Maternal, Fetal, and Neonatal Mortality
In many low-income countries, the maternal mortality rate (defined as the death of the mother during pregnancy or within 42 days of the birth) is 100 times greater than in high-income countries. Rates of stillbirth (defined as death in utero after 28 weeks of pregnancy) and neonatal mortality (defined as the death of a live-born baby within 28 days of life) are often 10 times greater or more than in high-income countries (Goldenberg and McClure, 2011; Ronsmans and Graham, 2006).1 The current rates in low-income countries are generally similar to the rates in high-income countries 80 years ago.
This chapter reviews the medical causes of maternal, fetal (stillbirth), and neonatal mortality in low-income countries; describes the similarities in these causes; and explores why the rates of each outcome have fallen in high-income countries. It then reviews the interventions proposed to reduce these mortalities.
Causes of Maternal, Fetal, and Neonatal Mortality in Low-Income Countries
In most developing countries, the major medical causes of maternal mortality are hemorrhage, the hypertensive diseases of pregnancy, and various types of maternal infections, among other things (IOM, 2003; Khan et al., 2006; Ronsmans and Graham, 2006; WHO and UNICEF, 2012)—see Figure 3-1. Hemorrhage (heavy bleeding), which is often categorized as antepartum (before delivery) or postpartum (after delivery), has many causes. The most common antenatal cause is premature placental separation (placental abruption), and the most common postpartum cause is failure of the uterus to contract after delivery—often called uterine atony. Women in their first pregnancy are more likely than those in subsequent pregnancies to develop a form of high blood pressure called preeclampsia. This condition may lead to a number of potentially deadly complications, including strokes and seizures (eclampsia). Women with obstructed or prolonged labor are more likely to die from hemorrhage and infection, as are women who undergo unsafe abortions.
Fetal mortality often occurs secondary to the hypertensive diseases of pregnancy, placental abruption associated with maternal hemorrhage, and various types of maternal infection, including syphilis and malaria (Black et al., 2010; Lawn et al., 2011)—see Figure 3-2. Most fetal deaths are due to asphyxia, or a reduction in the oxygen available to the fetus. Hypertensive diseases, abruption, prolonged labor, breech presentation (feet or buttocks presenting first), and umbilical cord accidents are often precursors of this type of mortality (Lawn et al., 2011).
Neonatal mortality in low-income countries is typically attributed to one of three major causes: infection, asphyxia, or prematurity (birth prior to 37 weeks gestation out of 40 weeks)—see Figure 3-3 and Lawn, Cousens, and Zupan (2005); Lawn, Wilczynska-Ketende, and Cousens (2006); and Black et al. (2010). It is crucial to understand that many of these deaths are either caused by one of the maternal con-
1Chapter 2 referred to maternal, infant, and neonatal morality rates. This chapter refers to maternal, fetal, and neonatal mortality rates because, although fetal deaths are not referenced in the Millennium Development Goals, the lives of unborn fetuses can be saved with some of the same measures as mothers and newborns.
ditions just described or are preventable by a maternal treatment (Lawn, Cousens, and Zupan, 2005). For example, neonatal deaths due to syphilis are preventable by maternal screening and treatment, and neonatal tetanus is preventable by maternal immunization. Most cases of neonatal asphyxia are preventable by the appropriate intrapartum care, especially if the mother has hypertensive disease or suffers a hemorrhage. The conditions that cause maternal, fetal, and neonatal mortality are frequently the same, and, as will be shown later, interventions that reduce one type of mortality often reduce the others.
History of Outcome Improvements in High-Income Countries
The reductions in maternal, fetal, and neonatal mortality in high-income and many middle-income countries have been impressive (see Figures 3-4, 3-5, and 3-6).
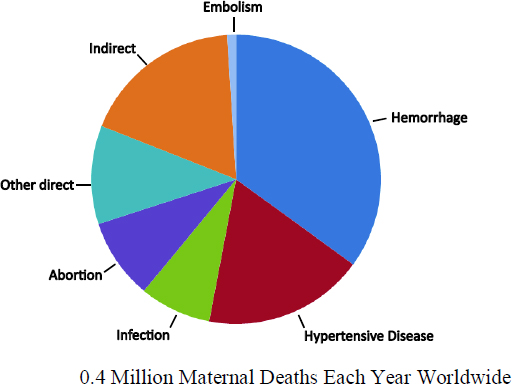
FIGURE 3-1 Main Causes of Maternal Mortality. SOURCE: Robert L. Goldenberg, as adapted from Khan et al. (2006).
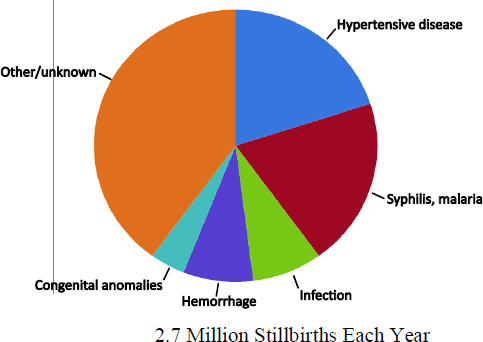
FIGURE 3-2 Main Causes of Fetal Mortality. SOURCE: Robert L. Goldenberg, as adapted from Lawn et al. (2009).
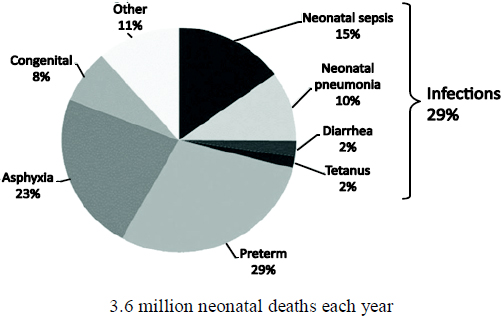
FIGURE 3-3 Causes of 3.6 Million Neonatal Deaths, 192 Countries, Based on Cause-Specific Mortality Data and Multi-cause Modeled Estimates. SOURCE: Based on data from Child Health Epidemiology Reference Group and WHO in Black et al. (2010). Reprinted from Seminar in Perinatology 34(6):371-386. Lawn, J.E., Kerber, K., Enweronu-Laryea, C., and Cousens, C. Copyright(2010), with permission of Elsevier.
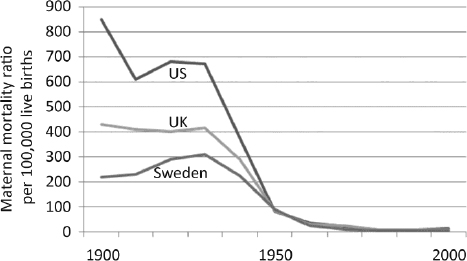
FIGURE 3-4 Maternal Mortality Ratios: United States, United Kingdom, and Sweden, 1900-2000. SOURCE: Goldenberg and McClure (2011). Reprinted from American Journal of Obstetrics & Gynecology 205(4):293-295, Goldenberg, R. L. and McClure, E.M., Copyright (2011), with permission of Elsevier.
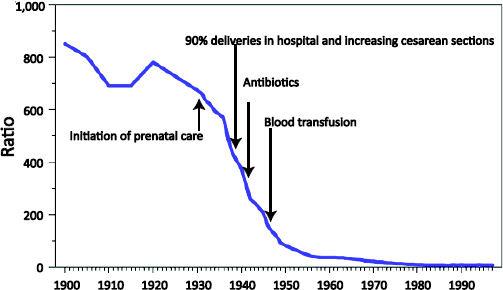
FIGURE 3-5 Maternal Mortality Ratio per 100,000 Live Births over Time and Interventions That Contributed to Decline, United States. SOURCE: Robert L. Goldenberg, adapted from Johnson (2001).
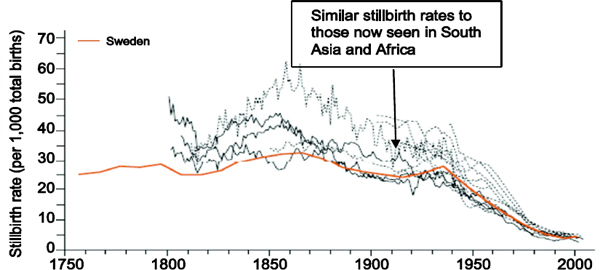
FIGURE 3-6 Long-Term Trends for Stillbirth Rates in 11 High-Income Countries, 1750-2000. SOURCE: Goldenberg et al. (2011). Reprinted from The Lancet 377(9779):1798-1805, Goldenberg, R.L., et al. Copyright (2011), with permission of Elsevier.
Maternal Mortality
In these countries, until about 1935 maternal mortality was frequently in the range of 500-1,000 deaths per 100,000 live births—that is, nearly 1 percent of mothers died with each pregnancy (Goldenberg and McClure, 2011). This situation had remained essentially unchanged for hundreds of years. In recent years, many high-income countries have reported maternal mortality ratios of about 10 per 100,000 live births, or a reduction of 99 percent (UNFPA et al., 2012). Several scholars have explored the history of this reduction, which in nearly all high-income countries began in about 1935 and was mostly completed by 1970-1980 (Figure 3-4)—see Goldenberg and McClure (2011) and Loudon (2000). The interventions responsible for the reductions in maternal mortality include the introduction of prenatal care and hospitalization for delivery, beginning in the 1930s; the availability of antibiotics to treat infection, beginning in the late 1930s and 1940s; uterotonics (drugs that cause the uterus to contract) in the late 1930s; and blood transfusion, beginning in the early 1940s (Figure 3-5).
The management of preeclampsia/eclampsia steadily improved with prenatal care to diagnose the condition, hospitalization to manage the condition, and, in 1940-1950 a change in management from watchful waiting to rapid delivery for severe or progressing disease. With antibiotics, blood availability, and improvements in anesthesia, cesarean sections became safer and were used more frequently to terminate pregnancies that threatened the life of the mother and fetus/neonate, such as those with prolonged labor or eclampsia. Thus new and effective interventions were introduced, first in high-income countries and later in some middle-income countries, and they were responsible for the nearly 99 percent reduction in maternal mortality. If these interventions were used widely and appropriately in low-income countries, there is no reason to believe that the results would not be similar to those in high-income countries.
Fetal Mortality
Until about 80 years ago in the United States and other high-income countries, fetal mortality rates generally ranged from 35 to 50 per 1,000 live births. In the 2011 Lancet series on stillbirths, nearly all high-income countries reported stillbirth rates of less than 5 per 1,000 live births, with several countries reporting rates as low as 2 per 1,000 live births, representing a reduction of approximately 95 percent (Flenady et al., 2011; Goldenberg et al., 2011). Like that for maternal mortality, the reduction in stillbirths
began about 1935, continued relatively rapidly until about 1980, and has continued more slowly until the present (Figure 3-6). Because the cause of a stillbirth is often less clear than that of a maternal death, it is somewhat more difficult to define the interventions responsible for most of the reduction. However, some of these interventions are clear. For example, in the 1920s syphilis was thought to be responsible for about 20 percent of U.S. stillbirths. Today in high-income countries, it is extremely rare for syphilis to cause a stillbirth. Eighty years ago, preeclampsia and eclampsia were the major killers of fetuses, but today they account for only a small percentage of a much smaller number of stillbirths. Stillbirths associated with placental abruption have also been much reduced. Monitoring the fetus for signs of asphyxia prenatally and during delivery and using various techniques such as fetal heart rate monitoring during delivery for signs of distress have reduced much of the asphyxia-related fetal mortality. The high rates of cesarean section and labor induction in many high-income countries are attributable in part to physicians’ concerns about stillbirth and their attempts to reduce this outcome. With appropriate care for the mother, it is possible to reduce stillbirth rates by as much as 90 percent. Overall, it is important to understand that the conditions that cause stillbirths are similar to those that cause maternal mortality, and the interventions needed to reduce stillbirths are the same as those that reduce maternal mortality.
Neonatal Mortality
Neonatal mortality in developed countries has also fallen substantially in the last 80 years. The reasons for the decline are related to the decreasing prevalence of some conditions and better treatment of others. About one-third of neonatal mortality is associated with preterm birth. In developed countries, the incidence of prematurity has, if anything, increased over the last decades, and so falling rates of preterm birth cannot be given credit for the lower mortality.
Preterm infants die from (1) respiratory distress syndrome (RDS), a condition caused by a lack of lung surfactant; (2) intraventricular hemorrhage (IVH), or bleeding into the newborn’s brain; (3) necrotizing enterocolitis, or breakdown of the infant’s bowel; and (4) many different types of infection. Beginning in about 1960, treatment of RDS with oxygen, and later with various types of ventilation, including with continuous positive airway pressure (CPAP) and, still later, with artificial surfactant used as either a prevention or a treatment, substantially lowered the death rate from RDS. Beginning in the 1990s in high-income countries, the increasing use of maternal corticosteroids prior to delivery substantially lowered the incidence of RDS. The incidence of preterm as well as term newborn infection was also reduced with better attention to clean care and the use of antibiotics prophylactically. Treatment with antibiotics for those newborns that did become infected saved many lives. Maternal treatment for syphilis and vaccination for tetanus contributed to the reduction of newborn infection-related mortality.
The incidence of newborn asphyxia was substantially reduced with better obstetric care, including monitoring for hypoxia prenatally and during labor, and the willingness to perform cesarean sections for signs of fetal distress. Newborn resuscitation techniques improved, and treatment of newborns with birth-related asphyxia also reduced mortality. Thus, as for maternal and fetal mortality, there were substantial reductions in neonatal mortality from about 1940 onward. Many of the interventions are the same or similar to those that reduce maternal and fetal mortality, and many are appropriate for use in low-income countries.
Interventions to Reduce Mortality
The interventions needed to reduce maternal, fetal, and neonatal mortality are thus well known, and if these were made available to pregnant women and their newborns in low-income countries, the pregnancy-related mortality rates in those countries would likely approach those in high-income countries. New interventions or technologies should not generally be necessary to achieve substantial reductions in these mortality rates in low- and middle-income countries. The question that arises instead is not what needs to be done, but how to make these interventions widely available and performed in an appropriate manner. All too often it is observed that an intervention is available but performed on the wrong patients
or performed poorly or too late. Thus, in addition to increasing the coverage of an intervention, ensuring quality performance is crucial. Finally, it has become clear that introducing one intervention at a time rarely results in a reduction in mortality. Instead, developing an understanding of the population that needs to be served and the capabilities within the system to provide care to that population is an appropriate starting point in any geographic area. Indeed, there is substantial evidence that the so-called systems approach—defining the population needing care, the goals of that care, and the resources and personnel available in clinics, hospitals, and homes, and then creating a system of care—is conducive to the most success in reducing maternal, fetal, and neonatal mortality.
This section reviews the conditions causing maternal, fetal, and neonatal mortality in low-income countries, and the section that follows describes the interventions that many experts believe should be directed at reducing maternal, fetal, and neonatal mortality in low-income countries.
Maternal Mortality
To forestall any particular condition that would threaten the life of a mother, fetus, or neonate, one must either prevent onset of the condition or diagnose and treat it in an appropriate and timely manner. Knowing the conditions from which mothers, fetuses, and newborns die and when they die is therefore crucial. For maternal mortality, the major causes of death in low- and middle-income countries are hemorrhage, preeclampsia/eclampsia, and infection (see Table 3-2 at the end of this chapter).
Prolonged labor and unsafe abortion are often precursors to infection and hemorrhage. Hemorrhage can therefore be prevented through a cesarean section for prolonged labor and the performance of safe compared with unsafe abortions. For abortion complications, uterine suction or a D&C (dilation and curettage), antibiotics, blood transfusion, and sometimes hysterectomy are necessary to save the woman’s life. The use of uterotonics such as misoprostol or oxytocin at delivery can prevent many of the postpartum hemorrhages associated with an atonic uterus. The case fatality rate associated with hemorrhage could be reduced by lowering the incidence of anemia, which is usually accomplished prenatally through iron and vitamin supplementation. However, major reductions in mortality from hemorrhage usually stem from the appropriate treatment of hemorrhage once it occurs. Treatment varies with the cause of the hemorrhage and may include surgery for lacerations or a ruptured uterus, manual placental removal or a D&C for a retained placenta or placental fragments, or uterotonics to treat hemorrhage from an atonic uterus. An antepartum hemorrhage due to placental abruption or previa calls for a cesarean section. A hysterectomy (surgical removal of the uterus) is the surgery of last resort for many types of hemorrhage. Also for a hemorrhage of any type, blood products are usually life-saving. Within the single condition of hemorrhage, then, there are many causes of hemorrhage, requiring many different types of prevention and treatment and substantial provider skills. No single intervention is likely to have a large impact on hemorrhage-related maternal mortality.
After hemorrhage, the major cause of maternal mortality is hypertension, particularly preeclampsia and eclampsia. Prevention may be possible by treating pregnant women with calcium and aspirin, but the effectiveness of these interventions remains uncertain. Women die from many complications of preeclampsia/eclampsia, including asphyxia during seizures, aspiration pneumonia, strokes, and cardiac, liver, or kidney failure, as well as hemorrhage secondary to clotting disorders and abruption. Because preeclampsia is often asymptomatic until late in its course, diagnosing this condition early is key to saving maternal lives. The condition is defined by hypertension and proteinuria (protein in the urine), and so tests for both, repeated in prenatal care throughout the pregnancy, are required to effectively diagnose this condition. Once preeclampsia is diagnosed, close observation, with delivery for worsening disease, is often life-saving. Induction of labor or a cesarean section is thus often necessary to substantially reduce mortality. Magnesium sulfate reduces initial and repeat seizures and may provide time to effect delivery prior to new or recurrent seizures. A combination of prenatal care to diagnose the condition and care in a facility with labor induction and cesarean section capabilities is necessary to reduce maternal mortality from this condition.
Infection is the third major killer of mothers. Malaria is important in endemic areas, and can be responsible for maternal deaths both during acute malarial episodes and afterward because of the effects of the disease on maternal anemia. Most important, however, are bacterial infections of the uterus. During and immediately after delivery, the uterus provides an excellent culture medium for bacteria, and uterine infections are very common. The use of nonsterile delivery techniques, prolonged labor, and instrumentation, including cesarean section, all increase the risk of infection. Prevention includes providing clean delivery sites, ensuring provider hand-washing, and avoiding prolonged labor and instrumentation whenever possible. Treatment generally consists of the timely administration of the appropriate antibiotics.
In summary, most maternal deaths can be prevented by reducing the prevalence of the conditions that cause the deaths or providing the appropriate treatment for those conditions. Because, with a few important exceptions, most complications leading to maternal death cannot be predicted or prevented, care for these conditions must be readily available to all women. Most maternal deaths occur during labor, delivery, and the immediate postpartum period. When the conditions causing maternal death are considered, it becomes clear that the treatments for these conditions need to be readily available during labor and after delivery. Few of these interventions are available in the home, and unfortunately in most low-income countries many of these interventions are not available in health clinics. For these reasons, this committee advocates having all births occur in facilities with all these interventions available.
Fetal Mortality
Most stillbirths can be attributed to intrauterine asphyxia, with a smaller percentage due to infection (Goldenberg et al., 2011). Many of the maternal conditions just described lead to fetal asphyxia, including prolonged labor, preeclampsia/eclampsia, and placental abruption. Pregnancy complications such as growth retardation, abnormal presentations, multiple fetuses, and cord complications also kill fetuses by way of asphyxia. The appropriate obstetric care for these conditions, plus monitoring of the fetus for signs of asphyxia, often with delivery by cesarean section for fetal distress, can prevent many stillbirths (WHO and UNICEF, 2012). Fetal deaths due to infection can at times be prevented. For example, diagnosing and treating maternal syphilis, preventing maternal malaria through the use of bednets and intermittent prophylaxis, and eliminating prolonged labor to reduce bacterial chorioamnionitis all have a role to play in reducing stillbirths (Goldenberg et al., 2011). With the possible exception of their use to treat group B streptococcus or membrane rupture, antibiotics have a smaller role than other interventions in preventing intrapartum stillbirths.
Neonatal Mortality
Neonates predominantly die from three major causes: asphyxia, infection, and preterm birth (Lawn, Wilczynska-Ketende, and Cousens, 2006). Asphyxia is best prevented, and the same interventions that reduce stillbirth also reduce the prevalence of neonatal asphyxia. Thus appropriate care for women with prolonged labor, placental abruption, preeclampsia/eclampsia, fetal growth retardation, etc. will reduce neonatal asphyxia and neonatal mortality as well. Neonatal resuscitation is important in reducing the deaths of infants born with mild asphyxia. Care for newborns with significant asphyxia, including respiratory, temperature, and nutritional support, can save some additional lives. Treatment of maternal conditions can have a profound impact on neonatal deaths from asphyxia.
Preventing deaths from neonatal infection also involves multiple approaches, some directed at the mother and some at the neonate. For example, preventing prolonged labor, treating the mother’s syphilis, immunizing her against tetanus, and administering antibiotics in the face of maternal infection with group B streptococcus or ruptured membranes will all likely reduce neonatal sepsis and mortality. Sterile/clean
techniques, including hand-washing at the time of delivery and in the newborn period, prophylactic antibiotics, especially in preterm infants, and the appropriate cord care—now often with chlorhexidine for home births—also reduce neonatal infections and mortality. For infections, antibiotic treatment frequently proves life-saving. Thus reductions in neonatal infection-related mortality depend on a wide variety of maternal and newborn interventions.
The predominant cause of preterm neonatal mortality is respiratory distress syndrome, but preterm infants also die from other causes, including necrotizing enterocolitis, intraventricular hemorrhage, and bacterial infection. Although few if any interventions effectively prevent preterm births, prevention of some of the conditions associated with preterm birth is possible. Giving the mother corticosteroids in the days prior to delivery may prevent up to 50 percent of RDS, as well as necrotizing enterocolitis, IVH, and neonatal mortality. For those neonates with RDS, oxygen, ventilatory support, and surfactant can substantially reduce mortality. Thus a program for reducing mortality from preterm birth also has maternal and neonatal components, and when they are applied appropriately, most preterm neonatal mortality can be eliminated.
Saving Lives
A number of publications and organizations have proposed lists of interventions appropriate for use in low- and middle-income countries to reduce maternal, fetal, and neonatal mortality; these lists are often accompanied by estimates of the lives potentially saved. Figure 3-7, from a recent Lancet issue, shows estimates of the number of maternal, fetal, and neonatal lives saved by using various interventions, there by illustrating the importance of basic and emergency care during labor and delivery (Pattinson et al., 2011). Because most deaths occur around the time of delivery, the United Nations promotes two strategies to reduce intrapartum maternal mortality: ensuring the presence of a skilled birth attendant at delivery and ensuring prompt access to emergency obstetric care. Ideally, skilled obstetric care should be available to all women. This must include intrapartum monitoring to enable early detection and management of complications, with referral where required (Table 3-1). In Table 3-1, basic essential emergency obstetric care comprises six nonsurgical functions: parenteral antibiotics, parenteral oxytocic drugs, parenteral anticonvulsants, manual removal of the placenta, removal of retained products of conception, and assisted vaginal delivery by forceps or vacuum. Neonatal resuscitation is often included in this proposed package. Comprehensive emergency obstetric care would add blood transfusions and cesarean sections to the list. Staffing for basic emergency obstetric care would include at least two skilled birth attendants available 24 hours a day, seven days a week, and assisted by trained staff. Comprehensive emergency obstetric care would require staff trained to provide a blood transfusion and perform a cesarean section. Because saving many maternal, fetal, and newborn lives requires a cesarean section, timely access to this intervention is crucial to achieving mortality rates comparable to those in high-income countries.
TABLE 3-1 Components of Basic and Comprehensive Obstetric Care
| Basica | Comprehensive |
| Parenteral antibiotics | All components of basic care |
| Parenteral oxytocic drugs | Blood transfusion |
| Parenteral anticonvulsants | Cesarean section |
| Manual removal of placenta | |
| Removal of retained products of conception | |
| Assisted vaginal delivery by forceps or vacuum |
aNeonatal resuscitation is often included in this proposed package.
SOURCE: Robert L. Goldenberg, adapted from Hofmeyr et al. (2009) and UNICEF, WHO, and UNFPA, (1997).
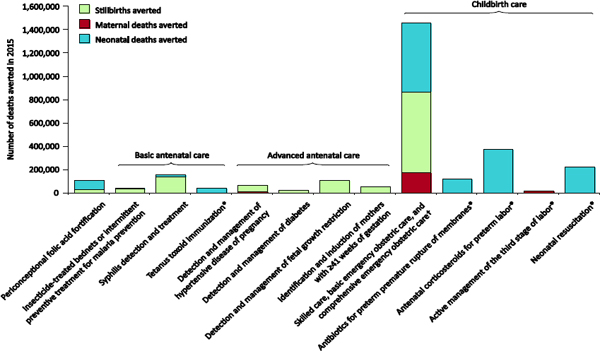
FIGURE 3-7 Various Interventions Appropriate to Reduce Maternal, Fetal, and Neonatal Mortality, with Estimates of Number of Lives Saved by 2015 at Full (99 percent) Coverage by Package. *Interventions specific to maternal and neonatal health and without an estimated direct effect on stillbirths. † Women receiving comprehensive emergency obstetric care are assumed to have access to the highest level of care during childbirth, including skilled care and basic emergency obstetric care if needed. SOURCE: Pattinson et al. (2011). Reprinted from The Lancet 377 9777:1610-1623. Copyright (2011), with permission from Elsevier.
Birth Attendant Capacity
Within any system of care, the capability of the birth attendant is considered crucial. In low-income countries, historically most often the birth attendant has been an unskilled or traditional birth attendant (TBA). For the most part, studies have shown that even with additional TBA training, maternal mortality rates do not decline, although with training in resuscitation there may be some reduction in stillbirths and neonatal mortality. For this reason, the World Health Organization and other groups have recommended the use of skilled birth attendants for delivery. However, the training and competencies of these “skilled attendants” vary widely, and most do not have the training necessary to perform a cesarean section, give blood, or administer antibiotics—interventions often necessary to save a life. Even the ability of the most skilled physician to save a life is limited if there is no blood or antibiotics, or if the facilities needed for a cesarean section are not available. Although this issue cannot be fully reviewed here, it should be obvious that those creating an effective system of care should pay great attention to the skill level of the birth attendant and the circumstances in which that attendant will attempt to provide life-saving care to the mother, fetus, and newborn.
Data
In low-income countries, one of the major obstacles to program development aimed at improving pregnancy-related outcomes is lack of reliable data on these outcomes, the causes of these outcomes, and
TABLE 3-2 Major Killers of Mothers, Fetuses, and Newborns in Low-Income Countries
| Condition | Mother | Fetus | Newborn |
| Hemorrhage | X | X | X |
| Preeclampsia/eclampsia | X | X | X |
| Intrauterine infection | X | X | X |
| Obstructed labor | X | X | X |
| Fetal Asphyxia | - | X | X |
| Preterm labor/birth | - | X | X |
| Syphilis | - | X | X |
| Malaria | X | X | - |
| Hemorrhage | X | X | X |
SOURCE: Goldenberg et al. (2011).
the coverage of the interventions that might improve these outcomes. Without these types of data and the ability of a hospital or clinic, a geographic area, or a political district to compare their outcomes with those of similar entities, the likelihood of improvements is much reduced. Without the ability of these entities to compare current outcomes with those achieved historically, the impacts of newly introduced programs or interventions will not be understood. Only when reliable data can be used to focus attention on the continuous quality of care improvement is it likely that sustained improvement will occur. In virtually every geographic area in which maternal, fetal, and neonatal mortality have been reduced, improvements in care and outcomes have gone hand in hand with the development of data systems that capture outcomes, the causes of poor outcomes, and intervention coverage.
Summary
If used widely and correctly, the substantial information available on medical interventions would save many maternal, fetal, and neonatal lives. However, this review of these interventions has emphasized that there is no magic bullet to saving lives. Instead, multiple interventions applied at various times during the pregnancy—including during prenatal care but especially at delivery—or even before (family planning) are needed to save lives. No single intervention by itself is likely to have much of an impact.
The causes of maternal, fetal, and neonatal deaths are often the same (Table 3-2), as are the treatments that prevent them. Thus it would be most efficient to have programs that focus on improving pregnancy outcomes rather than separate programs that focus on maternal mortality, stillbirth, or neonatal mortality reduction. Because many of the conditions that kill mothers, fetuses, and newborns cannot be predicted or prevented, ideally all women and newborns should have immediate or rapid access to the interventions that save lives, which for the most part are hospital-based. Creating a system by which these interventions can be appropriately applied to all women and newborns in a timely fashion is the challenge if any low-income country is to achieve substantial reductions in maternal, fetal, and neonatal mortality.
References
Black, Robert E., et al. 2010. Global, regional, and national causes of child mortality in 2008: A systematic analysis. Lancet 375(9730):1969-1987.
Flenady, Vicki, et al. 2011. “Stillbirths: The way forward in high-income countries.” Lancet 377(9778):1703-1717.
Goldenberg, Robert L., and Elizabeth M. McClure. 2011. Maternal mortality. American Journal of Obstetrics and Gynecology 205(4):293-295.
Goldenberg, Robert L., Elizabeth M. McClure, Zulfiqar A. Bhutta, José M. Belizán, Uma M. Reddy, Craig E. Rubens, Hillary Mabeya, Vicki Flenady, and Gary L. Darmstadt. 2011. Stillbirths: The vision for 2020.” Lancet 377(9779):1798-1805.
Hofmeyr, G. Justus, Rachel A. Haws, Staffan Bergström, Anne C.C. Lee, Pius Okong, Gary L. Darmstadt, Luke C. Mullany, Eh Kalu Shwe Oo, and Joy E. Lawn, 2009. Obstetric care in low-resource settings: What, who, and how to overcome challenges to scale up? International Journal of Gynecology and Obstetrics 107:S21-S45.
IOM (Institute of Medicine). 2003. Improving Birth Outcomes: Meeting the Challenge in the Developing World, Judith R. Bale, Barbara J. Stoll, and Adetokunbo O. Lucas, eds. Washington, DC: National Academies Press.
Johnson, John W. C. 2001. The millennial mark. American Journal of Obstetrics and Gynecology 185(2):261-267.
Khan, Khalid S., Daniel Wojdyla, Lale Say, A. Metin Gülmezoglu, and Paul F. A. Van Look. 2006. WHO analysis of causes of maternal death: A systematic review. Lancet 367(9516):1066-1074.
Lawn, Joy E., Hannah Blencowe, Robert Pattinson, Simon Cousens, Rajesh Kumar, Ibinabo Ibiebele, Jason Gardosi, Louise T. Day, and Cynthia Stanton. 2011. Stillbirths: Where? When? Why? How to make the data count? Lancet 377(9775):1448-1463.
Lawn, Joy E., Simon Cousens, and Jelka Zupan. 2005. 4 million neonatal deaths: When? Where? Why? Lancet 365(9462):891-900.
Lawn, Joy E., Katarzyna Wilczynska-Ketende, and Simon N. Cousens. 2006. Estimating the causes of 4 million neonatal deaths in the year 2000. International Journal of Epidemiology 35(3):706-718.
Lawn, J. E., M. Y. Yakoob, R. A. Haws, T. Soomro, G. L. Darmstadt, and Z. A. Bhutta. 2009. 3.2 million stillbirths: Epidemiology and overview of the evidence review. BMC Pregnancy Childbirth 9(Suppl. 1):S2.
Loudon, Irvine. 2000. Maternal mortality in the past and its relevance to developing countries today. American Journal of Clinical Nutrition 72(1):241s-246s.
Pattinson, Robert, et al. 2011. Stillbirths: How can health systems deliver for mothers and babies? Lancet 377(9777):1610-1623.
Ronsmans, Carine, and Wendy J. Graham. 2006. Maternal mortality: Who, when, where, and why. Lancet 368(9542):1189-1200.
UNFPA (United Nations Population Fund), UNICEF (United Nations Children’s Fund), WHO (World Health Organization), and World Bank. 2012. Trends in Maternal Mortality: 1990 to 2010. Geneva: WHO.
UNICEF (United Nations Children’s Fund), WHO (World Health Organization), and UNFPA (United Nations Population Fund). 1997. Guidelines for Monitoring the Availability and Use of Obstetric Services. New York: UNICEF.
WHO (World Health Organization) and UNICEF. 2012. Building a Future for Women and Children: The 2012 Report. Geneva: WHO.
This page intentionally left blank.












