Medical and Life Science Studies
(MRI, fMRI, MRS)
Enabled by 20 Tesla
Development of Human Magnetic Resonance Imaging and Spectroscopy
This section is focused on in vivo studies of human beings and animals in health and disease enabled by very-high-field magnetic resonance imaging and spectroscopy. Much of the material presented here is based on the expectation that large magnets with fields as high as 20 T (1 tesla is 10,000 gauss) can be produced with a homogeneity of 1 ppm over a sphere of 16 cm diameter. The clear bore necessary for humans is at least 65 cm in diameter for head and extremity studies and 85 cm for other body parts. These magnets are for scientific research on fundamental medical and physiological problems ranging from cognitive science to aging, heart disease, and cancer. The opportunities opened by much higher magnetic fields than exist today are tremendous, because many human health conditions cannot be approached by any other methods, as discussed in the body of this chapter. The technologies presented here are meant for research and not for routine clinical use.
It is expected that a total of 40,000 clinical systems will have been installed worldwide by 2012. The majority (64 percent) of new installations is for 1.5 T, with the remainder equally divided between 3.0 T and less than 1.5 T. The number of magnets that have been sold having field strength at or above 7 T is 50 at 7 T, 5 at 9.4 T, 1 at 10.5 T, and 2 at 11.7 T. One 14 T magnet for human brain imaging is being funded for South Korea (Z. Cho, personal communication). Animal research systems with small bores and high fields are also in increasing demand worldwide.
Below is a brief history of the development of magnetic resonance imaging (MRI) and in vivo magnetic resonance spectroscopy (MRS) magnets, followed by a discussion of the medical and life science opportunities enabled by higher field magnets with wider bores and homogeneity than currently exist anywhere in the world. Appendix F provides information on safety and potential health effects of MRI.
Chronology of High-Field Developments Leading to High-Field MRI and MRS
Magnetic resonance applications in experimental science started soon after the discovery of proton nuclear magnetic resonance (NMR) in the 1950s. NMR instruments became important for physicists and chemists because the NMR signal carried information about the chemical structure of molecules. The field of MRS is now of major importance, particularly to chemistry (see discussion in Chapter 3). In 1972, chemist Paul Lauterbur of Stony Brook University showed that one can image the spatial distribution of the hydrogen nucleus concentration (mainly water) in objects, and this led to MRI (Lauterbur, 1973). MRI initially and, 10 years later, functional magnetic resonance imaging (fMRI) have become major modalities for research and diagnostic medicine, as well as for animal physiology studies, since the mid-1980s. The growth internationally has been from a few low-field magnets in the United States, Scotland, and England in the mid-1970s to 40,000 installations worldwide in 2012.
The field of NMR spectroscopy being pursued by chemists and physicists for research in molecular structure and dynamics has followed a parallel path of development, but with higher fields and much smaller samples (Figure 4.1).
The initial medical applications used horizontal bore electromagnets with a field strength of 0.04 to 0.15 T in the late 1970s. In the 1980s commercialization was successful for superconducting systems at 0.35 T. In the mid-1980s General Electric marketed worldwide superconducting whole body systems for clinical medicine at 1.5 T. Safety and health effects studies commenced in the late 1970s and continue to the present time while keeping pace with new methods of acquisition of the magnetic resonance signals and the increases in magnetic field strength (Appendix F).
Throughout the development of MRI and MRS, “each substantive increase in field strength has in time led to dramatic improvements in the quality of images and spectra obtainable, and usually to ‘quantum leaps’ in the information available about tissue structure and function [Figure 4.2]. Each major increase in field has also introduced new technical challenges and problems that have required creative scientific and engineering solutions in order to realize the potential to improve image quality.” (Dula, 2010).
The evolution of higher field systems has continued. By 1988 success in development of a whole body 4 T system was reported (Barfuss et al., 1988; Bomsdorf
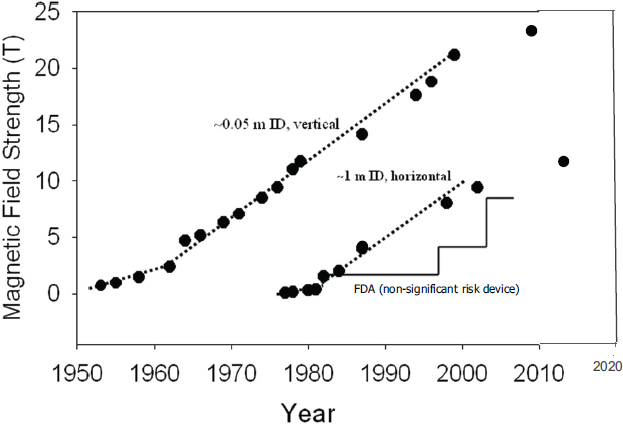
FIGURE 4.1 Flux density (tesla) of steady-state NMR-quality magnets relative to year first demonstrated. The upper group is magnets for analytical NMR applications. The lower set represents magnets suitable for human MRI/MRS. The step curve is the FDA guideline that does not denote a hazard threshold but only caution. SOURCE: Image used with permission from William Rooney, Oregon Health Sciences University; adapted from W.D. Rooney, G. Johnson, X. Li, E.R. Cohen, S.-G. Kim, K. Ugurbil, and C.S. Springer, 2007, The magnetic field and tissue dependences of human brain 1H2O longitudinal relaxation in vivo, Magnetic Resonance in Medicine 57:308-318.
et al., 1988; Schenck et al., 1992; Ugurbil et al., 1993), and commercial vendors made a small number of 4 T MRI magnets. However, ultimately the main industrial effort focused on developing scanners operating at 3 T, and these systems are replacing 1.5 T systems in many clinical applications. Much of the early 3 T developments emphasized brain imaging, partly motivated by the discovery of the benefits of blood oxygenation level-dependent susceptibility contrast as a measure of brain activity. This phenomenon is also known as fMRI.
The pioneering 8 T installation at Ohio State University (Robitaille et al., 1999) gave notice to the community that the current density limits of the magnet wire material then in use could be approached and that large bore magnets at 7 T could be developed with an expected commercial market. A compendium on ultrahigh-field magnetic resonance imaging was edited by Robitaille in 1988. Thus the major imaging equipment manufacturers, General Electric, Siemens, and Philips, along with vendors of research magnetic resonance (e.g., Varian) embarked on 7 T development, and academic researchers were successful in obtaining institutional and
federal grant support to install these large-bore high-field magnets for medical science research and applications (e.g., Ugurbil et al., 1999). As already mentioned, there are approximately 50 human scanners operating at 7 T in the world today. An example of the demonstrable improvement in image quality over the past 30 years is shown in Figure 4.2.
By 2004 two human imaging systems at 9.4 T with warm bores of 65 cm diameter were being tested at the University of Minnesota and the University of Illinois in Chicago. Smaller scanners operating at higher fields are in extensive use in animal research. Systems with warm bores of 21 and 40 cm operating at 11.7 and 9.4 T are in widespread use, while smaller systems (11 cm bore) are used to image mice at 15.2 T. The 11.7 T installations require operation in liquid helium with reduced pressure and temperature of 2.5 K. One can conclude that 11.7 T is a realistic limit for large NbTi superconducting magnets even at reduced temperatures. This chronology is graphed in Figure 4.1.
While magnet manufacture technology was progressing, in parallel there had to be improvements in shimming techniques and hardware. These include radiofrequency (RF) transmission and reception coils; gradient coils and power supplies; spectrometer design and performance; and the associated pulse sequences needed
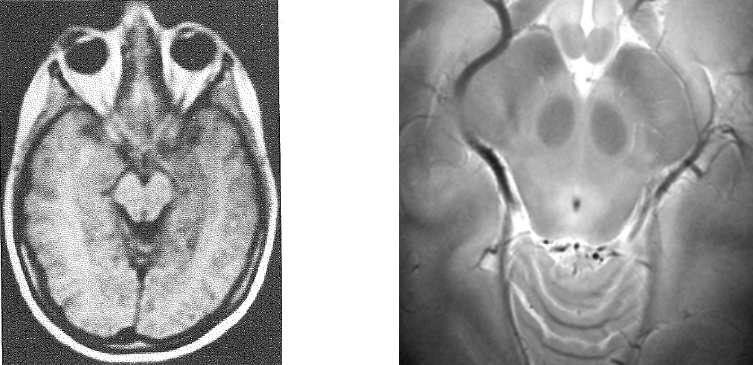
FIGURE 4.2 Increase in field and resolution for human brain proton MRI from 1984 to 2008. SOURCE: (Left) H.D. Sostman, D.D. Spencer, J.C. Gore, S.S. Spencer, W.G. Holcomb, P.D. Williamson, J. Prichard, C. Camputaro, R.H. Greenspan, and R.H. Mattson, 1984, Preliminary observations on NMR imaging in refractory epilepsy, Magnetic Resonance Imaging 2:301-306. (Right) Courtesy of John Gore, Vanderbilt University.
to obtain high-quality images with good contrast and high resolution in shorter experimental times. These advances have been and continue to be significant, with major new developments reported regularly. In the recent past, for example, parallel imaging using large numbers of receiver coils has emerged as an important improvement for imaging, providing new ways to reduce imaging times and increase signal to noise; at the same time, parallel transmission methods are also demonstrating how some limitations of RF fields at high frequencies can be overcome. Another notable innovation that arises only at very high fields (when RF wavelengths are shortened) is the use of traveling waves in MRI (e.g., Webb et al., 2010; Pang et al., 2011) to excite nuclei and detect their NMR signals using remote coils that are no longer closely coupled to the subject. This development suggests that a new class of experimental techniques may be developed, introducing some of the concepts of image formation from coherent optics such as holographic imaging and interferometry. These applications may develop significant importance as ultrahigh-field magnets and improvements in coil design arise.
The National High Magnetic Field Laboratory’s (NHMFL’s) role in meeting the needs of these research communities has been principally as a user facility. Currently, it has a range of wide-bore magnets at its Tallahassee and Gainesville facilities that are capable of performing MR using a variety of probes and scanners. The 21.1 T wide-bore spectrometer has been used for small-animal studies and is a resource for needed safety studies in planning for 20 T human studies. Other wide-bore magnets at NHMFL have sufficient homogeneity for exposures of animals in order to explore unforeseen physiological effects at static fields beyond 20 T.
The motivation for higher field imaging systems is to increase the signal-to-noise ratio (SNR) available for imaging, because the net nuclear magnetization induced in tissues scales linearly with the field, while the induced electromagnetic force (emf) in receiver coils also scales linearly with the frequency. Increased SNR leads to increased sensitivity for detecting changes within tissues, improved spatial resolution (imaging with smaller voxels), or shortening of data acquisition times. The main driver for development has been proton MRI, which largely depicts variations between tissues in proton (mainly water hydrogen nuclei) density and NMR relaxation times and provides exquisite anatomical images. In addition, there has been continual interest in the use of localized in vivo high-resolution NMR spectroscopy to study tissue metabolism and biochemistry. Proton magnetic resonance spectroscopy operates at the same frequency as proton MRI, but in addition there have been long-standing interest in and development of localized spectra from naturally occurring phosphorus-31 compounds (e.g., phosphomonesters, diesters, phosphocreatine, and adenosine phosphates involved in cellular energetics), as well as the metabolism of injected carbon-13-enriched agents. Next, the committee highlights the many opportunities for medical science advances using much higher fields than are currently available.
MEDICAL SCIENCE AND MAMMALIAN PHYSIOLOGY
Potentials for Studies of Eleven Species Other Than Protons
Whereas MRI and fMRI have been based on imaging proton spin density and intrinsic tissue relaxation rates as well as injected contrast-based relaxation rate changes, a major medical science window is opened by studies of other nuclei such as the spin ½ and spin 3/2 nuclei of carbon-13, oxygen-17, sodium-23, phosphorus-31, potassium-39, and other nuclei present in the mammalian body (see Table 4.1) along with the resonant frequencies at 20 T. Note that many of the anticipated problems for proton studies at 20 T disappear for the other nuclei listed, as they have lower gyromagnetic ratios, hence lower NMR frequencies. Physics of NMR for low gamma nuclei shows the time to acquire equivalent SNR data at 20 T will be reduced by a factor of 8 from that at 7 T and of 33 from that at 3 T, and spectral dispersion and relaxation time changes will allow investigations of metabolites in vivo that cannot be observed by any other method. As shown in Table 4.1, the sensitivities for detection of these nuclei of interest are very much lower than the sensitivity of protons. Moreover, not shown are the abundances in tissues of these nuclei, so it is important to adjust our expectations for applications based on knowledge of the local concentration of nuclei of interest. For example, sodium concentration within cells is 10 M and in extracellular fluid is 130 mM,
TABLE 4.1 Resonant Frequencies of Relevant NMR Nuclei at 20 Tesla
| Nucleus | Frequency (MHz) | Sensitivity | Spin |
| 1H | 852 | 1.00 | ½ |
| 7Li | 331 | 0.29 | |
| 13C | 214 | 0.02 | ½ |
| 15N | 184 | 0.001 | ½ |
| 17O | 115 | 0.03 | |
| 23Na | 225 | 0.09 | |
| 31P | 345 | 0.07 | ½ |
| 37Cl | 83 | 0.03 | |
| 39K | 40 | 0.0005 | |
| 57Fe | 28 | 0.00003 | ½ |
| 63Cu | 226 | 0.09 | |
| 67Zn | 53 | 0.003 | |
| 87Rb | 279 | 0.175 | |
but intracellular potassium is 150 mM and extracellular potassium is 5 mM. This information, plus an assumption that the SNR is proportional to field strength, gyromagnetic ratio, and the square root of time, allows us to calculate the time and resolution obtainable.
Carbon-13 Spectroscopy
13C MRS has great promise because of the richness of information to be gained by in vivo quantification of metabolites and the dynamics of metabolism. A principal advantage of 13C spectroscopy is the fact that the chemical shifts are 20 times those of proton spectroscopy; however, 13C’s low natural abundance of 1.1 percent has led to the need to inject large quantities of 13C-labeled substrates. Although hyperpolarized compounds offer significant improvements in detection, there are clear needs to improve the sensitivity for detecting 13C-labeled substrates limited to their Boltzmann magnetization. New insights into neurochemistry have emerged from the ability to study the metabolism of labeled compounds such as glucose and the kinetics of major brain neurotransmitters such as glutamate and GABA, while the synthesis and metabolism of other compounds such as glutamine and choline are of compelling importance in understanding the behaviors of many tumors. Measurements of glycogen production and use are of great interest in metabolic studies of the brain, muscle, and liver. However, at its natural abundance, measurements of glycogen levels are possible but currently take too long and are poorly resolved.
Oxygen-17 for Oxygen Utilization in Brain and Heart
17O is a stable isotope with a nuclear spin quantum number of 5/2 and is detectable in vivo although its natural abundance is only 0.037 percent, which is almost 30 times lower than that of 13C and about 3,000 times lower than that of protons. The gyromagnetic ratio is 7.4 times lower than that of 1H; thus the resonance frequency at 20 T is 115 MHz. The 17O nucleus possesses a quadrupolar moment that can interact with local electric field gradients. This mechanism leads to very short T1, T2, and T2* values (Zhu et al., 2005). In addition, relaxation times are field independent, so that 17O sensitivity gain with higher magnetic fields will enable imaging the dynamics of H 17O in vivo at 9.4 T and above (Zhu et al., 2001; 2 Atkinson and Thulborn, 2010). This is a new method for noninvasive measurement of cerebral metabolism of oxygen.
Sodium, Potassium, and Chloride Ions
The importance of clinical studies of the local concentrations of sodium and potassium is a compelling reason for making available wide-bore magnets with field strengths significantly greater than available currently. The transmembrane [Na+] gradient is one of the major reservoirs of metabolic energy. Detecting and measuring this gradient in vivo have been an unattainable goal of importance not only to fundamental biology but also to clinical issues such as mental disorders; tumor biology and response to therapy; heart failure; and diseases of the lungs and kidneys. Theoretically, sodium and potassium can be imaged in human subjects (Parrish et al., 1997) and experience with imaging at low fields and at 9.4 T has been reported (Thulborn et al., 2009; Kopp et al., 2012). However, because these studies have not enabled quantification of intracellular versus extracellular sodium, detection of [Na+] gradients has not been achieved except in limited experiments. Using multiple quantum NMR techniques, it has been shown that the concentrations of intracellular sodium and extracellular sodium can be evaluated during normal and pathologic function (e.g., perfused heart studies) (Schepkin et al., 1996). It is possible to discriminate cation resonances using the difference in longitudinal relaxation values, which if sufficiently different in intracellular and extracellular environments allow simple inversion recovery pulse sequences (e.g., potassium in perfused heart studies) (Kuki et al., 1990). The most recent 21.1 T rodent studies of brain sodium and proton diffusion demonstrate the potentials at high field to study sodium physiology in the live animal (Schepkin et al., 2010).
Twenty years ago the concept of 23Na relaxographic imaging was introduced to intracellular sodium imaging without the use of spectral shift reagents, most of which are toxic (Labadie et al., 1994). More recently, separation of signals representing intracellular and extracellular sodium has been demonstrated using longitudinal relaxometry on in vitro samples at 9.4 T (Zhang et al., 2010). Resolving the Nae/Nai components in human 23Na data will represent a major accomplishment for in vivo NMR. There is even the possibility for determining the instantaneous, homeostatic flux of active trans-membrane Na+ cycling (Charles Springer, personal communication).
An important potential clinical application is the investigation of migraine headaches, which affect 30 million U.S. citizens. Evidence for the importance of sodium extracellular concentration changes in migraine headache research comes from findings of an increase in [Na+] in cerebrospinal fluid of patients (Harrington et al., 2006) and the MRI demonstration of a significant increase in [Na+] in the brains of a rodent model of migraine at 21.1 T (Harrington et al., 2011). These are corroborated by the demonstration of increased excitability of neurons with increases of extracellular [Na+] (Arakaki et al., 2011).
Other recent studies have highlighted other compelling potential applications
for Na, Cl, and K imaging, including provocative results suggesting that in hypertension and renal disorders, there can be a chronic accumulation and storage of ions within tissues such as skin (Titze, 2003), so that interstitial and intravascular levels of sodium are not the same. This has important implications for therapy in renal disease and the management of hypertension (Kopp, 2012), a condition that affects 30 percent of U.S. adults. Although sodium imaging at 3 T has been used to investigate these phenomena, higher fields would allow better resolution, shorter imaging times, and, most importantly, the ability to measure levels of the other relevant ions, chlorine and potassium.
The promise of imaging sodium, potassium, and chlorine such that, in principle, estimates can be made of the resting membrane potentials of the brain and heart in health and disease depends on the availability of fields approaching 20 T.
Phosphorus-31 Spectroscopy at High Fields
Though the frequency of 31P at 20 T will present penetration and homogeneity problems greater than other spins, these problems are well understood from experience with 1H at 7 T (300 MHz). The promise for high-field chemical exchange saturation transfer (31P CEST) is that it could resolve all adenosine triphosphate (ATP), adenosine diphosphate (ADP), and adenosine monophosphate (AMP) signals and thus become an efficient, noninvasive diagnostic tool in heart disease. In addition, 13C-CEST has the potential to redefine metabolic pathway phenotypes in human beings. Furthermore, 20 T will allow measurement of pH, redox, metabolite levels, temperature, reactive oxygen species (ROS), singlet oxygen, and others through the use of PARACEST agents that have exchange rates too fast at lower fields (Cherry and Malloy, University of Texas Southwestern Medical Center, personal communication, 2012).
Selected Horizons for Proton Studies at 20 T
Anatomical Imaging and Spectroscopy Advantages
Although radio-frequency penetration for studying proton relaxation differences and chemical shift spectroscopy currently experiences engineering design difficulties up to the highest human magnet fields of 11.7 T, the benefits of increases in sensitivity, anatomic resolution, and spectroscopy dispersion motivate proton studies at 20 T. In animals including nonhuman primates, cortical anatomic imaging at 7 T and 9.4 T is routinely accomplished with spatial resolutions of about 100 μm and with fMRI resolutions of about 300 μm. In the human at 7 T the attainable spatial and fMRI resolutions are currently 500 μm and 1,000 μm, respectively.
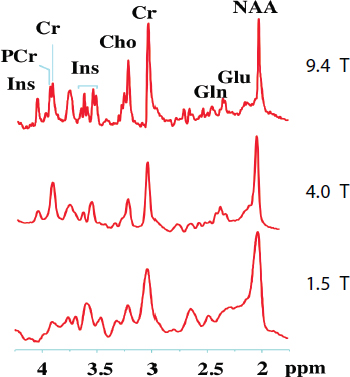
Figure 4.3 Comparison of the proton spectroscopy of a volume element of the brain that demonstrates the spectral resolution improvement in going from 1.5 T to 4 T to 9.4 T. The peaks of several of the compounds, such as glutamate (Glu) and glutamine (Gln), are just resolved at 9.4 T. SOURCE: Data courtesy of Michael Garwood, University of Minnesota, adapted from R. Gruetter, S.A. Weisdorf, V. Rajanayagan, M. Terpstra, et al., 1998, Resolution improvements in in vivo H NMR spectra with increased magnetic field strength, Journal of Magnetic Resonance 135:26-264.
There would be considerable value to being able to routinely image cortex with resolutions 2-4 times smaller—for example, to visualize cortical columns and cortical layers.
Experiments on small animals have resolutions of 50 μm at 21.1 T (Schepkin et al., 2010). Detailed anatomy, fMRI, and spectroscopic studies such as shown for lower fields in Figure 4.3 motivate higher fields than 7 T for proton MRI. One major important clinical goal would be to better understand dementia. Spectroscopic studies of the surface of the human heart for studies of congestive heart failure will most likely emphasize 13C and 31P. The gains in sensitivity for the lower gamma spins discussed above are substantially greater than will be experienced by the University of Minnesota when, as planned, it doubles the field of its human imaging magnet to 10.5 T. The dispersion increase can enable metabolic studies heretofore not possible.
Functional MRI (fMRI) at 20 T
During the past 20 years a method of mapping (imaging) the metabolic activity of the brain in response to activation uses signal changes associated with changes
in oxy- and deoxyhemoglobin concentrations. This technique, known as the BOLD (blood oxygen level dependent) technique (Ogawa et al., 1992), or fMRI, has opened new horizons in the cognitive sciences and neurophysiology (Ugurbil et al., 1999). Development of high-field MRI, such as 7 T, is now the high-end research platform in neurosciences with the goal of studying the fundamental computational units that reside in submillimeter organizations (Ugurbil, 2012). Imaging these units and their connectivity employs functional MRI that provides regional information on the neuronal activity changes in the brain. The feasibility of this goal at 7 T was demonstrated by imaging noninvasively the ocular dominance columns (Yacoub et al., 2007). However, magnetic fields much higher than 7 T are needed to achieve the SNR and data acquisition times required to decipher the neural code at the scale of fundamental computations. Even though “physiological noise” increases at high magnetic fields (Triantafyllou et al., 2005), for high-resolution imaging the noise in an fMRI time series is dominated by thermal noise; thus, the effective signal-to-noise ratio for fMRI will increase at least linearly with magnetic fields.
Though functional MRI uses proton frequencies of 852 MHz, penetration to depths of 3 cm in the human skull is not expected to be a problem at 20 T. Studies of RF safety for cell phone frequencies (which are 2 to 3 times higher) show the RF field can penetrate through bone and tissues to and beyond the cortex. In addition, fMRI is an approach that requires minimal power deposition and should be feasible even at 20 T. The main technical challenges of performing fMRI at high magnetic field strengths have been solved for 7 T, and currently the whole brain can be imaged in subsecond intervals (Moeller et al., 2010; Feinberg et al., 2010). Potential future applications using new rapid acquisition techniques include whole-brain connectivity analysis, including the dynamics of brain networks as recently demonstrated (Smith et al., 2012).
Chemical Exchange Saturation Transfer Horizon
One of the most important applications of 20 T is in the use of CEST, as it will allow detection of exchangeable -NH protons or -OH protons within cells and, for example, the imaging of liver glycogen (Sherry and Woods, 2008). Combined proton and 13C studies of lipid and amino acid metabolism in vivo in human subjects relative to nutrition, obesity, and diabetes is another area enabled by high-field studies of muscle and adipose tissue of the limbs.
New Opportunities for Exogenous Contrast Agents
Imaging the distribution of safe stable-isotope-based compounds at very high fields will open new horizons in the applications of contrast-enhanced MRI. The advances in MRI clinical applications have been enabled partly by advances in the
design of paramagnetic contrast agents such as those using gadolinium. When these agents are in the intravascular blood pool, they allow visualization of the vascular tree analogous to X-ray angiography, because the presence of the agent reduces the T1 relaxation of water protons in the blood. If a tissue region has increased permeability such that more contrast agent accumulates in that region (e.g., breast or brain tumor), there will occur a temporal decrease in the local T1 (increase in tissue water relaxation rate). This allows identification of different tissue pathologies by MRI imaging methods used to bring out signals related to the relative decrease in T1. The barrier to improving sensitivity of the injected compounds is the inability to effectively restrict fast local motions of contrast agents attached to slow-tumbling scaffolds.
Synthesis of new agents to reach the maximum sensitivity is theoretically attainable at 20 T because the slow-tumbling requirement does not apply. For T2 and fluorine agents, sensitivity can be increased by at least an order of magnitude compared to current experience at clinical field strengths of 3 T. This translates to being able to image targets at subnanomolar concentrations (e.g., cell surface receptors).
Metals other than gadolinium become competitive in terms of sensitivity at 20 T because their fast electronic relaxation times no longer represent a limitation. Consequently, completely new classes of contrast agents become possible. In addition, the increased Curie spin of paramagnetic ions may allow for new ways of engineering more sensitive contrast agents. For PARACEST agents, the larger chemical shifts at 20 T would make multiplexing less challenging.
Proton Transverse Relaxation Rate Changes with Field
The anomalous behavior of the proton transverse relaxation rate (R2 = 1/T2) per gram of protein solutions in water as a function of the measurement field to 7 T (300 MHz) and beyond is shown in Figure 4.4. The implication is that although R2 decreases monotonically as field strength increases to 3 T (as does R1), consistent with the theory of a gradual fall in effectiveness of inter- and intramolecular dipole-dipole couplings that promote relaxation, some other mechanism(s) lead to an increase in R2. The primary candidate for this relaxation is chemical exchange between labile protons (notably -OH and -NH, -NH2) and bulk water, which occurs at rates that depend on molecular structure, pH, and other factors and for which a quadratic field dependence may be predicted. Thus, Figure 4.4 suggests that the contrast mechanism of T2-weighted proton imaging is different than commonly observed at 3 T or at lower fields.
The underlying molecular properties that give rise to this contrast may be sensitive to a completely different set of influences than those known at lower fields. The transition to contrast that reflects chemical exchange is of particular relevance
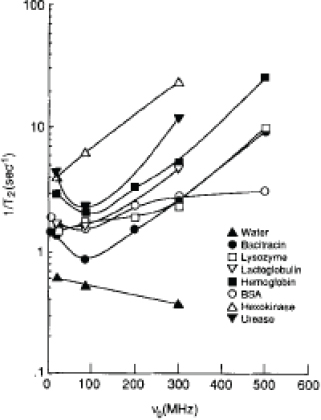
FIGURE 4.4 Transverse relaxation rates for protein solutions as a function of proton magnetic resonance frequencies. SOURCE: Reprinted with permission from John Wiley and Sons, Inc., J. Zhong, J.C. Gore, and I.M. Armitage, 1989, Contributions of chemical exchange and other relaxation mechanisms in protein solutions and tissues, Magnetic Resonance in Medicine 11(3):295-308. Copyright 1989 Wiley-Liss, Inc., A Wiley Company.
to new classes of molecular imaging techniques, including CEST and T1rho imaging. The manifestation of this mechanism will be at high magnetic fields.
Enhanced Contrast from Susceptibility Differences
Suppose two adjacent tissues have slightly different susceptibilities. This will result in a change in homogeneity that will increase linearly with field:
![]()
This local magnetic flux change leads to a signal phase difference over time:
![]()
The phase difference can lead to image distortions and voids due to phase cancellation effects. The effects noted at 4 T and 7 T will be greater by factors of more than 2 at 20 T. However, the distortion can be viewed as an effective contrast enhancement
depending on the amount of susceptibility difference and the volume of the tissue. This concept is already used at lower fields in susceptibility-weighted imaging, a technique that modulates the MRI signal intensity by local phase shifts to enhance vascular and other features. Moreover, tissue layers or domains having dimensions of tens of microns and small susceptibility differences from adjacent tissues might be visualized at higher fields than currently available. Animal experiments at very high fields can evaluate the extent of the benefits as well as problems of susceptibility differences between adjacent tissues because large differences in susceptibility can exist between paramagnetic tissues (e.g., ferritin-containing tissues) and adjacent normal diamagnetic tissues. The anisotropic magnetic susceptibility of neural tissues has already led to the development of imaging methods of the susceptibility tensor, from which new methods for mapping neural connectivity are emerging. Susceptibility anisotropy within macromolecules and assemblages of molecules is discussed in Appendix F.
Some of the potential benefits are related to the image contrast that results from bulk magnetic susceptibility differences in adjacent tissues due to compounds such as ferritin and myelin, both of which are found throughout brain tissue. In addition, the relative directional orientation of bundles of nerve fibers relative to the B0 field will give an associated frequency shift that translates to image contrast, as shown in Figure 4.5.
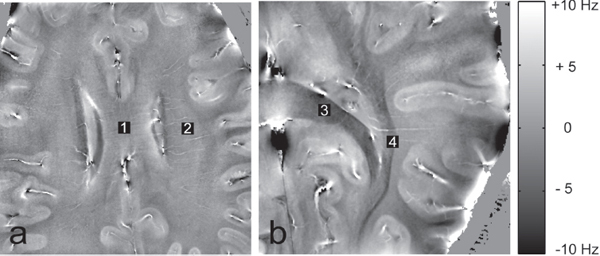
FIGURE 4.5 Contrast based on frequency differences from MRI at 7 T depends on susceptibility differences between adjacent tissues. In (a), the specimen is orientated with fiber tracks parallel to the B0 field and in (b), the tracks are oriented both parallel and orthogonal to the B0 field. The resolution is 0.25 × 0.25 mm for a 2 mm slice. SOURCE: J. Lee, K. Shmueli, M. Fukunaga, P. van Gelderen, H. Merkle, A. C. Silva, and J.H. Duyn, 2010, Sensitivity of MRI resonance frequency to the orientation of brain tissue microstructure, Proceedings of the National Academy of Sciences 107(11):5130-5135. Copyright 2010 National Academy of Sciences, U.S.A.
Potentials for Traveling Wave MRI at
Short-Wavelength Proton Magnetic Resonance
As mentioned above, at very high fields (when RF wavelengths are shortened) traveling waves (e.g., Webb et al., 2010) may be used to excite nuclei and detect their NMR signals using remote coils that are no longer closely coupled to the subject. This development suggests that a new class of experimental techniques may be developed, introducing some of the concepts of image formation from coherent optics such as holographic imaging and interferometry. There will continue to be complications from the nonuniform distributions of relevant electromagnetic properties of tissues, but these may also open up opportunities for mapping such properties as new forms of tissue contrast. For example, measuring the nonuniform distributions of RF fields at 7 T has provided preliminary images of distributions of tissue conductivity and permittivity, and at higher fields these variations should be more easily distinguished. Traveling wave methods may be adapted for such acquisitions, and the fact that image data can be detected at a distance from the object provides a compelling reason to continue to develop these methods at higher fields.
Interactions of High Magnetic Fields with Imaging Gradients
Because of the well-known frequency vs. resistance and inductance dependencies, one expects a smooth variation of impedance vs. frequency, but at higher fields the Lorentz forces due to current flow in the gradient coils within the magnetic field not only cause loud acoustic noise but also result in a frequency-dependent resistance change (Schmitt et al., 2006). The emf generated by moving conductors in the magnet oppose the voltage of the gradient power amplifier. This phenomenon will be more problematic at 20 T than experienced at 7 T even though nuclei of lower gyromagnetic ratios than those of protons are being studied because the effect is dependent on field and gradient strength rather than NMR frequency.
As magnetic resonance uses radio-frequency fields to excite nuclei, there are consequences from the interactions of the RF fields and the dielectric and resistive properties of the body (e.g., permittivity and conductivity) that vary with frequency and with tissue type. These effects alter the B1 transmitted field and spatially modulate the sensitivity of coils in reception, leading to spatial inhomogeneities (Kangarlu et al., 1999; Roschmann, 1987). At RFs of 300 MHz, the effective
wavelength in human tissue such as the brain with a dielectric coefficient of about 60 is 10 cm, so the wavelength is no longer larger than the object, leading to standing wave and interference effects. These can result in serious imaging artifacts. It is unknown how well one can deal with this problem at 20 T for protons at 852 MHz, where the limited penetration of RF at these frequencies is also more severe. Fortunately, previous studies at 7 T have coped with this problem for the proton frequency range of 300 MHz, and this range is appropriate for many of the other nuclei listed in Table 4.1 for 20 T.
Gradient Amplitude Requirements for Low γ Nuclei
Whereas very high fields open opportunities for imaging nuclei other than protons because the frequencies allow body penetration and to some extent reduce the RF power deposition, there is a penalty: The spatial resolution requires a spatially dependent frequency shift, and the magnitude of this shift is dependent on the product of the gyromagnetic ratio and the gradient. But this is not a serious consequence, because volumetric resolutions 10-50 times less than enjoyed from proton imaging can provide metabolic information not available by any other noninvasive method.
If magnet installations do not use active shielding, between 400 and 800 tons of iron are required for setting up the magnet site. In 2010, Varian completed an actively shielded 7 T system in Palo Alto, California. The feasibility, engineering, and costs for an actively shielded 20 T installation should be part of a design study.
The effects of fields and magnetic field gradients on sensory functions, the induction of E-fields from switched gradients, and absorbed power from the RF fields are discussed in Appendix F.
FINDING, CONCLUSION, AND RECOMMENDATION
Finding: The development of magnetic resonance imaging, functional magnetic resonance imaging, and magnetic resonance spectroscopy in human studies since 1973 has led to major new human physiology information and significant improvements in diagnoses and treatments. The fields employed for human studies have increased from 0.04 T to 11.74 T over the last 40 years, but other possibilities would be opened by still higher fields.
Conclusion: The current barriers in MRI medical science research would seem to call for an initiative to develop a 20 T magnet with capabilities to image and perform spectroscopy on the human head, large animals, and plants. Although this development would be for research and not for clinical applications, the research could lead to important clinical benefits.
The physics of NMR leads to an expectation that resolutions of 50 μm will be readily obtained in a field of 20 T, and the time to acquire equivalent SNR data will be reduced by a factor of 8 from that at 7 T and of 33 from that at 3 T. Changes in spectral dispersion and relaxation times will allow investigations of metabolites in vivo that cannot be observed by any other methods. A further horizon opened by 20 T is that of imaging nuclei such as 13C, 15N, 17O, 23Na, 31P, 37Cl, 39K, and nuclei other than 1H.
Limitations from absorbed power of RF frequencies restrict the depth of penetration for proton MRI and MRS, but this is not true for the other nuclei as the resonance frequencies of these nuclei at 20 T are in the range of currently successful proton MRI and MRS at lower fields. Potential human health effects at 20 T are not expected to go beyond temporary discomfort such as dizziness and short-term performance deficits. These observations from human subjects at fields up to 9.4 T do not present insurmountable barriers to safe studies. Continuation of human behavioral studies and animal research in small-bore magnets up to 20 T will lead to a better understanding of the mechanisms underlying the reversible symptoms and signs in animals and human subjects (cf. Appendix F).
A promising area will be 13C in vivo spectroscopy because the polarization from higher fields than currently available for in vivo studies will allow direct detection of metabolites we cannot monitor at lower fields.
Another important application of 20 T is in the use of CEST, as it will allow detection of almost any exchangeable -NH protons or -OH protons within cells, thereby allowing, for example, imaging of liver glycogen. Heart disease is the major cause of death in North America. High-field MRS in human subjects using 31P CEST can unlock the tissue and energy mysteries of heart failure (e.g., resolution of all ATP, ADP, and AMP signals). Studies using 13C-CEST have the potential to redefine metabolic pathway phenotypes in human beings. In addition, 20 T will allow measurement of pH, metabolite levels, temperature, reactive oxygen species, singlet oxygen, etc. through the use of PARACEST agents that have exchange rates too fast to detect at lower fields.
At 20 T, imaging the human cortex using proton MRI at 50-μm resolution will be possible even though RF penetration at 852 MHz is limited to a few centimeters. The susceptibility differences between Alzheimer’s plaques and adjacent tissues size should allow visualization of plaque-invested tissues even for particles of 20-μm
size. fMRI studies at 7 T give confidence that fMRI at 20 T and new rapid acquisition techniques will allow nearly whole-brain connectivity analyses.
Recommendation: A design and feasibility study should be conducted for the construction of a 20 T, wide-bore (65 cm diameter) magnet suitable for large animal and human subject research. The required homogeneity is 1 ppm or better over a 16 cm diameter sphere. The appropriate sponsorship might be multiple agencies (e.g., NIH, NSF, and DOE). In parallel, an engineering feasibility study should be undertaken to identify appropriate radio frequency, gradient coils, and power supplies that will enable MRI and MRS and an extension of current health and safety research currently being conducted at lower fields.
Arakaki X., H. Foster, L. Su, H. Do, A.J. Wain, A.N Fonteh, F. Zhou, and M.G. Harrington. 2011. Extracellular sodium modulates the excitability of cultured hippocampal pyramidal cells. Brain Research 1401:85.
Atkinson, I.C., and K.R. Thulborn. 2010. Feasibility of mapping the tissue mass corrected bioscale of cerebral metabolic rate of oxygen consumption using 17-oxygen and 23-sodium MR imaging in a human brain at 9.4 T. Neuroimage 51:723.
Barfuss, H., H. Fisher, D. Hentschel, R. Ladebeck, and J. Vetter. 1988. Whole-body MR imaging and spectroscopy with a 4T system. Radiology 169:811.
Bomsdorf, H., T. Helzel, D. Kunz, P. Roschmann, O. Tschendel, and J. Wieland. 1988. Spectroscopy and imaging with a 4 tesla whole-body MR system. NMR in Biomedicine 1(3):151-158.
Dula, A.N, E.B. Welch, J.L Creasy, J.C. Gatenby, E.A. Stringer, L.M. Chen, A.W. Anderson, M.J. Avison, and J.C. Gore. 2010. Challenges and opportunities of ultra-high field MRI. The Third International Conference on the Development of Biomedical Engineering in Vietnam IFMBE Proceedings 27:1-5.
Feinberg, D.A., S. Moeller, S.M. Smith, E. Auerbach, S. Ramanna, M. Gunther, M.F. Glasser, K.L. Miller, K. Ugurbil, and E. Yacoub. 2010. Multiplexed echo planar imaging for sub-second whole brain fMRI and fast diffusion imaging. PLoS ONE 5: e15710.
Harrington, M.G., A.N. Fonteh, R.P. Cowan, K. Perrine, J.M. Pogoda, R.G. Biringer, and A.F. Hühmer. 2006. Cerebrospinal fluid sodium increases in migraine. Headache 46:1128.
Harrington, M.G., E.Y. Chekmenev, V. Schepkin, A.N. Fonteh, and X. Arakaki. 2011. Sodium MRI in a rat migraine model and a NEURON simulation study support a role for sodium in migraine. Cephalalgia 31:1254.
Kangarlu, A., R.E. Burgess, H. Zhu, T. Nakayama, R.L. Hamlin, A.M. Abduljalil, and P.-M.L. Robitaille. 1999. Cognitive, cardiac and physiological safety studies in ultra high field magnetic resonance imaging. Journal of Magnetic Resonance Imaging 17:1407.
Kopp, C., P. Linz, L. Wachsmuth, A. Dahlmann, T. Horbach, C. Schofl, W. Renz, D. Santoro, T. Niendorf, D.N. Muller, M. Neininger, A. Cavallaro, K.U. Eckardt, R.E. Schmieder, F.C. Luft, M. Uder, and J. Titze. 2012. 23Na magnetic resonance imaging of tissue sodium. Hypertension 59:167.
Kuki, S., E. Suzuki, H. Watari, H. Takami, H. Matsuda, and Y. Kawashima. 1990. K-39 nuclear magnetic resonance observation of intracellular potassium without chemical shift reagents during metabolic inhibition in the isolated perfused rat heart. Circulation Research 67:401.
Labadie, C., J.-H. Lee, G. Vetek, and C.S. Springer. 1994. Relaxographic imaging. Journal of Magnetic Resonance B 105:99.
Lauterbur, P.C. 1973. Image formation by induced local interactions: Example employing magnetic resonance. Nature 242:190.
Moeller, S., E. Yacoub, C.A. Olman, E. Auerbach, J. Strupp, N. Harel, and K. Ugurbil. 2010. Multiband multislice GE-EPI at 7 Tesla, with 16-fold acceleration using partial parallel imaging with application to high spatial and temporal whole-brain fMRI. Magnetic Resonance in Medicine 63:1144.
Ogawa, S., D.W. Tank, R. Menon, J.M. Ellermann, S.G. Kim, H. Merkle, and K. Ugurbil. 1992. Intrinsic signal changes accompanying sensory stimulation: Functional brain mapping with magnetic resonance imaging. Proceedings of the National Academy of Sciences U.S.A. 89:5951.
Pang, Y., D.B. Vigneron, and X. Zhang. 2011. Parallel traveling-wave MRI: A feasibility study. Magnetic Resonance in Medicine 67:965.
Parrish, T.B., D.S. Fieno, S.W. Fitzgerald, and R.M. Judd. 1997. Theoretical basis for sodium and potassium MRI of the human heart at 1.5T. Magnetic Resonance in Medicine 38:653.
Robitaille, P.M.L., A.M. Abduljalil, A. Kangarlu, X. Zhang, Y. Yu, R. Burgess, S. Bair, P. Noa, L. Yang, H. Zhu, Z. Jiang, R.E. Bailey, W. Chung, Y. Somawiharja, P. Feynan, and D. Rayner. 1999. Design and assembly of an 8 Tesla whole body scanner. Journal of Computer Assisted Tomography 23:808.
Robitaille, P.M.(ed.). 2008. Ultra High Field Magnetic Resonance Imaging. Springer-Verlag, Berlin, Heidelberg, New York.
Roschmann, P. 1987. Radiofrequency penetration and absorption in the human body—Limitations to high field in whole-body nuclear magnetic resonance imaging. Medical Physics 14:922.
Schenck, J.F., C.L. Dumoulin, R.W. Redington, H.Y. Kressel, R.T. Elliot, and I.L. McDougall. 1992. Human exposure to 4.0-Tesla magnetic fields in a whole-body scanner. Medical Physics 19:1089.
Schepkin, V.D., U.O. Choy, and T.F. Budinger. 1996. Sodium alterations in isolated rat heart during cardioplegic arrest. Journal of Applied Physiology 81:2696.
Schepkin, V.D., W.W. Brey, P.L. Gor’kov, and S.C. Grant. 2010. Initial in vivorodent sodium and proton MR imaging at 21.1 T. Magnetic Resonance Imaging 28:400.
Schmitt, F.A., F. Potthast, C. Stoeckel, C.J. Triantafyllo, G. Wiggins, and L. Wiggins. 2006. Aspects of clinical imaging at 7T. Pp. 59-103 in Ultra High Field Magnetic Resonance Imaging. Springer-Verlag, Berlin, Heidelberg, New York.
Sherry, A.D., and M. Woods. 2008. Chemical exchange saturation transfer contrast agents for magnetic resonance imaging. Annual Review of Biomedical Engineering 10:391.
Smith, S.M., K.L. Miller, S. Moeller, J. Xu, E.J. Auerbach, M.W. Woolrich, C.F. Beckmann, M. Jenkinson, J. Andersson, M.F. Glasser, D.C. Van Essen, D.A. Feinberg, E.S. Yacoub, and K. Ugurbil. 2012. Temporally-independent functional modes of spontaneous brain activity. Proceedings of the National Academy of Sciences U.S.A. 109:3131.
Titze, J., R. Lang, C. Ilies, K.H. Schwind, K.A. Kirsch, P. Dietsch, F.C. Luft, and K.F. Hilgers. 2003. Osmotically inactive skin Na+ storage in rats. American Journal of Physiology and Renal Physiology 285:F1108.
Thulborn, K.R., A. Lu, I.C. Atkinson, F. Damen, and J.L. Villano. 2009. Quantitative sodium MR imaging and sodium bioscales for the management of brain tumors. Neuroimaging Clinics of North America 19:615.
Triantafyllou, C., R.D. Hoge, G. Krueger, C.J. Wiggins, A. Potthast, G.C. Wiggins, and L. Wald. 2005. Comparison of physiological noise at 1.5T, 3T, and 7T and optimization of MRI acquisition parameters. NeuroImage 26:243.
Ugurbil, K., M. Garwood, J. Ellermann, K. Hendrick, R. Hinke, X. Hu, S.G. Kim, R. Menon, H. Merkle, S. Ogawa, and R. Salmi. 1993. Imaging at high magnetic fields: Initial experiences at 4T. Magnetic Resonance Quarterly 9:259.
Ugurbil, K., X. Hu, W. Chen, X.-H. Zhu, S.-G. Kim, and A. Georgopoulos. 1999. Functional mapping in the human brain using high magnetic fields. Philosophical Transactions of the Royal Society 354:1195.
Ugurbil, K. 2012. The road to functional imaging and ultrahigh fields. NeuroImage 62:726.
Webb, A.G., C.M. Collins, M.J. Versluis, H.E. Kan, and N.B. Smith. 2010. MRI and localized proton spectroscopy in human leg muscle at 7 tesla using longitudinal traveling waves. Magnetic Resonance in Medicine. 63:297.
Yacoub, E., A. Shmuel, N. Logothetis, and K. Ugurbil. 2007. Robust detection of ocular dominance columns in humans using Hahn Spin Echo BOLD functional MRI at 7 tesla. Neuroimage 37:1161.
Zhang, Y., M. Poirer-Quinot, C.S. Springer, Jr., and J.A. Balschi. 2010. Discrimination of intra- and extracellular 23Na+ signals in yeast cell suspensions using longitudinal magnetic resonance relaxography. Journal of Magnetic Resonance 205:28.
Zhu, X.H., H. Merkle, J.H. Kwag, K. Ugurbil, and W. Chen. 2001. 17O relaxation time and NMR sensitivity of cerebral water and their field dependence. Magnetic Resonance in Medicine 45:543.
Zhu, X.H., N. Zhang, Y. Zhang, X. Zhang, K. Ugurbil, and W. Chen. 2005. In vivo 17O NMR approaches for brain study at high field. NMR in Biomedicine 18:83.




















