
3 Arctic Oil Spill Response Research
Large-scale work on oil spills in sea ice began in the early 1970s in Canada and the United States with the Beaufort Sea Project and the Outer Continental Shelf Environmental Assessment Program, respectively (e.g., Lewis, 1976). In both programs, laboratory and tank research was carried out, but the Beaufort Sea Project also involved the first significant field release of oil under growing sea ice during the winter season. More recently, in 2007-2010 an Oil in Ice Joint Industry Program1 (JIP) was managed by the Foundation for Scientific and Industrial Research (SINTEF). This large research effort was sponsored by six companies. It included a wide range of laboratory, tank, and field tests, including two cruises in the Norwegian Barents Sea where oil was deliberately released to assess weathering, burning, herding agents, skimmers, and in situ burning (ISB) (Sørstrøm et al., 2010). In 2012, an Arctic Oil Spill Response Technology JIP,2 with nine participating companies, launched a range of research projects on all aspects of responding to oil spills in the Arctic. This is the largest research program of its kind and is scheduled to continue through 2015 (Mullin, 2012). The U.S. Coast Guard (USCG) recently carried out a series of field trials with icebreakers in the Great Lakes and in the Arctic to test the limits and capabilities of response equipment and remote sensing platforms (Hansen and Lewandowski, 2011; USCG, 2013a,b).
This chapter describes the current state of oil spill response research. It begins with a discussion of a risk-based framework for thinking about oil spill impacts and then provides a summary of the state of knowledge governing our understanding of expected oil behavior in ice. The chapter then moves through a description of various oil spill response options and promising new concepts.
RISK-BASED FRAMEWORK
The committee chose to look at oil spill response in a risk-based framework. A classic risk matrix would depict the risk of an oil spill as a function of its probability (or likelihood) versus the magnitude of its impact (Figure 3.1). Therefore, there are two ways to reduce the risk related to oil
_____________
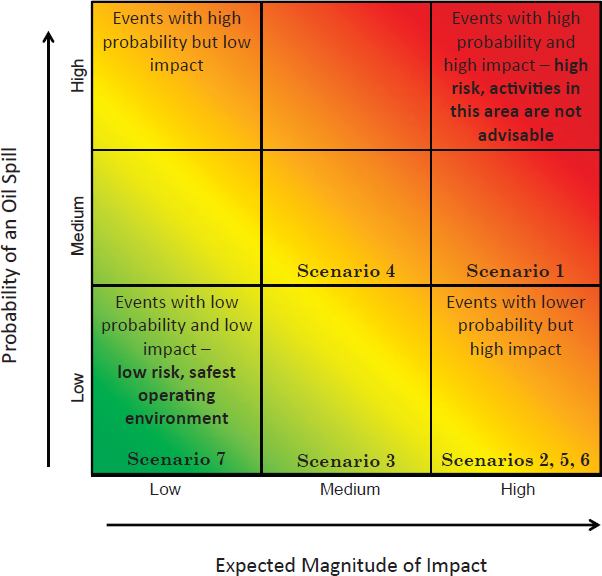
Figure 3.1 Oil spill risk matrix with low-probability/low-impact events in the lower left corner (low risk) and high-probability/high-impact events in the upper right corner (high risk). Risk increases as one departs from the lower left corner of the matrix. Scenarios related to different oil spill response events are mapped onto the risk matrix; numbers correspond to scenarios presented in Chapters 4 and 5. SOURCE: Committee.
spills—reduce the probability of an event, or reduce the magnitude of the impact. Environmental risks include not only low-probability, high-impact events like the Deepwater Horizon oil spill, but also small oil spill events with greater likelihoods. The probability of a spill is related to the type and condition of a vessel, pipeline, rig, or storage facility; the accuracy and availability of maps and charts; season, weather conditions, and presence or absence of ice; the behaviors, decisions, and levels of experience of key personnel; and the availability of infrastructure to support spill response (avail-
ability of a capping stack, for example). Spill impacts will be related to the amount and type of oil released; meteorological, oceanographic, and geologic conditions, including ice characteristics and cover; the degree of interaction between spilled oil and valued ecosystem elements; and the availability of response infrastructure and trained personnel. The choice and efficacy of oil spill response activities (commonly referred to as “countermeasures”) will also affect the magnitude of the impacts.
Risk scenarios were developed to highlight different facets of Arctic oil spill response. Although no formal risk assessments were undertaken, scenarios were developed for discussion purposes and to provide illustrations of the different types of events that could potentially result in an Arctic oil spill response. This is similar to processes followed in a variety of reports (e.g., Arctic Council, 2009, 2013). Scenarios are characterized as having relatively low to high probabilities—based on exposure, frequency, and relative risk levels—and are mapped onto the risk matrix shown in Figure 3.1. Impact levels are characterized as relatively low to high, based on potential oil spill volume or quantity and the ability of resources to reach and respond to the spill. Scenarios are briefly described below, with full discussions found in Chapters 4 and 5.
Scenarios 1 and 4—a passenger cruise or research ship accident and a barge separated from its tow—are determined to be of higher probability due to frequent seasonal operations, often in shallow, nearshore waters. The other scenarios—a large oil tanker accident (Scenario 2), a bulk carrier driven ashore (Scenario 3), a subsea pipeline break (Scenario 5), a well blowout (Scenario 6), and land-based oil tank spills (Scenario 7)—are thought to be less likely events due to existing containment and prevention systems, including established navigation routes for bulk ore carriers. Large tanker accidents, subsea pipeline breaks, and well blowouts are considered to have relatively high impacts, primarily because of large spill volumes and the remoteness of spill locations. The structural failure of a land-based oil tank would be relatively low impact because of the limited spill size, existing spill response equipment, and ability to reach the site with existing onshore resources.
OIL PROPERTIES
Crude oil is composed of a complex mixture of paraffinic, naphthenic, and aromatic hydrocarbons. Oils can differ from each other in a variety of ways, including density and sulfur content. The physical and chemical properties of an oil are not static but can vary between regions, within wells at the same location, and even within a given well over time (EPA, 2011). Key oil properties in cold water environments include measures of the American Petroleum Institute (API) gravity3 (an indicator of relative density in comparison to water), pour point (the temperature at which a fluid ceases to readily flow), and viscosity. As temperature decreases, viscosity increases and the possibility of going below the pour point becomes more likely. These properties are often considered in early stages of an oil spill response because they usually help define the most effective response options.
There are four standard groupings of oil types (ITOPF, 2013/2014). Group I oils, which include diesel fuel, are nonpersistent—they dissipate rapidly through evaporation and natural dispersion within a few hours and are unlikely to form emulsions, in which water droplets become entrained
_____________
3 API gravity is measured in degrees, and is calculated using the following equation: API gravity = (141.5/SG) – 131.5, where SG is the specific gravity of the petroleum liquid at 60°F.
in the oil through mixing. Group II and III oils will partially dissipate, losing up to 40% of their volume through evaporation. These oils are likely to increase in volume because of their tendency to form viscous water-in-oil emulsions. This also leads to a lack of natural dispersion, especially in Group III oils. Group IV oils have low volatility and are highly viscous. They are highly persistent and are unlikely to evaporate or disperse (ITOPF, 2013/2014). The properties of a fresh oil may change with time, as the petroleum reservoir changes during production. Because of this, a given set of measurements to characterize a fresh oil represents a snapshot in time that may need to be updated. Nevertheless, the classification of oils into specific groups allows broad understanding of how they will behave under different environmental circumstances.
The development of biofuels has somewhat complicated this scheme, as they represent a class of materials that does not readily fit into the categories developed for petroleum products. While the viscosity and relative density of biofuels may be similar to crude oils and petroleum-based fuels, other properties may be quite different, especially as they relate to effectiveness of oil spill response methods. Ethanol from plant sources such as corn, sugar beets, or sugar cane is quite different from crude oil-based fuels because of its infinite solubility in water. In the event of a spill, ethanol would be more akin to a chemical spill rather than an oil spill. Another common form of biofuel is biodiesel, which may be made from either plant- or animal-based materials—animal fats such as tallow and lard; plant oils such as corn, canola, sunflower, and rapeseed; and recycled grease and used cooking oils. Recently, the U.S. Air Force and Purdue University have focused on biofuel for jet aircraft use derived from the Camelina plant species.4
Biodiesel generally has higher viscosity, flash point, and pour point compared to petroleum-derived diesel, with similar specific gravity (NREL, 2009). Unlike conventional diesel, biodiesel may be suitable for mechanical collection in the event of a spill because of its higher flash point and pour point, especially in colder environments. However, its response to dispersants may be quite different from that of crude oil-derived products because the biodiesel’s range of molecular components is narrower.
NORTH SLOPE OIL PROPERTIES
Oil properties and characteristics for specific fields are provided to regulatory agencies by industry when applying for exploration drilling and development permits. For exploration drilling permits, oil properties are estimated from other oils that have been discovered in the region (see Figure 1.2 for locations of many of the fields mentioned in this section). Light (high API gravity or low density) crude oils with 32-57° API gravity and very low sulfur content (0.0-0.2%) have been encountered in Chukchi Sea wells (Burger, Popcorn, and Klondike). These petroleum hydrocarbons are similar to oils found at the Umiat, Alpine, Tarn, and related fields in northern Alaska. In the Beaufort Sea, several of the oil occurrences in nearshore wells like Liberty are related to the medium-gravity (25-30° API), high-sulfur (1.0-2.0%) Prudhoe Bay and Kuparuk oils of northern Alaska. Farther offshore, oil occurrences in the Beaufort Sea (Northstar, Kuvlum) are similar to the high-API gravity (30-40°),
_____________
4 See http://www.purdue.edu/newsroom/releases/2013/Q3/purdue-jet-to-fly-to-international-air-show-powered-bybiofuel.html.
low-sulfur (0.0-0.5%) oils known from the northernmost Arctic National Wildlife Refuge (described by Lillis et al., 1999). The offshore Hammerhead oil field (25 km north of Point Thomson) features an anomalously heavy crude oil (19° API) that has been altered by bacterial degradation, but is probably also related to the offshore Beaufort oils (Banet, 1994; Curiale, 1995; Lillis et al., 1999).
The Bureau of Ocean Energy Management (BOEM) has also identified 97 suspected natural oil seeps, 80% of which are in the Chukchi Sea, in unpublished data collected by industry in the 1980s and 1990s. These seep data could provide suggestions of where additional sampling or analysis might be targeted. However, the available seep data are too elementary for biomarker fingerprinting of oil types and making correlations to oils encountered in exploratory wells or oil fields (presentation by Kirk Sherwood, BOEM, March 2013).
OIL WEATHERING
While properties described in the above section can characterize a crude oil at a particular point in time, weathering after being spilled can change its overall chemical and physical properties. Weathering involves processes that are typically experienced in an open ocean environment—evaporation, dissolution, dispersion, oxidation, emulsification, biodegradation, and sedimentation (Sørstrøm et al., 2010). In the case of a subsea oil spill, surface weathering processes may not be significant if oil does not reach the surface. Key factors that impact weathering include air and water temperatures; the presence of waves, currents, and wind; exposure to sunlight; the presence of ice or snow; and the presence of natural sediments (Figure 3.2). Changes in an oil due to weathering affect spill response options and oil interactions with organisms and ecosystems.
Oil characteristics will also determine to what extent different weathering processes will be significant. Some processes affect oil quickly (e.g., evaporation in a warm environment), while others may take longer (e.g., biodegradation).5 Evaporation and dissolution cause the loss of the lightest chemical compounds in oil; these smaller-molecule, more volatile compounds contribute to lower viscosity, lower density, and greater solubility. Their loss from an oil slick can have a significant effect on the bulk characteristics of any remaining floating oil.
Evaporation is the most rapid weathering process. It accounts for the loss of 20-50% of many crude oils, 75% or more of some refined petroleum products, but only 10% or less of residual fuel oils (NRC, 2003, 2005). The evaporative loss of a light oil under three different ice coverage levels (open water, 30% ice coverage, and 90% ice coverage) at various current and wave height conditions with different air temperatures (−15°C to about −5°C) was studied by Brandvik and Faksness (2009). They reported that evaporative loss was estimated to be 30% for open water, 25% for the lighter ice coverage, and 19% for the heavier ice coverage, due to differences in oil film thicknesses.
Another important process is emulsification. As oil resides on the water’s surface, there is a general tendency for it to incorporate water and form an oil-in-water emulsion. The addition of mixing energy from waves can accelerate the process. As emulsification occurs, there is an increase in volume, viscosity, and water content, each of which can influence the efficiency of response options. For example, ISB loses its applicability once the water content of the emulsion begins to exceed
_____________
5 ITOPF online, 2012; see http://www.itopf.com/marine-spills/fate/weathering-process/.
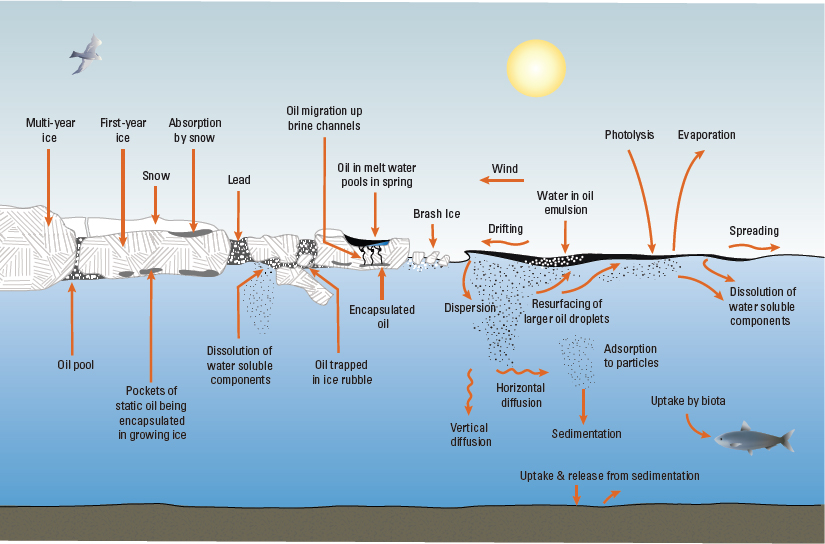
Figure 3.2 Environmental processes that affect oil behavior and weathering in open water and in ice. SOURCE: Modified from Daling et al. (1990) and A. Allen.
30-50%. Additionally, the use of dispersants may become less effective once the emulsion viscosity exceeds a threshold, although this will differ from one crude oil to another (Daling et al., 1990; Federal Interagency Solutions Group, 2010).6
Photo-oxidation is an additional process that contributes to the degradation and transformation of crude oil compounds after release to the environment (Garrett et al., 1998; Dutta and Harayama, 2000; Prince et al., 2003b). Light intensity near the water’s surface will be lower at northern latitudes due to a low angle of incidence, and the region has a wide range of daylight hours based on the season. Due to longer exposure times during the summer months, photo-oxidation may be a much more important process for oil degradation in the Arctic than in more temperate climates (Serova, 1992; Ivanov et al., 2005).
It should also be noted that not all of the chemicals that make up crude oil biodegrade at the same rate. Higher-molecular-weight components, including polycyclic aromatic hydrocarbons, cyclic alkanes, and naphtheno-aromatic hydrocarbons, may persist for some time.
While weathering trends (Table 3.1) provide some generalities, each real-world case will be different. Weathering may alter the potential toxicity of crude oil components through processes such as evaporation and dissolution of low-molecular-weight components, while photo-oxidation near the water surface could create products not initially present in the oil. There are a number of modeling tools available to assist in weathering predictions,7 although the underlying assumptions and potential limitations of models and the information from which they operate need to be understood and questioned when necessary. In general, evaporation rates take into account compositional details of specific crude oils. In the event that a crude oil released into the environment is not known completely, the use of another with similar characteristics may be used to gain an approximation of its behavior.8
OIL BEHAVIOR IN ICE
The presence of sea ice, discussed in Chapter 2, affects oil weathering processes and the overall behavior of oil in Arctic waters. In many cases, processes that affect traditional oil behavior in open water like evaporation, emulsification, and natural dispersion are slowed down or eliminated for extended periods of time. Laboratory, basin, and field experiments on oil behavior and weathering under Arctic conditions have been conducted independently in Canada and Norway (Sørstrøm et al., 1994, 2010; Brandvik and Faksness, 2009; Buist et al., 2009). Dickins (2011) summarized the behavior of oil in ice, derived from these findings and from direct observations from large-scale field trials dating back to 1972 (McMinn, 1972; NORCOR Engineering & Research Ltd., 1975;
_____________
6 Laboratory assessment of emulsions that were formed during the Macondo Well release indicated that they were susceptible to dispersion at reasonably low dispersant-to-oil ratios. See, for example, work done by SINTEF and documented within (Federal Interagency Solutions Group, 2010).
7 For example, see the following models: ADIOS2™, developed by the National Oceanic and Atmospheric Administration (NOAA) (Jones, 1997); the Type A model developed by ASA (Reed et al., 1989; French et al., 1996); the OWM model developed by SINTEF (Reed, Singsaas et al., 2001); the OSCAR model developed by SINTEF (1995, 1999).
8 Environment Canada developed an online database of crude oil properties that is still useful when attempting to determine weathering characteristics: http://www.etc-cte.ec.gc.ca/databases/Oilproperties/oil_prop_e.html.
Table 3.1 Chemical and Physical Changes to Crude Oil from Weathering
| Property | Potential Change upon Weathering | Cause | |||
| Oil viscosity | Increase (by an order of magnitude) | Loss of low-MW components from evaporation or dissolution | |||
| Increase (several orders of magnitude) | Formation of water-in-oil emulsions (mousse) | ||||
| Oil specific gravity | Increase | Loss of low-MW components from evaporation or dissolution | |||
| Oil slick volume | Decrease | Loss of low-MW components from evaporation or dissolution; loss of mass due to entrainment of oil droplets due to breaking waves | |||
| Increase (by magnitude factor of 3 to 5) | Formation of emulsions (mousse) | ||||
| Potential toxicity | Decrease | Loss of low-MW components from evaporation or dissolution | |||
| Increase | Formation of photo-oxidation products near water surface | ||||
NOTE: MW = molecular weight.
Dickins and Buist, 1981; Nelson and Allen, 1982; Buist and Dickins, 1987; Sørstrøm et al., 1994, 2010; Brandvik et al., 2006; Dickins et al., 2008). Figure 3.2 shows a schematic of different potential oil-ice interactions.
WEATHERING
Oil spilled during freeze-up will be affected by evaporation, dissolution, emulsification, and natural dispersion to some degree. Most oil spilled during the freeze-up period will remain on the surface of the ice or will migrate up, where it will be affected by evaporation. The evaporation rate is partially controlled by oil slick thickness—thicker oil slicks will evaporate more slowly than slicks in open water. Cold temperatures reduce evaporation rates, as would snow forming a thin film on
or covering the oil. Even when covered with snow, oil on an ice surface will lose approximately the same amount to evaporation as it would on water in more temperate waters (Buist et al., 2009).
Brandvik and Faksness (2009) reported that the formation of water-in-oil emulsions under Arctic conditions at a test site inside the ice pack, far removed from the effects of ocean swell, was significantly reduced. The presence of ice effectively dampens both wind wave activity and swell (depending on distance from the pack edge; e.g., Wadhams et al., 1988), increases the thickness of the oil slick, and reduces its surface area. In contrast, within the marginal ice zone at the edge of the pack, there is enough wave action to grind individual floes together and accelerate emulsification. This process was observed by Buist et al. (2009) and Payne et al. (1991) when they did a tank experiment with crude oil spilled in ice in an induced wave environment.
Movement and Drift Rates
Oil trapped within pack ice tends to move with the ice, which is in turn driven by currents and wind; oil in more open drift ice will be less strictly herded by the ice. Under-ice currents in most Arctic nearshore areas are not strong enough to spread the oil far beyond its initial contact with the ice. A 20 cm/s (~0.5 kn) current is needed to initiate and sustain movement of oil under the surface of the oil (Buist et al., 2009).
Spreading
Spreading behavior is one of the properties that is most different between oil spills in ice compared to open water. Oil spilled in the presence of ice is often naturally contained within a relatively small area, which has mostly positive implications for response and recovery options (Buist and Dickins, 1987). Table 3.2 compares the predicted areas and thicknesses covered by a 1,600 m3 (10,000 barrels [bbl]) crude oil spill on open water, under ice, and on smooth sea ice with and without snow. The table is intended for illustrative purposes to demonstrate the dramatic difference
| Open Water | Under Solid Midwinter Ice | On Smooth Ice | ||
| Ice | Snow | |||
| Final average oil thickness (mm) | 0.016 | 40 to 90 | 3 | 40 |
| Final area (ha) | 10,000 | 7 to 70 | 50 | 4 |
NOTE: The maximum pool depth under solid midwinter ice is determined by the depth of the under-ice depressions, which become deeper as the ice increases and deforms over winter. The final area is determined by both the volume of under-ice depressions and how they fill with oil.
SOURCE: SL Ross Environmental Research Ltd. et al., 2010.
in contaminated areas between oil on water, under ice, and on ice. Under solid midwinter ice, the maximum pool depth varies based on the depth of under-ice depressions, and the contaminated area is determined by both the volume of those depressions and the ways they fill with oil.
The spread of oil is reduced by ice and snow, with resulting oil slicks that are much thicker than those in open water (Dickins, 2011). In practice, an oil slick on open water will spread to cover areas with different equilibrium thickness after some time has elapsed. The majority of the oil is contained within a relatively small, thick patch, while the rest spreads out as a thin film or sheen over a much larger area. In contrast, the maximum contaminated areas under or on ice are predicted to be hundreds to thousands of times smaller. This can be a critical difference when looking at the potential for wildlife exposure, as thin films on the surface create significant risks to waterfowl.
The degree of natural oil containment provided by close pack ice depends on ice concentration and other variables. Generally, the presence of 6/10 ice concentration leads to a slick that is less than half the area of the same oil volume in open water. However, in open drift ice at concentrations less than 6/10, spreading rates approach those for open water (Buist and Dickins, 1987).
Encapsulation, Migration, and Release
Oil density and turbulence tend to govern how much oil becomes incorporated into growing ice, while the oil viscosity is a factor in how it will break down (Dickins, 2011). Heavier fuel oil particles can remain suspended at depth in slush ice; this was observed in the 1979 Kurdistan tanker oil spill between Nova Scotia and Newfoundland (Vandermeulen and Buckley, 1985).
Oil spilled under new ice will likely become encapsulated within 12 to 24 hours (NORCOR Engineering & Research Ltd., 1975; Dickins and Buist, 1999; Brandvik et al., 2006). New ice grows beneath the spilled oil, trapping the oil between layers of ice and isolating it from the ocean. Oil may migrate to the surface under thin new ice (less than ~10 cm thick) but will be trapped as the ice solidifies (Dickins, 2011). While lighter fuel oils may surface quickly, heavy fuel oils may be suspended in slush ice. Oil spilled under ice in late winter is unlikely to become encapsulated due to the slowdown in the rate of ice growth. In the spring, the trapped oil migrates vertically though brine channels in the ice sheet (NORCOR Engineering & Research Ltd., 1975; Brandvik et al., 2006; Dickins, 2011).
In an experiment that was part of the 1974-1975 Beaufort Sea Project, over 80% of spilled oil migrated to the ice surface and floated on melt pools by early summer (NORCOR Engineering & Research Ltd., 1975). The oil appeared as essentially fresh crude. Once the oil was exposed to the atmosphere, evaporation occurred at rates similar to spills in southern locations (NORCOR Engineering & Research Ltd., 1975). Oil that is distributed as fine droplets under the ice may migrate more slowly to the surface, a situation that could occur during a subsea blowout with a large gas volume (Dickins and Buist, 1981). In those cases, oil may not be exposed at the surface until the ice begins to melt (Dickins, 2011). As ice melts, encapsulated or surface oil can end up in the water, forming thin oil films before it is naturally dispersed due to wave action.
There are two areas where the knowledge of oil behavior is limited, due to a lack of field observations. The first is the behavior of oil spilled under multiyear ice. The only field experiment
designed to explore this behavior provided limited answers (Comfort and Purves, 1982). The second involves processes governing the interaction of oil with new and developing ice during and following freeze-up. This has been addressed by a tank study that was funded through the European Union (Wilkinson et al., 2014).
Developing knowledge of how to prepare for and respond to the possibility of Arctic oil spills is linked to the ability to safely conduct deliberate field-scale oil releases. A number of successful projects over the past four decades have demonstrated that having the ability to conduct deliberate and controlled releases of oil into the marine environment provides an important opportunity to advance the state of knowledge in all aspects of Arctic spill response. When done carefully, these field releases have few or no discernible negative environmental impacts. Important spinoffs from field trials include the opportunity for training of oil spill responders and public consultation to transfer understanding and knowledge.
Over the past 15 years, it has become much harder to obtain the necessary permits in the United States to conduct deliberate oil releases (Dickins, 2011). In the absence of any clear process to permit such work, scientists and engineers have looked toward other countries such as Norway, which has consistently supported this type of research by permitting spills in ice when clear research needs, methods, and goals, combined with responsible cleanup and monitoring plans, have been established (e.g., Sørstrøm et al., 1994, 2010). However, conditions are not completely equivalent to those encountered in the U.S. Arctic. Carefully planned field releases in areas potentially impacted by exploration, such as Alaska, Greenland, Canada, and Russia, could improve evaluation of new response strategies and understanding of oil-in-ice interactions. Dickins (2011) summarized the benefits of previous experimental field releases for research purposes. While there is broad support among industry and other stakeholders, there have been no successful permits to conduct a deliberate field release in U.S. waters since the early 1990s. There has been some interest from the federal government as well. The Interagency Coordinating Committee on Oil Pollution Research has been in discussion with the Environmental Protection Agency (EPA) and other federal agencies, with an aim to improve the permitting process for deliberate oil releases for experimental purposes (Eric Miller, personal communication, 2013). In 2013, the Bureau of Safety and Environmental Enforcement (BSEE) issued a solicitation for a joint industry project to assess the need for a field release of oil, dispersants, and natural gas in the U.S. outer continental shelf, with the possibility of an actual release. BSEE and EPA are also working together to develop a simulant that would successfully mimic the behavior of oil droplets in water.9
OIL SPILL SOURCES AND VOLUMES
Three potential point sources for oil spills in the U.S. Arctic are oil and gas wells and pipelines, ships (large oil tankers, bulk carriers, and fuel barges), and land-based municipal fuel storage tanks. The following section discusses likely spill volumes in each of these cases.
If a blowout were to occur in an exploratory well in the Chukchi or Beaufort Seas, the possible rate of uncontrolled flow would be dependent on a number of variables. These include the reser-
_____________
9 See http://www.bsee.gov/Research-and-Training/Oil-Spill-Response-Research/Projects/Project1029/.
| Model | Worst Case Discharge (bbl/day) | Oil Discharge at Day 30 (bbl) (Required by Regulations) | Relief Well Period (days) | Oil Discharge at End of Relief Well Period | Model Oil Gravity (aAPI) |
| BOEM Beaufort VLOS | 69,271 | 1,140,655 | 300 | 3,922,903 | 26 |
| BOEM Chukchi VLOSa | 61,672 | 1,148,300 | 46 | 1,552,400 | 35 |
NOTE: The relief well period for the Beaufort Sea assumes that ice conditions prevent access to the well throughout the winter.
a BOEM (2011).
SOURCE: Presentation by Kirk Sherwood, BOEM, March 2013.
voir characteristics (e.g., porosity, permeability, pressure, temperature, oil viscosity, gas content, and compressibility), wellbore configuration, and the ambient pressure at the blowout discharge point at the seafloor or at the rig. BOEM models “very large oil spills” (VLOSs) and “worst case discharges” using a finite-difference simulator, incorporating data from exploration wells, onshore and nearshore exploration and production wells, likely well designs, known or estimated properties of potential reservoir oils, and geophysical data from seismic surveys (presentation by Kirk Sherwood, BOEM, March 2013). VLOSs represent extreme (improbable, but geologically possible) scenarios that are intended to support assessments with very high environmental impacts. The models estimate daily discharge rates but also report the cumulative amount of oil that could be discharged over the estimated period of time required to drill a relief well (presentation by Kirk Sherwood, BOEM, March 2013). The results of two VLOS models are presented in Table 3.3. In practice, a successful well-capping operation could halt or significantly slow the flow of oil into the marine environment earlier than the time needed to drill a relief well.
Many types of ships have recently been utilizing the Arctic marine environment, including government vessels and icebreakers, container ships, general cargo ships, bulk carriers, tanker ships, passenger ships, tugs and barges, fishing vessels, and vessels related to oil and gas exploration (Arctic Council, 2009). A record of transits through the Northern Sea Route in 201310 indicates that some tanker ships carried over 800,000 bbl of oil as cargo, although smaller ships carried as little as 35,000 bbl of diesel fuel cargo. These numbers illustrate the broad range in volume of potential spills from cargo ships, which does not include the fuel oil they carry aboard. In the U.S. Arctic, doubled-hulled barges that provide fuel resupply for Alaskan villages can carry over 6,000 bbl of oil cargo.11
The villages store oil, diesel, and gasoline supplies for home and business heating, aviation fuel, and industrial needs for mining and oil and gas production. Because there are long periods between resupply due to sea and river ice, significant volumes of fuel may be stored in relatively close proximity
_____________
10 See http://www.arctic-lio.com/docs/nsr/transits/Transits_2013_final.pdf.
11 See http://www.marinelink.com/news/ecofriendly-operate335711.aspx.
to the shoreline. Examples include large storage facilities at the Red Dog Mine’s Delong Mountain Terminal and tank farms in the community of Barrow.
ARCTIC OIL SPILL COUNTERMEASURES
Arctic response strategies can leverage the natural behavior of oil in, on, and under ice. For instance, ice can bar the spread of oil, reducing spreading rates and leading to smaller contaminated areas (Sørstrøm et al., 1994, 2010; Potter et al., 2012); due to encapsulation or a lack of weathering, oil remains fresher for a longer time; and ice-covered areas generally have less severe wind and sea conditions. Despite the documented effects of climate change leading to later freeze-ups, greater extent of northerly ice edge retreat, and longer summer open water seasons, the Chukchi and Beaufort Sea coastlines are still buffered from oil spilled offshore by a fringe of fast ice for eight to nine months of the year. However, Arctic conditions impose many challenges for oil spill response—low temperatures and extended periods of darkness in the winter, oil that is encapsulated under ice or trapped in ridges and leads, oil spreading due to sea ice drift and surface currents, reduced effectiveness of conventional containment and recovery systems in measurable ice concentrations, and issues of life and safety of responders.
The following sections review the state of knowledge and recent advances regarding key response countermeasures and tools for oil removal under Arctic conditions: biodegradation (including dispersants), ISB, mechanical containment and recovery, detection and tracking, and oil spill trajectory modeling. It should be noted that the highlighted countermeasures are in addition to the “no response” option of natural recovery, a viable response option for some situations.
Although a number of new research developments are discussed in the following sections, this is not a comprehensive account of all ongoing efforts in the field of oil spill response. An extensive list of recent Arctic oil spill research can be found in Oil Spills in Arctic Waters (USARC, 2012). It summarizes not only federal efforts but also research and development efforts by non-profit organizations such as the Oil Spill Recovery Institute, industry such as Alaska Clean Seas and the Joint Industry Programs, and international efforts in Canada and Norway.
BIODEGRADATION AND DISPERSANT USE
Biodegradation by naturally occurring microbial communities is a major process contributing to the eventual removal of oil that enters the marine environment (Leahy and Colwell, 1990; Atlas and Bartha, 1992; Atlas, 1995; Brakstad and Bonaunet, 2006). While their numbers may be low in pristine environments, numerous scientific studies have shown that microbes have the ability to rapidly multiply following exposure to oil (Atlas, 1995). Biodegradation of petroleum hydrocarbons has been linked to a diverse community of microorganisms that have large surface-to-volume ratios and the ability to respond rapidly to changes to environmental conditions (ZoBell, 1973; Atlas, 1984; Leahy and Colwell, 1990).
Microbial responses to oil in marine environments are generally dominated by bacteria rather than archaea (Röling et al., 2004). Although fungi are known to degrade petroleum compounds
in some marine settings (Zinjarde and Pant, 2002) and their existence has been reported in high-latitude marine environments (Butinar et al., 2011), there have been no studies to evaluate their potential to degrade petroleum hydrocarbons in Arctic marine waters. Populations of indigenous oil-degrading bacteria are likely to be established in Arctic waters due to the presence of natural seeps (Landes, 1973).
The potential of indigenous microbes to degrade petroleum hydrocarbons in marine waters at low temperatures has been demonstrated in field and laboratory studies (Sveum and Ladousse, 1989; Bragg et al., 1994; Prince et al., 2003a; Brakstad et al., 2008; Lee et al., 2009b). Indigenous bacteria in Arctic waters have demonstrated the capacity to degrade petroleum hydrocarbons even at near-freezing temperatures (Brakstad and Bonaunet, 2006). Although the rates of oil biodegradation were lower than those observed at high temperatures (Margesin et al., 2003; Michaud et al., 2004), substantial levels of oil biodegradation by a consortium of bacteria including Pseudoalteromonas, Pseudomonas, Shewanella, Marinobacter, Psychrobacter, and Agreia were observed in nutrient-enriched Arctic seawater (Deppe et al., 2005; Brakstad and Bonaunet, 2006). However, it is important to note that Arctic surface waters are generally nutrient-depleted in summer. The presence of oil degraders does not appear to be a limiting factor in Arctic waters. Despite low population levels, the resident organisms are capable of rapidly responding to changes in environment and energy source. Newer studies suggest that indigenous bacteria in Arctic waters degrade oil faster than previously thought (Stapleton and Sayler, 2000; Whyte et al., 2002; Heiss-Blanquet et al., 2005; Prince et al., 2013). Recent studies with fresh Chukchi Sea water incubated at −1°C gave a biodegradation half-life of 60 days for Alaskan North Slope crude oil (McFarlin et al., 2014). Field trials on pristine Arctic and Antarctic beaches also reveal the presence of indigenous hydrocarbon-degrading bacteria (Grossman et al., 1999; Delille and Delille, 2000; Powell et al., 2005). The loss of oil from sediments at these sites was attributed to several processes, including physical removal, photo-oxidation, and biodegradation.
While microbial metabolism and motility have been measured in brine channels (Junge et al., 2002, 2003, 2004, 2006; Breezee et al., 2004; Faksness et al., 2011; Mykytczuk et al., 2013), biodegradation of oil in marine ice has not been fully investigated. During a preliminary winter field study with crude oil frozen into fjord ice in Svalbard, the bulk oil stimulated the growth of bacterial biomass and oil-degrading bacteria specifically (Brakstad et al., 2008). Another field study in Svalbard showed no significant degradation of oil in ice at subzero temperatures, but at 0°C, melt pool oil samples that were fertilized with inorganic nutrients had a substantial change in bacterial diversity (Gerdes and Dieckmann, 2005). The combination of low temperature and high salt content in the brine channels requires that microbes be both salt (halo-) and cold (psychro-) tolerant. A limiting factor for sealed brine pockets would be availability of oxygen, which would eventually halt biodegradation. Extremely halophilic or halotolerant microbes able to degrade oil have been reported (Diaz, 2008; Al-Mailem et al., 2010), but not yet in cold environments.
Influence of Environmental Factors
Many research programs have studied the influence of various environmental factors on biodegradation rates of oil spilled in Arctic waters. The effect of temperature on degradation rate in nature
is less than one might expect, since naturally occurring bacteria are adapted to the temperature in which they develop (Garrett et al., 2003; Macnaughton et al., 2003; Michaud et al., 2004; Brakstad and Bonaunet, 2006; Venosa and Holder, 2007). Psychrophilic bacteria, for example, may have metabolic rates comparable to those of organisms adapted to moderate temperatures. Recent studies using genomic, genetic, and physiological methods have shown that psychroactive bacteria have a number of molecular adaptations that facilitate their growth at subzero temperatures, including some down to −10°C (Bakermans et al., 2012).
A study using Antarctic water showed that the degree of degradation after 50 days of incubation differed only slightly between three tested temperatures (4°C, 10°C, and 20°C) (Delille et al., 2009). Nutrient (nitrogen and phosphorus) availability was identified in this study as the factor limiting oil biodegradation. In a study with low concentrations (2.5 mg/L; 2.5 parts per million [ppm]) of Alaska North Slope oil in Atlantic seawater, to counter the potential effects of nutrient depletion, it was noted that 80% of the saturates and 2- to 4-ring aromatics were biodegraded after 60 days at 8°C (Prince et al., 2013). There are, however, a number of confounding factors—for example, availability and type of bacteria, oil composition, and nutrient availability.
While the majority of marine bacteria thrive in a salinity range of 25-35 parts per thousand (ZoBell, 1973), species living in transition environments are adapted to salinity fluctuations. Diaz (2008) reported the isolation of a bacterial consortium from a North Sea crude oil sample that was capable of metabolizing hydrocarbons in salinities from 0 to 220 parts per thousand, although greater degradation occurred at lower salinities.
The availability of oxygen is important for more rapid removal of petroleum hydrocarbons (Leahy and Colwell, 1990). While a constant supply of oxygen is maintained at the sea surface by wind and waves, oxygen availability may be a limiting factor in subsurface sediments and within the water column. However, there is evidence of biodegradation of some petroleum hydrocarbons coupled with sulfate reduction in marine anoxic sediments (Lovley et al., 1997).
The composition of oil and its physical properties also influence its potential to be degraded by microbes. Saturates have been shown to have the highest rates of biodegradation, followed by light aromatics, while high-molecular-weight aromatics and polar compounds exhibit lower biodegradation rates (Prince, 2010). The same order is expected in cold Arctic waters. Changes to oil properties as a result of weathering have received considerable attention, as they influence the behavior of the oil and its biodegradation potential, and may render remaining components to be essentially nontoxic.
Compounds of low molecular weight in an oil slick are subject to two competing processes: evaporation and dissolution. In general, evaporation is slower, but still significant, in cold seawater compared to temperate seawater. Evaporation of the lower-molecular-weight components also results in increased viscosity of the residual oil (Faksness and Brandvik, 2008), which could negatively affect the ability of oil to disperse into very small droplets, thereby reducing biodegradation. Compared to evaporation, components dissolved from the oil phase are immediately available for microbes to degrade in the water column. In cold seawater, the dissolution of oil compounds is decreased compared to temperate water (Faksness and Brandvik, 2008). This decrease in solubility has been used as an explanation for the observed recalcitrance of hydrophobic compounds in cold water and Arctic conditions. The nonpolar compounds of the water-soluble fraction of crude oils are generally considered to be rapidly biodegraded in the marine environment (Brakstad and Faksness, 2000)
relative to other more persistent components such as those found in that grouping of components known as the “unresolved complex mixture” (Meredith et al., 2000; Han et al., 2008).
Garrett et al. (1998) demonstrated that photo-oxidation can create molecules with longer carbon chains (tending to contribute to stabilization of emulsions), while biodegradation tends to break down hydrocarbons into smaller, simpler units. Studies have also shown that the photo-oxidized compounds may have increased potential for biodegradation (Ni’matuzahroh et al., 1999; Dutta and Harayama, 2000; Maki et al., 2001).
Emulsification potentially reduces the biodegradation by lowering nutrient availability to oil-degrading bacteria (Brakstad et al., 2011; Cook et al., 2011). Furthermore, rapid emulsion formation could diminish the rates of evaporation and dissolution, causing retention of more toxic low-molecular-weight aromatic compounds within the residual oil. With sufficient mixing energy, oil associated with surface slicks breaks up into droplets that can become entrained, dissipated, and diluted in the water column (Delvigne and Sweeney, 1988; Fraser and Wicks, 1995; Lee et al., 2001). This natural dispersion process enhances oil biodegradation rates because droplet formation increases the surface area of the oil available for microbial attack and increases nutrient availability (Prince, 1993).
Oil spill trajectory models currently used to predict the fate of oil spills have not been calibrated for the full range of environmental factors encountered in the Arctic. At present, there are limitations in the usefulness of numerical models to predict oil biodegradation in cold marine environments. Detailed temperature-related biodegradation studies are needed to improve fate models, which often rely on inadequate datasets for cold climate spills.
Biodegradation and associated microbial studies from the Deepwater Horizon spill are instructive in that they show rapid growth of petroleum-degrading microbes as the oil disappeared, under in situ conditions of 4°C, nutrients, and oxygen. Oceanospirillales (a bacterial order) was dominant in the oil plume at one month (Hazen et al., 2010). By the second month, Cycloclasticus and Colwellia genera dominated, with Colwellia also dominant in flocs of oil and organic matter (Baelum et al., 2012). Colwellia strains isolated from the samples grew rapidly on Macondo oil at 4°C, confirming their capability to degrade oil components. When uncontaminated deep water from the Gulf of Mexico was incubated with Macondo oil, 25% of it was degraded at 4°C within 20 days; 60% was degraded when the dispersant Corexit was added. There was an associated enrichment of Oceanospirillales and Colwellia, showing the response of a very small population of indigenous biodegrading bacteria to the new petroleum hydrocarbon food source.
While much of the oil from the Deepwater Horizon spill was either recovered or degraded, some would have settled to the sediments. Sediments in the Deepwater Horizon spill region show an enrichment of Deltaproteobacteria, which are known to degrade hydrocarbons anaerobically, and metabolic signatures of anaerobic petroleum degradation (Kimes et al., 2013). The authors hypothesized that these organisms may degrade some of the more persistent oil components in the water column, though this remains to be tested. While the Gulf of Mexico has different characteristics than the Arctic Ocean, its cold deep waters and sediments may have similar bacterial populations. Furthermore, both have natural oil seeps that allow long-term selection for oil-degrading bacteria adapted to regional conditions.
A major oil spill in the Arctic could lead to a microbial community response similar to that ob-
served after the Deepwater Horizon spill, but it is not known how changes in microbial communities would impact the food web, especially the significant benthic communities.
Bioremediation
Research has been focused on the development of Arctic oil spill bioremediation strategies that accelerate the natural biodegradation process. Considering the logistical needs in mounting an Arctic spill response, bioremediation may be a very effective countermeasure as it does not require contaminated waste transport and disposal, large numbers of personnel to apply, or specialized equipment. In addition, it is considered by many to be a more environmentally friendly technology than some other responses. Most attempts have used biostimulation to overcome nutrient and oxygen limitation and to promote oil dispersion, increasing growth conditions for oil-degrading bacteria (Lee et al., 1993, 1997; Prince, 1993, 2005; Swannell et al., 1996; Lee and de Mora, 1999; Prince and Clark, 2004; Al-Darbi et al., 2005; Prince and Atlas, 2005).
Marine waters often contain low concentrations of nutrients (most significantly, nitrogen and phosphorus) that limit the hydrocarbon biodegradation (Lee et al., 1993; Prince, 1993; Lee and de Mora, 1999). Biostimulation activities to increase natural biodegradation in the marine environment have focused on applying fertilizers to overcome nutrient limitation (Prince, 1993; Bragg et al., 1994; Swannell et al., 1996; Venosa et al., 2002; Prince et al., 2003a). Its effectiveness was shown during cleanup operations following the Exxon Valdez oil spill, where the addition of fertilizer was shown to increase biodegradation rates by three to five times (Bragg et al., 1994; Atlas, 1995).
Application protocols need to be tailored to local conditions to account for differences in environmental parameters and oil characteristics at specific contaminated sites (Wrenn et al., 1994, 1997a,b, 2006; Venosa et al., 1996; Boufadel et al., 1999; Lee and de Mora, 1999). Some nutrient additions may inhibit microbial activity (Braddock et al., 1997), while excess nutrients can cause detrimental effects such as eutrophication. Certain microbial populations may require different nutrient ratios for optimal degradation of different hydrocarbons (Smith et al., 1998). A study that showed enhanced oil degradation in samples of ice-covered seawater also raised the prospect of treating oil within brine channels of sea ice (Delille et al., 1998).
Bioremediation efforts have been mainly carried out on shorelines and in wetlands, due to the difficulty of maintaining elevated nutrient concentrations in an open water system (Leahy and Colwell, 1990). Biostimulation trials conducted on Arctic beaches of Spitzbergen Island showed that application of a commercial nutrient formulation increased oil biodegradation in coarse but not fine sediments (Sveum and Ladousse, 1989). A later program expanded the scope of this study by testing the effectiveness of several other remediation methods including bioremediation, physical mixing, and surf washing (Lee et al., 1998a; Guénette et al., 2003; Sergy et al., 2003) to enhance the biodegradation of an intermediate fuel oil on sand and pebble shorelines. Over one year, biodegradation rates were approximately doubled in the oiled sediments that were treated with fertilizers, with no acute toxicity measured (Lee et al., 2003b; Prince et al., 2003a). By increasing sediment permeability, physical mixing also appeared to increase microbial activity (Owens et al., 2003). Commercial
and experimental nutrient mixtures also enhanced degradation of an Arabian crude oil stranded on intertidal sandy beaches on the Kerguelen Archipelago in the Antarctic (Pelletier et al., 2004).
Chemical Dispersants and Oil-Mineral Aggregate Formation The amount of oil that naturally disperses into the water column after a spill is dependent on the kind of oil spilled, ambient temperature, and the mixing energy and/or release conditions. For example, most if not all of the oil spilled in the midst of a gale with high sea states may be naturally dispersed, as in the 1993 Braer tanker incident (Spaulding et al., 1994). When the mixing energy that is provided by waves and wind overcomes surface tension at the oil-water interface, natural dispersion will occur and will break the oil film into variable-size droplets (Potter et al., 2012). Larger oil droplets resurface and coalesce, while smaller droplets stay suspended in the water column, where they will be diluted through turbulence and subsurface currents. These smaller droplets will eventually be subjected to processes of biodegradation.
Chemical dispersants are designed to reduce surface tension at the oil-water interface, which would allow waves to more easily break up the oil into small droplets (generally less than 100 microns) that are rapidly diluted in the water column, down to concentrations where natural nutrient levels are not rate limiting for microbial degradation of the residual hydrocarbons (Swannell and Daniel, 1999; Prince et al., 2013). Dispersant guidelines for the State of Alaska are contained in Annex F of the Unified Plan (ARRT, 2008). Alaska has no pre-approved dispersant use areas.
The biodegradation of chemical oil spill dispersants and the surfactants used in them has been studied for many years (Liu, 1983; Una and Garcia, 1983; Odokuma and Okpokwasili, 1992; Baumann et al., 1999; Lindstrom and Braddock, 2002; Venosa and Holder, 2007; Garcia et al., 2009). Varadaraj et al. (1995) noted that dispersants themselves can enhance the initial rate of hydrocarbon degradation, as oil-degrading bacteria can grow on the dispersant and then colonize dispersed oil droplets. This activity can cause a delay in oil biodegradation because the bacteria may preferentially utilize the dispersant first (Foght and Westlake, 1982; Bunch et al., 1983; Foght et al., 1983).
Dispersion will, in many cases, rapidly dilute oil concentrations below toxicity threshold limits and will increase the oil-water interface for greater microbial interaction and degradation. While primarily used on surface oil spills, in the Deepwater Horizon oil spill dispersant was applied via subsurface injection to the plume of oil released from the wellhead into 4oC waters at 1,500 m depth (Atlas and Hazen, 2011; Kujawinski et al., 2011). There has been considerable debate over the effectiveness of chemical dispersants on crude oil degradation at low seawater temperatures. The main concern is that as temperature decreases, chemical processes slow down and oil viscosity increases, making it more difficult to disperse. Generally, dispersants are effective on nonemulsified oil at freezing temperatures as long as viscosity does not increase significantly. Experiments to test the effectiveness of eight dispersants on South Louisiana crude oil (analogous to that released during Deepwater Horizon) at 5°C and 25°C revealed that temperature was less critical than expected (Venosa and Holder, 2013). To overcome the viscosity limits of conventional chemical dispersants in cold environments, recent research has focused on higher-viscosity products that increase contact time with the spilled oil (Nedwed et al., 2011) and those with higher concentrations of active ingredients. Wave-basin tests indicate that these products might be used to treat conventional oils with dispersant-to-oil ratios as low as 1:100 (compared to the currently recommended 1:20) and to disperse high-viscosity oils such as heavy crude and fuel oils (Nedwed, 2010).
Recently, the SINTEF Oil in Ice JIP (Daling et al., 2010; Sørstrøm et al., 2010) evaluated the effectiveness of dispersants under Arctic conditions, including cold air and water temperature, ice, and melting ice and river outflows. The presence of ice can increase the length of time that a dispersant is effective by slowing the rate of oil weathering and emulsification. Although wind-wave action that facilitates dispersion in open water is generally dampened by the presence of ice, the interaction of individual ice floes in response to winds and currents can lead to localized upper water column turbulence for more effective dispersion than would otherwise be possible without the presence of ice under similar wind conditions. Mechanical mixing may be needed to overcome the lack of turbulent mixing energy in scenarios involving significant ice cover—for example, vessel propellers or thrusters can provide artificial mixing energy while adding chemical dispersants to oil, an effect documented in tank tests and at sea (Nedwed et al., 2007; Daling et al., 2010). There are limitations to the surface application of dispersants, as vessels are able to cover only a limited amount of slick in a given time, while aircraft provide high coverage rates but cannot optimize dose rate to slick thickness.
There still remains some controversy over the use of chemical oil dispersants, due to concerns over their potential to induce toxic effects. A 2013 National Research Council (NRC) report summarized a number of recent studies on the toxicity of dispersants and dispersed oil on plankton, fish, and shrimp and determined that “there is some evidence that chemically dispersed oil and some dispersant compounds are toxic to some marine life, especially those in early life stages” (NRC, 2013). Contradictory evidence on the toxicity of chemically dispersed versus physically dispersed oil on marine organisms (NRC, 2013) exists as a result of differences in experimental conditions and methodology and the fact that actual exposure concentrations were not fully quantified in a large fraction of previous toxicity studies (NRC, 2005).
There are understandable concerns about the toxicity of the dispersant itself. However, in U.S. waters, only EPA pre-approved products that pass standardized product efficacy and toxicity tests are considered for use. Key factors determining toxicity for a given species are concentration and length of exposure time. Studies have shown that chemical dispersants can increase the exposure of oil to pelagic marine organisms. For example, Couillard et al. (2005) reported that the presence of dispersant caused a two- to fivefold increase in the concentration of total and high-molecular-weight polycyclic aromatic hydrocarbons (PAHs). However, while chemically dispersed oil may be more biologically available, dispersants themselves are rapidly diluted in the open ocean to less than 1 mg/L (1 ppm) within an hour, well below defined toxicity threshold limits (NRC, 1989; Lee et al., 2013).
Chemical dispersants do more than facilitate the transport of oil from surface oil slicks to the water column. Prince et al. (2013) recently suggested that biodegradation would be rapid and extensive when oil is present at concentrations expected with dispersant use. McFarlin et al. (2014) confirmed that Arctic microorganisms indigenous to Chukchi Sea water were capable of performing extensive biodegradation of chemically dispersed oil at an environmentally relevant temperature (−1°C) without nutrient additions. Indigenous microorganisms degraded both fresh and weathered oil at environmentally relevant concentrations, in both the presence and absence of dispersant, with oil losses ranging from 46% to 61% and up to 11% mineralization over 60 days without nutrient additions. When tested alone, 14% of 50 ppm of dispersant was mineralized within 60 days. Arctic species and their temperate region counterparts have been shown to have similar tolerance to dispersed oil, and the use of dispersant did not increase the toxicity of the oil (Gardiner et al., 2013).
There are at least two challenges to performing representative dispersed oil biodegradation studies in small-scale test systems (Lee et al., 2013). The first is how to conduct tests at the low dispersed oil concentrations that accurately represent field conditions. Many previous biodegradation studies were conducted in closed systems at unrealistically high concentrations (NRC, 2005). The second is how to maintain a stable dispersion in the laboratory. In order to reflect dispersion of oil in ocean conditions, biodegradation studies need formation of dispersed droplets of 70-100 microns and enough mixing energy to keep those droplets from resurfacing. This can be challenging in a closed system, especially during multiweek test periods, and demonstrates a weakness of many bench tests employed to study dispersant effectiveness and toxicity. They may accurately determine relative performance of different dispersants but are difficult to translate to real-world situations.
Formation of Oil-Mineral Aggregates In the marine environment, oil particles may not remain as discrete particles; they coalesce and rise back to the surface, or they can interact with suspended organic and/or inorganic particulate matter in the water column (Lee et al., 1985; Muschenheim and Lee, 2003; Owens and Lee, 2003) to form aggregate “flocs,” which include oil-mineral aggregates (OMAs). The formation of OMAs stabilizes the oil-water interface, with the suspended particulate matter acting as a surfactant. This favors droplet formation and enhances oil dispersion into the water column. The process of stabilizing oil droplets with fine clay particles has led to natural and proactive oil spill remediation strategies for shorelines (Bragg and Yang, 1995; Lunel et al., 1996; Owens, 1999; Lee et al., 2003a). Breaking waves on the beach provide sufficient mixing energy to form OMAs from fine sediments and spilled oil (Lee et al., 2003a). The OMAs transport oil away from the shore, simultaneously providing a microcosm for rapid bacterial biodegradation (Lee et al., 1998b; Stoffyn-Egli and Lee, 2002). OMA formation enhances the natural dispersion of oil and reduces its environmental persistence (Bragg and Yang, 1995; Lee et al., 2003a; Owens and Lee, 2003; Owens et al., 2003). Since OMA formation results from the stabilization of oil droplets by mineral fines, a synergistic effect may be achieved with the addition of dispersants (Li et al., 2007).
Laboratory studies have shown that OMAs can quickly form at near-freezing temperatures in seawater if high-energy mixing is applied (Cloutier et al., 2005; Khelifa et al., 2005). These results were confirmed in a mesoscale basin containing brackish water with slush and broken ice, in which 20-30 minutes of mixing dispersed about 50% of the spilled oil (Blouin and Lee, 2007; Cloutier and Doyon, 2008). These trials were reproduced at full scale in January 2008 in the St. Lawrence River, when 200 liters of fuel oil were mixed with chalk fines by an icebreaker propeller (Lee et al., 2009a, 2011a). After dispersion into the water column, the oil did not resurface. A control test with no added particles produced significant resurfacing oil. Water samples taken back to the laboratory revealed that OMAs were formed and that more than half of the total petroleum hydrocarbons had degraded after 56 days of incubation at 0.5°C (Lee et al., 2012).
Promising New Response Concepts for Dispersion and Biodegradation
The Deepwater Horizon response demonstrated that large-scale subsea dispersant injection may be an effective approach for wellhead blowout spill mitigation. A major benefit of direct sub-
sea dispersant injection is the ability to continuously respond independent of darkness, extreme temperatures, strong winds, rough seas, or the presence of ice. The dispersant volume could be substantially less than a surface application because of its ability to deliver dispersant very close to the oil release source, a key advantage with respect to enhancing oil encounter rate (how much oil a particular countermeasure can intercept or treat in a given time). This could be useful given the long and difficult logistics resupply chain in most Arctic areas, and, since less oil will come to the surface, subsea injection could lower volatile organic compounds encountered by responders at the sea surface. A comparison of response effectiveness showed that direct injection can disperse oil at rates significantly higher than those achievable by aerial dispersant application or other response methods (Federal Interagency Solutions Group, 2010). Another advantage of direct injection is higher efficiency, because the dispersant rapidly (within seconds) mixes with oil in a highly turbulent state at the discharge point. Because of the extremely rapid mixing under turbulent conditions, subsea injection is a potentially viable response even for wells in relatively shallow water, such as the Chukchi Sea. Based on research performed in a variety of mixing regimes (e.g., NRC, 2005; SL Ross Environmental Research Ltd. and MAR Inc., 2007, 2008; Reed et al., 2009), it is expected that a significant percentage of oil discharged from the Macondo well was converted to droplet sizes below 100 microns. Results from scaled-down laboratory experiments in vertical test tanks have demonstrated the potential generation of small oil droplets following dispersant additions to a subsurface discharge of oil at its point of release (Brandvik et al., 2013; Johansen et al., 2013). Still, more work needs to be done to understand the effectiveness, systems design, and short- and long-term impacts of subsea dispersant delivery. Rather than the ad hoc emergency injection system using remotely operated underwater vehicles and wands (as in the Deepwater Horizon response), future systems could involve pre-engineered injection lines directly into the well bore.
Mullin (2012) summarized a number of dispersant research studies undertaken by the current Arctic Response Technology JIP, while API and its industry companies have established a large-scale, multiple-year Subsea Dispersant Program, whose goal is to conduct controlled experiments on the effectiveness of subsea injection over various conditions, the effects of dispersed oil on deepwater marine environments, and numerical modeling needs for better prediction of oil fates.12 This program recently released an initial report summarizing the status of dispersant regulatory approval and conditions on the application of dispersants in different Arctic nations (SEA Consulting Group, 2013).
Concerns over the resurfacing of oil dispersed under ice are also being addressed by the Arctic Response Technology JIP (Mullin, 2012). Scientists in Norway, the United States, and Canada are assessing whether turbulence levels in the water column of the Arctic Ocean are sufficient to keep oil suspended for a sufficient time for effective biodegradation to occur. Results are expected for public release and publication in peer-reviewed journals in late 2014.
For dispersant application in ice-covered waters, newer gel formulations that are more effective due to reduced solvent concentrations are currently being developed by industry. The gel formulation could increase the window of usability by being able to treat a wider range of crude oils and by increasing the amount of oil that may be treated by a given volume of dispersant. Future possibilities include spraying gel on oil that rises to the surface of the ice in the spring rather than igniting it,
_____________
12 See www.api.org.
or adding gel dispersant to oil discharged from a surface blowout onto an ice cover (Nedwed et al., 2011).
Another recent effort involves the development of a jet aircraft–based dispersant delivery system that extends beyond the current use of Lockheed Hercules C-130As. This system was created in association with Oil Spill Response Limited at the recommendation of the International Association of Oil & Gas Producers’ (OGP’s) Global Industry Response Group. The Boeing 727 was selected because of its high transit speed, large payload, and extended range, which offers the possibility of effective response to spills in remote settings where other equipment may be less readily deployed (OGP, 2011; OGP and IPEICA, 2012). Aircraft are currently being configured for dispersant operation and will be put through an extensive number of tests to verify their performance.
Future approaches to enhance oil biodegradation include the application of nutrient-bearing treatment products (Kjeilen-Eilertsen et al., 2011) and the application of surfactants to surf-washing operations to increase the production of oil droplets and promote OMA formation. Another approach is bioaugmentation, where microbes with high biodegradation potentials are used to supplement existing microbial populations at contaminated sites (Bartha, 1986). This has often been proposed for use in situations where the indigenous microbial population cannot degrade petroleum, is stressed from recent exposure to an oil spill, has too small a population to maintain high biodegradation rates, or where seeding could reduce the lag period in growth prior to the onset of bioremediation (Forsyth et al., 1995). However, bioremediation field trials in open water environments have shown that bioaugmentation provides little or no benefit to treatment of spilled oil (K. Lee et al., 2005; Nichols and Venosa, 2008) and would not be expected to do so where natural seeps would have already enriched a larger number of adapted, oil-degrading microbes.
IN SITU BURNING
Controlled ISB of an oil slick as a response technology has been utilized for many years. While the first recorded use of ISB as a response countermeasure technique was in 1958 during a pipeline spill in the Mackenzie River, Northwest Territories (McLeod and McLeod, 1972), some important early work was carried out by the USCG in Alaska in the 1970s (McMinn, 1972). ISB is especially suited for use in the Arctic, where ice can often provide a natural barrier to maintain the necessary oil thicknesses for ignition, without the need for booms. A number of large-scale experiments successfully used ISB on oil that surfaced in spring melt pools after being spilled beneath the ice and trapped through a full winter. These experiments were carried out in the Canadian Beaufort Sea in 1975, 1980, and 1981, and in Svalbard in 2006 (NORCOR Engineering & Research Ltd., 1975; Dickins and Buist, 1981; Brandvik and Faksness, 2009). Overall removal rates ranged from 65% to greater than 90%, depending mainly on the size distribution of the melt pools. High efficiencies were documented for ISB of oil within fire-resistant booms in both Arctic ice-covered and southern open water environments (Potter et al., 2012).
ISB was used successfully on a trial basis during the Exxon Valdez response (Allen, 1990). In 1993, a U.S.-Canada experiment off Newfoundland burned crude oil in fire-resistant booms in the open ocean and monitored a large suite of environmental parameters including smoke composition,
residue toxicity, and upper-water-column impacts (Fingas et al., 1995). The most recent Arctic experiences with the use of ISB offshore were through the Oil in Ice JIP (Sørstrøm et al., 2010), in which oil that was allowed to drift and weather in very close pack ice for over a week was successfully ignited and burned (Brandvik et al., 2010). Most recently, the large-scale ISB operation in response to the Deepwater Horizon blowout resulted in a unique set of full-scale operational data applicable to response planning for Arctic offshore areas in the summer. A total of 411 controlled burns removed an estimated 220,000 to 310,000 barrels of oil from the Gulf of Mexico (Allen et al., 2011).
Experience with burning fresh, weathered, and emulsified oils and petroleum products in a range of ice conditions has led to some basic “rules of thumb” (Buist et al., 2003a). The most important parameter that determines the likelihood of success and expected removal efficiency is the oil thickness, since below a minimum thickness of about 3 mm the burn will self-extinguish due to heat loss to the water. In order to achieve 60%-80% removal efficiency in most situations, the starting thickness of crude oil needs to be on the order of 3-5 mm. With relatively fresh oil that is wind herded against an ice edge or on melt pools in the spring, removal efficiencies in excess of 90% are achievable. Another key operational constraint is wind speed; a maximum for successful ignition is about 10 m/s or 20 kn, based on experience gained in field testing (Buist et al., 2003b). Concentrated pack ice can enable ISB by keeping oil slicks at the appropriate thickness (Buist and Dickins, 1987). In very open drift ice conditions, oil can spread rapidly and become too thin for ignition (Potter et al., 2012). Fire booms can collect and keep slicks thick in open water; however, even light ice conditions make the use of booms challenging (Bronson et al., 2002). In spite of these challenges, Potter and Buist (2010) reported highly effective (~90%) burning of oil within small ice pieces and brash collected within a fire-resistant boom during 2009 field experiments in the Norwegian Barents Sea. Ice concentrations in these tests were between 1/10 and 3/10, with small boats used to corral the needed quantities of ice. Conventional booming techniques are most effective in ice concentrations up to 1/10, with some effectiveness in concentrations of 2/10-3/10 (Potter et al., 2012). Despite these test results, there is continued concern among some non-governmental organizations that actual spill conditions could reduce the effectiveness of ISB to far below the theoretical maximum (e.g., WWF, 2010; Goodyear and Beach, 2012). In practice, experiences with very large burns at sea have demonstrated that efficiencies increase with scale, as the oil is pulled into the burn area by strong radial inflow winds at the surface (Buist et al., 1994). This was shown in observations during the Deepwater Horizon response, where free-floating, organized burning slicks could be seen following the oil corralled within fire booms.
In 2004, a multiyear joint industry and government (Minerals Management Service, now managed by BSEE) project began to study oil-herding chemicals to thicken slicks for ISB, as an alternative to booms in open drift ice conditions. Small-scale laboratory experiments were followed by midscale testing in large basins (e.g., Buist et al., 2007). The cold-water herder effectively thickened oil slicks in ice concentrations of up to 70%. Herded slicks in excess of 3 mm thickness were routinely achieved, and were ignited and burned at air temperatures as low as −17°C. The burn efficiencies that were measured for the herded slicks were only slightly lower than theoretical maximums achievable for slicks that are equivalently sized and physically contained on open water (Buist et al., 2011). The concept of using chemical herders to burn free-drifting oil slicks in pack ice was successfully field tested for the first time in the Norwegian Barents Sea in 2008 as part of a JIP on Oil Spill
Contingency for Arctic and Ice-Covered Waters (Buist et al., 2010). Burn removal effectiveness in that test was estimated to be on the order of 90%. The residue floated readily and was recovered manually from the water surface and ice edges. Buist et al. (2011) summarized past research into chemical herders and concluded that oil spill responders should consider utilizing them to enhance ISB in light to medium ice concentrations.
In the 1990s, research efforts assessed the potential environmental impacts of ISB, primarily from smoke plume and burn residues (Fingas et al., 1995). The smoke plume emitted by burning an oil slick on water is often the primary ISB concern to the public and regulators, as low concentrations of smoke particles at ground or sea level can persist for a few kilometers downwind. In practice, smoke particulates and gases are rather quickly diluted to concentrations below levels of concern (Fingas et al., 2001). Work by Canadian and U.S. teams advanced the understanding of smoke constituents and how to predict downwind environmental impacts (McGrattan et al., 1995). This work included a series of medium-scale burns at fire test facilities in Alabama, as well as a highly documented large-scale burn at sea off the Canadian East Coast in 1993—the Newfoundland Oil Burn Experiment (Fingas et al., 1995).
Burn residue—the unburned oil that remains on the surface of the water after a fire extinguishes naturally—was also studied in the 1990s. Daykin et al. (1994) and Blenkinsopp et al. (1997) studied burn residue’s potential for aquatic toxicity, while an industry-funded research program examined the likelihood of burn residue sinking as it cooled (Buist et al., 1995; SL Ross Environmental Research Lt., 1998). Bioassays showed very little or no acute toxicity to oceanic organisms for either weathered oil or burn residue. These findings of little or no impact were validated with further studies by Gulec and Holdway (1999) and Gannon and Holdway (1999). Another area of historical research was to study the overall mass balance of PAHs consumed and created by ISB. During the Newfoundland Oil Burning Experiment, PAH concentrations were much lower in the plume and in particulate precipitation at ground level than they were in the initial oil composition, suggesting that PAHs are largely consumed by combustion (Fingas et al., 2001).
Numerous agencies, primarily in the United States, have established guidelines for the safe implementation of ISB as a countermeasure. The U.S. National Institute of Standards and Technology, NOAA, and Environment Canada have computer models used to predict safe distances for downwind smoke concentrations. In 1994, the Alaska Regional Response Team incorporated ISB guidelines for Alaska into its Unified Response Plan, becoming the first Arctic area to formally consider ISB as an oil spill countermeasure (ARRT, 2008). Their guidelines are considered the most fully developed to date. The American Society of Testing and Materials began developing standards associated with ISB in the late 1990s (ASTM, 2009), while the USCG produced an operations manual that details considerations and steps to be taken for open water ISB with fire booms (Buist et al., 2003a). New fire-resistant and fireproof boom designs were developed after a successful test burn during the Exxon Valdez spill (Allen, 1990, 1999). Several different types of fire booms were tested during the Deepwater Horizon oil spill, with some notable differences in their effectiveness for oil retention and durability in the face of fire intensity and sea state (Mabile, 2010).
All blowouts involve a mixture of oil and gas, mostly methane. While the casing of any wells drilled in the Chukchi or Beaufort OCS should extend below the shallow permafrost zones that could contain gas hydrates, ISB response strategies generally involve collecting and burning oil a
safe distance away from the discharge site. This ensures that sea surface gas concentrations are well below the limit that could lead to spontaneous ignition and combustion.
Promising New Concepts for Improving ISB
The Helitorch was originally developed for the U.S. Forest Service to set deliberate fires and was adopted by oil spill responders in the 1980s as a means to ignite oil slicks at sea and on ice (Allen, 1987). From an operational perspective, Helitorch use may be appreciably constrained by weather conditions, darkness, aircraft icing conditions, and distance to the spill site. In the mid-1990s, new formulations for Helitorch fuel improved the ignition of emulsified and hard-to-light slicks. More recently, gelled delivery systems that are potentially capable of being operated at higher speeds from a fixed-wing aircraft were tested in ground trials (Preli et al., 2011). API is sponsoring a program with the U.S. Forest Service and other agencies to evaluate safer, more effective alternatives to the Helitorch. The Arctic Response Technology JIP has as one of its research priorities the development of new high-speed aerial ignition systems (Mullin, 2012).
Other new ISB projects include a BSEE-funded Naval Research Laboratory study on the use of atomizers to burn emulsified oil,13 and validation and testing of an operational airborne application system for chemical herders planned under the Arctic Response Technology JIP (Mullin, 2012). In 2014, the JIP is also initiating a new project to evaluate the potential of chemical herders under different oil properties and weathering, as well as investigating windows of opportunity for their use.
MECHANICAL CONTAINMENT AND RECOVERY
Potter et al. (2012) define “containment and recovery” as actions taken to remove oil from the surface of water by containing the oil in a boom and/or recovering the oil with a skimming or direct suction device or sorbent material. After removal, the recovered mix of oil and water and contaminated materials needs to be stored offshore until it can be transferred to an approved disposal or recycling facility. Mechanical containment and recovery is often preferred over other oil spill countermeasures because they are viewed as directly removing oil from the marine environment.
However, there are problems using only mechanical containment and recovery, especially when dealing with large offshore spills in a remote area like the Arctic. Response to a large marine offshore spill is unlikely to rely only upon mechanical containment and recovery. Even though there were very good conditions for mechanical containment in the Deepwater Horizon spill, only an estimated 2% to 4% of the oil volume discharged was collected (Federal Interagency Solutions Group, 2010). This highlights a key drawback of mechanical containment and recovery systems when confronted by a large, rapidly spreading oil slick—there is an insufficient encounter rate to allow the skimmers to achieve a significant percentage of their theoretical recovery capacity (Allen, 1999). With any large spill in open water, oil rapidly spreads and forms a thin layer on the surface. Huge quantities of containment boom are needed to concentrate these thin slicks, requiring hundreds of vessels and skimmers to deploy and maintain. Skimmers can recover large volumes of water with the oil, result-
_____________
13 See http://www.bsee.gov/Research-and-Training/Oil-Spill-Response-Research/Projects/Project1012/.
ing in serious storage and disposal requirements that would be challenging in the Arctic (S. Ross Environmental Research Ltd. et al., 2010). With smaller, more contained spills in harbors and other protected waters, mechanical containment and recovery can be very effective. However, very large oil spills require a response approach that does not solely depend on mechanical recovery.
The use of booms and skimmers during the Arctic summer months, with open water, follows the same procedures practiced for decades in more temperate regions. The Arctic has extended daylight in the summer, which provides one great benefit for operation. However, the lack of approved disposal sites on land in many Arctic coastal areas, lack of port facilities to accept deep-draft vessels, and limited airlift capability to remote communities complicate the large-scale use of mechanical containment and recovery to respond to spills. Conventional booms and skimmers become increasingly ineffective as ice concentrations increase. Limited effectiveness is possible in very open drift ice (1/10 to 3/10) and in isolated polynyas within closer pack ice. The presence of ice interferes with boom operation and reduces flow to the skimmer head, greatly reducing overall effectiveness (Bronson et al., 2002; Potter et al., 2012).
Past research programs such as Mechanical Oil Recovery in Ice Infested Waters (Mullin et al., 2003), supported by industry and the Minerals Management Service, evaluated the concept of mechanically processing ice pieces, flushing the oil off, and returning the cleaned ice to the ocean. While it was successful at a very small scale of a few meters, scaling up to the size of realistic ice floe sizes led to a massive piece of equipment able to work in only a limited set of Arctic conditions. Finland developed specialized ship-based systems that process oiled ice, but they are tailored to handle very small floe sizes typical of frequently used shipping channels in the winter (Lampela, 2007). Scaling such systems up to handle Arctic ice is probably impractical, and limited by low encounter rates.
Currently, improvement of mechanical systems for Arctic applications focuses on cold temperature protection (e.g., heating and water injection systems to pump viscous oil at cold temperatures, and heating and enclosing sensitive skimmer components) and independent propulsion (e.g., allowing skimmers tethered to a mother ship to be guided into oiled leads). The SINTEF Oil in Ice JIP tested a number of new Arctic skimmer prototypes in tanks and offshore field trials in 2008 and 2009 (Sørstrøm et al., 2010). Several of these prototypes are now available commercially.
In situations involving small, contained spills in pack ice or larger spills under fast ice, mechanical recovery can provide a viable option. In the Baltic Sea, isolated pockets of oil contained in close pack ice have been mechanically recovered with over-the-side brush- or bucket-type skimmers (Lampela, 2007). Oil spilled under fast ice and contained by its undersurface roughness can be recovered mechanically through cutting, trenching, and drilling (ACS, 2012). Depending on location, oil recovered in this manner can be transported to shore over smooth ice or on ice roads for disposal and/or reinjection. These proven response strategies are most applicable to locations such as Prudhoe Bay, where stable fast ice extends tens of kilometers offshore.
Promising New Concepts in Mechanical Recovery
Future improvements in conventional mechanical recovery systems are likely to be incremental rather than transformative (Sørstrøm et al., 2010). The fundamental constraint of limited encounter
rate is made harder by the presence of ice and is not easily overcome with existing or proposed systems, although further modifications and improvements in cold-temperature operability are likely to continue. Although not generally considered in the category of mechanical recovery, well-capping systems can be viewed as a crucial component of mechanical recovery. They may be the most effective mechanical means of quickly stopping oil discharge from a subsea blowout, and they theoretically eliminate the need for any subsequent response.
Nonetheless, efforts have been undertaken to encourage the improvement of conventional mechanical recovery systems. One such effort was the Wendy Schmidt Oil Cleanup X Challenge,14 a competition designed to inspire innovative solutions that speed the pace of cleaning up marine oil spills from ships and other sources. The one-year competition ended in fall 2011, with the prize awarded to two teams that demonstrated oil recovery rates of over 2,500 gallons/minute, with efficiency rates of more than 70%. In 2014, BSEE is also planning to fund projects that potentially increase mechanical recovery of oil in drift ice.
ISSUES WITH OPERATIONAL LIMITS AND RECOVERY RATES OF COUNTERMEASURES
The knowledge gained from laboratory, tank, and field experiments under Arctic conditions can be used to determine operating limits for different countermeasures (Potter et al., 2012). However, operating limits by themselves are not good indicators of response effectiveness. For example, while it may be possible to ignite an oil slick in winds up to 10 m/s, the response will not necessarily achieve a high removal rate. Instead, removal rate will depend on a number of factors, such as oil thickness and degree of emulsification. Similarly, it is possible to deploy skimmers into isolated oil pools trapped between floes, but the recovery rate may be relatively low due to a need to continually relocate the skimmer and potential interference of ice with the skimmer intake (Bronson et al., 2002; Potter et al., 2012). Dispersants can theoretically be applied over a wide range of conditions, but unless a sufficient dose consistently reaches the thicker areas of an oil slick, overall effectiveness may be low.
Encounter rate may be the most critical factor for oil removal. This will depend on the speed of advance (e.g., vessels that tow boom, aircraft speed), the swath or sweep width (e.g., boom opening, aircraft or vessel spray width), the burn removal rate, and skimmer recovery rate. Allen et al. (1999) provides an in-depth discussion of the encounter rate as it applies to different response options. Calculating the expected recovery or removal rate for a particular response effort is a complex process, with no simple means of estimating spill response effectiveness. Instead, past experience with particular spills can help to assign ranges of recovery effectiveness for specific countermeasures. Net environmental benefit analysis (NEBA) can help responders, regulators, and stakeholders decide which oil spill response options could be recommended or advised against in a given situation. The principles of applying this type of analysis are discussed in Chapter 5.
As seen in the sections above, there are continuing efforts by industry and government to improve oil spill response both in the Arctic and elsewhere, but concerns remain regarding the ability to effectively mitigate a large spill in the Arctic or other remote locations. Members of the nongovernmental organization community maintain that even when used in conjunction, mechanical
_____________
14 See http://www.xprize.org/content/wendy-schmidt-oil-cleanup-x-challenge.
recovery, ISB, and biodegradation and dispersants may still only remove a small fraction of spilled oil (Goodyear and Beach, 2012; presentations by Stan Senner, Ocean Conservancy; Peter Van Tuyn, the Pew Charitable Trusts; and Chris Krenz, Oceana, June 2013). Furthermore, these groups call for rigorous testing under a range of Arctic conditions before using these techniques during a major spill.
DETECTION AND MONITORING
To mount an effective response, it is critical to know where spilled oil is at any given time. Finding and mapping oil in ice, even under favorable weather and light conditions, is far from straightforward. The lack of significant waves in the presence of ice complicates the use of marine or satellite radar systems, both of which depend on differences in surface roughness as a means of detection. With adverse weather or darkness, obtaining consistently reliable detection and mapping of contaminated areas becomes challenging and requires a mix of remote sensors operating in different parts of the electromagnetic spectrum. Assessments of remote sensing system capabilities for oil spills in ice have been forced to draw upon practical detection experiences of spills in open water environments (Coolbaugh, 2008; Dickins and Andersen, 2009; Fingas and Brown, 2011).
A wide range of sensor types have been tested for use in spill detection in ice. These include analytical, bench, and basin tests and field trials using a wide range of sensor types—acoustics, radar, ultraviolet fluorescence, infrared (IR), gamma ray, microwave radiometer, resonance scattering theory, gas sniffers, and ground penetrating radar (GPR) (e.g., Dickins, 2000; Goodman, 2008). Beginning in 2004, projects sponsored by the Minerals Management Service and industry (including the SINTEF Oil in Ice JIP) evaluated and tested a variety of sensors currently used to detect oil on open water to evaluate their potential for detecting oil in ice. In addition to the sensors above, these projects looked at side-looking airborne radar, synthetic aperture radar (SAR) satellites, forward-looking infrared (FLIR), trained dogs, and sonar (Bradford et al., 2010; Dickins et al., 2010). The current Arctic Response Technology JIP (2012-2015) is examining a range of these and other airborne, surface, and subsurface technologies in order to assign priorities for future development and testing (Puestow et al., 2013; Wilkinson et al., 2013).
Table 3.4 compares the capabilities of different sensors for remote sensing of oil spills in ice according to the platform and the oil/ice configuration over a range of ice environments (Dickins and Andersen, 2009). SINTEF JIP field experiments in 2008 and 2009 evaluated some of these technologies in close pack ice. Expected capabilities of different systems are based on information gathered during those experiments and from results of previous trials, not necessarily in the Arctic.
Dickins and Andersen (2009) concluded that current airborne systems are useful for detecting and mapping large spills in open ice but have much less potential as ice increases. Many of the non-radar sensors on airborne systems do not work well under Arctic conditions of darkness, cloudiness, fogginess, and rain for much of the year. A quantum leap in all-weather capability was realized in the late 1990s with the advent of commercially available, high-resolution SAR satellite systems, which are unaffected by darkness or cloud cover and can now resolve targets of a few meters (e.g., Radarsat, ERS-1, TerraSAR-X, COSMO-Skymed). First-generation SAR satellites mapped several large marine oil spills, including the Prestige, Nakodka, and Sea Empress (Hodgins et al., 1996; Lunel
et al., 1997). The ability of SAR satellites to detect and map oils slicks in the ocean with moderate wind conditions is likely to be practical for well-defined oil spills that spread in very open to open pack ice, where capillary waves can develop on the surface (Babiker et al., 2010).
The Deepwater Horizon oil spill provided an opportunity to utilize many of the latest detection technologies. Leifer et al. (2012) summarized how passive and active satellite and airborne marine remote sensing were applied to the spill. Slick thickness and oil-to-water emulsion ratios are key spill response parameters for containment and cleanup. These parameters were derived for thick (greater than 0.1 mm) slicks detected by the Airborne Visible/Infrared Imaging Spectrometer satellite. The Moderate Resolution Imaging Spectroradiometer (MODIS) satellite data allowed for detection of the total slick and was used to produce maps of oil thickness. Airborne and SAR provided synoptic data under all-sky conditions; however, SAR typically is not able to discriminate between thick oil slicks and thin sheens (0.1 mm or less). The Jet Propulsion Laboratory’s Uninhabited Aerial Vehicle SAR’s15 higher spatial resolution and signal-to-noise ratio led to better pattern discrimination.
At present, there is a lack of hard data to confirm theoretical assumptions about the performance of most remote sensing systems in a particular oil-in-ice scenario (GPR, discussed below, is one exception). SAR satellite imagery may be of use for detecting oil slicks in ice but will be dependent on factors such as the size of the spill, ice floe size and concentration, and wind speed. The main value of radar imagery in an Arctic spill incident lies in its ability to document changing ice conditions near a spill, which provides a valuable tactical tool for response (Potter et al., 2012). False positives or negatives are a concern with SAR imagery, because other conditions can create smooth surface areas that resemble oil (e.g., grease ice), while atmospheric phenomena can hide potential oil slicks (e.g., rain cells).
An important national issue is that the United States does not presently have its own commercial SAR satellite mission, so international partnerships are necessary. Access to images from international satellites typically only lasts about three days, which could be an issue for oil spill response (presentation by Nettie LaBelle-Hamer, University of Alaska Fairbanks, March 19, 2013). Classified SAR satellites from the National Geospatial-Intelligence Agency can be used for monitoring natural disasters and oil spills, but the information is only available to those people with appropriate clearances. However, derivative maps can be made using the data. Having emergency responders able to access necessary SAR data is of great importance for an integrated response.
In addition to rapidly developing remote sensing technologies, there will always be a need for well-trained observers flying in helicopters and fixed-wing aircraft to map oiled areas and to transmit critical information to response crews. Spotter aircraft were essential to the success of individual ISB operations during Deepwater Horizon (Allen et al., 2011).
Comprehensive tracking and long-term monitoring of oil released in ice require assimilating field data, plotting real-time observations, and integrating this information with forecasting tools such as weather models, ice drift algorithms, and oil spreading and weathering models. Commercially available ice-strengthened Global Positioning System (GPS) beacons and buoys have, for many years, been tracking ice movements during an entire winter season throughout the polar basin (Vaudrey and Dickins, 1996). Tracking oil spills accurately in a moving ice cover involves deploying large numbers
_____________
Table 3.4 Overview of Remote Sensing Systems for Detection of Oil in Ice
| Platform Sensor | Ice Surface | AUV | Shipborne | ||
| Dogs | GPR | Sonar | Marine Radar | FLIR | |
| OIL ON ICE | |||||
| Exposed on cold ice surface | Likely | NA | NA | Not likely | Likely* |
| Exposed on spring melt pools | Likely | NA | NA | Possible | Likely* |
| Buried under snow | Likely | Likely | NA | NA | Not likely* |
| OIL UNDER ICE | |||||
| Smooth fast ice | Possible | Likely | Likely | NA | NA |
| Deformed pack ice | Possible | Possible | Likely | NA | NA |
| OIL IN ICE | |||||
| Discrete encapsulated layer | Possible | Likely | Possible | NA | NA |
| Diffuse vertical saturation | Possible | Possible | Not likely | NA | NA |
| OIL BETWEEN ICE FLOES | |||||
| 1/10 to 3/10 concentration | NA | NA | Not likely | Likely | Likely* |
| 4/10 to 6/10 concentration | Not likely | NA | Not likely | Possible | Likely* |
| 7/10 to 9/10 concentration | Possible | NA | Not likely | Not likely | Likely* |
NOTE: An asterisk (*) denotes sensors blocked by dark/cloud/fog/precipitation.
SOURCE: Dickins and Andersen (2009).
| Airborne | Satellite | ||||
| GPR | Visible | UV | FLIR | SLAR | SAR |
| Likely | Likely* | Not likely* | Likely* | Not likely | Not likely |
| Not likely | Likely* | Possible* | Likely* | Possible | Not likely |
| Likely | Not likely* | Not likely* | Not likely* | Not likely | Not likely |
| Likely | NA | NA | NA | Not likely | Not likely |
| Possible | NA | NA | NA | Not likely | Not likely |
| Likely | NA | NA | NA | Not likely | Not likely |
| Possible | NA | NA | NA | Not likely | Not likely |
| Not likely | Likely* | Likely* | Likely* | Likely | Likely |
| Not likely | Likely* | Possible* | Likely* | Possible | Possible |
| Not likely | Likely* | Not likely | Likely* | Not likely | Not likely |
of beacons at regular intervals on the ice as oil moves away from the spill source, which can then be used to direct air and marine responders toward the spill. Closely spaced GPS beacons can follow the evolving pattern of spill fragmentation and divergence as the pack expands and contracts (Hirvi et al., 1987; Weingartner et al., 1998). There are also subsurface Lagrangian floats that can operate in ice-covered waters and be acoustically tracked while under sea ice, with capabilities including subsurface profiles down to 2,000 m water depth. These will be deployed under the Office of Naval Research Departmental Research Initiative Marginal Ice Zone Program.16 In the absence of sea ice or in reduced sea ice cover, the floats surface and send water-column profiles and subsurface position data via satellite; two-way communications allow the float’s drift depth and/or profiling frequency to be changed when the float surfaces.
Promising New Concepts in Detection and Monitoring
A number of systems, including GPR, are capable of both airborne and surface operation. Tank and field experiments from 2004 to 2006 showed that surface-based, commercially available GPR can detect and map oil sheens as thin as 1-3 cm underneath 1 m or more of solid ice or trapped as layers within ice (Dickins et al., 2008). In 2008, the same radar suspended beneath a helicopter traveling at speeds up to 20 kn and altitude up to 20 m successfully detected a thin layer of crude oil buried under hard-packed snow (Bradford et al., 2010). A prototype frequency-modulated continuous-wave radar designed to detect oil trapped under solid ice from a low-flying helicopter was developed in 2011-2012, with plans to test it as part of a new Arctic Response Technology JIP research program beginning in 2014.
More recently, consideration is being given to utilizing nuclear magnetic resonance as a potential means to detect oil trapped under or in ice in the future (Nedwed et al., 2008). Although further testing is needed to evaluate the practicality and effectiveness of an operational system, nuclear magnetic resonance has been successfully used to locate groundwater on multiple occasions.
IR systems (alone or in conjunction with high-speed marine radar and low-light-level video) can also be used from the surface, low-flying helicopters, aircraft (tracking high-resolution FLIR), or vessels. In 2009 SINTEF JIP tests, a basic uncooled hand-held IR sensor was able to discriminate between oil, open water, snow, and oil-free ice floes during daytime (Dickins et al., 2010). These experiments confirmed prior offshore releases in pack ice, in which IR differentiated warmer oil from cold water and ice (Singsaas et al., 1994).
Depending on ice conditions (e.g., floe size, thickness, stability), a variety of remote sensing systems that operate directly from the ice surface or from a nearby vessel could be deployed. As part of the SINTEF JIP, trained dogs on the ice tracked and located small oil spills buried under snow from a distance of 5 km and also determined the approximate dimensions of a larger oil spill (Brandvik and Buvik, 2009).
X-band marine radar has been used to detect slicks at sea in large-scale trials, and may be able to detect oil slicks in open ice (Dickins and Andersen, 2009). Integrated systems that combine high-
_____________
16 See http://www.apl.washington.edu/project/project.php?id=miz.
resolution FLIR and low-light cameras are now routinely deployed on response vessels in Norway as the SECurus system.17
Unmanned aerial vehicles (UAVs) and autonomous underwater vehicles (AUVs) already have the capability of carrying useful sensor packages over long distances (albeit at slow speed) for Arctic oil spill surveillance (Wadhams et al., 2006). Both single- and multi-beam sonar sensors successfully detected and mapped oiled boundaries and thicknesses under ice in a recent basin test at the U.S. Army Cold Regions Research and Engineering Laboratory (Wilkinson et al., 2014). A September 2013 exercise aboard the USCG Healey field tested UAVs, AUVs, and Arctic skimmers for response capabilities (USCG, 2013a). Further testing of different UAV sensors for oil spilled in ice is planned for 2014 and is being sponsored by the European Union and Arctic Response Technology JIP.
BSEE has recently partnered with the Army Research Development and Engineering Command to develop new sensing capabilities that could have applications during low-light periods,18 which could be especially useful for work in the Arctic.
Arctic nations such as Canada, Iceland, Finland, Denmark, Norway, and Sweden operate dedicated pollution surveillance aircraft. Canada dedicates one of its aircraft (a DHC-7) to Arctic missions. From 1983 to 2013, the USCG operated a fleet of Falcon Jets for maritime surveillance; most of these obsolete HU-25s have been decommissioned, with complete phase-out by 2014.19 They are being replaced by the HC-144A Ocean Sentries, which entered service in 2009 (there are currently 15). The search radars in USCG fixed-wing aircraft have a SAR setting that can be used for oil spill detection. There was some limited evaluation during Deepwater Horizon, but no rigorous testing has been performed and no testing with ice has yet occurred. The fixed-wing aircraft and helicopters have an electro-optical/IR system that may be useful in some Arctic conditions (Kurt Hansen, USCG, personal communication, February 3, 2014). A key aspect of the future effectiveness of remote sensing systems is the ability to integrate different datasets into a useful real-time or near-real-time product that responders can use with minimal interpretation. While there has been considerable progress on multispectral data fusion applied to pollution surveillance aircraft, an equivalent capability for Arctic spill surveillance has yet to be developed (Baschek, 2007). Lessons learned from Deepwater Horizon (Leifer et al., 2012) could be applied to optimizing future Arctic systems for detecting oil in ice.
TRAJECTORY MODELING
Several reviews of oil spill modeling technology are available (Huang, 1983; Spaulding, 1988; ASCE, 1996; Reed et al., 1999; Yapa et al., 2006; Drozdowski et al., 2011). Both earlier and more recent reviews recognize the challenges associated with oil spills in Arctic conditions—the presence of sea ice, cold and darkness in the winter, sparse observational networks for meteorological and oceanographic conditions, and a limited ability to respond to and monitor the physical and chemical evolution of a spill.
_____________
17 See www.aptomar.com.
18 See http://www.bsee.gov/Research-and-Training/Oil-Spill-Response-Research/Projects/Project1013/.
Oil in ice behaves in complex ways. Challenges associated with modeling the physics of ice growth, movement, and deformation on scales of meters or tens of meters are magnified when the details of oil behavior are incorporated. Background literature exists on oil-ice interaction studies over the past 40 years. DF Dickins Associates Ltd. and Fleet Technology Ltd. (1992) and SL Ross Environmental Research Ltd. et al. (2010) provide overviews of key studies. More recent work has largely focused on oil in and under ice (Yapa and Belaskas, 1993; El-Tahan and Venkatesh, 1994; Yapa and Weerasuriya, 1997), but mostly relies on small-scale, short-term laboratory studies. Ice lead dynamics that govern the spreading of oil in the field tend to not be included in these solutions, a significant shortcoming.
Understanding the effect of Arctic conditions on oil spill behavior and fate has increased significantly over the past decade (e.g., Gjøsteen et al., 2003; Faksness, 2007; Dickins et al., 2008; Brandvik and Faksness, 2009; Buist et al., 2009; Brandvik et al., 2010). Much of this knowledge has been garnered during mesoscale and field experiments (e.g., Singsaas et al., 1994; Buist et al., 2007; Sørstrøm et al., 2010), which show that in ice leads, evaporation, dispersion, and emulsification are slowed. The primary factors in determining observed weathering rates appear to be temperature, wave damping, and the presence of ice.
Despite these advances, there continues to be a need for additional Arctic data and for incorporation of these data into comprehensive oil spill models (Holland-Bartels and Pierce, 2011). A key problem in achieving such integration lies in a limited ability to model ice behavior at appropriate (meters) spatial scales. Forecasting (Reed and Aamo, 1994) and hindcasting (Johansen and Skognes, 1995) demonstrate the issues that can be encountered when oil-ice interaction models are used in the field. The limited capabilities of modeling ice behavior at scales of 1-10 m also significantly restrict the extent to which advances in oil spill modeling can be used. Ice coverage is dynamic and can change rapidly, with significant implications for oil weathering and transport.
Gjøsteen (2004) produced a model for spreading of oil in irregularly shaped simulated ice fields. Russian work in this field exists but is often not published in English, restricting its use and citation (e.g., Ovsienko et al., 1999). Both Gjøsteen and Ovsienko have developed spreading models that account for spreading of oil among ice floes. Incorporation into numerical models of these advances, as well as increased understanding of oil weathering processes in the presence of sea ice (e.g., Brandvik et al., 2004, 2005, 2010; Dickins et al., 2008; Faksness, 2007; Brandvik and Faksness, 2009; Buist et al., 2009), has been hindered by the interdisciplinary nature of the problem. Integration of knowledge of oil behavior and fates, ice cover, and hydrodynamic models is necessary for further significant advances in oil-ice interaction modeling.
Achieving higher spatial resolution using existing classic sea ice models (e.g., Hibler, 1979) is not sufficient, as robust oil spill models will need more detailed representations of sea ice (e.g., ice floe sizes, ice porosity, ice drift, ridging and growth rates, under-ice roughness). Oil spill models can then compute and retain fractions of oil on, in, and under ice floes, and allow for dynamic partitioning in both space and time. Advancement in this direction is needed for both sea ice and oil spill models, although neither is likely to be able to reliably perform at this level of detail in the near future.
The Arctic Oil Spill Response Technology JIP has recently initiated a research effort to improve oil spill trajectory modeling capability within the Arctic, with plans to develop new sea ice models related to ice dynamics. The models will be evaluated at regional scales using a high-resolution,
coupled ice-ocean model, and a new type of model will be developed to simulate sea ice dynamics in the marginal ice zone. All of the products developed during the JIP will be integrated into established oil spill trajectory models (Mullin, 2012).
BOEM currently uses a model called the Oil Spill Risk Analysis Model,20 which simulates the possible paths of oil spills through modeled ocean currents, winds, and sea ice. In March 2011, a workshop was held with the goal of improving BOEM oil spill models (SAIC, 2011). A main recommendation was the need for assimilating new oceanic, atmospheric, and sea ice data collected in the Arctic environment. Another was the requirement for validation and sensitivity testing of the model through integration with observational studies—for example, under programs such as the Forum for Arctic Ocean Modeling and Observational Synthesis.21
NOAA’s Office of Response and Restoration’s Emergency Response Division also employs an oil spill trajectory model, which is effectively a statistical model that is run many times to determine an uncertainty boundary where oil may be found. The General NOAA Operational Modeling Environment22 predicts possible trajectories of oil in water for given environmental forcing. An Arctic module contains information on tides, currents, bathymetry, and coastlines in the Arctic region, but does not include sea ice.
In 2013, BSEE and NOAA entered into an interagency agreement to adapt NOAA’s current models to better account for Arctic conditions. Their efforts are intended to complement the work being done by others, including the Arctic Oil Spill Response Technology JIP.
Promising New Concepts in Trajectory Modeling
Promising advances in sea ice modeling in the past decade include detailed models of brine-channel formation and drainage by Petrich et al. (2006, 2013). This approach allows for incorporation of oil into brine channels as well as bulk oil freezing into ice (Faksness and Brandvik, 2008; Faksness et al., 2011). Hopkins (1996) and Hopkins and Daly (2003) have developed a discrete element approach to modeling sea ice that allows for variably sized ice floes. These two advances together permit a parameterization of oil-ice interactions at a conceptual resolution that is significantly closer to reality than was previously possible. Wilkinson et al. (2007) have demonstrated the possibility of modeling the flow of oil under sea ice based on the topography of the under-ice surface. The authors relied on an AUV to map the under-ice contours, an approach that is probably not realistic over a long time frame and a dynamic ice field. Future ice models will need to produce an estimate of under-ice roughness if the spreading process is to be adequately represented. Other advances in oil spill modeling are occurring, although they are driven mostly by the Deepwater Horizon rather than by issues associated with the Arctic (e.g., the Gulf of Mexico Research Initiative23). There is a strong focus on underwater near-field plume modeling, including the effectiveness of dispersant injection at the wellhead.
_____________
20 See http://www.boem.gov/Environmental-Stewardship/Environmental-Assessment/Oil-Spill-Modeling/Oil-SpillRisk-Analysis-Model-%28OSRAM%29.aspx.
21 See http://www.whoi.edu/projects/famos/.
22 See http://response.restoration.noaa.gov/oil-and-chemical-spills/oil-spills/response-tools/gnome.html.
Uncertainty as Related to Numerical Tools for Decision Support
Numerical simulation models of coupled atmospheric, sea ice, and ocean physics are key components of today’s weather, ice, and ocean current forecasting operations. The success of these tools in providing accurate forecasts is defined by the degree to which model results reflect reality. This, in turn, depends on an understanding of the underlying processes, building this understanding into the models, and on available computational power. Simulation tools are also used for planning and training, support during a spill response, and evaluation of potential and actual environmental effects after an accident. These tools bring additional physical, chemical, and biological processes into the equation, along with their associated uncertainties.
Reliable forecast tools depend heavily on the assimilation of real-world observations because numerical models are by definition simplified representations of reality, relevant processes and their linkages are incompletely understood, and there are computational limitations on the ability to represent these processes over the full range of physical and temporal scales. The ability to supply observational data will remain a key element in reducing this uncertainty. For weather forecasts, such observations typically include wind speed, air temperature, humidity, and other environmental variables that are measured regularly at numerous locations on the Earth’s surface and in the overlying atmosphere. The density of similar measurements in the oceans and at the air-ocean interface is much less, but has increased significantly over the past two to three decades, particularly at lower latitudes. Such measurements are scarcer in the Arctic, where the complexity of the physical relationships is increased by seasonal growth, drift, and decay of sea ice. Adding spilled oil increases this complexity.
Simulation models are powerful integrators of complex interacting processes and events, and can be useful tools for decision support in both planning and executing emergency response actions. Statistical and ensemble analyses are two standard approaches to quantify the uncertainty of model results.
Statistical modeling is typically used in planning for a possible future event, when details of the event are not known beforehand. In oil spill contingency planning, for example, the timing, durations, and magnitudes of possible releases from a given production facility are unknown but can be estimated within certain bounds. One can then run a large number of simulations sampling from ranges of start times, release rates, and durations to produce probability maps for a variety of end points—for example, oil arrival time, the amount of oil on the sea surface, or the amount of oil along shorelines. Results can be used to support planning that protects priority resources. The probability of an impact on a given resource, weighted by the probable magnitude of the impacts, supplies measures of environmental risk that can in turn supply criteria for electing alternative response options.
Ensemble modeling simulates a given event using multiple models, then interprets the variability of the resulting outputs as a measure of intrinsic model-related uncertainty. This approach was used by both circulation and trajectory models during the Deepwater Horizon oil spill (e.g., Chang et al., 2011; Marianoa et al., 2011). It is also commonly applied in climate change modeling (e.g., Djalalova et al., 2010) and is most useful when there are multiple available models of similar complexity and focus.
Uncertainty in oil spill trajectory forecasting for open water conditions has been a recognized
challenge for decades (e.g., Galt, 1994) and will probably be greater in the presence of sea ice (Eicken, 2013). Trajectory analyses for decision support will need to account for this uncertainty in ways that are useful to response practitioners. Overlaying the trajectory forecast and the associated envelope of uncertainty on natural resource maps provides a graphical basis for decision making and reflects the present state of the art (e.g., NOAA Gulf Spill Restoration, 2012).
CHAPTER CONCLUSIONS AND RECOMMENDATIONS
Conclusion: Arctic oil spill research and development needs for improved decision support include:
- Determining and verifying biodegradation rates for hydrocarbons in offshore environments, in order to establish potential and capacities for natural attenuation or recovery and to determine which strategies can accelerate oil biodegradation;
- Improving technologies for dispersant application and induction of turbulence for oil spills in ice;
- Evaluating the toxicity of dispersants and chemically dispersed oil on key Arctic marine species, with appropriate experimental design and incorporation of real-world conditions and concentrations;
- Identifying and understanding ecosystem responses associated with changes in microbial biomass and species alterations;
- Further understanding of biogeochemical cycles, including particulate matter–petroleum chemical interactions;
- Improving ignition methods for in situ burning, focused specifically on the Arctic;
- Mapping the usefulness of chemical herders at different spatial scales, oil types, and weathered states, and in conjunction with other response options such as in situ burning;
- Understanding the limitations of mechanical recovery in both open water and ice, which will improve decision making regarding possible implementation of other response strategies;
- Investing in under-ice detection and response strategies, including remote sensing technology that will reliably detect oil in different ice conditions;
- Integrating remote sensing and observational techniques for detecting and tracking ice and oil, including UAVs, AUVs, SAR, and drifting buoys;
- Additional research into the physics of oil incorporation into developing ice;
- Establishing robust operational U.S. Arctic meteorological-oceanographic-ice and oil spill trajectory forecasting models for contingency planning and response support;
- Testing and evaluating risk-benefit decision processes, including NEBA, for use in the Arctic; and
- Summarizing relevant ongoing and planned research worldwide to achieve synergy and avoid unnecessary duplication.
Conclusion: Though much is known about the oil behavior and response technologies in ice-covered environments, there are areas where additional research is needed to make informed decisions about the most effective response strategies for different Arctic situations. In addition, there is a need to validate current and emerging oil spill response technologies on operational scales under realistic environmental conditions.
Conclusion: A systematic program of carefully planned and controlled field experiments that release oil in the U.S. Arctic is needed to advance understanding of oil behavior and response options.
Recommendation: A comprehensive, collaborative, long-term Arctic oil spill research and development program needs to be established. The program should focus on understanding oil spill behavior in the Arctic marine environment, including the relationship between oil and sea ice formation, transport, and fate. It should include assessment of oil spill response technologies and logistics, improvements to forecasting models and associated data needs, and controlled field releases under realistic conditions for research purposes. Industry, academic, government, non-governmental, grassroots, and international efforts should be integrated into the program, with a focus on peer review and transparency. An interagency permit approval process that will enable researchers to plan and execute deliberate releases in U.S. waters is also needed.
Recommendation: Priorities for oil spill research should leverage existing joint agreements and be addressed through a comprehensive, coordinated effort that links industry, government, academia, international and local experts, and non-governmental organizations. The Interagency Coordinating Committee on Oil Pollution Research, which is tasked to coordinate oil spill research and development among agencies and other partners, should lead the effort.
Conclusion: The oil spill response toolbox requires flexibility to evaluate and apply multiple response options, whether on their own or concurrently. No single technique will apply in all situations.
Recommendation: Dispersant pre-authorization in Alaska should be based on sound science, including research on fates and effects of chemically dispersed oil in the Arctic environment, experiments using oils that are representative of those in the Arctic, toxicity tests of chemically dispersed oil at realistic concentrations and exposures, and the use of representative microbial and lower-trophic benthic and pelagic Arctic species at appropriate temperatures and salinities.
Conclusion: Well-defined and tested decision processes for oil spill countermeasure deployment are critical to expedite review and approval. Decision processes need to include rapid research on countermeasures and be exercised regularly.






































