P. J., A. D. Okely, D. P. Cliff, R. A. Jones, and L. A. Baur. 2008. Correlates of objectively measured physical activity in obese children. Obesity 16(12):2634-2641.
Morris, F. L., G. A. Naughton, J. L. Gibbs, J. S. Carlson, and J. D. Wark. 1997. Prospective ten-month exercise intervention in premenarcheal girls: Positive effects on bone and lean mass. Journal of Bone and Mineral Research 12(9):1453-1462.
Morrison, J. A., L. A. Friedman, and C. Gray-McGuire. 2007. Metabolic syndrome in childhood predicts adult cardiovascular disease 25 years later: The Princeton Lipid Research Clinics Follow-up Study. Pediatrics 120(2):340-345.
Morrison, J. A., L. A. Friedman, P. Wang, and C. J. Glueck. 2008. Metabolic syndrome in childhood predicts adult metabolic syndrome and type 2 diabetes mellitus 25 to 30 years later. Journal of Pediatrics 152(2):201-206.
Nader, P. R., R. H. Bradley, R. M. Houts, S. L. McRitchie, and M. O’Brien. 2008. Moderate-to-vigorous physical activity from ages 9 to 15 years. Journal of the American Medical Association 300(3):295-305.
NASPE (National Association for Sport and Physical Education). 2001. Physical education is critical to a complete education—position statement. Reston, VA: NASPE.
Nassis, G. P., K. Papantakou, K. Skenderi, M. Triandafillopoulou, S. A. Kavouras, M. Yannakoulia, G. P. Chrousos, and L. S. Sidossis. 2005. Aerobic exercise training improves insulin sensitivity without changes in body weight, body fat, adiponectin, and inflammatory markers in overweight and obese girls. Metabolism: Clinical and Experimental 54(11):1472.
NIH (National Institutes of Health). 2001. Third Report of the National Cholesterol Education Program Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Bethesda, MD: NIH.
NRC (National Research Council)/IOM. 1999. Adolescent development and the biology of puberty: Summary of a workshop on new research. Washington, DC: National Academy Press.
Ogden, C. L., and K. M. Flegal. 2011. Smoothed percentage body fat percentiles for US children and adolescents, 1999-2004. National Health Statistics Report 43:1-7.
Ogden, C. L., M. D. Carroll, B. K. Kit, and K. M. Flegal. 2012. Prevalence of obesity and trends in body mass index among U.S. children and adolescents, 1999-2010. Journal of the American Medical Association 307(5):483-490.
Okely, A. D., M. L. Booth, and J. W. Patterson. 2001. Relationship between physical activity to fundamental movement skills among adolescents. Medicine and Science in Sports and Exercise 33(11):1899-1904.
Ondrak, K. S., R. G. McMurray, S. I. Bangdiwala, and J. S. Harrell. 2007. Influence of aerobic power and percent body fat on cardiovascular disease risk in youth. Journal of Adolescent Health 41(2):146-152.
Owens, S., B. Gutin, J. Allison, S. Riggs, M. Ferguson, M. Litaker, and W. Thompson. 1999. Effect of physical training on total and visceral fat in obese children. Medicine and Science in Sports and Exercise 31(1):143.
Pan, Y., and C. A. Pratt. 2008. Metabolic syndrome and its association with diet and physical activity in US adolescents. Journal of the American Dietetic Association 108(2):276.
Parfitt, A. 1994. The two faces of growth: Benefits and risks to bone integrity. Osteoporosis International 4(6):382-398.
Pate, R. R., C.-Y. Wang, M. Dowda, S. W. Farrell, and J. R. O’Neill. 2006. Cardiorespiratory fitness levels among US youth 12 to 19 years of age: Findings from the 1999-2002 National Health and Nutrition Examination Survey. Archives of Pediatrics & Adolescent Medicine 160(10):1005.
Pellegrini, A. D., P. D. Huberty, and I. Jones. 1995. The effects of recess timing on children’s playground and classroom behaviors. American Educational Research Journal 32(4):845-864.
Peluso, M. A., and L. H. Guerra de Andrade. 2005. Physical activity and mental health: The association between exercise and mood. Clinics (São Paulo, Brazil) 60(1):61-70.
Penedo, F. J., and J. R. Dahn. 2005. Exercise and well-being: A review of mental and physical health benefits associated with physical activity. Current Opinion in Psychiatry 18(2):189-193.
Petit, M., H. McKay, K. MacKelvie, A. Heinonen, K. Khan, and T. Beck. 2002. A randomized school-based jumping intervention confers site and maturity-specific benefits on bone structural properties in girls: A hip structural analysis study. Journal of Bone and Mineral Research 17(3):363-372.
Petty, K. H., C. L. Davis, J. Tkacz, D. Young-Hyman, and J. L. Waller. 2009. Exercise effects on depressive symptoms and self-worth in overweight children: A randomized controlled trial. Journal of Pediatric Psychology 34(9):929-939.
Platat, C., A. Wagner, T. Klumpp, B. Schweitzer, and C. Simon. 2006. Relationships of physical activity with metabolic syndrome features and low-grade inflammation in adolescents. Diabetologia 49(9):2078-2085.
Plowman, S. A. 1992. Physical activity, physical fitness, and low back pain. Exercise and Sport Sciences Review 20(1):221-242.
Primack, B. A., B. Swanier, A. M. Georgiopoulos, S. R. Land, and M. J. Fine. 2009. Association between media use in adolescence and depression in young adulthood: A longitudinal study. Archives of General Psychiatry 66(2):181-188.
Ramírez-Vélez, R., M. F. Suaréz-Ortegón, and A. C. Aguilar de Plata. 2012. Association between adiposity and cardiovascular risk factors in prepubertal children. Endocrinología y Nutrición (English Edition) 58(9):457-463.
Raudsepp, L., and P. Päll. 2006. The relationship between fundamental motor skills and outside-school physical activity of elementary school children. Pediatric Exercise Science 18(4):426-435.
Reed, J., A. Metzker, and D. Phillips. 2004. Relationships between physical activity and motor skills in middle school children. Perceptual and Motor Skills 99(2):483.
Robinson, L. E. 2011. Effect of a mastery climate motor program on object control skills and perceived physical competence in preschoolers. Research Quarterly for Exercise and Sport 82(2):355-359.
Robinson, L. E., and J. D. Goodway. 2009. Instructional climates in preschool children who are at risk. Part I: Object-control skill development. Research Quarterly for Exercise and Sport 80(3):533-542.
Robinson, L. E., D. D. Wadsworth, and C. M. Peoples. 2012. Correlates of school-day physical activity in preschoolers: A preliminary study. Research Quarterly for Exercise and Sport 83(1):20-26.
Robinson, T. N. 1999. Reducing children’s television viewing to prevent obesity. Journal of the American Medical Association 282(16):1561-1567.
Robinson, T. N., and D. L. G. Borzekowski. 2006. Effects of the smart classroom curriculum to reduce child and family screen time. Journal of Communication 56(1):1-26.
Rolland-Cachera, M., M. Deheeger, F. Bellisle, M. Sempe, M. Guilloud-Bataille, and E. Patois. 1984. Adiposity rebound in children: A simple indicator for predicting obesity. American Journal of Clinical Nutrition 39(1):129-135.
Rosenfield, R. L., R. B. Lipton, and M. L. Drum. 2009. Thelarche, pubarche, and menarche attainment in children with normal and elevated body mass index. Pediatrics 123(1):84-88.
Ross, R., and A. J. Bradshaw. 2009. The future of obesity reduction: Beyond weight loss. Nature Reviews Endocrinology 5(6):319-325.
Ross, R., and P. M. Janiszewski. 2008. Is weight loss the optimal target for obesity-related cardiovascular disease risk reduction? Canadian Journal of Cardiology 24(Suppl D):25D.
Ross, R., D. Dagnone, P. J. Jones, H. Smith, A. Paddags, R. Hudson, and I. Janssen. 2000.
Reduction in obesity and related comorbid conditions after diet-induced weight loss or exercise-induced weight loss in men. Annals of Internal Medicine 133(2):92-103.
Ross, R., I. Janssen, J. Dawson, A. M. Kungl, J. L. Kuk, S. L. Wong, T. B. Nguyen-Duy, S. Lee, K. Kilpatrick, and R. Hudson. 2012. Exercise-induced reduction in obesity and insulin resistance in women: A randomized controlled trial. Obesity Research 12(5):789-798.
Rowland, T. W. 1996. Developmental exercise physiology. Champaign, IL: Human Kinetics Publishers.
Rowland, T. W. 2005. Children’s exercise physiology. Champaign, IL: Human Kinetics Publishers.
Rowlands, A., D. Ingledew, and R. Eston. 2000. The effect of type of physical activity measure on the relationship between body fatness and habitual physical activity in children: A meta-analysis. Annals of Human Biology 27(5):479-497.
Rubin, D. A., R. G. McMurray, J. S. Harrell, A. C. Hackney, D. E. Thorpe, and A. M. Haqq. 2008. The association between insulin resistance and cytokines in adolescents: The role of weight status and exercise. Metabolism: Clinical and Experimental 57(5):683.
Ruiz, J., F. Ortega, J. Wärnberg, and M. Sjöström. 2007. Associations of low-grade inflammation with physical activity, fitness and fatness in prepubertal children: The European Youth Heart Study. International Journal of Obesity 31(10):1545-1551.
Ruiz, J. R., F. B. Ortega, J. Wärnberg, L. A. Moreno, J. J. Carrero, M. Gonzalez-Gross, A. Marcos, A. Gutierrez, and M. Sjöström. 2008. Inflammatory proteins and muscle strength in adolescents: The Avena Study. Archives of Pediatrics & Adolescent Medicine 162(5):462-468.
Sääkslahti, A., P. Numminen, H. Niinikoski, L. Rask-Nissilä, J. Viikari, J. Tuominen, and I. Välimäki. 1999. Is physical activity related to body size, fundamental motor skills, and CHD risk factors in early childhood? Pediatric Exercise Science 11:327-340.
Sallis, J. F., C. C. Berry, S. L. Broyles, and T. L. McKenzie. 1995. Variability and tracking of physical activity over 2 yrs in young children. Medicine and Science in Sports and Exercise 27(7):1042-1049.
Sallis, J. F., J. J. Prochaska, and W. C. Taylor. 2000. A review of correlates of physical activity of children and adolescents. Medicine and Science in Sports and Exercise 32(5):963-975.
Sardinha, L. B., L. B. Andersen, S. A. Anderssen, A. L. Quitério, R. Ornelas, K. Froberg, C. J. Riddoch, and U. Ekelund. 2008. Objectively measures time spent sedentary is associated with insulin resistance independent of overall and central body fat in 9- to 10-year-old Portuguese children. Diabetes Care 31(3):569-575.
Saris, W. H. M., J. W. H. Elvers, M. A. van’t Hof, and R. A. Binkhorst. 1986. Changes in physical activity of children aged 6 to 12 years. In Children and exercise XII, edited by J. Rutenfranz, R. Mocellin, and F. Klimt. Champaign, IL: Human Kinetics. Pp. 121-130.
Seefeldt, V. 1980. Developmental motor patterns: Implications for elementary school physical education. In Psychology of motor behavior and sport, edited by W. H. C. Nadeau, K. Newell, and G. Roberts. Champaign, IL: Human Kinetics. Pp. 314-323.
Sexton, H., A. Søgaard, and R. Olstad. 2001. How are mood and exercise related? Results from the Finnmark study. Social Psychiatry and Psychiatric Epidemiology 36(7):348-353.
Shaibi, G. Q., M. L. Cruz, G. D. C. Ball, M. J. Weigensberg, G. J. Salem, N. C. Crespo, and M. I. Goran. 2006. Effects of resistance training on insulin sensitivity in overweight Latino adolescent males. Medicine and Science in Sports and Exercise 38(7):1208.
Sigal, R. J., G. P. Kenny, N. G. Boulé, G. A. Wells, D. Prud’homme, M. Fortier, R. D. Reid, H. Tulloch, D. Coyle, and P. Phillips. 2007. Effects of aerobic training, resistance training, or both on glycemic control in type 2 diabetes. Annals of Internal Medicine 147(6):357-369.
Slaven, L., and C. Lee. 1997. Mood and symptom reporting among middle-aged women: The relationship between menopausal status, hormone replacement therapy, and exercise participation. Health Psychology 16(3):203.
Sollerhed, A.-C., E. Apitzsch, L. Råstam, and G. Ejlertsson. 2008. Factors associated with young children’s self-perceived physical competence and self-reported physical activity. Health Education Research 23(1):125-136.
Specker, B., and T. Binkley. 2003. Randomized trial of physical activity and calcium supplementation on bone mineral content in 3- to 5-year-old children. Journal of Bone and Mineral Research 18(5):885-892.
Steele, R. M., S. Brage, K. Corder, N. J. Wareham, and U. Ekelund. 2008. Physical activity, cardiorespiratory fitness, and the metabolic syndrome in youth. Journal of Applied Physiology 105(1):342-351.
Steene-Johannessen, J., S. A. Anderssen, E. Kolle, and L. B. Andersen. 2009. Low muscle fitness is associated with metabolic risk in youth. Medicine and Science in Sports and Exercise 41(7):1361-1367.
Stevens, J., C. Suchindran, K. Ring, C. D. Baggett, J. B. Jobe, M. Story, J. Thompson, S. B. Going, and B. Caballero. 2004. Physical activity as a predictor of body composition in American Indian children. Obesity Research 12(12):1974-1980.
Stodden, D. F., J. D. Goodway, S. J. Langendorfer, M. A. Roberton, M. E. Rudisill, C. Garcia, and L. E. Garcia. 2008. A developmental perspective on the role of motor skill competence in physical activity: An emergent relationship. Quest 60(2):290-306.
Stoedefalke, K., N. Armstrong, B. Kirby, and J. Welsman. 2000. Effect of training on peak oxygen uptake and blood lipids in 13- to 14-year-old girls. Acta Paediatrica 89(11):1290-1294.
Strong, W. B., R. M. Malina, C. J. Blimkie, S. R. Daniels, R. K. Dishman, B. Gutin, A. C. Hergenroeder, A. Must, P. A. Nixon, J. M. Pivarnik, T. Rowland, S. Trost, and F. Trudeau. 2005. Evidence based physical activity for school-age youth. Journal of Pediatrics 146(6):732-737.
Sundgot-Borgen, J., J. Rosenvinge, R. Bahr, and L. Schneider. 2002. The effect of exercise, cognitive therapy, and nutritional counseling in treating bulimia nervosa. Medicine and Science in Sports and Exercise 34(2):190.
Tanner, J. M. 1962. Growth at adolescence, 2nd ed. Oxford, England: Blackwell Scientific Publications.
Tanner, J., P. Hughes, and R. Whitehouse. 1981. Radiographically determined widths of bone muscle and fat in the upper arm and calf from age 3-18 years. Annals of Human Biology 8(6):495-517.
Thomas, J. R. 1994. Effects of training on gender differences in overhand throwing: A brief quantitative literature analysis. Research Quarterly for Exercise and Sport 65(1):67-71.
Thomas, J. R., and K. E. French. 1985. Gender differences across age in motor performance: A meta-analysis. Psychological Bulletin 98(2):260.
Thomas, J. R., and K. T. Thomas. 1988. Development of gender differences in physical activity. Quest 40(3):219-229.
Thomas, N., and D. Williams. 2008. Inflammatory factors, physical activity, and physical fitness in young people. Scandinavian Journal of Medicine & Science in Sports 18(5):543-556.
Tobias, J. H., C. D. Steer, C. G. Mattocks, C. Riddoch, and A. R. Ness. 2007. Habitual levels of physical activity influence bone mass in 11-year-old children from the United Kingdom: Findings from a large population-based cohort. Journal of Bone and Mineral Research 22(1):101-109.
Treuth, M. S., G. R. Hunter, R. Figueroa-Colon, and M. I. Goran. 1998. Effects of strength training on intra-abdominal adipose tissue in obese prepubertal girls. Medicine and Science in Sports and Exercise 30(12):1738.
Trotter, M., and B. B. Hixon. 1974. Sequential changes in weight, density, and percentage ash weight of human skeletons from an early fetal period through old age. Anatomical Record 179(1):1-18.
Trotter, M., and R. R. Peterson. 1970. Weight of the skeleton during postnatal development. American Journal of Physical Anthropology 33(3):313-323.
Turner, C. H., and A. G. Robling. 2003. Designing exercise regimens to increase bone strength. Exercise and Sport Science Reviews 31(1):45-50.
Umemura, Y., T. Ishiko, T. Yamauchi, M. Kurono, and S. Mashiko. 1997. Five jumps per day increase bone mass and breaking force in rats. Journal of Bone and Mineral Research 12(9):1480-1485.
U.S. Public Health Service. 2000. Report of the Surgeon General’s conference on children’s mental health: A national action agenda. Washington, DC: HHS.
Viner, R., and R. Booy. 2005. Epidemiology of health and illness. British Medical Journal 330(7488):411-414.
Wang, Q., H. Suominen, P. Nicholson, L. Zou, M. Alen, A. Koistinen, and S. Cheng. 2004. Influence of physical activity and maturation status on bone mass and geometry in early pubertal girls. Scandinavian Journal of Medicine & Science in Sports 15(2):100-106.
Wang, Q., S. Cheng, M. Alén, and E. Seeman. 2009. Bone’s structural diversity in adult females is established before puberty. Journal of Clinical Endocrinology and Metabolism 94(5):1555-1561.
Wärnberg, J., and A. Marcos. 2008. Low-grade inflammation and the metabolic syndrome in children and adolescents. Current Opinion in Lipidology 19(1):11-15.
Wärnberg, J., E. Nova, J. Romeo, L. A. Moreno, M. Sjöström, and A. Marcos. 2007. Lifestyle-related determinants of inflammation in adolescence. British Journal of Nutrition 98(Suppl 1):S116-S120.
Wärnberg, J., K. Cunningham, J. Romeo, and A. Marcos. 2010. Session 6: Role of physical activity on immune function physical activity, exercise and low-grade systemic inflammation. Proceedings of the Nutrition Society 69(3):400-406.
Watts, K., T. W. Jones, E. A. Davis, and D. Green. 2005. Exercise training in obese children and adolescents: Current concepts. Sports Medicine 35(5):375-392.
Weiss, R., and S. Caprio. 2005. The metabolic consequences of childhood obesity. Best Practice and Research Clinical Endocrinology and Metabolism 19(3):405-419.
Weiss, R., J. Dziura, T. S. Burgert, W. V. Tamborlane, S. E. Taksali, C. W. Yeckel, K. Allen, M. Lopes, M. Savoye, and J. Morrison. 2004. Obesity and the metabolic syndrome in children and adolescents. New England Journal of Medicine 350(23):2362-2374.
WHO (World Health Organization). 1986. Young people’s health—a challenge for society. Report of a study group on young people and health for all by the year 2000. http://whqlibdoc.who.int/trs/WHO_TRS_731.pdf (accessed March 1, 2013).
Williams, H. G. 1983. Perceptual and motor development. Englewood Cliffs, NJ: Prentice Hall.
Williams, H. G., K. A. Pfeiffer, J. R. O’Neill, M. Dowda, K. L. McIver, W. H. Brown, and R. R. Pate. 2008. Motor skill performance and physical activity in preschool children. Obesity 16(6):1421-1426.
Wrotniak, B. H., L. H. Epstein, J. M. Dorn, K. E. Jones, and V. A. Kondilis. 2006. The relationship between motor proficiency and physical activity in children. Pediatrics 118(6):e1758-e1765.
You, T., K. Murphy, M. Lyles, J. Demons, L. Lenchik, and B. Nicklas. 2006. Addition of aerobic exercise to dietary weight loss preferentially reduces abdominal adipocyte size. International Journal of Obesity 30(8):1211-1216.
Zeng, Q., S.-Y. Dong, X.-N. Sun, J. Xie, and Y. Cui. 2012. Percent body fat is a better predictor of cardiovascular risk factors than body mass index. Brazilian Journal of Medical and Biological Research 45(7):591-600.
Ziviani, J., A. Poulsen, and C. Hansen. 2009. Movement skills proficiency and physical activity: A case for Engaging and Coaching for Health (EACH)-child. Australian Occupational Therapy Journal 56(4):259-265.
































































