Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
CHAPTER 3 Ecological Relationships Knowledge of the ecological relationships between plant-parasitic nematodes and their environment is important for understanding some of the principles of nematode control. Agricultural land represents a specialized environment, ranging from a dry, barren waste to a moist, lush jungle of plant growth. Plant-parasitic nematodes are mostly those soil-inhabiting species that are capable of withstanding the frequent changes caused by man's agricultural practices. Some of these nematodes, such as species of spiral (Helicotylenchus spp.), stunt (Tylenchorhynchus spp.), and sheath (Hemicycliophora spp.) nematodes, can live in a wide variety of habitats. Some, such as the rice nematode (Hirschmanniella oryzae) in aquatic habitats, are widely distributed but are limited to particular combinations of environmental conditions. Still others, such as sting nematodes (Belonolaimus spp.) in the sandy soils of the southeastern United States, are found only in very special situations. There- fore, it is difficult to recommend control practices for such diverse kinds of nematodes without first knowing how they live and survive in the soil and in host plants. VERTICAL DISTRIBUTION OF NEMATODES The vertical distribution of nematodes in cultivated soil is usually irregular but is generally closely related to the distribution of plant roots and the area adjacent to roots, which is called the rhizosphere. Since the movement of nematodes in the soil by their own activities is limited at most to a few feet per year, it is obvious that the number of plant-parasitic nematodes is greater 17
18 FACTORS INFLUENCING NEMATODE CONTROL in soils containing plant roots than in soils without plant roots and is corre- lated with the distribution of roots of present and previous hosts. Nematodes are mainly concentrated in the top foot of soil, and as many as six billion have been estimated in the top inch of an acre of soil. Little information exists on the distribution of nematodes deeper than 1 foot in soil; however, a root-knot nematode (Meloidogyne incognita) has been found at a maximum depth of 17 feet in grape vineyards. Consequently, in a chemical treatment the soil must be treated to a greater depth for deep-rooted than for shallow- rooted crops, thus requiring more chemical and more expensive equipment. The fallowing of soil for an adequate period will generally reduce the number of nematodes present. Plant-parasitic nematodes survive longer in the absence of food sources than most nonparasitic nematodes. The cyst nematodes (Heterodera spp.), the most persistent of the plant-parasitic nematodes, de- cline in soil at a steady rate of 35 to 60 percent per year, depending on soil type, moisture, and temperature, regardless of the density of the initial popu- lation per unit of soil. Many questions remain unanswered concerning the in- fluence of soil type, moisture, aeration, and other factors on distribution and the response of nematodes to them. NEMATODE SURVIVAL Plant-parasitic nematodes are able to survive despite unfavorable conditions such as cold and dry periods between host crops. Except in the tropics and heated areas, such as greenhouses, they do not grow and reproduce through- out the year. Nematodes survive unfavorable environments in a dormant con- dition, which is a quiescent or inactive state that is often associated with a lowered metabolic rate. The length of the quiescent period is usually limited by the amount of food reserves in the nematode and the environmental con- ditions. Quiescence may serve to extend a comparatively short life cycle of 20 to 40 days to periods varying from a year for many plant parasites to 20 to 30 years for such nematodes as the stem nematode (Ditylenchus dipsaci) and the wheat nematode (Anguina tritici). NEMATODE POPULATIONS In agricultural soil, the upper population limit for any plant-parasitic nema- tode species depends on the nematode's reproductive potential, the host-plant species, and the length of time the nematode remains in an environment favorable for reproduction. Generally, the reproductive potential of the specialized endoparasites and aboveground parasites is greater than that of
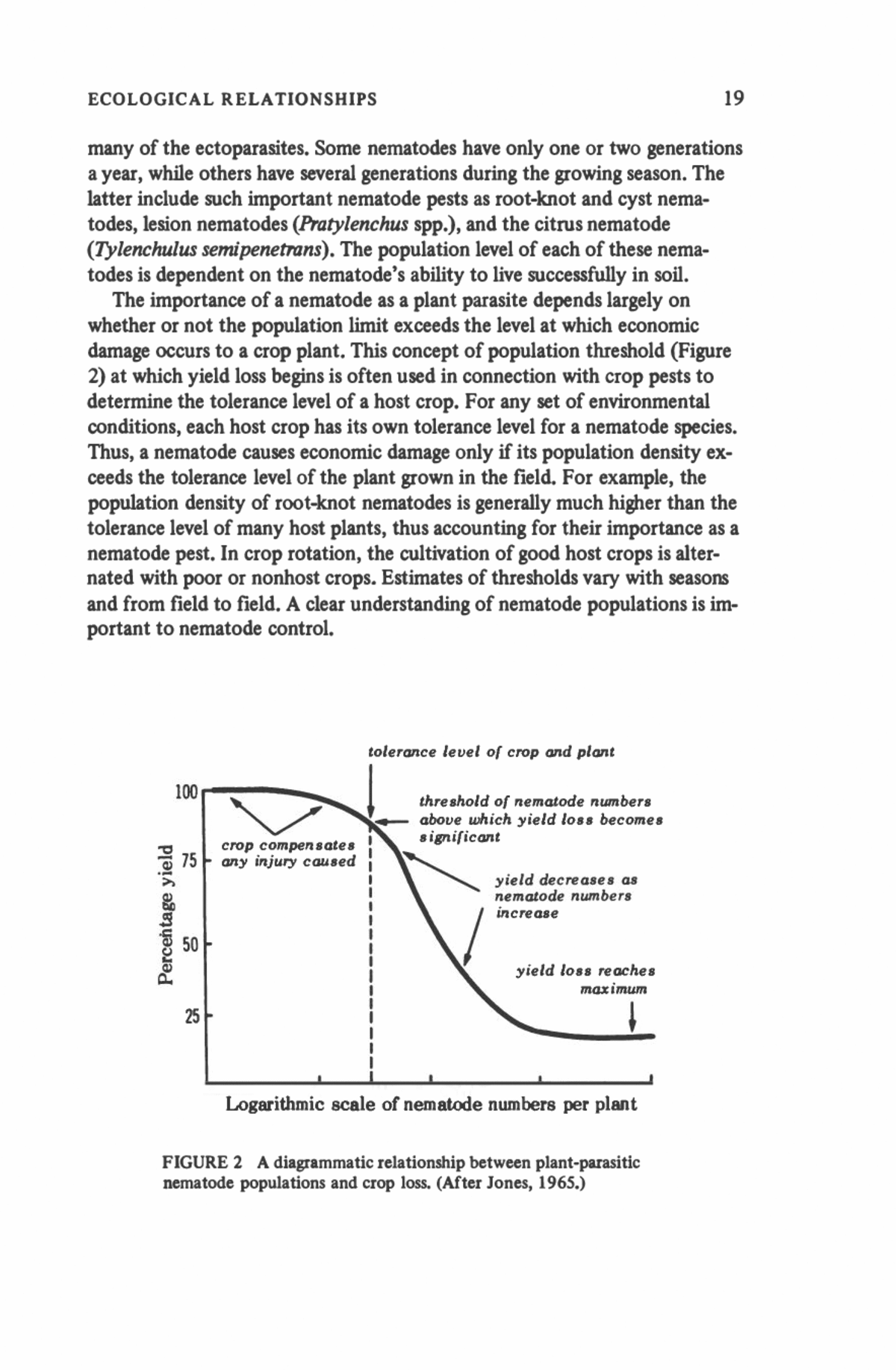
ECOLOGICAL RELATIONSHIPS 19 many of the ectoparasites. Some nematodes have only one or two generations a year, while others have several generations during the growing season. The latter include such important nematode pests as root-knot and cyst nema- todes, lesion nematodes (Pratylenchus spp.), and the citrus nematode (Tylenchulus semipenetrans). The population level of each of these nema- todes is dependent on the nematode's ability to live successfully in soil. The importance of a nematode as a plant parasite depends largely on whether or not the population limit exceeds the level at which economic damage occurs to a crop plant. This concept of population threshold (Figure 2) at which yield loss begins is often used in connection with crop pests to determine the tolerance level of a host crop. For any set of environmental conditions, each host crop has its own tolerance level for a nematode species. Thus, a nematode causes economic damage only if its population density ex- ceeds the tolerance level of the plant grown in the field. For example, the population density of root-knot nematodes is generally much higher than the tolerance level of many host plants, thus accounting for their importance as a nematode pest. In crop rotation, the cultivation of good host crops is alter- nated with poor or nonhost crops. Estimates of thresholds vary with seasons and from field to field. A clear understanding of nematode populations is im- portant to nematode control. 100 13 75 3 50 25 tolerance level of crop and plant threshold of nematode numbers above which yield loss becomes significant yield decreases as nematode numbers increase yield loss reaches maximum Logarithmic scale of nematode numbers per plant FIGURE 2 A diagrammatic relationship between plant-parasitic nematode populations and crop loss. (After Jones, 1965.)
20 FACTORS INFLUENCING NEMATODE CONTROL THE SOIL ENVIRONMENT All plant-parasitic nematodes inhabit soil for varying lengths of time during their life cycles (Figure 3). For example, the ectoparasitic nematodes spend their entire lives in the soil, usually in the rhizosphere of the plant. The more specialized endoparasites enter plant tissue and thereby spend less of their lives in the soil and rhizosphere. The aboveground parasites are mostly inside plant tissues and spend very little of their lives in the soil. Due to the nema- tode's life habitat in the soil, it is easier to control ectoparasitic than endo- parasitic nematodes. The principal factors in the nematode's soil environment are temperature, moisture, texture, aeration, and the chemistry of soil solution. Only in the Adult Larva 4 Larva 1 Larva 2 ECTOPARASITIC NEMATOOES 1. Hemicydiophora arenaria 2. Trichodorus christiei ENOOPARASITIC NEMATOOES 1. Heterodera schachtii 2. Meloidogyne javanica ABOVEGROUNO PARASITIC NEMATOOES 1. Oitylenchus dipsaci 2. Anguina tritici L2 ANGUINA FIGURE 3 The relationship between parasitic habits of some nematodes and the portion of their life cycle spent in the soil, plant, and rhizosphere.
ECOLOGICAL RELATIONSHIPS 21 laboratory is it possible to investigate this complex, constantly changing en- vironment; yet, from laboratory data it is difficult to relate varied factors such as nematode distribution, population levels, and pathogenicity to any one factor. Results of field-population studies are necessary to determine the influence of the interdependent and interacting environmental factors. TEMPERATURE Temperature affects nematode activities such as hatching, reproduction, movement, development, and survival and also affects the host plant. Most plant-parasitic nematodes become inactive at a low temperature range of 5 to 15°C, have an optimum range of 15 to 30°C, and become inactive at a high temperature range of 30 to 40°C. Temperatures outside these extremes may be lethal. Little information exists on the effect of constant or alternating temperatures on specific activities of individual nematode species. The Javanese root-knot nematode (M. javanica) is of little concern in the northern states, where it does not overwinter out-of-doors in deeply frozen soils, but the northern root-knot nematode (M. hapla) overwinters and may be a serious pest in these areas. Temperatures, however, do not limit the establishment of some nematodes: the sugar-beet nematode (Heterodera schachtii) is a serious pest in the north as well as in the south, where the soil temperature may ex- ceed 35°C. The less adaptable stem nematode is restricted to cool climates or to warm climates where the host is winter-grown. Determining the influence of temperature on nematode reproduction in plants is complicated, because temperature influences the growth of the plant itself. Changes in plant growth produce corresponding changes in root mor- phology and physiology. Temperature partially determines the choice of crop plantings and rotations. In areas of the United Kingdom, Europe, and the United States, some varieties of potatoes and sugar beets are grown in early spring, when the soils are too cold for reproduction of the potato-cyst and sugar-beet nematodes but are not too cold for the growth of the plants. Pro- tection from nematodes during the early part of the growing season reduces nematode damage at harvest. MOISTURE Fluctuating soil moisture due to rainfall or irrigation is one of the chief factors influencing nematode-population increases. Dry soil conditions may depress populations of a ring nematode (Criconomoides xenoplax), a dagger nematode (Xiphinema americanum), and root-knot and cyst nematodes. Although dry
22 FACTORS INFLUENCING NEMATODE CONTROL conditions may depress nematode activity and resulting populations, all nematodes may not be killed. Eggs of most nematodes as well as certain other nematode stages, such as preadult stages of pin nematodes (Paratylenchus spp.), survive drying. Dry fallowing of field soils may not be a practical con- trol measure except in some hot, dry regions, where it reduces the numbers of nematodes so that a profitable crop can be obtained. Saturated soils are not generally favorable for nematode pests of agricul- tural crops. In tropical rain belts and in flooded fields, populations of some species of root-knot, cyst, stunt, and pin nematodes have been reduced by excess water, lack of oxygen, and toxins of anaerobic organisms. However, high populations of some nematodes, such as species of Dolichodorus, Radopholus and Hirschmanniella, are found chiefly in wet locations. It is thought that nematodes are constantly active in soils that have a moisture content of between 40 and 60 percent of field capacity. In dry and wet soils they are quiescent for varying periods. Nematodes need free water films in the soil for hatching and movement, but the influence of moisture on the nematode is little known. Since the interrelationship of soil moisture and soil structure is responsible for the aeration properties of the soil, the oxygen level may be the fundamental factor influencing some activities of nematodes. As soil moisture increases, soil aeration decreases, so that soils become low in oxygen immediately after heavy rains, flooding, or irrigation. From studies of a few plant-parasitic nematodes, it appears that individual nematodes are capable of fermentative as well as oxidative metabolism, which enables them to survive for varying periods of time without oxygen. Low levels of oxygen may induce quiescence and enable nematodes to survive. Growth and develop- ment of nematodes, which are important in determining population levels, are oxygen-dependent; therefore, high populations are usually found in moist, well-aerated soils. SOIL TEXTURE AND STRUCTURE Soil texture describes the sizes of soil particles. A coarse-textured soil usually contains a high percent of sand and has large pores that drain more quickly than the small pores of fine-textured soil, which has a high proportion of clay and silt. Because of the wide variation of the biotic, physical, and chemical environments within textural categories, it is difficult to generalize among soil type, nematode activity, and distribution. Many of the cyst, root-knot, lesion, and stubby-root (Trichodorus spp.) nematodes are found in large numbers in coarse-textured sandy soils. However, the stem, sugar-beet cyst, and some species of lesion and stunt nematodes are numerous in
ECOLOGICAL RELATIONSHIPS 23 clay soils. Still others, such as the citrus nematode, occur frequently in both sandy and clay soils. The speed of nematode movement through soil is related to soil pore diameter, soil particle size, diameter of the nematode and its relative activity, and the thickness of the soil-water films. A nematode cannot move between soil particles when the pore diameters are less than the nematode width. As mentioned previously, soil structure, moisture, and aeration are interrelated. When the soil pores are full of water, a nematode moves inefficiently, and, when aeration becomes limiting, the nematode becomes inactive. In very dry soils, there is good aeration but not enough water to form films, so that the nematodes do not move. Only a soil of intermediate moisture has suffi- cient aeration and water films for efficient nematode movement. SOIL SOLUTION The chemical constitution of the soil solution, a major constituent of the soil environment, includes soil salinity, pH, organic matter, fertilizers, insecticides, and nematocides. Plant-parasitic nematodes probably derive few nutrients from the soil solution. The hatching of eggs and the survival of larvae may be influenced by various salts and ions. During dry and wet periods, soil nema- todes are subjected to variable salt concentrations in the soil solution. How- ever, nematodes can tolerate osmotic pressures up to about 10 atm, at least for short periods. This is considerably higher than the maximum 2 atm oc- curring in most agricultural soils. A soil pH ranging between 5.0 and 7.0 has little effect on nematodes. Lime, often used to neutralize soil acidity, causes no decrease in population. Fertilizers and organic matter may influence nema- tode populations indirectly by increasing host-plant growth. Occasionally, the use of nematocides and insecticides in soil may eliminate some nematode enemies, thus leading to an increase in population of a plant parasite. CLIMATE Rainfall and temperature above soil level are extremely important to the growth and development of both nematodes and plants. These factors are usually responsible for seasonal fluctuations in nematode populations and may even determine the success of a species in becoming established in a new habitat or region. Climatic factors affecting humidity are particularly im- portant to aboveground parasitic nematodes, which are able to invade seed- lings and move upward on plant surfaces covered by water films or droplets.
24 FACTORS INFLUENCING NEMATODE CONTROL These nematodes may be subjected to severe desiccation and great extremes of temperature owing to the more violent changes in aerial climate compared with soil climate. Perhaps, as an adaptation to this, certain stages of these nematodes, such as second-stage larvae in wheat nematodes and fourth-stage larvae in stem nematodes, are capable of withstanding long periods of desicca- tion. Specific information on the influence of the microclimate of the plant surface on nematode activities is lacking. THE PLANT ENVIRONMENT The plant-host environment, consisting of either root or stem and leaf tissue, greatly influences the endoparasitic nematodes. The plant tissues that are usually attacked are apical meristems that contain cells with thin walls and offer a chemically rich environment. The epidermis and cell wall offer mechanical barriers to nematode entrance and movement. The plant tissue protects endoparasitic nematodes from the soil environment and is their sole source of food, and the quality and quantity influence nematode growth and reproduction. Thus, host susceptibility, tolerance, and resistance to nematodes are dependent on properties of individual plant cells and tissues. Much remains unknown about the nature of these factors and their effect on the nematodes. The periderm and necrotic areas, which are formed in some plants in response to nematode feeding, may affect nematode growth and reproduction, because the quality and quantity of nutrients are deficient in these areas, or the nema- todes may be excluded from suitable plant cells by these areas. In plant para- sites such as root-knot, cyst, and citrus nematodes the host cells are modified to provide specialized feeding sites, and their physiological and nutritional dependence on the host become delicately balanced. Recently, this intricately balanced system has been studied to try to find ways of controlling nema- todes by the use of chemotherapeutic agents or antimetabolites to modify the host-plant environment to one unsuitable to nematodes. This area of re- search needs emphasis to gain an understanding of nematode nutrition and host-parasite relations and to aid in developing methods for systemic control of nematodes. THE RHIZOSPHERE In addition to serving as a source of food for nematodes, plant roots may also modify the soil environment by lowering the concentration of mineral nutri- ents, depleting moisture, increasing carbon dioxide, reducing oxygen, and contributing a variety of organic substances by exudation and sloughing off
ECOLOGICAL RELATIONSHIPS 25 of cells. The rhizosphere, the zone immediately around the plant roots, is a dynamic environment, where the relationships among nematode, host, and environment are often of a chemical nature. A root exudate stimulates the hatching of eggs of the cyst nematodes. Hatching is usually stimulated by chemicals from a wide range of plants, some of which are nonhosts. The com- position of hatching chemicals is unknown, despite more than 20 years of re- search by numerous workers. The eggs of root-knot nematodes, as well as most other plant-parasitic nematodes, hatch freely in water; but in soil, plant- root exudates significantly increase the hatching of root-knot nematode eggs as compared with a water hatch. They stimulate the metabolism of larvae after hatching and may account for their directional movement toward plant roots. The exudates also influence the molting of preadult larvae of the pin nematode. Such examples of stimulation by plant roots appear to be a refine- ment of parasitism. Root exudates and other chemicals may also inhibit egg-hatching or may repel nematodes. From a few observations, it appears that some plants, such as marigold, asparagus, and tobacco, produce chemicals that repel or even kill some species of nematodes. Little is known about the identity of these exu- dates and other chemicals, the nature of the reactions on nematodes, or the receptors in the nematodes. There is also evidence that nematodes may be repelled by small quantities of nematocides. Microorganisms in the rhizosphere may significantly influence nematodes in several crops by antagonism, by competition for food and oxygen, or by excretions that may stimulate or inhibit nematodes. Research in this area should not be overlooked. The ecological system that illustrates the complex interrelationships among plant-parasitic nematodes, plant, climate, and soil environment is summarized in Figure 4. Although the information now available is substantial, the inter- relationships for any one nematode-plant combination are not completely understood. Comprehensive coordinated information on nematode activities, such as length and stage of life cycle, and mechanisms for nematode survival in unfavorable environments and in the absence of a host is limited. A critical evaluation of vertical distribution of nematodes in soil, particu- larly at depths below 2 feet, is needed to determine the possible crop loss caused by these nematodes and the need for their control. Other areas need- ing ecological studies include host-parasite relations, mixed populations of plant parasites, the influence of other microorganisms in the rhizosphere, the influence of plant microclimate on aerial plant parasites, the influence of soil factors on population levels of plant parasites, and the application of nematode-hatching chemicals to infested soil. New methods, fresh approaches, and long-range programs aimed at developing integrated ecological concepts of plant-parasitic nematodes are necessary for progress toward more effective and economical control.
26 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 4 The ecological system, showing the complex interrelationships among plant- parasitic nematodes, plant, climate, and soil environment. BIBLIOGRAPHY Bird, A. F., and H. R. Wallace. The chemical ecology of Acanthocephala and Nematoda, In Chemical zoology. Academic Press Inc., New York. (In press) Hyman, L. H. 1951. The invertebrates. Vol. 3. McGraw-Hill Book Co., Inc., New York. 572 pp. Jones, F. G. W. 1959. Ecological relationships of nematodes. In C. S. Holton, G. W. Fisher, R. W. Fulton, H. Hart, and S. E. A. McCallan (eds.). Plant pathology problems and progress, pp. 395-411. Univ. Wisconsin Press, Madison. 588 pp. Jones, F. G. W. 1966. The population dynamics and population genetics of the potato cyst-nematode Heterodera rostochiensis Woll. on susceptible and resistant potatoes. Rep. Rothamsted Exp. Sta. 1965. pp. 301-316. Van Gundy, S. D. 1965. Factors in survival of nematodes. Ann. Rev. Phytopathol. 3:43-68. Wallace, H. R. 1964. The biology of plant parasitic nematodes. St. Martin's Press Inc., New York. 280 pp. Winslow, R. D. 1960. Some aspects of the ecology of free-living and plant parasitic nematodes. In J. N. Sasser and W. R. Jenkins (eds.). Nematology, fundamentals and recent advances, pp. 341-415. Univ. North Carolina Press, Chapel Hill. 480 pp.
CHAPTER The Physiology of Nematodes in Relation to Control Investigations of nematode metabolism and biochemistry, mechanisms of action of nematocidal agents, biochemical bases for plant resistance to nema- todes, and nematode nutrition are increasing. Few basic studies on nematode physiology were conducted before 1950, and, even now, relatively few in- vestigators are conducting research on such subjects. One reason for the limited research on many aspects of nematode physiology has been the diffi- culty of obtaining adequate quantities of specific plant-parasitic nematodes. While the information in this section may not bear directly on specific control measures, it may give insight as to why some of the commonly applied meth- ods are successful. CHEMICAL COMPOSITION Nematodes, like other animals, contain carbohydrates, proteins, lipids, nucleic acids, vitamins, hormones, minerals, and numerous other chemicals, but not much is known of the precise kinds or amounts of these substances present in nematodes. Although the composition of animal-parasitic nema- todes has received considerable study, it is doubtful if all these data are also applicable to the plant parasites. Chemical composition of nematodes, which affects longevity, degree of resistance to temperature extremes, desiccation, atmosphere, osmotic conditions, and chemicals, undoubtedly relates closely to the success of the various control measures utilized. Glucose, fructose, and 15 free and protein amino acids were identified from two species of plant-parasitic nematodes. Plant-parasitic nematodes 27
28 FACTORS INFLUENCING NEMATODE CONTROL generally contain far more lipid than do animal parasites. The high lipid con- tent of nematodes may have a bearing on the success of the lipophilic halo- genated hydrocarbons as nematocides and also on longevity of nematodes in the absence of host tissue. The cuticle, which protects nematodes from their environment, is composed primarily of protein but also contains lipids, polyphenols, enzymes, and nucleic acids. METABOLISM Intermediary metabolism in plant-parasitic nematodes is presently not under- stood well enough that we can make comparisons with the metabolism of animal parasites. Portions of the metabolic sequences of sugar breakdown and terminal oxidation in plant nematodes have been elucidated, but in no in- stance have all the steps in any one sequence been demonstrated. Research on these fundamental metabolic cycles is needed, since variations in the se- quences of chemical reactions might be important factors in governing the parasitic nature of nematodes. Such variations may also represent weak points in metabolism, toward which attempts to develop specific control measures could be directed. Synthesis of amino acids by two species of plant-parasitic nematodes incubated in 14C-labeled glucose and acetate solutions in the absence of plant tissues has been shown, illustrating that it is not imperative for nematodes to feed on cells in order to take in chemicals. Nematodes seem able to digest a variety of polymeric plant components outside the body. It has been reported that enzymes that break down starch, sucrose, pectin, cellulose, protein, and glycosides have been discharged by plant-parasitic nematodes. Presumably, these enzymes break down plant com- ponents outside the nematode body before they are ingested by the parasite. Homogenates or extracts of nematodes contained all the above plus other digestive enzymes. In addition to being active outside the nematode, such enzymes are very likely active in the further degradation of plant substances in the digestive tract of the nematode. Possession of specific digestive enzymes may determine if a nematode can parasitize higher plants or fungi or both. RESPIRATION Both plant- and animal-parasitic nematodes require oxygen. Animal parasites consume oxygen at a constant low rate, despite the ambient oxygen concen- tration, and build up an oxygen debt under low oxygen tensions. Nematodes may move actively in the presence of low oxygen, but such conditions may inhibit development and egg hatch.
THE PHYSIOLOGY OF NEMATODES IN RELATION TO CONTROL 29 Recent studies with plant-parasitic nematodes demonstrated that species differ in sensitivity to lack of oxygen. Egg production and survival of molting nematodes and males were most sensitive to low oxygen tension. Hatch of eggs was less sensitive and movement and survival of preadult and mature females were least sensitive to low oxygen. Control of nematodes through flooding of soil may be partially due to lowering of oxygen levels below those required for nematode survival. Oxygen consumption by intact specimens of several species of plant- parasitic nematodes has been measured by microrespirometry. Rates of oxygen uptake varied according to nematode species, condition of the nematodes, and carbon dioxide and osmotic concentrations of the incubation solution. Fresh homogenates of animal-parasitic nematodes initially consumed oxygen far more rapidly than did intact specimens, but the rate of uptake soon dropped off. This difference was probably caused by a low rate of diffusion of oxygen into intact nematodes. TEMPERATURE Since nematodes lack a means of controlling body temperature, metabolic and physical activity are dependent on the ambient temperature. In general, the optimum temperature for growth and development of plant-parasitic nematodes is in the range of 5 to 30°C. Desiccated nematodes can withstand both higher and lower temperatures than fully hydrated ones, probably be- cause enzymes are more resistant to temperature inactivation in the dehy- drated than in the hydrated condition. Moisture plays an important role in determining the thermal stresses that nematodes can withstand. In addition to its direct effects on survival, temperature influences other aspects of nematode biology. Infectivity, sex determination, and rate of de- velopment of plant-parasitic nematodes are affected by temperature. Cysts of the oat cyst nematode (Hetemdera avenae) are reported to require a period of exposure to low temperatures before eggs will hatch. In soybeans grown at 24°C, the developing soybean cyst nematodes (H. glycines) are mostly fe- males, while at 31°C they are mostly males. A few days of exposure of young infected plants to 35 °C followed by growth at 24° C was sufficient to cause most of the nematodes to develop as males. Root-knot nematodes (Meloido- gyne spp.) produce few viable eggs above 35°C, whereas 25 to 32°C is optimum for egg production; however, the golden nematode (Heterodera rosto- chiensis) develops and produces eggs most rapidly at 18°C. Optimum temper- atures for development of the host plant, as well as fluctuating temperatures, may also affect the rate of development of nematodes. Although temperature may influence nematode biology directly, other environmental factors may
30 FACTORS INFLUENCING NEMATODE CONTROL influence the effect of temperature. Advantage is taken of the temperature tolerance of nematodes when control measures such as hot-water treatments of nematode-infected plant material, heat sterilization of soil, and early plant- ing of crops to escape nematode damage are utilized. MOISTURE The natural environment of nematodes is aquatic. The soil solution that covers soil particles and is in soil pores is the medium in which nematodes live and through which they move to contact plant tissues. It is also the medium in which the physiological functions of nematodes occur, such as gas exchange and discharge of excretory products. For nematodes inside plant tissues, ample solution is available to satisfy their requirements. Water is also the medium in which nematocidal chemicals come in contact with the nematode body. Water moves freely into and out of the nematode body through the cuticle, although recent findings show that nematodes have at least some control over loss of body water under conditions of water stress. However, when desiccated, eggs and tanned cysts are resistant to water loss. Under conditions of ample moisture, solutes in the nematode body cavity maintain a constant turgor pressure within the body. All nematodes can probably withstand some water stress, but the degree of water stress which is lethal varies among species. Nematodes inhabiting the aboveground parts of plants, as well as certain soil- inhabiting nematodes such as the cyst stage of some Heterodera spp., are especially resistant to desiccation. Some nematodes will survive freeze-drying, while other species are killed when soil containing them is air-dried. When the moisture content of soil approaches the wilting point for plants, activity of nematodes in soil is curtailed. Desiccation, heat, and starvation are involved when fallow is used to control nematodes. Most free-living and plant-parasitic nematodes withstand severe and re- peated osmotic changes with little or no adverse effects. In hypertonic solu- tions, nematodes shrink and become inactive because of loss of water, but, when transferred to isotonic or hypotonic solutions, they quickly become turgid and active again. Eggs are not plasmolyzed in hypertonic solutions. Al- though solutions of high or fluctuating osmotic concentration may not kill nematodes, movement and egg hatch are inhibited at relatively low solute concentrations. Solute concentrations of 0.1 to 0.2 M inhibit the hatch of eggs of several plant-parasitic nematodes. Ions seem to move into and out of the nematode body with changes of osmotic concentration of the ambient solution, but movement of particular ions is somewhat selective. Specimens of the animal parasite, Ascaris sp., regulate the concentrations of certain ions
THE PHYSIOLOGY OF NEMATODES IN RELATION TO CONTROL 31 in the body fluid, particularly potassium, calcium, and magnesium. Although water and certain ions pass directly through the cuticle of all nematodes, the excretory system appears to regulate the osmotic concentration of the body fluid as well as functioning in the elimination of certain body wastes. Radio- active tracers may aid in elucidating mechanisms of controlling osmotic con- centration in nematodes. Finally, it must be remembered that water interacts with other physical and chemical factors affecting nematodes, as was de- scribed earlier. Soil is a complex medium in which the dynamic interaction of various physical and chemical factors combine to influence all aspects of nematode activity. DORMANCY AND LONGEVITY Several species of plant-parasitic nematodes become dormant during periods of unfavorable environmental conditions and then revive when conditions become favorable again. Nematodes that are parasitic on the aboveground parts of plants survive repeated periods of unfavorable conditions enroute to tissues upon which they will feed. Plant tissues suitable for growth and re- production of the wheat nematode (Anguina tritici) are available only for the short time of flower embryo development, after which weeks or months may elapse before plant tissues are again available. Larvae of the wheat nematode have been revived from galls after 28 years of storage, stem nematodes (Ditylenchus dipsaci) after 23 years of storage in infected plant material, and bentgrass nematodes (Anguina agrostis) after 4 years of storage. Quarantines, the use of nematode-free propagative materials, and rotations including non- host or resistant plants are some measures that are often used to control these types of nematodes. The developmental stage in which nematodes survive periods of dormancy varies with the species. Only second-stage larvae of the wheat nematode sur- vive dormancy, while in the stem, bud, and leaf nematodes (Aphelenchoides spp.) the resistant stages are the fourth-stage preadult larvae and the fifth-stage adults, respectively. Larvae inside eggs in cysts of the golden nematode and the sugar-beet nematode (Heterodera schachtii) remain alive for years when stored dry on the laboratory shelf. Larvae in cysts of certain other cystforming species are less resistant to drying. Dormant stages of nematodes are particu- larly resistant to the action of nematocidal chemicals because of the protec- tive cyst wall in cyst nematodes and the lowered metabolic activity or other physiological factors in both cyst and other nematode species. In the presence of growing host plants in a suitable environment, most nematodes can be maintained indefinitely. Longevity in the absence of host plants, an important factor in nematode control, is greatly influenced by
32 FACTORS INFLUENCING NEMATODE CONTROL environment. Nematodes of many species can survive in soil for at least a year in the absence of a suitable host, probably on body food reserves. In soil, nematodes survive over a wide temperature range, but they survive longer at low than at high temperatures. This is caused primarily by slow depletion of body food reserves and low metabolic rate. Certain plant-parasitic nematodes, such as the golden nematode, persist for years in soil in the absence of host plants. However, in some of the reports, total exclusion of possible hosts, such as particular weeds, that might main- tain populations was not certain. Populations of some cyst nematodes in soil without host plants declined at a rate of 35 to 60 percent per year, whereas populations of other types of nematodes were reduced by 75 to 95 percent in the first year. In most experiments, a few nematodes were still being re- covered from soil when the tests were terminated. The reduction of popula- tions of plant-parasitic nematodes in fallow soil thus follows a hyperbolic curve. Yet, even after extended periods of time, a few specimens, which are important for perpetuation of the species, may survive to serve as inoculum to rebuild populations once host plants are again available. HATCHING, MOLTING, GROWTH, AND SEX DETERMINATION Eggs of most nematode species hatch freely in water, soil solution, or other aqueous solutions of low osmotic concentration. However, hatch of eggs of certain cyst nematodes is greatly increased by incubation in solutions leached from the host-plant root system. This hatching factor has been studied most intensively with the golden nematode, in which the hatching reaction is highly specific. The cabbage cyst (Heterodera cruciferae), hop cyst (//. humuli), and carrot cyst (H. carotae) nematodes also respond to specific hatching factors. Egg hatch in several other cyst nematodes and some root-knot nematodes also may be stimulated by root exudate, but appreciable hatch occurs in water alone. In some cases, root exudate from nonhost plants stimulates egg hatch, and investigations indicate that more than one hatching factor occurs in some root exudates. In addition to substances in root exudates, many laboratory chemicals will stimulate the hatch of nematode eggs, but never to the degree obtained with root exudates. Chemicals stimulating hatch include various dyes, amino acids, sugars, and inorganic salts. No means of nematode control utilizing artificial stimulation of egg hatch has yet been developed. When an egg begins to hatch, the larva moves about within the eggshell, at which time enzymes secreted by the larva are thought to hydrolyze the inner layer of the eggshell. Destruction of this layer allows entrance of water into
THE PHYSIOLOGY OF NEMATODES IN RELATION TO CONTROL 33 the egg. This is then taken up by the larva and causes it to swell. Finally, it seems that a combination of enzymatic action weakening the eggshell and mechanical activity of the enlarged larva, both through bodily movement and stylet thrusts against the shell, are sufficient to rupture the eggshell and free the nematode larva. Hatching factors are thought to act by increasing the permeability of the eggshell, thereby hastening activation of the larva. The stimuli that induce molting in nematodes have been little investigated. Growth and molting of many plant-parasitic nematodes require feeding on plant tissues, but in species such as the reniform nematode (Rotylenchulus spp.), eggs will hatch and the nematodes will grow and molt to the preadult stage in a dish of water in the absence of host tissues. Complete development of adults and production of viable eggs, however, require feeding on host tissues. Molting, or exsheathing, in animal parasites seems to require enzymes produced by the nematode. The process of molting in animal- or plant- parasitic nematodes is not well understood, although certain root diffusates stimulated molting in some of the pin nematodes (Paratylenchus spp.). When food is in ample supply, growth in nematodes is a continuous process except during molts. The cuticle can make limited growth between molts. Since nematodes are determinate in cell number, growth is primarily ex- pressed as an increase in cell size. Growth may be accompanied by changes in body shape of the females, such as those that occur in root-knot, cyst, reni- form, and several other nematodes; but, in most kinds of nematodes, males and females remain vermiform throughout the life cycle. Sex determination in nematodes seems to be governed by both genetic and nutritional mechanisms. Results of several studies indicate that when the food supply becomes limited the sex ratio is shifted toward males. Males often predominate in nematode populations developed in the presence of resistant plants, which may be a reflection of nutritional deficiency. Possible control of sex by hormones in plant-parasitic nematodes has not been investigated. NUTRITION AND LABORATORY CULTURING Several free-living and animal-parasitic nematodes, and one stylet-bearing form, have been propagated in vitro. No nematode has been propagated on a completely defined medium, despite intensive efforts toward this goal. All media that support in vitro propagation of free-living nematodes have failed to support reproduction of plant-parasitic species. The free-living nematode Caenorhabditis briggsae is known to require six B-vitamins and ten amino acids. Although all plant-parasitic nematodes require living plant tissues for re- production, there is great variation in the host ranges of different nematodes.
34 FACTORS INFLUENCING NEMATODE CONTROL For instance, the golden nematode will reproduce only on a very few plants, while the host plants of certain root-knot nematodes number in the hundreds. Attempts at in vitro culturing of plant-parasitic nematodes would be most likely to succeed with species that do not induce specialized plant reactions and that have wide host ranges. Laboratory culturing of plant-parasitic nematodes on a scale adequate for biochemical studies has been successful with about 12 nematode species. Sterile root cultures, grown on nutrient agar media, were first utilized for culturing nematodes. Plant callus cultures, particularly of alfalfa, proved highly useful. This culturing technique is presently the most promising method of propagating large quantities of plant-parasitic nematodes in the laboratory. CONCLUSIONS The physiology and biochemistry of plant-parasitic nematodes is only now beginning to receive proper attention. To aid biochemical studies, supporting techniques such as those involving culturing and handling of many kinds of plant-parasitic nematodes still need to be developed. Indications are that as this area of research expands, novel and perhaps surprising variations from conventional metabolic patterns, which will contribute basic information not only to nematology but also to fundamental biochemistry, will be elucidated. Results of biological studies with plant-parasitic nematodes have indicated some very interesting phenomena, which, however, can only be explained by future biochemical and physiological investigations. Progress in biochemical nematology is presently hampered not only by a shortage of investigators but also by a lack of suitable positions and facilities for some of the few competent, enthusiastic, highly trained young graduates. Until more support is provided for such research, progress in this area will continue to be slow. BIBLIOGRAPHY Dougherty, E. C. 1960. Cultivation of Aschelminths, especially Rhabditid nematodes, pp. 297-318. In J. N. Sasser and W. R. Jenkins (eds.). Nematology, fundamentals and recent advances. Univ. North Carolina Press, Chapel Hill. 480 pp. Fairbairn, D. 1960. The physiology and biochemistry of nematodes, pp. 267-296. In J. N. Sasser and W. R. Jenkins (eds.). Nematology, fundamentals and recent ad- vances. Univ. North Carolina Press, Chapel Hill. 480 pp. Franklin, M. T. 1951. The cyst-forming species of Heterodera. Commonwealth Agr. Bur.. Famham Royal, England. 147 pp.
THE PHYSIOLOGY OF NEMATODES IN RELATION TO CONTROL 35 Jones, F. G. W. 1959. Ecological relationships of nematodes, pp. 395-411. In C. S. Holton, G. W. Fischer, R. W. Fulton, H. Hart, and S. E. A. McCallan (eds.). Plant pathology, problems and progress. Univ. Wisconsin Press, Madison. 588 pp. Krusberg, L. R. 1963. Host response to nematode infection. Ann. Rev. Phytopathol. 1:219-240. Lee, D. L. 1965. The physiology of nematodes. Oliver & Boyd, London, 154 pp. Rohde, R. A. 1960. Mechanisms of resistance to plant-parasitic nematodes, pp. 447-453. In J. N. Sasser and W. R. Jenkins (eds.). Nematology, fundamentals and recent ad- vances. Univ. North Carolina Press, Chapel Hill. 480 pp.
CHAPTER Pathogenic Relationships NEMATODE DISEASE SYMPTOMS AND DISEASE DIAGNOSIS The symptoms of nematode disease are commonly those of root impairment, such as growth reduction (Figure 5), increased wilting, mineral-deficiency symptoms, decreased winter-hardiness, and dieback in perennials. It is often difficult to prove conclusively whether nematodes, other microorganisms, other limiting factors, or combinations of these are the cause of root impair- ment. Before discussing the solution of this difficulty, however, those nema- tode disease symptoms that are easily recognized in plants should be considered. Top Symptoms and Signs Certain species of seedgall nematodes (Anguina spp.) transform floral parts, producing characteristic galls in place of normal seeds (Figure 6). Other species of Anguina produce galls and distortion in leaves and stem. The stem nematode (Ditylenchus dipsaci) causes swelling and distortion of stems and leaves (Figure 7). Bud and leaf nematodes (Aphelenchoides spp.) frequently cause foliar discoloration in a characteristic pattern (Figure 8). Where symp- toms suggest nematode infection, the actual presence of nematodes should be determined by dissecting or extracting nematodes from the tissues and identi- fying them. 36
PATHOGENIC RELATIONSHIPS 37 FIGURE 5 Reduced growth of corn associated with the stubby-root nematode, Trichodorus christiei. (After J. R. Christie. Courtesy of Phytopathology.) Root Symptoms and Signs The galling (Figure 9) caused by root-knot nematodes (Meloidogyne spp.) is easily recognized but can be confused with the more apical root-galling (Figure 10) caused by certain sheath nematodes (Hemicycliophora spp.) or with the bending and apical galling caused by dagger nematodes (Xiphinema spp.). Lesion nematodes (Pratylenchus spp.) produce characteristic lesions in the root cortex of some host plants (Figure 11). Female cyst nematodes (Heterodera spp.) can be seen on the roots of host plants (Figure 12) if the soil is carefully removed from the roots. Soil clings to a gelatinous material secreted by the citrus nematode (Tylenchulus semipenetrans), causing infected citrus roots to appear "dirtier" than uninfected ones. The stubby roots, ex- cessive root proliferation, and root necroses that accompany root infection by other kinds of root-feeding nematodes are often not sufficiently distinctive to permit sure diagnosis.
38 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 6 A normal seed head of wheat (left), and a head with seeds galled (right) by the wheat-gall nematode, Anguina tritici (Courtesy of Shell Development Company.) EXPERIMENTAL DETERMINATION OF PATHOGENICITY In general, because nematodes are unobtrusive plant parasites, their patho- genicity must be established experimentally. The procedures used to establish pathogenicity attempt to parallel those outlined in Koch's postulates. This at- tempt is not entirely successful, for two reasons. First, nematodes parasitic on higher plants are obligate parasites. Second, and more important, the activi- ties of most plant-parasitic nematodes occur in soil, a microbiologically com- plex medium. Nematologists are circumventing the first difficulty. Some of the more important plant-parasitic species are now maintained in plant-tissue cultures free of all other microorganisms. Large numbers of microbiologically sterile nematodes for experimental use can be obtained from such cultures. In time, this approach will probably succeed with many additional species. The second difficulty, extrapolation from results of simple experiments in- volving only nematode and plant to the complex field situation has not been
PATHOGENIC RELATIONSHIPS 39 overcome. This difficulty is shared by plant pathologists working with other soil microorganisms. In the absence of a satisfactory stepwise series of tests, such as Koch's rules of proof, nematologists make observations and conduct experiments to judge association with disease, include experimental treatments that will test involvement in disease, and conduct still other experiments to decide the role of a nematode in a disease. To judge association of a nematode with a plant disease, the nematode must first be identified to species. Nematodes show greater morphological specialization then do bacteria and fungi, and identification can usually be made with greater precision, although physiological races and morphologically similar species do occur. Specimens can be killed with hot water and pre- served in formalin or passed into glycerine and mounted for future reference. Methods for recovering and preserving nematodes are discussed in Chapter 7. Observation of the nematode feeding on or in the host is useful at the associa- tion stage. Differences in nematode population levels can be created by soil fumigation, by previous cropping, or by adding nematodes to uninfested soil. Subsequent plant growth in soil thus treated can be correlated with nematode population level. When adding nematodes to soil, it is useful to apply them in numbers on a logarithmic scale (e.g., 100; 1,000; 10,000; and 100,000). Such a FIGURE 7 Distortion of onion by the stem nematode, Ditylenchus dipsaci; one healthy onion on the left. (Courtesy of the Department of Nematology, University of California, Davis.)
40 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 8 Angular discoloration produced by the chrysanthemum foliar nematode, Aphelenchoides ritzemabosi, in chrysanthemum leaves. (Courtesy of Nematology Investi- gations, USDA.)
PATHOGENIC RELATIONSHIPS 41 *« . - - / . ⢠FIGURE 9 Galling of carrots by a root-knot nematode, Meloidogyne incognita, (Courtesy of the Department of Nematology, University of California, Riverside.)
42 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 10 Citrus root galled by the sheath nematode, Hemicydiophora arenaria. (After S. D. Van Gundy. Courtesy of Plant Disease Reporter.)
PATHOGENIC RELATIONSHIPS 43 FIGURE 11 Lesions on walnut root associated with a lesion nematode, Pratylen- chus vulnus. (Courtesy of the Department of Nematology, University of California, Davis.) FIGURE 12 Mature females of the golden nematode, Heterodera rostochiensis, on potato roots. (Courtesy of the Department of Plant Pathology, Cornell University.)
44 FACTORS INFLUENCING NEMATODE CONTROL series approximates various stages during the natural increase in a nematode population with time and enables determination of both the tolerance limit of the plant in terms of nematode numbers and the nematode population density that can maintain itself on the plant being tested. Depending on the similarity between field and experimental conditions, this information may be applied to field situations. Observations and experiments used to judge association with disease usually include either microorganisms other than nematodes or soil treatments that have microbiological or chemical effects in addition to the nematocidal effect. The data from these experiments can be used to judge association with disease of unknown cause but cannot be used to prove in- volvement in disease or its pathogenicity. Involvement of a nematode species in a plant disease may be judged by comparing plants inoculated with a water suspension of nematodes isolated from roots or soil with plants inoculated with an otherwise similar suspension but freed of nematodes by a technique such as sieving. Freeing the suspen- sion of nematodes by sieving provides a valid check on associated micro- organisms if these microorganisms pass through the sieves that are employed. The experimental goal would not be attained, for example, if fungus inoculum was present in the suspension in the form of mycelium, since mycelium, as well as the nematodes, would be removed by sieving. Bacteria and many fungus spores will pass through the fine sieves used in nematode extraction. If removal of the nematode (and nothing else) from the inoculum eliminates disease, that nematode must be involved in the disease. Other organisms may also be involved. Whether the nematode is a pathogen, an incitant, an aggra- vator, or a vector must be determined by further and different experimentation. The role of a nematode in plant disease can only be proved with nematodes that are freed of other microorganisms. This can often be accomplished by surface sterilization of the nematodes with chemicals, if the nematode sus- pension is free of organic fragments. Because surface sterilization frees plant- parasitic nematodes from other microorganisms, and because attempts to isolate organisms from inside these nematodes have usually failed, most plant- parasitic nematodes are believed to be internally free of live microorganisms. An exception may be the American dagger nematode (Xiphinema ameri- canum), in whose reproductive tract a bacterium was reported. Considered singly, studies of association, involvement, or role usually will not prove satisfactorily that a nematode is or is not the cause of a disease as it occurs in the field. Consideration of observations and results of experiments at all three of these levels are usually required to understand the relation of a nematode to a plant disease, but disease-control efforts need not await com- plete understanding of the disease. The initial judgment of association, and attempts at control by soil fumigation, often proceed together profitably.
PATHOGENIC RELATIONSHIPS 45 HISTOPATHOLOGY OF NEMATODE-PARASITIZED PLANTS Microscopic study of the tissue and cellular alterations associated with para- sitism is termed histopathology. Details of histopathological changes induced in tissues by nematode parasitism, starting with initial stages of parasitism and continuing until the interrelationship is well established, must be correlated with physiological and biochemical studies. By correlating findings from these studies, it may be possible to determine the fundamental basis for plant- tissue alterations that occur during nematode parasitism. For convenience, the gross symptoms of nematode parasitism of plant tissues may be separated into galling; necrosis, or death, of cells or tissues; distortion; and inhibition of growth. In many nematode-plant-tissue associa- tions a single symptom may be observed, but, in others, two, three, or all four symptoms may be seen. For instance, galling is the dominant symptom of infection in alfalfa seedlings by the stem nematode, but inhibition of apical growth and distortion of shoots may occur simultaneously with galling, and in older infections necrosis may develop. However, the only gross visible symp- tom of tobacco stunt nematode (Tylenchorhynchus claytoni) damage to plant roots may be retarded growth of the root system because of the inhibi- tion of growth of many root apices. Gall Formation Species of nematodes in several genera induce swellings, or plant galls, both in roots and aboveground parts of plants. The site of galling on the plant, i.e., seed, leaf, stem, or root, is determined by the feeding site of the particular nematode species involved. A plant gall possesses certain anatomical features that are more or less peculiar to the nematode species inducing the gall, al- though these features may vary somewhat, depending on the particular plant involved and environmental conditions such as soil type, fertilizer levels, moisture, and temperature. Perhaps the simplest types of plant galls induced by nematodes are those caused by dagger and sheath nematodes on the root apices of certain plants. These nematodes feed as ectoparasites, with only the stylet, and not the nema- tode body, penetrating plant tissues. Feeding occurs on a root apex, stimulat- ing hyperplasia (cell division) and hypertrophy (cell enlargement) of root-tip cells, which results in a gall composed of a compact mass of cells (Figure 13). Galls induced by the stem nematode on various plants and different above- ground parts are essentially similar histopathologically. Gall formation in alfalfa seedlings is well documented and serves as the example here (Figures 14 and 15). Within a few hours after inoculation, nematodes penetrate the epidermis in the axils of cotyledons, forming cavities in the cortical tissue just
46 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 13 Response of citrus-root cortical parenchyma tissue to feeding by a speci- men of Hemicycliophora arenaria shown with stylet penetrating plant root (arrow). (After S. D. Van Gundy. Courtesy of Phytopathology.) below the plant apex. One day after inoculation, swelling of the stem below the cotyledons is visible. Further gall development is principally an amplifica- tion of earlier anatomical changes. Galls enlarge by hyperplasia and hyper- trophy of cortical parenchyma cells, while nematodes are simultaneously destroying cortical cells and thus forming cavities. Galls may continue to en- large while the plant is actively growing. Many of the undestroyed paren- chyma cells become enlarged, misshapen, and separated from one another. Xylem tissue is little affected directly. Apical growth is usually inhibited, and, although nematodes do not penetrate the apex itself, these cells may become distorted when adjacent to galls. Although these nematodes penetrate roots in small numbers and form cavities in the cortex, no galling is induced.
PATHOGENIC RELATIONSHIPS 47 FIGURE 14 Cross section of an alfalfa stem galled by Ditylenchus dipsaci, 20 days after nematode inoculation (After L. R. Krusberg.)
48 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 15 Cross section of a normal alfalfa stem. (After L. R. Krusberg. Reproduced by permission of E. J. Brill.) The beach-grass root nematode, Ditylenchus radicicola, causes galls on roots of several grasses (Gramineae). These galls are determinate and develop only to specific size. The galls are histopathologically similar to galls caused by the stem nematode in aboveground parts of plants, except that hyper- trophy and hyperplasia are more pronounced (Figure 16). The most common and well-known gall-inducing nematodes are the root- knot nematodes. Second-stage larvae penetrate the epidermis near the root tip, and within 24 hours they move between or through cells to the feeding site in the vascular cylinder. A root tip may be penetrated by several larvae, in which case apical growth is usually inhibited. When larvae begin feeding, two to five cells in the vascular tissue around the head of the nematode enlarge, and nuclei within these cells may divide several times without division of the cytoplasm, thus resulting in multinucleate cells (Figure 17). Under the con- tinued influence of nematode feeding, walls between enlarging cells and ad- jacent cells dissolve, the protoplasts combine, and, as additional surrounding cells are incorporated, so-called giant cells are formed (Figure 18). Giant cells continue to enlarge for several days or weeks, and they provide food for the
PATHOGENIC RELATIONSHIPS 49 B FIGURE 16 A. Cross section of a normal root of Elymus arenarius. B. Cross section through a gall induced by Ditylenchus radicicola (N) on a root of E. arenarius. (After T. Goodey. Reproduced by permis- sion of/. Helminthol.) developing nematode. Typically, developed giant cells are multinucleate and contain viscous cytoplasm with many mitochondria, proplastids, and golgi bodies. They possess a well-developed endoplasmic reticulum and are very active metabolically. Walls of giant cells are usually thick except in areas where adjacent cells are being incorporated. Living nematodes are requited for initia- tion and continued development of giant cells. Giant cells may also form from cortex or storage parenchyma cells as well as from vascular tissues. Simultaneous with giant-cell formation, other changes occur in plant tissues surrounding the feeding root-knot nematode. Hypertrophy and hyper- plasia occur in the pericycle, cortex, and epidermis adjacent to the enlarging nematode, so that it is continually enveloped by plant tissues (Figure 18). As the nematode matures in small roots, the posterior portion extends close to the epidermis, so that extruded eggs lie outside the plant root. When egg-laying is nearly completed, degenerative changes such as granulation of giant-cell cytoplasm, dissolution and coalescence of giant-cell nuclei, cork formation around the nematode and giant-cell complex, and pronounced thickening of giant-cell walls may occur in gall tissues. When females die or mature males
50 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 17 Initiation of giant cells around the head of a larva of Meloidogyne incognita acrita in a sweet-potato root 3 days after nematode inoculation. Figure shows nematode larva (N) and initial formation of giant cells (GC). (After L. R. Krusberg. Courtesy of Phytopathology.) leave roots, there Is evidence that the plant resorbs the galled tissues to some degree. Cyst nematodes also induce giant cells in plant tissues, but galls are not formed, as there is little or no hypertrophy or hyperplasia in cells surrounding the nematode. Instead, the nematode breaks out of the root as it grows, so that only the head and neck are embedded in the root when the nematode reaches maturity (Figure 19). Giant cells induced by root-knot and cyst nematodes are similar in most respects. Leaf and seed galls induced by Anguina spp. vary in complexity. In general, a central cavity containing nematodes is surrounded by a few layers of paren- chymatous cells, which appear to serve as the source of food. Layers or groups of other types of cells, such as sclerenchyma, may surround the parenchyma tissue (Figure 20). Galls are determinate, and they form by hypertrophy and hyperplasia of parenchyma cells at the infection site. Nematodes of a few other genera, such as Nacobbus, Meloidodera, and Hypsoperine, also induce root-galling, which generally follows one or another of the patterns described above.
PATHOGENIC RELATIONSHIPS 51 FIGURE 18 Cross section of a tomato root gall showing a female, Meloidogyne sp. (N), giant cells (GC), and other abnormal tissues in the root. (Reproduced by permission of Rothamsted Experiment Station.)
52 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 19 Longitudinal section of a soybean root infected by a female soybean cyst nematode, Heterodera glycines, showing position of the nematode (N) and giant cells (GC) in root tissues. (After B. Y. Endo. Courtesy of Phytopathology.)
PATHOGENIC RELATIONSHIPS 53 FIGURE 20 Cross section through a gall induced byAnguina cecidoplastes in the lamina of a leaf of Andropogon pertusus, showing: MR, midribs; NC, nutritive cells; PVB, primary vascular bundles; S, sclerenchyma cells; SVB, secondary vascular bundles; and TC, thickened cells. (After T. Goodey. Reproduced by permission of/. Helminthol.) Lesion Formation Nematodes of several genera, including ectoparasitic and endoparasitic species, cause lesions in roots or aboveground parts of plants. Except in a few in- stances, little is known about progressive histopathological changes occurring in host tissues during penetration of nematodes into plants and establishment of the parasitic relationship. Lesion nematodes apparently penetrate almost anywhere along the roots. In the cortical parenchyma, the nematodes are present in cavities formed by
54 FACTORS INFLUENCING NEMATODE CONTROL their feeding. Root tissues of many but not all plants discolor quickly follow- ing nematode infection. In heavily infected roots, the cortex surrounding the vascular tissue may become permeated by cavities, resulting in sloughing off of the cortex and, finally, death of the root. Secondary microorganisms, such as bacteria and fungi, may aggravate root necrosis. Surface-sterilized lesion nematodes produce necrosis in sterile root tissues, but necrosis caused under field conditions is usually more extensive. The burrowing nematode (Radoph- olus similis) causes lesions in the cortical parenchyma of citrus roots similar to those caused by lesion nematodes, feeds on phloem and pericycle tissues, and may cause slight swelling of roots due to stimulation of cell division in the pericycle (Figure 21). A lance nematode (Hoplolaimus coronatus) also burrows into the cortex of roots, usually feeding in phloem tissue and occa- sionally penetrating xylem tissue. The potato rot nematode (Ditylenchus destructor) causes rot in under- ground parts of several plants. In potato tubers, nematodes feed on paren- chyma cells, creating a network of tunnels through the tissues and leaving vascular strands intact. Infected tissues shrink and crack, allowing secondary microorganisms to enter and add to the destruction. Potato tubers are success- fully colonized by the potato rot nematode only in the presence of fungi, al- though this nematode can be cultivated on callus cultures of several plants in the absence of fungi. The citrus nematode feeds primarily on cortical parenchyma cells of citrus- feeder roots. Occasionally, several second-stage larvae may enter a citrus root and form a cavity in the cortex. Usually, a second-, third-, or fourth-stage larva becomes partially embedded in a root, with the head several cells deep in the cortex, where it feeds on cortical parenchyma while maturation occurs. Only cells directly penetrated by the nematode body are destroyed; the cells on which the nematode feeds are altered but not destroyed (Figure 22). Localized cortical necrosis of plant roots is caused by spiral (Helicotylen- chus spp. and Rotylenchus spp.), pin (Paratylenchus spp.), ring (Criconemoides spp.), and other nematodes. These nematodes generally penetrate only a few cell layers into cortical tissues. The red ring disease of coconut palm, caused by Rhadinaphelenchus cocophilus, is characterized by discoloration in stems of this plant (Figure 23). Browning of chrysanthemum leaves is caused by infection with chrysan- themum foliar nematodes (Aphelenchoides ritzemabosi). Only older leaves become diseased, probably because intercellular spaces in the mesophyll are large enough to permit free movement of the nematode. Nematodes enter young leaves through the stomata but are restricted to the substomatal chamber and do not cause damage. Within a few days after infection, large numbers of mesophyll cells in the older leaves are destroyed by nematode feeding, causing large sections of leaf tissues to die, turn brown, and collapse.
PATHOGENIC RELATIONSHIPS 55 FIGURE 21 A cross section of a grapefruit root infected by the burrowing nematode, Radopholus similis, showing cavities in cortical parenchyma containing nematodes (N). (After DuCharme. Courtesy of Phytopathology.)
56 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 22 Cross section of a sour orange root, showing alteration of cells in the cortical parenchyma tissue by the citrus nematode, Tylenchulus semipenetrans (N). (After S. D. Van Gundy.) Inhibition of Apical Growth Inhibition of apical growth of plant tissues may accompany galling or necrotic responses to nematode parasitism. However, growth inhibition may be the only visible response to nematode feeding. For example, the awl (Dolicho- dorus spp.), sting (Belonolaimus spp.), and stubby-root nematodes (Tricho- dorus spp.) inhibit apical growth caused by ectoparasitic feeding on root apices. Cells in affected apices, including meristematic cells, become mature, enlarged, and vacuolate. In the presence of dense nematode populations, root apices may fee distorted and destroyed. Feeding by a stubby-root nematode (T. christiei) stimulates formation of lateral roots behind affected root tips. As these lateral roots emerge through the cortex, they also are attacked by nematodes, thus finally resulting in a severely restricted plant root system, consisting of numerous short, stubby roots (Figure 24). Stunt nematodes (Tylenchorhynchus spp.) and sometimes pin nematodes elicit a milder response in roots of many plants than do sting or stubby-root nematodes. Such plants, when grown in the presence of dense nematode popu- lations, merely have smaller root systems than do plants grown in nematode- free soil. These nematodes apparently feed on plant cells and reduce root
PATHOGENIC RELATIONSHIPS 57 FIGURE 23 Discoloration in the lower stem of a coconut palm, caused by Rhadinaphelenchus cocophilus. growth without destroying cells. No histopathological evidence of feeding was found in sections of alfalfa roots on which specimens of tobacco stunt nematode were microscopically observed feeding on epidermal cells. Certain nematodes feeding ectoparasitically near meristematic tissues of aboveground plant parts may also inhibit apical growth. Apical growth was stopped, and apical cells matured and became highly vacuolated, when alfalfa seedlings were inoculated with chrysanthemum foliar nematodes (Figure 25). This and other species of Aphelenchoides not only inhibit apical growth but also kill epidermal cell layers and cause malformation of young tissues.
58 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 24 Root system of a corn plant injured by the stubby-root nematode, Trichodorus christiei. (After J. R. Christie. Courtesy of Soil Science Society of Florida.) PHYSIOLOGY OF DISEASED TISSUES An increase in the respiratory rate of tissues is characteristic of plant diseases caused by fungi and bacteria. Because nematode-diseased tissues are reported to absorb oxygen more rapidly, at the same rate, or less rapidly than com- parable healthy plant tissues, no generalizations about the effect of nematodes on the respiratory rate of plant tissues are possible. Increased activity of oxidase enzymes, including the oxidases that destroy the plant growth hormone, indole-3-acetic acid (I AA), was found in extracts of plant galls caused by stem and root-knot nematodes. Solutions in which live nematodes were incubated also contained lAA-destroying enzymes. Such enzymes may be involved in inhibition of apical growth in plants affected by nematodes. Recent studies have elucidated some of the biochemical changes resulting from nematode infection of plant tissues. Results of histochemical tests on root-knot nematode galls demonstrate that giant-cell walls contain cellulose and pectin but no lignin, suberin, starch, or ninhydrin-positive substances. However, giant-cell protoplasm contains carbohydrate, fat, ribonucleic acid, and a large amount of protein. Starch disappears in galled tissues, while cellu- lose, sugars, phosphorylated intermediates, keto acids, free amino acids, protein, nucleic acids, phosphorus, and nitrogen increase when compared with
PATHOGENIC RELATIONSHIPS 59 V- MB' -\A rf337c ...â¢â¢ - FIGURE 25 Longitudinal section through the apical region of an alfalfa seedling inoculated with Aphelenchoides ritzemabosi, showing necrotic-epidermal and outer-cortical parenchyma tissue (NT), caused by nematode feeding. (After L. R. Krusberg. Reproduced by permission of E. J. Brill.) healthy tissues. Levels of several metals and sulfur remain the same as in com- parable healthy tissues. These findings, as well as results from histopathologi- cal studies, indicate that root galls induced by root-knot nematodes are very active metabolically. Levels of certain biochemicals in plant tissues affected by other nematodes were investigated. Free and protein amino acids accumulated more rapidly
60 FACTORS INFLUENCING NEMATODE CONTROL and to higher levels in shoot tissues of alfalfa and pea infected with stem nematode and in citrus roots infected with burrowing nematode than in com- parable healthy tissues, indicating stimulated synthesis of these substances. Plant roots infected with Nacobbus batatiformis accumulated starch, whereas in other nematode-diseased tissues starch usually disappears. In a few instances, radioactive tracers were used to study metabolism of root-knot nematode-galled plant tissues. Accumulation of 32P in galls oc- curred when infected roots were incubated in labeled phosphate solution. Results from recent studies demonstrated the incorporation of tritiated thymidine and uridine into giant-cell protoplasm. Uridine was incorporated into nuclei, nucleoli, and cytoplasm of giant cells regardless of the age of the nematode associated with the giant cells. However, thymidine was incorporated into nuclei of giant cells upon which larvae were apparently feeding at the time of treatment and only into those giant cells associated with growing nematodes, that is, second- to fourth-stage larvae. These results were inter- preted as an indication that DNA was synthesized in giant cells only when nematodes were feeding, but RNA was synthesized independent of nematode feeding. Little tritium was accumulated in nematode bodies. The mechanism of nematode-induced galling of plant tissues obviously in- volves plant-growth regulating mechanisms, but no incitant has been identi- fied. Most investigations concern host-parasite interactions of root-knot and stem nematodes. Based on paper chromatographic and biological activity assays, galled plant tissues were reported to contain more auxin or indole compounds than healthy tissues. In a recent study, eggs and larvae of three species of root-knot nematodes and tomato roots galled by each species con- tained indole plant-growth regulators. Occurrence of each of the four identi- fied growth regulators varied with the particular nematode species involved; those found in a specific nematode were also present in extracts of roots galled by that nematode species. Data from studies with plant-growth inhibi- tors also suggest that the mechanism of nematode-induced galling of plant tissues is growth-regulatory in nature. When applied to the tops of plants, the plant-growth inhibitor, maleic hydrazide, inhibited development of root-knot nematode galls on tobacco roots. The significance of these findings as related to nematode-induced galling has yet to be determined. A few studies have concerned mechanisms by which nematodes induce necro- sis in plant tissues. In peach roots infected by a lesion nematode, Paratylenchus penetrans, browning was due to breakdown of the plant glycoside, amygdalin, by (3-glycosidases of plant and nematode origin to release HCN, which was toxic to the root tissues. Phenolic compounds accumulate in lesion nematode- infected tobacco roots and stored-carrot root tissues, but the identities of these compounds and their mechanisms of formation and accumulation are unknown. In chrysanthemum leaves infected by chrysanthemum foliar nema-
PATHOGENIC RELATIONSHIPS 61 todes, rapid browning was thought to be caused by very active nematodes that quickly punctured and destroyed many mesophyll cells. Such tissues were thought to lack some nutrient that was required by the nematode and that stimulated hyperactive feeding. The rapid browning was interpreted as a sign of plant resistance. In chrysanthemum leaves which browned slowly, it was thought that nematodes were able to obtain required nutrients easily, so cells were punctured and destroyed at a slow rate. The interrelationships of nematode parasitism and mineral nutrition of plants has been the subject of many studies. In general, plants grown under conditions of marginal or deficient mineral supply are more susceptible to nematode injury than plants grown in the presence of optimal or excess mineral-nutrient supply. Nematode parasitism also may alter the balance of mineral elements in a parasitized plant, but the effects of nematode parasitism on specific nutrients in a plant are not well understood. CONCLUSIONS Judgments of nematode pathogenicity should be based on three comple- mentary kinds of evidence: nematode association with disease, nematode in- volvement in disease, and nematode role in disease. The majority of histopathological studies of nematode-plant inter- relationships have concerned tissues in which parasitic relationships were already established at the time of sampling. Although some fine detailed studies have been conducted, more are needed to develop a comprehensive picture of the plant-tissue alterations associated with plant diseases caused by different nematodes. Especially needed are studies of tissue changes dur- ing the critical period of the establishment of the host-parasite relationship; also needed are comparisons of reactions in resistant and susceptible plant tissues. Ideally, histopathological, cytological, and histochemical studies of plant tissues should be conducted simultaneously, and, where feasible, electron microscopy should be used. Such comparative studies, combined with physio- logical and biochemical studies of nematode-diseased plant tissues, should provide the information needed to understand the fundamental basis of nematode parasitism of plants. These kinds of studies should be encouraged. BIBLIOGRAPHY Christie, J. R. 1959. Plant nematodes, their bionomics and control. H. and W. B. Drew Co., Jacksonville, Florida. 256 pp.
62 FACTORS INFLUENCING NEMATODE CONTROL Goodey, J. B. 1963. Laboratory methods for work with plant and soil nematodes. Ministry of Agriculture, Fisheries, and Food. London. Tech. Bull. No. 2. 72 pp. Kirkpatrick, J. D., S. D. Van Gundy, and W. F. Mai. 1964. Interrelationships of plant nutrition, growth and parasitic nematodes. Plant Anal. Fert. Probl. 4:189-225. Krusberg, L. R. 1963. Host response to nematode infection. Ann. Rev. Phytopathol. 1:219-240. Mountain, W. B. 1960. Theoretical considerations of plant-nematode relationships, pp. 419-421. In J. N. Sasser and W. R. Jenkins (eds.), Nematology, fundamentals and recent advances. Univ. North Carolina Press, Chapel Hill. 480 pp. Oostenbrink, M. 1966. Major characteristics of the relation between nematodes and plants. Meded. Landb. Woogesch. Wageningen 66:3-46. Pitcher, R. S. 1965. Interrelationships of nematodes and other pathogens of plants. Helminthol. Abstr. 34:1-17. Wallace, H. R. 1963. The biology of plant parasitic nematodes. Edward Arnold Ltd., London. 280 pp.
CHAPTER 6 Nematode Interrelationships with Other Plant- Disease Organisms FUNGI AND BACTERIA One of the most significant developments in nematology and plant pathology during the last decade has been the demonstration of the many interactions between nematodes and other soil-inhabiting pathogens, including fungi, bacteria, and viruses, in causing plant diseases. The performance of several crop varieties that were selected for resistance to specific fungal or bacterial pathogens in nematode-infested fields figured heavily in this discovery. In- creases in growth of these disease-resistant varieties in fumigated soils over those in nonfumigated soils were surprisingly large. Controlled greenhouse tests proved conclusively that certain plant-pathogenic nematodes greatly en- hance development of diseases caused by fungi and bacteria in plants that are usually resistant to these diseases. Specific examples are the tobacco varieties developed for resistance to the black shank fungus, Phytophthora parasitica var. nicotianae (Figure 26), and the Granville wilt bacterium, Pseudomonas solanacearum (Figure 27), and cotton and tomato varieties resistant to the Fusarium wilts interacting with root-knot nematodes (Meloidogyne spp.). Although various theories of the role of the nematode have been advanced, the mechanisms that render nematode-infected plants susceptible to fungi and bacteria have not been elucidated. In some bacterial diseases, it is thought that endoparasitic nematodes, as they invade roots, merely provide avenues for entrance by bacteria. In some nematode-fungus-plant interactions, the role of the nematode is believed to be more complex than simple wounding of the plant, since wounds are not required for infection of plants by the fungi in- volved. Recent investigations indicate that host physiology is altered by 63
64 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 26 The interaction between the root-knot nematode, Meloidogyne incognita, and the black shank fungus, Phytophthora parasitica var. nicotianae, on the highly resistant black shank tobacco variety Coker 139. The plants on the right were grown in soil inocu- lated with the black shank fungus only; the plants on the left were grown in soil inoculated with the black shank fungus and root-knot nematodes. Root-knot nematodes alone, not shown, caused only slight stunting. (After Powell and Nusbaum. Courtesy of Phytopathology.) FIGURE 27 Dixie Bright 101 tobacco plants 21 days after inocula- tion with Pseudomonas solanacearum and Meloidogyne incognita. Left: root-knot nematodes added at transplanting and P. solanace- arum (concentrated suspension) added 24 hours after transplanting. Right: P. solanacearum alone (concentrated suspension) added to soil 24 hours after transplanting. Root-knot nematodes alone, not shown, caused only slight damage.
NEMATODE INTERRELATIONSHIPS WITH OTHER ORGANISMS 65 nematode infection of plant tissue. For example, the black shank fungus de- velops more vigorously and extensively in tobacco root tissues galled by root- knot nematodes than in nongalled roots of resistant varieties. Investigations on how nematode infections affect host physiology are needed, as well as basic studies on the interactions between nematodes and fungi. Synergistic relationships between phytopathogenic fungi and nematodes in increasing the severity of plant diseases have also been demonstrated. For ex- ample, the incidence and severity of wilt of eggplant, caused by the soilborne fungus Verticillium dahliae, were increased in the presence of a lesion nema- tode, Pratylenchus penetrans, and the number of nematodes within egg- plant roots was increased in the presence of the fungus. In addition to increas- ing the rate of nematode reproduction, fungus-infected roots were more attractive to and were more readily invaded by these nematodes than were noninfected roots. These relationships, however, are not well understood. Re- search is needed to determine the influence of individual environmental factors that affect development of pathogens and disease in plants simultaneously ex- posed to various combinations of organisms. NEMATODES Information of the relationship between two or more nematodes in causing a plant disease is fragmentary. One relationship that must exist is that of com- petition. It is not unusual to find that certain species are primary in a given disease situation; however, whether or not a given nematode is the predomi- nant species in causing disease in a plant will depend on factors such as the host, the initial population level of that nematode species as well as other nematodes, relative reproductive rates of the species involved, soil type, and other environmental factors. Furthermore, any advantage one nematode may have over another in a given situation is likely to be temporary and may change with the planting of a different crop and as the environment is other- wise modified. VIRUSES Soil treatments that kill nematodes reduce the incidence of certain diseases caused by soilborne viruses. When certain nematodes were allowed to feed on virus-infected plants and were then transferred to healthy plants, the healthy plants became diseased (Figure 28). Most of the soilborne viruses were really nematode-borne (nematode-transmitted) viruses. These fall into two distinct groups: the round- or polyhedral-shaped (NEPO-Viruses), transmitted only by
66 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 28 Chile peppers showing healthy and diseased plants. Left: healthy. Right: diseased, showing stunting and leaf mottle caused by tobacco rattle virus transmitted by the stubby-root nematode, Trichodorus allius. (Courtesy of the Department of Nema- tology, University of California, Riverside.) species of dagger (Xiphinema spp.) and needle nematodes (Longidorus spp.); and the rod- or tubular-shaped (NETU-Viruses), transmitted by species of stubby-root nematode (Trichodorus spp.). Examples of the NEPO-Viruses include tomato ring spot, tomato black ring, tobacco ring spot and the grape- vine fanleaf. The NETU-Viruses include pea early browning and tobacco rattle. Nematodes of some species retain the ability to transmit viruses for several weeks after feeding on an infected plant, indicating that the viruses are probably carried internally in the nematodes. The fanleaf disease of grapes was the first virus disease shown to be transmitted by nematodes. Nematode species in three generaâXiphinema, Trichodorus, and Longidorusâare now known to transmit some 20 viruses, many of economic importance, which together attack a wide range of host plants. PRACTICAL APPLICATIONS An understanding of interrelationships among various kinds of soil-inhabiting plant pathogens is extremely important. Most agricultural soils are infested with plant-parasitic nematodes of one to six or eight genera, as well as various
NEMATODE INTERRELATIONSHIPS WITH OTHER ORGANISMS 67 phytopathogenic fungi, bacteria, and viruses. Root systems of plants are often attacked simultaneously by several different soil-inhabiting organisms. Dis- eases resulting from such multiple infection are frequently classified as root- rot complexes. Their diagnosis is difficult. Since multiple infections of plant roots by several pathogens are common, it is necessary to gain a thorough understanding of the contributions of each component pathogen and the factors that favor their individual activity before it is possible to understand complex interrelationships. The breeder developing plant varieties resistant to specific diseases needs to be aware of plant-disease complexes caused by in- teracting pathogens and, if possible, to incorporate resistance to each of the pathogens involved. Recognition that nematodes interact with other pathogens in causing disease has to some extent promoted nematode control by the use of nema- tocides. Growers may elect to accept plant-yield losses caused by some nema- tode species, especially where the direct damage is slight, but they cannot afford the losses resulting from nematodes interacting with other organisms that cause such destructive diseases as black shank and Granville wilt of tobacco. In many instances, control of nematodes by soil fumigation results also in effective control of other diseases that would become economically damaging in the presence of the nematode. At present, soil nematocides are cheaper and more effective than soil fungicides or soil bactericides, and, for this reason, soil fumigation is cur- rently the cheapest means of control for many disease complexes. If less- expensive means for control of soil fungi or bacteria or if multiple resistance factors in plants are developed, the situation could change. FUTURE RESEARCH NEEDS Disease complexes involving plant-pathogenic nematodes and one or more other soil-inhabiting plant pathogens are now well recognized. Perhaps least understood is why the over-all effects of two pathogens interacting on a host are greater than the additive effects of two pathogens occurring singly. Also, the range and combinations of organisms involved, whether soilborne or aerial pathogens, are not known. For example, the effects of nematode parasitism of plant roots or other parts on leaf diseases caused by fungi, bacteria, and viruses as well as the physiological and biochemical bases for changes in susceptibility or disease expression need to be investigated.
68 FACTORS INFLUENCING NEMATODE CONTROL BIBLIOGRAPHY Harrison, B. D. 1964. The transmission of plant viruses in soil, pp. 118-144. In M. K. Corbett and H. D. Sisler (eds.). Plant virology. Univ. of Florida Press, Gainesville. 527 pp. Pitcher, R. S. 1963. Role of plant-parasitic nematodes in bacterial diseases. Phyto- pathology 53:35-39. Powell, N. T. 1963. The role of plant-parasitic nematodes in fungus diseases. Phyto- pathology 53:28-35. Raski, D. J., and W. B. Hewitt. 1963. Plant-parasitic nematodes as vectors of plant viruses. Phytopathology 53:39-47.
CHAPTER 7 Considerations Basic to Nematode Control The purpose of nematode control is to improve crop yields and quality. In a broader sense, nematode control aims to maximize the efficient and effective use of arable lands to meet the increasing need for food and fiber throughout the world. The biology of the nematode, its ecological relationships, methods of spread, value per acre of the host crop, and the cultural practices used in the particular area are important factors which must be considered in develop- ing control measures. NEMATODES-PRIMARILY A SOIL PROBLEM All plant parts, including roots, leaves, stems, buds, flowers, seeds, and even the trunks of trees, are subject to attack by nematodes. Most plant-parasitic species live either in the roots or in the soil around the roots of host plants, or in both. Even the foliar parasites spend at least part of their life cycle in the soil or in plant parts associated with the soil. Nematode eggs, larvae, or adults may be present in the soil and are usually more vulnerable to toxic chemicals or cultural practices than are nematodes within plant tissues. Certain developmental stages of some nematodes, such as the cysts of the sugar-beet nematode (Heterodera schachtii), are more re- sistant to desiccation or starvation than are active stages. However, it is not certain whether nematocidal chemicals are less effective against resistant than against active stages. More reliable information is needed in this important area of research. 69
70 FACTORS INFLUENCING NEMATODE CONTROL NEMATODE INFESTATIONS IN THE FIELD In some glasshouses or plant beds, where all the growing media can be treated with heat and chemicals or replaced with pasteurized soil, it is possible to eradicate nematode infestations. Under field conditions, however, eradication of nematodes is not possible. Suitable crop rotations, chemical treatments, or use of resistant varieties may reduce infestations to levels below those that cause economic losses in crop yield. But a few nematodes survive even under the most rigorous control programs, and, under favorable conditions and with repeated plantings of suitable host plants, these survivors inevitably build up again to damaging levels. For these reasons, it is justifiable to try to prevent the introduction of nematode parasites into noninfested fields or areas. The most common and efficient means of nematode dispersal are the activi- ties of man. Farm equipment moved from field to field, or even hundreds of miles across state boundaries, often carries viable nematodes in the soil or plant parts that cling to the equipment. Infected rootings, cuttings, bulbs, runners, and even seeds shipped for planting in other areas provide an ideal and direct means for nematodes to be carried from one site to another. HAZARDS OF MONOCULTURE Because it is difficult to detect low nematode populations in the soil and im- possible to eradicate an infestation once it is established, crop rotation is used to prevent the buildup of damaging infestations. It is a sound precaution to use crop rotations, even when repeated samplings fail to show the presence of plant-pathogenic nematodes. It may take years, even under monoculture, for a trace infestation of a particular species to build up to detectable and even- tually economically damaging levels. Once present, however, a high popula- tion is difficult to reduce and makes economic production of a susceptible crop extremely difficult. It is much harder to produce profitable crops once a severe nematode problem is present than it is to prevent the introduction and buildup of the pathogenic nematodes. Furthermore, the choice of a crop to use in a rotation is restricted when high levels of a destructive nematode are present. CONTROL METHODS AND LOCAL SITUATIONS Numerous measures may be successfully employed against a given nematode problem. As in any pest-control operation, local conditions may exclude some or all possibilities. Control measures must take into account the particular
CONSIDERATIONS BASIC TO NEMATODE CONTROL 71 area or situation involved. For example, it is important whether the host crop is a perennial or an annual and whether established plantings or future plant- ings are contemplated or planned. Variations in soil type are also important: dispersion of fumigants is greatly restricted in fine-textured clay, organic peat, or muck soils. This restricted dispersion limits the direct control of nematodes by fumigation, although practical economic responses by host plants are known from fumigation of both clay and peat types of soil. Further research is needed for a better understanding of factors affecting chemical control in these types of soil. Another example of the importance of local situations concerns resistant plant varieties. The first problem is availability of resistant varieties; certain resistant varieties are not always available in supply adequate to meet de- mand, and many plants have no known sources of resistance to important nematode species. Furthermore, resistance bred for one nematode species usually does not hold true for all populations of that species or for other species. Local differences in nematode populations or species may be an im- portant factor. The nature and extent of these differences are fruitful areas for research. The grape rootstocks Dogridge and Saltcreek, bred for resistance to root- knot nematode (Meloidogyne spp.), illustrate the importance of local condi- tions in the successful use of plant varieties for nematode control. These root- stocks, especially Saltcreek, are not suitable for use in fertile sandy loam or finer-textured soils, because they produce excessive vegetative growth, which makes control of fruit production difficult or impossible and affects the quality of fruit. Rotation schemes are highly effective control programs, but differences in local conditions must again be considered, because not all crops that are suitable for rotation can be used profitably. Differences in nematode life cycles and feeding habits must be considered in the development and application of control measures. Ectoparasitic nema- todes, which feed at root surfaces, are easily controlled in living plants by a side-dressing treatment with DBCP (l,2-dibromo-3-chloropropane). Control of the citrus nematode (Tylenchulus semipenetrans) on citrus and a dagger nematode (Xiphinema index) on grape are good examples of the successful use of DBCP. However, endoparasites, which feed and reproduce inside plant root tissues, such as root-knot nematodes, are not as readily killed in living plant roots; consequently, control has not been satisfactory when measured in growth response and yields. Another example of the importance of differences in life habits is en- countered when field soil is turned repeatedly during the summer to control nematodes. Theoretically, this treatment exposes nematodes to higher than normal temperatures and desiccation, either of which may be lethal. However,
72 FACTORS INFLUENCING NEMATODE CONTROL the larvae of some species of cyst nematodes, such as the sugar-beet nematode, are protected inside eggs held in the cysts; thus, they are better able than un- protected nematodes to resist desiccation and exposure to higher temperatures. The mortality of other species, such as the root-knot nematodes and ectopara- sitic species (Trichodorus, Xiphinema, and Criconemoides), is very high when the nematodes are exposed by soil cultivation. In a few situations, those species that attack the aerial parts of plants can be controlled by chemical sprays, or, if visual symptoms are well-defined and easily detected, the removal of infected plants may be practical. Parasites of roots present a different problem. Many root infections produce symptoms that are not diagnostic or that may be overlooked because they are under- ground. Nematodes in the soil are more difficult to recover for identification than those in plant tissues. Control of nematodes in perennial tree and vine crops is more difficult than in annual or herbaceous crops. The long-term nature of perennial crops makes rotation schemes, which are successfully used with annual crops, impractical. Furthermore, with annual crops, preplant soil fumigants can be applied every year or every other year, as needed, and the nematode kill does not have to be as complete as it must be for tree and vine crops. Often, successful annual crops can be grown following fumigation or fallow periods, even though nema- tode populations build up during that growing season to the same or higher levels than in untreated areas. With perennial crops, nematodes that survive fumigation or fallow have time to recover and build up to destructive levels. The cost of removing old plantings of tree or vine crops and bringing new plantings into bearing is very high. Surviving roots of excised plants provide a source of nutrient for nematodes and in part negate the effects of control practices. This has been studied in detail for grapevines, where it is known that some nematodes survive for four to five years after the vines are removed. There is also some evidence that citrus-nematode larvae survive for as long as nine years after removal of their tree hosts. IDENTIFICATION OF NEMATODES AND DEGREES OF INFESTATIONS The first step in diagnosing a crop-production problem in which nematodes may be a factor is to collect and identify the nematodes that are present in the involved soil or plant material. Accurate identification to species is es- sential because of differences in nematode life habits, host ranges, and patho- genic effects on various host plants. Distribution data are also important for determining whether the nematodes are associated or coincident with the disease problem.
CONSIDERATIONS BASIC TO NEMATODE CONTROL 73 COLLECTION OF SOIL AND PLANT SAMPLES -. The collection of soil may be made with a variety of augers, with tubes, or simply with a shovel (Figure 29). Moist soil, preferably in the vicinity of plant roots, rather than dry surface soil should be sampled. Each sample should con- tain feeder roots whenever possible. To avoid drying, plastic bags are prefer- able to other containers for soil and plant samples. Storage of samples at cool i temperatures is essential. EXTRACTION OF NEMATODES FROM SOIL AND PLANTS MECHANICAL ISOLATION Small pieces of plant parts, such as roots, stems, leaves, buds, and seeds, may be examined in clear water by tearing the tissue apart with dissecting needles. If nematodes are present, they will usually float free in the water and are readily seen. Food blenders (Figure 30) are sometimes used to macerate plant tissues to free the nematodes. The resulting suspension of plant parts and nematodes can be examined directly or can be fractionated with sieves before examination. SEPARATION BY SPECIFIC GRAVITY, SIZE, AND SHAPE Wet Sieving of Soil A volume of soil is mixed with approximately twice its volume of water in a pan. About 10 seconds are allowed for settling of coarse particles. Super- natant is then decanted through a series of sieves with openings ranging from 0.8 to 0.04 mm (Figure 31). The nematodes caught on the screens are washed into a beaker. The resultant suspension contains some small soil and plant particles along with the nematodes in water. This is examined under a binocular microscope equipped with a substage and a flat mirror to give dif- fuse, indirect light through a clear-glass stage (Figure 32). Final specific identi- fication is usually possible only by using a compound microscope with 900 to 1,000 magnification (oil immersion). Elutriation To increase precision, elutriation is sometimes substituted for the decanting of supernatant (Figure 33). Heterodera cysts are lighter and easier to separate
74 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 29 Equipment for collecting soil and root samples: a, shovel; b, Veihmeyer soil tube; c, hammer to drive Veihmeyer tube into ground; d, wrapped leather mallet to tap soil from bucket auger; e, 3-inch bucket-type auger; f, Dutch soil auger; g, extension used with e and f for deep samples.
CONSIDERATIONS BASIC TO NEMATODE CONTROL 75 FIGURE 30 Food blender for macerating plant parts.
76 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 31 Soil sieves, pans, and beakers for wet-sieving soil. FIGURE 32 Dissecting microscope with clear-glass stage and sub- stage mirror. Lamp with cover partly removed to show fluorescent bulbs.
CONSIDERATIONS BASIC TO NEMATODE CONTROL 77 FIGURE 33 Seinhorst soil elutriators. from soil than other nematode forms, and special apparatus have been devised for their recovery (e.g., Figure 34). Increasing the specific gravity of the soil solution with sugar and subsequent centrifugation is another commonly used method. SEPARATION BY NEMATODE MOVEMENT The equipment most widely used for separation by nematode movement is the Baermann funnel. A wire screen is fitted across a glass funnel about half
78 FACTORS INFLUENCING NEMATODE CONTROL FIGURE 34 Fenwick can to separate Heterodera cysts from soil. an inch below the top rim. A sample of soil, wet-sieve residue, or plant parts is placed on a paper tissue supported on the screen. The tissue used is pref- erably of the silicone-treated type, such as Kimwipes (Kimberly-Clark Corp., Neenah, Wisconsin), which do not disintegrate when wet. The bottom of the funnel may be extended by rubber or plastic tubing and closed with a pinch- cock or a small vial inserted in the end of the tubing (Figure 35). The funnel is then filled with water to a level to make contact with the sample on the wire. The nematodes move through the paper and screen into the water in the funnel. They settle by gravity to the bottom of the funnel, where they are collected for identification. A modification of this method is one set up inside a mist chamber (Figure 36). Substitution of a fine mist for the water bath allows better aeration and results in less microbial and chemical interference. In using a mist, the rubber tubing attached to a funnel can be inserted into a large test tube. The funnel is then placed in a mist chamber, where it is exposed to intermittent, regular fine sprays. The water filters through the sample, down the funnel, and into the test tube, where it accumulates and ultimately overflows. The nematodes carried in the water settle by gravity into the bottom of the tube. Both the Baermann-funnel and mist-chamber techniques depend on nematode activity and ability to move through soil or out of plant material and through the tissue paper into the funnel.
CONSIDERATIONS BASIC TO NEMATODE CONTROL 79 FIGURE 35 a, Large Baermann funnel fitted with several layers of cheesecloth to separate Xiphinema spp. from soil sievings; b, funnel showing wire screen in place; c, funnel with screen and tissues; d, funnel with pinchcock and water added; e and f, funnels with soil and root pieces on tissues; g and a are funnels fitted with small vials instead of pinchcocks. FIGURE 36 Baermann funnels on racks in mist chamber. Funnel tips are inserted directly into large test tubes instead of being closed by pinchcocks or vials.
80 FACTORS INFLUENCING NEMATODE CONTROL Endoparasitic species can be recovered from inside roots or other plant material by washing the plant parts free of soil and storing in a Mason jar or closed plastic bag (Figure 37). The lid of the Mason jar should be reversed and screwed down lightly enough to avoid desiccation but not so tightly as to lead to suffocation. The nematodes emerge from the roots, and after several days storage they are recovered by rinsing the jar with fresh water. The rinse water can be examined directly or cleared through the Baermann funnel. After the nematodes have been recovered by any of these or similar methods, identifications can be made from temporary mounts or specimens prepared in glycerine. The nematodes are picked up by a pipette, by a slender pick made of a bamboo splinter (Figure 38), or by nylon bristle from a tooth brush and transferred to a separate drop of water on a glass slide. Gentle heat from a small flame, sufficient only to stop movement, will kill the nematodes in the water. Before placing a cover glass on the drop of water, small glass rods are placed in the water to prevent the nematodes from flattening. A fingernail-polish or paraffin seal prevents drying and permits examination for several hours. Permanent mounts are made by kilhng the nematodes in water by gentle heat and fixing in 2V£ percent formalin for 24 hours. The nematodes are then transferred to 2V4 percent glycerine in 30 percent alcohol for 24 hours in a FIGURE 37 Moist roots in plastic bag or in Mason jar.
CONSIDERATIONS BASIC TO NEMATODE CONTROL 81 FIGURE 38 Tools and equipment for killing, fixing, preserving, and mounting nematodes for identification. sealed cavity slide and finally to 5 percent glycerine in 30 percent alcohol in a B.P.I. dish stored in a petri dish. After the mixture dries to a viscous condition (about 7 to 10 days), it is placed in a desiccator for 24 hours, and the speci- mens are mounted in dehydrated glycerine. Glass rod supports are again re- quired to prevent flattening. Zut slide ringing compound (Bennett's, Salt Lake City, Utah) is used to seal the cover slip. It is not always possible to prepare permanent slides for reference and re- study, in which case it is essential that mass collections be made whenever possible. Nematodes recovered in water are concentrated by allowing to settle for an hour or more, decanting the excess liquid, and adding hot 5 percent formalin in equal volume. Again allow to settle, decant, and store in small vials sealed by paraffin. Preserved material of this kind can be made into permanent mounts any time in the future when and if specific identification must be checked. The fact that there are usually mixtures of various plant- parasitic species in the same soil, often two or more species of the same genus that could be confused or overlooked, is further reason for keeping mass collections. Taxonomy of nematodes becomes increasingly complex and technical every year, as many new and closely related species are described. For this reason, increased training of scientists in this field is needed. Whenever practi- cal, there should be at least one trained taxonomist in every laboratory where nematological research is conducted.
82 FACTORS INFLUENCING NEMATODE CONTROL POPULATION LEVELS AND PREDICTION OF PLANT DAMAGE For most plant-parasitic nematode species, there is insufficient information on the levels of population or density of infestations that are likely to cause damage to particular crop plants. One notable exception is the golden nema- tode (Heterodera rostochiensis). In Great Britain and the Netherlands, advisory services are established for the control of this nematode by crop rotation. Soil samples are examined to calculate nematode populations surviving in the cyst stage. Based on the number of cysts with viable contents per gram of soil, ad- vice is given to growers as to whether to plant potatoes or to continue to plant nonhost crops. Similar advisory services are now available in the Netherlands for other nematodes pathogenic to various agricultural crops. The growers pay a fee for this service, which includes identification of nematode infestations in soil, calculations of nematode population densities, and advice as to types of crops to plant, based on the nematode analyses. Before such services can be developed further, much information is needed on the host-range preferences of many plant-parasitic nematode species, the minimum sampling procedures on which to judge infestations, and how to predict damage from population densities. Unfortunately, examination of soil samples is laborious, time-consuming, and expensive. Because of this high cost it is probable that similar advisory services will be developed only for agricultural crops of high acre value. BIBLIOGRAPHY Goodey, J. B. 1963. Laboratory methods for work with plant and soil nematodes. Ministry of Agriculture, Fisheries and Food. London. Tech. BulL No. 2. 72 pp. Goodey, T. Revised by J. B. Goodey. 1963. Soil and freshwater nematodes. Methuen and Co., Ltd., London. 544 pp. Southey, J. F. 1965. Plant nematology. Ministry of Agriculture, Fisheries and Food. London. Tech. Bull. No. 7. 282 pp. Thorne, G. 1961. Principles of nematology. McGraw-Hill Book Co., Inc., New York. 572pp.
PART BASIC PRINCIPLES OF CONTROL