

































Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
THE NONHUMAN PRIMATES OF COLOMBIA Jorge Hernandez-Camacho and Robert W. Cooper INTRODUCTION The faunal, floral, and physiographic diversity of Co- lombia is among the richest of the neotropics. Although many areas of the country are not as yet well studied, at least 12 genera and 22 species of nonhuman primates are presently known. Only Brazil, with about 16 genera and 32 species and 6.5 times more land area, has more nonhuman primate forms. It is not possible, based on presently available data, to say exactly where the suborder Platyrrhini first arose. However, the origin of primates in Colombia probably dates to the Tertiary period, when the first platyrrhines became established in South America. Of the five known familiesâCallitrichidae, Cal- limiconidae, Cebidae, Homunculidae (known from Argentina), and Xenothricidae (known from Jamaica)âonly the latter two are absent in the fossil and/or recent record of Colombia. From the middle Magdalena Valley three monotypic genera of Cebidae (Cebupithecia, Neosaimiri, and Stirtonia) are known from the late Miocene period (Hershkovitz, 1970). Even so the fossil record for neotropical primates is very limited at present, with no known material avail- able from Mexico and Central America and with the earliest South American records being those from the Tertiary period in Colombia, Bolivia, and Argentina. In historic times nonhuman primates were first documented in Colombia in some of the early chroni- cles following the Spanish conquest. Cotton-topped tamarins (Saguinus oedipus) and white-throated capuchins (Cebus capucinus)from Colombia and tufted capuchins (Cebus apella), squirrel monkeys (Saimiri sciureus), and black spider monkeys (Ateles paniscus) probably from the Guianas were first described by Linnaeus in 1758 based on several such early ac- counts. By 1766 Linnaeus had also described red howler monkeys (Alouatta seniculus) from Cartagena based on a seventeenth-century account of von Jac- quin. In 1807 Count von Hoffmansegg first described, from the Para region of Brazil, the two species of titis (Callicebus moloch and C. torquatus) that also occur in Colombia. The next major advance in descriptive knowledge of primates to occur in Colombia came with the travels of the famous German explorer Alexander von Humboldt in the early nineteenth century. He first reported the existence of night monkeys (Aotus trivirgatus), black-headed uakaris (Cacajao melano- cephalus), woolly monkeys (Lagothrix lagotricha), and white-fronted capuchins (Cebus albifrons). At about the same time (1812) Etienne Geoffroy-St. Hilaire described monk sakis (Pithecia monachus), probably based on specimens from Brazil. In 1823 von Spix, after 6 years in the Amazon region, reported the existence of pygmy marmosets (Cebuella pygmaea) and both saddle-backed and black-mantled, white- lipped tamarins (Saguinus fuscicollis and 5. nigricol- lis). By 1823, 11 of the 12 genera and 16 of the 22 species known in Colombia had been described. The genus Callimico and five additional species (Alouatta palliata, Saguinus geoffroyi, S. leucopus, S. graellsi, and S. inustus) were then reported over the next 128 years, the latest being 5. inustus in 1951. The development of descriptive, zoogeographic, and 35
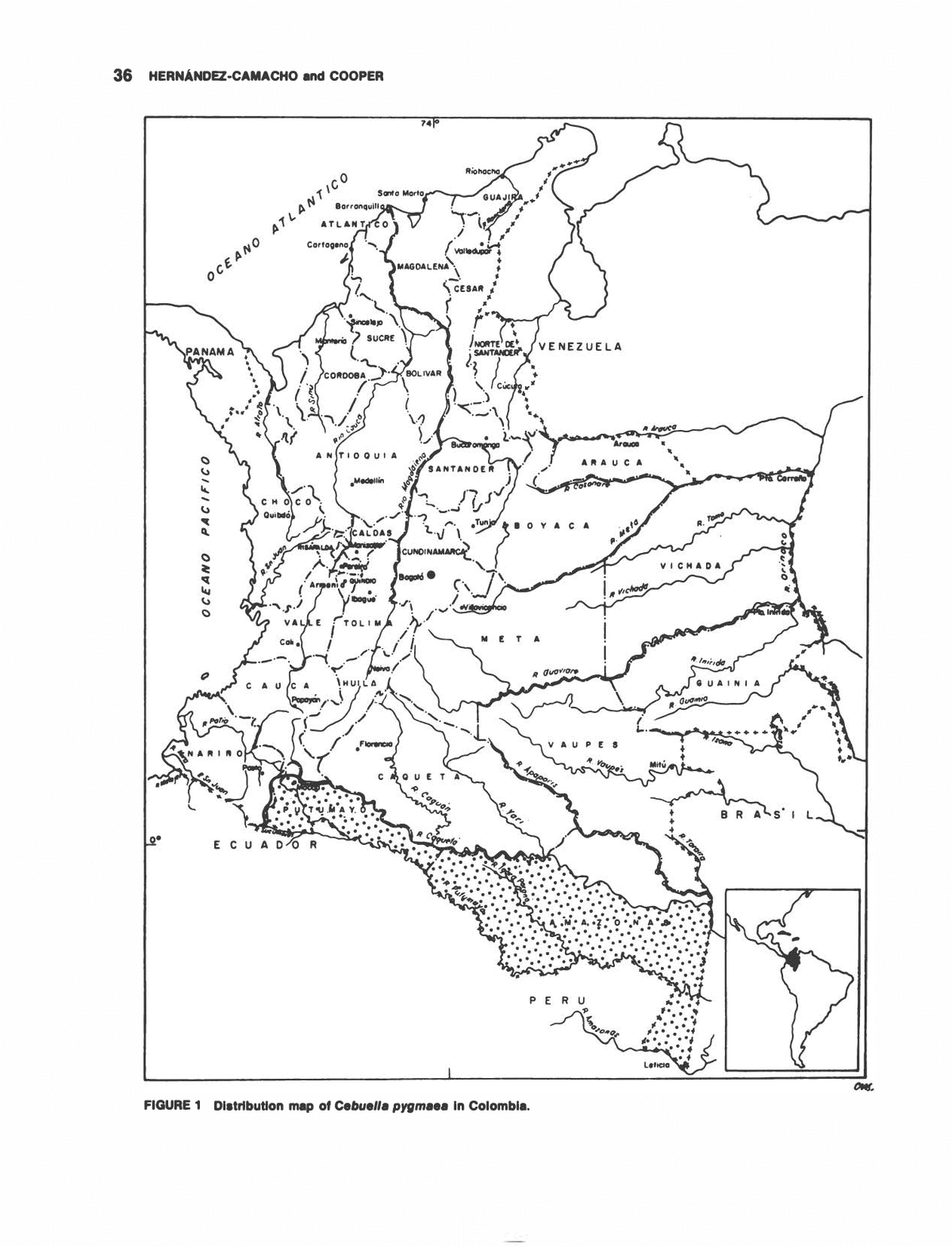
36 HERNANDEZ-CAMACHO and COOPER FIGURE 1 Distribution map of CebueIla pygmaea in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 37 natural historical information on neotropical primates has been relatively slow and sporadic, and the infor- mation produced in most areas of inquiry has been rather superficial. Unfortunately, this chapter will seldom depart far from that tradition. However, by summarizing some historical information, as well as more recent data based largely on the senior author's diverse field and museum experience in Colombia, we have attempted to add a few more bricks and possibly some missing mortar to the growing foundation of Colombian and general neotropical primate knowledge. Proceeding by approximate phylogenetic order of species, regional common names will be given, as well as known or probable distribution patterns, taxonomic notes and suggestions, general habitat preferences, some aspects of natural history, probable present population status, and important adverse population pressures. Our intention is to provide points of refer- ence and departure for students of primate biology in Colombia. By summarizing much of the basic informa- tion presently available to us, we hope to stimulate additions and corrections from the likely wealth of obscure published and unpublished information pres- ently known only to a few. In this spirit, we welcome constructive criticisms, new data, suggestions, etc., directed either to the authors and/or published for the benefit of all. 1. Cebuella pygmaea (Spix, 1823)âPygmy mar- mosets. COMMON NAMES: "Chichico" along the Putumayo River; "Leoncito" in the Putumayo and Amazon regions; and "Piel Roja" and "Mico de Bolsillo," popularized by animal dealers. DISTRIBUTION: (Figure 1) Cebuella is well known in the Colombian Amazon region south of the Caqueta River. Reports of its existence further north, in the upper Guaviare River region, remain to be documented. A captive specimen reportedly from Cano Morrocoy, on the south bank of the Guayabero River, about 2 km eastward from the town of El Refugio (also known as La Macarena), is the best evidence to date. In Colombia Cebuella is typically an inhabitant of mature, nonflooding forest. Its ecological association with the guarango tree (Parkia sp.) as a source of sap is one of the more striking dietary specializations known among neotropical primates. HABITS: Pygmy marmosets are rather difficult to find due to their small size, the camouflage of their coat color, their squirrellike habit of moving to the opposite side of a trunk when disturbed, and their lack of any conspicuous physical or vocal display. They have been observed in groups as large as 10 or 15 and are seldom, if ever, seen alone. They are most often found on the trunk or major branches of a guarango tree and, if disturbed while low on the trunk, are known to run to the ground for escape. Their terres- trial route to and from guarangos is evident by their intermittent presence in such trees in clearings a short distance from undisturbed forest. As mature guaran- gos are usually emergent over the forest canopy and somewhat free of climbers, it appears that such ter- restrial passage must be rather common. The stomach contents of specimens examined in March 1965 in the vicinity of Puerto Leguizamo, Comisaria of Putumayo, contained largely jellylike, dirty-whitish-colored guarango sap in addition to some finely crushed insects (mostly Coleoptera) and evidence of fruit pulp. The small sap-producing holes in the bark of the trunk or branches of guarango used by Cebuella appear to be produced largely, if not exclusively, by their procum- bent lower incisors. STATUS: As in many other Colombian primate species, the exact status of Cebuella is uncertain. It is fortunate that guarango is not a highly desirable timber species and is large enough to be left standing in many man-made clearings. Pygmy marmosets, of course, are unlikely to be hunted for their meat, but are sometimes collected for commercial purposes. In some areas the centuries old method of ringing a guarango trunk with a sticky resin to capture pygmy marmosets as they enter or leave the tree is used with considerable suc- cess. It is likely that this means of trapping is as ancient as the practice of some upper Putumayo River Indians who keep captive Cebuella to pick lice from their hair. Cebuella is also captured on occasion in banana-baited traps (intended forSaguinus spp.) near ground level. It is doubtful that such limited commercialization is nearly as threatening to the relatively prolific Cebuella as is habitat destruction. However, as discussed else- where in this volume (Moynihan, 1976), Cebuella seems to have remarkable adaptive abilities with regard to living as a near commensal of man in highly degraded habitat situations. 2. Saguinus nigricollis (Spix, 1823)âWhite-lipped, black-mantled tamarins. COMMON NAMES: "Leoncito" in the upper Putumayo River region; "Bebeleche" in Amazonian Colombia. DISTRIBUTION: (Figure 2) In ColombiaS. nigricol- lis occurs within about the same region as Cebuella pygmaea with the Caqueta River forming the northern limit. The population of the upper Putumayo River has a dull and brownish cast to the lower back and hind limbs, as well as some grizzled yellow and black in the saddle. This population is thus more reminiscent of S.
38 HERNANDEZ-CAMACHO and COOPER M> A/- // . « E T A 1.\ ^\ Saguinus nigricollis w Saguinus graellsi CMt FIGURE 2 Distribution map of the genus Saguinus (part) in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 39 fuscicollis than is the lower Putumayo and Leticia population, which has a rich ferrugineous cast to the lower back and hind limbs and no yellowish tones in the saddle area. Our material is inadequate to deter- mine whether or not these differences represent sub- specific distinctions. S. nigricollis in Colombia oc- cupies a wide range of rain forest habitats, from primary to secondary growth, and includes both sea- sonally flooded and nonflooding areas. HABITS: Group size seems to average between 5 and 10 individuals, whose high-pitched vocalizations are often heard well before the group is seen by an observer. They seem to prefer a middle canopy level and are most often seen at about 8 or 10 m above the forest floor. Stomach contents usually contain a mix- ture of fruits, berries, and insects. To date the only interspecific associations known to us are observa- tions of apparently mixed groups of S. nigricollis with S. fuscicollis in the area of Puerto Leguizamo. STATUS: For the past few years 5. nigricollis has been captured in the Leticia region for use in biomedi- cal investigation (principally virus cancer and infec- tious hepatitis studies). S. nigricollis was the most frequently encountered primate species during a field survey conducted in March 1972 from Leticia to a point some 70 km upriver on the Colombian bank. Within a 1,000-km2 region fronting on the Amazon and extending 15 to 18 km from its north bank, this species was encountered 10 times in a total of 75 hours of foot travel in undisturbed rain forest. It seems to be a rather adaptive species, living both close to human habitation and plantations and deep within relatively undisturbed primary forest. 3. Saguinus graellsi (Jimenez de la Espada, 1870). COMMON NAMES: "Bebeleche" in the Putumayo region. DISTRIBUTION: (Figure 2) 5. graellsi is known in Colombia from the Comisaria of Putumayo on the basis of a preserved specimen (Universidad Nacional de Colombia, Bogota), a number of reliable sightings (e.g., Moynihan, 1976), and captive specimens from the neighborhood of Puerto Asis eastward to the vicinity of Puerto Leguizamo. The northern limit is probably the southern bank of the Caqueta River. The eastern limit is unknown. 5. graellsi is undoubtedly sympatric with 5. fuscicollis fuscus throughout its range as well as with the population of S. nigricollis in the region of Puerto Leguizamo. HABITS: Unknown. STATUS: Unknown. 4. Saguinus fuscicollis (Spix, 1823)âWhite-lipped, saddle-backed tamarins. COMMON NAMES: "Bebeleche" throughout its range. DISTRIBUTION: (Figure 3) S. fuscicollis inhabits forested lowlands of the Amazon Basin in Colombia southward from the Caqueta River, northward in the Intendencia of Caqueta (eastward at least to the west- ern bank of the Yari River and northward to the southern bank of the Guayabero River in the southern Department of Meta), and eastward to the region around San Jose de Guaviare on the southern bank of the Guaviare River in the Comisaria of Vaupes. The species ranges upward to 500 m in the Andean pied- mont. The populations of the Putumayo River, including one specimen from the right bank of the Guayabero River in Angostura near the southern tip of the Macarena Mountains, are referrable to S. fuscicollis fuscus (Lesson, 1840). Unpreserved specimens from San Jose de Guaviare suggest that a presently unde- scribed subspecies inhabits this region. Live speci- mens recently examined in Leticia and reportedly from Puerto Narino on the Colombia bank of the Amazon are referrable to 5. fuscicollis triparitus (Milne-Edwards, 1878). 5. fuscicollis is not known in the immediate region of Leticia. Also, no specimens are available for the region between the Guamues and the Sucumbios Rivers of the southwestern Comisaria of Putumayo. HABITS: S. fuscicollis is usually encountered in groups of 5-20 individuals in primary forest, advanced second growth, or even areas of human activity. Stomach contents indicate a preference for various berries and insects. S. fuscicollis has been observed in mixed groups with 5. nigricollis in the region of Puerto Leguizamo. STATUS: S. fuscicollis is not seriously exploited in Colombia and not well known in all areas of its supposed range. Its population status is probably unthreatened and it may well have a somewhat inter- rupted distribution. 5. Saguinus inustus (Schwartz, 1951). COMMON NAMES: "Mico Diablo," "Diablito," and "Titi Diablito" in the Comisaria of Vaupes. DISTRIBUTION: (Figure 3) At present the species is known in Colombia from authentic records in the area of Mitii on both sides of the Vaupes River, as well as from San Jose Guaviare, on the southern bank of the Guaviare River, about 1 hour by boat downstream from the confluence of the Guayabero and Ariari rivers. Scattered reliable reports indicate its presence in other areas of the Comisaria of Vaupes and Guainia (Imâ¢rida and lower Guaviare rivers). It is likely that the Guaviare River forms the northern limit of the species, possibly as far west as some point between the towns
40 HERNANDEZ-CAMACHO and COOPER E C U A D'O Saguinus fuscicollis subspecies S. inustus geoffroyi oedipus leucopus S. S. S. FIGURE 3 Distribution map of the genus Saguinus (part) in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 41 of El Refugio and San Jose Guaviare; in 1959 the species was unknown as far north as the immediate vicinity of El Refugio. The southern limit is probably the Apaporis River, based in part on its history as an effective barrier for a number of other species and subspecies of terrestrial and arboreal vertebrates. It is also possible that its range may extend northward to the gallery forests of the Ariari from accounts of recent settlers living near Granada. HABITS: Although the species is found in rain forest in small groups, it is totally unstudied in Colom- bia. The possibility exists that its range is interrupted due to the heterogeneity of vegetational types (includ- ing chersophytic and chasmophytic dwarf forests, scrubs, and savannas) in Vaupes and Guainia. STATUS: Probably unthreatened but not known with certainty. 6. Saguinus geoffroyi (Pucheran, 1845)âGeoffroy's tamarins. COMMON NAMES: "Titi" and "Bichichi" in the Department of Choco. DISTRIBUTION: (Figure 3) S. geoffroyi occurs in Colombia from the Panamanian border probably as far south as the San Juan River, on the Pacific coast. The eastern limit is the western bank of the Atrato River. The upper altitudinal limit of its range is unknown, but it probably does not exist above 800 m of elevation. It seems more abundant in secondary forests or early secondary growth interspersed with plantations and seldom inhabits deep climax forest. Of the few museum specimens of S. geoffroyi known for Colombia as well as those seen in captivity (largely from the region of AcandO, a large percentage have distinct sulfur- yellowish underparts, including lightly pigmented areas of the limbs. This characteristic is seen in both juveniles and adults of either sex and appears to be individually variable, i.e., other specimens from the same region have perfectly white underparts, hands, and feet. HABITS: See 5. leucopus. STATUS: S. geoffroyi is very poorly known in Colombia due to the relative remoteness of the region in which it occurs. However, it is seldom exploited, and human population is neither very large nor ex- panding greatly within this area. 7. Saguinus oedipus (Linnaeus, 1758)âPinches or cotton-topped tamarins. COMMON NAMES: "Titi," "Titi Blanco," "Titi Leoncito," and "Titi Pielroja" throughout the range in northwestern Colombia. DISTRIBUTION: (Figure 3)5. oedipus is endemic to Colombia and occurs from the Uraba region of the northwestern Department of Antioquia southward at least to the Leon River and in the departments of Cordoba, Sucre, northern Bolivar, and Atlantico. The eastern limit is the western bank of the lower Mag- dalena and lower Cauca rivers, extending into north- central Antioquia. Recent investigation has disclosed that S. oedipus is absent from Mompos Island and is there replaced by S. leucopus. Cotton-topped tamarins occur in rain forest, deciduous forest, and second growth but are absent from xerophytic forest. Their highest reported localities are at elevations of about 400 m but they may range somewhat higher in the upper Sinu Valley. The vast majority of S. oedipus appear to have totally white underparts, forearms, hands, and feet. Only one museum specimen and several captive specimens from the San Jorge River basin have been observed to possess a sulfur-yellowish tinge to these areas. Another seemingly individually variable character is the shade and extent of the rufous coloration of the thighs and proximal tail. HABITS: See 5. leucopus. STATUS: S. oedipus has been heavily commer- cialized for at least 10-15 years. It is likely that as many as 30,000 to 40,000 cotton-topped tamarins have been exported during this period. Much of the species' habitat lies within a major cattle-grazing area of Co- lombia in which suitable forests have continually been cut or degraded. The present population status of 5. oedipus is unknown in absolute terms; however, con- cern for its survival is growing. Although the species is able to exist in small forest remnants, second growth, and in greatly altered areas, so much of its original habitat has been effectively destroyed that 5. oedipus has been protected by law in Colombia since 1969. 8. Saguinus leucopus (Giinther, 1876)âWhite-footed tamarins. COMMON NAMES: "Titi" and "Titi Gris" through- out the range in northcentral Colombia. DISTRIBUTION: (Figure 3) S. leucopus is endemic to Colombia and occurs in northeastern Antioquia (regions of Caceres, Valdivia, and the Nechf River Valley); southern Bolivar, including Mompos Island; and the western bank of the middle Magdalena River in the departments of Antioquia, Caldas, and northern Tolima (at least as far south as the vicinity of Mariquita). Its general limits are the eastern bank of the lower Cauca River, the western bank of the middle Magdalena River (including all of the larger river islands), and the foothills of the central Andes. Colora- tion in 5. leucopus seems to be rather constant, with the known exception of specimens from the extreme south- er n range of Parroquia de Bocaneme near Mariquita, whose basal portions of all colored hairs are decidedly darker brown than observed elsewhere. In addition
42 HERNANDEZ-CAMACHO and COOPER FIGURE 4 Distribution map of CalIimico goeldii in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 43 there are distinct dusky ulnar and tibial stripes and the tail is black rather than brown (except for the white tail tip). Specimens from several regions also demonstrate an individually variable, brown, elongated spot over the fourth metatarsal of each foot. HABITS: On the basis of scant information avail- able to us, no real distinction can be made between the habits and behavior of S. geoffroyi, S. oedipus, and S. leucopus. Group size reportedly ranges from 3 to 12 or more individuals. They are found at all levels of the forest and most frequently at its fringes near streams or other natural barriers and in areas of second growth. Food habits are diverse but include a strong preference for insects and fruits (particularly various types of berries found in the understory or in second growth). Also, on one occasion an adult male 5. oedipus in a semicaptive environment was observed to leap upon, decapitate with one bite, and consume in total a small Iguana iguana. It seems possible that the tendency of these species to be active in areas with more direct solar exposure may be related in part to their taste for insects and small lizards. Our observations in Colom- bia also lend support to the suggestion (Moynihan, 1970) that the decreasing abundance of S. geoffroyi on Barro Colorado Island in the Panama Canal Zone is probably related to the elimination of important ecological niches as the forest reaches an advanced successional stage. All of these closely related tamarins react in ap- parent fright to raptorial birds, including even rather small species. Interaction with other primate species is unknown to us, although all three forms live in or near habitat often occupied by Cebus capucinus or C. albifrons. Data on possible seasonal reproduction in the wild are limited, but it seems that such a tendency exists, based both on records in captivity (Cooper, unpublished data) and unconfirmed field reports. Al- though twin births are characteristic of tamarins and marmosets in general, people familiar with these species in the wild are seldom aware of this condition from field observation. STATUS: S. leucopus is little exploited, but its habitat has been greatly reduced due to clearing of forests, particularly during the past 20 years. 9. Callimico goeldii (Thomas, 1904)âGoeldi's mar- mosets. COMMON NAMES: None in Colombia. DISTRIBUTION: (Figure 4) In Colombia C. goeldii has been collected in two localities between the upper Putumayo and Caqueta rivers (Hernandez-Camacho and Barriga-Bonilla, 1966) and from a third locality in the lower Guamues River, a major southern tributary of the upper Putumayo. The northwesternmost collection was on the Igara-Parana River near La Chorrera; and the intermediate locality is Quebrada del Hacha, on the north bank of the Putumayo River. All specimens have been collected in nonflooding forest, either level or with low rolling hills. HABITS: According to the Kofan Indians in the Guamues region, Callimico is found most frequently in the understory and even on the ground, and its diet includes berries and small fruits. The single specimen collected near the mouth of the Quebrada del Hacha was associated with a group of Saguinus fuscicollis fuscus and was the only Callimico seen. STATUS: The species is poorly known over most of its supposed range in Colombia. The status of any local population is unknown. Occasional specimens that have been seen in the Leticia market are thought to be from upriver as far as the Ucayali, in Peru. 10. Saimiri sciureus (Linnaeus, 1758)âSquirrel mon- keys. COMMON NAME: "Titi" in the eastern plains, the Caqueta region, and the upper Magdalena Valley; "Vizcaino" in the Caqueta and the upper Guayabero River regions; "Mico Fraile," "Fraile," and "Frailecito" in the Putumayo River and Leticia re- gions; "Barizo" on occasion in the Leticia area (of Peruvian origin); "Saimiri" in the Leticia region (from Tupi Indian roots); "Menechino" (Tukano Indians, fide Olalla from specimen labels). DISTRIBUTION: (Figure 5) In Colombia Saimiri occupies all of the Colombian Amazon and the pied- mont of the eastern Andes and a considerable portion of the southeastern plains. The limit of its northeastern extension is not well defined, but it appears that increasing grasslands and decreasing gallery forest and rainfall are limiting factors. It occupies a wide range of forest types from gallery to low canopy sclerophyllous and hillside forests to palm forests (particularly associations of Mauritia flexuosa) and both seasonally flooded and nonflooding rain forests. In the upper Magdalena Valley it has a similar distribu- tion to Cebus apella, and its northernmost range is not well defined. The maximum elevation at which it has been observed is about 1500 m in this region. The llanos (eastern plains) and piedmont populations southward to the Caqueta River are rather uniform in appearance and differ from those of the Colombian Amazon mainly in having grizzled gray rather than yellow forearms and wrists. Their subspecific designa- tion is clearly S. s. caquetensis (J. A. Allen, 1916) but the subspecific status of the Amazon populations is awaiting careful revision of the entire species (see Cooper, 1968). HABITS: Group size in the gallery forests of the
44 HERNANDEZ-CAMACHO and COOPER FIGURE 5 Distribution map of Saimiri sciureus in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 45 eastern plains is relatively small, as noted by Thorington (1968) and by Baldwin (1971). Apparent single individuals are seen, and a maximum of about 30, mostly subadult, specimens was observed by Hernandez-Camacho traveling with a group of Cebus apella in piedmont forest near Villavicencio in June 1955. Group size in the Colombian Amazon is typically larger, but we are not aware of reliable accounts of more than 40 or 50 individuals. In all habitat types they seem to travel mostly beneath the highest available canopy but have been observed to come to the ground on rare occasions. Their food preferences are very broad and include fruits (e.g., Cecropia spp., Ficus spp., Euterpe spp., Rubiaceae, Campomanesia sp., etc.), berries, insects, spiders, etc. Thorington (1968) noticed births in the eastern plains in February or March, and Amazon populations seem to have about the same birth season (Cooper, 1968). STATUS: Squirrel monkeys are not seriously hunted for food in Colombia, but they are captured commercially in large numbers in the Leticia region. In March 1972 only one Saimiri group was encountered in nearly 75 hours of travel in undisturbed rain forest along the Colombian Amazon from Leticia upriver some 70 km. Although systematically collected data are lacking, it seems likely that numbers may be decreas- ing in local areas of intensive trapping and/or habitat destruction. The upper Magdalena Valley population is the most seriously endangered due to habitat de- struction, and protective measures are definitely needed. 11. Aotus trivirgatus (Humboldt, 1811)âNight mon- keys, owl monkeys, or Douroucoulis. COMMON NAMES: "Marta," "Martica," and "Marteja" in northern Colombia; "Marta" in the departments of Antioquia, Caldas, Quindio, Risaralda, Tolima, and northern Valle; "Mico Dormilon" in central Colombia; "Tutamono" and "Tutumono" in Metaand the Amazon; "Sorbehumo" in Meta; "Mico de Noche" in central Colombia and the Amazon; "Maco Cagao" in Santander. In general, except for "Maco Cagao," "Mico de Noche," "Mico Dor- milon," and "Sorbehumo," these names are also used for kinkajous (Potos flavus), olingos (Bassaricyon gabbii), woolly opossums (Caluromys spp.) and pygmy anteaters (Cyclopes didactylus). "Sorbehumo" is also applied to hawks like Buteo platypterus and Buteo swainsoni due to their habit of hunting at the edge of a fire (Aotus is said to approach campfires at night). The name "Douroucouli" is an onomatopoeic rendition of the vocalization of Aotus based upon the report of Justin Goudot (quoted by Alston, 1879). DISTRIBUTION: (Figure 6). Aotus is known in all of Colombia except for the Guajira Desert in the north- east, the altitudinal zones over 3,200 m, the northeast- ern plains, and several local savanna-and-scrub moun- tain areas of the comisarias of Guaima and Vaupes in the geological zone known as the Guayana Shield. With the exception of mangrove swamps, Aotus typi- cally inhabits every major forest zone of Colombia, including second growth and even well-shaded coffee plantations. Aotus trivirgatus subspecies, found throughout Colombia, may best be described accord- ing to relatively distinct regional characteristics of color pattern on the hands and feet and length of hair coat. A. t. griseimembra (Elliot, 1913) occurs in the lowlands of northern Colombia as well as the Santa Marta Mountains and extends westward to the neighborhood of the Simi River, the San Jorge River, the lower Cauca River, and the lowlands of the middle and upper Magdalena River Valley. The subspecies is typically characterized by a relatively short and somewhat adpressed hair coat, upperparts with a decidedly brownish or yellowish-brown cast, under- parts rather dull-to-light yellowish, and the dorsal surfaces of hands and feet light brownish with rather inconspicuously dark hair tips. Unfortunately, the type specimen with a strong dark admixture on the hands, and black sides on the feet, is exceptional; an exact topotype fits the more usual pattern as described previ- ously. The only other specimens seen at any significant variance with this general description are those of the Santa Marta Mountains at higher elevations (which have a tendency toward longer hair coats) and a single adult male from Ayacucho near La Gloria (500 ft) in the middle Magdalena Valley (which had very prominently dark hair tips on the dorsal hands and feet). Also, an adult pair and a subadult female from Sierra Negra (1000 m) in the Perija Mountains east of the Santa Marta Mountains near the Venezuelan border had longer hair coats, but the distal portion of the tail was dark brown instead of black, differing from all other known populations of A. trivirgatus. A. t. zonalis (Goldman, 1912) occurs in Colombia from the Panamanian border in the Pacific lowlands southward at least to the Raposo River just south of Buenaventura, the Uraba region and eastward to the Simi Valley, and possibly through the upper San Jorge Valley to the Puerto Valdivia region of northern Antioquia. This subspecies is completely homogeneous throughout its known range in Colombia as well as in eastern and central Panama. It resembles typical A. t. griseimembra in all respects except that the dorsal hair of its hands and feet appears dark brown or blackish. We do not relegate A. t. zonalis to a synonym of A. t. griseimembra as proposed by Hershkovitz (1949),
46 HERNANDEZ-CAMACHO and COOPER * E T A Aotus trivirgatus arise/membra A. t. zonalis A. t. lemurinus A. t. trivirgatus FIGURE 6 Distribution map of Aotus trivirgatus in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 47 because our examinations of literally hundreds of living specimens of the latter from the Barranquilla market have revealed only specimens with essentially light-colored hands and feet. A. t. lemurinus (I. Geoffroy-St. Hilaire, 1843) occurs in the three Andean mountain chains of Colombia at elevations from about 1,000-1,500 m upward to the timber line (3,000-3,200 m). It is a rather variable subspecies appearing quite often with two basic color phases, which can both be found in the same family group. One is decidedly grayish-brown, and the other is a richer, more reddish-brown in the upper parts. However, a range of intermediate coloration can be found. The underparts are always a rather dull yellow, indistinguishable from A. t. griseimembra and A. t. zonalis. The hair coat is extremely long and soft and is the most valuable distinguishing characteristic. The hands and feet of this species are remarkable in their color variation and lack of full correlation, even in indi- vidual specimens. Specimens examined from the west- ern Andes (around Cali) and most of the specimens from the central Andes have black-tipped hairs on the hands and feet (at least reaching the distal carpus and tarsus). A few specimens from the western Andes and a number of those from the central Andes appear variably grizzled in color on the metatarsal and metacarpal regions due to reduced extent of the dark tips, thus allowing exposure of the lighter hair bases. In the eastern Andes a full range of individual variation occurs with regard to this character, i.e., from extensive black hair tips to the very reduced tips typical of A. t. griseimembra. Some variation has also been observed between the hands and feet of the same individuals with regard to this characteristic. A. t. trivirgatus (Humboldt, 1812) occurs in the eastern plains and piedmont at least southward to the Guaviare River and probably extends continuously into the upper Amazon basin. It is characterized by a short-to-medium hair coat usually of a rather pure gray color with comparatively light-colored hands and feet due to rather short, dark, apical hair tips (as a rule only slightly more apparent than those of A. t. griseimembra). Although no specimens are available to us from the Guaviare to the Putumayo rivers, those from the Leticia area (as well as selected specimens from northwestern Brazil and southern Venezuela) have more richly golden-orange underparts in contrast with the more typically dull and lighter yellow under- parts of the specimens from the eastern plains and piedmont. Such specimens are also consistent with the characters claimed by Calvalho for the recognition of A. t. vociferans (Spix, 1823) from the Solimoes region (type locality Tabatinga, Brazil). However, more material from intermediate areas will be necessary to clarify the situation. It is noteworthy that in all subspecies of Colombian A. trivirgatus, coloration patterns of the head were found to be variable and totally unreliable taxonomic characteristics. HABITS: Aotus is typically encountered in pairs with one or two subadult offspring. On occasion associations of several adult pairs have been collected in the same nest. The nest sites of Aotus are located in tree hollows and/or in woody climbing vines in an accumulation of dry leaves and twigs. Such nests appear identical with those used by Potos flavus. Aside from the grossly similar colors and appearance of night monkeys, kinkajous, and olingos, the fact that they may all be encountered feeding in the same tree undoubtedly contributes to local confusion regarding their identities. Aotus become active just before dusk and have usually been observed in the field before midnight. This observation is not intended to preclude the possibility that another period of activity may begin in the morning before sunrise. In our experience, Aotus is seldom detectable from its vocalization. They have been encountered at almost every level beneath the canopy. An old observation of Goudot (in Alston, 1879, p. 14) states that Aotus returned regularly to the same area at night looking for guavas (Psidium guajava). STATUS: Aotus is presently one of the most suc- cessful primate forms in Colombia, due to its wide altitudinal and regional distribution, broad range of habitat (including rather degraded areas), and general lack of exploitation by man. The only region in which it is presently hunted extensively is the delta of the lower Cauca and neighboring middle Magdalena riv- ers. This trade has developed principally over the past 4 or 5 years in response to a growing biomedical research demand for this particular population of Aotus for use in malaria chemotherapy study (Cooper and Hernandez-Camacho, 1975). The effects on their local status caused by removal of several thousand specimens annually is not definitively known at this time. However, until reliable data become available on the trapping methods used, the specific areas of exploi- tation, the biological cycles of Aotus, and the size of the regional population, licenses for trapping and ex- portation will be carefully screened. Of historical interest is the fact that skins of Aotus, along with Lagothrix and Alouatta, have all been used locally as ornamentation on the brow bands of bridles for horses. 12. Callicebus moloch (Hoffmannsegg, 1807)âDusky titis.
48 HERNANDEZ-CAMACHO and COOPER 3. C. torquatus lugens 4. C. t. medemi FIGURE 7 Distribution map of the genus CaIIicebus in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 49 COMMON NAME: "Socay" or "Zocay" in Meta(C. m. ornatus) and in the upper Putumayo River region (C. m. discolor), probably of Quechua Indian origin; "Zogui-zogui" in the Amazon (C. m. discolor); also "MicoTocon," of Peruvian origin, in the Leticia area. DISTRIBUTION: (Figure 7) In the Department of Meta, C. m. ornatus (J. E. Gray, 1866) occurs in the piedmont to an upper altitudinal limit of about 500 m as well as in the adjacent lowland gallery forests. It is marginally sympatric with C. torquatus lugens along both banks of the Guayabero River at the southern extreme of its range. Its northernmost occurrence is the lower Upia River, a northern tributary of the Meta River. As pointed out by Mason (1966), C. m. ornatus is able to survive in even small isolated gallery forests of only a few hectares. C. m. discolor (I. Geoffroy-St. Hilaire, 1848) occurs in Amazon rain forest and has recently been collected on the southern bank of the Guamues River, a western tributary of the upper Putumayo. Its local existence in Colombia is limited to the region between the Guamues and the Sucumbios River, the next lower western Putumayo tributary. This population is proba- bly continuous with that along the southern bank of the Putumayo River in Ecuador and Peru and also is provisionally equivalent with the population that oc- curs in the trapezium area of the Amazonas Comisaria of Colombia between the Putumayo and Amazon rivers. HABITS: Both subspecies are usually encountered in groups of two to four animals. On one occasion (in June 1955) Hernandez-Camacho saw a total of seven individuals in a peninsula extending from a larger gallery forest on the Ocoa River, 7 km southeast of Villavicencio. Most sightings have been made in understory trees at heights of 3-8 m, and specimens have been collected on the ground (and crossing savanna in the case of C. m. ornatus). Callicebus is able to swim when forced and does so with a side-to- side undulation of the body and tail. C. m. ornatus often begins vocalization before sunrise, particularly on overcast days. It stops vocalizing somewhat sooner when the mornings are sunny than when they are over- cast. They have been observed in sloped piedmont forest with a canopy of no more than 10 m in the southern Macarena Mountains. C. moloch subspecies have not to our knowledge been observed in associa- tion with other primate species. Specimens of C. m. ornatus have produced stomach contents with a predominance of vegetable matter, including berries, seeds, and fruit pulp; leaf parts have never been observed. Also, most specimens (collected between January and March and in June and July) have included some Coleoptera and Orthoptera, as well as spiders and millipedes. STATUS: Clearing of level gallery forests and low piedmont areas for agricultural purposes in the Depart- ment of Meta northward from the Ariari River during the past 30 years has reduced by more than 50 percent the suitable habitat for this population of C. m. ornatus. Human population is continuing to grow in this area due to emigration from mountain regions to the west. However, Callicebus is seldom captured commercially or hunted for food in this region. It also displays an unusual ability to survive in isolated forests and in low mountainous areas. Its future is somewhat assured in the Macarena Mountains National Park (800.000 ha). As for the Colombian population of C. m. discolor, the region between the Guamues and Sucumbios rivers, now a major Colombian oilfield, is being rapidly set- tled, while the Colombian trapezium human population is increasing and some limited commercialization of the species is occurring, largely secondarily to food hunting. 13. Callicebus torquatus (Hoffmannsegg, 1807)â Widow monkeys and white-handed titis or collared titis. COMMON NAMES: "Macaco" in the Caqueta and Putumayo regions; "Macaco Caresebo" in the Guayabero River region; "Viuda" and "Viudita" in the Vaupes River and Orinoco River regions. DISTRIBUTION: (Figure 7)C. /. lugens occupies the area from the Guaviare and Guayabero rivers south- ward to the Putumayo and Colombian Amazon rivers, except for the habitats of C. t. medemi in the area between the upper Caqueta and Putumayo rivers. It also exists northward from the lower Guaviare River at least to the lower Tomo River in the Vichada Comi- saria. Within this entire area it is found in forest types ranging from gallery forests in the north to uninter- rupted rain forests in the Amazonian basin. C. t. medemi, most remarkable for its black hands, is known only from two localities in the vicinity of Puerto Leguizamo in the upper Putumayo basin. Two C. torquatus specimens from the southern bank of the lower Guamues River near Puerto Leguizamo have light-yellowish hands and distinctly ferrugineous un- derparts (including ventral tail surface). This pattern is in contrast with the blackish underparts of typical C. t. lugens and approaches the characteristics claimed for C. t. lucifer (Thomas, 1914), a subspecies not separa- ble from C. t. lugens according to Hershkovitz (1963). Recent collection (near Leticia) of a C. torquatus specimen with rather dull and somewhat brownish upperparts represents the only specimen with
50 HERNANDEZ-CAMACHO and COOPER E" .". '⢠â¢[ ^ -f FIGURE 8 Distribution map of the PIthecinae in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 51 adequate locality data known to us from the Colombian bank of the Amazon. This description is consistent with brownish individual varients of C. t. lugens seen almost throughout the range of the subspecies. HABITS: Observations have been limited to the northern extreme of C. t. lugens distribution (probably also the northernmost limit for the genus) between the Tomo and Tuparro rivers, as well as the area of C. t. lugens and C. m. ornatus sympatry on the upper Guayabero River. Observed group size has varied from two (Tuparro River) to a maximum of five (Guayabero River), and habitat has ranged from con- tinuous forests to narrow gallery forests. On one occasion a specimen was collected from a moving group of four after sunset (6:30 p.m.) in a wide gallery forest. On another occasion vocalization was heard at about 7:30 p.m. on the Guayabero River. Stomach contents (January to March) contained significantly less arthropod content than observed in C. m. ornatus in the same region (Guayabero River). Within the area of sympatry north of the Guayabero, only three groups of C. t. lugens were observed over a 3-month study period (as compared to nearly daily sightings of C. m. ornatus). On all occasions the C. t. lugens sightings were distant from any C. m. ornatus sightings. On the southern bank of the Guayabero during the same period, C. t. lugens were observed on the average of several times per week, while only one sighting (and collection) of C. m. ornatus was made. North of the Guayabero C. m. ornatus were observed in January and February to be carrying only older infants (with complete deciduous dentition) on occasions when parent-dependent specimens were seen or collected. During the same period, C. t. lugens on the southern bank were observed to be carrying considerably younger infants with adpressed hair and only deciduous incisors erupted in the specimens collected. STATUS: Considering the much greater range of C. torquatus subspecies in Colombia than of C. moloch subspecies, the status of the former is of less concern except in the more western areas of heavy settlement. Insofar as food hunting and commercial exploitation are concerned, the population pressures must be con- sidered as not significantly different from those of C. moloch subspecies. 14. Pithecia monachus (E. Geoffroy-st. Hilaire. 1812) âMonk sakis. COMMON NAME: "Volador" or "Mico Volador" in Colombia. DISTRIBUTION: (Figure 8) P. monachus (sub- specific designation problematic) occurs in the Colom- bian trapezius north of the Amazon and elsewhere in Amazonian Colombia north of the Putumayo River. Its northwestern limit is well documented as the piedmont of the eastern Andes as far north as the upper Caguan River to an elevation of 500 m. The northeastern extent of its range is uncertain but is at least to the southern bank of the Yari River and .below its mouth to the southern bank of the Caqueta River. Its habitat is always limited to primary, well-developed rain forest. HABITS: Monk sakis are usually observed as pairs or perhaps small family groups. They are high-canopy dwellers, shy and prone to rapid flight, which has resulted in practically nonexistent data on their be- havior and biology. Recent studies of primate ecology in the upper Putumayo River and Caqueta River basins by Kosei Izawa of the Japan Monkey Centre (see Izawa, 1972) will hopefully contribute to a better understanding of this and other primate species of the region. STATUS: Hunting Pithecia monachus for meat in Colombia is not known to us, nor have we seen dust brooms made from its tail as was observed by one of us (R. W. C.) in 1964 in Iquitos, Peru. Specimens, occa- sionally seen in dealers' compounds in Leticia, are exported from Colombia in very small numbers. Con- sidering its habits and requirements for undisturbed high rain forest, how Pithecia might be captured alive is unknown. Its population status in Colombia is undoubtedly better than that of such an obscure species as Cacajao melanocephalus, as monk sakis are relatively well known within their range in the Colom- bian Amazon basin. 15. Cacajao melanocephalus (Humboldt, 1812)â Black-headed uakaris. COMMON NAMES: "Chucuto" in the comisarias of Vaupes and Guainia; "Uacari" for occasional speci- mens arriving in Leticia (probably from the lower Rio Negro region in Brazil); "Piconturo" (Tukano Indians, fide Olalla from specimen labels). DISTRIBUTION: (Figure 8) Distribution is very poorly known. We have included the upper Rio Negro and the Inirida basins on the basis of reliable accounts of rubber hunters and the known occurrence of the species in the Amazonas Federal Territory of neighboring southern Venezuela. However, documen- tation exists for the Vaupes River and Apaporis River basins based upon preserved specimens from the mid- dle Apaporis near La Providencia (G. E. Erikson, personal collection; Universidad Nacional de Colom- bia, Bogota); from 10 specimens collected by the Olalla brothers on July 9, 1928, at "Tahuapunto" (Tauapunto), on the Brazilian bank of the Vaupes River at the border of Colombia (American Museum of Natural History); and from preserved Cacajao mandi-
52 HERNANDEZ-CAMACHO and COOPER Alouatta palliata A. seniculus FIGURE 9 Distribution map of the genus AIouatta in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 53 bles from contemporary Indian middens in the Mitu region of the Vaupes River. Dr. Frederico Medem (in lift.) has reported the occurrence of this species in the Cerro de las Pinturas and El Dorado Lake regions of the Vaupes Comisaria. The western extent of the probable range of C. melanocephalus in Colombia is documented by a specimen collected by Mr. Carlos A. Velasquez in 1942 during the Gilliard-Dillon expedi- tion of the American Museum (the specimen could not be located at the museum in August 1972) to the northern Macarena Mountains. In addition, Agapito, a famous chief of the Tinigua Indians, recounted in 1959 that he had killed a specimen many years earlier near La Angostura, on the southern bank of the Guayabero River, close to the southern tip of the Macarena Mountains. Accounts of a number of reliable infor- mants who are familiar with this species in the little- explored upper Vaupes basin are in agreement that it does not occur south of the Apaporis River. It appears likely that two subspecies of C. melanocephalus may exist, based on the plate pub- lished with Humboldt's original description (suggest- ing that the lower back and flanks are uniform in color with the hind limbs) and the description of Elliot (stating the back and sides to be reddish with a black admixture, as in the hind limbs). Specimens from the Maturaca River, a tributary of the Rio Negro, in northwestern Brazil near Venezuela, and from the Amazon Federal Territory of Venezuela, have a bright tawny back and flank color. On the other hand, all the Colombian specimens that have been examined, as well as a series from Tauapunto (Vaupes River), Brazil, and two specimens from the market in Leticia [probably originating in the area between the lower Caqueta River (or Yapura) and the lower Rio Negro on the northern side of the Amazon], are strikingly differ- ent in the markedly yellowish coloration of the sides and back without any obvious reddish tinge. If this distinction is valid, it appears that the correct sub- specific designation of the Colombian form would be C. m. ouakary (Spix, 1823). This subspecies would agree basically with the plate of Elliot (1913), although not with his verbal description. HABITS: It is reported by Frederico Medem, who collected the specimens from La Providencia in 1951, that C. melanocephalus is a high-canopy dweller, very shy, and found in groups of as many as 30 individuals. This account agrees in all respects with those of other informants and is consistent with the relatively large numbers of museum specimens collected in single localities on the same date. It is reportedly a highly prized food item by Indians of the region (e.g., Puinaves), possibly contributing to its apparent scar- city. Its food habits are essentially unknown. STATUS: The status of C. melanocephalus in Co- lombia must be considered very precarious. Its habits and apparently savory meat do not favor its coexis- tence with humans in areas of settlement and degraded forest. 16. Alouatta palliata (J. E. Gray, 1849)âMantled howlers. COMMON NAMES: "Mono Zambo" in northern Caribbean coastal Colombia; "Mono Negro" in the same area as well as along the Pacific coast (sometimes also applied to Ateles paniscus fusciceps)', "Mono Chongo" and "Chongon" in the lower Pacific coastal area adjacent to Ecuador. DISTRIBUTION: (Figure 9) The range of A. palliata in Colombia includes the entire Pacific coastal low- lands. It is absent from the coastal mangrove zone and probably from the adjacent natal zone (a swampy forest type dominated by "nato," Dimorphandra oleifera). It inhabits most of the morphologic classes of nonflooding forest, and its upper altitudinal limit in the piedmont zone is not known. In the northern junction between the Pacific and Atlantic oceans, it occurs in the Atrato River basin and in the Sinii Valley, where it is sympat- ric with A. seniculus. A specimen from Turbaco col- lected by Carriker in the early 1900's and reports by Dugand of the presence of the species in Los Pendales as late as the early 1950's (both localities in the Cartagena region) suggest that its northeastern exten- sion may even today be somewhat greater than we have chosen to indicate in our figure. However, recent field studies in the Maria Mountains in the depart- ments of Sucre and northern Bolivar have not revealed any evidence of A. palliata, although A. seniculus has been observed. In the Atrato River and Sinii River basins of the Caribbean coast it occurs from pluvial through rain forests to semievergreen forests. The Colombian population is referable to A. p. aequa- torialis (Festa, 1903). HABITS: Very little information is available on this species in Colombia, as most areas of its occurrence are remote and have not as yet been studied. STATUS: The status of A. palliata in Colombia is practically unknown, although meat hunting probably exerts more population pressure at this time than forest destruction, except in the area of its sympatry with A. seniculus, where intensive forest exploitation and destruction are occurring. 17. Alouatta seniculus (Linnaeus, 1766)âRed howlers. COMMON NAMES: "Mono," "Mono Colorado," "Mono Cotudo," and "Cotudo" in northern and cen- tral Colombia; "Roncador" in central Colombia;
54 HERNANDEZ-CAMACHO and COOPER "Araguate" in Arauca, and "Bonso," "Mono Ber- reador," or "Berriador" in Tolima and the eastern plains; "Bongo" and "Araguato" elsewhere in the eastern plains; "Mono," "Mono Cotudo," and "Cotumono" in the Amazon region; "Guariba Ver- melho" occasionally in the Leticia area (of Brazilian origin). DISTRIBUTION: (Figure 9) A. seniculus is absent on the Pacific coast of Colombia and the desert of the Guajira Peninsula and has not yet been reported from the Department of Narino (southwestern Andes near Ecuador). Otherwise it is present throughout the coun- try, except in nonforested areas and in mountainous regions above the cloud forest belt. Its upper altitudi- nal limits are known to be as high as 3,200 m locally in the central Andes. It is poorly known in the Atrato basin, but specimens have been collected on the west bank of the Atrato River near Unguia as well as on the east bank near Suatata. Its southern limits in this region are not known. Its habitat occupation is ex- tremely varied and includes the mangrove swamps of the Caribbean coast, the gallery forests of the eastern plains and other relatively dry regions, deciduous tropical forests, rain forests, cloud forests (including oak forests), and extremely small, isolated forest patches and second growth. Currently, the Colombian populations of A. seniculus are referred to the nominotypical subspecies of A. s. seniculus (Linnaeus, 1766). HABITS: The group size of this highly adaptive species ranges from 2 or 3 to 15 individuals with an apparent average of 6 to 8. The species has several remarkable and obvious behavior patterns. Groups can be heard at distances of 2 km or more giving their unique vocalizations from near dawn until about 8 a.m. on clear days, and they often begin again at 4:30 or 5 p.m., prior to retiring at dusk. On cloudy days vocali- zations may be heard at nearly any hour, and even on sunny days their calls may accompany the approach of a storm. Their basking behavior in exposed areas of the highest-available canopy (very often in defoliated Ceiba pentandra or other similarly emergent species) is also very striking. Considering their coat color (predominantly dark reddish) and the high ambient temperatures reached in nonshaded areas during mid- day, one wonders what body temperatures they might attain and what special cooling mechanisms they may possess beyond an extreme paucity of ventral hair. Their heavily pigmented skin undoubtedly protects them from ultraviolet radiation but should increase their absorption of heat while basking. When red howlers are disturbed, it is their habit to seek a high and concealed location in the canopy, where they remain immobile and very quiet. As has been reported in Alouatta palliata, red howlers will also "rain" excrement on observers below, particularly in areas where they are not actively persecuted. The diet of A. seniculus is varied in terms of plant species eaten, but generally consists of leaves and some soft fruits. Some of the principal dietary species include Anacardium excelsum, Cecropia spp.. Cassia moschata, Ficus spp., and Spondias mombin. Stomach contents are very finely masticated, making identification of food plant species more difficult than in most other Colombian primates. A. seniculus is not known to have interspecific associations with other primates in Colombia with the exception of Ateles paniscus belzebuth (Klein and Klein, 1973). In addi- tion, association between A. seniculus and A. palliata in the area of their sympatry is not known to us. Red howlers are able swimmers and have been known to cross bodies of water of 200 to 300 m in width. This ability may contribute to their success in the Cauca-Magdalena delta region and other such sparsely forested and seasonally flooded areas. The same adaptive significance may be seen in their ability and willingness to cross large expanses of treeless savanna on foot. In many areas of northern Colombia and the middle Magdalena Valley, A. seniculus is parasitized by botfly larvae of the genus Cuterebra. The preferred site is the ventral neck region, and large numbers of subcutaneous Cuterebra pupae can be ob- served at times in adult Alouatta. While filarid nematodes are frequent findings in the peritoneal cav- ity of red howlers from the north coastal and eastern plains regions of Colombia, they have not as yet been observed in howler populations from the middle Mag- dalena Valley. With regard to reproduction, A. seniculus is not a particularly seasonal breeder, as dependent infants have been observed in every month from January to August. Recently (May 1972), in southern Bolivar, collections included a female about 2 months pregnant and another carrying a 2-week-old baby in localities separated by only about 80 km. STATUS: It is difficult to generalize about the status of A. seniculus in Colombia. In most of the remaining forested lowlands, its call can still be heard daily. Forest destruction seems to be its principal enemy; but, due to its previously noted adaptive abilities, its continued presence is evident long after most other primate species have disappeared. Hunting of red howlers for meat is geographically variable, e.g., it is very seldom eaten in northern Colombia, but is occa- sionally eaten in the eastern plains by Indians and some immigrants from the mountainous interior. In the upper and middle Magdalena and Cauca valleys, it has long been prized as a food item by some segments of the human population, and today Alouatta is difficult to
THE NONHUMAN PRIMATES OF COLOMBIA 55 find in many suitable relic forests of this region. In these same interior areas, its hyoid bone is often used as a drinking device by campesinos who believe it to pos- sess therapeutic properties for curing goiters. The Spanish word for goiter is' 'coto,'' and from it is derived the regionally common name "cotudo" or "mono cotudo." In the Colombian Amazon the population status of Alouatta is more difficult to assess, since it seems not to be common around human settlements. In March 1972, during a 2-week survey of primary rain forest areas northwest from Leticia, it was never heard or seen. In regions of the country where the genera are both sympatric and hunted for food, Lagothrix and Ateles meat is always preferred to that of Alouatta. Howlers are not the basis of any commercial trade known to us in Colombia. Occasionally, live infant or juvenile specimens, produced by the hunting of adults, are reluctantly purchased by animal dealers, always at minuscule prices. Infrequently, A. seniculus skins are sold on a local basis, but hide hunting of this species is not presently common in Colombia. 18. Cebus capucinus (Linnaeus, 1758)âWhite- throated capuchins. COMMON NAMES: "Micro Negro" in the depart- ments of Cordoba, Sucre, and northern Bolivar; "Maicero Cariblanco," "Carita blanca," or "Mico" in the departments of northwestern Antioquia (Uraba region), Cordoba, Sucre, northern Bolivar, and Atlan- tico; "Machin" in the departments of Sucre and northern Bolivar; "Mico Maicero" in the Pacific coastal region. DISTRIBUTION: (Figure 10) Cebus capucinus oc- curs in Colombia from the Panama border south along the Pacific coast and western slope of the western Andes; Gorgona Island off the Pacific coast (Depart- ment of Narino); the upper Cauca Valley (very re- duced populations today); northwestern Department of Antioquia (Uraba region), and the departments of Cordoba, Sucre, northern Bolivar, and southwestern Atlantico eastward to the west bank of the lower Magdalena and the middle and lower San Jorge rivers. On the Pacific coast in the Department of Valle, C. capucinus is absent from the coastal alluvial plain (i.e., mangrove, natal, and lowland mixed forests), but appears in the piedmont. On Gorgona Island and along the Atlantic coast, the species occurs at sea level but is not known to inhabit mangrove forest. On the western slope of the western Andes, C. capucinus is found at elevations as high as 1,800 to 2,100 m; it is also likely that remaining populations on the eastern slope in the Cauca Valley may attain similar altitudes. Formerly, this latter population probably extended throughout the length of the western bank of the middle Cauca River to meet the populations of the upper San Jorge basin. Although C. capucinus is not known in the floodable (brackish) natal forests of the Pacific coast, it does occur in floodable (freshwater) forests elsewhere, e.g., the Atrato River basin. The species seems to prefer primary or advanced secondary forest, but it is also found in highly degraded remnant forests, includ- ing areas grazed by cattle in which some large trees and palms (particularly Scheelea magdalenica) sur- vive. Other than those already mentioned, the only forest types within its range in which C. capucinus is not known to subsist solely are those of xerophytic character. The taxonomic status of C. capucinus in Colombia suffers from lack of data in certain critical areas; however, three subspecies have been recognized, C. c. capucinus (Linnaeus, 1758), C. c. curtus (Bangs, 1905), and C. c. nigripectus (Elliot, 1909). The type locality of C. c. capucinus was fixed as northern Colombia by Goldman, based upon Simia hypoleuca (Humboldt, 1812), named from captive (nonpreserved) specimens seen at Puerto del Zapote northward from San Antero near the mouth of the Simi River in the Department of Cordoba. It is likely, how- ever, that the actual type locality was in the vicinity of Cartagena, because the species existed there during early colonial times (and was described in pre-Linnean literature), when Cartagena was the principal seaport within the range of the species. Although Hershkovitz (1949) first regarded Simia hypoleuca as a subspecies of Cebus albifrons, he later amended this interpreta- tion (1955) by proposing "that the name be disposed of as an unavailable synonym of C. albifrons." Cabrera (1957), however, recognized C. albifrons hypoleuca as a subspecies with synonomy, including C. malitiosus (C. a. malitiosus). A close examination of Humboldt's description of Simia hypoleuca conclusively shows it to be conspecific with C. capucinus populations with an overall brownish cast such as those of the upper San Jorge Valley. Also C. capucinus is known from the Sinii Valley (region of Monteria), as well as from the Tolii region not far from Puerto del Zapote in the coastal lowlands of the Gulf of Morrosquillo. As we will discuss later, such specimens can also be confused with particularly dark populations of C. albifrons from the middle Magdalena Valley. C. c. nigripectus was described from Las Pavas (1,400 m) on the western slope of the western Andes in the Department of Valle. Later, J. A. Allen (1916) referred additional specimens from the upper Cauca Valley to this subspecies. C. c. curtus is based on an adult pair collected on Gorgona Island. A comparison of specimens from northern and western Colombia,
56 HERNANDEZ-CAMACHO and COOPER 1. Cebus capucinus capucinus 2. C. albifrons malitiosus 3. C. a. cesare 4. C. a. (versicolor complex) 5. C. a. subspecies 6. C. a. albifrons 7. C. a. unicolor 8. C. a. yuracus FIGURE 10 Distribution map of the genus Cebus (part) in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 57 northwestern Ecuador, and eastern and central Panama with those of Gorgona Island shows no sig- nificant size differences in adults. Differences are demonstrable in color of the light parts (from nearly pure white to white strongly suffused with tawny ochraceous), the color of the chest and belly (black to black or dark drab strongly grizzled with light- yellowish white), the extension of a yellowish latero- ventral stripe to the genital region and/or medial thighs (as sparse yellowish hairs), the degree of development of yellow in the basal portion of the ventral tail hairs, and the development and coloration of the frontal tuft in females. However, these characters are so individu- ally variable as to make subspecific designation impos- sible based upon presently available material. Even the Central American populations examined by Hernandez-Camacho to date are subject to this same limitation. HABITS: The group size observed has been from about 6 to 15 individuals. As previously noted, they are found in a variety of forest conditions and can be seen from the highest canopy to very low shrublike trees and even on the ground. In this latter regard, they have been known to cross treeless expanses of several hundred meters to forage, for example, in cornfields. In degraded areas, it appears that palms such as Scheelea magdalenica are important in providing both food and shelter. Interspecific association of C. capucinus with other primates is not known in Colom- bia. As with other capuchins, they are omnivorous and are known to eat insects, eggs, small vertebrates, buds, leaves, berries, fruits, and (on Gorgona Island at low tide) even marine oysters, which are opened with the aid of stones (Dampier, 1697, quoted by Hill, 1960), in accounts agreeing with unpublished observa- tions of C. R. Carpenter in Panama (personal communi- cation, 1972). Specific seasons of reproduction for C. capucinus are not known. STATUS: The species has long been exploited lo- cally and exported as pets in small numbers in northern Colombia. It is also killed as an agricultural pest in corn-growing areas. However, as previously noted, C. capucinus is an adaptable primate, surviving in re- duced numbers even in badly degraded habitat. Its greatest threat, as demonstrated in the Cauca Valley and the northern Department of Bolivar, is the com- plete destruction of forests. 19. Cebus albifrons (Humboldt, 1812)âWhite-fronted capuchins. COMMON NAMES: "Mico," "Macaco," and "Cairara" in the Leticia region; "Mico Tangue" in the Caqueta River basin; "Maicero," "Maicero Cari- blanco," and "Mico Cariblanco" throughout its non- Amazonian range in Colombia; "Carita Blanca" and "Mico Bayo" in the departments of Magdalena, Cesar, and southeastern Bolivar; "Machin" in the middle Magdalena Valley; "Ouavapavi" by the Mai- pures Indians, according to Humboldt. DISTRIBUTION: (Figure 10) Cebus albifrons has a discontinuous (and in many areas unstudied) distribu- tion in Colombia. It occurs in the departments of Magdalena, Cesar, southern Guajira, southeastern Bolivar (including Mompos Island), eastern Sucre be- tween the San Jorge and lower Cauca rivers, Antioquia in the lowlands east of the Cauca River, the eastern lowlands of the Magdalena River basin, the lowlands of Santander, western Boyaca, eastern Caldas, western Cundinamarca and northern Tolima, the Catatumbo basin in North Santander Department, and the pied- mont forests of the Comisariaof Arauca. In the eastern plains, it is known only from the eastern Comisaria of Vichada (the Bita River near Puerto Carreno, the lower Tomo River, and Maipures, a former mission about 3 km southeastward from the confluence of the Tuparro and Orinoco rivers just above the Maipures rapids on the Colombian bank of the Orinoco). It is absent from the eastern Comisaria of Arauca, the Casanare plains of eastern Boyaca, the Department of Meta, and the northwestern Comisaria of Vichada. It is possible that the range of C. albifrons extends to the southern Comisaria of Vichada and to the comi- sarias of Guainia and Vaupes, since it has been re- ported from the southern bank of the lower Guayabero River by local informants (L. Klein, personal commu- nication, 1972) as well as to the forests of the northern bank of the Guaviare River in the Department of Meta. Yet, it is absent from the Ariari and Guayabero basins in Meta. It may also be present in the southeastern Intendencia of Caqueta and the northern Comisaria of Amazonas and is known in all the region south of the Caqueta River. Its altitudinal range is from sea level on the Caribbean coast to elevations of 1,500-2,000 m in the Department of Tolima. For detailed descriptions of the following subspecies, the reader is referred to Hershkovitz (1949). C. a. malitiosus (Elliot, 1909) is a well-defined subspecies inhabiting the deciduous and humid forests of the northern slopes of the Santa Marta mountains at least as high as 1,300 m. The eastern and southern limits of this population are not well defined. The subspecies is characterized largely by its rather dark- brownish overall coloration and rather light-yellowish shoulders. C. a. cesarae (Hershkovitz, 1949) is a very light- colored and well-defined subspecies occurring in the Department of Magdalena southward from the Cienaga Grande (it has been collected in brackish water man-
58 HERNANDEZ-CAMACHO and COOPER groves, Conocarpus erecta and Laguncularia racemosa, on the Palenque River) and in the lowlands of the Department of Cesar from the vicinity of El Banco and Tamalameque northward to the deciduous and gallery forests of the Rancheria River, in the southern Guajira Department. C. a. versicolor (Pucheran, 1845) is a "complex," including dark-colored populations extending along the Perija Mountains in the southern Department of Guajira and in the Department of Cesar and occurring in the middle Magdalena Valley, including Mompos Is- land; in the southeastern Department of Sucre be- tween the San Jorge and lower Cauca rivers; in the northern Department of Antioquia eastward from the Cauca River, including the Nechi River Valley; and in the humid lowlands of the Catatumbo basin in the Department of North Santander. Hershkovitz (1949) determined that three subspecies occupied this com- plete range: C. a. leucocephalus (Gray, 1865) from the eastern bank of the Magdalena River and in the Catatumbo region, characterized by a dark brown colgr and reduced reddish hues in the lower limbs; C. a. pleei (Hershkovitz, 1949) from the Norosi region in the southwest Department of Bolivar, characterized by reddishness, particularly in the limbs; and C. a. versicolor, recorded from the Department of Tolima and reminiscent of C. a. pleei but with overall light red coloring. However, in 1957 collections in the region of Barrancabermeja, on the eastern bank of the middle Magdalena River in the Department of Santander, demonstrated the presence of typical C. a. pleei and C. a. leucocephalus in localities only 10 km apart. Sub- sequent collections and field observations in the same region in 1958 revealed individuals typical of both supposed subspecies in the same groups as well as intermediate specimens approaching C. a. versicolor. As in many other populations of C. albifrons sub- species, a range of variation in shades of color from rather dull ("dark phase") to somewhat brighter ("light phase") individuals exists. This evidence strongly suggests that the "dark phase" (C. a. leucocephalus) and "light phase" (C. a. pleei) are extremes of the intermediate C. a. versicolor. This possibility must be further evaluated with specimens collected from several critical areas, particularly the western bank of the middle Magdalena River and the Lake Maracaibo drainage of Colombia. There is a pale and dull-colored population ofCebus albifrons in the piedmont forests of western Arauca Comisaria, the northern tip of Boyaca, and the eastern tip of North Santander. The available specimens from the Colombian bank of the Arauca River may repre- sent "light phase" animals of C. a. adustus, known from the Lake Maracaibo region of Venezuela. This possibility suggests the presence of C. a. adustus in the upper Apure basin of Venezuela. C. a. albifrons (Humboldt, 1812) does not have a preserved type specimen. Judging from the original description, it was duller and less yellowish ("dark phase") than specimens recently collected in eastern Vichada Comisaria near the type locality. The type specimen, according to Humboldt. had a dark-colored tail tip, unusual for C. albifrons and not characteristic of any recent specimens. The present subspecies is very light-colored with a strikingly yellow tone, resembling the population from Arauca but yellower, with an orange-yellow (not brown) dorsal surface on hands and feet. C. a. unicolor (Spix, 1823) is also a light-colored subsubspecies with a yellowish cast. It occurs in Amazonian Colombia south of the Caqueta River, except for the area of southwestern Putumayo Comi- saria south of the Guamues River, which is occupied by a light, dull-brownish population referable to C. a. yuracus (Hershkovitz, 1949). There is a great scarcity of Amazonian study specimens, and at this time the possibility exists that C. a. unicolor could be a synonym of C. a. albifrons. Selected specimens from the Amazonas Federal Territory of Venezuela are virtually indistinguishable from C. a. albifrons, but the population has been referred to C. a. unicolor by Hershkovitz (1949). During the past 2 years, several extremely dark- brown specimens of Cebus albifrons from the Barran- quilla market with reported provenience in the middle San Jorge Valley have been preserved. At first inspec- tion it is difficult to tell whether such specimens are referable to C. capucinus or C. albifrons. Inter- mediate characters include higher or more-recessed dark skull cap, more distinct baldness of the associated white forehead area, noticeably lighter shoulders and upper arms (all features suggesting the predominant pattern of C. capucinus), and an overall darker and uni- formly colored body, more so than even the extremely dark individuals of C. a. malitiosus. By the same token, some C. a. versicolor (pleei-lype) seen in the Magangue market and probably captured in the lower Cauca River region also show the above similar ten- dencies, except there is no noticeable increase in overall dark pigmentation. Based on these observa- tions, as well as the several dark-brown C. capucinus, "intermediate-like" specimens of unknown northern Colombian provenience, it seems quite likely to us that further studies in the San Jorge Valley and other zones at the interface of C. capucinus and C. albifrons distribution may ultimately show these forms to be conspecific. Another zone critical to such an analysis is the area in northwestern Ecuador in which C. a.
THE NONHUMAN PRIMATES OF COLOMBIA 59 equatorialis (J. A. Allen, 1914) and C. capucinus are both known to occur, but where sympatry or intergra- dation have yet to be determined. HABITS: In general, the habitat requirements of C. albifrons do not differ from those of C. capucinus. The same can be said for most other species characteristics of which we are aware, with the possible exception of maximum group size. It is our impression that groups of 20-30 C. albifrons are common in suitable habitat. In August 1957 Hernandez-Camacho observed a con- gregation of at least several hundred C. albifrons actively foraging on the leguminous fruits of Inga sp. at about 5:30 p.m. within a distance of about 300 m on both sides of the recently constructed road from El Centra to Quebrada Lizama in the municipality of Barrancabermeja. In this region another seemingly preferred foraging area is "guamera," a secondary forest about 15 years old with an 8-12 m canopy dominated by "guamo," also Inga sp. Additional important food items include the "guayabo de pava" (Bellucia axinanthera), which fruits throughout the year, and the continuously available food-bearing "pal- michales," large homogeneous associations of pal- miche (Euterpe sp.) common in floodable areas and boggy soils. STATUS: In the middle Magdalena Valley and the Amazon region, C. albifrons is hunted for food. Throughout their range they are persecuted in areas where they forage for corn. Some commercialization of the species occurs in the Amazon region and in the north coastal Cauca River and Magdalena River delta regions. However, C. albifrons is a rather cautious and adaptable species. It survives in even very reduced and degraded habitat and does not make its presence obvious by conspicuous vocal or visual displays. The greatest danger to the survival of many local popula- tions is the complete destruction of habitat for pur- poses of cattle-grazing and farming. 20. Cebus apella (Linnaeus, 1758)âBlack-capped or tufted capuchins. COMMON NAMES: "Maicero," or "Mico Maic- ero," is the most widespread name in Colombia; for tufted adults, "Mico" or "Maicero Cornudo," "Cachon," or "Cachudo"; in Leticia, "Mico" or "Macaco Prego"; "Aggue" (Tukano Indians, fide Olalla from specimen tickets). DISTRIBUTION: (Figure 11) Cebus apella occurs throughout the Colombian Amazon and in the entire lowlands and piedmont (to at least 1,300 m) of eastern Colombia. It also occurs in the upper Magdalena Valley in the Department of Huila to an elevation of 2,700 m (in the region of San Agustin) and in the region of Tierradentro in the Department of Cauca at altitudes of up to 2.500 m near Inza. A specimen in the British Museum labeled "Tolima Mountains" and collected prior to 1900 by White may have been collected in the Huila Department, as at that time the "State" of Tolima also included Huila (e.g., specimens of Lago- thrix lagorricha lugens collected by White and labeled "Tolima" included a latitudinal designation, 2° 20' N, which places them in the present-day Department of Huila). It is possible that C. apella and C. albifrons may have sympatric marginal contact in the Tolima Department. In the eastern plains, C. apella is found in virtually every type of humid forest from gallery to palm to rain forest and in both seasonally flooded and nonflooding habitats. They are also found in broken or isolated forest, in second growth, and in mountain areas (in cloud forest). They also cross open ground in passing from one forest segment to another. Subspecific identities of Colombian populations of C. apella are yet to be determined. If one follows closely the work of Hill (1960), it is necessary to accept a number of sympatric subspecies. Apparent taxonomic problems may result from well-known sex- ual, age-class, and individual variations in external features characteristic of this species. Until a thorough review of the situation can be accomplished, we con- sider it best to accept the earliest valid subspecies for northern South America, C. a. apella (Linnaeus, 1766), to represent all Colombian forms. We remain uncon- vinced, on the basis of examination of over 120 widely distributed museum specimens, that Cebus apella north of the Amazon from Colombia eastward exhibit phenotypic distinctions that would justify the recogni- tion of more than one subspecies throughout this large region. HABITS: Group size is variable, but 15-20 (at times 30) individuals seem about average for continuous forest. In isolated forest or second growth, 3-5 adults may form a group. Cebus apella is well known for its interspecific association with Saimiri sciureus (Thorington, 1967; Baldwin and Baldwin, 1971; Klein and Klein, 1973), and association with Ateles paniscus belzebuth has been observed in forests at the southern end of the Macarena Mountains (Hernandez- Camacho, unpublished data, 1959; Klein, personal communication, 1972). They are very noisy and, ex- cept at midday, can be easily sighted or heard in undisturbed lowland forest. In areas where they have not been hunted or otherwise molested, they exhibit considerable curiosity, often approaching to within a few meters of an observer. In addition, some adults have been seen by Hernandez-Camacho to throw feces and urine down toward an observer. When they travel in mature forest they seem to prefer a high route beneath the upper canopy. It appears that most birds leave an
60 HERNANDEZ-CAMACHO and COOPER ow. FIGURE 11 Distribution map of Cebus apeIla in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 61 area when a party of C. apella passes through. They have been observed by Hernandez-Camacho to ac- tively grasp at species such as Ara macao and Har- pagus bidentatus. On the other hand, they exhibit great fear, as evidenced by noisy flight, of such eagles as Harpia harpyja and Spizaetus ornatus. On one occasion a tayra (Eira barbara) was collected while in rapid pursuit of a troop of C. apella through the forest canopy. All large groups seem to be dominated by a large older male with younger males and juveniles occupying peripheral positions around the more cen- tral females and infants (i.e., an "age-graded male troop." Eisenberg et al., 1972). Small infants have been observed at almost every month of the year, suggesting that the species is not a seasonal breeder. In terms of food habits, they seem to eat largely fruits, including those of several palms (e.g., Euterpe sp., Jessenia polycarpa, Scheelea sp., and Syagrus inajai), of wild figs (Ficus spp.), of Araceae (Monstera sp.), wild plums (Spondias mom- bin), and berries of Isertia sp. (a shrub common in second growth) and ofPourouma sp. On occasion they have been observed to collect fallen fruits from the ground. They are also eager eaters of small Coleoptera and grubs, and it seems that they would eat any small vertebrate that they could catch. They are known for their agricultural thievery, but prefer immature corn to other fare, such as cultivated fruits. STATUS: The greatest pressures result from habitat destruction and from active hunting of C. apella as agricultural pests and in some areas as a source of meat. C. apella is a hardy and adaptive species with an extremely wide range in Colombia. Its numbers are being reduced in the upper Magdalena Valley and the major areas of immigration and settlement in the eastern plains. It is not a highly commercialized species in Colombia, the Amazon region providing the few specimens that are exported. 21. Lagothrix lagotricha (Humboldt, 1812)â Humboldt's woolly monkey. COMMON NAMES: "Mono Choyo," "Choyo," and "Choro" in the northern piedmont (Comisaria of Arauca and departments of Boyaca and Meta); "Churuco" or "Chuluco" in the upper Magdalena Valley and the comisarias of Caqueta, Vaupes, and Amazonas; "Barrigudo" in the Amazon. DISTRIBUTION: (Figure 12) Two apparently iso- lated populations, marking the northernmost extension of the genus, have recently been discovered in the upper San Jorge Valley in the Department of Cordoba, around San Pedro, and in the Serrania de San Lucas, in southeastern Bolivar (see Green, 1976). To date only juvenile specimens have been examined, and the subspecific allocation is difficult. These specimens have been very black in color with remarkably long and dense hair reminiscent of mountain populations of L. l. lugens. The known populations referable to L. /. lugens (Elliot, 1907) are confined almost entirely to Colombia. The exception is a recently discovered population in the area of dense piedmont forest in the Sarare River drainage of the State of Apure, adjacent to the Colom- bian border, in Venezuela. From this location, the distribution of L. /. lugens continues south and west to the eastern slopes of the Andes, including the pied- mont and neighboring wide gallery forests. Its range extends easterly in the plains along the Ariari River to include the Macarena Mountains. It is replaced by L. l. lagotricha on the northern bank of the lower Guayabero River. The subspecies of L. l. lugens extends westerly across the eastern Andes to include the upper Magdalena Valley, reaching an upper altitudi- nal limit of about 3,000 m. It is known as far north as the upper Saldana Valley on the west side of the Magdalena Valley in southern Tolima Department. On the east side of the Magdalena River, it occurs as far north as the mountains of the Pandi area, in southern Cundinamarca Department. As indicated in the dis- tribution map for this species, its existence is open to question along the eastern slope of the central Andes, which is a possible historical or present corridor to the recently discovered northernmost populations in southeastern Bolivar. The southernmost known occur- rence of L. /. lugens is in the area of Mocoa, in the piedmont forest of northwestern Putumayo Comisaria. As the adjacent southern piedmont region of the Putumayo basin and the neighboring Aguarico River drainage of Ecuador provides similar habitat and is poorly known, it is possible that L. l. lugens will yet be found in Ecuador. The populations of L. /. lagotricha in Colombia are continuous with those of northwestern Brazil, extreme northern Peru, and extreme northeastern Ecuador. Its northernmost extension in Colombia is at least to the northern bank of the Guaviare River of the Orinoco drainage. Its northwestern extension meets the south- eastern extension of L. /. lugens approximately at the junction of the piedmont forest with lowland rain forest. Questions of subspecific sympatry are prob- lematic, as observations in the region of Florencia and the Orteguaza River basin in the Intendencia of Caqueta suggest a gradual rather than an abrupt inter- gradation (represented by a changing frequency of buffy brown hair coats characteristic of typical L. l. lagotricha with the more grayish-to-blackish coat characteristic of typical L. l. lugens). Some individuals appear approximately intermediate, but specimens
62 HERNANDEZ-CAMACHO and COOPER Lagothrix lagotncha subspecies CMC FIGURE 12 Distribution map of the genus Lagothrix in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 63 showing all characteristics can be found in the same locality, if not in the same groups. The habitat of this species is always some type of humid forest, e.g., the gallery and palm (Mauritia flexuosa association) forests of the eastern plains, rain forest (seasonally flooded or not), and cloud forest. It can be observed in undisturbed primary forest or somewhat degraded forest, but we have never seen this species in second growth or young secondary forest. HABITS: Woolly monkeys have been seen in groups of four to six individuals (which have always included a rather old adult male) in piedmont forests of eastern Colombia and in the upper Magdalena Valley. They seem to be seen most frequently just beneath the upper canopy and sometimes in emergent trees. When disturbed by observers, they tend to move away from the area as opposed to hiding, a tactic more common in Alouatta. They also may void excreta and tend to use their hands in throwing it toward an observer. Adult males give threatening displays including branch shaking and grunting vocalizations, often with canines exposed. The diet of Lagothrix, insofar as we have observed, includes only vegetable matter but not leaves. In L. l. lagotricha from the eastern plains, stomach contents have always contained quantities of various palm fruits, particularly Mauritia flexuosa, Attalea regia (syn.: Maximiliana elegans), andJessenia polycarpa. During the fruiting season of Jessenia (February- April) and Attalea (June-July), areas of these palm populations are good places to encounter Lagothrix. A recently collected adult male specimen of L. /. lugens from the upper Magdalena Valley at more than 2,000-m elevation had stomach contents that included fruits of a Clusiacae (Clusia sp.), Ficus sp., and an unidentified species (probably a Sapotaceae). The morphology of its stomach (long and cylindrical with at least three distinct mucosal regions) was much like that of Ateles and quite different from that of Alouatta. Interspecific associations with other primates are not known in Colombia. In fact we have the distinct impression that Lagothrix and Ateles are largely mutu- ally exclusive. They do at times occur in the same region and in apparently identical habitat but have not been observed by Hernandez-Camacho closer to- gether than distances of 5 km or more. In the Caqueta and Putumayo regions, two species of eagles are known as "Aguilas Churuqueras" (Harpia harpyja and Morphnus guianensis) due to their reputation as Lagothrix predators. Carlos Lehmann (1959) has also reported the predation of Lagothrix by Isidore's eagle (Oroaetus isidori) in the Moscopan region near the Purace National Park in the central Andes. L. l. lagotricha collected from the eastern plains of Colom- bia by Hernandez-Camacho have always had an abun- dance of filarid nematodes in the abdominal cavity. STATUS: In total, L. lagotricha is probably the most persecuted primate species in Colombia. Its meat is highly prized as food by some people from the mountainous interior of Colombia (many of whom have emigrated to the eastern plains) and particularly by Indians and mestizos in the Amazon (see Izawa, 1972). The meat of L. l. lagotricha is particularly esteemed at the peak of the fruiting season of the palms on which they feed because of the subcutaneous fat the monkeys accumulate (see also Hershkovitz, 1972). As Lagothrix is known only in primary forest, its future is also closely linked to the survival of undis- turbed forests. Originally, as an adjunct to meat hunt- ing, immature Lagothrix were maintained and often sold as pets. Today it is likely that the situation is reversed in some areas of the Colombian Amazon, with meat being an adjunct to the collection of immature Lago- thrix for sale as pets. Recently, at the airport of Barranquilla, a man from Puerto Leguizamo, on the Putumayo River, was observed with three juvenile woolly monkeys and a few other small mammals and reptiles on his way to sell them to a local animal dealer. He had started his journey from Puerto Leguizamo by plane less than 12 hours earlier and would return home with a considerable profit (even after absorbing the price of round-trip plane fare), largely due to receipts from the three young Lagothrix. Of the more than 300 young woolly monkeys legally exported from Colom- bia in 1970, the majority probably came from the Putumayo and Caqueta regions. The Lagothrix ex- ported from Leticia are seldom, if ever, captured in Colombia; most come from Amazonian Brazil and Peru and represent L. l. poeppigi (Schinz, 1844). Recent field studies in the lower trapezium region revealed no populations of Lagothrix and no Colom- bian inhabitants of the area who knew where they might exist. That such reduced or absent populations have not always been the case is demonstrated by the report of Bates in 1863 that the Ticuna Indians of Tabatinga (a Brazilian town very near Leticia on the same bank of the Amazon) killed at least 1,200 woolly monkeys a year for food. 22. Ateles paniscus (Linnaeus, 1758)âSpider monkeys. COMMON NAMES: "Marimonda" and "Mari- munda" in northern, central, and eastern Co- lombia and in the Comisaria of Guainia; "Marimba" in the eastern plains as well as central and Amazonian
64 HERNANDEZ-CAMACHO and COOPER A teles paniscus belzebuth A. p. hybridus A. p. robustus A. p. grisescens C-Hf FIGURE 13 Distribution map of the genus AteIes in Colombia.
THE NONHUMAN PRIMATES OF COLOMBIA 65 Colombia; "Choiba" in the Department of Antioquia and the middle Magdalena Valley; "Braceadora" in the eastern plains; "Coata" in the Leticia region (Brazilian derivation); "Maquizapa" in the Putumayo and Leticia regions (of Keshwa origin); "Mono Negro" in the Department of Choco and on the Pacific coast; "Mica" in the Department of Bolivar; "Zamba" in the Department of Antioquia (for A. p. robustus). DISTRIBUTION: (Figure 13) In Colombia, Ateles is found from sea level to maximum elevations of 2,000- 2,500 m, the latter locally on the western slope of the western Andes and in southern Santander Depart- ment. It also occurs to 1.400 m in the Perija Mountains in the departments of Cesar and Guajira and to 1,300 m on the eastern slope of the eastern Andes in Boyaca; otherwise, it is generally encountered at lower than 800 m. Spider monkeys are notably absent in the dryer districts of the Caribbean coast, the northern and western wet slopes of the Santa Marta Mountains, the upper Cauca and Magdalena valleys, large areas of gallery forest in the northeastern plains, and locally over considerable extensions of the Amazon basin. A. paniscus belzebuth (Humboldt, 1812) in general has a naked face with black skin, sometimes slightly depigmented around the orbits and muzzle. It usually has a rather conspicuous submalar stripe of white hair that terminates in front of the base of the ear, as well as an often conspicuous triangular white-to-yellowish frontal hair patch, which may be infused with dark hairs (even to the point of disappearance). The upper parts, including the forelimbs and hands and most of the tail, are usually black. The tail tip is variably yellowish to black, although the undersurface is often buffy-yellowish to orangish, as are the underparts in general, usually including most of the leg. The feet are always black, as well as the hair covering the knees, which may also extend variable distances onto the anterolateral surfaces of the thigh and lower leg. A remarkable variation in many of these color characters may occur locally and individually. The range of A. p. belzebuth includes the Amazon lowlands northward at least to the Guaviare River and also the Macarena Mountains and the piedmont forests (locally to 1,300 m) northward to the Upia River drainage in southern Boyaca Department. The sub- specific identification of populations occurring in the piedmont north of the Upia River to the border of the Comisaria of Arauca (where A. p. hybridus occurs) is not yet known. In addition, A. p. belzebuth was recorded by Humboldt and Bonpland (1812) from both the Atures and Maipures rapids of the Orinoco River. As they did not mention on which bank of the river the species was observed or collected, it is uncertain whether this record is referable to Venezuela or to Colombia territory in the present Comisaria of Vichada. Furthermore, Brother Apolinar Maria (1913) re- corded a specimen from "Tolima" in the upper Mag- dalena Valley that he designated as Ateles "beel- zebuth," The specimen unfortunately is no longer available, but we are somewhat assured by his report in the same paper of A. "hybridus" from farther north in the Magdalena Valley (Minero River region of western Boyaca Department) that the former was a distinctly darker specimen and possibly identical with A. p. belzebuth of the eastern piedmont of the eastern Andes. Although no other records are known to us of A. p. belzebuth west of the eastern plains and pied- mont, a precedent exists for such "passage" into the upper Magdalena Valley in the cases ofCebus apella, Lagothrix lagotricha lugens, and Saimiri sciureus caquetensis, as well as several other species of ar- boreal and terrestrial vertebrates. A. paniscus hybridus (I. Geoffroy-St. Hilaire, 1829) in general also has an approximately naked face as in A. p. belzebuth but usually does not have conspicuous depigmented areas in adults. The submalar stripe and the frontal triangle are also variably present but in general not as conspicuously so as in A. p. belzebuth, The color of the back ranges from light grayish-brown (or avellaneous) to rather rich brown. The head, neck, forelimbs, and upper surface of the tail are invariably darker than the back and in some specimens approach blackish-brown. The hind limbs are usually lighter and similar in color to the back, with the exception of typically darker coloration over the knees. In the darkest specimens known, referable to the population of the western bank of the Magdalena River, the entire lateral surface of the hind limb is dark brown and relatively Uniform with the color of the back and feet. The underparts of A. p. hybridus vary from dirty-white to buffy, not contrasting markedly with the flanks in populations east of the Magdalena River. The ventral hair of the tail ranges from yellowish to buffy light brown. The contrast of the darker color of the ex- tremities, tail, and head with the lighter back is variably evident. The eye color (iris) of A. p. hybridus is usually light brown but is sometimes grayish-blue. Some specimens from the Cesar Valley have an overall bleached coloration, appearing light buffy-gray. An extremely light, uniformly buffy-colored specimen with bluish eyes was also seen in captivity by Hernandez-Camacho in Cano Muerto in the middle Magdalena Valley. In addition, a completely white specimen with pink skin and light blue eyes has been exhibited at the Barranquilla Zoological Garden and variously reported in popular literature by R. Tinoco, the Zoo Director.
66 HERNANDEZ-CAMACHO and COOPER A somewhat remarkable fact is that all known specimens west of the Magdalena River are considera- bly darker than those in essentially identical habitat east of the river. The range of A. p. hybridus includes the eastern bank of the lower Cauca River basin, the departments of Magdalena and Cesar (northward to the southernmost slopes of the Santa Marta Moun- tains), the northernmost extension of the Perija Moun- tains in the Department of Guajira, and the middle Magdalena River region at least to the northern de- partments of Caldas and Cundinamarca. There are also at least two additional populations in Colombia on both flanks of the eastern Andes Mountains at the border with Venezuela; one occurs in the Catatumbo River basin of North Santander Department and the other in the northeastern piedmont in the Comisaria of Arauca. A. paniscus robustus (J. A. Allen, 1913) also has a black face that is essentially hairless, at times with some depigmentation of the orbits and nasal orifices. The white submalar stripe is either entirely missing or represented by only a few scattered white hairs. The triangular frontal patch is usually entirely absent but apparently is present in some local populations. The hair coat is completely black, sometimes with very slight brownish shades on the head and back. Scat- tered yellowish hairs can often be found on the ventral surface of the body and inner surface of the thighs. One captive specimen, reputed to be from the Atrato River basin, was described as having reddish under- parts and given the name of A. "rufiventris" (Sclater, 1872). Eye color is usually dark brown for A. p. robustus, but rarely may be lighter to even bluish. The distribution of the subspecies covers the entire Pacific lowlands (except the region around Jurado in northwestern Choco Department); the Uraba region of northwestern Antioquia Department; the depart- ments of Coâ¢rdoba, Sucre, and northern Bolivar eastward to the lower Cauca River and along the western bank to south-central Antioquia (the most southerly record is from Concordia). In recent times the northernmost limit was the southern bank of the Canal del Dique in the Cartagena region; however, it probably formerly occurred northward to the Pendales region, where some luxurious hygrotropophytic forest still survives. This subspecies occupies the greatest range of habitat types of all of the Colombian forms of Ateles, from hygrotropophytic through pluvial to cloud forests. The southernmost specimen known in Colom- bia is from Barbacoas in the Department of Narino. Interestingly, it does not show any of the brownish coloration typical of the Ecuadorian population of A. p.fusciceps (a form that may yet be found in Colombia in the Mira River valley of Narino Department at the Ecuador border). Of historical interest is the fact that most old records of A. p. robustus are reported under the species designation of A. ater (F. Cuvier, 1823). A. p. grisescens (J. E. Gray, 1866) is characterized by a brownish or rusty-colored back (with black hair tips) and by completely black head, limbs, and tail. It could well be considered a traditional form between the bright yellowish-colored A. p. geoffroyi group of middle America and the largely jet black A. p. robustus of southeastern Panama and adjacent Colombia. A. p. grisescens is known in Colombia only from the vicinity of Jurado very near the Panamanian border on the Pacific coast. It is undoubtedly restricted by the Baudo Mountains to a narrow coastal strip that may extend as far south as Cabo Corrientes. From the above descriptions of Colombian Ateles populations, the fact that we consider all Ateles to be conspecific, as well as some of the logic underlying our conclusion, should be apparent. This position is also consistent with that of Hershkovitz (1969, 1970) and underscores the fact that all forms of the genus are allopatric with fundamental differences only in color. The characters of several supposedly distinct "species" recognized by Kellogg and Goldman (1944) show intergradation, e.g., Ateles geoffroyi intergrades towards Ateles fusciceps through A. g. grisescens; a specimen from Catival, San Jorge River, Colombia, representing A. fusciceps robustus shows a decided tendency toward A. belzebuth hybridus with a strong admixture of light-colored hairs on the back; A. belze- buth belzebuth has considerable individual and local variation in the development of dark brownish or black areas of the upperparts; and Bartlett (cited by Elliot, 1913) has recorded a specimen of A. paniscus chamek from Chamicuros in the Huallaga River of Peru, "which had the thighs and belly very gray or grizzled," thus approaching A. belzebuth, HABITS: Spider monkeys are known to occur in social groups of variable size. Some recent information (Klein, 1971a; Klein and Klein, 1976) suggests that groups of 15-30 may break into subunits for much of the day and regroup later in the afternoon. In remnant or degraded forests, it is not uncommon to find smaller groups that appear to consist of at least one adult pair and associated offspring and/or a few other individu- als. Activity can be observed from very early morning throughout the day to sunset. The animals are usually observed at middle or high-canopy levels, but it is not uncommon for them to forage on the ground in some circumstances. At times, when disturbed, adults are known to drop or throw twigs and small branches and to vocally threaten an observer. They are not as difficult to follow in level, open-floored forest as one might imagine, as they are exceptional neither in speed
THE NONHUMAN PRIMATES OF COLOMBIA 67 nor stealth. They tend to occur in more mature forest and are not usually found in historically isolated small forests (eastern plains) as are Cebus, Saimiri, and Callicebus at times. On the other hand, in north-coastal Colombia Ateles is often encountered in recently isolated, relatively mature forest tracts. They are essentially vegetarian and can become quite fat during the fruiting season of some plants (particularly a palm called "seje," Jes- senia polycarpa). Ateles also favors fruits of the genus Ficus and Brosimum, as well as Spondias mombin, which are often swallowed with little or no mastica- tion. As previously noted, their stomachs are remark- ably large and contain several distinct mucosal re- gions. In the middle Magdalena Valley, A. p. hybridus has been seen with newborn infants in August, while in the Macarena Mountains A. p. belzebuth has been observed with the same in February. It seems possible that spider monkeys are not highly seasonal breeders in Colombia, although definitive information is proba- bly lacking (see also Klein, 197Ib). STATUS: In large areas of Colombia, meat hunt- ing is a greater threat to Ateles than its habitat destruction, even though the latter pressure is often quite great. Spider monkeys seem not to be actively hunted for food in the Caribbean coastal region or by descendants of the original settlers of the eastern plains. However, they are hunted by recent settlers originating from the mountainous interior and by all known indigenous groups within their range, including those in the Colombian Amazon. In many areas of apparently suitable forest where Ateles is not found today, it seems possible that this absence could be related to historic hunting pressures, to certain rather subtle ecological deficiencies (see Klein and Klein, 1976). and/or to sylvatic yellow fever in recent times. Some hunting of Ateles for exportation does occur in the Caribbean coastal and Amazon regions, but this trade in Colombia is not very large. As with woolly monkeys, most spider monkeys in the Leticia market originate from Brazil or Peru and belong either to the A. p. chamek group of black spider monkeys or the A. p. belzebuth group with a prominent forehead triangle. 23. Species of Questionable Occurrence in Colombia A. Chiropotes satanas (Hoffmansegg, 1807)â Black saki. This primate form may yet be confirmed as occur- ring in Colombia based largely on records of its existence on the bank of the Orinoco River. All pertinent records known to us are from the Ven- ezuelan banks (in the Amazon Federal Territory of Venezuela) of the Orinoco (right bank), Casiquiare (eastern bank), and Negro (eastern bank) rivers, or it is uncertain from which bank such collections originated. If Chiropotes occurs in Colombia it would be on the eastern border of Guainia Comisaria or possibly the southeastern tip of Vichada Comisaria on the left banks of the Orinoco or Atabapo (Orinoco tributary) rivers or on the right bank of the upper Negro (or Guainia) River. A specimen in the American Museum from Pitado (5 km above San Fernando de Atabapo) on the Orinoco above the mouth of the Atabapo River is the closest known locality of C. satanas chiropotes (Humboldt, 1812) to Colombia. If the collection oc- curred on the left bank, the locality would be less than 10 km (without limiting geographic barriers) from Colombia. B. Other primate forms. It is possible, although not too likely, that Cacajao calvus (Spix, 1823) and/or Saguinus labiatus thomasi (Goeldi, 1907) exist as far west as the Amazonas Comisaria in Colombia between the Caqueta and Putumayo rivers. However, the nineteenth-century records of Saguinus mystax (Spix, 1823), 5. bicolor (Spix, 1823), and Cacajao rubicundus (I. Geoffroy-St. Hilaire, 1848) from Pebas, on the northern bank of the Amazon in the present Depart- ment of Loreto, Peru (which could imply an extension into the Amazonas Comisaria trapezium of Colombia), are incorrect. In addition, the type specimen of 5. fuscicollis illigeri (Pucheran, 1845) was incorrectly assumed to have come from Colombia. Another record (Elliot, 1913) for C. rubicundus from the northern bank of the Amazon opposite Sao Paulo de Olivenca, in Brazil (somewhat northeast from Leticia, Colombia), is also in error, as this form is not known north of the Amazon River. APPENDIX: SOME VERNACULAR PRIMATE NAMES USED BY VARIOUS INDIAN TRIBES IN COLOMBIA Achagua Indians, Comisaria of Arauca and eastern Department of Boyaca: A. seniculus, "Arabata" (L. M. Giron, 1883). Andaqui (AndakO Indians, southern Department of Cauca and extreme western Intendencia of Caqueta: A. p. belzebuth, "Fiaguai" (Castellvi, 1938). Catio Indians, northwestern Department of Antioquia: A. seniculus, "Kuara" (Fray P. del Santisimo Sacramento, in Ortiz, 1940). Choco Indians, Department of Choco: A. trivirgatus, "Una": A. seniculus, "Chipuro"; A. p. robustus, "Hierre," "Perre," "Yerre" (Vallejo, 1929). Coreguaje Indians. Intendencia of Caqueta: A. seniculus, "Emo," "Emu." "Emori." "Emuri"; Lagothrix. "Nase"; A. p. bel- zebuth, "Painaso" (Castellvi. 1938). Cubeo Indians. Mitu region. Comisaria of Vaupes: S. inustus(?), "Abujijiyo"; Saimiri, "Jijiyo"; C. torquatus, "Wao"; A. seniculus, "Emu"; C. albifrons, "Waja"; C. apella, "Taque"; Lagothrix, "Caparo" (Salser. 1972, Instituto Linguistico de Ver- ano).
68 HERNANDEZ-CAMACHO and COOPER GuahiboIndians.Comisariaof Vichada: Saimiri, "Tiii" andTsfile"; Aotus, "Muciiali"; C. torquatus, "Ojo-ojo"; A. seniculus, "Neje"; C. albifrons, "Vanali"; C. apella, "Pababe"; A. p. belzebuth, "Cuveri" and "Cuvairi"; Lagothrix, "Capalu" (Kondo, 1972, Instituto Linguistico de Verano), Saimiri, "Chuleyo" (Fernandez and Bartolome, 1895). Guake Indians: A. seniculus. "Arabata"; Lagothrix, "Arimina." "Arimina"; A. paniscus, "Jarachi" (Castellvi. 1938). Huitoto Indians, Curiplaya. Caqueta Intendencia: Lagothrix, "Jemo." La Chorrera, Igara-Parana River, Amazonas Comisaria: Lagothrix, "Xemma" (Castellvi, 1938). Karidaker Indians: Lagothrix, "Caparro" (Humboldt, 1812). Maipures Indians, vanished tribe of the Colombian bank of the Orinoco River, Comisaria of Vichada: Saimiri, "Bitschetchies," "Bichechies" (Humboldt, 1812). Miraria Indians (Bora group), southeastern Comisaria of Caqueta: A. seniculus, "Inome" (Castellvi, 1938). Muinane Indians* (Bora group). La ⢠Sabana and Araracuara, Amazonas Comisaria: Cebuella. "Jibiillaje"; 5. nigricollis and/or S. fuscicollis and Callimico, "Jiihusaryje"; Saimiri, "Till*-"; Aotus, "Tohomimi-"; C. torquatus. "Gaahi"; Pithecia, "Jiiubaiga"; A. seniculus. "iju"; C. albifrons, "Jimuhai"; C. apella "Chuyiyi"; A. p. belzebuth. "Meecu"; Lagothrix, "C«mi (Walton, 1972, Instituto Linguistico de Verano). Muzo Indians, a vanished tribe of western Department of Boyaca: Aotus, "Kubaime" (Simon, 1882-1892). Noanama Indians, Department of Choco: A. villosa, "Kotudii" (Ortiz, 1940). Puinave Indians. Comisaria of Vaupes: Saimiri. "Chiau," "Koka"; Aotus, "Macuriya" (also Maku Indians); Callicebus sp., "Tuii"; A. seniculus, "Caa"; Lagothrix, "Choicack"; A. p. belzebuth, "Chairi" (Cabrera, unpublished). Queehua (Keshwa), dialect of the Ingano Indians, Putumayo Co- misaria: Aotus, "Tutamono" (also Kamsa, Sibundoy Indians. Putumayo Comisaria); A. seniculus. "Koto," "Kotu," "Koto- monu" (Castellvi, 1938). Quimbaya Indians, a vanished tribe of the departments of Quindio and Risaralda: A. seniculus, "Chifumi" (Bastian, 1878). Siona Indians, Comisaria of Putumayo: Aotus, "Unamihue"; A. seniculus, "Emii"; Lagothrix. "Guao." "Mazo"; A. p. bel- zebuth, "Painnazo" (Castellvi, 1938). Tunebo Indians, Sarare River region, northern Department of Boyaca, southeastern Department of North Santander; Lagothrix, "Savaroma"; A. p. hybridus. "Citroa" (Castellvi, 1938). Yucuna Indians. El Deposito, Miriti Parana River, Amazonas Co- misaria: Saimiri, "Cuhuijru"; C. apella, "Calapichi"; Lagothrix, "Caparu" (Schauer, 1972, Instituto Linguistico de Verano. Also Vaupes Comisaria: Saimiri, "Cuhisii" (Cabrera, unpublished). Indians of uppermost San Juan River, Department of Risaralda: A. seniculus, "Trua" (Vallejo, 1929). *h = phonetic "glottal stop" of Muinane Indian language; e or 4 = phonetic high central unrounded vowel of Guahibo and Muinane languages. Bibliography to Appendix Fernandez. M., and M. Bartolome. 1895. Ensayo de gramatica hispanogoahiba. Bogota. Giron, L. M. 1883. Los Achaguas. Papel Periodico Ilustrado. Bogota. Humboldt. A. dc. and A. Bonpland. 1812. Recueil d'observations de zoologie et d'anatomie comparee. Smith and Gide, Paris. Kondo, R. 1972. Personal communication. Ortiz. S. E. 1940. Lingiiistica Colombiana, familia Choko. Univ. Catol. Bolivar., 6(18):46-77. Medellin. Salser, J. K. 1972. Personal communication. Schauer, J. 1972. Personal communication. Simon. Fray Pedro. 1882-1892. Noticias historiales de las conquis- tas de Tierra Firme en las Indias Occidentales. 5 vols. Bogota. Vallejo-E.. J. 1929. Vocabulario baudo. revista de Colombia. Bogota, p. 134. Reprod. in Ideariu.Ti. vol. I., Pasto. 1937. p. 259. Walton, J. 1972. Personal communication. REFERENCES Bastian, A. 1878. Die Kulturlander des alten Amerikas. T. I. Berlin, p. 243, Nota 1. Cabrera, I. Unpublished. Information based on recent field studies in Colombia. Castellvi, M. de. 1938. Materials para estudios glotologicos. Bol. Estud. Hist. ll(84):368-372. Pasto. Allen, J. A. 1916. Mammals collected on the Roosevelt Brazilian expedition, with field notes by Leo E. Miller. Bull. Am. Mus. Nat. Hist. 35:559-610. Alston, E. R. 1879. Mammalia. In Biologia Centrali-Americana, vol. 1, with an introduction by P. L. Sclater. pp. I-XX, 1-220 tables (col.) 1-22, Taylor and Francis, London. Baldwin, J. D., and J. I. Baldwin. 1971. Squirrel monkeys (Saimiri) in natural habitats in Panama. Colombia, Brazil and Peru. Pri- mates 12(l):45-61. Bates, H. W. 1863. The naturalist on the River Amazonas, vol. II. John Murray, London. Cabrera, A. 1957. Catalogo de los mamiferos de America del Sur Instituto Nacional de Investigacion de la Ciencias Naturales. Ciencio Zoologica. IV:No. 1, Buenos Aires and Peru. Cooper, R. W. 1968. Squirrel monkey taxonomy and supply. Pages 1-29 In L. A. Rosenblum and R. W. Cooper, eds. The squirrel monkey. Academic Press, New York. Cooper, R. W.. and J. Hernandez-Camacho. 1975. A current appraisal of Colombia's primate resources. Pages 37-66 in G. Bermant and D. G. Lindburg, eds. Primate utilization and conser- vation. John Wiley and Sons, New York. Eisenberg, J. F., N. A. Muckenhirn, and R. Rudran, 1972. The relation between ecology and social structure in primates. Science 176:863-*74. Elliot, D. G. 1913. A review of the primates, vols. I and II. American Museum of Natural History, New York. Erikson, G. E. 1963. Brachiation in New World monkeys and in anthropoid apes. Symp. Zool. Soc. Lond. 10:135-164. Fooden. J. 1963. A revision of the woolly monkeys (genus Lago- thrix). J. Mammal. 44:213-247. Fooden, J. 1964. Stomach contents and gastrointestinal proportions in wild-shot Guianan monkeys. Am. J. Phys. Anthropol. (n.s.) 22:227-231. Green, K. M. 1976. The nonhuman primate trade in Colombia. This volume. Hernandez-Camacho, J., and E. Barriga-Bonilla. 1966. Hallazgo del genero Callimico (Mammalia: Primates) en Colombia. Caldasia 9(44):365-3T7. Hershkovitz, P. 1949. Mammals of northern Colombia. Preliminary report no. 4: Monkeys (Primates), with taxonomic revision of some forms. Proc. U.S. Nat. Mus. 98:323-427. Hershkovitz, P. 1955. Notes on American monkeys of the genus Cebus. J. Mammal. 36(3):449-452. Hershkovitz, P. 1963. A systematic and zoogeographic account of
THE NONHUMAN PRIMATES OF COLOMBIA 69 the monkeys of the genus Callicebus (Cebidae) of the Amazonas and Orinoco River basins. Mammalia 27(1): 1-79. Hershkovitz, P. 1966. Taxonomic notes on tamarins; genus Saguinus (Callithricidae, Primates) with descriptions of four new forms. Folia Primatol. 4:381-395. Hershkovitz. P. 1968. Metachromism or the principle of evolution- ary change in mammalian tegumentary colors. Evolution 22:556-575. Hershkovitz. P. 1969. The evolution of mammals in southern continents. VI. The recent mammals of the neotropical region: a zoogeographic and ecological review. Q. Rev. Biol. 44(l):l-70. Hershkovitz. P. 1970. Notes on tertiary platyrrhine monkeys and description of a new genus from the late Miocene of Colombia. Folia Primatol. 12:1-37. Hershkovitz, P. 1972. Notes on New World monkeys. Int. Zoo Yearb. 12:3-12. Hill. W. C. O. 1960. Primates, IV. Cebidae, Part A, Edinburgh Univ. Press. Humboldt. A. de. and A. Bonpland. 1812. Recueild'observationsde zoologie et d'anatomie comparee. Smith and Gide. Paris. Izawa. K. 1972. Monkeys in the upper basin of Amazon. Monkey I6(2):5-18. (In Japanese) Kellogg, R.. and E. A. Goldman, 1944. Review of the spider monkeys. Proc. U.S. Nat. Mus. 96:1-45. Klein, L. L. 1971a. Ecological correlates of social grouping in Colombian spider monkeys. Paper presented at the Animal Be- havior Society Meeting. Logan, Utah. Klein. L. L. 19711' Observations on copulation and seasonal reproduction of two species of spider monkeys. Ateles belzebuth and A. geoffroyi, Folia Primatol. 15:233-248. Klein. L. L.. and D. J. Klein. 1973. Observations of two types of neotropical primate intertaxa associations. Am. J. Phys. An- thropol. 38(2):649-653. Klein. L. L., and D. J. Klein. 1974. Neotropical primates: aspects of habitat usage, population density, and regional distribution in La Macarena, Colombia. This volume. Lehmann. F. Carlos. 1959. Contribuciones al estudio de la fauna de Colombia. XIV. Neuvas observaciones sobre Oroaetus isidori (Des Murs). Nov. Colomb., Contrib. Cient. Mus. Hist. Nat. Univ. Cauca. Popayan 1(4): 169-195. Maria. Brother Appolinar. 1913. Catalogodel Museo (del Instituto de La Salle, Bogota). Bol. Soc. Cienc. Nat. Inst. La Salle, Bogota, 1(1). Mason, W. A. 1966. Social organization of the South American monkey Callicebus moloch: a preliminary report. Tulane Stud. Zool. 13:23-28. Mason, W. A. 1971. Field and laboratory studies of social organiza- tion in Saimiri and Callicebus. Primate Behavior 2:107-137. Moynihan, M. 1964. Some behavior patterns of platyrrhine mon- keys. I. The night monkey (Aotus trivirgatus). Smithson. Misc. Coll. 146(5): 1-84. Moynihan, M. 1966. Communication in the titi monkey. Callicebus J. Zool. (Lond.) 150:77-127. Moynihan, M. 1967. Comparative aspects of communication in New World Primates. In D. Morris, ed. Primate ethology. Weidenfield and Nicolson, London. Moynihan, M. 1970. Some behavior patterns of platyrrhine mon- keys. II. Saguinus geoffroyi and some other tamarins. Smithson. Contrib. Zool. 28:1-77. Moynihan. M. 1976. Notes on the ecology and behavior of the pygmy marmoset, Cebuella pygmaea. in Amazonian Colombia. This volume. Thorington, R. W., Jr. 1967. Feeding and activity of Cebus and Saimiri in a Colombian forest. Pages 180-184 In D. Starck, R. Schneider, and H. J. Kuhn, eds. Neue ergebnisse de primatologie. Fischer. Stuttgart. Thorington. R. W., Jr. 1968. Observations of squirrel monkeys in a Colombian forest. Pages 69-85 In L. A. Rosenblum and R. W. Cooper, eds. The squirrel monkey. Academic Press, New York. Thorington, R. W., Jr. 1970. Feeding behavior of nonhuman pri- mates in the wild. Pages 15-27 In R. S. Harris, ed. Feeding and nutrition of nonhuman primates. Academic Press. New York.
