Opportunities and Challenges in the Study of Biosocial Dynamics in Healthy Aging
INTRODUCTION
From the womb to the grave, the social world has a profound influence on individual health and well-being. As social animals, human bodies likely evolved to perceive and respond to social stimuli in ways that promote survival and adaptation. Thus, human physiology is attuned to characteristics of our social environment and interactions with others, and social behavior is likewise underpinned by complex biological processes. Social scientists have long been interested in identifying such biosocial dynamics and the roles these processes play in healthy or unhealthy patterns of aging across the life course. The objective of this chapter is to provide a brief overview of the current state of the study of biosocial processes in healthy aging. Research on the biological correlates of social status is reviewed to highlight promising methodological approaches for identifying biosocial connections and for probing biosocial theories of aging. Important methodological and analytical challenges that need to be addressed to significantly advance knowledge of biosocial processes involved in healthy aging are also discussed.
BIOSOCIAL INVESTIGATIONS: THE PROVINCE OF TWO TRADITIONS
The current state of study of biosocial processes involved in healthy human aging is primarily the confluence of accomplishments in two scientific fields: social epidemiology and social/health psychology. Social
epidemiologists have amassed a large body of evidence documenting social determinants of disease morbidity and mortality, including social inequality, socioeconomic status, social isolation, and a lack of social support and social resources (see, Berkman, 2009; House, Landis, and Umberson, 1988; Seeman and Crimmins, 2001). A particular strength of this research is evidence of prospective prediction of disease onset and mortality following measurement of social conditions, dampening concerns that such connections are primarily the result of selection processes (i.e., that poor health leads to unfavorable social conditions). The documentation of such links in large, nationally representative research cohorts also helps to assuage concerns that associations occur only in subsets of individuals with distinct social characteristics (e.g., those of abject poverty, the extremely isolated). Perhaps the most notable strength of this research is the observation that social risk factors rival or exceed traditional biomedical factors, such as smoking and cholesterol levels, in the power to predict poor health outcomes (Holt-Lunstad, Smith, and Layton, 2010; House et al., 1988). Although these observations have yet to lead to significant attention to social risk factors in clinical health care, social factors are now more centrally positioned in the cross-hairs of those focused on policies and interventions designed to improve public health.
As the evidence highlighting social conditions as key determinants of healthy aging has grown, so too has the motivation to understand how such conditions “get under the skin” to affect functioning and health (Adler and Ostrove, 1999; Seeman et al., 2010; Taylor, Repetti, and Seeman, 1997). In epidemiologic investigations, this motivation has fueled the addition of biological markers, or biomarkers, to study assessments in an effort to identify biological processes that might underlie links between social factors and health. This effort has been two-pronged: social studies of aging and health are increasingly incorporating biomarkers into study assessments, and biomedical studies are increasingly instituting more comprehensive measurement of social conditions. Examples of such studies are provided in Table 10-1. The result has been an exponentially increasing number of studies examining biosocial process that play a role in healthy aging.
Occurring contemporaneously with the increasing examination of biosocial connections in epidemiologic investigations, researchers in the fields of social and health psychology have been identifying the biological correlates of social factors in smaller-scale investigations with more fine-grained measures of social and biological processes than is typically possible in large-scale epidemiologic studies. This has included investigations of the biological correlates of psychosocial stressors and other social factors (e.g., quality of social relationships, presence of supportive ties), as well as experimental manipulations that examine the biological sequelae of varying social conditions (e.g., stressor experience in the presence or absence of
TABLE 10-1 Examples of Longitudinal Studies of Aging and Health Incorporating Social and Biomarker Assessments
|
Population-Based Surveys |
Community-Based or Cohort Surveys |
|
Health and Retirement Study (HRS) English Longitudinal Study of Ageing (ELSA) Social Environment and Biomarkers of Aging Study (SEBAS) National Social Life, Health and Aging Project (NSHAP) Study of Midlife in the United States (MIDUS) National Health and Nutrition Examination Surveys (NHANES) Cardiovascular Health Study (CHS) Cardiovascular Risk in Young Finns Study British Birth Cohort Studies (e.g., 1958, 1963) Costa Rican Longevity and Healthy Aging Study (CRELES) Twin Studies (SATSA, OCTO, SALT, GENDER, HARMONY) |
MacArthur Study of Successful Aging Whitehall Studies Normative Aging Study Women’s Health and Aging Studies Multiethnic Study of Atherosclerosis (MESA) Coronary Artery Risk Development in Young Adults (CARDIA) Study Chicago, Health, Aging and Social Relations Study (CHASRS) |
social support). Fueled by technological advances in the measurement of biomarkers in the field, a growing number of studies are also capturing the biological correlates of everyday social experiences as individuals go about their daily lives in their natural social environments.
Biological Targets of Biosocial Investigations
The social scientist seeking to understand how it is that a given social factor is linked to more or less healthy profiles of aging must first identify the biological pathways in the body that might play a role in differential health trajectories. Then, he or she must identify candidate biomarkers for assessing activity in target pathways and assess the feasibility of measuring such biomarkers. Various perspectives may shape the selection of targeted biological pathways. Disease-focused approaches typically target biomarkers (e.g., lipids, markers of inflammation) that play key roles in the pathophysiology of a given disease (e.g., atherosclerosis). Observation of an association between a social factor of interest and target biomarkers might then point to plausible pathways underlying connections between the social condition and the disease outcome. Biological theories of aging might also provide clues as to the pathways through which social factors
may influence health. A number of biological processes are implicated in theories that posit that the “deterioration and decline” of aging is a byproduct of damage versus repair forces occurring in the course of the physiology of life. These include oxidative damage and antioxidant defense or other repair mechanisms, as well as stochastic DNA damage in somatic cells and the efficacy of DNA repair mechanisms. Neuroendocrine, metabolic, and immune processes are also implicated in programmed aging processes (e.g., the neuroendocrine regulators of the Pacific salmon’s final upstream swim to mate, then die as a notable example), as well as in biological processes that may have been naturally selected for their reproductive and survival benefits in early life, but subsequently have negative consequences for wellbeing in later life. This concept is known as antagonistic pleiotropy; as an example, immune defense processes promoting survival, especially in the young, may upregulate inflammation burden and disease risk in the old. A discussion of the various biological processes hypothesized to underlie these mechanisms of “aging” are beyond the scope of this chapter, although readers should consult excellent available reviews (Finch and Ruvkun, 2001; Harman, 2003; Kirkwood and Austad, 2000; Martin, 2011; Parsons, 2003; Sohal and Orr, 2012; Sohal, Mockett, and Orr, 2002). The relevant point here is that the occurrence and nature of expression of such biological processes may well be sensitive to social environment input, and they represent mechanisms through which the social world may accelerate or decelerate healthy aging.
Biological targets of study might also be guided by biopsychosocial perspectives that postulate specific patterns of physiological activation through which social stimuli are transduced into electrochemical and biochemical signals in the body to orchestrate downstream physiology and behavior. This includes theoretical perspectives that posit specific biological signatures for specific psychosocial stimuli such as different patterns of autonomic activity in response to threatening versus challenging social situations (e.g., Blascovich et al., 2003) and specific neuroendocrine correlates of social status threats and accompanying cognitive and affective states (e.g., Dickerson, Gruenewald, and Kemeny, 2004; Gruenewald et al., 2004; Henry, 1993), as well as more general organizing frameworks for understanding biosocial connections, such as that found in the conceptualization of primary and secondary physiological regulatory systems (e.g., McEwen and Seeman, 1999). Primary regulators, notably the neuroendocrine and nervous systems, are those that act as communication systems between the brain, where thoughts and emotions regarding the social world are processed, and the downstream physiological systems that carry out behavior and physiology essential for dealing with social demands. These systems initiate changes in secondary regulatory systems, such as the cardiovascular, metabolic, and immune systems, which enable the body to address the demands of a social stimulus.
For example, it is nervous and neuroendocrine regulators, such as the sympathetic nervous system (SNS) and the hypothalamic-pituitary-adrenal (HPA) axis, that orchestrate downstream cardiovascular (e.g., increased heart rate, blood pressure) and metabolic activity (e.g., production of the body’s primary fuel, glucose) to enable the body to address the demands of a social stressor (e.g., an argument with one’s spouse).
The concept of allostasis has been proposed to explain how the activity of these primary and secondary regulators fluctuates to orchestrate physiological functioning to meet demands (McEwen, 1998; McEwen and Seeman, 1999; McEwen and Stellar, 1993; Sterling, 2004; Sterling and Eyer, 1988). As compared to the more tightly regulated setpoints of homeostatic processes (e.g., maintaining the body’s pH in a narrow range), allostatic processes are theorized to allow greater accommodation of physiological activity to varying demands (e.g., large increases in blood glucose to meet the energy demands of coping with a severe stressor). While often adaptive for addressing the demand at hand, allostatic activity may render individuals vulnerable to adverse functioning and health states under conditions when allostatic responses are engaged too often, are prolonged, initiate severe alterations in physiological activity, or are engaged under conditions with little adaptive benefit (e.g., activation of the HPA or SNS systems in response to worrying about a past social interaction). The wear and tear on body tissues and systems that can result from such allostatic states is referred to as “allostatic load” and represents one model of how social conditions that engage allostatic processes may increase risk of poor health.
What Is Learned from Biosocial Investigations?: An Example: Biological Correlates of Social Status
The allostatic correlates of a wide array of social factors have been explored in both small- and large-scale observational and experimental studies. In this section, the biological correlates of social status are highlighted to provide a flavor of the gains, the promises, and the challenges of biosocial investigations. One reason for this selection is that the connection between social status, most often conceptualized as some form of socioeconomic status (SES), and health has long been a central focus of social epidemiology. A large and relatively consistent body of empirical evidence indicates that those of lower SES experience poorer health, including increased incidence of most diseases, faster disease progression, and greater disease-specific and all-cause mortality risk, resulting in shorter length of life (see, Adler and Ostrove, 1999; Hemingway and Marmot, 1999; Kaplan and Keil, 1993; Marmot, 2006; Matthews and Gallo, 2011). These links have been documented for both prestige (e.g., occupational status) and
resource-based (e.g., income, assets) measures of SES, as well as indicators that tap both dimensions (e.g., education).
The consistently observed SES-health association has led to a hunt for the pathways that underlie the SES gradient in health, and biological pathways have been targets of increasing focus (Miller, Chen, and Cole, 2009; Seeman et al., 2010). A very general conceptualization of the routes through which SES might impact individual biological functioning is provided in Figure 10-1 and includes (1) SES-patterned environmental exposures, including exposure to pollutants, carcinogens, toxins, and adverse neighborhood/community characteristics; (2) SES variations in psychosocial exposures and processes, including psychosocial stress, cognitive-perceptual, and emotional processes, and psychosocial resources (e.g., access to social support, control over environment); and (3) SES-patterned health behavior, including smoking, physical activity, diet, and drug/alcohol use. The overarching hypothesis is that SES profoundly shapes individual thought, feeling, and behavior, as well as exposure to the slings and arrows of life, in turn, affecting allostatic processes and subsequent health-relevant biological wear and tear.
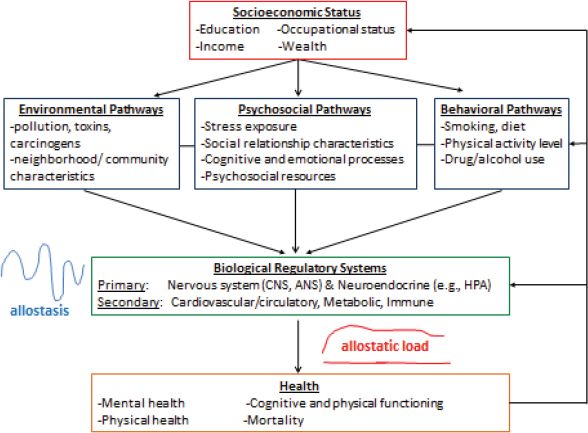
FIGURE 10-1 Conceptual model of potential pathways through which social status is linked to health.
Biomarker Correlates—Clarity or Confusion?
At first glance, the picture that emerges from the body of work examining biological correlates of SES is consistent with that examining health correlates: Those of lower SES have poorer biological profiles for most major biological regulatory systems, including nervous, neuroendocrine, cardiovascular, metabolic, and immune systems. For example, those of lower SES have been found to have higher levels of HPA and SNS hormones hypothesized to be elevated under conditions of stress (e.g., cortisol and catecholamines; Cohen, Doyle, and Baum, 2006; Janicki-Deverts et al., 2007; Steptoe et al., 2003), higher biomarker levels indicative of poor metabolic functioning (e.g., greater body mass index, higher fasting glucose and insulin and glycosylated hemoglobin, poorer lipid profiles; Danese et al., 2009; Loucks et al., 2007a,b; McLaren, 2007; Senese et al., 2009), and other indicators of cardiovascular disease risk (e.g., high blood pressure, low heart rate variability, high inflammation burden) (Brunner et al., 1996; Colhoun, Hemingway, and Poulter, 1998; Gruenewald et al., 2009; Hemingway et al., 2003; Koster et al., 2006; Sloan et al., 2005). Upon closer inspection, however, there are nuances in the consistency of findings in this literature that suggest that further attention to methods, measurement, and theory might be fruitful.
One interesting observation is the geographic and demographic variations in associations between SES and some biomarker indicators. For example, a review of 57 studies published over a 30-year period (1966-1996) indicated that a majority of investigations in the United States and Canada observed higher levels of blood pressure in those of lower SES (Colhoun et al., 1998), but associations were less consistent in the United Kingdom, Australasia, Asia, South Africa, and some European countries. Well-documented geographic and demographic (sex) variations have also been observed for associations between SES and body mass index or body weight (McLaren, 2007; Sobal and Stunkard, 1989).
These variations are notable because cross-study consistency in the validity and reliability of measurement of blood pressure and weight is likely considerably higher than is the case for other biomarkers that have to be assessed via saliva, blood, or urine samples and measured with complex assays (e.g., hormone or immune biomarkers). Thus, assuming fairly similar measurement of these biomarkers across samples, focus is shifted to other potential explanations for geographic and demographic variations in SES-biomarker associations, including characteristics of SES measurement, cultural variations in the meaning of SES, and its environmental, psychosocial, and behavioral correlates, and the role of social institutions and more macro social factors (e.g., universal access to health care) in moderating links between individual-level SES and biological functioning. A review of
research that sheds light on each of these possibilities is beyond the scope of this review, but these observations point to the need for increasing comprehensiveness and sophistication in the measurement and theorization of the social, psychological, and behavioral alongside the biological in biosocial investigations.
Biosocial Investigations—An Opportunity for Life Course Explorations?
Another aspect of research on SES gradients in biological functioning that is ripe for further attention is a better understanding of when, and how, SES becomes embodied across the life course. A number of life course frameworks have been proposed to explain the role of biosocial processes in healthy aging (Ben-Shlomo and Kuh, 2002; Pollitt, Rose, and Kaufman, 2005), with sensitive or critical period and accumulation of risk models receiving the most attention. The sensitive or critical period model suggests that connections between SES and biological functioning may vary depending on life course phase. Strict critical period models posit that events that occur within specific and narrow windows of development may permanently “tune” bodily systems and subsequent disease risk, which will be unaffected by risk exposures that occur outside the critical window. Sensitive period models posit that risk exposures at certain life course phases (e.g., early childhood) may simply have a stronger negative impact than those that occur in other life phases (e.g., late adulthood).
Growing evidence indicates that SES disadvantage in early life predicts poorer physiological functioning in later life, above and beyond the influence of more contemporary SES characteristics. The negative impact of early life disadvantage has been documented for cardiovascular and metabolic biomarkers (e.g., blood pressure, high-density lipoprotein cholesterol, insulin resistance; Blane et al., 1996; Kivimaki et al., 2006b; Poulton et al., 2002) and the HPA hormone cortisol (Li et al., 2007). When SES-adversity indicators from different life course phases are pitted against each other as predictors of later-life biological risk, sometimes childhood SES is a stronger predictor (Kivimaki et al., 2006b), while in other cases, more recent SES conditions exhibit greater predictive power (e.g., Blane et al., 1996). A significant limitation of these investigations is the lack of biological information from earlier life periods, rendering it difficult to discern whether SES-adversity experience actually alters biological functioning in earlier life course phases. An analysis where such information was available in the Cardiovascular Risk in Young Finns Study indicated that SES disparities in blood pressure emerged early in childhood and persisted across the life course, with early life alterations accounting for much of the association between childhood SES and adult blood pressure (Kivimaki et al., 2006a,b).
Exciting opportunities are on the horizon for identifying how SES experiences at different phases of the life course get under the skin to affect a wide array of biological processes and how SES disparities in biological functioning might track across the life time. Evidence of SES disparities in childhood of levels of primary neuroendocrine regulators (HPA and SNS hormones), as well as in downstream secondary regulatory systems (e.g., cardiovascular and metabolic biomarkers), is accumulating from cross-sectional and short-term longitudinal investigations (e.g., Evans and English, 2002; Evans and Kim, 2007; Goodman et al., 2005). These biological imprints of social adversity in childhood may provide clues as to the trajectories of healthy or unhealthy aging that lie ahead. Longitudinal, life course investigations that concurrently measure social and biological factors from childhood to adulthood will be particularly fruitful for understanding when social adversity is embodied for different systems, the permanency of biological imprints, and the genetic, psychosocial, and behavioral modifiers of these links. Of course, one needs to be mindful of the considerable burden involved in tracking cohorts for long periods of time in advocating for such designs.
Another life course model receiving growing attention in biosocial investigations is the accumulation of risk model. As applied to biosocial investigations, this model posits that greater overall exposure to adverse social conditions (e.g., low SES) across the life course accumulates to have a greater toll on biological functioning in later adulthood. The expansive battery of biomarkers recently added to the second wave of the Study of Midlife in the United States (MIDUS), a longitudinal study of health and aging, allowed for an exploration of the range of biological systems that might be sensitive to life course SES adversity experience. Analyzing data from MIDUS, Gruenewald and colleagues (2012b) documented greater dysregulation across seven different biological indices (SNS, parasympathetic nervous system, HPA system, cardiovascular system, glucose metabolism, lipid metabolism, inflammation) in adults with greater experience of SES adversity (assessed with multiple indicators of education, income, and financial strain) across childhood and adulthood. Figure 10-2 demonstrates that greater life course SES adversity left its mark in adulthood on all of the physiological indices examined. Aggregation of individual system risk indices into a multisystem measure of allostatic load (AL) also revealed a steep SES gradient in biological dysregulation. The difference in AL between those in the highest and lowest quintiles of lifetime SES adversity was equivalent to a 17-year age difference in AL, suggesting accelerated “biological aging” in those with greater cumulative adversity experience.
Support for the hypothesis that greater social adversity experience might accelerate the “aging” or “weathering” (Geronimus, 1992) of biological systems is also found in analyses of population-based surveys, such
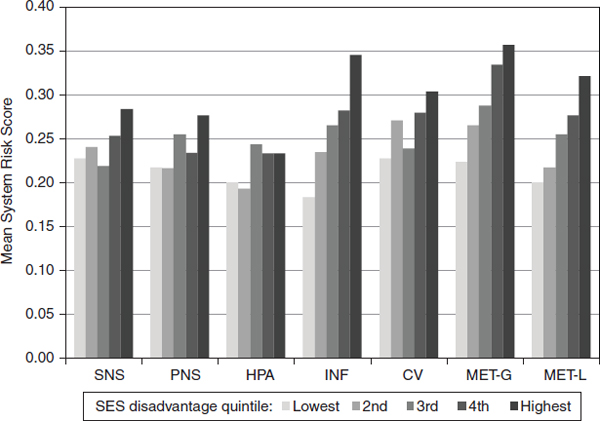
FIGURE 10-2 Mean levels of biological system dysregulation by quintiles of cumulative SES adversity (across childhood and adulthood) in the Study of Midlife in the United States (MIDUS).
NOTE: CV = cardiovascular system, HPA = hypothalamic-pituitary-adrenal axis, INF = inflammation, MET-G = glucose metabolism, MET-L = lipid metabolism, PNS = parasympathetic nervous system, SES = socioeconomic status, SNS = sympathetic nervous system.
SOURCE: Gruenewald et al. (2012b). Reprinted with permission.
as the National Health and Nutrition Examination Surveys (NHANES). Geronimus and colleagues (2006) found that poverty, ethnic minority status (“non-Hispanic Black”), and being female were each associated with a greater likelihood of experiencing high AL in NHANES III participants aged 18-64. Moreover, these factors interacted to predict the occurrence of high AL at earlier stages of the life course and disparities widened with age. In an analysis of both NHANES III and IV data, Crimmins, Kim, and Seeman (2009) replicated the finding of widening SES disparities in AL with age up until the 60s. However, they further documented the dramatic consequence of earlier weathering in the socially disadvantaged with analyses suggesting that SES disparities in biological risk disappear at older ages because the poor with high AL die before reaching older adulthood. These investigations are compelling examples of the value of biomarker data in understanding how life course characteristics of SES adversity may
shape disease risk and longevity. These analyses also highlight the complex interactions that may be occurring between SES and other demographic factors (gender, race), as well as other individual and social conditions as yet unexplored.
Promising Approaches
Time for a Confluence of Traditions?
An advantage of adding biomarker measures to large-scale, population-based studies of aging and health is the ability to examine biosocial associations with sufficient power in samples representative of the general population or specific subgroups. Another advantage is the opportunity to track the role of biosocial processes in the development of health conditions over time in longitudinal investigations. A disadvantage is that the design constraints of such investigations tend to allow only a limited “snapshot” measurement of psychosocial, behavioral and biological states at any given assessment. Many psychosocial, behavioral, and biological factors fluctuate considerably over time and context, within and between individuals, and important nuances of such fluctuations are not well captured in large-scale survey investigations.
These nuances have been investigated in smaller-scale observational and experimental studies over the past few decades, primarily carried out in the fields of social and health psychology. The impressive knowledge gained from these biosocial investigations could, and has, filled many volumes, so will not be reviewed in detail here. However, highlights include findings that biological functioning is intricately tied to characteristics of social interactions and social stressor experience (e.g., social conflict, discrimination, social-evaluative threat; Dickerson and Kemeny, 2004; Kiecolt-Glaser et al., 2002; Seeman and McEwen, 1996) and social characteristics that individuals bring with them to social interactions (e.g., background level of social integration and support, social conflict history, cultural norms; Uchino, Cacioppo, and Kiecolt-Glaser, 1996). What is emerging from this body of work is a picture of complex interconnections between our social, psychological, and biological worlds.
Despite the incredible advantage of such investigations in giving us a more detailed understanding of these complex interactions, the design constraints of these smaller-scale investigations typically limit the use of population-based samples, as well as samples of sufficient size to track and predict incident disease. Given that the relative strengths of the smaller-scale social and health psychology investigations are the weaknesses of the large-scale survey study, and vice-versa, a particularly exciting trend is the effort to combine these two designs within single studies. Although
still fairly rare, such efforts are taking the form of the addition of small substudies within larger-scale survey investigations in which more detailed assessments of biosocial processes are collected on subsets of participants.
Exploring Biosocial Connections in the Wild
One notable form of this substudy approach is the effort to collect data on biosocial processes in individuals’ natural social environments, as they navigate the challenges and social interactions of daily life. An example of such an approach comes from the Whitehall II Study of British civil servants, which has been a fruitful source of knowledge regarding the biosocial processes that may underlie social status gradients in health. A substudy of 202 Whitehall II participants examined cardiovascular and neuroendocrine parameters across a workday, with cardiovascular measurements occurring every 20 minutes and 10 saliva samples collected for the assay of the HPA hormone cortisol, at various points from waking to bedtime (Steptoe et al., 2003a). This investigation revealed that those of lower occupational status have higher blood pressure and heart rate levels, but only in the morning, while cortisol levels are higher primarily during working hours. This latter finding was reversed for females, with higher status women having higher levels of cortisol across the day. Taken alone, these findings merely add to those of other smaller-scale studies of more select samples, which document SES variations in cardiovascular activity across the workday (e.g., Gallo et al., 2004). However, one advantage of collecting such data within a larger longitudinal study of health and aging is the ability to integrate substudy findings within the larger web of observed biosocial and social-health associations in the parent cohort. Although yet to be done to a great extent, the possibilities of such analyses are numerous.
Despite the methodological challenges, ambulatory substudies of biosocial processes are growing in number. In addition to the Whitehall Study, numerous large-scale studies of health and aging, including MIDUS, the English Longitudinal Study of Ageing (ELSA), the Coronary Artery Risk Development in Young Adults (CARDIA) Study, and the Multi-ethnic Study of Atherosclerosis (MESA), have incorporated diurnal saliva sampling in participants’ natural environments. The focus has primarily been the measurement of the HPA hormone, cortisol, but the range of biomarkers that can be measured in saliva continues to expand. Our recent explorations of diurnal salivary cortisol variations in the MIDUS cohort indicate that greater SES adversity is linked to lower cortisol output in the morning but a flatter slope of decline across the day leading to higher evening levels (Gruenewald et al., 2012a). Similar SES variations in diurnal cortisol activity have been observed in other large cohort studies (Cohen et al., 2006; Hajat et al., 2010; Kumari et al., 2010). The changing nature of
SES variations in cortisol activity across the day (lower at some points but higher at others in the more disadvantaged) may render it difficult to discern SES differences with either single snapshot assessments or measures that aggregate information over longer time periods (and thus obscure within period variation). This may explain the less stark SES gradients for the HPA index in Figure 10-2, which included a 12-hour aggregate urinary measure of cortisol activity. The increasing inclusion of ambulatory and clinic-based assessments of biological activity (cortisol, cardiovascular) in large cohort studies will allow for a comparison of physiological measurement methods best able to capture the effect of social conditions on our physiology, as well as enhance our ability to understand the mediators and moderators of such links.
Integrating Laboratory-Based and Epidemiological Approaches
The addition of laboratory-based challenge substudies in large-scale, longitudinal studies of aging (e.g., Whitehall, MIDUS, CARDIA, MESA) is another example of the integration of epidemiological and social/health psychology approaches. Challenge paradigms expose participants to a standardized set of demanding and challenging activities (e.g., difficult cognitive tasks, public speaking) to examine the psychological and physiological consequences of “stressor” exposure. The advantage of such methods is that investigators can compare the psychobiological responses of individuals that vary on social characteristics to the same stimulus, negating concerns about variations in social characteristics “selecting” participants into certain stressors (e.g., conflictual interactions). Investigations of physiological reactivity within the Whitehall cohort have revealed that those of lower SES have delayed recovery of cardiovascular parameters (Steptoe et al., 2002, 2003b) and longer-lasting stress-associated increases in the inflammatory biomarker, interleukin-6 (Brydon et al., 2004). However, no reactivity and recovery differences emerged for other inflammatory (e.g., fibrinogen, tumor necrosis factor alpha) and immune (e.g., natural killer cell number; Owen et al., 2003) biomarkers. Thus, SES variations in stress reactivity are more complex than the simple message of those of lower status exhibiting greater reactivity and less recovery on all biomarker indicators. Lack of stress response differences for many biomarkers does not negate the possibility that stress is a mechanism through which SES variations in health occur, as those of lower SES may simply experience a greater absolute level of stress and its associated physiological correlates.
Another advantage of the laboratory challenge paradigm is the opportunity to more carefully pinpoint the psychological processes that might underlie SES variations in physiological reactivity. Chen and colleagues have documented that low SES children are more likely to interpret ambiguous
social situations as threatening than high SES children (Chen and Matthews, 1999; Chen et al., 2004), which may reflect cognitive interpretation tendencies shaped by an accumulation of less favorable social experiences. This tendency has also been shown to increase with aging (Chen and Matthews, 1999). Furthermore, such threat perception tendencies, and increases in these tendencies over time, partially explain the greater cardiovascular reactivity of low SES adolescents in laboratory challenge paradigms (Chen et al., 2004), as well as greater ambulatory cardiovascular activity during social interactions (Chen, Matthews, and Zhou, 2007) in individuals’ normal social environments. Increases in threat perception biases also played a more significant role in predicting future physiological reactivity in lower SES African American adolescents (Chen and Matthews, 2001), suggesting that social stressors (SES, minority status) may interact over time.
Moving Upstream
Another promising area of research is the effort to identify the neural processes that may mediate SES variations in the processing of social stimuli. There has been an explosive growth in the field of social neuroscience in the past decade, which seeks to understand the neural processes that mediate social behavior and social information processing. This has led to an increased understanding of the brain structures involved in these processes, including the prefontal cortex, the hippocampus, and the amygdala (Gianaros and Manuck, 2010; McEwen and Gianaros, 2010). One of the important roles these social brain structures play is in the modulation of activity of primary regulatory systems (nervous and neuroendocrine systems) in response to the processing of social and emotional stimuli. Thus, a better understanding of SES variations in neural activity may provide clues as to SES variations in the activity of downstream physiological systems.
Although preliminary, data are accumulating indicating SES correlates of neural activity. Gianaros and colleagues (2008) have found that young adults who perceive they came from lower status families exhibit greater amygdala activation in response to viewing angry faces in a functional magnetic resonance imaging (fMRI) investigation, consistent with the threat perceptions biases found in the work of Chen and colleagues. Gianaros and colleagues (2011) also recently reported that prefrontal cortex activity varies in midlife adults as a function of childhood SES, with those from lower SES backgrounds showing a lower cortical response to reward stimuli in fMRI assessments. These findings are intriguing in that they suggest differential patterns of activity in brain structures that modulate downstream stress regulatory systems. However, the links between patterns of functional neural activity in response to various stimuli (e.g., a threatening “social” stimulus in a scanner) and downstream physiology are only beginning to
be mapped out. Another concern is that almost all of this research has been conducted on small and select samples. Neuroscience assessments, including fMRI and EEG measurements, were recently assessed for a small subset of MIDUS participants, allowing linkage to the wide array of biomarkers and psychosocial information collected in the larger MIDUS II cohort. These data are ripe for analyses of how SES and other social factors may influence patterns of neural activity and subsequent downstream disease-relevant physiology.
Another approach through which to assess more “upstream” modulation of physiological functioning is through the study of social regulation of gene expression. Genetic potential is only realized when specific genes are turned on, or “expressed,” via transcription and translation. One of the first studies to show that a social factor could regulate gene expression was conducted by Cole and colleagues (2007) and examined a small subset of participants selected for high and low social loneliness from the Chicago Health Aging and Social Relations Study (CHASRS). Analyses indicated that of humans’ 20,000+ genes, 209 showed significant expression differences in older adults high and low in loneliness. An analysis of the functional activity of differentially expressed genes revealed three primary functional typologies—an upregulation of genes involved in inflammatory activity and a downregulation of genes involved in antibody production and in immune response to viral infection. These gene expression differences are notable because they may affect variations in downstream physiological functioning that explain the significantly greater risk for poor health outcomes in lonely versus nonlonely individuals (see Cacioppo et al., 2011; Hawkley and Cacioppo, 2010) and may represent a pattern of earlier “biological aging” in the lonely.
Gene expression profiles have also been found to vary as a function of SES. Collectively these studies suggest that SES adversity in childhood is associated with gene expression profiles in adolescence and adulthood characterized: (1) as proinflammatory, (2) by diminished glucocorticoid receptor expression and signaling, which may result in less effective control of HPA output and remove the glucocorticoid-mediated brake on inflammation, and (3) by upregulation of catecholamine associated transcription promoter pathways that suggest that the sympathetic nervous system may be playing a role in delivering proinflammatory signals to our genes (Chen et al., 2009; Miller and Chen, 2007; Miller et al., 2009). Miller and colleagues (2007) have labeled this a “defensive” phenotype and suggest that SES-adversity experiences early in life “program” these biological systems during sensitive or critical periods of early childhood, leading to heightened susceptibility in adulthood for the development of the many conditions associated with greater inflammatory and HPA activity. Are such processes a social form of antagonistic pleiotropy, whereby these biopsychosocial
processes confer reproductive or survival benefits in young adulthood but increase risk of ill-being in later adulthood? Or do these social experiences and biopsychosocial responses merely accelerate the forces of damage, and lessen the forces of repair, that shape trajectories of healthy aging across the life course?
A few aspects of these findings bear additional mention. The first is that low SES individuals with the “defensive” gene expression phenotype described above exhibited indicators of greater proinflammatory and HPA activity (Chen et al., 2011; Miller et al., 2009), suggesting that expression profiles bear a connection with more commonly measured “downstream” biomarkers. Second, there is increasing identification of the psychological correlates of expression profiles. For example, Chen and colleagues (2009) found that the threat perception style they had previously identified as more common in low SES children also appeared to underlie much of the association between SES and the defensive phenotype identified in gene expression studies. A third important point is that other social factors may moderate SES variations in gene expression profiles—those of low SES who experienced high levels of maternal warmth in childhood were less likely to show the more “risky” proinflammatory gene expression characteristic of the disadvantaged (Chen et al., 2011).
Attending to the Positive
One final suggestion for understanding SES variations in health comes in the form of turning attention toward understanding the social distribution of the positive goods in life. There has long been recognition that health is more than just the absence of ill-being and that it also encompasses various forms of social and psychological well-being (see Ryff, 1989; Ryff and Keyes, 1995). These include forms of social well-being (e.g., a sense of social connectedness, collective efficacy), hedonic well-being (e.g., feeling happy and satisfied), and eudaimonic well-being (having a sense of purpose in life, feeling engaged, valued, useful, in control), what some have characterized as indicators of flourishing or thriving (Keyes, 1998, 2002). A growing body of evidence also suggests social patterning of such forms of well-being with those with greater social disadvantage often reporting less of these goods (e.g., Diener et al., 2002; Keyes, Shmotkin, and Ryff, 2002). Beyond the injustice of social variations in these desired ends, social disparities in psychosocial well-being may also explain differential patterns of healthy aging. Lower hedonic and eudaimonic well-being predicts less favorable trajectories of cognitive and physical functioning, greater morbidity and mortality risk, and shorter length of life (Boyle et al., 2009, 2010; Cohen and Pressman, 2006; Gruenewald et al., 2007; Gruenewald, Liao, and Seeman, 2012; Okamoto and Tanaka, 2004; Pitkala et al., 2004;
Steptoe and Wardle, 2011). The biological pathways through which these forms of well-being may promote healthier trajectories of aging are just beginning to be elucidated, but accumulating evidence suggests neuroendocrine, cardiovascular, and inflammatory routes (Pressman and Cohen, 2005; Ryff et al., 2006; Ryff, Singer, and Dienberg Love, 2004; Steptoe, Dockray, and Wardle, 2009). The promising methodological approaches highlighted above, including identification of upstream physiological modulators and assessing biological correlates of social conditions in the lab and in the wild, may well help us better understand social disparities in flourishing and thriving and the implications for healthy aging.
SOME CHALLENGES OF BIOSOCIAL INVESTIGATIONS
Measurement
The potential promises of various methodological approaches in biosocial investigations are tempered by a number of challenges. One is the domain of measurement challenges. On the biological side, numerous challenges abound in collecting biomarker samples, including the relatively invasive nature of measurements (i.e., obtaining samples of blood, urine, or saliva, via physical measurement of the body), the detailed instruction needed for both research staff and participant, and for many biomarkers, sensitivity to the temporal and contextual characteristics of biomarker collection (e.g., time of day, whether to obtain “resting” or “challenge” measures). Biomarkers tend to be relatively expensive to measure (both in collection and processing costs), but costs tend to decrease somewhat with greater adoption in scientific and clinical realms. Technological advances have also led to a significant increase in the biomarkers that can be measured and the mediums (e.g., hair, finger-prick blood spots) and conditions (e.g., return of samples via unrefrigerated, postal mail) for capturing physiological samples. However, much work remains in establishing the reliability and validity of these newly developed methods.
Of course, most of the challenges of biomarker data collection are also the challenges of social factor data collection. Comprehensive measurement of social factors is often invasive (e.g., life experience interview, daily measurements) and requires detailed training and instruction to collect accurately. Social measures are also very sensitive to the temporal and contextual characteristics of data collection. Technological advances have also increased the mediums through which social measures are obtained (e.g., smartphones and other personal communication electronic devices, global positioning system or GPS tracking), and such data collection innovations are also associated with substantial monetary requirements.
Perhaps the biggest challenge is concurrent high-quality measurement of both social and biological characteristics within studies. Early large-scale biosocial investigations tended to add fairly crude measures of one or the other domain depending on the original study framework with the result that failure to observe significant or strong biosocial associations led to aspersions on the whole enterprise (i.e., not seeing the “value” of biomarker or social condition measurement). Fortunately, more focused investigations have continued to document rich biosocial connections, and efforts have been made to better “capture” both social and biological characteristics in larger-scale investigations. However, the considerable participant and investigator burden associated with such efforts continues to plague the field.
Capturing and Analyzing the Multilevel and Temporal Complexity
Another challenge for understanding biosocial processes is the methodological and analytical challenge of capturing and understanding multilevel and temporally complex processes. This overview has presented an incredibly simplistic directional and temporal model of biosocial connections, with the focus primarily at the individual level. Even at the individual level, this overview has neglected a discussion of the complex patterns of development or “aging” within biological, psychological, and social realms, to say nothing of intra-individual cross-level interactions. But most biosocial processes, even those that may be most tightly coupled during narrow windows of development, operate in a bidirectional, iterative process over time, nested within multiple levels of influences from the most micro biological to the most macro social. Biosocial studies of health and aging have particularly neglected measurement of bidirectional flows of influence between individuals and upward to larger units of social organization. Methodological advances, including linked ambulatory monitoring of experiences, activity, and interaction of multiple social actors over time within defined geographic boundaries, including measurement of group-level characteristics, may aid in understanding of such bidirectional flows of influence. However, careful measurement of multilevel characteristics of biosocial processes requires stepping outside one’s disciplinary comfort zone and working in multidisciplinary teams, which academic science has been slow to reward. The temporal and directional challenges also require time-, labor-, and monetary-intensive longitudinal investigations coupled with employment of sophisticated analytic techniques that can appropriately parse units of influence among a complicated web of associations. Nonetheless, the importance of such endeavors is being increasingly acknowledged by public health support systems, such as the National Institutes of Health, which has spurred scientific research initiatives for the study of social network analyses and multilevel systems processes in understanding health and aging.
Prognostic Significance of Social Variations in Biomarkers
The impetus for many biosocial investigations is to identify links between social factors and biological processes that may explain social gradients in health outcomes. As reviewed, social variations in biomarker levels and activity have been observed in many investigations. These variations in biological functioning can emerge early in life and persist across the lifetime. However, what is needed in this literature is commensurate evidence demonstrating that social disparities in biomarkers underlie social disparities in actual health outcomes. To date, investigations that have attempted to study each link in the chain simultaneously have examined the explanatory power of individual or clusters of biomarkers in explaining SES gradients in cardiovascular health outcomes or mortality. Individual biomarkers of inflammation (e.g., interleukin-6, C-reactive protein, fibrinogen) have been found to account for small to moderate proportions of SES gradients in incident cardiovascular disease/events and mortality, while more traditional cardiovascular risk factors (e.g., blood pressure, metabolic biomarkers) have been shown to play smaller, or no mediating roles (e.g., Loucks et al., 2009; Marmot et al., 2008; Ramsay et al., 2009; Rosvall et al., 2008). Greater explanatory power is observed when examining composites of cardiovascular or cardiovascular/metabolic biomarkers (Marmot et al., 2008), as well as for multisystem indices, such as captured in allostatic load measures (Seeman et al., 2004).
The increasing incorporation of biomarkers into longitudinal studies of aging will allow for these needed mediational analyses in the near future. However, the collection of valid incident disease information is difficult in large-scale survey studies. Mortality occurrence is somewhat easier to assess via links with mortality registries, but investigations must have in place appropriate human subject consent, as well as identifying information (e.g., Social Security number), to allow for identification of deaths through such systems. Nonetheless, the value of establishing the predictive validity of commonly assessed biomarkers is worth tackling these methodological challenges. Important benefits would be greater faith in using biomarkers as intermediate health endpoints, to better evaluate the success of health promotion interventions and policies, and to track the effect of social conditions on physical well-being.
LOOKING FORWARD
As this overview makes clear, the marriage of social and biological measurements in both large-scale, social epidemiological and smaller-scale, laboratory and observational investigations has been successfully achieved. Much has been learned from this union about the intricate ties between
humans’ social, psychological, and biological worlds. However, as in all marriages, there is still much to learn. One important focus for the future is the exploration of life course models of biosocial connections, including a greater understanding of the range of social conditions linked to biological processes, the characteristics of biosocial interactions at different phases of the life course, and how such processes operate across time to influence healthy aging. A second important focus for the future is a better understanding of the interaction of social and demographic factors, at both micro and macro levels, in shaping associations with biological factors. The currently limited understanding of geographic and demographic variations in biosocial associations suggests that additional theoretical and methodological development in this area is needed.
As discussed, there are a number of promising approaches that are poised to considerably advance knowledge of biosocial connections. Efforts to combine the methods and paradigms of social epidemiology and social and healthy psychology may be particularly fruitful. The field seems to be on the eve of incredible advancements in knowledge of the social regulation of “upstream” biological processes, including neural and genetic activity. Efforts in these areas, if appropriately coupled with continuing focus on “downstream” biological processes, will significantly propel our understanding of the biological pathways that underlie social disparities in healthy aging forward. It is necessary to engage these tools to understand not only the biology that underlies ill-being but also that which promotes flourishing and thriving.
As noted, there are also some challenges that may impede easy navigation of this journey. As with many areas of scientific inquiry, measurement challenges are considerable. The methodological and analytical challenges of adequately capturing the multilevel, bidirectional, and temporal characteristics of biosocial processes are also daunting. However, such challenges are not unique to study of biosocial processes and are characteristic of the study of many risk factors of unhealthy aging. For example, such challenges also plague the study of behavioral risk factors like smoking. It is doubtful that any in the health promotion realm would advocate avoiding scientific investigation of smoking because of the multilevel, multivariable, or temporal influences on smoking behavior.
The importance of establishing the prognostic significance of biomarkers and their role in explaining social disparities in health outcomes was also discussed. One should not confuse this with a need to establish the prognostic significance of social factors for healthy aging—decades of research have solidly established such connections. The value of biosocial research will be in the identification of the biological pathways that underlie these links, and in the potential use of biomarkers as surrogate endpoints or indicators that can be used to better understand the impact of adverse social
conditions on biological well-being, to track the efficacy of health-promotion policies and interventions, and to intervene at appropriate points in the life course to place individuals on more healthy trajectories of aging.
REFERENCES
Adler, N., and Ostrove, J. (1999). Socioeconomic status and health: What we know and what we don’t. Annals of the New York Academy of Sciences, 896, 3-15.
Ben-Shlomo, Y., and Kuh, D. (2002). A life course approach to chronic disease epidemiology: Conceptual models, empirical challenges and interdisciplinary perspectives. International Journal of Epidemiology, 31(2), 285-293.
Berkman, L.E. (2009). Social epidemiology: Social determinants of health in the United States: Are we losing ground? Annual Review of Public Health (vol. 30, pp. 27-41). Palo Alto, CA: Annual Reviews.
Blane, D., Hart, C.L., Smith, G.D., Gillis, C.R., Hole, D.J., and Hawthorne, V.M. (1996). Association of cardiovascular disease risk factors with socioeconomic position during childhood and during adulthood. British Medical Journal, 313(7,070), 1,434-1,438.
Blascovich, J., Mendes, W.B., Tomaka, J., Salomon, K., and Seery, M. (2003). The robust nature of the biopsychosocial model challenge and threat: A reply to Wright and Kirby. Personality and Social Psychology Review, 7(3), 234-243.
Boyle, P.A., Barnes, L.L., Buchman, A.S., and Bennett, D.A. (2009). Purpose in life is associated with mortality among community-dwelling older persons. Psychosomatic Medicine, 71(5), 574-579.
Boyle, P.A., Buchman, A.S., Barnes, L.L., and Bennett, D.A. (2010). Effect of a purpose in life on risk of incident Alzheimer disease and mild cognitive impairment in communitydwelling older persons. Archives of General Psychiatry, 67(3), 304-310.
Brunner, E., Smith, G.D., Marmot, M., Canner, R., Beksinska, M., and Obrien, J. (1996). Childhood social circumstances and psychosocial and behavioural factors as determinants of plasma fibrinogen. Lancet, 347(9,007), 1,008-1,013.
Brydon, L., Edwards, S., Mohamed-Ali, V., and Steptoe, A. (2004). Socioeconomic status and stress-induced increases in interleukin-6. Brain, Behavior, and Immunity, 18(3), 281-290.
Cacioppo, J.T., Hawkley, L.C., Norman, G.J., and Berntson, G.G. (2011). Social isolation. Annals of the New York Academy of Sciences, 1231(1), 17-22.
Chen, E., and Matthews, K.A. (1999). Socioeconomic differences in social information processing and cardiovascular reactivity. Annals of the New York Academy of Sciences, 896, 417-419.
Chen, E., and Matthews, K.A. (2001). Cognitive appraisal biases: An approach to understanding the relation between socioeconomic status and cardiovascular reactivity in children. Annals of Behavioral Medicine, 23(2), 101-111.
Chen, E., Langer, D.A., Raphaelson, Y.E., and Matthews, K.A. (2004). Socioeconomic status and health in adolescents: The role of stress interpretations. Child Development, 75(4), 1,039-1,052.
Chen, E., Matthews, K.A., and Zhou, F. (2007). Interpretations of ambiguous social situations and cardiovascular responses in adolescents. Annals of Behavioral Medicine, 34(1), 26-36.
Chen, E., Miller, G.E., Walker, H.A., Arevalo, J.M., Sung, C.Y., and Cole, S.W. (2009). Genome-wide transcriptional profiling linked to social class in asthma. Thorax, 64(1), 38-43.
Chen, E., Miller, G.E., Kobor, M.S., and Cole, S.W. (2011). Maternal warmth buffers the effects of low early-life socioeconomic status on pro-inflammatory signaling in adulthood. Molecular Psychiatry, 16(7), 729-737.
Cohen, S., and Pressman, S.D. (2006). Positive affect and health. [Article]. Current Directions in Psychological Science, 15(3), 122-125.
Cohen, S., Doyle, W.J., and Baum, A. (2006). Socioeconomic status is associated with stress hormones. Psychosomatic Medicine, 68(3), 414-420.
Cohen, S., Schwartz, J.E., Epel, E., Kirschbaum, C., Sidney, S., and Seeman, T. (2006). Socioeconomic status, race, and diurnal cortisol decline in the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Psychosomatic Medicine, 68(1), 41-50.
Cole, S.W., Hawkley, L.C., Arevalo, J.M., Sung, C.Y., Rose, R.M., and Cacioppo, J.T. (2007). Social regulation of gene expression in human leukocytes. Genome Biology, 8(9), R189.
Colhoun, H.M., Hemingway, H., and Poulter, N.R. (1998). Socioeconomic status and blood pressure: An overview analysis. Journal of Human Hypertension, 12(2), 91-110.
Crimmins, E.M., Kim, J. K., and Seeman, T.E. (2009). Poverty and biological risk: The earlier “aging” of the poor. The Journals of Gerontology: Series A Biological Sciences and Medical Sciences, 64(2), 286-292.
Danese, A., Moffitt, T.E., Harrington, H., Milne, B.J., Polanczyk, G., Pariante, C.M. et al. (2009). Adverse childhood experiences and adult risk factors for age-related disease depression, inflammation, and clustering of metabolic risk markers. Archives of Pediatric and Adolescent Medicine, 163(12), 1,135-1,143.
Dickerson, S.S., and Kemeny, M.E. (2004). Acute stressors and cortisol responses: A theoretical integration and synthesis of laboratory research. Psychological Bulletin, 130(3), 355-391.
Dickerson, S.S., Gruenewald, T.L., and Kemeny, M.E. (2004). When the social self is threatened: Shame, physiology, and health. Journal of Personality, 72(6), 1,191-1,216.
Diener, E., Ng, W., Harter, J., and Arora, R. (2002). Wealth and happiness across the world: Material prosperity predicts life evaluation, whereas psychosocial prosperity predicts positive feeling. Journal of Personality and Social Psychology, 99(1), 52-61.
Evans, G.W., and English, K. (2002). The environment of poverty: Multiple stressor exposure, psychophysiological stress, and socioemotional adjustment. Child Development, 73(4), 1,238-1,248.
Evans, G.W., and Kim, P. (2007). Childhood poverty and health—Cumulative risk exposure and stress dysregulation. Psychological Science, 18(11), 953-957.
Finch, C.E., and Ruvkun, G. (2001). The genetics of aging. Annual Review of Genomics and Human Genetics, 2, 435-462.
Gallo, L.C., Bogart, L.M., Vranceanu, A.M., and Walt, L.C. (2004). Job characteristics, occupational status, and ambulatory cardiovascular activity in women. Annals of Behavioral Medicine, 28(1), 62-73.
Geronimus, A.T. (1992). The weathering hypothesis and the health of African-American women and infants: Evidence and speculations. Ethnicity and Disease, 2(3), 207-221.
Geronimus, A.T., Hicken, M., Keene, D., and Bound, J. (2006). “Weathering” and age patterns of allostatic load scores among blacks and whites in the United States. American Journal of Public Health, 96(5), 826-833.
Gianaros, P.J., and Manuck, S.B. (2010). Neurobiological pathways linking socioeconomic position and health. Psychosomatic Medicine, 72(5), 450-461.
Gianaros, P.J., Horenstein, J.A., Hariri, A.R., Sheu, L.K., Manuck, S.B., Matthews, K.A., et al. (2008). Potential neural embedding of parental social standing. Social, Cognitive, and Affective Neuroscience, 3(2), 91-96.
Gianaros, P.J., Manuck, S.B., Sheu, L.K., Kuan, D.C., Votruba-Drzal, E., Craig, A.E., et al. (2011). Parental education predicts corticostriatal functionality in adulthood. Cerebral Cortex, 21(4), 896-910.
Goodman, E., McEwen, B.S., Huang, B., Dolan, L.M., and Adler, N.E. (2005). Social inequalities in biomarkers of cardiovascular risk in adolescence. Psychosomatic Medicine, 67(1), 9-15.
Gruenewald, T.L., Kemeny, M.E., Aziz, N., and Fahey, J.L. (2004). Acute threat to the social self: Shame, social self-esteem, and cortisol activity. Psychosomatic Medicine, 66(6), 915-924.
Gruenewald, T.L., Karlamangla, A.S., Greendale, G.A., Singer, B.H., and Seeman, T.E. (2007). Feelings of usefulness to others, disability, and mortality in older adults: The MacArthur Study of Successful Aging. The Journals of Gerontology: Series B Psychological Sciences and Social Sciences, 62(1), P28-P37.
Gruenewald, T.L., Cohen, S., Matthews, K.A., Tracy, R., and Seeman, T.E. (2009). Association of socioeconomic status with inflammation markers in black and white men and women in the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Social Science and Medicine, 69(3), 451-459.
Gruenewald, T.L., Liao, D., and Seeman, T.E. (2012). Contributing to others, contributing to oneself: Perceptions of generativity and health in later life. The Journals of Gerontology: Series B Psychological Sciences and Social Sciences, 67(6), 680-685.
Gruenewald, T., Karlamangla, A., Friedman, E., Almeida, D., Koretz, B., and Seeman, T. (2012a). Life Course Socioeconomic Adversity and Cortisol Activity in the Laboratory and Daily Life. Paper presented at the annual meeting of the American Psychosomatic Society, McLean, VA.
Gruenewald, T.L., Karlamangla, A.S., Hu, P., Stein-Merkin, S., Crandall, C.S., Koretz, B., et al. (2012b). History of socioeconomic disadvantage and allostatic load in later life. Social Science and Medicine, 74, 75-83.
Hajat, A., Diez-Roux, A., Franklin, T.G., Seeman, T., Shrager, S., Ranjit, N., et al. (2010). Socioeconomic and race/ethnic differences in daily salivary cortisol profiles: The multiethnic study of atherosclerosis. Psychoneuroendocrinology, 35(6), 932-943.
Harman, D. (2003). The free radical theory of aging. Antioxidants and Redox Signaling, 5(5), 557-561.
Hawkley, L.C., and Cacioppo, J.T. (2010). Loneliness matters: A theoretical and empirical review of consequences and mechanisms. Annals of Behavioral Medicine, 40(2), 218-227.
Hemingway, H., and Marmot, M. (1999). Evidence based cardiology: Psychosocial factors in the aetiology and prognosis of coronary heart disease. Systematic review of prospective cohort studies. British Medical Journal, 318(7,196), 1,460-1,467.
Hemingway, H., Shipley, M., Mullen, M.J., Kumari, M., Brunner, E., Taylor, M., et al. (2003). Social and psychosocial influences on inflammatory markers and vascular function in civil servants (the Whitehall II Study). American Journal of Cardiology, 92(8), 984-987.
Henry, J.P. (1993). Biological basis of the stress response. News in Physiological Sciences, 8, 69-73.
Holt-Lunstad, J., Smith, T.B., and Layton, J.B. (2010). Social relationships and mortality risk: A meta-analytic review. PLoS Medicine, 7(7), 1-20.
House, J.S., Landis, K.R., and Umberson, D. (1988). Social relationships and health. Science, 241(4865), 540-545.
Janicki-Deverts, D., Cohen, S., Adler, N.E., Schwartz, J.E., Matthews, A., and Seeman, T.E. (2007). Socioeconomic status is related to urinary catecholamines in the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Psychosomatic Medicine, 69(6), 514-520.
Kaplan, G.A., and Keil, J.E. (1993). Socioeconomic factors and cardiovascular disease: a review of the literature. Circulation, 88(4 Pt 1), 1,973-1,998.
Keyes, C.L.M. (1998). Social well-being. Social Psychology Quarterly, 61(2), 121-140.
Keyes, C.L. (2002). The mental health continuum: From languishing to flourishing in life. Journal of Health and Social Behavior, 43(2), 207-222.
Keyes, C.L., Shmotkin, D., and Ryff, C.D. (2002). Optimizing well-being: The empirical encounter of two traditions. Journal of Personality and Social Psychology, 82(6), 1,007-1,022.
Kiecolt-Glaser, J.K., McGuire, L., Robles, T.F., and Glaser, R. (2002). Psychoneuroimmunology: Psychological influences on immune function and health. Journal of Consulting and Clinical Psychology, 70(3), 537-547.
Kirkwood, T.B.L., and Austad, S.N. (2000). Why do we age? Nature, 408(6,809), 233-238.
Kivimaki, M., Lawlor, D.A., Smith, G.D., Keltikangas-Jarvinen, L., Elovainio, M., Vahtera, J., et al. (2006a). Early socioeconomic position and blood pressure in childhood and adulthood—The Cardiovascular Risk in Young Finns Study. Hypertension, 47(1), 39-44.
Kivimaki, M., Smith, G.D., Elovainio, M., Pulkki, L., Keltikangas-Jarvinen, L., Talttonen, L., et al. (2006b). Socioeconomic circumstances in childhood and blood pressure in adulthood: The Cardiovascular Risk in Young Finns Study. Annals of Epidemiology, 16(10), 737-742.
Koster, A., Bosma, H., Penninx, B.W., Newman, A.B., Harris, T.B., van Eijk, J.T., et al. (2006). Association of inflammatory markers with socioeconomic status. The Journals of Gerontology: Series A Biological Sciences and Medical Sciences, 61(3), 284-290.
Kumari, M., Badrick, E., Chandola, T., Adler, N.E., Epel, E., Seeman, T., et al. (2010). Measures of social position and cortisol secretion in an aging population: Findings from the Whitehall II Study. Psychosomatic Medicine, 72, 27-34.
Li, L., Power, C., Kelly, S., Kirschbaum, C., and Hertzman, C. (2007). Life-time socioeconomic position and cortisol patterns in mid-life. Psychoneuroendocrinology, 32(7), 824-833.
Loucks, E.B., Magnusson, K.T., Cook, S., Rehkopf, D.H., Ford, E.S., and Berkman, L.F. (2007a). Socioeconomic position and the metabolic syndrome in early, middle, and late life: Evidence from NHANES 1999-2002. Annals of Epidemiology, 17(10), 782-790.
Loucks, E.B., Rehkopf, D.H., Thurston, R.C., and Kawachi, I. (2007b). Socioeconomic disparities in metabolic syndrome differ by gender: Evidence from NHANES III. Annals of Epidemiology, 17(1), 19-26.
Loucks, E.B., Lynch, J.W., Pilote, L., Fuhrer, R., Almeida, N.D., Richard, H., et al. (2009). Life-course socioeconomic position and incidence of coronary heart disease. American Journal of Epidemiology, 169(7), 829-836.
Marmot, M. (2006). Health in an unequal world: Social circumstances, biology, and disease. Clinical Medicine, 6(6), 559-572.
Marmot, M.G., Shipley, M.J., Hemingway, H., Head, J., and Brunner, E.J. (2008). Biological and behavioural explanations of social inequalities in coronary heart disease: The Whitehall II Study. Diabetologia, 51(11), 1,980-1,988.
Martin, G.M. (2011). The biology of aging: 1985-2010 and beyond. FASEB Journal, 25(11), 3,756-3,762.
Matthews, K.A., and Gallo, L.C. (2011). Psychological perspectives on pathways linking socioeconomic status and physical health. In S.T. Fiske, D.L. Schacter, and S.E. Taylor (Eds.), Annual Review of Psychology (vol. 62, pp. 501-530). Palo Alto, CA: Annual Reviews.
McEwen, B. (1998). Stress, adaptation, and disease. Allostasis and allostatic load. Annals of the New York Academy of Sciences, 840, 33-44.
McEwen, B.S., and Gianaros, P.J. (2010). Central role of the brain in stress and adaptation: Links to socioeconomic status, health, and disease. Annals of the New York Academy of Sciences, 1(186), 190-222.
McEwen, B.S., and Seeman, T. (1999). Protective and damaging effects of mediators of stress. Elaborating and testing the concepts of allostasis and allostatic load. Annals of the New York Academy of Sciences, 896, 30-47.
McEwen, B., and Stellar, E. (1993). Stress and the individual. Mechanisms leading to disease. Archives of Internal Medicine, 153(18), 2,093-2,101.
McLaren, L. (2007). Socioeconomic status and obesity. Epidemiologic Reviews, 29, 29-48.
Miller, G., and Chen, E. (2007). Unfavorable socioeconomic conditions in early life presage expression of proinflammatory phenotype in adolescence. Psychosomatic Medicine, 69(5), 402-409.
Miller, G., Chen, E., and Cole, S.W. (2009). Health psychology: Developing biologically plausible models linking the social world and physical health. Annual Review of Psychology, 60, 501-524.
Miller, G.E., Chen, E., Fok, A.K., Walker, H., Lim, A., Nicholls, E.F., et al. (2009). Low early-life social class leaves a biological residue manifested by decreased glucocorticoid and increased proinflammatory signaling. Proceedings of the National Academy of Sciences of the United States of America, 106(34), 14,716-14,721.
Okamoto, K., and Tanaka, Y. (2004). Subjective usefulness and 6-year mortality risks among elderly persons in Japan. The Journals of Gerontology: Series B Psychological Sciences and Social Sciences, 59(5), P246-P249.
Owen, N., Poulton, T., Hay, F.C., Mohamed-Ali, V., and Steptoe, A. (2003). Socioeconomic status, C-reactive protein, immune factors, and responses to acute mental stress. Brain Behavior, and Immunity, 17(4), 286-295.
Parsons, P. A. (2003). From the stress theory of aging to energetic and evolutionary expectations for longevity. Biogerontology, 4(2), 63-73.
Pitkala, K.H., Laakkonen, M.L., Strandberg, T.E., and Tilvis, R.S. (2004). Positive life orientation as a predictor of 10-year outcome in an aged population. Journal of Clinical Epidemiology, 57(4), 409-414.
Pollitt, R.A., Rose, K.M., and Kaufman, J.S. (2005). Evaluating the evidence for models of life course socioeconomic factors and cardiovascular outcomes: A systematic review. BMC Public Health, 5(1), 7.
Poulton, R., Caspi, A., Milne, B.J., Thomson, W.M., Taylor, A., Sears, M.R., et al. (2002). Association between children’s experience of socioeconomic disadvantage and adult health: A life-course study. Lancet, 360(9,346), 1,640-1,645.
Pressman, S.D., and Cohen, S. (2005). Does positive affect influence health? Psychological Bulletin, 131(6), 925-971.
Ramsay, S.E., Morris, R.W., Whincup, P.H., Papacosta, O., Rumley, A., Lennon, L., et al. (2009). Socioeconomic inequalities in coronary heart disease risk in older age: Contribution of established and novel coronary risk factors. Journal of Thrombosis and Haemostasis, 7(11), 1,779-1,786.
Rosvall, M., Engstrom, G., Berglund, G., and Hedblad, B. (2008). C-reactive protein, established risk factors and social inequalities in cardiovascular disease—The significance of absolute versus relative measures of disease. BMC Public Health, 8, 10.
Ryff, C.D. (1989). Happiness is everything, or is it? Explorations on the meaning of psychological well-being. Journal of Personality and Social Psychology, 57(6), 1,069-1,081.
Ryff, C.D., and Keyes, C. L. (1995). The structure of psychological well-being revisited. Journal of Personality and Social Psychology, 69(4), 719-727.
Ryff, C.D., Singer, B.H., and Dienberg Love, G. (2004). Positive health: Connecting well-being with biology. Philosophical Transactions of the Royal Society B: Biological Sciences, 359(1,449), 1,383-1,394.
Ryff, C.D., Dienberg Love, G., Urry, H.L., Muller, D., Rosenkranz, M.A., Friedman, E.M., et al. (2006). Psychological well-being and ill-being: Do they have distinct or mirrored biological correlates? Psychotherapy and Psychosomatics, 75(2), 85-95.
Seeman, T.E., and Crimmins, E. (2001). Social environment effects on health and aging: Integrating epidemiologic and demographic approaches and perspectives. Annals of the New York Academy of Sciences, 954, 88-117.
Seeman, T., and McEwen, B. (1996). Impact of social environment characteristics on neuroendocrine regulation. Psychosomatic Medicine, 58(5), 459-471.
Seeman, T.E., Crimmins, E., Huang, M.H., Singer, B., Bucur, A., Gruenewald, T., et al. (2004). Cumulative biological risk and socio-economic differences in mortality: MacArthur Studies of Successful Aging. Social Science and Medicine, 58(10), 1,985-1,997.
Seeman, T., Epel, E., Gruenewald, T., Karlamangla, A., and McEwen, B.S. (2010). Socioeconomic differentials in peripheral biology: Cumulative allostatic load. Annals of the New York Academy of Sciences, 1,186, 223-239.
Senese, L.C., Almeida, N.D., Fath, A.K., Smith, B.T., and Loucks, E.B. (2009). Associations between childhood socioeconomic position and adulthood obesity. Epidemiologic Reviews, 31(1), 21-51.
Sloan, R.P., Huang, M.H., Sidney, S., Liu, K., Williams, O.D., and Seeman, T. (2005). Socioeconomic status and health: Is parasympathetic nervous system activity an intervening mechanism? International Journal of Epidemiology, 34(2), 309-315.
Sobal, J., and Stunkard, A. J. (1989). Socioeconomic status and obesity: A review of the literature. Psychological Bulletin, 105(2), 260-275.
Sohal, R.S., and Orr, W.C. (2012). The redox stress hypothesis of aging. Free Radical Biology and Medicine, 52(3), 539-555.
Sohal, R.S., Mockett, R.J., and Orr, W.C. (2002). Mechanisms of aging: An appraisal of the oxidative stress hypothesis. Free Radical Biology and Medicine, 33(5), 575-586.
Steptoe, A., and Wardle, J. (2011). Positive affect measured using ecological momentary assessment and survival in older men and women. Proceedings of the National Academy of Sciences of the United States of America, 108(45), 18,244-18,248.
Steptoe, A., Feldman, P.J., Kunz, S., Owen, N., Willemsen, G., and Marmot, M. (2002). Stress responsivity and socioeconomic status: A mechanism for increased cardiovascular disease risk? European Heart Journal, 23(22), 1,757-1,763.
Steptoe, A., Kunz-Ebrecht, S., Owen, N., Feldman, P. J., Willemsen, G., Kirschbaum, C., et al. (2003a). Socioeconomic status and stress-related biological responses over the working day. Psychosomatic Medicine, 65(3), 461-470.
Steptoe, A., Willemsen, G., Kunz-Ebrecht, S.R., and Owen, N. (2003b). Socioeconomic status and hemodynamic recovery from mental stress. Psychophysiology, 40(2), 184-191.
Steptoe, A., Dockray, S., and Wardle, J. (2009). Positive affect and psychobiological processes relevant to health. Journal of Personality, 77(6), 1,747-1,776.
Sterling, P. (2004). Principles of allostasis: Optimal design, predictive regulation, pathophysiology and rational therapeutics. In J. Schulkin (Ed.), Allostasis, Homeostasis, and the Costs of Adaptation. Cambridge, UK: Cambridge University Press.
Sterling, P., and Eyer, J. (1988). Allostasis: A new paradigm to explain arousal pathology. In S. Fisher and J. Reason (Eds.), Handbook of Life Stress, Cognition and Health (pp. 631-651). New York: John Wiley & Sons.
Taylor, S., Repetti, R., and Seeman, T. (1997). Health psychology: What is an unhealthy environment and how does it get under the skin? Annual Review of Psychology, 48, 411-447.
Uchino, B.N., Cacioppo, J.T., and Kiecolt-Glaser, J.K. (1996). The relationship between social support and physiological processes: A review with emphasis on underlying mechanisms and implications for health. Psychological Bulletin, 119(3), 488-531.


























